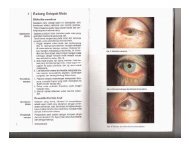
Clinical Pathways in Neuro-ophthalmology : An ... - E-Lib FK UWKS
Clinical Pathways in Neuro-ophthalmology : An ... - E-Lib FK UWKS
Clinical Pathways in Neuro-ophthalmology : An ... - E-Lib FK UWKS
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
This page <strong>in</strong>tentionally left blank
<strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong><br />
<strong>Neuro</strong>-Ophthalmology<br />
<strong>An</strong> Evidence-Based Approach<br />
Second Edition
This page <strong>in</strong>tentionally left blank
<strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong><br />
<strong>Neuro</strong>-Ophthalmology<br />
<strong>An</strong> Evidence-Based Approach<br />
Second Edition<br />
<strong>An</strong>drew G. Lee, M.D.<br />
Associate Professor of Ophthalmology, <strong>Neuro</strong>logy, and <strong>Neuro</strong>surgery<br />
University of Iowa Hospitals and Cl<strong>in</strong>ics<br />
Iowa City, Iowa<br />
Paul W. Brazis, M.D.<br />
Professor of <strong>Neuro</strong>logy<br />
Mayo School of Medic<strong>in</strong>e<br />
Consultant <strong>in</strong> <strong>Neuro</strong>logy and <strong>Neuro</strong>-Ophthalmology<br />
Mayo Cl<strong>in</strong>ic<br />
Jacksonville, Florida<br />
Thieme<br />
New York Stuttgart
Thieme New York<br />
333 Seventh Avenue<br />
New York, NY 10001<br />
Consult<strong>in</strong>g Medical Editor: Esther Gumpert<br />
Assistant Editor: Owen Zurhellen<br />
Director, Production and Manufactur<strong>in</strong>g: <strong>An</strong>ne V<strong>in</strong>nicombe<br />
Production Editor: David R. Stewart<br />
Market<strong>in</strong>g Director: Phyllis Gold<br />
Director of Sales: Ross Lumpk<strong>in</strong><br />
Chief F<strong>in</strong>ancial Officer: Peter van Woerden<br />
President: Brian D. Scanlan<br />
Compositor: Techset Composition Ltd., Salisbury, UK<br />
Pr<strong>in</strong>ter: Sheridan Books, Inc.<br />
<strong>Lib</strong>rary of Congress Catalog<strong>in</strong>g <strong>in</strong> Publication Data is available from the publisher<br />
Copyright # 2003 by Thieme Medical Publishers, Inc. This book, <strong>in</strong>clud<strong>in</strong>g all parts thereof, is legally protected<br />
by copyright. <strong>An</strong>y use, exploitation, or commercialization outside the narrow limits set by copyright legislation,<br />
without the publisher’s consent, is illegal and liable to prosecution. This applies <strong>in</strong> particular to photostat<br />
reproduction, copy<strong>in</strong>g, mimeograph<strong>in</strong>g or duplication of any k<strong>in</strong>d, translat<strong>in</strong>g, preparation of microfilms, and<br />
electronic data process<strong>in</strong>g and storage.<br />
Important note: Medical knowledge is ever-chang<strong>in</strong>g. As new research and cl<strong>in</strong>ical experience broaden our<br />
knowledge, changes <strong>in</strong> treatment and drug therapy may be required. The authors and editors of the material<br />
here<strong>in</strong> have consulted sources believed to be reliable <strong>in</strong> their efforts to provide <strong>in</strong>formation that is complete and <strong>in</strong><br />
accord with the standards accepted at the time of publication. However, <strong>in</strong> view of the possibility of human error<br />
by the authors, editors, or publisher of the work here<strong>in</strong>, or changes <strong>in</strong> medical knowledge, neither the authors,<br />
editors, or publisher, nor any other party who has been <strong>in</strong>volved <strong>in</strong> the preparation of this work, warrants that the<br />
<strong>in</strong>formation conta<strong>in</strong>ed here<strong>in</strong> is <strong>in</strong> every respect accurate or complete, and they are not responsible for any errors<br />
or omissions or for the results obta<strong>in</strong>ed from use of such <strong>in</strong>formation. Readers are encouraged to confirm the<br />
<strong>in</strong>formation conta<strong>in</strong>ed here<strong>in</strong> with other sources. For example, readers are advised to check the product<br />
<strong>in</strong>formation sheet <strong>in</strong>cluded <strong>in</strong> the package of each drug they plan to adm<strong>in</strong>ister to be certa<strong>in</strong> that the <strong>in</strong>formation<br />
conta<strong>in</strong>ed <strong>in</strong> this publication is accurate and that changes have not been made <strong>in</strong> the recommended dose or <strong>in</strong> the<br />
contra<strong>in</strong>dications for adm<strong>in</strong>istration. This recommendation is of particular importance <strong>in</strong> connection with new or<br />
<strong>in</strong>frequently used drugs.<br />
Some of the product names, patents, and registered designs referred to <strong>in</strong> this book are <strong>in</strong> fact registered<br />
trademarks or proprietary names even though specific reference to this fact is not always made <strong>in</strong> the text.<br />
Therefore, the appearance of a name without designation as proprietary is not to be construed as a representation<br />
by the publisher that it is <strong>in</strong> the public doma<strong>in</strong>.<br />
Pr<strong>in</strong>ted <strong>in</strong> the United States of America<br />
54321<br />
TNY ISBN 1-58890-136-X<br />
GTV ISBN 3-13-108642-4
Contents<br />
Foreword ........................................................ vii<br />
Preface ......................................................... ix<br />
1. The Diagnosis of Optic <strong>Neuro</strong>pathies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1<br />
2. Optic Neuritis. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 35<br />
3. Optic Disc Edema with a Macular Star and <strong>Neuro</strong>ret<strong>in</strong>itis . . . . . . . . . . . . . . 63<br />
4. Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy. . . . . . . . . . . . . . . . . . . . . . . . . . . . 73<br />
5. Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis . . . . . 93<br />
6. Traumatic Optic <strong>Neuro</strong>pathy . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 119<br />
7. Papilledema . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 129<br />
8. Transient Visual Loss . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 167<br />
9. Visual Field Defects . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 189<br />
10. Diplopia . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 215<br />
11. Third Nerve Palsies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 253<br />
12. Fourth Nerve Palsies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 281<br />
13. Sixth Nerve Palsies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 296<br />
14. Supranuclear Disorders of Gaze . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 311<br />
15. Ocular Myasthenia Gravis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 337<br />
16. Thyroid Eye Disease: Graves’ Ophthalmopathy . . . . . . . . . . . . . . . . . . . . . . 349<br />
17. Nystagmus and Other Ocular Oscillations . . . . . . . . . . . . . . . . . . . . . . . . . . 367<br />
18. Ptosis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 409<br />
19. Lid Retraction and Lid Lag. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 421<br />
20. <strong>An</strong>isocoria and Pupillary Abnormalities . . . . . . . . . . . . . . . . . . . . . . . . . . . . 433<br />
Index . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 464<br />
v
To our wives, Hilary and Liz<br />
and<br />
to our parents, Alberto C. Lee, M.D.,<br />
and Rosal<strong>in</strong>d G. Lee, M.D., and<br />
Dr. and Mrs. Peter T. Brazis
Foreword<br />
Never <strong>in</strong> the history of medic<strong>in</strong>e have physicians had so many ways to both diagnose<br />
and treat disease, and never have physicians had so many bureaucratic barriers to<br />
perform<strong>in</strong>g these activities. This paradox has necessitated a return to the days when<br />
cl<strong>in</strong>ical judgment was at least as important as diagnostic test<strong>in</strong>g. The challenge to all of<br />
us who care for patients is thus to understand the signs and symptoms that dist<strong>in</strong>guish<br />
among many different local and systemic disorders so that we can perform the most<br />
logical, expeditious, safe, and economic assessment.<br />
<strong>An</strong>drew G. Lee is an ophthalmologist and director of cl<strong>in</strong>ical neuro-<strong>ophthalmology</strong> at<br />
the University of Iowa Hospitals and Cl<strong>in</strong>ics <strong>in</strong> Iowa City, Iowa. Paul W. Brazis is a<br />
neurologist and head of the neuro-<strong>ophthalmology</strong> service at the Mayo Cl<strong>in</strong>ic <strong>in</strong><br />
Jacksonville, Florida. Both authors have extensive tra<strong>in</strong><strong>in</strong>g and experience <strong>in</strong> the field<br />
of neuro-<strong>ophthalmology</strong>, and both have contributed s<strong>in</strong>gly and together to the field of<br />
neuro-<strong>ophthalmology</strong> with numerous articles, chapters, and textbooks. In this book,<br />
these authors provide the reader with a triumverate of <strong>in</strong>formation. First, they describe<br />
the symptoms and signs of a variety of neuro-ophthalmologic disorders, such as<br />
anterior and retrobulbar optic neuropathies, ocular motor nerve pareses, and other<br />
disorders of ocular motility and alignment, and anisocoria. Second, they provide<br />
algorithms for differentiat<strong>in</strong>g, both <strong>in</strong> the office and us<strong>in</strong>g laboratory and neuroimag<strong>in</strong>g<br />
studies, among conditions that often have overlapp<strong>in</strong>g cl<strong>in</strong>ical manifestations. Third,<br />
they provide a basic set of references about each subject that the reader can use to<br />
expand his or her knowledge.<br />
By provid<strong>in</strong>g basic, cl<strong>in</strong>ically relevant <strong>in</strong>formation regard<strong>in</strong>g various disorders, their<br />
diagnosis, and treatment, this book teaches the reader how to approach a patient with a<br />
known or presumed neuro-ophthalmologic problem <strong>in</strong> a logical, straightforward, and<br />
cost-effective manner. As such, it is a welcome addition to the neuro-ophthalmologic<br />
repertoire.<br />
Neil R. Miller, M.D.<br />
Baltimore, Maryland<br />
vii
This page <strong>in</strong>tentionally left blank
Preface<br />
The primary goal of this book is to provide the reader with an easy-to-follow, heavily<br />
referenced guide to the management of common neuro-ophthalmologic conditions. We<br />
have specifically chosen to focus on recent (1990 to 2002) references, and we emphasize<br />
the best available cl<strong>in</strong>ical evidence. To this end, we have not <strong>in</strong>cluded letters or case<br />
reports unless they add significant new <strong>in</strong>formation. We <strong>in</strong>clude pre-1990 references<br />
only if they are of historical significance. We have tried to be <strong>in</strong>clusive, however, <strong>in</strong> the<br />
construction of our tables and charts, and provide the references as needed. The<br />
secondary goal of this book is to discuss and classify the available cl<strong>in</strong>ical evidence<br />
concern<strong>in</strong>g the evaluation and treatment of various neuro-ophthalmologic processes<br />
and grade the strength of any recommendations that are made. Readers will have to<br />
judge for themselves which is the best approach for the <strong>in</strong>dividual patient; the authors<br />
emphasize that these guidel<strong>in</strong>es are not meant to def<strong>in</strong>e any particular standard of care<br />
for these conditions.<br />
In this edition, we classify the cl<strong>in</strong>ical evidence <strong>in</strong>to the follow<strong>in</strong>g four<br />
categories. Where appropriate, we have summarized the class of evidence for each<br />
section.<br />
1. Class I: Well-designed, randomized, high statistical power controlled cl<strong>in</strong>ical trials<br />
<strong>in</strong>clud<strong>in</strong>g meta-analyses of such trials.<br />
2. Class II: Well-designed controlled studies without randomization <strong>in</strong>clud<strong>in</strong>g metaanalyses<br />
of such studies.<br />
3. Class III: Retrospective observational studies, cohort, or case-control studies, or<br />
multiple time series with or without <strong>in</strong>tervention.<br />
4. Class IV: Expert op<strong>in</strong>ion, case series, case reports.<br />
We grade the strength of the recommendations from each section as follows:<br />
1. Level A: A pr<strong>in</strong>ciple for patient management reflect<strong>in</strong>g a high degree of cl<strong>in</strong>ical<br />
certa<strong>in</strong>ty (usually requires class I evidence that directly addresses the cl<strong>in</strong>ical<br />
question).<br />
2. Level B: A recommendation reflect<strong>in</strong>g moderate cl<strong>in</strong>ical certa<strong>in</strong>ty based on either<br />
class II evidence or strong consensus of class III evidence with significant and<br />
consistent results.<br />
3. Level C: <strong>An</strong> acceptable practice option with low cl<strong>in</strong>ical certa<strong>in</strong>ty based on class III<br />
or class IV evidence.<br />
4. Level U: Inconclusive or conflict<strong>in</strong>g evidence, or op<strong>in</strong>ion that is <strong>in</strong>sufficient to<br />
support an evidence-based recommendation.<br />
ix
x Preface<br />
We would aga<strong>in</strong> like to thank our mentor, colleague, and friend Dr. Neil R. Miller for<br />
his encouragement and example. Dr. Brazis would also like to thank the follow<strong>in</strong>g<br />
<strong>in</strong>dividuals for their guidance: Drs. James Corbett, Jonathan Trobe, James Boll<strong>in</strong>g, and<br />
Frank Rub<strong>in</strong>o. He is appreciative of the encouragement and support of his family,<br />
especially Elizabeth, Erica, Paul, and Kelly Brazis.<br />
Dr. Lee acknowledges the support and encouragement of the three chairmen with<br />
whom he has served over the years: Drs. Mort Goldberg, Dan B. Jones, and Thomas<br />
We<strong>in</strong>geist. He is grateful to his colleagues at the University of Iowa, Drs. Randy Kardon,<br />
Michael Wall, and Stan Thompson, for their <strong>in</strong>sight, support, and friendship. He thanks<br />
his parents Drs. Alberto C. Lee and Rosal<strong>in</strong>d G. Lee for <strong>in</strong>still<strong>in</strong>g <strong>in</strong> a young man the<br />
thirst for knowledge and <strong>in</strong>tellectual curiosity. He is particularly thankful to his lov<strong>in</strong>g,<br />
patient, and tolerant wife, Dr. Hilary A. Beaver, who made a baby (Rachael E. Lee) while<br />
he made a book.<br />
We appreciate the assistance of our editors at Thieme Medical Publishers. We thank<br />
the faculty and residents of the Departments of Ophthalmology, <strong>Neuro</strong>logy, and<br />
<strong>Neuro</strong>surgery at the University of Iowa Hospitals and Cl<strong>in</strong>ics, and the Departments<br />
of <strong>Neuro</strong>logy and Ophthalmology at the Mayo Cl<strong>in</strong>ic <strong>in</strong> Jacksonville, Florida, for their<br />
academic stimulation. We especially appreciate and extend our love to our wives and<br />
families.<br />
<strong>An</strong>drew G. Lee, M.D.<br />
Paul W. Brazis, M.D.
1 r<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies<br />
The diagnosis of an optic neuropathy is usually made on cl<strong>in</strong>ical grounds alone. Several<br />
excellent references discuss <strong>in</strong> detail the anatomy of the optic nerve as well as<br />
exam<strong>in</strong>ation techniques (Burde, 1992; Miller, 1998; Trobe, 2001). The cl<strong>in</strong>ical features<br />
of optic neuropathies are summarized <strong>in</strong> Table 1–1. Other more sophisticated (and timeconsum<strong>in</strong>g)<br />
tests for optic neuropathy, such as visual evoked potentials, flicker fusion,<br />
formal color vision test<strong>in</strong>g, and contrast sensitivity, are not discussed.<br />
Once the diagnosis of optic neuropathy has been made, it is important to consider a<br />
wide differential diagnosis of possible etiologies, <strong>in</strong>clud<strong>in</strong>g hereditary, <strong>in</strong>flammatory,<br />
<strong>in</strong>filtrative, ischemic, demyel<strong>in</strong>at<strong>in</strong>g (optic neuritis), toxic, and compressive optic<br />
neuropathies. We refer the reader to the specific chapter on each type of optic<br />
neuropathy for further details.<br />
Can the Appearance of the Optic Nerve<br />
Differentiate Etiology?<br />
In general, the appearance of the optic nerve (e.g., normal, swollen, or pale) is not<br />
specific and cannot differentiate among various possible etiologies for optic neuropathy.<br />
Trobe et al reviewed 163 color fundus photographs of several entities result<strong>in</strong>g <strong>in</strong> optic<br />
Table 1–1. <strong>Cl<strong>in</strong>ical</strong> Features of an Optic <strong>Neuro</strong>pathy<br />
Decreased visual acuity<br />
Decreased color vision<br />
Visual field defect<br />
Ipsilateral relative afferent pupillary defect <strong>in</strong> unilateral or bilateral, asymmetric cases<br />
Light-near dissociation of the pupils <strong>in</strong> bilateral and symmetric cases<br />
Optic disc edema or disc atrophy (although the optic nerve may appear normal <strong>in</strong> retrobulbar optic<br />
neuropathy)<br />
1
2 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
atrophy, <strong>in</strong>clud<strong>in</strong>g glaucoma, central ret<strong>in</strong>al artery occlusion (CRAO), ischemic optic<br />
neuropathy (ION), optic neuritis (ON), hereditary optic neuropathy (Leber’s and non-<br />
Leber’s types), compressive optic neuropathy (CON), and traumatic optic neuropathy<br />
(TON) (Trobe, 1980). These photographs were reviewed by five ophthalmologists as<br />
‘‘unknowns.’’ Glaucoma, CRAO, and ION were correctly identified as the etiology by at<br />
least one of the five observers with an accuracy above 80%, but the rema<strong>in</strong><strong>in</strong>g etiologies<br />
were correctly identified <strong>in</strong> less than 50% of cases! Helpful features <strong>in</strong> differentiat<strong>in</strong>g the<br />
entities <strong>in</strong>cluded:<br />
1. The presence of ret<strong>in</strong>al arteriolar attenuation and sheath<strong>in</strong>g <strong>in</strong> ischemic lesions (e.g.,<br />
CRAO or ION).<br />
2. Temporal pallor <strong>in</strong> entities selectively <strong>in</strong>volv<strong>in</strong>g central vision and central visual field<br />
with spar<strong>in</strong>g of peripheral visual field (e.g., optic neuritis and toxic optic neuropathies).<br />
3. Superior or <strong>in</strong>ferior (sector) optic disc pallor <strong>in</strong> ION.<br />
Although optic disc cupp<strong>in</strong>g was often identified <strong>in</strong> glaucoma, it was also seen <strong>in</strong> 20%<br />
of cases not associated with glaucoma. Optic disc cupp<strong>in</strong>g <strong>in</strong> glaucoma cases, however,<br />
was more profound than <strong>in</strong> nonglaucomatous cases and greater neuroret<strong>in</strong>al rim pallor<br />
occurred <strong>in</strong> the nonglaucomatous cases. In patients with glaucoma, there is often<br />
absence of at least part of the neuroret<strong>in</strong>al rim, and the color of the rema<strong>in</strong><strong>in</strong>g rim is<br />
normal. With nonglaucomatous optic neuropathy, rarely is any area of the rim<br />
completely absent and the rema<strong>in</strong><strong>in</strong>g rim is often pale. Interest<strong>in</strong>gly, only 11% of<br />
these cases with a known history of papillitis or ION had sufficient clues to identify<br />
previous disc swell<strong>in</strong>g (Trobe, 1980).<br />
<strong>An</strong>other study suggested that optic disc appearance may help differentiate anterior<br />
ischemic optic neuropathy (AION) from ON, although there are overlapp<strong>in</strong>g features.<br />
Optic disc stereographs were reviewed by masked observers (87 AION and 68 ON)<br />
(Warner, 1997). Altitud<strong>in</strong>al disc swell<strong>in</strong>g was more than three times more common <strong>in</strong><br />
AION than ON, although most discs were diffusely swollen. Most patients with AION<br />
had hemorrhages, whereas most ON cases did not. Almost all discs with ON had<br />
normal color or were hyperemic; only 35% of discs with AION had pallid swell<strong>in</strong>g.<br />
Pallid swell<strong>in</strong>g was so rare <strong>in</strong> ON, however, that of discs with pallor, 93% had AION.<br />
Arterial attenuation was also much more typical of AION. AION was the cl<strong>in</strong>ical<br />
diagnosis <strong>in</strong> 82% of cases with altitud<strong>in</strong>al edema, 81% of cases with disc hemorrhage,<br />
93% of cases with pallid edema, and 90% of cases with arterial attenuation. A pale optic<br />
nerve with hemorrhage, regardless of type of edema, always represented AION (100%).<br />
A normal color nerve without hemorrhage reflected ON <strong>in</strong> 91% of cases, <strong>in</strong>creased from<br />
only 76% if hemorrhage was not considered. A hyperemic nerve with hemorrhage<br />
represented AION <strong>in</strong> 82% of cases, but if altitud<strong>in</strong>al edema was also present, AION<br />
<strong>in</strong>cidence <strong>in</strong>creased to 93%.<br />
In addition, numerous authors have stressed the localiz<strong>in</strong>g value to the optic chiasm<br />
or optic tract of a special type of optic atrophy caused by specific <strong>in</strong>volvement of the<br />
nerve fiber layer of the nasal and temporal ret<strong>in</strong>a, respectively. Involvement of these<br />
fibers results <strong>in</strong> atrophy of the nasal and temporal optic disc with spar<strong>in</strong>g of the <strong>in</strong>ferior<br />
and superior poles (‘‘band’’ or ‘‘bow tie’’ atrophy). Band atrophy occurs <strong>in</strong> the eye<br />
contralateral to the <strong>in</strong>volved optic tract and may be unilateral or bilateral with lesions of<br />
the optic chiasm.<br />
Neither the pattern (e.g., central scotoma, arcuate, altitud<strong>in</strong>al) of ipsilateral visual<br />
field impairment nor the severity of visual loss is pathognomonic for a specific optic
neuropathy, and virtually any visual field defect may occur with any optic neuropathy<br />
(Trobe, 1978). In their report on 35 eyes <strong>in</strong> 20 patients with CON and 70 eyes <strong>in</strong> 54<br />
patients with ON, Trobe and Glaser found central scotomas <strong>in</strong> 33% of cases of CON (vs.<br />
75% <strong>in</strong> ON) and felt that a central scotoma could not be used as a differentiat<strong>in</strong>g feature<br />
between the two entities (Trobe, 1978).<br />
The follow<strong>in</strong>g sections describe the evaluation of optic neuropathy; this approach is<br />
summarized <strong>in</strong> Figure 1–1. We beg<strong>in</strong> with an age-based differential diagnosis of an<br />
acute optic neuropathy. Two of the most common causes of acute optic neuropathy are<br />
AION and ON. Although there is considerable overlap <strong>in</strong> their cl<strong>in</strong>ical presentation, age<br />
can be used as an <strong>in</strong>itial differentiat<strong>in</strong>g feature <strong>in</strong> many cases (Rizzo, 1991). In younger<br />
patients (< 40 years old) with acute unilateral optic disc edema and evidence for an<br />
optic neuropathy, ON is more likely than AION. Conversely, <strong>in</strong> the older patient with<br />
acute optic disc edema and visual loss, AION is more common (class III).<br />
Is the <strong>Cl<strong>in</strong>ical</strong> Presentation Typical for<br />
<strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy?<br />
The features of typical AION are discussed <strong>in</strong> Chapter 4. If these features are present,<br />
the patient should undergo an evaluation for underly<strong>in</strong>g vasculopathic risk factors and<br />
giant cell arteritis (class III–IV, level B).<br />
Is the <strong>Cl<strong>in</strong>ical</strong> Presentation Typical for Optic<br />
Neuritis?<br />
The features and evaluation of typical ON are described <strong>in</strong> Chapter 2.<br />
Is the <strong>Cl<strong>in</strong>ical</strong> Presentation Consistent with<br />
Optic Disc Edema with a Macular Star<br />
(ODEMS)?<br />
The evaluation of optic disc edema with a macular star (ODEMS) is outl<strong>in</strong>ed <strong>in</strong><br />
Chapter 3.<br />
Is a Compressive Optic <strong>Neuro</strong>pathy Present?<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 3<br />
Compressive optic neuropathy (CON) usually causes pa<strong>in</strong>less, progressive, gradual loss<br />
of visual function (visual acuity, visual field, and color vision), a relative afferent<br />
pupillary defect (<strong>in</strong> unilateral or asymmetric cases), and optic disc edema or atrophy<br />
(but the optic disc may <strong>in</strong>itially appear normal) (Burde, 1992; Miller, 1998; Trobe, 1978).<br />
Unfortunately, CON may also present acutely or be steroid responsive and may<br />
masquerade as an <strong>in</strong>flammatory or demyel<strong>in</strong>at<strong>in</strong>g optic neuropathy.<br />
CON that is due to orbital or <strong>in</strong>tracanalicular lesions may result <strong>in</strong> ipsilateral optic<br />
disc edema followed by optic atrophy and may be associated with the development of<br />
abnormal blood vessels on the disc head called optociliary shunt vessels. These vessels
4 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 1–1. Evaluation of an optic neuropathy.
probably represent collateral circulation between the ret<strong>in</strong>al and choroidal venous<br />
circulation that allows venous blood to bypass the compression at the level of the<br />
optic nerve. The presence of an unexpla<strong>in</strong>ed relative afferent pupillary defect or<br />
unexpla<strong>in</strong>ed optic atrophy should prompt appropriate neuroimag<strong>in</strong>g studies (usually<br />
magnetic resonance imag<strong>in</strong>g of the <strong>in</strong>volved optic nerve) (Guy, 1990). Orbital signs such<br />
as proptosis, chemosis, or conjunctival <strong>in</strong>jection should direct the imag<strong>in</strong>g studies to<br />
the orbit (class III–IV, level B). Table 1–2 lists some possible causes of CON. Tables<br />
1–3, 1–4, 1–5, and 1–6, and Figures 1–2 and 1–3, review the ma<strong>in</strong> cl<strong>in</strong>ical features<br />
of men<strong>in</strong>gioma affect<strong>in</strong>g the anterior visual pathways, optic nerve glioma, and<br />
craniopharyngioma.<br />
Is There <strong>Cl<strong>in</strong>ical</strong> Evidence for an Infiltrative or<br />
Inflammatory Optic <strong>Neuro</strong>pathy?<br />
Infiltrative or <strong>in</strong>flammatory optic neuropathy may present with the typical features of<br />
an optic neuropathy discussed above. As described <strong>in</strong> Chapter 2, the cl<strong>in</strong>ical profile of<br />
typical ON (e.g., pa<strong>in</strong> with eye movement, typical age of onset, etc.) should be<br />
Table 1–2. Lesions Caus<strong>in</strong>g Compressive Optic <strong>Neuro</strong>pathy<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 5<br />
Intracranial or <strong>in</strong>traorbital benign and malignant tumors (Burde, 1992; Golnik, 1996; Katz, 1991;<br />
Kazim, 1992; Kodsi, 1993; Lee, 1997b)<br />
Men<strong>in</strong>gioma (see Table 1–3)<br />
Glioma (see Tables 1–4 and 1–5)<br />
Craniopharyngioma (see Table 1–6)<br />
Pituitary adenoma<br />
Lymphoma and leukemia (Brazis, 1995; Nygaard, 1991; Park, 1993; Roth, 2000)<br />
Germ<strong>in</strong>oma (Nakajima, 2001)<br />
S<strong>in</strong>us histocytosis with lymphadenopathy (Goldberg, 1998)<br />
Nasopharyngeal cancer<br />
Metastasis (Kattah, 1993; Newsom, 1999; Pengel, 1997)<br />
Extramedullary hematopoiesis (Aarabi, 1998)<br />
Orbital fractures<br />
Pneumatocele (We<strong>in</strong>, 1999)<br />
Inflammatory or <strong>in</strong>fectious diseases (e.g., mucoceles, scleros<strong>in</strong>g orbital <strong>in</strong>flammation) (Hao, 1994;<br />
Loehrl, 2000; Thorne, 2002; Yamaguchi, 1997)<br />
Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis (Tamai, 2000)<br />
Primary bone diseases (e.g., osteopetrosis, fibrous dysplasia, craniometaphyseal dysplasia,<br />
fibrosclerosis, Paget’s disease, aneurysmal bone cyst, pneumos<strong>in</strong>us dilatans, etc.) (Arroyo, 1991;<br />
Bland, 1992; Bocca, 1998; Caldermeyer, 1995; Chen, 1997; Daly, 1994; Grimm, 1995; Joseph, 1995;<br />
Katz, 1998; Michael, 2000; Saito, 1990; Schaffler, 2000; Skolnick, 2000; Steel, 1995; Stretch, 1992;<br />
Weisman, 1990)<br />
Vascular etiologies<br />
Orbital hemorrhage (Amrith, 1990; Buus, 1990; Dolman, 1991; Moorthy, 1992; Muthukumar, 1997)<br />
Orbital venous anomalies<br />
Carotid artery and anterior communicat<strong>in</strong>g artery aneurysms (Bakker, 1999; Miller, 1995; Misra,<br />
1991; Ortiz, 1991; Shutter, 1993; Vargas, 1994)<br />
Dolichoectasia of the carotid artery (Colap<strong>in</strong>to, 1996; Jacobson, 1999; Savy, 1996)<br />
(cont<strong>in</strong>ued)
6 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–2. (cont<strong>in</strong>ued)<br />
Compression by supracl<strong>in</strong>oid carotid artery (Ishikawa, 2000; Jacobson, 1999)<br />
Arteriovenous malformations<br />
Thyroid ophthalmopathy (see Chapter 16)<br />
Hydrocephalus<br />
Iatrogenic<br />
Intracranial catheters (Shults, 1993)<br />
Intranasal balloon catheter<br />
Intracranial oxidized cellulose hemostat<br />
Postoperative (e.g., post–optic canal decompression, s<strong>in</strong>us surgery) (Carter, 1998; Edelste<strong>in</strong>, 1998)<br />
Musl<strong>in</strong>oma (Bhatti, 2000; Lee, 1997a)<br />
differentiated from atypical ON (e.g., lack of pa<strong>in</strong>, atypical age of onset, anterior or<br />
posterior segment <strong>in</strong>flammation, etc.). Atypical cases should undergo an evaluation for<br />
<strong>in</strong>filtrative or <strong>in</strong>flammatory etiologies as listed <strong>in</strong> Table 1–7 (class IV, level C).<br />
Patients with <strong>in</strong>flammatory autoimmune optic neuropathy often have a progressive<br />
or recurrent steroid responsive or steroid dependent cl<strong>in</strong>ical course. A more detailed<br />
discussion of the evaluation of atypical ON and these alternative etiologies is found <strong>in</strong><br />
Chapter 2. In patients with a possible <strong>in</strong>flammatory or <strong>in</strong>filtrative optic neuropathy, a<br />
Table 1–3. <strong>Cl<strong>in</strong>ical</strong> Features of Men<strong>in</strong>giomas Affect<strong>in</strong>g the <strong>An</strong>terior Visual Pathway<br />
Most commonly middle aged (peak <strong>in</strong> the 5th decade)<br />
Female : male ¼ 3:1<br />
White > African-American<br />
Increased frequency <strong>in</strong> neurofibromatosis<br />
May grow <strong>in</strong> pregnancy<br />
Symptoms<br />
Pa<strong>in</strong>less (rarely retro-orbital pa<strong>in</strong>)<br />
Gradually progressive loss of vision or visual field defects<br />
If frontal, may have mental status changes<br />
May have diplopia if cavernous s<strong>in</strong>us <strong>in</strong>volvement<br />
Olfactory groove may have anosmia<br />
Ophthalmic signs<br />
May have relative afferent pupillary defect (RAPD)<br />
Optic disc edema (<strong>in</strong>clud<strong>in</strong>g papilledema) and=or optic atrophy<br />
May see optociliary shunt vessel (disc collaterals, visual loss, and optic atrophy—characteristic<br />
triad)<br />
Indocyan<strong>in</strong>e green videoangiography may show abnormal hemodynamics of choroidal circulation<br />
<strong>in</strong> patients with sheath men<strong>in</strong>giomas (Muci-Mendoza, 1999)<br />
Visual acuity loss or visual field defects<br />
Generalized depression or constriction (orbital=canal=sphenoid)<br />
Central, paracentral, or cecocentral (orbital=canal)<br />
Homonymous hemianopsia (suprasellar=sphenoid)<br />
Bitemporal hemianopsia (suprasellar=sphenoid)<br />
May have proptosis (orbital=sphenoid)<br />
Motility deficits
Table 1–3. (cont<strong>in</strong>ued)<br />
Sixth nerve palsy (most common), but any ocular motor palsy (third, fourth, sixth, comb<strong>in</strong>ation)<br />
Restrictive extraocular muscle mechanical limitation if orbital lesion<br />
Paretic pattern if suprasellar=sphenoid=cavernous s<strong>in</strong>us<br />
Differential diagnosis of optic nerve sheath men<strong>in</strong>gioma<br />
Sarcoidosis and other granulomatous diseases<br />
Optic nerve sheath men<strong>in</strong>gocele (Garrity, 1990)<br />
Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis<br />
Idiopathic <strong>in</strong>flammatory perioptic neuritis<br />
Metastasis (e.g., breast cancer) and other tumors (Newman, 1996)<br />
Source: Al-Mefty, 1990; Cunliffe, 1992; DeMonte, 1994; Dutton, 1991, 1992; Fayaz, 1999; F<strong>in</strong>eman, 1999; Garrity,<br />
1990; Goldsmith, 1994a,b; Grunberg, 1991; Hirsch, 1993; K<strong>in</strong>jo, 1995; Kl<strong>in</strong>k, 2000; Kotapka, 1994; Larson, 1995;<br />
Lee, 1996; Lee Wan, 1990; Lundsford, 1994; Mafee, 1999; Maroon, 1994; Moyer, 2000; Muci-Mendoza, 1999;<br />
Newman, 1994, 1996; Rub<strong>in</strong>ste<strong>in</strong>, 1994; Sadun, 1993; Stafford, 1998; Vaphiades, 2001; Weaver, 1993; Wilson, 1994;<br />
Wroe, 1991; Zimmerman, 1990b.<br />
Table 1–4. <strong>Cl<strong>in</strong>ical</strong> Features of Optic Glioma<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 7<br />
Age<br />
Can present at any age<br />
Usually < 10 years old (75%)<br />
Mean 8.8 years (90%< 20 years old)<br />
No gender predilection<br />
Association with neurofibromatosis type 1 (NF1)<br />
29% of optic nerve gliomas occur <strong>in</strong> sett<strong>in</strong>g of NF1<br />
15% of NF1 who have no visual symptoms have glioma<br />
Patients with NF1 may have borderl<strong>in</strong>e favorable or no different prognosis than patients without<br />
NF1<br />
Location of <strong>in</strong>filtration (topographic localization)<br />
One or both optic nerves (nerve alone <strong>in</strong> 24%)<br />
Optic disc (1.6%)<br />
Optic chiasm (75.7%) or tract<br />
In general, the more anterior the lesion, the better the prognosis<br />
Signs and symptoms<br />
Proptosis<br />
Pa<strong>in</strong>less progressive visual loss (optic neuropathy)<br />
Visual loss at presentation <strong>in</strong> 87.5%<br />
Hypothalamic symptoms (26%) or endocr<strong>in</strong>ologic: diabetes <strong>in</strong>sipidus, diencephalic wast<strong>in</strong>g,<br />
precocious puberty, somnolence, growth failure<br />
Disc swell<strong>in</strong>g (35%) or atrophy (59%)<br />
Rare optociliary shunt vessels<br />
Strabismus<br />
Nystagmus (23%) (spasmus nutans–like nystagmus)<br />
Visual field defects (central or bitemporal)<br />
Headache (23%)<br />
Intr<strong>in</strong>sic enlargement of optic nerve with variable contrast enhancement<br />
(cont<strong>in</strong>ued)
8 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–4. (cont<strong>in</strong>ued)<br />
<strong>Neuro</strong>imag<strong>in</strong>g<br />
Magnetic resonance (MR) scan with gadol<strong>in</strong>ium superior to computed tomography (CT) scan<br />
Differential diagnosis<br />
Parenchymal optic nerve enlargement<br />
Sarcoid<br />
Tuberculosis<br />
Syphilis<br />
Optic neuritis (e.g., multiple sclerosis)<br />
Optic nerve <strong>in</strong>filtration (e.g., leukemia, lymphoma)<br />
Extraparenchymal<br />
Optic nerve sheath men<strong>in</strong>gioma<br />
Optic nerve sheath metastasis<br />
Treatment<br />
Controversial<br />
Gliomas are often static lesions<br />
May enlarge and cause progressive visual loss <strong>in</strong> one or both eyes<br />
Treatment recommendations outl<strong>in</strong>ed <strong>in</strong> Figure 1–3 (Lee, 1999)<br />
Observation <strong>in</strong> stable cases is reasonable (class III–IV, level B)<br />
Radiation therapy (class III–IV, level C)<br />
Usually 5400 cGy <strong>in</strong> daily dose fractions 150–180 cGy<br />
Radiation risks <strong>in</strong>clude cerebrovascular disease, moyamoya disease, cerebral atrophy,<br />
subnormal <strong>in</strong>telligence or learn<strong>in</strong>g disabilities, secondary malignancies (e.g., astrocytomas),<br />
cataracts, radiation ret<strong>in</strong>opathy or optic neuropathy, endocr<strong>in</strong>opathy, hypothalamic<br />
dysfunction<br />
Chemotherapy—various agents <strong>in</strong> various comb<strong>in</strong>ations: act<strong>in</strong>omyc<strong>in</strong>-D, v<strong>in</strong>crist<strong>in</strong>e, 2 chloroethylcyclohexyl-1-nitrosourea<br />
(CCNU), 6-thioguan<strong>in</strong>e, procarbaz<strong>in</strong>e, dibromodulatol, topotecan,<br />
carboplat<strong>in</strong>, etoposide (class III–IV, level C)<br />
Surgical therapy (class III–IV, level C)<br />
Optic nerve glioma with no useful vision or progression may be resected<br />
Chiasmal hypothalamic, optic tract glioma cannot be resected<br />
Exophytic component of tumor may be debulked<br />
Hydrocephalus may require shunt<strong>in</strong>g procedure<br />
Prognosis<br />
80% have stable vision after an <strong>in</strong>itial period of visual loss<br />
10-year survival rate 85–100% <strong>in</strong> various series<br />
Spontaneous regression may occur<br />
Source: Brodovsky, 1997; Chateil, 2001; Créange, 1999; Cumm<strong>in</strong>gs, 2000; Deliganis, 1996; DiMario, 1993; Drake,<br />
1991; Dunn, 1990; Dutton, 1994; Epste<strong>in</strong>, 1992; Friedman, 1997; Fuss, 1999; Garvey, 1996; Gayre, 2001; Grill, 1999;<br />
Hoffman, 1993; Imes, 1991; Janss, 1995; Jenk<strong>in</strong>, 1993; Kestle, 1993; Kovalic, 1990; Lev<strong>in</strong>, 1992; Listernick, 1992,<br />
1994, 1997; Liu, 1992a, 2001; Moghrabi, 1993; Nishio, 1993; Oaks, 1990; Packer, 1993, 1994; Parsa, 2001; Petronio,<br />
1991; Pierce, 1990; Rodriguez, 1990; Shuper, 1997; Sutton, 1994, 1995; Wisoff, 1990a,b.<br />
lumbar puncture and additional laboratory studies (e.g., complete blood count, syphilis<br />
serology, ant<strong>in</strong>uclear antibody, Lyme titer, chest radiograph, etc.) should be considered.<br />
The appropriate specific laboratory studies should be directed by pert<strong>in</strong>ent history and<br />
exam<strong>in</strong>ation f<strong>in</strong>d<strong>in</strong>gs. Table 1–8 reviews the evaluation of an atypical or unexpla<strong>in</strong>ed<br />
optic neuropathy (class IV, level C).
Is There Evidence for Traumatic Optic<br />
<strong>Neuro</strong>pathy?<br />
The features and evaluation of TON are discussed <strong>in</strong> Chapter 6.<br />
Is There Evidence for a Toxic or Nutritional<br />
Optic <strong>Neuro</strong>pathy?<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 9<br />
Table 1–5. <strong>Cl<strong>in</strong>ical</strong> Features of Adult Malignant Gliomas of the <strong>An</strong>terior Visual Pathway<br />
Age at presentation: middle-age; range 6–79, mean 47.8; 73% were 40 or older<br />
Sex: 65% males and 35% females<br />
<strong>Cl<strong>in</strong>ical</strong> signs and symptoms<br />
Decreased vision<br />
Bilateral or unilateral<br />
Visual acuity usually falls to bl<strong>in</strong>dness over average of 11.1 weeks (range 1–60 weeks)<br />
Optic nerve visual field defects<br />
Normal discs, optic disc swell<strong>in</strong>g or atrophy<br />
Proptosis<br />
Ophthalmoplegia<br />
Retro-orbital pa<strong>in</strong> common<br />
Macular edema, cherry-red spot, and flame hemorrhage or hemorrhagic papillopathy may<br />
simulate central ret<strong>in</strong>al ve<strong>in</strong> occlusion (CRVO)<br />
Not associated with NF1 (neurofibromatosis)<br />
Location<br />
Involves chiasm and at least one contiguous optic nerve; often <strong>in</strong>volves hypothalamus, third<br />
ventricle, basal ganglia, temporal lobe<br />
Primarily affects chiasm and <strong>in</strong>tracranial optic nerves<br />
Treatment<br />
Radiation<br />
Chemotherapy<br />
Treatment may temporarily improve or rarely stabilize vision<br />
Pathology: malignant astrocytoma<br />
Prognosis<br />
Poor<br />
Overall mortality 97%<br />
Mean survival 8.7 months (3 to 24 months)<br />
Patients with toxic optic neuropathies usually present with pa<strong>in</strong>less, bilaterally<br />
symmetric, and slowly progressive visual loss. The visual field defect is typically<br />
bilateral central or cecocentral scotomas. The optic nerves may appear normal until<br />
late <strong>in</strong> the course of the disease when optic atrophy (often temporal pallor) usually<br />
develops. Occasionally the discs may be swollen and slightly hyperemic. A number of<br />
medications and tox<strong>in</strong>s may result <strong>in</strong> optic neuropathy (Brazis, 1998; Danesh-Meyer,<br />
2000; Sedwick, 1991, 1992). These are summarized <strong>in</strong> Table 1–9. Most of these etiologies<br />
can be excluded by a careful and detailed exposure and occupational history.
10 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–6. <strong>Cl<strong>in</strong>ical</strong> Feature of Craniopharyngiomas<br />
<strong>An</strong>y age<br />
Bimodal <strong>in</strong>cidence<br />
Peak age < 20 and 50 to 70 years old<br />
Equal sex distribution<br />
<strong>Cl<strong>in</strong>ical</strong><br />
Decreased visual acuity (optic nerve, chiasm, optic tract)<br />
In children—often decreased acuity and papilledema (50%)<br />
In adults—less commonly papilledema<br />
Signs of <strong>in</strong>creased <strong>in</strong>tracranial pressure (headache, nausea, vomit<strong>in</strong>g)<br />
Endocr<strong>in</strong>e<br />
Absent or precocious sexual development<br />
Growth disturbances<br />
Variable hypopituitarism<br />
Diabetes <strong>in</strong>sipidus<br />
Obesity<br />
Impotence<br />
Amenorrhea=galactorrhea<br />
Somnolence, confusion, or dementia (especially <strong>in</strong> older patients)<br />
Ocular f<strong>in</strong>d<strong>in</strong>gs<br />
Seesaw nystagmus<br />
Visual field defects<br />
Inferior bitemporal field defect (most have field defects)<br />
May have <strong>in</strong>congruous, asymmetric defect<br />
May <strong>in</strong>volve optic tract<br />
May cause ocular motor nerve palsies<br />
<strong>Neuro</strong>imag<strong>in</strong>g<br />
Magnetic resonance imag<strong>in</strong>g (MRI) del<strong>in</strong>eates tumor and <strong>in</strong>tracranial anatomy<br />
Computed tomography (CT) shows calcification better<br />
Occasionally may <strong>in</strong>filtrate optic nerve, chiasm tract, mimick<strong>in</strong>g primary <strong>in</strong>tr<strong>in</strong>sic tumor such as<br />
optic glioma (‘‘potbelly’’ appearance of optic nerve)<br />
Treatment<br />
Surgical: complete vs. partial resection<br />
Radiotherapy<br />
Cyst aspiration and P32 <strong>in</strong>stillation<br />
Consider <strong>in</strong>tracystic chemotherapy (bleomyc<strong>in</strong>)<br />
Secondary malignant glioma can develop after radiation<br />
therapy<br />
Source: Brummitt, 1992; Crotty, 1995; El-Mahdy, 1998; Fahlbusch, 1999; Honegger, 1999; Petito, 1996; Rao, 1995;<br />
We<strong>in</strong>er, 1994; Youl, 1990.<br />
Ethambutol is a commonly used medication that may cause toxic optic neuropathy.<br />
The mechanism of ethambutol toxicity is poorly understood but may be related to z<strong>in</strong>c<br />
depletion (Schild, 1991). The <strong>in</strong>cidence of toxicity is dose and duration dependent (Choi,<br />
1997; Harcombe, 1991; Kumar, 1993; Russo, 1994; Schild, 1991; Seth, 1991; Thomas, 1994;<br />
Tsai, 1997), with the <strong>in</strong>cidence of optic neuropathy be<strong>in</strong>g as high as 6% at doses of<br />
25 mg=kg=day. Doses less than 15 mg=kg=day are thought to be relatively safe, but optic<br />
neuropathy may occur even at ‘‘safe’’ doses.
Figure 1–2. Treatment algorithm for men<strong>in</strong>giomas affect<strong>in</strong>g optic pathway.<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 11<br />
Barron et al reported ethambutol optic neuropathy <strong>in</strong> 3 of 304 (0.99%) patients treated<br />
with ethambutol at 25 mg=kg=day for 60 days followed by 15 mg=kg=day (Barron,<br />
1974). Leibold described two types of visual loss due to ethambutol toxicity: a central<br />
toxicity (e.g., decreased visual acuity, central scotomas, and impaired color perception)<br />
and a periaxial toxicity (e.g., normal or almost normal visual acuity, normal color<br />
perception, and peripheral quadrantic scotomas or constriction) (Leibold, 1966). There<br />
was a 20% <strong>in</strong>cidence of central toxicity and an 11% <strong>in</strong>cidence of periaxial toxicity <strong>in</strong> 35<br />
patients receiv<strong>in</strong>g doses higher than 35 mg=kg=day for a m<strong>in</strong>imum of 185 days. A 5.3%<br />
<strong>in</strong>cidence of periaxial toxicity occurred <strong>in</strong> the 38 patients receiv<strong>in</strong>g less than<br />
35 mg=kg=day (Leibold, 1966). Although many authors feel that doses of 25 mg=kg=day<br />
=day for less than 2 months followed by ma<strong>in</strong>tenance doses of 15 mg=kg=day are safe,<br />
there are cases of visual loss even at ‘‘safe’’ doses (Alvarez, 1993; Thomas, 1994; Tsai,<br />
1997). Bronte-Stewart et al reported five patients with severe visual loss after<br />
25 mg=kg=day for 2 months followed by 15 mg=kg=day (Bronte-Stewart, 1976). Three
12 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 1–3. Treatment algorithm for optic pathway gliomas based on location. (Repr<strong>in</strong>ted from Lee,<br />
1999, with permission from # Swets & Zeitl<strong>in</strong>ger.)<br />
of these five patients had renal disease that may have <strong>in</strong>creased drug levels because 70%<br />
of the ethambutol dose is excreted by the kidneys (Citron, 1986). Tsai and Lee reported<br />
10 patients with ethambutol optic neuropathy from ‘‘safe’’ doses, stress<strong>in</strong>g that there is<br />
<strong>in</strong> fact no safe dose of ethambutol. Toxicity <strong>in</strong> this study was most prom<strong>in</strong>ent <strong>in</strong><br />
<strong>in</strong>dividuals over the age of 60 years, and thus this drug must be used with caution,<br />
especially <strong>in</strong> elderly patients (Tsai, 1997). Isoniazid (isonicot<strong>in</strong>ic acid hydrazide, INH),<br />
especially <strong>in</strong> comb<strong>in</strong>ation with ethambutol, has also been reported to cause a toxic optic<br />
neuropathy, and isoniazid toxicity should be suspected as the etiology <strong>in</strong> cases of<br />
persistent visual loss despite discont<strong>in</strong>uation of ethambutol (Jimenez-Lucho, 1987).<br />
Visual evoked potential studies may be useful <strong>in</strong> evaluat<strong>in</strong>g patients with early<br />
ethambutol toxicity (Kumar, 1993).<br />
Nutritional deficiencies may result <strong>in</strong> optic neuropathy (Bourne, 1998; Lessell, 1998).<br />
Some vitam<strong>in</strong> and nutrient deficiencies caus<strong>in</strong>g an optic neuropathy are listed <strong>in</strong> Table<br />
1–10.<br />
Pernicious anemia or dietary deficiency (e.g., vegetarian) may result <strong>in</strong> a vitam<strong>in</strong> B 12<br />
deficiency optic neuropathy. The pathophysiology of ‘‘alcohol amblyopia’’ is probably<br />
related to a deficiency (nutritional amblyopia) of B 12, thiam<strong>in</strong>e, and=or folate (rather<br />
than a direct toxic effect of alcohol). The ability of tobacco alone to cause a toxic optic<br />
neuropathy has been asserted by several authors (Samples and Younge, 1981). Samples<br />
and Younge (1981), for example, state that central and cecocentral scotomas may occur<br />
<strong>in</strong> association with smok<strong>in</strong>g alone, especially cigar smok<strong>in</strong>g. A toxic effect of cyanide<br />
may be the basis for tobacco optic neuropathy (Bronte-Stewart, 1976). Smok<strong>in</strong>g may<br />
also impair <strong>in</strong>test<strong>in</strong>al vitam<strong>in</strong> B 12 absorption.<br />
Patients suspected of harbor<strong>in</strong>g a toxic or nutritional optic neuropathy should be<br />
screened for nutritional deficiencies and treated with appropriate supplementation<br />
(class IV, level C). These patients should be urged to discont<strong>in</strong>ue alcohol and tobacco
Table 1–7. Infiltrative or Inflammatory Optic <strong>Neuro</strong>pathies<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 13<br />
Neoplastic<br />
Plasmacytoma and multiple myeloma (Ma<strong>in</strong>i, 1997)<br />
Carc<strong>in</strong>omatous men<strong>in</strong>gitis (Freilich, 1995; Ing, 1996; Katz, 1991; McFadzean, 1994; Sung, 1998;<br />
Teare, 1991)<br />
Leukemia (Brown, 1992a; Camera, 1993; Costagliola, 1992; Cramer, 1996; Horton, 1992; Pierro,<br />
1992; Shibasaki, 1992; Wallace, 1991)<br />
Lymphoma (Dunker, 1996; Fierz, 2001; Forman, 1998; Guyer, 1990; Noda, 1993; Siatkowski, 1992;<br />
Strom<strong>in</strong>ger, 1993; Yamamoto, 1994; Zaman, 1993)<br />
Infiltrative orbitopathy <strong>in</strong> POEMS syndrome<br />
Reactive lymphocytosis with pseudolymphoma from phenyto<strong>in</strong> (Galetta, 1991)<br />
Paraneoplastic disease (Ing, 1996; Lieberman, 1999; Luiz, 1998; Malik, 1992; Oohira, 1991;<br />
Thambisetty, 2001)<br />
Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis (Aylward, 1995; Botella, 1994; Girk<strong>in</strong>, 1998;<br />
Hamilton, 1993; Jacobson, 1996; Kawano, 1995; Lam, 1994; Lev<strong>in</strong>e, 1993; Mamelak, 1993;<br />
Nishizaki, 1997; Olmos, 1993; Parney, 1997; Rootman, 1994)<br />
Infectious etiologies<br />
Cryptococcal men<strong>in</strong>gitis (Cohen, 1993)<br />
Aspergillus (Brown, 1994; D<strong>in</strong>owitz, 2001; Hutnik, 1997; Johnson, 1999)<br />
Mucormycosis (Balch, 1997)<br />
Cysticercosis (Chandra, 2000; Gulliani, 2001; Gurha, 1999)<br />
Lyme disease (Lesser, 1990)<br />
Tuberculosis<br />
Toxoplasmosis (Song, 2002)<br />
Syphilis (Danesh-Meyer, 1999)<br />
Cat-scratch disease (Golnik, 1994b)<br />
HIV (AIDS) (Cacciatori, 1996)<br />
Inflammatory diseases (Burde, 1992)<br />
Churg-Strauss (Acheson, 1993)<br />
Contiguous s<strong>in</strong>us disease<br />
Behçet’s disease<br />
Sarcoidosis (Achiron, 1995; Beck, 1994; Carmody, 1994; DeBroff, 1993; Ing, 1997; Kosmorsky, 1996;<br />
Pelton, 1999; Sharma, 1991; Silver, 1994; Thorne, 1998)<br />
Wegener’s granulomatosis (Belden, 1993)<br />
Systemic lupus erythematosus (Ahmadieh, 1994; Rosenbaum, 1997; Siatkowski, 2001)<br />
Sjögren’s syndrome<br />
Relaps<strong>in</strong>g polychondritis<br />
Polyarteritis nodosa<br />
Inflammatory bowel disease<br />
Granulomatous hypophysitis (Arsava, 2001)<br />
Isolated optic nerve pseudotumor (Patankar, 2000)<br />
Scleros<strong>in</strong>g orbital <strong>in</strong>flammation (Thorne, 2002)<br />
use. Both serum and erythrocyte folate levels should be checked because there may be<br />
variability <strong>in</strong> the serum folate level alone (especially related to recent meals) (Golnik,<br />
1994).<br />
Toxic or nutritional optic neuropathies are pa<strong>in</strong>less, subacute <strong>in</strong> onset, and bilateral,<br />
and usually <strong>in</strong>volve central visual acuity and visual fields (e.g., central and cecocentral
14 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–8. Evaluation of an Atypical or Unexpla<strong>in</strong>ed Optic <strong>Neuro</strong>pathy<br />
First-l<strong>in</strong>e test<strong>in</strong>g<br />
Magnetic resonance imag<strong>in</strong>g of optic nerve(s)<br />
Erythrocyte sedimentation rate<br />
Complete blood count with differential<br />
Syphilis serology<br />
<strong>An</strong>t<strong>in</strong>uclear antibody (ANA)<br />
Chest radiograph<br />
<strong>An</strong>giotens<strong>in</strong>-convert<strong>in</strong>g enzyme (ant<strong>in</strong>eutrophil cytoplasmic antibody, ANCA)<br />
Lumbar puncture<br />
Second-l<strong>in</strong>e test<strong>in</strong>g<br />
Gallium scan if sarcoidosis suspected<br />
Purified prote<strong>in</strong> derivative (PPD) sk<strong>in</strong> test<strong>in</strong>g if tuberculosis suspected<br />
<strong>An</strong>ti–double-stranded DNA, complement levels, etc., if systemic lupus erythematosus or other<br />
collagen vascular disease suspected<br />
Leber’s hereditary optic neuropathy mutation blood test<br />
Heavy metal screen<br />
Serum vitam<strong>in</strong> B 12 and folate levels<br />
Lyme titer if endemic area or exposure history<br />
Paraneoplastic antibody profile (e.g., autoantibodies for collaps<strong>in</strong> response mediated prote<strong>in</strong><br />
(CRMP)-5 may be associated with optic neuropathy <strong>in</strong> patients with lung cancer, especially<br />
small-cell type, or thymoma) (Cross, 2002; Thambisetty, 2001; Yu, 2001)<br />
Consider more specific serologic studies if <strong>in</strong>fectious process suspect (e.g., Bartonella titers for<br />
cat-scratch disease, toxoplasmosis titers, toxocara titers, etc.)<br />
scotomas), but their cl<strong>in</strong>ical presentations may be variable. Unfortunately, CON may<br />
mimic the cl<strong>in</strong>ical presentation of toxic optic neuropathy, and neuroimag<strong>in</strong>g is recommended.<br />
The determ<strong>in</strong>ation of presumed toxic or nutritional optic neuropathy should<br />
<strong>in</strong>clude a complete evaluation to exclude other etiologies of bilateral, pa<strong>in</strong>less, and<br />
progressive optic neuropathies (e.g., hereditary optic neuropathy, bilateral compressive<br />
optic neuropathy, etc.). The evaluation of presumed toxic optic neuropathy is outl<strong>in</strong>ed<br />
<strong>in</strong> Table 1–11 (class IV, level C).<br />
Is There a History of Radiation Exposure to<br />
the Optic Nerves?<br />
Radiation optic neuropathy (RON) is thought to be an ischemic disorder of the optic<br />
nerve that usually results <strong>in</strong> irreversible severe visual loss months to years after<br />
radiation therapy to the bra<strong>in</strong> or orbit (Arnold, 1995; Borruat, 1993, 1996; Ebner, 1995;<br />
Girk<strong>in</strong>, 1997; Glantz, 1994; Goldsmith, 1992; Guy, 1991, 1995; Hudg<strong>in</strong>s, 1992; Jiang, 1994;<br />
Landau, 1996; Leber, 1998; Liu, 1992; McClellan, 1995; Parsons, 1994; Polak, 1995;<br />
Roden, 1990; Tachibana, 1990; Young, 1992; Zimmerman, 1990). It is most often a<br />
retrobulbar optic neuropathy, and thus the optic nerve may appear normal on <strong>in</strong>itial<br />
exam<strong>in</strong>ation. Approximately three fourths of patients have bilateral <strong>in</strong>volvement. The<br />
visual loss is characteristically rapid and progressive, with the disc becom<strong>in</strong>g pale over<br />
a period of 4 to 6 weeks. F<strong>in</strong>al vision is NLP <strong>in</strong> 45% and worse than 20=200 <strong>in</strong> an
Table 1–9. Etiologies for Toxic Optic <strong>Neuro</strong>pathy<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 15<br />
Common etiologies<br />
Ethambutol—tuberculosis therapy (Harcombe, 1991; Kumar, 1993;<br />
Russo, 1994; Schild, 1991; Seth, 1991; Thomas, 1994; Tsai, 1997)<br />
Ethanol and tobacco (tobacco alcohol amblyopia) (Danesh-Meyer, 2000; Sedwick, 1991, 1992)<br />
Less common etiologies<br />
Amantad<strong>in</strong>e—antiviral, Park<strong>in</strong>son’s disease<br />
Amiodarone (Cardarone)—cardiac disease (Macaluso, 1999; Sedwick, 1992; Speicher, 2000; Sreih,<br />
1999)<br />
Amoproxan—vasodilator and antiarrhythmic<br />
<strong>An</strong>il<strong>in</strong>e dyes<br />
Aspidium (male fern)<br />
Barbiturates—sedative, anticonvulsant<br />
Cafergot—headache<br />
Carbon disulfide—manufacture of viscose rayon fibers and cellophane films<br />
Carbon monoxide (Simmons, 1998)<br />
Carbon tetrachloride—manufactur<strong>in</strong>g of refrigerants and aerosols, dry-clean<strong>in</strong>g fluid, fat solvent,<br />
fire ext<strong>in</strong>guishers, <strong>in</strong>secticides, shampoo<br />
Cephalorid<strong>in</strong>e—antibiotic<br />
Chloramphenicol—antibiotic (Thomas, 1994)<br />
Chloronitrobenzene and d<strong>in</strong>itrobenzene—explosives<br />
Chlorpromaz<strong>in</strong>e (Thoraz<strong>in</strong>e)—antipsychotic<br />
Chlorpropamide (Diabenese)—diabetes<br />
Cimetid<strong>in</strong>e (Sa’adah, 1999)<br />
Ciprofloxac<strong>in</strong> (Cipro)—antibiotic (Vrabec, 1990)<br />
Cisplat<strong>in</strong> plus carboplat<strong>in</strong>—chemotherapy (Caraaceni, 1997)<br />
Cisplat<strong>in</strong> plus carmust<strong>in</strong>e—chemotherapy (Wang, 2000)<br />
Clioqu<strong>in</strong>ol—antibiotic<br />
Cobalt chloride<br />
Corticosteroids (Teus, 1991)<br />
Cyanide <strong>in</strong>toxication (dietary)<br />
Cyclospor<strong>in</strong>e—chemotherapy (Avery, 1991)<br />
D-penicillam<strong>in</strong>e—rheumatologic<br />
Deferoxam<strong>in</strong>e—for removal of excess iron <strong>in</strong> patients requir<strong>in</strong>g long-term transfusions (P<strong>in</strong>na,<br />
2001)<br />
Dichlorodiphenyltrichloroethane (DDT)—<strong>in</strong>secticide<br />
Digitalis (Digox<strong>in</strong>)—cardiac disease<br />
Diiodohydroxyqu<strong>in</strong>—amoebocide<br />
D<strong>in</strong>itrotoluene—explosive<br />
Disulfiram (<strong>An</strong>tabuse)—alcohol addiction<br />
Elcaton<strong>in</strong>—synthetic analogue of calciton<strong>in</strong> (Kimura, 1996)<br />
Emet<strong>in</strong>e—amoebocide<br />
Ethylchlorvynol (Placidyl)—hypnotic<br />
Ethylene glycol—antifreeze, moistener for tobacco, lacquer softener, solvent<br />
Etoposide phosphate and carboplat<strong>in</strong> (<strong>in</strong>tracarotid)—chemotherapy (Lauer, 1999)<br />
5-Fluorouracil—ant<strong>in</strong>eoplastic<br />
Gallium nitrate—ant<strong>in</strong>eoplastic (Csaky, 1997)<br />
(cont<strong>in</strong>ued)
16 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–9. (cont<strong>in</strong>ued)<br />
Halogenated hydroxyqu<strong>in</strong>olone (e.g., Clioqu<strong>in</strong>ol, boxyqu<strong>in</strong>olone [Colipar],<br />
chlorqu<strong>in</strong>adol [Sterox<strong>in</strong>], diiodohydroxyqu<strong>in</strong> [Diodoqu<strong>in</strong>], iodochlorhydroxyqu<strong>in</strong> [Entero-<br />
Vioform], Vioform)—gastro<strong>in</strong>test<strong>in</strong>al disorders<br />
Heavy metals (e.g., arsenic, lead, mercury)<br />
Hexachlorophene (Phiso-Hex)—detergent cleanser<br />
Iodoform—dis<strong>in</strong>fectant<br />
Iodopyracet (Diodrast)—radiologic contrast media<br />
Isoniazid (INH)—tuberculosis therapy (Thomas, 1994)<br />
Lead <strong>in</strong>toxication<br />
Lysol—dis<strong>in</strong>fectant<br />
Manganese—sk<strong>in</strong> exposure or <strong>in</strong>halation of fumes <strong>in</strong> pottery or electroplat<strong>in</strong>g <strong>in</strong>dustry (Lewis,<br />
2001)<br />
Melaton<strong>in</strong>, Zoloft (sertral<strong>in</strong>e), and a high-prote<strong>in</strong> diet (Lehman, 1999)<br />
Methamphetam<strong>in</strong>e (<strong>in</strong>tranasal abuse) (Wijaya, 1999)<br />
Methanol—wood alcohol, solvent, combustible, antifreeze, adulterant of alcohol (Sullivan-Mee,<br />
1998)<br />
Methotrexate—ant<strong>in</strong>eoplastic and rheumatologic (Johansson, 1992)<br />
Methyl acetate—solvent for nitrocellulose, res<strong>in</strong>s, and oils and manufacture of artificial leather<br />
Methyl bromide—fumigant, fire ext<strong>in</strong>guishers, refrigerant, <strong>in</strong>secticide<br />
Octamox<strong>in</strong>—monoam<strong>in</strong>e oxidase <strong>in</strong>hibitor<br />
Organophosphate pesticides<br />
Pamidronate—treatment of hypercalcemia (des Grottes, 1997)<br />
Penicillam<strong>in</strong>e (Cupram<strong>in</strong>e)—treatment of Wilson’s disease, rheumatologic diseases<br />
Phenazone (antipyr<strong>in</strong>e)—analgesic and antipyretic<br />
Phenipraz<strong>in</strong>e (Catron)—monoam<strong>in</strong>e oxidase <strong>in</strong>hibitor for hypertension and depression<br />
Plasmocid—antimalarial<br />
Qu<strong>in</strong><strong>in</strong>e—antimalarial, cramps<br />
Sodium fluoride<br />
Streptomyc<strong>in</strong>—tuberculosis therapy<br />
Styrene (v<strong>in</strong>yl benzyl)—synthetic rubber and fiberglass production<br />
Sulfonamides—antibiotics<br />
Tacrolimus (<strong>FK</strong> 506)—immunosuppressant (Brazis, 2000)<br />
Thallium—rodenticides and <strong>in</strong>secticides<br />
Tobutamide (Or<strong>in</strong>ase)—diabetes<br />
Toluene—glue sniff<strong>in</strong>g (Kiyokawa, 1999)<br />
Trichloroethylene—<strong>in</strong>dustrial solvent and degreas<strong>in</strong>g compound used <strong>in</strong> dry clean<strong>in</strong>g and <strong>in</strong><br />
manufacture of rubber<br />
Tricresyl phosphate—plasticizer and lubricant<br />
V<strong>in</strong>crist<strong>in</strong>e—ant<strong>in</strong>eoplastic agent<br />
additional 40% of affected eyes (i.e., 85% of eyes with RON have a f<strong>in</strong>al visual acuity of<br />
20=200 or worse). More rarely, RON may present as an anterior optic neuropathy with<br />
optic disc swell<strong>in</strong>g (Parsons, 1994). Such cases usually occur <strong>in</strong> the sett<strong>in</strong>g of radiation<br />
ret<strong>in</strong>opathy follow<strong>in</strong>g treatment of orbital or <strong>in</strong>traocular lesions. Associated f<strong>in</strong>d<strong>in</strong>gs of<br />
radiation ret<strong>in</strong>opathy resemble those of diabetic ret<strong>in</strong>opathy and variably <strong>in</strong>clude<br />
peripapillary hard exudates, hemorrhages, subret<strong>in</strong>al fluid, cotton-wool spots, focal
Table 1–10. Etiologies of Nutritional Optic <strong>Neuro</strong>pathy<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 17<br />
B 6 deficiency<br />
B 12 deficiency (e.g., pernicious anemia)<br />
Folate deficiency (Golnik, 1994a)<br />
Niac<strong>in</strong> deficiency<br />
Riboflav<strong>in</strong> deficiency<br />
Thiam<strong>in</strong>e (B 1)deficiency<br />
Iatrogenic malabsorption (e.g., post-biliopancreatic bypass procedure) (Smets, 1999)<br />
arteriolar narrow<strong>in</strong>g, macular edema, capillary nonperfusion, capillary telangiectasia,<br />
microaneurysms, neovascularization of disc and ret<strong>in</strong>a, perivascular sheath<strong>in</strong>g, vitreous<br />
hemorrhage, neovascular glaucoma, central ret<strong>in</strong>al artery occlusion, and central ret<strong>in</strong>al<br />
ve<strong>in</strong> occlusion. Loss of vision with anterior cases may be due to macular edema,<br />
macular hemorrhages, macular exudates, or perifoveal capillary nonperfusion, as<br />
well as from optic nerve <strong>in</strong>volvement. The cl<strong>in</strong>ical features of RON are outl<strong>in</strong>ed <strong>in</strong><br />
Table 1–12.<br />
The diagnosis of RON is suspected from the cl<strong>in</strong>ical sett<strong>in</strong>g and may usually be<br />
confirmed by magnetic resonance imag<strong>in</strong>g. In RON, the unenhanced T1- and T2weighted<br />
images show no abnormalities, but there is enhancement of the optic nerves,<br />
chiasm, and possibly the optic tracts <strong>in</strong> some cases (Borruat, 1993; Guy, 1991; Hudg<strong>in</strong>s,<br />
1992; McClellan, 1995; Tachibana, 1990; Young, 1992; Zimmerman, 1990). This enhancement<br />
usually resolves over several months. The differential diagnosis of RON is<br />
presented <strong>in</strong> Table 1–13. Recurrence of the primary tumor and radiation-<strong>in</strong>duced<br />
tumors must be especially considered.<br />
Patients with RON may rarely improve with corticosteroids. Hyperbaric oxygen<br />
therapy may be of benefit if given early <strong>in</strong> the course (e.g., with<strong>in</strong> 72 hours of onset of<br />
symptoms), although some patients show no improvement (Borruat, 1993, 1996; Liu,<br />
1992; Roden, 1990). <strong>An</strong>ticoagulation therapy was of no help <strong>in</strong> one case (Barbosa, 1999).<br />
There is no proven effective therapy for RON (class IV, level U).<br />
Table 1–11. Evaluation of Pa<strong>in</strong>less Progressive Bilateral Optic <strong>Neuro</strong>pathy (Presumed Toxic or<br />
Nutritional Optic <strong>Neuro</strong>pathy)<br />
Magnetic resonance imag<strong>in</strong>g of the optic nerves (exclude compressive optic neuropathy [CON])<br />
Vitam<strong>in</strong> B 12 level (serum)<br />
Folate level (serum and erythrocyte)<br />
Complete blood count with differential<br />
Ur<strong>in</strong>e heavy metal screen (mercury, lead, arsenic) if history suggestive<br />
Syphilis serology (e.g., rapid plasma reagent [RPR], fluorescent treponemal antibody absorption<br />
[FTA-ABS])<br />
Leber’s hereditary optic neuropathy mutational analysis<br />
Consider lumbar puncture and other laboratory studies (e.g., chest x-ray, ant<strong>in</strong>uclear antibody<br />
[ANA], sedimentation rate, angiotens<strong>in</strong>-1-convert<strong>in</strong>g enzyme [ACE], ant<strong>in</strong>eutrophil cytoplasmic<br />
antibody [ANCA], paraneoplastic antibody screen, etc.) if an <strong>in</strong>flammatory or <strong>in</strong>filtrative process is<br />
suspected
Table 1–12. <strong>Cl<strong>in</strong>ical</strong> Features of Radiation-Induced Optic <strong>Neuro</strong>pathy (RON)<br />
Acute onset of visual loss, gradual or rapidly progressive course<br />
Unilateral or bilateral ‘‘dimm<strong>in</strong>g’’ or ‘‘spotty vision’’; bilateral visual loss <strong>in</strong> 74%<br />
Variable acuity: 20=25–no light perception (NLP); often 20=200<br />
Variable color deficits<br />
Visual field loss<br />
Pa<strong>in</strong>less, progressive loss or constricted fields common<br />
May have altitud<strong>in</strong>al, central scotoma, junctional, or chiasmal field defects<br />
Monocular or b<strong>in</strong>ocular transient visual loss may precede by several weeks<br />
Fundus <strong>in</strong> RON<br />
Initially no optic nerve edema (or pallid edema); that is, almost always retrobulbar<br />
Rarely anterior but then associated with peripapillary hemorrhages and ret<strong>in</strong>al exudates,<br />
especially with radiation ret<strong>in</strong>opathy follow<strong>in</strong>g treatment of orbital or <strong>in</strong>traocular lesions<br />
Later atrophic nerve; may be pale <strong>in</strong>itially, optic atrophy over 4 to 6 weeks<br />
Occasional chiasmal syndrome<br />
Occasionally focal ret<strong>in</strong>al pigment epithelium (RPE) loss (choroidal compromise)<br />
<strong>Neuro</strong>imag<strong>in</strong>g<br />
Non-enhanced T1- and T2-weighted MRI images normal<br />
Often gadol<strong>in</strong>ium enhancement of optic nerves, chiasm, and even tracts<br />
Enhancement usually resolves <strong>in</strong> several months at which time visual function usually stabilizes<br />
Onset of visual loss<br />
Latency from radiation to onset of symptoms 1 to 144 months, with median delay 13 months after<br />
cessation of therapy<br />
Majority of cases occur with<strong>in</strong> 3 years of radiation therapy<br />
Occurs after radiation for paranasal s<strong>in</strong>us and other skull base malignancies, but also for pituitary<br />
adenomas, parasellar men<strong>in</strong>giomas, craniopharyngiomas, frontal or temporal gliomas, and<br />
<strong>in</strong>traocular tumors; rarely after radiation for thyroid orbitopathy (ma<strong>in</strong>ly <strong>in</strong> diabetics or patients<br />
with more than 2000–2500 cGy)<br />
Radiation dose<br />
Cumulative radiation dose of 2400–12,500 cGy<br />
75% have received total dose of 5000 or more cGy<br />
May occur with stereotactic radiosurgery (Girk<strong>in</strong>, 1997) if dosage to visual apparatus greater than<br />
8 Gy, large tumor volume, prior visual dysfunction, prior radiation exposure, or treatment<br />
isocenter with<strong>in</strong> 5 mm of anterior visual pathway<br />
Increased risk of RON<br />
Concomitant chemotherapy<br />
Patient with hormone secret<strong>in</strong>g pituitary adenoma<br />
Increased age<br />
Increased risk with <strong>in</strong>creased radiation exposure to optic nerve<br />
Treatment<br />
Unproven treatments<br />
Hyperbaric oxygen (HBO) if started early<br />
Corticosteroids<br />
Others (anticoagulation, antiplatelet agents)<br />
Prognosis<br />
Usually poor visual prognosis<br />
No light perception <strong>in</strong> 45%<br />
Worse than 20=200 <strong>in</strong> additional 40%<br />
85% have f<strong>in</strong>al visual acuity of 20=200 or worse<br />
Source: Arnold, 1995; Barbosa, 1999; Borruat, 1993, 1996; Ebner, 1995; Girk<strong>in</strong>, 1997; Glantz, 1994; Goldsmith, 1992;<br />
Gragoudas, 1999; Guy, 1991, 1995; Hudg<strong>in</strong>s, 1992; Jiang, 1994; Landau, 1996; Leber, 1998; Liu, 1992b; McClellan,<br />
1995; Mohamed, 2000; Parsons, 1994; Polak, 1995; Roden, 1990; Tachibana, 1990; Wijers, 1999; Toung, 1992;<br />
Zimmerman, 1990.<br />
18
Table 1–13. The Differential Diagnosis of Radiation Optic <strong>Neuro</strong>pathy<br />
Is There <strong>Cl<strong>in</strong>ical</strong> Evidence for a Hereditary<br />
Optic <strong>Neuro</strong>pathy?<br />
The Diagnosis of Optic <strong>Neuro</strong>pathies 19<br />
Recurrent tumor (ma<strong>in</strong> consideration!)<br />
Empty sella syndrome (arachnoiditis)<br />
Secondary new tumor <strong>in</strong> field of radiation (long latency 3 to 41 years; mean 15 years)<br />
Men<strong>in</strong>giomas or gliomas<br />
Dural tumors (e.g., fibrosarcoma)<br />
Cranial bone tumors (e.g., osteosarcoma)<br />
Peripheral nerve tumors (e.g., malignant schwannoma)<br />
<strong>Neuro</strong>fibromatosis type I and ataxia telangiectasia are risk factors<br />
Adhesive arachnoiditis<br />
Ischemic optic neuropathy<br />
Carc<strong>in</strong>omatous men<strong>in</strong>gitis<br />
Paraneoplastic optic neuropathy or ret<strong>in</strong>opathy<br />
Chemotherapy-related complications or toxicity<br />
Tamoxifen<br />
Cisplat<strong>in</strong>um<br />
Intraarterial bis 2-(chloroethyl)-1 nitrosourea (BCNU), etoposide phosphate and<br />
carboplat<strong>in</strong><br />
Primary optic nerve tumors<br />
Metastatic tumor<br />
Increased <strong>in</strong>tracranial pressure<br />
Venous s<strong>in</strong>us thrombosis<br />
Miller and Newman have divided the hereditary optic neuropathies <strong>in</strong>to three groups<br />
(Miller, 1998):<br />
1. Patients without associated neurologic signs and symptoms.<br />
2. Patients with neurologic signs and symptoms.<br />
3. Patients <strong>in</strong> whom the optic neuropathy is secondary to the underly<strong>in</strong>g systemic<br />
disease.<br />
The hereditary optic neuropathies may have an isolated, dom<strong>in</strong>ant (e.g., Kjer optic<br />
neuropathy), recessive, or mitochondrial (e.g., Leber’s hereditary optic neuropathy)<br />
<strong>in</strong>heritance pattern. The cl<strong>in</strong>ical features of Kjer autosomal-dom<strong>in</strong>ant optic neuropathy<br />
are outl<strong>in</strong>ed <strong>in</strong> Table 1–14, and the cl<strong>in</strong>ical features of Leber’s hereditary optic<br />
neuropathy are outl<strong>in</strong>ed <strong>in</strong> Table 1–15. We will not discuss other forms of hereditary<br />
optic neuropathy, as outl<strong>in</strong>ed <strong>in</strong> Table 1–16.<br />
Leber’s hereditary optic neuropathy (LHON) usually occurs <strong>in</strong> young males (up to 80<br />
to 90% of cases <strong>in</strong> the United States), although it may rarely occur <strong>in</strong> females and<br />
develop at any age (Ajax, 1998; Al-Salem, 1997; Bhatti, 1999; Cock, 1998; Hackett, 1997;<br />
Howell, 1997; Jacobson, 1998; Kerrison, 1997; MacMillan, 1998; Mashima, 1998; Purohit,<br />
1997; Saadati, 1998; Tsao, 1999).
20 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–14. The <strong>Cl<strong>in</strong>ical</strong> Features of Dom<strong>in</strong>ant Optic Atrophy (Kjer)<br />
Onset <strong>in</strong> first decade of life (usually 4 to 6 years) <strong>in</strong> 58% of patients<br />
Symptoms<br />
12.5% to 22.6% unaware of visual difficulties<br />
May be discovered to have optic atrophy as consequence of exam of another affected family<br />
member<br />
May have nystagmus<br />
Visual acuity<br />
20=20 to 20=60 <strong>in</strong> 40%<br />
20=200 to 20=600 <strong>in</strong> 15%<br />
Uncommonly hand motions or worse vision<br />
37% of patients 20=60 or better<br />
46% 20=60 to 20=200<br />
17% below 20=200<br />
Often <strong>in</strong>ability to perceive blue color (tritanopia) or generalized dyschromatopsia<br />
Central, paracentral, or cecocentral scotomas; may be characteristic <strong>in</strong>version of peripheral field, with<br />
the field be<strong>in</strong>g more constricted to blue isopters than red<br />
Optic atrophy: occasionally subtle; usually temporal; rarely diffuse<br />
Occasionally peripapillary atrophy, absent foveal reflex, mild macular pigmentary changes, arterial<br />
attenuation, and nonglaucomatous cupp<strong>in</strong>g<br />
Occasionally mental abnormality (10%) or hear<strong>in</strong>g loss<br />
Occasionally axonal, predom<strong>in</strong>antly sensory hereditary neuropathy (Chalmers, 1996)<br />
Visual evoked potential (VEP) may be reduced <strong>in</strong> amplitude and delayed<br />
Prognosis<br />
Visual prognosis is relatively good <strong>in</strong> Kjer’s dom<strong>in</strong>ant optic atrophy<br />
Stable or slow progression of visual loss<br />
Some families show<strong>in</strong>g evidence of l<strong>in</strong>kage to chromosome 3q and 18q<br />
Source: Bern<strong>in</strong>ger, 1991; Del Porto, 1994; Eigberg, 1994; Eliott, 1993; Johnston, 1997, 1999; Kerrison, 1999; Kjer,<br />
1996; Votruba, 1998.<br />
Table 1–15. <strong>Cl<strong>in</strong>ical</strong> Features of Leber’s Hereditary Optic <strong>Neuro</strong>pathy (LHON)<br />
Hereditary aspects<br />
‘‘Primary’’ mitochondrial DNA mutations (e.g., 11778, 3460, 14484)<br />
Mother’s egg sole provider of zygote’s cytoplasmic contents—mitochondria only extracellular<br />
source of DNA<br />
Every son and daughter of female carrier <strong>in</strong>herit LHON trait; only women pass trait<br />
Affected woman more likely to have affected children, especially daughters, than unaffected<br />
woman carrier<br />
20 to 83% of men at risk develop visual loss<br />
4 to 32% of women at risk develop visual loss<br />
Men affected more than women (80–90% males)<br />
Onset 13 to 35 years (range 5–80 years)<br />
Visual acuity loss<br />
Usually acute, rapid, unremitt<strong>in</strong>g, and pa<strong>in</strong>less<br />
Ultimately 20=200 to hand motions (20=20 to NLP range)<br />
Sequential bilateral <strong>in</strong>volvement (second eye <strong>in</strong> weeks to months later)<br />
Interval between onset <strong>in</strong> two eyes 0 to 15 months<br />
Simultaneous onset <strong>in</strong> both eyes 42–55%<br />
(cont<strong>in</strong>ued)
Table 1–15. (cont<strong>in</strong>ued)<br />
Rarely rema<strong>in</strong>s monocular<br />
Color vision severely affected<br />
Visual field loss (central or cecocentral scotomas; especially central 25–30 degrees)<br />
Occasionally positive Uhthoff’s phenomenon<br />
Fundus f<strong>in</strong>d<strong>in</strong>gs at the time of visual loss<br />
Small or absent cup (‘‘disc at risk’’) may predispose<br />
Triad of the suspect fundus<br />
Telangiectatic microangiopathy<br />
Apparent swell<strong>in</strong>g of nerve fiber layer around disc (‘‘pseudoedema’’)<br />
Fluoresce<strong>in</strong> angiogram often shows ‘‘pseudoedema’’ but may rarely see disc leakage<br />
Occasionally disc or ret<strong>in</strong>al hemorrhages, macular edema, or exudates<br />
Fundus appearance after visual loss<br />
Attenuated arterioles<br />
Nerve fiber layer loss especially papillomacular bundle<br />
Optic nerve pallor (temporally)<br />
May develop nonglaucomatous cupp<strong>in</strong>g<br />
MRI with acute visual loss may show <strong>in</strong>creased signal <strong>in</strong> middle or posterior <strong>in</strong>traorbital sections of<br />
optic nerves<br />
Prognosis—most patients rema<strong>in</strong> unchanged<br />
Some patients experience spontaneous improvement<br />
Improvement may occur gradually over 6 months to 1 year, or may suddenly improve up to 10<br />
years after onset<br />
Improved central vision—small island of vision with<strong>in</strong> large central scotoma<br />
Those that improve appear to have lower mean age of onset<br />
Better prognosis <strong>in</strong> mutation 14484 patients (37–65% improve) vs. mutation 11778 (4% improve)<br />
and mutation 3460 (22% improve) patients<br />
Associations<br />
Occasional cardiac conduction defects—Wolff-Park<strong>in</strong>son-White syndrome, Lawn-Ganong-Lev<strong>in</strong>e<br />
(LGL) syndrome, prolonged QT<br />
Dystonia described with 11778 and 3460 mutations<br />
Myoclonus with 11778 mutation (Carelli, 2001)<br />
Postural tremor occurs with <strong>in</strong>creased frequency <strong>in</strong> all forms<br />
Multiple sclerosis (MS)–like illness <strong>in</strong> up to 45% of females with 11778 mutation; rarely described<br />
<strong>in</strong> women with 3460 mutation or men with 11778 mutation<br />
Thoracic kyphosis <strong>in</strong> some patients with 3460 mutation<br />
Lesions <strong>in</strong> basal ganglia on MRI<br />
Spasticity<br />
Psychiatric disturbances<br />
Myelopathy<br />
Charcot-Marie-Tooth disease<br />
Treatment (class IV, level U)<br />
Medical therapy rema<strong>in</strong>s unproven<br />
Multivitam<strong>in</strong>s, folate, vitam<strong>in</strong> B 12, thiam<strong>in</strong>e 100 mg=day<br />
Coenzyme Q (Ubiqu<strong>in</strong>ate) 30 mg qid or 40 mg tid, ibed<strong>in</strong>one, and other coenzyme Q10 analogs<br />
Avoid alcohol, tobacco, and other environmental tox<strong>in</strong>s<br />
Discont<strong>in</strong>ue smok<strong>in</strong>g (Kerrison, 2000; Tsao, 1999)<br />
Consider electrocardiogram (ECG)<br />
Low vision assessment<br />
Source: Ajax, 1998; Al-Salem, 1997; Bhatti, 1999; Cock, 1998; Hackett, 1997; Howell, 1997; Jacobson, 1998;<br />
Kerrison, 1997, 2000; MacMillan, 1998; Mashima, 1998, 2000; Nakamura, 2000; Purohit, 1997; Saadati, 1998;<br />
Shaikh, 2001; Tsao, 1999; Vaphiades, 1999.<br />
21
22 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 1–16. Other Hereditary Optic <strong>Neuro</strong>pathies<br />
No associated neurologic deficits<br />
Congenital recessive optic atrophy (extremely rare; existence questioned)<br />
Apparent sex-l<strong>in</strong>ked optic atrophy<br />
Associated with other neurologic or systemic diseases<br />
Autosomal-dom<strong>in</strong>ant progressive optic atrophy with congenital deafness<br />
Autosomal-dom<strong>in</strong>ant progressive optic atrophy with progressive hear<strong>in</strong>g loss and ataxia<br />
Autosomal-dom<strong>in</strong>ant progressive optic atrophy with peripheral neuropathy<br />
Autosomal-dom<strong>in</strong>ant optic atrophy with ataxia and pes cavus<br />
Hereditary optic atrophy with progressive hear<strong>in</strong>g loss and polyneuropathy<br />
Familial bulbosp<strong>in</strong>al neuronopathy with optic atrophy<br />
Dom<strong>in</strong>ant optic atrophy, deafness, ophthalmoplegia, and myopathy<br />
Autosomal-recessive optic atrophy with progressive hear<strong>in</strong>g loss, spastic quadriplegia, mental<br />
deterioration, and death (opticocochleodentate degeneration)<br />
Opticoacoustic atrophy with dementia<br />
Sex-l<strong>in</strong>ked recessive optic atrophy, ataxia, deafness, tetraplegia, and areflexia<br />
Progressive encephalopathy with edema, hypsarrhythmia, and optic atrophy (PEHO syndrome)<br />
Juvenile diabetes <strong>in</strong>sipidus, diabetes mellitus, progressive optic atrophy, and deafness (Wolfram’s<br />
syndrome or DIDMOAD)<br />
Complicated hereditary <strong>in</strong>fantile optic atrophy (Behr’s syndrome)<br />
Optic atrophy with hereditary ataxias (Friedreich’s ataxia, Marie’s ataxia)<br />
Optic atrophy with Charcot-Marie-Tooth disease (hereditary sensorimotor neuropathy)<br />
Optic atrophy with myotonic muscular dystrophy (Gamez, 2001)<br />
Source: Barrett, 1997; Chalmers, 1996; Miller, 1998; Paradiso, 1996; Scold<strong>in</strong>g, 1996.<br />
Some patients with presumed ‘‘tobacco-alcohol amblyopia’’ or nutritional deficiency<br />
amblyopia may actual harbor a LHON mutation (Cullom, 1993; Purohit, 1997), and<br />
therefore test<strong>in</strong>g for Leber’s mutations may be <strong>in</strong>dicated <strong>in</strong> patients with presumed<br />
toxic or nutritional optic neuropathy. Although the diagnosis of LHON can be<br />
confirmed by serologic test<strong>in</strong>g for the known LHON mutations, little consensus exists<br />
regard<strong>in</strong>g the treatment of LHON. Some authors have recommended reduc<strong>in</strong>g metabolic<br />
stress on the optic nerve (e.g., discont<strong>in</strong>ue smok<strong>in</strong>g, alcohol use, known optic<br />
nerve tox<strong>in</strong>s, trauma, etc.). Medical therapy rema<strong>in</strong>s unproven, but some authors<br />
suggest supplementation with multivitam<strong>in</strong>s, folate, vitam<strong>in</strong> B 12, ibed<strong>in</strong>one and other<br />
coenzyme Q10 analogs, and thiam<strong>in</strong>e (class IV, level U).<br />
Is This an Atypical or Unexpla<strong>in</strong>ed Optic<br />
<strong>Neuro</strong>pathy?<br />
A number of patients with optic neuropathy do not fit <strong>in</strong>to the categories listed <strong>in</strong> our<br />
approach. For patients with unexpla<strong>in</strong>ed optic neuropathy or atypical optic neuropathy,<br />
a suggested evaluation is listed <strong>in</strong> Table 1–8.<br />
References<br />
Aarabi B, Haghshenas M, Rakeii V. (1998). Visual failure caused by suprasellar extramedullary hematopoiesis <strong>in</strong><br />
beta thalassemia: case report. <strong>Neuro</strong>surgery 42:922–925.
The Diagnosis of Optic <strong>Neuro</strong>pathies 23<br />
Acheson JF, Cockerell OC, Bentley CR, Sanders MD. (1993). Churg-Strauss vasculitis present<strong>in</strong>g with severe visual<br />
loss due to bilateral sequential optic neuropathy. Br J Ophthalmol 77:188–119.<br />
Achiron L, Strom<strong>in</strong>ger M, Witk<strong>in</strong> N, Prime S. (1995). Sarcoid optic neuropathy: a case report. J Am Optom Assoc<br />
66:646–651.<br />
Ahmadieh H, Roodpeyma S, Azarm<strong>in</strong>a M, Soheilian M, Sajjadi SH. (1994). Bilateral simultaneous optic neuritis <strong>in</strong><br />
childhood systemic lupus erythematosus. A case report. J <strong>Neuro</strong>-ophthalmol 14:84–86.<br />
Ajax ET, Kardon R. (1998). Late-onset Leber’s hereditary optic neuropathy. J <strong>Neuro</strong>-ophthalmol 18:30–31.<br />
Al-Mefty O, Kersh JE, Routh A, et al. (1990). The long term side effects of radiation therapy for benign bra<strong>in</strong><br />
tumors <strong>in</strong> adults. J <strong>Neuro</strong>surg 73:502–512.<br />
Al-Salem M. (1997). Leber’s congenital amaurosis <strong>in</strong> 22 affected members of one family. J Pediatr Ophthalmol<br />
Strabismus 34:254–257.<br />
Alvarez KL, Krop LC. (1993). Ethambutol-<strong>in</strong>duced ocular toxicity revisited. <strong>An</strong>n Pharmacother 27:102–103.<br />
Alvord EJ, Lofton S. (1988). Gliomas of the optic nerve or chiasm. Outcome by patient’s age, tumor site, and<br />
treatment. J <strong>Neuro</strong>surg 68:85–98.<br />
Amrith S, Baratham G, Khoo CY, et al. (1990). Spontaneous hematic cysts of the orbit present<strong>in</strong>g with acute<br />
proptosis. Ophthalmic Plast Reconstr Surg 6:273–277.<br />
Arnold AC. (1995). Radiation optic neuropathy. Presented at the North American <strong>Neuro</strong>-Ophthalmology meet<strong>in</strong>g,<br />
Tucson, Arizona.<br />
Arroyo JG, Lessell S, Montgomery WW. (1991). Steroid-<strong>in</strong>duced visual recovery <strong>in</strong> fibrous dysplasia. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:259–261.<br />
Arsava EM, Ulc K, Kansu T, et al. (2001). Granulomatous hypophysitis and bilateral optic neuropathy. J <strong>Neuro</strong>ophthalmol<br />
21:34–36.<br />
Avery R, Jabs DA, W<strong>in</strong>gard JR, et al. (1991). Optic disc edema after bone marrow transplantation. Possible role of<br />
cyclospor<strong>in</strong>e toxicity. Ophthalmology 98:1294–1301.<br />
Aylward GW, Sullivan TJ, Garner A, et al. (1995). Orbital <strong>in</strong>volvement <strong>in</strong> multifocal fibrosclerosis. Br J Ophthalmol<br />
79:246–249.<br />
Bakker SL, Hasan D, Bijvoet HW. (1999). Compression of the visual pathway by anterior cerebral artery<br />
aneurysm. Acta <strong>Neuro</strong>l Scand 99:204–207.<br />
Balch K, Phillips PH, Newman NJ. (1997). Pa<strong>in</strong>less orbital apex syndrome from mucormycosis. J <strong>Neuro</strong>-ophthalmol<br />
17:178–182.<br />
Barbosa AP, Carvalho D, Marques L, et al. (1999). Inefficiency of the anticoagulant therapy <strong>in</strong> the regression of the<br />
radiation-<strong>in</strong>duced optic neuropathy <strong>in</strong> Cush<strong>in</strong>g’s disease. J Endocr Invest 22:301–305.<br />
Barrett TG, Dundey SE, Fielder AR, Good PE. (1997). Optic atrophy <strong>in</strong> Wolfram (DIDMOAD) syndrome. Eye<br />
11:882–888.<br />
Barron CJ, Tepper L, Iov<strong>in</strong>g E. (1974). Ocular toxicity from ethambutol. Am J Ophthalmol 77:256–266.<br />
Beck AD, Newman NJ, Grossniklaus HE, et al. (1994). Optic nerve enlargement and chronic visual loss. Surv<br />
Ophthalmol 38:555–566.<br />
Belden CJ, Hamed LM, Mancuso AA. (1993). Bilateral isolated retrobulbar optic neuropathy <strong>in</strong> limited Wegener’s<br />
granulomatosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:119–123.<br />
Bern<strong>in</strong>ger TA, Jaeger W, Krastel M. (1991). Electrophysiology and colour perimetry <strong>in</strong> dom<strong>in</strong>ant <strong>in</strong>fantile optic<br />
atrophy. Br J Ophthalmol 75:49–53.<br />
Bhatti MT, Holder CA, Newman NJ, Hudg<strong>in</strong>s PA. (2000). MR characteristics of musl<strong>in</strong>-<strong>in</strong>duced optic neuropathy:<br />
report of two cases and review of the literature. AJNR 21:346–352.<br />
Bhatti MT, Newman NJ. (1999). A multiple sclerosis-like illness <strong>in</strong> a man harbor<strong>in</strong>g the mtDNA 14484 mutation. J<br />
<strong>Neuro</strong>-ophthalmol 19:28–33.<br />
Bland LI, Marchese MJ, McDonald JV. (1992). Acute monocular bl<strong>in</strong>dness secondary to fibrous dysplasia of the<br />
skull: a case report. <strong>An</strong>n Ophthalmol 24:263–266.<br />
Bocca G, de Vries J, Cruysberg JR, et al. (1998). Optic neuropathy <strong>in</strong> McCune-Albright syndrome: an <strong>in</strong>dication for<br />
aggressive treatment. Acta Paediatr 87:599–600.<br />
Borruat F-X, Schatz NJ, Glaser JS, Feun LG, Matos L. (1993). Visual recovery from radiation-<strong>in</strong>duced optic<br />
neuropathy. The role of hyperbaric oxygen therapy. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:98–101.<br />
Borruat F-X, Schatz NJ, Glaser JS, et al. (1996). Radiation optic neuropathy: report of cases, role of hyperbaric<br />
oxygen, and literature review. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:255–266.<br />
Botella C, Orozco M, Navarro J, Riesgo P. (1994). Idiopathic chronic hypertrophic craniocervical pachymen<strong>in</strong>gitis:<br />
case report. <strong>Neuro</strong>surgery 35:1144–1149.<br />
Bourne RR, Dol<strong>in</strong> PJ, Mtanda AT, et al. (1998). Epidemic optic neuropathy <strong>in</strong> primary school children <strong>in</strong> Dar es<br />
Salaam, Tanzania. Br J Ophthalmol 82:232–234.<br />
Brazis PW, Lee AG. (1998). <strong>Neuro</strong>-ophthalmic problems caused by medications. Focal Po<strong>in</strong>ts 16:1–13.<br />
Brazis PW, Menke DM, McLeish WM, et al. (1995). <strong>An</strong>giocentric T-cell lymphoma present<strong>in</strong>g with multiple cranial<br />
nerve palsies and retrobulbar optic neuropathy. J <strong>Neuro</strong>-ophthalmol 15:152–157.
24 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Brazis PW, Spivey JR, Boll<strong>in</strong>g JP, Steers JL. (2000). A case of bilateral optic neuropathy <strong>in</strong> a patient on Tacrolimus<br />
(<strong>FK</strong> 506) therapy after liver transplantation. Am J Ophthalmol 129:536–538.<br />
Brodovsky S, ten Hove MW, P<strong>in</strong>kerton RM, et al. (1997). <strong>An</strong> enhanc<strong>in</strong>g optic nerve lesion: malignant glioma of<br />
adulthood. Can J Ophthalmol 32:409–413.<br />
Bronte-Stewart J, Pettigrew AR, Foulds WS. (1976). Toxic optic neuropathy and its experimental production. Trans<br />
Ophthalmol Soc UK 96:355–358.<br />
Brown DM, Kimura AE, Oss<strong>in</strong>ig KC, We<strong>in</strong>er GJ. (1992a). Acute promyelocytic <strong>in</strong>filtration of the optic nerve<br />
treated by oral trans-ret<strong>in</strong>oic acid. Ophthalmology 99:1463–1467.<br />
Brown MD, Voljavec AS, Lott MT, et al. (1992b). Mitochondrial DNA complex I and III mutations associated with<br />
Leber’s hereditary optic neuropathy. Genetics 130:163–173.<br />
Brown P, Demaerel P, McNaught A, et al. (1994). <strong>Neuro</strong>-ophthalmological presentation of non-<strong>in</strong>vasive<br />
Aspergillus s<strong>in</strong>us disease <strong>in</strong> the non-immunocompromised host. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:234–237.<br />
Brummit ML, Kl<strong>in</strong>e LB, Wilson ER. (1992). Craniopharyngioma: pitfalls <strong>in</strong> diagnosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
12:77–81.<br />
Burde REM, Sav<strong>in</strong>o PJ, Trobe JD. (1992). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. St. Louis, Mosby-Year Book, pp.<br />
56–57.<br />
Buus DR, Tse DT, Farris BK. (1990). Ophthalmic complications of s<strong>in</strong>us surgery. Ophthalmology 97:612–619.<br />
Cacciatori M, L<strong>in</strong>g CS, Dhillon B. (1996). Retrobulbar neuritis <strong>in</strong> a patient with acquired immune deficiency<br />
syndrome. Acta Ophthalmol Scand 74:194–196.<br />
Caldemeyer KS, Smith RR, Edwards-Brown MK. (1995). Familial hypophosphatemic rickets caus<strong>in</strong>g ocular<br />
calcification and optic canal narrow<strong>in</strong>g. AJNR 16:1252–1254.<br />
Camera A, Piccirillo G, Cennamo G, et al. (1993). Optic nerve <strong>in</strong>volvement <strong>in</strong> acute lymphoblastic leukemia. Leuk<br />
Lymphoma 11:153–155.<br />
Caraaceni A, Mart<strong>in</strong>i C, Spatti G, et al. (1997). Recover<strong>in</strong>g optic neuritis dur<strong>in</strong>g systemic cisplat<strong>in</strong> and carboplat<strong>in</strong><br />
chemotherapy. Acta <strong>Neuro</strong>l Scand 96:260–261.<br />
Carelli V, Valent<strong>in</strong>o ML, Liguori R, et al. (2001). Leber’s hereditary optic neuropathy (LHON=1178) with<br />
myoclonus: report of two cases. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 71:813–816.<br />
Carmody RF, Mafee MF, Goodw<strong>in</strong> JA, et al. (1994). Orbital and optic pathway sarcoidosis: MR f<strong>in</strong>d<strong>in</strong>gs. AJNR<br />
15:775–783.<br />
Carter K, Lee AG, Tang RA, et al. (1998). <strong>Neuro</strong>-ophthalmologic complications of s<strong>in</strong>us surgery. <strong>Neuro</strong><strong>ophthalmology</strong><br />
19:75–82.<br />
Chalmers RM, Bird AC, Hard<strong>in</strong>g AE. (1996). Autosomal dom<strong>in</strong>ant optic atrophy with asymptomatic peripheral<br />
neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:195–196.<br />
Chandra S, Vashisht S, Menon V, et al. (2000). Optic nerve cysticercosis: imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs. AJNR 21:198–200.<br />
Chateil JF, Soussotte C, Pedespan JM, et al. (2001). MRI and cl<strong>in</strong>ical differences between optic pathway tumours <strong>in</strong><br />
children with and without neurofibromatosis. Br J Radiol 74:24–31.<br />
Chen YR, Breidahl A, Chang CN. (1997). Optic nerve decompression <strong>in</strong> fibrous dysplasia: <strong>in</strong>dications, efficacy,<br />
and safety. Plast Reconstr Surg 99:22–30.<br />
Choi SY, Hwang JM. (1997). Optic neuropathy associated with ethambutol <strong>in</strong> Koreans. Korean J Ophthalmol 11:106–<br />
110.<br />
Citron KM, Thomas GO. (1986). Ocular toxicity from ethambutol. Thorax 41:737–739.<br />
Cock HR, Tabrizi SJ, Cooper JM, Schapira AHV. (1998). The <strong>in</strong>fluence of nuclear background on the biochemical<br />
expression of 3460 Leber’s hereditary optic neuropathy. <strong>An</strong>n <strong>Neuro</strong>l 44:187–193.<br />
Cohen SB, Glasgow BT. (1993). Bilateral optic nerve cryptococcosis <strong>in</strong> sudden bl<strong>in</strong>dness <strong>in</strong> patients with acquired<br />
immune deficiency syndrome. Ophthalmology 100:1689–1694.<br />
Colap<strong>in</strong>to EV, Cabeen MA, Johnson LN. (1996). Optic nerve compression by a dolichoectatic <strong>in</strong>ternal carotid<br />
artery: case report. <strong>Neuro</strong>surgery 39:604–606.<br />
Costagliola C, R<strong>in</strong>aldi M, Cotticelli L, et al. (1992). Isolated optic nerve <strong>in</strong>volvement <strong>in</strong> chronic myeloid leukemia.<br />
Leuk Res 16:411–413.<br />
Cramer SC, Glaspy JA, Efird JT, Louis DN. (1996). Chronic lymphocytic leukemia and the central nervous system.<br />
A cl<strong>in</strong>ical and pathological study. <strong>Neuro</strong>logy 46:19–25.<br />
Créange A, Zeller J, Rosta<strong>in</strong>g-Rigattieri S, et al. (1999). <strong>Neuro</strong>logical complications of neurofibromatosis type 1 <strong>in</strong><br />
adulthood. Bra<strong>in</strong> 122:473–481.<br />
Cross SA, Salomao D, Lennon VA. (2002). A paraneoplastic syndrome of comb<strong>in</strong>ed optic neuritis and ret<strong>in</strong>itis<br />
def<strong>in</strong>ed serologically by CRMP-5-IgG. Presented at the 28th annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-<br />
Ophthalmology Society, Copper Mounta<strong>in</strong>, Colorado, February 9–14.<br />
Crotty TB, Scheithauer BW, Young WF Jr, et al. (1995). Papillary craniopharyngioma: a cl<strong>in</strong>icopathological study<br />
of 48 cases. J <strong>Neuro</strong>surg 83:206–214.<br />
Csaky KG, Caruso RC. (1997). Gallium nitrate optic neuropathy. Am J Ophthalmol 124:567–568.
The Diagnosis of Optic <strong>Neuro</strong>pathies 25<br />
Cullom ME, Heher KL, Miller NR, et al. (1993). Leber’s hereditary optic neuropathy masquerad<strong>in</strong>g as tobaccoalcohol<br />
amblyopia. Arch Ophthalmol 111:1482.<br />
Cumm<strong>in</strong>gs TJ, Provenzale JM, Hunter SB, et al. (2000). Gliomas of the optic nerve: histological, immunohistochemical<br />
(MIB-1 and p53), and MRI analysis. Acta <strong>Neuro</strong>pathol 99:563–570.<br />
Cunliffe IA, Moffat DA, Hardy DG, Moore AT. (1992). Bilateral optic nerve sheath men<strong>in</strong>giomas <strong>in</strong> a patient with<br />
neurofibromatosis type 2. Br J Ophthalmol 76:310–312.<br />
Daly BD, Chow CC, Cockram CS. (1994). Unusual manifestations of craniofacial dysplasia: cl<strong>in</strong>ical, endocr<strong>in</strong>ological,<br />
and computed tomographic features. Postgrad Med J 70:10–16.<br />
Danesh-Meyer H, Kubis KC, Sergott RC, et al. (1999). Not so progressive visual loss. Surv Ophthalmol 44:247–252.<br />
Danesh-Meyer H, Kubis KC, Wolf MA, Lessell S. (2000). Chiasmopathy? Surv Ophthalmol 44:329–335.<br />
DeBroff BM, Donahue SP. (1993). Bilateral optic neuropathy as the <strong>in</strong>itial manifestation of systemic sarcoidosis.<br />
Am J Ophthalmol 116:108–111.<br />
Deliganis AV, Geyer JR, Berger MS. (1996). Prognostic significance of type I neurofibromatosis (von Reckl<strong>in</strong>ghausen<br />
disease) <strong>in</strong> childhood optic glioma. <strong>Neuro</strong>surgery 38:1114–1118.<br />
Del Porto G, V<strong>in</strong>golo EM, Ste<strong>in</strong>dl K, et al. (1994). <strong>Cl<strong>in</strong>ical</strong> heterogeneity of dom<strong>in</strong>ant optic atrophy: the<br />
contribution of visual function <strong>in</strong>vestigations to diagnosis. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 32:717–727.<br />
DeMonte F, Smith HK, Al-Mefty O. (1994). Outcome of aggressive removal of cavernous s<strong>in</strong>us men<strong>in</strong>giomas. J<br />
<strong>Neuro</strong>surg 81:245–251.<br />
des Grottes JM, Schrooyen M, Dumon JC, Body JJ. (1997). Retrobulbar optic neuritis after pamidronate<br />
adm<strong>in</strong>istration <strong>in</strong> a patient with a history of cutaneous porphyria. Cl<strong>in</strong> Rheumatol 16:93–95.<br />
DiMario FJ, Ramsby G, Greenaste<strong>in</strong> R, et al. (1993). <strong>Neuro</strong>fibromatosis type I: magnetic resonance imag<strong>in</strong>g<br />
f<strong>in</strong>d<strong>in</strong>gs. J Child <strong>Neuro</strong>l 8:32–39.<br />
D<strong>in</strong>owitz M, Leen JS, Hameed M, et al. (2001). Sudden pa<strong>in</strong>less visual loss. Surv Ophthalmol 46:143–148.<br />
Dolman PJ, Glazer LC, Harris GJ, et al. (1991). Mechanisms of visual loss <strong>in</strong> severe proptosis. Ophthalmic Plast<br />
Reconstruct Surg 7:256–260.<br />
Drake JM, Joy M, Goldenberg A, et al. (1991). Computer and robot assisted resection of thalamic astrocytomas <strong>in</strong><br />
children. <strong>Neuro</strong>surgery 29:27–31.<br />
Dunker S, Reuter U, Rösler A, et al. (1996). Optic nerve <strong>in</strong>filtration <strong>in</strong> well-differentiated B-cell lymphoma.<br />
Ophthalmology 93:351–353.<br />
Dunn DW, Purv<strong>in</strong> V. (1990). Optic pathway gliomas <strong>in</strong> neurofibromatosis. Dev Med Child <strong>Neuro</strong>l 32:820–824.<br />
Dutton JJ. (1991). Optic nerve gliomas and men<strong>in</strong>giomas. <strong>Neuro</strong>l Cl<strong>in</strong> 9:163–177.<br />
Dutton JJ. (1992). Optic nerve sheath men<strong>in</strong>giomas. Surv Ophthalmol 37:167–183.<br />
Dutton JJ. (1994). Gliomas of the anterior visual pathway. Surv Ophthalmol 38:427–452.<br />
Dutton JJ, Tse DT, <strong>An</strong>derson RL. (1983). Compressive optic neuropathy follow<strong>in</strong>g use of <strong>in</strong>tracranial oxidized<br />
cellulose hemostat. Ophthalmic Surg 14:487–490.<br />
Ebner R, Slamovits TL, Friedlamd S, Pearlman JL, Fowble B. (1995). Visual loss follow<strong>in</strong>g treatment of sphenoid<br />
s<strong>in</strong>us cancer. Surv Ophthalmol 40:62–68.<br />
Edelste<strong>in</strong> C, Goldberg RA, Rub<strong>in</strong>o G. (1998). Unilateral bl<strong>in</strong>dness after ipsilateral prophylactic transcranial optic<br />
canal decompression for fibrous dysplasia. Am J Ophthalmol 126:469–471.<br />
Eigberg H, Kjer B, Kjer P, et al. (1994). Dom<strong>in</strong>ant optic atrophy (OPA 1) mapped to chromosome 3q region. I.<br />
L<strong>in</strong>kage analysis. Hum Mol Genet 3:977–980.<br />
Eliott D, Traboulski EI, Maumenee IH. (1993). Visual prognosis <strong>in</strong> autosomal dom<strong>in</strong>ant optic atrophy (Kjer type).<br />
Am J Ophthalmol 115:360–367.<br />
El-Mahdy W, Powell M. (1998). Transsphenoidal management of 28 symptomatic Rathke’s cleft cysts, with special<br />
reference to visual and hormonal recovery. <strong>Neuro</strong>surgery 42:7–17.<br />
Epste<strong>in</strong> MA, Packer RJ, Rorke LB, et al. (1992). Vascular malformation with radiation vasculopathy after treatment<br />
of chiasmatic hypothalamic glioma. Cancer 70:887–893.<br />
Fahlbusch R, Honegger J, Paulus W, et al. (1999). Surgical treatment of craniopharyngiomas: experience with 168<br />
patients. J <strong>Neuro</strong>surg 90:237–250.<br />
Fayaz I, Gentili F, MacKenzie IR. (1999). Optic nerve sheath men<strong>in</strong>gioma. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
67:408.<br />
Fierz AB, Sartoretti S, Thoelen AM. (2001). Optic neuropathy and central ret<strong>in</strong>al artery occlusion <strong>in</strong> non-<br />
Hodgk<strong>in</strong>’s lymphoma. J <strong>Neuro</strong>-ophthalmol 21:102–105.<br />
F<strong>in</strong>eman MS, Augsburger JJ, Lee AG, <strong>An</strong>drews DW. (1999). A new approach to an old problem. Surv Ophthalmol<br />
43:519–524.<br />
Forman S, Rosenbaum PS. (1998). Lymphomatoid granulomatosis present<strong>in</strong>g as an isolated unilateral optic<br />
neuropathy. A cl<strong>in</strong>icopathologic report. J <strong>Neuro</strong>-<strong>ophthalmology</strong> 18:150–152.<br />
Freilich RJ, Krol G, De<strong>An</strong>gelis LM. (1995). <strong>Neuro</strong>imag<strong>in</strong>g and cerebrosp<strong>in</strong>al fluid cytology <strong>in</strong> the diagnosis of<br />
leptomen<strong>in</strong>geal metastasis. <strong>An</strong>n <strong>Neuro</strong>l 38:51–57.
26 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Friedman JM, Birch P. (1997). <strong>An</strong> association between optic glioma and other tumours of the central nervous<br />
system <strong>in</strong> neurofibromatosis type I. <strong>Neuro</strong>pediatrics 28:131–132.<br />
Fuss M, Hug EB, Schaefer RA, et al. (1999). Proton radiation therapy (PRT) for pediatric optic pathway gliomas:<br />
comparison with 3D planned conventional photons and standard photon technique. Int J Radiat Oncol Biol<br />
Phys 45:1117–1126.<br />
Galetta SL, Stadtmauer EA, Hicks DG, et al. (1991). Reactive lymphohistiocytosis with recurrence <strong>in</strong> the optic<br />
chiasm. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 9:25–30.<br />
Gamez J, Montane D, Martorell L, et al. (2001). Bilateral optic nerve atrophy <strong>in</strong> myotonic dystrophy. Am J<br />
Ophthalmol 131:398–400.<br />
Garrity JA, Trautmann JC, Bartley GB, et al. (1990). Optic nerve sheath men<strong>in</strong>goceles. <strong>Cl<strong>in</strong>ical</strong> and radiographic<br />
features <strong>in</strong> 13 cases with a review of the literature. Ophthalmology 97:1519–1531.<br />
Garvey M, Packer RJ. (1996). <strong>An</strong> <strong>in</strong>tegrated approach to the treatment of chiasmatic hypothalamic gliomas. J<br />
<strong>Neuro</strong>-oncol 28:167–183.<br />
Gayre GS, Scott IU, Feuer W, et al. (2001). Long-term visual outcome <strong>in</strong> patients with anterior visual pathway<br />
gliomas. J <strong>Neuro</strong>-ophthalmol 21:1–7.<br />
Girk<strong>in</strong> CA, Comey CH, Lunsford D, et al. (1997). Radiation optic neuropathy after stereotactic radiosurgery.<br />
Ophthalmology 104:1634–1643.<br />
Girk<strong>in</strong> CA, Perry JD, Miller NR, Reich SG. (1998). Pachymen<strong>in</strong>gitis with multiple cranial neuropathies and<br />
unilateral optic neuropathy secondary to Pseudomonas aerug<strong>in</strong>osa. Case report and review. J <strong>Neuro</strong>-ophthalmol<br />
18:196–200.<br />
Glantz MJ, Burger PC, Friedman AH, et al. (1994). Treatment of radiation-<strong>in</strong>duced nervous system <strong>in</strong>jury with<br />
hepar<strong>in</strong> and warfar<strong>in</strong>. <strong>Neuro</strong>logy 44:2020–2027.<br />
Goldberg S, Mahadevia P, Lipton M, Rosenbaum PS. (1998). S<strong>in</strong>us histiocytosis with massive lymphadenopathy<br />
<strong>in</strong>volv<strong>in</strong>g orbit: reversal of compressive optic neuropathy after chemotherapy. J <strong>Neuro</strong>-Ophthalmol 18:270–<br />
275.<br />
Goldsmith BJ, Rosenthal SA, Wara WM, et al. (1992). Optic neuropathy after irradiation of men<strong>in</strong>gioma. Radiology<br />
185:71–76.<br />
Goldsmith BJ, Wara WM, Wilson CB, Larson DA. (1994a). Postoperative irradiation for subtotally resected<br />
men<strong>in</strong>giomas. A retrospective analysis of 140 patients treated from 1967 to 1990. J <strong>Neuro</strong>surg 80:195–201.<br />
Goldsmith BJ, Wara WM, Wilson CB, et al. (1994b). Postoperative irradiation of subtotally resected men<strong>in</strong>giomas.<br />
J <strong>Neuro</strong>surg 80:195–201.<br />
Golnik KC, Schaible ER. (1994a). Folate-responsive optic neuropathy. J <strong>Neuro</strong>-ophthalmol 14:163–169.<br />
Golnik KC, Hund PW, Stroman GA, Stewart WC. (1996). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> patients with<br />
unexpla<strong>in</strong>ed optic neuropathy. Ophthalmology 103:515–520.<br />
Golnik KC, Marotto ME, Fanous MM, et al. (1994b). Ophthalmic manifestations of Rochalimaea species. Am J<br />
Ophthalmol 118:145–151.<br />
Gragoudas ES, Li W, Lane AM, et al. (1999). Risk factors for radiation maculopathy and papillopathy after<br />
<strong>in</strong>traocular irradiation. Ophthalmology 106:1571–1578.<br />
Grill J, Couanet D, Cappelli C, et al. (1999). Radiation-<strong>in</strong>duced cerebral vasculopathy <strong>in</strong> children with<br />
neurofibromatosis and optic pathway glioma. <strong>An</strong>n <strong>Neuro</strong>l 45:393–396.<br />
Grimm MA, Hazelton T, Beck RW, et al. (1995). Postgadol<strong>in</strong>ium enhancement of a compressive neuropathy of the<br />
optic nerve. AJNR 16:779–781.<br />
Grunberg SM, Weiss MH, Spitz IM, et al. (1991). Treatment of unresectable men<strong>in</strong>giomas with the antiprogesterone<br />
agent mifepristone. J <strong>Neuro</strong>surg 74:861–866.<br />
Gulliani BP, Dadeya S, Malik KPS, Ja<strong>in</strong> DC. (2001). Bilateral cysticercosis of the optic nerve. J <strong>Neuro</strong>-ophthalmol<br />
21:217–218.<br />
Gurha N, Sood A, Dhar J, Gupta S. (1999). Optic nerve cysticercosis <strong>in</strong> the optic canal. Acta Ophthalmol Scand<br />
77:107–109.<br />
Guy J, Mancuso A, Beck R, et al. (1991). Radiation-<strong>in</strong>duced optic neuropathy: a magnetic resonance imag<strong>in</strong>g<br />
study. J <strong>Neuro</strong>surg 74:426–432.<br />
Guy J, Mancuso A, Quisl<strong>in</strong>g RG, et al. (1990). Gadol<strong>in</strong>ium-DTPA-enhanced magnetic resonance imag<strong>in</strong>g <strong>in</strong> optic<br />
neuropathies. Ophthalmology 97:592–600.<br />
Guy J, Schatz NJ. (1995). Radiation-<strong>in</strong>duced optic neuropathy. In: Tusa RJ, Newman SA, eds. <strong>Neuro</strong>-ophthalmological<br />
Disorders. New York, Marcel Dekker, pp. 437–450.<br />
Guyer DR, Green WR, Schachat AP, et al. (1990). Bilateral ischemic optic neuropathy and ret<strong>in</strong>al vascular<br />
occlusions associated with lymphoma and sepsis. Ophthalmology 97:882–888.<br />
Hackett SE. (1997). Leber’s hereditary optic neuropathy: a genetic disorder of the eye. Insight 22:94–96.<br />
Hamilton SR, Smith CH, Lessell S. (1993). Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
13:127–134.
The Diagnosis of Optic <strong>Neuro</strong>pathies 27<br />
Hao SP. (1994). Mucocele of the sphenoid s<strong>in</strong>us with acute bilateral bl<strong>in</strong>dness: report of two cases. J Formosan Med<br />
Assoc 93:519–521.<br />
Harcombe A, K<strong>in</strong>near W, Britton J, Macfarlane J. (1991). Ocular toxicity of ethambutol. Respir Med 85:151–153.<br />
Hedges TR. (1995). Two brothers with bilateral optic neuropathy. Surv Ophthalmol 39:417.<br />
Hirsch WL, Sekhar LN, Lanz<strong>in</strong>o G, et al. (1993). Men<strong>in</strong>giomas <strong>in</strong>volv<strong>in</strong>g the cavernous s<strong>in</strong>us: value of imag<strong>in</strong>g for<br />
predict<strong>in</strong>g surgical complications. AJR 160:1083–1088.<br />
Hoffman HJ, Humphreys RP, Drake JM, et al. (1993). Optic pathway=hypothalamic gliomas: a dilemma <strong>in</strong><br />
management. Pediatr <strong>Neuro</strong>surg 19:186–195.<br />
Honegger J, Buchfelder M, Fahlbusch R. (1999). Surgical treatment of craniopharyngiomas: endocr<strong>in</strong>ological<br />
results. J <strong>Neuro</strong>surg 90:251–257.<br />
Horton JC, Garcia EC, Becker EK. (1992). Magnetic resonance imag<strong>in</strong>g of leukemic <strong>in</strong>vasion of the optic nerve.<br />
Arch Ophthalmol 110:1207–1208.<br />
Howell N. (1997). Leber hereditary optic neuropathy: mitochondrial mutations and degeneration of the optic<br />
nerve. Vision Res 37:3495–3507.<br />
Howell N, Kubacka I, Halvorson S, Mackey D. (1993). Leber’s hereditary optic neuropathy: the etiological role of<br />
a mutation <strong>in</strong> the mitochondrial cytochrome b gene. Genetics 133:133–136.<br />
Hudg<strong>in</strong>s PA, Newman NJ, Dillon WP, et al. (1992). Radiation-<strong>in</strong>duced optic neuropathy: characteristic appearance<br />
on gadol<strong>in</strong>ium-enhanced MR. AJNR 13:235–238.<br />
Huopenen, et al. (1991). A new mtDNA mutation associated with Leber’s hereditary optic neuroret<strong>in</strong>opathy. Am J<br />
Hum Genet 48:1147.<br />
Hutnik CML, Nicolle DA, Munoz DG. (1997). Orbital aspergillosis. A fatal masquerader. J <strong>Neuro</strong>-ophthalmol<br />
17:257–261.<br />
Imes RK, Hoyt WF. (1991). Magnetic resonance imag<strong>in</strong>g signs of optic nerve gliomas <strong>in</strong> neurofibromatosis. Am J<br />
Ophthalmol 111:729–734.<br />
Ing EB, Augsburger JJ, Eagle RC. (1996). Lung cancer with visual loss. Surv Ophthalmol 40:505–510.<br />
Ing EB, Garrity JA, Cross SA, Ebersold MJ. (1997). Sarcoid masquerad<strong>in</strong>g as optic nerve sheath men<strong>in</strong>gioma. Mayo<br />
Cl<strong>in</strong> Proc 72:38–43.<br />
Ishikawa T, Ito T, Shoji E, Inukai K. (2000). Compressive optic nerve atrophy result<strong>in</strong>g from a distorted <strong>in</strong>ternal<br />
carotid artery. Pediatr <strong>Neuro</strong>l 22:322–324.<br />
Jacobson DM. (1999). Symptomatic compression of the optic nerve by the carotid artery. <strong>Cl<strong>in</strong>ical</strong> profile of 18<br />
patients with 24 affected eyes identified by magnetic resonance imag<strong>in</strong>g. Ophthalmology 106:1994–2004.<br />
Jacobson DM, <strong>An</strong>derson DR, Rupp GM, Warner JJ. (1996). Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis:<br />
cl<strong>in</strong>ical-radiological-pathological correlation of bone <strong>in</strong>volvement. J <strong>Neuro</strong>-ophthalmol 16:264–268.<br />
Jacobson DM, Stone EM, Miller NR, et al. (1998). Relative afferent pupillary defects <strong>in</strong> patients with Leber<br />
hereditary optic neuropathy and unilateral visual loss. Am J Ophthalmol 126:291–295.<br />
Janss AJ, Grundy R, Cnaan A, et al. (1995). Optic pathway and hypothalamic=chiasmatic gliomas <strong>in</strong> children<br />
younger than 5 years with a 6-year follow-up. Cancer 75:1052–1059.<br />
Jenk<strong>in</strong> D, <strong>An</strong>gyalfi S, Becker L, et al. (1993). Optic glioma <strong>in</strong> children, surveillance or irradiation? Int J Radiat Oncol<br />
Biol Phys 25:215–225.<br />
Jiang GL, Tucker SL, Guttenberger R, et al. (1994). Radiation-<strong>in</strong>duced <strong>in</strong>jury to the visual pathway. Radiother Oncol<br />
30:17–25.<br />
Jimenez-Lucho VE, Del Bustro R, Odel J. (1987). Isoniazid and ethambutol as a cause of optic neuropathy. Eur J<br />
Respir Dis 71:42–45.<br />
Johansson BA. (1992). Visual field defects dur<strong>in</strong>g low-dose methotrexate therapy. Doc Ophthalmol 79:91.<br />
Johns DR, Heher KL, Miller NR, Smith KH. (1993). LHON: cl<strong>in</strong>ical manifestations of the 14484 mutation. Arch<br />
Ophthalmol 111:495–498.<br />
Johns DR, Neufeld MJ. (1993). Cytochrome c oxidase mutations <strong>in</strong> Leber’s hereditary optic neuropathy. Biochem<br />
Biophys Res Commun 196:810–815.<br />
Johns DR, Smith KH, Miller NR. (1992). Leber’s hereditary optic neuropathy. <strong>Cl<strong>in</strong>ical</strong> manifestations of the 3460<br />
mutation. Arch Ophthalmol 110:1577–1581.<br />
Johns DR, Smith KH, Sav<strong>in</strong>o PJ, Miller NR. (1993). Leber’s hereditary optic neuropathy. <strong>Cl<strong>in</strong>ical</strong> manifestations of<br />
the 15257 mutation. Ophthalmology 100:981–986.<br />
Johnson TE, Casiano RR, Kronish JW, et al. (1999). S<strong>in</strong>o-orbital aspergillosis <strong>in</strong> acquired immunodeficiency<br />
syndrome. Arch Ophthalmol 117:57–64.<br />
Johnston RL, Burdon MA, Spalston DJ, et al. (1997). Dom<strong>in</strong>ant optic atrophy, Kjer type. L<strong>in</strong>kage analysis and<br />
cl<strong>in</strong>ical features <strong>in</strong> a large British pedigree. Arch Ophthalmol 115:100–103.<br />
Johnston RL, Seller MJ, Behnam JT, et al. (1999). Dom<strong>in</strong>ant optic atrophy. Ref<strong>in</strong><strong>in</strong>g the cl<strong>in</strong>ical diagnostic criteria <strong>in</strong><br />
light of genetic l<strong>in</strong>kage studies. Ophthalmology 106:123–128.
28 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Joseph MP. (1995). Commentary on Papay FA, Morales L Jr, Flaharty P, et al. Optic nerve decompression <strong>in</strong> cranial<br />
base fibrous dysplasia. J Craniofac. Surg 6:11–13.<br />
Kattah JC, Chrousos GC, Roberts J, et al. (1993). Metastatic prostate cancer to the optic canal. Ophthalmology<br />
100:1711–1715.<br />
Katz B. (1991). Disc edema, transient obscurations of vision, and a temporal fossa mass. Surv Ophthalmol 36:133–<br />
139.<br />
Katz BJ, Nerad JA. (1998). Ophthalmic manifestations of fibrous dysplasia. A disease of children and adults.<br />
Ophthalmology 105:2207–2215.<br />
Kawano Y, Kira JJ. (1995). Chronic hypertrophic cranial pachymen<strong>in</strong>gitis associated with HTLV-I <strong>in</strong>fection. J<br />
<strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 59:435–437.<br />
Kazim M, Kennerdell JS, Rothfus W, Marquardt M. (1992). Orbital lymphangioma. Correlation of magnetic<br />
resonance images and <strong>in</strong>traoperative f<strong>in</strong>d<strong>in</strong>gs. Ophthalmology 99:1588–1594.<br />
Kerrison JB, Arnould VJ, Ferraz Sallum JM, et al. (1999). Genetic heterogeneity of dom<strong>in</strong>ant optic atrophy, Kjer<br />
type. Identification of a second locus on chromosome 18q12.2-12.3. Arch Ophthalmol 117:805–810.<br />
Kerrison JB, Howell N, Miller NR, et al. (1995). Leber hereditary optic neuropathy: electron microscopy and<br />
molecular genetic analysis of a case. Ophthalmology 102:1509–1516.<br />
Kerrison JB, Miller NR, Hsu F-C, et al. (2000). A case-control study of tobacco and alcohol consumption <strong>in</strong> Leber<br />
hereditary optic neuropathy. Am J Ophthalmol 130:803–812.<br />
Kerrison JB, Newman NJ. (1997). <strong>Cl<strong>in</strong>ical</strong> spectrum of Leber’s hereditary optic neuropathy. Cl<strong>in</strong> <strong>Neuro</strong>sci 4:295–<br />
301.<br />
Kestle JRW, Hoffman HJ, Mock AR. (1993). Moya moya phenomenon after radiation for optic glioma. J <strong>Neuro</strong>surg<br />
79:32–35.<br />
Kimura H, Masai H, Kashii S. (1996). Optic neuropathy follow<strong>in</strong>g elcaton<strong>in</strong> therapy. J <strong>Neuro</strong>-ophthalmol 16:134–<br />
136.<br />
K<strong>in</strong>jo T, Al-Mefty O, Ciric I. (1995). Diaphragma sellae men<strong>in</strong>giomas. <strong>Neuro</strong>surgery 36:1082–1092.<br />
Kiyokawa M, Mizota A, Takasoh M, Adachi-Usami E. (1999). Pattern visual evoked cortical potentials <strong>in</strong> patients<br />
with toxic optic neuropathy caused by toluene abuse. Jpn J Ophthalmol 43:438–442.<br />
Kjer B, Eiberg H, Kjer P, et al. (1996). Dom<strong>in</strong>ant optic atrophy mapped to chromosome 3q region. II. <strong>Cl<strong>in</strong>ical</strong> and<br />
epidemiological aspects. Acta Ophthalmol Scand 74:3–7.<br />
Kl<strong>in</strong>k DF, Sampath P, Miller NR, et al. (2000). Long-term visual outcome after nonradical microsurgery <strong>in</strong> patients<br />
with parasellar and cavernous s<strong>in</strong>us men<strong>in</strong>giomas. <strong>Neuro</strong>surgery 47:24–32.<br />
Kodsi SR, Younge BR, Leavitt JA, et al. (1993). Intracranial plasma cell granuloma present<strong>in</strong>g as an optic<br />
neuropathy. Surv Ophthalmol 38:70–74.<br />
Kosmorsky GS, Prayson R. (1996). Primary optic pathway sarcoidosis <strong>in</strong> a 38-year-old white man. J <strong>Neuro</strong>ophthalmol<br />
16:188–190.<br />
Kotapka MJ, Kalia KK, Mart<strong>in</strong>ez AJ, Sekhar LN. (1994). Infiltration of the carotid artery by cavernous s<strong>in</strong>us<br />
men<strong>in</strong>gioma. J <strong>Neuro</strong>surg 81:252–255.<br />
Kovalic JJ, Grigsby PW, Shepard MJ, et al. (1990). Radiation therapy for gliomas of the optic nerve and chiasm. Int<br />
J Radiat Oncol Biol Phys 18:927–932.<br />
Kumar A, Sandramouli S, Verma L, Tewari HK, Khosla PK. (1993). Ocular ethambutol toxicity: Is it reversible? J<br />
Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:15–17.<br />
Lam BL, Barret DA, Glaser JS, et al. (1994). Visual loss from idiopathic <strong>in</strong>tracranial pachymen<strong>in</strong>gitis. <strong>Neuro</strong>logy<br />
44:694–698.<br />
Landau K, Killer HE. (1996). Radiation damage. <strong>Neuro</strong>logy 88:9.<br />
Larson JJ, van Loveren HR, Balko G, Tew JM Jr. (1995). Evidence of men<strong>in</strong>gioma <strong>in</strong>filtration <strong>in</strong>to cranial nerves:<br />
cl<strong>in</strong>ical implications for cavernous s<strong>in</strong>us men<strong>in</strong>giomas. J <strong>Neuro</strong>surg 83:596–599.<br />
Lauer AK, Wobig JL, Shults WT, et al. (1999). Severe ocular and orbital toxicity after <strong>in</strong>tracarotid etoposide<br />
phosphate and carboplat<strong>in</strong> therapy. Am J Ophthalmol 127:230–233.<br />
Leber KA, Berglöff J, Pendl G. (1998). Dose-response tolerance of the visual pathways and cranial nerves of the<br />
cavernous s<strong>in</strong>us to stereotactic radiosurgery. J <strong>Neuro</strong>surg 88:43–50.<br />
Lee AG, Cech DA, Rose JE, et al. (1997a). Recurrent visual loss due to musl<strong>in</strong>-<strong>in</strong>duced optochiasmatic<br />
arachnoiditis. <strong>Neuro</strong>-<strong>ophthalmology</strong> 18:199–204.<br />
Lee AG, Dutton JJ. (1999). A practice pathway for the management of gliomas of the anterior visual pathway: an<br />
update and evidence-based approach. <strong>Neuro</strong>-<strong>ophthalmology</strong> 22:139–155.<br />
Lee AG, Phillips PH, Newman NJ, et al. (1997b). <strong>Neuro</strong>-ophthalmologic manifestations of adenoid cystic<br />
carc<strong>in</strong>oma. J <strong>Neuro</strong>-ophthalmol 17:183–188.
The Diagnosis of Optic <strong>Neuro</strong>pathies 29<br />
Lee AG, Woo SY, Miller NR, et al. (1996). Improvement <strong>in</strong> visual function <strong>in</strong> an eye with a presumed optic nerve<br />
sheath men<strong>in</strong>gioma after treatment with three-dimensional conformal radiation therapy. J <strong>Neuro</strong>-ophthalmol<br />
16:247–251.<br />
Lee Wan W, Geller JL, Feldon SE, Sadun AA. (1990). Visual loss caused by rapidly progressive <strong>in</strong>tracranial<br />
men<strong>in</strong>giomas dur<strong>in</strong>g pregnancy. Ophthalmology 97:18–21.<br />
Lehman NL, Johnson LN. (1999). Toxic optic neuropathy after concomitant use of melaton<strong>in</strong>, Zoloft, and highprote<strong>in</strong><br />
diet. J <strong>Neuro</strong>-ophthalmol 19:232–234.<br />
Leibold JE. (1966). The ocular toxicity of ethambutol and its relationship to dose. <strong>An</strong>n NY Acad Sci 135:904–909.<br />
Lessell S. (1998). Nutritional amblyopia. J <strong>Neuro</strong>-ophthalmol 18:106–111.<br />
Lesser RL, Kornmehl EW, Pachner AR, et al. (1990). <strong>Neuro</strong>-ophthalmologic manifestations of Lyme disease.<br />
Ophthalmology 97:699–706.<br />
Lev<strong>in</strong> LA, Jakobiec FA. (1992). Optic nerve tumors of childhood: a decision-analytical approach to their diagnosis.<br />
Int Ophthalmol Cl<strong>in</strong> 32:223–240.<br />
Lev<strong>in</strong>e MR, Kaye L, Mair S, Bates J. (1993). Multifocal fibrosclerosis. Report of a case of bilateral idiopathic<br />
scleros<strong>in</strong>g pseudotumor and retroperitoneal fibrosis. Arch Ophthalmol 111:841–843.<br />
Lewis JR. (2001). Bilateral optic neuropathy secondary to manganese toxicity. Presented at the 27th <strong>An</strong>nual<br />
Meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Rancho Mirage, California, February 18–22.<br />
Lieberman FS, Odel J, Hirsh J, et al. (1999). Bilateral optic neuropathy with IgGk multiple myeloma improved<br />
after myeloablative chemotherapy. <strong>Neuro</strong>logy 52:414–416.<br />
Listernick R, Charrow J, Greenwald M. (1992). Emergence of optic pathway gliomas <strong>in</strong> children with neurofibromatosis<br />
type I after normal neuroimag<strong>in</strong>g results. J Pediatr 121:584–587.<br />
Listernick R, Charrow J, Greenwald M, et al. (1994). Optic gliomas I children with neurofibromatosis type I. J<br />
Pediatr 125:63–66.<br />
Listernick R, Louis DN, Packer RJ, Gutmann DH. (1997). Optic pathway gliomas <strong>in</strong> children with neurofibromatosis<br />
1: consensus statement from the NF1 Optic Pathway Glioma Task Force. <strong>An</strong>n <strong>Neuro</strong>l 41:143–149.<br />
Liu GT, Lessell S. (1992a). Spontaneous visual improvement <strong>in</strong> chiasmal gliomas. Am J Ophthalmol 114:193–201.<br />
Liu GT, Schmandt SM, Packer RJ. (2001). Visual loss <strong>in</strong> childhood. Surv Ophthalmol 46:35–42.<br />
Liu JL. (1992b). <strong>Cl<strong>in</strong>ical</strong> analysis of radiation optic neuropathy. Ch<strong>in</strong> J Ophthalmol 28:86–88.<br />
Loehrl TA, Leopold DA. (2000). Sphenoethmoidal mucocele present<strong>in</strong>g with bilateral visual compromise. <strong>An</strong>n<br />
Otol Rh<strong>in</strong>ol Laryngol 109:608–610.<br />
Luiz JE, Lee AG, Keltner JL, et al. (1998). Paraneoplastic optic neuropathy and autoantibody production <strong>in</strong> smallcell<br />
carc<strong>in</strong>oma of the lung. J <strong>Neuro</strong>-ophthalmol 18:187–181.<br />
Lundsford LD. (1994). Contemporary management of men<strong>in</strong>gioma: radiation therapy as an adjuvant and<br />
radiosurgery as an alternative to surgical removal? J <strong>Neuro</strong>surg 80:187–190.<br />
Macaluso DC, Shults WT, Fraunfelder FT. (1999). Features of amiodarone-<strong>in</strong>duced optic neuropathy. Am J<br />
Ophthalmol 127:610–612.<br />
Mackey DA, Buttery RC. (1992). LHON <strong>in</strong> Australia. Aust NZ J Ophthalmol 20:179.<br />
MacMillan C, Kirkham T, Fu K, et al. (1998). Pedigree analysis of French Canadian families with 14484C Leber’s<br />
hereditary optic neuropathy. <strong>Neuro</strong>logy 50:417–422.<br />
Mafee MF, Goodw<strong>in</strong> J, Dorodi S. (1999). Optic nerve sheath men<strong>in</strong>giomas. Role of MR imag<strong>in</strong>g. Radiol Cl<strong>in</strong> North<br />
Am 37:37–58.<br />
Ma<strong>in</strong>i R, Macewen CJ. (1997). Intracranial plasmacytoma present<strong>in</strong>g with optic nerve compression. Br J<br />
Ophthalmol 81:417–418.<br />
Malik S, Furlan AJ, Sweeney PJ, Kosmorsky GS, Wong M. (1992). Optic neuropathy: a rare paraneoplastic<br />
syndrome. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:137–141.<br />
Mamelak AN, Kelly WM, Davis RL, Rosenblum ML. (1993). Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis.<br />
Report of three cases. J <strong>Neuro</strong>surg 79:270–276.<br />
Maroon JC, Kennerdell JS, Vidovich DV, et al. (1994). Recurrent spheno-orbital men<strong>in</strong>gioma. J <strong>Neuro</strong>surg 80:202–<br />
208.<br />
Mashima Y, Hiida Y, Oguchi Y. (1992). Remission of Leber’s hereditary optic neuropathy with idebenone. Lancet<br />
340:368–369.<br />
Mashima Y, Hiida Y, Oguchi Y. (1995). Lack of differences among mitochondrial DNA <strong>in</strong> family members with<br />
Leber’s hereditary optic neuropathy and differ<strong>in</strong>g visual outcomes. J <strong>Neuro</strong>ophthalmol 15:15–19.<br />
Mashima Y, Kigasawa K, Wakakura M, Oguchi Y. (2000). Do idebenone and vitam<strong>in</strong> therapy shorten the time to<br />
achieve visual recovery <strong>in</strong> Leber hereditary optic neuropathy? J <strong>Neuro</strong>-ophthalmol 20:166–170.<br />
Mashima Y, Oshitari K, Imamura Y, et al. (1998). Orbital high resolution magnetic resonance imag<strong>in</strong>g with fast<br />
sp<strong>in</strong> echo <strong>in</strong> the acute stage of Leber’s hereditary optic neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 64:124–127.
30 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
McClellan RL, El Gammal T, Kl<strong>in</strong>e LB. (1995). Early bilateral radiation-<strong>in</strong>duced optic neuropathy with follow-up<br />
MRI. <strong>Neuro</strong>radiology 37:131–133.<br />
McFadzean R, Brosnahan D, Doyle D, et al. (1994). A diagnostic quartet <strong>in</strong> leptomen<strong>in</strong>geal <strong>in</strong>filtration of the optic<br />
nerve sheath. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 14:175–182.<br />
Michael CB, Lee AG, Patr<strong>in</strong>ely JR, et al. (2000). Visual loss associated with fibrous dysplasia of the anterior skull<br />
base. Case report and review of the literature. J <strong>Neuro</strong>surg 92:350–354.<br />
Miller NR, Newman NJ. (1998). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 5th ed. Baltimore, Williams &<br />
Wilk<strong>in</strong>s.<br />
Miller NR, Sav<strong>in</strong>o PJ, Schneider T. (1995). Rapid growth of an <strong>in</strong>tracranial aneurysm caus<strong>in</strong>g apparent retrobulbar<br />
optic neuritis. J <strong>Neuro</strong>-ophthalmol 15:212–218.<br />
Misra M, Mohanty AB, Rath S. (1991). Giant aneurysm of the <strong>in</strong>ternal carotid artery present<strong>in</strong>g features of<br />
retrobulbar neuritis. Ind J Ophthalmol 1:28–29.<br />
Moghrabi A, Friedman HS, Burger PC, et al. (1993). Carboplat<strong>in</strong> treatment of progressive optic pathway gliomas<br />
to delay radiotherapy. J <strong>Neuro</strong>surg 79:223–227.<br />
Mohamed IG, Roa W, Fulton D, et al. (2000). Optic nerve sheath fenestration for a reversible optic neuropathy <strong>in</strong><br />
radiation oncology. Am J Cl<strong>in</strong> Oncol 23:401–405.<br />
Moorthy RS, Yung CW, Nunery WR, et al. (1992). Spontaneous orbital subperiosteal hematomas <strong>in</strong> patients with<br />
liver disease. Ophthalmic Plast Reconstr Surg 8:150–152.<br />
Moyer PD, Golnik KC, Breneman J. (2000). Treatment of optic nerve sheath men<strong>in</strong>gioma with three-dimensional<br />
conformal radiation. Am J Ophthalmol 129:694–696.<br />
Muci-Mendoza R, Aravalo JF, Ramella M, et al. (1999). Optociliary ve<strong>in</strong>s <strong>in</strong> optic nerve sheath men<strong>in</strong>gioma.<br />
Indocyan<strong>in</strong>e green videoangiography f<strong>in</strong>d<strong>in</strong>gs. Ophthalmology 106:311–318.<br />
Muthukumar N. (1997). Traumatic hemorrhagic optic neuropathy: case report. Br J <strong>Neuro</strong>surg 11:166–167.<br />
Nakae K, Yamamoto S, Shigematsu I, Kono R. (1973). Relation between subacute myelo-optic neuropathy<br />
(SMON) and clioqu<strong>in</strong>ol: nationwide survey. Lancet 1:171–173.<br />
Nakajima T, Kumabe T, Jokura H, Yoshimoto T. (2001). Recurrent germ<strong>in</strong>oma <strong>in</strong> the optic nerve: report of two<br />
cases. <strong>Neuro</strong>surgery 48:214–218.<br />
Nakamura M, Yamamoto M. (2000). Variable pattern of visual recovery of Leber’s hereditary optic neuropathy. Br<br />
J Ophthalmol 84:534–535.<br />
Newman NJ. (1993). Leber’s hereditary optic neuropathy: new genetic considerations. Arch <strong>Neuro</strong>l 50:540–<br />
548.<br />
Newman NJ, Grossniklaus HE, Wojno TH. (1996). Breast carc<strong>in</strong>oma metastatic to the optic nerve. Arch Ophthalmol<br />
114:102–103.<br />
Newman NJ, Lott MT, Wallace DC. (1994). The cl<strong>in</strong>ical characteristics of pedigrees of Leber’s hereditary optic<br />
neuropathy with the 11778 mutation. Am J Ophthalmol 111:750.<br />
Newman SJ. (1994). Men<strong>in</strong>giomas: a quest for the optimum therapy. J <strong>Neuro</strong>surg 80:191–194.<br />
Newsom RSB, Simcock P, Zambarakji H. (1999). Cerebral metastasis present<strong>in</strong>g with altitud<strong>in</strong>al field defect. J<br />
<strong>Neuro</strong>-ophthalmol 19:10–11.<br />
Nishio S, Taheshita I, Fujiwara S, et al. (1993). Optico-hypothalamic glioma: an analysis of 16 cases. Childs Nerv<br />
Syst 9:334–338.<br />
Nishizaki T, Iwamoto F, Uesugi S, et al. (1997). Idiopathic cranial pachymen<strong>in</strong>goencephalitis focally affect<strong>in</strong>g the<br />
parietal dura mater and adjacent bra<strong>in</strong> parenchyma: case report. <strong>Neuro</strong>surgery 40:840–843.<br />
Noda S, Hayasaka S, Setogawa T. (1993). Intraocular lymphoma <strong>in</strong>vades the optic nerve and orbit. <strong>An</strong>n<br />
Ophthalmol 25:30–34.<br />
Nygaard R, Garwicz S, Haldorsen T, et al. (1991). Second malignant neoplasms <strong>in</strong> patients treated for childhood<br />
leukemia. Acta Pediatr Scand 80:1220–1228.<br />
Oaks W. (1990). Recent experience with resection of pilocytic astrocytomas of the hypothalamus. Concepts Pediatr<br />
<strong>Neuro</strong>surg 10:108–117.<br />
Olmos PR, Falko JM, Rea GL, et al. (1993). Fibros<strong>in</strong>g pseudotumor of the sella and parasellar area produc<strong>in</strong>g<br />
hypopituitarism and multiple cranial nerve palsies. <strong>Neuro</strong>surgery 32:1015–1021.<br />
Oohira A, Inoue T, Fukuda N, Uchida K-I. (1991). A case with paraneoplastic optic neuropathy present<strong>in</strong>g<br />
bitemporal hemianopsia. <strong>Neuro</strong>-<strong>ophthalmology</strong> 11:325–328.<br />
Ortiz JR, Newman NJ, Barrow DL. (1991). CREST-associated multiple <strong>in</strong>tracranial aneurysms and bilateral optic<br />
neuropathies. Ophthalmology 11:233–240.<br />
Packer RJ, Ater JC, Phillips P, et al. (1994). Efficacy of chemotherapy for children with newly diagnosed<br />
progressive low-grade glioma (abstract). <strong>An</strong>n <strong>Neuro</strong>l 36:496.<br />
Packer R, Lange B, Ater J, et al. (1993). Carboplat<strong>in</strong> and v<strong>in</strong>crist<strong>in</strong>e for recurrent and newly diagnosed low grade<br />
gliomas of childhood. J Cl<strong>in</strong> Oncol 11:850–860.
The Diagnosis of Optic <strong>Neuro</strong>pathies 31<br />
Paradiso G, Micheli F, Taratuto AL, Parera IC. (1996). Familial bulbosp<strong>in</strong>al neuronopathy with optic atrophy: a<br />
dist<strong>in</strong>ct entity. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 61:196–199.<br />
Park KL, Go<strong>in</strong>s KM. (1993). Hodgk<strong>in</strong>’s lymphoma of the orbit associated with acquired immunodeficiency<br />
syndrome. Am J Ophthalmol 116:111–112.<br />
Parney IF, Johnson ES, Allen PBR. (1997). ‘‘Idiopathic’’ cranial hypertrophic pachymen<strong>in</strong>gitis responsive to<br />
antituberculous therapy: case report. <strong>Neuro</strong>surgery 41:965–971.<br />
Parsa CF, Hoyt CS, Lesser RL, et al. (2001). Spontaneous regression of optic gliomas. Thirteen cases documented<br />
by serial neuroimag<strong>in</strong>g. Arch Ophthalmol 119:516–529.<br />
Parsons JT, Bova FJ, Fitzgerald CR, et al. (1994). Radiation optic neuropathy after megavoltage external-beam<br />
irradiation: analysis of time-dose factors. Int J Radiat Oncol Biol Phys 30:755–763.<br />
Patankar T, Prasad S, Krishnan A, Laxm<strong>in</strong>arayan R. (2000). Isolated optic nerve pseudotumor. Australas Radiol<br />
44:101–103.<br />
Peele KA, Kennerdell JS, Maroon JC, et al. (1996). The role of postoperative irradiation <strong>in</strong> the management of<br />
sphenoid w<strong>in</strong>g men<strong>in</strong>giomas. A prelim<strong>in</strong>ary report. Ophthalmology 103:1761–1767.<br />
Pelton RW, Lee AG, Orengo-Nania SD, Patr<strong>in</strong>ely JR. (1999). Bilateral optic disk edema caused by sarcoidosis<br />
mimick<strong>in</strong>g pseudotumor cerebri. Am J Ophthalmol 127:229–230.<br />
Pengel J, Crevits L, Wynants P, et al. (1997). Optic nerve metastasis simulat<strong>in</strong>g optic neuritis. Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
99:46–49.<br />
Petito CK. (1996). Craniopharyngioma: prognostic importance of histologic features. AJNR 17:1441–1442.<br />
Petronio J, Edwards M, Prados M, et al. (1991). Management of chiasmal and hypothalamic gliomas of <strong>in</strong>fancy<br />
and childhood with chemotherapy. J <strong>Neuro</strong>surg 74:701–708.<br />
Pierce SM, Barnes PD, Loeffler JS, et al. (1990). Def<strong>in</strong>itive radiation therapy <strong>in</strong> the management of symptomatic<br />
patients with optic glioma. Survival and long term effects. Cancer 65:45–52.<br />
Pierro L, Brancato R, Zagnanelli E, et al. (1992). Ocular <strong>in</strong>volvement <strong>in</strong> acute lymphoblastic leukemia: an<br />
echographic study. Int Ophthalmol 16:159–162.<br />
P<strong>in</strong>na A, Corda L, Carta F. (2001). Rapid recovery with oral z<strong>in</strong>c sulphate <strong>in</strong> deferoxam<strong>in</strong>e-<strong>in</strong>duced presumed<br />
optic neuropathy and hear<strong>in</strong>g loss. J <strong>Neuro</strong>-ophthalmol 21:32–33.<br />
Polak BCP, Wijngaarde R. (1995). Radiation neuropathy <strong>in</strong> patients with both diabetes mellitus and ophthalmic<br />
Graves’ disease. Orbit 14:71–74.<br />
Purohit SS, Tomsak RL. (1997). Nutritional deficiency amblyopia or Leber’s hereditary optic neuropathy? <strong>Neuro</strong><strong>ophthalmology</strong><br />
18:111–116.<br />
Rao GP, Blyth CP, Jefferys RV. (1995). Ophthalmic manifestations of Rathke’s cleft cysts. Am J Ophthalmol 119:86–<br />
91.<br />
Rizzo JF III, Lessell S. (1991). Optic neuritis and ischemic optic neuropathy: overlapp<strong>in</strong>g cl<strong>in</strong>ical problems. Arch<br />
Ophthalmol 109:1668–1672.<br />
Rizzo JF III, Lessell S. (1993). Tobacco amblyopia. Am J Ophthalmol 116:84–88.<br />
Roden D, Bosley TM, Fowble B, et al. (1990). Delayed radiation <strong>in</strong>jury to the retrobulbar optic nerves and chiasm.<br />
Ophthalmology 97:346–351.<br />
Rodriguez LA, Edwards MSB, Lev<strong>in</strong> VA. (1990). Management of hypothalamic gliomas <strong>in</strong> children: an analysis of<br />
33 cases. <strong>Neuro</strong>surgery 26:242–247.<br />
Rootman J, McCarthy M, White V, et al. (1994). Idiopathic scleros<strong>in</strong>g <strong>in</strong>flammation of the orbit. A dist<strong>in</strong>ct<br />
cl<strong>in</strong>icopathologic entity. Ophthalmology 101:570–584.<br />
Rosenbaum JT, Simpson T, Neuwelt CM. (1997). Successful treatment of optic neuropathy <strong>in</strong> association with<br />
systemic lupus erythematosus us<strong>in</strong>g <strong>in</strong>travenous cyclophosphamide. Br J Ophthalmol 81:130–132.<br />
Roth DB, Siatkowski RM. (2000). Bilateral bl<strong>in</strong>dness as the <strong>in</strong>itial presentation of lymphoma of the sphenoid s<strong>in</strong>us.<br />
Am J Ophthalmol 129:256–258.<br />
Rub<strong>in</strong>ste<strong>in</strong> AB, Loven D, Greir A, et al. (1994). Hormone receptors <strong>in</strong> <strong>in</strong>itially excised versus recurrent <strong>in</strong>tracranial<br />
men<strong>in</strong>giomas. J <strong>Neuro</strong>surg 81:184–187.<br />
Russo PA, Changlasian MA. (1994). Toxic optic neuropathy associated with ethambutol: implications for current<br />
therapy. J Am Optom Assoc 65:332–338.<br />
Sa’adah MA, Al Salem M, Ali AS, et al. (1999). Cimetid<strong>in</strong>e-associated optic neuropathy. Eur <strong>Neuro</strong>l 42:23–26.<br />
Saadati HG, Hsu HY, Heller KB, Sadun AA. (1998). A histopathologic and morphometric differentiation of nerves<br />
<strong>in</strong> optic nerve hypoplasia and Leber hereditary optic neuropathy. Arch Ophthalmol 116:911–916.<br />
Sadun AA, Weiss MH. (1993). Reversal of visual losses follow<strong>in</strong>g RU 486 therapy for men<strong>in</strong>gioma. Presented at<br />
the 19th annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Big Sky, Montana.<br />
Saito K, Suzuki Y, Nehashi K, et al. (1990). Unilateral extradural approach for bilateral optic canal release <strong>in</strong> a<br />
patient with fibrous dysplasia. Surg <strong>Neuro</strong>l 34:124–128.
32 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Samples JR, Younge BR. (1981). Tobacco-alcohol amblyopia. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:213–218.<br />
Savy LE, Moseley IF. (1996). Intracranial arterial calcification and ectasia <strong>in</strong> visual failure. Br J Radiol 69:394–401.<br />
Schaffler GJ, Simbrunner J, Lechner H, et al. (2000). Idiopathic sclerotic <strong>in</strong>flammation of the orbit with left optic<br />
nerve compression <strong>in</strong> a patient with multifocal fibrosclerosis. AJNR 21:194–197.<br />
Schild HS, Fox BC. (1991). Rapid-onset reversible ocular toxicity from ethambutol therapy. Am J Med 90:404–406.<br />
Scold<strong>in</strong>g NJ, Kellar-Wood HF, Shaw C, Shneerson JM, <strong>An</strong>toun N. (1996). Wolfram syndrome: hereditary diabetes<br />
mellitus with bra<strong>in</strong>stem and optic atrophy. <strong>An</strong>n <strong>Neuro</strong>l 39:352–360.<br />
Sedwick LA. (1991). The perils of Paul<strong>in</strong>e: visual loss <strong>in</strong> a tippler. Surv Ophthalmol 35:454–462.<br />
Sedwick LA. (1992). Gett<strong>in</strong>g to the heart of visual loss: when cardiac medication may be dangerous to the optic<br />
nerves. Surv Ophthalmol 36:366–372.<br />
Seth B, Khosla PK, Semwal OP, D’Monty V. (1991). Visual evoked responses <strong>in</strong> tuberculous children on<br />
ethambutol therapy. Indian Pediatr 28:713–717.<br />
Shaikh S, Ta C, Basham AA, Mansour S. (2001). Leber hereditary optic neuropathy associated with antiretroviral<br />
therapy for human immunodeficiency virus <strong>in</strong>fection. Am J Ophthalmol 131:143–145.<br />
Sharma OP, Sharma AM. (1991). Sarcoidosis of the nervous system: a cl<strong>in</strong>ical approach. Arch Intern Med 151:1317–<br />
1321.<br />
Shibasaki HS, Hayasaka S, Noda S, et al. (1992). Radiotherapy resolves leukemic <strong>in</strong>volvement of the optic nerves.<br />
<strong>An</strong>n Ophthalmol 24:395–397.<br />
Shoffner JM, Brown MD, Stugard C, et al. (1995). Leber’s hereditary optic neuropathy plus dystonia is caused by a<br />
mitochondrial DNA po<strong>in</strong>t mutation. <strong>An</strong>n <strong>Neuro</strong>l 38:163–169.<br />
Shults WT, Hamby S, Corbett JJ, et al. (1993). <strong>Neuro</strong>-ophthalmic complications of <strong>in</strong>tracranial catheters.<br />
<strong>Neuro</strong>surgery 33:135–138.<br />
Shuper A, Horeu G, Kornreich L. (1997). Visual pathway glioma: an erratic tumour with therapeutic dilemmas.<br />
Arch Dis Child 76:259–263.<br />
Shutter LA, Kl<strong>in</strong>e LB, Fisher WS. (1993). Visual loss and a suprasellar mass complicated by pregnancy. Surv<br />
Ophthalmol 38:63–69.<br />
Siatkowski RM, Lam BL, Schatz NJ, et al. (1992). Optic neuropathy <strong>in</strong> Hodgk<strong>in</strong>’s disease. Am J Ophthalmol<br />
114:625–629.<br />
Siatkowski RM, Scott IU, Verm AM, et al. (2001). Optic neuropathy and chiasmopathy <strong>in</strong> the diagnosis of systemic<br />
lupus erythematosus. J <strong>Neuro</strong>-ophthalmol 21:193–198.<br />
Silver MR, Messner LV. (1994). Sarcoidosis and its ocular manifestations. J Am Optom Assoc 65:321–327.<br />
Simmons IG, Good PA. (1998). Carbon monoxide poison<strong>in</strong>g causes optic neuropathy. Eye 12:809–814.<br />
Skolnick CA, Mafee MF, Goodw<strong>in</strong> JA. (2000). Pneumos<strong>in</strong>us dilatans of the sphenoid s<strong>in</strong>us present<strong>in</strong>g with visual<br />
loss. J <strong>Neuro</strong>-ophthalmol 20:259–263.<br />
Smets RM, Waeben M. (1999). Unusual comb<strong>in</strong>ation of night bl<strong>in</strong>dness and optic neuropathy after biliopancreatic<br />
bypass. Bull Soc Belge Ophtalmol 271:93–96.<br />
Song A, Scott IU, Davis JL, et al. (2002). Atypical anterior optic neuropathy caused by toxoplasmosis. Am J<br />
Ophthalmol 133:162–164.<br />
Speicher MA, Goldman MH, Ghrousos GA. (2000). Amiodarone optic neuropathy without disc edema. J <strong>Neuro</strong>ophthalmol<br />
20:171–172.<br />
Sreih AG, Schoenfeld MH, Marieb MA. (1999). Optic neuropathy follow<strong>in</strong>g amiodarone therapy. Pac<strong>in</strong>g Cl<strong>in</strong><br />
Electrophysiol 22:1108–1110.<br />
Stafford SL, Perry A, Leavitt JA, et al. (1998). <strong>An</strong>terior visual pathway men<strong>in</strong>giomas primarily resected between<br />
1978 and 1988. The Mayo Cl<strong>in</strong>ic Rochester experience. J <strong>Neuro</strong>-ophthalmol 18:206–210.<br />
Steel DHW, Potts MJ. (1995). Bilateral sudden visual loss <strong>in</strong> Albright’s syndrome. Br J Ophthalmol 79:1149.<br />
Stone EM, Newman NJ, Miller NR, et al. (1992). Visual recovery <strong>in</strong> patients with Leber’s hereditary optic<br />
neuropathy and the 11778 mutation. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:10–14.<br />
Stretch JR, Poole MD. (1992). Pneumos<strong>in</strong>us dilatans as the aetiology of progressive bl<strong>in</strong>dness. Br J Plast Surg<br />
45:469–473.<br />
Strom<strong>in</strong>ger MB, Schatz NJ, Glaser JS. (1993). Lymphomatous optic neuropathy. Am J Ophthalmol 116:774–776.<br />
Sung JU, Lam BL, Curt<strong>in</strong> VT, et al. (1998). Metastatic gastric carc<strong>in</strong>oma to the optic nerve. Arch Ophthalmol<br />
116:692–693.<br />
Sullivan-Mee M, Solis K. (1998). Methanol-<strong>in</strong>duced vision loss. J Am Optom Assoc 69:57–65.<br />
Sutton LN. (1994). Visual pathway gliomas of childhood. <strong>Neuro</strong>surgery 16:1–6.<br />
Sutton LN, Molloy PT, Senyak H. (1995). Long term outcome of hypothalamic=chiasmatic astrocytomas <strong>in</strong><br />
children treated with conservative surgery. J <strong>Neuro</strong>surg 83:583–589.<br />
Tachibana O, Yamashima T, Yamashita J. (1990). Radiation necrosis of the optic chiasm, optic tract, hypothalamus,
The Diagnosis of Optic <strong>Neuro</strong>pathies 33<br />
and upper pons after radiotherapy for pituitary adenoma detected by gadol<strong>in</strong>ium-enhanced, T1 weighted<br />
magnetic resonance imag<strong>in</strong>g: case report. <strong>Neuro</strong>surgery 27:640–643.<br />
Tamai H, Tamai K, Yuasa H. (2000). Pachymen<strong>in</strong>gitis with pseudo–Foster Kennedy syndrome. Am J Ophthalmol<br />
130:535–537.<br />
Teare JP, Whitehead M, Rake MO, Coker RJ. (1991). Rapid onset of bl<strong>in</strong>dness due to men<strong>in</strong>geal carc<strong>in</strong>omatosis<br />
from an oesophageal adenocarc<strong>in</strong>oma. Postgrad Med J 67:909–911.<br />
Teus MA, Teruel JL, Pascual J, et al. (1991). Corticosteroid <strong>in</strong>duced toxic optic neuropathy. Am J Ophthalmol<br />
112:605.<br />
Thambisetty MR, Scherzer CR, Yu Z, et al. (2001). Paraneoplastic optic neuropathy and cerebellar ataxia with<br />
small cell carc<strong>in</strong>oma of the lung. J <strong>Neuro</strong>-ophthalmol 21:164–167.<br />
Thomas RJ. (1994). <strong>Neuro</strong>toxicity of antibacterial therapy. South Med J 87:869–874.<br />
Thorne JE, Galetta SL. (1998). Disc edema and ret<strong>in</strong>al periphlebitis as the <strong>in</strong>itial manifestation of sarcoidosis. Arch<br />
<strong>Neuro</strong>l 55:862–863.<br />
Thorne JE, Volpe NJ, Wulc AE, Galetta SL. (2002). Caught by a masquerade: scleros<strong>in</strong>g orbital <strong>in</strong>flammation. Surv<br />
Ophthalmol 47:50–54.<br />
Trobe JD. (2001). The <strong>Neuro</strong>logy of Vision. Oxford, Oxford University Press.<br />
Trobe JD, Glaser JS. (1978). Quantitative perimetry <strong>in</strong> compressive optic neuropathy and optic neuritis. Arch<br />
Ophthalmol 96:1210–1216.<br />
Trobe JD, Glaser JS, Cassady JC. (1980). Optic atrophy: differential diagnosis by fundus observation. Arch<br />
Ophthalmol 98:1040–1045.<br />
Tsai RK, Lee UH. (1997). Reversibility of ethambutol optic neuropathy. J Ocul Pharmacol Ther 13:473–477.<br />
Tsao K, Aitken PA, Johns DR. (1999). Smok<strong>in</strong>g as an aetiological factor <strong>in</strong> a pedigree with Leber’s hereditary optic<br />
neuropathy. Br J Ophthalmol 83:577–581.<br />
Vaphiades MS. (2001). Disk edema and cranial MRI optic nerve enhancement: How long is too long? Surv<br />
Ophthalmol 46:56–58.<br />
Vaphiades MS, Newman NJ. (1999). Optic nerve enhancement on orbital magnetic resonance imag<strong>in</strong>g <strong>in</strong> Leber’s<br />
hereditary optic neuropathy. J <strong>Neuro</strong>-ophthalmol 19:238–239.<br />
Vargas ME, Kupersmith MJ, Setton A, et al. (1994). Endovascular treatment of giant aneurysm which cause visual<br />
loss. Ophthalmology 101:1091–1098.<br />
Votruba M, Fitzke FW, Holder GE, et al. (1998). <strong>Cl<strong>in</strong>ical</strong> features <strong>in</strong> affected <strong>in</strong>dividuals from 21 pedigrees with<br />
dom<strong>in</strong>ant optic atrophy. Arch Ophthalmol 116:351–358.<br />
Vrabec TA, Sergott AC, Jaeger EA, et al. (1990). Reversible visual loss <strong>in</strong> a patient receiv<strong>in</strong>g high-dose<br />
ciprofloxac<strong>in</strong> hydrochloride (cipro). Ophthalmology 97:707–710.<br />
Wallace RT, Shields JA, Shields CL, et al. (1991). Leukemic <strong>in</strong>filtration of the optic nerve. Arch Ophthalmol 109:1027.<br />
Wang MY, Arnold AC, V<strong>in</strong>ters HV, Glasgow BJ. (2000). Bilateral bl<strong>in</strong>dness and lumbosacral myelopathy<br />
associated with high-dose Carmust<strong>in</strong>e and Cisplat<strong>in</strong> therapy. Am J Ophthalmol 130:367–368.<br />
Warner JEA, Lessell S, Rizzo JF III, Newman NJ. (1997). Does optic disc appearance dist<strong>in</strong>guish ischemic optic<br />
neuropathy from optic neuritis? Arch Ophthalmol 115:1408–1410.<br />
Weaver DT, Garrity JA, Meyer FA, Laws ER. (1993). Visual prognosis <strong>in</strong> sphenoid ridge men<strong>in</strong>gioma. Presented at<br />
the 19th annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Big Sky, Montana.<br />
We<strong>in</strong> FB, Gans MS. (1999). The perils of a sneeze. J <strong>Neuro</strong>-ophthalmol 19:128–130.<br />
We<strong>in</strong>er HL, Wisoff JH, Rosenberg ME, et al. (1994). Craniopharyngiomas: a cl<strong>in</strong>icopathological analysis of factors<br />
predictive of recurrence and functional outcome. <strong>Neuro</strong>surgery 35:1001–1011.<br />
Weisman JS, Hepler RS, V<strong>in</strong>ters HV. (1990). Reversible visual loss caused by fibrous dysplasia. Am J Ophthalmol<br />
110:244–249.<br />
Wijaya J, Salu P, Leblanc A, Bervoets S. (1999). Acute unilateral visual loss due to a s<strong>in</strong>gle <strong>in</strong>tranasal<br />
methamphetam<strong>in</strong>e abuse. Bull Soc Belge Ophtalmol 271:19–25.<br />
Wijers OB, Levendag PC, Luyten GP, et al. (1999). Radiation-<strong>in</strong>duced bilateral optic neuropathy <strong>in</strong> cancer of the<br />
nasopharynx. Case failure analysis and review of the literature. Strahlenther Onkol 175:21–27.<br />
Wilson CB. (1994). Men<strong>in</strong>giomas: genetics, malignancy, and the role of radiation <strong>in</strong> <strong>in</strong>duction and treatment. J<br />
<strong>Neuro</strong>surg 81:666–675.<br />
Wisoff J. (1990a). Management of optic pathway tumors of childhood. Pediatr <strong>Neuro</strong>oncol 3:791.<br />
Wisoff JH, Abbott R, Epste<strong>in</strong> F. (1990b). Surgical management of exophytic chiasmatic-hypothalamic tumors of<br />
childhood. J <strong>Neuro</strong>surg 73:661–667.<br />
Wright JE, Sullivan TJ, Garner A, Wulc AE, Moseley IF. (1997). Orbital venous anomalies. Ophthalmology 104:905–<br />
913.<br />
Wroe SJ, Thompson AJ, McDonald WI. (1991). Pa<strong>in</strong>ful <strong>in</strong>traorbital men<strong>in</strong>giomas. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
54:1009–1010.
34 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Yamaguchi K, Ohnuma I, Takahashi S, et al. (1997). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> acute optic neuropathy by<br />
sphenoidal mucocele. Int Ophthalmol 21:9–11.<br />
Yamamoto N, Kiyosawa M, Kawasaki T, et al. (1994). Successfully treated optic nerve <strong>in</strong>filtration with adult T-cell<br />
lymphoma. J <strong>Neuro</strong>-ophthalmol 14:81–83.<br />
Youl BD, Plant GT, Stevens JM, et al. (1990). Three cases of craniopharyngioma show<strong>in</strong>g optic tract hypersignal on<br />
MRI. <strong>Neuro</strong>logy 40:1416–1419.<br />
Young WC, Thornton AF, Gebarski SS, et al. (1992). Radiation-<strong>in</strong>duced optic neuropathy: correlation of MR<br />
imag<strong>in</strong>g and radiation dosimetry. Radiology 185:904–907.<br />
Yu Z, Kryzer TJ, Greisman GE, et al. (2001). CRMP-5 neuronal autoantibody: marker of lung cancer and<br />
thymoma-related autoimmunity. <strong>An</strong>n <strong>Neuro</strong>l 49:146–154.<br />
Zaman AG, Graham EM, Sanders MD. (1993). <strong>An</strong>terior visual system <strong>in</strong>volvement <strong>in</strong> Hodgk<strong>in</strong>’s lymphoma. Br J<br />
Ophthalmol 77:184–187.<br />
Zimmerman CF, Schatz NJ. (1990a). Magnetic resonance imag<strong>in</strong>g of radiation optic neuropathy. Am J Ophthalmol<br />
110:389–394.<br />
Zimmerman CF, Schatz NJ, Glaser JS. (1990b). Magnetic resonance imag<strong>in</strong>g of optic nerve men<strong>in</strong>giomas.<br />
Enhancement with gadol<strong>in</strong>ium-DTPA. Ophthalmology 97:585–591.
2 r<br />
Optic Neuritis<br />
Optic neuritis (ON) is a general term for an optic neuropathy result<strong>in</strong>g from an idiopathic,<br />
<strong>in</strong>flammatory, <strong>in</strong>fectious, or demyel<strong>in</strong>at<strong>in</strong>g etiology. If the optic nerve is swollen on<br />
ophthalmoscopy, then the term papillitis or anterior ON is used. If the optic nerve is normal<br />
on ophthalmoscopy, then it is called retrobulbar ON. In cl<strong>in</strong>ical practice, most ophthalmologists<br />
use the term optic neuritis to describe idiopathic or demyel<strong>in</strong>at<strong>in</strong>g ON.<br />
What Are the Features of Typical Optic<br />
Neuritis?<br />
Patients with idiopathic or demyel<strong>in</strong>at<strong>in</strong>g ON usually present with a ‘‘typical’’ cl<strong>in</strong>ical<br />
profile as shown <strong>in</strong> Table 2–1 (Beck, 1992a, 1993a,c–e, 1994a; Cleary, 1993; Frederiksen,<br />
1991; Gerl<strong>in</strong>g, 1998a,b; J<strong>in</strong>, 1999; Keltner, 1993a, 1993b; Optic Neuritis Study Group,<br />
1991; Schneck, 1997; Slamovits, 1991a; Wakakuru, 1999b; Wall, 1998).<br />
The cl<strong>in</strong>ical characteristics of 455 patients with ON enrolled <strong>in</strong> the Optic Neuritis<br />
Treatment Trial (ONTT), a study sponsored by the National Eye Institute conducted at<br />
15 cl<strong>in</strong>ical centers <strong>in</strong> the United States between the years 1988 and 1991, are outl<strong>in</strong>ed <strong>in</strong><br />
Table 2–2.<br />
The majority of patients with ON with eye or ophthalmic trigem<strong>in</strong>al distribution pa<strong>in</strong><br />
or pa<strong>in</strong> with eye movement have <strong>in</strong>volvement of the orbital segment of the optic nerve<br />
(Kupersmith, 2002). The absence of pa<strong>in</strong>, particularly with eye movement, suggests<br />
the disorder is limited to the canalicular or <strong>in</strong>tracranial portion of the optic nerve<br />
(Kupersmith, 2002).<br />
What Visual Field Defects Are Noted with<br />
Optic Neuritis?<br />
<strong>An</strong>alysis of <strong>in</strong>itial perimetry <strong>in</strong> the ONTT showed that the most common present<strong>in</strong>g<br />
pattern was a diffuse field defect (48%), with altitud<strong>in</strong>al=arcuate defects <strong>in</strong> 20%, and<br />
35
36 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 2–1. Features of Typical Optic Neuritis (ON)<br />
Acute, usually unilateral loss of vision<br />
Visual acuity (variable visual loss 20=20 to no light perception (NLP)<br />
Visual field (variable optic nerve visual field defects)<br />
A relative afferent pupillary defect (RAPD) <strong>in</strong> unilateral or bilateral but asymmetric cases<br />
Periocular pa<strong>in</strong> (90%), especially with eye movement (Gerl<strong>in</strong>g, 1998a)<br />
Normal (65%) or swollen (35%) optic nerve head<br />
A young adult patient (
Optic Neuritis 37<br />
Table 2–2. The <strong>Cl<strong>in</strong>ical</strong> Profile of the Optic Neuritis Treatment Trial (ONTT)<br />
Patients<br />
<strong>Cl<strong>in</strong>ical</strong> characteristic Patients<br />
Female 77%<br />
White 85%<br />
Age (years) (mean SD) 32 6.7<br />
Mean days of visual symptoms before entry 5.0 1.6<br />
Ocular pa<strong>in</strong> present 92%<br />
Pa<strong>in</strong> worsened by eye movement 87%<br />
Ophthalmoscopic f<strong>in</strong>d<strong>in</strong>gs<br />
Optic disc appearance<br />
Optic disc swollen 35%<br />
Optic disc normal (retrobulbar) 65%<br />
Characteristics of swollen optic disc<br />
Mild and focal 28.6%<br />
Mild and diffuse 51%<br />
Severe and focal 3.1%<br />
Severe and diffuse 16.8%<br />
Ret<strong>in</strong>al or optic disc hemorrhage<br />
None 84.5%<br />
On disc 6.2%<br />
On ret<strong>in</strong>a 3.7%<br />
On both disc and ret<strong>in</strong>a 5.0%<br />
Vitreous<br />
Normal 93.8%<br />
Trace cells 6.2%<br />
More than trace cells 0%<br />
Ret<strong>in</strong>al exudates<br />
Present on or adjacent to disc 3.1%<br />
Present <strong>in</strong> the macula 0%<br />
Present elsewhere 0.6%<br />
Visual acuity<br />
20=20 or better 11%<br />
20=25–20=40 25%<br />
20=50–20=190 29%<br />
20=200–20=800 20%<br />
Count<strong>in</strong>g f<strong>in</strong>gers 4%<br />
Hand motions 6%<br />
Light perception 3%<br />
No light perception (NLP) 3%<br />
Visual field defects <strong>in</strong> <strong>in</strong>volved eye<br />
Pattern<br />
Diffuse 48%<br />
Altitud<strong>in</strong>al, arcuate, nasal step 20%<br />
Central, cecocentral 8%<br />
Other types 24%<br />
Chiasmal 5%<br />
(cont<strong>in</strong>ued)
38 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 2–2. (cont<strong>in</strong>ued)<br />
Retrochiasmal 9%<br />
Median visual field mean deviation (quartiles) 23.02 ( 31.90, 12.25)<br />
Visual function deficits <strong>in</strong> fellow eye 67%<br />
Visual acuity 14%<br />
Contrast sensitivity 15%<br />
Color vision 22%<br />
Visual field (mean deviation) 48%<br />
Abnormal MRI (one or more white matter lesion) 49%<br />
Percents represent the percent of patients with the characteristic.<br />
pure papillomacular <strong>in</strong>volvement <strong>in</strong> only 8%. Fang et al showed that ON affects the entire<br />
30 degrees (global field <strong>in</strong>volvement) even <strong>in</strong> patients who appear to have localized<br />
depression of visual threshold, <strong>in</strong>dicat<strong>in</strong>g that ON does not have a true predilection for the<br />
papillomacular bundle, or any specificnervefiber bundle (Fang, 1999a). In another study,<br />
Fang et al assessed specific nervefiber group <strong>in</strong>volvement by analyz<strong>in</strong>g recovery of field<br />
with<strong>in</strong> concentric field r<strong>in</strong>gs <strong>in</strong> the central 30 degrees and found that return of field function<br />
does not appear to differ between patients with diffuse or localized defects (Fang, 1999b).<br />
They postulate that reduced redundancy of axons <strong>in</strong> the periphery of the field compared<br />
with near fixation may be responsible for the greater recovery of threshold near fixation.<br />
What Are the Features of Atypical Optic<br />
Neuritis?<br />
Patients who meet the criteria listed <strong>in</strong> Table 2–1 are considered to have typical<br />
ON. Conversely, patients with the features listed <strong>in</strong> Table 2–3 have atypical ON<br />
(Beck, 1993a–e, 1994b; Biousse, 1999; Lee, 1998a; Moschos, 1990; Optic Neuritis Study<br />
Group, 1991). For example, the fundus features that should lead the exam<strong>in</strong>er to<br />
consider an alternate diagnosis to ON <strong>in</strong>clude lipid maculopathy, very severe disc<br />
edema with marked hemorrhages, cotton wools spots, vitreous cells, pale optic disc<br />
edema, ret<strong>in</strong>al arteriolar narrow<strong>in</strong>g, and ret<strong>in</strong>opathy.<br />
What Disorders May Be Associated with Optic<br />
Neuritis?<br />
Table 2–4 lists a number of disorders that may be associated with typical or atypical ON.<br />
The presence of one of these disorders is usually suggested by the historical or<br />
exam<strong>in</strong>ation f<strong>in</strong>d<strong>in</strong>gs.<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of Optic<br />
Neuritis <strong>in</strong> Children?<br />
The cl<strong>in</strong>ical features of ON <strong>in</strong> children differ from those <strong>in</strong> adults. Table 2–5 summarizes<br />
these features. Brady et al reviewed 25 cases and concluded that pediatric ON is usually
Table 2–3. Features of Atypical Optic Neuritis (ON)<br />
associated with visual recovery; however, a significant number of patients (22%) rema<strong>in</strong><br />
visually disabled. A normal magnetic resonance (MR) image of the bra<strong>in</strong> may be<br />
associated with a better outcome. Younger patients are more likely to have bilateral<br />
disease and a better visual prognosis (Brady, 1999).<br />
In another study of 47 children with multiple sclerosis, 38 (80.9%) had ON at least<br />
once, and 10 (21.3%) had two or more attacks of ON (Boiko, 2000). The presence of<br />
tumor necrosis factor a7 (TNF-a7) locus on chromosome 6 was proposed as a possible<br />
marker of early multiple sclerosis (MS) onset <strong>in</strong> these patients.<br />
What Is the Evaluation of Optic Neuritis?<br />
Optic Neuritis 39<br />
Bilateral simultaneous onset of ON <strong>in</strong> an adult patient<br />
Lack of pa<strong>in</strong><br />
Severe headache (e.g., sphenoid s<strong>in</strong>usitis)<br />
Ocular f<strong>in</strong>d<strong>in</strong>gs suggestive of an <strong>in</strong>flammatory process<br />
<strong>An</strong>terior uveitis<br />
Posterior chamber <strong>in</strong>flammation more than trace<br />
Macular exudate or star figure<br />
Ret<strong>in</strong>al <strong>in</strong>filtrate or ret<strong>in</strong>al <strong>in</strong>flammation<br />
Severe disc swell<strong>in</strong>g<br />
Marked hemorrhages<br />
Lack of significant improvement of visual function or worsen<strong>in</strong>g of visual function after 30 days<br />
Lack of at least one l<strong>in</strong>e of visual acuity improvement with<strong>in</strong> the first 3 weeks after onset of<br />
symptoms<br />
Age greater than 50 years<br />
Preexist<strong>in</strong>g diagnosis or evidence of other systemic condition<br />
Inflammatory (e.g., sarcoidosis, Wegener’s granulomatosis, systemic lupus erythematosus)<br />
Infectious disease (e.g., Lyme disease, tuberculosis, human immunodeficiency virus <strong>in</strong>fection)<br />
Severe hypertension, diabetes, or other systemic vasculopathy<br />
Exquisitely steroid sensitive or steroid-dependent optic neuropathy<br />
In atypical cases, consideration should be given to do<strong>in</strong>g a lumbar puncture and<br />
additional laboratory studies; <strong>in</strong> the ONTT, syphilis serology, ant<strong>in</strong>uclear antibody,<br />
and chest x-ray were performed. The required evaluation depends on the history and<br />
exam<strong>in</strong>ation, with specific attention to <strong>in</strong>fectious or <strong>in</strong>flammatory etiologies as listed <strong>in</strong><br />
Table 2–4. In addition, patients with <strong>in</strong>flammatory autoimmune ON often have<br />
progressive or recurrent steroid-responsive or steroid-dependent optic neuropathy<br />
(Beck, 1994a; Bielory, 1993; Riedel, 1998).<br />
The association of acute or subacute loss of vision <strong>in</strong> one or both eyes caused by optic<br />
neuropathy preceded or followed by a transverse or ascend<strong>in</strong>g myelopathy is referred<br />
to as neuromyelitis optica (Devic’s disease). The cl<strong>in</strong>ical features of Devic’s disease are<br />
outl<strong>in</strong>ed <strong>in</strong> Table 2–6.
40 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 2–4. Disorders Associated with Optic Neuritis<br />
Polyneuropathies<br />
Guilla<strong>in</strong>-Barré syndrome (Nadkarni, 1993; Ropper, 1991)<br />
Miller Fisher syndrome (Chan, 2002)<br />
Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (CIDP) (Kaufman, 1998; Lee, 1999)<br />
Infections<br />
Bacteria<br />
Syphilis (Frohman, 1997)<br />
Tuberculosis (Mansour, 1998)<br />
Lyme disease (Arnold, 1993; Jacobson, 1991; Karma, 1995; Lesser, 1990; W<strong>in</strong>terkorn, 1990)<br />
Bartonella henselae (Cat-scratch disease) (Brazis, 1986; Schwartzman, 1994, 1995)<br />
Mycoplasma (Nadkarni, 1993; Salzman, 1992; Sheth, 1993)<br />
Whipple’s disease<br />
Brucellosis (Abd Elrazek, 1991; McLean, 1992)<br />
b-Hemolytic streptococcus<br />
Men<strong>in</strong>gococcus (Miller, 1995)<br />
Propionibacterium acnes (Kouyoumdjian, 2001)<br />
Fungi<br />
Aspergillus<br />
Histoplasmosis (Perry, 1999; Yau, 1996)<br />
Cryptococcus (Golnik, 1991)<br />
Rickettsiae (e.g., Q fever, epidemic typhus)<br />
Protozoa<br />
Toxoplasmosis (Banta, 2002; Falcone, 1993; Grossniklaus, 1990; Pierce, 1993; Rose, 1991; Song,<br />
2002)<br />
Parasites<br />
Toxocariasis (Komiyama, 1995)<br />
Cysticercosis (Chang, 2001; Menon, 2000)<br />
Viruses<br />
Adenovirus<br />
Hepatitis A (McKibb<strong>in</strong>, 1995)<br />
Hepatitis B (Achiron, 1994)<br />
Cytomegalovirus (CMV) (Hark<strong>in</strong>s, 1992; Ho, 1995; Mansour, 1997; Patel, 1996; Roarty, 1993)<br />
Coxsackie B<br />
Rubella<br />
Chickenpox (Lee, 1997)<br />
Herpes zoster (Deane, 1995; Greven, 2001; Gunduz, 1994; Lee, 1997; Miyashita, 1993; Mori, 1997;<br />
Nakazawa, 1999)<br />
Herpes simplex virus 1 (Tornerup, 2000)<br />
Epste<strong>in</strong>-Barr (EB) virus (<strong>in</strong>fectious mononucleosis) (<strong>An</strong>derson, 1994; Beiran, 2000; Corssmit,<br />
1997; Straussberg, 1993)<br />
Measles (Totan, 1999)<br />
Mumps (Sugita, 1991)<br />
Influenza<br />
HTLV-1 (Lehky, 1996; Merle, 1997; Yoshida, 1998)<br />
Prions (Jakob-Creutzfeldt disease)<br />
HIV (AIDS)-related (Friedman, 1991; Nichols, 1992)<br />
(cont<strong>in</strong>ued)
Table 2–4. (cont<strong>in</strong>ued)<br />
Optic Neuritis 41<br />
Primary HIV-related optic neuritis (Burton, 1998; Malessa, 1995; Newman, 1992; Quicenco, 1992;<br />
Sadun, 1995; Sweeney, 1993)<br />
Syphilis (McLeish, 1990)<br />
Cat-scratch disease (Bartonella henselae) (Schwartzman, 1994, 1995)<br />
Cryptococcus (Golnik, 1991)<br />
Histoplasmosis (Yau, 1996)<br />
Cytomegalovirus (CMV) (Mansour, 1997; Patel, 1996; Roarty, 1993)<br />
Herpes zoster (Friedlander, 1996; Lee, 1998b; Litoff, 1990; Margolis, 1998; Meenken, 1998;<br />
Shayegani, 1996)<br />
Hepatitis B<br />
Toxoplasmosis (Falcone, 1993)<br />
Postvacc<strong>in</strong>ation (Albitar, 1997; Hull, 1997; Kerrison, 2001; L<strong>in</strong>ssen, 1997; Stewart, 1999; Topaloglu<br />
1992; van de Geijn, 1994; Yen, 1991)<br />
Smallpox<br />
Tetanus<br />
Rabies<br />
Influenza<br />
Hepatitis B<br />
Bacille Calmette-Guér<strong>in</strong> (BCG)<br />
<strong>An</strong>thrax (Kerrison, 2002)<br />
Trivalent measles-mumps-rubella vacc<strong>in</strong>e<br />
Mantoux tubercul<strong>in</strong> sk<strong>in</strong> test<br />
Focal <strong>in</strong>fection or <strong>in</strong>flammation (Bath, 1998; Farris, 1990; Fujimoto, 1999; Moorman, 1999)<br />
Paranasal s<strong>in</strong>usitis<br />
Mucocele<br />
Post<strong>in</strong>fectious<br />
Malignant otitis externa<br />
Systemic <strong>in</strong>flammations and diseases<br />
Behçet’s disease (Vaphiades, 1999)<br />
Inflammatory bowel disease (Hutnik, 1996)<br />
Reiter’s syndrome<br />
Sarcoidosis (Beck, 1994; Case Records Massachusetts General Hospital, 1996; DeBroft, 1993;<br />
Haupert, 1997; Kosmorsky, 1996)<br />
Systemic lupus erythematosus (Ahmadieh, 1994; Gal<strong>in</strong>do-Rodriguez, 1999; Giorgi, 1999a,b;<br />
N<strong>in</strong>omiya, 1990; Ohnuma, 1996; Rosenbaum, 1997)<br />
Sjögren’s syndrome<br />
Mixed connective tissue disease<br />
Rheumatoid arthritis (Agildere, 1999)<br />
Miscellaneous<br />
Multifocal choroiditis<br />
Birdshot chorioret<strong>in</strong>opathy<br />
Acute posterior multifocal placoid pigment epitheliopathy (APMPPE) (Wolf, 1990)<br />
Autoimmune optic neuropathy (Bielory, 1993; Riedel, 1998)<br />
Familial Mediterranean fever (Lossos, 1993)<br />
Bee or wasp st<strong>in</strong>g (Berrios, 1994; Choi, 2000; Maltzman, 2000; Song, 1991)<br />
Snake bite (Menon, 1997)<br />
(cont<strong>in</strong>ued)
42 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 2–4. (cont<strong>in</strong>ued)<br />
Postpartum optic neuritis (Leiba, 2000)<br />
Retrobulbar optic neuritis with ret<strong>in</strong>itis pigmentosa s<strong>in</strong>e pigmento (Hatta, 2000)<br />
<strong>Neuro</strong>myelitis optica (Devic’s disease) (Ahasan, 1994; Al-Deeb, 1993; Barkhoff, 1991b;<br />
Ha<strong>in</strong>fellner, 1992; Hershewe, 1990; Igarishi, 1994; Ja<strong>in</strong>, 1994; Jeffrey, 1996; Khan, 1990; Mandler,<br />
1993, 1998; O’Riordan, 1996; Piccolo, 1990; Ramelli, 1992; Silber, 1990)<br />
Recurrent optic neuromyelitis with endocr<strong>in</strong>opathies (Vernant, 1997)<br />
Table 2–5. Features of Childhood Optic Neuritis Compared<br />
with Adult Optic Neuritis<br />
More likely to be bilateral<br />
More likely to have papillitis<br />
May have worse present<strong>in</strong>g vision (later presentation?)<br />
More likely to be associated with viral=para<strong>in</strong>fectious etiology<br />
Less likely to be associated with multiple sclerosis<br />
Table 2–6. <strong>Cl<strong>in</strong>ical</strong> Features of Devic’s Disease<br />
Age: Typically younger patients<br />
Gender: Affects men and women equally<br />
Race<br />
May be more common <strong>in</strong> African Americans who develop ON (Phillips, 1998)<br />
May be more common <strong>in</strong> Asians who develop ON (Sakuma, 1999)<br />
Recurrent optic neuromyelitis with endocr<strong>in</strong>opathies <strong>in</strong> eight <strong>An</strong>tillean women from Mart<strong>in</strong>ique<br />
and Guadeloupe (Vernant, 1997)<br />
Familial cases: rare (Yamakawa, 2000)<br />
Pathology: Differs from multiple sclerosis (MS)<br />
Cerebellum is almost never affected<br />
Excavation of affected tissue with formation of cavities common <strong>in</strong> Devic’s but rare <strong>in</strong> MS<br />
Gliosis characteristic of MS absent or m<strong>in</strong>imal with Devic’s<br />
Arcuate fibers <strong>in</strong> cerebral subcortex relatively unaffected <strong>in</strong> Devic’s but severely damaged <strong>in</strong> MS<br />
<strong>Cl<strong>in</strong>ical</strong> features<br />
May have prodrome of fever, sore throat, and headache<br />
Visual loss<br />
May precede or follow paraplegia<br />
Usually bilateral (hours, days, or rarely weeks between eyes)<br />
Rapid and usually severe (complete bl<strong>in</strong>dness not uncommon)<br />
Central scotoma most common visual field defect<br />
Ophthalmoscopy<br />
Majority have mild disc swell<strong>in</strong>g of both discs but may be normal<br />
Occasional severe swell<strong>in</strong>g with dilation of ve<strong>in</strong>s and extensive peripapillary exudates<br />
May have slight narrow<strong>in</strong>g of ret<strong>in</strong>al vessels<br />
(cont<strong>in</strong>ued)
Table 2–6. (cont<strong>in</strong>ued)<br />
What Were the Results of the Optic Neuritis<br />
Treatment Trial (ONTT)?<br />
Optic Neuritis 43<br />
Visual prognosis<br />
Usually some recovery of vision<br />
Often recovers with<strong>in</strong> weeks to months<br />
Some cases severe and permanent<br />
Paraplegia (transverse myelitis)<br />
Usually sudden and severe<br />
Often recover to some degree but may be permanent complete paralysis<br />
Sp<strong>in</strong>al cord MRI often shows abnormality extend<strong>in</strong>g over three or more segments<br />
May have Lhermitte’s symptom, paroxysmal tonic spasms, and radicular pa<strong>in</strong><br />
Course: monophasic or relaps<strong>in</strong>g<br />
Associations<br />
Rarely associated with demyel<strong>in</strong>at<strong>in</strong>g peripheral neuropathy<br />
Rarely associated with HIV-1 <strong>in</strong>fection, systemic lupus erythematosus, antiphospholipid antibody<br />
syndrome, and pulmonary tuberculosis<br />
Laboratory studies<br />
Often cerebrosp<strong>in</strong>al fluid (CSF) pleocytosis (e.g., >50 WBC, often polymorphonuclear cells)<br />
Oligoclonal bands uncommon<br />
Rare <strong>in</strong>creased <strong>in</strong>tracranial pressure<br />
Treatment<br />
Possible response to IV steroids<br />
IV gamma globul<strong>in</strong><br />
Mortality less than 10 to 33%<br />
Source: Ahasan, 1994; Al-Deeb, 1993; Barkhoff, 1991b; Blanche, 2000; Filippi, 1999; Ha<strong>in</strong>fellner, 1992; Hershewe,<br />
1990; Igarishi, 1994; Ja<strong>in</strong>, 1994; Jeffrey, 1996; Khan, 1990; Mandler, 1993, 1998; O’Riordan, 1996; Phillips, 1998;<br />
Piccolo, 1990; Ramelli, 1992; Silber, 1990; Vernant, 1997; W<strong>in</strong>gerchuk, 1999; Yamakawa, 2000.<br />
The ONTT was developed to evaluate the efficacy of corticosteroid treatment for acute<br />
ON and to <strong>in</strong>vestigate the relationship between ON and MS (Beck, 1992a, 1993a–e,<br />
1995a). The ONTT was sponsored by the National Eye Institute as a randomized,<br />
controlled cl<strong>in</strong>ical trial that enrolled 457 patients at 15 cl<strong>in</strong>ical centers <strong>in</strong> the United<br />
States between the years 1988 and 1991. The ONTT entry criteria specified that patients<br />
be between the ages of 18 and 46 years, that they have a relative afferent pupillary<br />
defect as well as a visual field defect <strong>in</strong> the affected eye, and that they were exam<strong>in</strong>ed<br />
with<strong>in</strong> 8 days of the onset of visual symptoms of a first attack of acute unilateral ON.<br />
Patients were excluded if they had previous episodes of ON <strong>in</strong> the affected eye,<br />
previous corticosteroid treatment for ON or MS, or systemic disease other than MS<br />
that might be a cause of the ON (Beck, 1992a, 1993a–e, 1995a). The cl<strong>in</strong>ical features of<br />
the ONTT patients are outl<strong>in</strong>ed <strong>in</strong> Table 2–2.<br />
In the ONTT, all patients underwent test<strong>in</strong>g for collagen vascular disease (ant<strong>in</strong>uclear<br />
antibody [ANA]), serologic test<strong>in</strong>g for syphilis (fluorescent treponemal antibody<br />
absorption [FTA-ABS]), and a chest radiograph for sarcoidosis. Lumbar puncture was<br />
optional. <strong>An</strong> ANA test was positive <strong>in</strong> a titer less than 1:320 <strong>in</strong> 13% of patients, and
44 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
1:320 or greater <strong>in</strong> 3%. Only one patient was eventually diagnosed with a collagen<br />
vascular disease.<br />
Visual and neurologic outcomes <strong>in</strong> these patients were no different from those of the<br />
other ONTT patients. The FTA-ABS was positive <strong>in</strong> six patients (1.3%), but none had<br />
syphilis. A chest radiograph did not reveal sarcoidosis <strong>in</strong> any patient. However, <strong>in</strong> a<br />
separate report, Jacobson et al described 4 of 20 patients with isolated ON with a<br />
positive serology for Lyme disease (Jacobson, 1991). These authors recommended<br />
serologic test<strong>in</strong>g for Lyme disease <strong>in</strong> patients with ON, with or without the typical<br />
rash of erythema migrans, who live <strong>in</strong> or have visited Lyme endemic areas. Cerebrosp<strong>in</strong>al<br />
fluid (CSF) analysis was recommended for patients with positive serology and<br />
<strong>in</strong>travenous (IV) antibiotic therapy for unexpla<strong>in</strong>ed pleocytosis (Jacobson, 1991). We do<br />
not order Lyme titers for patients with ON from nonendemic regions (class IV, level C).<br />
The evaluation recommendations of the ONTT study group for patients with typical<br />
acute ON are listed <strong>in</strong> Table 2–7.<br />
What Are the <strong>Neuro</strong>imag<strong>in</strong>g F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Optic<br />
Neuritis?<br />
Periventricular white matter signal abnormalities on magnetic resonance imag<strong>in</strong>g (MRI)<br />
consistent with MS (Baumhefner, 1990; Jacobs, 1991) have been reported <strong>in</strong> 40 to 70% of<br />
cases of isolated ON (Christiansen, 1992; Fe<strong>in</strong>ste<strong>in</strong>, 1992; Francis, 1995; Frederiksen,<br />
1991a; Jacobs, 1991; Morrissey, 1993). MRI with gadol<strong>in</strong>ium may show enhanc<strong>in</strong>g<br />
lesions <strong>in</strong> 26 to 37% of patients with isolated ON (Christiansen, 1992; Merandi, 1991)<br />
and may <strong>in</strong>crease the detection of disease activity (Guy, 1990; Merandi, 1991; Thompson,<br />
1990).<br />
Although computed tomography (CT) scan of the head may also show abnormalities<br />
<strong>in</strong> MS and ON, CT has been relatively <strong>in</strong>sensitive to the detection of MS plaques<br />
compared to MRI. MRI is a very sensitive test for detect<strong>in</strong>g lesions consistent with MS<br />
(Baumhefner, 1990). Paty reported 19 cases of cl<strong>in</strong>ically def<strong>in</strong>ite MS (CDMS) out of 200<br />
consecutive patients with suspected MS compar<strong>in</strong>g predictive value of MR scann<strong>in</strong>g<br />
Table 2–7. Modified Evaluation Recommendations of the Optic Neuritis Treatment Trial<br />
(ONTT) for Optic Neuritis (ON)<br />
No laboratory studies or lumbar puncture required for typical optic neuritis<br />
Potential test<strong>in</strong>g for atypical optic neuritis<br />
Chest radiograph<br />
Syphilis serology<br />
Collagen vascular disease screen<br />
Serum chemistries<br />
Complete blood counts<br />
Lumbar puncture<br />
Lyme serology <strong>in</strong> patients from endemic areas<br />
<strong>Neuro</strong>imag<strong>in</strong>g<br />
MR imag<strong>in</strong>g of the bra<strong>in</strong> for all optic neuritis (class I–II, level B)<br />
Consider MR of head and orbit with fat suppression views to exam<strong>in</strong>e optic nerve course,<br />
especially <strong>in</strong> atypical optic neuritis
Optic Neuritis 45<br />
with CT scann<strong>in</strong>g, evoked potentials (EPs), and CSF analysis for oligoclonal bands (Paty,<br />
1988). Eighteen of these 19 (95%) patients had MR scans that were ‘‘strongly suggestive<br />
of MS’’ at first evaluation. Fourteen of 19 (74%) patients had positive oligoclonal bands.<br />
Ten of 19 (53%) patients had abnormal somatosensory EPs, 9 of 19 (47%) patients had<br />
abnormal visual EPs (VEPs), and 9 of 19 (47%) patients had abnormal CT scans.<br />
Comb<strong>in</strong><strong>in</strong>g multiple reports, the risk of develop<strong>in</strong>g MS with<strong>in</strong> 1 to 4 years is about<br />
30% (range 23–35%) <strong>in</strong> patients with isolated ON and an abnormal MR scan (Beck,<br />
1993a; Frederiksen, 1992; Jacobs, 1997; Söderström, 1998). Morrisey et al reported 89<br />
patients (44 with ON, 17 with bra<strong>in</strong>stem <strong>in</strong>volvement, and 28 with sp<strong>in</strong>al cord<br />
<strong>in</strong>volvement) with an acute cl<strong>in</strong>ical demyel<strong>in</strong>at<strong>in</strong>g attack (Morrisey, 1993). Of these 89<br />
patients, 57 (64%) had one or more MR scan abnormalities and 32 had no MR scan<br />
abnormalities. Only one of the 32 patients with normal MR scans developed MS, versus<br />
development of MS <strong>in</strong> 37 of 57 patients (65%) with an abnormal MR scan. Of the three<br />
isolated cl<strong>in</strong>ical syndromes (optic nerve, bra<strong>in</strong>stem, sp<strong>in</strong>al cord), ON with an abnormal<br />
MR scan had the highest rate of progression to MS—82%. Jacobs et al reported 42<br />
patients with isolated monosymptomatic optic neuritis (Jacobs, 1991). Dur<strong>in</strong>g 5.6 years<br />
of follow-up, 21 patients developed MS. Of these 21 patients, 16 (76%) had abnormal<br />
MR scans and 5 had normal MR scans (Jacobs, 1991).<br />
Söderström et al performed a prospective study of 147 consecutive patients with<br />
acute monosymptomatic ON (Söderström, 1998). Of 116 patients exam<strong>in</strong>ed with MR<br />
scans, 64 (55%) had three or more high signal lesions, 11 (9%) had one or two high signal<br />
lesions, and 41 (35%) had a normal MRI. Among 146 patients undergo<strong>in</strong>g CSF studies,<br />
oligoclonal bands were demonstrated <strong>in</strong> 103 (71%) patients. Dur<strong>in</strong>g the 6-year study<br />
period, 53 patients (36%) developed CDMS. Three or more MS lesions on MR scan or<br />
CSF oligoclonal bands were strongly associated with MS. Jacobs et al found that 42 of 74<br />
(57%) patients with isolated monosymptomatic ON had 1 to 20 bra<strong>in</strong> lesions by MR<br />
scans (Jacobs, 1997). All of the bra<strong>in</strong> lesions were cl<strong>in</strong>ically silent and had characteristics<br />
consistent with MS. Dur<strong>in</strong>g 5.6 years of follow-up, 21 patients (28%) developed CDMS.<br />
Sixteen of the 21 convert<strong>in</strong>g patients (76%) had abnormal MR scans; the other 5 (24%)<br />
had MR scans that were normal <strong>in</strong>itially (when they had ON only) and normal <strong>in</strong> 4 of<br />
the 5 when repeated after they had developed cl<strong>in</strong>ical MS. Of the 53 patients who had<br />
not developed CDMS, 26 (49%) had abnormal MR scans and 27 (51%) had normal MR<br />
scans. The authors concluded that the f<strong>in</strong>d<strong>in</strong>gs of an abnormal MR scan at the time of<br />
ON was significantly related to the subsequent development of MS. The <strong>in</strong>terpretation<br />
of the strength of that relationship must be tempered by the fact that some of the<br />
convert<strong>in</strong>g patients had normal MR scans and approximately half of the patients who<br />
did not develop cl<strong>in</strong>ical MS had abnormal MR scans. Thus, it should be emphasized<br />
that MS is a cl<strong>in</strong>ical diagnosis that cannot be made on the basis of MR scan<br />
abnormalities alone (Guy, 1994; Paty, 1993), and the absence of MR scan abnormalities<br />
does not protect aga<strong>in</strong>st the future development of MS (Beck, 1993d; Jacobs, 1991).<br />
The ONTT prospectively studied 388 patients who did not have probable or def<strong>in</strong>ite<br />
MS at study entry and who were followed for the development of CDMS (Optic<br />
Neuritis Study Group, 1997a). The 5-year cumulative probability of CDMS was 30% and<br />
did not differ by treatment group (see below). <strong>Neuro</strong>logic impairment <strong>in</strong> patients who<br />
developed CDMS was generally mild. Bra<strong>in</strong> MR scans performed at study entry was a<br />
strong predictor of CDMS, with the 5-year risk of CDMS rang<strong>in</strong>g from 16% <strong>in</strong> 202<br />
patients with no MR lesions to 51% <strong>in</strong> 89 patients with three or more MR lesions. The<br />
5-year risk of CDMS follow<strong>in</strong>g optic neuritis is dependent on the number of lesions
46 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
present on bra<strong>in</strong> MR scan. Even a normal bra<strong>in</strong> MRI, however, did not preclude the<br />
development of CDMS.<br />
MR scans may demonstrate contrast-enhanc<strong>in</strong>g lesions with<strong>in</strong> the optic nerve <strong>in</strong><br />
patients with ON (Cornblath, 1997; Dunker, 1996; Kupersmith, 2002a). Less complete<br />
visual recovery <strong>in</strong> ON was associated with longer lesions of the optic nerve and with<br />
<strong>in</strong>volvement of the <strong>in</strong>tracanicular segment <strong>in</strong> one study (Dunker, 1996). In another<br />
study, however, lesions <strong>in</strong>volv<strong>in</strong>g the canal or longer segments of the optic nerve had<br />
worse start<strong>in</strong>g vision, but the location and length of enhancement were not predictive of<br />
recovery (Kupersmith, 2002a).<br />
Should a Lumbar Puncture Be Performed <strong>in</strong><br />
Patients with Optic Neuritis?<br />
Patients with ON may show abnormalities <strong>in</strong> CSF analysis consistent with MS. These<br />
CSF abnormalities <strong>in</strong>clude <strong>in</strong>creased cell count (>5 cells per cubic millimeter), <strong>in</strong>creased<br />
total prote<strong>in</strong>, <strong>in</strong>creased CSF–immunoglobul<strong>in</strong> G (IgG) concentration, oligoclonal bands,<br />
antibodies to myel<strong>in</strong> basic prote<strong>in</strong> (MBP) and proteolipid prote<strong>in</strong> (PLP), and <strong>in</strong>creased<br />
CSF MBP levels (Cole, 1998; Fredericksen, 1992; Jacobs, 1997; Sellebjerg, 1994, 1995;<br />
Simon, 2000; Söderström, 1993, 1998; Warren, 1994). Lumbar puncture, however, did not<br />
produce any additional unsuspected diagnosis <strong>in</strong> the 141 patients <strong>in</strong> the ONTT undergo<strong>in</strong>g<br />
CSF analysis. In addition, a normal <strong>in</strong>itial CSF after ON did not preclude the<br />
eventual development of MS (Sandberg-Wollheim, 1975).<br />
Cole et al <strong>in</strong>vestigated the predictive value of CSF oligoclonal band<strong>in</strong>g for MS 5 years<br />
after optic neuritis <strong>in</strong> patients enrolled <strong>in</strong> the ONTT (Cole, 1998). In 76 patients, the<br />
presence of oligoclonal bands was associated with development of CDMS. However,<br />
the results suggested that CSF analysis was useful <strong>in</strong> the risk assessment of optic<br />
neuritis patients only when the bra<strong>in</strong> MR scan was normal and was not of predictive<br />
value when bra<strong>in</strong> MR scan lesions were present at the time of optic neuritis. CDMS<br />
developed with<strong>in</strong> 5 years <strong>in</strong> 22 of the 76 patients (29%); <strong>in</strong> 16 of 38 patients (42%) with<br />
oligoclonal bands present and <strong>in</strong> 6 of 38 patients (16%) without bands. Among the 39<br />
patients with normal MR scans, CDMS developed <strong>in</strong> three of 11 patients (27%) with<br />
bands present but <strong>in</strong> only one patient (4%) without bands. In contrast, among 37<br />
patients with abnormal MR scans, CDMS developed <strong>in</strong> 13 of 27 (48%) with bands and 5<br />
of 10 (50%) without bands. The positive predictive value of bands was 42% and the<br />
negative predictive value was 84%. Among the 39 patients with normal MR scans, the<br />
positive predictive value was 27% and the negative predictive value was 96%, whereas<br />
among the 37 patients with abnormal MR scans the positive predictive value was 48%<br />
and the negative predictive value was 50%.<br />
Although several authors have reported that abnormal CSF results may be predictive<br />
of eventual MS (Cole, 1998; Frederiksen, 1992; Jacobs, 1997; Sandberg-Wollheim, 1990;<br />
Söderström, 1998), others have not found CSF abnormalities to have predictive value<br />
(Sandberg-Wollheim, 1975). Although a lumbar puncture was optional <strong>in</strong> the ONTT, it<br />
should be considered <strong>in</strong> atypical ON or <strong>in</strong> cases where the diagnosis of MS might be<br />
clarified by CSF analysis (class I–II, level B).
Should Visual Evoked Potentials Be<br />
Performed on Patients with Optic Neuritis?<br />
Although the VEP is often abnormal <strong>in</strong> patients with ON (Ashworth, 1994; Brusa, 1999;<br />
Fotiou, 1999; Frederiksen, 1999; Fuhr, 2001; Honan, 1990; R<strong>in</strong>alduzzi, 2001), an abnormal<br />
VEP <strong>in</strong> the sett<strong>in</strong>g of a cl<strong>in</strong>ically diagnosed ON does not alter the diagnostic or<br />
treatment plan. The VEP does not provide additional prognostic <strong>in</strong>formation for visual<br />
recovery or for the development of MS. We do not recommend rout<strong>in</strong>e use of VEP <strong>in</strong><br />
typical ON (class III–IV, level C). VEP may be useful <strong>in</strong> identify<strong>in</strong>g a second site of<br />
neurologic <strong>in</strong>volvement (previous ON) to strengthen the cl<strong>in</strong>ical diagnosis of MS <strong>in</strong><br />
patients with no history or exam<strong>in</strong>ation f<strong>in</strong>d<strong>in</strong>gs of an optic neuropathy (Celesia, 1990).<br />
What Is the Treatment of Optic Neuritis?<br />
Optic Neuritis 47<br />
Although corticosteroids have been used for acute ON (Lessell, 1992; Sellebjerg, 1999;<br />
Silberberg, 1993) and have been shown to improve symptoms <strong>in</strong> MS (Kapoor, 1998),<br />
well-controlled data to support the treatment efficacy of steroids <strong>in</strong> ON have been<br />
lack<strong>in</strong>g until recently (Beck, 1992a). IV methylprednisolone (MP) treatment has been<br />
reported to decrease CSF anti-MBP levels, <strong>in</strong>trathecal IgG synthesis, and CSF oligoclonal<br />
bands; to decrease gadol<strong>in</strong>ium enhancement of MS plaques (and presumably blood–<br />
bra<strong>in</strong> barrier disruption) on MR scan; and to improve cl<strong>in</strong>ical disability. Modulation of<br />
the function of <strong>in</strong>flammatory cells may also contribute to the cl<strong>in</strong>ical efficacy or highdose<br />
corticosteroids (Sellebjerg, 2000). The cl<strong>in</strong>ical effect of treatment might be due to<br />
reduction of <strong>in</strong>flammation and myel<strong>in</strong> breakdown (Barkhoff, 1991a, 1992; Warren,<br />
1994).<br />
Rawson et al reported a more rapid visual recovery, but no difference <strong>in</strong> visual<br />
outcome after 1 year <strong>in</strong> a double-bl<strong>in</strong>d, placebo-controlled, prospective study of 50<br />
patients with ON treated with adrenocorticotropic hormone (ACTH) (Rawson, 1966).<br />
Rose et al observed similarly more rapid improvement <strong>in</strong> patients with ON treated with<br />
ACTH compared with placebo (Rose, 1970). Bowden et al, however, reported no benefit<br />
from ACTH compared with placebo <strong>in</strong> 54 patients with ON (Bowden, 1974). Gould et al<br />
reported a prospective, s<strong>in</strong>gle-bl<strong>in</strong>d, controlled, randomized cl<strong>in</strong>ical trial of 74 patients<br />
with ON who experienced more rapid improvement with a retrobulbar <strong>in</strong>jection of<br />
triamc<strong>in</strong>olone, but patients had no difference <strong>in</strong> outcome after 6 months (Gould, 1977).<br />
In the ONTT, the patients were randomly assigned to one of three treatment arms <strong>in</strong><br />
the study:<br />
1. IV methylprednisolone sodium succ<strong>in</strong>ate (250 mg every 6 hours for 3 days) followed<br />
by oral prednisone (1 mg=kg daily) for 11 days.<br />
2. Oral prednisone (1 mg=kg daily) for 14 days.<br />
3. Oral placebo for 14 days, followed by a short oral taper.<br />
The major conclusions of the ONTT related to treatment are summarized <strong>in</strong> Table 2–8.<br />
Wakakura et al also performed a randomized trial of IV megadose methylprednisolone<br />
<strong>in</strong> ON and found that treatment with steroids improved visual recovery at 3 weeks.<br />
Visual function at 12 weeks and at 1 year, however, was the same as <strong>in</strong> control patients<br />
(Wakakura, 1999a). Sellebjerg et al performed a randomized, controlled trial of oral
48 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 2–8. Summary of the ONTT F<strong>in</strong>d<strong>in</strong>gs<br />
Intravenous (IV) steroids followed by oral corticosteroids accelerated visual recovery but<br />
provided no long-term visual benefit.<br />
‘‘Standard dose’’ oral prednisone alone did not improve the visual outcome and was associated<br />
with an <strong>in</strong>creased rate of new attacks of ON.<br />
IV followed by oral corticosteroids reduced the rate of development of cl<strong>in</strong>ically def<strong>in</strong>ite MS<br />
(CDMS) dur<strong>in</strong>g the first 2 years, but by 3 years the effect had subsided (Beck, 1993b, 1995a).<br />
MR f<strong>in</strong>d<strong>in</strong>gs were of prognostic significance for MS.<br />
Treatment was well tolerated with few major side effects.<br />
high-dose methylprednisolone (500 mg daily for 5 days with a 10-day taper) <strong>in</strong> 30<br />
patients compared to 30 control patients (Sellebjerg, 1999). The visual analog scale but<br />
not spatial visual function was better <strong>in</strong> the steroid group at 3 weeks. After 8 weeks, the<br />
visual analog scale and spatial visual function were comparable <strong>in</strong> both groups. The risk<br />
of new demyel<strong>in</strong>at<strong>in</strong>g attacks with<strong>in</strong> 1 year was unaffected by treatment. In another<br />
study, 55 patients with acute ON received IV sal<strong>in</strong>e or IV MP and were assessed at 6<br />
months (Kapoor, 1998). Patients with short lesions of the optic nerve on MR scan<br />
presented earlier than those with long lesions (<strong>in</strong>volv<strong>in</strong>g three or more 5-mm-thick<br />
slices of any part of the optic nerve, as well as its <strong>in</strong>tracanalicular portion). Lesion length<br />
was significantly less <strong>in</strong> patients present<strong>in</strong>g with<strong>in</strong> a week of onset of symptoms.<br />
Treatment did not limit lesion length <strong>in</strong> either the long or short lesion subgroups and<br />
had no significant effect on f<strong>in</strong>al visual outcome. The authors conclude that steroids do<br />
not improve visual outcome or lesion length <strong>in</strong> patients with acute ON (Kapoor, 1998).<br />
Based on the ONTT results, it is recommended that treatment with oral prednisone <strong>in</strong><br />
standard doses be avoided <strong>in</strong> ON (Kaufman, 2000) (class I, level A). Treatment with IV<br />
MP should be considered <strong>in</strong> patients with abnormal MR scans of the bra<strong>in</strong> or a<br />
particular need (e.g., monocular patient or occupational requirement) to recover<br />
visual function more rapidly (class I, level B). Beck et al thought that although bra<strong>in</strong><br />
MR scan may not be necessary for the diagnosis of ON, imag<strong>in</strong>g was valuable for<br />
prognostic purposes (Beck, 1992, 1993a–e). In the ONTT, patients with multiple signal<br />
abnormalities on MR scans most clearly benefited from IV corticosteroid therapy <strong>in</strong><br />
terms of development of MS. The rate of development of MS was too low <strong>in</strong> the patients<br />
with normal MR scans to assess treatment benefit <strong>in</strong> this group. ON patients <strong>in</strong> the<br />
ONTT had MR scans with<strong>in</strong> 9 days of the onset of visual loss. Some authors have<br />
suggested that patients present<strong>in</strong>g later than this <strong>in</strong>terval with an abnormal MR scan<br />
may still benefit from treatment with IV MP with<strong>in</strong> a treatment w<strong>in</strong>dow of about 2<br />
months (Guy, 1994). The results of the ONTT have led to a reduction <strong>in</strong> the use or oral<br />
corticosteroids <strong>in</strong> the treatment of ON (Trobe, 1999).<br />
Even though the ONTT was a large, well-designed study, several criticisms have been<br />
raised:<br />
1. The lack of an <strong>in</strong>travenous control group.<br />
2. Incomplete mask<strong>in</strong>g of all patients (i.e., <strong>in</strong>-hospital IV-treated patients knew they had<br />
received IV MP).<br />
3. Data regard<strong>in</strong>g treatment effect of IV MP on the development of MS was obta<strong>in</strong>ed<br />
from a retrospective analysis that was primarily designed for a different purpose (to<br />
evaluate the treatment effect).
4. The role of retrobulbar steroids was not assessed.<br />
5. The role of higher doses of MP, such as 30 mg per kg dose suggested for the treatment<br />
of acute sp<strong>in</strong>al cord <strong>in</strong>jury, was not determ<strong>in</strong>ed.<br />
6. The efficacy of oral prednisone at higher doses was not assessed.<br />
7. The need or lack of need for oral taper<strong>in</strong>g doses of corticosteroids follow<strong>in</strong>g IV MP<br />
was not addressed.<br />
Despite these concerns, the ONTT is the best well-controlled prospective cl<strong>in</strong>ical trial<br />
(class I) available <strong>in</strong> the literature to date on the treatment and evaluation of ON. We<br />
follow the evaluation and the treatment recommendations of the ONTT (class I, level B).<br />
Should Interferon Therapy Be Instituted <strong>in</strong><br />
Patients with Optic Neuritis?<br />
In a double-bl<strong>in</strong>d, randomized trial, 383 patients who had a first acute demyel<strong>in</strong>at<strong>in</strong>g<br />
event (optic neuritis, <strong>in</strong>complete transverse myelitis, or a bra<strong>in</strong>stem or cerebellar<br />
syndrome) were studied. All had evidence of prior subcl<strong>in</strong>ical demyel<strong>in</strong>ation on MR<br />
imag<strong>in</strong>g of the bra<strong>in</strong> (two or more silent lesions of at least 3 mm <strong>in</strong> diameter thought<br />
characteristic of MS). Patients received either weekly <strong>in</strong>tramuscular <strong>in</strong>jections of 30 mgof<br />
<strong>in</strong>terferon-b-1a (193 patients) or placebo (190 patients) (CHAMPS Study Group, 2001;<br />
Jacobs, 2000). The patients had received <strong>in</strong>itial treatment with corticosteroids. Dur<strong>in</strong>g 3<br />
years of follow-up, the cumulative probability of the development of CDMS was<br />
significantly lower <strong>in</strong> the <strong>in</strong>terferon-b-1a group than <strong>in</strong> the placebo group (rate ratio,<br />
0.56). At 3 years, the cumulative probability was 35% <strong>in</strong> the <strong>in</strong>terferon-b-1a group and<br />
50% <strong>in</strong> the placebo group. As compared with the patients <strong>in</strong> the placebo group, patients<br />
<strong>in</strong> the <strong>in</strong>terferon-b-1a group had a relative reduction <strong>in</strong> the volume of bra<strong>in</strong> lesions,<br />
fewer new lesions or enlarg<strong>in</strong>g lesions, and fewer gadol<strong>in</strong>ium-enhanc<strong>in</strong>g lesions at 18<br />
months. The authors concluded that <strong>in</strong>itiat<strong>in</strong>g treatment with <strong>in</strong>terferon-b-1a at the time<br />
of a first demyel<strong>in</strong>at<strong>in</strong>g event is beneficial for patients with bra<strong>in</strong> lesions on MRI that<br />
<strong>in</strong>dicate high risk of CDMS (CHAMPS Study Group, 2001; Jacobs, 2000).<br />
Are There Treatments Other than Steroids for<br />
Optic Neuritis?<br />
Intravenous immunoglobul<strong>in</strong> (IVIg) had been <strong>in</strong>itially reported to improve visual acuity<br />
<strong>in</strong> an uncontrolled study of five patients with def<strong>in</strong>ite MS and unilateral or bilateral but<br />
stable demyel<strong>in</strong>at<strong>in</strong>g ON (van Engelen, 1992). However, <strong>in</strong> a randomized trial <strong>in</strong> 55<br />
patients, this agent did not reverse persistent visual loss from ON to a degree that merits<br />
general use (Noseworthy, 2001).<br />
What Is the Long-Term Vision Prognosis of<br />
Patients with Optic Neuritis?<br />
Optic Neuritis 49<br />
In patients with ON, visual recovery generally beg<strong>in</strong>s with<strong>in</strong> the first 2 weeks, with<br />
much of the recovery occurr<strong>in</strong>g by the end of 1 month. If recovery is <strong>in</strong>complete at 6<br />
months, some further improvement may cont<strong>in</strong>ue for up to 1 year.
50 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
In the ONTT, there was no significant difference <strong>in</strong> visual acuity compar<strong>in</strong>g the three<br />
treatment groups at 6 months. After 12 months, visual acuity was 20=40 or greater <strong>in</strong><br />
93% of patients, greater than 20=20 <strong>in</strong> 69%, and 20=200 or lower <strong>in</strong> 3%. Results were<br />
similar <strong>in</strong> each treatment group. The only predictor of poor visual outcome was poor<br />
visual acuity at the time of study entry; even so, of 160 patients start<strong>in</strong>g with a visual<br />
acuity of 20=200 or worse, all had at least some improvement and only 8 (5%) had<br />
visual acuities that were still 20=200 or worse at 6 months. Of 30 patients whose <strong>in</strong>itial<br />
visual acuity was light perception (LP) or no light perception (NLP), 20 (67%) recovered<br />
to 20=40 or better. Basel<strong>in</strong>e acuity was the best predictor of 6-month visual acuity<br />
outcome; older age was statistically associated with a slightly worse outcome but this<br />
appeared to be of no cl<strong>in</strong>ical importance.<br />
Thus, <strong>in</strong> most patients with ON, visual recovery is rapid. The only factor of value <strong>in</strong><br />
predict<strong>in</strong>g the visual outcome is <strong>in</strong>itial severity of visual loss. However, even when<br />
<strong>in</strong>itial loss is severe, visual recovery is still good <strong>in</strong> most patients. Patients not follow<strong>in</strong>g<br />
the usual course of visual recovery should be considered atypical and further <strong>in</strong>vestigation<br />
<strong>in</strong> regard to etiology of the visual loss is appropriate.<br />
At the 5-year follow-up for 347 (64%) of 545 patients <strong>in</strong> the ONTT, the affected eyes<br />
had normal or only slightly abnormal visual acuities <strong>in</strong> most patients, and results did<br />
not significantly differ by treatment group (Optic Neuritis Study Group, 1997b). Visual<br />
acuity <strong>in</strong> affected eyes was 20=25 or better <strong>in</strong> 87%, 20=25 to 20=40 <strong>in</strong> 7%, 20=50 to 20=190<br />
<strong>in</strong> 3%, and 20=200 or worse <strong>in</strong> 3%. Recurrence of ON <strong>in</strong> either eye occurred <strong>in</strong> 28% of<br />
patients and was more frequent <strong>in</strong> patients with MS and <strong>in</strong> patients without MS who<br />
were <strong>in</strong> the prednisone treatment group. Most eyes with a recurrence reta<strong>in</strong>ed normal or<br />
almost normal visual function. In conclusion, most patients with ON reta<strong>in</strong> good or<br />
excellent vision 5 years follow<strong>in</strong>g an attack of ON, even if the ON recurs. Recurrences<br />
are more frequent <strong>in</strong> patients with MS and <strong>in</strong> those treated with oral prednisone alone.<br />
Recurrence of ON <strong>in</strong> either eye occurs <strong>in</strong> 28% of patients and are twofold more frequent<br />
<strong>in</strong> patients who had or developed CDMS (46%) compared with patients without CDMS<br />
(22%).<br />
What Is the Risk of Develop<strong>in</strong>g Multiple<br />
Sclerosis follow<strong>in</strong>g Optic Neuritis?<br />
The risk for the development of MS follow<strong>in</strong>g ON is quite variable <strong>in</strong> the literature, with<br />
reports rang<strong>in</strong>g from 8 to 85% (Cole, 1998; Frith, 2000; Jacobs, 1997; Optic Neuritis<br />
Study Group, 1997a; Rodriguez, 1995; Söderström, 1998; Sorensen, 1999). Most<br />
studies <strong>in</strong>dicate a 25 to 35% risk of patients with ON develop<strong>in</strong>g MS. This variability<br />
is probably related to numerous factors <strong>in</strong>clud<strong>in</strong>g:<br />
Differences <strong>in</strong> patient populations (e.g., cl<strong>in</strong>ic or hospital versus population based);<br />
sample sizes; study design (retrospective versus prospective)<br />
Duration of follow-up (longer <strong>in</strong>terval studies tend to report higher <strong>in</strong>cidence rates)<br />
Differences <strong>in</strong> selection criteria and diagnostic evaluation of ON cases<br />
Different study diagnostic criteria for both ON and MS<br />
Rodriguez et al found a cumulative probability of develop<strong>in</strong>g CDMS of 24% after 5<br />
years and 39% after 10 years and noted no difference <strong>in</strong> the risk of develop<strong>in</strong>g MS
etween men and women (Rodriguez, 1995). Rizzo and Lessell studied 60 patients with<br />
ON, with a mean follow-up of 14.9 years (Rizzo, 1988). Life table analysis <strong>in</strong>dicated that<br />
74% of the women and 34% of the men developed MS 15 years after their attack of ON,<br />
and 91.3% of the women and 44.8% of the men would develop MS after 20 years. MR<br />
scan abnormalities may be the best predictor for the eventual development of MS after<br />
ON. As noted above, <strong>in</strong> the ONTT prospective study of 388 patients who did not have<br />
probable or def<strong>in</strong>ite MS at study entry, the 5-year cumulative probability of CDMS was<br />
30% (Optic Neuritis Study Group, 1997a). Bra<strong>in</strong> MR scans performed at study entry<br />
were a strong predictor of CDMS, with the 5-year risk of CDMS rang<strong>in</strong>g from 16% <strong>in</strong><br />
202 patients with no MR lesions to 51% <strong>in</strong> 89 patients with three or more MR lesions.<br />
The 5-year risk of CDMS follow<strong>in</strong>g ON is highly dependent on the number of lesions<br />
present on bra<strong>in</strong> MR scan. Sorensen et al studied the predictive value on survival of ON<br />
as onset manifestation of MS and concluded that ON as onset manifestation of MS (vs.<br />
another or unknown onset manifestation of MS) <strong>in</strong>dicates a more favorable prognosis of<br />
survival of MS <strong>in</strong> women (Sorensen, 1999). ON was the present<strong>in</strong>g manifestation of MS<br />
<strong>in</strong> 10% of MS cases.<br />
Brex et al performed high-resolution, multisequence bra<strong>in</strong> and sp<strong>in</strong>al cord MRI <strong>in</strong> 60<br />
patients after their first demyel<strong>in</strong>at<strong>in</strong>g event, <strong>in</strong>clud<strong>in</strong>g 38 patients with ON (Brex,<br />
1999). At basel<strong>in</strong>e, 73% of patients had lesions on T2-weighted fast sp<strong>in</strong>-echo (FSE) bra<strong>in</strong><br />
images and 42% had asymptomatic sp<strong>in</strong>al cord lesions. Of the 38 patients with ON, 29<br />
had lesions <strong>in</strong> the bra<strong>in</strong> on FSE images and 16 had sp<strong>in</strong>al cord lesions. Repeat MRI<br />
demonstrated new FSE lesions <strong>in</strong> 43% of the patients overall. After 1 year, 26% of the<br />
patients developed MS. The MRI features that provided the best comb<strong>in</strong>ation of<br />
sensitivity and specificity for the development of MS were new FSE lesions at followup<br />
and enhanc<strong>in</strong>g lesions at basel<strong>in</strong>e. The authors concluded that the comb<strong>in</strong>ation of<br />
basel<strong>in</strong>e MR abnormalities on multisequence MRI and new lesions at follow-up,<br />
Table 2–9. Risk Factors for Develop<strong>in</strong>g Multiple Sclerosis Follow<strong>in</strong>g Optic Neuritis<br />
Optic Neuritis 51<br />
Factor<br />
Increased risk<br />
References<br />
Abnormal MR scan (three or more lesions) Jacobs, 1997; Optic Neuritis Study Group, 1997a;<br />
Söderström, 1998<br />
Prior nonspecific neurologic symptoms ONTT<br />
Increased CSF oligoclonal bands Cole, 1998<br />
Increased CSF IgG Jacobs, 1997; Söderström, 1998<br />
Previous optic neuritis ONTT<br />
HLA-DR2 and HLA-B7<br />
Decreased risk<br />
Morrissey, 1993<br />
Normal MR scan ONTT<br />
Absence of pa<strong>in</strong>* ONTT<br />
Marked disc edema* ONTT<br />
Ret<strong>in</strong>al exudates or macular star* ONTT<br />
Bilateral simultaneous onset* Frederiksen, 1997a<br />
Onset <strong>in</strong> childhood* Lucch<strong>in</strong>etti, 1997<br />
*We consider these f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a patient with ON to be atypical and thus likely require further evaluation.<br />
ONTT, Optic Neuritis Treatment Trial.
52 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
<strong>in</strong>dicat<strong>in</strong>g dissem<strong>in</strong>ation <strong>in</strong> space and time, are associated with a high sensitivity and<br />
specificity for the early development of cl<strong>in</strong>ical MS.<br />
Ghezzi et al evaluated the risk of SDMS after acute isolated ON <strong>in</strong> 102 patients with<br />
follow-up duration 6.3 years (10 patients were lost to follow-up) (Ghezzi, 1999, 2000).<br />
The risk of develop<strong>in</strong>g SDMS was 13% after 2 years, 30% after 4 years, 37% after 6 years,<br />
and 42% after 8 and 10 years. Gender, age, and season of ON onset did not affect the<br />
risk. CDMS occurred <strong>in</strong> 37 of 71 patients (52.1%) with one MRI lesion or more; no<br />
Figure 2-1. Evaluation of optic neuritis.
patient with a normal MRI developed CDMS. CDMS developed more frequently <strong>in</strong><br />
patients with <strong>in</strong>trathecal IgG synthesis than <strong>in</strong> those without (43% vs. 28%), but the<br />
difference was not statistically significant (Ghezzi, 1999, 2000).<br />
Increased risk of MS has been reported <strong>in</strong> patients with human leukocyte antigen<br />
HLA-DR2 and HLA-B7 tissue types, but we do not recommend rout<strong>in</strong>e HLA screen<strong>in</strong>g<br />
for ON (Hauser, 2000; Morrissey, 1993). Risk factors for develop<strong>in</strong>g MS follow<strong>in</strong>g ON<br />
are outl<strong>in</strong>ed <strong>in</strong> Table 2–9.<br />
Our approach to ON is summarized <strong>in</strong> Figure 2–1.<br />
References<br />
Optic Neuritis 53<br />
Abd Elrazek M. (1991). Brucella optic neuritis. Arch Intern Med 151:776–778.<br />
Achiron LR. (1994). Post<strong>in</strong>fectious hepatitis B optic neuritis. Optom Vis Sci 71:53–56.<br />
Agildere AM, Tutar NU, Yucel E, et al. (1999). Pachymen<strong>in</strong>gitis and optic neuritis <strong>in</strong> rheumatoid arthritis: MRI<br />
f<strong>in</strong>d<strong>in</strong>gs. Br J Radiol 72:404–407.<br />
Ahasan HA, Jafiqueudd<strong>in</strong> AK, Chowdrhury MA, et al. (1994). <strong>Neuro</strong>myelitis optica (Devic’s disease) follow<strong>in</strong>g<br />
chicken pox. Trop Doct 24:75–76.<br />
Ahmadieh H, Roodpeyma S, Azarm<strong>in</strong>a M, et al. (1994). Bilateral simultaneous optic neuritis <strong>in</strong> childhood<br />
systemic lupus erythematosus. A case report. J <strong>Neuro</strong>-ophthalmol 14:84–86.<br />
Albitar S, Bourgeon B, Gen<strong>in</strong> R, et al. (1997). Bilateral retrobulbar optic neuritis with hepatitis B vacc<strong>in</strong>ation.<br />
Nephrol Dial Transplant 12:2169–2170.<br />
Al-Deeb SM, Yaqub BA, Khoja WO. (1993). Devic’s neuromyelitis optica and varicella. J <strong>Neuro</strong>l 240:450–451.<br />
<strong>An</strong>derson MD, Kennedy CA, Lewis AW, Christensen GR. (1994). Retrobulbar neuritis complicat<strong>in</strong>g acute Epste<strong>in</strong>-<br />
Barr virus <strong>in</strong>fection. Cl<strong>in</strong> Infect Dis 18:799–801.<br />
Arnold AC. (1999). Visual field defects <strong>in</strong> the Optic Neuritis Treatment Trial: central vs peripheral, focal vs global.<br />
Am J Ophthalmol 128:632–634.<br />
Arnold RW, Schriever G. (1993). Lyme amaurosis <strong>in</strong> a child. J Pediatr Ophthalmol Strabismus 30:268–270.<br />
Ashworth B, Asp<strong>in</strong>all PA, Mitchell JD. (1994). Visual function <strong>in</strong> multiple sclerosis. Doc Ophthalmol 73:209–224.<br />
Banta JT, Lam BL. (2002). Toxoplasmic anterior optic neuropathy. Presented at the 28th annual meet<strong>in</strong>g of the<br />
North American <strong>Neuro</strong>-Ophthalmology Society. Copper Mounta<strong>in</strong>, Colorado, February 9–14.<br />
Barkhoff F, Frequ<strong>in</strong> STFM, Hommes OR, et al. (1992). A correlative triad of gadol<strong>in</strong>ium-DTPA MRI, EDSS, and<br />
CSF-MBP <strong>in</strong> relaps<strong>in</strong>g multiple sclerosis patients treated with high-dose <strong>in</strong>travenous methylprednisolone.<br />
<strong>Neuro</strong>logy 42:63–67.<br />
Barkhoff F, Hommes OR, Lamers KJB, et al. (1991a). High dose <strong>in</strong>travenous methylprednisolone <strong>in</strong> multiple<br />
sclerosis. <strong>Neuro</strong>logy 41:1219–1222.<br />
Barkhoff F, Scheltens P, Valk J, et al. (1991b). Serial quantitative MR assessment of optic neuritis <strong>in</strong> a case of<br />
neuromyelitis optica, us<strong>in</strong>g gadol<strong>in</strong>ium-‘‘enhanced’’ STIR imag<strong>in</strong>g. <strong>Neuro</strong>radiology 33:70–71.<br />
Bath AP, Rowe JR, Innes AJ. (1998). Malignant otitis externa with optic neuritis. J Laryngol Otol 112:274–277.<br />
Baumhefner RO, Tourtellotte WW, Syndulko K, et al. (1990). Quantitative multiple sclerosis plaque assessment<br />
with magnetic resonance imag<strong>in</strong>g: its correlation with cl<strong>in</strong>ical parameters, evoked potentials, and <strong>in</strong>trablood-barrier<br />
IgG synthesis. Arch <strong>Neuro</strong>1 47:19–26.<br />
Beck AD, Newman NJ, Grossniklaus HE, et al. (1994a). Optic nerve enlargement and chronic visual loss. Surv<br />
Ophthalmol 38:555–566.<br />
Beck RW. (1992a). Corticosteroid treatment of optic neuritis. <strong>Neuro</strong>logy 42:1133–1135.<br />
Beck RW. (1995a). The optic neuritis treatment trial: three year follow-up results. Arch Ophthalmol 113:136–137.<br />
Beck RW, Arr<strong>in</strong>gton I, Murtagh FR, et al. (1993a). Bra<strong>in</strong> magnetic resonance imag<strong>in</strong>g <strong>in</strong> acute optic neuritis:<br />
experience of the optic neuritis study group. Arch <strong>Neuro</strong>l 50:841–846.<br />
Beck RW, Cleary PA. (1993b). Recovery from severe visual loss <strong>in</strong> optic neuritis. Arch Ophthalmol 111:300.<br />
Beck RW, Cleary PA. (1993c). Optic neuritis treatment trial. One year follow-up results. Arch Ophthalmol<br />
111:773–775.<br />
Beck RW, Cleary PA, <strong>An</strong>derson MA, et al. (1992b). A randomized, controlled trial of corticosteroids <strong>in</strong> the<br />
treatment of acute optic neuritis. N Engl J Med 326:581–588.<br />
Beck RW, Cleary PA, Backlund JC. (1994b). The course of visual recovery after optic neuritis. Ophthalmology<br />
101:1771–1778.
54 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Beck RW, Cleary PA, Trobe JD, et al. (1993d). The effect of corticosteroids for acute optic neuritis on the subsequent<br />
development of multiple sclerosis. N Engl J Med 329:1764–1769.<br />
Beck RW, Kupersmith MJ, Cleary PA, Katz B. (1993e). Fellow eye abnormalities <strong>in</strong> acute unilateral optic neuritis.<br />
Ophthalmology 100:691–698.<br />
Beck RW, Trobe J. (1995b). What we have learned from the optic neuritis treatment trial. Ophthalmology<br />
102:1504–1508.<br />
Beiran I, Krasnitz I, Zimhoni-Eibsitz M, et al. (2000). Paediatric chiasmal neuritis—typical of post-Epste<strong>in</strong>-Barr<br />
virus <strong>in</strong>fection? Acta Ophthalmol 78:226–227.<br />
Berrios RR, Serrano LA. (1994). Bilateral optic neuritis after a bee st<strong>in</strong>g. Am J Ophthalmol 117:677–678.<br />
Bielory L, Kupersmith M, Warren F, et al. (1993). Sk<strong>in</strong> biopsies <strong>in</strong> the evaluation of atypical optic neuropathies.<br />
Ocul Immunol Inflam 1:231–241.<br />
Biousse V, Trichet C, Bloch-Michel E, Roullet E. (1999). Multiple sclerosis associated with uveitis <strong>in</strong> two large<br />
cl<strong>in</strong>ic-based series. <strong>Neuro</strong>logy 52:179–181.<br />
Blanche P, Diaz E, Gombert B, et al. (2000). Devic’s neuromyelitis optica and HIV-1 <strong>in</strong>fection. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 68:795–796.<br />
Boiko AN, Guseva ME, Guseva MR, et al. (2000). Cl<strong>in</strong>ico-immunogenetic characteristics of multiple sclerosis optic<br />
neuritis <strong>in</strong> children. J <strong>Neuro</strong>virol 6(suppl 2):S152–S155.<br />
Bowden AN, Bowden PMA, Friedmann AI, et al. (1974). A trial of corticotroph<strong>in</strong> gelat<strong>in</strong> <strong>in</strong>jection <strong>in</strong> acute optic<br />
neuritis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 37:869–873.<br />
Brady KM, Brar AS, Lee AG, et al. (1999). Optic neuritis <strong>in</strong> children: cl<strong>in</strong>ical features and visual outcome. JAm<br />
Assoc Pediatr Ophthalmol Strabismus 3:98–103.<br />
Brazis PW, Stokes HR, Erv<strong>in</strong> FR. (1986). Optic neuritis <strong>in</strong> cat scratch disease. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 6:172–174.<br />
Brex PA, O’Riordan JI, Miszkiel KA, et al. (1999). Multisequence MRI <strong>in</strong> cl<strong>in</strong>ically isolated syndromes and the<br />
early development of MS. <strong>Neuro</strong>logy 53:1184–1190.<br />
Brodsky MC, Beck RW. (1994). The chang<strong>in</strong>g role of MR imag<strong>in</strong>g <strong>in</strong> the evaluation of acute optic neuritis.<br />
Radiology 192:22–23.<br />
Brusa A, Jones SJ, Kapoor R, et al. (1999). Long-term recovery and fellow eye deterioration after optic neuritis,<br />
determ<strong>in</strong>ed by serial visual evoked potentials. J <strong>Neuro</strong>l 246:776–782.<br />
Burton BJL, Leff AP, Plant GT. (1998). Steroid-responsive HIV optic neuropathy. J <strong>Neuro</strong>-ophthalmol 18:25–29.<br />
Case records of the Massachusetts General Hospital. Case 37-1996. (1996). N Engl J Med 335:1668–1674.<br />
Celesia GG, Kaufman DI, Brigell M, et al. (1990). Optic neuritis: A prospective study. <strong>Neuro</strong>logy 40:919–923.<br />
CHAMPS Study Group. (2001). Interferon beta-1a for optic neuritis patients at high risk for multiple sclerosis.<br />
Am J Ophthalmol 132:463–471.<br />
Chan JW. (2002). Optic neuritis <strong>in</strong> anti-GQ1b positive Miller Fisher syndrome. Presented at the 28th annual<br />
meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Copper Mounta<strong>in</strong>, Colorado, February 9–14.<br />
Chang GY, Keane JR. (2001). Visual loss <strong>in</strong> cysticercosis: analysis of 23 patients. <strong>Neuro</strong>logy 57:545–548.<br />
Choi MY, Cho SH. (2000). Optic neuritis after bee st<strong>in</strong>g. Korean J Ophthalmol 14:49–52.<br />
Christiansen P, Frederiksen JL, Henriksen O, et al. (1992). Gd-DTPA-enhanced lesions <strong>in</strong> the bra<strong>in</strong> of patients with<br />
acute optic neuritis. Acta <strong>Neuro</strong>l Scand 85:141–146.<br />
Cleary PA, Beck RW, <strong>An</strong>derson MM, et al. (1993). Design, methods and conduct of the Optic Neuritis Treatment<br />
Trial. Controlled Cl<strong>in</strong> Trials 24:123–142.<br />
Cleary PA, Beck RW, Bourque LB, et al. (1997). Visual symptoms after optic neuritis: results from the Optic<br />
Neuritis Treatment Trial. J <strong>Neuro</strong>-ophthalmol 17:1–28.<br />
Cole SR, Beck RW, Moke PS, et al. (1998). The predictive value of CSF oligoclonal band<strong>in</strong>g for MS 5 years after<br />
optic neuritis. <strong>Neuro</strong>logy 51:885–887.<br />
Cornblath WT, Qu<strong>in</strong>t DJ. (1997). MRI of optic nerve enlargement <strong>in</strong> optic neuritis. <strong>Neuro</strong>logy 48:821–825.<br />
Corssmit EP, Leverste<strong>in</strong>-van Hall MA, Portegies P, Bakker P. (1997). Severe neurological complications <strong>in</strong><br />
association with Epste<strong>in</strong>-Barr virus <strong>in</strong>fection. J <strong>Neuro</strong>virol 3:460–464.<br />
Deane JS, Bibby K. (1995). Bilateral opiic neuritis follow<strong>in</strong>g herpes zoster ophthalmicus. Arch Ophthalmol<br />
113:972–973.<br />
DeBroft BM, Donahue SP. (1993). Bilateral optic neuropathy as the <strong>in</strong>itial manifestation of systemic sarcoidosis.<br />
Am J Ophthalmol 116:108–110.<br />
Dunker S, Wiegand W. (1996). Prognostic value of magnetic resonance imag<strong>in</strong>g <strong>in</strong> monosymptomatic optic<br />
neuritis. Ophthalmology 103:1768–1773.<br />
Falcone PM, Notis C, Merhige K. (1993). Toxoplasmosis papillitis as the <strong>in</strong>itial manifestation of acquired<br />
immunodeficiency syndrome. <strong>An</strong>n Ophthalmol 25:56–57.<br />
Fang JP, Donahue SP, L<strong>in</strong> RH. (1999a). Global visual field <strong>in</strong>volvement <strong>in</strong> acute unilateral optic neuritis.<br />
Am J Ophthalmol 128:554–565.
Optic Neuritis 55<br />
Fang JP, L<strong>in</strong> RH, Donahue SP. (1999b). Recovery of visual field function <strong>in</strong> the Optic Neuritis Treatment Trial.<br />
Am J Ophthalmol 128:566–572.<br />
Farris BK, Pickard DJ. (1990). Bilateral post<strong>in</strong>fectious optic neuritis and <strong>in</strong>travenous steroid therapy <strong>in</strong> children.<br />
Ophthalmology 97:339–345.<br />
Fe<strong>in</strong>ste<strong>in</strong> A, Youl B, Ron M. (1992). Acute optic neuritis. Bra<strong>in</strong> 115:1403–1415.<br />
Filippi M, Rocca MA, Moiola L, et al. (1999). MRI and magnetization transfer imag<strong>in</strong>g changes <strong>in</strong> the bra<strong>in</strong> and<br />
cervical cord of patients with Devic’s neuromyelitis optica. <strong>Neuro</strong>logy 53:1705–1710.<br />
Fotiou F, Koutlas E, Tsorl<strong>in</strong>is I, et al. (1999). The value of neurophysiological and MRI assessment <strong>in</strong><br />
demyel<strong>in</strong>at<strong>in</strong>g optic neuritis. Electromyograph Cl<strong>in</strong> <strong>Neuro</strong>physiol 39:397–404.<br />
Francis CS, Evens AC, Arnold DL. (1995). <strong>Neuro</strong>imag<strong>in</strong>g <strong>in</strong> multiple sclerosis. <strong>Neuro</strong>l Cl<strong>in</strong> 23:147–170.<br />
Frederiksen JL. (1997a). Bilateral acute optic neuritis: prospective cl<strong>in</strong>ical, MRI, CSF, neurophysiological and HLA<br />
f<strong>in</strong>d<strong>in</strong>gs. <strong>Neuro</strong>-<strong>ophthalmology</strong> 17:175–183.<br />
Frederiksen JL, Larsson HBW, Olesen J. (1992). Correlation of magnetic resonance imag<strong>in</strong>g and CSF f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong><br />
patients with acute monosymptomatic optic neuritis. Acta <strong>Neuro</strong>l Scand 86:317–322.<br />
Frederiksen JL, Larsson HBW, Olesen J, Stigsby B. (1991a). MR, VEP, SEP, and biothesiometry suggest<br />
monosymptomatic acute optic neuritis to be a first manifestation of multiple sclerosis. Acta <strong>Neuro</strong>l Scand<br />
83:34–350.<br />
Frederiksen JL, Larsson HBW, Ottovay E, Stigsby B, Olesen J. (1991b). Acute optic neuritis with normal visual<br />
acuity. Acta Ophthalmol 69:357–366.<br />
Frederiksen JL, Petrera J. (1999). Serial visual evoked potentials <strong>in</strong> 90 untreated patients with acute optic neuritis.<br />
Surv Ophthalmol 44(suppl 1):S54–S62.<br />
Frederiksen JL, Sorensen TL, Sellebjerg FT. (1997b). Residual symptoms and signs after untreated acute optic<br />
neuritis. Acta Ophthalmol Scand 75:544–547.<br />
Friedlander SM, Rahhal FM, Ericson L, et al. (1996). Optic neuropathy preced<strong>in</strong>g acute ret<strong>in</strong>al necrosis <strong>in</strong> acquired<br />
immunodeficiency syndrome. Arch Ophthalmol 114:1481–1485.<br />
Friedman DI. (1991). <strong>Neuro</strong>-ophthalmic manifestations of human immunodeficiency virus <strong>in</strong>fection. <strong>Neuro</strong>l Cl<strong>in</strong><br />
9:55–72.<br />
Frith JA, McLeod JG, Hely M. (2000). Acute optic neuritis <strong>in</strong> Australia: a 13 year prospective study. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 68:246.<br />
Frohman L, Wolansky L. (1997). Magnetic resonance imag<strong>in</strong>g of syphilitic optic neuritis=per<strong>in</strong>euritis. J <strong>Neuro</strong>ophthalmol<br />
17:57–59.<br />
Fuhr P, Borggrefe-Chappuis A, Sch<strong>in</strong>dler C, Kappos L. (2001). Visual and motor evoked potentials <strong>in</strong> the course of<br />
multiple sclerosis. Bra<strong>in</strong> 124:2162–2168.<br />
Fujimoto N, Adachi-Usami E, Saito E, Nagata H. (1999). Optic nerve bl<strong>in</strong>dness due to paranasal s<strong>in</strong>us disease.<br />
Ophthalmologica 213:262–264.<br />
Gal<strong>in</strong>do-Rodriguez G, Av<strong>in</strong>a-Zubieta JA, Pizarro S, et al. (1999). Cyclophosphamide pulse therapy <strong>in</strong> optic<br />
neuritis due to systemic lupus erythematosus. Am J Med 106:65–69.<br />
Gerl<strong>in</strong>g J, Jancknecht P, Kommerell G. (1998a). Orbital pa<strong>in</strong> <strong>in</strong> optic neuritis and anterior ischemic<br />
optic neuropathy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:93–99.<br />
Gerl<strong>in</strong>g J, Meyer JH, Kommerell G. (1998b). Visual field defects <strong>in</strong> optic neuritis and anterior ischemic optic<br />
neuropathy: dist<strong>in</strong>ctive features. Graefes Arch Cl<strong>in</strong> Experimental Ophthalmol 236:188–192.<br />
Ghezzi A, Mart<strong>in</strong>elli V, Rodegher M, et al. (2000). The prognosis of idiopathic optic neutritis. <strong>Neuro</strong>l Sci 21:<br />
S865–S869.<br />
Ghezzi A, Mart<strong>in</strong>elli V, Torri V. (1999). Long-term follow-up of isolated optic neuritis: the risk of develop<strong>in</strong>g<br />
multiple sclerosis, its outcome, and the prognostic role of paracl<strong>in</strong>ical tests. J <strong>Neuro</strong>l 246:770–775.<br />
Giorgi D, Balacco Gabrieli C. (1999a). Optic neuropathy <strong>in</strong> systemic lupus erythematosus and antiphospholipid<br />
syndrome (APS): cl<strong>in</strong>ical features, pathogenesis, review of the literature and proposed ophthalmological<br />
criteria for APS diagnosis. Cl<strong>in</strong> Rheumatol 18:124–131.<br />
Giorgi D, Balacco Gabrieli C, Bonomo L. (1999b). The association of optic neuropathy with transverse myelitis <strong>in</strong><br />
systemic lupus erythematosus. Rheumatology 38:191–192.<br />
Golnik KC, Newman SA, Wispelway B. (1991). Cryptococcal optic neuropathy <strong>in</strong> acquired deficiency syndrome.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:96–103<br />
Gould ES, Bird AC, Leaver PK, McDonald WI. (1977). Treatment of optic neuritis by retrobulbar <strong>in</strong>jection of<br />
triamc<strong>in</strong>olone. Br Med J 1:1495–1497.<br />
Greven CM, S<strong>in</strong>gh T, Stanton CA, Mart<strong>in</strong> TJ. (2001). Optic chiasm, optic nerve, and ret<strong>in</strong>al <strong>in</strong>volvement secondary<br />
to Varicella-zoster virus. Arch Ophthalmol 119:608–610.<br />
Grossniklaus HE, Specht CS, Allaire G, et al. (1990). Toxoplasma gondii ret<strong>in</strong>ochoroiditis and optic neuritis <strong>in</strong><br />
acquired immune deficiency syndrome. Ophthalmology 97:1342–1346.
56 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Gunduz K, Ozdermir O. (1994). Bilateral retrobulbar neuritis follow<strong>in</strong>g unilateral herpes tester ophthalmicus.<br />
Ophthalmologica 208:61–64.<br />
Guy JR, Mancuso AA, Quisl<strong>in</strong>g R. (1994). The role of magnetic resonance imag<strong>in</strong>g <strong>in</strong> optic neuritis. Ophthalmol<br />
Cl<strong>in</strong> North Am 7:449–458.<br />
Guy J, Mancuso A, Quisl<strong>in</strong>g R, Beck R, Moster M. (1990). Gadol<strong>in</strong>ium-DTPA enhanced magnetic resonance<br />
imag<strong>in</strong>g <strong>in</strong> optic neuropathies. Ophthalmology 97:592–599.<br />
Hark<strong>in</strong>s T, Ma<strong>in</strong>o JH. (1992). Cytomegalovirus ret<strong>in</strong>itis complicated by optic neuropathy: a longitud<strong>in</strong>al study.<br />
J Am Optom Assoc 63:21–27.<br />
Hatta M, Hayasaka S, Kato T, Kadoi C. (2000). Retrobulbar optic neuritis and rhegmatogenous ret<strong>in</strong>al detachment<br />
<strong>in</strong> a fourteen-year-old girl with ret<strong>in</strong>itis pigmentosa s<strong>in</strong>e pigmento. Ophthalmologica 214:153–155.<br />
Haupert CL, Newman NJ. (1997). Prolonged Uhthoff phenomenon <strong>in</strong> sarcoidosis. Am J Ophthalmol 124:564–566<br />
Hauser SL, Oksenberg JR, L<strong>in</strong>coln R, et al. (2000). Interaction between HLA-DR2 and abnormal bra<strong>in</strong> MRI <strong>in</strong> optic<br />
neuritis and early MS. <strong>Neuro</strong>logy 54:1859–1861.<br />
Ha<strong>in</strong>fellner JA, Schmidbauer M, Schmutzhard E, et al. (1992). Devic’s neuromyelitis optica and Schilder’s<br />
myel<strong>in</strong>oclastic diffuse sclerosis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 55:1194–1196.<br />
Hershewe GL, Corbett JJ, Thompson HS. (1990). The NANOS Devic’s Study Group. Presented at the North<br />
American <strong>Neuro</strong>-Ophthalmology Society Meet<strong>in</strong>g, Steamboat Spr<strong>in</strong>gs, Colorado, February 4–8.<br />
Ho M. (1995). Cytomegalovirus. In: Mandell GL, Bennett JE, Dol<strong>in</strong> R, eds. Pr<strong>in</strong>ciples and Practice of Infectious<br />
Disease. 4th ed. New York, Churchill Liv<strong>in</strong>gstone, pp. 1351–1364.<br />
Honan WP, Heron JR, Foster DH, et al. (1990). Visual loss <strong>in</strong> multiple sclerosis and its relation to previous optic<br />
neuritis, disease duration, and cl<strong>in</strong>ical classification. Bra<strong>in</strong> 113:975–987.<br />
Hull TP, Bates JH. (1997). Optic neuritis after <strong>in</strong>fluenza vacc<strong>in</strong>ation. Am J Ophthalmol 124:703–704.<br />
Hutnik CML, Nicolle DA, Canny CLB. (1996). Papillitis: a rare <strong>in</strong>itial presentation of Crohn’s disease.<br />
Can J Ophthalmol 31:373–376.<br />
Igarishi Y, Oyachi H, Nakamura Y, et al. (1994). <strong>Neuro</strong>myelitis optica. Ophthalmology 208:226–229.<br />
Jacobs LD, Beck RW, Simon JH, et al. (2000). Intramuscular <strong>in</strong>terferon beta-1a therapy <strong>in</strong>itiated dur<strong>in</strong>g a first<br />
demyel<strong>in</strong>at<strong>in</strong>g event <strong>in</strong> multiple sclerosis. N Eng J Med 343:898–904.<br />
Jacobs LD, Kaba SE, Miller CM, et al. (1997). Correlation of cl<strong>in</strong>ical, magnetic resonance imag<strong>in</strong>g, and<br />
cerebrosp<strong>in</strong>al fluid f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> optic neuritis. <strong>An</strong>n <strong>Neuro</strong>l 41:392–398<br />
Jacobs L, Munschauer FE. Kaba SE. (1991). <strong>Cl<strong>in</strong>ical</strong> and magnetic resonance imag<strong>in</strong>g <strong>in</strong> optic neuritis. <strong>Neuro</strong>logy<br />
41:15–19.<br />
Jacobson DM, Marx JJ, Dlesk A. (1991). Frequency and cl<strong>in</strong>ical significance of Lyme seropositivity <strong>in</strong> patients with<br />
isolated optic neuritis. <strong>Neuro</strong>logy 41:706–711.<br />
Ja<strong>in</strong> S, Hiran S, Sarma PS. (1994). Devic’s disease. J Assoc Phys India 42:166.<br />
Jeffrey AR, Buncic JR. (1996). Pediatric Devic’s neuromyelitis optica. J Pediatr Ophthalmol Strabismus 33:223–229.<br />
J<strong>in</strong> Y-P, de Pedro-Cuesta J, Soderstrom M, L<strong>in</strong>k H. (1999). Incidence of optic neuritis <strong>in</strong> Stockholm, Sweden,<br />
1990–1995. II. Time and space patterns. Arch <strong>Neuro</strong>l 56:975–980.<br />
Kakisu Y, Adachi-Usami E, Fujimoto N. (1991). Pattern visually evoked cortical potential and magnetic resonance<br />
Imag<strong>in</strong>g <strong>in</strong> optic neuritis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:205–212.<br />
Kapoor R, Miller DH, Jones SJ, et al. (1998). Effects of <strong>in</strong>travenous methylprednisolone on outcome <strong>in</strong> MRI-based<br />
prognostic subgroups <strong>in</strong> acute optic neuritis. <strong>Neuro</strong>logy 50:230–237.<br />
Karma A, Seppala I, Mikkila H, et al. (1995). Diagnosis and cl<strong>in</strong>ical characteristics of ocular Lyme borreliosis.<br />
Am J Ophthalmol 119:127–135.<br />
Kaufman DI. (1998). Peripheral demyel<strong>in</strong>at<strong>in</strong>g and axonal disorders. In: Miller NR, Newman NJ, eds. Walsh and<br />
Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 5th ed. Baltimore, Williams and Wilk<strong>in</strong>s, pp. 5677–5719.<br />
Kaufman DI, Trobe JD, Eggenberger ER, Whitaker JN. (2000). Practice parameter: the role of corticosteroids <strong>in</strong> the<br />
management of acute monosymptomatic optic neuritis. Report of the Quality Standards Subcommittee of the<br />
American Academy of <strong>Neuro</strong>logy. <strong>Neuro</strong>logy 54:2039–2044.<br />
Keltner JL, Johnson CA, Beck RW, Clearly PA, Spurr JO, and the Optic Neuritis Study Group. (1993a). Quality<br />
control functions of the visual field read<strong>in</strong>g center (VFRC) for the optic neuritis treatment trial (ONTT).<br />
Controlled Cl<strong>in</strong> Trials 14:143–159.<br />
Keltner JL, Johnson CA, Spurr JO, Beck RW. (1993b). Basel<strong>in</strong>e visual field profile of optic neuritis: the experience of<br />
the Optic Neuritis Treatment Trial. Arch Ophthalmol 111:231–234.<br />
Keltner JL, Johnson CA, Spurr JO, Beck RW, for the Optic Neuritis Study Group. (1999). Comparison of central<br />
and peripheral visual field properties <strong>in</strong> the Optic Neuritis Treatment Trial. Am J Ophthalmol 128:543–553.<br />
Kerrison JB, Lounsbury D, Lane G, et al. (2001). Optic neuritis follow<strong>in</strong>g anthrax vacc<strong>in</strong>ation. Presented at the 27th
Optic Neuritis 57<br />
annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Rancho Mirage, California, February<br />
18–22.<br />
Kerrison JB, Lounsbury D, Thirkill CE, et al. (2002). Optic neuritis after anthrax vacc<strong>in</strong>ation. Ophthalmology<br />
109:99–104.<br />
Khan MA, Mahar PS, Raghuraman VU. (1990). <strong>Neuro</strong>myelitis optica (Devic’s disease). Br J Cl<strong>in</strong> Pract 44:667–668.<br />
Komiyama A, Hasegawa O, Nakamura S, et al. (1995). Optic neuritis <strong>in</strong> cerebral toxocariasis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 59:197–198.<br />
Kosmorsky GS, Prayson R. (1996). Primary optic pathway sarcoidosis <strong>in</strong> a 38-year-old white man.<br />
J <strong>Neuro</strong>-ophthalmol 16:188–190.<br />
Kouyoumdjian GA, Lark<strong>in</strong> TP, Blackburn PJ, Mandava N. (2001). Optic disk edema as a presentation of<br />
propionibacterium acnes endophthalmitis. Am J Ophthalmol 132:259–261.<br />
Kupersmith MJ, Alban T, Zeiffer B, Lefton D. (2002a). Contrast-enhanced MRI <strong>in</strong> acute optic neuritis: relationship<br />
to visual performance. Bra<strong>in</strong> 125:812–822.<br />
Kupersmith MJ, Fazzone HE, Lefton D. (2002b). Localization of the pa<strong>in</strong> with optic neuritis. Presented at the 28th<br />
annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Copper Mounta<strong>in</strong>, Colorado,<br />
February 9–14.<br />
Lee AG, Brazis PW. (1998a). The 5-year risk of MS after optic neuritis. <strong>Neuro</strong>logy 51:1236–1238.<br />
Lee AG, Galetta SL, Lepore FE, Appel SH. (1999). Optic atrophy and chronic acquired polyneuropathy.<br />
J <strong>Neuro</strong>-ophthalmol 19:67–69.<br />
Lee CC, Venketasubramanian N, Lam MS. (1997). Optic neuritis: a rare complication of primary varicella<br />
<strong>in</strong>fection. Cl<strong>in</strong> Infect Dis 24:515–516.<br />
Lee MS, Cooney EL, Stoessel KM, Gariano RF. (1998b). Varicella zoster virus retrobulbar optic neuritis preced<strong>in</strong>g<br />
ret<strong>in</strong>itis <strong>in</strong> patients with acquired deficiency syndrome. Ophthalmology 105:467–471.<br />
Lehky TJ, Flerlage N, Katz D, et al. (1996). Human T-cell lymphotropic virus type II-associated myelopathy:<br />
cl<strong>in</strong>ical and immunologic profiles. <strong>An</strong>n <strong>Neuro</strong>l 40:714–723.<br />
Leiba H, Glaser JS, Schatz NJ, Siatkowski RM. (2000). Postpartum optic neuritis: etiologic and pathophysiologic<br />
considerations. J <strong>Neuro</strong>-ophthalmol 20:85–88.<br />
Lessell S. (1992). Corticosteroid treatment of acute optic neuritis. N Engl J Med 326:634–635.<br />
Lesser RL, Kornmehl EW, Pachner WR, et al. (1990). <strong>Neuro</strong>-ophthalmologic manifestations of Lyme disease.<br />
Ophthalmology 97:699–706.<br />
L<strong>in</strong>ssen WH, Kruisdijk JJ, Barkhof F, Smit LM. (1997). Severe irreversible optic neuritis follow<strong>in</strong>g Mantoux<br />
tubercul<strong>in</strong> sk<strong>in</strong> test <strong>in</strong> child with multiple sclerosis—a case report. <strong>Neuro</strong>pediatrics 28:338–340.<br />
Litoff D, Catalano RA. (1990). Herpes zoster optic neuritis <strong>in</strong> human immunodeficiency syndrome virus <strong>in</strong>fection.<br />
Arch Ophthalmol 108:782–783.<br />
Lossos A, Eliashiv S, Ben-Chetrit E, et al. (1993). Optic neuritis associated with familial Mediterranean fever.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:141–143.<br />
Lucch<strong>in</strong>etti CF, Kiers L, O’Duffy A, et al. (1997). Risk factors for develop<strong>in</strong>g multiple sclerosis after childhood<br />
optic neuritis. <strong>Neuro</strong>logy 49:1413–1418.<br />
Malessa R, Agel<strong>in</strong>k MW, Diener H. (1995). Dysfunction of visual pathways <strong>in</strong> HIV-1 <strong>in</strong>fection. J <strong>Neuro</strong>l Sci<br />
130:82–87.<br />
Maltzman JS, Lee AG, and Miller NR. (2000). Optic neuropathy occurr<strong>in</strong>g after bee and wasp st<strong>in</strong>g. Ophthalmology<br />
107:193–195.<br />
Mandler RN, Davis LE, Jeffrey DR, Kornfeld M. (1993). Devic’s neuromyelitis optica: a cl<strong>in</strong>icopathological study<br />
of 8 patients. <strong>An</strong>n <strong>Neuro</strong>l 34:162–168.<br />
Mandler RN, Ahmed W, Dencoff JE. (1998). Devic’s neuromyelitis optica: a prospective study of seven patients<br />
treated with prednisone and azathiopr<strong>in</strong>e. <strong>Neuro</strong>logy 51:1219–1220.<br />
Mansour AM. (1997). Cytomegalovirus optic neuritis. Curr Op<strong>in</strong> Ophthalmol 8:55–58<br />
Mansour AM. (1998). Optic disk tubercle. J <strong>Neuro</strong>-ophthalmol 18:201–203.<br />
Margolis TP, Milner MS, Shama A, et al. (1998). Herpes zoster ophthalmicus <strong>in</strong> patients with human immunodeficiency<br />
virus <strong>in</strong>fection. Am J Ophthalmol 125:285–291.<br />
McKibb<strong>in</strong> M, Cleland PG, Morgan SJ. (1995). Bilateral optic neuritis after hepatitis A. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
58:508.<br />
McLean DR, Russell N, Khan MY. (1992). <strong>Neuro</strong>brucellosis: cl<strong>in</strong>ical and therapeutic features. Cl<strong>in</strong> Infect Dis<br />
15:582–590.<br />
McLeish WM, Pulido JS, Holland S, et al. (1990). The ocular manifestations of syphilis <strong>in</strong> the human<br />
immunodeficiency virus type 1-<strong>in</strong>fected host. Ophthalmology 97:196–203.
58 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Meenken C, van den Horn GJ, de Smer MD, van der Meer JTM. (1998). Optic neuritis herald<strong>in</strong>g varicella zoster<br />
virus ret<strong>in</strong>itis <strong>in</strong> a patient with acquired immunodeficiency syndrome. <strong>An</strong>n <strong>Neuro</strong>l 43:534–536<br />
Menon V, Tandon R, Khanna S, et al. (2000). Cysticercosis of the optic nerve. J <strong>Neuro</strong>-ophthalmol 20:59–60.<br />
Menon V, Tandon R, Sharma T, Gupta A. (1997). Optic neuritis follow<strong>in</strong>g snake bite. Indian J Ophthalmol<br />
45:236–237.<br />
Merandi SF, Kudryk BT, Murtagh FR, Arr<strong>in</strong>gton JA. (1991). Contrast-enhanced MR imag<strong>in</strong>g of optic nerve lesions<br />
<strong>in</strong> patients with acute optic neuritis. AJNR 12:923–926.<br />
Merle D, Smadja D, Bera O, et al. (1997). Uveitis and papillitis <strong>in</strong> association with HTLV-I associated myelopathy.<br />
<strong>An</strong>n Ophthalmol 29:258–261.<br />
Miller NR. (1995). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed., vol. 5, pt 1. Baltimore, Williams and<br />
Wilk<strong>in</strong>s, pp. 2895–2896.<br />
Miyashita K, Kigasawa K, Mashima Y, Fuj<strong>in</strong>o T. (1993). Superior altitud<strong>in</strong>al hemianopia and herpes zoster.<br />
<strong>An</strong>n Ophthalmol 25:20–23.<br />
Moorman CM, <strong>An</strong>slow P, Elston JS. (1999). Is sphenoid s<strong>in</strong>us opacity significant <strong>in</strong> patients with optic neuritis. Eye<br />
13:76–82.<br />
Mori T, Terai T, Hatano M, et al. (1997). Stellate ganglion block improved loss of visual acuity caused by<br />
retrobulbar optic neuritis after herpes zoster. <strong>An</strong>esth <strong>An</strong>alg 85:870–871.<br />
Morrissey SP, Miller DH, Kendall BE, et al. (1993). The significance of bra<strong>in</strong> magnetic resonance imag<strong>in</strong>g<br />
abnormalities at presentation with cl<strong>in</strong>ically isolated syndromes suggestive of multiple sclerosis. A 5-year<br />
follow-up study. Bra<strong>in</strong> 116:135–146.<br />
Moschos M. (1990). Acute bilateral optic neuritis. Doc Ophthalmol 73:225–230.<br />
Nadkarni N, Lisak RP. (1993). Guilla<strong>in</strong>-Barré syndrome (GBS) with bilateral optic neuritis and central white<br />
matter disease. <strong>Neuro</strong>logy 43:842–843.<br />
Nakazawa T, Abe T, Ohmura M. (1999). Varicella zoster-associated optic neuropathy with choroidal <strong>in</strong>volvement.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 21:39–45.<br />
Newman NJ, Lessell S. (1992). Bilateral optic neuropathies with remission <strong>in</strong> two HIV-positive men.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:1–5.<br />
Newman NJ, Lessell S, W<strong>in</strong>terkorn JS. (1991). Optic chiasmal neuritis. <strong>Neuro</strong>logy 41:1203–1210.<br />
Nichols JW, Goodw<strong>in</strong> JA. (1992). <strong>Neuro</strong>-ophthalmologic complications of AIDS. Sem<strong>in</strong> Ophthalmol 7:24–29.<br />
N<strong>in</strong>omiya M, Ohashi K, Sasaki N, et al. (1990). A case of optic neuritis accompany<strong>in</strong>g systemic lupus<br />
erythematosus several years after onset. Folia Ophthalmol Jpn 41:636.<br />
Noseworthy JH, O’Brien PC, Petterson TM, et al. (2001). A randomized trial of <strong>in</strong>travenous immunoglobul<strong>in</strong> <strong>in</strong><br />
<strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g optic neuritis. <strong>Neuro</strong>logy 56:1514–1522.<br />
Ohnuma I, Yamaguchi K, Takahashi S. (1996). Retrobulbar neuritis <strong>in</strong> a patient with mixed connective tissue<br />
disease. Folia Ophthalmol Jpn 47:828–831.<br />
Optic Neuritis Study Group. (1991). The cl<strong>in</strong>ical profile of optic neuritis. Experience of the Optic Neuritis<br />
Treatment Trial. Arch Ophthalmol 109:1673–1678.<br />
Optic Neuritis Study Group. (1997a). The 5-year risk of MS after optic neuritis. Experience of the Optic<br />
Neuritis Treatment Trial. <strong>Neuro</strong>logy 49:1404–1413.<br />
Optic Neuritis Study Group. (1997b). Visual function 5 years after optic neuritis. Experience of the Optic Neuritis<br />
Treatment Trial. Arch Ophthalmol 115:1545–1552<br />
O’Riordan JI, Gallagher HL, Thompson AJ, et al. (1996). <strong>Cl<strong>in</strong>ical</strong>, CSF, and MRI f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Devic’s neuromyelitis<br />
optica. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:382–387.<br />
Patel SS, Rutzen AR, Marx JL, et al. (1996). Cytomegalovirus papillitis <strong>in</strong> patients with acquired immune<br />
deficiency syndrome: visual prognosis of patients treated ganciclovir and foscarnet. Ophthalmology<br />
103:1476–1482.<br />
Paty DW, Li DKB, the UBC MS=MRI Study Group, et al. (1993). Interferon beta-1b is effective <strong>in</strong> relaps<strong>in</strong>gremitt<strong>in</strong>g<br />
multiple sclerosis, II: MRI analysis results of a multicenter, randomized, double-bl<strong>in</strong>d, placebocontrolled<br />
trial. <strong>Neuro</strong>logy 43:662–667.<br />
Paty DW, Oger JJF, Kastrukoff LF, et al. (1988). MRI <strong>in</strong> the diagnosis of MS: a prospective study with comparison<br />
of cl<strong>in</strong>ical evaluation, evoked potentials, oligoclonal band<strong>in</strong>g and CT. <strong>Neuro</strong>logy 38:180–185.<br />
Percy AK, Norbrega FT, Kurland LT. (1992). Optic neuritis and multiple sclerosis. Arch Ophthalmol 87:<br />
135–139.<br />
Perry JD, Girk<strong>in</strong> CA, Miller NR, Mann RB. (1999). Dissem<strong>in</strong>ated histoplasmosis caus<strong>in</strong>g reversible gaze palsy and<br />
optic neuropathy. J <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:140–143.<br />
Phillips PH, Newman NJ, Lynn MJ. (1998). Optic neuritis <strong>in</strong> African Americans. Arch <strong>Neuro</strong>l 55:186–192.<br />
Piccolo G, Franciotta DM, Camana C, et al. (1990). Devic’s neuromyelitis optica: long-term follow-up and serial<br />
CSF f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> two cases. J <strong>Neuro</strong>l 237:262–264.
Optic Neuritis 59<br />
Pierce EA, D’Amico DJ. (1993). Ocular toxoplasmosis: pathogenesis, diagnosis, and management. Sem<strong>in</strong><br />
Ophthalmol 8:40–52.<br />
Quicenco JI, Capparelli E, Sadun AA, et al. (1992). Visual dysfunction without ret<strong>in</strong>itis <strong>in</strong> patients with acquired<br />
immunodeficiency syndrome. Am J Ophthalmol 113:8–13.<br />
Ramelli GP, Deonna T, Roulet E, et al. (1992). Transverse myelitis and optic neuromyelitis <strong>in</strong> children: apropos of 3<br />
case reports. Schweiz Rundsch Med Prax 81:661–663.<br />
Rawson MD, Liversedge LA, Goldfarb G. (1966). Treatment of acute retrobuibar neuritis with corticotroph<strong>in</strong>.<br />
Lancet 2:1044–1046.<br />
Riedel P, Wall M, Grey A, et al. (1998). Autoimmune optic neuropathy. Arch <strong>Neuro</strong>l 116:1121–1124.<br />
R<strong>in</strong>alduzzi S, Brusa A, Jones SJ. (2001). Variation of visual evoked potential delay to stimulation of central, nasal,<br />
and temporal regions of the macula <strong>in</strong> optic neuritis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 70:28–35.<br />
Rizzo JF, Lessell S. (1988). Risk of develop<strong>in</strong>g multiple sclerosis after uncomplicated optic neuritis: a long term<br />
prospective study. <strong>Neuro</strong>logy 38:185–190.<br />
Roarty JD, Fisher EJ, Nussbaum JJ. (1993). Long-term morbidity of cytomegalovirus ret<strong>in</strong>itis <strong>in</strong> patients with<br />
acquired immune deficiency syndrome. Ophthalmology 100:1685–1688.<br />
Rodriguez M, Siva A, Cross SA, et al. (1995). Optic neuritis: a population based study <strong>in</strong> Olmsted County,<br />
M<strong>in</strong>nesota. <strong>Neuro</strong>logy 45:244–250.<br />
Ropper AH, Wijdicks EFM, Truax BT. (1991). Guilla<strong>in</strong>-Barré Syndrome. Philadelphia, FA Davis.<br />
Rose AS, Kuzma JW, Kurtzke JF, et al. (1970). Cooperative study <strong>in</strong> the evaluation of therapy <strong>in</strong> multiple sclerosis.<br />
ACTH vs. placebo—f<strong>in</strong>al report. <strong>Neuro</strong>logy 20:1–59.<br />
Rose GE. (1991). Papillitis, ret<strong>in</strong>al neovascularization and recurrent ret<strong>in</strong>al ve<strong>in</strong> occlusion <strong>in</strong> Toxoplasma<br />
ret<strong>in</strong>ochoroiditis: a case report with uncommon cl<strong>in</strong>ical signs. Aust NZ J Ophthalmol 19:155–157.<br />
Rosenbaum JT, Simpson J, Neuwelt CM. (1997). Successful treatment of optic neuropathy <strong>in</strong> association with<br />
systemic lupus erythematosus us<strong>in</strong>g <strong>in</strong>travenous cyclophosphamide. Br J Ophthalmol 81:130–132.<br />
Sadun AA, Pepose JS, Madigan MC, et al. (1995). AIDS-related optic neuropathy: a histological, virological and<br />
ultrastructural study. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 233:387–398.<br />
Sakuma R, Fujihara K, Sato N, et al. (1999). Optic-sp<strong>in</strong>al form of multiple sclerosis and anti-thyroid autoantibodies.<br />
J <strong>Neuro</strong>l 246:449–453.<br />
Salzman MB, Sood SK, Slav<strong>in</strong> ML, et al. (1992). Ocular manifestations of Mycoplasma pneumoniae <strong>in</strong>fection.<br />
Cl<strong>in</strong> Infect Dis 14:1137–1139.<br />
Sandberg-Wollheim M. (1975). Optic neuritis: studies on the cerebrosp<strong>in</strong>al fluid <strong>in</strong> relation to cl<strong>in</strong>ical course <strong>in</strong><br />
61 patients. Acta <strong>Neuro</strong>l Scand 52:167–178.<br />
Sandberg-Wollheim M, Bynke H, Cronqvist S, et al. (1990). A long-term prospective study of optic neuritis:<br />
evaluation of risk factors. <strong>An</strong>n <strong>Neuro</strong>l 27:386–393.<br />
Schneck ME, Haegerstrom-Portnoy G. (1997). Color vision defect type and spatial vision <strong>in</strong> the Optic Neuritis<br />
Treatment Trial. Invest Ophthalmol Vis Sci 38:2278–2289.<br />
Schwartzman WA, Patnaik M, <strong>An</strong>gula FJ, et al. (1995). Bartonella (Rochalimaea) antibodies, dementia, and cat<br />
ownership among men <strong>in</strong>fected with immunodeficiency virus. Cl<strong>in</strong> Infect Dis 21:954–959.<br />
Schwartzman WA, Patnaik M, Barka NE, et al. (1994). Rochalimara antibodies <strong>in</strong> HIV associated neurologic<br />
disease. <strong>Neuro</strong>logy 44:1312–1316.<br />
Sedwick LA. (1991). Optic neuritis. <strong>Neuro</strong>l Cl<strong>in</strong> 9:97–112.<br />
Sellebjerg F, Christiansen M, Jensen J, Frederiksen JL. (2000). Immunological effects of oral high-dose methylprednisolone<br />
<strong>in</strong> acute optic neuritis and multiple sclerosis. Eur J <strong>Neuro</strong>l 7:281–289.<br />
Sellebjerg FT, Frederkisen JT. Olsson T. (1994). <strong>An</strong>ti-myel<strong>in</strong> basic prote<strong>in</strong> and anti-proteolipid prote<strong>in</strong> antibodysecret<strong>in</strong>g<br />
cells <strong>in</strong> the cerebrosp<strong>in</strong>al fluid of patients with acute optic neuritis. Arch <strong>Neuro</strong>l 51:1032–1036.<br />
Sellebjerg F, Madsen HO, Frederiksen JL, et al. (1995). Acute optic neuritis: myel<strong>in</strong> basic prote<strong>in</strong> and proteolipid<br />
prote<strong>in</strong> antibodies, aff<strong>in</strong>ity and the HLA system. <strong>An</strong>n <strong>Neuro</strong>l 38:943–950.<br />
Sellebjerg F, Nielsen S, Frederiksen JL, Olesen J. (1999). A randomized, controlled trial of oral high-dose<br />
methylprednisolone <strong>in</strong> acute optic neuritis. <strong>Neuro</strong>logy 52:1479–1484.<br />
Shayegani A, Odel JG, Kazim M, et al. (1996). Varicella-zoster virus retrobulbar optic neuritis <strong>in</strong> a patient with<br />
human immunodeficiency virus. Am J Ophthalmol 112:586–588.<br />
Sheth RD, Goulden KJ, Pryse-Phillips WE. (1993). The focal encephalopathies associated with Mycoplasma<br />
pneumoniae. Can J <strong>Neuro</strong>l Sci 20:319–323.<br />
Silber MH, Willcox PA, Bowen RM, et al. (1990). <strong>Neuro</strong>myelitis optica (Devic’s disease) and pulmonary<br />
tuberculosis. <strong>Neuro</strong>logy 40:934–938.<br />
Silberberg DH. (1993). Corticosteroids and optic neuritis. N Engl J Med 329:1808–1810.
60 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Simon JH, McDonald WI. (2000). Assessment of optic nerve damage <strong>in</strong> multiple sclerosis us<strong>in</strong>g magnetic<br />
resonance imag<strong>in</strong>g. J <strong>Neuro</strong>l Sci 172(suppl 1):S23–S26.<br />
Slamovits TL, Mackl<strong>in</strong> R. (1991a). What to tell the patient with optic neuritis about multiple sclerosis. Surv<br />
Ophthalmol 36:47–50.<br />
Slamovits TL, Rosen CE, Cheng KP, Striph GG. (1991b). Visual recovery <strong>in</strong> patients with optic neuritis and visual<br />
loss to no light perception. Am J Ophthalmol 111:209–214.<br />
Söderström M, L<strong>in</strong>k H, Sun J-B, et al. (1994). Autoimmune T cell repertoire <strong>in</strong> optic neuritis and multiple sclerosis:<br />
T cells recogniz<strong>in</strong>g multiple myel<strong>in</strong> prote<strong>in</strong>s are accumulated <strong>in</strong> cerebrosp<strong>in</strong>al fluid. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 57:544–551.<br />
Söderström M, L<strong>in</strong>k H, Xu Z, Fredriksson S. (1993). Optic neuritis and multiple sclerosis: anti-MBP and anti-MBP<br />
peptide antibody-secret<strong>in</strong>g cells are accumulated <strong>in</strong> CSF. <strong>Neuro</strong>logy 43:1215–1222.<br />
Söderström M, Ya-P<strong>in</strong>g J, Hillert J, L<strong>in</strong>k H. (1998). Optic neuritis. Prognosis for multiple sclerosis from MRI, CSF,<br />
and HLA f<strong>in</strong>d<strong>in</strong>gs. <strong>Neuro</strong>logy 50:708–714.<br />
Song A, Scott IU, Davis JL, Lam BL. (2002). Atypical anterior optic neuropathy caused by toxoplasmosis. Am J<br />
Ophthalmol 133:162–164.<br />
Song HS, Wray SH. (1991). Bee st<strong>in</strong>g optic neuritis. A case report with visual evoked potentials. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:45–49.<br />
Sorensen TL, Frederiksen JL, Bronnum-Hansen H, Petersen HC. (1999). Optic neuritis as onset manifestation of<br />
multiple sclerosis. A nationwide, long-term survey. <strong>Neuro</strong>logy 53:473–478.<br />
Staedt D, Kappos L, RohrbachE, Heun R, Ratzka M. (1990). Occurrence of MRI abnormalities <strong>in</strong> patients with<br />
isolated optic neuritis. Eur <strong>Neuro</strong>l 30:305–309.<br />
Steel DH, Waldock A. (1998). Measurement of the ret<strong>in</strong>al nerve fibre layer with scann<strong>in</strong>g laser polarimetry <strong>in</strong><br />
patients with previous demyel<strong>in</strong>at<strong>in</strong>g optic neuritis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 64:505–509.<br />
Stewart O, Chang B, Bradbury J. (1999). Simultaneous adm<strong>in</strong>istration of hepatitis B and polio vacc<strong>in</strong>es associated<br />
with bilateral optic neuritis. Br J Ophthalmol 83:1200–1201.<br />
Straussberg R, Amir J, Cohen HA, et al. (1993). Epste<strong>in</strong>-Barr virus <strong>in</strong>fection associated with encephalitis and optic<br />
neuritis. J Pediatr Ophthalmol Strabismus 30:262–263.<br />
Sugita K, <strong>An</strong>do M, M<strong>in</strong>amitani K, et al. (1991). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> a case of mumps post<strong>in</strong>fectious<br />
encephalitis with asymptomatic optic neuritis. Eur J Pediatr 150:773–775.<br />
Sweeney BJ, Manji H, Gilson RJC, et al. (1993). Optic neuritis and HIV-1 <strong>in</strong>fection. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
56:705–707.<br />
Thompson AJ, Kermode A, MacManus DG, et al. (1990). Patterns of disease activity <strong>in</strong> multiple sclerosis: cl<strong>in</strong>ical<br />
and magnetic resonance imag<strong>in</strong>g study. Br Med J 300:631–634.<br />
Thompson AJ, Miller D, Youl B, et al. (1992). Serial gadol<strong>in</strong>ium-enhanced MRI <strong>in</strong> relaps<strong>in</strong>g=remitt<strong>in</strong>g multiple<br />
sclerosis of vary<strong>in</strong>g disease duration. <strong>Neuro</strong>logy 42:60–63.<br />
Tomsak RL, Lystad LD, Katirji MB, et al. (1992). Rapid response of syphilitic optic neuritis to posterior sub-<br />
Tenon’s steroid <strong>in</strong>jection. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:6–7.<br />
Topaloglu H, Berker M, Kansu T, et al. (1992). Optic neuritis and myelitis after booster tetanus toxoid vacc<strong>in</strong>ation.<br />
Lancet 339:178–179.<br />
Tornerup NR, Fomsgaard A, Nielsen NV. (2000). HSV-1-<strong>in</strong>duced acute ret<strong>in</strong>al necrosis syndrome present<strong>in</strong>g with<br />
severe <strong>in</strong>flammatory orbitopathy, proptosis, and optic nerve <strong>in</strong>volvement. Ophthalmology 107:397–401.<br />
Totan Y, Cekic O. (1999). Bilateral retrobulbar neuritis follow<strong>in</strong>g measles <strong>in</strong> an adult. Eye 13:383–384.<br />
Trobe JD. (1994). High-dose corticosteroid regimen retards development of multiple sclerosis <strong>in</strong> optic neuritis<br />
treatment trial. Arch Ophthalmol 112:35–36.<br />
Trobe JD, Siev<strong>in</strong>g PC, Guire KE, Fendrick AM. (1999). The impact of the Optic Neuritis Treatment Trial on the<br />
practices of ophthalmologists and neurologists. Ophthalmology 106:2047–2053.<br />
van de Geijn EJ, Tukkie R, van Phillips LA, Punt H. (1994). Bilateral optic neuritis with branch ret<strong>in</strong>al artery<br />
occlusion associated with vacc<strong>in</strong>ation. Doc Ophthalmol 86:403–408.<br />
van Engelen BGM, Hommes OR, P<strong>in</strong>ckers A, et al. (1992). Improved vision after <strong>in</strong>travenous immunoglobul<strong>in</strong> <strong>in</strong><br />
stable demyel<strong>in</strong>at<strong>in</strong>g optic neuritis. <strong>An</strong>n <strong>Neuro</strong>l 32:834.<br />
Vaphiades MS, Lee AG. (1999). A bad eye and a sore lip. Surv Ophthalmol 44:148–152.<br />
Vernant J-C, Cabre P, Smadje D, et al. (1997). Recurrent optic neuromyelitis with endocr<strong>in</strong>opathies: a new<br />
syndrome. <strong>Neuro</strong>logy 48:58–64.<br />
Wakakura M, Mashimo K, Oono S, et al. (1999a). Multicenter cl<strong>in</strong>ical trial for evaluat<strong>in</strong>g methylprednisolone<br />
pulse treatment of idiopathic optic neuritis <strong>in</strong> Japan. Jpn J Ophthalmol 43:133–138.<br />
Wakakura M, M<strong>in</strong>ei-Higa R, Oono S, et al. (1999b). Basel<strong>in</strong>e features of idiopathic optic neuritis as determ<strong>in</strong>ed by<br />
a multicenter treatment trial <strong>in</strong> Japan. Jpn J Ophthalmol 43:127–132.
Optic Neuritis 61<br />
Wall M, Johnson CA, Kutzko KE, et al. (1998). Long- and short-term variability of automated perimetry results <strong>in</strong><br />
patients with optic neuritis and healthy subjects. Arch Ophthalmol 116:53–61.<br />
Warren KG, Catz I, Bauer C. (1988). Cerebrosp<strong>in</strong>al fluid antibodies to myel<strong>in</strong> basic prote<strong>in</strong> <strong>in</strong> acute idiopathic<br />
optic neuritis. <strong>An</strong>n <strong>Neuro</strong>l 23:297–299.<br />
Warren KG, Catz I, Johnson E, Mielke B. (1994). <strong>An</strong>ti-myel<strong>in</strong> basic prote<strong>in</strong> and anti-proteolipid prote<strong>in</strong> specific<br />
forms of multiple sclerosis. <strong>An</strong>n <strong>Neuro</strong>l 35:280–289.<br />
W<strong>in</strong>gerchuk DM, Hogancamp WF, O’Brien PC, We<strong>in</strong>shenker BG. (1999). The cl<strong>in</strong>ical course of neuromyelitis<br />
optica (Devic’s disease). <strong>Neuro</strong>logy 53:1107–1114.<br />
W<strong>in</strong>terkorn JMS. (1990). Lyme disease: neurologic and ophthalmologic manifestations. Surv Ophthalmol<br />
35:191–204.<br />
Wolf MD, Folk JC, Goeken NE. (1990). Acute posterior multifocal pigment epitheliopathy and optic neuritis <strong>in</strong> a<br />
family. Am J Ophthalmol 110:89–90.<br />
Yamakawa K, Kuroda H, Fujihara K, et al. (2000). Familial neuromyelitis optica (Devic’s syndrome) with late<br />
onset <strong>in</strong> Japan. <strong>Neuro</strong>logy 55:318–320.<br />
Yau TH, Rivera-Velasquez PM, Mark AS, et al. (1996). Unilateral optic neuritis caused by Histoplasma capsulatum <strong>in</strong><br />
a patient with acquired immunodeficiency syndrome. Am J Ophthalmol 121:324–326.<br />
Yen MY, Liu JH. (1991). Bilateral optic neuritis follow<strong>in</strong>g bacille Calmette-Guér<strong>in</strong> (BCG) vacc<strong>in</strong>ation. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:246–249.<br />
Yoshida Y, Saiga T, Takahashi H, Hara A. (1998). Optic neuritis and human T-lymphotropic virus type<br />
1-associated myelopathy: a case report. Ophthalmologica 212:73–76.
This page <strong>in</strong>tentionally left blank
3 r<br />
Optic Disc Edema with a Macular<br />
Star and <strong>Neuro</strong>ret<strong>in</strong>itis<br />
Optic disc edema with a macular star (ODEMS) is a descriptive term encompass<strong>in</strong>g a<br />
heterogeneous group of disorders. In 1916, Leber described patients with idiopathic<br />
unilateral visual loss, optic disc edema, and macular exudate (Leber, 1916). He<br />
<strong>in</strong>correctly theorized that the pathologic process was primarily ret<strong>in</strong>al and called the<br />
condition ‘‘stellate ret<strong>in</strong>opathy.’’ The condition subsequently has been called Leber’s<br />
stellate maculopathy, Leber’s idiopathic stellate neuroret<strong>in</strong>itis, or simply neuroret<strong>in</strong>itis.<br />
In 1977, Gass suggested that this syndrome was caused by a prelam<strong>in</strong>ar disc vasculitis<br />
that results <strong>in</strong> leakage of disc capillaries and concluded that this entity was not a ret<strong>in</strong>al<br />
vasculopathy but a primary optic neuropathy (Gass, 1977).<br />
This syndrome is characterized by swell<strong>in</strong>g of the optic disc, peripapillary and<br />
macular hard exudates that often occur <strong>in</strong> a star pattern, and (often) vitreous cells.<br />
Because the macular exudate likely results from primary optic nerve disease and not a<br />
true ret<strong>in</strong>itis, we prefer the term idiopathic optic disc edema with a macular star (ODEMS)<br />
for idiopathic cases, and use the term neuroret<strong>in</strong>itis when optic disc swell<strong>in</strong>g and a<br />
macular star are associated with ret<strong>in</strong>itis, especially if an <strong>in</strong>fectious cause is documented<br />
(Brazis, 1996).<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of ODEMS<br />
and <strong>Neuro</strong>ret<strong>in</strong>itis?<br />
The cl<strong>in</strong>ical features of ODEMS have been described by a number of authors (Brazis,<br />
1996; Hamard, 1994; K<strong>in</strong>g, 1991) and are summarized <strong>in</strong> Table 3–1. Patients are usually<br />
children or young adults, with the average age of onset be<strong>in</strong>g 20 to 40 years. Men and<br />
women are affected equally. Most cases are unilateral, but bilateral <strong>in</strong>volvement has<br />
been noted to occur <strong>in</strong> up to a third of the cases. Most patients present with acute<br />
unilateral loss of vision. The condition is often pa<strong>in</strong>less, but retrobulbar pa<strong>in</strong>, pa<strong>in</strong> on<br />
eye movement, or associated headache may occur. A nonspecific viral illness precedes<br />
or accompanies the visual loss <strong>in</strong> approximately half of the cases.<br />
63
64 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 3–1. <strong>Cl<strong>in</strong>ical</strong> Characteristics of Optic Disc Edema with Macular Star (ODEMS)<br />
Age at onset: childhood to young adult (6 to 50 years of age)<br />
Gender: men and women affected equally<br />
Bilateral <strong>in</strong>volvement: 5–33%<br />
Pa<strong>in</strong>: occasional<br />
<strong>An</strong>tecedent viral illness: approximately 50%<br />
Initial visual acuity: variable (20=20–light perception)<br />
Dyschromatopsia: often prom<strong>in</strong>ent<br />
Visual field test<strong>in</strong>g: central, cecocentral, arcuate, or altitud<strong>in</strong>al defects; possible generalized<br />
constriction<br />
Relative afferent pupil defect present; but may be absent if bilateral <strong>in</strong>volvement<br />
Optic disc swell<strong>in</strong>g present with subsequent optic atrophy<br />
Macular star present but may take 1 or 2 weeks to develop<br />
Vitreous cells common (90%)<br />
Visual acuity with ODEMS may range from 20=20 to light perception, but most cases<br />
are <strong>in</strong> the 20=40 to 20=200 range. Dyschromatopsia is often present. Perimetry most<br />
often reveals a central or cecocentral scotoma, but other ‘‘optic nerve–type’’ field<br />
abnormalities may occur, <strong>in</strong>clud<strong>in</strong>g arcuate and altitud<strong>in</strong>al defects or generalized<br />
constriction. Most patients have a relative afferent papillary defect unless <strong>in</strong>volvement<br />
is bilateral and relatively symmetric.<br />
Optic disc edema is the earliest sign of ODEMS and may be severe. The disc edema<br />
tends to resolve over 2 weeks to 2 months, but <strong>in</strong> some patients optic atrophy ensues.<br />
Optic disc edema is associated with leakage of disc capillaries with the fluid spread<strong>in</strong>g<br />
from the disc through the outer plexiform layer of the ret<strong>in</strong>a. The serous component of<br />
the fluid accumulation <strong>in</strong> Henle’s layer is reabsorbed, and the lipid precipitate forms a<br />
macular star. The macular star may be present at the onset of visual loss or may be<br />
noted only after 1 to 2 weeks follow<strong>in</strong>g development of the disc edema. The macular<br />
star may even be observed only after the disc swell<strong>in</strong>g is start<strong>in</strong>g to resolve. Patients<br />
with acute disc swell<strong>in</strong>g with a normal macula should thus be reexam<strong>in</strong>ed with<strong>in</strong> 2<br />
weeks to search for the presence of a macular star, especially because it is of prognostic<br />
importance for the patient’s subsequent risk of develop<strong>in</strong>g multiple sclerosis (see<br />
below). Fluoresce<strong>in</strong> angiography typically shows leakage from the optic disc <strong>in</strong> the<br />
middle to late phases, with abnormal permeability of the deep capillaries <strong>in</strong> the optic<br />
nerve head but no perifoveal leakage (Ray, 2001).<br />
ODEMS is often associated with cells <strong>in</strong> the vitreous. Other occasional f<strong>in</strong>d<strong>in</strong>gs<br />
<strong>in</strong>clude cells <strong>in</strong> the anterior chamber, chorioret<strong>in</strong>itis, <strong>in</strong>flammatory sheath<strong>in</strong>g of the<br />
peripapillary ve<strong>in</strong>s, scleritis and uveitis, and (rarely) central or branch ret<strong>in</strong>al artery<br />
occlusions (May, 1995). The association of ODEMS with these latter f<strong>in</strong>d<strong>in</strong>gs suggests a<br />
more diffuse vasculitis or an <strong>in</strong>fectious cause.<br />
What Is the Etiology and Differential of<br />
ODEMS and <strong>Neuro</strong>ret<strong>in</strong>itis?<br />
Most cases of ODEMS are idiopathic and thought to be the result of nonspecific viral<br />
<strong>in</strong>fection or some immune-mediated process. In general, ODEMS is usually a benign,
Optic Disc Edema with a Macular Star and <strong>Neuro</strong>ret<strong>in</strong>itis 65<br />
self-limited <strong>in</strong>flammatory process. A number of <strong>in</strong>fectious agents and <strong>in</strong>flammatory<br />
diseases, however, have been reported to cause ODEMS and neuroret<strong>in</strong>itis. Infectious<br />
etiologies are listed <strong>in</strong> Table 3–2. Some of these <strong>in</strong>fectious agents have been implicated <strong>in</strong><br />
s<strong>in</strong>gle case reports, but it appears that syphilis, cat-scratch disease, Lyme disease, and<br />
perhaps toxoplasmosis are the most common causes of ODEMS and neuroret<strong>in</strong>itis <strong>in</strong><br />
cases where an etiologic agent can be identified. Infectious agents should be aggressively<br />
sought <strong>in</strong> cases of ODEMS and neuroret<strong>in</strong>itis because appropriate antibiotic<br />
treatment might be <strong>in</strong>dicated. Ray and Gragoudas recommended special emphasis on<br />
recent patient travel history (Lyme endemic areas), consumption of unpasteurized or<br />
uncooked foods (toxoplasmosis), sexually transmitted disease exposure (syphilis), and<br />
animal contacts (cat scratch) (Ray, 2001).<br />
ODEMS or neuroret<strong>in</strong>itis may occur as part of syphilitic men<strong>in</strong>gitis (usually bilateral),<br />
or may occur as an isolated entity <strong>in</strong> patients with secondary syphilis, <strong>in</strong> which case it<br />
may be associated with unilateral or bilateral uveitis (Halper<strong>in</strong>, 1992; McCleish, 1990;<br />
N<strong>in</strong>omiya, 1990). ODEMS or neuroret<strong>in</strong>itis is common manifestation of cat-scratch<br />
disease (Bar, 1990; Bhatti, 2001; Carithers, 1991; Chrousos, 1990; Cunn<strong>in</strong>gham, 2000;<br />
Earhart, 2000; Fish, 1992; Ghauri, 1998; Golnik, 1994; Gray, 1999; K<strong>in</strong>g, 1991; Labalette,<br />
Table 3–2. Infectious Etiologies of Optic Disc Edema with Macular Star or <strong>Neuro</strong>ret<strong>in</strong>itis<br />
Viral<br />
Hepatitis B<br />
Herpes simplex<br />
Herpes zoster (Dhar, 1997)<br />
Epste<strong>in</strong>-Barr virus<br />
Influenza A<br />
Mumps (Foster, 1990)<br />
Coxsackie B<br />
Bacteria<br />
Cat-scratch disease (Bartonella henselae) (Bar, 1990; Bhatti, 2001; Carithers, 1991; Chrousos, 1990;<br />
Earhart, 2000; Fish, 1992; Ghauri, 1998; Golnik, 1994; Gray, 1999; K<strong>in</strong>g, 1991; Labalette, 2001;<br />
May, 1995; McCrary, 1994, 1997; Ormerod, 1998; Reed, 1998; Rosen, 1999; Schwartzman, 1994,<br />
1995; Solley, 1999; Suhler, 2000; Thompson, 1999; Ulrich, 1992; Wade, 1999; Zhao, 1991);<br />
Bartonella elizabethae (O’Halloran, 1998); Bartonella grahamii (Kerkoff, 1999)<br />
Tuberculosis (Stechschulte, 1999)<br />
Salmonella<br />
Lyme disease (Bialasiewicz, 1992; Karma, 1995; Lesser, 1990; Schönherr, 1990, 1991; Miller, 1995a)<br />
Syphilis (Halper<strong>in</strong>, 1992; McCleish, 1990; N<strong>in</strong>omiya, 1990)<br />
Leptospirosis<br />
Fungi<br />
Histoplasmosis<br />
Parasites and Protozoa<br />
Toxoplasmosis (Fish, 1993; Moreno, 1992)<br />
Toxocara<br />
Other<br />
Sarcoidosis (Kosmorsky, 1995; Miller, 1995b)
66 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
2001; May, 1995; McCrary, 1994, 1997; Ormerod, 1998; Reed, 1998; Rosen, 1999;<br />
Schwartzman, 1994, 1995; Solley, 1999; Suhler, 2000; Ulrich, 1992; Wade, 1999; Zhao,<br />
1991). In fact, optic neuritis (papillitis) without a macular star has only rarely been<br />
reported with this disease (Brazis, 1986; Golnik, 1994). Cat-scratch disease may cause<br />
ODEMS or may cause a neuroret<strong>in</strong>itis with chorioret<strong>in</strong>itis at times associated with<br />
uveitis, cells <strong>in</strong> the anterior chamber, and even branch or central ret<strong>in</strong>al artery occlusions<br />
(Fish, 1992; Golnik, 1994; May, 1995; Ulrich, 1992; Zhao, 1991). This disease may also<br />
cause a multifocal ret<strong>in</strong>itis with optic disc edema (without macular star), branch ret<strong>in</strong>al<br />
artery occlusion, and vitreitis (Cohen, 1995). Optic disc edema associated with peripapillary<br />
serous ret<strong>in</strong>al detachment, even without macular star formation, may be an<br />
early sign of cat-scratch disease (Wade, 2000). Solley et al studied 24 patients (35 eyes)<br />
with choroidal, ret<strong>in</strong>al, or optic disc manifestations of cat-scratch disease and found that<br />
discrete white ret<strong>in</strong>al or choroidal lesions were the most common posterior segment<br />
f<strong>in</strong>d<strong>in</strong>g (46% of eyes, 63% of patients) followed by macular star (43% of eyes, 63% of<br />
patients) (Solley, 1999). Vascular-occlusive events were also seen (14% of eyes, 21% of<br />
patients) and the site of occlusion was found to be <strong>in</strong>timately associated with the<br />
aforementioned ret<strong>in</strong>al lesions. F<strong>in</strong>al visual acuity was 20=25 or better <strong>in</strong> 26 (74%) of 35<br />
eyes and was similar <strong>in</strong> both treated and untreated patients. Cat-scratch disease,<br />
therefore, should be considered <strong>in</strong> any patient who presents with ODEMS or neuroret<strong>in</strong>itis,<br />
especially if there is associated lymphadenopathy or ret<strong>in</strong>al artery occlusion<br />
(class III, level B). The treatment (Conrad, 2001) of cat-scratch disease is quite variable <strong>in</strong><br />
the literature and has <strong>in</strong>cluded various antibiotic regimens <strong>in</strong>clud<strong>in</strong>g penicill<strong>in</strong>s,<br />
cephalospor<strong>in</strong>s, am<strong>in</strong>oglycosides, tetracycl<strong>in</strong>es, macrolides, qu<strong>in</strong>olones, trimethoprimsulfamethoxazole,<br />
and rifamp<strong>in</strong> (class III, level C). Reed et al reported seven cases, and<br />
concluded that, compared to historic controls, doxycycl<strong>in</strong>e and rifamp<strong>in</strong> shortened the<br />
course of the disease and improved visual recovery (class III, level C) (Reed, 1998). The<br />
ophthalmologic manifestations of cat-scratch disease are outl<strong>in</strong>ed <strong>in</strong> Table 3–3.<br />
ODEMS may also occur with stage II Lyme disease (Bialasiewicz, 1992; Karma, 1995;<br />
Lesser, 1990; Miller, 1995a; Schönherr, 1990, 1991). Toxoplasmosis may also cause<br />
ODEMS or neuroret<strong>in</strong>itis (Fish, 1993; Moreno, 1992). Two features that often occur<br />
with toxoplasmosis neuroret<strong>in</strong>itis, but that are uncommon with idiopathic ODEMS, are<br />
a prom<strong>in</strong>ent anterior chamber <strong>in</strong>flammation and the presence of toxoplasmosis chorioret<strong>in</strong>al<br />
scars. Toxoplasmosis neuroret<strong>in</strong>itis is perhaps more likely to cause recurrent<br />
episodes of ODEMS or neuroret<strong>in</strong>itis, compared to the usual monophasic course of<br />
idiopathic ODEMS.<br />
ODEMS and neuroret<strong>in</strong>itis must be differentiated from other entities <strong>in</strong> which optic<br />
disc swell<strong>in</strong>g occurs with or without macular star formation, <strong>in</strong>clud<strong>in</strong>g vascular causes<br />
(e.g., anterior ischemic optic neuropathy, posterior hyaloid detachment, branch or<br />
central ret<strong>in</strong>al artery occlusion, hypertension, diabetes, polyarteritis nodosa, <strong>in</strong>flammatory<br />
bowel disease, and Eales’ disease), papilledema from <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure, optic nerve tumors or <strong>in</strong>filtrative processes, diffuse unilateral subacute<br />
neuroret<strong>in</strong>itis (DUSN), and the acute neuroret<strong>in</strong>opathy that may occur associated<br />
with progressive facial hemiatrophy (Parry-Romberg syndrome) (Akura, 2001,<br />
Friedrich, 2001; Garcia-Arumi, 1994; Gass, 1991; Leavitt, 1997; Lee, 2002; Matsuda,<br />
1994; Verm, 1997). With many of these etiologies macular stars are rarely seen<br />
and usually the differential diagnosis is not difficult on cl<strong>in</strong>ical grounds. A<br />
suggested workup for patients with optic disc edema with macular star is outl<strong>in</strong>ed <strong>in</strong><br />
Figure 3–1.
What Is the Prognosis <strong>in</strong> Cases of ODEMS?<br />
Optic Disc Edema with a Macular Star and <strong>Neuro</strong>ret<strong>in</strong>itis 67<br />
Table 3–3. Ophthalmologic Abnormalities <strong>in</strong> Cat-Scratch Disease<br />
(May Be Seen <strong>in</strong> Various Comb<strong>in</strong>ations)<br />
<strong>An</strong>terior segment<br />
Par<strong>in</strong>aud’s oculoglandular syndrome<br />
Follicular conjunctivitis<br />
Conjunctival bacillary angiomatosis<br />
Posterior segment<br />
Optic nerve<br />
Optic disc edema with macular star (ODEMS) or neuroret<strong>in</strong>itis<br />
Optic neuritis (papillitis)<br />
Optic nerve granuloma<br />
Optic disc edema with peripapillary serous ret<strong>in</strong>al detachment<br />
Bilateral disc edema with multifocal ret<strong>in</strong>itis<br />
Vitreous<br />
Vitreitis with or without anterior uveitis<br />
Vitreous hemorrhage<br />
Intermediate uveitis with or without ret<strong>in</strong>al vasculitis<br />
Ret<strong>in</strong>al<br />
Ret<strong>in</strong>al white spot syndrome<br />
Focal choroidal <strong>in</strong>filtrates<br />
Acute multifocal ret<strong>in</strong>itis<br />
Focal or diffuse ret<strong>in</strong>al vasculitis<br />
Branch or central ret<strong>in</strong>al artery occlusions<br />
Branch ret<strong>in</strong>al ve<strong>in</strong> occlusions<br />
Macular edema<br />
Atrophic scar <strong>in</strong> ret<strong>in</strong>al pigment epithelium<br />
Serous ret<strong>in</strong>al detachment<br />
Subret<strong>in</strong>al angiomatous mass lesion<br />
Source: Bafna, 1996; Bar, 1990; Bhatti, 2001; Brazis, 1986; Carithers, 1991;<br />
Chrousos, 1990; Cohen, 1995; Cunn<strong>in</strong>gham, 1997a,b, 2000; Earhart, 2000; Fish,<br />
1992; Gass, 1977; Ghauri, 1998; Golnik, 1994; Gray, 1999; K<strong>in</strong>g, 1991; Lee,<br />
1994; May, 1995; McCrary, 1994, 1997; Ormerod, 1998, 1999; Reed, 1998;<br />
Schwartzman, 1994, 1995; Soheilian, 1996; Solley, 1999; Suhler, 2000; Ulrich,<br />
1992; Wade, 2000; Zacchei, 1995; Zhao, 1991.<br />
ODEMS is usually a benign condition that resolves spontaneously without treatment<br />
(Rosen, 1999). The disc edema and peripapillary ret<strong>in</strong>al detachment tend to resolve over<br />
a period of 2 to 3 months, while the macular star usually beg<strong>in</strong>s to disappear after 1<br />
month. The macular star, however, may persist for up to 1 year. Optic atrophy and<br />
macular ret<strong>in</strong>al pigment epithelial changes may be residuals of previous ODEMS. The<br />
prognosis for visual recovery <strong>in</strong> ODEMS is usually good, but significant residual visual<br />
disability may occasionally occur (Lee, 1998a). Recurrences of ODEMS or neuroret<strong>in</strong>itis<br />
<strong>in</strong> the same or fellow eye have been described <strong>in</strong> idiopathic as well as <strong>in</strong>fectious cases,<br />
especially <strong>in</strong> patients with toxoplasmosis (Fish, 1993; Purv<strong>in</strong>, 1994). Also, Purv<strong>in</strong> and
68 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 3–1. Evaluation of optic disc edema with a macular star (ODEMS) or neuroret<strong>in</strong>itis.
Chioran described an apparently dist<strong>in</strong>ct type of recurrent ODEMS <strong>in</strong> patients who<br />
experienced from two to seven attacks of ODEMS at <strong>in</strong>tervals rang<strong>in</strong>g from 1 to 10 years<br />
(Purv<strong>in</strong>, 1994). The attacks often affected both eyes but never simultaneously. Visual<br />
field defects were of the nerve fiber bundle type <strong>in</strong>stead of the central or cecocentral<br />
scotomas that are most often noted <strong>in</strong> benign ODEMS. Patients with the recurrent form<br />
of the disease may not experience significant improvement <strong>in</strong> optic nerve function.<br />
Although optic neuritis is a risk factor for the development of multiple sclerosis (see<br />
Chapter 2), ODEMS or neuroret<strong>in</strong>itis is not (Optic Neuritis Study Group, 1997). For<br />
example, <strong>in</strong> the Optic Neuritis Treatment Trial (see Chapter 2), among patients with<br />
swollen discs, cl<strong>in</strong>ically def<strong>in</strong>ite multiple sclerosis did not develop <strong>in</strong> any patient who<br />
had macular exudates (Optic Neuritis Study Group, 1997). Because a macular exudate<br />
may not develop <strong>in</strong> cases of ODEMS until 2 weeks after presentation, patients who<br />
demonstrate acute papillitis with a normal macula should be reevaluated with<strong>in</strong> 2 weeks<br />
for the development of a macular star. Its presence makes the subsequent development<br />
of multiple sclerosis extremely unlikely.<br />
In summary, there is no class I or class II evidence for the diagnosis and treatment of<br />
ODEMS. Individual history and exam<strong>in</strong>ation should guide the evaluation focus<strong>in</strong>g on<br />
exposure history (syphilis, Lyme disease, tuberculosis), systemic f<strong>in</strong>d<strong>in</strong>gs (typhus, viral<br />
illness, fungi, tuberculosis, Leptospira), or typical ophthalmoscopic features (e.g., adjacent<br />
chorioret<strong>in</strong>al scar <strong>in</strong> toxoplasmosis). In typical ODEMS, however, most of the cases<br />
rema<strong>in</strong> idiopathic. Cat-scratch disease is emerg<strong>in</strong>g as a common etiology <strong>in</strong> cases with a<br />
proven cause and perform<strong>in</strong>g a Bartonella henselae titer is a reasonable practice option <strong>in</strong><br />
ODEMS (class III, level C). In the absence of risk factors or cl<strong>in</strong>ical suspicion, the yield<br />
for test<strong>in</strong>g for other <strong>in</strong>fectious etiologies is low (class III, level U). Rout<strong>in</strong>e test<strong>in</strong>g even<br />
for treatable disorders such as syphilis (serology), Lyme disease, or tuberculosis (chest<br />
radiography, purified prote<strong>in</strong> derivative sk<strong>in</strong> test) are practice options of uncerta<strong>in</strong> yield<br />
(class III, level U).<br />
There is no proven treatment for idiopathic ODEMS (class III, level C). Steroids have<br />
been used <strong>in</strong> some cases with unclear effect (class III, level U). Aggressive immunosuppressive<br />
agents may be considered <strong>in</strong> the rare recurrent cases (class IV) (Purv<strong>in</strong>,<br />
1994). If a specific <strong>in</strong>fectious agent is discovered, then appropriate antibiotics should be<br />
considered, but the data are limited (class III, level B). Specific attention, however,<br />
should focus on treatable (e.g., cat-scratch disease, Lyme, syphilis, and tuberculosis)<br />
<strong>in</strong>fectious etiologies (class III, level B). Atypical cases (e.g., bilateral) might require<br />
further evaluation (e.g., neuroimag<strong>in</strong>g or lumbar puncture to exclude papilledema) to<br />
exclude other causes of ODEMS (class III).<br />
References<br />
Optic Disc Edema with a Macular Star and <strong>Neuro</strong>ret<strong>in</strong>itis 69<br />
Akura J, Ikeda T, Sato K, Ikeda N. (2001). Macular star associated with posterior hyaloid detachment. Acta<br />
Ophthalmol Scand 79:317–318.<br />
Bafna S, Lee AG. (1996). Bilateral optic disc edema and multifocal ret<strong>in</strong>al lesions without loss of vision <strong>in</strong> cat<br />
scratch disease. Arch Ophthalmol 114:1016–1017.<br />
Bar S, Segal M, Shapira R, Savir H. (1990). <strong>Neuro</strong>ret<strong>in</strong>itis associated with cat scratch disease. Am J Ophthalmol<br />
110:703–705.<br />
Bhatti MT, Asif R, Bhatti LB. (2001). Macular star <strong>in</strong> neuroret<strong>in</strong>itis. Arch <strong>Neuro</strong>l 58:1008–1009.<br />
Bialasiewicz AA. (1992). Augenbefunde bei Lyme-Borreliose. Ophthalmologe 89:W47–W59.<br />
Brazis PW, Lee AG. (1996). Optic disk edema with a macular star. Mayo Cl<strong>in</strong> Proc 71:1162–1166.
70 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Brazis PW, Stokes HR, Erv<strong>in</strong> FR. (1986). Optic neuritis <strong>in</strong> cat scratch disease. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 6:172–174.<br />
Carithers HA, Margileth AM. (1991). Cat-scratch disease. Acute encephalopathy and other neurologic manifestations.<br />
Am J Dis Child 145:98–101.<br />
Chrousos GA, Drack AV, Young M, et al. (1990). <strong>Neuro</strong>ret<strong>in</strong>itis <strong>in</strong> cat scratch disease. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
10:92–94.<br />
Cohen SM, Davis JL, Gass DM. (1995). Branch ret<strong>in</strong>al arterial occlusions <strong>in</strong> multifocal ret<strong>in</strong>itis with optic nerve<br />
edema. Arch Ophthalmol 113:1271–1276.<br />
Conrad DA. (2001). Treatment of cat-scratch disease. Curr Op<strong>in</strong> Pediatr 13:56–59.<br />
Cunn<strong>in</strong>gham ET, Koehler JE. (2000). Perspective. Ocular bartonellosis. Am J Ophthalmol 130:340–349.<br />
Cunn<strong>in</strong>gham ET Jr, Schatz H, McDonald HR, Johnson RN. (1997a). Acute multifocal ret<strong>in</strong>itis. Am J Ophthalmol<br />
123:347–357.<br />
Cunn<strong>in</strong>gham ET Jr, McDonald HR, Schatz H, et al. (1997b). Inflammatory mass of the optic nerve head associated<br />
with systemic Bartonella henselae <strong>in</strong>fection. Arch Ophthalmol 115:1596–1597.<br />
Dhar MY, Goel JL, Sota LD. (1997). Optic neuroret<strong>in</strong>itis, a rare manifestation of herpes zoster ophthalmicus: a case<br />
report. J Commun Dis 29:57–61.<br />
Earhart KC, Power MH. (2000). Bartonella neuroret<strong>in</strong>itis. N Engl J Med 343:1459.<br />
Fish RH, Hogan RN, Night<strong>in</strong>gale SD, <strong>An</strong>and R. (1992). Peripapillary angiomatosis associated with cat-scratch<br />
neuroret<strong>in</strong>itis. Arch Ophthalmol 110:323.<br />
Fish RH, Hosk<strong>in</strong>s JC, Kl<strong>in</strong>e LB. (1993). Toxoplasmosis neuroret<strong>in</strong>itis. Ophthalmology 100:1177–1182.<br />
Foster RE, Lowder CY, Meisler DM, et al. (1990). Mumps neuroret<strong>in</strong>itis <strong>in</strong> an adolescent. Am J Ophthalmol<br />
110:91–93.<br />
Friedrich Y, Fe<strong>in</strong>er M, Gawi H, Friedman Z. (2001). Diabetic papillopathy with macular star mimick<strong>in</strong>g cl<strong>in</strong>ically<br />
significant diabetic macular edema. Ret<strong>in</strong>a 21:80–82.<br />
Garcia-Arumi J, Salvador F, Corcostegui B, Mateo C. (1994). <strong>Neuro</strong>ret<strong>in</strong>itis associated with melanocytoma of the<br />
optic disk. Ret<strong>in</strong>a 14:173–176.<br />
Gass JDM. (1977). Diseases of the optic nerve that may simulate macular disease. Trans Am Acad Ophthalmol<br />
Otolaryngol 83:763–770.<br />
Gass JDM, Harb<strong>in</strong> TS Jr, Del Piero EJ. (1991). Exudative stellate neuroret<strong>in</strong>opathy and Coat’s syndrome <strong>in</strong> patients<br />
with progressive hemifacial atrophy. Eur J Ophthalmol 1:2–10.<br />
Ghauri RR, Lee AG, Purv<strong>in</strong> V. (1998). Optic disk edema with a macular star. Surv Ophthalmol 43:270–274.<br />
Golnik KC, Marotto ME, Fanous MM, et al. (1994). Ophthalmic manifestations of Rochalimaea species.<br />
Am J Ophthalmol 118:145–151.<br />
Gray AV, Reed JB, Wendel RT, Morse LS. (1999). Bartonella henselae <strong>in</strong>fection associated with peripapillary<br />
angioma, branch ret<strong>in</strong>al artery occlusion, and severe visual loss. Am J Ophthalmol 127:223–224.<br />
Halper<strong>in</strong> LS. (1992). <strong>Neuro</strong>ret<strong>in</strong>itis due to seronegative syphilis associated with human immunodeficiency virus.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:171–172.<br />
Hamard P, Hamard H, Ngohou S. (1994). La neuroret<strong>in</strong>ite stellaire idiopathique de Leber. A propos de neuf cas.<br />
J Fr Ophtalmol 17:116–123.<br />
Karma A, Seppala I, Mikkila H, et al. (1995). Diagnosis and cl<strong>in</strong>ical characteristics of ocular Lyme borreliosis.<br />
Am J Ophthalmol 119:127–135.<br />
Kerkoff FT, Bergmans AM, van Der Zee A, Rothova A. (1999). Demonstration of Bartonella grahamii DNA <strong>in</strong><br />
ocular fluids of a patient with neuroret<strong>in</strong>itis. J Cl<strong>in</strong> Microbiol 37:4034–4038.<br />
K<strong>in</strong>g MH, Cartwright MJ, Carney MD. (1991). Leber’s idiopathic stellate neuroret<strong>in</strong>itis. <strong>An</strong>n Ophthalmol 23:58–60.<br />
Kosmorsky GS, Prayson R. (1995). Primary optic pathway sarcoidosis <strong>in</strong> a 38-year-old white man.<br />
J <strong>Neuro</strong>-ophthalmol 16:188–190.<br />
Labalette P, Bermond D, Dedes V, Savage C. (2001). Cat-scratch disease neuroret<strong>in</strong>itis diagnosed by a polymerase<br />
cha<strong>in</strong> reaction approach. Am J Ophthalmology 132:575–576.<br />
Leavitt JA, Pruthi S, Morgenstern BZ. (1997). Hypertensive ret<strong>in</strong>opathy mimick<strong>in</strong>g neuroret<strong>in</strong>itis <strong>in</strong> a twelve-yearold<br />
girl. Surv Ophthalmol 41:477–480.<br />
Leber T. (1916). Die pseudonephritischen Netzhauterkrankungen, die Ret<strong>in</strong>itis stellata: die Purtschersche<br />
Netzhautaffektion nach schwere Schädelverletzung. In: Graefe AC, Saemisch T, eds. Graefe-Saemisch<br />
Handbuch der Gesamten Augenheilkunde. 2nd ed., vol. 7, pt. 2. Leipzig, East Germany, Engelmann.<br />
Lee AG, Beaver HA, Monsul NT, Miller NR. (2002). Acute bilateral optic disk edema with a macular figure <strong>in</strong> a<br />
12-year-old girl. Surv Ophthalmol 47:42–49.<br />
Lee AG, Brazis PW. (1998a). Poor visual outcome follow<strong>in</strong>g optic disc edema with a macular star (neuroret<strong>in</strong>itis).<br />
J <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:57–61.<br />
Lee AG, Orenga-Nania SD, Brazis PW, Lech EM. (1998b). Poor visual outcome follow<strong>in</strong>g optic disc edema with a<br />
macular star (‘‘neuroret<strong>in</strong>itis’’). <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:57–61.
Optic Disc Edema with a Macular Star and <strong>Neuro</strong>ret<strong>in</strong>itis 71<br />
Lee WR, Chawla JC, Reid R. (1994). Bacillary angiomatosis of the conjunctiva. Am J Ophthalmol 118:152–157.<br />
Lesser RL, Kornmehl EW, Pachner AR, et al. (1990). <strong>Neuro</strong>-ophthalmologic manifestations of Lyme disease.<br />
Ophthalmology 97:699–706.<br />
Matsuda A, Ch<strong>in</strong> S, Ohashi T. (1994). A case of neuroret<strong>in</strong>itis associated with long-stand<strong>in</strong>g polyarteritis nodosa.<br />
Ophthalmologica 208:168–171.<br />
May EF, Levi L, Ng JD, Truxal AR. (1995). Rochalimaea neuroret<strong>in</strong>itis and ret<strong>in</strong>al vasculitis. Presented at the<br />
meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Tucson, Arizona.<br />
McCleish WM, Pulido JS, Holland S, et al. (1990). The ocular manifestations of syphilis <strong>in</strong> the human<br />
immunodeficiency virus type 1–<strong>in</strong>fected host. Ophthalmology 97:196–203.<br />
McCrary B, Cockerhan W, Pierce P. (1994). <strong>Neuro</strong>ret<strong>in</strong>itis <strong>in</strong> cat-scratch disease associated with the macular star.<br />
Pediatr Infect Dis J 13:938–939.<br />
McCrary B, Cockerham W, Pierce P. (1997). <strong>Neuro</strong>ret<strong>in</strong>itis <strong>in</strong> cat scratch disease associated with macular star.<br />
J Miss State Med Assoc 38:158–159.<br />
Miller NR. (1995a). In: Miller NR, ed. Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. Baltimore: Williams & Wilk<strong>in</strong>s,<br />
pp. 3657–3658.<br />
Miller NR. (1995b). Miller NR, ed. Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. Baltimore: Williams & Wilk<strong>in</strong>s,<br />
pp. 4487–4489.<br />
Moreno RJ, Weisman J, Waller S. (1992). <strong>Neuro</strong>ret<strong>in</strong>itis: an unusual presentation of ocular toxoplasmosis.<br />
<strong>An</strong>n Ophthalmol 24:68–70.<br />
N<strong>in</strong>omiya H, Hamada T, Akiya S, Kazama H. (1990). Three cases of acute syphilitic neuroret<strong>in</strong>itis. Folia Ophthalmol<br />
Jpn 41:2088–2094.<br />
O’Halloran HS, Draud K, M<strong>in</strong>ix M, et al. (1998). Leber’s neuroret<strong>in</strong>itis <strong>in</strong> a patient with serologic evidence of<br />
Bartonella elizabethae. Ret<strong>in</strong>a 18:276–278.<br />
Optic Neuritis Study Group. (1997). The 5-year risk of MS after optic neuritis. Experience of the Optic Neuritis<br />
Treatment Trial. <strong>Neuro</strong>logy 49:1404–1413.<br />
Ormerod LD, Dailey JP (1999). Ocular manifestations of cat-scratch disease. Curr Op<strong>in</strong> Ophthalmol 10:209–216.<br />
Ormerod LD, Skolnick KA, Menosky MM, et al. (1998). Ret<strong>in</strong>al and choroidal manifestations of cat-scratch<br />
disease. Ophthalmology 105:1024–1031.<br />
Purv<strong>in</strong> VA, Chioran G. (1994). Recurrent neuroret<strong>in</strong>itis. Arch Ophthalmol 112:365–371.<br />
Ray S, Gragoudas E. (2001). <strong>Neuro</strong>ret<strong>in</strong>itis. Int Ophthalmol Cl<strong>in</strong> 41:83–102.<br />
Reed JB, Scales DK, Wong MT, et al. (1998). Bartonella henselae neuroret<strong>in</strong>itis <strong>in</strong> cat scratch disease. Diagnosis,<br />
management, and sequelae. Ophthalmology 105:459–466.<br />
Rosen BS, Barry CJ, Nicoll AM, Constable IJ. (1999). Conservative management of documented neuroret<strong>in</strong>itis <strong>in</strong><br />
cat scratch disease associated with Bartonella henselae. Aust NZ J Ophthalmol 27:153–156.<br />
Schönherr U, Lang GE, Maythaler FH. (1991). Bilaterale Lebersche <strong>Neuro</strong>ret<strong>in</strong>itis stellata bei Borrelia burgdorferi-<br />
Serokonversion. Kl<strong>in</strong> Monatsbl Augenheilkd 198:44–47.<br />
Schönherr U, Wilk CM, Lang GE, Naumann GOH. (1990). Intraocular manifestations of Lyme borreliosis.<br />
Presented at the Fourth International Conference on Borreliosis, Stockholm, Sweden, June 18–21.<br />
Schwartzman WA, Patnaik M, Barka NE, et al. (1994). Rochalimara antibodies <strong>in</strong> HIV associated neurologic<br />
disease. <strong>Neuro</strong>logy 44:1312–1316.<br />
Schwartzman WA, Patnaik M, <strong>An</strong>gula FJ, et al. (1995). Bartonella (Rochalimaea) antibodies, dementia, and cat<br />
ownership among men <strong>in</strong>fected with immunodeficiency virus. Cl<strong>in</strong> Infect Dis 21:954–959.<br />
Soheilian M, Markomichelakis N, Foster CS. (1996). Intermediate uveitis and ret<strong>in</strong>al vasculitis as manifestations of<br />
cat scratch disease. Am J Ophthalmol 122:582–584.<br />
Solley WA, Mart<strong>in</strong> DF, Newman NJ, et al. (1999). Cat scratch disease. Posterior segment manifestations.<br />
Ophthalmology 106:1546–1553.<br />
Stechschulte SU, Kim RY, Cunn<strong>in</strong>gham ET. (1999). Tuberculous neuroret<strong>in</strong>itis. J <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:201–204.<br />
Suhler EB, Lauer AK, Rosenbaum JT. (2000). Prevalence of serologic evidence of cat scratch disease <strong>in</strong> patients<br />
with neuroret<strong>in</strong>itis. Ophthalmology 107:871–876.<br />
Thompson PK, Vaphiades MS, Sacccente M. (1999). Cat-scratch disease present<strong>in</strong>g as neuroret<strong>in</strong>itis and peripheral<br />
facial palsy. J <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:240–241.<br />
Ulrich GG, Waecker NJ Jr, Meister SJ, et al. (1992). Cat scratch disease associated with neuroret<strong>in</strong>itis <strong>in</strong> a 6-year-old<br />
girl. Ophthalmology 99:246–249.<br />
Verm A, Lee AG. (1997). Bilateral optic disk edema with macular exudates as the manifest<strong>in</strong>g sign of a cerebral<br />
arteriovenous malformation. Am J Ophthalmol 123:422–424.<br />
Wade NK, Po S, Wong IG, Cunn<strong>in</strong>gham ET Jr. (1999). Bilateral Bartonella-associated neuroret<strong>in</strong>itis. Ret<strong>in</strong>a<br />
19:355–356.
72 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Wade NK, Levi L, Jones MR, et al. (2000). Optic disk edema associated with peripapillary serous ret<strong>in</strong>al<br />
detachment: an early sign of systemic Bartonella henselae <strong>in</strong>fection. Am J Ophthalmol 130:327–334.<br />
Zacchei AC, Newman NJ, Sternberg P. (1995). Serous ret<strong>in</strong>al detachment of the macula associated with cat scratch<br />
disease. Am J Ophthalmol 120:796–797.<br />
Zhao X, Ge B. (1991). Treatment of papillo-ret<strong>in</strong>itis and uveitis with cat scratch disease by comb<strong>in</strong>ation of TCM<br />
and modern drugs. J Trad Ch<strong>in</strong> Med 11:184–186.
4 r<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features for Typical<br />
Nonarteritic AION?<br />
<strong>An</strong>terior ischemic optic neuropathy (AION) is characterized cl<strong>in</strong>ically by the acute onset<br />
of usually unilateral visual loss. Although pa<strong>in</strong> may occur <strong>in</strong> approximately 10% (range<br />
8–30% <strong>in</strong> various series) of patients, the visual loss is typically pa<strong>in</strong>less. Middle-aged to<br />
older patients (usually older than 50) are the predom<strong>in</strong>ant populations at risk for AION.<br />
The ocular exam<strong>in</strong>ation <strong>in</strong> these patients reveals the follow<strong>in</strong>g: (1) ipsilateral visual<br />
acuity and visual field loss; (2) a relative afferent pupillary defect; and (3) edema of the<br />
optic nerve head with or without peripapillary hemorrhages (Arnold, 1994b; Feldon,<br />
1999; Friedland, 1996; Gerl<strong>in</strong>g, 1998a,b; Grosvenor, 1993; Hattenhauer, 1997; Hayreh,<br />
1990a, 1997a; Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial, 1995, 1996; Johnson,<br />
1993, 1994a,b, 1996a,b; Kay, 1991; Lessell, 1999; Moro, 1990; Rizzo, 1991; Sawle, 1990).<br />
The presence of optic disc edema (anterior optic neuropathy) <strong>in</strong> the acute phase is<br />
essential for the diagnosis of AION to be made. Rarely, AION may present with<br />
asymptomatic disc edema without visual loss or field defect (Gordon, 1997) or be<br />
associated with macular edema (Tomsak, 1998). After resolution of the disc edema, the<br />
optic disc develops sector or diffuse pallor. The typical cl<strong>in</strong>ical features of nonarteritic<br />
(NA-AION) are outl<strong>in</strong>ed <strong>in</strong> Table 4–1.<br />
The optic disc appearance may help differentiate AION from optic neuritis (ON),<br />
although there are overlapp<strong>in</strong>g features. Optic disc stereo photos were reviewed by<br />
masked observers (87 AION and 68 ON) (Warner, 1997). Altitud<strong>in</strong>al disc swell<strong>in</strong>g was<br />
more than three times more common <strong>in</strong> AION than ON, although most discs were<br />
diffusely swollen. Most patients with AION had hemorrhages, whereas most ON cases<br />
did not. Almost all discs with ON had normal color or were hyperemic, and only 35% of<br />
discs with AION had pallid swell<strong>in</strong>g. Pallid swell<strong>in</strong>g was so rare <strong>in</strong> ON, however, that<br />
of discs with pallor, 93% had AION. Arterial attenuation was also much more typical of<br />
AION. AION was the cl<strong>in</strong>ical diagnosis <strong>in</strong> 82% of cases with altitud<strong>in</strong>al edema, 81% of<br />
the cases with disc hemorrhage, 93% of the cases with pallid edema, and 90% of the<br />
73
74 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 4–1. Typical <strong>Cl<strong>in</strong>ical</strong> Features of Nonarteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy<br />
(NA-AION)<br />
Age usually over 40 years<br />
Unilateral variable loss of visual acuity and=or visual field<br />
Visual field defects consistent with an optic neuropathy (e.g., central, cecocentral, arcuate, or<br />
altitud<strong>in</strong>al)<br />
Optic disc edema (usually pallid edema) <strong>in</strong> the acute phase followed by optic atrophy that may<br />
be sector or diffuse<br />
Relative afferent pupillary defect <strong>in</strong> unilateral or bilateral but asymmetric cases<br />
Small cup and cup to disc ratio (less than 0.2) (Burde, 1993; Feldon, 1999; Lav<strong>in</strong>, 1994; Salomon,<br />
1999a)<br />
Often associated with underly<strong>in</strong>g vasculopathic risk factors (e.g., hypertension, diabetes, smok<strong>in</strong>g,<br />
ischemic heart disease, hypercholesterolemia) (Chung, 1994; Feldon, 1999; Hayreh, 1994;<br />
Salomon, 1999a)<br />
Lack of premonitory symptoms (e.g., transient visual loss)<br />
Usually visual loss rema<strong>in</strong>s static but may improve slightly or progress<br />
End-stage optic disc appearance is segmental or diffuse pallor without significant cupp<strong>in</strong>g<br />
(unlike arteritic AION) (Danesh-Meyer, 2001)<br />
cases with arterial attenuation. A pale nerve with hemorrhage, regardless of type of<br />
edema, always represented AION (100%). A normal color nerve without hemorrhage<br />
reflected ON <strong>in</strong> 91% of the cases, <strong>in</strong>creased from only 76% if hemorrhage was not<br />
considered. A hyperemic nerve with hemorrhage represented AION <strong>in</strong> 82% of cases, but<br />
if altitud<strong>in</strong>al edema was also present, AION <strong>in</strong>cidence <strong>in</strong>creased to 93%.<br />
Ischemic optic neuropathy (ION) without acute disc edema is referred to as posterior<br />
ischemic optic neuropathy (PION). PION is an atypical presentation of ION but it may<br />
occur <strong>in</strong> several conditions as listed <strong>in</strong> Table 4–2.<br />
Younger patients (less than 40 years of age) with diabetes (Inoue, 1997; Jacobson,<br />
1997), migra<strong>in</strong>e, severe hypertension <strong>in</strong>clud<strong>in</strong>g preeclampsia, or oral contraceptive use<br />
Table 4–2. Conditions Associated with Posterior Ischemic Optic <strong>Neuro</strong>pathy<br />
Atherosclerosis and arteriosclerosis (Sadda, 2001)<br />
Severe hypotension or blood loss<br />
Diabetes (Inoue, 1997)<br />
Collagen vascular disorders (e.g., systemic lupus erythematosus)<br />
Giant cell (temporal) arteritis (Sadda, 2001)<br />
Hematologic disorders<br />
Infection (e.g., Aspergillus, herpes zoster)<br />
Internal carotid artery occlusion or dissection (Biousse, 1998b; Kerty, 1999)<br />
Malignant hypertension<br />
Migra<strong>in</strong>e<br />
After surgical procedures (hypotension, anemia) (Sadda, 2001)<br />
Severe anemia<br />
Radiation therapy<br />
Thromboembolism (e.g., <strong>in</strong>ternal carotid artery disease)<br />
Source: Lee, 1995a, 1996; Sadda, 2001.
may also develop ION. We consider the development of ION <strong>in</strong> patients less than 40<br />
years old to be an atypical presentation (R<strong>in</strong>aldi, 1990). A cl<strong>in</strong>ical presentation of AION<br />
may occur <strong>in</strong> young patients without any known vasculopathic risk factors and has<br />
been termed AION of the young (AIONY). AIONY differs from typical AION <strong>in</strong> that<br />
recurrent attacks are more common than with typical NA-AION.<br />
Bilateral simultaneous <strong>in</strong>volvement may occur <strong>in</strong> NA-AION (up to 15% of cases), but<br />
we consider this also an atypical f<strong>in</strong>d<strong>in</strong>g. Giant cell arteritis (as well as other causes of a<br />
bilateral optic neuropathy) should be excluded <strong>in</strong> these cases (Hayreh, 1998).<br />
Diabetic papillopathy is probably an atypical form of AION described <strong>in</strong> diabetics<br />
who present with m<strong>in</strong>imal visual symptoms. This entity usually resolves <strong>in</strong> weeks to<br />
months. The cl<strong>in</strong>ical features of diabetic papillopathy are outl<strong>in</strong>ed <strong>in</strong> Table 4–3.<br />
What Other Conditions Are Associated with<br />
Ischemic Optic <strong>Neuro</strong>pathy?<br />
ION has been reported <strong>in</strong> association with a number of systemic conditions listed <strong>in</strong><br />
Table 4–4.<br />
What <strong>Cl<strong>in</strong>ical</strong> Features Are Atypical for<br />
<strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy?<br />
Patients with an atypical AION should undergo a complete evaluation to exclude other<br />
causes of an optic neuropathy (e.g., <strong>in</strong>flammatory, <strong>in</strong>filtrative, compressive optic<br />
neuropathies). The cl<strong>in</strong>ical features that are atypical for AION are listed <strong>in</strong> Table 4–5.<br />
Recurrence of NA-AION <strong>in</strong> the same eye is uncommon. Hayreh et al studied 594<br />
consecutive patients with a diagnosis of NA-AION and found that recurrence occurred<br />
<strong>in</strong> the same eye <strong>in</strong> 45 patients (7.6%) with a median follow-up of 3.1 years (Hayreh,<br />
2001b). Although it is uncommon for NA-AION to recur <strong>in</strong> the same eye, it may <strong>in</strong>volve<br />
Table 4–3. <strong>Cl<strong>in</strong>ical</strong> Features of Diabetic Papillopathy<br />
May be unilateral or bilateral (simultaneous or sequential)<br />
May have relative afferent pupillary defect if unilateral or bilateral but asymmetric<br />
May be associated with type I or type II diabetes<br />
Disc swell<strong>in</strong>g is mild to moderate and the disc is consistently hyperemic<br />
Disc edema usually resolves with<strong>in</strong> 1 to 10 months<br />
Macular edema and capillary nonperfusion are frequent associated f<strong>in</strong>d<strong>in</strong>gs<br />
Small cup-to-disc ratio <strong>in</strong> un<strong>in</strong>volved fellow eyes (the ‘‘disc at risk’’)<br />
Significant (55 seconds) delay <strong>in</strong> fluoresce<strong>in</strong> fill<strong>in</strong>g of all or a portion of the optic disc may occur<br />
M<strong>in</strong>imal if any visual symptoms<br />
May have enlarged bl<strong>in</strong>d spot or arcuate defect<br />
Residual visual loss due to associated macular edema and ret<strong>in</strong>opathy<br />
Occasionally residual mild optic atrophy<br />
Source: Arnold, 1997; Burde, 1993; Katz, 1990a; Regillo, 1995; Vaphiades, 2002.<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 75
76 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 4–4. Conditions Associated with <strong>An</strong>terior (and Posterior) Ischemic Optic <strong>Neuro</strong>pathy<br />
Systemic vasculopathy<br />
Common<br />
Hypertension (Feldon, 1999; Hayreh, 1990b, 1994b; Salomon, 1999b)<br />
Hypotension<br />
Diabetes mellitus (Feldon, 1999; Hayreh, 1994b; Inoue, 1997; Salomon, 1999b)<br />
Arteriosclerosis, atherosclerosis, and ischemic heart disease (Hayreh, 1994b; Sadda, 2001;<br />
Salomon, 1999b)<br />
Hypercholesterolemia (Salomon, 1999b)<br />
Uncommon<br />
Female carrier of Fabry’s disease (Abe, 1992)<br />
Takayasu’s arteritis (Schmidt, 1997)<br />
Carotid occlusion and dissection (Biousse, 1998a,b; Gotte, 2000; Kerty, 1999; Mokri, 1996;<br />
Rivk<strong>in</strong>, 1990; Strome, 1997)<br />
Carotid artery hypoplasia (Horowitz, 2001)<br />
Thromboangiitis obliterans<br />
Vasospasm (Hayreh, 1997a; Kaiser, 1996)<br />
Migra<strong>in</strong>e<br />
Raynaud’s disease<br />
Acute blood loss or hypotension (Brown, 1994; Chun, 1997; Connolly, 1994; Cull<strong>in</strong>ane, 2000;<br />
Hayreh, 1999; Lee, 1995b; Shaked, 1998; Teshome, 1999)<br />
Systemic <strong>in</strong>flammatory response syndrome (survivors of severe <strong>in</strong>juries) (Cul<strong>in</strong>ane, 2000)<br />
Postsurgical (Sadda, 2001; Williams, 1995)<br />
Cardiopulmonary bypass procedures (Lund, 1994; Moster, 1998; Shapira, 1996; Spoor, 1991)<br />
Lumbar sp<strong>in</strong>e surgery (Alexandrakis, 1999; Brown, 1994; Cheng, 2000; Dilger, 1998; Katz, 1994;<br />
Lee, 1995b; Loftman, 1996; Myers, 1997; Roth, 1997; Smith, 1996; Stevens, 1997)<br />
Abdom<strong>in</strong>al surgery<br />
Radical neck dissection (Fenton, 2001; Kirkali, 1990; Marks, 1990; Nawa, 1992; Schnobel, 1995;<br />
Wilson, 1991)<br />
Leg ve<strong>in</strong> bypass surgery (Remigio, 2000)<br />
Mitral valve surgery<br />
Nasal surgery (<strong>in</strong>tranasal anesthetic) (Sav<strong>in</strong>o, 1990)<br />
Cholecystectomy<br />
Parathyroidectomy<br />
Radical prostatectomy (Williams, 1999)<br />
Liver transplant (Janicki, 2001)<br />
Coronary angiography<br />
After treatment for malignant hypertension (Connolly, 1994)<br />
Hemodialysis (Connolly, 1994)<br />
Nocturnal hypotension (Hayreh, 1994b, 1997c, 1999; Landau, 1996)<br />
Therapeutic phlebotomy<br />
Cardiac arrest<br />
Surgical (nonhypotensive or nonanemic)<br />
Cataract surgery (McCulley, 2001; Perez-Santonja, 1993)<br />
Laser <strong>in</strong> situ keratomileusis (LASIK) (Cameron, 2001; Cornblath, 2002; Lee, 2000)<br />
Secondary <strong>in</strong>traocular lens implantation<br />
After lower lid blepharoplasty (Good, 1999)<br />
(cont<strong>in</strong>ued)
Table 4–4. (cont<strong>in</strong>ued)<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 77<br />
After general surgery without significant blood loss<br />
Ret<strong>in</strong>al surgery<br />
Infectious<br />
Aspergillus<br />
Herpes zoster (Atmaca, 1992; Borruat, 1992; Kothe, 1990; Lexa, 1993)<br />
Lyme disease<br />
Recurrent herpes labialis (Johnson, 1996b)<br />
Staphylococcal cavernous s<strong>in</strong>us thrombosis<br />
Syphilis<br />
Acquired immune deficiency syndrome (AIDS)<br />
Elevated titers of IgG antibodies to Chlamydia pneumoniae (Weger, 2002)<br />
Inflammatory disorders (Coppetto, 1992)<br />
Allergic vasculitis<br />
Behçet’s disease<br />
Buerger’s disease<br />
Churg-Strauss disease (Acheson, 1993; Kattah, 1994; Sehgal, 1995; Vitali, 1996)<br />
Crohn’s disease<br />
Mixed connective tissue disease<br />
Polyarteritis nodosa<br />
Postviral vasculitis<br />
Relaps<strong>in</strong>g polychondritis (Massry, 1995)<br />
Rheumatoid arthritis<br />
Sjögren’s syndrome (Mochizuki, 2000; Rosler, 1995)<br />
Systemic lupus erythematosus (Siatkowski, 2001)<br />
HLA-B27 associated anterior uveitis and ankylos<strong>in</strong>g spondylitis (Tham, 2001)<br />
Ocular<br />
Hyperopia (Katz, 1993)<br />
Optic disc drusen (Lee, 2002; Liew, 1999)<br />
Papilledema<br />
Elevated <strong>in</strong>traocular pressure (Kalenak, 1991; Katz, 1992)<br />
Acute angle-closure glaucoma (Slav<strong>in</strong>, 2001)<br />
Birdshot ret<strong>in</strong>ochoroidopathy<br />
Hematologic abnormalities<br />
<strong>An</strong>emia (e.g., iron deficiency anemia) (Golnik, 1990; Kacer, 2001)<br />
Hyperhomocyste<strong>in</strong>emia (Kawasaki, 1999; Pianka, 2000; Weger, 2001)<br />
<strong>An</strong>tiphospholipid antibodies (Aziz, 2000; Galetta, 1991; Ohte, 1995; Re<strong>in</strong>o, 1997; Rosler, 1995)<br />
<strong>An</strong>tiphospholipid antibodies with factor V Leiden mutation (Sr<strong>in</strong>ivasan, 2001)<br />
Activated prote<strong>in</strong> C resistance (Worrall, 1997)<br />
Decreased concentrations of prote<strong>in</strong> C, prote<strong>in</strong> S, or antithromb<strong>in</strong> III (Bertram, 1995)<br />
G-6-PD deficiency syndrome<br />
Leukemia<br />
Lipid abnormalities (Giuffre, 1990; Talks, 1995)<br />
Lupus anticoagulant<br />
Pernicious anemia<br />
Polycythemia vera<br />
(cont<strong>in</strong>ued)
78 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 4–4. (cont<strong>in</strong>ued)<br />
Sickle cell trait and disease (Perlman, 1994)<br />
Thrombocytopenic purpura (Killer, 2000)<br />
Waldenström’s macroglobul<strong>in</strong>emia<br />
Embolic (Horton, 1995)<br />
Miscellaneous<br />
Acute <strong>in</strong>termittent porphyria<br />
Allergic disorders<br />
Serum sickness<br />
Bacille Calmette-Guér<strong>in</strong> (BCG) vacc<strong>in</strong>ation<br />
Urticaria<br />
Qu<strong>in</strong>cke’s edema<br />
Cardiac valvular disease (Hayreh, 1994a)<br />
Cavernous s<strong>in</strong>us thrombosis (Gupta, 1990)<br />
Cervical discopathies and vasospasm<br />
Favism<br />
Gastro<strong>in</strong>test<strong>in</strong>al ulcers (Hayreh, 1994a)<br />
Graves’ disease (Dosso, 1994)<br />
Human lymphocyte antigen-A29 (Johnson, 1993)<br />
Medications<br />
Interferon-alfa treatment (Purv<strong>in</strong>, 1995; Tang, 1995)<br />
Intracarotid carmust<strong>in</strong>e<br />
Sumatriptan for migra<strong>in</strong>e (Chiari, 1994)<br />
Omeprazole for gastric ulcer treatment (Schönhofer, 1997)<br />
Amiodarone (Mäntyjärvi, 1998)<br />
Oxymetazol<strong>in</strong>e nasal spray (nasal decongestant) (Fivgas, 1999)<br />
Sildenafil (Viagra) (Cunn<strong>in</strong>gham, 2001; Egan, 2000; Pomeranz, 2002)<br />
Lymphoma and sepsis (Guyer, 1990)<br />
Migra<strong>in</strong>e (Chiari, 1994)<br />
Postimmunization<br />
Radiation necrosis (Kawasaki, 1998; Parsons, 1994)<br />
Renal failure and uremia (Haider, 1993; Korzets, 1998; W<strong>in</strong>kelmayer, 2001)<br />
Smok<strong>in</strong>g (Chung, 1994; Johnson, 1994b; Talks, 1995)<br />
Trauma (Gadkari, 1990)<br />
After trans-Atlantic airplane journey (Kaiserman, 2002)<br />
Familial AION (Sadun, 1996)<br />
the fellow eye <strong>in</strong> 10 to 73% of cases (Boone, 1996). Beri et al evaluated 438 patients with<br />
AION (Beri, 1987); 388 had NA-AION and 50 had arteritic AION. The risk of bilateral<br />
<strong>in</strong>volvement for the arteritic form was 1.9 times the risk for NA-AION. At 3 years, Beri<br />
et al calculated an <strong>in</strong>cidence of bilateral NA-AION of 26% (Beri, 1987). In patients with<br />
bilateral disease, some authors have noted that the f<strong>in</strong>al outcome between eyes is<br />
similar for acuity, color vision, and visual fields (Boone, 1996). In another study, visual<br />
function <strong>in</strong> the second eye <strong>in</strong> patients with bilateral NA-AION correlated poorly with<br />
the first eye (WuDunn, 1997). In this study, older patients ( > 50 years) with bilateral
NA-AION reta<strong>in</strong>ed better visual function <strong>in</strong> the second eye, whereas <strong>in</strong> younger<br />
patients the extent of visual loss <strong>in</strong> the second eye could not be predicted based on<br />
the visual loss <strong>in</strong> the first eye. Kupersmith et al also reported poor correlation of visual<br />
acuity and field defects <strong>in</strong> the second eye compared to the first <strong>in</strong>volved eye<br />
(Kupersmith, 1997).<br />
The visual loss <strong>in</strong> NA-AION is usually acute and rema<strong>in</strong>s relatively static, but may<br />
spontaneously improve <strong>in</strong> up to 42.7% of patients (Aiello, 1992; Barrett, 1992; IONDT,<br />
1995; Movsas, 1991; Rizzo, 1991). In up to 25% of patients, visual loss may be<br />
progressive over several weeks. In our op<strong>in</strong>ion, gradual and progressive visual loss<br />
should prompt further evaluation, <strong>in</strong>clud<strong>in</strong>g neuroimag<strong>in</strong>g, to exclude other causes of a<br />
cont<strong>in</strong>u<strong>in</strong>g optic neuropathy (e.g., optic nerve sheath men<strong>in</strong>gioma).<br />
What Is the Evaluation and Treatment for<br />
AION?<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 79<br />
Table 4–5. <strong>Cl<strong>in</strong>ical</strong> Features Atypical for Nonarteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy<br />
Age younger than 40 years<br />
Bilateral simultaneous onset<br />
Visual field defect not consistent with an optic neuropathy (e.g., bitemporal hemianopsia,<br />
homonymous hemianopsia)<br />
Lack of optic disc edema <strong>in</strong> the acute phase<br />
Lack of relative afferent pupillary defect<br />
Large cup to disc ratio (Parsa, 1998)<br />
End-stage optic disc appearance of cupped disc (present <strong>in</strong> 2% of patients with nonarteritic-AION<br />
vs. 92% of patients with arteritic AION) (Danesh-Meyer, 2001)<br />
Lack of vasculopathic risk factors<br />
Presence of premonitory symptoms of transient visual loss (amaurosis fugax)<br />
Progression of visual loss beyond 2 to 4 weeks<br />
Recurrent episodes <strong>in</strong> the same eye<br />
<strong>An</strong>terior or posterior segment <strong>in</strong>flammation (e.g., vitreous cells)<br />
Patients with typical features of AION (e.g., acute onset, unilateral visual loss, ipsilateral<br />
optic disc edema, older=aged patient) do not require neuroimag<strong>in</strong>g (class II–III, level B).<br />
The major entity that must be excluded <strong>in</strong> AION is giant cell arteritis (GCA) (see<br />
Chapter 5). <strong>An</strong> erythrocyte sedimentation rate (and=or C-reactive prote<strong>in</strong>) and other<br />
appropriate evaluation for GCA should be considered <strong>in</strong> cases of AION <strong>in</strong> patients over<br />
age 50 (class II, level B). Patients with atypical features (Table 4–5) should be evaluated<br />
for other etiologies of an optic neuropathy (see Chapter 1).<br />
Further laboratory studies to <strong>in</strong>vestigate the presence of a hypercoagulable state<br />
could be considered <strong>in</strong> patients with NA-AION who do not have the typical risk factors,<br />
such as older age, diabetes, hypertension, or tobacco use, or <strong>in</strong> young patients with<br />
bilateral or recurrent attacks of NA-AION, but the data are conflict<strong>in</strong>g (class III, level C).<br />
Some authors have recommended that laboratory tests for a hypercoagulable state <strong>in</strong><br />
the follow<strong>in</strong>g patients: (1) young (less than age 45 years) patients with NA-AION;
80 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
(2) NA-AION without a small cup to disc ratio (‘‘disc at risk’’) <strong>in</strong> the fellow eye; (3)<br />
bilateral simultaneous NA-AION; (4) recurrent NA-AION <strong>in</strong> the same eye; (5) NA-<br />
AION <strong>in</strong> a patient with a previous history or family history of recurrent thrombotic<br />
events (Lee, 1999) (class III, level U). Hyperhomocyste<strong>in</strong>emia was discovered <strong>in</strong> two of<br />
12 nondiabetic patients with NA-AION before the age of 50 years (Kawasaki, 1999).<br />
Both of these two patients had experienced NA-AION <strong>in</strong> both eyes with recurrent<br />
episodes (class III).<br />
Unfortunately, although corticosteroids (systemic, retrobulbar, sub-Tenons), anticoagulation,<br />
dipyridamole, acetazolamide, hemodilution, vasodilators, vasopressors,<br />
atrop<strong>in</strong>e, norep<strong>in</strong>ephr<strong>in</strong>e, diphenylhydanto<strong>in</strong>, and hyperbaric oxygen have been tried<br />
<strong>in</strong> the past, there is no proven therapy for NA-AION (Arnold, 1996; Wolfe, 1993). The<br />
natural history of NA-AION <strong>in</strong> the past has been difficult to def<strong>in</strong>e. In the Ischemic<br />
Optic <strong>Neuro</strong>pathy Decompression Trial (IONDT), there was an unexpectedly high rate<br />
of spontaneous (three or more l<strong>in</strong>es from basel<strong>in</strong>e at 6 months) improvement of 42.7%<br />
(class I, level A). This rate is higher than that noted <strong>in</strong> the literature on AION before<br />
1989 (less than 10%). In the literature s<strong>in</strong>ce then, visual improvement rates as high as<br />
33% have been reported (Arnold, 1994b; Rizzo, 1991; Yee, 1993).<br />
We believe that any future treatments for NA-AION will have to prove better than the<br />
natural history data of the IONDT. Most previously published reports on treatment for<br />
NA-AION are limited by retrospective design, nonstandardized methods of data<br />
collection or measurement, small sample sizes, and variable (usually relatively short)<br />
lengths of follow-up.<br />
Medical control of underly<strong>in</strong>g hypertension, diabetes, and other presumed etiologic<br />
vasculopathic risk factors (such as smok<strong>in</strong>g cessation) has been recommended (Chung,<br />
1994; Hayreh, 1994b), but no well-controlled data on the efficacy of such measures <strong>in</strong><br />
reduc<strong>in</strong>g fellow-eye <strong>in</strong>volvement exist (class III, level C). In addition, overaggressive<br />
control of arterial hypertension may be potentially dangerous <strong>in</strong> patients <strong>in</strong> whom acute<br />
and=or nocturnal hypotension is an underly<strong>in</strong>g etiology for NA-AION (Hayreh, 1994b)<br />
(class III, level U). Patients with malignant hypertension <strong>in</strong> whom the blood pressure<br />
is lowered too rapidly may also be at risk for the precipitation of NA-AION <strong>in</strong> the<br />
fellow eye.<br />
Are Additional Studies (e.g., Non<strong>in</strong>vasive<br />
Carotid Doppler Studies, Cardiac Studies,<br />
<strong>Neuro</strong>imag<strong>in</strong>g) Warranted <strong>in</strong> Patients with<br />
NA-AION?<br />
Although Guyer et al reported a significantly higher <strong>in</strong>cidence of cerebrovascular and<br />
cardiovascular disease <strong>in</strong> 200 patients with idiopathic AION (Guyer, 1985, 1988),<br />
Hayreh et al have found no <strong>in</strong>creased risk for subsequent cerebrovascular or cardiovascular<br />
disease (Hayreh, 1994b). Some authors have found no <strong>in</strong>creased <strong>in</strong>cidence of<br />
generalized cerebral vascular disease on magnetic resonance imag<strong>in</strong>g (MRI) of the head<br />
<strong>in</strong> n<strong>in</strong>e patients with NA-AION, but Arnold et al reported an <strong>in</strong>creased number of<br />
central nervous system white matter lesions on bra<strong>in</strong> MRI <strong>in</strong> patients with NA-AION<br />
(Arnold, 1995). Fry et al found no significant difference <strong>in</strong> carotid stenosis <strong>in</strong> 15 patients<br />
with AION versus controls (Fry, 1993). Several authors have reported no significant
association between AION and extracranial carotid artery occlusive disease. AION has<br />
rarely been attributed to embolic disease (Horton, 1995). We do not perform additional<br />
non<strong>in</strong>vasive evaluation of the carotid or cardiac systems <strong>in</strong> patients with NA-AION<br />
unless there are other signs of carotid disease, such as ocular ischemic syndrome or<br />
ret<strong>in</strong>al emboli, or a history of transient or persistent focal neurologic deficits (Horton,<br />
1995) (class III, level U). We also consider MR angiography <strong>in</strong> patients with NA-AION<br />
with associated ipsilateral head or neck pa<strong>in</strong> to evaluate for carotid artery dissection<br />
(Biousse, 1998a,b). <strong>Neuro</strong>imag<strong>in</strong>g studies of the head are not <strong>in</strong>dicated <strong>in</strong> patients with<br />
typical unilateral NA-AION (Arnold, 1995) (class II–III, level B).<br />
Should the Patient with NA-AION Be Placed<br />
on Aspir<strong>in</strong> Therapy?<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 81<br />
Aspir<strong>in</strong> is often given to patients follow<strong>in</strong>g the development of NA-AION, but there<br />
does not seem to be any beneficial effect of treatment on eventual visual outcome<br />
(Botelho, 1996). Some authors, however, have suggested that aspir<strong>in</strong> therapy may<br />
reduce the risk of NA-AION <strong>in</strong> the fellow eye (Beck, 1997; Sanderson, 1995). Sanderson<br />
et al performed a retrospective review of 101 patients with AION for over 3 years<br />
(Sanderson, 1995). Fellow-eye <strong>in</strong>volvement occurred <strong>in</strong> 33 patients, of whom 23 did not<br />
take aspir<strong>in</strong> (compared with 47 patients on aspir<strong>in</strong> out of 68 patients without fellow eye<br />
<strong>in</strong>volvement). These authors estimated a threefold reduction of second eye <strong>in</strong>volvement<br />
(p ¼ 0.0005) <strong>in</strong> the aspir<strong>in</strong>-treated group and concluded that aspir<strong>in</strong> therapy significantly<br />
reduces the relative risk of NA-AION <strong>in</strong> the fellow eye. Beck reported on the results of a<br />
survey (270 of 350 neuro-ophthalmologist respondents) that among 5188 ophthalmologists,<br />
60% usually or always prescribed aspir<strong>in</strong> (usually 325 mg per day); 6%<br />
prescribed aspir<strong>in</strong> about half the time; and 34% occasionally or never prescribed<br />
aspir<strong>in</strong>. Among 582 neurologists, the percentages were 71%, 10%, and 19%, respectively<br />
(Beck, 1997). In a retrospective study of 431 patients, Beck et al found that the<br />
cumulative probability of NA-AION <strong>in</strong> the fellow eye was 7% <strong>in</strong> an aspir<strong>in</strong> group<br />
and 15% <strong>in</strong> a nonaspir<strong>in</strong> group and the 5-year cumulative probabilities were 17% and<br />
20%, respectively (Beck, 1997). This study thus suggests a possible short-term benefit of<br />
aspir<strong>in</strong> <strong>in</strong> reduc<strong>in</strong>g the risk of NA-AION <strong>in</strong> the fellow eye. Kupersmith et al found that<br />
aspir<strong>in</strong> taken two or more times per week decreased the <strong>in</strong>cidence (17.5% vs. 53.5%) of<br />
second eye <strong>in</strong>volvement <strong>in</strong> patients with unilateral NA-AION regardless of risk factors<br />
(Kupersmith, 1997). Salomon et al retrospectively evaluated 52 patients (Salomon,<br />
1999b). Second eye <strong>in</strong>volvement was noted <strong>in</strong> 8 of 16 patients (50%) who did not<br />
receive aspir<strong>in</strong>, <strong>in</strong> 3 of 8 patients (38%) who received 100 mg=day aspir<strong>in</strong>, and <strong>in</strong> only 5<br />
of 28 patients (18%) who received aspir<strong>in</strong> 325 mg=day. Moreover, the mean time to<br />
second eye <strong>in</strong>volvement was 63 months <strong>in</strong> patients who did not receive aspir<strong>in</strong>, versus<br />
156 months <strong>in</strong> patients who received aspir<strong>in</strong> 325 mg=day. The authors concluded that<br />
aspir<strong>in</strong> 325 mg=day may be effective <strong>in</strong> reduc<strong>in</strong>g the frequency of second eye <strong>in</strong>volvement<br />
<strong>in</strong> NA-AION (Salomon, 1999b). In light of the possible association between NA-<br />
AION and cerebrovascular and cardiac vasculopathic risk factors (e.g., hypertension,<br />
diabetes) and the recognized reduction <strong>in</strong> morbidity and mortality for patients with<br />
cerebrovascular disease and cardiac disease (e.g., myocardial <strong>in</strong>farction) (Roth, 1994)<br />
treated with aspir<strong>in</strong>, our current practice (until a prospective trial is performed) is to
82 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
offer oral aspir<strong>in</strong> therapy to patients (who have no contra<strong>in</strong>dications to aspir<strong>in</strong>) with<br />
NA-AION (class II–III, level U).<br />
Are There Other Treatments for NA-AION?<br />
A pilot cl<strong>in</strong>ical trial on the efficacy of levodopa <strong>in</strong> NA-AION was published with<br />
<strong>in</strong>terest<strong>in</strong>g results. Johnson et al reported a prospective, randomized, double-masked,<br />
placebo-controlled, cl<strong>in</strong>ical trial of 20 subjects with NA-AION of 30 months’ mean<br />
duration (Johnson, 1996a). Subjects were randomized to low-dose levodopa and<br />
carbidopa or placebo for 3 weeks. At 12 weeks, the levodopa group was provided a<br />
higher, conventional dose of levodopa and carbidopa for three additional weeks. At<br />
12 weeks, the levodopa group experienced a significant (p ¼ 0.16) mean difference <strong>in</strong><br />
improvement of visual acuity of 5.9 letters from the placebo group, and at 24 weeks the<br />
treatment effect rema<strong>in</strong>ed (p ¼ 0.36). There was a mean ga<strong>in</strong> of 7.5 letters <strong>in</strong> the<br />
levodopa group compared to the placebo group, and three subjects experienced a<br />
doubl<strong>in</strong>g of the visual angle, as denoted by a ga<strong>in</strong> of at least 15 letters. No significant<br />
improvement was noted for color vision or visual field (class II–III, level U).<br />
In a follow-up study, Johnson et al further studied the effect of levodopa on visual<br />
function <strong>in</strong> patients treated with<strong>in</strong> 45 days of onset of NA-AION (Johnson, 2000). In a<br />
nonrandomized, retrospective study <strong>in</strong>volv<strong>in</strong>g 37 patients, 18 were treated with<br />
S<strong>in</strong>emet 25–100 (100 mg levodopa=25 mg carbidopa), whereas 19 patients served as<br />
controls. The proportions of patients with worsened, unchanged, and improved visual<br />
acuity at 6 months were compared <strong>in</strong> the two groups. A higher proportion of the<br />
patients <strong>in</strong> the levodopa group had improved visual acuity with a correspond<strong>in</strong>g<br />
lower proportion hav<strong>in</strong>g worsened acuity as compared to control patients. Ten of the<br />
13 patients (76.9%) <strong>in</strong> the levodopa group with 20=40 visual acuity or worse at basel<strong>in</strong>e<br />
had improved visual acuity at 6 months, and none of the 18 patients had worsened<br />
acuity. In contrast, 3 of 10 control patients (30%) with 20=40 visual acuity or worse at<br />
basel<strong>in</strong>e had improved visual acuity at 6 months, and 3 of 19 control patients (15.8%)<br />
had worsened visual acuity. The proportion of patients with worsened, unchanged,<br />
and improved visual fields at 6 months was compared for the two groups and there<br />
was no significant difference. The authors concluded that patients treated with<br />
levodopa with<strong>in</strong> 45 days of onset of NA-AION were more likely to experience<br />
improvement and less likely to have worsened visual acuity than untreated patients<br />
(Johnson, 2000).<br />
Unfortunately, there are many flaws <strong>in</strong> this latter study, and the conclusions are<br />
controversial and may well be erroneous for the follow<strong>in</strong>g reasons (Cox, 2000) (class<br />
II–III, level U). Cox (2000) summarized the controversial po<strong>in</strong>ts:<br />
1. The study was retrospective, unplanned, nonrandomized, and based on a small<br />
sample size.<br />
2. The treatment and control groups were very different, ma<strong>in</strong>ly <strong>in</strong> basel<strong>in</strong>e visual<br />
functions. The control group actually had better mean acuities and mean field scores<br />
at basel<strong>in</strong>e and the imbalance between the groups at basel<strong>in</strong>e makes any results<br />
essentially un<strong>in</strong>terpretable.<br />
3. The study was not randomized, and selection and measurement bias may have been<br />
present.
4. The patients placed on the drug may have expected a better visual outcome and,<br />
thus, ‘‘tested better’’ than the nontreated group.<br />
5. The statistical analysis used was flawed.<br />
It is not the practice of the authors to recommend levodopa for NA-AION based on<br />
the available evidence (class II–III, level U).<br />
Does Optic Nerve Sheath Fenestration<br />
Improve Visual Outcomes <strong>in</strong> NA-AION?<br />
Initial reports of visual improvement follow<strong>in</strong>g optic nerve sheath fenestration (ONSF)<br />
for NA-AION were encourag<strong>in</strong>g, but anecdotal (Kelman, 1991; Manor, 1990; Sergott,<br />
1990; Spoor, 1991). Other reports followed with mixed results (Flaharty, 1993; Glaser,<br />
Table 4–6. Characteristics of Patients with Nonarteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy<br />
Eligible for the Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial<br />
420 patients (258 randomized; 162 not randomized)<br />
62% men; 95% white<br />
Mean age at onset 66.0 (peak age range 60–69)<br />
Hypertension 47%; diabetes 24%<br />
42% recalled onset of visual symptoms with<strong>in</strong> 2 hours of awaken<strong>in</strong>g<br />
Initial VA 20=20 to light perception (LP), with 49% patients better than 20=64, and 34% 20=200<br />
or worse<br />
Mean Westergan erythrocyte sedimentation rate (ESR) 18.4 with 9% greater than 40<br />
Nonrandomized patients were younger, 72% were male, and had lower prevalence of<br />
hypertension and diabetes<br />
45% of patients reported worsen<strong>in</strong>g (subjective) of vision between onset and basel<strong>in</strong>e exam<br />
29% of eligible patients with basel<strong>in</strong>e VA > 20=64 had documented progression to 20=64 or<br />
worse dur<strong>in</strong>g 30-day period<br />
15% of randomized patients smoked or discont<strong>in</strong>ued smok<strong>in</strong>g < 1 year before onset<br />
Earliest symptoms<br />
Intermittent blurr<strong>in</strong>g 5.0%<br />
Blurred vision 36.1%<br />
Scotoma 45.4%<br />
Complete loss of vision 3.8%<br />
Optic disc of affected eye<br />
Swollen 100.0%<br />
Diffuse 75.4%<br />
Focal 24.6%<br />
Disc or ret<strong>in</strong>al hemorrhage 71.8%<br />
Exudates 6.5%<br />
Abnormal ret<strong>in</strong>al vasculature 19.1%<br />
Optic disc nonstudy eye<br />
Pallor 22.5%<br />
Swollen 0.5%<br />
All percentages ¼ randomized þ nonrandomized patients.<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 83
84 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
1994; Hayreh, 1990a; Jablons, 1993; Kelman, 1991; McHenry, 1993a,b; Mutlukan, 1990;<br />
Sadun, 1993; Sergott, 1990; Spoor, 1991, 1993; Wall, 1991; Wilson, 1990; Yee, 1993).<br />
Subsequently, a well-designed, masked, prospective, randomized Ischemic Optic<br />
<strong>Neuro</strong>pathy Decompression Trial at 25 cl<strong>in</strong>ical centers was <strong>in</strong>itiated with the support<br />
of the National Eye Institute (Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial [IONDT]<br />
Research Group, 1995). The study <strong>in</strong>clusion criteria were as follows: cl<strong>in</strong>ical syndrome<br />
consistent with NA-AION (e.g., acute, unilateral visual loss, relative afferent pupillary<br />
defect, swollen optic nerve, etc.); age greater than 50 years; visual symptoms for less<br />
than 14 days from onset; and visual acuity of 20=64 or worse. Patients were randomly<br />
assigned to either ONSF (119 patients) or a control group (125 patients). Experienced<br />
Figure 4–1. Evaluation of anterior ischemic optic neuropathy.
protocol-certified study surgeons performed all the surgeries. The primary outcome<br />
measure was a three or more l<strong>in</strong>e improvement of visual acuity after 6 months, and<br />
visual field mean deviation on the Humphrey Field <strong>An</strong>alyzer (Program 24-2) was a<br />
secondary outcome measure. Recruitment was halted <strong>in</strong> September 1994 on the<br />
recommendation of the Data and Safety Monitor<strong>in</strong>g Committee. The cl<strong>in</strong>ical characteristics<br />
of the patients recruited are summarized <strong>in</strong> Table 4–6. After 6 months, 32.6% of the<br />
ONSF (surgery) group had improved three or more l<strong>in</strong>es of visual acuity compared with<br />
42.7% of the control group; but 23.9% of the ONSF group had lost three or more l<strong>in</strong>es of<br />
visual acuity compared with only 12.4% of the control group. Likewise, visual field data<br />
confirmed a lack of benefit for surgery. The 3-month, 12-month, and 24-month data<br />
confirmed the f<strong>in</strong>d<strong>in</strong>gs of the 6-month data (Ischemic Optic <strong>Neuro</strong>pathy Decompression<br />
Trial Research Group, 2000). In addition, there was no <strong>in</strong>dication of benefit from ONSF<br />
<strong>in</strong> the subgroup of patients with progressive visual loss. The authors concluded that<br />
‘‘ONSF is not effective and may be harmful <strong>in</strong> NA-AION’’ (IONDT, 1995), but were<br />
careful to state that they could ‘‘offer no recommendation regard<strong>in</strong>g the safety and<br />
efficacy of this surgery for other conditions’’ (IONDT, 1995). We agree that ONSF should<br />
not be performed for NA-AION (Beck, 1995; IONDT, 1995; Lessell, 1995; Smith, 1995)<br />
(class I, level A).<br />
Our approach to NA-AION is outl<strong>in</strong>ed <strong>in</strong> Figure 4–1.<br />
References<br />
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 85<br />
Abe H, Sakai T, Sawaguchi S, et al. (1992). Ischemic optic neuropathy <strong>in</strong> a female carrier with Fabry’s disease.<br />
Ophthalmologica 205:83–88.<br />
Acheson JF, Cockrell OC, Bentley CR, Sanders MD. (1993). Churg-Strauss vasculitis present<strong>in</strong>g with severe visual<br />
loss due to bilateral sequential optic neuropathy. Br J Ophthalmol 77:118–119.<br />
Aiello AL, Sadun AA, Feldon SE. (1992). Spontaneous improvement of progressive anterior ischemic optic<br />
neuropathy. Report of two cases. Arch Ophthalmol 110:1197–1199.<br />
Alexandrakis G, Lam BL. (1999). Bilateral posterior ischemic optic neuropathy after sp<strong>in</strong>al surgery. Am J<br />
Ophthalmol 127:354–355.<br />
Arnold AC, Badr MA, Hepler RS. (1996). Fluoresce<strong>in</strong> angiography <strong>in</strong> nonischemic optic disc edema. Arch<br />
Ophthalmol 114:293–298.<br />
Arnold AC, Hepler RS. (1994a). Fluoresce<strong>in</strong> angiography <strong>in</strong> acute nonarteritic anterior ischemic optic neuropathy.<br />
Am J Ophthalmol 117:222–230.<br />
Arnold AC, Hepler RS. (1994b). Natural history of nonarteritic anterior ischemic optic neuropathy. J <strong>Neuro</strong>ophthalmol<br />
14:66–69.<br />
Arnold AC, Hepler RS, Hamilton DR, Lufk<strong>in</strong> RB. (1995). Magnetic resonance imag<strong>in</strong>g of the bra<strong>in</strong> <strong>in</strong> nonarteritic<br />
anterior ischemic optic neuropathy. J <strong>Neuro</strong>-ophthalmol 15:158–160.<br />
Arnold AC, Hepler RS, Lieber M, Alexander JM. (1996). Hyperbaric oxygen therapy for nonarteritic anterior<br />
ischemic optic neuropathy. Am J Ophthalmol 122:535–541.<br />
Arnold AC, Petrovich M. (1997). Diabetic papillopathy: cl<strong>in</strong>ical features and fluoresce<strong>in</strong> angiographic evidence of<br />
optic disc ischemia. Presented at the 23rd annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology<br />
Society, Keystone, Colorado, February 9–13.<br />
Atmaca LS, Ozmert E. (1992). Optic neuropathy and central ret<strong>in</strong>al artery occlusion <strong>in</strong> a patient with herpes zoster<br />
ophthalmicus. <strong>An</strong>n Ophthalmol 24:50–53.<br />
Aziz A, Coneay MD, Robertson HJ, et al. (2000). Acute optic neuropathy and transverse myelopathy <strong>in</strong> patients<br />
with antiphospholipid antibody syndrome: favorable outcome after treatment with anticoagulants and<br />
glucocorticoids. Lupus 9:301–310.<br />
Barrett DA, Glaser JS, Schatz NJ, W<strong>in</strong>terkorn JMS. (1992). Spontaneous recovery of vision <strong>in</strong> progressive anterior<br />
ischemic optic neuropathy. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:219–225.<br />
Beck RW. (1992). Optic neuritis or anterior ischemic optic neuropathy? Arch Ophthalmol 110:1357.<br />
Beck RW. (1995). Optic nerve sheath fenestration for anterior ischemic optic neuropathy? The answer is <strong>in</strong><br />
(editorial). J <strong>Neuro</strong>-ophthalmol 15:61–62.
86 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Beck RW, Corbett JR, Thompson HS, et al. (1985). Decreased visual acuity from optic disc drusen. Arch Ophthalmol<br />
103:1155–1159.<br />
Beck RW, Hayreh SS, Podhajsky PA, Tan E-S, Moke PS. (1997). Aspir<strong>in</strong> therapy <strong>in</strong> nonarteritic anterior ischemic<br />
optic neuropathy. Am J Ophthalmol 123:212–217.<br />
Beri M, Klugman MR, Kohler JA, Hayreh SS. (1987). <strong>An</strong>terior ischemic optic neuropathy VII. Incidence of<br />
bilaterality and various <strong>in</strong>fluenc<strong>in</strong>g factors. Ophthalmology 94:1020–1028.<br />
Bertram B, Remky A, Arend O, et al. (1995). Prote<strong>in</strong> C, prote<strong>in</strong> S, and antithromb<strong>in</strong> III <strong>in</strong> acute ocular occlusive<br />
disease. German J Ophthalmol 4:332–335.<br />
Biousse V, Schaison M, Touboul P-J, et al. (1998a). Ischemic optic neuropathy associated with <strong>in</strong>ternal carotid<br />
artery dissection. Arch <strong>Neuro</strong>l 55:715–719.<br />
Biousse V, Touboul P-J, D’<strong>An</strong>glejan-Chatillon J, et al. (1998b). Ophthalmic manifestations of <strong>in</strong>ternal carotid<br />
dissection. Am J Ophthalmol 126:565–577.<br />
Boone MI, Massry GG, Frankel RA, Holds JB, Chung SM. (1996). Visual outcome <strong>in</strong> bilateral nonarteritic anterior<br />
ischemic optic neuropathy. Ophthalmology 103:1223–1228.<br />
Borruat FX, Herbort CP. (1992). Herpes zoster ophthalmicus. <strong>An</strong>terior ischemic optic neuropathy and acyclovir.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:37–40.<br />
Botelho PJ, Johnson LN, Arnold AC. (1996). The effect of aspir<strong>in</strong> on the visual outcome of nonarteritic anterior<br />
ischemic optic neuropathy. Am J Ophthalmol 121:450–451.<br />
Brown RH, Schauble JF, Miller NR. (1994). <strong>An</strong>emia and hypotension as contributors to perioperative loss of<br />
vision. <strong>An</strong>esthesiology 80:222–226.<br />
Burde RM. (1993). Optic disk risk factors for nonarteritic anterior ischemic optic neuropathy (review). Am J<br />
Ophthalmol 116:759–764.<br />
Cameron BD, Saffra NA, Strom<strong>in</strong>ger MB. (2001). Laser <strong>in</strong> situ keratomileusis-<strong>in</strong>duced optic neuropathy.<br />
Ophthalmology 108:660–665.<br />
Cheng MA, Sigurdson W, Tempelhoff R, Lauryssen C. (2000). Visual loss after sp<strong>in</strong>e surgery: a survey.<br />
<strong>Neuro</strong>surgery 46:625–631.<br />
Chiari M, Manzoni GC, Van de Geijn EJ. (1994). Ischemic optic neuropathy after sumatriptan <strong>in</strong> a migra<strong>in</strong>e with<br />
aura patient (letter). Headache 34:237–238.<br />
Chun DM, Lev<strong>in</strong> DK. (1997). Ischemic optic neuropathy after hemorrhage from a cornual ectopic gestation. Am J<br />
Obstet Gynecol 177:1550–1552.<br />
Chung SM, Gay CA, McCrary JA III. (1994). Nonarteritic ischemic optic neuropathy. The impact of tobacco use.<br />
Ophthalmology 101:779–782.<br />
Connolly SE, Gordon KB, Horton JC. (1994). Salvage of vision after hypotension-<strong>in</strong>duced ischemic optic<br />
neuropathy. Am J Ophthalmol 117:235–242.<br />
Coppetto JR, Greco TP. (1992). Autoimmune ischemic optic neuropathy associated with positive rheumatoid<br />
factor and transient nephrosis. <strong>An</strong>n Ophthalmol 24:434–438.<br />
Cornblath WT, Warren F, Tang R. (2002). Optic neuropathy after LASIK. Presented at the 28th annual meet<strong>in</strong>g of<br />
the North American <strong>Neuro</strong>-Ophthalmology Society, Copper Mounta<strong>in</strong>, Colorado, February 9–14.<br />
Cox TA, Beck RW, Ferris FL, Hayreh SS. (2000). Does Levopopa improve visual function <strong>in</strong> NAION?<br />
Ophthalmology 107:1431–1438.<br />
Cull<strong>in</strong>ane DC, Jenk<strong>in</strong>s JM, Reddy S, et al. (2000). <strong>An</strong>terior ischemic optic neuropathy: a complication after<br />
systemic <strong>in</strong>flammatory response syndrome. J Trauma Injury Infect Crit Care 48:381–386.<br />
Cunn<strong>in</strong>gham AV, Smith KH (2001). <strong>An</strong>terior ischemic optic neuropathy associated with Viagra. J <strong>Neuro</strong>-ophthalmol<br />
21:22–25.<br />
Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Sergott RC. (2001). The prevalence of cupp<strong>in</strong>g <strong>in</strong> end-stage arteritic and nonarteritic<br />
anterior ischemic optic neuropathy. Ophthalmology 108:593–598.<br />
Diegel JT. (1994). The ischemic optic neuropathy decompression trial (letter). Arch Ophthalmol 112:1275–1276.<br />
Dilger JA, Tetzlaff JE, Bell GR, et al. (1998). Ischaemic optic neuropathy after sp<strong>in</strong>al fusion. Can J <strong>An</strong>aesth<br />
45:63–66.<br />
Dosso A, Safran AB, Sunaric G, Burger A. (1994). <strong>An</strong>terior ischemic optic neuropathy <strong>in</strong> Graves’ disease.<br />
J <strong>Neuro</strong>-ophthalmol 14:170–174.<br />
Egan R, Pomeranz H. (2000). Sidenafil (Viagra) associated anterior ischemic optic neuropathy. Arch Ophthalmol<br />
118:291–292.<br />
Feldon SE. (1999). <strong>An</strong>terior ischemic optic neuropathy: trouble wait<strong>in</strong>g to happen. Ophthalmology 106:651–652.<br />
Fenton S, Fenton JE, Browne M, et al. (2001). Ischaemic optic neuropathy follow<strong>in</strong>g bilateral neck dissection.<br />
J Laryngol Otol 115:158–160.<br />
Fivgas GD, Newman NJ. (1999). <strong>An</strong>terior ischemic optic neuropathy follow<strong>in</strong>g the use of a nasal decongestant.<br />
Am J Ophthalmol 127:104–106.
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 87<br />
Flaharty PM, Sergott RC, Lieb W, Bosley TM, Sav<strong>in</strong>o PJ. (1993). Optic nerve sheath decompression may improve<br />
blood flow <strong>in</strong> anterior ischemic optic neuropathy. Ophthalmology 100:297–302.<br />
Friedland S, W<strong>in</strong>terkorn JM, Burde RM. (1996). Luxury perfusion follow<strong>in</strong>g anterior ischemic optic neuropathy.<br />
J <strong>Neuro</strong>-ophthalmol 16:163–171.<br />
Fry CL, Carter JE, Kanter MC, Tegeler CH, Tuley MR. (1993). <strong>An</strong>terior ischemic optic neuropathy is not associated<br />
with carotid artery atherosclerosis. Stroke 24:539–542.<br />
Gadkari SS, Ladi DS, Gupta S, et al. (1990). Traumatic ischaemic optic neuropathy (a case report). J Postgrad Med<br />
37:179–180.<br />
Galetta SL, Plock GL, Kushner MJ, Wyszynski RE, Brucker AJ. (1991). Ocular thrombosis associated with<br />
antiphospholipid antibodies. <strong>An</strong>n Ophthalmol 23:207–212.<br />
Gerl<strong>in</strong>g J, Jancknecht P, Kommerell G. (1998a). Orbital pa<strong>in</strong> <strong>in</strong> optic neuritis and anterior ischemic optic<br />
neuropathy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:93–99.<br />
Gerl<strong>in</strong>g J, Meyer JH, Kommerell G. (1998b). Visual field defects <strong>in</strong> optic neuritis and anterior ischemic optic<br />
neuropathy: dist<strong>in</strong>ctive features. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 236:188–192.<br />
Giuffre G. (1990). Hematological risk factors for anterior ischemic optic neuropathy. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
10:197–203.<br />
Glaser JS. (1991). Optic neuritis and ischemic neuropathy: What we thought we already knew. Arch Ophthalmol<br />
109:1666–1667.<br />
Glaser JS, Teimory M, Schatz NJ. (1994). Optic nerve sheath fenestration for progressive ischemic optic<br />
neuropathy. Arch Ophthalmol 112:1047–1050.<br />
Golnik KC, Newman SA. (1990). <strong>An</strong>terior ischemic optic neuropathy associated with macrocytic anemia. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 10:244–247.<br />
Good CD, Cassidy LM, Moseley IF, Sanders MD. (1999). Posterior optic nerve <strong>in</strong>farction after lower lid<br />
blepharoplast. J <strong>Neuro</strong>-Ophthalmol 19:176–179.<br />
Gordon RN, Burde RM, Slamovits T. (1997). Asymptomatic optic disc edema. J <strong>Neuro</strong>-Ophthalmol 17:29–32.<br />
Gotte K, Riedel F, Knorz MC, Hormann K. (2000). Delayed anterior ischemic optic neuropathy after neck<br />
dissection. Arch Otolaryngol Head Neck Surg 126:220–223.<br />
Grosvenor T, Mal<strong>in</strong>ovsky V, Gelv<strong>in</strong> J, Tonekaboni K. (1993). Diagnosis and management of temporal arteritis: a<br />
review and case report. Optom Vision Sci 70:771–777.<br />
Gupta A, Jalali S, Bansal RK, Grewal SP. (1990). <strong>An</strong>terior ischemic optic neuropathy and branch ret<strong>in</strong>al artery<br />
occlusion <strong>in</strong> cavernous s<strong>in</strong>us thrombosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:193–196.<br />
Guyer DR, Green WR, Schachat AP, Bastacky S, Miller NR. (1990). Bilateral ischemic optic neuropathy and ret<strong>in</strong>al<br />
vascular occlusions associated with lymphoma and sepsis. Ophthalmol 97:882–888.<br />
Guyer DR, Miller NR, Enger CL, F<strong>in</strong>e SL. (1985). The risk of cerebrovascular and cardiovascular disease <strong>in</strong><br />
patients with anterior ischemic optic neuropathy. Arch Ophthalmol 103:1136–1142.<br />
Guyer DR, Miller NR, Enger CL, F<strong>in</strong>e SL. (1988). Incidence of subcortical lesions not <strong>in</strong>creased <strong>in</strong><br />
nonarteritic anterior ischemic optic neuropathy on magnetic resonance imag<strong>in</strong>g (letter). Am J Ophthalmol<br />
105:324.<br />
Haider S, Asbury NJ, and Hamilton DV. (1993). Optic neuropathy <strong>in</strong> uraemic patients on dialysis. Eye 7:148–151.<br />
Hattenhauer MG, Leavitt JA, Hodge DO, et al. (1997). Incidence of nonarteritic ischemic optic neuropathy. Am J<br />
Ophthalmol 123:103–107.<br />
Hayreh SS. (1990a). The role of optic nerve sheath fenestration <strong>in</strong> management of anterior ischemic optic<br />
neuropathy (letter). Arch Ophthalmol 108:1063–1064.<br />
Hayreh SS. (1990b). <strong>An</strong>terior ischaemic optic neuropathy. Differentiation of arteritic from nonarteritic type and its<br />
management. Eye 4:25–41.<br />
Hayreh SS. (1996). Acute ischemic disorders of the optic nerve: pathogenesis, cl<strong>in</strong>ical manifestations, and<br />
management. Ophthalmol Cl<strong>in</strong> North Am 9:407–442.<br />
Hayreh SS. (1997a). <strong>An</strong>terior ischemic optic neuropathy. Cl<strong>in</strong> <strong>Neuro</strong>sci 4:251–263.<br />
Hayreh SS, Jonas JB (2001a). Optic disc morphology after arteritic anterior ischemic optic neuropathy. Ophthalmology<br />
108:1586–1594.<br />
Hayreh SS, Joos KM, Podhajsky PA, Long CR. (1994a). Systemic diseases associated with nonarteritic anterior<br />
ischemic optic neuropathy. Am J Ophthalmol 118:776–780.<br />
Hayreh SS, Piegors DJ, Heistad DD. (1997b). Seroton<strong>in</strong>-<strong>in</strong>duced constriction of ocular arteries <strong>in</strong> atherosclerotic<br />
monkeys: implications for ischemic disorders of the ret<strong>in</strong>a and optic nerve head. Arch Ophthalmol<br />
115:220–228.<br />
Hayreh SS, Podhajsky P, Zimmerman MB. (1999). Role of nocturnal arterial hypotension <strong>in</strong> optic nerve head<br />
ischemic disorders. Ophthalmologica 213:76–96.
88 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Hayreh SS, Podhajsky PS, Zimmerman B. (2001b). Ipsilateral recurrence of nonarteritic anterior ischemic optic<br />
neuropathy. Am J Ophthalmol 132:734–742.<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1997c). Nonarteritic anterior ischemic optic neuropathy: time of onset<br />
of visual loss. Am J Ophthalmol 124:641–647.<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1998). Occult giant cell arteritis: ocular manifestations. Am J Ophthalmol<br />
125:521–526.<br />
Hayreh SS, Zimmerman BM, Podhajsky P, Alward WLM. (1994b). Nocturnal arterial hypotension and its role <strong>in</strong><br />
optic nerve head and ocular ischemic disorders. Am J Ophthalmol 117:603–624.<br />
Horowitz J, Melamud A, Sela L, et al. (2001). Internal carotid artery hypoplasia present<strong>in</strong>g as anterior ischemic<br />
optic neuropathy. Am J Ophthalmol 131:673–674.<br />
Horton JC. (1995). Embolic cilioret<strong>in</strong>al artery occlusion with atherosclerosis of the ipsilateral carotid artery. Ret<strong>in</strong>a<br />
15:441–444.<br />
Inoue M, Tsukahara Y. (1997). Vascular optic neuropathy <strong>in</strong> diabetes mellitus. Jpn J Ophthalmol 41:328–331.<br />
Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial Research Group (IONDT). (1995). Optic nerve decompression<br />
for nonarteritic anterior ischemic optic neuropathy (AION) is not effective and may be harmful. JAMA<br />
273:625–632.<br />
Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial Research Group. (IONDT). (1996). Characteristics of patients<br />
with nonarteritic anterior ischemic optic neuropathy eligible for the Ischemic optic neuropathy decompression<br />
trial. Arch Ophthalmol 114:1366–1374.<br />
Ischemic Optic <strong>Neuro</strong>pathy Decompression Trial Research Group. (2000). Ischemic Optic <strong>Neuro</strong>pathy Decompression<br />
Trial. Twenty-four month update. Arch Ophthalmol 118:793–798.<br />
Jablons MM, Glaser JS, Schatz NJ, et al. (1993). Optic nerve sheath fenestration for treatment of progressive<br />
ischemic optic neuropathy: results <strong>in</strong> 26 patients. Arch Ophthalmol 111:84–87.<br />
Jackson TL, Farmer CKT, K<strong>in</strong>gswood C, Vickers S. (1999). Hypotensive ischemic optic neuropathy and peritoneal<br />
dialysis. Am J Ophthalmol 128:109–111.<br />
Jacobson DM, Vierkant RA, Belongia EA. (1997). Nonarteritic anterior ischemic optic neuropathy. A case-control<br />
study of potential risk factors. Arch Ophthalmol 115:1403–1407.<br />
Janicki PK, Pai R, Kelly Wright J, et al. (2001). Ischemic optic neuropathy after liver transplantation. <strong>An</strong>esthesiology<br />
94:361–363.<br />
Johnson LN, Arnold AC. (1994a). Incidence of non-arteritic anterior ischemic optic neuropathy: population based<br />
study <strong>in</strong> the state of Missouri and Los <strong>An</strong>geles County, California. J <strong>Neuro</strong>-ophthalmol 14:38–44.<br />
Johnson LN, Botelho PJ, Kuo HC. (1994b). Is smok<strong>in</strong>g a risk factor for NAAION? (letter). Ophthalmology<br />
101:1322–1324.<br />
Johnson LN, Gould TJ, Krohel GB. (1996a). Effect of levodopa and carbidopa on recovery of visual function <strong>in</strong><br />
patients with nonarteritic anterior ischemic optic neuropathy of longer than six months duration. Am J<br />
Ophthalmol 121:77–83.<br />
Johnson LN, Guy ME, Krohel GB, Madsen RW. (2000). Levodopa may improve vision loss <strong>in</strong> recent-onset,<br />
nonarteritic anterior ischemic optic neuropathy. Ophthalmology 107:521–526.<br />
Johnson LN, Krohel GB, Allen SD, Mozayeni R. (1996b). Recurrent herpes labialis as a potential risk factor for<br />
nonarteritic anterior ischemic optic neuropathy. J Natl Med Assoc 88:369–373.<br />
Johnson LN, Kuo HC, Arnold AC. (1993). HLA-A29 as a potential risk factor for nonarteritic anterior ischemic<br />
optic neuropathy (letter). Am J Ophthalmol 115:540–542.<br />
Kacer B, Hattenbach LO, Horle S, et al. (2001). Central ret<strong>in</strong>al ve<strong>in</strong> occlusion and nonarteritic ischemic optic<br />
neuropathy <strong>in</strong> 2 patients with mild iron deficiency anemia. Ophthalmologica 215:128–131.<br />
Kaiser HJ, Flammer J, Messerli J. (1996). Vasospasm: a risk factor for non-arteritic anterior ischemic optic<br />
neuropathy? <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:5–10.<br />
Kaiserman I, Frucht-Pery J. (2002). <strong>An</strong>terior ischemic optic neuropathy after a trans-Atlantic airplane journey. Am J<br />
Ophthalmol 133:581–583.<br />
Kalenak JW, Kosmorsky GS, Rockwood EJ. (1991). Nonarteritic anterior ischemic optic neuropathy and<br />
<strong>in</strong>traocular pressure. Arch Ophthalmol 109:660–661.<br />
Kattah JC, Chrousos GA, Katz PA, McCasland B, Kolsky MP. (1994). <strong>An</strong>terior ischemic optic neuropathy <strong>in</strong><br />
Churg-Strauss syndrome. <strong>Neuro</strong>logy 44:2200–2202.<br />
Katz B. (1990a). Disc swell<strong>in</strong>g <strong>in</strong> an adult diabetic patient. Surv Ophthalmol 25:158–163.<br />
Katz B. (1992). <strong>An</strong>terior ischemic optic neuropathy and <strong>in</strong>traocular pressure (letter). Arch Ophthalmol 110:596–597.<br />
Katz B, Spencer WH. (1993). Hyperopia as a risk factor for nonarteritic anterior ischemic optic neuropathy. Am J<br />
Ophthalmol 116:754–758.<br />
Katz B, We<strong>in</strong>reb RN, Wheeler DT, Klauber MR. (1990b). <strong>An</strong>terior ischemic optic neuropathy and <strong>in</strong>traocular<br />
pressure. Br J Ophthalmol 74:99–102.
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 89<br />
Katz DM, Trobe JD, Cornblath WT, Kl<strong>in</strong>e LB. (1994). Ischemic optic neuropathy after lumbar sp<strong>in</strong>e surgery. Arch<br />
Ophthalmol 112:925–931.<br />
Kawasaki A, Purv<strong>in</strong> VA, Burgett RA. (1999). Hyperhomocyste<strong>in</strong>aemia <strong>in</strong> young patients with non-arteritic<br />
anterior ischaemic optic neuropathy. Br J Ophthalmol 83:1287–1290.<br />
Kawasaki A, Purv<strong>in</strong> VA, Tang R. (1998). Bilateral anterior ischemic optic neuropathy follow<strong>in</strong>g <strong>in</strong>fluenza<br />
vacc<strong>in</strong>ation. J <strong>Neuro</strong>-ophthalmol 18:56–59.<br />
Kay MC. (1991). Ischemic optic neuropathy. <strong>Neuro</strong>l Cl<strong>in</strong> 9:115–129.<br />
Keely KA, Yip B. (1997). Diabetic papillopathy: two case reports <strong>in</strong> <strong>in</strong>dividuals with adult onset diabetes mellitus.<br />
J Am Optom Assoc 68:595–603.<br />
Kelman SE, Elman MJ. (1991). Optic nerve sheath decompression for nonarteritic ischemic optic neuropathy<br />
improves multiple visual function measurements. Arch Ophthalmol 109:667–671.<br />
Kerty E. (1999). The <strong>ophthalmology</strong> of <strong>in</strong>ternal carotid artery dissection. Acta Ophthalmol Scand 77:418–421.<br />
Killer HE, Huber A, Portman C, et al. (2000). Bilateral non-arteritic anterior ischemic optic neuropathy <strong>in</strong> a patient<br />
with autoimmune thrombocytopenia. Eur J Ophthalmol 10:180–182.<br />
Kirkali P, Kansu T. (1990). A case of unilateral posterior ischemic optic neuropathy after radical neck dissection.<br />
<strong>An</strong>n Ophthalmol 22:297–298.<br />
Korzets Z, Zeltzer E, Rathaus M, et al. (1998). Uremic optic neuropathy. A uremic manifestation mandat<strong>in</strong>g<br />
dialysis. Am J Nephrol 18:240–242.<br />
Kosmorsky G, Straga J, Knight C, et al. (1998). The role of transcranial Doppler <strong>in</strong> nonarteritic ischemic optic<br />
neuropathy. Am J Ophthalmol 126:288–290.<br />
Kothe AC, Flanagan J, Trev<strong>in</strong>o RC. (1990). True posterior ischemic optic neuropathy associated with herpes zoster<br />
ophthalmicus. Optom Vis Sci 67:845–849.<br />
Kupersmith MJ, Frohman LP, Sanderson MC, et al. (1997). Aspir<strong>in</strong> reduces the <strong>in</strong>cidence of second eye NAION: a<br />
retrospective study. J <strong>Neuro</strong>-ophthalmol 17:250–253.<br />
Landau K, W<strong>in</strong>terkorn JMS, Mailloux LU, Vetter W, Napolitano B. (1996). 24-hour blood pressure monitor<strong>in</strong>g <strong>in</strong><br />
patients with anterior ischemic optic neuropathy. Arch Ophthalmol 114:570–575.<br />
Lav<strong>in</strong> PJ. (1994). Optic disk risk factors for nonarteritic anterior ischemic optic neuropathy (letter). Am J<br />
Ophthalmol 117:822.<br />
Lee AG. (1995a). Reversible loss of vision due to posterior ischemic optic neuropathy. Can J Ophthalmol 30:327–329.<br />
Lee AG. (1995b). Ischemic optic neuropathy follow<strong>in</strong>g lumbar sp<strong>in</strong>e surgery. J <strong>Neuro</strong>surg 83:348–349.<br />
Lee AG. (1999). Prothrombotic and vascular risk factors <strong>in</strong> nonarteritic anterior ischemic optic neuropathy.<br />
Ophthalmology 106:2231.<br />
Lee AG, Brazis PW, Miller NR. (1996). Posterior ischemic optic neuropathy associated with migra<strong>in</strong>e. Headache<br />
36:506–509.<br />
Lee AG, Kohnen T, Ebner R, et al. (2000). Optic neuropathy associated with laser <strong>in</strong> situ keratomileusis. J Cataract<br />
Refract Surg 26:1581–1584.<br />
Lee AG, Lyle C, Tang R, et al. (2002). Optic nerve head drusen and ischemic optic neuropathy. Presented at the<br />
28th annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society. Copper Mounta<strong>in</strong>, Colorado,<br />
February 9–14.<br />
Lessell S. (1995). Surgery for ischemic optic neuropathy (editorial). Arch Ophthalmol 113:273–274.<br />
Lessell S. (1999). Nonarteritic anterior ischemic optic neuropathy. Enigma variations. Arch Ophthalmol 117:386–388.<br />
Lexa FJ, Galetta SL, Yousem DM, et al. (1993). Herpes zoster ophthalmicus with orbital pseudotumor syndrome<br />
complicated by optic nerve <strong>in</strong>farction and cerebral granulomatous angiitis: MR-pathologic correlation. AJ<br />
NR 14:185–190.<br />
Liew SC, Mitchell P. (1999). <strong>An</strong>terior ischaemic optic neuropathy <strong>in</strong> a patient with optic disc drusen. Aust N Z J<br />
Ophthalmol 27:157–160.<br />
Loftman BA, Shapiro J. (1996). Ischemic optic neuropathy (letter). J <strong>Neuro</strong>surg 84:306.<br />
Lund PE, Madsen K. (1994). Bilateral bl<strong>in</strong>dness after cardiopulmonary bypass. J Cardiothorac Vasc <strong>An</strong>esth<br />
8:448–450.<br />
Luscavage LE, Volpe NJ, Liss R (2001). Posterior ischemic optic neuropathy after uncomplicated cataract<br />
extraction. Am J Ophthalmol 132:408–409.<br />
Manor RS. (1990). Nonarteritic ischemic optic neuropathy <strong>in</strong> identical female tw<strong>in</strong>s: improvement of visual<br />
outcome <strong>in</strong> one by optic nerve decompression. (letter). Arch Ophthalmol 108:1067–1068.<br />
Mäntyjärvi M, Tuppura<strong>in</strong>en K, Ikäheimo K. (1998). Ocular side effects of Amiodarone. Surv Ophthalmol<br />
42:360–366.<br />
Marks SC, Jaques DA, Hirata RM, Saunders JR. (1990). Bl<strong>in</strong>dness follow<strong>in</strong>g bilateral radical neck dissection. Head<br />
Neck 12:342–345.
90 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Massry GG, Chung SM, Selhorst JB. (1995). Optic neuropathy, headache, and diplopia with MRI suggestive of<br />
cerebral arteritis <strong>in</strong> relaps<strong>in</strong>g polychondritis. J <strong>Neuro</strong>-ophthalmol 15:171–175.<br />
McCulley TJ, Lam BL, Feuer WJ. (2001). Incidence of nonarteritic anterior ischemic optic neuropathy associated<br />
with cataract extraction. Ophthalmology 108:1275–1278.<br />
McHenry JG, Spoor TC. (1993a). Optic nerve sheath fenestration for treatment of progressive ischemic optic<br />
neuropathy. Arch Ophthalmol 111:1601–1602, 1602–1603.<br />
McHenry JG, Spoor TC. (1993b). The efficacy of optic nerve sheath fenestration for anterior ischemic optic<br />
neuropathy and other optic neuropathies (letter). Am J Ophthalmol 116:254–256.<br />
Mochizuki A, Hayashi A, Hisahara S, Shoji S. (2000). Steroid-responsive Devic’s variant <strong>in</strong> Sjögren’s syndrome.<br />
<strong>Neuro</strong>logy 54:1319–1392.<br />
Mokri B, Silbert PL, Shiev<strong>in</strong>k WI, Piepgras DG. (1996). Cranial nerve palsy <strong>in</strong> spontaneous dissection of the<br />
extracranial <strong>in</strong>ternal carotid artery. <strong>Neuro</strong>logy 46:356–359.<br />
Moody TA, Irv<strong>in</strong>e AR, Cahn PH, Susac JO, Horton JC. (1993). Sudden visual field constriction associated with<br />
optic disc drusen. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:8–13.<br />
Moro F, Doro D, Mantovani E, Sala M. (1990). Ischemic optic neuropathies. Metab Pediatr Syst Ophthalmol 13:75–78.<br />
Moster ML, Katz JB, Sedwick LA. (1998). Visual loss after coronary bypass surgery. Surv Ophthalmol 42:453–457.<br />
Movsas T, Kelman SE, Elman MJ, Miller NR, Dickers<strong>in</strong> K, M<strong>in</strong> Y-I. (1991). The natural course of non-arteritic<br />
ischemic optic neuropathy (abstract). Invest Ophthalmol Vis Sci 42:951.<br />
Mutlukan E, Cullen JF. (1990). Can empty sella syndrome be mistaken for a progressive form of nonarteritic<br />
ischemic optic neuropathy? (letter). Arch Ophthalmol 108:1066–1067.<br />
Myers MA, Hamilton SR, Bogosian AJ, et al. (1997). Visual loss as a complication of sp<strong>in</strong>e surgery: a review of 37<br />
cases. Sp<strong>in</strong>e 22:1325–1329.<br />
Nawa Y, Jaques JD, Miller NR, et al. (1992). Bilateral posterior optic neuropathy after bilateral radical neck<br />
dissection and hypotension. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 230:301–308.<br />
Ohte A, Kimura T, Kimura W, et al. (1995). A case of optic disc <strong>in</strong>farction due to lupus anticoagulant. Folia<br />
Ophthalmol Jpn 46:783–787.<br />
Parsa CF, Muci-Mendoza R, Hoyt WF. (1998). <strong>An</strong>terior ischemic optic neuropathy <strong>in</strong> a disc with a cup. <strong>An</strong><br />
exception to the rule. J <strong>Neuro</strong>-ophthalmol 18:169–170.<br />
Parsons JT, Bova FL, Fitzgerald CR, Mendenhall WM, Million RR. (1994). Radiation optic neuropathy after<br />
megavoltage external-beam irradiation: analysis of time-dose factors. Int J Radiat Oncol Biol Phys 30:755–763.<br />
Perez-Santonja JJ, Bueno JL, Meza J, Garcia-Sandoval B, Serrano JM, Zato MA. (1993). Ischemic optic neuropathy<br />
after <strong>in</strong>traocular lens implantation to correct high myopia <strong>in</strong> a phakic patient. J Cataract Refract Surg<br />
19:651–654.<br />
Perlman JI, Forman S, Gonzalez ER. (1994). Retrobulbar ischemic optic neuropathy associated with sickle cell<br />
disease. J <strong>Neuro</strong>-ophthalmol 14:45–48.<br />
Pianka P, Almog Y, Man O, et al. (2000). Hyperhomocyst<strong>in</strong>emia <strong>in</strong> patients with nonarteritic anterior ischemic<br />
optic neuropathy, central ret<strong>in</strong>al artery occlusion, and central ret<strong>in</strong>al ve<strong>in</strong> occlusion. Ophthalmology<br />
107:1588–1592.<br />
Pomeranz HD, Smith KH, Hart WM Jr, Egan RA. (2002). Sildenafil-associated nonarteritic anterior ischemic optic<br />
neuropathy. Ophthalmology 109:584–587.<br />
Purv<strong>in</strong> VA. (1995). <strong>An</strong>terior ischemic optic neuropathy secondary to <strong>in</strong>terferon alpha. Arch Ophthalmol<br />
113:1041–1044.<br />
Regillo CD, Brown GC, Sav<strong>in</strong>o PJ, et al. (1995). Diabetic papillopathy. Patient characteristics and fundus f<strong>in</strong>d<strong>in</strong>gs.<br />
Arch Ophthalmol 113:889–895.<br />
Re<strong>in</strong>o S, Munoz-Rodriguez FJ, Cervera R, et al. (1997). Optic neuropathy <strong>in</strong> the primary antiphospholipid<br />
syndrome: report of a case and review of the literature. Cl<strong>in</strong> Rheumatol 16:629–631.<br />
Remigio D, Wertenbaker C, Katz DM. (2000). Post-operative bilateral vision loss. Surv Ophthalmol 44:426–432.<br />
R<strong>in</strong>aldi G, Pastori G, Ammirati M, Bellavitis A. (1990). Considerations upon a non-typical anterior ischemic optic<br />
neuropathy. Metab Pediatr Syst Ophthalmol 13:92–95.<br />
Rivk<strong>in</strong> MJ, Hedges TR III, Logigian EL. (1990). Carotid dissection present<strong>in</strong>g as posterior ischemic optic<br />
neuropathy. <strong>Neuro</strong>logy 40:1469.<br />
Rizzo JF III, Lessell S. (1991). Optic neuritis and ischemic optic neuropathy: overlapp<strong>in</strong>g cl<strong>in</strong>ical profiles. Arch<br />
Ophthalmol 109:1668–1672.<br />
Rosler SH, Conway MD, <strong>An</strong>aya JM, et al. (1995). Ischemic optic neuropathy and high-level anticardiolip<strong>in</strong><br />
antibodies <strong>in</strong> primary Sjogren’s syndrome. Lupus 4:155–157.<br />
Roth GJ, Calverly DC. (1994). Aspir<strong>in</strong>, platelets, and thrombosis: theory and practice. Blood 83:885–898.<br />
Roth S, Nunez R, Schreider BD. (1997). Unexpla<strong>in</strong>ed visual loss after lumbar sp<strong>in</strong>al fusion. J <strong>Neuro</strong>surg <strong>An</strong>esthesiol<br />
9:346–348.
Nonarteritic Ischemic Optic <strong>Neuro</strong>pathy 91<br />
Sadda SR, Nee M, Miller NR, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> spectrum of posterior ischemic optic neuropathy. Am J<br />
Ophthalmol 132:743–750.<br />
Sadun AA. (1993). The efficacy of optic nerve sheath decompression for anterior ischemic optic neuropathy and<br />
other optic neuropathies. Am J Ophthalmol 115:384–389.<br />
Sadun F, Wang M, Lev<strong>in</strong> LB, Feldon S. (1996). Familial nonarteritic ischemic optic neuropathy. Presented at the<br />
meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society.<br />
Salomon O, Huna-Brown R, Kurtz S, et al. (1999a). <strong>An</strong>alysis of prothrombotic and vascular factors <strong>in</strong> patients with<br />
nonarteritic anterior ischemic optic neuropathy. Ophthalmology 106:739–742.<br />
Salomon O, Huna-Baron R, Ste<strong>in</strong>berg DM, et al. (1999b). Role of aspir<strong>in</strong> <strong>in</strong> reduc<strong>in</strong>g the frequency of second eye<br />
<strong>in</strong>volvement <strong>in</strong> patients with non-arteritic anterior ischaemic optic neuropathy. Eye 13:357–359.<br />
Sanderson M, Kupersmith M, Frohman L, et al. (1995). Aspir<strong>in</strong> reduces anterior ischemic optic neuropathy <strong>in</strong> the<br />
second eye. ARVO abstracts. Invest Ophthalmol Vis Sci 36:S196.<br />
Sav<strong>in</strong>o PJ, Burde RM, Mills RP. (1990). Visual loss follow<strong>in</strong>g <strong>in</strong>tranasal anesthetic <strong>in</strong>jection. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
10:140–144.<br />
Sawle GV, James CB, Ross-Russell RW. (1990). The natural history of nonarteritic anterior ischemic optic<br />
neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 53:830–833.<br />
Schmidt MH, Fox AJ, Nicolle DA. (1997). Bilateral anterior ischemic optic neuropathy as a presentation of<br />
Takayasu’s disease. J <strong>Neuro</strong>-ophthalmol 17:156–161.<br />
Schnobel GA, Schmidbauer M, Millesi W, Undt G. (1995). Posterior ischemic optic neuropathy follow<strong>in</strong>g bilateral<br />
radical neck dissection. Int J Oral Maxillofac Surg 24:283–287.<br />
Schönhofer PS, Werner B, Tröger U. (1997). Ocular damage associated with proton pump <strong>in</strong>hibitors. Br Med J<br />
314:1805.<br />
Segato T, Peirmarochhi S, Midena E. (1990). The role of fluoresce<strong>in</strong> angiography <strong>in</strong> the <strong>in</strong>terpretation of optic<br />
nerve head diseases. Metab Pediatr Syst Ophthalmol 13:111–114.<br />
Sehgal M, Swanson JW, DeRemee RA, Colby TV. (1995). <strong>Neuro</strong>logic manifestations of Churg-Strauss syndrome.<br />
Mayo Cl<strong>in</strong> Proc 70:337–341.<br />
Sergott RC, Sav<strong>in</strong>o PJ, Bosley TM. (1990). Optic nerve sheath decompression: a cl<strong>in</strong>ical review and proposed<br />
pathophysiologic mechanism. Aust N Z J Ophthalmol 18:365–373.<br />
Shaked G, Gavriel A, Roy-Shapira A. (1998). <strong>An</strong>terior ischemic optic neuropathy after hemorrhagic shock. J Trauma<br />
44:923–925.<br />
Shapira OM, Kimmel WA, L<strong>in</strong>dsey PS, Shahian DM. (1996). <strong>An</strong>terior ischemic optic neuropathy after open heart<br />
operations. <strong>An</strong>n Thoracic Surg 61:660–666.<br />
Siatkowski RM, Scott IU, Verm AM, et al. (2001). Optic neuropathy and chiasmopathy <strong>in</strong> the diagnosis of systemic<br />
lupus erythematosus. J <strong>Neuro</strong>-<strong>ophthalmology</strong> 21:193–198.<br />
Slav<strong>in</strong> ML, Margulis M. (2001). <strong>An</strong>terior ischemic optic neuropathy follow<strong>in</strong>g acute angle-closure glaucoma. Arch<br />
Ophthalmol 119:1215.<br />
Smith DB. (1995). Ischemic optic neuropathy decompression trial (letter). JAMA 274:612.<br />
Smith FP. (1996). Ischemic optic neuropathy (letter). J <strong>Neuro</strong>surg 84:149–150.<br />
Spoor TC, McHenry JG, Lau-Sickon L. (1993). Progressive and static nonarteritic ischemic optic neuropathy<br />
treated by optic nerve sheath decompression. Ophthalmology 100:306–311.<br />
Spoor TC, Wilk<strong>in</strong>son MJ, Ramocki JM. (1991). Optic nerve sheath decompression for the treatment of progressive<br />
nonarteritic ischemic optic neuropathy. Am J Ophthalmol 111:724–728.<br />
Sr<strong>in</strong>ivasan S, Fern A, Watson WH, McColl MD. (2001). Reversal of nonarteritic anterior ischemic optic neuropathy<br />
associated with coexist<strong>in</strong>g primary antiphospholipid syndrome and factor V Leiden mutation. Am J<br />
Ophthalmol 131:671–673.<br />
Stevens WR, Glazer PA, Kelley SD, et al. (1997). Ophthalmic complications after sp<strong>in</strong>al surgery. Sp<strong>in</strong>e 22:1319–1324.<br />
Strome SE, Hill JS, Burnst<strong>in</strong>e MA, et al. (1997). <strong>An</strong>terior ischemic optic neuropathy follow<strong>in</strong>g neck dissection. Head<br />
& Neck 19:148–152.<br />
Swartz NG, Beck RW, Sav<strong>in</strong>o RC, et al. (1995). Pa<strong>in</strong> <strong>in</strong> anterior ischemic optic neuropathy. J <strong>Neuro</strong>-ophthalmol<br />
15:9–10.<br />
Talks SJ, Chong NHV, Gibson JM, et al. (1995). Fibr<strong>in</strong>ogen, cholesterol, and smok<strong>in</strong>g as risk factors for nonarteritic<br />
anterior ischemic optic neuropathy. Eye 9:85.<br />
Tang RA. (1995). Interferon: friend or foe? Arch Ophthalmol 113:987.<br />
Teshome T and Alemayehu W. (1999). Loss of vision from distant haemorrhage: report of four cases. East Afr Med J<br />
76:706–708.<br />
Tham M-B, Cunn<strong>in</strong>gham ET Jr. (2001). <strong>An</strong>terior ischaemic optic neuropathy <strong>in</strong> a patient with HLA-B27 associated<br />
anterior uveitis and ankylos<strong>in</strong>g spondylitis. Br J Ophthalmol 85:754.
92 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Tomsak RL, Zakov ZN. (1998). Nonarteritic anterior ischemic optic neuropathy with macular edema. Visual<br />
improvement and fluoresce<strong>in</strong> angiographic characteristics. J <strong>Neuro</strong>-ophthalmol 18:166–168.<br />
Vaphiades MS, Regillo CD, Arnold AC. (2002). The disk edema dilemma. Surv Ophthalmol 47:183–188.<br />
Vitali C, Genovesi-Ebert F, Romani A, et al. (1996). Ophthalmological and neuro-ophthalmological <strong>in</strong>volvement <strong>in</strong><br />
Churg-Strauss syndrome: a case report. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 234:404–408.<br />
Wall M, Newman SA. (1991). Optic nerve sheath decompression for the treatment of progressive nonarteritic<br />
ischemic optic neuropathy (letter). Am J Ophthalmol 112:741–742.<br />
Wang MY, Sadun F, Lev<strong>in</strong> LB, et al. (1999). Occurrence of familial nonarteritic anterior ischemic optic neuropathy<br />
<strong>in</strong> a case series. J <strong>Neuro</strong>-ophthalmol 19:144–147.<br />
Warner JEA, Lessell S, Rizzo JF III, Newman NJ. (1997). Does optic disc appearance dist<strong>in</strong>guish ischemic optic<br />
neuropathy from optic neuritis? Arch Ophthalmol 115:1408–1410.<br />
Weger M, Stranger O, Deutschmann H, et al. (2001). Hyperhomocyst(e)<strong>in</strong>aemia, but not MTHFR C677T mutation,<br />
as a risk factor for non-arteritic ischaemic optic neuropathy. Br J Ophthalmology 85:803–806.<br />
Weger M, Haas A, Stanger O, et al. (2002). Chlamydia pneumoniae seropositivity and the risk of nonarteritic<br />
ischemic optic neuropathy. Ophthalmology 109:749–752.<br />
Williams GC, Lee AG, Adler HL, et al. (1999). Bilateral anterior ischemic optic neuropathy and branch ret<strong>in</strong>al<br />
artery occlusion after radical prostatectomy. J Urol 162:1384–1385.<br />
Williams EL, Hart WM Jr, Tempelhoff R. (1995). Postoperative ischemic optic neuropathy. <strong>An</strong>esth <strong>An</strong>alg<br />
80:1018–1029.<br />
Wilson JF, Freeman SB, Breene DP. (1991). <strong>An</strong>terior ischemic optic neuropathy caus<strong>in</strong>g bl<strong>in</strong>dness <strong>in</strong> the head and<br />
neck surgery patient. Arch Otolaryngol Head Neck Surg 117:1304–1306.<br />
Wilson WB. (1990). Does optic nerve sheath decompression help progressive ischemic optic neuropathy? (letter).<br />
Arch Ophthalmol 108:1065–1066.<br />
W<strong>in</strong>kelmayer WC, Eigner M, Berger O, et al. (2001). Optic neuropathy <strong>in</strong> uremia: an <strong>in</strong>terdiscipl<strong>in</strong>ary emergency.<br />
Am J Kidney Dis 37:E23.<br />
Wolfe S, Schulte-Strake U, Bertram B, et al. (1993). Hemodilution therapy <strong>in</strong> patients with acute anterior ischemic<br />
optic neuropathy. Ophthalmology 90:21–26.<br />
Worrall BB, Moazami G, Odel JG, Behrens MM. (1997). <strong>An</strong>terior ischemic optic neuropathy and activated prote<strong>in</strong><br />
C resistance. A case report and review of the literature. J <strong>Neuro</strong>-ophthalmol 17:162–165.<br />
WuDunn D, Zimmerman K, Sadun AA, Feldon SE. (1997). Comparison of visual function <strong>in</strong> fellow eyes after<br />
bilateral nonarteritic anterior ischemic optic neuropathy. Ophthalmology 104:104–111.<br />
Yee RD, Selky AK, Purv<strong>in</strong> VA. (1993). Outcomes of surgical and nonsurgical management of nonarteritic ischemic<br />
optic neuropathy. Trans Am Ophthalmol Soc 91:227–243.
5 r<br />
Arteritic <strong>An</strong>terior Ischemic Optic<br />
<strong>Neuro</strong>pathy and Giant Cell Arteritis<br />
Giant cell (temporal or cranial) arteritis (GCA) is an <strong>in</strong>flammatory vasculopathy of the<br />
elderly that affects medium- to large-sized arteries. GCA may present with numerous<br />
systemic and ocular manifestations (Aburahma, 1992, 1996; Aiello, 1993; Astion, 1994;<br />
Barton, 1991; Berlit, 1992; Buchb<strong>in</strong>der, 1992; Cid, 1998; Cleark<strong>in</strong>, 1992b; Diamond, 1991,<br />
1993; DiBartolomeo, 1992; Evans, 1993; Gabriel, 1995; Gaynes, 1994; Glutz von<br />
Blotsheim, 1997; Grosser, 1999; Hayreh, 1991, 1998b,c; Heathcote, 1999; Hellman,<br />
1993; Hunder, 1990a; Kachroo, 1996; Kattah, 1999; Kyle, 1993; Matzk<strong>in</strong>, 1992; Mizen,<br />
1991; Myles, 1992; Nordborg, 1990; Postel, 1993; Pounta<strong>in</strong>, 1995; Rousseau, 1994;<br />
Salvarani, 1995; Schmidt, 1994; Siatkowski, 1993; Sonnenblick, 1994; Stevens, 1995;<br />
We<strong>in</strong>berg, 1994). Here we concentrate on the ocular manifestations, diagnosis, and<br />
treatment of GCA. Less emphasis is placed on nonocular <strong>in</strong>volvement by GCA.<br />
What <strong>Cl<strong>in</strong>ical</strong> Features Suggest Giant<br />
Cell Arteritis?<br />
GCA usually causes visual loss due to anterior ischemic optic neuropathy (AION). All<br />
patients older than age 50 years with AION should be suspected of hav<strong>in</strong>g GCA. The<br />
<strong>in</strong>dex of suspicion is greater with <strong>in</strong>creas<strong>in</strong>g numbers of typical features of GCA listed<br />
<strong>in</strong> Table 5–1 (Aburahma, 1992, 1996; Aiello, 1993; Astion, 1994; Barton, 1991; Berlit, 1992;<br />
Buchb<strong>in</strong>der, 1992; Cleark<strong>in</strong>, 1992b; Diamond, 1991, 1993; DiBartolomeo, 1992; Evans,<br />
1993; Gabriel, 1995; Gaynes, 1994; Glutz von Blotsheim, 1997; Goh, 2000; Hayreh,<br />
1991, 1998b,c; Hellman, 1993; Hunder, 1990a; Kachroo, 1996; Kyle, 1993; Liu, 1994;<br />
Matzk<strong>in</strong>, 1992; Mizen, 1991; Myles, 1992; Nordborg, 1990; Postel, 1993; Pounta<strong>in</strong>, 1995;<br />
Rousseau, 1994; Salvarani, 1995; Siatkowski, 1993; Sonnenblick, 1994; Stevens, 1995;<br />
We<strong>in</strong>berg, 1994).<br />
93
94 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 5–1. Typical Features of Giant Cell Arteritis (GCA)<br />
Age greater than 50 years (median 75 years)<br />
Acute, often severe, visual loss (usually anterior ischemic optic neuropathy [AION])<br />
Unilateral or bilateral visual loss (higher <strong>in</strong>cidence of bilateral than nonarteritic [NA]-AION)<br />
Pallid swell<strong>in</strong>g of the optic nerve (may be ‘‘chalk white’’)<br />
Optic atrophy eventually (usually <strong>in</strong> 6 to 8 weeks) often with end-stage optic disc appearance of<br />
cupp<strong>in</strong>g with pallor and loss of neuroret<strong>in</strong>al rim (Danesh-Meyer, 2001; Hayreh, 1998c, 2001)<br />
Constitutional signs and symptoms<br />
Headache (4–100%)<br />
Scalp or temporal artery tenderness (28–91%)<br />
Weight loss (16–76%)<br />
Jaw claudication (4–67%) (Lee, 1995)<br />
<strong>An</strong>orexia (14–69%)<br />
Fever (low grade) and diaphoresis (Fife, 1994)<br />
Proximal muscle aches or weakness (28–86%)<br />
Polymyalgia rheumatica<br />
Morn<strong>in</strong>g stiffness last<strong>in</strong>g 30 m<strong>in</strong>utes or more<br />
Proximal jo<strong>in</strong>t pa<strong>in</strong> (e.g., shoulders, hips, neck, or torso)<br />
Fatigue and malaise (12–97%)<br />
Leg claudication (2–43%)<br />
Elevated erythrocyte sedimentation rate (usually > 50 mm per hour by Westergren method)<br />
Temporal artery biopsy positive<br />
Is the <strong>Cl<strong>in</strong>ical</strong> Suspicion for GCA High?<br />
In 1990 the American College of Rheumatology (Hunder, 1990a) analyzed 214 patients<br />
with GCA (196 proven by positive temporal artery biopsy) and compared them with<br />
593 patients with other forms of vasculitis. In their analysis of 33 criteria, the highest<br />
sensitivity criteria for GCA were the follow<strong>in</strong>g:<br />
1. Age >50 years (mean age 69 years, 90% >60 years)<br />
2. Westergren erythrocyte sedimentation rate (ESR) >50 mm=hour<br />
3. Abnormal temporal artery biopsy (TAB)<br />
The highest specificity cl<strong>in</strong>ical criteria were the follow<strong>in</strong>g:<br />
1. Jaw and=or tongue claudication<br />
2. Visual abnormalities (e.g., AION, amaurosis, optic atrophy)<br />
3. Temporal artery abnormalities (e.g., decreased pulse, tenderness, or nodules)<br />
If at least three or more criteria of the follow<strong>in</strong>g five were met, the specificity of<br />
diagnosis was 91.2% and the sensitivity was 93.5%:<br />
1. Age >50 years<br />
2. New headache (localized)<br />
3. Temporal artery abnormality (see above)<br />
4. Elevated ESR ( >50 mm=hour)<br />
5. Abnormal temporal artery biopsy (e.g., necrotiz<strong>in</strong>g arteritis, mult<strong>in</strong>ucleated giant cells)
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 95<br />
One of these diagnostic criteria (positive temporal artery biopsy) makes the<br />
diagnosis with high specificity and is the ‘‘gold standard’’ for diagnosis. Fernandez-<br />
Herlihy <strong>in</strong>creased the specificity for diagnosis of GCA by def<strong>in</strong><strong>in</strong>g symptom clusters,<br />
for example, jaw claudication with any of the follow<strong>in</strong>g (Fernandez-Herlihy,<br />
1988):<br />
1. Recent headaches and scalp tenderness<br />
2. Scalp tenderness and ESR >50 mm=hour<br />
3. Visual symptoms and ESR >50 mm=hour<br />
A specificity of 90 to 100% could be obta<strong>in</strong>ed if the cluster <strong>in</strong>cluded elevated ESR, scalp<br />
tenderness, jaw claudication, recent visual changes, polymyalgia rheumatica, and a<br />
good response to steroid therapy. A 94.8% sensitivity and 100% specificity were<br />
obta<strong>in</strong>ed if the symptom cluster <strong>in</strong>cluded new-onset headache, jaw claudication, and<br />
abnormal temporal artery exam<strong>in</strong>ation (Mizen, 1991). Vilaseca et al found that simultaneous<br />
jaw claudication, abnormal temporal arteries on exam, and new headache had a<br />
specificity of 94.8% for positive TAB (Vilaseca, 1987). Chmelewski et al compared the<br />
<strong>in</strong>itial cl<strong>in</strong>ical features of 30 patients with positive TAB and 68 with negative TAB<br />
(Chmelewski, 1992). TAB-positive patients had significantly <strong>in</strong>creased <strong>in</strong>cidence of<br />
headache (93% vs. 62%) and jaw claudication (50% vs. 18%). Jaw claudication had a<br />
specificity of 56% as a differentiat<strong>in</strong>g feature, but the specificity of headache was low<br />
(40%). Hayreh et al reported that jaw claudication (p ¼ 0.001) and neck pa<strong>in</strong> (mostly <strong>in</strong><br />
the occipital and back parts of the neck; p ¼ 0.0003) were significant <strong>in</strong>dicators of a<br />
positive TAB <strong>in</strong>dependent of ESR and age, and that these cl<strong>in</strong>ical signs were more<br />
highly correlated to a positive TAB than anorexia, weight loss, fever, and scalp<br />
tenderness (Hayreh, 1997). Hayreh et al felt that the odds of a positive TAB were<br />
9.0 times greater with jaw claudication, 3.3 times greater with neck pa<strong>in</strong>, 3.2 times<br />
greater with a C-reactive prote<strong>in</strong> (CRP) >2.45 mg=dL, 2.1 times greater with an ESR of<br />
47 to 107 mm=hour, 2.7 times greater with an ESR >107 mm=hour, and 2.0 times greater<br />
when the patient was greater than 75 years old (compared with age below 75 years).<br />
The typical features of GCA are listed <strong>in</strong> Table 5–1.<br />
Acute visual loss is reported <strong>in</strong> 7 to 60% (average 36%) of patients with GCA.<br />
Although the usual cause of visual loss <strong>in</strong> GCA is AION or central ret<strong>in</strong>al artery<br />
occlusion (CRAO) (Charness, 1991; Cleark<strong>in</strong>, 1992a; Liu, 1994), cilioret<strong>in</strong>al artery<br />
occlusion, ocular ischemic syndrome, posterior ischemic optic neuropathy (PION),<br />
choroidal ischemia, or rarely occipital lobe ischemia may also occur (Miller, 1991;<br />
Sadda, 2001). In a prospective study of 170 patients with biopsy-proven GCA, 85 (50%)<br />
presented with ocular <strong>in</strong>volvement (Hayreh, 1998b). The ocular f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> this study<br />
are outl<strong>in</strong>ed <strong>in</strong> Table 5–2.<br />
Although visual loss and AION <strong>in</strong> GCA tends to be more severe than that seen <strong>in</strong><br />
NA-AION (Hayreh, 1998b), the lack of severe visual loss is not a differentiat<strong>in</strong>g feature.<br />
Patients with AION <strong>in</strong> GCA may have little or no visual loss. On the other hand, very<br />
severe visual loss with AION is a ‘‘red flag’’ for GCA. In a study by Hayreh et al, 54% of<br />
patients with arteritic AION had <strong>in</strong>itial visual acuity of count<strong>in</strong>g f<strong>in</strong>gers to no light<br />
perception (compared to 26% of patients with NA-AION). Light perception was present<br />
<strong>in</strong> 29% and no light perception <strong>in</strong> 4% of AION due to GCA (Hayreh, 1998c). Therefore,<br />
massive early visual loss <strong>in</strong> AION is suggestive of GCA. Up to 25% of GCA patients<br />
have visual acuities of 20=40 or better and 20% of NA-AION patients have <strong>in</strong>itial visual<br />
acuities of count<strong>in</strong>g f<strong>in</strong>gers or worse (Hayreh, 1990). The cl<strong>in</strong>ical features favor<strong>in</strong>g
96 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 5–2. Ocular F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> 85 Patients with Biopsy-Proven<br />
GCA (Hayreh, 1998b) and Ocular Involvement<br />
F<strong>in</strong>d<strong>in</strong>g<br />
Ocular symptoms<br />
Number of Patients (%)<br />
Visual loss of vary<strong>in</strong>g severity 83 (97.7%)<br />
Amaurosis fugax 26 (30.6%)<br />
Diplopia 5 (5.9%)<br />
Eye pa<strong>in</strong><br />
Ocular signs<br />
7 (8.2%)<br />
Arteritic AION 69 (81.2%)<br />
Central ret<strong>in</strong>al artery occlusion 12 (14.1%)<br />
Cilioret<strong>in</strong>al artery occlusion 12 (14.1%)<br />
Posterior ION 6 (7.1%)<br />
Ocular ischemic syndrome 1 (1.2%)<br />
n ¼ 85 with ocular <strong>in</strong>volvement.<br />
arteritic AION over NA-AION are listed <strong>in</strong> Table 5–3. Other less common ocular<br />
features of GCA are listed <strong>in</strong> Table 5–4.<br />
The differential diagnosis for these ocular conditions (especially unexpla<strong>in</strong>ed diplopia,<br />
ret<strong>in</strong>al or choroidal ischemia, central ret<strong>in</strong>al artery occlusion without visible emboli, or<br />
transient visual loss) should <strong>in</strong>clude GCA. Goldberg reviewed the literature <strong>in</strong> 1983 on<br />
ocular motor paresis <strong>in</strong> GCA and found ocular muscle <strong>in</strong>volvement was reported <strong>in</strong> 59<br />
patients (Goldberg, 1983). The duration of symptoms was transitory to several months.<br />
Many cases had other signs to suggest GCA (e.g., headache, scalp tenderness, optic<br />
nerve, or ret<strong>in</strong>al <strong>in</strong>volvement). The diplopia was often transient, variable, and some-<br />
Table 5–3. Features Suggestive of Arteritic AION Rather than NA-AION<br />
Elderly patients with constitutional symptoms (especially scalp tenderness or jaw claudication)<br />
Polymyalgia rheumatica<br />
Elevated erythrocyte sedimentation rate (ESR) and=or C-reactive prote<strong>in</strong> (CRP)<br />
Amaurosis fugax—likely transient optic nerve ischemia rather than ret<strong>in</strong>al ischemia (Hayreh,<br />
1998b; Liu, 1994; Ronchetto, 1992)<br />
Ocular f<strong>in</strong>d<strong>in</strong>gs (Hayreh, 1990, 1997, 1998b,c; Sadda, 2001):<br />
Posterior ischemic optic neuropathy (PION)<br />
Cup to disc ratio greater than 0.2 <strong>in</strong> fellow eye<br />
Early massive or bilateral simultaneous visual loss<br />
Markedly pallid optic disc edema (chalky white <strong>in</strong> 68.7%)<br />
End-stage optic disc appearance of cupp<strong>in</strong>g (seen <strong>in</strong> 92% of eyes with arteritic AION vs. 2% of<br />
eyes with NA-AION) (Danesh-Meyer, 2001)<br />
Fluoresce<strong>in</strong> angiography f<strong>in</strong>d<strong>in</strong>gs of choroidal nonperfusion or delayed choroidal fill<strong>in</strong>g<br />
(<strong>in</strong>docyan<strong>in</strong>e green angiography provides no additional <strong>in</strong>formation) (Hayreh, 1990; Mack,<br />
1991; Segato, 1990; Siatkowski, 1993; Valmaggia, 1999)<br />
AION associated with choroidal nonfill<strong>in</strong>g<br />
Simultaneous AION with nonembolic cilioret<strong>in</strong>al artery occlusion (CRAO)<br />
Simultaneous AION with choroidal or ret<strong>in</strong>al <strong>in</strong>farction
Table 5–4. Other Ocular Features of GCA<br />
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 97<br />
Visual loss<br />
Transient visual loss (Hayreh, 1998b; Liu, 1994; Thystrup, 1994)<br />
Alternat<strong>in</strong>g transient visual loss (F<strong>in</strong>elli, 1997)<br />
Alternat<strong>in</strong>g transient visual loss <strong>in</strong>duced by bright light (Galetta, 1997b)<br />
Posture related ret<strong>in</strong>al ischemia<br />
Bilateral transient visual loss with change <strong>in</strong> posture due to vertebrobasilar <strong>in</strong>volvement (Diego,<br />
1998)<br />
Bilateral transient visual loss with change <strong>in</strong> posture due to impend<strong>in</strong>g AION (Diego, 1998)<br />
Nonembolic branch or central ret<strong>in</strong>al artery occlusion (F<strong>in</strong>eman, 1996; Glutz von Blotsheim, 1997;<br />
Hayreh, 1998b; Liu, 1994; Miller, 1991; We<strong>in</strong>, 2000)<br />
Comb<strong>in</strong>ed central ret<strong>in</strong>al artery and ve<strong>in</strong> occlusion<br />
Ophthalmic artery occlusion<br />
Ophthalmic artery microembolism (Schauble, 2000)<br />
Choroidal or ret<strong>in</strong>al ischemia (Glutz von Blotsheim, 1997; Quillen, 1993; Slav<strong>in</strong>, 1994)<br />
Cotton wool spots (Hayreh, 1998b; MacLeod, 1993; Melberg, 1995; Thystrup, 1994)<br />
General anesthesia <strong>in</strong>duced ischemic optic neuropathy<br />
Pre- and perichiasmal ischemia and visual field defects<br />
Postchiasmal ischemic visual field defects (rare)<br />
<strong>An</strong>terior segment ischemia (Birt, 1994)<br />
Episcleritis and scleritis<br />
Iritis<br />
Panuveitis (Rajesh, 2000)<br />
Conjunctivitis<br />
Glaucoma (e.g., acute angle closure glaucoma)<br />
Uveitic glaucoma (Tomsak, 1997)<br />
Transient bilateral corneal edema<br />
Acute hypotony<br />
Marg<strong>in</strong>al corneal ulceration (Tomsak, 1997)<br />
Autonomic pupil abnormalities<br />
Tonic pupil<br />
Light-near dissociation<br />
Horner syndrome (Pascual-Sedano, 1998)<br />
Miosis<br />
Mydriasis<br />
Diplopia<br />
Orbital ischemia<br />
Ophthalmoplegia (Goadsby, 1991) due to ischemia to cranial nerves III, IV, and=or VI (Bondeson,<br />
1997; Diamond, 1991; Killer, 2000)<br />
Bra<strong>in</strong>stem ischemia (rare)<br />
Internuclear ophthalmoplegia (Ahmed, 1999; Askari, 1993; Eggenberger, 1998; Johnston, 1992;<br />
Trend, 1990)<br />
Internuclear ophthalmoplegia with facial nerve palsy (‘‘eight-and-a-half syndrome’’) (Eggenberger,<br />
1998)<br />
One-and-a-half syndrome (Galetta, 1997b)<br />
Nystagmus<br />
(cont<strong>in</strong>ued)
98 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 5–4. (cont<strong>in</strong>ued)<br />
Subjective diplopia by history<br />
Transient diplopia with or without ptosis (Hayreh, 1998b; Liu, 1994)<br />
Divergence <strong>in</strong>sufficiency (Jacobson, 2000)<br />
Transient oculomotor synk<strong>in</strong>esis<br />
Laboratory measures of ischemia<br />
Color Doppler hemodynamics (Ho, 1994)<br />
Decreased ocular pulse<br />
Decreased ocular pulse amplitudes<br />
Orbital <strong>in</strong>volvement<br />
Orbital pseudotumor (Chertok, 1990; de Heide, 1999; Laidlaw, 1990; Lee, 2001; Looney, 1999)<br />
Orbital <strong>in</strong>farction (Borruat, 1993; Chertok, 1990; Laidlaw, 1990)<br />
Ocular ischemic syndrome (Casson, 2001; Hamed, 1992; Hayreh, 1998b; Hwang, 1999); may<br />
be bilateral (Casson, 2001)<br />
Reversible bruit<br />
Optic nerve enhancement on MR imag<strong>in</strong>g (may help <strong>in</strong> differentiation from NA-AION)<br />
(Lee, 1999a)<br />
times not associated with motility exam<strong>in</strong>ation abnormalities. The optic nerve or central<br />
ret<strong>in</strong>al artery <strong>in</strong>volvement followed with<strong>in</strong> several days <strong>in</strong> many patients. Graham<br />
described 10 GCA patients with ophthalmoplegia (four pupil-spar<strong>in</strong>g third nerve<br />
palsies, four sixth nerve palsies, and two multiple ocular motor nerve palsies)<br />
(Graham, 1980). Bondeson described a patient with pupil-spar<strong>in</strong>g third nerve palsy<br />
secondary to GCA (Bondeson, 1997). Brilakis and Lee reviewed 18 previous reports<br />
(81 patients) of diplopia with GCA (Brilakis, 1998). Of these 81 patients, 60 (74%) had<br />
other signs and symptoms of GCA and 21 (26%) had <strong>in</strong>sufficient cl<strong>in</strong>ical <strong>in</strong>formation to<br />
determ<strong>in</strong>e if other signs and symptoms of GCA were present.<br />
Liu et al noted that transient monocular bl<strong>in</strong>dness (18% of patients) and transient<br />
diplopia (15% of patients) were the most common premonitory visual compla<strong>in</strong>t <strong>in</strong><br />
GCA (Liu, 1994). Hayreh also described transient diplopia <strong>in</strong> 5.9% of patients with GCA<br />
(Hayreh, 1998b) and noted that all of the extraocular muscles and the levator palpebrae<br />
superioris are supplied by more than one and up to five vascular branches of the<br />
ophthalmic artery, except for the <strong>in</strong>ferior oblique (with only one branch). This collateral<br />
vascular supply may expla<strong>in</strong> the usual transient nature of diplopia <strong>in</strong> GCA, which is<br />
thought due to ischemia of one or more of the extraocular muscles due to arteritic<br />
occlusion of one or more of the muscular arteries (Hayreh, 1998a).<br />
We do not rout<strong>in</strong>ely obta<strong>in</strong> an ESR on patients with transient or persistent diplopia<br />
without systemic signs of GCA <strong>in</strong> whom there is a clear alternative etiology (e.g., other<br />
vasculopathic risk factors). Nevertheless, we consider the diagnosis of GCA <strong>in</strong> all<br />
patients over 55 years with unexpla<strong>in</strong>ed diplopia (class III, level U). It is our current<br />
practice to evaluate for GCA <strong>in</strong> elderly patients with diplopia that is ill def<strong>in</strong>ed or<br />
transient or if there are other signs or symptoms of GCA (class III, level U).<br />
Caselli and Hunder reviewed the neurologic aspects of GCA and emphasized the<br />
often underrecognized fact that GCA affects the aortic arch and its branches, not just the<br />
superficial temporal arteries. Although GCA does not cause a widespread <strong>in</strong>tracranial<br />
vasculitis, it may <strong>in</strong>volve the cervicocephalic arteries <strong>in</strong>clud<strong>in</strong>g the carotid artery and
vertebral arteries (Caselli, 1993). Less commonly recognized f<strong>in</strong>d<strong>in</strong>gs of GCA are listed<br />
<strong>in</strong> Table 5–5.<br />
Is the ESR Always Elevated <strong>in</strong> GCA?<br />
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 99<br />
Although the ESR is often elevated <strong>in</strong> GCA (Britta<strong>in</strong>, 1991; We<strong>in</strong>ste<strong>in</strong>, 1994), patients<br />
with biopsy-proven GCA may have a normal ESR (2–30%) (Brigden, 1998; Britta<strong>in</strong>,<br />
1991; Glutz von Blotsheim, 1997; Grodum, 1990; Hayreh, 1997; Jundt, 1991; Litw<strong>in</strong>,<br />
1992; Liu, 1994; Neish, 1991; Salvarani, 2001; Wise, 1991; Zweegman, 1993). Cullen<br />
found an average ESR of 84 mm=hour <strong>in</strong> TAB proven GCA (Cullen, 1967).<br />
What Is the Normal Value for an ESR?<br />
The ESR rises with <strong>in</strong>creas<strong>in</strong>g age. The Westergren method is preferred over the<br />
W<strong>in</strong>trobe method because of the more limited scale of the W<strong>in</strong>trobe ESR. Boyd and<br />
Hoffbrand reported a Westergren ESR normal of 40 mm=hour for persons over age<br />
65 years (Boyd, 1966). Bottiger and Svedberg felt that 30 mm=hour for women and<br />
20 mm=hour for men was a reasonable limit (Bottiger, 1967). Hayreh concluded that a<br />
patient with an ESR >40 mm=hour should be considered to ‘‘suffer from temporal<br />
arteritis, unless proven otherwise.’’<br />
Miller et al measured Westergren ESR <strong>in</strong> 27,912 adults aged 20 to 65 years (Miller,<br />
1983). None of the subjects were anemic. A series of curves of ESR versus age were<br />
derived for men and women with maximum values for 98% of the population.<br />
<strong>An</strong> empiric formula (98% curve) for deriv<strong>in</strong>g the maximum ESR normal is listed as<br />
follows (Miller, 1983; Sox, 1986): for men, age divided by 2; for women, age þ10 divided<br />
by 2.<br />
Hayreh et al suggested a cut-off criterion for an elevated ESR of 33 mm=hour for men<br />
and 35 mm=hour for women with a sensitivity and specificity of 92% (Hayreh, 1997).<br />
In addition, the ESR value at the time of diagnosis may not correlate with the cl<strong>in</strong>ical<br />
features or prognosis for visual loss <strong>in</strong> GCA. Other markers (e.g., CRP, von Willebrand<br />
factor) have also been proposed <strong>in</strong> the evaluation of GCA. Jacobson and Slamovits<br />
found an <strong>in</strong>verse correlation between ESR and hematocrit and felt that the ‘‘ESR may<br />
not reliably <strong>in</strong>dicate active disease <strong>in</strong> a patient with a normal hematocrit’’ (Jacobson,<br />
1987). F<strong>in</strong>ally, it should be emphasized that the diagnosis of GCA is a cl<strong>in</strong>ical diagnosis,<br />
and reliance for such a diagnosis should not be placed on the ESR alone. If the cl<strong>in</strong>ical<br />
suspicion for GCA is high, a repeat ESR, TAB, and treatment with empiric prednisone<br />
should beg<strong>in</strong> regardless of the <strong>in</strong>itial ESR value.<br />
Are There Other Hematologic Tests for the<br />
Diagnosis of GCA?<br />
<strong>An</strong>other acute-phase reactant, CRP, has also been advocated as a marker for GCA<br />
(Hayreh, 1997). Hayreh et al felt that an elevated CRP (above 2.45 mg=dL) was more<br />
sensitive (100%) than the ESR (92%) for the detection of GCA, and that a CRP comb<strong>in</strong>ed<br />
with an ESR gave the best specificity for diagnosis (97%) (Hayreh, 1997).<br />
Other hematologic tests listed <strong>in</strong> Table 5–6 have been reported <strong>in</strong> association with<br />
GCA, but are of uncerta<strong>in</strong> significance (e.g., serum amyloid, von Willebrand’s factor,
100 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 5–5. Less Common Features of GCA<br />
Large vessel <strong>in</strong>volvement (Butt, 1991; Lambert, 1996)<br />
Carotid siphon<br />
Bruits (Caselli, 1988)<br />
Facial artery (Achkar, 1995)<br />
Pa<strong>in</strong> on palpation of external carotid artery (Gonzalez-Gay, 1998b)<br />
Occipital artery pa<strong>in</strong> and occipital neuralgia ( Jundt, 1991)<br />
Subclavian or axillary artery (N<strong>in</strong>et, 1990)<br />
Aortitis or aortic rupture (Evans, 1995; Gersbach, 1993; Lagrand, 1996; Lie, 1995a; Liu, 1995;<br />
Mitnich, 1990; Richardson, 1996)<br />
Aortic aneurysm (Hamano, 1999)<br />
Limb claudication or gangrene (Desmond, 1999; Lie, 1995a; Walz-Leblanc, 1991)<br />
Upper or lower limb ischemia (Garcia Vazquez, 1999)<br />
Unilateral distal extremity swell<strong>in</strong>g and edema (Kontoyianni, 1999)<br />
Raynaud’s phenomenon (Mallia, 1999)<br />
<strong>Neuro</strong>logic features<br />
Central nervous system arteritis (Caselli, 1988, 1990, 1993; Husse<strong>in</strong>, 1990; Reich, 1990)<br />
Acute encephalopathy (Caselli, 1990; Tomer, 1992)<br />
Aseptic men<strong>in</strong>gitis<br />
Cerebellar <strong>in</strong>farction (McLean, 1993)<br />
Diabetes <strong>in</strong>sipidus<br />
Occipital <strong>in</strong>farction and cortical bl<strong>in</strong>dness<br />
Multifocal dural enhancement and enhancement of temporalis muscles on MR imag<strong>in</strong>g ( Joelson, 2000)<br />
Myelopathy (Caselli, 1988)<br />
Cervical radiculopathy (Rivest, 1995)<br />
Quadriplegia (Brennan, 1982)<br />
Transverse myelopathy<br />
Sp<strong>in</strong>al cord <strong>in</strong>farction (Galetta, 1997a)<br />
<strong>Neuro</strong>psychiatric syndromes<br />
Halluc<strong>in</strong>ations<br />
Depression<br />
Behavioral changes<br />
Psychosis and confusion<br />
Seizures<br />
Transient ischemic attacks (Caselli, 1988)<br />
Tremor<br />
Dysarthria precipitated by chew<strong>in</strong>g or prolonged talk<strong>in</strong>g (Lee, 1999b)<br />
Numb ch<strong>in</strong> syndrome (Genereau, 1999)<br />
Proximal muscle weakness with skeletal muscle vasculitis (Lacomas, 1999)<br />
<strong>Neuro</strong>-otologic symptoms (Caselli, 1988)<br />
Deafness (Caselli, 1988; Reich, 1990)<br />
T<strong>in</strong>nitus<br />
Vertigo<br />
Bra<strong>in</strong>stem (Dick, 1991; Gonzalez-Gay, 1998a)<br />
Ataxia, nystagmus, upgaze palsy<br />
Lateral medullary syndrome (Shanahan, 1999)<br />
Vertebrobasilar <strong>in</strong>volvement (Sheehan, 1993)<br />
(cont<strong>in</strong>ued)
Table 5–5. (cont<strong>in</strong>ued)<br />
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 101<br />
Acute confusional states (Caselli, 1988, 1990)<br />
Cluster headache ( Jiménez-Jiménez, 1998)<br />
Peripheral neuropathies (Caselli, 1988)<br />
Sciatic neuropathy<br />
Carpal tunnel syndrome (Dennis, 1996)<br />
Vernet’s syndrome (affection of n<strong>in</strong>th, tenth, and eleventh cranial nerves due to ischemia of<br />
ascend<strong>in</strong>g pharyngeal artery) (Gout, 1998)<br />
Pa<strong>in</strong> syndromes (headache, neck pa<strong>in</strong>, backache) (Caselli, 1993)<br />
Respiratory tract (Gur, 1996; Rischmueller, 1996; Zenone, 1994)<br />
Cough (Lim, 1999; Olopade, 1997)<br />
Hoarseness<br />
Diaphragmatic weakness (Burton, 1999)<br />
Tongue ischemia (Caselli, 1988)<br />
Seronegative polyarthritis<br />
Coronary arteritis and myocardial <strong>in</strong>farction (Freddo, 1999)<br />
Visceral <strong>in</strong>volvement<br />
Renal <strong>in</strong>volvement (L<strong>in</strong>, 1995)<br />
Visceral angiitis<br />
Liver <strong>in</strong>volvement (Ilan, 1993; Killer, 2000)<br />
Small bowel <strong>in</strong>farction (Phelan, 1993)<br />
Tongue necrosis (Llorente, 1994)<br />
Submandibular swell<strong>in</strong>g (Ruiz-Masera, 1995)<br />
Secondary amyloidosis (Altlparmak, 2001; Stebb<strong>in</strong>g, 1999)<br />
Ischemic sk<strong>in</strong> lesions (Hansen, 1995) and scalp necrosis (Botella-Estrada, 1999; Currey, 1997; Rudd,<br />
1998)<br />
Association with parvovirus B19 <strong>in</strong>fection (Gabriel, 1999; Straud, 1996)<br />
Mortality (Bisgard, 1991; Matteson, 1996)<br />
Myocardial <strong>in</strong>farction and mesenteric <strong>in</strong>farction<br />
plasma viscosity, ant<strong>in</strong>eutrophil cytoplasmic antibodies, and various human lymphocyte<br />
antigen [HLA] types). <strong>An</strong>ticardiolip<strong>in</strong> antibodies were present at the onset <strong>in</strong> 19 of<br />
40 patients with GCA and polymyalgia rheumatica (Manna, 1998). In 56% of these<br />
patients, these antibodies disappeared dur<strong>in</strong>g steroid treatment. Thrombocytosis<br />
occurred <strong>in</strong> 44% of 34 patients <strong>in</strong> one series and the platelet count was reduced by<br />
corticosteroid therapy (Gonzalez-Alegre, 2001). No association was found between the<br />
platelet count and ischemic complications of the disease.<br />
Is a TAB Necessary <strong>in</strong> a Patient with a High<br />
<strong>Cl<strong>in</strong>ical</strong> Suspicion for GCA? Should a<br />
Unilateral or Bilateral TAB Be Performed?<br />
TAB is a relatively easy and safe procedure to perform with low morbidity. There is one<br />
reported case of stroke after TAB, but this is a rare complication (Haist, 1985).
102 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 5–6. Laboratory Abnormalities <strong>in</strong> GCA<br />
<strong>An</strong>ticardiolip<strong>in</strong> antibodies (Kerleau, 1994; Manna, 1998; McHugh, 1990)<br />
<strong>An</strong>t<strong>in</strong>eutrophilic antibodies (Bosch, 1991; McHugh, 1990)<br />
Mild to moderate normochromic, normocytic anemia (Weiss, 1995)<br />
Elevated white blood cell count and platelet count<br />
Thrombocytosis (Gonzalez-Alegre, 2001; L<strong>in</strong>coff, 2000)<br />
Elevated acute-phase reactant prote<strong>in</strong>s (e.g., fibr<strong>in</strong>ogen, von Willebrand factor) (Pounta<strong>in</strong>, 1994)<br />
Abnormal plasma viscosity (Gudmundsson, 1993; Orrell, 1993)<br />
Serum prote<strong>in</strong> electrophoresis abnormalities<br />
Hepatic dysfunction<br />
Elevated endothel<strong>in</strong>-1 plasma levels (Pache, 2002)<br />
Multiple immunologic abnormalities (Bosch, 1991; Radda, 1981; Salvarani, 1991; Wawryk, 1991;<br />
Weyand, 1992, 1994, 1995, 1997)<br />
Immune complexes<br />
T-cell abnormalities<br />
Immunohistochemical abnormalities<br />
HLA-DR4 and -DR3 (Combe, 1998; Gros, 1998)<br />
Complications of TAB <strong>in</strong>clude <strong>in</strong>fection, chronic sk<strong>in</strong> ulceration, transient brow droop,<br />
hemorrhage, damage to the facial nerve, and stroke (Bhatti, 2001b; Danish-Meyer, 2000;<br />
Miller, 2000). Several authors have reported various techniques (Cleark<strong>in</strong>, 1991; Hedges,<br />
1992; Tomsak, 1991). Temporal arteries that are difficult to locate may require the use of<br />
<strong>in</strong>traoperative Doppler for localization (Beckman, 1990). Hall et al performed 134 TABs,<br />
and 46 (34%) showed GCA (Hall, 1983). Of the 88 TABs (66%) that were normal (over a<br />
70-month follow-up period), only eight patients required steroid therapy. Thus, a<br />
negative TAB predicted the absence of steroid therapy requirement <strong>in</strong> 91% and<br />
helped determ<strong>in</strong>e the appropriate treatment <strong>in</strong> 94% of cases. These and other authors<br />
thought that a TAB should be done before patients are committed to long-term<br />
corticosteroid therapy (Hall, 1983) because of the associated significant side effects of<br />
chronic steroid use <strong>in</strong>clud<strong>in</strong>g cush<strong>in</strong>goid features, hypertension, diabetes, osteoporosis,<br />
compression fractures (up to 25% of patients), steroid myopathy, steroid psychosis, and<br />
fluid retention requir<strong>in</strong>g diuretics (Nesher, 1994). Nadeau reviewed four different series<br />
(162 patients) of steroid therapy <strong>in</strong> GCA and found a 38% <strong>in</strong>cidence of complications<br />
(range 12.7 to 60.6%) (Nadeau, 1988). Us<strong>in</strong>g a cl<strong>in</strong>ical decision analysis approach,<br />
Nadeau concluded that when steroid complications were likely, a TAB was useful even<br />
with ‘‘fairly high pre-biopsy probabilities of disease’’ (Nadeau, 1988). In addition,<br />
Hedges et al thought that no laboratory test (i.e., ESR) or frequently observed symptom<br />
or sign of GCA alone or <strong>in</strong> comb<strong>in</strong>ation with other f<strong>in</strong>d<strong>in</strong>gs (e.g., jaw claudication) had<br />
the diagnostic specificity or sensitivity of the TAB (Hedges, 1983).<br />
Patients with a negative unilateral TAB <strong>in</strong> whom there is a strong cl<strong>in</strong>ical suspicion<br />
(see cl<strong>in</strong>ical features and symptom clusters, above) for GCA should be considered for a<br />
contralateral TAB (Coppetto, 1990; Hunder, 1990a). To m<strong>in</strong>imize costs, some authors<br />
have advocated that a frozen section be performed on the symptomatic-side TAB and, if<br />
it is normal, proceed at the same sitt<strong>in</strong>g with a contralateral TAB (Hall, 1984).<br />
Ponge et al analyzed 200 patients who underwent 200 bilateral TAB, all of which were<br />
preceded by Doppler flow studies. Forty-two TABs were positive, 20 bilaterally and 22<br />
unilaterally (Ponge, 1988). In their analysis, they discovered that four patients with
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 103<br />
GCA would not have been diagnosed if only a unilateral TAB had been performed.<br />
Unilaterally positive TABs have been demonstrated <strong>in</strong> 8 to 14% of retrospective bilateral<br />
TAB series (Hall, 1984). Hall and Hunder retrospectively reviewed 652 TABs at Mayo<br />
Cl<strong>in</strong>ic (Hall, 1984). Of these, 234 (36%) revealed GCA, and 193 (82%) were positive on<br />
unilateral TAB. Bilateral TABs were performed <strong>in</strong> 41 cases (18%) because frozen section<br />
was normal on the first TAB. Of the 193 unilateral TABs, frozen section was abnormal<br />
<strong>in</strong> 188 and normal <strong>in</strong> 5. Thus, 86% of the 234 cases would have been diagnosed by<br />
unilateral TAB alone and 14% were diagnosed only because a TAB was performed on<br />
the contralateral side. Hayreh et al reported 76 of 363 patients who underwent a second<br />
TAB because of a strong cl<strong>in</strong>ical <strong>in</strong>dex of suspicion for GCA (Hayreh, 1997). Seven of<br />
these 76 patients had a positive contralateral TAB. Of the rema<strong>in</strong><strong>in</strong>g 257 patients with a<br />
negative TAB, none developed signs of GCA on follow-up and these authors thought<br />
that this was <strong>in</strong>dicative that a second TAB would not have been positive.<br />
Boyev et al performed a retrospective study to determ<strong>in</strong>e the utility of unilateral<br />
versus bilateral TABs <strong>in</strong> detect<strong>in</strong>g the pathologic changes of GCA (Boyev, 1999). Of<br />
908 specimens exam<strong>in</strong>ed from 758 patients, 300 specimens were simultaneous bilateral<br />
biopsies from 150 patients, 72 specimens were bilateral sequential biopsies from<br />
36 patients, and the rema<strong>in</strong><strong>in</strong>g 536 specimens were unilateral biopsies from 536 patients.<br />
Of the 186 patients who had bilateral simultaneous or nonsimultaneous biopsies,<br />
176 had identical diagnoses on both sides. In four patients, no artery was obta<strong>in</strong>ed<br />
on one side. In each of the rema<strong>in</strong><strong>in</strong>g six patients, five of whom had bilateral<br />
simultaneous biopsies and one of whom had bilateral sequential biopsies performed<br />
8 days apart, the biopsy specimen from one side was <strong>in</strong>terpreted as show<strong>in</strong>g only<br />
arteriosclerotic changes with no evidence of active or healed arteritis, whereas the other<br />
specimen was <strong>in</strong>terpreted as show<strong>in</strong>g either probable healed arteritis (three specimens)<br />
or possible early arteritis (three cases). In none of the six patients with differ<strong>in</strong>g<br />
diagnoses between the two sides was one side <strong>in</strong>terpreted as show<strong>in</strong>g def<strong>in</strong>ite, active<br />
GCA. Five of six patients were subsequently determ<strong>in</strong>ed to have GCA, based on a<br />
comb<strong>in</strong>ation of cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs, ESR, and response to treatment with corticosteroids.<br />
The authors concluded that perform<strong>in</strong>g simultaneous or sequential TABs improves the<br />
diagnostic yield <strong>in</strong> at least 3% of cases of GCA, whereas <strong>in</strong> 97% of cases the two<br />
specimens show the same f<strong>in</strong>d<strong>in</strong>gs. Thus, <strong>in</strong> patients <strong>in</strong> whom only one artery can be<br />
biopsied, there is a high probability of obta<strong>in</strong><strong>in</strong>g the correct diagnosis. Nevertheless,<br />
although the improvement <strong>in</strong> diagnostic yield of bilateral TABs is low, the consequences<br />
of both delayed diagnosis and treatment of GCA as well as the use of systemic<br />
corticosteroids <strong>in</strong> patients who do not have GCA are of such severity that consideration<br />
should always be given to perform<strong>in</strong>g bilateral TABs <strong>in</strong> patients suspected of hav<strong>in</strong>g<br />
the disease.<br />
Pless et al reviewed 60 bilateral TAB results and reported a 5% chance of obta<strong>in</strong><strong>in</strong>g a<br />
positive biopsy result on one side and a negative biopsy result on the other side (Pless,<br />
2000), whereas Danesh-Meyer et al found a 1% discordance among 91 bilateral<br />
TABs (Danesh-Meyer, 2000). Danesh-Meyer et al performed a meta-analysis of exist<strong>in</strong>g<br />
literature and concluded that the overall chance of discordance is about 4% (Danesh-<br />
Meyer, 2000). Danesh-Meyer et al suggest that ‘‘consideration of simultaneous bilateral<br />
TABs appears to be a safe and prudent approach for diagnosis of GCA’’ (Danesh-Meyer,<br />
2000), and Pless et al suggest that ‘‘it is reasonable to biopsy both sides at the same<br />
session <strong>in</strong> order to <strong>in</strong>crease the likelihood of achievement of a correct diagnosis’’ (Pless,<br />
2000). In editorials follow<strong>in</strong>g the papers of Danesh-Meyer et al and Pless et al, the<br />
follow<strong>in</strong>g suggestions were noted:
104 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
1. Miller suggested that bilateral TABs should be considered <strong>in</strong> all patients <strong>in</strong> whom the<br />
diagnosis of GCA is suspected. ‘‘The biopsies can be simultaneous or sequential’’<br />
(Miller, 2000).<br />
2. Lessell suggests that ‘‘it makes sense to rout<strong>in</strong>ely perform bilateral biopsies or to<br />
biopsy the other side if the first side has a negative result <strong>in</strong> patients whose<br />
symptoms, signs, and laboratory results po<strong>in</strong>t to the diagnosis of giant cell arteritis’’<br />
(Lessell, 2000).<br />
3. Sav<strong>in</strong>o suggests that ‘‘performance of bilateral temporal artery biopsy, with or<br />
without the aid of frozen sections, appears to be the safest strategy’’ (Sav<strong>in</strong>o, 2000).<br />
We perform unilateral TAB <strong>in</strong> patients <strong>in</strong> all patients (class II–III, level C). If the pre-<br />
TAB <strong>in</strong>dex of suspicion for GCA is low, then we do not perform a second TAB. If the<br />
pre-TAB <strong>in</strong>dex of suspicion for GCA is high, then we consider a contralateral TAB. In<br />
the cases of moderate suspicion we <strong>in</strong>dividualize the decision for contralateral TAB<br />
(class III, level C).<br />
TAB has a variable sensitivity for GCA <strong>in</strong> the literature rang<strong>in</strong>g from 56 to 93%<br />
(Gonzalez-Gay, 2001; Mizen, 1991). The sensitivity improves to 85 to 90% when clear<br />
criteria for negative TAB are established (Nadeau, 1988). Skip lesions may occur<br />
pathologically (even <strong>in</strong> bilateral TAB) and may produce a false-negative rate of at<br />
least 4 to 5% (Kle<strong>in</strong>, 1976). A large segment TAB of a length of at least 2 to 5 cm is often<br />
recommended (Kle<strong>in</strong>, 1976), but other authors have found that even TAB as short as<br />
4 mm (if serially sectioned properly at 1 mm segments and with a m<strong>in</strong>imum of n<strong>in</strong>e<br />
sections from each segment) may result <strong>in</strong> a less than 1% false-negative rate (99%<br />
probability of detect<strong>in</strong>g any evidence of GCA) (Chambers, 1988). Short-length TAB,<br />
<strong>in</strong>sufficient section<strong>in</strong>g (0.25–0.5 mm cross sections through the entire specimen are<br />
recommended) (McDonnell, 1986), and variability <strong>in</strong> the quality and availability of<br />
good ophthalmic pathologic <strong>in</strong>terpretation of specimens contribute to a high falsenegative<br />
rate of 9 to 61% (Mizen, 1991). In addition, although steroid therapy may<br />
produce a false-negative result, TABs may be performed up to a few weeks (or more) of<br />
start<strong>in</strong>g steroid treatment (Achkar, 1994; To, 1994). Rarely, TAB may be positive even<br />
after 6 months of prednisone treatment (Guevara, 1998). Thus, even <strong>in</strong> the sett<strong>in</strong>g of a<br />
negative unilateral (or bilateral) TAB, the patient with a high cl<strong>in</strong>ical suspicion for GCA<br />
should be treated with cont<strong>in</strong>ued empiric corticosteroids (oral prednisone 80 to<br />
120 mg=day). Consideration could be given to a third biopsy of other arteries (e.g.,<br />
occipital, facial, or frontal artery) if the cl<strong>in</strong>ical suspicion for GCA rema<strong>in</strong>s high and<br />
pathologic confirmation is desired (Kattah, 1991; Weems, 1992). We have rarely had to<br />
resort to a third biopsy (class IV, level U). It should also be noted that TABs may<br />
occasionally reveal etiologies other than GCA (e.g., sarcoidosis or Wegener’s granulomatosis)<br />
for temporal artery vasculitis (Levy, 1994; Nish<strong>in</strong>o, 1993) and a tender superficial<br />
temporal artery and decreased pulse on palpation may occur with <strong>in</strong>timal fibrosis<br />
(Petzold, 2002). In a patient thought to have arteritic posterior ION, a TAB revealed lung<br />
adenocarc<strong>in</strong>oma as a cause for the optic neuropathy (Bhatti, 2001a).<br />
What Is the Evaluation for a Patient with a<br />
Moderate <strong>Cl<strong>in</strong>ical</strong> Suspicion for GCA?<br />
Patients with moderate cl<strong>in</strong>ical suspicion for GCA should undergo an ESR and a TAB<br />
(class II–III, level B). Unfortunately, constitutional symptoms and signs may be absent <strong>in</strong>
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 105<br />
up to 21.2% of cases (‘‘occult’’ GCA) (Desmet, 1990; Hayreh, 1998c; Liu, 1994). In a study<br />
of 85 patients with biopsy proven GCA, occult GCA occurred <strong>in</strong> 18 patients (21.5%)<br />
(Hayreh, 1998c). Ocular symptoms <strong>in</strong> these patients with occult GCA <strong>in</strong>cluded visual<br />
loss <strong>in</strong> 18 patients (100%), amaurosis fugax <strong>in</strong> 6 (33.3%), diplopia <strong>in</strong> 2 (11.1%), and eye<br />
pa<strong>in</strong> <strong>in</strong> 1 (5.6%), whereas ocular ischemia lesions <strong>in</strong>cluded AION <strong>in</strong> 17 (94.4%), central<br />
ret<strong>in</strong>al artery occlusion <strong>in</strong> 2 (11.1%), and cilioret<strong>in</strong>al artery occlusion <strong>in</strong> 2 (11.1%). If the<br />
ESR is elevated, and if a unilateral TAB is negative, then a contralateral TAB should be<br />
performed. Alternatively, a frozen section of the TAB on the symptomatic side could be<br />
performed and, if negative, a simultaneous contralateral TAB is done (Hall, 1984). If<br />
both TABs are negative, alternative etiologies of the elevated ESR should be considered,<br />
such as <strong>in</strong>fections, connective tissue disease, renal disease (especially nephrotic<br />
syndrome and uremia) (Gruener, 1992), malignant neoplasm (21% of negative TAB<br />
were cancer <strong>in</strong> Hedges series), diabetes mellitus (Hedges, 1983), and diffuse dissem<strong>in</strong>ated<br />
atheroembolism (Coppetto, 1984). If the ESR (and=or CRP) are normal, a unilateral<br />
TAB is negative, and the patient has few or nonspecific constitutional symptoms,<br />
then the steroid therapy can be tapered or a contralateral TAB can be performed. If both<br />
TABs are negative, the steroids can be tapered.<br />
What Is the Evaluation of the Patient with a<br />
Low <strong>Cl<strong>in</strong>ical</strong> Suspicion for GCA?<br />
In patients with low cl<strong>in</strong>ical suspicion for GCA (e.g., typical AION <strong>in</strong> a known<br />
vasculopathic patient with no constitutional signs or symptoms), alternative etiologies<br />
(e.g., <strong>in</strong>fection, <strong>in</strong>flammation, collagen vascular disease, underly<strong>in</strong>g malignancy,<br />
diabetes) for a high or even borderl<strong>in</strong>e high ESR should be <strong>in</strong>vestigated (Hedges,<br />
1983). Bedell and Bush suggested that patients with markedly elevated ESR (i.e.,<br />
ESR >100 mm=hour) should be evaluated for underly<strong>in</strong>g disease (Bedell, 1985). Based<br />
<strong>in</strong> part on these recommendations, we suggest the evaluation outl<strong>in</strong>ed <strong>in</strong> Table 5–7 for<br />
patients with an elevated ESR and a low cl<strong>in</strong>ical suspicion for GCA. In patients with a<br />
low cl<strong>in</strong>ical suspicion for GCA and an elevated ESR, a unilateral TAB could be<br />
performed and, if negative, no further evaluation or treatment for GCA is needed<br />
(class III, level C).<br />
Table 5–7. Laboratory Evaluation for Markedly Elevated ESR<br />
Complete blood count with differential<br />
Blood urea nitrogen and creat<strong>in</strong><strong>in</strong>e<br />
Alkal<strong>in</strong>e phosphatase<br />
Serum prote<strong>in</strong> electrophoresis<br />
Serum cholesterol<br />
Pregnancy test<br />
Chest radiography<br />
Consider mammogram and other evaluation for underly<strong>in</strong>g malignancy<br />
Ur<strong>in</strong>e analysis<br />
Purified prote<strong>in</strong> derivative (ppd) and control sk<strong>in</strong> test<strong>in</strong>g<br />
Guaiac tests of stools (six determ<strong>in</strong>ations)
106 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What Is the Preferred Treatment Regimen<br />
for GCA?<br />
Untreated GCA may result <strong>in</strong> significant visual loss <strong>in</strong> one or both eyes. Therefore, it is<br />
imperative that corticosteroid therapy beg<strong>in</strong> immediately upon cl<strong>in</strong>ical suspicion of<br />
GCA (class II, level B) to prevent visual loss (i.e., before TAB and laboratory confirmation).<br />
Most authors have recommended an <strong>in</strong>itial dose of oral prednisone of 1.0 mg=kg<br />
to 1.5 mg=kg=d (60 to 100 mg per day) (Laidlaw, 1990; Lundberg, 1990; Myles, 1992;<br />
Weisman, 1995) (class III, level C). Although some authors (Aiello, 1993) have reported<br />
that an <strong>in</strong>itial lower dose of 40 mg per day may be adequate to control cl<strong>in</strong>ical<br />
symptoms (Myles, 1992), patients with visual loss probably require higher doses.<br />
Some anecdotal cases of visual improvement have been reported follow<strong>in</strong>g <strong>in</strong>travenous<br />
(IV) corticosteroids for patients with visual loss and GCA (Diamond, 1991; Liu, 1994;<br />
Matzk<strong>in</strong>, 1992; Postel, 1993). Many patients note improvement <strong>in</strong> symptoms with<strong>in</strong> 1 to<br />
2 days of start<strong>in</strong>g steroid therapy, but other patients may cont<strong>in</strong>ue to experience<br />
symptoms of GCA <strong>in</strong>clud<strong>in</strong>g visual loss despite adequate corticosteroid therapy<br />
(Aiello, 1993; Evans, 1994; Liu, 1994; Rauser, 1995). A rapid or premature reduction<br />
of steroid therapy <strong>in</strong> GCA may also precipitate visual loss (Cullen, 1967). Occasionally<br />
new AION may occur <strong>in</strong> patients on ‘‘adequate’’ doses of corticosteroids (Rauser, 1995).<br />
Hwang et al reported a patient who developed bilateral ocular ischemic syndrome<br />
despite corticosteroid treatment (Hwang, 1999).<br />
Jover et al reported a randomized, double-bl<strong>in</strong>d, placebo-controlled study compar<strong>in</strong>g<br />
corticosteroids alone versus corticosteroids comb<strong>in</strong>ed with methotrexate <strong>in</strong> 42 patients<br />
with new-onset GCA (Jover, 2001). The prednisone plus methotrexate group experienced<br />
fewer relapses than the prednisone with placebo group, whereas the rate and<br />
severity of adverse events were similar <strong>in</strong> both groups. The authors suggested that<br />
methotrexate plus corticosteroids is a safe alternative to corticosteroids alone for GCA<br />
and is more effective <strong>in</strong> controll<strong>in</strong>g disease ( Jover, 2001).<br />
Stauton et al described a patient with GCA whose cl<strong>in</strong>ical condition deteriorated<br />
steadily with signs suggest<strong>in</strong>g an evolv<strong>in</strong>g vertebrobasilar stroke dur<strong>in</strong>g corticosteroid<br />
treatment (Stauton, 2000). The authors theorized that the cl<strong>in</strong>ical deterioration might<br />
have actually been <strong>in</strong>duced by the <strong>in</strong>itiation of the corticosteroids.<br />
Should Oral or IV Corticosteroids Be Used<br />
for GCA?<br />
Liu et al reported a 34% chance of visual improvement after corticosteroid therapy.<br />
Additional visual loss occurred <strong>in</strong> 7 of 41 (17%) patients despite corticosteroids (Liu,<br />
1994). Three patients experienced fellow-eye <strong>in</strong>volvement after oral therapy, but none of<br />
those treated with IV steroids developed fellow-eye <strong>in</strong>volvement. Based on these<br />
results, these authors recommended IV therapy (methylprednisolone 250 mg four<br />
times daily for 3 to 5 days) <strong>in</strong> patients with visual loss due to GCA (Liu, 1994).<br />
Matzk<strong>in</strong> et al reported visual recovery <strong>in</strong> two patients with central ret<strong>in</strong>al artery<br />
occlusions due to GCA after treatment with high-dose IV methylprednisolone (Matzk<strong>in</strong>,<br />
1992). Other authors have described anecdotal cases of visual improvement follow<strong>in</strong>g<br />
IV corticosteroids for patients with visual loss and GCA (Aiello, 1993; Diamond, 1991;
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 107<br />
Liu, 1994; Matzk<strong>in</strong>, 1992; Postel, 1993). Unfortunately, well-controlled prospective data<br />
on oral versus IV corticosteroids are lack<strong>in</strong>g (Cleark<strong>in</strong>, 1992a; Cornblath, 1997).<br />
Cornblath and Eggenberger reviewed charts from two centers and reviewed all<br />
previously reported cases of GCA treated with IV methylprednisolone (Cornblath,<br />
1997). Four patients with GCA exhibited severe progressive visual loss after at least<br />
48 hours of high-dose IV methylprednisolone, and a fifth patient had further loss <strong>in</strong> one<br />
eye and improvement <strong>in</strong> the other eye after 24 hours of treatment. They noted that <strong>in</strong><br />
previous reports of IV methylprednisolone for GCA, 4 patients lost vision and<br />
14 patients recovered vision. They concluded that the results of IV methylprednisolone<br />
treatment of patients with visual loss from GCA are similar to the results of treatment<br />
with oral corticosteroids, with IV methylprednisolone treatment be<strong>in</strong>g more costly and<br />
hav<strong>in</strong>g a small risk of sudden death. In a retrospective study, Chan et al reported visual<br />
acuity improvement <strong>in</strong> 21 of 73 (29%) patients treated promptly with oral or <strong>in</strong>travenous<br />
corticosteroids (Chan, 2001). There was an <strong>in</strong>creased likelihood of improved<br />
vision <strong>in</strong> the group given <strong>in</strong>travenous corticosteroids (40%) compared with those who<br />
received oral steroids (13%). Patients with GCA treated with oral or IV corticosteroids<br />
can have visual loss <strong>in</strong> a previously <strong>in</strong>volved eye or an un<strong>in</strong>volved eye, or can have<br />
visual recovery. Nevertheless, we favor IV steroids <strong>in</strong> patients with severe visual loss of<br />
less than 48 hours’ duration due to GCA, especially if there is bilateral <strong>in</strong>volvement, if<br />
the patient is monocular, or if the patient has lost vision dur<strong>in</strong>g oral steroid therapy<br />
(Hayreh, 1990; Slamovits, 1992) (class III, level C).<br />
Although the visual prognosis after AION or CRAO due to GCA is poor, significant<br />
visual improvement after steroid therapy has been reported <strong>in</strong> a small percentage<br />
of patients (Aiello, 1993; Cleark<strong>in</strong>, 1992a,b; Diamond, 1991). Aiello et al reviewed<br />
245 patients over a 5-year period at the Mayo Cl<strong>in</strong>ic (Aiello, 1993). Of these 245 patients,<br />
34 (14%) permanently lost vision due to GCA. After 5 years, the probability of visual<br />
loss after start<strong>in</strong>g steroid therapy (oral) was 1%. These authors reviewed an additional<br />
857 patients from the literature. Of these 857 patients, 174 (20%) lost vision due to GCA,<br />
and 31 of these 174 patients had visual loss or progression on steroid therapy.<br />
Kupersmith et al studied 22 patients with GCA, 7 of whom (9 eyes) had ischemic<br />
optic nerve <strong>in</strong>jury (Kupersmith, 1999). Four eyes had improved visual acuity of two<br />
l<strong>in</strong>es or more with<strong>in</strong> 1 year of start<strong>in</strong>g corticosteroids, and no patient developed visual<br />
loss as the steroids were reduced. At 1 year, visual acuity, contrast sensitivity, color<br />
vision, and threshold perimetry were not significantly different from 4- to 5-week<br />
determ<strong>in</strong>ations. At 1 year, no significant cataractous or glaucomatous changes were<br />
noted. The authors concluded that patients with GCA-related visual loss can improve<br />
with treatment (start<strong>in</strong>g doses 60 to 1000 mg per day with reduction to daily doses of<br />
40 to 50 mg per day given for 4 to 6 weeks) and that gradual reduction of dose<br />
thereafter, as cl<strong>in</strong>ically permitted, did not result <strong>in</strong> delayed visual loss. There were no<br />
significant dose-related ophthalmic complications.<br />
Gonzalez-Gay et al noted visual <strong>in</strong>volvement <strong>in</strong> 69 of 239 patients with GCA with<br />
predictors of permanent visual loss <strong>in</strong>clud<strong>in</strong>g transient visual loss, jaw claudications,<br />
normal liver enzymes, and absence of constitutional syndrome (Gonzalez-Gay, 1998a).<br />
Partial improvement of vision was noted <strong>in</strong> eight patients, with the only predictor of<br />
improvement be<strong>in</strong>g early corticosteroid treatment (oral or <strong>in</strong>travenous) with<strong>in</strong> the first<br />
day of visual loss.<br />
Consultation with an <strong>in</strong>ternist or rheumatologist is recommended for the detection of<br />
constitutional signs, monitor<strong>in</strong>g of ESR, and management of steroid therapy and
108 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
potential side effects <strong>in</strong> patients with GCA (Mizen, 1991). Hunder emphasized that the<br />
goal of therapy is to use the lowest dose of steroid over the shortest period of time to<br />
adequately control the symptoms of GCA (Hunder, 1990a). Unfortunately, the dosage<br />
and duration of therapy are variable among patients and must be determ<strong>in</strong>ed on an<br />
<strong>in</strong>dividual and empirical basis (Hunder, 1990a). Gradual taper<strong>in</strong>g of the steroid dose by<br />
10% per week as long as symptoms and ESR rema<strong>in</strong> stable is a reasonable approach.<br />
Most patients can be tapered off steroids with<strong>in</strong> 1 year, but some patients (especially<br />
those with neurologic or other systemic symptoms) may require prolonged (years) or<br />
<strong>in</strong>def<strong>in</strong>ite therapy. Recurrences may also occur years later. Turner et al reported a<br />
normalization of the ESR <strong>in</strong> 87% of 47 patients with<strong>in</strong> 4 weeks of treatment (Turner,<br />
1974). Huston et al reported a duration of steroid treatment of 1 to 77 months (median 7<br />
months) (Huston, 1978). Cullen and Coleiro felt that an ESR of less than 20 mm=hour<br />
was a desirable goal (Cullen, 1976). Delecoeullerie et al reported a mean duration of<br />
therapy of 30.9 months <strong>in</strong> 210 patients (Delecoeullerie, 1988), but <strong>An</strong>derson et al<br />
reported an average duration of 5 years (<strong>An</strong>derson, 1986). Taper<strong>in</strong>g of steroids is<br />
equally important as start<strong>in</strong>g therapy <strong>in</strong> GCA because the risk of fracture is <strong>in</strong>creased<br />
sixfold and that of cataract formation fourfold after 5 years of steroid therapy (Robb-<br />
Nicholson, 1988). Recurrent symptoms and=or ESR elevation may prompt <strong>in</strong>creas<strong>in</strong>g<br />
the steroid dosage, but cl<strong>in</strong>ical symptoms may be a better <strong>in</strong>dicator than the ESR alone<br />
(Britta<strong>in</strong>, 1991). As corticosteroid doses are lowered, the ESR may rise and, if it <strong>in</strong>creases<br />
to above normal rates, the taper<strong>in</strong>g schedule may be <strong>in</strong>terrupted for 2 to 4 weeks to<br />
allow stabilization of the ESR. Although a rise <strong>in</strong> the ESR is often associated with cl<strong>in</strong>ical<br />
recurrence of GCA, ischemic complications may occur on steroid therapy despite a<br />
stable ESR. Every-other-day steroid therapy does not seem to sufficiently control disease<br />
activity but may eventually be used once active GCA is under control.<br />
If Major Steroid Complications Occur,<br />
Therapy Is Prolonged, or the Disease Is Still<br />
Active, What Should Be Done?<br />
A repeat TAB could be considered, but it subjects the patient to a second surgical<br />
procedure. In addition, as already stated, a negative TAB (first or second), especially<br />
after prolonged steroid therapy, does not exclude GCA. Nevertheless, Cohen reported<br />
13 patients with known GCA and a ris<strong>in</strong>g ESR with any attempt at taper<strong>in</strong>g steroid<br />
therapy (Cohen, 1973). N<strong>in</strong>e of these patients underwent a second TAB and one of those<br />
had a third TAB to determ<strong>in</strong>e if active GCA was present histologically or whether the<br />
ESR was elevated due to alternative etiologies. Steroid-related complications (e.g.,<br />
cush<strong>in</strong>goid appearance, melena, hematuria, osteoporosis) had developed <strong>in</strong> n<strong>in</strong>e<br />
patients. Five of the 10 second TABs (50%) cont<strong>in</strong>ued to show active <strong>in</strong>flammation,<br />
and patients were cont<strong>in</strong>ued on steroid therapy, whereas the five (50%) patients with<br />
<strong>in</strong>active TAB were tapered off steroids.<br />
If a second TAB is not performed or the cl<strong>in</strong>ical suspicion for active disease rema<strong>in</strong>s<br />
high, then patients with significant contra<strong>in</strong>dications to steroid therapy, those who fail<br />
steroid therapy (Wilke, 1995), and those who develop steroid complications may benefit<br />
from other immunosuppressive agents. Steroid derivatives such as deflazacort<br />
(Cimm<strong>in</strong>o, 1994) and other immunosuppressive regimens such as cyclophosphamide
Figure 5–1. Evaluation of giant cell arteritis.<br />
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 109
110 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
(Cytoxan), azathiopr<strong>in</strong>e (Imuran), dapsone, and cyclospor<strong>in</strong>e (Sand-immune) have been<br />
employed <strong>in</strong> GCA, but there is little controlled cl<strong>in</strong>ical data regard<strong>in</strong>g their efficacy <strong>in</strong><br />
GCA (de Vita, 1992). No additive effect of cyclospor<strong>in</strong>e compared to corticosteroids<br />
alone was apparent <strong>in</strong> 22 patients with GCA requir<strong>in</strong>g more than 5 mg per day of<br />
prednisone 1 year after onset (Schaufelberger, 1998). Several authors (Hernandez-<br />
Garcia, 1994; van der Veen, 1996) have reported variable responses to methotrexate <strong>in</strong><br />
addition to steroid therapy <strong>in</strong> GCA, and this agent may prove to be a useful steroidspar<strong>in</strong>g<br />
medication for GCA <strong>in</strong> the future. A double-bl<strong>in</strong>d study of methotrexate <strong>in</strong><br />
GCA is under way (Langford, 1997). In addition, DeSilva and Hazleman reported the<br />
use of azathiopr<strong>in</strong>e <strong>in</strong> a double-bl<strong>in</strong>d, placebo-controlled study. There was a statistically<br />
significant reduction <strong>in</strong> mean prednisolone dose after 52 weeks <strong>in</strong> the azathiopr<strong>in</strong>etreated<br />
group (DeSilva, 1986).<br />
Our approach to the patient with GCA is outl<strong>in</strong>ed <strong>in</strong> Figure 5–1.<br />
References<br />
Aburahma AF, Thaxton L. (1996). Temporal arteritis: diagnostic and therapeutic considerations. Am Surg<br />
62:449–451.<br />
Aburahma AF, Wistberger TA. (1992). Diagnos<strong>in</strong>g giant cell temporal arteritis. West Virg<strong>in</strong>ia Med J 88:188–193.<br />
Achkar AA, Lie JT, Gabriel SE, Hunder GG. (1995). Giant cell arteritis <strong>in</strong>volv<strong>in</strong>g the facial artery. J Rheumatol<br />
22:360–362.<br />
Achkar AA, Lie JT, Hunder GG, et al. (1994). How does previous corticosteroid treatment affect the biopsy<br />
f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> giant cell (temporal arteritis). <strong>An</strong>n Intern Med 120:987.<br />
Ahmed I, Zaman M. (1999). Bilateral <strong>in</strong>ternuclear ophthalmoplegia: an <strong>in</strong>itial present<strong>in</strong>g sign of giant cell arteritis.<br />
J Am Geriatr Soc 47:734–736.<br />
Aiello PD, Trautmann JC, McPhee TJ, et al. (1993). Visual prognosis <strong>in</strong> giant cell arteritis. Ophthalmology<br />
100:550–555.<br />
Altlparmak MR, Tabak F, Pamuk ON, et al. (2001). Giant cell arteritis and secondary amyloidosis: the natural<br />
history. Scand J Rheumatol 30:114–116.<br />
<strong>An</strong>derson R, Malwall B, Bengtsson B-A. (1986). Long-term survival <strong>in</strong> giant cell arteritis <strong>in</strong>clud<strong>in</strong>g temporal<br />
arteritis and polymyalgia rheumatica. Acta Med Scand 220:361–364,<br />
<strong>An</strong>derson R, Malwall B, Bengtsson B-A. (1988). Long-term corticosteroid treatment <strong>in</strong> giant cell arteritis. Cl<strong>in</strong> Exp<br />
Immunol 73:82.<br />
Askari A, Jolobe OM, Shepherd DI. (1993). Internuclear ophthalmoplegia and Horner’s syndrome due to<br />
presumed giant cell arteritis. J R Soc Med 86:362.<br />
Astion ML, Wener MH, Thomas RG, Hunder GG, Bloch DA. (1994). Application of neural networks to the<br />
classification of giant cell arteritis. Arthritis Rheum 37:760–770.<br />
Barton JJ, Corbett JJ. (1991). <strong>Neuro</strong>-ophthalmologic vascular emergencies <strong>in</strong> the elderly. Cl<strong>in</strong> Geriatr Med<br />
7:525–548.<br />
Beckman RL, Hartmann BM. (1990). The use of a Doppler flow meter to identify the course of the temporal artery<br />
(letter). J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:304.<br />
Bedell SE, Bush BT. (1985). Erythrocyte sedimentation rate: from folklore to facts. Am J Med 78:1001–1009.<br />
Berlit P. (1992). <strong>Cl<strong>in</strong>ical</strong> and laboratory f<strong>in</strong>d<strong>in</strong>gs with giant cell arteritis. J <strong>Neuro</strong>l Sci 111:1–12.<br />
Bhatti MT, Furman J, Gupta S, et al. (2001a). Superficial temporal artery biopsy diagnostic for lung carc<strong>in</strong>oma.<br />
Am J Ophthalmol 132:135–138.<br />
Bhatti MT, Goldste<strong>in</strong> MH. (2001b). Facial nerve <strong>in</strong>jury follow<strong>in</strong>g superficial temporal artery biopsy. Dermatol Surg<br />
27:15–17.<br />
Birt CM, Slomovic A, Motolko M, Buncic R. (1994). <strong>An</strong>terior segment ischemia <strong>in</strong> giant cell arteritis.<br />
Can J Ophthalmol 29:93–94.<br />
Bisgard C, Sloth H, Keid<strong>in</strong>g N, Juel K. (1991). Excess mortality <strong>in</strong> giant cell arteritis. J Intern Med 230:119–123.<br />
Bondeson J, Asman P. (1997). Giant cell arteritis present<strong>in</strong>g with oculomotor nerve palsy. Scand J Rheumatol<br />
26:327–328.<br />
Borruat FX, Bogousslavsky J, Uffer S, Kla<strong>in</strong>guti G, Schatz NJ. (1993). Orbital <strong>in</strong>farction syndrome. Ophthalmology<br />
100:562–568.
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 111<br />
Bosch X, Font J, Mirapeix E, et al. (1991). <strong>An</strong>t<strong>in</strong>eutrophil cytoplasmic antibodies <strong>in</strong> giant cell arteritis (letter).<br />
J Rheumatol 18:787.<br />
Botella-Estrada R, Sammart<strong>in</strong> O, Mart<strong>in</strong>ez V, et al. (1999). Magnetic resonance angiography <strong>in</strong> the diagnosis of a<br />
case of giant cell arteritis manifest<strong>in</strong>g as scalp necrosis. Arch Dermatol 135:769–771.<br />
Bottiger LE, Svedberg CA. (1967). Normal erythrocyte sedimentation rate and age. Br Med J 2:85–87.<br />
Boyd RV, Hoffbrand BI. (1966). Erythrocyte sedimentation rate <strong>in</strong> elderly hospital <strong>in</strong>-patients. Br Med J 1:901–902.<br />
Boyev L, Miller NR, Green WR. (1999). Efficacy of unilateral versus bilateral temporal artery biopsies for the<br />
diagnosis of giant cell arteritis. Am J Ophthalmol 128:211–215.<br />
Brigden M. (1998). The erythrocyte sedimentation rate: still a helpful test when used judiciously. Postgrad Med<br />
103:257–262.<br />
Brilakis H, Lee AG. (1998). Ophthalmoplegia <strong>in</strong> treated polymyalgia rheumatica and healed giant cell arteritis.<br />
Strabismus 6:71–75.<br />
Britta<strong>in</strong> GP, McIlwa<strong>in</strong>e GG, Bell JA, Gibson JM. (1991). Plasma viscosity or erythrocyte sedimentation rate <strong>in</strong> the<br />
diagnosis of giant cell arteritis? Br J Ophthalmol 75:656–659.<br />
Buchb<strong>in</strong>der R, Detsky AS. (1992). Management of suspected giant cell arteritis: a decision analysis. J Rheumatol<br />
19:1220–1228.<br />
Burton EA, W<strong>in</strong>er JB, Barber PC. (1999). Giant cell arteritis of the cervical radicular vessels present<strong>in</strong>g with<br />
diaphragmatic weakness. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 67:223–226.<br />
Butt Z, Cullen JF, Mutlukan E. (1991). Pattern of arterial <strong>in</strong>volvement of the head, neck and eyes <strong>in</strong> giant cell<br />
arteritis: three case reports. Br J Ophthalmol 75:368–371.<br />
Caselli RJ. (1990). Giant cell (temporal) arteritis: a treatable cause of multi-<strong>in</strong>farct dementia. <strong>Neuro</strong>logy 40:753–755.<br />
Caselli RJ, Hunder GG. (1993). <strong>Neuro</strong>logic aspects of giant cell (temporal) arteritis. Rheum Dis Cl<strong>in</strong> North Am<br />
19:941–953.<br />
Caselli RJ, Hunder GG, Whisnant JP. (1988). <strong>Neuro</strong>logic disease <strong>in</strong> biopsy-proven giant cell arteritis. <strong>Neuro</strong>logy<br />
38:352–359.<br />
Casson RJ, Flem<strong>in</strong>g <strong>FK</strong>, Shaikh A, James B. (2001). Bilateral ocular ischemic syndrome secondary to giant cell<br />
arteritis. Arch Ophthalmol 119:306–307.<br />
Chambers W, Bernard<strong>in</strong>o V. (1988). Specimen length <strong>in</strong> temporal artery biopsies. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
8:121–125.<br />
Chan CCK, Pa<strong>in</strong>e M, O’Day J. (2001). Steroid management <strong>in</strong> giant cell arteritis. Br J Ophthalmol 85:1061–1064.<br />
Charness ME, Liu GT. (1991). Central ret<strong>in</strong>al artery occlusion <strong>in</strong> giant cell arteritis. Treatment with nitroglycer<strong>in</strong>.<br />
<strong>Neuro</strong>logy 41:1698–1699.<br />
Chertok P, Leroux JL, LeMarchand M, et al. (1990). Orbital pseudotumor <strong>in</strong> temporal arteritis revealed by<br />
computed tomography. Cl<strong>in</strong> Exp Rheumatol 8:587–589.<br />
Chmelewski WL, McKnight KM, Agudelo CA, Wise CM. (1992). Present<strong>in</strong>g features and outcome <strong>in</strong> patients<br />
undergo<strong>in</strong>g temporal artery biopsy: a review of 98 patients. Arch Intern Med 152:1690–1695.<br />
Cid MC, Font C, Oristrell J, et al. (1998). Association between strong <strong>in</strong>flammatory response and low risk of<br />
develop<strong>in</strong>g visual loss and other cranial ischemic complications <strong>in</strong> giant cell (temporal) arteritis. Arthritis<br />
Rheum 41:26–32.<br />
Cimm<strong>in</strong>o MA, Moggiana G, Montecucco C, et al. (1994). Long term treatment of polymyalgia rheumatica with<br />
deflazacort. <strong>An</strong>n Rheum Dis 43:331.<br />
Cleark<strong>in</strong> LG. (1992a). IV steroids for central ret<strong>in</strong>al artery occlusion <strong>in</strong> giant-cell arteritis (letter). Ophthalmology<br />
99:1482–1483.<br />
Cleark<strong>in</strong> LG, Caballero J. (1992b). Recovery of visual function <strong>in</strong> anterior ischemic optic neuropathy due to giant<br />
cell arteritis. Am J Med 92:703–704.<br />
Cleark<strong>in</strong> LG, Watts MT. (1991). How to perform a temporal artery biopsy. Br J Hosp Med 46:172–174.<br />
Cohen DN. (1973). Temporal arteritis: improvement <strong>in</strong> visual prognosis and management with repeat biopsies.<br />
Trans Am Acad Ophthalmol Otolaryngol 77:74–85.<br />
Combe B, Sany J, Le Quellac A, et al. (1998). Distribution of HLA-DRB1 alleles of patients with polymyalgia<br />
rheumatica and giant cell arteritis <strong>in</strong> a Mediterranean population. J Rheumatol 25:94–98.<br />
Coppetto JR, Lessell S, Lessell IM, Greco TP, Eisenberg MS. (1984). Diffuse dissem<strong>in</strong>ated atheroembolism. Three<br />
cases with neuro-ophthalmic manifestations. Arch Ophthalmol 102:224–228.<br />
Coppetto JR, Monteiro M. (1990). Diagnosis of highly occult temporal arteritis by repeat temporal artery biopsies.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 10:217–218.<br />
Cornblath WT, Eggenberger ER. (1997). Progressive visual loss from giant cell arteritis despite high-dose<br />
<strong>in</strong>travenous methylprednisolone. Ophthalmology 104:854–858.<br />
Costa MM, Romeu JC, da Silva P, de Queiroz V. (1995). Successful treatment of ischaemic optic neuropathy<br />
secondary to giant cell arteritis with <strong>in</strong>travenous pulse of methylprednisolone (letter). Cl<strong>in</strong> Rheumatol<br />
14:713–714.
112 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Cullen JF. (1967). Occult temporal arteritis. A common cause of bl<strong>in</strong>dness <strong>in</strong> old age. Br J Ophthalmol 51:513–525.<br />
Cullen JF, Coleiro JA. (1976). Ophthalmic complications of giant cell arteritis. Surv Ophthalmol 20:247–260.<br />
Currey J. (1997). Scalp necrosis <strong>in</strong> giant cell arteritis and review of the literature. Br J Rheumatol 36:814–816.<br />
Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Eagle RC Jr, et al. (2000). Low diagnostic yield with second biopsies <strong>in</strong> suspected<br />
giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 20:213–215.<br />
Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Sergott RC. (2001). The prevalence of cupp<strong>in</strong>g <strong>in</strong> end-stage arteritic and nonarteritic<br />
anterior ischemic optic neuropathy. Ophthalmology 108:593–598.<br />
de Heide LJ, Talsma MA. (1999). Giant-cell arteritis present<strong>in</strong>g as an orbital pseudotumor. Neth J Med 55:196–198.<br />
Delecoeullerie G, Joly P, DeLara AC, et al. (1988). Polymyalgia rheumatica and temporal arteritis: a retrospective<br />
analysis of prognostic features and different corticosteroid regimens (11 year survey of 210 patients). <strong>An</strong>n<br />
Rheum Dis 47:733–739.<br />
Dennis RH II, Ransome JR. (1996). Giant cell arteritis present<strong>in</strong>g as a carpal tunnel syndrome. J Natl Med Assoc<br />
88:524–525.<br />
DeSilva M, Hazleman BL. (1986). Azathiopr<strong>in</strong>e <strong>in</strong> giant cell arteritis=polymyalgia rheumatica: a double-bl<strong>in</strong>d<br />
study. <strong>An</strong>n Rheum Dis 45:136–138.<br />
Desmet GD, Knockaert DC, Bobbaers HJ. (1990). Temporal arteritis: the silent presentation and delay <strong>in</strong> diagnosis.<br />
J Intern Med 227:237–240.<br />
Desmond J, Hussa<strong>in</strong> ST, Col<strong>in</strong> JF. (1999). Giant cell arteritis as a cause of <strong>in</strong>termittent claudications. Hosp Med<br />
60:302.<br />
de Vita S, Tavoni A, Jeracitano G, Gemignani G, Dolcher MP, Bombardieri S. (1992). Treatment of giant cell<br />
arteritis with cyclophosphamide pulses (letter). J Intern Med 232:373–375.<br />
Diamond JP. (1991). Treatable bl<strong>in</strong>dness <strong>in</strong> temporal arteritis. Br J Ophthalmol 75:432.<br />
Diamond JP. (1993). IV steroid treatment <strong>in</strong> giant cell arteritis (letter). Ophthalmology 100:291–292.<br />
DiBartolomeo AG, Brick JE. (1992). Giant cell arteritis and polymyalgia rheumatica. Postgrad Med 91:107–109.<br />
Dick AD, Millar A, Johnson N. (1991). Bra<strong>in</strong>stem vascular accidents and cranial arteritis. Scott Med J 36:85.<br />
Diego M, Margo CE. (1998). Postural vision loss <strong>in</strong> giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 18:124–126.<br />
Egge K, Midtbo A, Westby R. (1996). Arteritis temporalis. Acta Ophthalmol 44:49–56.<br />
Eggenberger E. (1998). Eight-and-a-half syndrome: one-and-a-half syndrome plus cranial nerve VII palsy.<br />
J <strong>Neuro</strong>ophthalmol 18:114–116.<br />
Ellis JD, Munro P, McGettrick P. (1994). Bl<strong>in</strong>dness with a normal erythrocyte sedimentation rate <strong>in</strong> giant cell<br />
arteritis. Br J Hosp Med 52:358–359.<br />
Evans JM, Batts KP, Hunder GG. (1994). Persistent giant cell arteritis despite corticosteroid treatment. Mayo Cl<strong>in</strong><br />
Proc 69:1060–1061.<br />
Evans JM, O’Fallon WM, Hunder GG. (1995). Increased <strong>in</strong>cidence of aortic aneurysm and dissection <strong>in</strong> giant cell<br />
(temporal) arteritis. A population-based study. <strong>An</strong>n Intern Med 122:502–507.<br />
Evans JM, Vukov LF, Hunder GG. (1993). Polymyalgia rheumatica and giant cell arteritis <strong>in</strong> emergency<br />
department patients. <strong>An</strong>n Emerg Med 22:1633–1635.<br />
Fernandez-Herlihy L. (1988). Temporal arteritis: cl<strong>in</strong>ical aids to diagnosis. J Rheumatol 15:1797.<br />
Fife A, Dorrell L, Snow MH, Ong EL. (1994). Giant cell arteritis—a cause of pyrexia of unknown orig<strong>in</strong>. Scott Med J<br />
39:114–115.<br />
F<strong>in</strong>elli PF. (1997). Alternat<strong>in</strong>g amaurosis fugax and temporal arteritis. Am J Ophthalmol 123:850–851.<br />
F<strong>in</strong>eman MS, Sav<strong>in</strong>o PJ, Federman JL, Eagle RC, Jr. (1996). Branch ret<strong>in</strong>al artery occlusion as the <strong>in</strong>itial sign of<br />
giant cell arteritis. Am J Ophthalmol 122:428–430.<br />
Freddo T, Price M, Kase C, Goldste<strong>in</strong> MP. (1999). Myocardial <strong>in</strong>farction and coronary artery <strong>in</strong>volvement <strong>in</strong> giant<br />
cell arteritis. Optom Vis Sci 76:14–18.<br />
Gabriel S, Espy M, Erdman DD, et al. (1999). The role of parvovirus B19 <strong>in</strong> the pathogenesis of giant cell arteritis.<br />
A prelim<strong>in</strong>ary evaluation. Arthritis Rheum 42:1255–1258.<br />
Gabriel SE, O’Fallon WM, Achkar AA, Lie JT, Hunder GG. (1995). The use of cl<strong>in</strong>ical characteristics to predict the<br />
results of temporal artery biopsy among patients with suspected giant cell arteritis. J Rheumatol 22:93–96.<br />
Galetta SL, Balcer LJ, Lieberman AP, et al. (1997a). Refractory giant cell arteritis with sp<strong>in</strong>al cord <strong>in</strong>farction.<br />
<strong>Neuro</strong>logy 49:1720–1723.<br />
Galetta SL, Balcer LJ, Liu GT. (1997b). Giant cell arteritis with unusual flow-related neuro-ophthalmologic<br />
manifestations. <strong>Neuro</strong>logy 49:1463–1465.<br />
Galetta SL, Raps EC, Wulc AE, et al. (1990). Conjugal temporal arteritis. <strong>Neuro</strong>logy 40:1839–1842.<br />
Garcia Vazquez JM, Carreira JM, Seoane C, Vidal JJ. (1999). Superior and <strong>in</strong>ferior limb ischemia <strong>in</strong> giant cell<br />
arteritis: angiography follow-up. Cl<strong>in</strong> Rheumatol 18:61–65.<br />
Gaynes BI. (1994). Occult giant cell arteritis: a diagnosis of suspicion. J Am Optom Assoc 65:564–571.
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 113<br />
Genereau T, Lotholary O, Biousse V, Guillev<strong>in</strong> L. (1999). Numb ch<strong>in</strong> syndrome as first sign of temporal arteritis.<br />
J Rheumatol 26:1425–1426.<br />
Gersbach P, Lang H, Kipfer B, Meyer R, Schupbach P. (1993). Impend<strong>in</strong>g rupture of the ascend<strong>in</strong>g aorta due to<br />
giant cell arteritis. Eur J Cardiothorac Surg 7:667–670.<br />
Glutz von Blotsheim S, Borruat FX. (1997). <strong>Neuro</strong>-ophthalmic complications of biopsy-proven giant cell arteritis.<br />
Eur J Ophthalmol 7:375–382.<br />
Goadsby PJ, Mossman S. (1991). Giant cell arteritis and ophthalmoplegia (letter). Aust NZ J Med 21:930.<br />
Goh KY, Lim TH. (2000). Giant cell arteritis caus<strong>in</strong>g bilateral sequential anterior ischaemic optic neuropathy—a<br />
case report. S<strong>in</strong>gapore Med J 41:32–33.<br />
Goldberg RT. (1983). Ocular muscle paresis and cranial arteritis—an unusual case. <strong>An</strong>n Ophthalmol 15:240–243.<br />
Gonzalez-Alegre P, Ruiz-Lopez AD, Abarca-Costalago M, Gonzalez-Santos P. (2001). Increment of the platelet<br />
count <strong>in</strong> temporal arteritis: response to therapy and ischemic complications. Eur <strong>Neuro</strong>l 45:43–45.<br />
Gonzalez-Gay MA, Blanco R, Rodriguez-Valverde V, et al. (1998a). Permanent visual loss and cerebrovascular<br />
accidents <strong>in</strong> giant cell arteritis. Predictors and response to treatment. Arthritis Rheum 41:1497–1504.<br />
Gonzalez-Gay MA, Garcia-Porrua C. (1998b). Carotid tenderness: an om<strong>in</strong>ous sign of giant cell arteritis? Scand J<br />
Rheumatol 27:154–156.<br />
Gonzalez-Gay MA, Garcia-Porrua C, Llorca J, et al. (2001). Biopsy-proven giant cell arteritis: cl<strong>in</strong>ical spectrum and<br />
predictive value for positive temporal artery biopsy. Sem<strong>in</strong> Arthritis Rheum 30:249–256.<br />
Gout O, Viala K, Lyon-Caen O. (1998). Giant cell arteritis and Vernet’s syndrome. <strong>An</strong>n <strong>Neuro</strong>l 50:1862–1864.<br />
Graham E. (1980). Survival <strong>in</strong> temporal arteritis. Trans Ophthalmol Soc UK 100:108–110.<br />
Grodum E, Petersen HA. (1990). Temporal arteritis with normal erythrocyte sedimentation rates present<strong>in</strong>g as<br />
occipital neuralgia. J Intern Med 227:279–280.<br />
Gros F, Maillefert JF, Beh<strong>in</strong> A, et al. (1998). Giant cell arteritis with ocular complications discovered simultaneously<br />
<strong>in</strong> two sisters. Cl<strong>in</strong> Rheumatol 17:58–61.<br />
Grosser SJ, Reddy RK, Tomsak RL, Katz<strong>in</strong> WE. (1999). Temporal arteritis <strong>in</strong> African Americans.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 21:25–31.<br />
Grosvenor T, Mal<strong>in</strong>ovsky V, Gelv<strong>in</strong> J, Tonekaboni K. (1993). Diagnosis and management of temporal arteritis: a<br />
review and case report. Optom Vis Sci 70:771–777.<br />
Gruener G, Merchut MP. (1992). Renal causes of elevated sedimentation rate <strong>in</strong> suspected temporal arteritis. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 12:272–274.<br />
Gudmundsson M, Nordborg E, Bengtsson B-A, et al. (1993). Plasma viscosity <strong>in</strong> giant cell arteritis as a predictor of<br />
disease activity. <strong>An</strong>n Rheum Dis 52:104.<br />
Guevara RA, Newman NJ, Grossniklaus HE. (1998). Positive temporal artery biopsy 6 months after prednisone<br />
treatment. Arch Ophthalmol 116:1252–1253.<br />
Gur H, Whrenfeld M, Izsak E. (1996). Pleural effusion as a present<strong>in</strong>g manifestation of giant cell arteritis. Cl<strong>in</strong><br />
Rheumatol 15:200–203.<br />
Haist SA. (1985). Stroke after temporal artery biopsy. Mayo Cl<strong>in</strong> Proc 60:538.<br />
Hall S, Hunder GG. (1984) Is temporal artery biopsy prudent? Mayo Cl<strong>in</strong> Proc 59:793–796.<br />
Hall S, Lie JT, Kurland LT, Persell<strong>in</strong> S, O’Brien PC, Hunder GG. (1983). The therapeutic impact of temporal artery<br />
biopsy. Lancet 2:1217–1220.<br />
Hamano K, Gohra H, Katoh T, et al. (1999). <strong>An</strong> ascend<strong>in</strong>g aortic aneurysm caused by giant cell arteritis: report of a<br />
case. Surg Today 29:957–959.<br />
Hamed LM, Guy JR, Moster ML, Bosley T. (1992). Giant cell arteritis <strong>in</strong> the ocular ischemic syndrome. Am J<br />
Ophthalmol 113:702–705.<br />
Hansen BL, Junker P. (1995). Giant cell arteritis present<strong>in</strong>g with ischaemic sk<strong>in</strong> lesions of the neck. Br J Rheumatol<br />
34:1182–1184.<br />
Hayreh SS. (1990). <strong>An</strong>terior ischaemic optic neuropathy. Differentiation of arteritic from nonarteritic type and its<br />
management. Eye 4:25–41.<br />
Hayreh SS. (1991). Ophthalmic features of giant cell arteritis. Baillieres Cl<strong>in</strong> Rheumatol 5:431–459.<br />
Hayreh SS. (1998a). Ocular manifestations of giant cell arteritis (reply to correspondence). Am J Ophthalmol<br />
126:742–744.<br />
Hayreh SS, Jonas JB. (2001). Optic disc morphology after arteritic anterior ischemic optic neuropathy.<br />
Ophthalmology 108:1586–1594.<br />
Hayreh SS, Podhajsky PA, Raman R, Zimmerman B. (1997). Giant cell arteritis: validity and reliability of various<br />
diagnostic criteria. Am J Ophthalmol 123:285–296.<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1998b). Ocular manifestations of giant cell arteritis. Am J Ophthalmol<br />
125:509–520.
114 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1998c). Occult giant cell arteritis: ocular manifestations. Am J<br />
Ophthalmol 125:521–526.<br />
Heathcote JG. (1999). Update <strong>in</strong> pathology: temporal arteritis and its ocular manifestations. Can J Ophthalmol<br />
34:63–68.<br />
Hedges TR III. (1992). The importance of temporal artery biopsy <strong>in</strong> the diagnosis of giant cell arteritis. Arch<br />
Ophthalmol 110:1377.<br />
Hedges TR III, Gieger GL, Albert DM. (1983). The cl<strong>in</strong>ical value of negative temporal artery biopsy specimens.<br />
Arch Ophthalmol 101:1251–1254.<br />
Hellman DB. (1993). Immunopathogenesis, diagnosis, and treatment of giant cell arteritis, temporal arteritis,<br />
polymyalgia rheumatica and Takayasu’s arteritis. Curr Op<strong>in</strong> Rheum 5:25–32.<br />
Hernandez-Garcia C, Soriano C, Morado C, et al. (1994). Methotrexate treatment <strong>in</strong> the management of giant cell<br />
arteritis. Scand J Rheum 23:295–298.<br />
Ho AC, Sergott RC, Regillo CD, et al. (1994). Color Doppler hemodynamics of giant cell arteritis. Arch Ophthalmol<br />
112:938–945.<br />
Hunder GG. (1990a). Giant cell (temporal) arteritis. Rheum Dis Cl<strong>in</strong> North Am 16:399.<br />
Hunder GG, Bloch DA, Michel BA, et al. (1990b). The American College of Rheumatology, 1990 criteria for the<br />
classification of giant cell arteritis. Arthritis Rheum 33:1122–1128.<br />
Husse<strong>in</strong> AMA, Hay N. (1990). Case report: cerebral arteritis with unusual distribution. Cl<strong>in</strong> Radiol 41:353.<br />
Huston KA, Hunder GG, Lie JT, Kennedy RH, Elveback LR. (1978). Temporal arteritis: a 25-year epidemiologic,<br />
cl<strong>in</strong>ical, and pathologic study. <strong>An</strong>n Intern Med 88:162–167.<br />
Hwang J-M, Girk<strong>in</strong> CA, Perry JD, et al. (1999). Bilateral ocular ischemic syndrome secondary to giant cell arteritis<br />
progress<strong>in</strong>g despite corticosteroid treatment. Am J Ophthalmol 127:102–104.<br />
Ilan Y, Ben-Chetrit E. (1993). Liver <strong>in</strong>volvement <strong>in</strong> giant cell arteritis. Cl<strong>in</strong> Rheumatol 12:219–222.<br />
Imakita M, Yutani C, Ishibashi-Ueda H. (1993). Giant cell arteritis <strong>in</strong>volv<strong>in</strong>g the cerebral artery. Arch Pathol Lab<br />
Med 117:729–733.<br />
Jacobson DM. (2000). Divergence <strong>in</strong>sufficiency revisited. Natural history of idiopathic cases and neurologic<br />
associations. Arch Ophthalmol 118:1237–1241.<br />
Jacobson DM, Slamovits TL. (1987). Erythrocyte sedimentation rate and its relationship to hematocrit <strong>in</strong> giant cell<br />
arteritis. Arch Ophthalmol 105:965–967.<br />
Jiménez-Jiménez FJ, Garcia-Albea E, Zurdo M, et al. (1998). Giant cell arteritis present<strong>in</strong>g as cluster headache.<br />
<strong>Neuro</strong>logy 51:1787–1788.<br />
Joelson E, Ruthrauff B, Ali F, et al. (2000). Multifocal dural enhancement associated with temporal arteritis.<br />
Arch <strong>Neuro</strong>l 57:119–122.<br />
Johnston JL, Thomson GT, Sharpe JA, Inman RD. (1992). Internuclear ophthalmoplegia <strong>in</strong> giant cell arteritis<br />
(letter). J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 55:84–85.<br />
Jover JA, Hernandez-Garcia C, Morado IC, et al. (2001). Comb<strong>in</strong>ed treatment of giant-cell arteritis with<br />
methotrexate and prednisone. A randomized, double-bl<strong>in</strong>d, placebo-controlled trial. <strong>An</strong>n Intern Med<br />
134:106–114.<br />
Jundt JW, Mock D. (1991). Temporal arteritis with normal erythrocyte sedimentation rates present<strong>in</strong>g as occipital<br />
neuralgia. Arthritis Rheum 34:217–219.<br />
Kachroo A, Tello C, Bais R, Panush RS. (1996). Giant cell arteritis: diagnosis and management. Bull Rheum Dis<br />
45:2–5.<br />
Kattah JC, Cupps T, Manz HJ, el Khodary A, Caputy A. (1991). Occipital artery biopsy: a diagnostic alternative <strong>in</strong><br />
giant cell arteritis. <strong>Neuro</strong>logy 41:949–950.<br />
Kattah JC, Mejico L, Chrousos GA, et al. (1999). Pathologic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a steroid-responsive optic nerve <strong>in</strong>farct <strong>in</strong><br />
giant-cell arteritis. <strong>Neuro</strong>logy 53:177–180.<br />
Kerleau JM, Levesque H, Delpech A, et al. (1994). Prevalence and evolution of anticardiolip<strong>in</strong> antibodies <strong>in</strong> giant<br />
cell arteritis dur<strong>in</strong>g corticosteroid therapy. A prospective study of 20 cases. Br J Rheumatol 33:648.<br />
Killer HE, Holtz DJ, Kaiser HJ, Laeng RH. (2000). Diplopia, ptosis, and hepatitis as present<strong>in</strong>g signs and<br />
symptoms of giant cell arteritis. Br J Ophthalmol 84:1318.<br />
Kle<strong>in</strong> RG, Campbell RJ, Hunder GG, Carney JA. (1976). Skip lesions <strong>in</strong> temporal arteritis. Mayo Cl<strong>in</strong> Proc<br />
51:504–510.<br />
Kontoyianni A, Maragou M, Alvanou E, et al. (1999). Unilateral distal extremity swell<strong>in</strong>g with pitt<strong>in</strong>g oedema <strong>in</strong><br />
giant cell arteritis. Cl<strong>in</strong> Rheumatol 18:82–84.<br />
Kranemann CF, Buys YM. (1997). Acute angle-closure glaucoma <strong>in</strong> giant cell arteritis. Can J Ophthalmol 32:389–391.<br />
Kupersmith MJ, Langer R, Mitnick H, et al. (1999). Visual performance <strong>in</strong> giant cell arteritis (temporal arteritis)<br />
after 1 year of therapy. Br J Ophthalmol 83:796–801.
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 115<br />
Kyle V, Hanzleman BL. (1993). The cl<strong>in</strong>ical and laboratory course of polymyalgia rheumatica=giant cell arteritis<br />
after the first two months of treatment. <strong>An</strong>n Rheum Dis 52:847–850.<br />
Lacomas D, Giuliani MJ, Wasko MC, Oddis CV. (1999). Giant cell arteritis present<strong>in</strong>g with proximal weakness and<br />
skeletal muscle vasculitis. Muscle Nerve 22:142–144.<br />
Lagrand WK, Hoogendoorn M, Bakker K, teVelde J, Labrie A. (1996). Aortoduodenal fistula as an unusual<br />
manifestation of giant-cell arteritis. Eur J Vasc Endovasc Surg 11:502–503.<br />
Laidlaw PAH, Smith PEM, Hudgson P. (1990). Orbital pseudotumor secondary to giant cell arteritis: an<br />
unreported condition. Br Med J 300:784.<br />
Lambert M, Weber A, Boland B, DePlaen JF, Donckier J. (1996). Large vessel vasculitis without temporal artery<br />
<strong>in</strong>volvement: isolated form of giant cell arteritis? Cl<strong>in</strong> Rheumatol 15:174–180.<br />
Langford CA, Sneller MC, Hoffman GS. (1997). Methotrexate use <strong>in</strong> systemic vasculitis. Rheum Dis Cl<strong>in</strong> North Am<br />
23:841–853.<br />
Lee AG. (1995). Jaw claudication: a sign of giant cell arteritis. J Am Dent Assoc 126:1028–1029.<br />
Lee AG, Eggenberger ER, Kaufman DI, Manrique C. (1999a). Optic nerve enhancement on magnetic resonance<br />
imag<strong>in</strong>g <strong>in</strong> arteritic ischemic optic neuropathy. J <strong>Neuro</strong>-ophthalmol 19:235–237.<br />
Lee AG, Tang RA, Feldon SE, et al. (2001). Orbital presentations of giant cell arteritis. Graefes Arch Cl<strong>in</strong> Exp<br />
Ophthalmol 239:509–513.<br />
Lee CC, Su WW, Hunder GG. (1999b). Dysarthria associated with giant cell arteritis. J Rheumatol 26:931–932.<br />
Lessell S. (1995). Surgery for ischemic optic neuropathy (editorial). Arch Ophthalmol 113:273–274.<br />
Lessell S. (2000). Bilateral temporal artery biopsies <strong>in</strong> giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 20:220–221.<br />
Lev<strong>in</strong> LA. (1995). Clarify<strong>in</strong>g the treatment of nonarteritic anterior ischemic optic neuropathy (editorial).<br />
JAMA 273:666–667.<br />
Levy MH, Margo CE. (1994). Temporal artery biopsy and sarcoidosis. Am J Ophthalmol 117:409–410.<br />
Lie JT. (1995a). Aortic and extracranial large vessel giant cell arteritis: a review of 72 cases with histopathologic<br />
documentation. Sem<strong>in</strong> Arthritis Rheum 24:422–431.<br />
Lie JT, Tokugawa DA. (1995b). Bilateral lower limb gangrene and stroke as <strong>in</strong>itial manifestations of systemic giant<br />
cell arteritis <strong>in</strong> an African-American. J Rheumatol 22:363–366.<br />
Lim KH, Liam CK, Vasudevan AE, Wong CM. (1999). Giant cell arteritis present<strong>in</strong>g as chronic cough and<br />
prolonged fever. Respirology 4:299–301.<br />
L<strong>in</strong> JL, Hsueh S. (1995). Giant cell arteritis <strong>in</strong>duced renal artery aneurysm (letter). Cl<strong>in</strong> Nephrol 43:66–68.<br />
L<strong>in</strong>coff NS, Erlich PD, Brass LS. (2000). Thrombocytosis <strong>in</strong> temporal arteritis. Ris<strong>in</strong>g platelet counts: a red flag for<br />
giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 20:67–72.<br />
Litw<strong>in</strong> MS, Henderson DR, Kirkham B. (1992). Normal sedimentation rates and giant cell arteritis. Arch Intern Med<br />
152:209.<br />
Liu GT, Glaser JS, Schatz NJ, Smith JL. (1994). Visual morbidity <strong>in</strong> giant cell arteritis. Ophthalmology 101:1779–1785.<br />
Liu G, Shupak R, Chiu BK. (1995). Aortic dissection <strong>in</strong> giant-cell arteritis. Sem<strong>in</strong> Arthritis Rheum 25:160–171.<br />
Liu NH, LaBree LD, Feldon SE, Rao NA. (2001). The epidemiology of giant cell arteritis. A 12-year retrospective<br />
study. Ophthalmology 108:1145–1149.<br />
Llorente Pendas S, DeVicente Rodriguez JC, et al. (1994). Tongue necrosis as a complication of giant cell arteritis.<br />
Oral Surg Oral Med Oral Pathol 78:448–451.<br />
Looney BD. (1999). Unilateral proptosis result<strong>in</strong>g from giant-cell arteritis. J Am Optom Assoc 70:443–449.<br />
Lundberg I, Hedfors E. (1990). Restricted dose and duration of corticosteroid treatment <strong>in</strong> patients with<br />
polymyalgia rheumatica and temporal arteritis. J Rheumatol 17:1340–1345.<br />
Mack HG, O’Day J, Currie JN. (1991) Delayed choroidal perfusion <strong>in</strong> giant cell arteritis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
11:221–227.<br />
MacLeod JD, Rizk SN. (1993). Cotton-wool spots <strong>in</strong> giant cell arteritis (letter). Eye 7:715–716.<br />
Mallia C, Coleiro B, Crockford M, Ellul B. (1999). Raynaud’s phenomenon caused by giant cell arteritis. Adv Exp<br />
Med Biol 455:517–520.<br />
Manna R, Latteri M, Cristiano G, et al. (1998). <strong>An</strong>ti-cardiolip<strong>in</strong> antibodies <strong>in</strong> giant cell arteritis and polymyalgia<br />
rheumatica: a study of 40 cases. Br J Rheumatol 315:549–550.<br />
Matteson EL, Gold KN, Block DA, Hunder GG. (1996). Long-term survival of patients with giant cell arteritis <strong>in</strong><br />
the American College of Rheumatology giant cell arteritis classification criteria cohort. Am J Med<br />
100:193–196.<br />
Matzk<strong>in</strong> DC, Slamovits TL, Sachs R, Burde RM. (1992). Visual recovery <strong>in</strong> two patients after <strong>in</strong>travenous<br />
methylprednisolone treatment of central ret<strong>in</strong>al artery occlusion secondary to giant-cell arteritis.<br />
Ophthalmology 99:68–71.<br />
McDonnell PJ, Moore GW, Miller NR, Hutch<strong>in</strong>s GM, Green WR. (1986). Temporal arteritis: a cl<strong>in</strong>icopathologic<br />
study. Ophthalmology 93:518–530.
116 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
McHugh NJ, James IE, Plant GT. (1990). <strong>An</strong>ticardiolip<strong>in</strong> and ant<strong>in</strong>eutrophil antibodies <strong>in</strong> giant cell arteritis.<br />
J Rheumatol 17:916–922.<br />
McLean CA, Gonzales MF, Dowl<strong>in</strong>g JP. (1993). Systemic giant cell arteritis and cerebellar <strong>in</strong>farction. Stroke<br />
24:899–902.<br />
Melberg NS, Grand MG, Diekert JP, et al. (1995). Cotton-wool spots and the early diagnosis of giant cell arteritis.<br />
Ophthalmology 102:1611–1614.<br />
Miller A, Green M. (1983). Simple rule for calculat<strong>in</strong>g normal erythrocyte sedimentation rate. Br Med J 286:266.<br />
Miller NR. (1991). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams & Wilk<strong>in</strong>s,<br />
pp. 2601–2627.<br />
Miller NR. (2000). Giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 20:219–221.<br />
Mitnich HJ, Tunick PA, Rotterdam H, et al. (1990). <strong>An</strong>temortem diagnosis of giant cell aortitis. J Rheumatol 17:708.<br />
Mizen TR. (1991). Giant cell arteritis: diagnostic and therapeutic considerations. Ophthalmol Cl<strong>in</strong> North Am 4:547–556.<br />
Myles AB, Perera T, Ridley MG. (1992). Prevention of bl<strong>in</strong>dness <strong>in</strong> giant cell arteritis by corticosteroid treatment.<br />
Br J Rheumatol 31:103–105.<br />
Nadeau SE. (1988). Temporal arteritis: a decision-analytical approach to temporal artery biopsy. Acta <strong>Neuro</strong>l Scand<br />
78:90–100.<br />
Neish PR, Sergent JS. (1991). Giant cell arteritis: a case with unusual neurologic manifestations and a normal<br />
sedimentation rate. Arch Intern Med 151:378–380.<br />
Nesher G, Sonnenblick M, Friedlander Y. (1994). <strong>An</strong>alysis of steroid related complications and mortality <strong>in</strong><br />
temporal arteritis: a 15 year survey of 43 patients. J Rheumatol 21:1283.<br />
N<strong>in</strong>et JP, Bachet P, Dumontet CM, et al. (1990). Subclavian and axillary <strong>in</strong>volvement <strong>in</strong> temporal arteritis and<br />
polymyalgia rheumatica. Am J Med 88:13.<br />
Nish<strong>in</strong>o H, DeRemee RA, Rub<strong>in</strong>o FA, Parisi JE. (1993). Wegener’s granulomatosis associated with vasculitis of the<br />
temporal artery: report of five cases. Mayo Cl<strong>in</strong> Proc 68:115–121.<br />
Nordborg E, Bengtsson BA. (1990). Epidemiology of biopsy-proven giant cell arteritis (GCA). J Intern Med 227:233.<br />
Olopade CO, Sekosan M, Schraufnagel DE. (1997). Giant cell arteritis manifest<strong>in</strong>g as chronic cough and fever of<br />
unknown orig<strong>in</strong>. Mayo Cl<strong>in</strong> Proc 72:1048–1050.<br />
Orgull S, Gass A, Flammer J. (1994). Optic disc cupp<strong>in</strong>g <strong>in</strong> arteritic anterior ischemic optic neuropathy.<br />
Ophthalmologica 208:336–338.<br />
Orrell RW, Johnson MH. (1993). Plasma viscosity and the diagnosis of giant cell arteritis. Br J Cl<strong>in</strong> Pract 47:71–72.<br />
Pache M, Kaiser HJ, Haufschild T, et al. (2002). Increased endothel<strong>in</strong>-1 plasma levels <strong>in</strong> giant cell arteritis: a report<br />
on four patients. Am J Ophthalmol 133:160–162.<br />
Pascual-Sedano B, Roig C. (1998). Horner’s syndrome due to giant cell arteritis. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:75–77.<br />
Pedro-Botet J, Coll J, Lopez MJ, Grau JM. (1996). Pericardial effusion and giant cell arteritis (letter). Br J Rheumatol<br />
35:194–195.<br />
Petzold A, Plant GT, Scaravilli F. (2002). Rapidly develop<strong>in</strong>g <strong>in</strong>timal fibrosis mimick<strong>in</strong>g giant cell arteritis. Br J<br />
Ophthalmol 86:114–115.<br />
Phelan MJ, Kok K, Burrow C, Thompson RN. (1993). Small bowel <strong>in</strong>farction <strong>in</strong> association with giant cell arteritis.<br />
Br J Rheumatol 32:63–65.<br />
Pless M, Rizzo JF III, Lamk<strong>in</strong> JC, Lessell S. (2000). Concordance of bilateral temporal artery biopsy <strong>in</strong> giant cell<br />
arteritis. J <strong>Neuro</strong>-ophthalmol 20:216–218.<br />
Ponge T, Barrier JH, Grolleau JY, et al. (1988). The efficacy of selective unilateral temporal artery biopsy versus<br />
bilateral biopsies for diagnosis of giant cell arteritis. J Rheumatol 15:997.<br />
Postel EA, Pollock SC. (1993). Recovery of vision <strong>in</strong> a 47-year-old man with fulm<strong>in</strong>ant giant cell arteritis. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 13:262–270.<br />
Pounta<strong>in</strong> G, Calv<strong>in</strong> J, Hazleman BL. (1994). Alpha 1-antichymotryps<strong>in</strong>, C-reactive prote<strong>in</strong> and erythrocyte<br />
sedimentation rate <strong>in</strong> polymyalgia rheumatica and giant cell arteritis. Br J Rheumatol 33:550–554.<br />
Pounta<strong>in</strong> G, Hazleman B. (1995). ABC of rheumatology. Polymyalgia rheumatica and giant cell arteritis. Br Med J<br />
310:1057–1059.<br />
Quillen DA, Cantore WA, Schwartz SR, Brod RD, Saddani JW. (1993). Choroidal nonperfusion <strong>in</strong> giant cell<br />
arteritis. Am J Ophthalmol 116:171–175.<br />
Radda TM, Pehamberger H, Smolen J, Menzel J. (1981). Ocular manifestation of temporal arteritis: immunological<br />
studies. Arch Ophthalmol 99:487–488.<br />
Rajesh CV, Cole M. (2000). Panuveitis as a present<strong>in</strong>g feature of giant cell arteritis. Br J Ophthalmol 84:340.<br />
Rauser M, Rismondo V. (1995). Ischemic optic neuropathy dur<strong>in</strong>g corticosteroid therapy for giant cell arteritis<br />
(letter). Arch Ophthalmol 113:707–708.<br />
Reich KA, Giansiracusa DF, Strongwater SL. (1990). <strong>Neuro</strong>logic manifestations of giant cell arteritis. Am J Med<br />
89:67–72.
Arteritic <strong>An</strong>terior Ischemic Optic <strong>Neuro</strong>pathy and Giant Cell Arteritis 117<br />
Richardson MP, Lever AM, F<strong>in</strong>k AM, Dixon AK, Hazleman BL. (1996). Survival after aortic dissection <strong>in</strong> giant cell<br />
arteritis (letter). <strong>An</strong>n Rheum Dis 55:332–333.<br />
Rischmueller M, Davies RP, Smith MD. (1996). Three year follow-up of a case of giant cell arteritis present<strong>in</strong>g with<br />
a chronic cough and upper limb ischaemic symptoms. Br J Rheumatol 35:800–802.<br />
Rivest D, Brunet D, Desbiens R, Bouchard JP. (1995). C-5 radiculopathy as a manifestation of giant cell arteritis.<br />
<strong>Neuro</strong>logy 45:1222–1224.<br />
Robb-Nicholson C, Chang RW, <strong>An</strong>derson S, et al. (1988). Diagnostic value of history and exam<strong>in</strong>ation <strong>in</strong> giant cell<br />
arteritis: a cl<strong>in</strong>ical pathological study of 81 temporal artery biopsies. J Rheumatol 15:1793.<br />
Ronchetto F. (1992). Transient monocular bl<strong>in</strong>dness <strong>in</strong> a patient with giant-cell arteritis. Pathogenetic and<br />
therapeutic considerations (letter). Recent Prog Med 83:241–242.<br />
Rousseau P. (1994). Giant cell arteritis. Arch Fam Med 3:628–632.<br />
Rudd JC. (1998). Ischemic scalp necrosis preced<strong>in</strong>g loss of visual acuity <strong>in</strong> giant cell arteritis. Arch Ophthalmol<br />
116:1690–1691.<br />
Ruiz-Masera JJ, Alamillos-Granados FJ, Dean-Ferrer A, et al. (1995). Submandibular swell<strong>in</strong>g as the first<br />
manifestation of giant cell arteritis. Report of a case. J Craniomaxillofac Surg 23:119–121.<br />
Russell RW. (1996). Giant cell arteritis—a cl<strong>in</strong>ical review. Curr Med Drugs 7:3–8.<br />
Sadda SR, Nee M, Miller NR, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> spectrum of posterior ischemic optic neuropathy. Am J<br />
Ophthalmol 132:743–750.<br />
Salvarani C, Gabriel SE, O’Fallon WM, Hunder GG. (1995). The <strong>in</strong>cidence of giant cell arteritis <strong>in</strong> Olmsted County,<br />
M<strong>in</strong>nesota: apparent fluctuations <strong>in</strong> cyclic pattern. <strong>An</strong>n Intern Med 123:192–194.<br />
Salvarani C, Hunder GG. (2001). Giant cell arteritis with low erythrocyte sedimentation rate: frequency of<br />
occurrence <strong>in</strong> a population-based study. Arthritis Rheum 45:140–145.<br />
Salvarani C, Macchioni P, Zizzi F, et al. (1991). Epidemiologic and immunogenic aspects of polymyalgia<br />
rheumatica and giant cell arteritis <strong>in</strong> northern Italy. Arthritis Rheum 34:351–356.<br />
Sav<strong>in</strong>o PJ. (2000). Giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 20:221.<br />
Schauble B, Wijman CAC, Kole<strong>in</strong>i B, Babikian VL. (2000). Ophthalmic artery microembolism <strong>in</strong> giant cell arteritis.<br />
J <strong>Neuro</strong>-ophthalmol 20:273–275.<br />
Schaufelberger C. (1998). No additive effect of cyclospor<strong>in</strong> A compared with glucocorticoid treatment alone <strong>in</strong><br />
giant cell arteritis: results of an open, controlled, randomized study. Br J Rheumatol 37:464–465.<br />
Schmidt D, Loffler KU. (1994). Temporal arteritis. Comparison of histological and cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs. Acta Ophthalmol<br />
72:319–325.<br />
Schwartz NG, Beck RW, Sav<strong>in</strong>o PJ, et al. (1995). Pa<strong>in</strong> <strong>in</strong> anterior ischemic optic neuropathy. J <strong>Neuro</strong>-ophthalmol<br />
15:9–10.<br />
Segato T, Piermarocchi S, Midena E. (1990). The role of fluoresce<strong>in</strong> angiography <strong>in</strong> the <strong>in</strong>terpretation of optic nerve<br />
head diseases. Metab Pediatr Syst Ophthalmol 13:111–114.<br />
Sehgal M, Swanson JW, DeRemee RA, Cobly TV. (1995). <strong>Neuro</strong>logic manifestations of Churg-Strauss syndrome.<br />
Mayo Cl<strong>in</strong> Proc 70:337–341.<br />
Shanahan EM, Hutch<strong>in</strong>son M, Hanley SD, Bresnihan B. (1999). Giant cell arteritis present<strong>in</strong>g as lateral medullary<br />
syndrome. Rheumatology 38:188–189.<br />
Sheehan MM, Keohane C, Twomey C. (1993). Fatal vertebral giant cell arteritis. J Cl<strong>in</strong> Pathol 46:1129–1131.<br />
Siatkowski RM, Gass JDM, Glaser JS, Smith JL, Schatz NJ, Schiffman J. (1993). Fluoresce<strong>in</strong> angiography <strong>in</strong> the<br />
diagnosis of giant cell arteritis. Am J Ophthalmol 115:57–63.<br />
Slamovits TL, Matzk<strong>in</strong> SC, Burde RM, Sachs R. (1992). IV steroids for central ret<strong>in</strong>al artery occlusion <strong>in</strong> giant cell<br />
arteritis. (letter). Ophthalmology 99:1482–1484.<br />
Slav<strong>in</strong> ML, Barnondes MJ. (1994). Visual loss caused by choroidal ischemia preced<strong>in</strong>g anterior ischemic optic<br />
neuropathy <strong>in</strong> giant cell arteritis. Am J Ophthalmol 117:81–86.<br />
Sonnenblick M, Nesher G, Friedlander Y, Rub<strong>in</strong>ow A. (1994). Giant cell arteritis <strong>in</strong> Jerusalem: a 12-year<br />
epidemiologic study. Br J Rheumatol 33:938–941.<br />
Sox HC, Liang MH. (1986). The erythrocyte sedimentation rate: guidel<strong>in</strong>es for rational use. <strong>An</strong>n Intern Med<br />
104:515–523.<br />
Stauton H, Safford F, Leader M, O’Riorda<strong>in</strong> D. (2000). Deterioration of giant cell arteritis with corticosteroid<br />
therapy. Arch <strong>Neuro</strong>l 57:581–584.<br />
Stebb<strong>in</strong>g J, Buetens O, Hellmann D, Stone J. (1999). Secondary amyloidosis associated with giant cell<br />
arteritis=polymyalgia rheumatica. J Rheumatol 26:2698–2700.<br />
Stevens RJ, Hughes RA. (1995). The aetiopathogenesis of giant cell arteritis. Br J Rheumatol 34:960.<br />
Straud R, Corman LC. (1996). Association of parvovirus B19 <strong>in</strong>fection with giant cell arteritis. Cl<strong>in</strong> Infect Dis<br />
22:1123.
118 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Thystrup J, Knudsen GM, Mogensen AM, Fledelius HC. (1994). Atypical visual loss <strong>in</strong> giant cell arteritis.<br />
Acta Ophthalmol 72:759–764.<br />
To KW, Enzer YR, Tsiaras WG. (1994). Temporal artery biopsy after one month of corticosteroid therapy.<br />
Am J Ophthalmol 117:265.<br />
Tomer Y, Neufeld MY, Shoenfeld Y. (1992). Coma with triphasic wave pattern <strong>in</strong> EEG as a complication of<br />
temporal arteritis. <strong>Neuro</strong>logy 42:439.<br />
Tomsak RL. (1997). Handbook of Treatment <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. Newton, MA, Butterworth-He<strong>in</strong>emann.<br />
Tomsak RL. (1991). Superficial temporal artery biopsy: a simplified technique. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:<br />
202–204.<br />
Trend P, Graham E. (1990). Internuclear ophthalmoplegia <strong>in</strong> giant cell arteritis (letter). J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 53:532.<br />
Turner RG, Henry J, Friedmann AI, et al. (1974). Giant cell arteritis. Postgrad Med J 50:265.<br />
Valmaggia C, Speiser P, Bischoff P, Neiderberger H. (1999). Indocyan<strong>in</strong>e green versus fluoresce<strong>in</strong> angiography <strong>in</strong><br />
the differential diagnosis of arteritic and nonarteritic anterior ischemic optic neuropathy. Ret<strong>in</strong>a 19:131–134.<br />
van der Veen MJ, D<strong>in</strong>ant HJ, van Booma-Frankfort C, van Albada-Kuipers GA, Biljsma JW. (1996). Can<br />
methotrexate be used as a steroid spar<strong>in</strong>g agent <strong>in</strong> the treatment of polymyalgia rheumatica and giant<br />
cell arteritis? <strong>An</strong>n Rheum Dis 55:219–223.<br />
Vilaseca J, Gonzalez A, Cid MC, Lopez-Vivancos J, Ortega A. (1987). <strong>Cl<strong>in</strong>ical</strong> usefulness of temporal artery biopsy.<br />
<strong>An</strong>n Rheum Dis 46:282–285.<br />
Walz-Leblanc BA, Ameli FM, Keystone EC. (1991). Giant cell arteritis present<strong>in</strong>g as limb claudication. Report and<br />
review of the literature. J Rheumatol 18:470–472.<br />
Wawryk SO, Ayberk H, Boyd AW, Rode J. (1991). <strong>An</strong>alysis of adhesion molecules <strong>in</strong> the immunopathogenesis of<br />
giant cell arteritis. J Cl<strong>in</strong> Pathol 44:497–501.<br />
Weems JJ Jr. (1992). Diagnosis of giant cell arteritis by occipital artery biopsy (letter). Am J Med 93:231–232.<br />
We<strong>in</strong> FB, Miller NR. (2000). Unilateral central ret<strong>in</strong>al artery occlusion followed by contralateral anterior ischemic<br />
optic neuropathy <strong>in</strong> giant cell arteritis. Ret<strong>in</strong>a 20:301–303.<br />
We<strong>in</strong>berg DA, Sav<strong>in</strong>o PJ, Sergott RC, Bosley TM. (1994). Giant cell arteritis. Corticosteroids, temporal artery<br />
biopsy and bl<strong>in</strong>dness. Arch Fam Med 3:623–627.<br />
We<strong>in</strong>ste<strong>in</strong> A, Del Guidice J. (1994). The erythrocyte sedimentation rate—time honored and tradition bound.<br />
J Rheumatol 21:1177.<br />
Weisman MH. (1995). Corticosteroids <strong>in</strong> the treatment of rheumatologic diseases. Curr Op<strong>in</strong> Rheumatol 7:183–190.<br />
Weiss LM, Gonzalez E, Miller SB, Agudelo CA. (1995). Severe anemia as the present<strong>in</strong>g manifestation of giant cell<br />
arteritis. Arthritis Rheum 38:434–436.<br />
Weyand CM, Bartley GB. (1997). Giant cell arteritis: new concepts <strong>in</strong> pathogenesis and implications for management.<br />
Am J Ophthalmol 123:392–395.<br />
Weyand CM, Gorozny JJ. (1995). Giant cell arteritis as an antigen driven disease. Rheum Dis Cl<strong>in</strong> North Am<br />
21:1027–1039.<br />
Weyand CM, Hicok KC, Hunder GG, Goronzy JJ. (1992). The HLA-DRB1 locus as a genetic component <strong>in</strong> giant<br />
cell arteritis: mapp<strong>in</strong>g of a disease-l<strong>in</strong>ked sequence motif to the antigen-b<strong>in</strong>d<strong>in</strong>g site of the HLA-DR<br />
molecule. J Cl<strong>in</strong> Invest 90:2355–2361.<br />
Weyand CM, Schonberger J, Ippitz U, Hunder NNH, Hicok KC, Goronzy JJ. (1994). Dist<strong>in</strong>ct vascular lesions <strong>in</strong><br />
giant cell arteritis share identical T cell clonotypes. J Exp Med 179:695–703.<br />
Wilke WS, Hoffman GS. (1995). Treatment of corticosteroid-resistant giant cell arteritis. Rheum Dis Cl<strong>in</strong> North Am<br />
21:59–71.<br />
Wise CM, Agudelo CA, Chmeleski WL, McKnight KM. (1991). Temporal arteritis with low erythrocyte<br />
sedimentation rate: a review of five cases. Arthritis Rheum 34:217–219.<br />
Zenone T, Souquet PJ, Bohas C, Vital Durand D, Bernard JP. (1994). Unusual manifestations of giant cell arteritis:<br />
pulmonary nodules, cough, conjunctivitis and otitis with deafness. Eur Respir J 7:2252–2254.<br />
Zweegman S, Makk<strong>in</strong>k B, Stehouwer CD. (1993). Giant-cell arteritis with normal erythrocyte sedimentation rate:<br />
case report and review of the literature. Nether J Med 42:128–131.
6 r<br />
Traumatic Optic <strong>Neuro</strong>pathy<br />
What Is the Traumatic Optic <strong>Neuro</strong>pathy?<br />
Traumatic optic neuropathy (TON) is a cl<strong>in</strong>ical diagnosis that presents with typical<br />
cl<strong>in</strong>ical features. Table 6–1 summarizes these features. The <strong>in</strong>cidence of TON after<br />
craniofacial trauma is probably 2 to 5%. Multiple mechanisms have been proposed <strong>in</strong><br />
TON. Table 6–2 lists the major theories for pathogenesis of TON.<br />
What Is the Evaluation of Traumatic Optic<br />
<strong>Neuro</strong>pathy?<br />
Once the cl<strong>in</strong>ical diagnosis of TON is made, neuroimag<strong>in</strong>g should be performed if<br />
possible. The <strong>in</strong>cidence of visible canal fracture <strong>in</strong> TON is variable and does not<br />
correlate well with the severity of visual loss (Goldberg, 1992; Seiff, 1990; Ste<strong>in</strong>sapir,<br />
Table 6–1. <strong>Cl<strong>in</strong>ical</strong> Features of Traumatic Optic <strong>Neuro</strong>pathy<br />
History of direct or <strong>in</strong>direct impact <strong>in</strong>jury to the head, face, or orbit<br />
Unilateral or bilateral visual loss<br />
Variable loss of visual acuity (range 20=20 to no light perception)<br />
Variable loss of visual field<br />
Relative afferent pupillary defect (unilateral or bilateral but asymmetric cases)<br />
Commonly normal or less commonly swollen optic nerve (Brodsky, 1995)<br />
Eventual ipsilateral optic atrophy<br />
Exclusion of other etiologies of visual loss <strong>in</strong> the sett<strong>in</strong>g of trauma:<br />
Open globe<br />
Traumatic cataract<br />
Vitreous hemorrhage<br />
Ret<strong>in</strong>al detachment<br />
119
120 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 6–2. Proposed Mechanisms of Traumatic<br />
Optic <strong>Neuro</strong>pathy<br />
Compressive or direct mechanical <strong>in</strong>jury<br />
Laceration<br />
Optic nerve contusion, edema, and swell<strong>in</strong>g<br />
Avulsion or transection<br />
Bone fragment or fracture<br />
Hemorrhage<br />
Retrobulbar with <strong>in</strong>creased <strong>in</strong>traorbital pressure<br />
Subperiosteal hematoma<br />
Optic nerve sheath hematoma<br />
Vascular <strong>in</strong>jury<br />
Vasospasm<br />
Ischemia<br />
Infarction<br />
Source: Aitken, 1991; Mauriello, 1992; Miller, 1990;<br />
Ste<strong>in</strong>sapir, 1994a; Volpe, 1991; Wol<strong>in</strong>, 1990.<br />
1994a). Computed tomography (CT) may be the best imag<strong>in</strong>g study for the evaluation<br />
of TON, detailed exam<strong>in</strong>ation for bone fractures, evaluation of bone anatomy (Goldberg,<br />
1992), and detection of acute hemorrhage (Knox, 1990; Seiff, 1990). Crowe et al<br />
described a case of an <strong>in</strong>trasheath and <strong>in</strong>trachiasmal hemorrhage and delayed visual<br />
loss (Crowe, 1989). Chou et al <strong>in</strong> 1996 summarized the literature on TON from 1922 to<br />
1990 and reported optic canal fracture <strong>in</strong> 92 of 431 cases (21%) (Chou, 1996).<br />
The role of magnetic resonance imag<strong>in</strong>g (MRI) <strong>in</strong> TON has yet to be clearly def<strong>in</strong>ed<br />
(Takehara, 1994). In addition, MRI is generally not available <strong>in</strong> the acute sett<strong>in</strong>g and is<br />
less useful than CT imag<strong>in</strong>g for the detection of acute hemorrhage, canal fractures, and<br />
bone anatomy (class III, level C).<br />
What Is the Treatment of Traumatic Optic<br />
<strong>Neuro</strong>pathy?<br />
The natural history of TON is not well def<strong>in</strong>ed but up to 20 to 38% of untreated patients<br />
may improve over time. Hughes described 56 cases of untreated TON, of which 44%<br />
were permanently bl<strong>in</strong>d and 16% ga<strong>in</strong>ed useful vision (Hughes, 1962). There is,<br />
however, no large, well-controlled randomized prospective data regard<strong>in</strong>g the treatment<br />
of TON (class III, level U). The literature on medical and surgical treatment of<br />
TON is difficult to summarize accurately because of the variations <strong>in</strong> cl<strong>in</strong>ical presentation,<br />
treatment modalities (e.g., steroids alone, steroids with surgery, surgery alone),<br />
surgical techniques and approaches, study <strong>in</strong>clusion criteria, and outcome measures,<br />
and because of recruitment bias and small sample sizes (class III–IV, level U). Cook et al<br />
<strong>in</strong> 1996 reviewed all cases of TON published <strong>in</strong> the English-language literature and<br />
performed a meta-analysis of treatment results (Cook, 1996). Patients were classified<br />
<strong>in</strong>to one of four grades (Table 6–3) depend<strong>in</strong>g on visual acuity and the location and type<br />
of fracture. Recovery of vision was significantly better <strong>in</strong> patients who underwent<br />
treatment compared with observation alone. No significant difference <strong>in</strong> improvement<br />
was noted <strong>in</strong> patients treated with corticosteroids alone, surgical decompression alone,
Table 6–3. Grades of Traumatic Optic <strong>Neuro</strong>pathy<br />
Grade 1: Acuity better than 20=200 without posterior orbital<br />
fracture<br />
Grade 2: Acuity 20=200 to light perception (LP) without a<br />
posterior orbital fracture<br />
Grade 3: Acuity of no light perception (NLP) or presence of<br />
nondisplaced posterior orbital fracture and some<br />
rema<strong>in</strong><strong>in</strong>g vision<br />
Grade 4: NLP and a displaced posterior orbital fracture<br />
Source: Cook, 1996.<br />
Traumatic Optic <strong>Neuro</strong>pathy 121<br />
or a comb<strong>in</strong>ation of those modalities. The prognosis for visual recovery worsened with<br />
<strong>in</strong>creas<strong>in</strong>g severity of grade. Recovery of vision was better <strong>in</strong> patients without orbital<br />
fractures and <strong>in</strong> those with anterior rather than posterior fractures.<br />
Chou et al <strong>in</strong> 1996 summarized the treatment results from the literature (28 reports)<br />
and found improvement <strong>in</strong> 94 (53%) of 176 medical treatment patients; 219 (46%) of 477<br />
surgical treatment patients; and 25 of 81 (31%) patients without treatment (Chou, 1996).<br />
These authors divided the patients undergo<strong>in</strong>g medical and surgical treatment <strong>in</strong>to two<br />
groups: patients with no light perception (NLP) vision and those with better than light<br />
perception (LP) vision. They reported that the NLP group had an improvement rate of<br />
36% (14 of 39 patients) follow<strong>in</strong>g medical treatment and 34% (19 of 56 patients)<br />
follow<strong>in</strong>g surgical treatment, versus the better than LP group that had an improvement<br />
rate of 70% (55 of 79) after medical treatment and 70% (69 of 98) after surgical treatment<br />
(class II, level C) (Chou, 1996).<br />
Lev<strong>in</strong> et al studied a total of 133 patients with TON (127 unilateral and 6 bilateral)<br />
who had <strong>in</strong>itial visual assessment with<strong>in</strong> 3 days of <strong>in</strong>jury and at least 1 month of followup<br />
(Lev<strong>in</strong>, 1999). On the basis of treatment received with<strong>in</strong> 7 days of <strong>in</strong>jury, patients<br />
with unilateral <strong>in</strong>juries were categorized as be<strong>in</strong>g <strong>in</strong> one of three treatment groups: (1)<br />
untreated (n ¼ 9), (2) corticosteroids (n ¼ 85), or (3) optic canal decompression (n ¼ 33).<br />
Corticosteroid therapy was categorized accord<strong>in</strong>g to <strong>in</strong>itial daily dose of methylprednisolone<br />
(or equivalent corticosteroid) as (1) megadose for 55400 mg (40%), (2) very<br />
high dose for 2000–5399 mg (18%), (3) high dose for 500–1999 mg (16%), (4) moderate<br />
dose for 100–499 mg (9%), and (5) low dose for
122 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
subgroups could have been missed. These results were thought to provide sufficient<br />
evidence to conclude that neither corticosteroid treatment nor optic canal surgery<br />
should be considered the standard of care for patients with TON. The authors felt<br />
that it is therefore cl<strong>in</strong>ically reasonable to treat or not treat on an <strong>in</strong>dividual patient basis<br />
(class II, level C).<br />
The study of Lev<strong>in</strong> et al had several potential problems:<br />
1. The study was not randomized, controlled, or masked, and treatment decisions<br />
followed the <strong>in</strong>vestigators ‘‘customary practice.’’<br />
2. Selection bias may have been present.<br />
3. Some patients were <strong>in</strong>itially treated with corticosteroids, and it is possible that the<br />
decision to perform surgery was related to a lack of positive response to the steroid<br />
treatment. This could have biased the results by remov<strong>in</strong>g nonresponders from the<br />
steroid group and add<strong>in</strong>g patients less likely to improve to the surgery group.<br />
4. Although the data suggested that neither the presence nor the absence of any<br />
particular CT f<strong>in</strong>d<strong>in</strong>g (e.g., optic nerve compression from a bone fragment or<br />
comm<strong>in</strong>uted canal fracture) affected visual outcome, a standardized methodology<br />
was not used for either CT technique or grad<strong>in</strong>g, and the number of patients with<br />
specific CTf<strong>in</strong>d<strong>in</strong>gs was small.<br />
How Much and What Dose of Corticosteroids<br />
Should Be Used?<br />
Although the ma<strong>in</strong>stay of medical treatment for TON has been corticosteroids, there is<br />
no prospective well-controlled study (i.e., no class I evidence) to support the efficacy of<br />
treatment or the validity of the various steroid preparations, dosages, or duration of<br />
therapy (<strong>An</strong>derson, 1982; Lam, 1990; Mauriello, 1992; Volpe, 1991). <strong>An</strong>derson et al<br />
proposed dexamethasone 3 to 5 mg=kg=day for all patients with TON and advocated<br />
surgery for patients with delayed visual loss who failed medical treatment or those with<br />
<strong>in</strong>itial visual improvement followed by worsen<strong>in</strong>g despite medical treatment (<strong>An</strong>derson,<br />
1982). Three (50%) of six patients had visual recovery after steroids, and four patients<br />
underwent transethmoidal-sphenoidal decompression with return of vision <strong>in</strong> one case<br />
(25%). Seiff reported a nonconsecutive, nonrandomized retrospective series of 36<br />
patients with TON (Seiff, 1990). Eighteen patients experienced visual improvement,<br />
<strong>in</strong>clud<strong>in</strong>g 5 of 15 (33%) patients who did not receive corticosteroids, and 13 of 21 (62%)<br />
patients treated with dexamethasone 1 mg=kg=day. This difference was not found to be<br />
statistically significant. Spoor et al reported an uncontrolled, nonconsecutive, retrospective<br />
series of 22 eyes <strong>in</strong> 21 patients with TON (Spoor, 1990). Of these 21 patients, 8<br />
received <strong>in</strong>travenous (IV) dexamethasone 20 mg every 6 hours and 13 received IV<br />
methylprednisolone (MP) 30 mg=kg load followed by 15 mg=kg every 6 hours. Visual<br />
improvement occurred <strong>in</strong> 7 of 9 patients <strong>in</strong> the dexamethasone group, and 12 of the 13<br />
patients <strong>in</strong> the MP group. Lessell described 33 cases of TON. Vision improved <strong>in</strong> 5 of 25<br />
untreated cases, 1 of 4 treated with corticosteroids, and 3 of 4 treated with transethmoidal<br />
decompression (Lessell, 1989). Kitthawees<strong>in</strong> and Yospaiboon (2001) performed a<br />
randomized, double-bl<strong>in</strong>d study compar<strong>in</strong>g dexamethasone and methylprednisolone <strong>in</strong><br />
20 patients with TON. There were no significant differences <strong>in</strong> visual improvement
etween the two groups. Chen et al (1998) reviewed 30 cases of TON (Chen, 1998).<br />
Thirteen of 21 cases treated with IV methylprednisolone improved and patients with<br />
vision better than light perception had a better prognosis.<br />
Because there are no double-masked, placebo-controlled, prospective, randomized<br />
data for the treatment of TON, many authors have advocated high doses of IV<br />
corticosteroids for TON, extrapolat<strong>in</strong>g the data on the use of higher dose MP for central<br />
nervous system (CNS) <strong>in</strong>jury (Bracken, 1990, 1993). The first National Acute Sp<strong>in</strong>al<br />
Cord Injury Study (NASCIS 1) (Bracken, 1993) was a non–placebo-controlled study that<br />
concluded there was no beneficial effect of MP 1000 mg bolus followed by 1000 mg per<br />
day for 10 days (‘‘high dose’’) compared with MP 100 mg bolus, then 100 mg per day for<br />
10 days (‘‘standard dose’’). NASCIS 2 was a multicenter, placebo-controlled, randomized,<br />
double-masked study of acute sp<strong>in</strong>al cord <strong>in</strong>jury that showed that treatment<br />
with<strong>in</strong> 8 hours with MP 30 mg=kg bolus followed by 5.4 mg=kg=hour for 24 hours<br />
resulted <strong>in</strong> significant improvement <strong>in</strong> motor and sensory function compared to<br />
placebo. MP delivered after 8 hours did not improve neurologic outcome. It was<br />
thought that MP <strong>in</strong> the 15 to 30 mg=kg dose range had a different pharmacologic effect<br />
on CNS <strong>in</strong>jury parameters <strong>in</strong>clud<strong>in</strong>g blood flow, calcium homeostasis, energy metabolism,<br />
and cl<strong>in</strong>ical outcome (Bracken, 1990, 1993). The traditional dose calculation for an<br />
equivalent dose of dexamethasone compared with MP has been based on the glucocorticoid<br />
potency of 5:1. Ste<strong>in</strong>sapir and Goldberg emphasized <strong>in</strong> 1994 that the potency<br />
ratio for dexamethasone to MP <strong>in</strong> CNS <strong>in</strong>jury may be closer to 2:1 and therefore that<br />
dexamethasone 15 mg=kg may be required (compared to the dose of 3 to 6 mg=kg<br />
recommended by <strong>An</strong>derson and other authors) for the adequate treatment of TON<br />
(Ste<strong>in</strong>sapir, 1994a). In a more recent review, Ste<strong>in</strong>sapir (1999) questioned the evidence<br />
that high-dose methylprednisolone is beneficial <strong>in</strong> TON. In one study us<strong>in</strong>g a crush<br />
<strong>in</strong>jury model <strong>in</strong> rats, there was a dose-dependent decrease <strong>in</strong> the number of axons <strong>in</strong> the<br />
methylprednisolone-treated animals compared with sal<strong>in</strong>e-treated controls (Ste<strong>in</strong>sapir,<br />
1994a). Despite these limitations, we summarize <strong>in</strong> Table 6–4 one protocol for the<br />
treatment of TON (class II–IV, level C).<br />
Table 6–4. Traumatic Optic <strong>Neuro</strong>pathy Protocol (class II–IV, level C)<br />
Diagnose TON appropriately (exclude alternative etiologies <strong>in</strong>clud<strong>in</strong>g open globe) (class III, level B).<br />
Perform canthotomy or cantholysis if the orbit is tense. Dra<strong>in</strong> subperiosteal hematoma if present<br />
(class III–IV, level C).<br />
Consider start<strong>in</strong>g IV corticosteroids (one regimen: methylprednisolone 30 mg=kg IV bolus, then<br />
5.4 mg=kg=hour IV for 48 hours or 15 mg=kg every 6 hours) even <strong>in</strong> patients with NLP vision<br />
(Joseph, 1990; Lessell, 1989; Spoor, 1990) (class III, level C).<br />
Perform high-resolution computed tomography (CT) scan of the optic canal and orbit. Consider optic<br />
nerve decompression if bony fragments imp<strong>in</strong>g<strong>in</strong>g on the optic nerve present (class III, level U).<br />
If vision improves on IV methylprednisolone after 48 hours, then start rapid oral taper of prednisone<br />
(class III, level C).<br />
If there is no cl<strong>in</strong>ical response after 48 hours or if vision deteriorates dur<strong>in</strong>g the steroid taper, then<br />
surgical decompression of the optic canal is offered especially for patients with severe visual loss<br />
(worse than 20=800) (class III, level C).<br />
Source: Repr<strong>in</strong>ted from Ste<strong>in</strong>sapir, 1994a, with permission from Elsevier Science.<br />
Traumatic Optic <strong>Neuro</strong>pathy 123
124 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What Is the Surgical Treatment of Traumatic<br />
Optic <strong>Neuro</strong>pathy?<br />
Multiple surgical approaches (e.g., lateral facial, transantral, transconjunctival=<br />
<strong>in</strong>tranasal endoscopic, sublabial transnasal, transfrontal, transethmoidal, or a comb<strong>in</strong>ation<br />
of these approaches, extracranial versus <strong>in</strong>tracranial, etc.) and surgical <strong>in</strong>dications<br />
have been offered for the treatment of TON. Unfortunately, there is no well-controlled<br />
prospective class I data to support the use of any one surgical approach to the optic<br />
nerve over another (<strong>An</strong>and, 1991; Fernandez, 1994; Friedman, 1991; Girard, 1992;<br />
Joseph, 1990; Knox, 1990; Kuppersmith, 1997; Lev<strong>in</strong>, 1994; Luxenberger, 1998;<br />
Ste<strong>in</strong>sapir, 1994a). Of particular <strong>in</strong>terest is the literature from Japan concern<strong>in</strong>g TON.<br />
Several papers have suggested that TON is much more common <strong>in</strong> Japan and more<br />
responsive to surgical treatment. Fukado reported 460 canal fractures on stereoscopic<br />
radiography of the optic canal <strong>in</strong> 500 patients with loss of vision follow<strong>in</strong>g head trauma<br />
(Fukado, 1972, 1975). Of 400 patients who underwent transethmoidal canal decompression,<br />
almost 100% had improvement. Several authors have raised serious questions<br />
about these studies, <strong>in</strong>clud<strong>in</strong>g the validity of the diagnostic criteria for canal fracture,<br />
the lack of complete ocular exam<strong>in</strong>ation data <strong>in</strong>clud<strong>in</strong>g visual field <strong>in</strong>formation, the<br />
paucity of bilateral cases, the high percentage of improvement after surgery, and the<br />
suspiciously high frequency of canal fracture (Kennerdell, 1976). Niho et al reported an<br />
80% success rate <strong>in</strong> 25 patients with TON and transsphenoidal decompression of the<br />
canal (Niho, 1970). Matsuzaki et al reported optic canal fractures <strong>in</strong> 52% of 33 patients<br />
with TON (Matsuzaki, 1982). Vision improved <strong>in</strong> 36% of the 11 cases undergo<strong>in</strong>g<br />
surgical decompression of the canal (8 transcranial and 3 transethmoidal). Vision<br />
improved <strong>in</strong> 50% of the 22 patients treated medically with prednisone (40–<br />
100 mg=day for 5 to 7 days), mannitol, and urok<strong>in</strong>ase (if per<strong>in</strong>eural hematoma was<br />
suspected). Fujitani et al reported 110 cases of TON, of which 43 cases underwent<br />
medical therapy with prednisone 60 mg=day and 70 eyes underwent transethmoidal<br />
decompression. The medically treated group had a 44% improvement rate versus a 47%<br />
improvement rate after surgery (Fujitani, 1986). M<strong>in</strong>e et al studied 34 patients with<br />
<strong>in</strong>direct TON (M<strong>in</strong>e, 1999). Twelve cases (13 eyes) underwent surgery and 24 patients<br />
(24 eyes) were managed without surgery. When <strong>in</strong>itial visual acuity was hand motions<br />
or better, vision improved significantly more <strong>in</strong> patients with surgery than <strong>in</strong> those<br />
without surgery. Age and optic canal fracture did not affect visual improvement or<br />
<strong>in</strong>fluence the decision for or aga<strong>in</strong>st surgery.<br />
Joseph et al reported 14 patients <strong>in</strong> a retrospective, nonconsecutive study with TON<br />
treated with transethmoidal-sphenoidal canal decompression and dexamethasone preand<br />
postoperatively. Eleven of the 14 patients improved, <strong>in</strong>clud<strong>in</strong>g 3 of 5 patients who<br />
presented with NLP vision ( Joseph, 1990). Luxenberger et al retrospectively studied 14<br />
patients who underwent optic nerve decompression surgery (with<strong>in</strong> 48 hours <strong>in</strong> 67%)<br />
and megadose corticosteroid therapy and noted improvement <strong>in</strong> 7 patients (50%)<br />
(Luxenberger, 1998). However, <strong>in</strong> this study there was no formal measurement of <strong>in</strong>itial<br />
vision, the def<strong>in</strong>ition of visual improvement was not stated, and the length of follow-up<br />
was not stated. Li et al reported the results of 45 consecutive patients treated with<br />
extracranial optic nerve decompression after at least 12 to 24 hours of corticosteroid<br />
therapy without improvement and noted visual improvement <strong>in</strong> 32 patients after<br />
surgery (71%) (Li, 1999b). Wang et al (2001) reviewed 61 consecutive, nonrandomized
Figure 6–1. Evaluation and treatment of traumatic optic neuropathy.<br />
Traumatic Optic <strong>Neuro</strong>pathy 125
126 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
patients with TON. There was no significant difference <strong>in</strong> visual improvement <strong>in</strong><br />
patients treated with surgical versus nonsurgical means. No light perception vision,<br />
however, or the presence of an orbital fracture (presumably a marker of more severe<br />
trauma), were poor prognostic <strong>in</strong>dicators. In this series, 29 of 34 patients (85%) with<br />
orbital fractures presented with no light perception. Lubben et al (2001) reported a<br />
retrospective analysis of 65 cases of TON who underwent optic nerve decompression.<br />
Thirteen of their 65 patients were comatose and the surgical <strong>in</strong>dication for TON was<br />
based on the f<strong>in</strong>d<strong>in</strong>g of a canal or orbital apex lesion. We generally do not recommend<br />
surgery for comatose patients who cannot provide visual <strong>in</strong>formation. Kountakis et al<br />
(2000) performed a retrospective review of TON treated with endoscopic optic nerve<br />
decompression. Eleven of 34 patients treated with high-dose steroids improved and 23<br />
did not improve. Of these 23 patients, 17 underwent endoscopic optic nerve decompression<br />
and 14 of 17 (82%) had improved visual acuity. These authors suggested that<br />
patients with visual acuity better than 20=200 had a better prognosis with steroids alone<br />
than patients with worse than 20=400 visual acuity.<br />
Unfortunately, until a randomized, prospective, double-masked, placebo-controlled<br />
cl<strong>in</strong>ical trial is performed, the treatment of TON will rema<strong>in</strong> controversial (class II–III,<br />
level U). The approach to TON is outl<strong>in</strong>ed <strong>in</strong> Figure 6–1.<br />
References<br />
Agarwal A, Mahapatra AK. (1999). Visual outcome <strong>in</strong> optic nerve <strong>in</strong>jury patients without <strong>in</strong>itial light perception.<br />
Ind J Ophthalmol 47:233–236.<br />
Aitken P, Sofferman R. (1991). Traumatic optic neuropathy. Ophthalmol Cl<strong>in</strong> North Am 4:479–490.<br />
<strong>An</strong>and VK, Sherwood C, Al-Mefty O. (1991). Optic nerve decompression via transethmoidal and supraorbital<br />
approaches. Op Tech Otolaryngol Head Neck Surg 2:157–166.<br />
<strong>An</strong>derson RL, Panje WR, Gross CE. (1982). Optic nerve bl<strong>in</strong>dness follow<strong>in</strong>g blunt forehead trauma. Ophthalmology<br />
89:445–455.<br />
Berestka JS, Rizzo JF III. (1994). Controversy <strong>in</strong> the management of traumatic optic neuropathy. Int Ophthalmol<br />
Cl<strong>in</strong> 34:87–96.<br />
Bilyk JR, Joseph MP. (1994). Traumatic optic neuropathy. Sem<strong>in</strong> Ophthalmol 9:200–211.<br />
Bracken MB, Holford TR. (1993). Effects of tim<strong>in</strong>g of methylprednisolone or naloxone adm<strong>in</strong>istration on recovery<br />
of segmental and long-tract neurologic function <strong>in</strong> NASCIS 2. J <strong>Neuro</strong>surg 79:500–507.<br />
Bracken MB, Shepard MJ, Coll<strong>in</strong>s WF, et al. (1990). A randomized controlled trial of methylprednisolone or<br />
naloxone <strong>in</strong> the treatment of acute sp<strong>in</strong>al cord <strong>in</strong>jury. Results of the Second National Acute Sp<strong>in</strong>al Cord<br />
Injury Study. N Engl J Med 322:1405–1411.<br />
Brodsky MC, Wald KJ, Chen S, Weiter JJ. (1995). Protracted posttraumatic optic disc swell<strong>in</strong>g. Ophthalmology<br />
192:1628–1631.<br />
Chen HY, Tsai RK, Wang HZ (1998). Intravenous methylprednisolone <strong>in</strong> treatment of traumatic optic neuropathy<br />
(abstract). Kaosh<strong>in</strong>g J Med Sci 14:577–583.<br />
Chou PI, Sadun AA, Chen YC, Su WY, L<strong>in</strong> SZ, Lee CC. (1996). <strong>Cl<strong>in</strong>ical</strong> experiences <strong>in</strong> the management of<br />
traumatic optic neuropathy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 18:325–336.<br />
Cook MW, Lev<strong>in</strong> LA, Joseph MP, P<strong>in</strong>czower EF. (1996). Traumatic optic neuropathy. A meta-analysis. Arch<br />
Otolaryngol Head Neck Surg 122:389–392.<br />
Crowe NW, Nickles TP, Troost T, Elster AD. (1989). Intrachiasmal hemorrhage: a cause of delayed post-traumatic<br />
bl<strong>in</strong>dness. <strong>Neuro</strong>logy 39:863–865.<br />
Fernandez CIS, Rollon A, Padilla JDG. (1994). Posttraumatic amaurosis with partial return of visual acuity. J Oral<br />
Maxillofacial Surg 52:1077–1079.<br />
Friedman M. (1991). Optic nerve decompression. Op Tech Otolaryngol Head Neck Surg 2:149.<br />
Fujitani T, Inoue K, Takahashi T, et al. (1986). Indirect traumatic optic nerve neuropathy-visual outcome of<br />
operative and non-operative cases. Jpn J Ophthalmol 30:125–134.<br />
Fukado Y. (1972). Results <strong>in</strong> 350 cases of surgical decompression of the optic nerve. Trans Asia Pacif Acad<br />
Ophthalmol 4:96–99.
Traumatic Optic <strong>Neuro</strong>pathy 127<br />
Fukado Y. (1975). Results <strong>in</strong> 400 cases of surgical decompression of the optic nerve. Mod Prob Ophthalmol 14:474–<br />
481.<br />
Girard BC, Bouzas EA, Lama SG, et al. (1992). Visual improvement after transethmoidal-sphenoid decompression<br />
<strong>in</strong> optic nerve <strong>in</strong>juries. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:142.<br />
Goldberg RA, Hannani K, Toga AW. (1992). Microanatomy of the orbital apex: computed tomography and<br />
microplann<strong>in</strong>g of soft and hard tissue. Ophthalmology 99:1447–1452.<br />
Hughes B. (1962). Indirect <strong>in</strong>jury of the optic nerve and chiasm. Bull Johns Hopk<strong>in</strong>s Hosp 111:98–126.<br />
Joseph MP, Lessell S, Rizzo J, Momose KJ. (1990). Extracranial optic nerve decompression for traumatic optic<br />
neuropathy. Arch Ophthalmol 108:1091–1093.<br />
Kennerdell JS, Amsbaugh GA, Myers EN. (1976). Transantral-ethmoidal decompression of optic canal fracture.<br />
Arch Ophthalmol 94:1040–1043.<br />
Kitthawees<strong>in</strong> K, Yospaiboon. (2001). Dexamethasone and methylprednisolone <strong>in</strong> treatment of <strong>in</strong>direct traumatic<br />
optic neuropathy (abstract). J Med Assoc Thai 84:628–634.<br />
Knox BE, Gates GA, Berry SM. (1990). Optic nerve decompression via the lateral facial approach. Laryngoscope<br />
100:458–462.<br />
Kountakis SE, Maillard AA, El-Harazi SM, et al. (2000). Endoscopic optic nerve decompression for traumatic<br />
bl<strong>in</strong>dness. Otolaryngol Head Neck Surg 123:34–37.<br />
Kuppersmith RB, Alford EL, Patr<strong>in</strong>ely JR, Lee AG, Parke RB, Holds JB. (1997). Comb<strong>in</strong>ed transconjunctival=<br />
<strong>in</strong>tranasal endoscopic approach to the optic canal <strong>in</strong> traumatic optic neuropathy. Laryngoscope 107:<br />
311–315.<br />
Lam BL, We<strong>in</strong>geist TA. (1990). Corticosteroid-responsive traumatic optic neuropathy. Am J Ophthalmol 109:99–101.<br />
Lessell S. (1989). Indirect optic nerve trauma. Arch Ophthalmol 107:382–386.<br />
Lev<strong>in</strong> LA, Beck RW, Joseph MP, et al. (The International Optic Nerve Trauma Study Group). (1999). The treatment<br />
of traumatic optic neuropathy. The International Optic Nerve Trauma Study. Ophthalmology 106:168–1277.<br />
Lev<strong>in</strong> LA, Joseph MP, Rizzo JF III. (1994). Optic canal decompression <strong>in</strong> <strong>in</strong>direct optic nerve trauma. Ophthalmology<br />
101:566.<br />
Li KK, Teknos TN, Lai A, et al. (1999a). Extracranial optic nerve decompression: a 10-year review of 92 patients. J<br />
Craniofac Surg 10:454–459.<br />
Li KK, Teknos TN, Lai A, et al. (1999b). Traumatic optic neuropathy: results <strong>in</strong> 45 consecutive surgically treated<br />
patients. Otolaryngol Head Neck Surg 120:5–11.<br />
Lubben B, Stoll W, Grenzebach U. (2001). Optic nerve decompression <strong>in</strong> the comatose and conscious patients after<br />
trauma. Laryngoscope 111:320–328.<br />
Luxenberger W, Stammberger H, Jebeles JA, Walch C. (1998). Endoscopic optic nerve decompression: the Granz<br />
experience. Laryngoscope 108:873–882.<br />
Mahapatra AK, Tandon DA. (1993). Traumatic optic neuropathy <strong>in</strong> children—a prospective study. Pediatr<br />
<strong>Neuro</strong>surg 19:34.<br />
Matsuzaki H, Kunita M, Kawai K. (1982). Optic nerve damage <strong>in</strong> head trauma: cl<strong>in</strong>ical and experimental studies.<br />
Jpn J Ophthalmol 26:447–461.<br />
Mauriello JA, DeLuca J, Krieger A, Schulder M, Frohman L. (1992). Management of traumatic optic neuropathy—<br />
a study of 23 patients. Br J Ophthalmol 76:349–352.<br />
Miller NR. (1990). The management of traumatic optic neuropathy (editorial). Arch Ophthalmol 108:1086–1087.<br />
M<strong>in</strong>e S, Yamakami I, Yamaura A, et al. (1999). Outcome of traumatic optic neuropathy. Comparison between<br />
surgical and nonsurgical treatment. Acta <strong>Neuro</strong>chir 141:27–30.<br />
Niho S, Niho M, Niho K. (1970). Decompression of the optic canal by the transethmoidal route and decompression<br />
of the superior orbital fissure. Can J Ophthalmol 5:22–40.<br />
Pomeranz HD, Rizzo JF, Lessell S. (1999). Treatment of traumatic optic neuropathy. Int Ophthalmol Cl<strong>in</strong> 39:185–194.<br />
Seiff RR. (1990). High dose corticosteroids for treatment of vision loss due to <strong>in</strong>direct <strong>in</strong>jury to the optic nerve.<br />
Ophthalmic Surg 21:389–395.<br />
Spoor TC, Hartel WC, Lens<strong>in</strong>k DB, Wilk<strong>in</strong>son MJ. (1990). Treatment of traumatic optic neuropathy with<br />
corticosteroids. Am J Ophthalmol 110:665–669.<br />
Ste<strong>in</strong>sapir KD. (1999). Traumatic optic neuropathy. Curr Op<strong>in</strong> Ophthalmol 10:340–342.<br />
Ste<strong>in</strong>sapir KD, Goldberg RA. (1994a). Traumatic optic neuropathy. Surv Ophthalmol 38:487–518.<br />
Ste<strong>in</strong>sapir KD, S<strong>in</strong>ha S, Hovda DA, Goldberg RA. (1994b). Axonal loss and dynamic changes <strong>in</strong> cerebral glucose<br />
metabolism follow<strong>in</strong>g optic nerve trauma. Invest Ophthalmol Vis Sci 35:1544.<br />
Sullivan G, Helveston EM. (1969). Optic atrophy after seem<strong>in</strong>gly trivial trauma. Arch Ophthalmol 81:159–161.<br />
Takehara S, Tanaka T, Uemura K, et al. (1994). Optic nerve <strong>in</strong>jury demonstrated by MRI with STIR sequences.<br />
<strong>Neuro</strong>radiology 36:512.
128 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Volpe N, Lessell S, Kl<strong>in</strong>e L. (1991). Traumatic optic neuropathy: diagnosis and management. Int Ophthalmol Cl<strong>in</strong><br />
31:142–156.<br />
Wang BH, Robertson BC, Girotto JA, et al. (2001). Traumatic optic neuropathy: a review of 61 patients. Plast<br />
Reconstr Surg 107:1655–1664.<br />
Wol<strong>in</strong> M, Lav<strong>in</strong> P. (1990). Spontaneous visual recovery from traumatic optic neuropathy after blunt head <strong>in</strong>jury.<br />
Am J Ophthalmol 109:430–435.
7 r<br />
Papilledema<br />
What Is the Def<strong>in</strong>ition of Papilledema?<br />
The term papilledema is frequently applied to optic disc swell<strong>in</strong>g from any cause, but it<br />
should be used cl<strong>in</strong>ically only for disc swell<strong>in</strong>g that results from <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure. The ma<strong>in</strong> reason for apply<strong>in</strong>g the term judiciously is that to most neuroophthalmologists<br />
papilledema implies an etiology, and thus <strong>in</strong>appropriate or unnecessary<br />
test<strong>in</strong>g could result from miscommunication between providers.<br />
Other forms of optic disc swell<strong>in</strong>g due to local or systemic etiologies should be named<br />
accord<strong>in</strong>g to their presumed etiology (e.g., optic neuritis, anterior ischemic optic<br />
neuropathy, etc). It is usually not possible to determ<strong>in</strong>e the etiology of disc swell<strong>in</strong>g<br />
from the ophthalmoscopic appearance of the disc alone. The history and neuroophthalmologic<br />
exam<strong>in</strong>ation, especially the visual fields, are necessary to reach an<br />
etiologic diagnosis. It is also important to note that optic disc swell<strong>in</strong>g may not develop<br />
if optic atrophy is present. For example, <strong>in</strong> patients with prior ‘‘bow-tie atrophy’’ of the<br />
optic nerve from a suprasellar mass, disc swell<strong>in</strong>g may affect only the superior and<br />
<strong>in</strong>ferior aspects of the nerve (‘‘tw<strong>in</strong> peaks papilledema’’) (Ing, 1996).<br />
The symptoms associated with optic disc swell<strong>in</strong>g depend on the underly<strong>in</strong>g etiology.<br />
In general, swollen optic discs from any cause may be associated with transient visual<br />
obscurations (see Chapter 8) (Sadun, 1984). These are typically unilateral or bilateral<br />
dimm<strong>in</strong>g or black<strong>in</strong>g out of vision that usually lasts seconds and may be precipitated by<br />
changes <strong>in</strong> posture (e.g., bend<strong>in</strong>g or straighten<strong>in</strong>g).<br />
What Are the Features that Dist<strong>in</strong>guish Real<br />
Papilledema from Pseudopapilledema?<br />
True disc swell<strong>in</strong>g must be dist<strong>in</strong>guished from pseudopapilledema (e.g., anomalously<br />
elevated discs caused by optic nerve head drusen) (Kurz-Lev<strong>in</strong>, 1999). Pseudopapilledema<br />
is a relatively common f<strong>in</strong>d<strong>in</strong>g, and optic disc drusen are among the most<br />
frequent etiologies. Drusen of the disc may be obvious, t<strong>in</strong>y, or buried. Other disc<br />
129
130 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
anomalies that may be mistaken for papilledema <strong>in</strong>clude small, ‘‘crowded’’ hyperopic<br />
discs and tilted or anomalous discs. With pseudopapilledema the peripapillary nerve<br />
fiber layer is normal, venous pulsations are usually present, there is no vascular<br />
engorgement or hemorrhages, there are no cotton-wool spots, and the discs do not<br />
leak dye on fluoresce<strong>in</strong> angiography. Myel<strong>in</strong>ated nerve fibers may occasionally resemble<br />
disc swell<strong>in</strong>g but are characterized by a white feathery nerve fiber layer appearance.<br />
Hyaloid traction on the optic disc and epipapillary glial tissue may occasionally also be<br />
mistaken for disc swell<strong>in</strong>g. Ophthalmoscopic criteria that might dist<strong>in</strong>guish pseudopapilledema<br />
from true papilledema <strong>in</strong>clude the follow<strong>in</strong>g (Glaser, 1990):<br />
1. <strong>An</strong> absent central cup with a small disc diameter<br />
2. Vessels aris<strong>in</strong>g from the central apex of the disc<br />
3. <strong>An</strong>omalous branch<strong>in</strong>g of vessels (e.g., bifurcations, trifurcations) with <strong>in</strong>creased<br />
number of disc vessels<br />
4. Visible ‘‘glow’’ of drusen seen with disc transillum<strong>in</strong>ation<br />
5. Irregular optic disc marg<strong>in</strong>s with derangement of peripapillary ret<strong>in</strong>al pigment<br />
epithelium<br />
6. Absence of superficial capillary telangiectasia on the optic disc head<br />
7. No hemorrhages (although subret<strong>in</strong>al hemorrhages may occur with disc drusen)<br />
8. No exudates or cotton-wool spots<br />
What Evaluation Is Necessary for Optic Disc<br />
Drusen?<br />
Most cases of pseudopapilledema can be diagnosed cl<strong>in</strong>ically and simply documented<br />
photographically. In difficult cases, further test<strong>in</strong>g may be useful <strong>in</strong> the diagnosis of<br />
drusen. Disc drusen may show autofluorescence noted prior to <strong>in</strong>jection of fluoresce<strong>in</strong><br />
angiography dye. Although generally not required for the diagnosis, computed<br />
tomography (CT) imag<strong>in</strong>g may demonstrate the calcified drusen <strong>in</strong> the optic nerve.<br />
Buried drusen may also be visible on orbital ultrasound.<br />
Kurz-Lev<strong>in</strong> and Landau retrospectively reviewed 142 patients (261 eyes) with<br />
suspected optic disc drusen (Kurz-Lev<strong>in</strong>, 1999). Evaluations <strong>in</strong>cluded B-scan echography,<br />
orbital CT scan, and=or pre<strong>in</strong>jection control photography for autofluorescence.<br />
Thirty-six of the 261 eyes were evaluated us<strong>in</strong>g all three techniques, and drusen of the<br />
optic nerve head were diagnosed <strong>in</strong> 21 eyes. B-scan ultrasonography was positive <strong>in</strong> all<br />
21 eyes. N<strong>in</strong>e cases had positive CT scans f<strong>in</strong>d<strong>in</strong>gs, and 10 had positive pre<strong>in</strong>jection<br />
control photographs. In 82 eyes with suspected buried drusen of the optic nerve head,<br />
B-scan echography showed drusen <strong>in</strong> 39 eyes, compared with 15 eyes <strong>in</strong> which drusen<br />
were shown us<strong>in</strong>g pre<strong>in</strong>jection control photography. No drusen were seen on pre<strong>in</strong>jection<br />
control photography or CT scan that were missed on B-scan echography. The<br />
authors concluded that drusen of the optic nerve head are diagnosed most reliably<br />
us<strong>in</strong>g B-scan echography compared with both pre<strong>in</strong>jection control photography and CT<br />
scans. Pre<strong>in</strong>jection control photography is usually positive when there are visible<br />
drusen of the optic disc, and therefore its cl<strong>in</strong>ical use is limited. Likewise, CT scan is<br />
an expensive and less sensitive test for the detection of buried drusen of the optic nerve<br />
head. We recommend B-scan ultrasonography for the detection of buried drusen as the<br />
<strong>in</strong>itial diagnostic study (class III, level C).
Is the Disc Swell<strong>in</strong>g Caused by Optic<br />
<strong>Neuro</strong>pathy or Papilledema?<br />
Disc swell<strong>in</strong>g due to raised <strong>in</strong>tracranial pressure (i.e., papilledema) is usually bilateral<br />
and symmetric <strong>in</strong> both eyes. Unilateral disc swell<strong>in</strong>g is most commonly caused by local<br />
pathology with<strong>in</strong> the optic nerve or orbit. Unilateral papilledema, however, can occur,<br />
although most of these cases are actually bilateral but asymmetric disc swell<strong>in</strong>g (Chari,<br />
1991; Huna-Baron, 2001; Killer, 2001; Lepore, 1992; Strom<strong>in</strong>ger, 1992; To, 1990). If one<br />
optic nerve is atrophic, it may not swell, and unilateral disc swell<strong>in</strong>g may occur from<br />
<strong>in</strong>creased <strong>in</strong>tracranial pressure <strong>in</strong> these cases (e.g., Foster Kennedy syndrome). These<br />
optic neuropathies are discussed <strong>in</strong> Chapters 1 through 6. Processes caus<strong>in</strong>g optic<br />
neuropathies associated with disc swell<strong>in</strong>g are usually unilateral, but may be bilateral,<br />
and are listed <strong>in</strong> Table 7–1. Other processes that may mimic papilledema and that may<br />
present with bilateral optic disc swell<strong>in</strong>g with little or no visual acuity impairment, color<br />
vision loss, or visual field defects and normal <strong>in</strong>tracranial pressure are listed <strong>in</strong> Table 7–2.<br />
Certa<strong>in</strong> <strong>in</strong>flammatory or <strong>in</strong>fectious processes, such as syphilis, sarcoidosis, HIVassociated<br />
men<strong>in</strong>goradiculitis, and viral men<strong>in</strong>goencephalitis that affect the men<strong>in</strong>ges<br />
may cause optic disc swell<strong>in</strong>g due to per<strong>in</strong>euritis (Hyk<strong>in</strong>, 1991; Nakamura, 1999;<br />
Prevett, 1997). Cat-scratch disease and Lyme disease may also cause bilateral disc<br />
edema with normal visual fields and vision (Bafna, 1996; Fedorowski, 1996, Rothermel,<br />
2001).<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of<br />
Papilledema?<br />
The cl<strong>in</strong>ical features and stages of papilledema are outl<strong>in</strong>ed <strong>in</strong> Tables 7–3 and 7–4. The<br />
Frisen papilledema grad<strong>in</strong>g scale is listed <strong>in</strong> Table 7–5. Features helpful <strong>in</strong> differentiat<strong>in</strong>g<br />
true optic disc edema from pseudo-disc edema (e.g., buried disc drusen) are<br />
outl<strong>in</strong>ed <strong>in</strong> Table 7–6.<br />
Table 7–1. Bilateral Optic <strong>Neuro</strong>pathies with Optic Disc Edema<br />
Papilledema 131<br />
Infectious (e.g., <strong>in</strong>fectious optic neuritis, men<strong>in</strong>gitis, neuroret<strong>in</strong>itis, uveitis associated disc edema,<br />
cat-scratch disease, Lyme disease)<br />
Demyel<strong>in</strong>at<strong>in</strong>g (e.g., multiple sclerosis)<br />
Inflammatory (e.g., systemic lupus erythematosus, sarcoidosis) (Sherman, 1999)<br />
Vascular conditions, <strong>in</strong>clud<strong>in</strong>g arteritic and nonarteritic anterior ischemic optic neuropathy, disc<br />
swell<strong>in</strong>g <strong>in</strong> diabetics (diabetic papillopathy), central ret<strong>in</strong>al ve<strong>in</strong> occlusion, and carotidcavernous<br />
s<strong>in</strong>us fistula<br />
Infiltrative (e.g., carc<strong>in</strong>omatous men<strong>in</strong>gitis, sarcoid)<br />
Compressive (e.g., neoplastic thyroid ophthalmopathy)<br />
Hereditary (e.g., Leber’s hereditary optic neuropathy)<br />
Traumatic (rare)<br />
Paraneoplastic optic neuropathy<br />
Mechanical (e.g., hypotony)<br />
Chronic respiratory disease (O’Halloran, 1999)
132 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 7–2. Etiologies for Bilateral Optic Disc Edema with Normal Visual Function<br />
Hypertensive optic neuropathy and ret<strong>in</strong>opathy (Lee, 2002a; Wall, 1995a)<br />
Blood dyscrasias (e.g., anemia, polycythemia, dysprote<strong>in</strong>emia)<br />
Cyanotic congenital heart disease: disc swell<strong>in</strong>g may be due to decreased arterial oxygen saturation<br />
and polycythemia<br />
Sleep apnea: probably by a mechanism similar to that <strong>in</strong> congenital cyanotic heart disease<br />
(Purv<strong>in</strong>, 2000)<br />
Sp<strong>in</strong>al cord tumors (often with myelopathy: e.g., back pa<strong>in</strong>, leg weakness, sensory changes,<br />
bladder <strong>in</strong>volvement, etc.)<br />
Acute <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (AIDP or Guilla<strong>in</strong>-Barré syndrome) and<br />
chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (CIDP) (Morrison, 1999)<br />
POEMS (peripheral neuropathy, organomegaly, endocr<strong>in</strong>opathy, monoclonal gammopathy, and<br />
sk<strong>in</strong> changes)<br />
Crow-Fukase syndrome (peripheral polyneuropathy, organomegaly, lymphadenopathy,<br />
Castleman’s disease, endocr<strong>in</strong>opathy, gammopathy, or myeloma and sk<strong>in</strong> changes (Boll<strong>in</strong>g,<br />
1990; Wong, 1998)<br />
Hypoparathyroidism (primary or surgically <strong>in</strong>duced) (McLean, 1998)<br />
Uremia—these patients may have normal or <strong>in</strong>creased <strong>in</strong>tracranial pressure<br />
Hypoxemia and anemia<br />
Diabetic papillopathy (see Chapter 4)<br />
What Studies Should Be Performed to<br />
Investigate the Patient with Papilledema?<br />
All patients with papilledema require a thorough neurologic and neuro-ophthalmologic<br />
history and physical exam<strong>in</strong>ation. In general, the syndromes caus<strong>in</strong>g <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure are listed <strong>in</strong> Table 7–7.<br />
Table 7–3. The <strong>Cl<strong>in</strong>ical</strong> Features of Papilledema<br />
Usually bilateral but may be unilateral or asymmetric (Killer, 2001)<br />
Usually preserved visual acuity and color vision early<br />
May have transient visual loss last<strong>in</strong>g seconds (obscurations of vision)<br />
Visual field defects<br />
Enlarged bl<strong>in</strong>d spot<br />
Generalized constriction<br />
Glaucomatous-like defects<br />
Eventual peripheral constriction, especially nasally<br />
No afferent pupillary defect unless severe and asymmetric disc edema<br />
Fluoresce<strong>in</strong> angiography<br />
Early disc capillary dilation, dye leakage, and microaneurysm formation<br />
Late leakage of dye beyond disc marg<strong>in</strong>s<br />
May be normal <strong>in</strong> early papilledema<br />
Echography may show <strong>in</strong>creased diameter of optic nerve with fluid <strong>in</strong> the optic nerve sheath
Table 7–4. The Stages of Papilledema<br />
Papilledema 133<br />
Early papilledema<br />
M<strong>in</strong>imal disc hyperemia with capillary dilation<br />
Early opacification of nerve fiber layer (peripapillary ret<strong>in</strong>a loses its superficial l<strong>in</strong>ear and<br />
curvil<strong>in</strong>ear light reflex and appears red without luster)<br />
Early swell<strong>in</strong>g of disc<br />
Absence of venous pulsations<br />
Peripapillary ret<strong>in</strong>al nerve fiber layer hemorrhage<br />
Fully developed papilledema<br />
Engorged and tortuous ret<strong>in</strong>al ve<strong>in</strong>s<br />
May have spl<strong>in</strong>ter hemorrhages at or adjacent to the disc marg<strong>in</strong><br />
Disc surface grossly elevated<br />
Surface vessels become obscured by now opaque nerve fiber layer<br />
May have cotton wool spots<br />
Paton’s l<strong>in</strong>es (circumferential ret<strong>in</strong>al folds) or choroidal folds<br />
May have exudates (e.g., macular star or hemistar)<br />
May have hemorrhages or fluid <strong>in</strong> the macula that may decrease vision<br />
In acute cases (e.g., subarachnoid hemorrhage), subhyaloid hemorrhages may occur that may<br />
break <strong>in</strong>to vitreous (Terson’s syndrome)<br />
Rarely macular or peripapillary subret<strong>in</strong>al neovascularization<br />
Chronic papilledema<br />
Hemorrhages and exudates slowly resolve<br />
Central cup, which is <strong>in</strong>itially reta<strong>in</strong>ed even <strong>in</strong> severe cases, ultimately becomes obliterated<br />
Initial disc hyperemia changes to a milky gray<br />
Small hard exudates that are refractile and drusen-like may appear on disc surface<br />
Visual field loss <strong>in</strong>clud<strong>in</strong>g nerve fiber layer defects may develop<br />
Optociliary ‘‘shunt’’ (collaterals) vessels may develop<br />
Atrophic papilledema (pale disc edema)<br />
Optic disc pallor with nerve fiber bundle visual field defects<br />
Ret<strong>in</strong>al vessels become narrow and sheathed<br />
Occasional pigmentary changes or choroidal folds <strong>in</strong> macula<br />
Selective loss of peripheral axons while spar<strong>in</strong>g central axons (usually preservation of good central<br />
visual acuity)<br />
In all patients with bilateral optic disc swell<strong>in</strong>g, the blood pressure should be checked<br />
to evaluate for possible malignant hypertension. Blood dyscrasia should be considered<br />
if there are other suggestive ret<strong>in</strong>al vascular f<strong>in</strong>d<strong>in</strong>gs (e.g., <strong>in</strong>complete or complete<br />
central ret<strong>in</strong>al ve<strong>in</strong> occlusion with optic disc edema). <strong>Neuro</strong>imag<strong>in</strong>g is required <strong>in</strong> all<br />
patients (class II, level B).<br />
CT imag<strong>in</strong>g is the preferred study <strong>in</strong> evaluat<strong>in</strong>g acute vascular processes<br />
(e.g., subarachnoid, epidural, subdural, or <strong>in</strong>tracerebral hemorrhage, acute <strong>in</strong>farction)<br />
or <strong>in</strong> acute head trauma (e.g., rule out fracture, acute bleed). CT scan may be used <strong>in</strong><br />
patients with contra<strong>in</strong>dications to magnetic resonance imag<strong>in</strong>g (MRI) (e.g., pacemakers,<br />
metallic clips <strong>in</strong> head, metallic foreign bodies), and obese or claustrophobic patients.<br />
Otherwise, MRI is the modality of choice <strong>in</strong> papilledema. MR angiography or MR<br />
venography may be useful for suspected arterial disease or venous obstruction. If
134 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 7–5. Frisen Papilledema Grad<strong>in</strong>g System<br />
Stage 0: Normal optic disc<br />
Stage 1<br />
Obscuration of the nasal border of the disc<br />
No elevation of the disc borders<br />
Disruption of the normal radial nerve fiber layer (NFL) arrangement with grayish opacity<br />
accentuat<strong>in</strong>g nerve fiber bundles<br />
Normal temporal disc marg<strong>in</strong><br />
Subtle grayish halo with temporal gap<br />
Stage 2<br />
Obscuration of all borders<br />
Elevation of nasal border<br />
Complete peripapillary halo<br />
Stage 3<br />
Obscuration of all borders<br />
Elevation of all borders<br />
Increased diameter of the optic nerve head<br />
Obscuration of one or more segments of major blood vessels leav<strong>in</strong>g the disc<br />
Peripapillary halo—irregular outer fr<strong>in</strong>ge with f<strong>in</strong>ger-like extensions<br />
Stage 4<br />
Elevation of entire nerve head<br />
Obscuration of all borders<br />
Peripapillary halo<br />
Total obscuration on the disc of a segment of a major blood vessel<br />
Stage 5<br />
Dome-shaped protrusions represent<strong>in</strong>g anterior expansion of the optic nerve head<br />
Peripapillary halo is narrow and smoothly demarcated<br />
Total obscuration of a segment of a major blood vessel may or may be present<br />
Obliteration of the optic cup<br />
Source: Repr<strong>in</strong>ted from Friedman, 2001, with permission from Elsevier Science.<br />
Table 7–6. Differentiat<strong>in</strong>g True Optic Disc Edema from Pseudopapilledema<br />
Optic Disc Edema Pseudopapilledema<br />
Disc vasculature obscured Disc marg<strong>in</strong> vasculature clear<br />
Elevation of peripapillary NFL Elevation conf<strong>in</strong>ed to disc<br />
Obscured peripapillary NFL Sharp peripapillary NFL<br />
Venous congestion No venous congestion<br />
Exudates=cotton wool spots No exudates=cotton wool spots<br />
Loss of cup late Small cupless disc<br />
Normal disc vessels <strong>An</strong>omalous disc vessels<br />
No circumpapillary light reflex Crescent circumpapillary light reflex<br />
Absent venous pulsations With or without spontaneous venous pulsations<br />
NFL, nerve fiber layer.
Table 7–7. Syndromes Caus<strong>in</strong>g Increased Intracranial Pressure<br />
Primary causes<br />
Idiopathic pseudotumor cerebri syndrome (idiopathic <strong>in</strong>tracranial hypertension) with papilledema<br />
or without papilledema<br />
Secondary causes<br />
Hydrocephalus<br />
Shunt failure <strong>in</strong> patient with hydrocephalus (ventriculomegaly may be absent)<br />
Mass lesions—tumor, hemorrhage, large <strong>in</strong>farction, abscess<br />
Men<strong>in</strong>gitis=encephalitis<br />
Subarachnoid hemorrhage<br />
Trauma<br />
Arteriovenous malformations with high blood flow overload<strong>in</strong>g venous return<br />
Intracranial or extracranial venous obstruction<br />
Secondary pseudotumor cerebri syndrome due to certa<strong>in</strong> systemic diseases, drugs, or pregnancy<br />
Source: J.J. Corbett, personal communication.<br />
neuroimag<strong>in</strong>g shows no structural lesion or hydrocephalus, then lumbar puncture is<br />
warranted. Studies should <strong>in</strong>clude an accurate open<strong>in</strong>g pressure, cerebrosp<strong>in</strong>al fluid<br />
(CSF) cell count and differential, glucose, prote<strong>in</strong>, cytology, Venereal Disease Research<br />
Laboratory (VDRL) test, and appropriate studies for microbial agents.<br />
Patients with a history of a ventriculoperitoneal shunt for hydrocephalus may<br />
develop papilledema, visual loss, or signs of a dorsal midbra<strong>in</strong> syndrome (see<br />
Chapter 14) due to shunt failure. Usually CT or MRI reveals recurrence of the<br />
hydrocephalus. Shunt malfunction may occur without ventriculomegaly, perhaps due<br />
to poor ventricular compliance and ‘‘stiff ventricles’’ (Katz, 1994; Lee, 1996; Newman,<br />
1994a). Thus shunt revision is <strong>in</strong>dicted when there are signs or symptoms of <strong>in</strong>creased<br />
<strong>in</strong>tracranial pressure, even if ventriculomegaly is absent, to prevent deterioration of<br />
visual function and potentially irreversible visual loss.<br />
What Is the Pseudotumor Cerebri Syndrome?<br />
Papilledema 135<br />
Pseudotumor cerebri is a diagnosis of exclusion. The modified Dandy criteria <strong>in</strong>clude (1)<br />
normal neuroimag<strong>in</strong>g studies (usually MRI); (2) normal CSF contents; (3) elevated<br />
open<strong>in</strong>g pressure; and (4) signs and symptoms related only to <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure (e.g., headache, papilledema, nonlocaliz<strong>in</strong>g sixth nerve palsy). Pseudotumor<br />
cerebri (PTC) is usually idiopathic but may be due to certa<strong>in</strong> systemic diseases, drugs,<br />
pregnancy, and <strong>in</strong>tracranial or extracranial venous obstruction.<br />
Obstruction or impairment of <strong>in</strong>tracranial venous dra<strong>in</strong>age may result <strong>in</strong> cerebral<br />
edema with <strong>in</strong>creased <strong>in</strong>tracranial pressure and papilledema. Tumors that occlude the<br />
posterior portion of the superior sagittal s<strong>in</strong>us or other cerebral venous s<strong>in</strong>uses may<br />
cause <strong>in</strong>creased <strong>in</strong>tracranial pressure. Septic or aseptic thrombosis or ligation of the<br />
cavernous s<strong>in</strong>us, lateral s<strong>in</strong>us, sigmoid s<strong>in</strong>us, or superior sagittal s<strong>in</strong>us may mimic PTC<br />
(Çelebisoy, 1999; Couban, 1991; Cremer, 1996; Daif, 1995; Gironell, 1997; Horton, 1992;<br />
Kim, 2000; Lam, 1992; Van den Br<strong>in</strong>k, 1996). A patient with neurofibromatosis type 2<br />
developed papilledema from obstruction of cerebrosp<strong>in</strong>al outflow at the arachnoid
136 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
granulations by diffuse convexity men<strong>in</strong>giomatosis (Thomas, 1999). Kieper et al noted<br />
that 5 of 107 patients who underwent suboccipital craniotomy or translabyr<strong>in</strong>th<strong>in</strong>e<br />
craniectomy developed PTC (Kieper, 1999). In each patient, the transverse s<strong>in</strong>us on the<br />
treated side was thrombosed, and patency of the contralateral s<strong>in</strong>us was confirmed on<br />
MRI. PTC has also been described after arteriovenous malformation embolization<br />
(Kollar, 1999). Sluggish flow <strong>in</strong> a venous varix after embolization, result<strong>in</strong>g <strong>in</strong><br />
thrombosis that was propagated to ve<strong>in</strong> of Galen, was the proposed mechanism.<br />
Ligation of one or both jugular ve<strong>in</strong>s (e.g., radical neck dissection), thrombosis of a<br />
central <strong>in</strong>travenous catheter <strong>in</strong> the chest or neck, subclavian ve<strong>in</strong> catheterization and<br />
arteriovenous fistula, the superior vena cava syndrome, or a glomus jugular tumor<br />
impair<strong>in</strong>g venous dra<strong>in</strong>age may also cause <strong>in</strong>creased <strong>in</strong>tracranial pressure. Osteopetrosis<br />
caus<strong>in</strong>g obstruction of venous outflow at the jugular foramen has also been<br />
reported (Ageli, 1994; Kiers, 1991; Lam, 1992; Siatkowski, 1999). Venous s<strong>in</strong>us thrombosis<br />
may be the mechanism for PTC reported <strong>in</strong> several conditions <strong>in</strong>clud<strong>in</strong>g systemic<br />
lupus erythematosus, essential thrombocythemia, prote<strong>in</strong> S deficiency, antithromb<strong>in</strong> III<br />
deficiency, the antiphospholipid antibody syndrome, activated prote<strong>in</strong> C resistance,<br />
paroxysmal nocturnal hemoglob<strong>in</strong>uria, Behçet’s disease, men<strong>in</strong>geal sarcoidosis,<br />
lymphoma, hypervitam<strong>in</strong>osis A, mastoiditis, and trich<strong>in</strong>osis (Akova, 1993; Biousse,<br />
1999; Daif, 1995; Farah, 1998; Gironell, 1997; Hauser, 1996; Leker, 1998; Mokri, 1993;<br />
Pelton, 1999; Provenzale, 1998). In fact, elevated <strong>in</strong>tracranial venous pressure is thought<br />
by some authors to be the universal mechanism of PTC of vary<strong>in</strong>g etiologies, <strong>in</strong>clud<strong>in</strong>g<br />
idiopathic PTC (Cremer, 1996; Karahalios, 1996; K<strong>in</strong>g, 1995). Higg<strong>in</strong>s et al presented a<br />
case of PTC thought secondary to bilateral transverse s<strong>in</strong>us stenosis discovered on<br />
venography that was treated successfully by <strong>in</strong>sert<strong>in</strong>g a self-expand<strong>in</strong>g stent across<br />
the stenosis <strong>in</strong> the right transverse s<strong>in</strong>us (Higg<strong>in</strong>s, 2002). These authors suggest that the<br />
transverse s<strong>in</strong>us pathology was not thrombosis but an idiopathic narrow<strong>in</strong>g of the<br />
transverse s<strong>in</strong>us bilaterally.<br />
Biousse et al noted that central venous thrombosis (CVT) can present with all the<br />
classic criteria for idiopathic pseudotumor cerebri, <strong>in</strong>clud<strong>in</strong>g normal CT imag<strong>in</strong>g and<br />
CSF contents (Biousse, 1999). Of 160 consecutive patients with CVT, 59 patients (37%)<br />
presented with isolated <strong>in</strong>tracranial hypertension. <strong>Neuro</strong>imag<strong>in</strong>g revealed <strong>in</strong>volvement<br />
of more than one venous s<strong>in</strong>us <strong>in</strong> 35 patients (59%); CT imag<strong>in</strong>g was normal <strong>in</strong> 27 of 50<br />
patients (54%). The superior sagittal s<strong>in</strong>us was <strong>in</strong>volved <strong>in</strong> 32 patients (54%) (isolated <strong>in</strong><br />
7) and the lateral s<strong>in</strong>us <strong>in</strong> 47 (80%) (isolated <strong>in</strong> 17). The straight s<strong>in</strong>us was thrombosed<br />
<strong>in</strong> eight patients, cortical ve<strong>in</strong>s were <strong>in</strong>volved <strong>in</strong> two patients, and deep cerebral<br />
ve<strong>in</strong>s <strong>in</strong> three, always <strong>in</strong> association with thrombosis <strong>in</strong> the superior sagittal s<strong>in</strong>us<br />
or lateral s<strong>in</strong>uses. Lumbar puncture was performed <strong>in</strong> 44 patients and showed elevated<br />
open<strong>in</strong>g pressure <strong>in</strong> 25 of 32 (78%) and abnormal CSF contents <strong>in</strong> 11 (25%). Etiologic<br />
risk factors <strong>in</strong>cluded local causes (7), surgery (1), <strong>in</strong>flammatory disease (18), <strong>in</strong>fection<br />
(2), cancer (1), postpartum (1), coagulopathies (11), and oral contraception (7).<br />
The cause was unknown <strong>in</strong> 11 cases (19%). <strong>An</strong>ticoagulants were used <strong>in</strong> 41 of 59<br />
patients (69%), steroids or acetazolamide <strong>in</strong> 26 (44%), therapeutic lumbar puncture <strong>in</strong> 44<br />
(75%), and surgical shunt <strong>in</strong> 1. Three patients had optic atrophy with severe visual loss,<br />
one died from carc<strong>in</strong>omatous men<strong>in</strong>gitis, and 55 (93%) had complete recovery<br />
(although visual field test<strong>in</strong>g was not systematically performed). The authors emphasized<br />
that MRI and MR venography should be considered <strong>in</strong> presumed isolated<br />
<strong>in</strong>tracranial hypertension.
Papilledema 137<br />
Among the 59 patients with isolated <strong>in</strong>creased <strong>in</strong>tracranial hypertension, 33 (56%)<br />
were female, but the authors did not record the patients’ weights. They note, however,<br />
that be<strong>in</strong>g a young, obese woman does not protect a patient from develop<strong>in</strong>g CVT, and<br />
therefore should not be used on an <strong>in</strong>dividual basis to rule out CVT. When MRI is not<br />
available, the authors suggest that conventional angiography be performed and, <strong>in</strong>deed,<br />
<strong>in</strong> another prospective study of 24 patients with apparently idiopathic PTC, angiography<br />
disclosed CVT <strong>in</strong> six patients (Teh<strong>in</strong>drazanarivelo, 1992). Increased blood flow<br />
and venous hypertension have also been implicated as the mechanism of papilledema<br />
noted <strong>in</strong> some patients with cerebral arteriovenous malformations (AVMs), especially<br />
dural AVMs and fistulas (Adelman, 1998; Çelebisoy, 1999; Chimowitz, 1990; Cockerell,<br />
1993; Cognard, 1998; David, 1995; Mart<strong>in</strong>, 1998; Rosenfield, 1991). Thus, we consider<br />
MR venography (and, <strong>in</strong> selected cases, MR angiography or even formal angiography)<br />
to <strong>in</strong>vestigate the possibility of venous s<strong>in</strong>us occlusion <strong>in</strong> patients with PTC, especially<br />
<strong>in</strong> patients with features not typical for idiopathic PTC (e.g., <strong>in</strong> th<strong>in</strong> patients, men, the<br />
elderly) (class III, level C). However, we found MR venography to be normal <strong>in</strong> 22<br />
consecutive obese females with idiopathic PTC (Lee and Brazis, 2000).<br />
K<strong>in</strong>g et al found that when transducer-measured <strong>in</strong>tracranial venous pressure is high<br />
<strong>in</strong> patients with idiopathic PTC, reduction of CSF pressure by removal of CSF<br />
predictably lowers the venous s<strong>in</strong>us pressure (K<strong>in</strong>g, 2002). This study <strong>in</strong>dicates that<br />
the <strong>in</strong>creased venous pressure <strong>in</strong> idiopathic PTC patients is caused by the elevated<br />
<strong>in</strong>tracranial pressure and not the reverse. Accord<strong>in</strong>g to Corbett and Digre, ‘‘The chicken<br />
is the CSF pressure elevation and the egg is the venous s<strong>in</strong>us pressure elevation’’<br />
(Corbett, 2002).<br />
The idiopathic narrow<strong>in</strong>g of the venous s<strong>in</strong>uses bilaterally noted <strong>in</strong> the case of PTC<br />
described by Higg<strong>in</strong>s et al may conceivably have been transverse s<strong>in</strong>us compression<br />
from <strong>in</strong>creased <strong>in</strong>tracranial pressure (Higg<strong>in</strong>s, 2002). Thus, venous occlusive disease<br />
and elevated venous pressure may well not be the mechanism of PTC <strong>in</strong> most idiopathic<br />
cases.<br />
Many systemic diseases, drugs, vitam<strong>in</strong> deficiencies and excesses, pregnancy, and<br />
hereditary conditions have been associated with the pseudotumor cerebri syndrome<br />
(secondary pseudotumor cerebri). These reported etiologies are listed <strong>in</strong> Table 7–8. In<br />
general, many of these reported associations may be co<strong>in</strong>cidental and anecdotal. Of<br />
those listed <strong>in</strong> Table 7–8, the etiologies most firmly associated with pseudotumor cerebri<br />
<strong>in</strong>clude drugs and systemic diseases (Ireland, 1990).<br />
Drugs<br />
The drugs or drug conditions associated with pseudotumor cerebri are hypervitam<strong>in</strong>osis<br />
A, steroid withdrawal, anabolic steroids, lithium, nalidixic acid, the <strong>in</strong>secticide<br />
chlordecone (Kepone), isoret<strong>in</strong>o<strong>in</strong>, ketoprofen (Orudis) or <strong>in</strong>domethac<strong>in</strong> <strong>in</strong> Bartter’s<br />
syndrome, thyroid replacement <strong>in</strong> hypothyroid children, danazol, all-trans-ret<strong>in</strong>oic<br />
acid (ATRA) or tret<strong>in</strong>o<strong>in</strong>, cyclospor<strong>in</strong>e, exogenous growth hormone, and probably<br />
tetracycl<strong>in</strong>e and m<strong>in</strong>ocycl<strong>in</strong>e.<br />
Systemic Diseases<br />
The systemic diseases or syndromes associated with pseudotumor cerebri are Behçet’s<br />
syndrome, renal failure, Addison’s disease, hypoparathyroidism, systemic lupus
138 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 7–8. Reported Etiologies of Secondary Pseudotumor Cerebri (PTC)<br />
Nutritional causes<br />
Hypervitam<strong>in</strong>osis A (Alemayehu, 1995; Donahue, 2000; Moskowitz, 1993; Scott, 1997; Sharieff,<br />
1996; Sirdofsky, 1994)<br />
Excessive carrot <strong>in</strong>take to ma<strong>in</strong>ta<strong>in</strong> weight loss likely exacerbated papilledema <strong>in</strong> one patient with<br />
PTC, due to high vitam<strong>in</strong> A levels (Donahue, 2000)<br />
Hypovitam<strong>in</strong>osis A (Panozzo, 1998)<br />
Vitam<strong>in</strong> D–deficient rickets (Alpan, 1991)<br />
Multiple vitam<strong>in</strong> deficiencies (Scott, 1997; Van Gelder, 1991)<br />
Parenteral hyperalimentation<br />
Drugs and other exogenous agents<br />
Nalidixic acid (Mukherjee, 1990; Scott, 1997)<br />
Tetracycl<strong>in</strong>e (Cuddihy, 1994; Gardner, 1995; Scott, 1997)<br />
M<strong>in</strong>ocycl<strong>in</strong>e (Chiu, 1998; Donnet, 1992; Lewis, 1997; Moskowitz, 1993; Torres, 1997)<br />
Nitrofuranto<strong>in</strong><br />
Penicill<strong>in</strong><br />
Ofloxac<strong>in</strong> (Getenet, 1993)<br />
Ciprofloxac<strong>in</strong> (W<strong>in</strong>row, 1990)<br />
Amiodarone (Ahmad, 1996; Borruat, 1993)<br />
Lithium (Ames, 1994; Dommisse, 1991; Lev<strong>in</strong>e, 1990)<br />
Phenyto<strong>in</strong><br />
Cytos<strong>in</strong>e arab<strong>in</strong>oside (Sacchi, 1999)<br />
Etret<strong>in</strong>ate<br />
Leuprorel<strong>in</strong> acetate (Arber, 1990)<br />
Ketam<strong>in</strong>e<br />
Indomethac<strong>in</strong> <strong>in</strong> Bartter’s syndrome<br />
Ketoprofen <strong>in</strong> Bartter’s syndrome<br />
Insecticide exposure: l<strong>in</strong>dane, chlordecone (Kepone) (Verderber, 1991)<br />
Steroids, <strong>in</strong>clud<strong>in</strong>g topical steroid and anabolic steroids (Scott, 1997)<br />
Steroid withdrawal (Liu, 1994; Scott, 1997)<br />
Oxytoc<strong>in</strong> (Mayer-Hubner, 1996)<br />
Growth hormone (Blethen, 1995; Francois, 1997; Koller, 1997; Malozowski, 1995; Maneatis, 2000;<br />
Rogers, 1999)<br />
Beta-human chorionic gonadotrop<strong>in</strong> (Haller, 1993)<br />
Depo-Provera (depot medroxyprogesterone)<br />
L-thyrox<strong>in</strong>e therapy for juvenile hypothyroidism (Campos, 1995; Misra, 1992; Raghavan, 1997)<br />
Endocr<strong>in</strong>e and metabolic dysfunction and pregnancy<br />
Pregnancy (<strong>in</strong>clud<strong>in</strong>g ectopic pregnancy) and postpartum (Daif, 1995; Koppel, 1990; McDonnell,<br />
1997; Shapiro, 1995)<br />
Menarche<br />
Turner syndrome<br />
Hyperthyroidism<br />
Hypothyroidism (Adams, 1994)<br />
Addison’s disease and crisis (Alexandrakis, 1993; Condulis, 1997; Leggio, 1995)<br />
Hypoparathyroidism and pseudohypoparathyroidism (Mada Mohan, 1993)<br />
Cush<strong>in</strong>g’s disease and post–pituitary surgery for Cush<strong>in</strong>g’s disease (Parfitt, 1994)<br />
Polycystic ovaries (Au Eong, 1997)<br />
(cont<strong>in</strong>ued)
Table 7–8. (cont<strong>in</strong>ued)<br />
Papilledema 139<br />
Catch-up growth follow<strong>in</strong>g severe nonorganic (physical and emotional abuse <strong>in</strong>clud<strong>in</strong>g food<br />
deprivation) failure to thrive (Alison, 1997)<br />
Familial hypomagnesemia-hypercalcuria (Gregoric, 2000)<br />
Rickets (Salaria, 2001)<br />
Systemic illnesses (<strong>in</strong>clud<strong>in</strong>g some caus<strong>in</strong>g venous occlusion)<br />
Systemic lupus erythematosus (Chaves-Carballo, 1999; Chevalier, 1992; Daif, 1995; Green, 1995;<br />
Horoshovski, 1995; Scott, 1997; Vachvanichsanong, 1992)<br />
Behçet’s syndrome (Bosch, 1995; Daif, 1995; Farah, 1998; Kansu, 1991)<br />
Cystic fibrosis (Bikangaga, 1996; Lucidi, 1993; Nasr, 1995; Scott, 1997)<br />
Reye’s syndrome<br />
<strong>An</strong>tiphospholipid antibody syndrome (Daif, 1995; Leker, 1998; Mokri, 1993; Orefice, 1995)<br />
Hematologic abnormalities and malignancies<br />
Iron-deficiency anemia (Scott, 1997; Tugal, 1994)<br />
Pernicious anemia and other megaloblastic anemias (Van Gelder, 1991)<br />
Thrombocythemia and thrombocytosis (Sussman, 1997; Teh<strong>in</strong>drazanarivelo, 1990)<br />
Cryofibr<strong>in</strong>ogenemia<br />
Abnormal fibr<strong>in</strong>ogen or <strong>in</strong>creased serum fibr<strong>in</strong>ogen (Sussman, 1997)<br />
Cryoglobul<strong>in</strong>emia<br />
Hodgk<strong>in</strong>’s disease<br />
Castleman’s disease<br />
Leukemia (Guymer, 1993; Saitoh, 2000)<br />
Myeloma (Wasan, 1992)<br />
Prote<strong>in</strong> S deficiency (Daif, 1995)<br />
Activated prote<strong>in</strong> C resistance (Provenzale, 1998)<br />
<strong>An</strong>tithromb<strong>in</strong> III deficiency (Daif, 1995; Sussman, 1997)<br />
<strong>An</strong>ticardiolip<strong>in</strong> antibodies (Kesler, 2000)<br />
Hemophilia A (factor VIII deficiency) (Jacome, 2001)<br />
Multicentric angiofollicular lymph node hyperplasia (Feigert, 1990)<br />
Paroxysmal nocturnal hemoglob<strong>in</strong>uria (Hauser, 1996)<br />
Thrombocytopenic purpura<br />
Polycythemia (Sussman, 1997)<br />
Chronic respiratory <strong>in</strong>sufficiency and the Pickwickian syndrome (Wol<strong>in</strong>, 1995)<br />
Sleep apnea (Lee, 2002b)<br />
Chronic renal failure and uremia (Chang, 1992; Guy, 1990; Scott, 1997)<br />
Renal or bone marrow transplantation (Avery, 1991, Katz, 1997; Obeid, 1997; Sheth, 1994)<br />
Infections and <strong>in</strong>flammatory diseases<br />
HIV <strong>in</strong>fection and AIDS (Gross, 1991; Javeed, 1995; Schwarz, 1995; Travero, 1993)<br />
Lyme disease (Kan, 1998; Scott, 1997)<br />
Typhoid fever (Moodley, 1990; Vargas, 1990)<br />
Familial Mediterranean fever (Gokalp, 1992)<br />
Trich<strong>in</strong>osis<br />
Chickenpox<br />
Otitis media (Scott, 1997)<br />
Acute purulent s<strong>in</strong>usitis (Kumar, 1999)<br />
<strong>Neuro</strong>sarcoidosis (Akova, 1993; Pelton, 1999; Redwood, 1990)<br />
Tolosa-Hunt syndrome (Nezu, 1995)<br />
(cont<strong>in</strong>ued)
140 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 7–8. (cont<strong>in</strong>ued)<br />
Mucopolysaccharidoses (Sheridan, 1994)<br />
After occipitocervical arthrodesis and immobilization <strong>in</strong> a halo vest (Daftari, 1995)<br />
Chiari I malformation (Milhorat, 1999)<br />
Guilla<strong>in</strong>-Barré syndrome (Weiss, 1991)<br />
Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (CIDP) (Fant<strong>in</strong>, 1993; Kaufman,<br />
1998; Midroni, 1996)<br />
Multiple sclerosis (Newman, 1994b)<br />
Crohn’s disease (Scott, 1997)<br />
Peripheral nerve sheath tumor of thigh (Hills, 1998)<br />
Familial pseudotumor cerebri (possible autosomal recessive or dom<strong>in</strong>ant transmission) (Fujiwara,<br />
1997; Kharode, 1992; Sant<strong>in</strong>elli, 1998)<br />
Homozygous tw<strong>in</strong>s (Fujiwara, 1997)<br />
erythematosus, and sarcoidosis (most of these likely cause pseudotumor cerebri<br />
syndrome by venous s<strong>in</strong>us obstruction or impairment of venous s<strong>in</strong>us dra<strong>in</strong>age).<br />
What Is Idiopathic Pseudotumor Cerebri?<br />
Idiopathic PTC (idiopathic <strong>in</strong>tracranial hypertension) is a disease typically of obese<br />
women <strong>in</strong> the childbear<strong>in</strong>g years (Arseni, 1992; Corbett, 1982; Ireland, 1990; Ja<strong>in</strong>, 1992;<br />
Kesler, 2001a; Radhakrishnan, 1994; Soler, 1998; Walker, 2001; Wall, 1991). Approximately<br />
10 to 15% of cases are male (Digre, 1988), and, when it occurs <strong>in</strong> children, there is<br />
usually no gender preference (Balcar, 1999; C<strong>in</strong>cirip<strong>in</strong>i, 1999; Lessell, 1992; Scott, 1997),<br />
although <strong>in</strong> some series girls outnumber boys (Gordon, 1997). Children with PTC,<br />
especially younger children, are less likely to be obese than adults with PTC (Balcar,<br />
1999; C<strong>in</strong>cirip<strong>in</strong>i, 1999; Scott, 1997). Even though men with PTC are less likely to be<br />
obese than woman, they tend to be more obese than controls (Digre, 1988). In a study<br />
from Israel, 18 of 134 patients with idiopathic PTC were men and 25% of the men were<br />
significantly overweight, as compared to 78% of the women (Kesler, 2001b). The<br />
occurrence of PTC <strong>in</strong> a man, especially a th<strong>in</strong> man, should raise the possibility of<br />
venous occlusive disease or a secondary PTC syndrome. African-American men appear<br />
to be at greater risk of visual loss. The <strong>in</strong>cidence of idiopathic PTC is approximately 1 or<br />
2 per 100,000, with a higher <strong>in</strong>cidence <strong>in</strong> obese women between the ages of 15 and 44<br />
years (4 to 21 per 100,000) (Kesler, 2001a; Radhakrishnan, 1993a,b). Table 7–9 lists the<br />
diagnostic criteria for idiopathic PTC.<br />
What Are the Risk Factors and <strong>Cl<strong>in</strong>ical</strong><br />
Characteristics of Idiopathic PTC?<br />
The most important risk factors for the development of idiopathic PTC <strong>in</strong>clude female<br />
sex, obesity, and recent weight ga<strong>in</strong> (Giuseffi, 1991; Ireland, 1990). Several conditions<br />
previously associated with idiopathic PTC are no more common <strong>in</strong> PTC than <strong>in</strong><br />
controls. In a retrospective case-control study of 40 patients with idiopathic PTC and<br />
39 age- and sex-matched controls, all forms of menstrual abnormalities, <strong>in</strong>cidence of
Table 7–9. Criteria for the Diagnosis of Idiopathic PTC<br />
Papilledema 141<br />
Increased <strong>in</strong>tracranial pressure must be documented <strong>in</strong> an alert and oriented patient without<br />
localiz<strong>in</strong>g neurologic f<strong>in</strong>d<strong>in</strong>gs (except for cranial nerve VI palsy)<br />
Sp<strong>in</strong>al fluid pressures between 200 and 250 mm H 2O may occur normally <strong>in</strong> obese patients, and<br />
when elevated sp<strong>in</strong>al fluid pressure is suspected, confirmation requires values greater than<br />
250 mm H2O (Corbett, 1983)<br />
The cerebrosp<strong>in</strong>al fluid should have normal contents (<strong>in</strong>clud<strong>in</strong>g prote<strong>in</strong> and glucose) with no<br />
cytologic abnormalities; occasionally the cerebrosp<strong>in</strong>al fluid prote<strong>in</strong> level may be low<br />
<strong>Neuro</strong>imag<strong>in</strong>g (MR imag<strong>in</strong>g with and without contrast and possibly MR venography) should be<br />
normal with no evidence of hydrocephalus, mass lesion, men<strong>in</strong>geal enhancement, or venous<br />
occlusive disease; neuroimag<strong>in</strong>g may often show the follow<strong>in</strong>g, which may be helpful <strong>in</strong><br />
establish<strong>in</strong>g the diagnosis of PTC (percentages from Brodsky and Vaphiades, 1998):<br />
Flatten<strong>in</strong>g of the posterior sclera (80% of patients)<br />
Distention of perioptic subarachnoid space (50% of patients)<br />
Enhancement (with gadol<strong>in</strong>ium) of the prelam<strong>in</strong>ar optic nerve (45% of patients)<br />
Empty sella (70% of patients)<br />
Intraocular protrusion of the prelam<strong>in</strong>ar optic nerve (30% of patients)<br />
Vertical tortuosity of the orbital optic nerve (40% of patients) (Brodsky, 1998; Gibby, 1993; Jacobson,<br />
1990; Manfre, 1995)<br />
No secondary cause (secondary PTC) is evident<br />
pregnancy, antibiotic use, and oral contraceptive use were equal <strong>in</strong> both groups (Ireland,<br />
1990). In another study compar<strong>in</strong>g 50 PTC patients with 100 age-matched controls, iron<br />
deficiency anemia, thyroid dysfunction, pregnancy, antibiotic <strong>in</strong>take, and the use of oral<br />
contraceptives were no more common <strong>in</strong> PTC patients than <strong>in</strong> controls (Giuseffi, 1991).<br />
The reason that obesity predisposes to PTC is unclear. Central obesity may raise<br />
<strong>in</strong>traabdom<strong>in</strong>al pressure, which <strong>in</strong>creases pleural pressure and cardiac fill<strong>in</strong>g pressure,<br />
<strong>in</strong>clud<strong>in</strong>g central venous pressure, lead<strong>in</strong>g to <strong>in</strong>creased <strong>in</strong>tracranial venous pressure<br />
and <strong>in</strong>creased <strong>in</strong>tracranial pressure (Sugerman, 1997). As noted above, elevated<br />
<strong>in</strong>tracranial venous pressure is thought by some authors to be the universal mechanism<br />
of PTC of various etiologies, <strong>in</strong>clud<strong>in</strong>g idiopathic PTC. However, the study of K<strong>in</strong>g et al<br />
cited above <strong>in</strong>dicates that the <strong>in</strong>creased venous pressure <strong>in</strong> idiopathic PTC patients is<br />
caused by the elevated <strong>in</strong>tracranial pressure and not the reverse (K<strong>in</strong>g, 2002). Idiopathic<br />
PTC may share a common pathogenesis with orthostatic edema, a condition <strong>in</strong> which<br />
there is evidence of dependent edema after prolonged stand<strong>in</strong>g (Friedman, 1998b).<br />
Seventy-seven percent of PTC patients had evidence of peripheral edema and 80% had<br />
significant orthostatic retention of sodium and water. Excretions of a standard sal<strong>in</strong>e<br />
load and of a tap water load were significantly impaired <strong>in</strong> the upright posture <strong>in</strong> the<br />
PTC patients with orthostatic edema compared to lean and obese but otherwise normal<br />
subjects. Orthostatic retention of water and sodium and consequent edema is similar <strong>in</strong><br />
patients with idiopathic PTC and orthostatic edema. This suggests that these two<br />
disorders may have a common pathogenesis.<br />
Elevated vitam<strong>in</strong> A levels have been noted <strong>in</strong> patients with idiopathic PTC (Jacobson,<br />
1999). Serum ret<strong>in</strong>ol concentrations were significantly higher <strong>in</strong> patients with idiopathic<br />
PTC compared to controls (Selhorst, 2000), even after adjust<strong>in</strong>g for age and body mass<br />
<strong>in</strong>dex. Patients may <strong>in</strong>gest an abnormally large amount of vitam<strong>in</strong> A, metabolize it
142 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
abnormally, or be unusually sensitive to its effects. Alternatively, elevated levels of<br />
serum ret<strong>in</strong>ol may reflect an epiphenomenon of another variable not measured or a<br />
nonspecific effect of elevated ret<strong>in</strong>ol b<strong>in</strong>d<strong>in</strong>g capacity (Jacobson, 1999).<br />
Endocr<strong>in</strong>ologic abnormalities may be more common <strong>in</strong> men with PTC (Lee, 2002c). In<br />
a study of eight men with PTC, two had abnormal estradiol levels, four had abnormal<br />
follicle-stimulat<strong>in</strong>g hormone (FSH) and lute<strong>in</strong>iz<strong>in</strong>g hormone (LH) levels, and seven had<br />
low testosterone levels (Lee, 2002c).<br />
What Are the Symptoms of PTC?<br />
The most common symptoms of PTC <strong>in</strong>clude headache, transient obscurations of<br />
vision, pulsatile t<strong>in</strong>nitus, and diplopia (Giuseffi, 1991; Ireland, 1990; Wall, 1991). In a<br />
prospective study of 50 idiopathic PTC patients (92% women; mean age, 32 years; 92%<br />
obese), symptoms <strong>in</strong>cluded headache (94%), transient visual obscurations (68%),<br />
<strong>in</strong>tracranial noises (58%), susta<strong>in</strong>ed visual loss (26%), photopsia (54%), diplopia<br />
(38%), and retrobulbar pa<strong>in</strong> (44%) (Wall, 1991). The headaches <strong>in</strong> patients with PTC<br />
may be constant or <strong>in</strong>termittent, and <strong>in</strong> 93% of patients they are reported to be the most<br />
severe headache ever (Wall, 1990). The headache may often be pulsatile, be of gradually<br />
<strong>in</strong>creas<strong>in</strong>g <strong>in</strong>tensity dur<strong>in</strong>g the day, awaken the patient at night, be precipitated by<br />
changes <strong>in</strong> posture, and be transiently relieved by lumbar puncture (Wall, 1990). Pa<strong>in</strong> <strong>in</strong><br />
a cervical nerve root distribution (possibly from a dilated nerve root sleeve) or retroocular<br />
pa<strong>in</strong> with eye movement, uncommon with other headache disorders, may help<br />
to differentiate this headache syndrome (Wall, 1990). There is no clear correlation<br />
between the height of CSF pressure and the severity of the headache. Transient visual<br />
obscurations last seconds, may be unilateral or bilateral, and related to changes <strong>in</strong><br />
posture. They do not correlate with the degree of <strong>in</strong>tracranial hypertension or the extent<br />
of disc swell<strong>in</strong>g, and are not considered to be harb<strong>in</strong>gers of permanent visual loss<br />
(Corbett, 1982; Giuseffi, 1991). Intracranial noises are common with PTC and are<br />
perhaps due to transmission of <strong>in</strong>tensified vascular pulsations via CSF under high<br />
pressure to the walls of the venous s<strong>in</strong>uses (Sismanis, 1990). The pulsatile t<strong>in</strong>nitus may<br />
be audible to others (Biousse, 1998). In fact, PTC without papilledema has been reported<br />
<strong>in</strong> patients with pulsatile t<strong>in</strong>nitus (Felton, 1995; Wang, 1996). Diplopia is often mild and<br />
usually due to a sixth cranial nerve palsy, presumably a nonlocaliz<strong>in</strong>g sign of raised<br />
<strong>in</strong>tracranial pressure.<br />
In a study of 101 patients with PTC, other m<strong>in</strong>or symptoms <strong>in</strong>cluded neck stiffness <strong>in</strong><br />
31 patients, distal extremity paresthesias <strong>in</strong> 31, t<strong>in</strong>nitus <strong>in</strong> 27, jo<strong>in</strong>t pa<strong>in</strong>s <strong>in</strong> 13, low back<br />
pa<strong>in</strong> <strong>in</strong> 13, and gait <strong>in</strong>stability <strong>in</strong> 4 (Round, 1988). These m<strong>in</strong>or symptoms resolved<br />
promptly upon lower<strong>in</strong>g of the <strong>in</strong>tracranial pressure. Stiff neck and strabismus may be<br />
the most common present<strong>in</strong>g symptoms <strong>in</strong> children with PTC (C<strong>in</strong>cirip<strong>in</strong>i, 1999). Sleeprelated<br />
breath<strong>in</strong>g problems are common <strong>in</strong> PTC patients and may be a risk factor<br />
(Marcus, 2001). Patients with idiopathic PTC are significantly more affected by hardships<br />
associated with health problems than age- and weight-matched controls and have<br />
higher levels of depression and anxiety (Kle<strong>in</strong>schmidt, 2000). Other rare and exceptional<br />
cl<strong>in</strong>ical abnormalities that have been described <strong>in</strong> patients with PTC are listed <strong>in</strong><br />
Table 7–10.
Table 7–10. Rare and Exceptional F<strong>in</strong>d<strong>in</strong>gs Described with PTC<br />
What Are the Signs of Pseudotumor Cerebri?<br />
Papilledema 143<br />
Fourth cranial nerve palsy (Lee, 1995; Speer, 1999)<br />
Third cranial nerve palsy<br />
Sixth cranial nerve palsy (unilateral) without papilledema (Krishna, 1998)<br />
Bilateral sixth and fourth cranial nerve palsies (Patton, 2000)<br />
Skew deviation<br />
Complete external ophthalmoplegia (Friedman, 1998a)<br />
Bilateral total <strong>in</strong>ternal and external ophthalmoplegia<br />
Internuclear ophthalmoplegia with vertical gaze paresis with or without ptosis (Friedman, 1997,<br />
1998a)<br />
Vertical gaze palsy (Friedman, 1998a)<br />
Divergence <strong>in</strong>sufficiency (Jacobson, 2000)<br />
Sensory exotropia or comitant esotropia <strong>in</strong> children (C<strong>in</strong>cirip<strong>in</strong>i, 1999)<br />
Ptosis (Friedman, 1998a)<br />
Lid retraction (Friedman, 1998a)<br />
Trigem<strong>in</strong>al neuropathy (Davenport, 1994)<br />
Unilateral or bilateral facial nerve palsy (Bakshi, 1992; Capobianco, 1997; Selky, 1994a)<br />
Hemifacial spasm (Mayer, 1996; Selky, 1994a)<br />
Cerebrosp<strong>in</strong>al fluid rh<strong>in</strong>orrhea (Camras, 1998; Clark, 1994)<br />
Transient partial pituitary deficiency (De Paepe, 1993)<br />
Fatal tonsillar herniation after lumbar puncture (Sullivan, 1991)<br />
Atypical ophthalmoscopic f<strong>in</strong>d<strong>in</strong>gs and visual abnormalities:<br />
Visual field loss despite resolution of papilledema (Golnik, 1999)<br />
Gaze-evoked amaurosis (O’Duffy, 1998)<br />
Acute visual loss secondary to anterior ischemic optic neuropathy<br />
Acute visual loss due to central ret<strong>in</strong>al artery occlusion<br />
Acute visual loss due to branch ret<strong>in</strong>al artery occlusion (Lam, 1992; Liu, 1994)<br />
Acute visual loss due to central ret<strong>in</strong>al ve<strong>in</strong> occlusion (Chern, 1991)<br />
Visual loss (occasionally acute) due to macular disease, <strong>in</strong>clud<strong>in</strong>g chorioret<strong>in</strong>al striae, pigmentary<br />
disturbances, exudates, macular edema, nerve fiber layer hemorrhages, subret<strong>in</strong>al<br />
hemorrhages from neovascular membranes, or subret<strong>in</strong>al scars (Akova, 1994; Carter, 1995;<br />
Liu, 1994; Talks, 1998)<br />
Acquired hyperopia with choroidal folds (Jacobson, 1995; Talks, 1998)<br />
Coexistence with optic nerve head drusen (Krasnitz, 1997)<br />
Optociliary ‘‘shunt’’ (collaterals) vessels (may resolve after ONSF) (Brazier, 1996)<br />
Papilledema is found <strong>in</strong> most cases of PTC. It may be asymmetric, rarely unilateral, and<br />
even occasionally absent (Chari, 1991; Greenfield, 1997; Krishna, 1998; Marcelis, 1991;<br />
Mathew, 1996; Saito, 1999; Tourn, 1996; Wall, 1998; Wang, 1996). In one series, 10% of<br />
478 patients with PTC had asymmetric papilledema, and visual loss was most<br />
pronounced <strong>in</strong> the eye with the higher grade of papilledema (Wall, 1998). If optic<br />
atrophy is present unilaterally, the disc swell<strong>in</strong>g will be unilateral <strong>in</strong> the opposite eye,<br />
thus mimick<strong>in</strong>g the Foster Kennedy syndrome (Tourn, 1996). In the patient described by
144 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Saito et al with unilateral disc swell<strong>in</strong>g as a manifestation of PTC related to cyclospor<strong>in</strong><br />
therapy for leukemia, the right disc was thought to be spared because of leukemic<br />
<strong>in</strong>filtration caus<strong>in</strong>g constriction of the optic nerve sheath (Saito, 1999). If no papilledema<br />
is evident, there is no risk of visual loss no matter how high the <strong>in</strong>tracranial pressure; <strong>in</strong><br />
the <strong>in</strong>dividual patient, the severity of papilledema cannot predict the severity of visual<br />
loss. Isolated <strong>in</strong>tracranial pressure without papilledema may present as chronic daily<br />
headache. Quattrone et al <strong>in</strong>vestigated 114 consecutive patients with chronic daily<br />
headache with MR venography and found that 11 (9.6%) had cerebral venous thrombosis<br />
affect<strong>in</strong>g one or both transverse s<strong>in</strong>uses and half of these patients had isolated<br />
<strong>in</strong>tracranial hypertension without papilledema (Quattrone, 2001).<br />
Visual field loss is the major causes of morbidity <strong>in</strong> PTC (Corbett, 1982; Wall, 1991).<br />
Visual acuity loss and optic atrophy may occur <strong>in</strong> some patients. Hypertension and<br />
recent weight ga<strong>in</strong> have been reported to be significant risk factors for visual loss<br />
(Corbett, 1982). Often the patient is unaware of peripheral visual field dysfunction, and<br />
Snellen acuity test<strong>in</strong>g is a poor <strong>in</strong>dicator of early visual deficit <strong>in</strong> PTC. The papilledema<br />
causes optic nerve fiber loss that results <strong>in</strong> field constriction and nerve fiber bundle<br />
defects (Corbett, 1982; Wall, 1991). Optic nerve diameter changes on ultrasound <strong>in</strong> PTC<br />
are associated with perimetric threshold loss; PTC functional deficits may thus be<br />
related to the degree of distention of optic nerve sheaths as a result of an <strong>in</strong>creased CSF<br />
pressure (Salgarello, 1996). Bl<strong>in</strong>d spot enlargement is commonly encountered, yet it is<br />
more a reflection of the disc swell<strong>in</strong>g itself <strong>in</strong>stead of optic nerve damage and is<br />
improved with refraction. Acute visual acuity loss is rare but may occur by the<br />
mechanisms listed <strong>in</strong> Table 7–10 (e.g., anterior ischemic optic neuropathy, ret<strong>in</strong>al<br />
artery or ve<strong>in</strong> occlusion, or subret<strong>in</strong>al hemorrhage from neovascular membranes).<br />
The frequency of visual field loss and acuity loss with PTC is variable, but <strong>in</strong> one<br />
study field loss was noted <strong>in</strong> 75% of eyes us<strong>in</strong>g manual strategies and 78% of eyes us<strong>in</strong>g<br />
automated threshold perimetry (Wall, 1987). Motion perimetry abnormalities correlate<br />
well with static perimetry abnormalities <strong>in</strong> patients with PTC, and <strong>in</strong>deed may identify<br />
nerve fiber bundle defects not detected with conventional perimetry (Wall, 1995).<br />
Contrast sensitivity test<strong>in</strong>g is also a relatively sensitive means of assess<strong>in</strong>g optic<br />
nerve damage <strong>in</strong> patients with PTC (Stavroua, 1997). Rowe and Sarkies, however,<br />
noted that visual field test<strong>in</strong>g, as opposed to visual acuity and contrast sensitivity<br />
test<strong>in</strong>g, is the most sensitive <strong>in</strong>dicator of visual loss <strong>in</strong> PTC patients (Rowe, 1998).<br />
What Is the Evaluation of Pseudotumor<br />
Cerebri?<br />
All patients with PTC require a thorough history, especially regard<strong>in</strong>g medication use,<br />
pregnancy, <strong>in</strong>tercurrent illnesses, and recent weight ga<strong>in</strong>. Most cases do not require<br />
laboratory evaluation (class II, level C). Some patients may require blood work (e.g.,<br />
sedimentation rate, complete blood count, syphilis serology, calcium, phosphate,<br />
creat<strong>in</strong><strong>in</strong>e, and electrolytes). All patients should undergo complete ophthalmologic<br />
exam<strong>in</strong>ations <strong>in</strong>clud<strong>in</strong>g formal perimetry (e.g., Goldmann and=or automated) and optic<br />
disc stereo photography. Secondary causes of PTC should especially be considered <strong>in</strong><br />
men, <strong>in</strong> th<strong>in</strong> patients, and <strong>in</strong> patients younger than 15 years or older than 45 years<br />
(atypical PTC).
Cognard et al noted that dural arteriovenous fistulas may present with isolated<br />
<strong>in</strong>tracranial hypertension, mimick<strong>in</strong>g idiopathic PTC, and thought that cerebral angiography<br />
should be performed to evaluate for their presence <strong>in</strong> all patients with PTC<br />
(Cognard, 1998). Biousse et al noted that CVT may present with PTC, and that this<br />
diagnosis should be considered even <strong>in</strong> women with the typical body habitus of<br />
idiopathic PTC (Biousse, 1999). In another prospective study of 24 patients with<br />
apparently idiopathic pseudotumor cerebri, angiography disclosed CVT <strong>in</strong> six patients<br />
(Teh<strong>in</strong>drazanarivelo, 1992). We recommend that patients with PTC undergo MRI of the<br />
head (class II–III, level B) with MR venography to evaluate for venous occlusive disease,<br />
but we recommend cerebral angiography only <strong>in</strong> select cases (class III, level C). If<br />
venous occlusive disease is discovered, then evaluation for a hypercoagulable state and<br />
vasculitis should be performed (class III, level C).<br />
What Is the Treatment for PTC?<br />
The treatment of PTC has two major goals: the alleviation of symptoms and preservation<br />
of visual function. We suggest a management plan, adapted from that<br />
recommended by Corbett and Thompson, which is outl<strong>in</strong>ed <strong>in</strong> Table 7–11.<br />
What Is the Medical Management of PTC?<br />
Papilledema 145<br />
Some patients require no treatment if symptoms are m<strong>in</strong>imal and visual function is<br />
normal. All patients require serial monitor<strong>in</strong>g of visual function, especially visual fields,<br />
to observe closely for signs of visual impairment. The use of visual evoked responses<br />
and repeated measurements of <strong>in</strong>tracranial pressure by lumbar puncture do not provide<br />
data that help to guide therapeutic decisions (Corbett, 1989). Weight reduction, <strong>in</strong>clud<strong>in</strong>g<br />
surgically <strong>in</strong>duced weight reduction <strong>in</strong> morbidly obese patients, may improve the<br />
papilledema and reduce <strong>in</strong>tracranial pressure (Johnson, 1998; Kupersmith, 1997, 1998;<br />
Newborg, 1974; Sugerman, 1995). For example, <strong>in</strong> one study, n<strong>in</strong>e patients placed on a<br />
salt-restricted, rapid weight reduction rice diet showed improvement <strong>in</strong> papilledema<br />
(mean weight 261 pounds before treatment and 187 pounds after treatment) (Newborg,<br />
1974). In another study, all eight morbidly obese patients with PTC who underwent<br />
gastric surgery had complete resolution of their papilledema. There was resolution or<br />
marked reduction <strong>in</strong> headache, resolution of t<strong>in</strong>nitus, and a decrease <strong>in</strong> CSF pressure<br />
from a mean of 353 to a mean of 168 mm of H2O follow<strong>in</strong>g a mean weight loss of 57 kg<br />
when measured at 34 months after surgery (Sugerman, 1995). In another study of 24<br />
severely obese women with idiopathic PTC, 23 were treated by gastric bypass surgery<br />
and one underwent laparoscopic adjustable gastric b<strong>in</strong>d<strong>in</strong>g (Sugerman, 1999). At 1 year<br />
after surgery, 19 patients lost an average of 45 12 kg, which was 71% 18% of their<br />
excess weight. Five patients were lost to follow-up and four were followed for less than<br />
1 year after their surgery. Surgically <strong>in</strong>duced weight loss was associated with resolution<br />
of headache and pulsatile t<strong>in</strong>nitus <strong>in</strong> all but one patient with<strong>in</strong> 4 months of the<br />
procedure. Of the 19 patients not lost to follow-up, 2 rega<strong>in</strong>ed the weight with<br />
recurrence of their headache and pulsatile t<strong>in</strong>nitus. The authors concluded that bariatric<br />
surgery should be considered the ‘‘procedure of choice for severely obese patients<br />
with PTC and is shown to have a much higher rate of success than CSF-peritoneal
146 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 7–11. Management of Idiopathic PTC<br />
Confirm cl<strong>in</strong>ical diagnosis (diagnosis of exclusion)<br />
Medical treatment recommendations<br />
Acetazolamide (e.g., Diamox sequels 500 mg qhs for 3 days then 500 mg bid—up to 2 to<br />
4 g per day) if no contra<strong>in</strong>dications<br />
Consider furosemide (Lasix) if acetazolamide-<strong>in</strong>tolerant<br />
Other medications have not been proven but may <strong>in</strong>deed be useful (e.g., topiramate,<br />
methazolamide)<br />
Avoid corticosteroids if possible (cause weight ga<strong>in</strong> and other side effects) except possibly IV<br />
steroids for acute visual loss<br />
Expla<strong>in</strong> medication side effects of acetazolamide<br />
Paresthesias, anorexia, malaise, t<strong>in</strong>-like taste, and fatigue may limit use<br />
May cause nausea and vomit<strong>in</strong>g, electrolyte changes, kidney stones<br />
Avoid if severe allergic reaction to sulfa drugs (obta<strong>in</strong> <strong>in</strong>formation about type of reaction)<br />
Caution dur<strong>in</strong>g pregnancy:<br />
Relatively contra<strong>in</strong>dicated, especially dur<strong>in</strong>g first 20 weeks<br />
Potential teratogenicity (category C agent)<br />
Consult with obstetrics and gynecology if benefit outweighs risk<br />
Usually avoid diuretics and caloric restriction if pregnant<br />
Encourage weight reduction<br />
Treat headache symptomatically<br />
Consider diagnosis of and treat associated sleep apnea<br />
Surgical treatment (if fail, <strong>in</strong>tolerant to, or noncompliant with maximal medical therapy)<br />
Optic nerve sheath fenestration<br />
Lumboperitoneal shunt procedure<br />
Indications for surgery:<br />
New worsen<strong>in</strong>g of visual field defect*<br />
Enlargement of previously exist<strong>in</strong>g visual field defect*<br />
Reduced visual acuity not due to macular edema<br />
Presence of severe visual loss (20=40 or worse) <strong>in</strong> one or both eyes at time of <strong>in</strong>itial exam<strong>in</strong>ation<br />
<strong>An</strong>ticipated hypotension <strong>in</strong>duced by treatment of high blood pressure or renal dialysis<br />
Psychosocial reasons, such as patient’s <strong>in</strong>ability to perform visual field studies, noncompliance<br />
with medications, or it<strong>in</strong>erant lifestyle<br />
Headache unresponsive to standard headache medications<br />
Follow-up visit <strong>in</strong>tervals<br />
Return monthly (similar <strong>in</strong>terval) until disc edema resolved (usually several months)<br />
Perform formal visual fields and complete eye exam<br />
Stereo optic disc photographs (at <strong>in</strong>itial evaluation and with changes <strong>in</strong> disc)<br />
*Bl<strong>in</strong>d spot enlargement should not be considered significant visual loss (refractive).<br />
shunt<strong>in</strong>g ... as well as provid<strong>in</strong>g resolution of additional obesity co-morbidity’’<br />
(Sugerman, 1999). In a retrospective study, Kupersmith et al noted that weight reduction<br />
sped recovery from PTC <strong>in</strong> women but may not have def<strong>in</strong>itely improved the eventual<br />
visual outcome (Kupersmith, 1997). Kupersmith et al later noted that weight reduction<br />
was associated with a more rapid recovery of papilledema and visual field dysfunction<br />
<strong>in</strong> patients with idiopathic PTC (weight loss greater than or equal to 2.5 kg dur<strong>in</strong>g any
3-month <strong>in</strong>terval <strong>in</strong> the study) (Kupersmith, 1998). Johnson et al noted that approximately<br />
6% weight loss was associated with resolution of marked papilledema <strong>in</strong> obese<br />
PTC patients (Johnson, 1998).<br />
Medical treatments for PTC <strong>in</strong>clude carbonic anhydrase <strong>in</strong>hibitors (e.g., acetazolamide),<br />
loop diuretics, and corticosteroids. Acetazolamide <strong>in</strong> doses up to 2 to 4 g per<br />
day has proven effective <strong>in</strong> some patients with PTC (Schoeman, 1994). Acetazolamide<br />
should probably be avoided dur<strong>in</strong>g pregnancy, especially dur<strong>in</strong>g the first 20 weeks,<br />
because of potential teratogenic effects <strong>in</strong> animals. The teratogenic effect <strong>in</strong> humans is<br />
not well documented. Caloric restriction and the use of other diuretics are also relatively<br />
contra<strong>in</strong>dicated dur<strong>in</strong>g pregnancy. Other carbonic anhydrase <strong>in</strong>hibitors, such as methazolamide<br />
(Neptazane), are often used <strong>in</strong> acetazolamide-<strong>in</strong>tolerant patients but their<br />
efficacy has not been proven. Furosemide (Lasix) <strong>in</strong>hibits CSF production and may have<br />
an additive effect with acetazolamide. The use of furosemide alone has not been<br />
systematically studied. There is one report of eight children treated with comb<strong>in</strong>ed<br />
therapy of acetazolamide and furosemide. All patients had a rapid cl<strong>in</strong>ical response<br />
with resolution of papilledema, reduction <strong>in</strong> the mean CSF pressure after the first<br />
week of treatment, and normalization of pressure with<strong>in</strong> 6 weeks of start<strong>in</strong>g therapy<br />
(Schoeman, 1994).<br />
Digox<strong>in</strong> has also been reported to be a successful treatment <strong>in</strong> one small group of<br />
patients (Goodw<strong>in</strong>, 1990). Intravenous <strong>in</strong>domethac<strong>in</strong> transiently reduced <strong>in</strong>tracranial<br />
pressure <strong>in</strong> seven patients with PTC, and five patients treated with oral <strong>in</strong>domethac<strong>in</strong><br />
(75 mg per day) along with acetazolamide reported improvement <strong>in</strong> headaches and<br />
t<strong>in</strong>nitus and improvement <strong>in</strong> papilledema and visual fields (Forderreuther, 2000). This<br />
drug might be an alternative for the treatment of PTC. Corticosteroids may be<br />
efficacious <strong>in</strong> the short run, but the complications of this medication, especially <strong>in</strong> the<br />
chronic treatment of an already obese <strong>in</strong>dividual, have resulted <strong>in</strong> most cl<strong>in</strong>icians<br />
suggest<strong>in</strong>g that their use be avoided (Corbett, 1989). Liu et al treated four patients with<br />
acute, severe visual loss associated with PTC with a comb<strong>in</strong>ation of high-dose<br />
methylprednisolone (250 mg four times a day for 5 days followed by an oral taper),<br />
acetazolamide, and ranitid<strong>in</strong>e (Liu, 1994). In addition to severe disc edema, one patient<br />
had a serous detachment of both maculae and lipid deposition, one had unilateral<br />
macular star, and one had a monocular branch ret<strong>in</strong>al artery occlusion. These latter<br />
three patients experienced rapid and last<strong>in</strong>g improvement <strong>in</strong> visual acuity, visual fields,<br />
papilledema, and symptoms, whereas the fourth patient did not improve and required<br />
optic nerve sheath fenestration. The authors suggested this comb<strong>in</strong>ation treatment for<br />
patients with acute, severe visual loss associated with florid papilledema and suggested<br />
surgical treatment if no immediate improvement occurs (Liu, 1994).<br />
What About Repeated Lumbar Punctures?<br />
Papilledema 147<br />
Repeated lumbar punctures have never been systematically studied for the treatment of<br />
PTC. As these procedures are uncomfortable, of questionable benefit, and potentially<br />
associated with complications (e.g., <strong>in</strong>fection, <strong>in</strong>trasp<strong>in</strong>al epidermoid tumors [Corbett,<br />
1983]), we feel that they should not be performed therapeutically, except perhaps with<br />
PTC <strong>in</strong> pregnancy (class III, level C).
148 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What Is the Surgical Management of PTC?<br />
When medical therapy fails or when visual dysfunction deteriorates, surgical therapies<br />
for PTC should be considered (Corbett, 1989). The <strong>in</strong>dications for surgical therapy, as<br />
suggested by Corbett and Thompson, are outl<strong>in</strong>ed <strong>in</strong> Table 7–11 (Corbett, 1989). The<br />
two ma<strong>in</strong> procedures performed <strong>in</strong>clude lumboperitoneal shunt (LPS) and optic nerve<br />
sheath fenestration (ONSF). Various authorities have vehemently advocated one or the<br />
other procedure and both have their advantages and disadvantages, but there has been<br />
no prospective study compar<strong>in</strong>g the efficacy of the two procedures.<br />
Lumboperitoneal shunt can relieve headache, diplopia, and papilledema, and can<br />
reverse visual loss (<strong>An</strong>giari, 1992; Burgett, 1997; Eggenberger, 1996; Johnston, 1988a,b;<br />
Lundar, 1990; Rosenberg, 1993). This procedure may be performed if warranted <strong>in</strong><br />
pregnancy (Shapiro, 1995). Eggenberger et al retrospectively studied 27 patients with<br />
PTC treated with at least one LPS to ascerta<strong>in</strong> the efficacy of this treatment<br />
(Eggenberger, 1996). The <strong>in</strong>dications for LPS were <strong>in</strong>tractable headache <strong>in</strong> 18 patients<br />
(67%) and progressive optic neuropathy <strong>in</strong> 14 patients (52%). Visual function returned<br />
to normal <strong>in</strong> both eyes of six patients, showed no change <strong>in</strong> either eye <strong>in</strong> four patients,<br />
and improved <strong>in</strong> at least one eye <strong>in</strong> the rema<strong>in</strong><strong>in</strong>g four. Four patients had unilateral and<br />
one had bilateral sixth nerve palsies; all completely resolved postsurgery. The average<br />
duration of follow-up for this population was 77 months (mean 47 months). A<br />
function<strong>in</strong>g LPS was successful <strong>in</strong> alleviat<strong>in</strong>g symptoms <strong>in</strong> all of the patients studied<br />
and no patient with a function<strong>in</strong>g shunt compla<strong>in</strong>ed of shunt-related symptoms, such as<br />
low-pressure headache or abdom<strong>in</strong>al pa<strong>in</strong>, with<strong>in</strong> 2 months after the shunt was<br />
performed.<br />
The major complication of LPS is shunt failure requir<strong>in</strong>g revision. The authors<br />
concluded that placement of a LPS is a satisfactory treatment for the majority of<br />
patients with PTC who require surgical therapy for the disorder even though some<br />
patients ultimately require multiple shunt revisions.<br />
Rosenberg et al reviewed the efficacy of cerebrosp<strong>in</strong>al diversion procedures for PTC<br />
<strong>in</strong> patients from six different <strong>in</strong>stitutions (Rosenberg, 1993). Thirty-seven patients<br />
underwent a total of 73 LPS and 10 ventricular shunts. Only 14 patients rema<strong>in</strong>ed<br />
‘‘cured’’ after a s<strong>in</strong>gle surgical procedure. The average time between shunt <strong>in</strong>sertion and<br />
shunt replacement was 9 months, although 64% of the shunts lasted less than 6 months.<br />
Shunt failure (recurrent papilledema or <strong>in</strong>creased CSF pressure on lumbar puncture)<br />
(55%) and low-pressure headaches (21%) were the most common <strong>in</strong>dications for<br />
reoperation. Other reasons for shunt replacement <strong>in</strong>cluded <strong>in</strong>fection, abdom<strong>in</strong>al pa<strong>in</strong>,<br />
radicular pa<strong>in</strong>, operative complications, and CSF leak. The vision of most patients<br />
improved (13) or stabilized (13) postoperatively. However, three patients who had<br />
<strong>in</strong>itially improved subsequently lost vision, six had a postoperative decrease <strong>in</strong> vision,<br />
two patients improved <strong>in</strong> one eye but worsened postoperatively <strong>in</strong> the other, and four<br />
lost vision despite apparently adequate shunt function. Shunt failure with relapse of<br />
PTC occurred as late as 7 years after <strong>in</strong>sertion. The authors concluded that CSF<br />
diversion procedures have a significant failure rate as well as a high frequency of<br />
side effects. Johnston et al reported 36 patients who dur<strong>in</strong>g follow-up required a total<br />
of 85 shunt<strong>in</strong>g procedures with an overall complication rate of 52% and a failure rate of<br />
48% (Johnston, 1988a).<br />
Burgett et al retrospectively analyzed cl<strong>in</strong>ical data from 30 patients who underwent<br />
LPS for PTC and found LPS an effective means of acutely lower<strong>in</strong>g <strong>in</strong>tracranial pressure
(Burgett, 1997). Symptoms of <strong>in</strong>creased <strong>in</strong>tracranial pressure improved <strong>in</strong> 82% of<br />
patients, and five patients (29%) demonstrated total resolution of all symptoms.<br />
Among 14 patients with impaired visual acuity, 10 (71%) improved by at least two<br />
Snellen l<strong>in</strong>es. Worsen<strong>in</strong>g of vision occurred <strong>in</strong> only one eye. Of 28 eyes with abnormal<br />
Goldmann perimetry, 18 (64%) improved and none worsened. The <strong>in</strong>cidence of serious<br />
complications was low, but the major drawback was a need for frequent revisions <strong>in</strong> a<br />
few patients (30 patients underwent a total of 126 revisions with the mean revision rate<br />
of 4.2 per patient). The authors suggested that LPS should be considered the first<br />
surgical procedure for patients with PTC with severe visual loss at presentation or with<br />
<strong>in</strong>tractable headache (with or without visual loss). After shunt<strong>in</strong>g, it is important to<br />
identify patients who are shunt <strong>in</strong>tolerant (Burgett, 1997).<br />
Thus, CSF diversion procedures, especially LPS, are often effective <strong>in</strong> controll<strong>in</strong>g PTC,<br />
and although placement of the shunt is generally safe, any operation performed under<br />
general anesthesia carries some risk, and there is at least one perioperative death<br />
reported follow<strong>in</strong>g LPS (Eisenberg, 1971). Shunt obstruction is the most common<br />
complication (Burgett, 1997; Chumas, 1993b; Eggenberger, 1996; Rosenberg, 1993)<br />
followed by secondary <strong>in</strong>tracranial hypotension caused by excessive dra<strong>in</strong>age of the<br />
CSF via the LPS (Burgett, 1997; Chumas, 1993b; Eggenberger, 1996; Johnston, 1988a,b;<br />
Rosenberg, 1993; Sell, 1995). Symptoms of <strong>in</strong>tracranial hypotension <strong>in</strong>clude nausea and<br />
vomit<strong>in</strong>g, nuchal rigidity, disturbances of vision, vertigo, t<strong>in</strong>nitus, and reduced hear<strong>in</strong>g<br />
(the latter three are thought due to a decreased <strong>in</strong>tralabyr<strong>in</strong>th<strong>in</strong>e pressure gradient<br />
across the cochlear aqueduct). Complications of LPS are listed <strong>in</strong> Table 7–12.<br />
Optic nerve sheath fenestration has been proven to prevent deterioration <strong>in</strong> vision<br />
and, <strong>in</strong> some cases, improve visual function <strong>in</strong> patients with PTC (Acheson, 1994;<br />
<strong>An</strong>derson, 1992; Bourman, 1988; Brazier, 1996; Carter, 1995; Corbett, 1988b; Goh, 1997;<br />
Horton, 1992; Kelman, 1991, 1992; Lee, 1998; Liu, 1994; Mauriello, 1995; Mittra, 1993;<br />
Pearson, 1991; Sergott, 1988; Spoor, 1991, 1993, 1995; Talks, 1998). For example, <strong>in</strong> one<br />
study 26 patients underwent 40 ONSFs for relief of visual loss or to preserve vision (16<br />
unilateral and 12 bilateral operations) (Knight, 1986). Papilledema disappeared or was<br />
Table 7–12. Complications of Lumboperitoneal Shunts (LPSs)<br />
Papilledema 149<br />
Shunt obstruction (Burgett, 1997; Chumas, 1993b; Eggenberger, 1996; Rosenberg, 1993)<br />
Intracranial hypotension (Burgett, 1997; Chumas, 1993b; Eggenberger, 1996; Johnston, 1988a,b;<br />
Rosenberg, 1993; Sell, 1995)<br />
Cerebrosp<strong>in</strong>al fluid leak<br />
Lumbar radiculopathy (Eggenberger, 1996; Johnston, 1988a,b; Rosenberg, 1993; Sell, 1995)<br />
Shunt or disc space <strong>in</strong>fection (Cabezudo, 1990; Eggenberger, 1996; Johnston, 1988a,b; Rosenberg,<br />
1993)<br />
Abdom<strong>in</strong>al pa<strong>in</strong>, bowel perforation, or migration or dislocation of the peritoneal end of the catheter<br />
(Chumas, 1993b; Eggenberger, 1996; Rosenberg, 1993)<br />
Tonsillar herniation (symptomatic or asymptomatic) and syr<strong>in</strong>gomyelia (Chumas, 1993a,b;<br />
Cognard, 1998)<br />
Subdural hemorrhage<br />
Visual loss from ret<strong>in</strong>al ischemia<br />
Bilateral visual loss and simultagnosia from bilateral parieto-occipital <strong>in</strong>farction related to rupture of<br />
a previously asymptomatic <strong>in</strong>tracranial aneurysm after LPS (Miller, 1997)<br />
Rarely, death (Eisenberg, 1971)
150 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
strik<strong>in</strong>gly reduced <strong>in</strong> 24 of 28 patients. The other four patients had gliotic discs (two<br />
patients) or were followed up for only a short time. Visual acuity improved <strong>in</strong> 12 of 40<br />
eyes and rema<strong>in</strong>ed the same <strong>in</strong> 22 of 40 eyes. In six eyes, the visual acuity decreased.<br />
The visual fields improved <strong>in</strong> 21 of 40 eyes and rema<strong>in</strong>ed the same <strong>in</strong> 10 eyes; five of the<br />
10 eyes that did not change had poor vision before surgery. Eight eyes <strong>in</strong> five patients<br />
cont<strong>in</strong>ued to lose acuity postoperatively. <strong>An</strong> additional two eyes developed visual field<br />
loss with preserved visual acuity. In another study, 23 patients with chronic papilledema<br />
had ONSF and 21 of the 23 patients demonstrated improvement <strong>in</strong> visual function<br />
(Sergott, 1988). Twelve of 21 patients with bilateral visual loss had improved visual<br />
function bilaterally after unilateral surgery, and 6 of 21 patients needed bilateral surgery.<br />
ONSF improved vision <strong>in</strong> six patients who failed to recover vision after LPS.<br />
Kelman et al performed ONSF on 17 patients with severe visual acuity or field loss<br />
(Kelman, 1992). Postoperatively, visual acuity improved or stabilized <strong>in</strong> 33 of 34 eyes<br />
(97%) and the visual fields improved <strong>in</strong> 20 of 21 eyes that underwent surgery. Kelman<br />
et al also performed ONSF <strong>in</strong> 12 patients (16 eyes) with function<strong>in</strong>g LPS and progressive<br />
visual loss (Kelman, 1991), and all patients demonstrated improvement <strong>in</strong> visual<br />
function. Liu et al reported a woman with PTC treated with an LPS who developed<br />
acute pallid disc swell<strong>in</strong>g with peripapillary hemorrhages and visual acuity of no light<br />
perception (NLP) OD and 20=70 OS <strong>in</strong> association with LPS failure (Liu, 1996). The<br />
patient underwent ONSF and LPS revision and her visual acuity improved to 20=20 OU<br />
and the papilledema resolved. Pearson et al operated upon n<strong>in</strong>e patients (14 eyes), and<br />
visual function showed significant improvement or stabilized <strong>in</strong> all but one patient<br />
(Pearson, 1991). Spoor et al performed ONSF <strong>in</strong> 53 patients (101 eyes) with PTC and<br />
visual loss (Spoor, 1991). Sixty-n<strong>in</strong>e eyes (85 patients) with acute papilledema uniformly<br />
had improved visual function after ONSF. Of the 32 eyes with chronic papilledema<br />
(18 patients), only 10 improved. Thirteen eyes required secondary or tertiary ONSF after<br />
an <strong>in</strong>itial successful result. Eleven of 13 eyes had improved visual function after repeat<br />
ONSF. Goh et al described 29 eyes of patients with PTC who underwent ONSF for<br />
visual loss <strong>in</strong> spite of Diamox treatment (Goh, 1997). Visual acuity and visual fields were<br />
compared before and after operation (with<strong>in</strong> 1 and 6 months). The mean follow-up of<br />
this study was 15.7 months (range 1 to 50 months). Visual acuity improved <strong>in</strong> four eyes<br />
(14%), was unchanged <strong>in</strong> 22 eyes (76%), and worsened <strong>in</strong> three eyes (10%). Visual fields<br />
improved <strong>in</strong> 10 (48%) eyes, rema<strong>in</strong>ed unchanged <strong>in</strong> 8 (38%) eyes, and worsened <strong>in</strong> 3<br />
(14%) eyes (6 lost to follow-up). There were four repeat surgeries <strong>in</strong> which vision was<br />
lost <strong>in</strong> one eye.<br />
Banta et al reported 158 ONSFs <strong>in</strong> 86 patients with PTC with visual loss despite<br />
medical treatment (Banta, 2000). Visual acuity stabilized or improved <strong>in</strong> 148 of 158<br />
(94%) eyes and visual fields stabilized or improved <strong>in</strong> 71 of 81 (88%) eyes. Surgical<br />
complications, most often benign and transient, occurred <strong>in</strong> 39 of 86 patients. Diplopia<br />
occurred <strong>in</strong> 30 patients, with 87% resolv<strong>in</strong>g spontaneously (2 patients required<br />
prismatic correction, and 2 other patients underwent subsequent strabismus surgery).<br />
Only one eye <strong>in</strong> one patient had permanent severe visual loss (count f<strong>in</strong>gers acuity)<br />
secondary to an operative complication (presumed traumatic optic neuropathy). One<br />
patient had total ophthalmoplegia and bl<strong>in</strong>dness after surgery (orbital apex compression<br />
syndrome) that completely resolved over 1 month with steroid therapy. Visual loss<br />
occurred <strong>in</strong> 16 of 158 (10%) eyes after <strong>in</strong>itially successful primary ONSF with time from<br />
surgery to failure variable (up to 5 years postsurgery). No specific risk factors that<br />
predisposed patients to ONSF failure were discovered. N<strong>in</strong>e eyes <strong>in</strong> six patients
Papilledema 151<br />
underwent repeat ONSF for progressive visual loss after an <strong>in</strong>itially successful ONSF.<br />
The only complication encountered on repeat ONSF was transient diplopia <strong>in</strong> two<br />
patients. Two patients who underwent repeat ONSF required a CSF diversion procedure<br />
to halt progressive visual loss, and two patients with stable visual function after<br />
repeat ONSF required CSF diversion procedures for <strong>in</strong>tractable headaches. Three<br />
patients with progressive visual loss after <strong>in</strong>itially successful primary ONSF underwent<br />
CSF diversion procedures <strong>in</strong>stead of repeat ONSF. After ONSF, many patients <strong>in</strong>itially<br />
had symptomatic improvement of headaches, but only 8 of 61 (13%) patients with<br />
headache as a present<strong>in</strong>g symptom had subjective improvement. N<strong>in</strong>e patients underwent<br />
CSF diversion procedures for <strong>in</strong>tractable headaches after ONSF despite stable<br />
visual parameters. The authors noted that the patient population with a significant<br />
headache component would likely benefit from an <strong>in</strong>itial CSF diversion procedure. The<br />
authors concluded that ONSF is a safe and effective means of stabiliz<strong>in</strong>g visual acuity<br />
and visual fields <strong>in</strong> patients with PTC with progressive visual loss despite maximum<br />
medical therapy.<br />
Mittra et al exam<strong>in</strong>ed changes <strong>in</strong> color Doppler imag<strong>in</strong>g before and after ONSF for<br />
PTC (Mittra, 1993). Their results suggest that some of the visual loss from chronic<br />
papilledema may be due to ischemia, and worsen<strong>in</strong>g visual acuity correlates with<br />
greater impairment of the retrobulbar circulation. One of the mechanisms by which<br />
ONSF improves visual function may thus be reversal of this ischemic process.<br />
Talks et al reported 24 patients with PTC who required ONSF (Talks, 1998). Twentyone<br />
of the 48 eyes (44%) had macular changes, <strong>in</strong>clud<strong>in</strong>g choroidal folds (n<strong>in</strong>e patients),<br />
circumferential (Paton’s) l<strong>in</strong>es (four), nerve fiber layer hemorrhages (three), macular<br />
stars (five), macular edema (six), ret<strong>in</strong>al pigment epithelial changes (four), and subret<strong>in</strong>al<br />
hemorrhage lead<strong>in</strong>g to a macular scar (one). Significant visual loss attributable to<br />
the macular changes was found <strong>in</strong> five eyes <strong>in</strong> the short term and three eyes <strong>in</strong> the long<br />
term. The two eyes that improved had macular stars; of the three eyes that did not<br />
improve, two had ret<strong>in</strong>al pigment epithelial changes and one had subret<strong>in</strong>al hemorrhage<br />
lead<strong>in</strong>g to a macular scar. The authors concluded that the majority of macular<br />
changes <strong>in</strong> PTC patients resolve and do not add to visual loss from optic nerve damage.<br />
Patients with marked macular edema, however, are at the most risk for permanent<br />
visual loss and should be considered for early surgical treatment.<br />
ONSF has also been effective <strong>in</strong> children with PTC (Lee, 1998). Of 12 patients with<br />
PTC (less than 16 years of age) reviewed, 67% had improved visual acuity, 33% had<br />
improved visual fields, and 17% had worsen<strong>in</strong>g of visual acuity and visual fields<br />
postoperatively (Lee, 1998).<br />
Headaches may be relieved <strong>in</strong> over half of the patients with PTC undergo<strong>in</strong>g ONSF<br />
(Corbett, 1989). For example, with unilateral decompression, headaches were improved<br />
or were relieved <strong>in</strong> 13 of 17 patients <strong>in</strong> one series (Sergott, 1988) and <strong>in</strong> 10 of 16 patients<br />
<strong>in</strong> another study (Corbett, 1989), whereas 91% of patients (10=11) had relief of headache<br />
after ONSF <strong>in</strong> a third study (Kosmorsky, 1993). ONSF may also relieve papilledema and<br />
improve vision when performed on patients with PTC secondary to occlusion of the<br />
dural s<strong>in</strong>uses (Horton, 1992; Mittra, 1993).<br />
Thus, some reports have suggested that ONSF is more effective and associated with<br />
fewer complications than LPS (Corbett, 1988b; Sergott, 1988). Because of these reports,<br />
many physicians have abandoned LPS <strong>in</strong> favor of ONSF for the majority of their<br />
patients with PTC who require surgery (Eggenberger, 1996). Long-term follow-up data<br />
suggest, however, that ONSF may not be as effective as orig<strong>in</strong>ally claimed. Up to 33% of
152 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
patients undergo<strong>in</strong>g ONSF for PTC who show <strong>in</strong>itial improvement <strong>in</strong> visual function<br />
later show deterioration <strong>in</strong> visual field and acuity (Spoor, 1993, 1995). In a study of the<br />
long-term effectiveness of ONSF for PTC, Spoor and McHenry reviewed 32 series of<br />
postoperative visual fields <strong>in</strong> patients who were undergo<strong>in</strong>g ONSF for PTC who had<br />
stable visual acuity and four or more fields dur<strong>in</strong>g 6 to 60 months of follow-up (Spoor,<br />
1993). The authors then extended the review to <strong>in</strong>clude all patients (54 patients, 75 eyes)<br />
who underwent ONSF for PTC, who were followed up with serial automated perimetry.<br />
Fifty-two eyes (68%) showed improvement (36%) or stabilization (32%) of visual<br />
function, whereas 24 eyes (32%) experienced deterioration of visual function after an<br />
<strong>in</strong>itially successful ONSF. The probability of failure from 3 to 5 years was 0.35 by lifetable<br />
analysis. The authors concluded that ONSF effectively stabilizes or improves<br />
visual function <strong>in</strong> the majority of patients with PTC and visual loss. It may fail at any<br />
time after surgery, however, and patients need rout<strong>in</strong>e follow-up with automated<br />
perimetry to detect deterioration of visual function. Some of these late failures may<br />
be prevented by better and different operative techniques (Sergott, 1988; Spoor, 1995).<br />
Also, Acheson et al reported 14 patients (11 with idiopathic PTC and three with dural<br />
venous s<strong>in</strong>us occlusion) who underwent eight unilateral and six bilateral ONSF<br />
(Acheson, 1994). Visual acuity and fields either improved or stabilized <strong>in</strong> 17 out of 20<br />
eyes, and three deteriorated. Of the eight patients undergo<strong>in</strong>g unilateral surgery, the<br />
other eye rema<strong>in</strong>ed stable <strong>in</strong> seven and deteriorated <strong>in</strong> one. Four patients required<br />
ONSF despite previous shunt<strong>in</strong>g or subtemporal decompression. Five patients required<br />
shunt<strong>in</strong>g or subtemporal decompression after ONSF because of persistent headache <strong>in</strong><br />
three cases and for uncontrolled visual failure <strong>in</strong> two cases. No patient lost vision as a<br />
direct complication of surgery.<br />
Thus, vision can be saved after shunt failure, and <strong>in</strong> other cases may be ma<strong>in</strong>ta<strong>in</strong>ed<br />
without the need for a shunt. Shunt<strong>in</strong>g may still be required, however, after ONSF,<br />
especially for persistent headache. Mauriello et al reviewed the records of 108 patients<br />
with pseudotumor who underwent ONSF and who showed visual loss with<strong>in</strong> 1 month<br />
of surgery (Mauriello, 1995). Five patients, <strong>in</strong>clud<strong>in</strong>g two with renal failure and<br />
hypertension, had visual loss with<strong>in</strong> 1 month of ONSF. The first had an abrupt decrease<br />
<strong>in</strong> vision 6 days after ONSF, and <strong>in</strong> this patient a vessel on the nerve sheath bled <strong>in</strong>to the<br />
surgical site. After high-dose <strong>in</strong>travenous (IV) steroids failed to improve vision,<br />
emergency LPS resulted <strong>in</strong> full visual recovery. <strong>An</strong> apparent <strong>in</strong>fectious optic neuropathy<br />
developed <strong>in</strong> the second patient 3 days after surgery. After 72 hours of IV antibiotics,<br />
visual acuity improved from 20=600 to 20=15. The other three patients had gradual<br />
visual loss after ONSF, which stabilized after LPSs. These authors reviewed ONSF<br />
failures <strong>in</strong> the literature and showed that four of seven patients with abrupt visual loss<br />
with<strong>in</strong> the first 2 weeks of ONSF had no improvement <strong>in</strong> vision despite various<br />
treatments, <strong>in</strong>clud<strong>in</strong>g shunts. The series of Corbett et al of 40 ONSF <strong>in</strong> 28 patients<br />
<strong>in</strong>cluded six patients who lost vision with<strong>in</strong> the first 2 weeks of surgery (Corbett,<br />
1988b). Only one of these six patients had return of vision, and this patient had a<br />
dramatic decrease of vision from 20=30 <strong>in</strong> the <strong>in</strong>volved eye to NLP 3 hours postoperatively<br />
after retrobulbar hemorrhage, with acuity improv<strong>in</strong>g to 20=20 after surgical<br />
dra<strong>in</strong>age of the retrobulbar hematoma. The other five patients had no visual recovery<br />
despite LPS, cont<strong>in</strong>uous lumbar dra<strong>in</strong>age, and repeat ONSF <strong>in</strong> one patient who had<br />
<strong>in</strong>trasheath hemorrhage due to cough<strong>in</strong>g (this patient went from 20=30 to 20=200 10 days<br />
postoperatively). Intravenous steroids appeared to enhance visual recovery <strong>in</strong> one<br />
patient of Flynn et al who went from 20=400 to NLP 5 hours postoperatively but
who improved to 20=800 after <strong>in</strong>travenous dexamethasone (Flynn, 1994). Mauriello et al<br />
concluded that avoidance of bleed<strong>in</strong>g dur<strong>in</strong>g ONSF may prevent fibrous occlusion of<br />
the surgical site, and that patients with no identifiable cause for visual loss after ONSF<br />
who do not respond to IV steroids should be evaluated for emergency LPS (Mauriello,<br />
1995). Also, postoperative <strong>in</strong>fectious optic neuropathy should be considered <strong>in</strong> the<br />
differential of abrupt visual loss after ONSF. If ONSF fails, the authors favor LPS rather<br />
than repeat ONSF.<br />
Numerous complications have also been reported after ONSF (Brodsky, 1997; Corbett,<br />
1988b; Flynn, 1994; Plotnik, 1993; Smith, 1992). Plotnik and Kosmorsky reported<br />
postoperative complications <strong>in</strong> 15 of the 38 eyes (39%) undergo<strong>in</strong>g ONSF (Plotnik,<br />
1993). Temporary motility disorders (due to extraocular muscle damage or damage to<br />
their nerve or blood supply) occurred <strong>in</strong> 29% and all resolved, the longest by 9 weeks.<br />
Pupillary dysfunction occurred <strong>in</strong> four eyes (11%) and consisted of sectorial tonic pupils<br />
(due to damage to short ciliary nerves or their blood supply caus<strong>in</strong>g iris sph<strong>in</strong>cter palsy)<br />
that lasted 2 to 8 weeks <strong>in</strong> three eyes but persisted <strong>in</strong> one eye for 12 weeks. Four eyes<br />
(11%) had postoperative vascular complications, <strong>in</strong>clud<strong>in</strong>g two with central ret<strong>in</strong>al<br />
artery occlusions, one supertemporal branch ret<strong>in</strong>al artery occlusion, and one episode of<br />
transient outer ret<strong>in</strong>al ischemia. Both eyes with the central ret<strong>in</strong>al artery occlusions had<br />
poor visual outcomes, and eyes that had undergone prior ONSF were significantly more<br />
likely to have vascular complications than those without a previous operation. The<br />
<strong>in</strong>cidence of vascular complications was 67% <strong>in</strong> eyes that had undergone prior ONSF<br />
and 6% <strong>in</strong> those that had never undergone a previous ONSF. The complications<br />
reported with ONSF are listed <strong>in</strong> Table 7–13.<br />
Table 7–13. Complications of Optic Nerve Sheath Fenestration (ONSF)<br />
Papilledema 153<br />
Ocular motility disorders (e.g., temporary horizontal motility disorder caused by dis<strong>in</strong>sertion of the<br />
medial rectus muscle or comb<strong>in</strong>ed third and sixth nerve palsies)<br />
Transient or permanent diffuse or sectorial tonic pupils<br />
Conjunctival blebs with dellen formation<br />
Chemosis<br />
Chorioret<strong>in</strong>al scar from excessive traction on the globe<br />
Peripapillary hemorrhages thought secondary to short ciliary vessel <strong>in</strong>jury<br />
Orbital hemorrhage<br />
Trauma to the optic nerve<br />
Myel<strong>in</strong>ated nerve fibers (noted 5 months and 6 years postoperatively, thought to be stimulated by<br />
trauma associated with surgery)<br />
Microhyphemas<br />
Orbital apex syndrome (? steroid responsive)<br />
Subconjunctival Tenon’s cysts<br />
Streptococcal corneal ulcers<br />
Dacryocystitis<br />
Intraoperative angle closure glaucoma<br />
Deterioration of visual function, transient bl<strong>in</strong>dness, choroidal <strong>in</strong>farction (fundus changes with<br />
choroidal <strong>in</strong>farction may not be evident for several weeks after operation)<br />
Central or branch ret<strong>in</strong>al artery occlusion<br />
Death<br />
Source: Banta, 2000; Bourman, 1988; Brodsky, 1997; Corbett, 1988b; Flynn, 1994; Plotnik, 1993; Smith, 1992.
154 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
From the above summary, it is evident that both ONSF and LPS may improve vision<br />
and prevent deterioration of vision <strong>in</strong> patients with PTC (class II–III, level B). Both<br />
procedures have their advantages and disadvantages, and either may fail with time no<br />
matter what procedure is used. Approximately one third of patients undergo<strong>in</strong>g ONSF<br />
will not experience headache relief and only about 75% of ONSFs appear to be<br />
function<strong>in</strong>g 6 months after surgery. The probability of function<strong>in</strong>g of ONSF steadily<br />
decreases thereafter such that 66% are function<strong>in</strong>g at 12 months, 55% at 3 years, 38% at<br />
Figure 7–1. Evaluation of bilateral optic disc swell<strong>in</strong>g.
5 years, and 16% at 6 years after surgery (Spoor, 1993). Thus, these patients must have<br />
their visual function followed for years as deterioration may require repeat procedures<br />
for ONSF failures. Although patients may be treated with a second ONSF after <strong>in</strong>itial<br />
failure, eyes that have more than one ONSF are less likely to improve after surgery and<br />
more likely to experience significant vascular complication than are eyes that undergo a<br />
s<strong>in</strong>gle surgery (Plotnik, 1993). On the other hand, LPS is fraught with many complications,<br />
although headache due to PTC is probably better controlled by LPS. Also, LPS<br />
failure is common, although most shunt failures occur with<strong>in</strong> 2 to 3 months of the <strong>in</strong>itial<br />
LPS (cumulative risk, 37%) and only rarely is the first shunt revision required more than<br />
1 year after <strong>in</strong>itial LPS (Eggenberger, 1996). Thus, a patient with PTC who undergoes an<br />
LPS and who ma<strong>in</strong>ta<strong>in</strong>s a function<strong>in</strong>g shunt for more than 1 year has a lower risk of<br />
requir<strong>in</strong>g a shunt revision over subsequent years (Eggenberger, 1996). Patients undergo<strong>in</strong>g<br />
LPS, however, also need careful follow-up after their procedure because of the<br />
possibility of late failures. LPS failure has been reportedly successfully treated by repeat<br />
LPS or by ONSF.<br />
The evaluation of the patient with optic disc swell<strong>in</strong>g is summarized <strong>in</strong> Figure 7–1.<br />
References<br />
Papilledema 155<br />
Acheson JF, Green WT, Sanders MD. (1994). Optic nerve sheath decompression for the treatment of visual failure<br />
<strong>in</strong> chronic raised <strong>in</strong>tracranial pressure. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:1426–1429.<br />
Adams C, Dean HJ, Israels SJ, et al. (1994). Primary hypothyroidism with <strong>in</strong>tracranial hypertension and pituitary<br />
hyperplasia. Pediatr <strong>Neuro</strong>l 10:166–168.<br />
Adelman JU. (1998). Headache and papilledema secondary to dural arteriovenous malformation. Headache<br />
38:621–623.<br />
Ageli SI, Sato Y, Gantz BJ. (1994). Glomus jugulare tumors masquerad<strong>in</strong>g as benign <strong>in</strong>tracranial hypertension.<br />
Arch Otolaryngol 120:1277–1280.<br />
Ahmad S. (1996). Amiodarone and reversible benign <strong>in</strong>tracranial hypertension. Cardiology 87:90.<br />
Akova YA, Kansu T, Duman S. (1993). Pseudotumor cerebri secondary to dural s<strong>in</strong>us thrombosis <strong>in</strong> neurosarcoidosis.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:188–189.<br />
Akova YA, Kansu T, Yazar Z, et al. (1994). Macular subret<strong>in</strong>al neovascular membrane associated with<br />
pseudotumor cerebri. J <strong>Neuro</strong>-ophthalmol 14:193–195.<br />
Alemayehu W. (1995). Pseudotumor cerebri (toxic effect of the ‘‘magic bullet’’). Ethiop Med J 33:265–270.<br />
Alexandrakis G, Filatov V, Walsh T. (1993). Pseudotumor cerebri <strong>in</strong> a 12-year-old boy with Addison’s disease.<br />
Am J Ophthalmol 116:650–651.<br />
Alison L, Hobbs CJ, Hanks HG, Butler G. (1997). Non-organic failure to thrive complicated by benign <strong>in</strong>tracranial<br />
hypertension dur<strong>in</strong>g catch-up growth. Acta Paediatr 86:1141–1143.<br />
Alpan G, Glick B, Peleg O, Eyal F. (1991). Pseudotumor cerebri and coma <strong>in</strong> vitam<strong>in</strong> D–dependent rickets.<br />
Cl<strong>in</strong> Pediatr 30:254–256.<br />
Ames D, Wirsh<strong>in</strong>g WC, Cokely HT, Lo LL. (1994). The natural course of pseudotumor cerebri <strong>in</strong> lithium-treated<br />
patients. J Cl<strong>in</strong> Psychpharmacol 14:286–287.<br />
<strong>An</strong>derson RL, Flaharty PM. (1992). Treatment of pseudotumor cerebri by primary and secondary optic nerve<br />
sheath decompression. Am J Ophthalmol 113:599–601.<br />
<strong>An</strong>giari P, Corrad<strong>in</strong>i L, Corsi M, Merli GA. (1992). Pseudotumor cerebri. Lumboperitoneal shunt <strong>in</strong> long last<strong>in</strong>g<br />
cases. J <strong>Neuro</strong>surg Sci 36:145–149.<br />
Arber N, Shir<strong>in</strong> H, Fadial R, et al. (1990). Pseudotumor cerebri associated with leuprorel<strong>in</strong> acetate. Lancet 335:668.<br />
Arseni C, Simoca I, Jipescu I, et al. (1992). Pseudotumor cerebri: risk factors, cl<strong>in</strong>ical course, prognostic criteria.<br />
Romanian J <strong>Neuro</strong>l Psychiatry 30:115–132.<br />
Au Eong KG, Hariharan S, Chau EC, et al. (1997). Idiopathic <strong>in</strong>tracranial hypertension, empty sella turcica and<br />
polycystic ovary syndrome—a case report. S<strong>in</strong>gapore Med J 38:129–130.<br />
Avery R, Jabs D, W<strong>in</strong>gard J, et al. (1991). Optic disk edema after bone marrow transplantation. Possible role of<br />
cyclospor<strong>in</strong>e toxicity. Ophthalmology 98:1294–1301.
156 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Bafna S, Lee AG. (1996). Bilateral optic disc edema and multifocal ret<strong>in</strong>al lesions without loss of vision <strong>in</strong> cat<br />
scratch disease. Arch Ophthalmol 114:1016–1017.<br />
Bakshi SK, Oak JL, Chawla KP, et al. (1992). Facial nerve <strong>in</strong>volvement <strong>in</strong> pseudotumor cerebri. J Postgrad Med<br />
38:144–145.<br />
Balcar LJ, Liu GT, Forman S, et al. (1999). Idiopathic <strong>in</strong>tracranial hypertension: relation of age and obesity <strong>in</strong><br />
children. <strong>Neuro</strong>logy 52:870–872.<br />
Bandyopadhyay S. (2001). Pseudotumor cerebri. Arch <strong>Neuro</strong>l 58:1699–1701.<br />
Banta JT, Farris BK. (2000). Pseudotumor cerebri and optic nerve sheath decompression. Ophthalmology<br />
107:1907–1912.<br />
Bikangaga P, Canny GJ. (1996). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> <strong>in</strong>fants with cystic fibrosis. Arch Pediatr Adolesc<br />
Med 150:551–552.<br />
Biousse V, Ameri A, Bousser M-G. (1999). Isolated <strong>in</strong>tracranial hypertension as the only sign of cerebral venous<br />
thrombosis. <strong>Neuro</strong>logy 53:1537–1542.<br />
Biousse V, Newman NJ, Lessell S. (1998). Audible pulsatile t<strong>in</strong>nitus <strong>in</strong> idiopathic <strong>in</strong>tracranial hypertension.<br />
<strong>Neuro</strong>logy 50:1185–1186.<br />
Blethen SL. (1995). Complications of growth hormone therapy <strong>in</strong> children. Curr Op<strong>in</strong> Pediatr 7:466–471.<br />
Boll<strong>in</strong>g JP, Brazis PW. (1990). Optic disk swell<strong>in</strong>g with peripheral neuropathy, organomegaly, endocr<strong>in</strong>opathy,<br />
monoclonal gammopathy, and sk<strong>in</strong> changes (POEMS syndrome). Am J Ophthalmol 109:503–510.<br />
Borruat FX, Regli F. (1993). Pseudotumor cerebri as a complication of amiodarone therapy. Am J Ophthalmol<br />
116:776–777.<br />
Bosch JA, Valdes M, Solans R, et al. (1995). Sk<strong>in</strong> hyper-reactivity <strong>in</strong> a patient with benign <strong>in</strong>tracranial hypertension<br />
as an early manifestation of Behçet’s disease. Br J Rheumatol 34:184.<br />
Bourman ND, Spoor TC, Ramocki JM. (1988). Optic nerve sheath decompression for pseudotumor cerebri. Arch<br />
Ophthalmol 106:1378–1383.<br />
Brazier DJ, Sanders MD. (1996). Disappearance of optociliary shunt vessels after optic nerve sheath decompression.<br />
Br J Ophthalmol 80:186–187.<br />
Brodsky MC, Rettele GA. (1997). Protracted postsurgical bl<strong>in</strong>dness with visual recovery follow<strong>in</strong>g optic nerve<br />
sheath fenestration. Arch Ophthalmol 115:1473–1474.<br />
Brodsky MC, Vaphiades M. (1998). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> pseudotumor cerebri. Ophthalmology<br />
105:1686–1693.<br />
Burgett RA, Purv<strong>in</strong> VA, Kawasaki A. (1997). Lumboperitoneal shunt<strong>in</strong>g for pseudotumor cerebri. <strong>Neuro</strong>logy<br />
49:734–739.<br />
Cabezudo JM, Olabe J, Bacci F. (1990). Infection of the <strong>in</strong>tervertebral disc space after placement of a percutaneous<br />
lumboperitoneal shunt for benign <strong>in</strong>tracranial hypertension. <strong>Neuro</strong>surgery 26:1008–1009.<br />
Campos SP, Olitsky S. (1995). Idiopathic <strong>in</strong>tracranial hypertension after L-thyrox<strong>in</strong>e therapy for acquired primary<br />
hypothyroidism. Cl<strong>in</strong> Pediatr 34:334–337.<br />
Camras LR, Ecanow JS, Abood CA. (1998). Spontaneous cerebrosp<strong>in</strong>al fluid rh<strong>in</strong>orrhea <strong>in</strong> a patient with<br />
pseudotumor cerebri. J <strong>Neuro</strong>imag<strong>in</strong>g 8:41–42.<br />
Capobianco DJ, Brazis PW, Chesire WP. (1997). Idiopathic <strong>in</strong>tracranial hypertension and seventh nerve palsy.<br />
Headache 37:286–288.<br />
Carter SR, Seiff SR. (1995). Macular changes <strong>in</strong> pseudotumor cerebri before and after optic nerve sheath<br />
fenestration. Ophthalmology 102:937–941.<br />
Çelebisoy N, Seçil Y, Yüceyar N, Ertek<strong>in</strong> C. (1999). Occult cerebral vascular causes of pseudotumor cerebri.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 21:157–163.<br />
Chang D, Nagamoto G, Smith WE. (1992). Benign <strong>in</strong>tracranial hypertension and chronic renal failure. Cleve Cl<strong>in</strong> J<br />
Med 59:419–422.<br />
Chari C, Rao NS. (1991). Benign <strong>in</strong>tracranial hypertension—its unusual manifestations. Headache 31:599–600.<br />
Chaves-Carballo E, Dabbagh O, Bahabri S. (1999). Pseudotumor cerebri and leukoencephalopathy <strong>in</strong> childhood<br />
lupus. Lupus 8:81–84.<br />
Chen HY, Tsai RK, Di Capua HM, Rosati P, et al. (1993). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> an older child with<br />
cystic fibrosis. Pediatr <strong>Neuro</strong>l 9:494–495.<br />
Chen HY, Tsai RK, Huang SM. (1998). ATRA-<strong>in</strong>duced pseudotumor cerebri—one case report. Kaohsiung J Med Sci<br />
14:58–60.<br />
Chern S, Margargal LE, Brav SS. (1991). Bilateral central ret<strong>in</strong>al ve<strong>in</strong> occlusion as an <strong>in</strong>itial manifestation of<br />
pseudotumor cerebri. <strong>An</strong>n Ophthalmol 23:54–57.<br />
Chevalier X, de Bandt M, Bourgeois P, Kahn MF. (1992). Primary Sjögren’s syndrome preced<strong>in</strong>g the presentation<br />
of systemic lupus erythematosus as a benign <strong>in</strong>tracranial hypertension syndrome. <strong>An</strong>n Rheum Dis<br />
51:808–809.
Papilledema 157<br />
Chimowitz MI, Little JR, Awad IA, et al. (1990). Intracranial hypertension associated with unruptured cerebral<br />
arteriovenous malformations. <strong>An</strong>n <strong>Neuro</strong>l 27:474–479.<br />
Chiu AM, Chuenkongkaew L, Cornblath WT, et al. (1998). M<strong>in</strong>ocycl<strong>in</strong>e treatment and pseudotumor cerebri<br />
syndrome. Am J Ophthalmol 126:116–121.<br />
Chumas PD, Armstrong DC, Drake JM, et al. (1993a). Tonsillar herniation: the rule rather than the exception after<br />
lumboperitoneal shunt<strong>in</strong>g <strong>in</strong> the pediatric population. J <strong>Neuro</strong>surg 78:568–573.<br />
Chumas PD, Kulkarni AV, Drake JM, et al. (1993b). Lumboperitoneal shunt<strong>in</strong>g: a retrospective study <strong>in</strong> the<br />
pediatric population. <strong>Neuro</strong>surgery 32:376–383.<br />
C<strong>in</strong>cirip<strong>in</strong>i GS, Donahue S, Borchert MS. (1999). Idiopathic <strong>in</strong>tracranial hypertension <strong>in</strong> prepubertal pediatric<br />
patients: characteristics, treatment, and outcome. Am J Ophthalmol 127:178–182.<br />
Clark D, Bullock T, Hui T, Firth J. (1994). Benign <strong>in</strong>tracranial hypertension: a cause of CSF rh<strong>in</strong>orrhea. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 57:847–849.<br />
Cockerell OC, Lai HM, Ross-Russell RW. (1993). Pseudotumor cerebri associated with arteriovenous malformations.<br />
Postgrad Med J 69:637–640.<br />
Cognard C, Casasco A, Toevi M, et al. (1998). Dural arteriovenous fistulas as a cause of <strong>in</strong>tracranial hypertension<br />
due to impairment of cranial venous outflow. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 65:308–316.<br />
Condulis N, Germa<strong>in</strong> G, Charest N, et al. (1997). Pseudotumor cerebri: a present<strong>in</strong>g manifestation of Addison’s<br />
disease. Cl<strong>in</strong> Pediatr 36:711–713.<br />
Corbett JJ, Digre K. (2002). Idiopathic <strong>in</strong>tracranial hypertension. <strong>An</strong> answer to, ‘‘the chicken or the egg?’’ <strong>Neuro</strong>logy<br />
58:5–6.<br />
Corbett JJ, Jacobson DM, Maurer RC, et al. (1988a). Enlargement of the bl<strong>in</strong>d spot caused by papilledema.<br />
Am J Ophthalmol 105:261–265.<br />
Corbett JJ, Mehta MP. (1983). Cerebrosp<strong>in</strong>al fluid pressure <strong>in</strong> normal obese subjects and patients with<br />
pseudotumor cerebri. <strong>Neuro</strong>logy 33:1386–1388.<br />
Corbett JJ, Nerad JA, Tse D, et al. (1988b). Result of optic nerve sheath fenestration for pseudotumor cerebri. The<br />
lateral orbitotomy approach. Arch Ophthalmol 106:1391–1397.<br />
Corbett JJ, Thompson HS. (1989). The rational management of idiopathic <strong>in</strong>tracranial hypertension. Arch <strong>Neuro</strong>l<br />
46:1049–1051.<br />
Corbett JJ, Sav<strong>in</strong>o PJ, Thompson HS, et al. (1982). Visual loss <strong>in</strong> pseudotumor cerebri: follow-up of 57 patients<br />
from 5 to 41 years and a profile of 14 patients with permanent severe visual loss. Arch <strong>Neuro</strong>l 39:461–474.<br />
Couban S, Maxner CE. (1991). Cerebral venous s<strong>in</strong>us thrombosis present<strong>in</strong>g as idiopathic <strong>in</strong>tracranial hypertension.<br />
Can Med Assoc J 145:657–659.<br />
Cremer PD, Thompson EO, Johnston IH, Halmagyi GM. (1996). Pseudotumor cerebri and cerebral venous<br />
hypertension. <strong>Neuro</strong>logy 47:1602–1603.<br />
Cruz OA, Fogg SG, Roper-Hall G. (1996). Pseudotumor cerebri associated with cyclospor<strong>in</strong>e use. Am J Ophthalmol<br />
122:436–437.<br />
Cuddihy J. (1994). Case report of benign <strong>in</strong>tracranial hypertension secondary to tetracycl<strong>in</strong>e. Irish Med J 87:90.<br />
Daftari TK, Heller JG, Newman NJ. (1995). Pseudotumor cerebri after occipitocervical arthrodesis and immobilization<br />
<strong>in</strong> a halo vest. A case report. J Bone Jo<strong>in</strong>t Surg 77A:455–458.<br />
Daif A, Awada A, al-Rajeh S, et al. (1995). Cerebral venous thrombosis <strong>in</strong> adults. A study of 40 cases from Saudi<br />
Arabia. Stroke 26:1193–1195.<br />
Davenport RJ, Will RG, Galloway PJ. (1994). Isolated <strong>in</strong>tracranial hypertension present<strong>in</strong>g with trigem<strong>in</strong>al<br />
neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:381.<br />
David CA, Peerless SJ. (1995). Pseudotumor syndrome result<strong>in</strong>g from cerebral arteriovenous malformation: case<br />
report. <strong>Neuro</strong>surgery 36:588–590.<br />
De Paepe L, Abs R, Verlooy J, et al. (1993). Benign <strong>in</strong>tracranial hypertension as a cause of transient partial pituitary<br />
deficiency. J <strong>Neuro</strong>l Sci 114:152–155.<br />
Digre KB, Corbett JJ. (1988). Pseudotumor cerebri <strong>in</strong> men. Arch <strong>Neuro</strong>l 45:866–872.<br />
Dommisse J. (1991). Pseudotumor cerebri associated with lithium therapy <strong>in</strong> two patients. J Cl<strong>in</strong> Psychiatry 52:239.<br />
Donahue SP. (2000). Recurrence of idiopathic <strong>in</strong>tracranial hypertension after weight loss: the carrot craver.<br />
Am J Ophthalmol 130:850–851.<br />
Donnet A, Dufour H, Graziani N, Grisoli F. (1992). M<strong>in</strong>ocycl<strong>in</strong>e and benign <strong>in</strong>tracranial hypertension. Biomed<br />
Pharmacother 46:171–172.<br />
Dudenhoefer EJ, Cornblath WT, Schatz MP. (1998). Scalp necrosis with giant cell arteritis. Ophthalmology 105:1875–<br />
1878.<br />
Eggenberger ER, Miller NR, Vitale S. (1996). Lumboperitoneal shunt for the treatment of pseudotumor cerebri.<br />
<strong>Neuro</strong>logy 46:1524–1530.<br />
Eisenberg HM, Davidson RI, Shillito J. (1971). Lumboperitoneal shunts: review of 34 cases. J <strong>Neuro</strong>surg 35:427–431.
158 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Evers JP, Jacobson RJ, P<strong>in</strong>cus J, Zwiebel JA. (1992). Pseudotumor cerebri follow<strong>in</strong>g high-dose cytos<strong>in</strong>e arab<strong>in</strong>oside.<br />
Br J Haematol 80:559–560.<br />
Fanoous M, Hamed LM, Margo CE. (1991). Pseudotumor cerebri associated with danazol withdrawal. JAMA<br />
266:1218–1219.<br />
Fant<strong>in</strong> A, Feist RM, Reddy CV. (1993). Intracranial hypertension and papilloedema <strong>in</strong> chronic <strong>in</strong>flammatory<br />
demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy. Br J Ophthalmol 77:193.<br />
Farah S, Al-Shubaili A, Montaser A, et al. (1998). Behcet’s syndrome: a report of 41 patients with emphasis on<br />
neurological manifestations. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 64:382–384.<br />
Fedorowski JJ, Hyman C. (1996). Optic disk edema as the present<strong>in</strong>g sign of Lyme disease. Cl<strong>in</strong> Infect Dis 23:639–<br />
640.<br />
Feigert JM, Sweet DL, Coleman M, et al. (1990). Multicentric angiofollicular lymph node hyperplasia with<br />
peripheral neuropathy, pseudotumor cerebri, IgA dysprote<strong>in</strong>emia, and thrombocytosis <strong>in</strong> women. A dist<strong>in</strong>ct<br />
syndrome. <strong>An</strong>n Intern Med 113:362–367.<br />
Felton WL, Sismanis A. (1995). Idiopathic <strong>in</strong>tracranial hypertension without papilledema <strong>in</strong> patients with<br />
pulsatile t<strong>in</strong>nitus. Presented at the North American <strong>Neuro</strong>-Ophthalmology meet<strong>in</strong>g, Tucson, Arizona.<br />
Flynn WJ, Westfall CT, Weisman JS. (1994). Transient bl<strong>in</strong>dness after optic nerve sheath fenestration.<br />
Am J Ophthalmol 117:678–679.<br />
Forderreuther S, Straube A. (2000). Indomethac<strong>in</strong> reduces CSF pressure <strong>in</strong> <strong>in</strong>tracranial hypertension. <strong>Neuro</strong>logy<br />
55:1043–1045.<br />
Fort JA, Smith LD. (1999). Pseudotumor cerebri secondary to <strong>in</strong>termediate-dose cytarab<strong>in</strong>e HCl. <strong>An</strong>n Pharmacother<br />
33:576–578.<br />
Francois I, Casteels I, Silberste<strong>in</strong> J, et al. (1997). Empty sella, growth hormone deficiency and pseudotumor cerebri:<br />
effect of <strong>in</strong>itiation, withdrawal and resumption of growth hormone therapy. Eur J Pediatr 156:69–70.<br />
Friedman DI. (2001). Papilledema and pseudotumor cerebri. Ophthalmol Cl<strong>in</strong> North Am 14:129–147.<br />
Friedman DI, Forman S, Levi L, et al. (1997). Unusual ocular motility disturbances <strong>in</strong> pseudotumor cerebri.<br />
Presented at the 23rd annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Keystone,<br />
Colorado, February 9–13.<br />
Friedman DI, Forman S, Levi L, et al. (1998a). Unusual ocular motility disturbances with <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure. <strong>Neuro</strong>logy 50:1893–1896.<br />
Friedman DI, Streeten DHP. (1998b). Idiopathic <strong>in</strong>tracranial hypertension and orthostatic edema may share a<br />
common pathogenesis. <strong>Neuro</strong>logy 50:1099–1104.<br />
Fujiwara S, Sawamura Y, Kato T, Abe H, Katusima H. (1997). Idiopathic <strong>in</strong>tracranial hypertension <strong>in</strong> female<br />
homozygous tw<strong>in</strong>s. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 62:652–654.<br />
Galm O, Fabry U, Osieka R. (2000). Pseudotumor cerebri after treatment of relapsed acute promyelocytic leukemia<br />
with arsenic trioxide. Leukemia 14:343–344.<br />
Gardner K, Cox T, Digre KB. (1995). Idiopathic <strong>in</strong>tracranial hypertension associated with tetracycl<strong>in</strong>e use <strong>in</strong><br />
fraternal tw<strong>in</strong>s: case report and review. <strong>Neuro</strong>logy 45:6–10.<br />
Getenet JC, Croisile B, Vighetto A, et al. (1993). Idiopathic <strong>in</strong>tracranial hypertension after ofloxac<strong>in</strong> treatment.<br />
Acta <strong>Neuro</strong>l Scand 87:503–504.<br />
Gibby WA, Cohen MS, Goldberg HI, Sergott RC. (1993). Pseudotumor cerebri: CT f<strong>in</strong>d<strong>in</strong>gs and correlation with<br />
vision loss. Am J Radiol 160:143–146.<br />
Gironell A, Marti-Fabregas J, Bello J, Avila A. (1997). Non-Hodgk<strong>in</strong>’s lymphoma as a new cause of non-thrombotic<br />
superior sagittal s<strong>in</strong>us occlusion. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 63:121–122.<br />
Giuseffi V, Wall M, Siegel PZ, Roojas PB. (1991). Symptoms and disease associations <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension (pseudotumor cerebri): a case-control study. <strong>Neuro</strong>logy 41:239–244.<br />
Glaser JS. (1990). <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. Philadelphia, JP Lipp<strong>in</strong>cott, p. 106.<br />
Goh KY, Schatz NJ, Glaser JS. (1997). Optic nerve sheath fenestration for pseudotumor cerebri. J <strong>Neuro</strong><strong>ophthalmology</strong><br />
17:86–91.<br />
Gokalp HZ, Baskaya MK, Ayd<strong>in</strong> V. (1992). Pseudotumor cerebri with familial Mediterranean fever. Cl<strong>in</strong> <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg 94:261–263.<br />
Golnik KC, Devoto TM, Kersten RC, Kulw<strong>in</strong> D. (1999). Visual loss <strong>in</strong> idiopathic <strong>in</strong>tracranial hypertension after<br />
resolution of papilledema. Ophthalmic Plast Reconstr Surg 15:442–444.<br />
Goodw<strong>in</strong> JA. (1990). Treatment of idiopathic <strong>in</strong>tracranial hypertension with digox<strong>in</strong>. <strong>An</strong>n <strong>Neuro</strong>l 28:248.<br />
Gordon K. (1997). Pediatric pseudotumor cerebri: descriptive epidemiology. Can J <strong>Neuro</strong>l Sci 24:2192–21.<br />
Graybill JR, Sobel J, Saag M, et al. (2000). Diagnosis and management of <strong>in</strong>creased <strong>in</strong>tracranial pressure <strong>in</strong> patients<br />
with AIDS and cryptococcal men<strong>in</strong>gitis. The NIAID Mycoses study group and AIDS cooperative treatment<br />
groups. Cl<strong>in</strong> Infect Dis 30:47–54.
Papilledema 159<br />
Green L, V<strong>in</strong>ker S, Amital H, et al. (1995). Pseudotumor cerebri <strong>in</strong> systemic lupus erythematosus. Sem<strong>in</strong> Arthritis<br />
Rheum 25:103–108.<br />
Greenfield DS, Wanichwecharungruang B, Liebman JM, Ritch R. (1997). Pseudotumor cerebri appear<strong>in</strong>g with<br />
unilateral papilledema after trabeculectomy. Arch Ophthalmol 115:423–426.<br />
Gregoric A, Bracic K, Novljan G, Marcun-Varda N. (2000). Pseudotumor cerebri <strong>in</strong> a child with familial<br />
hypomagnesemia-hypercalciuria. Pediatr Nephrol 14:269–270.<br />
Gross FJ, M<strong>in</strong>del JS. (1991). Pseudotumor cerebri and Guilla<strong>in</strong>-Barré syndrome associated with human immunodeficiency<br />
virus <strong>in</strong>fection. <strong>Neuro</strong>logy 41:1845–1846.<br />
Guy J, Johnston PK, Corbett JJ, Day AL, Glaser JS. (1990). Treatment of visual loss <strong>in</strong> pseudotumor cerebri<br />
associated with uremia. <strong>Neuro</strong>logy 40:28–32.<br />
Guymer RH, Cairns JD, O’Day J. (1993). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> chronic myeloid leukemia. Aust N Z J<br />
Ophthalmol 21:181–185.<br />
Haller JS, Meyer DR, Cromie W, Fagles N, Hayes S. (1993). Pseudotumor cerebri follow<strong>in</strong>g beta-human chorionic<br />
gonadotrop<strong>in</strong> hormone treatment for undescended testicles. <strong>Neuro</strong>logy 43:448–449.<br />
Hauser D, Barzilai N, Zalish M, et al. (1996). Bilateral papilledema with ret<strong>in</strong>al hemorrhages <strong>in</strong> association<br />
with cerebral venous s<strong>in</strong>us thrombosis and paroxysmal nocturnal hemoglob<strong>in</strong>uria. Am J Ophthalmol 122:<br />
592–593.<br />
Hedges TR III, Legge RH, Peli E, Yardley CJ. (1995). Ret<strong>in</strong>al nerve fiber layer changes and visual field loss <strong>in</strong><br />
idiopathic <strong>in</strong>tracranial hypertension. Ophthalmology 102:1242–1247.<br />
Higg<strong>in</strong>s JN, Owler BK, Cous<strong>in</strong>s C, Pickard JD. (2002). Venous s<strong>in</strong>us stent<strong>in</strong>g for refractory benign <strong>in</strong>tracranial<br />
hypertension. Lancet 359:228–230.<br />
Hills C, Sohn RS. (1998). Peripheral nerve sheath tumor presents as idiopathic <strong>in</strong>tracranial hypertension. <strong>Neuro</strong>logy<br />
50:308–309.<br />
Horoshovski D, Amital H, Katz M, Shoenfeld Y. (1995). Pseudotumor cerebri <strong>in</strong> SLE. Cl<strong>in</strong> Rheumatol 14:708–710.<br />
Horton JC, Seiff SR, Pitts LH, et al. (1992). Decompression of the optic nerve sheath for vision-threaten<strong>in</strong>g<br />
papilledema caused by dural s<strong>in</strong>us occlusion. <strong>Neuro</strong>surgery 31:203–212.<br />
Huna-Baron R, Landau K, Rosenberg M, et al. (2001). Unilateral swollen disc due to <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure. <strong>Neuro</strong>logy 56:1588–1590.<br />
Hyk<strong>in</strong> PG, Spalton DJ. (1991). Bilateral per<strong>in</strong>euritis of the optic nerves. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:375–376.<br />
Ing EB, Leavitt JA, Younge BR. (1996). Papilledema follow<strong>in</strong>g bowtie optic atrophy. Arch Ophthalmol 114:356–357.<br />
Ireland B, Corbett JJ, Wallace RB. (1990). The search for the cause of idiopathic <strong>in</strong>tracranial hypertension: a<br />
prelim<strong>in</strong>ary case-control study. Arch <strong>Neuro</strong>l 47:315–320.<br />
Jacobson DM. (1995). Intracranial hypertension and the syndrome of acquired hyperopia with choroidal folds.<br />
J <strong>Neuro</strong>-ophthalmol 15:178–185.<br />
Jacobson DM. (2000). Divergence <strong>in</strong>sufficiency revisited. Natural history of idiopathic cases and neurologic<br />
associations. Arch Ophthalmol 118:1237–1241.<br />
Jacobson DM, Berg R, Wall M, et al. (1999). Serum vitam<strong>in</strong> A concentration is elevated <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension. <strong>Neuro</strong>logy 53:1114–1118.<br />
Jacobson DM, Karanjia PN, Olson KA, Warner JJ. (1990). Computed tomography ventricular size has no<br />
predictive value <strong>in</strong> diagnos<strong>in</strong>g pseudotumor cerebri. <strong>Neuro</strong>logy 40:1454–1455.<br />
Jacome DE. (2001). Idiopathic <strong>in</strong>tracranial hypertension and hemophilia A. Headache 41:595–598.<br />
Ja<strong>in</strong> N, Rosner F. (1992). Idiopathic <strong>in</strong>tracranial hypertension: report of seven cases. Am J Med 93:391–395.<br />
Javeed N, Shaikh J, Jayaram S. (1995). Recurrent pseudotumor cerebri <strong>in</strong> an HIV-positive patient. AIDS 9:817–819.<br />
Johnson LN, Krohel GB, Madsen RW, March GA Jr. (1998). The role of weight loss and acetazolamide <strong>in</strong> the<br />
treatment of idiopathic <strong>in</strong>tracranial hypertension (pseudotumor cerebri). Ophthalmology 105:2313–2317.<br />
Johnston I, Besser M, Morgan M. (1988a). Cerebrosp<strong>in</strong>al fluid diversion <strong>in</strong> the treatment of benign <strong>in</strong>tracranial<br />
hypertension. J <strong>Neuro</strong>surg 69:195–202.<br />
Johnston I, Paterson A, Besser M. (1988b). The treatment of benign <strong>in</strong>tracranial hypertension: a review of 134<br />
cases. Surg <strong>Neuro</strong>l 69:195–202.<br />
Kan L, Sood SK, Maytal J. (1998). Pseudotumor cerebri <strong>in</strong> Lyme disease: a case report and literature review. Pediatr<br />
<strong>Neuro</strong>l 18:439–441.<br />
Kansu T, Kansu E, Zileli T, Kirkali P. (1991). <strong>Neuro</strong>-ophthalmologic manifestations of Behçet’s disease.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 11:7–11.<br />
Karahalios D, Rekate HL, Khayata MH, Apostolides, PJ. (1996). Elevated <strong>in</strong>tracranial venous pressure as a<br />
universal mechanism <strong>in</strong> pseudotumor cerebri of vary<strong>in</strong>g etiologies. <strong>Neuro</strong>logy 46:198–202.<br />
Katz B, Moster ML, Slav<strong>in</strong> ML. (1997). Disk edema subsequent to renal transplantation. Surv Ophthalmol<br />
41:315–320.
160 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Katz DM, Trobe JD, Muraszko KM, Dauser RC. (1994). Shunt failure without ventriculomegaly proclaimed by<br />
ophthalmic f<strong>in</strong>d<strong>in</strong>gs. J <strong>Neuro</strong>surg 81:721–725.<br />
Kaufman DI. (1998). Peripheral demyel<strong>in</strong>at<strong>in</strong>g and axonal disorders. In: Miller NR, Newman NJ, eds. Walsh and<br />
Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 5th ed. Baltimore, Williams & Wilk<strong>in</strong>s, pp. 5677–5719.<br />
Kelman SE, Heaps R, Wolf A, Elman MJ. (1992). Optic nerve decompression surgery improves visual function <strong>in</strong><br />
patients with pseudotumor cerebri. <strong>Neuro</strong>surgery 30:391–395.<br />
Kelman SE, Sergott RC, Cioffi GA, et al. (1991). Modified optic nerve sheath decompression <strong>in</strong> patients with<br />
function<strong>in</strong>g lumboperitoneal shunts and progressive visual loss. Ophthalmology 98:1449–1453.<br />
Kesler A, Ellis MH, Reshef T, et al. (2000). Idiopathic <strong>in</strong>tracranial hypertension and anticardiolip<strong>in</strong> antibodies.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 68:379–380.<br />
Kesler A, Gadoth N. (2001a). Epidemiology of idiopathic <strong>in</strong>tracranial hypertension. J <strong>Neuro</strong>-ophthalmol 21:12–14.<br />
Kesler A, Goldhammer Y, Gadoth N. (2001b). Do men with pseudotumor cerebri share the same characteristics as<br />
women? A retrospective review of 141 cases. J <strong>Neuro</strong>-ophthalmol 21:15–17.<br />
Kessler LA, Novelli PM, Reigel DH. (1998). Surgical treatment of benign <strong>in</strong>tracranial hypertension—subtemporal<br />
decompression revisited. Surg <strong>Neuro</strong>l 50:73–76.<br />
Kharode C, McAbee G, Sherman J, Kaufman M. (1992). Familial <strong>in</strong>tracranial hypertension: report of a case and<br />
review of the literature. J Child <strong>Neuro</strong>l 7:196–198.<br />
Kieper GL, Sherman JD, Tomsick TA, Tew JM JR. (1999). Dural s<strong>in</strong>us thrombosis and pseudotumor cerebri: unexpected<br />
complications of suboccipital craniotomy and translabyr<strong>in</strong>th<strong>in</strong>e craniectomy. J <strong>Neuro</strong>surg 91:192–199.<br />
Kiers L, K<strong>in</strong>g JO. (1991). Increased <strong>in</strong>tracranial pressure follow<strong>in</strong>g bilateral neck dissection and radiotherapy.<br />
Aust NZ J Surg 61:459–461.<br />
Killer HE, Flammer J (2001). Unilateral papilledema caused by a fronto-temporo-parietal arachnoid cyst.<br />
Am J Ophthalmol 132:589–591.<br />
Kim AW, Trobe JD. (2000). Syndrome simulat<strong>in</strong>g pseudotumor cerebri caused by partial transverse s<strong>in</strong>us<br />
obstruction <strong>in</strong> metastatic prostate cancer. Am J Ophthalmol 129:254–256.<br />
K<strong>in</strong>g JO, Mitchell PJ, Thompson KR, Tress BM. (1995). Cerebral venography and manometry <strong>in</strong> idiopathic<br />
<strong>in</strong>tracranial hypertension. <strong>Neuro</strong>logy 45:2224–2228.<br />
K<strong>in</strong>g JO, Mitchell PJ, Thompson KR, Tress BM. (2002). Manometry comb<strong>in</strong>ed with cervical puncture <strong>in</strong> idiopathic<br />
<strong>in</strong>tracranial hypertension. <strong>Neuro</strong>logy 58:26–30.<br />
Kle<strong>in</strong>schmidt JJ, Digre KB, Hanover R. (2000). Idiopathic <strong>in</strong>tracranial hypertension. Relationship to depression,<br />
anxiety, and quality of life. <strong>Neuro</strong>logy 54:319–324.<br />
Knight RS, Fielder AR, Firth JL. (1986). Benign <strong>in</strong>tracranial hypertension: visual loss and optic nerve sheath<br />
fenestration. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 49:243–250.<br />
Kollar C, Parker G, Johnston I. (2001). Endovascular treatment of cranial venous s<strong>in</strong>us obstruction result<strong>in</strong>g <strong>in</strong><br />
pseudotumor syndrome. Report of three cases. J <strong>Neuro</strong>surg 94:646–651.<br />
Kollar CD, Johnston IH. (1999). Pseudotumor cerebri after arteriovenous malformation embolization. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 67:249.<br />
Koller EA, Stadel BV, Malozowski SN. (1997). Papilledema <strong>in</strong> 15 renally compromised patients treated with<br />
growth hormone. Pediatr Nephrol 11:451–454.<br />
Koppel BS, Kaunitz AM, Tuchman AJ. (1990). Pseudotumor cerebri follow<strong>in</strong>g eclampsia. Eur <strong>Neuro</strong>l 30:6–8.<br />
Kosmorsky GS, Boyle KA. (1993). Relief of headache after ONSD. 19th annual meet<strong>in</strong>g of the North American<br />
<strong>Neuro</strong>-Ophthalmologic Society, Big Sky, Montana.<br />
Krasnitz I, Beiran I, Mezer E, Miller B. (1997). Coexistence of optic nerve head drusen and pseudotumor cerebri: a<br />
cl<strong>in</strong>ical dilemma. Eur J Ophthalmol 7:383–386.<br />
Krishna R, Kosmorsky GS, Wright KW. (1998). Pseudotumor cerebri s<strong>in</strong>e papilledema with unilateral sixth nerve<br />
palsy. J <strong>Neuro</strong>-ophthalmol 18:53–55.<br />
Kumar RK, Ghali M, Dragojevic F, Young F. (1999). Papilloedema secondary to acute purulent s<strong>in</strong>usitis. J Paedriatr<br />
Child Health 35:396–398.<br />
Kupersmith MJ, Gemell L, Turb<strong>in</strong> R, et al. (1997). Effect of weight loss on pseudotumor cerebri <strong>in</strong> women.<br />
<strong>Neuro</strong>logy 48(suppl):A386.<br />
Kupersmith MJ, Gamell L, Turb<strong>in</strong> R, et al. (1998). Effects of weight loss on the course of idiopathic <strong>in</strong>tracranial<br />
hypertension. <strong>Neuro</strong>logy 50:1094–1098.<br />
Kurz-Lev<strong>in</strong> MM, Landau K. (1999). A comparison of imag<strong>in</strong>g techniques for diagnos<strong>in</strong>g drusen of the optic nerve<br />
head. Arch Ophthalmol 117:1045–1049.<br />
Lam BL, Schatz NJ, Glaser JS, Bowen BC. (1992). Pseudotumor cerebri from cranial venous obstruction.<br />
Ophthalmology 99:706–712.<br />
Lam BL, Siatkowski RM, Fox GM, Glaser JS. (1992). Visual loss <strong>in</strong> pseudotumor cerebri from branch ret<strong>in</strong>al artery<br />
occlusion. Am J Ophthalmol 113:334–336.
Papilledema 161<br />
Lee AG. (1995). Fourth nerve palsy <strong>in</strong> pseudotumor cerebri. Strabismus 3:57–59.<br />
Lee AG. (1996). Visual loss as the manifest<strong>in</strong>g symptom of ventriculoperitoneal shunt malfunction. Am J<br />
Ophthalmol 122:127–129.<br />
Lee AG, Beaver HA, Monsul NT, Miller NR. (2002a). Acute bilateral optic disk edema with a macular star figure <strong>in</strong><br />
a 12-year-old girl. Surv Ophthalmol 47:42–49.<br />
Lee AG, Brazis PW. (2000). Magnetic resonance venography <strong>in</strong> idiopathic pseudotumor cerebri. J <strong>Neuro</strong>-ophthalmol<br />
20:12–13.<br />
Lee AG, Golnik K, Kardon R, et al. (2002b). Sleep apnea and <strong>in</strong>tracranial hypertension <strong>in</strong> men. Ophthalmology<br />
109:482–485.<br />
Lee AG, Kardon RH, Wall M, Schlechte J. (2002c). Endocr<strong>in</strong>ologic abnormalities <strong>in</strong> pseudotumor cerebri <strong>in</strong> men.<br />
Presented at the 28th annual meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society. Copper<br />
Mounta<strong>in</strong>, Colorado, February 9–14, 2002.<br />
Lee AG, Patr<strong>in</strong>ely JR, Edmond JC. (1998). Optic nerve sheath decompression <strong>in</strong> pediatric pseudotumor cerebri.<br />
Ophthalmic Surg Lasers 29:514–517.<br />
Leggio MG, Cappa A, Mol<strong>in</strong>ari M, et al. (1995). Pseudotumor cerebri as present<strong>in</strong>g syndrome of Addisonian crisis.<br />
Ital J <strong>Neuro</strong>l Sci 16:387–389.<br />
Leker RR, Ste<strong>in</strong>er I. (1998). <strong>An</strong>ticardiolip<strong>in</strong> antibodies are frequently present <strong>in</strong> patients with idiopathic<br />
<strong>in</strong>tracranial hypertension. Arch <strong>Neuro</strong>l 55:817–820.<br />
Lepore FE. (1992). Unilateral and highly asymmetric papilledema <strong>in</strong> pseudotumor cerebri. <strong>Neuro</strong>logy 42:676–678.<br />
Lessell S. (1992). Pediatric pseudotumor cerebri (idiopathic <strong>in</strong>tracranial hypertension). Surv Ophthalmol<br />
37:155–166.<br />
Lev<strong>in</strong>e SH, Puchalski C. (1990). Pseudotumor cerebri associated with lithium therapy <strong>in</strong> two patients. J Cl<strong>in</strong><br />
Psychiatry 51:251–253.<br />
Lewis PA, Kearrney PJ. (1997). Pseudotumor cerebri <strong>in</strong>duced by m<strong>in</strong>ocycl<strong>in</strong>e treatment for acne vulgaris. Acta<br />
Derm Venereol 77:83.<br />
Liu GT, Kay MD, Bienfang DC, Schatz NJ. (1994). Pseudotumor cerebri associated with corticosteroid withdrawal<br />
<strong>in</strong> <strong>in</strong>flammatory bowel disease. Am J Ophthalmol 15:352–357.<br />
Liu GT, Volpe NJ, Schatz NJ, Galetta SL, Farrar JT, Raps EC. (1996). Severe sudden visual loss caused by<br />
pseudotumor cerebri and lumboperitoneal shunt failure. Am J Ophthalmol 122:129–131.<br />
Lucidi V, Di Capua M, Rosati P, Papadatou B, Castro M. (1993). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> an older child<br />
with cystic fibrosis. Pediatr <strong>Neuro</strong>l 9:494–495.<br />
Lundar T, Nornes H. (1990). Pseudotumor cerebri. <strong>Neuro</strong>surgical considerations. Acta <strong>Neuro</strong>chir 51:366–368.<br />
Mada Mohan P, Noushad TP, Sarita P, et al. (1993). Hypoparathyroidism with benign <strong>in</strong>tracranial hypertension.<br />
J Assoc Physicians India 41:752–753.<br />
Mahmoud HH, Hurwitz CA, Roberts WM, et al. (1993). Tret<strong>in</strong>o<strong>in</strong> toxicity <strong>in</strong> children with acute promyelocytic<br />
leukaemia. Lancet 342:1394–1395.<br />
Malozowski S, Tanner LA, Wysowski DK, et al. (1995). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> children with growth<br />
hormone deficiency treated with growth hormone. J Pediatr 126:996–999.<br />
Maneatis T, Baptista J, Connelly K, Blethen S. (2000). Growth hormone safety update from the National<br />
Cooperative Growth Study. J Pediatr Endocr<strong>in</strong>ol 13(suppl 2):1035–1044.<br />
Manfre L, Lagalla R, Mangiameli A, et al. (1995). Idiopathic <strong>in</strong>tracranial hypertension: Orbital MRI. <strong>Neuro</strong>radiology<br />
37:459–461.<br />
Marcelis J, Silberste<strong>in</strong> SD. (1991). Idiopathic <strong>in</strong>tracranial hypertension without papilledema. Arch <strong>Neuro</strong>l<br />
48:392–399.<br />
Marcus DM, Lynn J, Miller JJ, et al. (2001). Sleep disorders: a risk factor for pseudotumor cerebri?<br />
J <strong>Neuro</strong>-<strong>ophthalmology</strong> 21:121–123.<br />
Mart<strong>in</strong> TJ, Bell DA, Wilson JA. (1998). Papilledema <strong>in</strong> a man with an ‘‘occult’’ dural arteriovenous malformation.<br />
J <strong>Neuro</strong>-ophthalmol 18:49–52.<br />
Mathew NT, Ravishankar K, San<strong>in</strong> LC. (1996). Coexistence of migra<strong>in</strong>e and idiopathic <strong>in</strong>tracranial hypertension<br />
without papilledema. <strong>Neuro</strong>logy 46:1226–1230.<br />
Mauriello JA, Shaderowfsky P, Gizzi M, Frohman L. (1995). Management of visual loss after optic nerve sheath<br />
decompression <strong>in</strong> patients with pseudotumor cerebri. Ophthalmology 102:441–445.<br />
Mayer Benegas N, Volpe NJ, Liu GT, Galetta SL. (1996). Hemifacial spasm and idiopathic <strong>in</strong>tracranial hypertension.<br />
J <strong>Neuro</strong>-ophthalmol 16:70.<br />
Mayer-Hubner B. (1996). Pseudotumor cerebri from <strong>in</strong>tranasal oxytoc<strong>in</strong> and excessive fluid <strong>in</strong>take. Lancet 347:623<br />
McDonnell GV, Patterson VH, McK<strong>in</strong>stry S. (1997). Cerebral venous thrombosis occurr<strong>in</strong>g dur<strong>in</strong>g an ectopic<br />
pregnancy and complicated by <strong>in</strong>tracranial hypertension. Br J Cl<strong>in</strong> Pract 51:194–197.
162 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
McLean C, Lobo R, Brazier OJ. (1998). Optic disc <strong>in</strong>volvement <strong>in</strong> hypocalcaemia with hypoparathyroidism:<br />
papilloedema or optic neuropathy? <strong>Neuro</strong>-<strong>ophthalmology</strong> 117–124.<br />
Midroni G, Dyck PJ. (1996). Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy: unusual cl<strong>in</strong>ical features and<br />
therapeutic responses. <strong>Neuro</strong>logy 46:1206–1212.<br />
Milhorat TH, Chou MW, Tr<strong>in</strong>idad EM, et al. (1999). Chiari I malformation redef<strong>in</strong>ed: cl<strong>in</strong>ical and radiographic<br />
f<strong>in</strong>d<strong>in</strong>gs for 354 symptomatic patients. <strong>Neuro</strong>surgery 44:1005–1017.<br />
Miller NR. (1997). Bilateral visual loss and simultagnosia after lumboperitoneal shunt for pseudotumor cerebri.<br />
J <strong>Neuro</strong>-ophthalmol 17:36–38.<br />
Misra M, Khan GM, Rath S. (1992). Eltrox<strong>in</strong> <strong>in</strong>duced pseudotumor cerebri—a case report. Indian J Ophthalmol 40:117.<br />
Mittra RA, Sergott RC, Flaharty PM, et al. (1993). Optic nerve decompression improves hemodynamic parameters<br />
<strong>in</strong> papilledema. Ophthalmology 100:987–997.<br />
Mokri B, Jack CR Jr, Petty GW. (1993). Pseudotumor syndrome associated with cerebral venous s<strong>in</strong>us occlusion<br />
and antiphospholipid antibodies. Stroke 24:469–472.<br />
Moodley M, Coovadia HM. (1990). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> typhoid fever. A case report. South Afr Med<br />
J78:608–609.<br />
Morrison KE, Davies PTG. (1999). Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy present<strong>in</strong>g with headache<br />
and papilledema. Headache 39:299–300.<br />
Moskowitz Y, Leibowitz E, Ronen M, Aviel E. (1993). Pseudotumor cerebri <strong>in</strong>duced by vitam<strong>in</strong> A comb<strong>in</strong>ed with<br />
m<strong>in</strong>ocycl<strong>in</strong>e. <strong>An</strong>n Ophthalmol 25:306–308.<br />
Mukherjee A, Dutta P, Lahiri M, et al. (1990). Benign <strong>in</strong>tracranial hypertension after nalidixic acid overdose <strong>in</strong><br />
<strong>in</strong>fant. Lancet 335:1602.<br />
Naderi S, Nukala S, Marruenda F, et al. (1999). Pseudotumor cererbi <strong>in</strong> acute promyelocytic leukemia:<br />
improvement despite cont<strong>in</strong>ued ATRA therapy. <strong>An</strong>n Hematol 78:333–334.<br />
Nagamoto G, Smith WE. (1992). Benign <strong>in</strong>tracranial hypertension and chronic renal failure. Cleve Cl<strong>in</strong> J Med<br />
59:419–422.<br />
Nakamura N, Hara R, Kimura R, et al. (1999). Optic per<strong>in</strong>euritis not associated with syphilitic <strong>in</strong>fection.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 21:135–145.<br />
Nasr SZ, Schaffert D. (1995). Symptomatic <strong>in</strong>crease <strong>in</strong> <strong>in</strong>tracranial pressure follow<strong>in</strong>g pancreatic enzyme<br />
replacement therapy for cystic fibrosis. Pediatr Pulmonol 19:396–397.<br />
Newborg B. (1974). Pseudotumor cerebri treated with rice reduction diet. Arch Intern Med 133:802–807.<br />
Newman NJ, Sedwick LA, Boghen DR. (1994a). Bilateral visual loss and disc edema <strong>in</strong> a 15-year-old girl.<br />
Surv Ophthalmol 38:365–370.<br />
Newman NJ, Selzer KA, Bell RA. (1994b). Association of multiple sclerosis and <strong>in</strong>tracranial hypertension. J <strong>Neuro</strong><strong>ophthalmology</strong><br />
14:189–192.<br />
Nezu A, Kimura S, Osaka H. (1995). Tolosa-Hunt syndrome with pseudotumor cerebri. Report of an unusual case.<br />
Bra<strong>in</strong> Dev 17:216–218.<br />
Obeid T, Awada A, Huraib S, et al. (1997). Pseudotumor cerebri <strong>in</strong> renal transplant recipients: a diagnostic<br />
challenge. J Nephrol 10:258–260.<br />
O’Duffy D, James B, Elston J. (1998). Idiopathic <strong>in</strong>tracranial hypertension present<strong>in</strong>g with gaze-evoked amaurosis.<br />
Acta Ophthalmol Scand 76:119–120.<br />
O’Halloran HS, Berger JR, Baker RS, et al. (1999). Optic nerve edema as a consequence of respiratory disease.<br />
<strong>Neuro</strong>logy 53:2204–2205.<br />
Orefice G, De Joanna G, Coppola M, et al. (1995). Benign <strong>in</strong>tracranial hypertension: a non-thrombotic complication<br />
of the primary antiphospholipid syndrome? Lupus 4:324–326.<br />
Panozzo G, Babighian S, Bonora A. (1998). Association of xerophthalmia, flecked ret<strong>in</strong>a, and pseudotumor cerebri<br />
caused by hypovitam<strong>in</strong>osis A. Am J Ophthalmol 125:708–710.<br />
Parfitt VJ, Dearlove JC, Savage D, et al. (1994). Benign <strong>in</strong>tracranial hypertension after pituitary surgery for<br />
Cush<strong>in</strong>g’s disease. Postgrad Med J 70:115–117.<br />
Patton N, Beatty S, Lloyd IC. (2000). Bilateral sixth and fourth nerve palsies <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension. J R Soc Med 93:80–81.<br />
Pears J, Sandercock PA. (1990). Benign <strong>in</strong>tracranial hypertension associated with danazol. Scott Med J 35:49.<br />
Pearson PA, Baker RS, Khorram D, Smith TJ. (1991). Evaluation of optic nerve sheath fenestration <strong>in</strong> pseudotumor<br />
cerebri us<strong>in</strong>g automated perimetry. Ophthalmology 98:99–105.<br />
Pelton RW, Lee AG, Orengo-Nania SD, Patr<strong>in</strong>ely JR. (1999). Bilateral optic disk edema caused by sarcoidosis<br />
mimick<strong>in</strong>g pseudotumor cerebri. Am J Ophthalmol 127:229–230.<br />
Plotnik, JL, Kosmorsky, GS. (1993). Operative complications of optic nerve sheath decompression. Ophthalmology<br />
100:683–690.
Papilledema 163<br />
Prevett MC, Plant GT. (1997). Intracranial hypertension and HIV associated men<strong>in</strong>goradiculitis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 62:407–409.<br />
Provenzale JM, Barboriak DP, Ortel TL. (1998). Dural s<strong>in</strong>us thrombosis associated with activated prote<strong>in</strong> C<br />
resistance: MR imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs and proband identification. Am J Radiol 170:499–502.<br />
Purv<strong>in</strong> VA, Kawasaki A, Yee RD (2000). Papilledema and obstructive sleep apnea syndrome. Arch Ophthalmol<br />
118:1626–1630.<br />
Quattrone A, Bono F, Oliveri RL, et al. (2001). Cerebral venous thrombosis and isolated <strong>in</strong>tracranial hypertension<br />
without papilledema <strong>in</strong> CDH. <strong>Neuro</strong>logy 57:31–36.<br />
Raghavan S, DiMart<strong>in</strong>o-Nardi J, Saenger P, L<strong>in</strong>der B. (1997). Pseudotumor cerebri <strong>in</strong> an <strong>in</strong>fant after L-thyrox<strong>in</strong>e<br />
therapy for transient neonatal hypothyroidism. J Pediatr 130:478–480.<br />
Redwood MD, W<strong>in</strong>er JB, Rossor M. (1990). <strong>Neuro</strong>sarcoidosis present<strong>in</strong>g as benign <strong>in</strong>tracranial hypertension. Eur<br />
<strong>Neuro</strong>l 30:282–283.<br />
Radhakrishnan K, Ahlskog JE, Garrity JA, Kurland LT. (1994). Idiopathic <strong>in</strong>tracranial hypertension. Mayo Cl<strong>in</strong> Proc<br />
69:169–180.<br />
Radhakrishnan K, Ahlskog JE, Cross SA, Kurland LT, O’Fallon WM. (1993a). Idiopathic <strong>in</strong>tracranial<br />
hypertension (pseudotumor cerebri): descriptive epidemiology <strong>in</strong> Rochester, M<strong>in</strong>n, 1976 to 1990. Arch<br />
<strong>Neuro</strong>l 50:78–80.<br />
Radhakrishnan K, Thacker AK, Bohlaga NH, Maloo JC, Gerryo SE. (1993b). Epidemiology of idiopathic<br />
<strong>in</strong>tracranial hypertension: a prospective and case-control study. J <strong>Neuro</strong>l Sci 116:18–28.<br />
Rogers AH, Rogers GL, Bremer DL, McGregor ML (1999). Pseudotumor cerebri <strong>in</strong> children receiv<strong>in</strong>g recomb<strong>in</strong>ant<br />
human growth hormone. Ophthalmology 106:1186–1190.<br />
Rosenberg, ML, Corbett, JJ, Smith, C, et al. (1993). Cerebrosp<strong>in</strong>al fluid diversion procedures <strong>in</strong> pseudotumor<br />
cerebri. <strong>Neuro</strong>logy 43:1071–1072.<br />
Rosenfeld JV, Widaa HA, Adams CB. (1991). Cerebral arteriovenous malformation caus<strong>in</strong>g benign <strong>in</strong>tracranial<br />
hypertension - case report. <strong>Neuro</strong>l Med Chir 31:523–525.<br />
Rothermel H, Hedges TR 3rd, Steere AC (2001). Optic neuropathy <strong>in</strong> children with Lyme disease. Pediatrics<br />
108:477–481.<br />
Round R, Keane JR. (1988). The m<strong>in</strong>or symptoms of <strong>in</strong>creased <strong>in</strong>tracranial pressure: 101 patients with benign<br />
<strong>in</strong>tracranial hypertension. <strong>Neuro</strong>logy 38:1461–1464.<br />
Rowe FJ, Sarkies NJ. (1998). Assessment of visual function <strong>in</strong> idiopathic <strong>in</strong>tracranial hypertension: a prospective<br />
study. Eye 12:111–118.<br />
Sacchi S, Kantarjian HM, Freireich EJ, et al. (1999). Unexpected high <strong>in</strong>cidence of severe toxicities associated with<br />
alpha <strong>in</strong>terferon, low-dose cytos<strong>in</strong>e arab<strong>in</strong>oside and all-trans ret<strong>in</strong>oic acid <strong>in</strong> patients with chronic<br />
myelogenous leukemia. Leuk Lymphoma 35:483–489.<br />
Sadun AA, Currie JN, Lessell S. (1984). Transient visual obscurations with elevated discs. <strong>An</strong>n <strong>Neuro</strong>l 16:489–494.<br />
Saito J, Kami M, Taniguchi F, et al. (1999). Case report. Unilateral papilledema after bone marrow transplantation.<br />
Bone Marrow Transplant 23:963–965.<br />
Saitoh S, Momoi MY, Gunji Y. (2000). Pseudotumor cerebri manifest<strong>in</strong>g as a symptom of acute promyelocytic<br />
leukemia. Pediatr Int 42:97–99.<br />
Sakamaki Y, Nakamura R, Uchida M, et al. (1990). A case of pseudotumor cerebri follow<strong>in</strong>g glucocorticoid<br />
therapy <strong>in</strong> which warfar<strong>in</strong> prevented recurrence. Jpn J Med 29:566–570.<br />
Salaria M, Poddar B, Parmar V. (2001). Rickets present<strong>in</strong>g as pseudotumor cerebri and seizures. Indian J Pediatr<br />
68:181.<br />
Salgarello T, Tamburrelli C, Fals<strong>in</strong>i B, et al. (1996). Optic nerve diameters and perimetric thresholds <strong>in</strong> idiopathic<br />
<strong>in</strong>tracranial hypertension. Br J Ophthalmol 80:509–514.<br />
Sant<strong>in</strong>elli R, Tolone C, Toraldo R, et al. (1998). Familial idiopathic <strong>in</strong>tracranial hypertension with sp<strong>in</strong>al and<br />
radicular pa<strong>in</strong>. Arch <strong>Neuro</strong>l 55:854–856.<br />
Schoeman JF. (1994). Childhood pseudotumor cerebri: cl<strong>in</strong>ical and <strong>in</strong>tracranial pressure response to acetazolamide<br />
and furosemide treatment <strong>in</strong> a case series. J Child <strong>Neuro</strong>l 9:130–134.<br />
Schroeter T, Lanvers C, Herd<strong>in</strong>g H, Suttorp M. (2000). Pseudotumor cerebri <strong>in</strong>duced by all-trans-ret<strong>in</strong>oic acid <strong>in</strong> a<br />
child treated for acute promyelocytic leukemia. Med Pediatr Oncol 34:284–286.<br />
Schwarz S, Husstedt IW, Georgiadis D, et al. (1995). Benign <strong>in</strong>tracranial hypertension <strong>in</strong> an HIV-<strong>in</strong>fected patient:<br />
headache as the only present<strong>in</strong>g sign. AIDS 9:657–658.<br />
Scott IU, Siatkowski RM, Eneyni M, Brodsky MC, Lam BL. (1997). Idiopathic <strong>in</strong>tracranial hypertension <strong>in</strong> children<br />
and adolescents. Am J Ophthalmol 124:253–255.<br />
Selhorst JB, Kulkantrakorn K, Corbett JJ, et al. (2000). Ret<strong>in</strong>ol-b<strong>in</strong>d<strong>in</strong>g prote<strong>in</strong> <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension (IIH). J <strong>Neuro</strong>-ophthalmol 20:250–252.<br />
Selky AK, Dobyns WB, Yee RD. (1994a). Idiopathic <strong>in</strong>tracranial hypertension and facial diplegia. <strong>Neuro</strong>logy 44:457.
164 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Selky AK, Purv<strong>in</strong> VA. (1994b). Hemifacial spasm. <strong>An</strong> unusual manifestation of idiopathic <strong>in</strong>tracranial hypertension.<br />
J <strong>Neuro</strong>-ophthalmol 14:196–198.<br />
Sell JJ, Ruppe FW, Orrison WW Jr. (1995). Iatrogenically <strong>in</strong>duced <strong>in</strong>tracranial hypotension syndrome. Am J Radiol<br />
165:1513–1515.<br />
Selleri C, Pane F, Notaro R, et al. (1996). All-trans-ret<strong>in</strong>oic acid (ATRA) responsive sk<strong>in</strong> relapses of acute<br />
promyelocytic leukaemia followed by ATRA-<strong>in</strong>duced pseudotumor cerebri. Br J Haematol 92:937–940.<br />
Sergott RC, Sav<strong>in</strong>o PJ, Bosley TM. (1988). Optic nerve sheath decompression provides long-term visual<br />
improvement for pseudotumor cerebri. Arch Ophthalmol 106:1384–1390.<br />
Shapiro S, Yee R, Brown H. (1995). Surgical management of pseudotumor cerebri <strong>in</strong> pregnancy: case report.<br />
<strong>Neuro</strong>surgery 37:829–831.<br />
Sharieff GQ, Hanten K. (1996). Pseudotumor cerebri and hypercalcemia result<strong>in</strong>g from vitam<strong>in</strong> A toxicity.<br />
<strong>An</strong>n Emerg Med 27:518–521.<br />
Sheridan M, Johnston I. (1994). Hydrocephalus and pseudotumor cerebri <strong>in</strong> the mucopolysaccharidoses. Childs<br />
Nerv Syst 10:148–150.<br />
Sherman MD, Own KH. (1999). Interstitial nephritis and uveitis present<strong>in</strong>g with bilateral optic disk edema. Am J<br />
Ophthalmol 127:609–610.<br />
Sheth KJ, Kivl<strong>in</strong> JD, Leichter HE, et al. (1994). Pseudotumor cerebri with vision impairment <strong>in</strong> two children with<br />
renal transplantation. Pediatr Nephrol 8:91–93.<br />
Siatkowski RM, Vilar NF, Sternau L, Co<strong>in</strong> CG. (1999). Bl<strong>in</strong>dness from bad bones. Surv Ophthalmol 43:487–490.<br />
Sirdofsky M, Kattah J, Macedo P. (1994). Intracranial hypertension <strong>in</strong> a diet<strong>in</strong>g patient. J <strong>Neuro</strong>-ophthalmol 14:9–11.<br />
Sismanis A, Butts FM, Hughes GB. (1990). Objective t<strong>in</strong>nitus <strong>in</strong> benign <strong>in</strong>tracranial hypertension. <strong>An</strong> update.<br />
Laryngoscope 100:33–36.<br />
Smith KH, Wilk<strong>in</strong>son JT, Br<strong>in</strong>dley GO. (1992). Comb<strong>in</strong>ed third and sixth nerve paresis follow<strong>in</strong>g optic nerve<br />
sheath fenestration. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:85–87.<br />
Soler D, Cox T, Bullock P, et al. (1998). Diagnosis and management of benign <strong>in</strong>tracranial hypertension. Arch Dis<br />
Child 78:89–94.<br />
Speer C, Pearlman J, Phillips PH, et al. (1999). Fourth nerve palsy <strong>in</strong> pediatric pseudotumor cerebri. Am J<br />
Ophthalmol 127:236–237.<br />
Spoor TC, McHenry JG. (1993). Long-term effectiveness of optic nerve sheath decompression for pseudotumor<br />
cerebri. Arch Ophthalmol 111:632–635.<br />
Spoor TC, McHenry JG, Sh<strong>in</strong> DH. (1995). Long-term results us<strong>in</strong>g adjunctive mitomyc<strong>in</strong> C <strong>in</strong> optic nerve sheath<br />
decompression for pseudotumor cerebri. Ophthalmology 102:2024–2028.<br />
Spoor TC, Ramocki JM, Madison MP, et al. (1991). Treatment of pseudotumor cerebri by primary and secondary<br />
optic nerve sheath decompression. Am J Ophthalmol 112:177–185.<br />
Stavroua P, Honan WP. (1997). Contrast sensitivity <strong>in</strong> benign <strong>in</strong>tracranial hypertension. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
17:127–134.<br />
Strom<strong>in</strong>ger MB, Weiss GB, Mehler MF. (1992). Asymptomatic unilateral papilledema <strong>in</strong> pseudotumor cerebri. J<br />
Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:238–241.<br />
Sugerman HJ, DeMaria EJ, Felton WL, et al. (1997). Increased <strong>in</strong>tra-abdom<strong>in</strong>al pressure and cardiac fill<strong>in</strong>g<br />
pressure <strong>in</strong> obesity-associated pseudotumor cerebri. <strong>Neuro</strong>logy 49:507–511.<br />
Sugerman HJ, Felton WL 3rd, Salvant JB Jr, et al. (1995). Effects of surgically <strong>in</strong>duced weight loss on idiopathic<br />
<strong>in</strong>tracranial hypertension <strong>in</strong> morbid obesity. <strong>Neuro</strong>logy 45:1655–1659.<br />
Sugerman HJ, Felton WL 3rd, Sismanis A, et al. (1999). Gastric surgery for pseudotumor cerebri associated with<br />
severe obesity. <strong>An</strong>n Surg 229:634–640.<br />
Sullivan HC. (1991). Fatal tonsillar herniation <strong>in</strong> pseudotumor cerebri. <strong>Neuro</strong>logy 41:1142–1144.<br />
Sussman J, Leach M, Greaves M, et al. (1997). Potentially prothrombotic abnormalities of coagulation <strong>in</strong> benign<br />
<strong>in</strong>tracranial hypertension. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 62:229–233.<br />
Suzuki H, Takanashi J, Kobayashi K, et al (2001). MR imag<strong>in</strong>g of idiopathic <strong>in</strong>tracranial hypertension. AJNR<br />
22:196–199.<br />
Talks SJ, Mossa F, Elston JS. (1998). The contribution of macular changes to visual loss <strong>in</strong> benign <strong>in</strong>tracranial<br />
hypertension. Eye 12:806–808.<br />
Teh<strong>in</strong>drazanarivelo A, Bousser MG. (1990). Possible benign <strong>in</strong>tracranial hypertension and essential thrombocythaemia.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 53:819.<br />
Teh<strong>in</strong>drazanarivelo A, Evard S, Schaison M, et al. (1992). Prospective study of cerebral s<strong>in</strong>us venous thrombosis <strong>in</strong><br />
patients present<strong>in</strong>g with benign <strong>in</strong>tracranial hypertension. Cerebrovasc Dis 2:22–27.<br />
Thomas DA, Trobe JD, Cornblath WT. (1999). Visual loss secondary to <strong>in</strong>creased <strong>in</strong>tracranial pressure <strong>in</strong><br />
neurofibromatosis type 2. Arch Ophthalmol 117:1650–1653.<br />
To KW, Warren FA. (1990). Unilateral papilledema <strong>in</strong> pseudotumor cerebri. Arch Ophthalmol 108:644–645.
Papilledema 165<br />
Torres M, May E, Watanabe F, et al. (1997). Intracranial hypertension associated with m<strong>in</strong>ocycl<strong>in</strong>e. Presented at the<br />
23rd <strong>An</strong>nual Meet<strong>in</strong>g of the North American <strong>Neuro</strong> Ophthalmology Society, Keystone, Colorado, February<br />
9–13.<br />
Tourn N, Sharpe JA. (1996). Pseudotumor cerebri mimick<strong>in</strong>g Foster Kennedy syndrome. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
16:55–57.<br />
Travero F, Stagnaro R, Fazio B. (1993). Benign <strong>in</strong>tracranial hypertension with HIV <strong>in</strong>fection. Eur <strong>Neuro</strong>l 33:191–192.<br />
Tugal O, Jacobson R, Berez<strong>in</strong> S, et al. (1994). Recurrent benign <strong>in</strong>tracranial hypertension due to iron deficiency<br />
anemia. Case report and review of the literature. Am J Pediatr Hematol Oncol 16:266–270.<br />
Tulipan N, Lav<strong>in</strong> PJ, Copeland M. (1998). Stereotactic ventriculoperitoneal shunt for idiopathic <strong>in</strong>tracranial<br />
hypertension: technical note. <strong>Neuro</strong>surgery 43:175–176.<br />
Vachvanichsanong P, Dissaneewate P, Vasikananont P. (1992). Pseudotumor cerebri <strong>in</strong> a boy with systemic lupus<br />
erythematosus. Am J Dis Child 146:1417–1419.<br />
Van den Br<strong>in</strong>k WA, Pieterman H, Avezaat CJ. (1996). Sagittal s<strong>in</strong>us occlusion, caused by an overly<strong>in</strong>g depressed<br />
cranial fracture, present<strong>in</strong>g with late signs and symptoms of <strong>in</strong>tracranial hypertension: case report.<br />
<strong>Neuro</strong>surgery 38:1044–1046.<br />
Van Gelder T, van Gemert HM, Tjiong HL. (1991). A patient with megaloblastic anaemia and idiopathic<br />
<strong>in</strong>tracranial hypertension. Case history. Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg 93:321–322.<br />
Varadi G, Lossos A, Or R, et al. (1995). Successful allogenic bone marrow transplantation <strong>in</strong> a patient with<br />
ATRA-<strong>in</strong>duced pseudotumor cerebri. Am J Hematol 50:147–148.<br />
Vargas JA, Garcia-Mer<strong>in</strong>o A, Rodriguez E, Villagra A. (1990). Pseudotumor cerebri complicat<strong>in</strong>g typhoid fever.<br />
Eur <strong>Neuro</strong>l 30:345–346.<br />
Verderber L, Lav<strong>in</strong> P, Wesley R. (1991). Pseudotumor cerebri and chronic benzene hexacloride (l<strong>in</strong>dane) exposure.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:113.<br />
Verm A, Lee AG. (1997). Bilateral optic disk edema with macular exudates as the manifest<strong>in</strong>g sign of a cerebral<br />
arteriovenous malformation. Am J Ophthalmol 123:422–424.<br />
Visani G, Bontempo G, Manfroi S, et al. (1996). All-trans-ret<strong>in</strong>oic acid and pseudotumor cerebri <strong>in</strong> a young adult<br />
with acute promyelocytic leukemia: a possible disease association. Haematologica 81:152–154.<br />
Walker RWH. (2001). Idiopathic <strong>in</strong>tracranial hypertension: any light on the mechanism of the raised pressure?<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 71:1–5.<br />
Wall M. (1990). The headache profile of idiopathic <strong>in</strong>tracranial hypertension. Cephalgia 10:331–335.<br />
Wall M. (2000). Idiopathic <strong>in</strong>tracranial hypertension: mechanisms of visual loss and disease management.<br />
Sem<strong>in</strong> <strong>Neuro</strong>l 20:89–95.<br />
Wall M, Breen L, W<strong>in</strong>terkorn J. (1995a). Optic disk edema with cotton-wool spots. Surv Ophthalmol 39:502–508.<br />
Wall M, George D. (1987). Visual loss <strong>in</strong> pseudotumor cerebri. Incidence and defects related to visual field<br />
strategy. Arch <strong>Neuro</strong>l 44:170–175.<br />
Wall M, George D. (1991). Idiopathic <strong>in</strong>tracranial hypertension. A prospective study of 50 patients. Bra<strong>in</strong><br />
114:155–180.<br />
Wall M, Montgomery EB. (1995b). Us<strong>in</strong>g motion perimetry to detect visual field defects <strong>in</strong> patients with idiopathic<br />
<strong>in</strong>tracranial hypertension: a comparison with conventional automated perimetry. <strong>Neuro</strong>logy 45:1169–1175.<br />
Wall M, White WN 2nd. (1998). Asymmetric papilledema <strong>in</strong> idiopathic <strong>in</strong>tracranial hypertension: prospective<br />
<strong>in</strong>terocular comparison of sensory visual function. Invest Ophthalmol Vis Sci 39:134–142.<br />
Wang S-J, Silberste<strong>in</strong> SD, Patterson S, Young WB. (1996). Idiopathic <strong>in</strong>tracranial hypertension without papilledema.<br />
A case-control study <strong>in</strong> a headache center. <strong>Neuro</strong>logy 51:245–249.<br />
Wasan H, Mansi JL, Benjam<strong>in</strong> S, et al. (1992). Myeloma and benign <strong>in</strong>tracranial hypertension. Br Med J 304:685.<br />
Watnick RL, Trobe JD. (1989). Bilateral optic nerve compression as a mechanism for the Foster-Kennedy<br />
syndrome. Ophthalmology 96:1793–1798.<br />
Weiss GB, Bajwa ZH, Mehler MF. (1991). Co-occurrence of pseudotumor cerebri and Guilla<strong>in</strong>-Barré syndrome <strong>in</strong><br />
an adult. <strong>Neuro</strong>logy 41:603–604.<br />
W<strong>in</strong>row AP, Supramaniam G. (1990). Benign <strong>in</strong>tracranial hypertension after ciprofloxac<strong>in</strong> adm<strong>in</strong>istration. Arch Dis<br />
Child 65:1165–1166.<br />
Wol<strong>in</strong> MJ, Brannon WL. (1995). Disk edema <strong>in</strong> an overweight woman. Surv Ophthalmol 39:307–314.<br />
Wong VA, Wade NK. (1998). POEMS syndrome: an unusual cause of bilateral disk swell<strong>in</strong>g. Am J Ophthalmol<br />
126:452–454.<br />
Yokokura M, Hatake K, Komatsu N, et al. (1994). Toxicity of tret<strong>in</strong>o<strong>in</strong> <strong>in</strong> acute promyelocytic leukaemia. Lancet<br />
343:361–362.
This page <strong>in</strong>tentionally left blank
8 r<br />
Transient Visual Loss<br />
What Questions Should Be Asked of a Patient<br />
with Transient Visual Loss?<br />
The most important questions that need to be addressed <strong>in</strong> the assessment of the patient<br />
with transient visual loss (TVL) <strong>in</strong>clude the follow<strong>in</strong>g:<br />
1. Is the visual loss monocular or b<strong>in</strong>ocular? Monocular TVL implies disease of the eye,<br />
ret<strong>in</strong>a, optic nerve, orbit, circulation to the eye (e.g., heart, aorta, carotid artery,<br />
ophthalmic artery, central ret<strong>in</strong>al artery), or migra<strong>in</strong>e. B<strong>in</strong>ocular TVL implies bilateral<br />
eye disease, disease affect<strong>in</strong>g the circulation to both eyes (e.g., bilateral carotid<br />
stenosis), <strong>in</strong>creased <strong>in</strong>tracranial pressure with papilledema, vertebrobasilar ischemia<br />
or <strong>in</strong>sufficiency, or migra<strong>in</strong>e.<br />
2. What is the temporal profile of the transient loss of vision? For example, TVL <strong>in</strong> one<br />
eye last<strong>in</strong>g seconds is characteristic of transient obscurations of vision result<strong>in</strong>g from<br />
optic nerve ischemia or papilledema. Monocular TVL last<strong>in</strong>g 2 to 30 m<strong>in</strong>utes is<br />
characteristic of TVL associated with carotid artery disease.<br />
3. What are the precipitants of the visual loss? For example, patients with an<br />
<strong>in</strong>traorbital mass may develop TVL only <strong>in</strong> certa<strong>in</strong> eye positions due to the mass<br />
compress<strong>in</strong>g the ipsilateral optic nerve or optic nerve circulation (gaze-evoked<br />
amaurosis). Monocular or b<strong>in</strong>ocular TVL due to carotid disease may occur follow<strong>in</strong>g<br />
exposure to bright light.<br />
4. Are optic nerve or ret<strong>in</strong>al vessel abnormalities evident on funduscopic exam<strong>in</strong>ation?<br />
For example, the fundus exam may reveal papilledema <strong>in</strong> transient obscurations<br />
of vision, ret<strong>in</strong>al emboli <strong>in</strong> carotid or cardiac disease, and disc anomalies <strong>in</strong><br />
monocular TVL.<br />
This chapter discusses various entities that may cause monocular or b<strong>in</strong>ocular TVL.<br />
Approaches to patients with monocular and b<strong>in</strong>ocular transient visual loss are given <strong>in</strong><br />
Figures 8–1 and 8–2, respectively.<br />
167
168 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 8–1. Evaluation of monocular transient visual loss (TVL).<br />
Does Monocular TVL Occur Only <strong>in</strong> Certa<strong>in</strong><br />
Positions of Gaze (Gaze-Evoked TVL)?<br />
Patients who experience TVL evoked by eccentric position of gaze (gaze-evoked TVL)<br />
usually have an <strong>in</strong>traorbital mass that <strong>in</strong>termittently compresses the circulation to the<br />
optic nerve or ret<strong>in</strong>a (Bremner, 1999; Danish-Meyer, 2001; Knapp, 1992; Kohmoto, 1993;<br />
Mezer, 1997; Smith, 1998). The visual loss immediately clears when the direction of gaze<br />
is changed. The most common lesions are orbital cavernous hemangiomas or optic<br />
nerve sheath men<strong>in</strong>giomas. Other orbital lesions produc<strong>in</strong>g this sign <strong>in</strong>clude osteomas,<br />
neurofibromas, gliomas, medial rectus granular myoblastoma, metastases, varices,<br />
orbital trauma, thyroid eye disease, and <strong>in</strong>traocular foreign body (buckshot pellet).<br />
The exam<strong>in</strong>ation may be normal or show evidence of an optic neuropathy with an<br />
afferent pupil defect, color vision impairment, disc edema, and optociliary collateral<br />
vessels. Other signs of orbital tumor, such as proptosis, limitation of extraocular muscle
Figure 8–2. Evaluation of b<strong>in</strong>ocular transient visual loss (TVL).<br />
Transient Visual Loss 169
170 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 8–2. (cont<strong>in</strong>ued )<br />
movement, swell<strong>in</strong>g of the eyelids, chemosis, and conjunctival congestion, may be<br />
evident. Evaluation requires magnetic resonance imag<strong>in</strong>g (MRI) or computed tomography<br />
(CT) scans of the orbital structures. Intermittent visual loss and exophthalmos<br />
may occur with bend<strong>in</strong>g over or the Valsalva maneuver (Sobottka Ventura, 2001). Gazeevoked<br />
monocular TVL has also been noted <strong>in</strong> patients with pseudotumor cerebri
(O’Duffy, 1998). It has been hypothesized that <strong>in</strong> an eccentric position of gaze, ischemic<br />
compression of a tense dilated optic nerve sheath results <strong>in</strong> elevation of <strong>in</strong>trasheath<br />
pressure compromis<strong>in</strong>g blood flow to the ret<strong>in</strong>a or optic nerve (Miller, 1991; O’Duffy,<br />
1998).<br />
Does the Visual Loss Occur After Prolonged<br />
Read<strong>in</strong>g (Read<strong>in</strong>g-Evoked TVL)?<br />
Read<strong>in</strong>g may also <strong>in</strong>duce monocular TVL. Manor et al described a 49-year-old man with<br />
a 5-year history of dimm<strong>in</strong>g of central vision <strong>in</strong> the left eye provoked only dur<strong>in</strong>g<br />
read<strong>in</strong>g (Manor, 1996). <strong>An</strong> orbital apex tumor situated lateral to and above the optic<br />
nerve was found. This read<strong>in</strong>g-evoked visual dimm<strong>in</strong>g may be a variant of gaze-evoked<br />
TVL. The optic nerve, displaced laterally and superiorly and stretched by the act of<br />
read<strong>in</strong>g, may have been compressed between the tumor and the contracted <strong>in</strong>ferior<br />
rectus muscle. Thus, orbital neuroimag<strong>in</strong>g is appropriate <strong>in</strong> patients with read<strong>in</strong>g<strong>in</strong>duced<br />
TVL.<br />
Intermittent angle closure glaucoma may cause TVL, and read<strong>in</strong>g-<strong>in</strong>duced TVL has<br />
been reported <strong>in</strong> one case. O’Sullivan et al described a 66-year-old patient with episodes<br />
of monocular TVL last<strong>in</strong>g 3 m<strong>in</strong>utes to several hours that were precipitated by read<strong>in</strong>g,<br />
writ<strong>in</strong>g, or watch<strong>in</strong>g television (O’Sullivan, 1995). Ophthalmologic exam was normal<br />
but read<strong>in</strong>g over 4 hours <strong>in</strong>duced corneal edema, a poorly reactive semidilated pupil,<br />
and a shallow anterior chamber with <strong>in</strong>traocular pressure of 50 mm Hg. The <strong>in</strong>termittent<br />
angle closure glaucoma and the patient’s symptoms were treated successfully<br />
by iridotomies.<br />
Do the Episodes of Monocular TVL Last<br />
Seconds?<br />
Transient Visual Loss 171<br />
Episodes of TVL last<strong>in</strong>g less than 60 seconds may occur <strong>in</strong> patients with papilledema<br />
(Wall, 1991). These transient obscurations of vision may occur <strong>in</strong> one or both eyes<br />
(<strong>in</strong>dividually or simultaneously) and typically last only a few seconds, though <strong>in</strong> rare<br />
cases they may last for hours. The episodes may be precipitated by changes <strong>in</strong> position,<br />
and are thought to be related to the effects of <strong>in</strong>creased <strong>in</strong>tracranial pressure on the flow<br />
of blood to the eye, perhaps where the central ret<strong>in</strong>al artery penetrates the optic nerve<br />
sheath to enter the substance of the nerve (Miller, 1991). Similar monocular TVL last<strong>in</strong>g<br />
seconds may occur <strong>in</strong> optic nerve sheath men<strong>in</strong>giomas unrelated to <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure. The pathogenesis of these episodes <strong>in</strong> men<strong>in</strong>gioma is unknown and<br />
may be caused by the effect of the men<strong>in</strong>gioma on the central ret<strong>in</strong>al artery where it<br />
enters the optic nerve (Miller, 1991). Transient obscurations of vision may also occur <strong>in</strong><br />
an eye with congenital abnormalities of the optic disc, such as peripapillary staphyloma<br />
(see below), or optic disc drusen. A case of ice-pick headaches associated with<br />
monocular visual loss with sc<strong>in</strong>tillat<strong>in</strong>g scotoma last<strong>in</strong>g seconds has been described<br />
<strong>in</strong> a patient with a history of migra<strong>in</strong>e with visual aura (Ammache, 2000). The patient<br />
was treated with oxygen <strong>in</strong>halation and <strong>in</strong>domethac<strong>in</strong> with complete resolution of the<br />
symptoms. F<strong>in</strong>ally, carotid atherosclerotic disease may rarely cause very brief episodes
172 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
of transient visual loss, but more often attacks of TVL with carotid disease last 2 to 15<br />
m<strong>in</strong>utes (see below).<br />
Patients with transient visual obscurations first require ophthalmologic exam<strong>in</strong>ation.<br />
If papilledema is evident (Chapter 7), these patients must have an MRI scan of the bra<strong>in</strong>.<br />
If this study is normal, a sp<strong>in</strong>al tap is <strong>in</strong>dicated to <strong>in</strong>vestigate the possibility of <strong>in</strong>fection<br />
or pseudotumor cerebri (idiopathic <strong>in</strong>tracranial hypertension). Patients with drusen or<br />
other optic disc anomalies caus<strong>in</strong>g monocular TVL may require no further evaluation. If<br />
there are signs of an optic neuropathy on the side of the TVL (e.g., relative afferent<br />
pupillary defect, ipsilateral swollen or atrophic optic nerve, etc.), then MRI with<br />
attention to the orbit is warranted to evaluate a compressive lesion. Patients without<br />
apparent disc abnormalities should be screened for carotid atherosclerotic disease or<br />
other sources of emboli (see below). In selected cases, MRI should be performed to<br />
<strong>in</strong>vestigate the possibility of a structural bra<strong>in</strong> lesion such as optic nerve sheath<br />
men<strong>in</strong>gioma.<br />
Do the Episodes of Monocular TVL Last<br />
M<strong>in</strong>utes?<br />
Monocular TVL last<strong>in</strong>g 5 to 60 m<strong>in</strong>utes (usually 2 to 30 m<strong>in</strong>utes) is strongly suggestive<br />
of thromboembolic disease. Ret<strong>in</strong>al emboli may arise from the aorta (Romano, 1998), the<br />
carotid artery, or the heart. Patients often describe the TVL as a veil or shade descend<strong>in</strong>g<br />
or ascend<strong>in</strong>g over a portion of their visual field. Other patients compla<strong>in</strong> of patchy<br />
visual loss (‘‘Swiss cheese’’ pattern) or peripheral constriction with central visual<br />
spar<strong>in</strong>g (Bruno, 1990). Some episodes of monocular TVL are accompanied by a<br />
sensation of color or other photopsias. These may superficially be similar to migra<strong>in</strong>e,<br />
consist<strong>in</strong>g of showers of stationary flecks of light that disperse quickly (Bruno, 1990;<br />
Goodw<strong>in</strong>, 1987; Pess<strong>in</strong>, 1977). Most episodes of embolic monocular TVL last 2 to 30<br />
m<strong>in</strong>utes. Marshall and Meadows found that <strong>in</strong> 51 of 67 patients (76%) episodes lasted 30<br />
m<strong>in</strong>utes or less, with 29 patients (43%) experienc<strong>in</strong>g episodes last<strong>in</strong>g 5 m<strong>in</strong>utes or less<br />
(Marshall, 1968). Pess<strong>in</strong> et al noted that attacks lasted less than 15 m<strong>in</strong>utes <strong>in</strong> 30 of 33<br />
patients, and <strong>in</strong> 14 patients (42%) the episodes lasted 5 m<strong>in</strong>utes or less (Pess<strong>in</strong>, 1977).<br />
Among 35 patients evaluated by Goodw<strong>in</strong> et al, 22 patients (63%) had attacks last<strong>in</strong>g 5<br />
m<strong>in</strong>utes or less, 8 (23%) had episodes last<strong>in</strong>g 6 to 15 m<strong>in</strong>utes, and 6 patients (17%) had<br />
episodes last<strong>in</strong>g more than 15 m<strong>in</strong>utes (Goodw<strong>in</strong>, 1987). Episodes of monocular TVL<br />
due to thromboembolic disease rarely last several hours.<br />
Patients with thromboembolic disease may demonstrate emboli with<strong>in</strong> the ret<strong>in</strong>al<br />
vessels. Emboli may be composed of clotted blood, fibr<strong>in</strong>, platelets, atheromatous tissue,<br />
white cells, calcium, <strong>in</strong>fectious organisms (septic emboli), air, fat, tumor cells, amniotic<br />
fluid, or foreign materials (e.g., talc, artificial valve material, catheters, silicone,<br />
cornstarch, mercury, corticosteroids). The most common types of emboli seen <strong>in</strong><br />
atherosclerotic disease of the aorta=carotid arteries or cardiac disease <strong>in</strong>clude the<br />
follow<strong>in</strong>g:<br />
1. Cholesterol emboli (Hollenhorst plaques) are bright, glisten<strong>in</strong>g, yellow or coppercolored<br />
fragments, most often seen <strong>in</strong> peripheral arterioles <strong>in</strong> the temporal fundus.<br />
These emboli most often arise from atheromatous plaques <strong>in</strong> the aorta or carotid<br />
bifurcation.
Transient Visual Loss 173<br />
2. Platelet-fibr<strong>in</strong> emboli are dull, white, gray, often elongated, and subject to fragmentation<br />
and distal movement. These emboli most often lodge at bifurcations of ret<strong>in</strong>al<br />
vessels and arise from the walls of atherosclerotic arteries or from the heart,<br />
especially from heart valves. They may also be seen <strong>in</strong> coagulopathies.<br />
3. Calcific emboli tend to be large, ovoid or rectangular, and chalky-white. These emboli<br />
often occur over or adjacent to the optic disc. They usually arise from cardiac (aortic<br />
or mitral) valves and less often from the aorta or carotid artery. Unlike cholesterol<br />
emboli, which often disappear <strong>in</strong> a few days, calcific emboli may rema<strong>in</strong> permanently<br />
visible.<br />
Sharma et al found the sensitivity and specificity of visible ret<strong>in</strong>al emboli for the<br />
detection of hemodynamically significant (def<strong>in</strong>ed as greater than or equal to 60%)<br />
carotid stenosis to be 39% and 68%, respectively, <strong>in</strong> patients with acute ret<strong>in</strong>al artery<br />
occlusion (Sharma, 1998). The presence of a visible embolus generated a likelihood ratio<br />
of 1.24, whereas the absence of a visible embolus generated a likelihood of 0.88. The<br />
authors concluded that the presence of a visible embolus is a poor diagnostic test for<br />
the detection of hemodynamically significant carotid artery stenosis <strong>in</strong> the sett<strong>in</strong>g of<br />
acute ret<strong>in</strong>al artery occlusion. Kle<strong>in</strong> et al described the prevalence at basel<strong>in</strong>e and the<br />
5-year <strong>in</strong>cidence of ret<strong>in</strong>al emboli <strong>in</strong> the Beaver Dam Study. They reported the<br />
associated risk factors, the relationship of ret<strong>in</strong>al emboli at basel<strong>in</strong>e to stroke, and<br />
ischemic heart disease mortality <strong>in</strong> these patients. The study consisted of 4,926 patients,<br />
aged 43 to 86 years at basel<strong>in</strong>e (Kle<strong>in</strong>, 1999). The prevalence of ret<strong>in</strong>al emboli at basel<strong>in</strong>e<br />
was 1.3% and the 5-year <strong>in</strong>cidence was 0.9%. The prevalence of ret<strong>in</strong>al emboli was<br />
associated with high pulse pressure, hypertension, diabetes mellitus, past and current<br />
smok<strong>in</strong>g, cardiovascular disease, and the presence of ret<strong>in</strong>opathy. Patients with ret<strong>in</strong>al<br />
emboli had a significantly higher risk of dy<strong>in</strong>g with stroke than those without ret<strong>in</strong>al<br />
emboli.<br />
TVL may also occur from ocular hypoperfusion rather than embolization. In some<br />
patients, monocular TVL may occur when the patient is exposed to bright light. These<br />
patients usually have severe, ipsilateral carotid occlusive disease. Bilateral, simultaneous<br />
TVL <strong>in</strong>duced by exposure to bright light may rarely occur with bilateral severe<br />
carotid stenosis or occlusion (Kaiboriboon, 2001). The light-<strong>in</strong>duced TVL probably<br />
reflects the <strong>in</strong>ability of a borderl<strong>in</strong>e ocular circulation to susta<strong>in</strong> the <strong>in</strong>creased ret<strong>in</strong>al<br />
metabolic activity associated with light exposure. Alternat<strong>in</strong>g transient visual loss to<br />
bright light has also been described with giant cell arteritis (Galetta, 1997).<br />
One prospective study assessed the cl<strong>in</strong>ical features of monocular TVL and the<br />
likelihood of atherosclerotic lesions of the <strong>in</strong>ternal carotid artery (ICA) (Donders, 2001).<br />
Of the 337 patients, 159 had a normal ICA on the relevant side, 33 had a stenosis of 0 to<br />
69%, 100 had a stenosis of 70 to 99%, and 45 had an ICA occlusion. <strong>An</strong> altitud<strong>in</strong>al onset<br />
or disappearance of symptoms was associated with atherosclerotic lesions of the<br />
ipsilateral ICA. A severe (70 to 99%) stenosis was also associated with duration of<br />
TVL between 1 and 10 m<strong>in</strong>utes, and with a speed of onset <strong>in</strong> seconds. <strong>An</strong> ICA occlusion<br />
was associated with attacks be<strong>in</strong>g provoked by light, an altitud<strong>in</strong>al character, and the<br />
occurrence of more than 10 attacks.<br />
TVL may also occur with carotid artery dissection. In a review of the cl<strong>in</strong>ical features<br />
of 146 patients with extracranial carotid artery dissection, 41 patients (28%) had<br />
monocular TVL. The TVL was pa<strong>in</strong>ful <strong>in</strong> 31 cases, associated with a Horner’s syndrome<br />
<strong>in</strong> 13 cases, and described as ‘‘sc<strong>in</strong>tillations’’ or ‘‘flash<strong>in</strong>g lights’’ (often related to
174 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
postural changes suggest<strong>in</strong>g choroidal hypoperfusion) <strong>in</strong> 23 cases (Biousse, 1998b). Two<br />
of 23 patients with spontaneous carotid artery dissection experienced transient monocular<br />
bl<strong>in</strong>dness; <strong>in</strong> one of these patients, episodes were provoked by sitt<strong>in</strong>g up from a<br />
sup<strong>in</strong>e position (Kerty, 1999).<br />
Postprandial transient visual loss has also been described (Lev<strong>in</strong>, 1997). In one<br />
patient, episodes of splotchy visual loss occurred unilaterally on the left 1 hour after<br />
eat<strong>in</strong>g her largest meal of the day. The episodes lasted approximately 3 hours and were<br />
occasionally accompanied by numbness and weakness of the contralateral arm. Severe<br />
left carotid stenosis was noted. In a second patient, blotchy bilateral transient visual loss<br />
episodes last<strong>in</strong>g 2 m<strong>in</strong>utes to 1.5 hours were precipitated by eat<strong>in</strong>g or stand<strong>in</strong>g from a<br />
sitt<strong>in</strong>g or ly<strong>in</strong>g position. This second patient was found to have complete occlusion of<br />
the right carotid artery and moderate stenosis of the left carotid artery. The authors<br />
proposed that postprandial visual loss may be a symptom of critical carotid stenosis,<br />
with ret<strong>in</strong>al and choroidal hypoperfusion probably caused by a comb<strong>in</strong>ation of mesenteric<br />
steal, decreased cardiac output, and abnormal vasomotor control (Lev<strong>in</strong>, 1997).<br />
Venous stasis ret<strong>in</strong>opathy (hypotensive ret<strong>in</strong>opathy), associated with severe carotid<br />
or ophthalmic artery occlusive disease, may also be associated with TVL (Gass, 1997).<br />
This syndrome is characterized by visual loss and ischemic ret<strong>in</strong>al <strong>in</strong>farction often<br />
accompanied by signs of ciliary artery obstruction, pallor of the disc, and hypotony.<br />
Venous stasis ret<strong>in</strong>opathy may simulate Purtscher’s ret<strong>in</strong>opathy (multifocal areas of<br />
ischemia) and be associated with a variety of fundus pictures (Gass, 1997):<br />
1. M<strong>in</strong>imal or no ophthalmoscopic changes <strong>in</strong> some patients with monocular TVL.<br />
2. Few widely scattered blot and dot hemorrhages and mild dilation of ret<strong>in</strong>al ve<strong>in</strong>s<br />
(venous stasis ret<strong>in</strong>opathy), usually <strong>in</strong> patients with m<strong>in</strong>imal visual compla<strong>in</strong>ts.<br />
3. Dilation of the ret<strong>in</strong>al arterial tree, dilation of the ret<strong>in</strong>al ve<strong>in</strong>s, and cotton-wool<br />
patches.<br />
4. Ret<strong>in</strong>al capillary changes, <strong>in</strong>clud<strong>in</strong>g microaneurysms, cystoid macular edema, and<br />
angiographic evidence of areas of capillary nonperfusion that may be conf<strong>in</strong>ed to the<br />
areas along the horizontal raphe.<br />
5. Larger areas of peripheral capillary nonperfusion, ret<strong>in</strong>al neovascularization, and<br />
hemorrhage.<br />
6. <strong>An</strong>y degree of branch ret<strong>in</strong>al ve<strong>in</strong> occlusion, branch ret<strong>in</strong>al ve<strong>in</strong> occlusion, branch<br />
ret<strong>in</strong>al artery occlusion, and central ret<strong>in</strong>al artery occlusion.<br />
7. Ischemic optic neuropathy.<br />
8. Fluoresce<strong>in</strong> angiography show<strong>in</strong>g diffuse ret<strong>in</strong>al capillary telangiectasia, delayed<br />
ret<strong>in</strong>al artery circulation time, late sta<strong>in</strong><strong>in</strong>g of the disc, and aggregations of microaneurysms<br />
around the preequatorial zone mimick<strong>in</strong>g idiopathic juxtafoveal ret<strong>in</strong>al<br />
telangiectasia.<br />
9. <strong>An</strong>y of the above associated with panuveitis, neovascular glaucoma, and a rapidly<br />
progress<strong>in</strong>g cataract (ocular ischemic syndrome).<br />
Venous stasis ret<strong>in</strong>opathy may be difficult to differentiate from central ret<strong>in</strong>al ve<strong>in</strong><br />
occlusion (CRVO). Helpful differentiat<strong>in</strong>g features <strong>in</strong>clude the follow<strong>in</strong>g:<br />
1. The ret<strong>in</strong>al ve<strong>in</strong>s are irregular <strong>in</strong> caliber with venous stasis ret<strong>in</strong>opathy.<br />
2. Hemorrhages, microaneurysms, and capillary dilations are often peripheral rather<br />
than <strong>in</strong> the posterior pole with venous stasis ret<strong>in</strong>opathy (with CRVO these changes<br />
are often diffuse rather than peripheral).
Transient Visual Loss 175<br />
3. Venous stasis ret<strong>in</strong>opathy is not associated with disc edema or optociliary ve<strong>in</strong>s<br />
(compared with CRVO).<br />
The ocular ischemic syndrome (Gass, 1997; Malhotra, 2000) is a progressive disorder<br />
due to hypoperfusion of eye that may be associated with TVL and ocular discomfort or<br />
frank pa<strong>in</strong> localized to the orbit and upper face that is often decreased when the patient<br />
lies down. Rubeosis iridis <strong>in</strong> an older nondiabetic patient without evidence of venous<br />
obstructive disease or other predispos<strong>in</strong>g cause is suggestive of the ocular ischemic<br />
syndrome. In persons over the age of 50 with new-onset iritis, the possibility of ocular<br />
ischemic syndrome should be considered. It is usually due to atherosclerotic carotid or<br />
ophthalmic artery disease. Other less common causes for venous stasis ret<strong>in</strong>opathy and<br />
the ocular ischemic syndrome <strong>in</strong>clude giant cell arteritis, carotid artery dissection,<br />
cavernous s<strong>in</strong>us thrombosis, Takayasu’s disease, fibromuscular dysplasia, mucormycosis,<br />
herpes zoster ophthalmicus, myelofibrosis, vasospasm, and postaneurysm repair<br />
(Borruat, 1993; Casson, 2001; Gupta, 1990; Hamed, 1992; Hwang, 1999; Lewis, 1993;<br />
Meire, 1991; W<strong>in</strong>terkorn, 1995; Zimmerman, 1995). Four of seven patients with<br />
maxillofacial arteriovenous malformations (AVMs) that had been treated previously<br />
with proximal ligation of the supply<strong>in</strong>g external carotid artery had signs of ocular<br />
ischemia (<strong>An</strong>dracchi, 2000). These four patients had significant ophthalmic artery<br />
supply by the malformations, suggest<strong>in</strong>g that when the ophthalmic arterial blood<br />
supply is recruited, an ophthalmic artery ‘‘steal’’ phenomenon occurs, caus<strong>in</strong>g ocular<br />
ischemia. This ‘‘steal’’ may be precipitated or worsened by previous surgical proximal<br />
ligation of external carotid arterial branches that are potential collaterals with the<br />
ophthalmic artery but fail to occlude the arteriovenous shunt.<br />
Giant cell arteritis (GCA) may produce attacks of TVL last<strong>in</strong>g m<strong>in</strong>utes to hours<br />
<strong>in</strong>dist<strong>in</strong>guishable from those produced by atheromatous disease (Hayreh, 1998a,b) (see<br />
Chapter 5). TVL probably results from <strong>in</strong>termittent <strong>in</strong>flammatory occlusion of the<br />
ophthalmic, posterior ciliary, or central ret<strong>in</strong>al arteries. A postural form of TVL has<br />
been described <strong>in</strong> giant cell arteritis and a tenuous optic disc perfusion (Wykes, 1984).<br />
Alternat<strong>in</strong>g monocular TVL may occur with GCA (F<strong>in</strong>elli, 1997) and may be <strong>in</strong>duced by<br />
bright light (Galetta, 1997).<br />
TVL may also occur <strong>in</strong> association with antiphospholipid antibodies, hyperviscosity<br />
and hypercoagulable states, polycythemia vera, systemic lupus erythematosus (SLE),<br />
and hepatitis C–associated type II cryoglobul<strong>in</strong>emia-mediated systemic vasculitis with<br />
mononeuritis multiplex. AVMs may divert blood flow from or reduce blood flow <strong>in</strong> the<br />
ophthalmic artery (ophthalmic steal syndrome) (Case Records of the MGH, 1999;<br />
Donders, 1998; Lev<strong>in</strong>e, 1990). The TVL may alternate from eye to eye. Donders et al<br />
noted that TVL occurred <strong>in</strong> 6% of patients with SLE (Donders, 1998). In patients with<br />
SLE, there was no relationship between TVL and the presence of antiphospholipid<br />
antibodies or livedo reticularis. Five of 10 patients with SLE had TVL <strong>in</strong> either eye<br />
(alternat<strong>in</strong>g TVL).<br />
Vasospasm, especially associated with migra<strong>in</strong>e, may also produce TVL without any<br />
of the visual phenomena typically seen dur<strong>in</strong>g a migra<strong>in</strong>e attack (Bernard, 1999; Booy,<br />
1990; Burger, 1991; O’Sullivan, 1992; W<strong>in</strong>terkorn, 1993). Vasospasm of the ret<strong>in</strong>al vessels<br />
has been documented by ophthalmoscopy dur<strong>in</strong>g some attacks of monocular TVL. TVL,<br />
likely due to vasospasm and migra<strong>in</strong>e, may be <strong>in</strong>duced by exercise or sexual <strong>in</strong>tercourse<br />
(Jehn, 2002; Teman, 1995). Exercise-<strong>in</strong>duced TVL may last m<strong>in</strong>utes to hours (Jehn, 2002).<br />
TVL <strong>in</strong> young <strong>in</strong>dividuals is often benign and related to migra<strong>in</strong>e. Tipp<strong>in</strong> et al reviewed
176 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
83 cases of TVL or ocular <strong>in</strong>farction before age 45 years. These authors found that<br />
cerebral transient ischemic attacks occurred <strong>in</strong> n<strong>in</strong>e patients but no case of stroke was<br />
found (Tipp<strong>in</strong>, 1989). Forty-one percent of the patients had headaches or orbital pa<strong>in</strong><br />
accompany<strong>in</strong>g their TVL spells and an additional 25.3% had severe headaches <strong>in</strong>dependent<br />
of the visual loss. Of the orig<strong>in</strong>al 83 patients, 42 were reexam<strong>in</strong>ed after a mean<br />
period of 5.8 years. None of the patients <strong>in</strong> this group had a stroke. The cl<strong>in</strong>ical status at<br />
follow-up did not correlate with duration of visual loss (TVL or ocular <strong>in</strong>farction),<br />
frequency (s<strong>in</strong>gle or recurrent episodes), gender, presence of headache or heart disease,<br />
cigarette smok<strong>in</strong>g, use of oral contraceptives, or abnormal f<strong>in</strong>d<strong>in</strong>gs on echocardiogram<br />
or blood studies. The authors concluded that TVL and ocular <strong>in</strong>farction occurr<strong>in</strong>g <strong>in</strong> the<br />
younger patient are probably associated with a more benign cl<strong>in</strong>ical course than that<br />
seen <strong>in</strong> older persons, and that migra<strong>in</strong>e is a likely cause for visual loss <strong>in</strong> a majority <strong>in</strong><br />
this group. O’Sullivan et al described n<strong>in</strong>e young adults (median age 19.5 years) who<br />
suffered from TVL (O’Sullivan, 1992). The attacks of TVL were short <strong>in</strong> duration and<br />
associated with premonitory symptoms <strong>in</strong> five patients and a migra<strong>in</strong>ous headache <strong>in</strong><br />
two. In five patients the visual loss progressed <strong>in</strong> a lacunar pattern (vision was lost <strong>in</strong> a<br />
series of blobs), unlike the ‘‘curta<strong>in</strong>’’ pattern characteristic of TVL <strong>in</strong> older patients.<br />
Investigation revealed no evidence of an embolic or atheromatous etiology. In two<br />
patients a m<strong>in</strong>or abnormality was found on echocardiography. The authors conclude<br />
that TVL <strong>in</strong> young adults has a different cl<strong>in</strong>ical pattern and may have a different<br />
etiology, possibly migra<strong>in</strong>e, compared with that seen <strong>in</strong> older patients. The pattern of<br />
visual loss <strong>in</strong> some of the cases suggests that the choroidal circulation rather than the<br />
ret<strong>in</strong>al circulation is primarily affected.<br />
TVL last<strong>in</strong>g 15 to 20 m<strong>in</strong>utes (occasionally up to 7 hours) may occur dur<strong>in</strong>g episodes<br />
of spontaneous anterior chamber hemorrhage (hyphema) (Kosmorsky, 1985; Miller,<br />
1991). In these patients TVL may be associated with erythropsia (see<strong>in</strong>g red) and color<br />
desaturation. Such hemorrhages are most likely to occur after cataract extraction and are<br />
particularly apt to occur after placement of an iris fixation lens implant. Other potential<br />
causes of spontaneous anterior chamber hemorrhages <strong>in</strong>clude vascular anomalies of the<br />
iris (e.g., <strong>in</strong> myotonic dystrophy or Sturge-Weber syndrome), microhemangiomas,<br />
diffuse hemangiomatosis of childhood, neoplasms (e.g., melanoma or ret<strong>in</strong>oblastoma),<br />
diseases of blood or vessels (e.g., leukemia, hemophilia, scurvy, lymphoma), rubeosis<br />
iridis, severe iritis, fibrovascular membranes, juvenile xanthogranuloma, occult trauma<br />
or delayed bleed<strong>in</strong>g after trauma, hydro-ophthalmos, malignant exophthalmos, histiocytosis<br />
X, and postsclerotomy with cautery (Kosmorsky, 1985). Episodes of TVL last<strong>in</strong>g<br />
up to 24 hours have been described with recurrent hyphema after deep sclerotomy with<br />
collagen implant (DSCI) (Ambres<strong>in</strong>, 2001). The uveitis-glaucoma-hyphema (UGH)<br />
syndrome is an unusual cause of monocular TVL follow<strong>in</strong>g cataract extraction and<br />
<strong>in</strong>traocular lens implantation (Cates, 1998). Patients may present with the full triad or<br />
with its <strong>in</strong>dividual elements, with symptoms often develop<strong>in</strong>g at an <strong>in</strong>terval, often<br />
years, after cataract surgery. Table 8–1 compares the symptoms of TVL <strong>in</strong> ret<strong>in</strong>al emboli<br />
compared with the UGH syndrome (Cates, 1998).<br />
Intermittent angle closure glaucoma may also cause brief episodes of monocular TVL<br />
that are usually, though not always, associated with ipsilateral eye pa<strong>in</strong> and occasionally<br />
simultaneous dilation of the pupil (Miller, 1991). Exercise-<strong>in</strong>duced visual disturbances<br />
may also occur dur<strong>in</strong>g attacks of pigmentary glaucoma (Jehn, 2002). Episodes of<br />
monocular TVL last<strong>in</strong>g 2 to 3 m<strong>in</strong>utes <strong>in</strong>duced by changes <strong>in</strong> posture have been<br />
described follow<strong>in</strong>g scleral buckle procedure, likely due to <strong>in</strong>termittent obstruction of
Transient Visual Loss 177<br />
Table 8–1. Comparison Between the Classic Symptoms of Visual Loss <strong>in</strong> Patients with<br />
Transient Visual Loss (TVL) Due to Ret<strong>in</strong>al Emboli and the Uveitis-Glaucoma-Hyphema<br />
(UGH) Syndrome<br />
TVL Due to Ret<strong>in</strong>al Embolus TVL Due to UGH Syndrome<br />
Speed of onset Sudden (seconds) Gradual (m<strong>in</strong>utes)<br />
Recovery Rapid (seconds to m<strong>in</strong>utes) Slow (hours to days)<br />
Character Dark curta<strong>in</strong> over vision Gradual mist<strong>in</strong>g of vision<br />
Erythropsia (red vision)<br />
Location Sector loss Diffuse<br />
Pa<strong>in</strong> None With or without ache <strong>in</strong> affected eye<br />
the central ret<strong>in</strong>al artery blood flow by the encircl<strong>in</strong>g element (F<strong>in</strong>eman, 1999). F<strong>in</strong>ally,<br />
TVL may also be associated with the congenital anomalies, peripapillary staphyloma,<br />
and morn<strong>in</strong>g glory syndrome (Ebner, 1995; Gass, 1997; Zarnegar, 1995). Episodes of<br />
TVL with these anomalies may last 15 to 20 seconds (obscurations of vision) or up to 20<br />
m<strong>in</strong>utes, the latter mimick<strong>in</strong>g TVL with thromboembolic disease. The episodes of TVL<br />
<strong>in</strong> peripapillary staphyloma may be associated with <strong>in</strong>termittent dilation of the ret<strong>in</strong>al<br />
ve<strong>in</strong>s and may be orthostatic.<br />
Patients with monocular TVL last<strong>in</strong>g m<strong>in</strong>utes associated with visible ret<strong>in</strong>al emboli<br />
need to be evaluated for carotid and aortic vascular disease and cardiac valvular<br />
disease. Stroke risk factors (e.g., smok<strong>in</strong>g, hypertension, diabetes mellitus, hyperlipidemia,<br />
etc.) should be evaluated and controlled. Studies to evaluate the carotid arteries<br />
<strong>in</strong>clude carotid Doppler and ultrasound. Some patients may require MR angiography<br />
and conventional angiography. Cardiac <strong>in</strong>vestigations <strong>in</strong>clude transthoracic and transesophageal<br />
echocardiography and cardiac MRI. In a study of 18 patients with branch or<br />
central ret<strong>in</strong>al artery occlusion, transesophageal echocardiogram revealed a possible<br />
cardiac or thoracic source of embolus <strong>in</strong> 13 patients (72%), whereas a potential carotid<br />
source of embolus was present <strong>in</strong> three of 16 patients (19%) (Kramer, 2001).<br />
Hurwitz et al performed a prospective cl<strong>in</strong>ical and arteriographic study compar<strong>in</strong>g<br />
patients with monocular TVL and patients with other transient hemispheral cerebral<br />
ischemic attacks (Hurwitz, 1985). In their 93 patients with monocular TVL, a potentially<br />
operable atherosclerotic carotid lesion (def<strong>in</strong>ed as 5 50% stenosis or ulceration on the<br />
side of TVL) was found <strong>in</strong> 66% of the patients, and the 7-year cumulative rate of cerebral<br />
<strong>in</strong>farction <strong>in</strong> these patients was 14%. In 212 patients with other hemispheric transient<br />
ischemic attacks, an operable carotid lesion was found <strong>in</strong> 51% of patients, with the<br />
7-year cumulative rate of <strong>in</strong>farction 27%. Therefore, <strong>in</strong> approximately two thirds of<br />
patients with monocular TVL, a potentially operable carotid lesion may be found.<br />
In patients with monocular TVL (or other carotid distribution transient ischemic<br />
attacks or nondisabl<strong>in</strong>g stroke) and ipsilateral carotid stenosis of 70 to 99%, carotid<br />
endarterectomy may be <strong>in</strong>dicated. Surgery may be recommended <strong>in</strong> this sett<strong>in</strong>g if the<br />
patient is a good surgical candidate and the perioperative morbidity and mortality of<br />
the surgeon is <strong>in</strong> the 2% or less range (North American Symptomatic Carotid<br />
Endarterectomy Trial Collaborators, 1991). Carotid endarterectomy <strong>in</strong> this group<br />
reduces the 2-year ipsilateral stroke rate from 26 to 9%, and decreases the major or<br />
fatal ipsilateral stroke rate from 13.1 to 2.5%. The benefit of surgery <strong>in</strong> the 70% or greater
178 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
stenosis patients is greatest among men, <strong>in</strong> patients with a recent stroke as a qualify<strong>in</strong>g<br />
event, and <strong>in</strong> patients with hemispheric (versus visual) symptoms (Barnett, 1998). The<br />
benefit of surgery is twice as great for patients with 90 to 99% carotid stenosis versus 70<br />
to 79% stenosis. The frequency of major functional impairment was much lower <strong>in</strong> the<br />
surgical group than <strong>in</strong> the medical group (Haynes, 1994). In patients with monocular<br />
TVL, other carotid distribution transient ischemic attack or nondisabl<strong>in</strong>g stroke, and 50<br />
to 69% ipsilateral carotid stenosis, the 5-year rate of any ipsilateral stroke was 15.7% <strong>in</strong><br />
the surgical group and 22.2% <strong>in</strong> the medical group (Barnett, 1998). Among patients<br />
with < 50% stenosis, the stroke rate was not significantly lower <strong>in</strong> the surgery group<br />
(14.9%) than <strong>in</strong> the medical group (18.7%). Therefore, carotid endarterectomy <strong>in</strong> patients<br />
with symptomatic carotid stenosis of 50 to 69% yields only moderate reduction <strong>in</strong> stroke<br />
risk, with the absolute risk reduction be<strong>in</strong>g about 10% at 5 years. Among patients with<br />
<strong>in</strong>ternal carotid artery stenosis, the prognosis is better for those present<strong>in</strong>g with<br />
transient monocular bl<strong>in</strong>dness than for those present<strong>in</strong>g with hemispheric transient<br />
ischemic attacks (Benavente, 2001). Decisions about treatment must take <strong>in</strong>to account<br />
the recognized risk patient factors, and the surgical perioperative complication rates<br />
must be 6% or less. In the patient with less than 50% carotid stenosis, a cardiac or aortic<br />
embolic source should be sought and, if none is found, the treatment is aspir<strong>in</strong> plus<br />
control of stroke risk factors. In patients with emboli from a cardiac valvular source,<br />
especially those with cardiac dysrhythmias such as atrial fibrillation, anticoagulation is<br />
warranted if the patient is an appropriate medical candidate. Patients older than 55<br />
years with a history of monocular TVL last<strong>in</strong>g m<strong>in</strong>utes without visible ret<strong>in</strong>al emboli<br />
should have an evaluation for giant cell arteritis (e.g., erythrocyte sedimentation rate,<br />
temporal artery biopsy) (class II, level C).<br />
Patients with evidence of monocular TVL result<strong>in</strong>g from ocular hypoperfusion (e.g.,<br />
venous stasis ret<strong>in</strong>opathy and the ocular ischemic syndrome) might have decreased<br />
ret<strong>in</strong>al artery pressure on ophthalmodynamometry. The patient should be <strong>in</strong>vestigated<br />
for carotid stenosis and, if this is <strong>in</strong>significant, ophthalmic artery stenosis or occlusion is<br />
<strong>in</strong>ferred. When carotid stenosis is severe, endarterectomy may be used to reestablish<br />
flow (Kawaguchi, 2001; Rennie, 2002); when the <strong>in</strong>ternal carotid artery is totally<br />
occluded, a superficial temporal artery to middle cerebral artery bypass procedure<br />
may be considered if the external carotid is patent (Kawaguchi, 1999). With early<br />
treatment, resolution of the hypoperfusion syndrome may occur; unfortunately, no<br />
therapy is clearly effective. In one study, carotid endarterectomy was effective for<br />
improv<strong>in</strong>g or prevent<strong>in</strong>g the progress of chronic ocular ischemia caused by <strong>in</strong>ternal<br />
carotid stenosis; visual acuity improved <strong>in</strong> 5 of 11 patients and had not worsened <strong>in</strong> the<br />
other 6 (Kawaguchi, 2001). Reestablishment of flow <strong>in</strong> a previously stenotic <strong>in</strong>ternal<br />
carotid artery may actually produce further visual difficulties by <strong>in</strong>creas<strong>in</strong>g perfusion to<br />
the ciliary arteries and caus<strong>in</strong>g dramatic <strong>in</strong>crease <strong>in</strong> <strong>in</strong>traocular pressure. Carotid<br />
endarterectomy or superficial temporal artery to middle cerebral artery bypass procedure<br />
have been comb<strong>in</strong>ed with laser panret<strong>in</strong>al photocoagulation, peripheral ret<strong>in</strong>al<br />
cryotherapy, or both. These latter procedures are thought to decrease the oxygen<br />
requirement of the eye and thus reduce the drive for neovascularization. Rarely, the<br />
ocular ischemic syndrome may be improved by the calcium channel blocker, verapamil<br />
(W<strong>in</strong>terkorn, 1995).<br />
If no thromboembolic source for the episodes of TVL is documented, then further<br />
studies should be considered. These <strong>in</strong>clude MRI of the bra<strong>in</strong> with MR angiography to<br />
<strong>in</strong>vestigate for possible bra<strong>in</strong> ischemia or less likely a vascular malformation, and
laboratory studies, <strong>in</strong>clud<strong>in</strong>g sedimentation rate, complete blood count, antiphospholipid<br />
antibodies, ant<strong>in</strong>uclear antibodies, collagen vascular disease profile, and studies to<br />
<strong>in</strong>vestigate the presence of dysprote<strong>in</strong>emia (class III–IV, level U).<br />
Young patients (< 45 years old) with monocular TVL are unlikely to have significant<br />
carotid disease. A cardiac embolic source as well as a vasculitis or coagulopathy must be<br />
sought. As noted above, monocular TVL <strong>in</strong> younger patients has a more benign cl<strong>in</strong>ical<br />
course than that found <strong>in</strong> an older population, and migra<strong>in</strong>e is a likely cause for many<br />
episodes. Calcium channel blockers (e.g., verapamil or nifedip<strong>in</strong>e), if not otherwise<br />
contra<strong>in</strong>dicated, may be considered <strong>in</strong> some of these patients to reduce the frequency of<br />
episodes of TVL (Teman, 1995; W<strong>in</strong>terkorn, 1993).<br />
F<strong>in</strong>ally, all patients with monocular TVL last<strong>in</strong>g m<strong>in</strong>utes should have a complete<br />
ophthalmoscopic exam<strong>in</strong>ation to <strong>in</strong>vestigate such conditions as <strong>in</strong>termittent angle<br />
closure glaucoma, morn<strong>in</strong>g glory syndrome, and peripapillary staphyloma. Spontaneous<br />
anterior chamber hemorrhage (hyphema) should also be considered, especially <strong>in</strong><br />
patients with associated erythropsia and <strong>in</strong> those who have undergone cataract<br />
extraction.<br />
Episodes of monocular TVL last<strong>in</strong>g hours are rare. However, such spells may occur<br />
with thromboembolic disease, as a postprandial phenomenon associated with critical<br />
carotid stenosis, and with migra<strong>in</strong>e. Monocular TVL last<strong>in</strong>g hours may be a symptom of<br />
impend<strong>in</strong>g central ret<strong>in</strong>al ve<strong>in</strong> occlusion (Biousse, 1997).<br />
<strong>An</strong> approach to the evaluation of patients with monocular TVL is presented <strong>in</strong><br />
Figure 8–1.<br />
Are the Episodes of TVL B<strong>in</strong>ocular?<br />
Transient Visual Loss 179<br />
Transient visual obscurations last<strong>in</strong>g seconds may occur <strong>in</strong> one or both eyes <strong>in</strong> patients<br />
with <strong>in</strong>creased <strong>in</strong>tracranial pressure and papilledema. Also, patients with bilateral<br />
severe carotid occlusive disease may rarely have bilateral TVL on exposure to bright<br />
light. Otherwise, episodes of bilateral simultaneous TVL are usually due to migra<strong>in</strong>e,<br />
bilateral occipital lobe ischemia (e.g., vertebrobasilar <strong>in</strong>sufficiency), or other occipital<br />
lesions.<br />
The presence of a small area of visual loss or a mild disturbance of vision that<br />
progressively <strong>in</strong>creases over 15 m<strong>in</strong>utes or longer (march and buildup) is highly<br />
characteristic of migra<strong>in</strong>e (Russell, 1996). This visual abnormality is usually bilateral<br />
and homonymous. The patient need not have headaches for this diagnosis to be made.<br />
Most patients describe abnormal positive visual symptoms associated with the<br />
episodes. Most commonly, fortification spectra are described around an area of scotoma.<br />
These sc<strong>in</strong>tillations or distortions with<strong>in</strong> the area of visual disturbance may resemble<br />
‘‘heat waves’’ or ‘‘water runn<strong>in</strong>g down a glass.’’ The typical migra<strong>in</strong>e visual aura starts<br />
as a flicker<strong>in</strong>g, uncolored, zigzag l<strong>in</strong>e <strong>in</strong> the center of the visual field that gradually<br />
progresses and expands toward the periphery of one hemifield and often leaves a<br />
temporary scotoma (Fisher, 1999). A migra<strong>in</strong>ous visual accompaniment often occurs <strong>in</strong><br />
<strong>in</strong>dividuals over age 50 and often occurs <strong>in</strong> the absence of headache <strong>in</strong> this age group<br />
(Wijman, 1998). These episodes probably are not associated with an <strong>in</strong>creased stroke<br />
risk. The spells are usually stereotyped, beg<strong>in</strong> gradually, and progress, last several<br />
m<strong>in</strong>utes to 1 hour, and usually <strong>in</strong>clude positive visual phenomena (bright images,<br />
colors, movement of images) and affect both eyes. In a study by Wijman et al, the
180 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
migra<strong>in</strong>ous visual accompaniments were never accompanied by headache <strong>in</strong> 58% of<br />
patients, and 42% of <strong>in</strong>dividuals had no history of recurrent headache (Wijman, 1998).<br />
The risk of stroke <strong>in</strong> these patients was 11.5%, significantly less than the 33.3% noted <strong>in</strong><br />
patients with transient ischemic attacks but not significantly different from the rate of<br />
13.6% <strong>in</strong> those with neither migra<strong>in</strong>ous accompaniments nor transient ischemic attacks.<br />
Associated symptoms may <strong>in</strong>clude nausea, aphasia, eye pa<strong>in</strong>, diplopia, dizz<strong>in</strong>ess,<br />
t<strong>in</strong>nitus, numbness, and paresthesias.<br />
Rarely, the positive visual phenomena of migra<strong>in</strong>e may persist for months to years,<br />
unassociated with electroencephalographic or MRI f<strong>in</strong>d<strong>in</strong>gs (Liu, 1995). Patients with<br />
persistent migra<strong>in</strong>ous visual phenomena (migra<strong>in</strong>e aura status) may demonstrate<br />
occipital hypoperfusion on bra<strong>in</strong> s<strong>in</strong>gle photon emission tomography (SPECT) (Chen,<br />
2001; Luda, 1991). This persistent migra<strong>in</strong>ous phenomena may be responsive to<br />
lamotrig<strong>in</strong>e (Chen, 2001). In another form of migra<strong>in</strong>e aura status, patients may<br />
experience a large number of consecutive (mostly) visual auras, very often without<br />
headache. Between the auras, the patient is without symptoms. Episodes can last for<br />
weeks, and with<strong>in</strong> this period several migra<strong>in</strong>e auras can occur on one day. Haan et al<br />
described three such patients with migra<strong>in</strong>e aura status treated successfully with<br />
acetazolamide (Haan, 2000).<br />
Abnormal visual disturbances similar to those with migra<strong>in</strong>e, often associated with<br />
headache, may rarely occur with cerebral structural lesions, such as AVMs of the<br />
occipital lobe or bra<strong>in</strong> tumors, but these usually do not have the characteristic buildup<br />
and resolution of visual symptoms. Instead, these lesions usually produce symptoms<br />
that steadily <strong>in</strong>crease <strong>in</strong> frequency and duration until they are present daily.<br />
Occipital lobe tumors may rarely produce sc<strong>in</strong>tillat<strong>in</strong>g scotomas that mimic migra<strong>in</strong>e<br />
(Biousse, 1998a; Miller, 1991; Pep<strong>in</strong>, 1990; Riaz, 1991). In most of these cases, the tumors<br />
were diagnosed only after the patients eventually developed papilledema or when a<br />
homonymous visual field defect was documented. Riaz et al described three patients<br />
with classical migra<strong>in</strong>e for many years’ duration that preceded the diagnosis of<br />
men<strong>in</strong>gioma (Riaz, 1991). In two patients, the tumors were occipital and <strong>in</strong> one<br />
frontotemporal. Visual symptoms <strong>in</strong> two of these patients were exceptional by their<br />
constant localization to the same hemianopic field, whereas <strong>in</strong> the third patient they<br />
<strong>in</strong>volved either hemianopic field. The visual phenomena sometimes occurred <strong>in</strong>dependent<br />
of headache.<br />
Arteriovenous malformations of the occipital lobes may also produce visual symptoms<br />
and headache that may simulate migra<strong>in</strong>e (Haas, 1991; Kupersmith, 1996, 1999;<br />
Kurita, 2000; Spier<strong>in</strong>gs, 2001). Visual symptoms with occipital AVMs are usually brief,<br />
episodic, unformed, and not associated with the angular, sc<strong>in</strong>tillat<strong>in</strong>g figures that occur<br />
with migra<strong>in</strong>e. They also tend to occur consistently <strong>in</strong> the same visual field. However,<br />
the cl<strong>in</strong>ical symptoms classically noted with migra<strong>in</strong>e may occasionally occur with<br />
occipital AVMs. Kupersmith et al described the cl<strong>in</strong>ical presentations of 70 patients with<br />
occipital AVMs (Kupersmith, 1996). At the time of presentation, headache was present<br />
<strong>in</strong> 39 (56%); the headache was throbb<strong>in</strong>g <strong>in</strong> 19 cases (27%) with preced<strong>in</strong>g homonymous<br />
positive visual phenomena with migra<strong>in</strong>e-like features <strong>in</strong> the field contralateral to the<br />
AVM <strong>in</strong> 15 cases. A visual disturbance <strong>in</strong> the opposite field, not necessarily associated<br />
with headache, occurred <strong>in</strong> 39 patients (56%). Patients often described episodes of<br />
sc<strong>in</strong>tillat<strong>in</strong>g scotomas, jagged flicker<strong>in</strong>g fortification images, transient and permanent<br />
homonymous hemianopia, blurred vision <strong>in</strong> a hemifield, hemifield spots, tunnel vision,<br />
and diplopia. Three patients had transient field loss as a prodrome to grand mal
Transient Visual Loss 181<br />
seizures and two others had episodes of flicker<strong>in</strong>g vision associated with seizure<br />
activity on electroencephalography. Only 5 of the 23 patients with visual symptoms<br />
who had a homonymous field defect did not have recurrent headaches. Fifteen<br />
additional patients without visual symptoms, 8 of whom had no recurrent headaches,<br />
had homonymous visual field defects. The authors concluded that if ‘‘migra<strong>in</strong>e’’<br />
headache or visual symptoms are restricted to one side of the head (even if the visual<br />
field exam is normal), then a neuroimag<strong>in</strong>g study should be performed to <strong>in</strong>vestigate<br />
the possibility of an occipital AVM. Migra<strong>in</strong>e <strong>in</strong> this sett<strong>in</strong>g is a diagnosis of exclusion.<br />
Whereas some features of headache and visual symptoms are similar for occipital AVMs<br />
and migra<strong>in</strong>e, the two disorders are usually dist<strong>in</strong>guishable. Kurita and Sh<strong>in</strong> described<br />
a man with periodic right-sided throbb<strong>in</strong>g headaches heralded by a visual prodrome of<br />
sc<strong>in</strong>tillat<strong>in</strong>g bright lights <strong>in</strong> the left visual field last<strong>in</strong>g several m<strong>in</strong>utes (Kurita, 2000).<br />
The headaches decreased 18 months after radiosurgery for a right occipital AVM.<br />
Positive visual phenomena resembl<strong>in</strong>g migra<strong>in</strong>e have also been described with cerebral<br />
venous s<strong>in</strong>us thrombosis (Newman, 1989). F<strong>in</strong>ally, sc<strong>in</strong>tillat<strong>in</strong>g scotomas occasionally<br />
occur <strong>in</strong> patients with SLE, but it is not clear if they are a manifestation of a<br />
cerebrovascular disorder related to lupus or simply the coexistence of two separate<br />
disease processes (Miller, 1991).<br />
Panayiotopoulos et al described n<strong>in</strong>e patients with idiopathic occipital epilepsy and<br />
visual seizures (Panayiotopoulos, 1999). The ictal elementary visual halluc<strong>in</strong>ations were<br />
stereotyped for each patient, usually last<strong>in</strong>g seconds. They consisted of ma<strong>in</strong>ly multiple,<br />
bright colored, small circular spots, circles, or balls. Mostly, they appeared <strong>in</strong> a temporal<br />
hemifield, often mov<strong>in</strong>g contralaterally or <strong>in</strong> the center, where they may be flash<strong>in</strong>g.<br />
They may be multiple and <strong>in</strong>crease <strong>in</strong> size <strong>in</strong> the course of a seizure and may progress to<br />
extraoccipital manifestations and convulsions. Bl<strong>in</strong>dness occurred usually from the<br />
beg<strong>in</strong>n<strong>in</strong>g and postictal headache, often <strong>in</strong>dist<strong>in</strong>guishable from migra<strong>in</strong>e, was common.<br />
Three of n<strong>in</strong>e patients had ictal bl<strong>in</strong>dness as the only seizure manifestation. Most<br />
patients responded to carbamazep<strong>in</strong>e. Elementary visual halluc<strong>in</strong>ations <strong>in</strong> occipital<br />
seizures are entirely different from the visual aura of migra<strong>in</strong>e. They are ma<strong>in</strong>ly colored,<br />
have a circular pattern, have the same onset regard<strong>in</strong>g localization, are often brief<br />
(last<strong>in</strong>g seconds, occasionally m<strong>in</strong>utes), develop rapidly, and then <strong>in</strong>dividual components<br />
may multiply or move together to the contralateral side. They often occur daily<br />
and may be associated with other seizure manifestations. Conversely, the visual aura of<br />
migra<strong>in</strong>e start with predom<strong>in</strong>antly flicker<strong>in</strong>g achromatic or black and white (rarely<br />
colored) l<strong>in</strong>ear and zigzag patterns <strong>in</strong> the center of vision that gradually expand over<br />
m<strong>in</strong>utes toward the periphery of one hemifield and often leave a scotoma. Migra<strong>in</strong>e<br />
rarely occurs daily.<br />
Dreier et al described two patients with migra<strong>in</strong>e who experienced migra<strong>in</strong>ous auralike<br />
symptoms several m<strong>in</strong>utes after the onset of acute headache <strong>in</strong>duced by subarachnoid<br />
hemorrhage (Dreier, 2001). The cases suggest that subarachnoid hemorrhage<br />
is a trigger for migra<strong>in</strong>ous aura.<br />
Symptoms similar to the sc<strong>in</strong>tillat<strong>in</strong>g scotomas of migra<strong>in</strong>e may also occur with acute<br />
vitreous or ret<strong>in</strong>al detachment (Miller, 1991). In these patients, the visual symptoms are<br />
clearly monocular, last longer than typical migra<strong>in</strong>ous visual aura, and occur without<br />
any associated headache. Sc<strong>in</strong>tillat<strong>in</strong>g scotomas, as well as monocular TVLs, have also<br />
been described associated with <strong>in</strong>ternal carotid artery dissection (Biousse, 1998b;<br />
Ramadan, 1991). The first of the three patients described by Ramadan et al developed<br />
sudden severe right occipital headache followed m<strong>in</strong>utes later by nausea and bright
182 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
dots <strong>in</strong> both visual fields that spread centrifugally dur<strong>in</strong>g a 10-m<strong>in</strong>ute period and<br />
persisted for several hours (Ramadan, 1991). The second perceived sc<strong>in</strong>tillat<strong>in</strong>g and<br />
nonmarch<strong>in</strong>g ‘‘snowflakes’’ <strong>in</strong> the entire visual field of the right eye that lasted 10<br />
m<strong>in</strong>utes, dur<strong>in</strong>g which time the right eye lost vision. This was followed by right<br />
frontotemporal sharp pa<strong>in</strong> that lasted for another hour. The third patient noted the<br />
abrupt onset of see<strong>in</strong>g stationary, sharp-edged gray shapes (triangles, squares, and<br />
zigzag l<strong>in</strong>es), outl<strong>in</strong>ed <strong>in</strong> bright red and blue and superimposed on a glar<strong>in</strong>g background.<br />
These positive visual phenomena were perceived <strong>in</strong> the left eye and lasted for 3<br />
days. She later developed another episode of visual phenomena <strong>in</strong> the left eye<br />
associated with left supraorbital and temporal throbb<strong>in</strong>g headache. The first patient’s<br />
episode was b<strong>in</strong>ocular but atypical for classic migra<strong>in</strong>e <strong>in</strong> that the positive visual<br />
phenomena lasted for hours; <strong>in</strong> the other two patients the symptoms were monocular,<br />
and <strong>in</strong> one of these the positive symptoms lasted for days, aga<strong>in</strong> atypical features for<br />
classic migra<strong>in</strong>e. As noted above, <strong>in</strong> a study of 146 patients with extracranial carotid<br />
artery dissection, 41 patients (28%) had transient monocular visual loss that was<br />
described as ‘‘sc<strong>in</strong>tillations’’ or ‘‘flash<strong>in</strong>g lights’’ (often related to postural changes<br />
suggest<strong>in</strong>g choroidal hypoperfusion) <strong>in</strong> 23 cases (Biousse, 1998b).<br />
Patients with restrictive thyroid ophthalmopathy may occasionally compla<strong>in</strong> of<br />
flash<strong>in</strong>g lights <strong>in</strong> the superior visual field on upgaze, possibly phosphenes as a result<br />
of either compression of the globe by a tight <strong>in</strong>ferior rectus muscle or traction on the<br />
<strong>in</strong>sertion of the <strong>in</strong>ferior rectus muscle (Danks, 1998). Twelve of 30 patients with thyroid<br />
ophthalmopathy had flash<strong>in</strong>g lights on upward gaze and all had tight <strong>in</strong>ferior rectus<br />
muscles (Danks, 1998).<br />
B<strong>in</strong>ocular episodes of TVL may be due to bilateral occipital ischemia secondary to<br />
disease of the vertebrobasilar circulation (rarely bilateral ret<strong>in</strong>al ischemia from systemic<br />
hypotension or bilateral carotid disease). Episodes of visual loss or blurr<strong>in</strong>g <strong>in</strong> patients<br />
with vertebrobasilar transient ischemic attacks (TIAs) usually occur <strong>in</strong> association with<br />
other symptoms of transient bra<strong>in</strong>stem, cerebellar, or posterior cerebral ischemia,<br />
<strong>in</strong>clud<strong>in</strong>g vertigo, dysarthria, dysphagia, diplopia, weakness, sensory disturbances<br />
(especially perioral numbness), coord<strong>in</strong>ation difficulties, and gait <strong>in</strong>stability. Visual<br />
loss or blurr<strong>in</strong>g of vision <strong>in</strong> these patients is bilateral and symmetric, may be<br />
hemianopic or diffuse, and usually lasts several m<strong>in</strong>utes or occasionally less than a<br />
m<strong>in</strong>ute (but not seconds, as noted with obscurations of vision noted with papilledema<br />
and <strong>in</strong>creased <strong>in</strong>tracranial pressure). The sc<strong>in</strong>tillat<strong>in</strong>g and expand<strong>in</strong>g scotomas of<br />
migra<strong>in</strong>e rarely occur with vertebrobasilar TIAs, and migra<strong>in</strong>ous visual phenomena<br />
usually last 20 to 30 m<strong>in</strong>utes, somewhat longer than visual loss noted with vertebrobasilar<br />
TIAs. Also, Hilton-Jones et al described a patient with a large frontal lobe tumor<br />
who experienced frequent, stereotyped episodes of bilateral, simultaneous visual loss<br />
last<strong>in</strong>g 5 to 30 m<strong>in</strong>utes (Hilton-Jones, 1982). This patient reportedly did not have<br />
papilledema.<br />
Other unusual causes of transient bilateral visual loss should be mentioned. For<br />
example, transient bilateral bl<strong>in</strong>dness last<strong>in</strong>g m<strong>in</strong>utes to hours may rarely occur with<br />
giant cell arteritis, due to either vertebrobasilar <strong>in</strong>sufficiency or bilateral impend<strong>in</strong>g<br />
anterior ischemic optic neuropathy (Diego, 1998). Bilateral blurred vision last<strong>in</strong>g<br />
m<strong>in</strong>utes to several hours dur<strong>in</strong>g sexual arousal may be associated with narrow-angle<br />
glaucoma (Friedberg, 1999). As noted above, transient bilateral bl<strong>in</strong>dness may be the<br />
sole manifestation of occipital epilepsy (Panayiotopoulos, 1999). In fact, prolonged (48<br />
hour) visual loss may occur with occipital seizures (status epilepticus amauroticus)
(Sawchuk, 1997). Transient bilateral cortical bl<strong>in</strong>dness last<strong>in</strong>g 24 hours has been<br />
described with preeclampsia (Kesler, 1998), and transient cortical bl<strong>in</strong>dness last<strong>in</strong>g<br />
hours, days, or even several weeks may occur after cerebral angiography (Gibson, 1982).<br />
Temporary bilateral bl<strong>in</strong>dness (pupils normal or nonreactive) may occur with irritability,<br />
confusion, bradycardia, nausea, hypertension, dyspnea, and seizures dur<strong>in</strong>g or<br />
after transurethral prostatic resection (TURP) (Barletta, 1994). This TURP syndrome is<br />
thought due to excessive absorption of nonelectrolyte irrigat<strong>in</strong>g fluid through the<br />
prostatic venous s<strong>in</strong>uses <strong>in</strong>to the general circulation. Glyc<strong>in</strong>e toxicity on the optic<br />
nerves or cortex, due to excessive glyc<strong>in</strong>e absorption, is the likely mechanism of visual<br />
loss. The symptoms and signs of the TURP syndrome resolve with<strong>in</strong> 24 hours with<br />
<strong>in</strong>travenous pyridox<strong>in</strong>e and arg<strong>in</strong><strong>in</strong>e hydrochloride.<br />
Bilateral TVL last<strong>in</strong>g several weeks may occur with the reversible posterior leukoencephalopathy<br />
due to immunosuppressive therapy (cyclospor<strong>in</strong>e or tacrolimus) after<br />
transplantation. Patients on <strong>in</strong>terferon-a for myeloma or <strong>in</strong>terleuk<strong>in</strong>-2 therapy for<br />
malignancy or HIV disease may develop TVL. TVL may occur <strong>in</strong> eclampsia, acute<br />
hypertensive encephalopathy associated with renal disease, or acute <strong>in</strong>termittent<br />
porphyria (H<strong>in</strong>chey, 1996; Karp, 1996; Kuperschmidt, 1995).<br />
What Is the Evaluation for B<strong>in</strong>ocular TVL?<br />
Transient Visual Loss 183<br />
The evaluation of patients with bilateral TVL depends on a thorough history, especially<br />
directed at the characteristics and temporal course of the episodes of TVL and any<br />
associated symptoms, and a complete neuro-ophthalmologic exam<strong>in</strong>ation, <strong>in</strong>clud<strong>in</strong>g<br />
visual field test<strong>in</strong>g. If the episodes last seconds and papilledema is present, then MRI is<br />
<strong>in</strong>dicated. If MRI is negative, then a sp<strong>in</strong>al tap is warranted. If episodes of bilateral<br />
visual loss occur only on exposure to bright light, then evaluation of the carotid arteries<br />
is <strong>in</strong>dicated. Patients with typical expand<strong>in</strong>g migra<strong>in</strong>e sc<strong>in</strong>tillations and positive<br />
phenomena last<strong>in</strong>g 20 to 30 m<strong>in</strong>utes that have been noted to occur on different sides<br />
at different times and headaches that have been documented to occur on different sides<br />
at different times usually do not require further workup. Abnormalities on visual field<br />
exam<strong>in</strong>ation suggest<strong>in</strong>g a retrochiasmal lesion or atypical migra<strong>in</strong>e-like phenomena<br />
should prompt neuroimag<strong>in</strong>g (class III–IV, level C). Patients with visual symptoms that<br />
are brief, episodic, unformed, and not associated with the angular, sc<strong>in</strong>tillat<strong>in</strong>g figures<br />
might also require MRI or MR angiography (class III–IV, level U). When either<br />
‘‘migra<strong>in</strong>e’’ headache or visual symptoms are restricted to one side of the head (even<br />
if the visual field exam is normal), a neuroimag<strong>in</strong>g study for occipital AVM is reasonable<br />
(class III–IV, level U). Patients with migra<strong>in</strong>e and symptoms or signs of collagen<br />
vascular disease require a collagen vascular disease profile. Electroencephalography or<br />
a trial of anticonvulsant medications is warranted if occipital epilepsy is likely (class III,<br />
level U).<br />
The evaluation and treatment of patients with vertebrobasilar TIAs is controversial.<br />
MRI and MR angiography are usually suggested, especially to evaluate the vertebrobasilar<br />
circulation, and <strong>in</strong>traarterial angiography may be considered. A cardiac embolic<br />
source should always be considered and, if warranted, transthoracic or transesophageal<br />
echocardiography may be performed (class III–IV, level C). Treatment <strong>in</strong>cludes control<br />
of stroke risk factors and antiplatelet drugs or anticoagulation.
184 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
<strong>An</strong> approach to the evaluation of patients with bilateral TVL is presented <strong>in</strong><br />
Figure 8–2.<br />
References<br />
Ambres<strong>in</strong> A, Borruat F-X, Mermoud A. (2001). Recurrent transient visual loss after deep sclerotomy. Arch<br />
Ophthalmol 119:1213–1214.<br />
Ammache Z, Graber M, Davis P. (2000). Idiopathic stabb<strong>in</strong>g headache associated with monocular visual loss. Arch<br />
<strong>Neuro</strong>l 57:745–746.<br />
<strong>An</strong>dracchi S, Kupersmith MJ, Nelson MJ, et al. (2000). Visual loss from arterial steal <strong>in</strong> patients with maxillofacial<br />
arteriovenous malformation. Ophthalmology 107:730–736.<br />
Barletta JP, Fanous MM, Hamed LM. (1994). Temporary bl<strong>in</strong>dness <strong>in</strong> the TUR syndrome. J <strong>Neuro</strong>-ophthalmol<br />
14:6–8.<br />
Barnett HJM, Taylor DW, Eliasziw M, et al. (1998). Benefit of carotid endarterectomy <strong>in</strong> patients with symptomatic<br />
moderate or severe stenosis. New Engl J Med 339:1415–1425.<br />
Benavente O, Eliasziw M, Streifler JY, et al. (2001). Prognosis after transient monocular bl<strong>in</strong>dness with carotidartery<br />
stenosis. New Engl J Med 345:1084–1090.<br />
Bernard GA, Bennett JL. (1999). Vasospastic amaurosis fugax. Arch Ophthalmol 117:1568.<br />
Biousse V, Newman NJ, Lee AG, et al. (1998a). Intracranial Ew<strong>in</strong>g’s sarcoma. J <strong>Neuro</strong>-ophthalmol 18:187–191.<br />
Biousse V, Newman NJ, Sternberg P Jr. (1997). Ret<strong>in</strong>al ve<strong>in</strong> occlusion and transient monocular visual loss<br />
associated with hyperhomocyst<strong>in</strong>emia. Am J Ophthalmol 1997;124:257–260.<br />
Biousse V, Touboul P-J, D’<strong>An</strong>glejan-Chatillon J, et al. (1998b). Ophthalmic manifestations of <strong>in</strong>ternal carotid<br />
dissection. Am J Ophthalmol 126:565–577.<br />
Booy R. (1990). Amaurosis fugax <strong>in</strong> young women. Lancet 335:1538.<br />
Borruat F-X, Bogousslavsky J, Uffer S, et al. (1993). Orbital <strong>in</strong>farction syndrome. Ophthalmology 100:562–568.<br />
Bremner FD, Sanders MD, Sranford MR. (1999). Gaze-evoked amaurosis <strong>in</strong> dysthyroid orbitopathy. Br J<br />
Ophthalmol 83:501.<br />
Bruno A, Corbett JJ, Boller J, et al. (1990). Transient monocular visual loss patterns and associated vascular<br />
abnormalities. Stroke 21:34–39.<br />
Burger SK, Saul RF, Selhorst JB, Thurston SE. (1991). Transient monocular bl<strong>in</strong>dness caused by vasospasm. N Engl<br />
J Med 325:870–873.<br />
Case Records of the Massachusetts General Hospital. (1999). Case 3-1999. N Engl J Med 340:300–307.<br />
Casson RJ, Flem<strong>in</strong>g <strong>FK</strong>, Shaikh A, James B. (2001). Bilateral ocular ischemic syndrome secondary to giant cell<br />
arteritis. Arch Ophthalmol 119:306–307.<br />
Cates CA, Newman DK. (1998). Transient monocular visual loss due to uveitis-glaucoma-hyphaema (UGH)<br />
syndrome. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 65:131–132.<br />
Chen W-T, Fuh J-L, Lu S-R, Wang S-J. (2001). Persistent migra<strong>in</strong>ous phenomena might be responsive to<br />
lamotrig<strong>in</strong>e. Headache 41:823–825.<br />
Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Bilyk JR, et al. (2001). Gaze-evoked amaurosis produced by <strong>in</strong>traorbital buckshot<br />
pellet. Ophthalmology 108:201–206.<br />
Danks JJ, Harrad RA. (1998). Flash<strong>in</strong>g lights <strong>in</strong> thyroid eye disease: a new symptom described and (possibly)<br />
expla<strong>in</strong>ed. Br J Ophthalmol 82:1309–1311.<br />
Diego M, Margo CE. (1998). Postural vision loss <strong>in</strong> giant cell arteritis. J <strong>Neuro</strong>-ophthalmol 18:124–126.<br />
Donders RCJM for the Dutch TMB Study Group. (2001). <strong>Cl<strong>in</strong>ical</strong> features of transient monocular bl<strong>in</strong>dness and<br />
the likelihood of atherosclerotic lesions of the <strong>in</strong>ternal carotid artery. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
71:247–249.<br />
Donders RCJM, Kappelle LJ, Derksen RHWM, et al. (1998). Transient monocular bl<strong>in</strong>dness and antiphospholipid<br />
antibodies <strong>in</strong> systemic lupus erythematosus. <strong>Neuro</strong>logy 51:535–540.<br />
Dreier JP, Sakowitz OW, Unterberg AW, et al. (2001). Migra<strong>in</strong>ous aura start<strong>in</strong>g several m<strong>in</strong>utes after the onset of<br />
subarachnoid hemorrhage. <strong>Neuro</strong>logy 57:1344–1345.<br />
Ebner R. (1995). Morn<strong>in</strong>g glory syndrome, amaurosis fugax, and cortical laser tomography. Presented at the North<br />
American <strong>Neuro</strong>-Ophthalmology Society annual meet<strong>in</strong>g, Tucson, Arizona.<br />
F<strong>in</strong>elli PF. (1997). Alternat<strong>in</strong>g amaurosis fugax and temporal arteritis. Am J Ophthalmol 123:850–851.<br />
F<strong>in</strong>eman MS, Regillo CD, Sergott RC, et al. (1999). Transient visual loss and decreased ocular blood flow velocities<br />
follow<strong>in</strong>g a scleral buckl<strong>in</strong>g procedure. Arch Ophthalmol 117:1647–1648.
Transient Visual Loss 185<br />
Fisher CM. (1999). Late-life (migra<strong>in</strong>ous) sc<strong>in</strong>tillat<strong>in</strong>g zigzags without headache: one person’s 27-year experience.<br />
Headache 39:391–397.<br />
Friedberg DN, Fox LE. (1999). Blurred vision dur<strong>in</strong>g sexual arousal associated with narrow-angle glaucoma. Am J<br />
Ophthalmol 128:647–648.<br />
Galetta SL, Balcar LJ, Liu GT. (1997). Giant cell arteritis with unusual flow-related neuro-ophthalmologic<br />
manifestations. <strong>Neuro</strong>logy 49:1463–1465.<br />
Gass JDM. (1997). Stereoscopic Atlas of Macular Disease. Diagnosis and Treatment. 4th ed. St. Louis, Mosby, pp.<br />
464–466, 984–985.<br />
Gibson JM, Cullen JF. (1982). Bl<strong>in</strong>dness and visual field defects follow<strong>in</strong>g cerebral angiography. <strong>Neuro</strong><strong>ophthalmology</strong><br />
2:297–303.<br />
Goodw<strong>in</strong> JA, Gorelick P, Helgason CM. (1987). Symptoms of amaurosis fugax <strong>in</strong> atherosclerotic carotid artery<br />
disease. <strong>Neuro</strong>logy 37:829–832.<br />
Gupta A, Jalali S, Bansal RK, Grewal SPS. (1990). <strong>An</strong>terior ischemic optic neuropathy and branch ret<strong>in</strong>al artery<br />
occlusion <strong>in</strong> cavernous s<strong>in</strong>us thrombosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:193–196.<br />
Haan J, Sluis P, Sluis LH, Ferrari MD. (2000). Acetazolamide treatment for migra<strong>in</strong>e aura status. <strong>Neuro</strong>logy<br />
55:1588–1589.<br />
Haas DC. (1991). Arteriovenous malformations and migra<strong>in</strong>e: case reports and analysis of the relationship.<br />
Headache 31:509–513.<br />
Hamed LM, Guy JR, Moster ML, Bosley T. (1992). Giant cell arteritis <strong>in</strong> the ocular ischemic syndrome. Am J<br />
Ophthalmol 113:702–705.<br />
Hardy TG, O’Day J. (1998). Ret<strong>in</strong>al arteriovenous malformation with fluctuat<strong>in</strong>g vision and ischemic central<br />
ret<strong>in</strong>al ve<strong>in</strong> occlusion and its sequelae: 25-year follow-up of a case. J <strong>Neuro</strong>-ophthalmol 18:233–236.<br />
Haynes RB, Taylor DW, Sackett DL, et al. (1994). Prevention of functional impairment by endarterectomy for<br />
symptomatic high-grade carotid stenosis. JAMA 271:1256–1259.<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1998a). Ocular manifestations of giant cell arteritis. Am J Ophthalmol<br />
125:509–520.<br />
Hayreh SS, Podhajsky PA, Zimmerman B. (1998b). Occult giant cell arteritis: ocular manifestations. Am J<br />
Ophthalmol 125:521–526.<br />
Hilton-Jones D, Ponsford JR, Graham N. (1982). Transient visual obscurations, without papilloedema. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 45:832–834.<br />
H<strong>in</strong>chey J, Chaves C, Appignani B, et al. (1996). A reversible posterior leukoencephalopathy syndrome. N Engl<br />
J Med 334:494–500.<br />
Hurwitz BJ, Heyman A, Wilk<strong>in</strong>son WE, et al. (1985). Comparison of amaurosis fugax and transient cerebral<br />
ischemia. A prospective cl<strong>in</strong>ical, arteriographic study. <strong>An</strong>n <strong>Neuro</strong>l 18:698–704.<br />
Hwang J-M, Girk<strong>in</strong> CA, Perry JD, et al. (1999). Bilateral ocular ischemic syndrome secondary to giant cell arteritis<br />
progress<strong>in</strong>g despite corticosteroid treatment. Am J Ophthalmol 127:102–104.<br />
Jehn A, Dettwiler BF, Fleischhauer J, et al. (2002). Exercise-<strong>in</strong>duced vasospastic amaurosis fugax. Arch Ophthalmol<br />
120:220–222.<br />
Kaiboriboon K, Piriyawat P, Selhorst JB. (2001). Light-<strong>in</strong>duced amaurosis fugax. Am J Ophthalmol 131:674–676.<br />
Karp BI, Yang JC, Khorsand M, et al. (1996). Multiple cerebral lesions complicat<strong>in</strong>g therapy with <strong>in</strong>terleuk<strong>in</strong>-2.<br />
<strong>Neuro</strong>logy 47:417–424.<br />
Kawaguchi S, Sakaki T, Morimoto T, et al. (1999). Effects of bypass on ocular ischaemic syndrome caused by<br />
reversed flow <strong>in</strong> the ophthalmic artery. Lancet 354:2052–2053.<br />
Kawaguchi S, Okuno S, Sakaki T, Nishikawa N. (2001). Effect of carotid endarterectomy on chronic ocular<br />
ischemic syndrome due to <strong>in</strong>ternal carotid artery stenosis. <strong>Neuro</strong>surgery 48:328–333.<br />
Kerty E. (1999). The <strong>ophthalmology</strong> of <strong>in</strong>ternal carotid artery dissection. Acta Ophthalmol Scand 77:418–421.<br />
Kesler A, Kaneti H, Kidron D. (1998). Transient cortical bl<strong>in</strong>dness <strong>in</strong> preeclampsia with <strong>in</strong>dication of generalized<br />
vascular endothelial damage. J <strong>Neuro</strong>-ophthalmol 18:163–165.<br />
Kle<strong>in</strong> R, Kle<strong>in</strong> BEK, Jensen SC, et al. (1999). Ret<strong>in</strong>al emboli and stroke. The Beaver Dam Eye Study. Arch<br />
Ophthalmol 117:1063–1068.<br />
Knapp ME, Flaharty PM, Sergott RC, et al. (1992). Gaze-<strong>in</strong>duced amaurosis from central ret<strong>in</strong>al artery<br />
compression. Ophthalmology 99:238–240.<br />
Kohmoto H, Oohira A. (1993). Gaze-evoked scotomata <strong>in</strong> metastatic orbital tumor. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
13:223–226.<br />
Kosmorsky GS, Rosenfeld SI, Burde RM. (1985). Transient monocular obscuration? Amaurosis fugax: a case<br />
report. Br J Ophthalmol 69:688–690.<br />
Kramer M, Goldenberg-Cohen N, Shapira Y, et al. (2001). Role of transesophageal echocardiography <strong>in</strong> the<br />
evaluation of patients with ret<strong>in</strong>al artery occlusion. Ophthalmology 108:1461–1464.
186 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Kuperschmidt H, Bont A, Schnorf H, et al. (1995). Transient cortical bl<strong>in</strong>dness and bioccipital bra<strong>in</strong> lesions <strong>in</strong> two<br />
patients with acute <strong>in</strong>termittent porphyria. <strong>An</strong>n Intern Med 123:598–600.<br />
Kupersmith MJ, Berenste<strong>in</strong> A, Nelson PK, et al. (1999). Visual symptoms with dural arteriovenous malformations<br />
dra<strong>in</strong><strong>in</strong>g <strong>in</strong>to occipital ve<strong>in</strong>s. <strong>Neuro</strong>logy 52:156–162.<br />
Kupersmith MJ, Vargas ME, Yashar A, et al. (1996). Occipital arteriovenous malformations: visual disturbances<br />
and presentation. <strong>Neuro</strong>logy 46:953–957.<br />
Kurita H, Sh<strong>in</strong> M. (2000). Resolution of migra<strong>in</strong>e with aura caused by an occipital arteriovenous malformation.<br />
Arch <strong>Neuro</strong>l 57:1219–1220.<br />
Lev<strong>in</strong> LA, Mootha VV. (1997). Postprandial transient visual loss. A symptom of critical carotid stenosis.<br />
Ophthalmology 104:297–401.<br />
Lev<strong>in</strong>e SR, Deegan MJ, Futrell N, Welch KMA. (1990). Cerebrovascular and neurologic disease associated with<br />
antiphospholipid antibodies. 48 cases. <strong>Neuro</strong>logy 40:1181–1189.<br />
Lewis JR, Glaser JS, Schatz NJ, Hutson DG. (1993). Pulseless (Takayasu) disease with ophthalmic manifestations.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:242–249.<br />
Liu GT, Schatz NJ, Galetta SL, et al. (1995). Persistent positive visual phenomena <strong>in</strong> migra<strong>in</strong>e. <strong>Neuro</strong>logy<br />
45:664–668.<br />
Luda E, Bo E, Sicuro L, et al. (1991). Susta<strong>in</strong>ed visual aura: a totally new variation of migra<strong>in</strong>e. Headache<br />
31:582–583.<br />
Malhotra R, Gregory-Evans K. (2000). Management of ocular ischemic syndrome. Br J Ophthalmol 84:1428–1431.<br />
Manor RS, Yassur Y, Hoyt WF. (1996). Read<strong>in</strong>g-<strong>in</strong>duced visual dimm<strong>in</strong>g. Am J Ophthalmol 121:212–213.<br />
Marshall J, Meadows S. (1968). The natural history of amaurosis fugax. Bra<strong>in</strong> 91:419–434.<br />
Meire FM, De Laey JJ, van Thienen MN, Schudd<strong>in</strong>ck L. (1991). Ret<strong>in</strong>al manifestations <strong>in</strong> fibromuscular dysplasia.<br />
Eur J Ophthalmol 1:63–68.<br />
Mezer E, Gdal-On M, Miller B. (1997). Orbital metastasis of renal cell carc<strong>in</strong>oma masquerad<strong>in</strong>g as amaurosis<br />
fugax. Eur J Ophthalmol 7:301–304.<br />
Miller NR. (1991). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams & Wilk<strong>in</strong>s,<br />
pp. 2300–2307, 2526–2528.<br />
Newman DS, Lev<strong>in</strong>e SR, Curtis VL, Welch KMA. (1989). Migra<strong>in</strong>e-like visual phenomena associated with cerebral<br />
venous thrombosis. Headache 29:82–85.<br />
North American Symptomatic Carotid Endarterectomy Trial Collaborators. (1991). Beneficial effects of carotid<br />
endarterectomy <strong>in</strong> symptomatic patients with high-grade carotid stenosis. N Engl J Med 325:445–453.<br />
O’Duffy D, James B, Elston J. (1998). Idiopathic <strong>in</strong>tracranial hypertension present<strong>in</strong>g with gaze-evoked amaurosis.<br />
Acta Ophthalmol Scand 76:119–120.<br />
O’Sullivan E, Shaunak S, Matthews T, et al. (1995). Transient monocular bl<strong>in</strong>dness. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
59:559.<br />
O’Sullivan E, Rossor M, Elston JS. (1992). Amaurosis fugax <strong>in</strong> young people. Br J Ophthalmol 76:660–662.<br />
Panayiotopoulos CP. (1999). Elementary visual halluc<strong>in</strong>ations, bl<strong>in</strong>dness, and headache <strong>in</strong> idiopathic occipital<br />
epilepsy: differentiation from migra<strong>in</strong>e. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 66:536–540.<br />
Pep<strong>in</strong> EP. (1990). Cerebral metastasis present<strong>in</strong>g as migra<strong>in</strong>e with aura. Lancet 336:127–128.<br />
Pess<strong>in</strong> MS, Duncan GD, Mohr JP, Poskanzer DC. (1977). <strong>Cl<strong>in</strong>ical</strong> and angiographic features of carotid transient<br />
ischemic attacks. N Engl J Med 296:358–362.<br />
Ramadan NM, Tietjen GE, Lev<strong>in</strong>e SR, Welch KMA. (1991). Sc<strong>in</strong>tillat<strong>in</strong>g scotomata associated with <strong>in</strong>ternal carotid<br />
artery dissection. Report of three cases. <strong>Neuro</strong>logy 41:1084–1087.<br />
Rennie CA, Flanagan DW. (2002). Resolution of proliferative venous stasis ret<strong>in</strong>opathy after carotid endarterectomy.<br />
Br J Ophthalmol 86:117–118.<br />
Riaz G, Selhorst JB, Hennessey, JJ. (1991). Men<strong>in</strong>geal lesions mimick<strong>in</strong>g migra<strong>in</strong>e. <strong>Neuro</strong>-<strong>ophthalmology</strong> 11:41–48.<br />
Romano JG, Babikian VL, Wijman CAC, Hedges TR III. (1998). Ret<strong>in</strong>al ischemia <strong>in</strong> aortic arch syndrome. J <strong>Neuro</strong>ophthalmol<br />
18:237–241.<br />
Russell MB, Olesen J. (1996). A nosographic analysis of the migra<strong>in</strong>e aura <strong>in</strong> a general population. Bra<strong>in</strong><br />
119:355–361.<br />
Sawchuk KSLJ, Churchill S, Feldman E, Drury I. (1997). Status epilepticus amauroticus. <strong>Neuro</strong>logy 49:1467–1469.<br />
Sharma S, Brown GC, Pater JL, Cruess AF. (1998). Does a visible ret<strong>in</strong>al embolus <strong>in</strong>crease the likelihood of<br />
hemodynamically significant carotid artery stenosis <strong>in</strong> patients with acute ret<strong>in</strong>al arterial occlusion? Arch<br />
Ophthalmol 116:1602–1606.<br />
Smith L, Kriss A, Gregson R, et al. (1998). Gaze evoked amaurosis <strong>in</strong> neurofibromatosis type II. Br J Ophthalmol<br />
82:584–585.
Transient Visual Loss 187<br />
Sobottka Ventura AC, Remi<strong>in</strong>da L, Mojon DS. (2001). Intermittent visual loss and exophthalmos due to the blue<br />
rubber bleb nevus syndrome. Am J Ophthalmol 132:132–135.<br />
Spier<strong>in</strong>gs ELH. (2001). Daily migra<strong>in</strong>e with visual aura associated with an occipital arteriovenous malformation.<br />
Headache 41:193–197.<br />
Teman AJ, W<strong>in</strong>terkorn JMS, We<strong>in</strong>er D. (1995). Transient monocular bl<strong>in</strong>dness associated with sexual <strong>in</strong>tercourse.<br />
N Engl J Med 333:393.<br />
Tipp<strong>in</strong> J, Corbett JJ, Kerber RE, et al. (1989). Amaurosis fugax and ocular <strong>in</strong>farction <strong>in</strong> adolescents and young<br />
adults. <strong>An</strong>n <strong>Neuro</strong>l 26:69–77.<br />
Wall M, George D. (1991). Idiopathic <strong>in</strong>tracranial hypertension: a prospective study of 50 patients. Bra<strong>in</strong><br />
114:155–180.<br />
Wijman CAC, Wolf PA, et al. (1998). Migra<strong>in</strong>ous visual accompaniments are not rare <strong>in</strong> late life: the Fram<strong>in</strong>gham<br />
Study. Stroke 29:1539–1543.<br />
W<strong>in</strong>terkorn JMS, Beckman RL. (1995). Recovery from ocular ischemic syndrome after treatment with verapamil.<br />
J <strong>Neuro</strong>-ophthalmol 15:209–211.<br />
W<strong>in</strong>terkorn JMS, Kupersmith, MJ, Wirtschafter, JD, Forman, S. (1993). Brief report: treatment of vasospastic<br />
amaurosis fugax with calcium-channel blockers. N Engl J Med 329:396–398.<br />
Wykes WN, Adams GGW, Cullen JF. (1984). Temporal arteritis: visual loss associated with posture. <strong>Neuro</strong><strong>ophthalmology</strong><br />
4:107–109.<br />
Zarnegar SR, Chung S, Selhorst JB. (1995). <strong>An</strong> unusual cause of amaurosis fugax. Presented at the North American<br />
<strong>Neuro</strong>-Ophthalmology annual meet<strong>in</strong>g, Tucson, Arizona.<br />
Zimmerman CF, Van Patten PD, Golnik KC, et al. (1995). Orbital <strong>in</strong>farction syndrome after surgery for <strong>in</strong>tracranial<br />
aneurysms. Ophthalmology 102:594–598.
This page <strong>in</strong>tentionally left blank
9 r<br />
Visual Field Defects<br />
What Is the Topographical Diagnosis of Visual<br />
Field Defects?<br />
The localization of visual field defects is outl<strong>in</strong>ed <strong>in</strong> Figure 9–1.<br />
Is the Visual Field Defect Unilateral?<br />
Lesions affect<strong>in</strong>g the ret<strong>in</strong>a, nerve fiber layer, or optic nerve produce visual field defects<br />
<strong>in</strong> the ipsilateral eye that correspond <strong>in</strong> position, shape, extent, and <strong>in</strong>tensity to the<br />
lesion. The lesion may be <strong>in</strong>flammatory, ischemic, degenerative, or neoplastic. Because<br />
the nerve fiber layer aris<strong>in</strong>g from the peripheral ret<strong>in</strong>a arches over the fovea, superior or<br />
<strong>in</strong>ferior nerve fiber layer damage results <strong>in</strong> arcuate-shaped visual field defects. Rarely,<br />
patients with a lesion of the anterior occipital lobe may have a unilateral, contralateral<br />
visual field defect (see monocular temporal crescent).<br />
Is a Ret<strong>in</strong>al Lesion Responsible for the Visual<br />
Field Defect?<br />
Almost all ret<strong>in</strong>al lesions result<strong>in</strong>g <strong>in</strong> visual field loss are visible ophthalmoscopically.<br />
Careful attention should be directed to the ret<strong>in</strong>a and ret<strong>in</strong>al nerve fiber layer<br />
correspond<strong>in</strong>g to the visual field defect. Patients with macular disease also may<br />
compla<strong>in</strong> of metamorphopsia, micropsia, and positive photopsias (e.g., flash<strong>in</strong>g<br />
lights) that are unusual <strong>in</strong> patients with optic neuropathies. Easily visible ret<strong>in</strong>al lesions<br />
are not discussed <strong>in</strong> detail. Table 9–1 lists some ret<strong>in</strong>al disorders that may be difficult to<br />
visualize without careful attention to the macula with high magnification and stereoscopic<br />
view<strong>in</strong>g. In some cases the ret<strong>in</strong>a may appear normal or near normal, and<br />
ancillary test<strong>in</strong>g may be required to def<strong>in</strong>e the etiology as ret<strong>in</strong>al (e.g., fluoresce<strong>in</strong><br />
angiography or focal, multi-focal, or full field electroret<strong>in</strong>ography).<br />
189
190 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 9–1. Evaluation of visual field defects.
Table 9–1. Subtle Ret<strong>in</strong>al Diseases that May Cause Visual<br />
Field Loss<br />
Cystoid macular edema<br />
Epiret<strong>in</strong>al membrane<br />
Outer ret<strong>in</strong>al <strong>in</strong>flammatory diseases<br />
Multiple evanescent white dot syndrome (MEWDS)<br />
Acute macular neuroret<strong>in</strong>itis (AMN)<br />
Acute ret<strong>in</strong>al pigment epitheliitis<br />
Acute multifocal placoid pigment epitheliopathy (AMPPE)<br />
Acute zonal occult outer ret<strong>in</strong>opathy (AZOOR)<br />
Serous detachment of the macula<br />
Cone-rod dystrophy<br />
Ret<strong>in</strong>itis pigmentosa s<strong>in</strong>e pigmento<br />
Cancer-associated ret<strong>in</strong>opathy (CAR)<br />
Melanoma-associated ret<strong>in</strong>opathy (MAR)<br />
<strong>An</strong>nular or r<strong>in</strong>g scotomas may occur with ret<strong>in</strong>opathies or optic neuropathies.<br />
Etiologies of annular or r<strong>in</strong>g scotomas <strong>in</strong>clude pigmentary ret<strong>in</strong>opathies, ret<strong>in</strong>itis,<br />
choroiditis, bl<strong>in</strong>d<strong>in</strong>g diffuse light, ret<strong>in</strong>al migra<strong>in</strong>e, myopia, cancer-associated ret<strong>in</strong>opathy<br />
(CAR), open angle glaucoma (from coalescence of upper and lower arcuate<br />
scotomas), and optic neuropathies (e.g., anterior ischemic optic neuropathy, AION).<br />
Bilateral annular or r<strong>in</strong>g scotomas may be due to bilateral ret<strong>in</strong>al or optic nerve disease<br />
but may also occur with bilateral occipital pole damage or occur on a functional<br />
(nonorganic) basis.<br />
Is There Evidence for an Optic <strong>Neuro</strong>pathy?<br />
Visual Field Defects 191<br />
Central visual field defects (unilateral or bilateral) are the result of damage to the<br />
papillomacular bundle or optic nerve. <strong>An</strong>y visual field defect produced by a ret<strong>in</strong>al<br />
lesion may be produced by a lesion of the optic nerve (Miller, 1998) and virtually any<br />
etiology may be responsible (e.g., glaucomatous, degenerative, ischemic, traumatic,<br />
<strong>in</strong>flammatory, <strong>in</strong>filtrative, compressive, vascular optic neuropathy). Patients with a<br />
unilateral visual field defect and evidence for an optic neuropathy should undergo<br />
evaluation for an optic neuropathy (see Chapter 1). Patients with a unilateral hemianopic<br />
visual field defect (junctional scotoma of Traquair) may harbor a lesion of the<br />
optic nerve at the junction of the optic nerve and chiasm.<br />
In assess<strong>in</strong>g optic nerve–related visual field defects, several anatomic po<strong>in</strong>ts are<br />
worth remember<strong>in</strong>g:<br />
1. Fibers from peripheral ganglion cells occupy a more peripheral position of the optic<br />
disc, whereas fibers from ganglion cells located closer to the disc occupy a more<br />
central position.<br />
2. Peripheral fibers course peripherally through the entire extent of the optic nerve.<br />
3. The papillomacular bundle occupies a large sector-shaped region of the temporal<br />
disc. This bundle of fibers moves centrally <strong>in</strong> the more distal (posterior) portions of<br />
orbital optic nerve.
192 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
4. All ret<strong>in</strong>al fibers reta<strong>in</strong> their relative positions throughout the visual pathways except<br />
<strong>in</strong> the optic tract and at the lateral geniculate nucleus, where there is a rotation of 90<br />
degrees that becomes ‘‘straightened out’’ <strong>in</strong> the optic radiations.<br />
Is the Monocular Temporal Crescent Affected<br />
<strong>in</strong> Isolation?<br />
Although monocular peripheral temporal visual field defects are most often the result of<br />
ret<strong>in</strong>al or optic nerve disease, a lesion of the peripheral nasal fibers <strong>in</strong> the anterior<br />
occipital lobe may also produce a unilateral (monocular) temporal crescent-shaped<br />
visual field defect from 60 to 90 degrees (‘‘half-moon syndrome’’) (Chavis, 1997).<br />
Although retrochiasmal lesions <strong>in</strong> the visual pathway usually result <strong>in</strong> homonymous<br />
visual field loss, the monocular temporal crescent is the one exception. Homonymous<br />
visual field loss with spar<strong>in</strong>g of this temporal crescent or selective <strong>in</strong>volvement of<br />
this area localizes the lesion to the occipital lobe, and neuroimag<strong>in</strong>g should be directed<br />
to the contralateral calcar<strong>in</strong>e cortex (Landau, 1995; Lepore, 2001).<br />
Is the Visual Field Defect Bilateral?<br />
Bilateral lesions of the ret<strong>in</strong>a or optic nerve may result <strong>in</strong> bilateral visual field defects.<br />
Table 9–2 reviews the cl<strong>in</strong>ical features of bilateral superior or <strong>in</strong>ferior altitud<strong>in</strong>al defects<br />
and bilateral central or cecocentral scotomas. In the absence of evidence for bilateral<br />
ret<strong>in</strong>al disease or bilateral optic neuropathy, bilateral visual field loss results from<br />
disease affect<strong>in</strong>g the optic chiasm or retrochiasmal pathways.<br />
Is the Visual Field Defect Junctional <strong>in</strong><br />
Nature?<br />
Nerve fibers orig<strong>in</strong>at<strong>in</strong>g <strong>in</strong> the ret<strong>in</strong>a follow a specific topographic arrangement <strong>in</strong> the<br />
optic nerve and chiasm. Compressive lesions at the junction of the <strong>in</strong>tracranial optic<br />
nerve and optic chiasm may produce characteristic visual field defects. Optic nerve<br />
<strong>in</strong>volvement at the junction of the optic chiasm results <strong>in</strong> unilateral visual field loss<br />
(optic neuropathy). If fibers from the <strong>in</strong>feronasal ret<strong>in</strong>a of the contralateral eye<br />
(Wilbrand’s knee) are <strong>in</strong>volved, there will also be a superotemporal visual field defect<br />
<strong>in</strong> the contralateral eye.<br />
The <strong>in</strong>tracranial optic nerves extend posteriorly from the optic foramen and jo<strong>in</strong> at the<br />
optic chiasm. With<strong>in</strong> the chiasm, fibers from the nasal ret<strong>in</strong>a of each eye cross <strong>in</strong>to<br />
the contralateral optic tract, and fibers from the temporal ret<strong>in</strong>a pass uncrossed <strong>in</strong>to the<br />
ipsilateral optic tract. With<strong>in</strong> the <strong>in</strong>tracranial optic nerve, the crossed (nasal ret<strong>in</strong>al) and<br />
uncrossed (temporal ret<strong>in</strong>al) fibers are anatomically separated at the junction of the<br />
optic nerve and chiasm. In addition, <strong>in</strong>ferior nasal cross<strong>in</strong>g fibers may loop anteriorly<br />
for a short distance <strong>in</strong>to the contralateral optic nerve. These fibers are often referred to as<br />
the anterior knee or Wilbrand’s knee.
Visual Field Defects 193<br />
Table 9–2. <strong>Cl<strong>in</strong>ical</strong> Features and Etiologies of Bilateral Superior or Inferior Altitud<strong>in</strong>al<br />
Defects and Bilateral Central or Cecocentral Scotomas<br />
Bilateral superior or <strong>in</strong>ferior (altitud<strong>in</strong>al hemianopia)<br />
Most caused by bilateral optic nerve or ret<strong>in</strong>al disease<br />
Rarely large prechiasmal lesion compresses both nerves <strong>in</strong>feriorly to cause bilateral superior<br />
hemianopia; compression of nerves from below may also elevate them aga<strong>in</strong>st the dural<br />
shelves extend<strong>in</strong>g out from the <strong>in</strong>tracranial end of the optic canals and cause bilateral <strong>in</strong>ferior<br />
altitud<strong>in</strong>al defects<br />
Bilateral symmetric damage to postchiasmal pathways may cause bilateral altitud<strong>in</strong>al defects;<br />
bilateral lesions of medial aspect of lateral geniculate body may cause bilateral <strong>in</strong>ferior<br />
hemianopias<br />
Bilateral occipital lesions may cause bilateral <strong>in</strong>ferior, and less often, superior hemianopias<br />
Bilateral ‘‘checker board’’ altitud<strong>in</strong>al hemianopia—superior defect <strong>in</strong> one eye and <strong>in</strong>ferior <strong>in</strong> the<br />
other eye<br />
Bilateral optic neuropathies<br />
May cause b<strong>in</strong>ocular diplopia because of vertical hemifield slide—preexist<strong>in</strong>g m<strong>in</strong>or phoria<br />
becomes a tropia because of vertical or horizontal separation or overlap of two nonoverlapp<strong>in</strong>g<br />
hemifields; this phenomenon may also occur with broad arcuate defects<br />
Bilateral cecocentral or central scotomas<br />
May be bilateral optic neuropathy of any cause (e.g., compressive) but more common etiologies<br />
<strong>in</strong>clude:<br />
Toxic or nutritional amblyopia<br />
Bilateral demyel<strong>in</strong>at<strong>in</strong>g optic neuritis<br />
Syphilis<br />
Leber’s hereditary optic neuropathy<br />
Bilateral macular disease<br />
Bilateral occipital lesions <strong>in</strong>volv<strong>in</strong>g macular projections<br />
Lesions at the junction of the optic nerve and chiasm may produce specific types of<br />
visual field defects that allow topographic localization. Selective compression of the<br />
crossed or uncrossed visual fibers at the junction may result <strong>in</strong> a unilateral temporal or<br />
nasal hemianopic field defect, respectively. In addition, <strong>in</strong>volvement of the <strong>in</strong>feronasal<br />
fibers of the anterior knee (Wilbrand’s knee) results <strong>in</strong> a superotemporal visual field<br />
defect contralateral to the lesion.<br />
In 1927, H. M. Traquair used the term junctional scotoma to refer to a unilateral<br />
temporal hemicentral field defect due to compression of the nasal fibers cross<strong>in</strong>g <strong>in</strong> the<br />
<strong>in</strong>tracranial optic nerve at the junction of the optic nerve and chiasm. Miller emphasized<br />
that the junctional scotoma described by Traquair refers to a strictly unilateral temporal<br />
scotoma that is assumed to arise from a lesion at the junction of the optic nerve and<br />
chiasm (Miller, 1998). Unfortunately, some confusion has arisen regard<strong>in</strong>g the use of the<br />
term junctional scotoma. As opposed to the defect described by Traquair, some authors<br />
have used the term to refer to an ipsilateral optic neuropathy with a contralateral<br />
superotemporal visual field defect. This superotemporal defect is caused by compression<br />
of the <strong>in</strong>feronasal fibers from the contralateral eye travel<strong>in</strong>g <strong>in</strong> Wilbrand’s knee.<br />
To clarify this dist<strong>in</strong>ction, Miller, cit<strong>in</strong>g J. Lawton Smith, recommended that the<br />
unilateral temporal visual field defect described by Traquair should be referred to as the<br />
‘‘junctional scotoma of Traquair’’ to differentiate it from the contralateral superotemporal<br />
defect more commonly referred to as the ‘‘junctional scotoma’’ (Miller, 1998).
194 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Recently, the existence of Wilbrand’s knee has come <strong>in</strong>to question. Wilbrand was<br />
restricted to exam<strong>in</strong><strong>in</strong>g human subjects who had undergone enucleation. In the<br />
enucleated eye, the nerve fibers atrophied and became dist<strong>in</strong>ct from the nerve fibers<br />
of the normal eye as seen on myel<strong>in</strong> sta<strong>in</strong><strong>in</strong>g. Horton, utiliz<strong>in</strong>g axon label<strong>in</strong>g techniques<br />
<strong>in</strong> nonenucleated monkeys, was unable to demonstrate cross<strong>in</strong>g fibers loop<strong>in</strong>g <strong>in</strong>to the<br />
contralateral optic nerve (Wilbrand’s knee) (Horton, 1997). In one monkey that had<br />
undergone enucleation 4 years previously, however, nerve fiber topography similar to<br />
that described by Wilbrand was found. Horton hypothesized that Wilbrand’s knee may<br />
be an artifact of enucleation caused by atrophy of the optic nerve and not a normal<br />
anatomic f<strong>in</strong>d<strong>in</strong>g. Nevertheless, whether Wilbrand’s knee exists anatomically, the<br />
localiz<strong>in</strong>g value of junctional visual field loss to the junction of the optic nerve and<br />
chiasm rema<strong>in</strong>s undim<strong>in</strong>ished because chiasmal compression alone may result <strong>in</strong> the<br />
contralateral superotemporal visual field defect (junctional scotoma). Karanjia and<br />
Jacobson described a junctional scotoma due to a focal lesion (pituitary tumor) of the<br />
prechiasmatic segment of the distal optic nerve and stressed the ‘‘exquisite localiz<strong>in</strong>g<br />
value’’ of a junctional scotoma (Karanjia, 1999).<br />
Trobe and Glaser noted that junctional visual field loss was due to a mass lesion <strong>in</strong> 98<br />
out of 100 cases (Trobe, 1983). The differential diagnosis of a junctional syndrome<br />
<strong>in</strong>cludes pituitary tumors, suprasellar men<strong>in</strong>giomas, supracl<strong>in</strong>oid aneurysms, craniopharyngiomas,<br />
and gliomas (Hershenfeld, 1993). Chiasmal neuritis, pachymen<strong>in</strong>gitis,<br />
and trauma are rare etiologies of the junctional syndrome. Junctional visual field<br />
abnormalities may also occur on a functional (nonorganic) basis.<br />
Patients with the junctional scotoma of Traquair or the junctional scotoma should be<br />
considered to have a compressive lesion at the junction of the optic nerve and chiasm<br />
until proven otherwise. <strong>Neuro</strong>imag<strong>in</strong>g studies, preferably magnetic resonance imag<strong>in</strong>g<br />
(MRI), should be directed to this location. Patients with junctional scotoma may be<br />
unaware of a small superotemporal visual field defect, and patients present<strong>in</strong>g with<br />
strictly unilateral visual compla<strong>in</strong>ts may be misdiagnosed as hav<strong>in</strong>g an optic neuritis or<br />
other unilateral optic neuropathy. Therefore, <strong>in</strong> any patient with presumed unilateral<br />
visual loss, careful visual field test<strong>in</strong>g should be performed <strong>in</strong> the contralateral<br />
asymptomatic eye.<br />
Is a Bitemporal Hemianopsia Present?<br />
Bitemporal hemianopsia may be peripheral, paracentral, or central. The visual field<br />
defect may ‘‘split’’ or ‘‘spare’’ the macular central field. The bitemporal defect usually is<br />
the result of a compressive mass lesion at the level of the optic chiasm (Miller, 1998).<br />
Tables 9–3 and 9–4 list the possible etiologies of a chiasmal lesion. Pseudochiasmal<br />
visual field defects (i.e., bitemporal defects that do not respect the vertical midl<strong>in</strong>e) may<br />
be due to tilted discs, colobomas, bilateral nasal ret<strong>in</strong>al disease (e.g., schisis), glaucoma,<br />
and bilateral optic neuropathies. <strong>Neuro</strong>imag<strong>in</strong>g (preferably MRI) should be directed at<br />
the optic chiasm <strong>in</strong> all patients with bitemporal defects that respect the vertical midl<strong>in</strong>e.<br />
Certa<strong>in</strong> anatomic relationships are important <strong>in</strong> evaluat<strong>in</strong>g chiasmal visual field<br />
defects:
Table 9–3. Compressive Chiasmal Syndromes<br />
Visual Field Defects 195<br />
Most common<br />
Pituitary apoplexy (Bills, 1993; Biousse, 2001; Embil, 1997)<br />
Pituitary tumor (especially pituitary adenoma) (Abe, 1998; Ikeda, 1995; Kerrison, 2000;<br />
Kupersmith, 1994; Lee, 1998; Peter, 1995; Petruson, 1995)<br />
Optic chiasm diastasis from pituitary tumor (Duru, 1999)<br />
Men<strong>in</strong>gioma (K<strong>in</strong>jo, 1995)<br />
Craniopharyngioma (Fahlbusch, 1999; Honegger, 1999; Mikelberg, 1993)<br />
Dysgerm<strong>in</strong>oma<br />
Suprasellar aneurysm<br />
Chiasmal glioma (Cirak, 2000; Miyairi, 2000; Rossi, 1994)<br />
Less common<br />
Abscess<br />
<strong>An</strong>aplastic astrocytoma (Miyairi, 2000)<br />
Arachnoid cyst<br />
Aspergillosis<br />
Cavernous hemangioma (Cobbs, 2001; Hwang, 1993)<br />
Chiasmal hematoma (chiasmal apoplexy) (Pakzaban, 2000)<br />
Chondroma<br />
Chordoma<br />
Choristomas<br />
Colloid cyst of the third ventricle<br />
Dermoid<br />
Dolichoectatic <strong>in</strong>ternal carotid arteries (Jacobson, 1999; Slav<strong>in</strong>, 1990b)<br />
Ependymoma<br />
Epidermoid<br />
Esthesioneuroblastoma<br />
Extramedullary hematopoiesis (Aarabi, 1998)<br />
Fibrous dysplasia<br />
Gangliocytoma (McCowen, 1999)<br />
Ganglioglioma (Liu, 1996)<br />
Giant cell tumor of bone<br />
Glioma<br />
Granular cell myoblastoma<br />
Hemangioblastoma (Saw<strong>in</strong>, 1996)<br />
Hemangioma (Bourekas, 2000)<br />
Hemangiopericytoma<br />
Histiocytosis X<br />
Hydrocephalus and distention of the third ventricle<br />
Intrasellar<br />
Langerhans’ cell histiocytoma (Job, 1999)<br />
Leukemia and lymphoma (Lee, 2001; McFadzean, 1990)<br />
Lipoma<br />
Lymphocytic hypophysitis (Abe, 1995; Beressi, 1994; Honegger, 1997; Jabre, 1997; Kerrison, 1997;<br />
Kristof, 1999; Lee, 1994; Naik, 1994; Nishioka, 1997; Stelmach, 1991; Thodou, 1995; Tubridy,<br />
2001)<br />
(cont<strong>in</strong>ued)
196 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 9–3. (cont<strong>in</strong>ued)<br />
Lymphohistiocytosis (Galetta, 1991)<br />
Melanoma (Aub<strong>in</strong>, 1997)<br />
Men<strong>in</strong>geal carc<strong>in</strong>omatosis<br />
Metastatic disease to bra<strong>in</strong> or pituitary gland (Baeesa, 1999)<br />
Mucocele or mucopyocele<br />
Multiple myeloma<br />
Nasopharyngeal cancer<br />
Nonneoplastic pituitary gland enlargement<br />
Paraganglioma<br />
Plasmacytoma<br />
Rathke cleft cyst (El-Mahdy, 1998; Fischer, 1994; Rao, 1995; Voelker, 1991; Yamamoto, 1993)<br />
Rhabdomyosarcoma (Arita, 2001)<br />
Sarcoid granuloma<br />
Sarcoma<br />
Schwannoma<br />
Septum pellucidum cyst<br />
S<strong>in</strong>us tumors<br />
Sphenoid s<strong>in</strong>us<br />
Syphilitic granuloma<br />
Teratoma<br />
Vascular malformation<br />
Venous aneurysm aris<strong>in</strong>g from carotid-cavernous s<strong>in</strong>us fistula (Wolansky, 1997)<br />
Venous angioma<br />
The ratio of crossed to uncrossed fibers is 53:47.<br />
Uncrossed fibers, both dorsal and ventral, ma<strong>in</strong>ta<strong>in</strong> their relative position at the<br />
lateral aspects of the chiasm and pass directly <strong>in</strong>to the ipsilateral optic tract.<br />
Dorsal extramacular cross<strong>in</strong>g fibers from each eye decussate posteriorly <strong>in</strong> the chiasm<br />
and then directly enter the dorsomedial aspect of contralateral optic tract.<br />
Macular fibers that cross do so <strong>in</strong> the central and posterior portions of chiasm.<br />
Some <strong>in</strong>feronasal ret<strong>in</strong>a fibers, primarily peripheral fibers, may loop <strong>in</strong> Wilbrand’s<br />
loop (although anatomic existence of this structure is questioned).<br />
Is a B<strong>in</strong>asal Hemianopsia Present?<br />
Most organic nasal visual field defects are actually arcuate <strong>in</strong> nature. Bilateral irregular<br />
nasal defects may be associated with optic disc drusen, but such defects do not obey the<br />
vertical midl<strong>in</strong>e and <strong>in</strong> fact are generally arcuate <strong>in</strong> nature. B<strong>in</strong>asal defects are usually<br />
due to bilateral <strong>in</strong>traocular disease of the ret<strong>in</strong>a or optic nerve (e.g., chronic papilledema,<br />
ischemic optic neuropathy, glaucoma, optic nerve drusen, or ret<strong>in</strong>al disease such<br />
as sector ret<strong>in</strong>itis pigmentosa or ret<strong>in</strong>oschisis). Rarely compression of the lateral chiasm<br />
may result <strong>in</strong> a b<strong>in</strong>asal defect. Bilateral nasal defect may occur with hydrocephalus with<br />
third ventricle enlargement caus<strong>in</strong>g lateral displacement of optic nerves aga<strong>in</strong>st the
Table 9–4. Other Causes of Chiasmal Syndrome<br />
Visual Field Defects 197<br />
Hydrocephalus (Bogdanovic, 2000)<br />
Cobalam<strong>in</strong> deficiency (Wilhelm, 1993)<br />
Demyel<strong>in</strong>at<strong>in</strong>g disease (Newman, 1991; Wilhelm, 1993)<br />
Empty sella syndrome (primary or secondary) (Kosmorsky, 1997)<br />
Chiasmal ischemia<br />
Optochiasmatic arachnoiditis<br />
Foreign body–<strong>in</strong>duced granuloma (e.g., musl<strong>in</strong>)<br />
Idiopathic<br />
Infection<br />
Chronic fungal <strong>in</strong>fection<br />
Cryptococcus<br />
Cysticercosis (Chang, 2001)<br />
Encephalitis<br />
Epste<strong>in</strong>-Barr virus (Beiran, 2000)<br />
Men<strong>in</strong>gitis<br />
Mucormycosis (Lee, 1996)<br />
Nasopharyngeal and s<strong>in</strong>us <strong>in</strong>fections<br />
Syphilis<br />
Tuberculosis<br />
Inflammatory<br />
Collagen vascular disease (e.g., systemic lupus erythematosus) (Frohman, 2001; Siatkowski,<br />
2001)<br />
Rheumatoid pachymen<strong>in</strong>gitis<br />
Sarcoid<br />
Multiple sclerosis<br />
Posthemorrhagic<br />
Posttraumatic<br />
Radiation necrosis<br />
Shunt catheter<br />
Toxic (see toxic optic neuropathies)<br />
Tobacco-alcohol toxicity (Danesh-Meyer, 2000)<br />
Ethchlorvynol (Placidyl)<br />
Phenipraz<strong>in</strong>e (Catron)<br />
Trauma, <strong>in</strong>clud<strong>in</strong>g postsurgical (Carter, 1998; Dom<strong>in</strong>go, 1993; He<strong>in</strong>z, 1994)<br />
Fat pack<strong>in</strong>g after transsphenoidal hypophysectomy (McHenry, 1993)<br />
Tether<strong>in</strong>g scar tissue caus<strong>in</strong>g delayed visual deterioration after pituitary surgery (Czech, 1999)<br />
Vascular occlusion<br />
Vasculitis (Wilhelm, 1993)<br />
Hereditary (probably autosomal recessive) chiasmal optic neuropathy (Pomeranz, 1999)<br />
Nonorganic (functional) (Miele, 2000)
198 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
supracl<strong>in</strong>oid portion of the <strong>in</strong>ternal carotid arteries. B<strong>in</strong>asal defects have also been<br />
described <strong>in</strong> patients with primary empty sella syndrome and with other suprasellar<br />
lesions (Charteris, 1996).<br />
<strong>An</strong> unusual b<strong>in</strong>asal visual field impairment has been noted with spontaneous<br />
<strong>in</strong>tracranial hypotension from a dural cerebrosp<strong>in</strong>al fluid leak (Horton, 1994). Some<br />
of these patients have a b<strong>in</strong>asal defect with peripheral depressions most severe <strong>in</strong><br />
the upper nasal quadrants but also <strong>in</strong>volv<strong>in</strong>g the lower nasal and upper temporal<br />
quadrants.<br />
Is a Homonymous Hemianopsia Present?<br />
Homonymous visual field impairments appear with lesions of the retrochiasmal pathways.<br />
Those affect<strong>in</strong>g the optic tract and lateral geniculate body tend to be <strong>in</strong>congruous,<br />
but the more posteriorly the lesion is located <strong>in</strong> the optic radiation, the greater the<br />
congruity of the defects. In general, tumors produce slop<strong>in</strong>g field defects, whereas<br />
vascular lesions produce sharp field defects. The localization of homonymous field<br />
defects depends on the nature of the field defect and associated neuro-ophthalmologic<br />
and neurologic f<strong>in</strong>d<strong>in</strong>gs. Homonymous field defects may be caused by lesions affect<strong>in</strong>g<br />
the optic tract, lateral geniculate body, optic radiations, or occipital lobe. Rarely, an<br />
occipital lesion may cause a monocular field defect (see monocular crescent, above). In<br />
general, complete homonymous hemianopias are nonlocaliz<strong>in</strong>g and may be seen with<br />
any lesions of the retrochiasmal pathway, <strong>in</strong>clud<strong>in</strong>g lesions of the lateral geniculate<br />
body, optic radiations, and striate cortex.<br />
Is the Homonymous Hemianopia Caused by<br />
an Optic Tract Lesion?<br />
In the optic tract, macular fibers lie dorsolaterally, peripheral fibers from the upper<br />
ret<strong>in</strong>a are situated dorsomedially, and peripheral fibers from the lower ret<strong>in</strong>a run<br />
ventrolaterally. Complete unilateral optic tract lesions cause a complete macular<br />
splitt<strong>in</strong>g homonymous hemianopia, usually without impaired visual acuity, unless<br />
the lesion extends to <strong>in</strong>volve the optic chiasm or nerve. Partial optic tract lesions are<br />
more common than complete lesions and result <strong>in</strong> an <strong>in</strong>congruous field defect that may<br />
be scotomatous. (The only other postchiasmatic location for a lesion caus<strong>in</strong>g a<br />
scotomatous hemianopic visual field defect is the occipital lobe.) Optic tract lesions<br />
are often associated with a relative afferent pupillary defect (RAPD) <strong>in</strong> the eye with the<br />
temporal field loss (contralateral to the side of the lesion). <strong>An</strong> afferent pupillary defect <strong>in</strong><br />
the contralateral eye <strong>in</strong> a patient with normal visual acuity bilaterally and a complete<br />
homonymous hemianopia is usually <strong>in</strong>dicative of optic tract <strong>in</strong>volvement (Miller, 1998).<br />
Wilhelm et al described a possible exception to this cl<strong>in</strong>ical rule. These authors<br />
described a RAPD contralateral to the lesions <strong>in</strong> 16 of 43 patients with congruous<br />
homonymous hemianopias (optic tract lesions excluded) (Wilhelm, 1996). Responsible<br />
lesions were postgeniculate and closer than 10 mm to the lateral geniculate nucleus. A<br />
RAPD did not occur <strong>in</strong> lesions farther than 18 mm from the lateral geniculate nucleus.<br />
The authors postulated that the RAPD was probably not caused by a lesion of the visual<br />
pathway per se, but by a lesion of <strong>in</strong>tercalated neurons between the visual pathways
and the pupillomotor centers <strong>in</strong> the pretectal area of the midbra<strong>in</strong>. <strong>An</strong>other abnormality<br />
of the pupil that may occur with optic tract lesions is due to concurrent third nerve<br />
<strong>in</strong>volvement by the pathologic process caus<strong>in</strong>g the tract damage. In these cases, the<br />
pupil ipsilateral to the lesion may be large and poorly reactive. F<strong>in</strong>ally, many patients<br />
with chronic optic tract lesions develop bilateral optic atrophy with a characteristic<br />
‘‘wedge,’’ ‘‘band,’’ or ‘‘bow-tie’’ pallor <strong>in</strong> the contralateral eye (identical to that seen <strong>in</strong><br />
some patients with bitemporal visual field loss from chiasmal lesions), and a more<br />
generalized pallor <strong>in</strong> the ipsilateral optic nerve associated with loss of nerve fiber layer<br />
<strong>in</strong> the superior and <strong>in</strong>ferior arcuate regions correspond<strong>in</strong>g to the bulk of temporal fibers<br />
subserv<strong>in</strong>g the nasal visual fields (hemianopic optic atrophy) (Miller, 1998). Hemianopic<br />
optic atrophy <strong>in</strong>dicates postchiasmal, preoptic radiation <strong>in</strong>volvement (i.e., optic tract or<br />
lateral geniculate body damage), but has also been rarely described <strong>in</strong> congenital<br />
retrogeniculate lesions (Miller, 1998).<br />
Etiologies of optic tract lesions <strong>in</strong>clude space-occupy<strong>in</strong>g lesions (e.g., glioma, men<strong>in</strong>gioma,<br />
craniopharyngioma, metastasis, pituitary adenoma, ectopic p<strong>in</strong>ealoma, abscess,<br />
sella arachnoidal cyst), aneurysms, arteriovenous malformations, dolichoectatic basilar<br />
arteries, demyel<strong>in</strong>at<strong>in</strong>g disease, and trauma, <strong>in</strong>clud<strong>in</strong>g neurosurgical procedures (e.g.,<br />
temporal lobectomy, <strong>in</strong>sertion of <strong>in</strong>traventricular shunt) (Chun, 1998; Freitag, 2000;<br />
Groomm, 1997; Guirgis, 2001; Liu, 1997; Miller, 1998; Molia, 1996; Shults, 1993; Slav<strong>in</strong>,<br />
1990a; Vargas, 1994). Patients undergo<strong>in</strong>g posterior pallidotomy for park<strong>in</strong>sonism may<br />
develop mild to moderate contralateral homonymous superior quadrantanopias associated<br />
with small paracentral scotomas likely due to optic tract damage (Biousse, 1998).<br />
A congenital optic tract syndrome has also been described (Murphy, 1997). A complete<br />
neurologic exam<strong>in</strong>ation and MRI, with specific attention to the optic tract region,<br />
are warranted <strong>in</strong> all patients suspected of hav<strong>in</strong>g an optic tract lesion. If MRI fails to<br />
reveal the responsible lesion, then MR angiography or cerebral angiography may be<br />
warranted <strong>in</strong> nontraumatic cases to <strong>in</strong>vestigate the presence of vascular lesions (e.g.,<br />
aneurysm).<br />
Is the Homonymous Hemianopia Caused by a<br />
Lesion of the Lateral Geniculate Body?<br />
Visual Field Defects 199<br />
In the lateral geniculate body, axons from ganglion cells superior to fovea are located<br />
medially, axons orig<strong>in</strong>at<strong>in</strong>g from ganglion cells <strong>in</strong>ferior to fovea are located laterally,<br />
and macular fibers term<strong>in</strong>ate <strong>in</strong> a large central area. As axons leave the lateral geniculate<br />
body they re-rotate back to their orig<strong>in</strong>al positions so that with<strong>in</strong> the optic radiations<br />
and the striate cortex, fibers that have synapsed with axons from superior ret<strong>in</strong>as are<br />
located <strong>in</strong> superior radiations and above the calcar<strong>in</strong>e fissure <strong>in</strong> the striate cortex,<br />
whereas fibers that have synapsed with axons from the <strong>in</strong>ferior ret<strong>in</strong>as are located <strong>in</strong> the<br />
<strong>in</strong>ferior optic radiations and below the calcar<strong>in</strong>e fissure. Upper field fibers orig<strong>in</strong>ate <strong>in</strong><br />
the medial aspect of lateral geniculate nucleus and travel through the parietal lobes,<br />
whereas lower fields orig<strong>in</strong>ate from the lateral aspect of the lateral geniculate body and<br />
make a loop <strong>in</strong> the temporal lobe (Meyer’s loop or the Meyer-Archambault loop).<br />
Lateral geniculate body lesions may also cause a complete macular splitt<strong>in</strong>g homonymous<br />
hemianopia (Miller, 1998). Partial lesions result <strong>in</strong> an <strong>in</strong>congruous homonymous<br />
field defect. Hemianopic optic atrophy may develop and no RAPD is usually evident.
200 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Although the study of Wilhelm et al suggests that a RAPD may occasionally be present<br />
with lateral geniculate body or parageniculate optic radiation lesions (Wilhelm, 1996),<br />
this observation has not been confirmed by other <strong>in</strong>vestigators.<br />
Although lesions of the optic tract or lateral geniculate body often cause <strong>in</strong>congruous<br />
field defects, two relatively specific patterns of congruous homonymous field defects<br />
with abruptly slop<strong>in</strong>g borders, associated with sectorial optic atrophy, have been<br />
attributed to focal lesions of the lateral geniculate body caused by <strong>in</strong>farction <strong>in</strong> the<br />
territory of specific arteries. Occlusion of the anterior choroidal artery may cause a<br />
homonymous defect <strong>in</strong> the upper and lower quadrants with spar<strong>in</strong>g of a horizontal<br />
sector (quadruple sectoranopia) (Luco, 1992). This defect occurs because the lateral<br />
geniculate body is organized <strong>in</strong> projection columns oriented vertically that represent<br />
sectors of the field parallel to the horizontal meridians, and the anterior choroidal artery<br />
supplies the hilum and anterolateral part of the nucleus. Bilateral lateral geniculate<br />
lesions may therefore cause bilateral hourglass-shaped visual field defects (Donahue,<br />
1995) or bilateral bl<strong>in</strong>dness. In three reported cases of isolated bilateral <strong>in</strong>volvement of<br />
the lateral geniculate bodies, the pathogenesis <strong>in</strong>cluded anterior choroidal syphilitic<br />
arteritis, methanol toxicity-produc<strong>in</strong>g coagulative necrosis of the lateral geniculate body,<br />
and geniculate myel<strong>in</strong>olysis associated with the rapid correction of hyponatremia,<br />
respectively (Donahue, 1995). Barton described another patient with bilateral sectoranopia<br />
(‘‘hourglass’’ pattern) due to probable osmotic demyel<strong>in</strong>ation (Barton, 2001).<br />
Interruption of the posterior lateral choroidal artery that perfuses the central portion of<br />
the lateral geniculate causes a horizontal homonymous sector defect (wedge shaped)<br />
(Borruat, 1995; Luco, 1992; Neau, 1996; We<strong>in</strong>, 2000). In posterior lateral choroidal<br />
territory <strong>in</strong>farction, the homonymous quadrantanopia may be associated with hemisensory<br />
loss and neuropsychological dysfunction (transcortical aphasia, memory disturbances),<br />
and delayed contralateral abnormal movements (Neau, 1996). A homonymous<br />
horizontal sectoranopia is not diagnostic of a lateral geniculate body lesion, however, as<br />
a similar sector defect may occur with lesions affect<strong>in</strong>g the optic radiations (Carter, 1985)<br />
or, rarely, the occipital cortex <strong>in</strong> the region of the calcar<strong>in</strong>e fissure (Grossman, 1990),<br />
the temporooccipital junction, the parietotemporal region, or <strong>in</strong> the distribution of<br />
the superficial sylvian artery territory (Growchowicki, 1991). F<strong>in</strong>ally, a patient has<br />
been described with bilateral lateral geniculate lesions with bilateral sector defects<br />
with preservation of the visual fields <strong>in</strong> an hourglass distribution (Greenfield, 1996).<br />
The patient was a 28-year-old woman who developed <strong>in</strong>congruous b<strong>in</strong>asal and bitemporal<br />
visual field defects 1 week after hav<strong>in</strong>g a febrile gastroenteritis, characterized<br />
by severe diarrhea, while travel<strong>in</strong>g <strong>in</strong> Mexico. MRI demonstrated bilaterally <strong>in</strong>creased<br />
signal <strong>in</strong>tensity with<strong>in</strong> the lateral geniculate bodies. The severe diarrhea was thought to<br />
be associated with an aseptic bilateral lateral geniculitis result<strong>in</strong>g <strong>in</strong> the hourglassshaped<br />
visual fields.<br />
Patients with lesions of the lateral geniculate body may have no other signs or<br />
symptoms of neurologic <strong>in</strong>volvement or may have associated f<strong>in</strong>d<strong>in</strong>gs related to<br />
thalamic or corticosp<strong>in</strong>al tract <strong>in</strong>volvement. Etiologies for lateral geniculate damage<br />
<strong>in</strong>clude <strong>in</strong>farction, arteriovenous malformation, trauma, tumor, <strong>in</strong>flammatory disorders,<br />
demyel<strong>in</strong>at<strong>in</strong>g disease, and toxic exposure (e.g., methanol) (Borruat, 1995; Donahue,<br />
1995; Greenfield, 1996; Groomm, 1997; Kosmorsky, 1998; Luco, 1992; Neau, 1996). MRI,<br />
with attention to the lateral geniculate region, is <strong>in</strong>dicated <strong>in</strong> all cases (Borruat, 1995;<br />
Horton, 1990; Neau, 1996).
Is the Lesion Caus<strong>in</strong>g the Homonymous<br />
Hemianopia Located <strong>in</strong> the Optic Radiations?<br />
Visual Field Defects 201<br />
Lesions of the proximal portion of the optic radiations may result <strong>in</strong> a complete<br />
homonymous hemianopia with macular splitt<strong>in</strong>g. Superior homonymous quadrantic<br />
defects (‘‘pie-<strong>in</strong>-the-sky’’ field defects) may result from a lesion <strong>in</strong> the temporal<br />
(Meyer’s) loop of the optic radiations or <strong>in</strong> the <strong>in</strong>ferior bank of the calcar<strong>in</strong>e fissure.<br />
In a study of 30 patients with superior quadrantanopias, lesions were occipital <strong>in</strong> 83%,<br />
temporal <strong>in</strong> 13%, and parietal <strong>in</strong> 3% (Jacobson, 1997). In temporal lobe lesions, the<br />
superior quadrantic defect is usually, but not always, <strong>in</strong>congruous (Miller, 1998), and<br />
the <strong>in</strong>ferior marg<strong>in</strong>s of the defects may have slop<strong>in</strong>g borders and may cross beyond the<br />
horizontal midl<strong>in</strong>e. Also, the ipsilateral nasal field defect is often denser and comes<br />
closer to fixation than the defect <strong>in</strong> the contralateral eye. Macular vision may or may not<br />
be <strong>in</strong>volved with the quadrantic defect (Miller, 1998).<br />
Although visual field defects often may occur <strong>in</strong> isolation with temporal lobe lesions<br />
(Jacobson, 1997), other signs of neurologic impairment may be evident (Brazis, 2001).<br />
With dom<strong>in</strong>ant temporal lobe <strong>in</strong>volvement, aphasic syndromes may occur, whereas<br />
nondom<strong>in</strong>ant lesions may be associated with impaired recognition of facial emotional<br />
expression, sensory amusia (<strong>in</strong>ability to appreciate various characteristics of music), and<br />
aprosodias (impaired appreciation of emotional overtones of spoken language). Other<br />
abnormalities seen with temporal lobe dysfunction <strong>in</strong>clude memory impairment and<br />
seizures. Etiologies for temporal lobe dysfunction <strong>in</strong>clude space-occupy<strong>in</strong>g lesions (e.g.,<br />
tumors, abscesses, hemorrhage), arteriovenous malformations, <strong>in</strong>farction, <strong>in</strong>fections,<br />
congenital malformations, demyel<strong>in</strong>at<strong>in</strong>g disease, and trauma (e.g., temporal<br />
lobectomy) (Hughes, 1999; Slav<strong>in</strong>, 1990b). MRI is required <strong>in</strong> all patients.<br />
Hughes et al studied the visual field defects <strong>in</strong> 32 patients after temporal lobe<br />
resection (Hughes, 1999). Visual field defects were present <strong>in</strong> 31 of the 32 patients,<br />
but none of the patients were aware of the deficits. Po<strong>in</strong>ts nearest fixation were<br />
relatively spared, and defects were greatest <strong>in</strong> the sector closest to the vertical meridian<br />
<strong>in</strong> the eye ipsilateral to the resection. Ipsilateral and contralateral field defects differed <strong>in</strong><br />
topography and <strong>in</strong> depth. Thus this study demonstrated that certa<strong>in</strong> fibers from the<br />
ipsilateral eye travel more anteriorly and laterally <strong>in</strong> Meyer’s loop and supports the<br />
hypothesis that visual field defects due to anterior retrogeniculate lesions are <strong>in</strong>congruous<br />
because of anatomic differences <strong>in</strong> the afferent pathway (Hughes, 1999). There is<br />
no difference <strong>in</strong> the <strong>in</strong>cidence of visual field defects produced by anterior temporal<br />
lobectomy versus amygdalohippocampectomy (which spares lateral temporal anatomy)<br />
for patients with <strong>in</strong>tractable epilepsy (Egan, 2000).<br />
Involvement of the optic radiations <strong>in</strong> the depth of the parietal lobe gives rise to a<br />
congruous homonymous hemianopia, denser below than above (‘‘pie-on-the-floor’’<br />
defect). Such defects are usually more congruous than those produced by lesions of<br />
the temporal lobe, and because the entire optic radiation passes through the parietal<br />
lobe, large lesions may produce complete homonymous hemianopia with macular<br />
splitt<strong>in</strong>g (Miller, 1998). Patients may often be unaware of their visual field defects.<br />
Patients do not have associated pupillary abnormalities, and optic atrophy does not<br />
occur unless the responsible lesion is congenital.<br />
In a study of 41 patients with <strong>in</strong>ferior quadrantanopias, 76% were due to occipital<br />
lesions, 22% to parietal lesions, and 2% to temporal lesions (Jacobson, 1997). In patients
202 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
with occipital lesions, the field defects often occurred <strong>in</strong> isolation, whereas other<br />
localiz<strong>in</strong>g signs of parietal <strong>in</strong>volvement were evident <strong>in</strong> 89% of patients with parietal<br />
lesions. Thus, although visual field defects may occur <strong>in</strong> relative isolation with<br />
parietal lobe lesions, lesions <strong>in</strong> this location more often betray themselves by other<br />
signs of neurologic dysfunction (Brazis, 2001). Parietal lobe lesions may be associated<br />
with contralateral somatosensory impairment, <strong>in</strong>clud<strong>in</strong>g impaired object recognition,<br />
impaired position sense, impaired touch and pa<strong>in</strong> sensation, and tactile ext<strong>in</strong>ction.<br />
Dom<strong>in</strong>ant parietal lesions may cause apraxia, f<strong>in</strong>ger agnosia, acalculia, right-left<br />
disorientation, alexia, and aphasic disturbances, whereas nondom<strong>in</strong>ant lesions may<br />
be associated with anosognosia (denial of neurologic impairment), autotopagnosia<br />
(failure to recognize hemiplegic limbs as belong<strong>in</strong>g to self), spatial disorientation,<br />
hemispatial neglect, constructional apraxia (abnormal draw<strong>in</strong>g and copy<strong>in</strong>g), and<br />
dress<strong>in</strong>g apraxia. Pathologic processes associated with parietal dysfunction are essentially<br />
the same as those that may cause temporal lobe dysfunction and are best<br />
evaluated by MRI.<br />
Lepore studied n<strong>in</strong>e patients with alexia without agraphia and found that three had<br />
complete right homonymous hemianopia, two had complete right homonymous hemianopia<br />
with additional b<strong>in</strong>ocular or monocular left field loss, two had right superior<br />
quadrantanopia, and the last two had bilateral superior or <strong>in</strong>ferior quadrantanopia<br />
(Lepore, 1998). Right superior quadrant vision was impaired <strong>in</strong> eight patients, and no<br />
patient demonstrated an isolated right <strong>in</strong>ferior quadrantanopia or an isolated left<br />
homonymous field defect. No patient atta<strong>in</strong>ed 20=20 visual acuity bilaterally. Lepore<br />
concluded that bilateral visual field loss and decreased visual acuity occur <strong>in</strong> many cases<br />
of alexia without agraphia. The frequent presence of a right superior quadrantic field<br />
defect implies a critical role <strong>in</strong> read<strong>in</strong>g for the ventral outflow pathways of the dom<strong>in</strong>ant<br />
calcar<strong>in</strong>e cortex. Although right homonymous hemianopia and a left occipital lobe and<br />
splenium lesions rema<strong>in</strong> the paradigm for alexia without agraphia, bilateral field loss,<br />
decreased visual acuity, and bihemispheric disease are common and may adversely<br />
affect the <strong>in</strong>tegrity of neural read<strong>in</strong>g mechanisms.<br />
Is the Visual Field Defect Caused by an<br />
Occipital Lesion?<br />
Homonymous quadrantic visual field defects may occur with unilateral occipital lesions<br />
(Horton, 1991a). Superior quadrantic defects may be seen with <strong>in</strong>ferior calcar<strong>in</strong>e lesions,<br />
and <strong>in</strong>ferior quadrantic defects may occur with superior calcar<strong>in</strong>e lesions. A patient<br />
with a neurologically isolated quadrantanopia is likely to have a lesion <strong>in</strong> the occipital<br />
lobe, although, <strong>in</strong> the case of a superior quadrantanopia, the possibility of a temporal<br />
lobe lesion cannot be excluded us<strong>in</strong>g cl<strong>in</strong>ical criteria only (Jacobson, 1997). As noted<br />
above, quadrantanopias caused by lesions of the parietal lobe usually are associated<br />
with other localiz<strong>in</strong>g signs (Jacobson, 1997). Often field defects due to calcar<strong>in</strong>e lesions<br />
have a sharp horizontal edge that would not be caused by tumors or missile <strong>in</strong>juries<br />
because it is unlikely that they would <strong>in</strong>jure only one bank of the calcar<strong>in</strong>e fissure and<br />
leave the fellow calcar<strong>in</strong>e bank untouched. Horton and Hoyt suggest that a lesion of the<br />
extrastriate cortex (areas V2 and V3) would be more likely to expla<strong>in</strong> the sharp<br />
horizontal edge of the defect because areas V2 and V3 are divided along the horizontal
Visual Field Defects 203<br />
meridian <strong>in</strong>to separate halves flank<strong>in</strong>g the striate (V1) cortex and, consequently, the<br />
upper and lower quadrants <strong>in</strong> extrastriate cortex are physically isolated on opposite<br />
sides of the striate cortex (Horton, 1991b). Although a lesion <strong>in</strong> this location (e.g., a<br />
tumor) may have irregular marg<strong>in</strong>s, if it crosses the representation of the horizontal<br />
meridian <strong>in</strong> extrastriate cortex it will produce a quadrantic visual field defect with a<br />
sharp horizontal border because of the split layout of the upper and lower quadrants of<br />
V2=V3. Thus, a homonymous quadrantanopia respect<strong>in</strong>g the horizontal meridian is not<br />
a ‘‘pathognomonic’’ sign of extrastriate cortical disease but may occur with striate<br />
lesions (McFadzean, 1997). A congruous <strong>in</strong>ferior quadrantanopia with borders aligned<br />
on both the vertical and horizontal meridians has also been described with a lesion of<br />
the superior fibers of the optic radiations near the contralateral trigone where<br />
the fascicles of visual axons become compact as they approach the calcar<strong>in</strong>e cortex<br />
(Borruat, 1993).<br />
Gray et al report two patients with unique homonymous hemianopias from occipital<br />
lesions (Gray, 1998). One patient had vertical meridian spar<strong>in</strong>g and the other displayed<br />
horizontal meridian spar<strong>in</strong>g. MRI correlation with the defects confirmed that the<br />
vertical hemianopic meridian is represented along the border of the calcar<strong>in</strong>e lip and<br />
the horizontal meridian lies at the base of the calcar<strong>in</strong>e banks deep with<strong>in</strong> the calcar<strong>in</strong>e<br />
fissure. Galetta and Grossman reported two patients further demonstrat<strong>in</strong>g that the<br />
horizontal meridian is represented at the calcar<strong>in</strong>e fissure base <strong>in</strong> the primary visual<br />
cortex (Galetta, 2000).<br />
Medial occipital lesions cause highly congruous homonymous field defects (Horton,<br />
1994; Pess<strong>in</strong>, 1987a,b). When both the upper and the lower calcar<strong>in</strong>e cortices are<br />
affected, a complete homonymous hemianopia, usually with macular spar<strong>in</strong>g, develops.<br />
Spar<strong>in</strong>g of the central 5 degrees of vision (macular spar<strong>in</strong>g) is common with occipital<br />
lesions, probably due to a comb<strong>in</strong>ation of a large macular representation and dual blood<br />
supply (Miller, 1998). The central 10 to 15 degrees of vision fill a majority of the total<br />
surface area of the occipital cortex (as much as 50 to 60%) (Gray, 1997; Horton, 1994;<br />
McFadzean, 1994; Wong, 1999). We consider macular spar<strong>in</strong>g to be present when at least<br />
5 degrees of central visual field is spared; macular spar<strong>in</strong>g of 3 degrees or less may be<br />
due to wander<strong>in</strong>g fixation and may not be cl<strong>in</strong>ically mean<strong>in</strong>gful. Patients with purely<br />
occipital lesions are often aware of the hemianopia, whereas patients with larger or<br />
more anterior lesions, affect<strong>in</strong>g parietal regions or associative pathways to the primary<br />
or secondary visual association cortex, may be unaware of their deficit. Celesia et al,<br />
however, prospectively studied 32 consecutive patients with homonymous field defects<br />
due to ischemic <strong>in</strong>farcts and found hemianopic anosognosia, def<strong>in</strong>ed as the unawareness<br />
of visual loss <strong>in</strong> the homonymous hemifield (or hemiquadrant) <strong>in</strong> 20 patients (63%)<br />
(Celesia, 1997). Hemianopic anosognosia occurred predom<strong>in</strong>antly <strong>in</strong> right-sided lesions<br />
(16 of 26 patients or 62%), but also was present <strong>in</strong> 4 of 6 patients (67%) with left-sided<br />
lesions. Hemianopic anosognosia was associated with somatic anosognosia <strong>in</strong> 9 patients<br />
and hem<strong>in</strong>eglect <strong>in</strong> 17 patients. Eight patients had pure homonymous hemianopia<br />
without cognitive, motor, or somatosensory deficits; four of these patients had awareness<br />
of visual defect and three had hemianopic anosognosia. Patients <strong>in</strong> these two<br />
groups had similar anatomic lesions. Patients with phosphenes, photopsias, or visual<br />
halluc<strong>in</strong>ations were usually aware of their visual field loss. The authors suggest that<br />
hemianopic anosognosia is most often related to failure of discovery of the deficits,<br />
occasionally with severe visual hem<strong>in</strong>eglect, sometimes to generalized cognitive impairment,<br />
or to a comb<strong>in</strong>ation of these factors. The authors further conclude (1) there is no
204 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
specific cortical area for conscious visual perception; (2) visual awareness is processed<br />
by a distributed network <strong>in</strong>clud<strong>in</strong>g multiple visual cortices, parietal and frontal lobes,<br />
the pulv<strong>in</strong>ar, and the lateral geniculate bodies (lesions localized at various nodes or<br />
centers <strong>in</strong> the network may produce similar phenomena); and (3) both hemispheres are<br />
<strong>in</strong>volved <strong>in</strong> visual process<strong>in</strong>g and conscious awareness.<br />
Lesions of the striate cortex may be classified as anterior, <strong>in</strong>termediate, or posterior<br />
(Horton, 1994; Landau, 1995; McFadzean, 1994; Miller, 1998). <strong>An</strong>terior lesions lie<br />
adjacent to the parieto-occipital fissure and affect the monocular temporal crescent of<br />
the contralateral visual field (temporal crescent or half-moon syndrome). This area<br />
constitutes less than 10% of the total surface area of the striate cortex and the defect<br />
beg<strong>in</strong>s approximately 60 degrees from fixation. Both upper and lower temporal<br />
crescents may be scotomatous <strong>in</strong> the field of one eye, or only the upper or lower<br />
temporal crescent may be <strong>in</strong>volved. Conversely, the temporal crescent may be spared<br />
with lesions that destroy the entire calcar<strong>in</strong>e cortex except for the anterior tip (Landau,<br />
1995; Lepore, 2001). Posterior lesions are located <strong>in</strong> the posterior 50 to 60% of the striate<br />
cortex, <strong>in</strong>clud<strong>in</strong>g the occipital pole and operculum, affect macular vision (i.e., the central<br />
10 degrees <strong>in</strong> the contralateral hemifield), and therefore cause scotomatous defects.<br />
Intermediate lesions lie between the anterior and posterior conf<strong>in</strong>es and affect from 10<br />
to 60 degrees <strong>in</strong> the contralateral hemifield.<br />
The most common cause of unilateral occipital disease is <strong>in</strong>farction <strong>in</strong> the distribution<br />
of the posterior cerebral artery (Belden, 1999; Pess<strong>in</strong>, 1987a,b). Other etiologies <strong>in</strong>clude<br />
venous <strong>in</strong>farction, hemorrhage, arteriovenous malformation and fistulas, tumor,<br />
abscess, and trauma (Bartolomei, 1998; Kupersmith, 1996, 1999; Liu, 1997; Molia,<br />
1996). Thus, MRI is warranted <strong>in</strong> all patients.<br />
Bilateral occipital lobe lesions may occur from a s<strong>in</strong>gle or from consecutive events and<br />
may cause bilateral homonymous scotomas, usually with some macular spar<strong>in</strong>g (‘‘r<strong>in</strong>g’’<br />
scotomas) that respects the vertical midl<strong>in</strong>e (Miller, 1998). In some cases there may be<br />
‘‘tunnel’’ or ‘‘keyhole’’ fields with bilateral complete homonymous hemianopias except<br />
for macular spar<strong>in</strong>g. Careful test<strong>in</strong>g <strong>in</strong> these cases reveals that the macular spar<strong>in</strong>g<br />
respects the vertical midl<strong>in</strong>e. Bilateral lesions affect<strong>in</strong>g the superior or <strong>in</strong>ferior calcar<strong>in</strong>e<br />
cortices may produce bilateral altitud<strong>in</strong>al defects that may mimic the visual field<br />
abnormalities seen with bilateral optic nerve or ret<strong>in</strong>al disease (Hansen, 1993;<br />
Lakhanpal, 1990). Bilateral upper calcar<strong>in</strong>e bank lesions may have associated neurologic<br />
f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clud<strong>in</strong>g Bal<strong>in</strong>t’s syndrome (apraxia of gaze, optic ataxia, decreased visual<br />
attention, and simultanagnosia), abnormal depth perception, defective revisualization<br />
of spatial relations, topographic disorientation, and disorientation to place (Brazis, 2001;<br />
Caplan, 1990). Bilateral lesions of the <strong>in</strong>ferior banks of the calcar<strong>in</strong>e fissure may be<br />
associated with prosopagnosia (<strong>in</strong>ability to identify faces visually), cerebral dyschromatopsia,<br />
amnesia, and difficulty revisualiz<strong>in</strong>g the morphology and appearance of people<br />
and objects (Brazis, 2001; Caplan, 1990). Bilateral lesions of the visual cortices, often due<br />
to large bilateral posterior cerebral artery <strong>in</strong>farcts <strong>in</strong>volv<strong>in</strong>g both banks of the calcar<strong>in</strong>e<br />
fissure and both temporal lobes, cause cortical bl<strong>in</strong>dness often associated with agitated<br />
delirium and amnesia (Brazis, 2001; Caplan, 1990).<br />
Cortical bl<strong>in</strong>dness implies visual impairment due to discrete <strong>in</strong>volvement of the<br />
occipital cortices bilaterally, whereas cerebral bl<strong>in</strong>dness is a more general term <strong>in</strong>dicat<strong>in</strong>g<br />
visual loss from any process affect<strong>in</strong>g the retrogeniculate visual pathways. The essential<br />
features of cortical and cerebral bl<strong>in</strong>dness <strong>in</strong>clude complete loss of all visual sensation<br />
<strong>in</strong>clud<strong>in</strong>g all appreciation of light and dark; loss of reflex lid closure to bright
illum<strong>in</strong>ation and to threaten<strong>in</strong>g gestures; retention of the reflex constriction of the pupils<br />
to illum<strong>in</strong>ation and to convergence movements; and <strong>in</strong>tegrity of the normal structure of<br />
the ret<strong>in</strong>a as verified by ophthalmoscopy (Miller, 1998).<br />
There are many etiologies of cerebral and cortical bl<strong>in</strong>dness, <strong>in</strong>clud<strong>in</strong>g hypoxia,<br />
<strong>in</strong>farction, hemorrhage, eclampsia, hypertensive encephalopathy, tentorial herniation<br />
from cerebral mass, tumor, arteriovenous malformation, <strong>in</strong>fection (e.g., progressive<br />
multifocal leukoencephalopathy, Jakob-Creutzfeldt disease, subacute scleros<strong>in</strong>g panencephalitis,<br />
HIV encephalitis, syphilis, encephalitis, abscess), <strong>in</strong>flammation (e.g., sarcoidosis),<br />
demyel<strong>in</strong>at<strong>in</strong>g disease, trauma, metabolic disorders (e.g., adrenoleukodystrophy,<br />
hypoglycemia, porphyria, mitochondrial encephalopathies), tox<strong>in</strong>s (e.g., lead, mercury,<br />
ethanol, carbon monoxide), medications (e.g., cyclospor<strong>in</strong>e, tacrolimus, <strong>in</strong>terleuk<strong>in</strong>-2),<br />
radiation encephalopathy, Alzheimer’s disease, postictal after seizures, and complications<br />
of cerebral angiography (Belden, 1999; Blake, 1999; H<strong>in</strong>chey, 1996; Karp, 1996;<br />
Kuperschmidt, 1995; Miller, 1998; Ormerod, 1996; Pomeranz, 1998; Steg, 1999). Occasionally,<br />
patients with cortical bl<strong>in</strong>dness deny their visual defect (<strong>An</strong>ton’s syndrome).<br />
What If a Homonymous Visual Field Defect Is<br />
Present But <strong>Neuro</strong>imag<strong>in</strong>g Is Normal?<br />
Visual Field Defects 205<br />
As noted above, MRI is <strong>in</strong>dicated <strong>in</strong> all patients with a homonymous visual field defect,<br />
except <strong>in</strong> acute or traumatic cases, <strong>in</strong> which computed tomography (CT) imag<strong>in</strong>g is<br />
usually adequate, or <strong>in</strong> patients <strong>in</strong> whom MRI is contra<strong>in</strong>dicated (e.g., ferromagnetic<br />
aneurysmal clip, metallic fragments, pacemakers, etc.). There are several cl<strong>in</strong>ical<br />
situations <strong>in</strong> which MRI may be normal <strong>in</strong> a patient with a homonymous hemianopia<br />
(Brazis, 2000):<br />
1. Homonymous hemianopia or cortical bl<strong>in</strong>dness may be an early or <strong>in</strong>itial f<strong>in</strong>d<strong>in</strong>g <strong>in</strong><br />
some patients with the Heidenha<strong>in</strong> variant of Jakob-Creutzfeldt disease, and <strong>in</strong> most<br />
of these patients rout<strong>in</strong>e MRI or CT is normal (Aguglia, 1991; Brazis, 2000; Felton,<br />
1996; Jacobs, 2001; Vargas, 1995; Warren, 1992). Some patients, however, will have<br />
symmetric hyper<strong>in</strong>tensities <strong>in</strong> the basal ganglia and=or gray matter of the occipital<br />
cortex on T2-weighted and proton-weighted images (Kropp, 1999), and some will<br />
have abnormalities <strong>in</strong> the cortex, basal ganglia, and thalamus on diffusion-weighted<br />
MRI (Bahn, 1997, 1999; Jacobs, 2001; Mittal, 2002; Na, 1999; Zerr, 2000a,b). Bilateral<br />
symmetric, high signal <strong>in</strong>tensities on T2-weighted images were present <strong>in</strong> the basal<br />
ganglia of 109 (67%) of 162 patients with Jakob-Creutzfeldt disease, and thus MRI<br />
was thought to be reasonably sensitive (67%) and highly specific (93%) <strong>in</strong> the<br />
diagnosis of this entity (Schroter, 2000). The electroencephalogram (EEG) is often<br />
<strong>in</strong>itially normal <strong>in</strong> these patients, although it usually eventually shows characteristic<br />
periodic complexes <strong>in</strong> most patients. Patients soon also develop mentation impairment,<br />
myoclonus, and other signs of Jakob-Creutzfeldt disease, but <strong>in</strong>itially the<br />
diagnosis may be quite difficult. Abnormalities <strong>in</strong> the cerebrosp<strong>in</strong>al fluid, such as the<br />
presence of 14-3-3 prote<strong>in</strong> or neuron-specific enolase, may assist <strong>in</strong> the diagnosis<br />
(Aksamit, 2001; Green, 2001; Hsich, 1996; Kropp, 1999; Lemstra, 2000; Poser, 1999;<br />
Zerr, 1995, 1998, 2000a,b). For example, the presence of the 14-3-3 bra<strong>in</strong> prote<strong>in</strong> <strong>in</strong> the<br />
cerebrosp<strong>in</strong>al fluid has a positive predictive value for Jakob-Creutzfeldt disease of<br />
94.7%, whereas its absence has a negative predictive value of 92.4% (Hsich, 1996;
206 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Zerr, 1998). In other studies, the sensitivity was 94 to 97% and the specificity 74 to<br />
87% (Lemstra, 2000; Zerr, 2000a,b). False negatives have been documented (Chapman,<br />
2000) and false positives have been noted with herpes simplex encephalitis,<br />
men<strong>in</strong>goencephalitis, stroke, hypoxic bra<strong>in</strong> <strong>in</strong>jury, carc<strong>in</strong>omatous men<strong>in</strong>gitis, vascular<br />
dementia, Hashimoto’s encephalopathy, <strong>in</strong>tracerebral metastasis, frontotemporal<br />
dementia, dementia with Lewy bodies, and Alzheimer’s disease (Burkhard, 2001;<br />
Chapman, 2000; Hernandez Echebarria, 2000; Lemstra, 2000; Zerr, 1998). The 14-3-3<br />
prote<strong>in</strong> assay may be positive <strong>in</strong> paraneoplastic neurologic disorders that may mimic<br />
Jakob-Creutzfeldt disease, but the immunoblast<strong>in</strong>g pattern of this prote<strong>in</strong> dist<strong>in</strong>guishes<br />
most patients with paraneoplastic disorders from those with Jakob-<br />
Creutzfeldt disease (Saiz, 1999).<br />
2. Some patients with Alzheimer’s disease or Lewy body disease may develop a<br />
homonymous field defect (Bashir, 1998; Brazis, 2000; Trick, 1995). MRI may be<br />
normal or show only diffuse atrophy, and the EEG is normal or shows only mild<br />
diffuse slow<strong>in</strong>g. This diagnosis is suspected <strong>in</strong> patients with a slowly progressive<br />
dementia without other ‘‘focal’’ neurologic f<strong>in</strong>d<strong>in</strong>gs, and the dementia usually far<br />
outweighs the visual field impairment.<br />
3. Most patients with field defects from cerebral <strong>in</strong>farction or hypoxia demonstrate MRI<br />
changes compatible with ischemia. However, Moster et al described two patients,<br />
one with bilateral homonymous congruous hemianopic central scotomata after<br />
carbon monoxide poison<strong>in</strong>g and the other with bilateral congruous <strong>in</strong>ferior visual<br />
scotomata after global hypoxia, who were <strong>in</strong>itially diagnosed with ‘‘functional’’<br />
visual loss (Moster, 1996). Neither CT nor MRI adequately demonstrated the source<br />
of the visual dysfunction, but s<strong>in</strong>gle photon emission computed tomography<br />
(SPECT) <strong>in</strong> one patient and positron emission tomography (PET) imag<strong>in</strong>g <strong>in</strong> the<br />
other confirmed the organic substrate of the visual impairment. Wang et al also<br />
reported two patients with organophosphate <strong>in</strong>toxication associated with cortical<br />
visual loss who had normal MRI but abnormal hypometabolism of the visual cortex<br />
demonstrated on PET scann<strong>in</strong>g (Wang, 1999). Brazis et al also presented a patient<br />
with a homonymous field defect secondary to cerebral <strong>in</strong>farction with normal MRI<br />
(Brazis, 2000). Functional imag<strong>in</strong>g techniques, such as SPECT or PET, should thus be<br />
considered <strong>in</strong> patients with suspected cortical visual loss and normal CT or MRI<br />
studies. Functional MRI is also a promis<strong>in</strong>g method for the objective detection of<br />
abnormalities <strong>in</strong> the afferent visual system (Miki, 1996).<br />
4. Transient homonymous hemianopia with normal CT imag<strong>in</strong>g has rarely been<br />
reported with nonketotic hyperglycemia (Brazis, 2000; Harden, 1991). These patients<br />
had other positive visual phenomena associated with a homonymous hemianopia.<br />
Thus, nonketotic hyperglycemia may present with positive visual phenomena<br />
associated with a homonymous field defect and normal neuroimag<strong>in</strong>g.<br />
5. Functional (nonorganic) hemianopias are associated with normal imag<strong>in</strong>g studies<br />
(Keane, 1998; Mart<strong>in</strong>, 1998; Thompson, 1996). One method of determ<strong>in</strong><strong>in</strong>g if a field<br />
defect is nonorganic is to test saccadic eye movements <strong>in</strong>to the supposedly absent<br />
portion of the field, with the patient assum<strong>in</strong>g that eye movements and not visual<br />
fields are be<strong>in</strong>g tested. Demonstrat<strong>in</strong>g ‘‘hemianopic’’ defects with both eyes open is<br />
often useful (Keane, 1998). <strong>An</strong>other method is to place a 30-diopter Fresnel prism <strong>in</strong>to<br />
the upper quadrants of a trial frame (Carlow, 1995). After visual fields are obta<strong>in</strong>ed<br />
without the prism, the prism is placed first base-out and then base-<strong>in</strong> and with each<br />
change the fields are repeated. Patients with pathologic hemianopsias shift their
superior field 15 degrees to the right or to the left of the central vertical meridian with<br />
the prism base <strong>in</strong> or out, respectively, whereas patients with suspected functional<br />
hemifield defect do not shift their superior sectors <strong>in</strong> a similar fashion.<br />
What Treatments Can Be Offered to Patients<br />
with Homonymous Hemianopias?<br />
Treatment of processes caus<strong>in</strong>g visual field impairment is directed at the underly<strong>in</strong>g<br />
etiology. Unfortunately, patients with homonymous hemianopias have a consistently<br />
poor rehabilitation outcome, with no more than 20% of patients undergo<strong>in</strong>g spontaneous<br />
recovery with<strong>in</strong> the first several months of bra<strong>in</strong> <strong>in</strong>jury (Kerkhoff, 1992, 1994).<br />
Smith suggested the use of Fresnel press-on prisms <strong>in</strong> patients with homonymous<br />
hemianopia (Smith, 1981). The prism is placed on the outside half of the lens ipsilateral<br />
to the hemianopia with the base toward that side (e.g., for a patient with a right<br />
homonymous hemianopia, a 15- to 30-diopter prism is placed base-out on the right half<br />
of the right lens). The goal is to <strong>in</strong>crease the patient’s scann<strong>in</strong>g skills. Although prisms<br />
may help some patients, and although patients with the prisms perform significantly<br />
better than controls on visual perception tasks, there is overall no difference <strong>in</strong> activities<br />
of daily liv<strong>in</strong>g function<strong>in</strong>g (Rossi, 1990).<br />
Read<strong>in</strong>g problems are common <strong>in</strong> patients with homonymous field defects (Leff,<br />
2000). Patients with right hemianopias cannot see which letters or words follow those<br />
they have already read, and patients with left hemianopias often lose their place when<br />
read<strong>in</strong>g, often beg<strong>in</strong>n<strong>in</strong>g aga<strong>in</strong> on an unrelated l<strong>in</strong>e. A right homonymous hemianopia<br />
also disrupts the motor preparation of read<strong>in</strong>g saccades dur<strong>in</strong>g text read<strong>in</strong>g (Leff, 2000).<br />
A ruler to guide the patient’s vision is often useful, and some patients with hemianopias<br />
can improve their read<strong>in</strong>g by turn<strong>in</strong>g the material 90 degrees and read<strong>in</strong>g vertically <strong>in</strong><br />
their <strong>in</strong>tact hemifields. Hemianopic patients may also be tra<strong>in</strong>ed to perform large<br />
saccades <strong>in</strong>to the bl<strong>in</strong>d field and to search their entire field <strong>in</strong> various patterns, result<strong>in</strong>g<br />
<strong>in</strong> some visual improvement (Kerkoff, 1994, 2000).<br />
Patients with cortical or cerebral bl<strong>in</strong>dness with some visual preservation may benefit<br />
by referral to low-vision specialists for <strong>in</strong>struction <strong>in</strong> various visual aids to assist read<strong>in</strong>g<br />
and other daily activities.<br />
What Should Be Done with an Unexpla<strong>in</strong>ed<br />
Visual Field Defect?<br />
Visual Field Defects 207<br />
Patients with an unexpla<strong>in</strong>ed unilateral or bilateral visual field defect should have<br />
careful attention paid to the correspond<strong>in</strong>g areas of the ret<strong>in</strong>a and optic nerve on<br />
ophthalmoscopy. Some patients may have occult ret<strong>in</strong>al and=or choroidal vascular<br />
disease that may be detected only by timed and directed (to the location predicted by<br />
the visual field defect) fluoresce<strong>in</strong> angiography (Rizzo, 1993). Visual defects respect<strong>in</strong>g<br />
the vertical midl<strong>in</strong>e with a bitemporal or homonymous ‘‘flavor’’ should undergo a<br />
neuroimag<strong>in</strong>g study. Rarely, patients with unilateral or bilateral nasal defects respect<strong>in</strong>g<br />
the vertical midl<strong>in</strong>e may harbor an underly<strong>in</strong>g compressive lesion (Miller, 1998). In
208 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
addition, apparently altitud<strong>in</strong>al (superior or <strong>in</strong>ferior) bilateral visual field defects may<br />
actually represent bilateral lesions of the optic nerves or retrochiasmal pathway.<br />
Constriction of the visual fields may occur <strong>in</strong> media opacities, miotic pupils, or<br />
uncorrected refractive error; as an artifact of test<strong>in</strong>g; <strong>in</strong> occult ret<strong>in</strong>al disease (e.g.,<br />
ret<strong>in</strong>itis pigmentosa, cancer-associated ret<strong>in</strong>opathy, etc.); with any optic neuropathy<br />
(e.g., optic neuritis, ischemic optic neuropathy, glaucoma, etc.); with bilateral retrochiasmal<br />
lesions (e.g., occipital stroke); or <strong>in</strong> nonorganic patients. In fact, any comb<strong>in</strong>ation<br />
of these entities (e.g., an optic neuropathy and a retrochiasmal homonymous<br />
hemianopsia) may produce any number of comb<strong>in</strong>ations of associated visual field<br />
defects. The simple algorithm presented obviously cannot account for every one of these<br />
comb<strong>in</strong>ations. Electrophysiologic test<strong>in</strong>g such as electroret<strong>in</strong>ography, visual evoked<br />
potentials, and other ancillary test<strong>in</strong>g such as fluoresce<strong>in</strong> angiography may disclose an<br />
abnormality <strong>in</strong> the ret<strong>in</strong>a or optic nerve even <strong>in</strong> the absence of an ophthalmoscopically<br />
visible lesion. Pattern visual evoked potentials aid <strong>in</strong> the diagnosis of functional visual<br />
loss (Xu, 2001).<br />
References<br />
Aarabi B, Haghshenas M, Rakeii V. (1998). Visual failure caused by suprasellar extramedullary hematopoiesis <strong>in</strong><br />
beta thalassemia: case report. <strong>Neuro</strong>surgery 42:922–926.<br />
Abe T, Matsumoto K, Kuwazawa J, et al. (1998). Headache associated with pituitary adenoma. Headache<br />
38:782–786.<br />
Abe T, Matsumoto K, Sanno N, Osamura Y. (1995). Lymphocytic hypophysitis: case report. <strong>Neuro</strong>surgery<br />
36:1016–1019.<br />
Aguglia U, Gambarelli D, Farnarier G, Quattrone A. (1991). Different susceptibilities of the geniculate and<br />
extrageniculate visual pathways to human Creutzfeldt-Jacob disease (a comb<strong>in</strong>ed neurophysiologicalneuropathological<br />
study). Electroencephalogr Cl<strong>in</strong> <strong>Neuro</strong>physiol 78:413–423.<br />
Aksamit AJ, Preissner CM, Homburger A. (2001). Quantitation of 14-3-3 and neuron-specific enolase prote<strong>in</strong>s <strong>in</strong><br />
CSF <strong>in</strong> Creutzfeldt-Jakob disease. <strong>Neuro</strong>logy 57:728–730.<br />
<strong>An</strong>derson JR, <strong>An</strong>toun N, Burnet N, et al. (1999). <strong>Neuro</strong>logy of the pituitary gland. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
66:703–721.<br />
Arita K, Sugiyama K, Tom<strong>in</strong>aga A, Yamasaki F. (2001). Intrasellar rhabdomyosarcoma: case report. <strong>Neuro</strong>surgery<br />
48:677–680.<br />
Aub<strong>in</strong> MJ, Hardy J, Comtois R. (1997). Primary sellar haemorrhagic melanoma: case report and review of the<br />
literature. Br J <strong>Neuro</strong>surg 11:80–83.<br />
Baeesa SS, Benoit BG. (1999). Solitary metastasis of breast carc<strong>in</strong>oma <strong>in</strong> the optic chiasm. Br J <strong>Neuro</strong>surg<br />
13:319–321.<br />
Bahn MM, Kado DK, L<strong>in</strong> W, Pearlman AL. (1997). Bra<strong>in</strong> magnetic resonance diffusion abnormalities <strong>in</strong><br />
Creutzfeldt-Jacob disease. Arch <strong>Neuro</strong>l 54:1411–1415.<br />
Bahn MM, Parchi P. (1999). Abnormal diffusion-weighted magnetic resonance images <strong>in</strong> Creutzfeldt-Jakob<br />
disease. Arch <strong>Neuro</strong>l 56:577–583.<br />
Bartolomei J, Wecht DA, Chaloupka J, et al. (1998). Occipital lobe vascular malformations: prevalence of visual<br />
field deficits and prognosis after therapeutic <strong>in</strong>tervention. <strong>Neuro</strong>surgery 43:415–423.<br />
Barton JJS. (2001). Bilateral sectoranopia from probable osmotic demyel<strong>in</strong>ation. <strong>Neuro</strong>logy 57:2318–2319.<br />
Bashir K, Elble RJ, Ghobrial M, Struble RG. (1998). Hemianopsia <strong>in</strong> dementia with Lewy bodies. Arch <strong>Neuro</strong>l<br />
55:1132–1135.<br />
Beiran I, Krasnitz I, Zimhoni-Eibsitz M, et al. (2000). Paediatric chiasmal neuritis—typical of post-Epste<strong>in</strong>-Barr<br />
virus <strong>in</strong>fection? Acta Ophthalmol 78:226–227.<br />
Belden JR, Caplan LR, Pess<strong>in</strong> MS, Kwan E. (1999). Mechanism and cl<strong>in</strong>ical features of posterior border-zone<br />
<strong>in</strong>farcts. <strong>Neuro</strong>logy 53:1312–1318.<br />
Beressi N, Cohen R, Beressi J-P, et al. (1994). Pseudotumoral lymphocytic hypophysitis successfully treated by<br />
corticosteroid alone: first case report. <strong>Neuro</strong>surgery 35:505–508.<br />
Bills DC, Meyer FB, Laws ER, et al. (1993). A retrospective analysis of pituitary apoplexy. <strong>Neuro</strong>surgery 33:602–609.
Visual Field Defects 209<br />
Biousse V, Newman NJ, Carroll C, et al. (1998). Visual fields <strong>in</strong> patients with posterior Gpi pallidotomy. <strong>Neuro</strong>logy<br />
50:258–265.<br />
Biousse V, Newman NJ, Oyesiku NM. (2001). Precipitat<strong>in</strong>g factors <strong>in</strong> pituitary apoplexy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 71:542–545.<br />
Blake PY, Miller NR. (1999). Progressive bilateral homonymous visual field defects caused by a left hemispheral<br />
arteriovenous malformation: resolution after embolization. <strong>Neuro</strong>-Ophthalmology 21:17–23.<br />
Bogdanovic MD, Plant GT. (2000). Chiasmal compression due to obstructive hydrocephalus. J <strong>Neuro</strong>-ophthalmol<br />
20:266–267.<br />
Borchert MS, Lessell S, Hoyt WF. (1996). Hemifield slide diplopia from altitud<strong>in</strong>al visual field defects. J <strong>Neuro</strong>ophthalmol<br />
16:107–109.<br />
Borruat F-X, Maeder P. (1995). Sectoranopia after head trauma: evidence of lateral geniculate body lesion on MRI.<br />
<strong>Neuro</strong>logy 45:590–592.<br />
Borruat F-X, Siatkowski RM, Schatz NJ, Glaser JS. (1993). Congruous quadrantanopia and optic radiation lesion.<br />
<strong>Neuro</strong>logy 43:1430–1432.<br />
Bourekas EC, Tzalonikou M, Christofordis GA. (2000). Case 1. Cavernous hemangioma of the optic chiasm. AJR<br />
175:888; 891.<br />
Brazis PW, Lee AG, Graff-Radford N, et al. (2000). Homonymous visual field defects <strong>in</strong> patients without<br />
correspond<strong>in</strong>g structural lesions on neuroimag<strong>in</strong>g. J <strong>Neuro</strong>-Ophthalmol 20:92–96.<br />
Brazis PW, Masdeu JC, Biller J. (2001). Localization <strong>in</strong> <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>logy. 4th ed. Philadelphia, Lipp<strong>in</strong>cott Williams<br />
& Wilk<strong>in</strong>s, pp. 453–521.<br />
Burkhard PR, Sanchez, J-C, Landis T, Hochstrasser DF. (2001). CSF detection of the 14-3-3 prote<strong>in</strong> <strong>in</strong> unselected<br />
patients with dementia. <strong>Neuro</strong>logy 56:1528–1533.<br />
Caplan LR. (1990). Visual perception abnormalities. Presented at the 42nd annual meet<strong>in</strong>g of the American<br />
Academy of <strong>Neuro</strong>logy, Miami, Florida.<br />
Carlow TJ. (1995). Functional hemianopsia: identified with Fresnel prisms and quantitative perimetry. Presented<br />
at the North American <strong>Neuro</strong>-Ophthalmology Society meet<strong>in</strong>g, Tucson, Arizona.<br />
Carter JE, O’Connor P, Shacklett D, Rosenberg M. (1985). Lesions of the optic radiations mimick<strong>in</strong>g lateral<br />
geniculate nucleus visual field defects. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 48:982–988.<br />
Carter K, Lee AG, Tang RA, et al. (1998). <strong>Neuro</strong>-ophthalmologic complications of s<strong>in</strong>us surgery. <strong>Neuro</strong><strong>ophthalmology</strong><br />
19:75–82.<br />
Celesia GG, Brigell MG, Vaphiades MS. (1997). Hemianopic anosognosia. <strong>Neuro</strong>logy 49:88–97.<br />
Chang GY, Keane JR. (2001). Visual loss <strong>in</strong> cysticercosis: analysis of 23 patients. <strong>Neuro</strong>logy 57:545–548.<br />
Chapman T, McKeel DW Jr, Morris JC. (2000). Mislead<strong>in</strong>g results with the 14-3-3 assay for the diagnosis of<br />
Creutzfeldt-Jakob disease. <strong>Neuro</strong>logy 55:1396–1397.<br />
Charteris DG, Cullen JF. (1996). B<strong>in</strong>asal field defects <strong>in</strong> primary empty sella syndrome. J <strong>Neuro</strong>-ophthalmol<br />
16:110–114.<br />
Chavis PS, Al-Hazmi A, Clunie D, Hoyt WF. (1997). Temporal crescent syndrome with magnetic resonance<br />
correlation. J <strong>Neuro</strong>-Ophthalmol 17:151–155<br />
Chun BB, Lee AG, Coughl<strong>in</strong> WF, et al. (1998). Unusual presentation of sellar arachnoid cyst. J <strong>Neuro</strong>-Ophthalmol<br />
18:246–249.<br />
Cirak B, Unal O, Arslan H, C<strong>in</strong>ai A. (2000). Chiasmatic glioblastoma of childhood. A case report. Acta Radiol<br />
41:375–376.<br />
Cobbs CS, Wilson CB. (2001). Intrasellar cavernous hemangioma. J <strong>Neuro</strong>surg 94:520–522.<br />
Czech T, Wolfsberger S, Reitner A, Gorzer H. (1999). Delayed visual deterioration after surgery for pituitary<br />
adenoma. Acta <strong>Neuro</strong>chir 141:45–51.<br />
Danesh-Meyer H, Kubis KC, Wolf MA, Lessell S. (2000). Chiasmopathy? Surv Ophthalmol 44:329–335.<br />
Dom<strong>in</strong>go Z, de Villiers JC. (1993). Post-traumatic chiasmatic disruption. Br J <strong>Neuro</strong>surg 7:141–147.<br />
Donahue SP, Kardon RH, Thompson HS. (1995). Hourglass-shaped visual fields as a sign of bilateral lateral<br />
geniculate myel<strong>in</strong>olysis. Am J Ophthalmol 119:378–380.<br />
Duru S, Ceylan S, Ceylan S. (1999). Optic chiasm diastasis <strong>in</strong> a pituitary tumor. Case illustration. J <strong>Neuro</strong>surg<br />
90:363.<br />
Egan RA, Shults WT, So N, et al. (2000). Visual field deficits <strong>in</strong> conventional anterior temporal lobectomy versus<br />
amygdalohippocampectomy. <strong>Neuro</strong>logy 55:1818–1822.<br />
El-Mahdy W, Powell M. (1998). Transsphenoidal management of 28 symptomatic Rathke’s cleft cysts, with special<br />
reference to visual and hormonal recovery. <strong>Neuro</strong>surgery 42:7–17.<br />
Embil JM, Kramer M, K<strong>in</strong>near S, Bruce Light R. (1997). A bl<strong>in</strong>d<strong>in</strong>g headache. Lancet 349:182.<br />
Fahlbusch R, Honegger J, Paulus W, et al. (1999). Surgical treatment of craniopharyngiomas: experience with 168<br />
patients. J <strong>Neuro</strong>surg 90:237–250.
210 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Felton WL. (1996). Presented at the 28th <strong>An</strong>nual Meet<strong>in</strong>g of the Frank B. Walsh Meet<strong>in</strong>g, Salt Lake City, Utah,<br />
February 10–11.<br />
Fischer EG, DeGirolami U, Suojanen JN. (1994). Reversible visual deficit follow<strong>in</strong>g debulk<strong>in</strong>g of a Rathke’s cleft<br />
cyst: a tethered chiasm? J <strong>Neuro</strong>surg 81:459–462.<br />
Freitag SK, Miller NR, Kosmorsky G. (2000). Visual loss <strong>in</strong> a 42-year-old man. Surv Ophthalmol 44:507–512.<br />
Frohman LP, Frieman BJ, Wolansky L. (2001). Reversible bl<strong>in</strong>dness result<strong>in</strong>g from optic chiasmitis secondary to<br />
systemic lupus erythematosus. J <strong>Neuro</strong>-Ophthalmol 21:18–21.<br />
Galetta SL, Grossman RI. (2000). The representation of the horizontal meridian <strong>in</strong> the primary visual cortex.<br />
J <strong>Neuro</strong>-Ophthalmol 20:89–91.<br />
Galetta SL, Stadmauer EA, Hicks DA, et al. (1991). Reactive lymphohistiocytosis with recurrence <strong>in</strong> the optic<br />
chiasm. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:25–30.<br />
Gray LG, Galetta SL, Schatz NJ. (1998). Vertical and horizontal meridian spar<strong>in</strong>g <strong>in</strong> occipital lobe homonymous<br />
hemianopias. <strong>Neuro</strong>logy 50:1170–1173.<br />
Gray LG, Galetta SL, Siegal T, Schatz NJ. (1997). The central visual field <strong>in</strong> homonymous hemianopia. Evidence<br />
for unilateral foveal representation. Arch <strong>Neuro</strong>l 54:312–317.<br />
Green AJE, Thompson EJ, Stewart GE, et al. (2001). Use of 14-3-3 and other bra<strong>in</strong>-specific prote<strong>in</strong>s <strong>in</strong> CSF <strong>in</strong> the<br />
diagnosis of variant Creutzfeldt-Jacob disease. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 70:744–748.<br />
Greenfield DS, Siatkowski RM, Schatz NJ, Glaser JS. (1996). Bilateral lateral geniculitis associated with severe<br />
diarrhea. Am J Ophthalmol 122:280–281.<br />
Groomm, Kay MD, Vic<strong>in</strong>anza-Adami C, Sant<strong>in</strong>i R. (1997). Optic tract syndrome secondary to metastatic breast<br />
cancer. Am J Ophthalmol 125:115–118.<br />
Grossman M, Galetta SL, Nichols CW, Grossman RI. (1990). Horizontal homonymous sectoral field defect after<br />
ischemic <strong>in</strong>farction of the occipital lobe. Am J Ophthalmol 109:234–236.<br />
Growchowicki M, Vighetto A. (1991). Homonymous horizontal sectoranopia: report of four cases. Br J Ophthalmol 75:624.<br />
Guirgis MF, Lam BL, Falcone SF. (2001). Optic tract compression from dolichoectatic basilar artery. Am J<br />
Ophthalmol 132:282–286.<br />
Hansen HV. (1993). Bilateral <strong>in</strong>ferior altitud<strong>in</strong>al hemianopia. <strong>Neuro</strong>-<strong>ophthalmology</strong> 13:81.<br />
Harden CL, Rosenbaum DH, Daras M. (1991). Hyperglycemia present<strong>in</strong>g with occipital seizures. Epilepsia<br />
32:215–220.<br />
He<strong>in</strong>z GW, Nunery WR, Grossman CB. (1994). Traumatic chiasmal syndrome associated with midl<strong>in</strong>e basilar<br />
skull fractures. Am J Ophthalmol 117:90–96.<br />
Hernandez Echebarria LE, Saiz A, Graus F, et al. (2000). Detection of 14-3-3 prote<strong>in</strong> <strong>in</strong> the CSF of a patient with<br />
Hashimoto’s encephalopathy. <strong>Neuro</strong>logy 54:1539–1540.<br />
Hershenfeld SA, Sharpe JS. (1993). Monocular temporal hemianopia. Br J Ophthalmol 77:422–427.<br />
H<strong>in</strong>chey J, Chaves C, Appignani B, et al. (1996). A reversible posterior leukoencephalopathy syndrome. N Engl<br />
J Med 334:494–500.<br />
Honegger J, Fahlbusch R, Bornemann A, et al. (1997). Lymphocytic and granulomatous hypophysitis: experience<br />
with n<strong>in</strong>e cases. <strong>Neuro</strong>surgery 40:713–723.<br />
Honegger J, Buchfelder M, Fahlbusch R. (1999). Surgical treatment of craniopharyngiomas: endocr<strong>in</strong>ological<br />
results. J <strong>Neuro</strong>surg 90:251–257.<br />
Horton JC. (1997). Wilbrand’s knee of the primate optic chiasm is an artefact of monocular enucleation. Trans Am<br />
Ophthalmol Soc 95:579–609.<br />
Horton JC, Fishman RA. (1994). <strong>Neuro</strong>visual f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> the syndrome of spontaneous <strong>in</strong>tracranial hypotension<br />
from dural cerebrosp<strong>in</strong>al fluid leak. Ophthalmology 101:244–251.<br />
Horton JC, Hoyt WF. (1991a). Quadrantic visual field defects. A hallmark of lesions <strong>in</strong> extrastriate (V2=V3) cortex.<br />
Bra<strong>in</strong> 114:1703–1718.<br />
Horton JC, Hoyt WF. (1991b). The representation of the visual field <strong>in</strong> human striate cortex. A revision of the<br />
classic Holmes map. Arch Ophthalmol 109:816–824.<br />
Horton JC, Landau K, Maeder P, Hoyt WF. (1990). Magnetic resonance imag<strong>in</strong>g of the human lateral geniculate<br />
body. Arch <strong>Neuro</strong>l 47:1201–1206.<br />
Hsich G, Kenney K, Gibbs CJ, et al. (1996). The 14-3-3 bra<strong>in</strong> prote<strong>in</strong> <strong>in</strong> cerebrosp<strong>in</strong>al fluid as a marker for<br />
transmissible spongiform encephalopathy. N Engl J Med 335:924–930.<br />
Hughes TS, Abou-Khalil B, Lav<strong>in</strong> PJM, et al. (1999). Visual field defects after temporal lobe resection.<br />
A prospective quantitative analysis. <strong>Neuro</strong>logy 53:167–172.<br />
Hwang JF, Yau CW, Huang KI, Tasi CY. (1993). Apoplectic optochiasmal syndrome due to <strong>in</strong>tr<strong>in</strong>sic cavernous<br />
hemangioma. Case report. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:232–236.<br />
Ikeda H, Yoshimoto T. (1995). Visual disturbances <strong>in</strong> patients with pituitary adenoma. Acta <strong>Neuro</strong>l Scand<br />
92:157–160.
Visual Field Defects 211<br />
Jabre A, Rosales R, Reed JE, Spatz EL. (1997). Lymphocytic hypophysitis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 63:672–673.<br />
Jacobs DA, Lesser RL, Mourelatos Z, et al. (2001). The Heidenha<strong>in</strong> variant of Creutzfeldt-Jakob disease: cl<strong>in</strong>ical,<br />
pathologic, and neuroimag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs. J <strong>Neuro</strong>-Ophthalmol 21:99–102.<br />
Jacobson DM. (1997). The localiz<strong>in</strong>g value of a quadrantanopsia. Arch <strong>Neuro</strong>l 54:401–404.<br />
Jacobson, DM. (1999). Symptomatic compression of the optic nerve by the carotid artery. <strong>Cl<strong>in</strong>ical</strong> profile of 18<br />
patients with 24 affected eyes identified by magnetic resonance imag<strong>in</strong>g. Ophthalmology 106:1994–2004.<br />
Job OM, Schatz NJ, Glaser JS. (1999). Visual loss with Langerhans cell histiocytosis: multifocal central nervous<br />
system <strong>in</strong>volvement. J <strong>Neuro</strong>-Ophthalmol 19:49–53.<br />
Karanjia N, Jacobson DM. (1999). Compression of the prechiasmatic optic nerve produces a junctional scotoma.<br />
Am J Ophthalmol 128:256–258.<br />
Karp BI, Yang JC, Khorsand M, et al. (1996). Multiple cerebral lesions complicat<strong>in</strong>g therapy with <strong>in</strong>terleuk<strong>in</strong>-2.<br />
<strong>Neuro</strong>logy 47:417–424.<br />
Keane JR. (1998). Patterns of hysterical hemianopia. <strong>Neuro</strong>logy 51:1230–1231.<br />
Kerkhoff G. (2000). <strong>Neuro</strong>visual rehabilitation: recent developments and future directions. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 68:691–706.<br />
Kerkhoff G, MunB<strong>in</strong>ger U, Meier EK. (1994). <strong>Neuro</strong>visual rehabilitation <strong>in</strong> cerebral bl<strong>in</strong>dness. Arch <strong>Neuro</strong>l<br />
51:474–481.<br />
Kerkhoff G, MunB<strong>in</strong>ger U, Haaf E, et al. (1992). Rehabilitation of homonymous scotomata <strong>in</strong> patients with<br />
postgeniculate damage of the visual system: saccadic compensation tra<strong>in</strong><strong>in</strong>g. Restorative <strong>Neuro</strong>l <strong>Neuro</strong>sci<br />
4:245–254.<br />
Kerrison JB, Lee AG, We<strong>in</strong>ste<strong>in</strong> JM. (1997). Acute loss of vision dur<strong>in</strong>g pregnancy due to a suprasellar mass. Surv<br />
Ophthalmol 41:402–408.<br />
Kerrison JB, Lynn MJ, Baer CA, et al. (2000). Stages of improvement <strong>in</strong> visual fields after pituitary tumor resection.<br />
Am J Ophthalmol 130:813–820.<br />
K<strong>in</strong>jo T, al-Mefty O, Ciric I. (1995). Diaphragma sellae men<strong>in</strong>giomas. <strong>Neuro</strong>surgery 36:1082–1092.<br />
Kosmorsky G, Lancione RR Jr. (1998). When fight<strong>in</strong>g makes you see black holes <strong>in</strong>stead of stars. J <strong>Neuro</strong>-<br />
Ophthalmol 18:255–257.<br />
Kosmorsky GS, Straga JM. (1997). A descent th<strong>in</strong>g to do for the chiasm. J <strong>Neuro</strong>-ophthalmol 17:53–56.<br />
Kristof RA, Van Roost D, Kl<strong>in</strong>gmüller D, et al. (1999). Lymphocytic hypophysitis: non-<strong>in</strong>vasive diagnosis and<br />
treatment by high dose methylprednisolone pulse therapy? J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 67:398–402.<br />
Kropp S, Schulz-Schaeffer WJ, F<strong>in</strong>kenstaedt M, et al. (1999). The Heidenha<strong>in</strong> variant of Creutzfeldt-Jacob disease.<br />
Arch <strong>Neuro</strong>l 56:55–61.<br />
Kuperschmidt H, Bont A, Schnorf H, et al. (1995). Transient cortical bl<strong>in</strong>dness and bioccipital bra<strong>in</strong> lesions <strong>in</strong> two<br />
patients with acute <strong>in</strong>termittent porphyria. <strong>An</strong>n Intern Med 123:598–600.<br />
Kupersmith MJ, Berenste<strong>in</strong> A, Nelson PK, et al. (1999). Visual symptoms with dural arteriovenous malformations<br />
dra<strong>in</strong><strong>in</strong>g <strong>in</strong>to occipital ve<strong>in</strong>s. <strong>Neuro</strong>logy 52:156–162.<br />
Kupersmith MJ, Rosenberg C, Kle<strong>in</strong>berg D. (1994). Visual loss <strong>in</strong> pregnant women with pituitary adenomas. <strong>An</strong>n<br />
Intern Med 121:473–477.<br />
Kupersmith MJ, Vargas ME, Yashar A, et al. (1996). Occipital arteriovenous malformations: visual disturbances<br />
and presentation. <strong>Neuro</strong>logy 46:953–957.<br />
Lakhanpal A, Selhorst JB. (1990). Bilateral altitud<strong>in</strong>al visual fields. <strong>An</strong>n Ophthalmol 22:112–117.<br />
Landau K, Wichmann W, Valavanis A. (1995). The miss<strong>in</strong>g temporal crescent. Am J Ophthalmol 119:345–349.<br />
Lee AG, Sforza PD, Fard AK, et al. (1998). Pituitary adenoma <strong>in</strong> children. J <strong>Neuro</strong>-Ophthalmol 18:102–105.<br />
Lee AG, Tang RA, Roberts D, et al. (2001). Primary central nervous system lymphoma <strong>in</strong>volv<strong>in</strong>g the optic chiasm<br />
<strong>in</strong> AIDS. J <strong>Neuro</strong>-Ophthalmol 21:95–98.<br />
Lee BL, Holland GN, Glasgow BJO. (1996). Chiasmal <strong>in</strong>farction and sudden bl<strong>in</strong>dness caused by mucormycosis <strong>in</strong><br />
AIDS and diabetes mellitus. Am J Ophthalmol 122:895–896.<br />
Lee J-H, Laws ER, Guthrie BL, et al. (1994). Lymphocytic hypophysitis: occurrence <strong>in</strong> two men. <strong>Neuro</strong>surgery<br />
34:159–163.<br />
Leff AP, Scott SK, Crewes H, et al. (2000). Impaired read<strong>in</strong>g <strong>in</strong> patients with right hemianopia. <strong>An</strong>n <strong>Neuro</strong>l<br />
47:171–178.<br />
Lemstra AW, van Meegen MT, Vreyl<strong>in</strong>g JP, et al. (2000). 14-3-3 test<strong>in</strong>g <strong>in</strong> diagnos<strong>in</strong>g Creutzfeldt-Jacob disease. A<br />
prospective study <strong>in</strong> 112 patients. <strong>Neuro</strong>logy 55:514–516.<br />
Lepore FE. (2001). The preserved temporal crescent: the cl<strong>in</strong>ical implications of an ‘‘endangered’’ f<strong>in</strong>d<strong>in</strong>g.<br />
<strong>Neuro</strong>logy 57:1918–1921.<br />
Lepore FE. (1998). Visual deficits <strong>in</strong> alexia without agraphia. <strong>Neuro</strong>-Ophthalmology 19:1–6.<br />
Liu GT, Galetta SL. (1997). Homonymous hemifield loss <strong>in</strong> childhood. <strong>Neuro</strong>logy 49:1748–1749.
212 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Liu GT, Galetta SL, Rorke LB, et al. (1996). Gangliogliomas <strong>in</strong>volv<strong>in</strong>g the optic chiasm. <strong>Neuro</strong>logy 46:1669–1673.<br />
Luco C, Hoppe A, Schweitzer M, et al. (1992). Visual field defects <strong>in</strong> vascular lesions of the lateral geniculate body.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 55:12–15.<br />
Mart<strong>in</strong> TJ. (1998). Threshold perimetry of each eye with both eyes open <strong>in</strong> patients with monocular functional<br />
(nonorganic) and organic vision loss. Am J Ophthalmol 125:857–864.<br />
McCowen KC, Glickman JN, Black PM, et al. (1999). Gangliocytoma masquerad<strong>in</strong>g as a prolact<strong>in</strong>oma. Case<br />
report. <strong>Neuro</strong>logy 91:490–495.<br />
McFadzean R, Brosnahan D, Hadley D, Mutlukan E. (1994). Representation of the visual field <strong>in</strong> the occipital<br />
striate cortex. Br J Ophthalmol 78:185–190.<br />
McFadzean RM, Hadley DM. (1997). Homonymous quadrantanopia respect<strong>in</strong>g the horizontal meridian. A feature<br />
of striate and extrastriate cortical disease. <strong>Neuro</strong>logy 49:1741–1746.<br />
McFadzean RM, McIlwa<strong>in</strong>e GG, McLellan D. (1990). Hodgk<strong>in</strong>’s disease at the optic chiasm. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
10:248–254.<br />
McHenry JG, Spoor TC. (1993). Chiasmal compression from fat pack<strong>in</strong>g after transsphenoidal resection of<br />
<strong>in</strong>trasellar tumor <strong>in</strong> two patients. Am J Ophthalmol 116:253.<br />
Miele DL, Odel JG, Behrens MM, et al. (2000). Functional bitemporal quadrantopia and the multifocal visual<br />
evoked potential. J <strong>Neuro</strong>-Ophthalmol 20:159–162.<br />
Mikelberg FS, Yidegiligne HM. (1993). Axonal loss <strong>in</strong> band atrophy of the optic nerve <strong>in</strong> craniopharyngioma: a<br />
quantitative analysis. Can J Ophthalmol 28:69–71.<br />
Miki A, Nakajima T, Fujita M, et al. (1996). Functional magnetic resonance imag<strong>in</strong>g <strong>in</strong> homonymous hemianopsia.<br />
Am J Ophthalmol 121:258–266.<br />
Miller NR, Newman NJ. (1998). Topical diagnosis of lesions <strong>in</strong> the visual sensory pathway. In: Miller NR,<br />
Newman NJ, eds. Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 5th ed. Baltimore, Williams & Wilk<strong>in</strong>s,<br />
pp. 237–386.<br />
Mittal S, Farmer P, Kal<strong>in</strong>a P, et al. (2002). Correlation of diffusion-weighted magnetic resonance imag<strong>in</strong>g with<br />
neuropathology <strong>in</strong> Creutzfeldt-Jakob disease. Arch <strong>Neuro</strong>l 59:128–134.<br />
Miyairi Y, Tada T, Tanaka Y, et al. (2000). <strong>An</strong>aplastic astrocytoma <strong>in</strong>vad<strong>in</strong>g the optic chiasm through the optic<br />
pathway. Case illustration. J <strong>Neuro</strong>surg 93:716.<br />
Mojon DS, Odel JG, Rios RJ, Hirano M. (1997). Pituitary adenoma revealed by paracentral junctional scotoma of<br />
Traquair. Ophthalmologica 211:104–108.<br />
Molia L, W<strong>in</strong>terkorn JMS, Schneider SJ. (1996). Hemianopic visual field defects <strong>in</strong> children with <strong>in</strong>tracranial<br />
shunts: report of two cases. <strong>Neuro</strong>surgery 39:599–603.<br />
Morita A, Meyer FB, Laws ER Jr. (1998). Symptomatic pituitary metastases. J <strong>Neuro</strong>surg 89:69–73.<br />
Moster ML, Galetta SI, Schatz NJ. (1996). Physiologic functional imag<strong>in</strong>g <strong>in</strong> ‘‘functional’’ visual loss. Surv<br />
Ophthalmol 40:395–399.<br />
Murphy MA, Grosof DH, Hart WM Jr. (1997). Congenital optic tract syndrome: magnetic resonance imag<strong>in</strong>g and<br />
scann<strong>in</strong>g laser ophthalmoscopy f<strong>in</strong>d<strong>in</strong>gs. J <strong>Neuro</strong>-Ophthalmol 17:226–230.<br />
Na DL, Suh CK, Choi SH, et al. (1999). Diffusion-weighted magnetic resonance imag<strong>in</strong>g <strong>in</strong> probable Creutzfeldt-<br />
Jacob disease. A cl<strong>in</strong>ical-anatomic correlation. Arch <strong>Neuro</strong>l 56:951–957.<br />
Naik RG, Amm<strong>in</strong>i A, Shah P, et al. (1994). Lymphocytic hypophysitis. Case report. J <strong>Neuro</strong>surg 80:925–927.<br />
Neau J-P, Bogousslavsky J. (1996). The syndrome of posterior choroidal artery territory <strong>in</strong>farction. <strong>An</strong>n <strong>Neuro</strong>l<br />
39:779–788.<br />
Newman NJ, Lessell S, W<strong>in</strong>terkorn MS. (1991). Optic chiasmal neuritis. <strong>Neuro</strong>logy 41:1203–1210.<br />
Nishioka H, Ito H, Fuskushima C. (1997). Recurrent lymphocytic hypophysitis. <strong>Neuro</strong>surgery 41:684–687.<br />
Ormerod LD, Rhodes RH, Gross SA, et al. (1996). Ophthalmologic manifestations of acquired immune deficiency<br />
syndrome-associated progressive multifocal leukoencephalopathy. Ophthalmology 103:899–906.<br />
Pakzaban P, Westmark K, Westmark R. (2000). Chiasmal apoplexy due to hemorrhage from a pituitary adenoma<br />
<strong>in</strong>to the optic chiasm: case report. <strong>Neuro</strong>surgery 46:1511–1514.<br />
Pess<strong>in</strong> MS, Kwan ES, DeWitt LD, et al. (1987a). Posterior cerebral artery stenosis. <strong>An</strong>n <strong>Neuro</strong>l 1:85–89.<br />
Pess<strong>in</strong> MS, Lathi ES, Cohen MB, et al. (1987b). <strong>Cl<strong>in</strong>ical</strong> features and mechanism of occipital <strong>in</strong>farction. <strong>An</strong>n <strong>Neuro</strong>l<br />
21:290–299.<br />
Peter M, DeTribolet N. (1995). Visual outcome after transsphenoidal surgery for pituitary adenomas. Br J<br />
<strong>Neuro</strong>surg 9:151–157.<br />
Petruson B, Jakobsson KE, Elfverson J, Bengtsson BA. (1995). Five-year follow-up of nonsecret<strong>in</strong>g pituitary<br />
adenomas. Arch Otolaryngol Head Neck Surg 121:317–322.<br />
Pomeranz HD, Henson JW, Lessell S. (1998). Radiation-associated cerebral bl<strong>in</strong>dness. Am J Ophthalmol<br />
126:609–611.
Visual Field Defects 213<br />
Pomeranz HD, Lessell S. (1999). A hereditary chiasmal optic neuropathy. Arch Ophthalmol 117:128–131.<br />
Poon A, McNeill P, Harper A, O’Day J. (1995). Patterns of visual loss associated with pituitary macroadenomas.<br />
Aust NZ J Ophthalmol 23:107–115.<br />
Poser S, Mollenhauer B, Kraub A, et al. (1999). How to improve the cl<strong>in</strong>ical diagnosis of Creutzfeldt-Jacob disease.<br />
Bra<strong>in</strong> 122:2345–2351.<br />
Rao GP, Blyth CP, Jeffreys R. (1995). Ophthalmic manifestations of Rathke’s cleft cysts. Am J Ophthalmol 119:86–91.<br />
Rizzo JF III. (1993). Occult ret<strong>in</strong>al and choroidal vascular disease. The value of timed and directed fluoresce<strong>in</strong><br />
angiography. Ophthalmology 100:1407–1416.<br />
Rossi LN, Pastor<strong>in</strong>o G, Scotti G, et al. (1994). Early diagnosis of optic glioma <strong>in</strong> children with neurofibromatosis<br />
type 1. Childs Nerv Syst 10:426–429.<br />
Rossi PW, Keyfets S, Red<strong>in</strong>g MJ. (1990). Fresnel prisms improve visual perception <strong>in</strong> stroke patients with<br />
homonymous hemianopia or unilateral visual neglect. <strong>Neuro</strong>logy 40:1597–1599.<br />
Saiz A, Graus F, Dalmau J, et al. (1999). Detection of 14-3-3 bra<strong>in</strong> prote<strong>in</strong> <strong>in</strong> the cerebrosp<strong>in</strong>al fluid of patients with<br />
paraneoplastic neurological disorders. <strong>An</strong>n <strong>Neuro</strong>l 46:774–777.<br />
Saw<strong>in</strong> PD, Follett KA, Wen BC, Laws ER Jr. (1996). Symptomatic <strong>in</strong>trasellar hemangioblastoma <strong>in</strong> a child treated<br />
with subtotal resection and adjuvant radiosurgery. Case report. J <strong>Neuro</strong>surg 84:1046–1050.<br />
Schroter A, Zerr I, Henkel K, et al. (2000). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> the cl<strong>in</strong>ical diagnosis of Creutzfeldt-<br />
Jacob disease. Arch <strong>Neuro</strong>l 57:1751–1757.<br />
Shults WT, Hamby S, Corbett JJ, et al. (1993). <strong>Neuro</strong>-ophthalmic complications of <strong>in</strong>tracranial catheters.<br />
<strong>Neuro</strong>surgery 33:135–138.<br />
Siatkowski RM, Scott IU, Verm AM, et al. (2001). Optic neuropathy and chiasmopathy <strong>in</strong> the diagnosis of systemic<br />
lupus erythematosus. J <strong>Neuro</strong>-ophthalmol 21:193–198.<br />
Slav<strong>in</strong> ML. (1990a). Acute homonymous field loss: really a diagnostic dilemma. Surv Ophthalmol 34:399–407.<br />
Slav<strong>in</strong> ML. (1990b). Bitemporal hemianopia associated with dolichoectasia of the <strong>in</strong>tracranial carotid arteries. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 10:80–81.<br />
Slav<strong>in</strong> ML, Lam BL, Decker RE, et al. (1993). Chiasmal compression from fat pack<strong>in</strong>g after transsphenoidal<br />
resection of <strong>in</strong>trasellar tumor <strong>in</strong> two patients. Am J Ophthalmol 115:368–371.<br />
Smith JL. (1981). New pearls check list. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 1:78.<br />
Steg RE, Kess<strong>in</strong>ger A, Wszolek ZK. (1999). Case report. Cortical bl<strong>in</strong>dness and seizures <strong>in</strong> a patient receiv<strong>in</strong>g<br />
<strong>FK</strong>506 after bone marrow transplantation. Bone Marrow Transplant 23:959–962.<br />
Stelmach M, O’Day J. (1991). Rapid change <strong>in</strong> visual fields associated with suprasellar lymphocytic hypophysitis.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:19–24.<br />
Thodou E, Asa SL, Kontogeorgos G, et al. (1995). <strong>Cl<strong>in</strong>ical</strong> case sem<strong>in</strong>ar: lymphocytic hypophysitis: cl<strong>in</strong>icopathologic<br />
f<strong>in</strong>d<strong>in</strong>gs. J Cl<strong>in</strong> Endocr<strong>in</strong>ol MeTab 80:2302–2311.<br />
Thompson JC, Kosmorsky GS, Ellis BD. (1996). Fields of dreamers and dreamed-up fields. Functional and fake<br />
perimetry. Ophthalmology 103:117–125.<br />
Trick GL, Trick LR, Morris P, Wolf M. (1995). Visual field loss <strong>in</strong> senile dementia of the Alzheimer’s type. <strong>Neuro</strong>logy<br />
45:68–74.<br />
Trobe JD, Glaser JS. (1983). The Visual Fields Manual: A Practical Guide to Test<strong>in</strong>g and Interpretation. Ga<strong>in</strong>esville,<br />
Triad, p. 176.<br />
Tubridy N, Molloy J, Saunders D, et al. (2001). Postpartum pituitary hypophysitis. J <strong>Neuro</strong>-Ophthalmol 21:106–108.<br />
Vargas ME, Kupersmith MJ, Sav<strong>in</strong>o PJ, et al. (1995). Homonymous field defect as the first manifestation of<br />
Creutzfeldt-Jacob disease. Am J Ophthalmol 119:497–504.<br />
Vargas ME, Kupersmith MJ, Setton, A, et al. (1994). Endovascular treatment of giant aneurysm which cause visual<br />
loss. Ophthalmology 101:1091–1098.<br />
Voelker JL, Campbell RL, Muller J. (1991). <strong>Cl<strong>in</strong>ical</strong>, radiographic, and pathological features of symptomatic<br />
Rathke’s cleft cysts. J <strong>Neuro</strong>surg 74:535–544.<br />
Wang A-G, Liu R-S, Liu J-H, et al. (1999). Positron emission tomography scan <strong>in</strong> cortical visual loss <strong>in</strong> patients<br />
with organophosphate <strong>in</strong>toxication. Ophthalmology 106:1287–1291.<br />
Warren FE, Vargas ME, Seidman I, Kupersmith MJ. (1992). Homonymous field defect <strong>in</strong> an HIV negative, at risk<br />
<strong>in</strong>dividual. Presented at the 24th <strong>An</strong>nual Frank B. Walsh Society meet<strong>in</strong>g, Los <strong>An</strong>geles, California, February<br />
28–29.<br />
We<strong>in</strong> F, Miller NR, Vaphiades MS. (2000). <strong>An</strong> unusual homonymous visual field defect. Surv Ophthalmol<br />
44:324–328.<br />
Wilhelm H, Grodd W, Schiefer U, Zrenner E. (1993). Uncommon chiasmal lesions: demyel<strong>in</strong>at<strong>in</strong>g disease,<br />
vasculitis, and cobalam<strong>in</strong> deficiency. Germ J Ophthalmol 2:234–240.
214 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Wilhelm H, Wilhelm B, Petersen D, et al. (1996). Relative afferent pupillary defects <strong>in</strong> patients with geniculate and<br />
retrogeniculate lesions. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:219–224.<br />
Wolansky LJ, Shaderowsky PD, Sander R, et al. (1997). Optic chiasmal compression by venous aneurysm:<br />
magnetic resonance imag<strong>in</strong>g diagnosis. J <strong>Neuro</strong>imag<strong>in</strong>g 7:46–47.<br />
Wong AMF, Sharpe JA. (1999). Representation of the visual field <strong>in</strong> the human occipital cortex. A magnetic<br />
resonance imag<strong>in</strong>g and perimetric correlation. <strong>Neuro</strong>logy 117:208–217.<br />
Xu S, Meyer D, Yoser S, et al. (2001). Pattern visual evoked potential <strong>in</strong> the diagnosis of functional visual loss.<br />
Ophthalmology 108:76–81.<br />
Yamamoto M, Jimbo M, Ide M, et al. (1993). Recurrence of symptomatic Rathke’s cleft cyst: a case report. Surg<br />
<strong>Neuro</strong>l 39:263–268.<br />
Zerr I, Bodemer M, Gefeller O, et al. (1998). Detection of 14-3-3 prote<strong>in</strong> <strong>in</strong> the cerebrosp<strong>in</strong>al fluid supports the<br />
diagnosis of Creutzfeldt-Jakob disease. <strong>An</strong>n <strong>Neuro</strong>l 43:32-40.<br />
Zerr I, Bodemer M, Racker S, et al. (1995). Cerebrosp<strong>in</strong>al concentration of neuron-specific enolase <strong>in</strong> diagnosis of<br />
Creutzfeldt-Jacob disease. Lancet 345:1609–1610.<br />
Zerr I, Pocchiari M, Coll<strong>in</strong>s S, et al. (2000a). <strong>An</strong>alysis of EEG and CSF 14-3-3 prote<strong>in</strong>s as aids to the diagnosis of<br />
Creutzfeldt-Jakob disease. <strong>Neuro</strong>logy 55:811–815.<br />
Zerr I, Schulz-Schaeffer WJ, Giese A, et al. (2000b). Current cl<strong>in</strong>ical diagnosis <strong>in</strong> Creutzfeldt-Jakob disease:<br />
identification of uncommon variants. <strong>An</strong>n <strong>Neuro</strong>l 48:323–329.
10 r<br />
Diplopia<br />
In this chapter we divide diplopia <strong>in</strong>to several categories: monocular versus b<strong>in</strong>ocular<br />
and horizontal versus vertical. The evaluation for diplopia is outl<strong>in</strong>ed <strong>in</strong> Figure 10–1.<br />
Is the Diplopia Monocular?<br />
Monocular diplopia can usually be diagnosed by the history alone. Diplopia that is<br />
present monocularly rema<strong>in</strong>s present despite cover<strong>in</strong>g the fellow eye and then disappears<br />
when the <strong>in</strong>volved eye is occluded. It may occur unilaterally or bilaterally. The<br />
second image is often described as a less clear and partially superimposed ‘‘ghost<br />
image’’ or a ‘‘halo’’ on the first image. A p<strong>in</strong>hole may dramatically reduce the patient’s<br />
symptoms. Patients without a clear history of monocular diplopia can be asked to keep<br />
a diary of their symptoms with specific <strong>in</strong>structions to document the details for review<br />
at a future visit. A p<strong>in</strong>hole can be given to patients with suspected monocular diplopia<br />
to try at home. This ‘‘take home’’ p<strong>in</strong>hole can be made <strong>in</strong> the office out of a bus<strong>in</strong>ess<br />
card or a note card us<strong>in</strong>g a pen or pencil to make a small-diameter hole. The patient can<br />
then try the p<strong>in</strong>hole at home dur<strong>in</strong>g the episode of diplopia to test if it resolves the<br />
symptoms.<br />
Monocular diplopia usually implies a problem with<strong>in</strong> the eye itself and may respond<br />
to refraction, artificial tear trial, or contact lens trial. Table 10–1 lists the ocular causes of<br />
monocular diplopia. Monocular diplopia usually does not require any further neuroophthalmologic<br />
evaluation.<br />
<strong>An</strong>other less common form of monocular diplopia is cerebral polyopia (Jones, 1999).<br />
Cerebral polyopia usually can be dist<strong>in</strong>guished from monocular diplopia due to ocular<br />
disease because all of the images are seen with equal clarity, the multiple images do not<br />
resolve with a p<strong>in</strong>hole, and the images are unchanged <strong>in</strong> appearance whether the<br />
patient is view<strong>in</strong>g b<strong>in</strong>ocularly or monocularly. Some patients see only two images,<br />
whereas others may see many or even hundreds of images occurr<strong>in</strong>g <strong>in</strong> a grid-like<br />
pattern (‘‘entomopia’’ or ‘‘<strong>in</strong>sect eye’’) (Lopez, 1993). Some patients experience the<br />
215
216 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 10–1. Evaluation of diplopia.
polyopia only <strong>in</strong> certa<strong>in</strong> positions of gaze. Patients with cerebral polyopia often have<br />
associated signs of occipital or parieto-occipital region damage, such as homonymous<br />
visual field defects, difficulty with visually guided reach<strong>in</strong>g, cerebral achromatopsia or<br />
dyschromatopsia, object agnosia, and abnormal visual afterimages. These patients<br />
require neuroimag<strong>in</strong>g (e.g., magnetic resonance imag<strong>in</strong>g, MRI), to <strong>in</strong>vestigate the<br />
etiology of the polyopia. Cerebral <strong>in</strong>farction is the most common etiology, although<br />
cerebral polyopia may also occur with tumors, multiple sclerosis, encephalitis, seizures,<br />
and with migra<strong>in</strong>e (Jones, 1999).<br />
Is the Diplopia B<strong>in</strong>ocular?<br />
Table 10–1. Ocular Causes of Monocular Diplopia<br />
Refractive error (Woods, 1996) <strong>in</strong>clud<strong>in</strong>g astigmatism<br />
Poorly fitt<strong>in</strong>g contact lens<br />
Corneal abnormalities<br />
Keratoconus<br />
Corneal surface abnormality<br />
Tear film disorders <strong>in</strong>clud<strong>in</strong>g dry eye<br />
Refractive surgery<br />
Corneal transplant<br />
Lid abnormalities (e.g., chalazion, lid position abnormalities)<br />
Iris abnormalities (e.g., iridotomy=iridectomy, miotic pupils)<br />
Lens abnormalities<br />
Cataract<br />
Subluxation or dislocation<br />
Intraocular lens (e.g., position<strong>in</strong>g holes, decentered lens)<br />
Ret<strong>in</strong>al abnormalities (e.g., epiret<strong>in</strong>al membrane, scar)<br />
A history of b<strong>in</strong>ocular diplopia is associated with ocular misalignment. Identification of<br />
specific cl<strong>in</strong>ical signs and symptoms may allow identification of specific etiologies for<br />
the diplopia.<br />
Is the Diplopia Transient or Persistent?<br />
Diplopia 217<br />
Diplopia may be noted only <strong>in</strong> certa<strong>in</strong> fields of gaze (e.g., only on look<strong>in</strong>g down <strong>in</strong> some<br />
patients with fourth nerve palsies) and may fluctuate dur<strong>in</strong>g the day (e.g., diplopia <strong>in</strong><br />
thyroid ophthalmopathy may be more apparent <strong>in</strong> the early morn<strong>in</strong>g). Patients with<br />
truly <strong>in</strong>termittent diplopia may be asymptomatic at exam<strong>in</strong>ation and eye misalignment<br />
may be subtle or not demonstrated. Table 10–2 lists the causes of transient diplopia.<br />
These etiologies are discussed <strong>in</strong> more detail <strong>in</strong> subsequent sections on horizontal and<br />
vertical diplopia.
218 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Is This an Ocular Motor Cranial <strong>Neuro</strong>pathy?<br />
Ocular motor cranial nerve palsies are discussed <strong>in</strong> the chapters on third nerve palsies<br />
(Chapter 11), fourth nerve palsies (Chapter 12), and sixth nerve palsies (Chapter 13).<br />
Is There Evidence for a Restrictive<br />
Ophthalmoplegia Due to Orbital Disease?<br />
Orbital signs, such as proptosis, chemosis, and <strong>in</strong>jection, should be looked for <strong>in</strong><br />
patients with diplopia. Forced ductions may reveal a restrictive component to the<br />
diplopia. Orbital wall fracture, orbital tumors, orbital <strong>in</strong>flammatory disease, or trauma<br />
may result <strong>in</strong> a restrictive strabismus. Orbital imag<strong>in</strong>g is <strong>in</strong>dicated <strong>in</strong> these patients.<br />
Is This Myasthenia Gravis?<br />
Table 10–2. Causes of Transient Diplopia<br />
Transient ischemia<br />
Transient ocular muscle ischemia (e.g., giant cell arteritis)<br />
Vertebrobasilar artery ischemia<br />
Decompensation of preexist<strong>in</strong>g phoria<br />
Ret<strong>in</strong>al hemifield slide phenomena<br />
Myasthenia gravis<br />
Muscle or mechanical<br />
Thyroid ophthalmopathy<br />
Brown’s syndrome<br />
Silent s<strong>in</strong>us syndrome<br />
Intermittent phenomena<br />
Migra<strong>in</strong>e<br />
<strong>Neuro</strong>myotonia<br />
Intermittent or paroxysmal skew deviation<br />
Superior oblique myokymia<br />
Paroxysmal superior rectus and levator palpebrae spasm<br />
Increased <strong>in</strong>tracranial pressure<br />
Multiple sclerosis (days to weeks)<br />
The evaluation and management of myasthenia gravis is discussed <strong>in</strong> Chapter 15. <strong>An</strong>y<br />
patient with pa<strong>in</strong>less, pupil-spar<strong>in</strong>g, nonproptotic ophthalmoplegia or diplopia should<br />
be evaluated for the possible diagnosis of myasthenia gravis.<br />
Is This Thyroid Eye Disease?<br />
Although transient or persistent diplopia and ophthalmoplegia may occur without<br />
other signs of thyroid eye disease, identification of the dist<strong>in</strong>ctive signs of thyroid
disease as described <strong>in</strong> Chapter 16 is essential <strong>in</strong> the evaluation of any patient with<br />
diplopia.<br />
Is This a Supranuclear Process?<br />
In a patient with negative forced ductions and no evidence for restrictive ophthalmoplegia,<br />
the doll’s-head maneuver (rapid forced head movements horizontally<br />
and vertically to activate the vestibulo-ocular reflex) may overcome a supranuclear<br />
ophthalmoplegia (see Chapter 14). Failure of the doll’s-head maneuver to overcome the<br />
ophthalmoplegia suggests an <strong>in</strong>franuclear etiology.<br />
Is the Deviation Vertical or Horizontal?<br />
If there are no dist<strong>in</strong>ctive or obvious signs to <strong>in</strong>dicate diagnosis of a specific etiology for<br />
the diplopia, then the vertical or horizontal nature of the deviation may allow further<br />
classification of the problem.<br />
What Are Phorias and Tropias? How Does the<br />
Exam<strong>in</strong>er Assess Horizontal Eye Muscle<br />
Function?<br />
A phoria is a latent ocular misalignment that is kept <strong>in</strong> check by fusion. Fusion is the<br />
process of merg<strong>in</strong>g images from each eye <strong>in</strong>to a s<strong>in</strong>gle perception. Sensory fusion is the<br />
cortical <strong>in</strong>tegration of two images, whereas motor fusion represents the corrective<br />
movements of the eyes required to ma<strong>in</strong>ta<strong>in</strong> eye alignment on the target of regard.<br />
Breakdown of fusion due to fatigue, stress, or illness may allow a preexist<strong>in</strong>g phoria to<br />
become an <strong>in</strong>termittent or manifest tropia. The degree of eye deviation may be<br />
approximately equal <strong>in</strong> all directions of gaze (comitant) or less commonly may vary<br />
<strong>in</strong> different positions of gaze (<strong>in</strong>comitant). Horizontal deviations from decompensation<br />
of prior childhood strabismus are typically comitant. Breakdown of acquired deviations,<br />
such as an old fourth nerve palsy, may be <strong>in</strong>comitant.<br />
Ductions (each eye mov<strong>in</strong>g separately) and versions (the eyes mov<strong>in</strong>g conjugately)<br />
must always be assessed. In assess<strong>in</strong>g normal eye excursion, an imag<strong>in</strong>ary vertical l<strong>in</strong>e<br />
through the lower lacrimal punctum should co<strong>in</strong>cide with a boundary l<strong>in</strong>e between the<br />
<strong>in</strong>ner third and outer two thirds of cornea. If more cornea is hidden, adduction is<br />
excessive; if more cornea is visible and if some sclera is visible, adduction is limited. If<br />
abduction is normal, the corneal limbus should touch the outer canthus. If the limbus<br />
passes that po<strong>in</strong>t and some of the cornea is hidden, abduction is excessive; if some of the<br />
sclera rema<strong>in</strong>s visible, abduction is limited (von Noorden, 1996).<br />
What Are the Causes of B<strong>in</strong>ocular Horizontal<br />
Diplopia (Esotropia and Exotropia)?<br />
Diplopia 219<br />
Horizontal b<strong>in</strong>ocular diplopia is usually due to disease processes affect<strong>in</strong>g the medial<br />
and=or lateral rectus muscles, the <strong>in</strong>nervation of these muscles (<strong>in</strong>clud<strong>in</strong>g ocular motor
220 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
cranial nerves and neuromuscular junction), or processes affect<strong>in</strong>g fusion or convergence<br />
and divergence mechanisms (Brazis, 1999). By def<strong>in</strong>ition, patients with horizontal<br />
diplopia compla<strong>in</strong> that the two images are side by side. The separation of images may<br />
vary or rema<strong>in</strong> unchanged at far or near fixation. For example, the image separation<br />
from a left abducens nerve palsy is typically worse at a distance than at close range and<br />
worse on left gaze.<br />
Table 10–3 categorizes the causes of b<strong>in</strong>ocular horizontal diplopia as either disorders<br />
caus<strong>in</strong>g esotropia (ET) or disorders caus<strong>in</strong>g exotropia (XT). Congenital and childhood<br />
strabismus syndromes (Table 10–4) are mentioned but not discussed <strong>in</strong> depth. For a<br />
thorough discussion of childhood strabismus syndromes, the reader is referred to the<br />
excellent text of von Noorden (von Noorden, 1996).<br />
What Are the Childhood Strabismus<br />
Syndromes Caus<strong>in</strong>g Esotropia and Exotropia?<br />
Childhood strabismus syndromes may be confused with acquired causes of ET and XT<br />
<strong>in</strong> adulthood. Most childhood ETs are comitant and present at an early age with<br />
‘‘crossed-eyes’’ or amblyopia. Childhood comitant ETs may be due to hyperopia or<br />
impaired accommodation or convergence (Mohney, 2001). Incomitant childhood ETs<br />
<strong>in</strong>clude A-pattern and V-pattern esodeviations, <strong>in</strong> which the esodeviation is worse on<br />
upward and downward gaze, respectively, retraction syndromes (see below), and<br />
mechanical-restrictive esodeviation due to congenital fibrosis of the medial rectus<br />
muscle. Some patients with congenital nystagmus are able to decrease the amplitude<br />
or frequency of their nystagmus by convergence (nystagmus blockage syndrome) and<br />
thus an esotropia develops.<br />
Occasionally, adults with a long-stand<strong>in</strong>g, essentially asymptomatic, esophoria may<br />
present with diplopia due to ‘‘decompensation’’ (Kushner, 2001). This decompensation<br />
of a long-stand<strong>in</strong>g esophoria may occur after head trauma, with chang<strong>in</strong>g refractive<br />
needs, when the patient receives drugs that depress the central nervous system<br />
(e.g., alcohol or sedatives), with systemic illnesses, or for unclear reason. History and<br />
exam<strong>in</strong>ation often reveal supportive evidence for a long-stand<strong>in</strong>g strabismus, <strong>in</strong>clud<strong>in</strong>g<br />
a history of childhood strabismus or patch<strong>in</strong>g, the presence of an old head turn, and<br />
horizontal comitance.<br />
Childhood XT is less frequent than childhood ET. The XT may be <strong>in</strong>termittent or<br />
persistent, and sometimes adults with exophoria or <strong>in</strong>termittent XT may present with<br />
diplopia due to the <strong>in</strong>ability to adequately compensate for the eye misalignment<br />
(decompensation of exophoria).<br />
Duane’s retraction syndrome is characterized by a narrow<strong>in</strong>g of the palpebral fissure<br />
and globe retraction on adduction (Chung, 2000). Three forms have been described<br />
(DeResp<strong>in</strong>is, 1993). In type I, abduction is limited but adduction is normal or only<br />
slightly limited. In type II, adduction is impaired but abduction is normal or slightly<br />
limited. In type III, both adduction and abduction are impaired. Eye deviation may or<br />
may not be present <strong>in</strong> primary position but if it is present, then ET is usually present <strong>in</strong><br />
type I and III patients, whereas XT is more frequent <strong>in</strong> type II patients. Although many<br />
patients adopt a head turn to ma<strong>in</strong>ta<strong>in</strong> s<strong>in</strong>gle b<strong>in</strong>ocular vision, these patients rarely<br />
compla<strong>in</strong> of spontaneous diplopia. They do have diplopia recognition, if specifically
Table 10–3. Etiologies of Esotropia=Exotropia and Acquired Horizontal Diplopia<br />
Diplopia 221<br />
Esotropia<br />
Childhood strabismus syndromes (see Table 10–4)<br />
Change of angle of preexist<strong>in</strong>g childhood strabismus or loss of suppression scotoma<br />
Decompensation of a long-stand<strong>in</strong>g esophoria<br />
Consecutive esotropia (after strabismus surgery)<br />
Optical causes (e.g., optical center change <strong>in</strong> glasses, over-m<strong>in</strong>us <strong>in</strong> accommodative<br />
esophoria)<br />
Sensory esotropia (usually not associated with diplopia)<br />
Disorders of muscle and restrictive syndromes<br />
Orbital myositis (orbital pseudotumor)<br />
Thyroid eye disease<br />
Medial orbital wall fracture<br />
Postsurgical esotropia<br />
Isolated lateral rectus weakness<br />
Muscle trauma<br />
Progressive external ophthalmoplegia syndromes<br />
<strong>An</strong>omalous orbital structures, such as extraocular muscles <strong>in</strong>sert<strong>in</strong>g <strong>in</strong>to an abnormal location,<br />
fibrous bands, and discrete anomalous muscles (Lueder, 2002)<br />
Other orbital disease processes<br />
Disorders of the neuromuscular junction (e.g., myasthenia gravis)<br />
Disorders of cranial nerves<br />
Sixth nerve palsy<br />
Ocular neuromyotonia<br />
Central disorders<br />
Cyclic esotropia<br />
Periodic alternat<strong>in</strong>g esotropia<br />
Divergence <strong>in</strong>sufficiency or paralysis<br />
Acute acquired comitant esotropia<br />
Spasm of the near reflex<br />
Midbra<strong>in</strong> pseudo–sixth nerve palsy<br />
Thalamic esotropia<br />
Acquired motor fusion deficiency<br />
Hemifield slide phenomena<br />
Exotropia<br />
Childhood strabismus syndromes (see Table 10–4)<br />
Change of angle of preexist<strong>in</strong>g childhood strabismus or loss of suppression scotoma<br />
Decompensation of a long-stand<strong>in</strong>g exophoria<br />
Consecutive exotropia (after strabismus surgery)<br />
Exotropia secondary to vitreous hemorrhage<br />
Optical causes<br />
Sensory exotropia (often not associated with diplopia)<br />
Disorders of the muscle<br />
Orbital myositis (orbital pseudotumor)<br />
Thyroid eye disease (uncommon)<br />
Postsurgical exotropia<br />
(cont<strong>in</strong>ued)
222 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 10–3. (cont<strong>in</strong>ued)<br />
Isolated medial rectus weakness<br />
Muscle trauma<br />
Progressive external ophthalmoplegia syndromes<br />
Other orbital disease processes<br />
Disorders of the neuromuscular junction (e.g., myasthenia gravis)<br />
Disorders of cranial nerves<br />
Third nerve palsy<br />
Ocular neuromyotonia<br />
Central disorders<br />
Acquired motor fusion deficiency<br />
Internuclear ophthalmoplegia (WEMINO syndrome and WEBINO syndrome) and the one-anda-half<br />
syndrome (paralytic pont<strong>in</strong>e exotropia)<br />
Vitam<strong>in</strong> E deficiency (e.g., abetalipoprote<strong>in</strong>emia)<br />
Convergence <strong>in</strong>sufficiency and paralysis<br />
Hemifield slide phenomena<br />
asked, and state that they do recognize two images when their eyes are misaligned. In<br />
all types, there may be a vertical deviation of the adduct<strong>in</strong>g eye with ‘‘up-shoots’’ and<br />
‘‘down-shoots.’’ Duane’s retraction syndrome is predom<strong>in</strong>antly congenital and is<br />
thought to be due to anomalous <strong>in</strong>nervation of the lateral rectus muscle by the <strong>in</strong>ferior<br />
division of the oculomotor nerve (DeResp<strong>in</strong>is, 1993). <strong>An</strong> acquired Duane’s-like<br />
syndrome has been described <strong>in</strong> patients with pont<strong>in</strong>e glioma, with rheumatoid<br />
arthritis, follow<strong>in</strong>g trigem<strong>in</strong>al rhizotomy, and after removal of an orbital cavernous<br />
hemangioma by lateral orbitotomy.<br />
Consecutive esotropia refers to esodeviation that occurs iatrogenically after surgical<br />
overcorrection of an exodeviation (patients who are surgically undercorrected may<br />
also still be diplopic postoperatively). Consecutive exotropia similarly results from<br />
surgical overcorrection of ET or may arise spontaneously <strong>in</strong> a previously esotropic<br />
patient, especially <strong>in</strong> association with poor vision <strong>in</strong> the deviat<strong>in</strong>g eye (sensory<br />
exotropia).<br />
What Are Sensory Esotropia and Sensory<br />
Exotropia?<br />
Sensory deviations <strong>in</strong>clud<strong>in</strong>g ET or XT result from reduced visual acuity <strong>in</strong> one eye.<br />
These patients do not compla<strong>in</strong> of diplopia because of the visual loss. Loss of fusion <strong>in</strong><br />
cases of visual loss may allow a preexist<strong>in</strong>g phoria to become manifest. Sidikaro and<br />
von Noorden reported 121 patients with sensory heterotropias and noted that ET and<br />
XT occurred with almost equal frequency when the onset of visual impairment occurred<br />
at birth or between birth and age 5 (Sidikaro, 1982). Sensory XT, however, predom<strong>in</strong>ates<br />
<strong>in</strong> older children and adults.
Table 10–4. Classification of Childhood Strabismus Syndromes<br />
Esodeviations<br />
Comitant esodeviation<br />
Accommodative esotropia<br />
Refractive<br />
Nonrefractive<br />
Hypoaccommodative<br />
Partially accommodative esotropia<br />
Nonaccommodative esotropia<br />
Infantile (onset birth to 6 months)<br />
Acquired (<strong>in</strong>cludes esotropia with myopia, cyclic esotropia, and some cases of divergence<br />
<strong>in</strong>sufficiency)<br />
Microtropia<br />
Nystagmus blockage syndrome<br />
Incomitant esodeviation<br />
A- and V-pattern esotropia<br />
Duane’s retraction syndrome type I and III<br />
Congenital mechanical-restrictive esodeviations (e.g., congenital fibrosis)<br />
Secondary esodeviation<br />
Sensory esotropia<br />
Consecutive esotropia (after strabismus surgery)<br />
Exodeviations<br />
Primary<br />
Duane’s syndrome type II<br />
Restrictive—congenital fibrosis<br />
Secondary<br />
Sensory exotropia<br />
Consecutive exotropia (after strabismus surgery)<br />
Source: Repr<strong>in</strong>ted from von Noorden, 1996, with permission from Elsevier Science.<br />
What Disorders of the Extraocular Muscles<br />
Are Associated with Horizontal Diplopia?<br />
Diplopia 223<br />
Orbital pseudotumor is an idiopathic orbital <strong>in</strong>flammatory condition characterized<br />
by the follow<strong>in</strong>g features: (1) typically unilateral but may be bilateral; (2) cl<strong>in</strong>ical signs<br />
of orbital mass effect and <strong>in</strong>flammation (e.g., proptosis, chemosis, pa<strong>in</strong>, <strong>in</strong>jection,<br />
ophthalmoplegia); (3) orbital imag<strong>in</strong>g shows focal or diffuse <strong>in</strong>flammatory lesion; (4)<br />
histopathology reveals a fibro-<strong>in</strong>flammatory lesion; and (5) no other identifiable local or<br />
systemic causes (Lacey, 1999; Mombaerts, 1996).<br />
When the <strong>in</strong>flammatory process is conf<strong>in</strong>ed to one or multiple extraocular muscles,<br />
the process is referred to as orbital myositis, although some authors feel that orbital<br />
pseudotumor and orbital myositis may be dist<strong>in</strong>ct cl<strong>in</strong>icotherapeutic entities<br />
(Mombaerts, 1997). Patients present with acute or subacute orbital pa<strong>in</strong> and diplopia.<br />
F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clude conjunctival chemosis and <strong>in</strong>jection, ptosis, and proptosis. <strong>An</strong>gleclosure<br />
glaucoma may rarely occur (Bernard<strong>in</strong>o, 2001). The process may be unilateral or
224 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
bilateral and usually resolves with corticosteroid therapy (Mombaerts, 1997) or radiation<br />
therapy. The illness is often monophasic but recurrent episodes may occur.<br />
Characteristics associated with recurrences <strong>in</strong>clude male gender, lack of proptosis,<br />
eyelid retraction, horizontal extraocular muscle <strong>in</strong>volvement, multiple or bilateral<br />
extraocular muscle <strong>in</strong>volvement, muscle tendon spar<strong>in</strong>g on neuroimag<strong>in</strong>g, and lack<br />
of response to steroids or nonsteroidal anti<strong>in</strong>flammatory agents (Mannor, 1997). Orbital<br />
myositis may be associated with systemic diseases, such as Crohn’s disease (Squires,<br />
1991), celiac disease, Churg-Strauss syndrome (Takahashi, 2001), systemic lupus erythematosus<br />
(Lacey, 1999; Serop, 1994), Whipple’s disease (Orssaud, 1992), rheumatoid<br />
arthritis, l<strong>in</strong>ear scleroderma (Ramboer, 1997; Serup, 1994; Suttorp-Schulten, 1990),<br />
and Wegener’s granulomatosis. Recurrent orbital myositis may occasionally be<br />
familial (Maurer, 1999) and orbital myositis may occasionally be paraneoplastic<br />
(Harris, 1994).<br />
<strong>Neuro</strong>imag<strong>in</strong>g reveals enlarged, irregular muscles usually with tend<strong>in</strong>ous <strong>in</strong>sertion<br />
<strong>in</strong>volvement (as opposed to tendon spar<strong>in</strong>g <strong>in</strong> thyroid ophthalmopathy). Intracranial<br />
extension of the <strong>in</strong>flammatory process is rare (De Jesús, 1996). The differential diagnosis<br />
of orbital pseudotumor is outl<strong>in</strong>ed <strong>in</strong> Table 10–5.<br />
Orbital polymyositis and giant cell myocarditis is a rare, dist<strong>in</strong>ct nosologic entity<br />
characterized by progressive, often pa<strong>in</strong>ful bilateral ophthalmoplegia with thickened<br />
extraocular muscles and cardiac arrhythmia often lead<strong>in</strong>g to death (Kattah, 1990; Leib,<br />
1994; Stevens, 1996). Pathologically, the extraocular and cardiac muscles showed diffuse<br />
mononuclear and giant cell <strong>in</strong>flammation. Cardiac transplantation may be lifesav<strong>in</strong>g<br />
(Leib, 1994).<br />
Biopsy may be required to exclude other diseases, except <strong>in</strong> pure myositic locations,<br />
<strong>in</strong> which the cl<strong>in</strong>icopathologic picture is rather unique and surgical biopsy may damage<br />
the muscle, and <strong>in</strong> posterior locations, <strong>in</strong> which the optic nerve may be at risk dur<strong>in</strong>g<br />
surgery (Mombaerts, 1996). Pathologic studies <strong>in</strong> orbital myositis reveal <strong>in</strong>flammatory<br />
<strong>in</strong>filtrate composed ma<strong>in</strong>ly of small well-differentiated mature lymphocytes, admixed<br />
with plasma cells, <strong>in</strong> a diffuse or multifocal pattern. The muscle fibers are swollen and<br />
separated by edema and fibrosis with loss of normal striations and degeneration of<br />
muscle fibers (Mombaerts, 1996). Other atypical histopathologic patterns, such as<br />
extensive sclerosis, true vasculitis, granulomatous <strong>in</strong>flammation, and tissue eos<strong>in</strong>ophilia,<br />
can be used for subclassification of orbital pseudotumor <strong>in</strong> general (Mombaerts,<br />
1996). There is no unequivocal correlation between cl<strong>in</strong>icotherapeutic outcome and<br />
these atypical f<strong>in</strong>d<strong>in</strong>gs.<br />
Thyroid eye disease (thyroid orbitopathy, thyroid ophthalmopathy, or Graves’<br />
disease) is a disorder characterized cl<strong>in</strong>ically by lid retraction, lid lag <strong>in</strong> downward<br />
gaze, exophthalmos, diplopia (due to extraocular muscle <strong>in</strong>flammation or fibrosis),<br />
potential visual loss due to compressive optic neuropathy or corneal damage, and signs<br />
and symptoms of orbital congestion (Bartley, 1994, 1995a,b, 1996a,b). The restrictive<br />
extraocular muscle <strong>in</strong>volvement may be confirmed by impaired ocular motility dur<strong>in</strong>g<br />
the forced duction test. The extraocular muscles predom<strong>in</strong>antly affected <strong>in</strong>clude the<br />
<strong>in</strong>ferior, medial, and superior rectus muscles, and as the process causes muscle tightness<br />
or restriction, the diplopia is worse <strong>in</strong> the direction opposite to that of the <strong>in</strong>volved<br />
muscle(s) action. Thus, hypertropia and esotropia are quite common <strong>in</strong> thyroid eye<br />
disease but exotropia is uncommon because lateral rectus muscle is usually not<br />
markedly <strong>in</strong>volved. In fact, if a patient with thyroid eye disease is noted to be exotropic,<br />
superimposed myasthenia gravis should be considered, as there is an <strong>in</strong>creased risk of
Table 10–5. Differential Diagnosis of Orbital Pseudotumor<br />
Diplopia 225<br />
Thyroid eye disease (see Table 10–6)<br />
Orbital cellulitis (e.g., orbital apex syndrome)<br />
Bacterial<br />
Fungal<br />
Aspergillosis (Hutnik, 1997; Lev<strong>in</strong>, 1996; Slav<strong>in</strong>, 1991)<br />
Mucormycosis (Balch, 1997; Dooley, 1992; Downie, 1993; Johnson, 1999)<br />
Bipolaris hawaiiensis (Mask<strong>in</strong>, 1989)<br />
Act<strong>in</strong>omycosis (Sullivan, 1992)<br />
Cysticercosis (Lacey, 1999)<br />
Trich<strong>in</strong>osis (Behrens-Baumann, 1990)<br />
Low-flow dural-cavernous s<strong>in</strong>us fistula<br />
Neoplastic<br />
Metastatic<br />
Breast cancer (false ‘‘orbital pseudotumor’’ presentation) (Goldberg, 1990a,b; Lacey, 1999;<br />
Toller, 1998)<br />
Lymphoid hyperplasia<br />
Non-Hodgk<strong>in</strong>’s lymphoma and Hodgk<strong>in</strong>’s disease<br />
S<strong>in</strong>us histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease)<br />
Sem<strong>in</strong>oma (bilateral nonspecific <strong>in</strong>flammatory or Graves-like orbitopathy not due to direct<br />
orbital metastasis)<br />
Infiltrative<br />
Erdheim-Chester disease (idiopathic <strong>in</strong>filtration of the heart, lungs, retroperitoneum, bones, and<br />
other tissues by xanthomatous histiocytes and Touton giant cells) (Esmaeli, 2001; Shields, 1991;<br />
Valmaggia, 1997)<br />
Orbital amyloidosis (Çeviker, 1997; Lacey, 1999; Murdoch, 1996)<br />
Inflammatory<br />
Sarcoidosis (Cornblath, 1993; Lacey, 1999; Patel, 1994; Segal, 2000; Takahashi, 2000)<br />
Giant cell arteritis (de Heide, 1999)<br />
Orbital polymyositis and giant cell myocarditis<br />
Systemic <strong>in</strong>flammatory diseases (Wegener’s granulomatosis, systemic lupus erythematosus)<br />
(Woo, 2001)<br />
myasthenia gravis <strong>in</strong> patients with thyroid eye disease (Lee 1997; Vargas, 1993). Thyroid<br />
eye disease is further discussed <strong>in</strong> Chapter 16. Thyroid eye disease and orbital myositis<br />
may resemble each other cl<strong>in</strong>ically. Differential features are outl<strong>in</strong>ed <strong>in</strong> Table 10–6.<br />
The diagnosis of myasthenia gravis (MG) should be considered <strong>in</strong> all patients with<br />
pa<strong>in</strong>less ptosis and=or ocular motor weakness without pupillary <strong>in</strong>volvement or<br />
proptosis. Weakness and fatigue conf<strong>in</strong>ed to the extraocular muscles or lids comb<strong>in</strong>ed<br />
with orbicularis oculi paresis is especially suggestive of this diagnosis. MG may cause<br />
hypertropia, esotropia, or exotropia, and can mimic many neurogenic conditions<br />
<strong>in</strong>clud<strong>in</strong>g abducens nerve palsies, gaze abnormalities, divergence paresis, and <strong>in</strong>ternuclear<br />
ophthalmoplegia. Therefore, <strong>in</strong> any patient with an abnormality of horizontal<br />
gaze, MG should at least be considered. MG is discussed further <strong>in</strong> Chapter 15.<br />
Orbital trauma may result <strong>in</strong> horizontal diplopia due to a fracture of the medial<br />
orbital wall <strong>in</strong> isolation or accompanied by fracture of the orbital floor or other orbital
226 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 10–6. <strong>Cl<strong>in</strong>ical</strong> Differential Diagnosis of Orbital<br />
Myositis and Thyroid Eye Disease<br />
Orbital Myositis Thyroid Eye Disease<br />
Males and females<br />
equally affected<br />
Females predom<strong>in</strong>ate<br />
Acute or subacute onset Gradual onset<br />
Often severe orbital pa<strong>in</strong> Pa<strong>in</strong>less or ‘‘foreign<br />
body’’ sensation<br />
Motility problems early Motility problems late<br />
May have limited ductions Restrictive ductions<br />
No lid lag or retraction Lid lag and retraction<br />
<strong>Neuro</strong>imag<strong>in</strong>g of orbit<br />
Enlarged muscles irregular Enlarged muscles<br />
often smooth<br />
Tendon spared Tendon may be <strong>in</strong>volved<br />
Often unilateral Often bilateral<br />
bones (Eitzen, 1991; Merle, 1998; von Noorden, 1996). Medial rectus muscle <strong>in</strong>carceration<br />
may lead to ET with impaired abduction or XT with impaired adduction. Medial<br />
orbital wall <strong>in</strong>jury may occur iatrogenically dur<strong>in</strong>g endoscopic transnasal s<strong>in</strong>us surgery<br />
(Eitzen, 1991; von Noorden, 1996). Also, medial or lateral orbital surgery (e.g., optic<br />
nerve sheath fenestration) may directly <strong>in</strong>jure the medial or lateral rectus muscles,<br />
result<strong>in</strong>g <strong>in</strong> <strong>in</strong>itial muscle paresis, sometimes followed eventually by scarr<strong>in</strong>g and<br />
restrictive ET or XT, respectively. Other ocular surgeries (e.g., pterygium surgery, scleral<br />
buckle, and glaucoma setons) may also result <strong>in</strong> horizontal diplopia.<br />
Isolated medial rectus paresis is rare and results <strong>in</strong> XT, due to unopposed action of the<br />
lateral rectus muscle. The XT is worse on gaze to the opposite side and is associated<br />
with impaired adduction on the side of the weak muscle. Impaired monocular<br />
adduction is more often noted with <strong>in</strong>ternuclear ophthalmoplegia than isolated<br />
medial rectus palsy due to a partial third nerve palsy. Isolated medial rectus muscle<br />
paresis may occur with MG, orbital myositis, muscle trauma, or orbital disease. Lesions<br />
of the oculomotor nerve cause medial rectus paresis but not <strong>in</strong> isolation. Because the<br />
neurons controll<strong>in</strong>g the medial rectus muscle probably lie at three different locations<br />
with<strong>in</strong> the oculomotor nucleus, it is unlikely that a medial rectus paralysis could be the<br />
sole manifestation of a bra<strong>in</strong>stem oculomotor nuclear lesion.<br />
Although isolated lateral rectus paresis is most often due to lesions of the sixth cranial<br />
nerve, other processes, <strong>in</strong>clud<strong>in</strong>g MG, orbital myositis, muscle trauma, and orbital<br />
lesions, may impair the muscle directly.<br />
What Disorders of the Cranial Nerves Cause<br />
Horizontal Diplopia?<br />
Unilateral sixth cranial nerve <strong>in</strong>jury results <strong>in</strong> an <strong>in</strong>comitant esodeviation that is<br />
worsened with gaze <strong>in</strong>to the field of the weak lateral rectus muscle. Patients may
employ a compensatory face turn <strong>in</strong> the direction of the paralyzed lateral rectus muscle<br />
to limit diplopia. Abduction is often limited on the side of the lesion. With bilateral<br />
paralysis, both eyes may be <strong>in</strong> a position of adduction and the esotropia <strong>in</strong>creases upon<br />
look<strong>in</strong>g to the left or right. MG may mimic an isolated sixth nerve palsy, so <strong>in</strong> some<br />
patients with isolated abduction paresis a Tensilon test should be considered, especially<br />
if there are signs of fatigability of the muscle paresis or associated ptosis. Sixth cranial<br />
nerve palsies are further discussed <strong>in</strong> Chapter 13.<br />
Lesions of the third cranial nerve may cause an XT because of weakness of the medial<br />
rectus muscle with the eye deviat<strong>in</strong>g toward the side of the preserved lateral rectus<br />
muscle. This XT is usually associated with other signs of third nerve affection, <strong>in</strong>clud<strong>in</strong>g<br />
paresis of eye elevation and depression, ptosis, and pupillary <strong>in</strong>volvement. Third cranial<br />
nerve palsies are further discussed <strong>in</strong> Chapter 11.<br />
Ocular neuromyotonia (ONM) is a rare disorder characterized by episodic (last<strong>in</strong>g<br />
seconds to m<strong>in</strong>utes) horizontal or vertical diplopia, occurr<strong>in</strong>g either spontaneously or<br />
follow<strong>in</strong>g susta<strong>in</strong>ed (10 to 20 seconds) eccentric gaze (Abdulla, 1999; Barroso, 1993;<br />
Chung, 1997; Ezra, 1996b; Frohman, 1995; Fu, 1995; Haupert, 1997; Helmchen, 1992;<br />
Morrow, 1996; Newman, 1993; Yee, 1998). Most patients have had prior radiation<br />
therapy to the sellar or parasellar region (months to years before onset of the ONM) for<br />
tumors, <strong>in</strong>clud<strong>in</strong>g chordoma, pituitary tumors, craniopharyngioma, chondrosarcoma,<br />
rhabdomyosarcoma, thalamic glioma, s<strong>in</strong>onasal carc<strong>in</strong>oma, and medulloblastoma. In<br />
some cases, however, no responsible structural lesion or history of radiation therapy is<br />
noted. Rarely ONM may be due to a compressive lesion, such as an aneurysm (Abdulla,<br />
1999; Ezra, 1996b), dolichoectatic basilar artery (Tilikete, 2000), thyroid eye disease<br />
(Chung, 1997), Paget’s disease of bone (Boschi, 1997), or after cavernous s<strong>in</strong>us<br />
thrombosis secondary to mucormycosis (Harrison, 1997). One patient had fourth<br />
nerve <strong>in</strong>volvement where spasms of the superior oblique muscle were <strong>in</strong>duced only<br />
by alcohol <strong>in</strong>take (Ezra, 1996b), whereas another developed ONM several years after<br />
myelography with thorium dioxide (Thorotrast) (Yee, 1998).<br />
ONM is thought to reflect impaired muscle relaxation due to <strong>in</strong>appropriate<br />
discharges from oculomotor, trochlear, or abducens neurons or axons with unstable<br />
cellular membranes. Patients with ONM often benefit from membrane stabiliz<strong>in</strong>g agents<br />
such as carbamazep<strong>in</strong>e. One patient noted that she could term<strong>in</strong>ate episodes of<br />
episodic ocular depression <strong>in</strong>stantly by forcefully direct<strong>in</strong>g her gaze upward, and<br />
thus stretch<strong>in</strong>g the affected muscle might also prove to be an effective way of end<strong>in</strong>g<br />
attacks (Safran, 1998). Patients with unexpla<strong>in</strong>ed transient episodic diplopia should thus<br />
be specifically tested for diplopia and ocular misalignment follow<strong>in</strong>g susta<strong>in</strong>ed<br />
eccentric gaze.<br />
What Central Disorders Cause Horizontal<br />
Diplopia?<br />
Diplopia 227<br />
Central disorders caus<strong>in</strong>g horizontal diplopia <strong>in</strong>clude cyclic esotropia, periodic alternat<strong>in</strong>g<br />
esotropia, divergence <strong>in</strong>sufficiency and paralysis, convergence spasm, convergence<br />
<strong>in</strong>sufficiency and paralysis, acquired motor fusion deficiency, <strong>in</strong>ternuclear<br />
ophthalmoplegia and the one-and-a-half syndrome, vitam<strong>in</strong> E deficiency, and the<br />
hemifield slip phenomenon. Exotropia due to vitreous hemorrhage is <strong>in</strong>cluded here,
228 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
as the diplopia may be due to impaired fusional mechanisms. Internuclear ophthalmoplegia,<br />
the one-and-a-half syndrome, and the motility disorder associated with vitam<strong>in</strong> E<br />
deficiency (abetalipoprote<strong>in</strong>emia) may all cause horizontal diplopia (and occasionally<br />
vertical diplopia when associated with skew deviation) and are discussed <strong>in</strong> Chapter 17.<br />
What Is Cyclic Esotropia?<br />
Cyclic esotropia is a rare condition characterized by a regularly recurr<strong>in</strong>g ET that often<br />
occurs with regular 48-hour cycles (Riordan-Eva, 1993; Tapiero, 1995). There is often a<br />
24-hour period of normal b<strong>in</strong>ocular vision followed by a 24-hour period of manifest ET;<br />
72-hour and 96-hour cycles have also been reported. The ET may eventually become<br />
constant. Cyclic ET usually appears <strong>in</strong> young children but may also occur <strong>in</strong> adults<br />
(Riordan-Eva, 1993; Tapiero, 1995). The condition usually starts without precipitant but<br />
has been described after strabismus surgery for <strong>in</strong>termittent XT, after cataract surgery,<br />
after traumatic aphakia, after surgical removal of a third ventricular astrocytoma, and <strong>in</strong><br />
association with optic atrophy or ret<strong>in</strong>al detachment (Riordan-Eva, 1993). The etiology<br />
of this condition is unknown, with possible causes be<strong>in</strong>g oculomotor nerve hyperactivity<br />
(although there are no associated abnormalities of the pupil or lid), abducens nerve<br />
dysfunction, strabismus be<strong>in</strong>g <strong>in</strong>terrupted by periodic <strong>in</strong>tervals of fusion, or, most<br />
likely, a disorder of central mechanisms.<br />
What Is Periodic Alternat<strong>in</strong>g Esotropia?<br />
Periodic alternat<strong>in</strong>g esotropia (PAE) is a rare cyclic disorder typically associated with<br />
periodic alternat<strong>in</strong>g nystagmus or periodic alternat<strong>in</strong>g gaze (PAG) (Hamed, 1992).<br />
While one eye ma<strong>in</strong>ta<strong>in</strong>s fixation, the other eye undergoes a phase of wax<strong>in</strong>g then<br />
wan<strong>in</strong>g <strong>in</strong>ward deviation. The cycle is completed by a phase of vary<strong>in</strong>g <strong>in</strong>ward<br />
deviation <strong>in</strong> the eye that was <strong>in</strong>itially fixat<strong>in</strong>g after a transition period of orthotropia<br />
dur<strong>in</strong>g which fixation changes. This condition is <strong>in</strong>variably associated with severe bra<strong>in</strong><br />
dysfunction and is especially noted <strong>in</strong> young children with ataxia or hydrocephalus.<br />
What Constitutes Divergence Insufficiency<br />
and Divergence Paralysis?<br />
Weakness of divergence is characterized by <strong>in</strong>termittent or constant ET at distance with<br />
fusion at near (Akman, 1995; Arai, 1990; Fril<strong>in</strong>g, 1993; Jacobson, 2000; Lepore, 1999;<br />
Lewis, 1996; Schanzer, 1998; von Noorden, 1996). Abduction by duction and version<br />
test<strong>in</strong>g is relatively full bilaterally. The angle of strabismus rema<strong>in</strong>s unchanged<br />
(comitant) or may be decreased on gaze to either side. Fusional divergence is reduced<br />
or absent. Fusional divergence is measured by plac<strong>in</strong>g prisms of progressively larger<br />
strength base-<strong>in</strong> over one eye while the subject is fixat<strong>in</strong>g at distance and near and<br />
not<strong>in</strong>g when the fixation image appears double (break po<strong>in</strong>t). Patients with divergence<br />
weakness should also demonstrate normal speed and amplitude of horizontal saccades<br />
(Leigh, 1999).
Diplopia 229<br />
When ET at distance due to divergence impairment occurs <strong>in</strong> an otherwise healthy<br />
<strong>in</strong>dividual, it is referred to as ‘‘divergence <strong>in</strong>sufficiency’’ or ‘‘primary divergence<br />
<strong>in</strong>sufficiency,’’ whereas when it occurs associated with neurologic disease it is called<br />
‘‘divergence paralysis’’ or ‘‘secondary divergence <strong>in</strong>sufficieny.’’ Divergence <strong>in</strong>sufficiency<br />
(primary) is usually observed <strong>in</strong> young adults, is self-limited, and may be treated with<br />
base-out prisms or occasionally surgery (Akman, 1995; Arai, 1990; Fril<strong>in</strong>g, 1993;<br />
Jacobson, 2000; Lewis, 1996; Schanzer, 1998; von Noorden, 1996). In one study, 95%<br />
of patients with primary divergence <strong>in</strong>sufficiency were older than 50 years and<br />
symptoms resolved <strong>in</strong> 40% of patients after a median of 5 months (Jacobson, 2000).<br />
Divergence paralysis (secondary divergence <strong>in</strong>sufficiency) is usually noted with bra<strong>in</strong>stem<br />
disease. It has been reported with multiple sclerosis, <strong>in</strong>tracranial masses (e.g.,<br />
pontomedullary glioma), bra<strong>in</strong>stem hemorrhage or <strong>in</strong>farction, head trauma, <strong>in</strong>creased<br />
<strong>in</strong>tracranial pressure (e.g., pseudotumor cerebri, neurobrucellosis, frontal lobe glioblastoma),<br />
the spontaneous <strong>in</strong>tracranial hypotension syndrome, cerebellar lesions, craniocervical<br />
junction abnormalities (e.g., Chiari malformation), hydrocephalus, men<strong>in</strong>gitis,<br />
encephalitis, syphilis, clivus lymphoma, acute lymphoblastic leukemia, chronic lymphocytic<br />
leukemia, diazepam <strong>in</strong>gestion, giant cell arteritis, Fisher’s syndrome, Wernicke’s<br />
encephalopathy, Park<strong>in</strong>son’s disease, Machado-Joseph disease, progressive supranuclear<br />
palsy, and after lumbar puncture or epidural block (Akman, 1995; Arai, 1990;<br />
Brown, 1999; Fril<strong>in</strong>g, 1993; Horton, 1994; Jacobson, 2000; Lepore, 1999; Lewis, 1996;<br />
Mokri, 1997; Ohyagi, 2000; Schanzer, 1998; Tekeli, 1999; Vers<strong>in</strong>o, 1996). Abducens nerve<br />
palsy may also cause esotropia that is worse at a distance than near, and <strong>in</strong>deed some<br />
authors believe that divergence paralysis does not exist and that all such cases actually<br />
represent bilateral abducens nerve palsies. However, three f<strong>in</strong>d<strong>in</strong>gs occur with divergence<br />
paralysis but not with bilateral sixth nerve palsies: (1) fusional divergence is<br />
reduced or absent, (2) the esotropia not only rema<strong>in</strong>s unchanged dur<strong>in</strong>g horizontal gaze<br />
but may even decrease, and (3) saccadic velocities are normal.<br />
In a study of 17 adults with divergence weakness, none of the patients were found to<br />
have concurrent neurologic disease (Wigg<strong>in</strong>s, 1999). Thirteen patients rema<strong>in</strong>ed stable,<br />
three improved, and one progressed. Sixteen patients were treated successfully: 12 with<br />
prisms and four with strabismus surgery; one patient was not treated. The authors<br />
concluded that divergence weakness is usually an isolated condition that tends to<br />
rema<strong>in</strong> stable and respond to treatment with prisms or strabismus surgery. In another<br />
study of 15 patients with divergence paresis, three cases were idiopathic and the rest<br />
were associated with diverse central nervous system diseases (Lepore, 1999). Although<br />
six of these patients had posterior fossa disease, neuroimag<strong>in</strong>g showed no common<br />
circumscribed lesion site or evidence of <strong>in</strong>creased <strong>in</strong>tracranial pressure, and thus<br />
divergence paresis was considered a nonlocaliz<strong>in</strong>g cause of horizontal diplopia.<br />
Divergence paresis may be mimicked by myasthenia gravis (Lepore, 1999).<br />
As noted above, unilateral or bilateral abducens nerve paresis may cause an acute ET<br />
with the deviation greater at distance than near. This deviation may eventually become<br />
comitant (‘‘spread of comitance’’), <strong>in</strong> which case it will be difficult to recognize the<br />
paretic element. Acute acquired comitant ET may occur <strong>in</strong> childhood and may be<br />
benign (von Noorden, 1996). For example, it may develop after artificial <strong>in</strong>terruption of<br />
fusion by occlusion of one eye. A previous esophoria may decompensate and become<br />
manifest after fusion has been disrupted. Acquired comitant ET <strong>in</strong> childhood may also<br />
occur with central nervous system tumors, especially bra<strong>in</strong>stem and cerebellar tumors<br />
and tumors of the corpus callosum, and with Chiari I malformation. Thus, these
230 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
patients must undergo neuroimag<strong>in</strong>g (Biousse, 2000; Hoyt, 1995; Lewis, 1996; Lopresto<br />
Weeks, 1999; Simon, 1996). It has also been described after head trauma <strong>in</strong> a child on<br />
carbamazep<strong>in</strong>e (Fukuo, 1998). The mechanism of acute acquired comitant ET is<br />
unknown.<br />
What Is Convergence Spasm?<br />
Convergence spasm usually occurs on a functional basis. Patients exhibit <strong>in</strong>termittent<br />
episodes of susta<strong>in</strong>ed maximal convergence associated with accommodative spasm<br />
(<strong>in</strong>duced myopia) and miosis (Al-D<strong>in</strong>, 1994; Goldste<strong>in</strong>, 1996; von Noorden, 1996). The<br />
spasm may be triggered by ask<strong>in</strong>g the patient to fixate an object held closely before the<br />
eyes; after the fixation object has been removed, the eyes will rema<strong>in</strong> <strong>in</strong> a convergent<br />
position (von Noorden, 1996). Quick saccades back and forth <strong>in</strong> the horizontal plane<br />
may also <strong>in</strong>duce the spasm.<br />
Patients with spasm of the near reflex often compla<strong>in</strong> of headache, diplopia,<br />
photophobia, and blurred vision and often have tunnel visual fields. Patients may<br />
<strong>in</strong>itially be thought to have unilateral or bilateral abducens nerve paresis or myasthenia<br />
gravis. Observation of miosis dur<strong>in</strong>g the spasm <strong>in</strong> a patient with apparent unilateral or<br />
bilateral limitation of abduction and severe myopia (8 to 10 diopters) <strong>in</strong>dicates the<br />
correct diagnosis (Al-D<strong>in</strong>, 1994; Goldste<strong>in</strong>, 1996; Newman, 1989; Postert, 1997). This<br />
miosis generally resolves as soon as either eye is occluded (Newman, 1989). Also,<br />
despite apparent weakness of abduction, patients have full abduction when one eye is<br />
patched or dur<strong>in</strong>g duction test<strong>in</strong>g. Convergence spasm may occasionally be confused<br />
with divergence paralysis <strong>in</strong> that <strong>in</strong> both <strong>in</strong>stances there is ET at distance fixation. With<br />
convergence spasm, however, fusional divergence is normal and visual acuity at<br />
distance is decreased.<br />
Spasm of the near reflex may rarely be associated with organic disease of the central<br />
and ocular motor system. Increased or susta<strong>in</strong>ed convergence may be seen with lesions<br />
of the diencephalic-mesencephalic junction. For example, a pseudo–sixth nerve palsy<br />
may occur from midbra<strong>in</strong> lesions (midbra<strong>in</strong> pseudo–sixth nerve palsy), perhaps due to<br />
an excess of convergence tone (Pullic<strong>in</strong>o, 2000). In a study of patients with pseudoabducens<br />
palsy and ‘‘top-of-the-basilar’’ <strong>in</strong>farcts, the smallest <strong>in</strong>farcts produc<strong>in</strong>g an<br />
ipsilateral pseudoabducens palsy were located just rostral to the oculomotor nucleus,<br />
near the midbra<strong>in</strong>-diencephalic junction (Pullic<strong>in</strong>o, 2000). Two patients with only<br />
contralateral pseudoabducens palsy had subthalamic and thalamic <strong>in</strong>farction, and<br />
four patients with bilateral pseudoabducens palsy had larger <strong>in</strong>farcts <strong>in</strong>volv<strong>in</strong>g the<br />
midbra<strong>in</strong>. All patients with pseudoabducens palsy had upgaze palsy. The authors<br />
concluded that lesions near the midbra<strong>in</strong>-diencephalic junction are important for the<br />
development of pseudoabducens palsy and that this abnormality and convergenceretraction<br />
nystagmus are both manifestations of abnormal vergence activity. Inhibitory<br />
descend<strong>in</strong>g pathways for convergence may pass through the thalamus and decussate <strong>in</strong><br />
the subthalamic region (Pullic<strong>in</strong>o, 2000). A case of pseudo–sixth nerve palsy has been<br />
ascribed to bra<strong>in</strong>stem <strong>in</strong>farction due to deep cerebral venous thrombosis (Bernste<strong>in</strong>,<br />
2001). Acute esotropia has been described with contralateral thalamic <strong>in</strong>farction <strong>in</strong> the<br />
territory of the mesencephalic artery (acute thalamic esotropia). Tonic activation of the<br />
medial rectus muscle <strong>in</strong> these cases could result from damage to direct <strong>in</strong>hibitory<br />
projections from the thalamus or impairments of <strong>in</strong>puts to midbra<strong>in</strong> neurons <strong>in</strong>volved
<strong>in</strong> vergence control. Acute thalamic hemorrhage may cause bilateral asymmetric<br />
esotropia with the contralateral eye more affected than the ipsilateral eye<br />
(Hertle, 1990). Bilateral pseudo–sixth nerve palsies have been described with symmetric<br />
bilateral paramedian thalamic lesions without midbra<strong>in</strong> <strong>in</strong>volvement (Wiest, 2000).<br />
Other etiologies of <strong>in</strong>creased or susta<strong>in</strong>ed spasm of the near reflex <strong>in</strong>clude Wernicke-<br />
Korsakoff syndrome, Arnold-Chiari malformation, encephalitis, hepatic encephalopathy,<br />
neurosyphilis, vertebrobasilar ischemia, multiple sclerosis, labyr<strong>in</strong>th<strong>in</strong>e fistula,<br />
trauma, posterior fossa tumor, pituitary adenoma, phenyto<strong>in</strong> <strong>in</strong>toxication, cyclic oculomotor<br />
palsy, Raeder’s paratrigem<strong>in</strong>al syndrome, ocular <strong>in</strong>flammation, Fisher’s<br />
syndrome, and ocular myasthenia gravis (Al-D<strong>in</strong>, 1994; Goldste<strong>in</strong>, 1996; Hertle, 1990;<br />
Postert, 1997; Thompson, 1998; von Noorden, 1996).<br />
What Constitutes Convergence Insufficiency<br />
and Convergence Paralysis?<br />
Patients with an exodeviation greater at close range than at a distance have convergence<br />
<strong>in</strong>sufficiency type XT. Adduction is usually normal, there is a remote near po<strong>in</strong>t of<br />
convergence, and fusional convergence is decreased at near fixation (von Noorden,<br />
1996). This condition is common among teenagers and college students, especially those<br />
with an <strong>in</strong>creased visual work load, but may also be seen <strong>in</strong> the elderly. It often<br />
develops at times of stress or fatigue, but also may be noted dur<strong>in</strong>g systemic <strong>in</strong>fection or<br />
after head trauma. Acquired cerebral lesions, especially affect<strong>in</strong>g the nondom<strong>in</strong>ant<br />
parietal lobe, may rarely be responsible (Ohtsuka, 1988). Patients with convergence<br />
<strong>in</strong>sufficiency typically compla<strong>in</strong> of eyestra<strong>in</strong> and ache. After brief periods of read<strong>in</strong>g,<br />
the letters will blur and run together and often diplopia occurs dur<strong>in</strong>g near work.<br />
Typically, the patient will close or cover one eye while read<strong>in</strong>g to obta<strong>in</strong> relief from<br />
visual fatigue.<br />
Patients with convergence paralysis, as opposed to convergence <strong>in</strong>sufficiency, often<br />
harbor a lesion of the midbra<strong>in</strong>. Diplopia exists only at near fixation, adduction is<br />
normal, and the patient is unable to converge. Preservation of accommodation or<br />
pupillary miosis at close range confirms an organic etiology. Other signs of midbra<strong>in</strong><br />
damage usually are present <strong>in</strong>clud<strong>in</strong>g impaired vertical gaze, upbeat or downbeat<br />
nystagmus, convergence-retraction nystagmus, and eyelid retraction. Many conditions<br />
are associated with convergence paralysis, <strong>in</strong>clud<strong>in</strong>g Park<strong>in</strong>son’s disease, progressive<br />
supranuclear palsy, dorsal midbra<strong>in</strong> tumors, midbra<strong>in</strong> hemorrhage or <strong>in</strong>farction, multiple<br />
sclerosis, encephalitis, metabolic causes, trauma, subdural hematoma, and drugs<br />
(Racette, 1999; Spierer, 1995). Dissociated unilateral convergence paralysis has been<br />
described with thalamotectal hemorrhage (L<strong>in</strong>dner, 1992). Selective convergence loss<br />
with light-near dissociation has been described with bilateral paramedian thalamic<br />
<strong>in</strong>farction (Wiest, 2000).<br />
What Is Acquired Motor Fusion Deficiency?<br />
Diplopia 231<br />
Motor fusion is a function of the extrafoveal ret<strong>in</strong>al periphery (von Noorden, 1996).<br />
Acquired motor fusional deficiency is a rare condition that represents loss of both<br />
fusional convergence and divergence that may occur after head trauma, stroke, bra<strong>in</strong>
232 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
tumor, or neurosurgery (von Noorden, 1996). It is assumed to be due to midbra<strong>in</strong><br />
damage. The patients compla<strong>in</strong> of eyestra<strong>in</strong> and are unable to ma<strong>in</strong>ta<strong>in</strong> s<strong>in</strong>gle vision for<br />
any length of time. Despite apparent ocular alignment, patients compla<strong>in</strong> of transient or<br />
permanent diplopia. Fusional amplitudes are absent, but stereopsis and sensory fusion<br />
are <strong>in</strong>tact dur<strong>in</strong>g brief episodes of fusion. There is no effective therapy.<br />
Why Is Vitreous Hemorrhage Sometimes<br />
Associated with a Secondary Exotropia?<br />
Fujikado et al described eight patients with exotropia and b<strong>in</strong>ocular diplopia after<br />
recovery from vitreous hemorrhage (Fujikado, 1997). Vitreous hemorrhages were<br />
bilateral <strong>in</strong> three patients and unilateral <strong>in</strong> five patients. Diplopia occurred <strong>in</strong> all<br />
patients after vitrectomy. Exotropia was present <strong>in</strong> all of the patients, and seven of<br />
the eight also had vertical strabismus with an average deviation of six prism diopters.<br />
The authors concluded that diplopia after vitrectomy for long-stand<strong>in</strong>g vitreous<br />
hemorrhage may occur due to fusion impairment comparable to that occasionally<br />
seen after surgery for traumatic cataract.<br />
What Is the Hemifield Slide Phenomenon?<br />
The hemifield slide or slip phenomenon is a rare cause of <strong>in</strong>termittent b<strong>in</strong>ocular<br />
diplopia noted <strong>in</strong> some patients with lesions of the optic chiasm. This phenomenon<br />
occurs with complete or nearly complete bitemporal hemianopic visual field defects<br />
with disruption of ocular fusion and decompensation of a previous phoria. The<br />
underly<strong>in</strong>g pathophysiology is loss of b<strong>in</strong>ocularity due to lack of cortical representation<br />
of correspond<strong>in</strong>g po<strong>in</strong>ts <strong>in</strong> the visual field from each eye (i.e., transection of the chiasm<br />
creates two <strong>in</strong>dependent, free-float<strong>in</strong>g hemifields, with each eye project<strong>in</strong>g only to the<br />
ipsilateral visual cortex). Patients compla<strong>in</strong> of <strong>in</strong>termittent diplopia and difficulty with<br />
near work (e.g., thread<strong>in</strong>g a needle or sew<strong>in</strong>g). On exam<strong>in</strong>ation, no ocular motor palsy<br />
is noted despite the patients’ compla<strong>in</strong>ts of diplopia. If the eyes <strong>in</strong>termittently converge<br />
(esotropia), a blank space is produced between the vertical meridians as the eyes ‘‘slip’’<br />
<strong>in</strong>ward and the rema<strong>in</strong><strong>in</strong>g hemifields drift apart horizontally. Ocular divergence<br />
(exotropia) causes overlapp<strong>in</strong>g of the vertical meridians, result<strong>in</strong>g <strong>in</strong> superimposition<br />
of images from noncorrespond<strong>in</strong>g ret<strong>in</strong>al areas. Hemifield slide diplopia may also occur<br />
from altitud<strong>in</strong>al visual field defects (Borchert, 1996). Two cases have been described <strong>in</strong><br />
which heteronymous altitud<strong>in</strong>al field defects resulted <strong>in</strong> loss of fusion and transient<br />
overlap of preserved hemifields. This phenomenon resulted <strong>in</strong> compla<strong>in</strong>ts of diplopia<br />
similar to that described with bitemporal hemianopsia. The patients had superior<br />
altitud<strong>in</strong>al field defects <strong>in</strong> one eye and <strong>in</strong>ferior defects <strong>in</strong> the other. One compla<strong>in</strong>ed<br />
of vertical diplopia and the other had vertical and horizontal diplopia.<br />
How Does One Exam<strong>in</strong>e a Patient with the<br />
Compla<strong>in</strong>t of Vertical Diplopia?<br />
Patients with vertical diplopia compla<strong>in</strong> of see<strong>in</strong>g two images, one atop or diagonally<br />
displaced from the other. The evaluation of these patients starts with a careful history,
Diplopia 233<br />
<strong>in</strong>clud<strong>in</strong>g queries concern<strong>in</strong>g previous eye muscle surgeries, childhood strabismus, and<br />
history of patch<strong>in</strong>g or orthoptic exercises. The exam<strong>in</strong>er must question whether the<br />
diplopia is monocular, and thus usually due to abnormalities of the ocular media<br />
<strong>in</strong>clud<strong>in</strong>g refractive error, or b<strong>in</strong>ocular and due to impaired ocular motility. Often, by<br />
ask<strong>in</strong>g some pert<strong>in</strong>ent questions and by observ<strong>in</strong>g the posture of the patient’s head, a<br />
likely diagnosis can be reached even before physical exam<strong>in</strong>ation beg<strong>in</strong>s (Spector, 1993;<br />
von Noorden, 1996). If the patient compla<strong>in</strong>s of vertical diplopia <strong>in</strong> primary gaze, often<br />
one of the vertically act<strong>in</strong>g extraocular muscles is underact<strong>in</strong>g: the right and=or left<br />
<strong>in</strong>ferior rectus, superior rectus, <strong>in</strong>ferior oblique, or superior oblique. Then one asks if the<br />
vertical separation between images is worse on gaze to the left or right. If, for example,<br />
vertical separation of images is worse on gaze to the right, then four of the eight<br />
extraocular muscles may be underact<strong>in</strong>g (the right superior or <strong>in</strong>ferior rectus or the left<br />
<strong>in</strong>ferior or superior oblique). If the separation is worse on gaze to the right and down,<br />
the right <strong>in</strong>ferior rectus or left superior oblique must be underact<strong>in</strong>g; if the image<br />
separation worsens or improves with left or right head tilt, torsional abnormalities,<br />
especially due to oblique muscle <strong>in</strong>volvement, should be suspected.<br />
Patients with b<strong>in</strong>ocular vertical diplopia may adopt a compensatory head, face, or<br />
ch<strong>in</strong> position to move their eyes <strong>in</strong>to a gaze angle that achieves b<strong>in</strong>ocular s<strong>in</strong>gle vision.<br />
Underaction of the superior or <strong>in</strong>ferior rectus muscles is compensated by neck flexion or<br />
extension (ch<strong>in</strong> down or ch<strong>in</strong> up), which seeks to avoid the eye position of maximum<br />
image separation. Torsional diplopia is usually caused by underaction of the superior or<br />
<strong>in</strong>ferior oblique muscles and may be associated with an angular head tilt. This head tilt<br />
is assumed to avoid the vertical and torsional image separation.<br />
The three-step test is important <strong>in</strong> the evaluation of vertical diplopia:<br />
1. Determ<strong>in</strong>e whether there is a right or left hypertropia or hyperphoria <strong>in</strong> primary<br />
position. For example, if there is a right hypertropia <strong>in</strong> primary position, there is<br />
paresis of the right eye depressors (right <strong>in</strong>ferior rectus or superior oblique) or left<br />
eye elevators (left superior rectus or <strong>in</strong>ferior oblique).<br />
2. Compare the amount of vertical deviation <strong>in</strong> right and left gaze. For example, if the<br />
right hypertropia <strong>in</strong>creases <strong>in</strong> left gaze, either the right superior oblique or the left<br />
superior rectus is underact<strong>in</strong>g.<br />
3. Compare the vertical deviation <strong>in</strong> right head tilt and left head tilt (Bielschowsky<br />
maneuver). For example, if the vertical deviation <strong>in</strong>creases with right head tilt, the<br />
right superior oblique must be weak; if the hyperdeviation <strong>in</strong>creases on left head tilt,<br />
the left superior rectus is weak.<br />
Ocular torsion may be measured with the double Maddox rod test, which utilizes a<br />
red Maddox rod over the right eye and a white Maddox rod over the left eye <strong>in</strong> a trial<br />
frame. A th<strong>in</strong> base-down prism may be placed before one eye to separate the horizontal<br />
l<strong>in</strong>es <strong>in</strong>duced. The tilt of the ret<strong>in</strong>al image is opposite the tilt of the horizontal l<strong>in</strong>e, as<br />
seen by the patient. Therefore, when the l<strong>in</strong>e is seen slanted toward the nose, an<br />
excyclodeviation is present, whereas if the tilt is toward the temple, an <strong>in</strong>cyclodeviation<br />
is present. A simple mnemonic is that the l<strong>in</strong>e is always tilted <strong>in</strong> the direction <strong>in</strong> which<br />
the offend<strong>in</strong>g muscle would rotate the eye if it were act<strong>in</strong>g alone (von Noorden, 1996).<br />
For example, a patient with right superior oblique muscle palsy will describe the red<br />
l<strong>in</strong>e to be lower than the white l<strong>in</strong>e and relatively <strong>in</strong>torted or slanted toward the nose.<br />
The Maddox rod is then turned until the two l<strong>in</strong>es are parallel and the magnitude of the<br />
cyclotropia can be read off the trial frame. Cyclodeviation may also be noted with
234 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
<strong>in</strong>direct ophthalmoscopy (von Noorden, 1996). Normally, the average location of the<br />
fovea <strong>in</strong> relation to the optic nerve head is 0.3 disc diameters below a horizontal l<strong>in</strong>e<br />
extend<strong>in</strong>g through the geometric center of the optic disc. From this position, an<br />
imag<strong>in</strong>ary horizontal l<strong>in</strong>e will cross the optic nerve head just below the halfway<br />
po<strong>in</strong>t between its geometric center and lower pole. Incyclotropia is present when the<br />
fovea appears above a l<strong>in</strong>e extend<strong>in</strong>g horizontally from the center of the optic nerve<br />
head, and excyclotropia is present when the fovea is below a l<strong>in</strong>e extend<strong>in</strong>g horizontally<br />
from just below the lower pole of the optic disc. F<strong>in</strong>ally, if restrictive ophthalmopathy is<br />
thought to be responsible for vertical misalignment, the forced duction test can be used<br />
to differentiate whether limitation of ocular movement is due to paresis of extraocular<br />
muscle or tether<strong>in</strong>g of the antagonist extraocular muscle.<br />
What Are the Etiologies of Vertical Diplopia?<br />
B<strong>in</strong>ocular vertical diplopia may be due to supranuclear processes, ocular motor nerve<br />
dysfunction, neuromuscular junction disease, diseases of eye muscle, mechanical<br />
processes caus<strong>in</strong>g vertical eye misalignment, and even ret<strong>in</strong>al disease (Brazis, 1998a).<br />
Etiologies responsible for vertical b<strong>in</strong>ocular diplopia and hypertropia=hyperphoria are<br />
outl<strong>in</strong>ed <strong>in</strong> Table 10–7.<br />
What Supranuclear Processes May Cause<br />
Vertical Diplopia?<br />
Monocular elevation paresis (‘‘double elevator palsy’’) may occur on a peripheral basis<br />
(e.g., due to primary <strong>in</strong>ferior rectus restriction, primary superior rectus palsy, myasthenia<br />
gravis, or a fascicular third nerve lesion) or with pretectal supranuclear lesions.<br />
Supranuclear monocular elevation paresis may be congenital (Bell, 1990; Ziffer, 1992) or<br />
acquired, with the latter due to a lesion contralateral to the paretic eye or ipsilateral to<br />
the paretic eye that <strong>in</strong>terrupt efferents from the rostral <strong>in</strong>terstitial nucleus of the MLF<br />
to the superior rectus and <strong>in</strong>ferior oblique subnuclei (Hommel, 1990; Thomke, 1992).<br />
Double elevator palsy may simply be an asymmetric upgaze palsy that cl<strong>in</strong>ically<br />
presents as monocular elevation paresis <strong>in</strong> the more severely affected eye (Thomke,<br />
1992). Patients do not have subjective diplopia <strong>in</strong> primary position and demonstrate<br />
limitation of monocular elevation that is the same from primary position, adduction, or<br />
abduction, confirm<strong>in</strong>g a supranuclear basis for the elevation impairment. Patients may<br />
have associated pupillary abnormalities, convergence impairment, and other neurologic<br />
signs of bra<strong>in</strong>stem <strong>in</strong>volvement but do not have ptosis, lid retraction, proptosis, positive<br />
forced ductions, or a positive Tensilon test. A vertical one-and-a-half syndrome, with<br />
vertical upgaze palsy and monocular paresis of downgaze on the side of the lesion or<br />
contralateral to the lesion, and skew deviation, vertical misalignment result<strong>in</strong>g from<br />
supranuclear derangements, may also cause vertical diplopia and are described <strong>in</strong><br />
Chapter 14. The ocular tilt reaction (OTR), discussed <strong>in</strong> Chapter 14, may also be<br />
associated with vertical diplopia. A tonic OTR may simulate superior oblique palsy<br />
(Donahue, 1999). Five patients with OTR had a three-step test suggest<strong>in</strong>g superior<br />
oblique palsy (bilateral <strong>in</strong> one patient). However, no patient had the expected excyclotorsion<br />
of the hypertropic eye. Two patients had conjugate ocular torsion (<strong>in</strong>torsion of
Table 10–7. Etiologies of B<strong>in</strong>ocular Vertical Diplopia and Hypertropia=Hyperphoria<br />
Diplopia 235<br />
Supranuclear causes<br />
Supranuclear monocular elevation paresis (congenital or acquired)<br />
Skew deviation<br />
Vertical one-and-a-half syndrome<br />
Wernicke’s syndrome<br />
Paroxysmal superior rectus and levator palpebrae spasm with multiple sclerosis<br />
Vitreous hemorrhage<br />
Ocular motor nerve dysfunction<br />
Third nerve palsy<br />
Fourth nerve palsy<br />
Hypertropia (small) accompany<strong>in</strong>g sixth nerve palsy<br />
Superior oblique myokymia<br />
Ocular neuromyotonia<br />
Ophthalmoplegic migra<strong>in</strong>e<br />
Wernicke’s syndrome<br />
Fisher’s syndrome<br />
Guilla<strong>in</strong>-Barré syndrome<br />
Decompensation of a long-stand<strong>in</strong>g phoria<br />
Increased <strong>in</strong>tracranial pressure<br />
<strong>Neuro</strong>muscular junction disease<br />
Myasthenia gravis<br />
Botulism<br />
Diseases of the eye muscle<br />
Isolated paresis of a vertical-act<strong>in</strong>g extraocular muscle (e.g., due to congenital causes, myasthenia<br />
gravis, Graves’ disease, botulism, trauma, postsurgery, trochleitis, orbital metastasis, orbital<br />
pseudotumor, muscle ischemia form giant cell arteritis, etc.)<br />
Superior oblique muscle<br />
Inferior oblique muscle<br />
Superior rectus muscle<br />
Inferior rectus muscle<br />
Decompensation of a long-stand<strong>in</strong>g phoria<br />
Graves’ disease<br />
Chronic progressive external ophthalmoplegia (CPEO) syndromes<br />
After surgery (e.g., cataract operation)<br />
Congenital strabismus syndromes<br />
Dissociated vertical deviation (DVD)<br />
Congenital ‘‘double elevator’’ palsy (monocular elevation deficiency)<br />
Double depressor paralysis (unilateral paralysis of the <strong>in</strong>ferior rectus and superior oblique; may be<br />
congenital or acquired)<br />
Physiologic hyperdeviation on lateral gaze (asymptomatic)<br />
Mechanical processes caus<strong>in</strong>g vertical eye misalignment<br />
Graves’ disease<br />
Brown’s superior oblique tendon sheath syndrome<br />
Congenital<br />
Acquired (e.g., superomedial orbital trauma, tenosynovitis or myositis, adhesions, metastasis to<br />
the superior oblique muscle, frontal s<strong>in</strong>us osteoma, pans<strong>in</strong>usitis, psoriasis, peribulbar<br />
anesthesia, blepharoplasty, maxillofacial or s<strong>in</strong>us surgery, and superior oblique tuck)<br />
(cont<strong>in</strong>ued)
236 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 10–7. (cont<strong>in</strong>ued)<br />
Superior oblique click syndrome (e.g., due to schwannoma or giant cell tumor of sheath of<br />
superior oblique tendon)<br />
Acquired Brown’s syndrome associated with underaction of the ipsilateral superior oblique muscle<br />
(‘‘can<strong>in</strong>e tooth syndrome’’)<br />
Orbital floor blowout fracture<br />
Maxillary s<strong>in</strong>usitis (silent s<strong>in</strong>us syndrome)<br />
Direct trauma to the extraocular muscles (e.g., <strong>in</strong>tramuscular hematoma)<br />
Congenital <strong>in</strong>ferior rectus fibrosis<br />
<strong>An</strong>omalous orbital structures, such as extraocular muscles <strong>in</strong>sert<strong>in</strong>g <strong>in</strong>to an abnormal location,<br />
fibrous bands, and discrete anomalous muscles (Lueder, 2002)<br />
Strabismus fixus (generalized fibrosis of extraocular muscles)<br />
Postoperative sequelae (<strong>in</strong>clud<strong>in</strong>g ret<strong>in</strong>al detachment surgery, orbital surgery, strabismus surgery,<br />
and cataract surgery)<br />
Orbital <strong>in</strong>flammation (myositis) and pseudotumor<br />
Metastatic <strong>in</strong>filtration of extraocular muscles<br />
Orbital tumors<br />
Fallen eye syndrome (long-stand<strong>in</strong>g superior oblique muscle paresis <strong>in</strong> patients who habitually<br />
fixate with the paretic eye may develop hypodeviation of the un<strong>in</strong>volved eye caused by<br />
contracture of the contralateral <strong>in</strong>ferior rectus muscle)<br />
Ris<strong>in</strong>g eye syndrome (long-stand<strong>in</strong>g <strong>in</strong>ferior oblique muscle palsy may result <strong>in</strong> contracture and<br />
fibrosis of the contralateral superior rectus<br />
Miscellaneous<br />
Hemifield slip phenomenon from dense bitemporal hemianopsia or heteronymous altitud<strong>in</strong>al field<br />
defects<br />
Foveal displacement syndrome (e.g., due to subret<strong>in</strong>al or preret<strong>in</strong>al neovascular membranes)<br />
the hypertropic eye and extorsion of the hypotropic eye) and two patients had only<br />
<strong>in</strong>torsion of the hypotropic eye. All had neurologic deficits consistent with more<br />
widespread bra<strong>in</strong>stem disease. The authors concluded that vertical ocular deviations<br />
that three-step to a superior oblique palsy are not always caused by fourth nerve<br />
weakness. When a patient with an apparent fourth nerve palsy has ocular torsion<br />
<strong>in</strong>consistent with a superior oblique (SO) palsy, OTR should be suspected, especially if<br />
posterior fossa or vestibular dysfunction coexist. Because results of the Bielschowsky<br />
head tilt test may be positive <strong>in</strong> patients with the OTR, the feature dist<strong>in</strong>guish<strong>in</strong>g OTR<br />
from SO palsy is the direction of torsion. The authors advocate a fourth step—<br />
evaluation of ocular torsion—<strong>in</strong> addition to the standard three steps.<br />
Wernicke’s syndrome is due to thiam<strong>in</strong>e deficiency and is especially seen with chronic<br />
alcohol abuse. Patients with Wernicke’s encephalopathy may compla<strong>in</strong> of vertical<br />
diplopia due to a supranuclear or nuclear lesion. This is associated with other signs<br />
of bra<strong>in</strong>stem and cerebellar dysfunction (e.g., nystagmus, gaze palsies, gait ataxia),<br />
confusion, memory impairment, and peripheral polyneuropathy.<br />
Paroxysmal superior rectus and levator palpebrae spasm is a rare and unique disorder<br />
described <strong>in</strong> a s<strong>in</strong>gle patient with multiple sclerosis (Ezra, 1996a). Paroxysms of vertical<br />
diplopia and lid retraction <strong>in</strong> this patient lasted 3 to 4 seconds and exam<strong>in</strong>ation revealed<br />
<strong>in</strong>termittent right hypertropia, lid retraction, and restriction of downgaze. MRI revealed<br />
multiple lesions consistent with multiple sclerosis, <strong>in</strong>clud<strong>in</strong>g a lesion <strong>in</strong> the midbra<strong>in</strong> <strong>in</strong>
the region of the third nerve fascicle. Carbamazep<strong>in</strong>e stopped all of the symptoms that<br />
were believed due to spontaneous spasm of the superior rectus=levator complex.<br />
What Cranial Nerve Impairments Cause<br />
Vertical Diplopia?<br />
Diplopia 237<br />
Third nerve palsies may cause vertical and horizontal b<strong>in</strong>ocular diplopia and are<br />
discussed <strong>in</strong> Chapter 11. Fourth cranial nerve palsies are a common cause of acquired<br />
b<strong>in</strong>ocular vertical diplopia (von Noorden, 1986, 1996) and are discussed <strong>in</strong> Chapter 12.<br />
Increased <strong>in</strong>tracranial pressure may rarely cause transient diplopia (Harr<strong>in</strong>gton,<br />
1953). A patient has been described with recurrent attacks of a right third nerve palsy<br />
caus<strong>in</strong>g diplopia <strong>in</strong> a sett<strong>in</strong>g of headaches, papilledema, periodic ur<strong>in</strong>ary <strong>in</strong>cont<strong>in</strong>ence,<br />
and other neurologic f<strong>in</strong>d<strong>in</strong>gs. The transient third nerve palsy lasted about 5 m<strong>in</strong>utes<br />
and eventually the patient developed a persistent third nerve palsy. At autopsy,<br />
the patient had right frontal and temporal bra<strong>in</strong> metastases with herniation of the<br />
hippocampal gyrus that stretched the right third nerve.<br />
Superior oblique myokymia (SOM) is a rare disorder of unknown etiology characterized<br />
symptomatically by oscillopsia, episodic vertical or torsional diplopia, or both. This<br />
disorder is discussed <strong>in</strong> Chapter 17. Ocular neuromyotonia (ONM) may also cause<br />
vertical diplopia and is discussed above.<br />
Ophthalmoplegic migra<strong>in</strong>e usually starts <strong>in</strong> the first decade of life and usually affects<br />
the oculomotor nerve, although rare trochlear nerve or multiple ocular motor nerve<br />
<strong>in</strong>volvement has been described (Miller, 1991). <strong>Cl<strong>in</strong>ical</strong> criteria essential for the<br />
diagnosis of ophthalmoplegic migra<strong>in</strong>e <strong>in</strong>clude (1) a history of typical migra<strong>in</strong>e<br />
headache (severe, throbb<strong>in</strong>g, unilateral but occasionally bilateral or alternat<strong>in</strong>g), with<br />
headaches possibly last<strong>in</strong>g hours or days; (2) ophthalmoplegia that may <strong>in</strong>clude one or<br />
more nerves and may alternate sides with attacks (extraocular muscle paralysis may<br />
occur with the first attack of headache or, rarely, precede it; usually, however, the<br />
paralysis appears subsequent to an established migra<strong>in</strong>e pattern; and (3) exclusion of<br />
other causes, by neuroimag<strong>in</strong>g, surgery, or autopsy. Friedman et al studied 5000 patients<br />
with migra<strong>in</strong>e and found eight examples (0.16%) of ophthalmoplegic migra<strong>in</strong>e (Friedman,<br />
1962). All eight patients had recurrent attacks of headache (usually orbital),<br />
usually accompanied by nausea and vomit<strong>in</strong>g, and an ipsilateral third nerve palsy.<br />
The third nerve paresis reached a maximum as the headache began to resolve and<br />
persisted for 1 to 4 weeks. The third nerve paralysis dur<strong>in</strong>g the attack is often complete<br />
or nearly so, but partial third nerve paresis, <strong>in</strong>clud<strong>in</strong>g superior division third nerve<br />
paresis, may occur (Katz, 1989). Most patients have normal neuro-ophthalmologic<br />
exam<strong>in</strong>ations between attacks, but some patients may demonstrate partial third nerve<br />
paresis or even signs of aberrant regeneration. The differential diagnosis of ophthalmoplegic<br />
migra<strong>in</strong>e is that of pa<strong>in</strong>ful ophthalmoplegia <strong>in</strong> general; dur<strong>in</strong>g the <strong>in</strong>itial attack,<br />
structural lesions, especially aneurysms, should be suspected, and the evaluation is that<br />
as described for third nerve palsies (Chapter 11). The diagnosis should thus be made<br />
with caution, especially if the first attack occurs <strong>in</strong> adulthood, and only after other<br />
causes of pa<strong>in</strong>ful ophthalmoplegia have been excluded by appropriate laboratory and<br />
neuroimag<strong>in</strong>g studies (Harr<strong>in</strong>gton, 1953).
238 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Fisher’s syndrome (ophthalmoplegia associated with ataxia and areflexia) or<br />
Guilla<strong>in</strong>-Barré syndrome (associated with diffuse muscle paresis, areflexia, etc.) may<br />
also be associated with vertical diplopia (Spector, 1993). Miller Fisher syndrome,<br />
Guilla<strong>in</strong>-Barré syndrome with ophthalmoplegia, Bickerstaff’s bra<strong>in</strong>stem encephalitis,<br />
and acute ophthalmoparesis without ataxia are all commonly associated with a<br />
common autoantibody (anti-GQ1b IgG antibodies) (Odaka, 2001; Yuki, 2001).<br />
Although patients with sixth cranial nerve palsies ma<strong>in</strong>ly compla<strong>in</strong> of horizontal<br />
b<strong>in</strong>ocular diplopia with esotropia or esophoria on exam<strong>in</strong>ation, some patients compla<strong>in</strong><br />
of vertical as well as horizontal diplopia (Slav<strong>in</strong>, 1989). Hyperdeviation with sixth nerve<br />
palsies may occur <strong>in</strong> primary gaze but is usually most prom<strong>in</strong>ent to the side of the palsy<br />
with the hyperdeviation measur<strong>in</strong>g 4 to 16 prism diopters. Vertical ductions <strong>in</strong> patients<br />
with isolated sixth nerve palsies with hyperdeviation are normal, no torsion is evident<br />
on double Maddox rod test<strong>in</strong>g, and head tilt test is almost always negative. In rare<br />
cases, up- or down-shoot<strong>in</strong>g of the paretic eye may be noted on attempted abduction.<br />
The hyperdeviation may be due to mechanical factors (decreased vertical stabilization<br />
of globe due to weak lateral rectus) or vertical substitution movement <strong>in</strong> face of one<br />
paretic muscle (Slav<strong>in</strong>, 1989). Although hyperdeviation may occur with isolated sixth<br />
nerve palsies, one must always be concerned that the hyperdeviation <strong>in</strong> a patient with a<br />
sixth nerve palsy reflects concomitant <strong>in</strong>volvement of the third or fourth cranial nerve<br />
(e.g., with cavernous s<strong>in</strong>us pathology), associated skew deviation, or myasthenia gravis.<br />
Conditions suggest<strong>in</strong>g additional causes for a hyperdeviation with a sixth nerve palsy<br />
<strong>in</strong>clude a positive head tilt test, cyclotropia on double Maddox rod test<strong>in</strong>g, concomitant<br />
nystagmus or other signs of bra<strong>in</strong>stem dysfunction, associated ptosis, or decreased<br />
vertical muscle ductions.<br />
What Disease Processes Affect<strong>in</strong>g the<br />
<strong>Neuro</strong>muscular Junction Cause Vertical<br />
Diplopia?<br />
A common cause of <strong>in</strong>termittent diplopia is myasthenia gravis. The ocular misalignment<br />
worsens with fatigue and improves with rest or Tensilon <strong>in</strong>jection. Myasthenia<br />
gravis may masquerade as a fourth nerve palsy with vertical duction limitations,<br />
cyclotropia, and a positive head tilt test. Increased vertical deviation with gaze hold<strong>in</strong>g<br />
and improvement of the deviation after eye closure <strong>in</strong> these cases are suggestive of<br />
myasthenia gravis. Botulism may also be associated with vertical diplopia.<br />
What Disease Processes Affect<strong>in</strong>g the<br />
Extraocular Muscles Cause Vertical Diplopia?<br />
Isolated paresis of a vertical-act<strong>in</strong>g extraocular muscle may cause vertical b<strong>in</strong>ocular<br />
diplopia. In all such cases, myasthenia gravis should be considered. Other etiologies of<br />
isolated vertical-act<strong>in</strong>g extraocular muscle palsy <strong>in</strong>clude local trauma (e.g., cataract<br />
surgery), vascular disease (especially muscle ischemia with giant cell arteritis), thyroid<br />
ophthalmopathy, congenital causes, and the etiologies of restrictive ophthalmopathy<br />
noted below (von Noorden, 1991, 1996). Isolated superior rectus palsy causes ipsilateral
Diplopia 239<br />
hypotropia <strong>in</strong> primary position, impaired eye elevation <strong>in</strong> abduction, small excyclotropia,<br />
and absent Bell’s phenomenon. Head tilt is usually toward the sound side but<br />
may be toward the side of palsy. Superior rectus palsy may be associated with<br />
ipsilateral ptosis, especially <strong>in</strong> congenital cases. Also, a pseudoptosis may be noted<br />
on the side of hypotropia when the nonparetic eye fixates. Isolated <strong>in</strong>ferior rectus palsy<br />
is often congenital and results <strong>in</strong> hypertropia <strong>in</strong> primary gaze, <strong>in</strong>cyclotropia, impaired<br />
depression of the eye <strong>in</strong> abduction, pseudoptosis of the sound eye when the paretic eye<br />
fixates, and a head tilt to either side. Isolated <strong>in</strong>ferior oblique muscle paresis is rare and<br />
results <strong>in</strong> hypotropia <strong>in</strong> primary gaze, impaired elevation <strong>in</strong> adduction, <strong>in</strong>cyclotropia,<br />
and a head tilt, most often toward the paralyzed side. The head tilt test is positive on<br />
tilt<strong>in</strong>g the head toward the normal side. Although superior oblique muscle paresis is<br />
most often due to fourth nerve palsies, this muscle may also be affected by myasthenia<br />
gravis, botulism, trochleitis, orbital metastasis, orbital pseudotumor (myositis), and<br />
trauma to the trochlea (Moster, 1992; Spector, 1993; von Noorden, 1986). Occasionally<br />
damage to the trochlea (e.g., due to dog bite or frontal s<strong>in</strong>us surgery) may cause<br />
acquired Brown’s syndrome (see below) associated with underaction of the ipsilateral<br />
superior oblique muscle referred to as the ‘‘can<strong>in</strong>e tooth syndrome.’’ Isolated superior<br />
oblique myositis may cause mild limitation of elevation of the eye <strong>in</strong> adduction<br />
(Stidham, 1998).<br />
Rarely, vertical b<strong>in</strong>ocular diplopia may occur from chronic progressive external<br />
ophthalmoplegia (often associated with ptosis, orbicularis oculi paresis, and occasionally<br />
pigmentary ret<strong>in</strong>opathy). These patients more often have m<strong>in</strong>imal or no diplopia<br />
despite prom<strong>in</strong>ent external ophthalmoplegia.<br />
Decompensation of a long-stand<strong>in</strong>g phoria may cause hypertropia and vertical<br />
diplopia (Burde, 1991). A phoria will become manifest and break down <strong>in</strong>to a tropia<br />
if fusion is broken. This occurs transiently by occlud<strong>in</strong>g or blurr<strong>in</strong>g vision <strong>in</strong> one eye,<br />
when a patient is tired, when a patient has taken a central nervous system depressant<br />
such as alcohol or sedative medications, or dur<strong>in</strong>g a febrile illness. More persistent<br />
decompensation may occur after head trauma, with chang<strong>in</strong>g refractive needs, and<br />
often for unclear reasons. <strong>Neuro</strong>-ophthalmologic history and exam<strong>in</strong>ation often reveal<br />
supportive evidence for a long-stand<strong>in</strong>g strabismus <strong>in</strong>clud<strong>in</strong>g a history of childhood<br />
strabismus or patch<strong>in</strong>g, the presence of a head tilt or turn (old photos), and large vertical<br />
fusional amplitude (6 to 20 prism diopters). Vertical fusional amplitudes are measured<br />
by present<strong>in</strong>g vertically oriented prisms of gradually <strong>in</strong>creas<strong>in</strong>g strength <strong>in</strong> front of one<br />
eye after first neutraliz<strong>in</strong>g any manifest tropia. The amount of prism needed to produce<br />
diplopia over that needed to neutralize the tropia (if present) represents the fusional<br />
amplitude (normal vertical fusional amplitudes are two to four prism diopters).<br />
A number of congenital conditions may be associated with vertical deviation of the<br />
eyes without vertical diplopia. These conditions <strong>in</strong>clude congenital strabismus<br />
syndromes, dissociated vertical deviation, congenital double elevator or double depressor<br />
palsy, and asymptomatic physiologic hyperdeviation on lateral gaze. Congenital<br />
strabismus syndromes may be associated with overaction of the <strong>in</strong>ferior or superior<br />
oblique muscles, caus<strong>in</strong>g V-pattern exotropia or esotropia and A-pattern exotropia or<br />
esotropia, respectively. Patients with these patterns of congenital strabismus do not<br />
have hyperdeviation <strong>in</strong> primary gaze, do not have a positive head tilt test, do not have<br />
ductional limitation, and do not have cyclodeviation. Dissociated vertical (hyper)deviation<br />
(DVD), characterized by upward turn of the nonfixat<strong>in</strong>g eye, may also accompany<br />
congenital strabismus, especially esotropia. Monocular occlusion of either eye produces
240 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
elevation of the occluded eye without correspond<strong>in</strong>g depression of the uncovered eye<br />
(i.e., DVD does not follow Her<strong>in</strong>g’s law). The eye under cover ‘‘floats’’ up and out and<br />
may also excyclotort. After removal of the cover, the eye makes a slow downward<br />
movement to reach midl<strong>in</strong>e accompanied by <strong>in</strong>cycloduction. The deviation is often<br />
variable, bilateral, and asymmetric. There is no ductional limitation, head tilt test is<br />
negative, and the deviation is not gaze dependent. Congenital ‘‘double elevator’’ palsy<br />
(monocular elevation deficiency) may be due to <strong>in</strong>ferior rectus restriction (with positive<br />
forced ductions to elevation, no muscle paralysis, and normal saccades of the superior<br />
rectus), elevator weakness (with negative forced ductions, evidence of paralysis of<br />
vertical muscles, and reduced saccadic velocities <strong>in</strong> upgaze of the affected eye), or a<br />
comb<strong>in</strong>ation of <strong>in</strong>ferior rectus restriction and weak elevators (with positive forced<br />
ductions <strong>in</strong> elevation, reduced upward vertical saccadic velocities <strong>in</strong> <strong>in</strong>volved eye, and<br />
variable muscle paresis). Von Noorden noted that one must consider the possibility that<br />
double elevator paralysis is a misnomer and that generalized weakness of elevation is<br />
caused by a superior rectus palsy of long stand<strong>in</strong>g, the deviation hav<strong>in</strong>g spread<br />
throughout the entire upward field of gaze and the <strong>in</strong>ferior rectus hav<strong>in</strong>g become<br />
contracted (von Noorden, 1996). Double depressor paralysis (unilateral paralysis of the<br />
<strong>in</strong>ferior rectus and superior oblique) is rare and may be congenital or acquired (von<br />
Noorden, 1996). Aga<strong>in</strong>, von Noorden suspects that so-called double depressor paralyses<br />
are caused by <strong>in</strong>ferior rectus muscle paralysis of long stand<strong>in</strong>g and secondary superior<br />
rectus contracture (von Noorden, 1996).<br />
It should be noted that many patients may have asymptomatic physiologic hyperdeviation<br />
on lateral gaze. Slav<strong>in</strong> et al noted a physiologic hyperdeviation of greater than<br />
two prism diopters that simulates overaction of the <strong>in</strong>ferior oblique muscle, <strong>in</strong> 77% of<br />
normal subjects (Slav<strong>in</strong>, 1988). The hyperdeviation occurred <strong>in</strong> any field of gaze and<br />
never measured greater than 10 prism diopters. Forty-seven percent of the patients<br />
showed an isolated left hyperdeviation <strong>in</strong> right upgaze and right hyperdeviation <strong>in</strong> left<br />
upgaze, 32% had either a right hyperdeviation <strong>in</strong> left upgaze or a left hyperdeviation <strong>in</strong><br />
right upgaze, and <strong>in</strong> 85% of patients a V pattern of less than 15 prism diopters was<br />
detected. No patient had hyperdeviation <strong>in</strong> primary gaze or hyperdeviation <strong>in</strong>duced by<br />
head tilt, and no patients compla<strong>in</strong>ed of vertical diplopia. Thus, physiologic hyperdeviation<br />
should not be considered to expla<strong>in</strong> a hyperdeviation if there is a primary<br />
position hyperdeviation, if hyperdeviation is <strong>in</strong>duced by head tilt <strong>in</strong> primary gaze, if<br />
there is a significant downgaze hyperdeviation, if there is duction limitation, or if there<br />
is an uncrossed hyperdeviation <strong>in</strong> peripheral gaze (e.g., a left hyperdeviation on gaze to<br />
the left and up).<br />
What Mechanical Processes Cause Vertical<br />
Eye Misalignment?<br />
Restrictive ophthalmopathy may result <strong>in</strong> vertical b<strong>in</strong>ocular diplopia. Restrictive<br />
ophthalmopathy is def<strong>in</strong>ed as limitation of eye movement associated with a positive<br />
forced duction test. <strong>An</strong> <strong>in</strong>crease <strong>in</strong> <strong>in</strong>traocular pressure (>5 mm) <strong>in</strong> the direction aga<strong>in</strong>st<br />
the restriction is <strong>in</strong>direct evidence of restriction (differential <strong>in</strong>traocular pressure).<br />
Normal saccadic velocities favor a restrictive ophthalmopathy as a cause for diplopia<br />
rather than an ocular motor nerve palsy (Spector, 1993; von Noorden, 1996).
Diplopia 241<br />
Thyroid ophthalmopathy (Graves’ disease) is a common cause of horizontal or<br />
vertical diplopia. ‘‘Tightness’’ and restriction of the extraocular muscles preferentially<br />
affects the <strong>in</strong>ferior rectus, medial rectus, and superior rectus, <strong>in</strong> that order. Limitation of<br />
elevation <strong>in</strong> one or both eyes is by far the most common defect of ocular motility.<br />
Vertical misalignment with thyroid ophthalmopathy is usually associated with other<br />
characteristic signs, such as lid lag, lid retraction, and proptosis. Patients often have<br />
ductional limitation <strong>in</strong> the vertical plane and may also have cyclodeviation and a<br />
positive head tilt test. The forced duction test is often positive and the diagnosis is aided<br />
by demonstrat<strong>in</strong>g appropriate extraocular muscle enlargement with orbital echography,<br />
computed tomography (CT), or MRI.<br />
Thyroid ophthalmopathy may present as an apparent superior oblique muscle paresis<br />
on the three-step test and thus be confused with a fourth nerve palsy (Moster, 1992;<br />
Slav<strong>in</strong>, 1988). This cl<strong>in</strong>ical picture is caused by the restrictive process affect<strong>in</strong>g the<br />
opposite <strong>in</strong>ferior rectus muscle (hypotropic eye) with the hypotropia greatest <strong>in</strong> the field<br />
of superior rectus. Clues to the diagnosis of thyroid ophthalmopathy, rather than<br />
superior oblique weakness, <strong>in</strong> a patient with hyperdeviation, <strong>in</strong>clude the follow<strong>in</strong>g:<br />
1. Increased vertical deviation <strong>in</strong> upgaze.<br />
2. Increased <strong>in</strong>traocular tension (>5 mm) <strong>in</strong> upgaze.<br />
3. When a ‘‘subacute’’ or ‘‘chronic’’ superior oblique palsy is diagnosed (i.e., when the<br />
hyperdeviation is greater on upgaze), consider that a restrictive process may be<br />
operative; if ductions are normal, differential <strong>in</strong>traocular pressure should be done.<br />
4. If a patient with acute diplopia is found to have a hypertropia greater on upgaze<br />
rather than downgaze, a diagnosis of superior oblique palsy should be withheld.<br />
With Brown’s superior oblique tendon sheath syndrome, there is an <strong>in</strong>ability to<br />
elevate the adducted eye above the midhorizontal plane (von Noorden, 1996). This<br />
condition may be bilateral <strong>in</strong> about 10% of patients. A mechanical restriction to free<br />
movement of the superior oblique tendon at the pulley may prevent the upward and<br />
<strong>in</strong>ward movement of the globe, thus mimick<strong>in</strong>g paresis of the <strong>in</strong>ferior oblique muscle.<br />
Episodic vertical diplopia results from entrapment of the eye on gaze downward and<br />
<strong>in</strong>ward or <strong>in</strong> the field of action of the superior oblique. The eye may then release<br />
suddenly, occasionally associated with the sensation or actual hear<strong>in</strong>g of a click. Some<br />
m<strong>in</strong>imal restriction of elevation may persist even <strong>in</strong> full abduction and there may be<br />
slight down-shoot of the adducted eye mimick<strong>in</strong>g superior oblique overaction. Hypotropia<br />
may be evident <strong>in</strong> primary gaze, forced ductions are positive, there may be a<br />
compensatory head posture (ch<strong>in</strong> up due to hypotropia with a head turn toward the<br />
<strong>in</strong>volved side), and a V-pattern exotropia may be noted. This syndrome is often<br />
congenital but may be acquired due to superomedial orbital trauma, tenosynovitis or<br />
myositis, adhesions, metastasis to the superior oblique muscle, frontal s<strong>in</strong>us osteoma,<br />
pans<strong>in</strong>usitis, psoriasis, peribulbar anesthesia, blepharoplasty, implantation of an<br />
Ahmed valve glaucoma implant, maxillofacial or s<strong>in</strong>us surgery, and superior oblique<br />
tuck (Alonso-Valdivielso, 1993; Baker, 1991; Coats, 1999; Erie, 1990; Saunders, 1990;<br />
Thorne, 1999; von Noorden, 1996). In congenital cases, MRI may show enlargement of<br />
the tendon-trochlea complex with this complex be<strong>in</strong>g of irregular shape and of<br />
<strong>in</strong>termediate signal <strong>in</strong>tensity (Sener, 1996).<br />
The superior oblique click syndrome is a form of <strong>in</strong>termittent acquired Brown’s<br />
syndrome with a cl<strong>in</strong>ical picture that alternates between a Brown’s-type syndrome and<br />
a superior oblique muscle palsy (White, 1999). The cl<strong>in</strong>ical features depend on the
242 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
direction <strong>in</strong> which the muscle is impeded. The click, often audible to the patient and=or<br />
the exam<strong>in</strong>er, may signal the release of the restriction. The click is palpable <strong>in</strong> the<br />
superonasal orbit. Lesions are located with<strong>in</strong> the sheath of the anterior superior oblique<br />
tendon, and <strong>in</strong>clude schwannoma and giant cell tumor of the tendon.<br />
The differential of Brown’s syndrome <strong>in</strong>cludes primarily a paralysis of the <strong>in</strong>ferior<br />
oblique muscle. Forced ductions separate these syndromes. Other restrictions of<br />
elevation (such as thyroid ophthalmopathy, fibrosis of the <strong>in</strong>ferior rectus muscle,<br />
double elevator palsy, and orbital floor fractures) usually cause restriction of elevation<br />
from any gaze position and are not limited to restriction of elevation <strong>in</strong> adduction (von<br />
Noorden, 1996). However, orbital floor fracture and thyroid ophthalmopathy may<br />
simulate Brown’s syndrome (Hudson, 1992; Hughes, 1992; von Noorden, 1996).<br />
Orbital blow-out fractures frequently <strong>in</strong>carcerate the <strong>in</strong>ferior rectus muscle and its<br />
surround<strong>in</strong>g tissue. Characteristics f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clude the follow<strong>in</strong>g (Baker, 1991; Egbert,<br />
2000):<br />
1. Ecchymosis of the <strong>in</strong>volved eye.<br />
2. Diplopia often present <strong>in</strong> all positions of gaze immediately posttrauma. This diplopia<br />
may persist <strong>in</strong> upgaze or downgaze.<br />
3. Paresthesia of the <strong>in</strong>fraorbital area due to damage to the <strong>in</strong>fraorbital nerve.<br />
4. Enophthalmos, either early or late.<br />
5. Entrapment of the <strong>in</strong>ferior rectus, <strong>in</strong>ferior oblique, and=or surround<strong>in</strong>g tissue. This<br />
results <strong>in</strong> restriction of upward gaze with positive forced duction test<strong>in</strong>g. Inferior<br />
rectus paresis, result<strong>in</strong>g <strong>in</strong> hypertropia <strong>in</strong> primary position <strong>in</strong> the <strong>in</strong>volved eye, may<br />
also occur due to direct nerve or muscle trauma.<br />
6. Hypotropia <strong>in</strong> primary position that <strong>in</strong>creases <strong>in</strong> upgaze.<br />
7. Frequent <strong>in</strong>traocular damage.<br />
Other causes of restrictive ophthalmopathy <strong>in</strong>clude direct trauma to the extraocular<br />
muscles (e.g., <strong>in</strong>tramuscular hematoma), congenital <strong>in</strong>ferior rectus fibrosis (often with<br />
ipsilateral ptosis), strabismus fixus (generalized fibrosis of extraocular muscles), postoperative<br />
sequelae (<strong>in</strong>clud<strong>in</strong>g ret<strong>in</strong>al detachment surgery, orbital surgery, strabismus<br />
surgery, s<strong>in</strong>us surgery, and cataract surgery), orbital <strong>in</strong>flammation (myositis) and<br />
pseudotumor, metastatic <strong>in</strong>filtration of extraocular muscles, and other orbital tumors<br />
(Carter, 1998; Engle, 1997; Hupp, 1990; Spector, 1993). Orbital lesions are usually<br />
associated with pa<strong>in</strong>, proptosis, chemosis, or other f<strong>in</strong>d<strong>in</strong>gs that betray their location.<br />
Also, with long-stand<strong>in</strong>g muscle paralysis, the antagonist muscle may become contractured<br />
and fibrotic. Thus, patients with long-stand<strong>in</strong>g superior oblique muscle paresis<br />
who habitually fixate with the paretic eye may develop the fallen eye syndrome<br />
(Spector, 1993). This syndrome manifests as a unilateral superior oblique muscle paresis<br />
present<strong>in</strong>g with hypodeviation of the un<strong>in</strong>volved eye that worsens <strong>in</strong> abduction caused<br />
by contracture of the contralateral <strong>in</strong>ferior rectus muscle. Conversely, long-stand<strong>in</strong>g<br />
<strong>in</strong>ferior oblique muscle palsy may result <strong>in</strong> the ris<strong>in</strong>g eye syndrome due to contracture<br />
and fibrosis of the contralateral superior rectus muscle (the contralateral eye rises<br />
dur<strong>in</strong>g attempted abduction) (Spector, 1993). Transient recurrent vertical diplopia, likely<br />
due to <strong>in</strong>termittent transient fusion impairment, has been described with maxillary<br />
s<strong>in</strong>usitis associated with lower<strong>in</strong>g of the orbital floor (silent s<strong>in</strong>us syndrome) (Borruat,<br />
1999; Kubis, 2000; Wan, 2000). The spectrum of silent s<strong>in</strong>us syndrome <strong>in</strong>cludes<br />
enophthalmos, hypoglobus, transient vertical diplopia, lid retraction, lagophthalmos,<br />
and blurred vision (Kubis, 2000; Wan, 2000).
Diplopia 243<br />
The congenital <strong>in</strong>ferior rectus fibrosis syndrome is a rare familial or sporadic<br />
syndrome manifested by downward fixation of one or both eyes associated with<br />
marked ptosis, restricted eye elevation (of equal magnitude from adduction, primary<br />
position, and abduction), positive forced ductions, and a backward (ch<strong>in</strong>-up) head tilt<br />
(Engle, 1997; von Noorden, 1996). CT imag<strong>in</strong>g may show atrophy of the <strong>in</strong>volved<br />
<strong>in</strong>ferior rectus muscle (Hupp, 1990). In some cases pathologic studies have shown<br />
absence of the superior division of the oculomotor nerve and its correspond<strong>in</strong>g alpha<br />
motor neurons, and abnormalities of the levator palpebrae superioris and rectus<br />
superior (the muscles <strong>in</strong>nervated by the superior division of the oculomotor nerve).<br />
Thus, congenital fibrosis of the extraocular muscles likely results from an abnormality <strong>in</strong><br />
the development of the extraocular muscle lower motor neuron system (Engle, 1997).<br />
Vertical diplopia after cataract surgery requires some comment. Three categories of<br />
strabismus or diplopia have been noted after cataract surgery (Hunter, 1995):<br />
1. Preexist<strong>in</strong>g condition (e.g., thyroid eye disease) <strong>in</strong> which misalignment was masked<br />
by a dense cataract.<br />
2. Conditions secondary to the prolonged occlusion by the cataract (e.g., sensory<br />
deprivation). Disruption of b<strong>in</strong>ocularity may be caused by long-term occlusion of<br />
vision by the cataract. Also, fusional amplitude can be reduced by the occlud<strong>in</strong>g<br />
cataract. Here the improved vision after surgery renders the preexist<strong>in</strong>g ocular<br />
conditions symptomatic.<br />
3. Surgical trauma to extraocular muscles or orbital soft tissue (<strong>in</strong>jury to <strong>in</strong>ferior rectus<br />
muscle caus<strong>in</strong>g paresis or contracture is most common).<br />
Capó and Guyton studied 19 patients with vertical strabismus after cataract surgery<br />
and noted that the vertical deviation was greater <strong>in</strong> the field of action of the presumed<br />
tight muscle <strong>in</strong> 16 of the 19 patients (Capó, 1996a). <strong>An</strong> ipsilateral hypertropia with<br />
superior rectus muscle overaction subsequently developed <strong>in</strong> two patients with an<br />
<strong>in</strong>itial hypotropia. The authors noted that myotoxicity from direct <strong>in</strong>jection of local<br />
anesthetics <strong>in</strong>to an extraocular muscle probably causes transient paresis followed by<br />
segmental contracture of the <strong>in</strong>volved muscle. Mild contractures result <strong>in</strong> strabismus<br />
with a motility pattern of an overactive muscle, whereas larger amounts of contracture<br />
lead to restrictive strabismus. In another prospective study of 20 consecutive patients<br />
with acquired vertical diplopia after cataract surgery, Capó et al noted that 50% of<br />
<strong>in</strong>volved muscles were overactive, 39% were restricted, and 11% were paretic (Capó,<br />
1996b). In overactive strabismus, versions showed overaction of the affected muscle<br />
with no significant underaction of its antagonist, the deviation <strong>in</strong>creased <strong>in</strong> the field of<br />
action of the affected muscle, and forced ductions were negative or mildly positive. In<br />
restrictive cases, the affected muscle was tight by forced duction test<strong>in</strong>g and the<br />
deviation was either comitant or worse <strong>in</strong> the field of action of the antagonist muscle.<br />
In paretic cases, the diagnosis was reached by limited ductions <strong>in</strong> the field of action of<br />
the affected muscle, accompanied by negative forced-duction test<strong>in</strong>g. The <strong>in</strong>ferior rectus<br />
was <strong>in</strong>volved <strong>in</strong> 17 patients (61%) and superior rectus muscle <strong>in</strong> 11 (39%). The odds of<br />
damag<strong>in</strong>g the <strong>in</strong>ferior rectus, as opposed to superior rectus, with peribulbar anesthesia<br />
was 4.8 times higher than with retrobulbar blocks. The authors drew the follow<strong>in</strong>g<br />
conclusions concern<strong>in</strong>g motility disturbances caus<strong>in</strong>g acquired strabismus after cataract<br />
surgery:<br />
1. Myotoxic effects of local anesthesia could result <strong>in</strong> temporary or permanent muscle<br />
weakness.
244 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
2. Superior rectus overaction may occur from superior rectus contracture secondary to<br />
temporary paresis of the <strong>in</strong>ferior rectus muscle caused by local anesthetic.<br />
3. Inferior rectus muscle contracture may result from direct penetration by needle, with<br />
elevated tissue pressure due to hematoma or a large amount of anesthetic with<strong>in</strong> the<br />
muscle, followed by secondary vascular compromise and ensu<strong>in</strong>g muscle fibrosis.<br />
4. In the series, restrictive and overactive motility disorders predom<strong>in</strong>ate, suggest<strong>in</strong>g<br />
that most cases with persistent vertical strabismus after cataract surgery result from<br />
muscle contracture rather than from permanent muscle paresis.<br />
The authors concluded that <strong>in</strong> this patient population, permanent vertical strabismus<br />
after cataract surgery results more often from overact<strong>in</strong>g or restricted muscles than from<br />
primary muscle paresis. Both the superior and <strong>in</strong>ferior recti can be <strong>in</strong>jured with<br />
retrobulbar anesthesia, but peribulbar <strong>in</strong>jections affect the <strong>in</strong>ferior rectus muscle more<br />
frequently. In the discussion of this paper, Guyton noted that vertical strabismus after<br />
cataract surgery is mostly caused by the local anesthetic block (Capó, 1996b). He stated<br />
that local anesthetic ‘‘kills’’ muscle, and that the muscle regenerates <strong>in</strong> young people<br />
with no sequelae, but <strong>in</strong> the elderly, regeneration is m<strong>in</strong>imal, a scar forms, and<br />
contracture occurs, caus<strong>in</strong>g strabismus. Occasionally, a permanent extraocular muscle<br />
paresis occurs, possibly because of nerve damage, but this mechanism is still not clear.<br />
Also, Corboy and Jiang reported 31 cases of postoperative hypotropia follow<strong>in</strong>g 2143<br />
cataract operations and noted that myotoxicity or perimuscular <strong>in</strong>flammation from<br />
anesthesia likely produced contracture hypotropia and restricted elevation of the globe<br />
(Corboy, 1997).<br />
Koide et al reported 18 eyes of 17 patients with diplopia after retrobulbar anesthesia<br />
for cataract surgery (Koide, 2000). Several cases showed superior or <strong>in</strong>ferior deviations,<br />
but most patients had nonuniform disturbances of eye movements. In another study,<br />
orthoptic evaluations were carried out <strong>in</strong> 118 cataract surgery (all retrobulbar anesthesia<br />
<strong>in</strong>jections) patients with<strong>in</strong> 1 month before and 1 day, 1 week, and 1 month after surgery<br />
(Golnik, 2000). Preoperatively, 16 patients had ocular misalignment; 10 were phoric, 4<br />
were <strong>in</strong>termittently tropic, and 2 were tropic. Follow-up evaluation was obta<strong>in</strong>ed for<br />
101 patients (86%) at 1 day, 91 (77%) at 1 week, and 88 (75%) at 1 month. A change <strong>in</strong><br />
ocular alignment occurred <strong>in</strong> 22 of 101 patients (22%) at 1 day, 9 of 91 (10%) at 1 week,<br />
and 6 of 88 (7%) at 1 month. Only 1 patient who had a change <strong>in</strong> alignment at 1 month<br />
was symptomatic. The authors concluded that change <strong>in</strong> ocular alignment after<br />
uneventful cataract surgery occurred <strong>in</strong> 7% of patients, but symptomatic diplopia<br />
was uncommon (1 <strong>in</strong> 118) <strong>in</strong> this small series. Johnson noted persistent vertical diplopia<br />
after cataract surgery <strong>in</strong> 0.23% of patients <strong>in</strong> whom retrobulbar anesthesia was<br />
performed (Johnson, 2001). No cases were found after topical anesthesia.<br />
Vertical strabismus after cataract surgery may also result from <strong>in</strong>ferior oblique muscle<br />
<strong>in</strong>jury from local anesthesia. Hunter et al described four patients without preexist<strong>in</strong>g<br />
strabismus who developed diplopia after cataract surgery (Hunter, 1995). Three had<br />
delayed onset hypertropia with fundus extorsion <strong>in</strong> the eye that underwent surgery,<br />
consistent with <strong>in</strong>ferior oblique muscle overaction secondary to presumed contracture.<br />
The fourth patient had an <strong>in</strong>termediate-onset hypotropia with fundus <strong>in</strong>torsion <strong>in</strong> the<br />
eye that underwent surgery, consistent with <strong>in</strong>ferior oblique paresis. The <strong>in</strong>ferior<br />
oblique muscle contracture observed <strong>in</strong> three patients may have been caused by local<br />
anesthetic myotoxicity, whereas the early paresis observed <strong>in</strong> one patient may have been<br />
due to mechanical trauma or anesthetic toxicity directly to the nerve <strong>in</strong>nervat<strong>in</strong>g the
muscle. Inferior oblique muscle or nerve <strong>in</strong>jury should be considered as another<br />
possible cause of postoperative vertical strabismus, especially when significant<br />
fundus torsion accompanies a vertical deviation. As noted above, a Brown’s syndrome<br />
may also occur after cataract surgery.<br />
What Is the Foveal Displacement Syndrome?<br />
Diplopia 245<br />
B<strong>in</strong>ocular diplopia may occasionally occur with ret<strong>in</strong>al disease. Burgess et al described<br />
11 patients with subret<strong>in</strong>al neovascular membranes <strong>in</strong> one eye who developed b<strong>in</strong>ocular<br />
diplopia before and after effective photocoagulation therapy (the foveal displacement<br />
syndrome) (Burgess, 1980). The diplopia was thought to be due to a rivalry between<br />
central and peripheral fusional mechanisms. The subret<strong>in</strong>al neovascular produced shift<br />
of the photoreceptor array toward the proliferat<strong>in</strong>g neovascular complex; if the lesion<br />
was <strong>in</strong>ferior to the fovea, the foveal receptors were shifted toward the membrane and<br />
with both eyes open the superior ret<strong>in</strong>a relative to the fovea was stimulated <strong>in</strong> the<br />
affected eye. The diplopic image produced by the stimulated superior ret<strong>in</strong>al receptors<br />
is projected <strong>in</strong>feriorly <strong>in</strong> space. If the unaffected eye is covered, the affected eye will<br />
have to elevate the fovea, produc<strong>in</strong>g a downward motion of the cornea mimick<strong>in</strong>g a<br />
true hypertropia. For example, an <strong>in</strong>ferior foveal lesion will mimic a hypertropia <strong>in</strong> the<br />
affected eye. All of the patients demonstrated the follow<strong>in</strong>g:<br />
1. The affected eye deviated (measured tropia) away from the position of the ret<strong>in</strong>al<br />
lesion (e.g., a lesion <strong>in</strong>ferior to the fovea produces a superior scotoma).<br />
2. The affected eye deviated upward (toward the scotoma).<br />
3. The distal diplopic image was downward (toward the ret<strong>in</strong>al image).<br />
The diplopia <strong>in</strong> this condition responds only transiently to prisms. Surgical removal<br />
of the subret<strong>in</strong>al neovascular membrane may correct the diplopia, at least transiently<br />
(Brazis, 1998b). Foveal displacement syndrome may also occur <strong>in</strong> patients with<br />
preret<strong>in</strong>al membranes (Benegas, 1999; Silverberg, 1999). Benegas et al described seven<br />
patients with b<strong>in</strong>ocular diplopia concurrent with macular disease, <strong>in</strong>clud<strong>in</strong>g epiret<strong>in</strong>al<br />
membranes (six patients) and vitreomacular traction (one patient) (Benegas, 1999).<br />
All seven patients had aniseikonia and all had concomitant small-angle strabismus. The<br />
response to treatment with prisms was variable. The authors concluded that aniseikonia,<br />
caused by separation or compression of photoreceptors, is likely a contribut<strong>in</strong>g<br />
factor to the existence of diplopia and the <strong>in</strong>ability to fuse <strong>in</strong> patients with macular<br />
disease. Silverberg et al presented seven patients with b<strong>in</strong>ocular diplopia due to<br />
macular disease, <strong>in</strong>clud<strong>in</strong>g subret<strong>in</strong>al neovascularization, epiret<strong>in</strong>al membrane, and<br />
central serous ret<strong>in</strong>opathy (Silverberg, 1999). All except one had a small-angle, comitant<br />
hyperdeviation with no muscle paresis. Neither prism correction nor manipulation<br />
of the refractive errors corrected the diplopia. However, a partially occlusive foil<br />
(Bangerter) of density rang<strong>in</strong>g from 0.4 to 1.0 placed <strong>in</strong> front of the affected eye<br />
provided an effective treatment, allow<strong>in</strong>g peripheral fusion to be ma<strong>in</strong>ta<strong>in</strong>ed.<br />
The hemifield slip phenomenon, a rare cause of <strong>in</strong>termittent b<strong>in</strong>ocular vertical<br />
diplopia noted <strong>in</strong> some patients with lesions of the optic chiasm, is discussed above.<br />
F<strong>in</strong>ally, vertical or horizontal diplopia may also occur on a nonorganic basis (fictitious<br />
diplopia).
246 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
References<br />
Abdulla N, Eustace P. (1999). A case of ocular neuromyotonia with tonic pupil. J <strong>Neuro</strong>-ophthalmol 19:125–127.<br />
Akman A, Dayanir V, Sefik Sanaç A, Kansu T. (1995). Acquired esotropia as present<strong>in</strong>g sign of cranio-cervical<br />
junction anomalies. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:311–314.<br />
Al-D<strong>in</strong> AN, <strong>An</strong>derson M, Eeg-Olofsson O, et al. (1994). <strong>Neuro</strong>-ophthalmic manifestations of the syndrome of<br />
ophthalmoplegia, ataxia, and areflexia. A review. Acta <strong>Neuro</strong>l Scand 89:157–163.<br />
Alonso-Valdivielso JL, Alvarez Lario B, Alegre Lopez J, et al. (1993). Acquired Brown’s syndrome <strong>in</strong> a patient with<br />
systemic lupus erythematosus. <strong>An</strong>n Rheum Dis 52:63–64.<br />
Arai M, Fuji S. (1990). Divergence paralysis associated with the <strong>in</strong>gestion of diazepam. J <strong>Neuro</strong>l 237:45–46.<br />
Baker RS, Epste<strong>in</strong> AD. (1991). Ocular motor abnormalities from head trauma. Surv Ophthalmol 35:245–267.<br />
Balch K, Phillips PH, Newman NJ. (1997). Pa<strong>in</strong>less orbital apex syndrome from mucormycosis. J <strong>Neuro</strong>-ophthalmol<br />
17:178–182.<br />
Barroso LH, Hoyt WF. (1993). Episodic exotropia form lateral rectus neuromyotonia—appearance and remission<br />
after radiation therapy for a thalamic glioma. J Pediatr Ophthalmol Strabismus 30:56–57.<br />
Bartley GB. (1994). The epidemiologic characteristics and cl<strong>in</strong>ical course of ophthalmopathy associated with<br />
autoimmune thyroid disease <strong>in</strong> Olmsted County, M<strong>in</strong>nesota. Trans Am Ophthalmol Soc 92:477–588.<br />
Bartley GB, Fatourechi V, Kadrmas EF, et al. (1995a). The <strong>in</strong>cidence of Graves’ ophthalmopathy <strong>in</strong> Olmsted<br />
County, M<strong>in</strong>nesota. Am J Ophthalmol 120:511–517.<br />
Bartley GB, Fatourechi V, Kadrmas EF, et al. (1996a). <strong>Cl<strong>in</strong>ical</strong> features of Graves’ ophthalmopathy <strong>in</strong> an <strong>in</strong>cidence<br />
cohort. Am J Ophthalmol 121:284–290.<br />
Bartley GB, Fatourechi V, Kadrmas EF, et al. (1996b). The chronology of Graves’ ophthalmopathy <strong>in</strong> an <strong>in</strong>cidence<br />
cohort. Am J Ophthalmol 121:426–434.<br />
Bartley GB, Gorman CA. (1995b). Diagnostic criteria for Graves’ ophthalmopathy. Am J Ophthalmol 119:792–795.<br />
Behrens-Baumann W, Freissler G. (1990). Computed tomographic appearance of extraocular muscle calcification<br />
<strong>in</strong> a patient with seropositive trich<strong>in</strong>osis. Am J Ophthalmol 110:709–710.<br />
Bell JA, Fielder AR, V<strong>in</strong>ey S. (1990). Congenital double elevator palsy <strong>in</strong> identical tw<strong>in</strong>s. J Cl<strong>in</strong> <strong>Neuro</strong>-Ophthalmol<br />
10:32–34.<br />
Benegas NM, Egbert J, Engel WK, Kushner BJ. (1999). Diplopia secondary to aniseikonia associated with macular<br />
disease. Arch Ophthalmol 117:896–899.<br />
Bernard<strong>in</strong>o CR, Davidson RS, Maus M, Spaeth GL. (2001). <strong>An</strong>gle-closure glaucoma <strong>in</strong> association with orbital<br />
pseudotumor. Ophthalmology 108:1603–1606.<br />
Bernste<strong>in</strong> R, Bernard<strong>in</strong>i GL. (2001). Abnormal vergence with upper bra<strong>in</strong>stem <strong>in</strong>farcts: pseudoabducens palsy.<br />
<strong>Neuro</strong>logy 56:424–425.<br />
Biousse V, Newman NJ, Petermann SH, Lambert SR. (2000). Isolated comitant esotropia and Chiari I malformation.<br />
Am J Ophthalmol 130:216–220.<br />
Borchert MS, Lessell S, Hoyt WF. (1996). Hemifield slide diplopia from altitud<strong>in</strong>al visual field defect. J <strong>Neuro</strong>ophthalmol<br />
16:107–109.<br />
Borruat F-X, Jacques B, Dürig J. (1999). Transient vertical diplopia and silent s<strong>in</strong>us disorder. J <strong>Neuro</strong>-ophthalmol<br />
19:173–175.<br />
Boschi A, Spiritus M, Cioffi M, et al. (1997). Ocular neuromyotonia <strong>in</strong> a case of Paget’s disease of bone. <strong>Neuro</strong><strong>ophthalmology</strong><br />
18:67–71.<br />
Brazis PW, Lee AG. (1998a). B<strong>in</strong>ocular vertical diplopia. Mayo Cl<strong>in</strong> Proc 73:55–66.<br />
Brazis PW, Lee AG. (1999). Acquired b<strong>in</strong>ocular horizontal diplopia. Mayo Cl<strong>in</strong> Proc 74:907–916.<br />
Brazis PW, Lee AG, Boll<strong>in</strong>g JP. (1998b). B<strong>in</strong>ocular vertical diplopia due to subret<strong>in</strong>al neovascular membrane.<br />
Strabismus 6:127–131.<br />
Brown SM, Iacuone JJ. (1999). Intact sensory fusion <strong>in</strong> a child with divergence paresis caused by a pont<strong>in</strong>e glioma.<br />
Am J Ophthalmol 128:528–530.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1991). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. St Louis, Mosby<br />
Yearbook, p. 227.<br />
Burgess D, Roper-Hall G, Burde RM. (1980). B<strong>in</strong>ocular diplopia associated with subret<strong>in</strong>al neovascular<br />
membranes. Arch Ophthalmol 98:311–317.<br />
Capó H, Guyton DL. (1996a). Ipsilateral hypertropia after cataract surgery. Ophthalmology 103:721–730.<br />
Capó H, Roth E, Johnson T, et al. (1996b). Vertical strabismus after cataract surgery. Ophthalmology 103:<br />
918–921.<br />
Carter K, Lee AG, Tang RA, et al. (1998). <strong>Neuro</strong>-ophthalmologic complications of s<strong>in</strong>us surgery. <strong>Neuro</strong><strong>ophthalmology</strong><br />
19:75–82.
Diplopia 247<br />
Çeviker N, Baykaner K, Akata F, et al. (1997). Primary amyloidosis of an extraocular muscle. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
18:147–148.<br />
Chung M, Stout JT, Borchert MS. (2000). <strong>Cl<strong>in</strong>ical</strong> diversity of hereditary Duane’s retraction syndrome. Ophthalmology<br />
107:500–503.<br />
Chung SM, Lee AG, Holds JB, et al. (1997). Ocular neuromyotonia <strong>in</strong> Graves’ dysthyroid orbitopathy. Arch<br />
Ophthalmol 115:365–370.<br />
Coats DK, Paysse EA, Orgenga-Nania S. (1999). Acquired pseudo-Brown’s syndrome immediately follow<strong>in</strong>g<br />
Ahmed valve glaucoma implant. Ophthalmic Surg Lasers 30:396–397.<br />
Corboy JM, Jiang X. (1997). Postanesthetic hypotropia: a unique syndrome <strong>in</strong> the left eye. J Cataract Refract Surg<br />
23:1394–1398.<br />
Cornblath WT, Elner V, Rolfe M. (1993). Extraocular muscle <strong>in</strong>volvement <strong>in</strong> sarcoidosis. Ophthalmology 100:501–<br />
505.<br />
de Heide LJ, Talsma MA. (1999). Giant-cell arteritis present<strong>in</strong>g as an orbital pseudotumor. Neth J Med 55:<br />
196–198.<br />
De Jesús O, Inserni JA, Gonzalez A, Colón L. (1996). Idiopathic orbital <strong>in</strong>flammation with <strong>in</strong>tracranial extension.<br />
Case report. J <strong>Neuro</strong>surg 85:510–513.<br />
DeResp<strong>in</strong>is PA, Caputo AR, Wagner RS, Guo S. (1993). Duane’s retraction syndrome. Surv Ophthalmol 38:257–288.<br />
Donahue SP, Lev<strong>in</strong> PJM, Hamed LM. (1999). Tonic ocular tilt reaction simulat<strong>in</strong>g a superior oblique palsy.<br />
Diagnostic confusion with the 3-step test. Arch Ophthalmol 117:347–352.<br />
Dooley DP, Hollsten DA, Grimes SR, Moss J. (1992). Indolent orbital apex syndrome caused by occult<br />
mucormycosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:245–249.<br />
Downie JA, Francis IC, Arnold JJ, et al. (1993). Sudden bl<strong>in</strong>dness and total ophthalmoplegia <strong>in</strong> mucormycosis. A<br />
cl<strong>in</strong>icopathologic correlation. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:27–34.<br />
Durno CA, Ehrlich R, Taylor R, et al. (1997). Keep<strong>in</strong>g an eye on Crohn’s disease: orbital myositis as the present<strong>in</strong>g<br />
symptom. Can J Gastroenterol 11:497–500.<br />
Egbert JE, May K, Kersten RC, Kulw<strong>in</strong> DR. (2000). Pediatric orbital floor fracture. Direct extraocular muscle<br />
<strong>in</strong>volvement. Ophthalmology 107:1875–1879.<br />
Eitzen JP, Elsas FJ. (1991). Strabismus follow<strong>in</strong>g endoscopic <strong>in</strong>tranasal s<strong>in</strong>us surgery. J Pediatr Ophthalmol<br />
Strabismus 28:168–170.<br />
Engle EC, Goumnerov BC, McKeown CA, et al. (1997). Oculomotor nerve and muscle abnormalities <strong>in</strong> congenital<br />
fibrosis of the extraocular muscles. <strong>An</strong>n <strong>Neuro</strong>l 41:314–325.<br />
Erie JC. (1990). Acquired Brown’s syndrome after peribulbar anesthesia. Am J Ophthalmol 109:349–350.<br />
Esmaeli B, Ahmadi A, Tang R, et al. (2001). Interferon therapy for orbital <strong>in</strong>filtration secondary to Erdheim-<br />
Chester disease. Am J Ophthalmol 132:945–947.<br />
Ezra E, Plant GT. (1996a). Paroxysmal superior rectus and levator palpebrae spasm: a unique presentation of<br />
multiple sclerosis. Br J Ophthalmol 80:187–188.<br />
Ezra E, Spalton D, Sanders MD, Graham EM, Plant GT. (1996b). Ocular neuromyotonia. Br J Ophthalmol 80:350–<br />
355.<br />
Friedman AP, Harter DH, Merritt HH. (1962). Ophthalmoplegic migra<strong>in</strong>e. Arch <strong>Neuro</strong>l 7:320–327.<br />
Fril<strong>in</strong>g R, Yassur Y, Merk<strong>in</strong> L, et al. (1993). Divergence paralysis versus bilateral sixth nerve palsy <strong>in</strong> an <strong>in</strong>complete<br />
Miller-Fisher syndrome. <strong>Neuro</strong>-Ophthalmology 13:215–217.<br />
Frohman EM, Zee DS. (1995). Ocular neuromyotonia: cl<strong>in</strong>ical features, physiological mechanisms, and response to<br />
therapy. <strong>An</strong>n <strong>Neuro</strong>l 37:620–626.<br />
Fu ER. (1995). Ocular neuromyotonia—an unusual ocular motility complication after radiation therapy for<br />
nasopharyngeal carc<strong>in</strong>oma. <strong>An</strong>n Acad Med S<strong>in</strong>gapore 24:895–897.<br />
Fujikado T, Ohmi G, Ikeda T, et al. (1997). Exotropia secondary to vitreous hemorrhage. Graefes Arch Cl<strong>in</strong> Exp<br />
Ophthalmol 235:143–148.<br />
Fukuo Y, Abe T, Hayasaka S. (1998). Acute comitant esotropia <strong>in</strong> a boy with head trauma and convulsions<br />
receiv<strong>in</strong>g carbamazep<strong>in</strong>e. Ophthalmologica 212:61–62.<br />
Goldberg RA, Rootman J. (1990a). <strong>Cl<strong>in</strong>ical</strong> characteristics of metastatic orbital tumors. Ophthalmology 97:620–624.<br />
Goldberg RA, Rootman J, Cl<strong>in</strong>e RA. (1990b). Tumors metastatic to the orbit: a chang<strong>in</strong>g picture. Surv Ophthalmol.<br />
35:1–24.<br />
Goldste<strong>in</strong> JH, Schneekloth BB. (1996). Spasm of the near reflex: a spectrum of anomalies. Surv Ophthalmol 40:269–<br />
278.<br />
Golnik KC, West CE, Kaye E, et al. (2000). Incidence of ocular misalignment and diplopia after uneventful cataract<br />
surgery. J Cataract Refract Surg 26:1205–1209.<br />
Hamed LM, Silbiger J. (1992). Periodic alternat<strong>in</strong>g esotropia. J Pediatr Ophthalmol Strabismus 29:240–242.
248 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Harr<strong>in</strong>gton DO, Flocks M. (1953). Ophthalmoplegic migra<strong>in</strong>e. Pathogenesis: report of pathologic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a case<br />
of recurrent oculomotor paralysis. Arch Ophthalmol 49:643–655.<br />
Harris GJ, Murphy ML, Schmidt EW, et al. (1994). Orbital myositis as a paraneoplastic syndrome. Arch Ophthalmol<br />
112:380–386.<br />
Harrison AR, Wirtschafter JD. (1997). Ocular neuromyotonia <strong>in</strong> a patient with cavernous s<strong>in</strong>us thrombosis<br />
secondary to mucormycosis. Am J Ophthalmol 124:122–123.<br />
Haupert CL, Newman NJ. (1997). Ocular neuromyotonia 18 years after radiation therapy. Arch Ophthalmol<br />
115:1331–1332.<br />
Helmchen C, Dieterich M, Straube A, et al. (1992). Abducens neuromyotonia with partial oculomotor paralysis.<br />
Nervenartz 63:625–629.<br />
Hertle RW, Bienfang DC. (1990). Oculographic analysis of acute esotropia secondary to a thalamic hemorrhage. J<br />
Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:21–26.<br />
Hommel M, Bogousslavsky DC. (1990). The spectrum of vertical gaze palsy follow<strong>in</strong>g unilateral bra<strong>in</strong>stem stroke.<br />
<strong>Neuro</strong>logy 41:1229–1234.<br />
Horton JC, Fishman RA. (1994). <strong>Neuro</strong>visual f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> the syndrome of spontaneous <strong>in</strong>tracranial hypotension<br />
from dural cerebrosp<strong>in</strong>al fluid leak. Ophthalmology 101:244–251.<br />
Hoyt CS, Good WV. (1995). Acute onset concomitant esotropia: when is it a sign of serious neurological disease?<br />
Br J Ophthalmol 79:498–501.<br />
Hudson HL, Feldon SE. (1992). Late overcorrection of hypotropia <strong>in</strong> Graves’ ophthalmopathy. Ophthalmology<br />
99:356–360.<br />
Hughes DS, Beck L, Hill R, Plenty J. (1992). Dysthyroid eye disease present<strong>in</strong>g as Brown’s syndrome. Acta<br />
Ophthalmol 70:262–265.<br />
Hunter DG, Lam GC, Guyton DL. (1995). Inferior oblique muscle <strong>in</strong>jury from local anesthesia for cataract surgery.<br />
Ophthalmology 102:501–509.<br />
Hupp SL, Williams JP, Curran JE. (1990). Computerized tomography <strong>in</strong> the diagnosis of congenital fibrosis<br />
syndrome. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:135–139.<br />
Hutnik CML, Nicolle DA, Munoz DG. (1997). Orbital aspergillosis. A fatal masquerader. J <strong>Neuro</strong>-ophthalmol<br />
17:257–261.<br />
Jacobson DM. (2000). Divergence <strong>in</strong>sufficiency revisited. Natural history of idiopathic cases and neurologic<br />
associations. Arch Ophthalmol 118:1237–1241.<br />
Johnson DA. (2001). Persistent vertical b<strong>in</strong>ocular diplopia after cataract surgery. Am J Ophthalmol 132:831–835.<br />
Johnson TE, Casiano RR, Kronish JW, et al. (1999). S<strong>in</strong>o-orbital aspergillosis <strong>in</strong> acquired immunodeficiency<br />
syndrome. Arch Ophthalmol 117:57–64.<br />
Jones MR, Waggoner R, Hoyt WF. (1999). Cerebral polyopia with extrastriate quadrantanopia: report of a case<br />
with magnetic resonance documentation of V2=V3 cortical <strong>in</strong>farction. J <strong>Neuro</strong>-ophthalmol 19:1–6.<br />
Kattah JC, Zimmerman LE, Kolsky MP, et al. (1990). Bilateral orbital <strong>in</strong>volvement <strong>in</strong> fatal giant cell polymyositis.<br />
Ophthalmology 97:520–525.<br />
Katz B, Rimmer S. (1989). Ophthalmoplegic migra<strong>in</strong>e with superior ramus oculomotor paresis. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
9:181–183.<br />
Kim M, Provias, J, Bernste<strong>in</strong> M. (1995). Rosai-Dorfman disease mimick<strong>in</strong>g multiple men<strong>in</strong>giomas: case report.<br />
<strong>Neuro</strong>surgery 36:1185–1187.<br />
Koide R, Hnda Y, Ozawa T. (2000). Diplopia after cataract surgery. J Cataract Refract Surg 26:1198–1204.<br />
Kubis KC, Danesh-Meyer H, Bilyk JR, et al. (2000). Unilateral lid retraction dur<strong>in</strong>g pregnancy. Surv Ophthalmol<br />
45:69–76.<br />
Kushner BJ. (2001). Recently acquired diplopia <strong>in</strong> adults with long-stand<strong>in</strong>g strabismus. Arch Ophthalmol<br />
119:1795–1801.<br />
Lacey B, Chang W, Rootman J. (1999). Nonthyroid causes of extraocular muscle disease. Surv Ophthalmol 44:187–<br />
213.<br />
Lee AG, Brazis PW. (1997). Therapeutic neuro-<strong>ophthalmology</strong>. In: Appel SH, ed. Current <strong>Neuro</strong>logy. Vol 17.<br />
Amsterdam, IOS Press, pp. 265–292.<br />
Leib ML, Odel JG, Cooney MJ. (1994). Orbital polymyositis and giant cell myocarditis. Ophthalmology 101:950–954.<br />
Leigh RJ, Zee DS. (1999). The <strong>Neuro</strong>logy of Eye Movements. 3rd ed. New York, Oxford University Press.<br />
Lepore FE. (1999). Divergence paresis: a nonlocaliz<strong>in</strong>g cause of diplopia. J <strong>Neuro</strong>-ophthalmol 19:242–245.<br />
Lev<strong>in</strong> LA, Avery R, Shore JW, et al. (1996). The spectrum of orbital aspergillosis: a cl<strong>in</strong>icopathological review. Surv<br />
Ophthalmol 41:142–154.<br />
Lewis AR, Kl<strong>in</strong>e LB, Sharpe JA. (1996). Acquired esotropia due to Arnold-Chiari I malformation. J <strong>Neuro</strong>ophthalmol<br />
16:49–54.
Diplopia 249<br />
L<strong>in</strong>dner K, Hitzenberger P, Drlicek M, Grisold W. (1992). Dissociated unilateral convergence paralysis <strong>in</strong> a patient<br />
with thalamotectal hemorrhage. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 55:731–733.<br />
Lopez JR, Adornato BT, Hoyt WF. (1993). ‘‘Entomopia’’: a remarkable case of cerebral polyopia. <strong>Neuro</strong>logy<br />
43:2145–2146.<br />
Lopresto Weeks C, Hamed LM. (1999). Treatment of acute comitant esotropia <strong>in</strong> Chiari I malformation.<br />
Ophthalmology 106:2368–2371.<br />
Lueder GT. (2002). <strong>An</strong>omalous orbital structures result<strong>in</strong>g <strong>in</strong> unusual strabismus. Surv Opthalmol 47:27–35.<br />
Mannor GE, Rose GE, Moseley IF, Wright JE. (1997). Outcome of orbital myositis. <strong>Cl<strong>in</strong>ical</strong> features associated with<br />
recurrence. Ophthalmology 104:409–414.<br />
Mask<strong>in</strong> SL, Fetchick RJ, Leone CR, et al. (1989). Bipolaris hawaiiensis-caused phaeohypomycotic orbitopathy. A<br />
devastat<strong>in</strong>g fungal s<strong>in</strong>usitis <strong>in</strong> an apparently immunocompetent host. Ophthalmology 96:175–179.<br />
Massry GG, Hornblass A, Harrison W. (1996). Itraconazole <strong>in</strong> the treatment of orbital aspergillosis. Ophthalmology<br />
103:1467–1470.<br />
Maurer I, Zierz S. (1999). Recurrent orbital myositis. Report of a familial <strong>in</strong>cidence. Arch <strong>Neuro</strong>l 56:1407–1409.<br />
Merle H, Gerard M, Raynaud M. (1998). Isolated medial orbital blow-out fracture with medial rectus entrapment.<br />
Acta Ophthalmol Scand 76:378–379.<br />
Miller NR. (1991). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th Edition. Baltimore, Williams & Wilk<strong>in</strong>s, pp.<br />
2533–2538.<br />
Mohney BG. (2001). Common forms of childhood esotropia. Ophthalmology 108:805–809.<br />
Mokri B, Peipgras DG, Miller GM. (1997). Syndrome of orthostatic headaches and diffuse pachymen<strong>in</strong>geal<br />
gadol<strong>in</strong>ium enhancement. Mayo Cl<strong>in</strong> Proc 72:400–413.<br />
Mombaerts I, Goldschmed<strong>in</strong>g R, Schl<strong>in</strong>gemann RO, Koornneef L. (1996). What is orbital pseudotumor? Surv<br />
Ophthalmol 41:66–78.<br />
Mombaerts I, Koornneef L. (1997). Current status of treatment of orbital myositis. Ophthalmology 104:402–408.<br />
Morrow MJ, Kao GW, Arnold AC. (1996). Bilateral ocular neuromyotonia: oculographic correlations. <strong>Neuro</strong>logy<br />
46:264–266.<br />
Moster ML, Bosley TM, Slav<strong>in</strong> ML, Rub<strong>in</strong> SE. (1992). Thyroid ophthalmopathy present<strong>in</strong>g as superior oblique<br />
paresis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:94–97.<br />
Murdoch IE, Sullivan TJ, Moseley I, et al. (1996). Primary localized amyloidosis of the orbit. Br J Ophthalmol<br />
80:1083–1086.<br />
Newman NJ, Lessell S. (1989). Pupillary dilation with monocular occlusion as a sign of nonorganic oculomotor<br />
dysfunction. Am J Ophthalmol 108:461–462.<br />
Newman SA. (1993). Gaze-<strong>in</strong>duced strabismus (cl<strong>in</strong>ical conference). Surv Ophthalmol 38:303–309.<br />
Odaka M, Yuki N, Hirata K. (2001). <strong>An</strong>ti-GQib IgG antibody syndrome: cl<strong>in</strong>ical and immunological range. J<br />
<strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 70:50–55.<br />
Ohtsuka K, Maekawa H, Takeda M, et al. (1988). Accommodation and convergence <strong>in</strong>sufficiency with left middle<br />
cerebral artery occlusion. Am J Ophthalmol 106:60–64.<br />
Ohyagi Y, Yamada T, Okayama A, et al. (2000). Vergence disorders <strong>in</strong> patients with sp<strong>in</strong>ocerebellar ataxia<br />
3=Machado-Joseph disease: a synoptophore study. J <strong>Neuro</strong>l Sci 173:120–123.<br />
Orssaud C, Poisson M, Gardeur D. (1992). Orbital myositis, recurrence of Whipple’s disease (French). J Fr<br />
Ophtalmol 15:205–208.<br />
Patel AS, Kelman SE, Duncan GW, Rismondo V. (1994). Pa<strong>in</strong>less diplopia caused by extraocular muscle sarcoid.<br />
Arch Ophthalmol 112:879–880.<br />
Postert T, Büttner T, McMonagle U, Przuntek H. (1997). Spasm of the near reflex: case report and review of the<br />
literature. <strong>Neuro</strong>-<strong>ophthalmology</strong> 17:149–152.<br />
Pullic<strong>in</strong>o P, L<strong>in</strong>coff N, Truax BT. (2000). Abnormal vergence with upper bra<strong>in</strong>stem <strong>in</strong>farcts: pseudoabducens palsy.<br />
<strong>Neuro</strong>logy 55:352–358.<br />
Racette BA, Gokden MS, Tychsen LS, Perlmuttter JS. (1999). Convergence <strong>in</strong>sufficiency <strong>in</strong> idiopathic Park<strong>in</strong>son’s<br />
disease responsive to levodopa. Strabismus 7:169–174.<br />
Ramboer K, Demaerel P, Baert AL, et al. (1997). L<strong>in</strong>ear scleroderma with orbital <strong>in</strong>volvement: follow up and<br />
magnetic resonance imag<strong>in</strong>g. Br J Ophthalmol 81:90–91.<br />
Riordan-Eva P, Vickers SF, McCrarry B, et al. (1993). Cyclic strabismus without b<strong>in</strong>ocular function. J Pediatr<br />
Ophthalmol Strabismus 30:106–108.<br />
Safran AB, Magistris M. (1998). Term<strong>in</strong>at<strong>in</strong>g attacks of ocular neuromyotonia. J <strong>Neuro</strong>-ophthalmol 18:47–48.<br />
Saunders RA, Stratas BA, Gordon RA, Holgate RC. (1990). Acute-onset Brown’s syndrome associated with<br />
pans<strong>in</strong>usitis. Arch Ophthalmol 108:58–60.<br />
Schanzer B, Bordaberry M, Jeffrey AR, et al. (1998). The child with divergence paresis. Surv Ophthalmol 42:571–576.
250 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Segal EI, Tang R, Lee AG, et al. (2000). Orbital apex lesion as the present<strong>in</strong>g manifestation of sarcoidosis. J <strong>Neuro</strong>-<br />
Ophthalmol 20:156–158.<br />
Sener EC, Özkan SB, Aribal ME, et al. (1996). Evaluation of congenital Brown’s syndrome with magnetic<br />
resonance imag<strong>in</strong>g. Eye 10:492–496.<br />
Serop S, Vianna RN, Claeys M, De Laey JJ. (1994). Orbital myositis secondary to systemic lupus erythematosus.<br />
Acta Ophthalmol (Copenh) 72:520–523.<br />
Shields JA, Karcioglu ZA, Shields CL, et al. (1991). Orbital and eyelid <strong>in</strong>volvement with Erdheim-Chester disease.<br />
A report of two cases. Arch Ophthalmol 109:850–854.<br />
Sidikaro Y, von Noorden GK. (1982). Observations <strong>in</strong> sensory heterotropia. J Pediatr Ophthalmol Strabismus 19:12–<br />
19.<br />
Silverberg M, Schular E, Veronneau-Troutman S, et al. (1999). Nonsurgical management of b<strong>in</strong>ocular diplopia<br />
<strong>in</strong>duced by macular pathology. Arch Ophthalmol 117:900–903.<br />
Simon JW, Waldman JB, Couture KC. (1996). Cerebellar astrocytoma manifest<strong>in</strong>g as isolated, comitant esotropia <strong>in</strong><br />
childhood. Am J Ophthalmol 121:584–586.<br />
Slav<strong>in</strong> ML. (1989). Hyperdeviation associated with isolated unilateral abducens palsy. Ophthalmology 96:512–<br />
516.<br />
Slav<strong>in</strong> ML. (1991). Primary aspergillosis of the orbital apex. Arch <strong>Neuro</strong>l 109:1502.<br />
Slav<strong>in</strong> ML, Potash SD, Rub<strong>in</strong> SE. (1988). Asymptomatic physiologic hyperdeviation <strong>in</strong> peripheral gaze.<br />
Ophthalmology 95:778–781.<br />
Spector RH. (1993). Vertical diplopia. Surv Ophthalmol 38:31–62.<br />
Spierer A, Huna R, Rechtman C, Lapidot D. (1995). Convergence <strong>in</strong>sufficiency secondary to subdural hematoma.<br />
Am. J. Ophthalmol. 120:258–260.<br />
Squires RH Jr, Zwiener RJ, Kennedy RH. (1991). Orbital myositis and Crohn’s disease. J Pediatr Gastroenterol Nutr<br />
15:448–451.<br />
Stevens AW, Grossman ME, Barr ML. (1996). Orbital myositis, vitiligo, and giant cell myocarditis. J Am Acad<br />
Dermatol 35:310–312.<br />
Stidham DB, Sondhi N, Plager D, Helveston E. (1998). Presumed isolated <strong>in</strong>flammation of the superior oblique<br />
muscle <strong>in</strong> idiopathic orbital myositis. Ophthalmology 105:2216–2219.<br />
Sullivan TJ, Aylward GW, Wright JE. (1992). Act<strong>in</strong>omycosis of the orbit. Br J Ophthalmol 76:505–506.<br />
Suttorp-Schulten MS, Koornneef L. (1990). L<strong>in</strong>ear scleroderma associated with ptosis and motility disorders. Br J<br />
Ophthalmol 74:694–695.<br />
Takahashi T, Fujita N, Takeda K, et al. (2000). A case of sarcoid myopathy with external ocular <strong>in</strong>volvement—<br />
diagnosis and follow-up study with 99mTc pyrophosphate sc<strong>in</strong>tigraphy. R<strong>in</strong>sho Sh<strong>in</strong>keigaku 40:145–148.<br />
Takahashi T, Uchida S, Arita M, et al. (2001). Orbital <strong>in</strong>flammatory pseudotumor and ischemic vasculitis <strong>in</strong> Churg-<br />
Strauss syndrome. Report of two cases and review of the literature. Ophthalmology 108:1129–1133.<br />
Tapiero B, Pedespan JM, Rougier MB, et al. (1995). Cyclic strabismus. Presentation of two new cases and critical<br />
review of the literature. J Fr Ophthalmol 18:411–420.<br />
Tekeli O, Tomac S, Gursel E, Hasiripi H. (1999). Divergence paralysis and <strong>in</strong>tracranial hypertension due to<br />
neurobrucellosis. A case report. B<strong>in</strong>ocular Vis Strabismus Q 14:117–118.<br />
Thomke F, Hopf HC. (1992). Acquired monocular elevation paresis. <strong>An</strong> asymmetric up-gaze palsy. Bra<strong>in</strong> 115:1901–<br />
1910.<br />
Thompson SH, Miller NR. (1998). Disorders of pupillary function, accommodation, and lacrimation. In: Miller<br />
NR, Newman NJ, eds. Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-Ophthalmology. 5th ed. Baltimore, Williams & Wilk<strong>in</strong>s,<br />
pp. 1016–1018.<br />
Thorne JE, Volpe NJ, Liu GT. (1999). Magnetic resonance imag<strong>in</strong>g of acquired Brown syndrome <strong>in</strong> a patient with<br />
psoriasis. Am J Ophthalmol 127:233–235.<br />
Tilikete C, Vial C, Niederlaender M, et al. (2000). Idiopathic ocular neuromyotonia: a neurovascular compression<br />
syndrome? J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 69:642–644.<br />
Toller KK, Gigantelli JW, Spald<strong>in</strong>g J. (1998). Bilateral orbital metastases from breast carc<strong>in</strong>oma. A case of false<br />
pseudotumor. Ophthalmology 105:1897–1901.<br />
Valmaggia C, Neuweiler J, Fretz C, Gottlob I. (1997). A case of Erdheim-Chester disease with orbital <strong>in</strong>volvement.<br />
Arch Ophthalmol 115:1467–1468.<br />
Vargas ME, Warren FA, Kupersmith MJ. (1993). Exotropia as a sign of myasthenia gravis <strong>in</strong> dysthyroid<br />
ophthalmopathy. Br J Ophthalmol 77:822–823.<br />
Vers<strong>in</strong>o M, Hurko O, Zee DS. (1996). Disorders of b<strong>in</strong>ocular control of eye movements <strong>in</strong> patients with cerebellar<br />
dysfunction. Bra<strong>in</strong> 119:1933–1950.<br />
von Noorden GK. (1996). B<strong>in</strong>ocular Vision and Ocular Motility. 5th ed. St Louis, Mosby.
Diplopia 251<br />
von Noorden GK, Hansell R. (1991). <strong>Cl<strong>in</strong>ical</strong> characteristics and treatment of isolated <strong>in</strong>ferior rectus paralysis.<br />
Ophthalmol 98:253–257.<br />
von Noorden GK, Murray E, Wong SY. (1986). Superior oblique paralysis. A review of 270 cases. Arch Ophthalmol<br />
104:1771–1776.<br />
Wan MK, Francis IC, Carter PR, et al. (2000). The spectrum of presentation of silent s<strong>in</strong>us syndrome. J <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:207–212.<br />
White VA, Cl<strong>in</strong>e RA. (1999). Pathologic causes of the superior oblique click syndrome. Ophthalmology 106:1292–<br />
1295.<br />
Wiest G, Mallek R, Baumgartner C. (2000). Selective loss of vergence control secondary to bilateral paramedian<br />
thalamic <strong>in</strong>farction. <strong>Neuro</strong>logy 54:1997–1999.<br />
Wigg<strong>in</strong>s RE Jr, Baumgartner S. (1999). Diagnosis and management of divergence weakness <strong>in</strong> adults. Ophthalmology<br />
106:1353–1356.<br />
Woo TL, Francis IC, Wilcsek GA, et al. (2001). Australasian orbital and adnexal Wegener’s granulomatosis.<br />
Ophthalmology 108:1535–1543.<br />
Woods RL, Bradley A, Atchison DA. (1996). Monocular diplopia caused by ocular aberrations and hyperopic<br />
defocus. Vis Res 36:3597–3606.<br />
Yee RD, Purv<strong>in</strong> VA. (1998). Ocular neuromyotonia: three case reports with eye movement record<strong>in</strong>gs. J <strong>Neuro</strong>ophthalmol<br />
18:1–8.<br />
Yuki N, Odaka M, Hirata K. (2001). Acute ophthalmoparesis (without ataxia) associated with anti-GQ1b IgG<br />
antibody. <strong>Cl<strong>in</strong>ical</strong> features. Ophthalmology 108:196–200.<br />
Ziffer AJ, Rosenbaum AL, Demer JL, Yee RD. (1992). Congenital double elevator palsy: vertical saccadic velocity<br />
utiliz<strong>in</strong>g the scleral search coil technique. J Pediatr Ophthalmol Strabismus 29:142–149.
This page <strong>in</strong>tentionally left blank
11 r<br />
Third Nerve Palsies<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of a Third<br />
Cranial Nerve Palsy?<br />
The oculomotor nerve (third cranial nerve) supplies four extraocular muscles (medial,<br />
superior and <strong>in</strong>ferior recti, and <strong>in</strong>ferior oblique) as well as the levator of the lid, and<br />
conta<strong>in</strong>s parasympathetic fibers that supply the sph<strong>in</strong>cter of the pupil and the ciliary<br />
body. A complete peripheral third nerve palsy (TNP) thus causes ptosis, a fixed and<br />
dilated pupil, and a down (hypotropic) and out (exotropic) rest<strong>in</strong>g eye position.<br />
Partial TNPs may cause (<strong>in</strong> comb<strong>in</strong>ation or isolation) variable ptosis; variable paresis<br />
of eye adduction, elevation, and depression; and variable pupillary <strong>in</strong>volvement.<br />
In this section, we discuss the localization of TNPs associated with other neurologic<br />
signs (nonisolated TNPs) and TNPs without other associated neurologic or neuroophthalmologic<br />
deficits (isolated TNPs) (Lee, 1999).<br />
Is the TNP Isolated or Nonisolated? Can the<br />
TNP Be Localized?<br />
We classify TNPs as either nonisolated or isolated. The isolated TNPs were def<strong>in</strong>ed as<br />
TNPs without associated neurologic f<strong>in</strong>d<strong>in</strong>gs (e.g., headache, other cranial neuropathies).<br />
Patients with evidence for myasthenia gravis (e.g., variability, fatigue,<br />
Cogan’s lid twitch sign, enhancement of ptosis) are not <strong>in</strong>cluded <strong>in</strong> the isolated TNP<br />
group. We def<strong>in</strong>e six types of TNP <strong>in</strong> Table 11–1. The localization of TNP is outl<strong>in</strong>ed <strong>in</strong><br />
Table 11–2. Etiologies of TNPs by localization are outl<strong>in</strong>ed <strong>in</strong> Table 11–3.<br />
253
254 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 11–1. Def<strong>in</strong>itions of the Six Types of Third Nerve Palsy (TNP)<br />
Type 1: nonisolated<br />
TNP is considered nonisolated if it has the follow<strong>in</strong>g features:<br />
Orbital disease (e.g., chemosis, proptosis, lid swell<strong>in</strong>g, <strong>in</strong>jection, and positive forced ductions)<br />
Evidence to suggest myasthenia gravis (e.g., fatigability of the motility defect, Cogan’s lid twitch<br />
sign, orbicularis oculi weakness)<br />
Multiple cranial nerve palsies (<strong>in</strong>clud<strong>in</strong>g bilateral TNP) or radiculopathy<br />
Bra<strong>in</strong>stem signs (e.g., hemiplegia, cerebellar signs, other cranial nerve deficits)<br />
Systemic, <strong>in</strong>fectious, or <strong>in</strong>flammatory risk factors for TNP (e.g., history of previous malignancy,<br />
giant cell arteritis, collagen vascular disease)<br />
Severe headache<br />
Type 2: traumatic<br />
Isolated unilateral TNP, which has a clearly established temporal relationship to significant<br />
previous head trauma and does not progress, is considered traumatic <strong>in</strong> orig<strong>in</strong>; patients with<br />
m<strong>in</strong>or head trauma are not <strong>in</strong>cluded<br />
Type 3: congenital<br />
Patient born with an isolated TNP<br />
Type 4: acquired, nontraumatic isolated<br />
Type 4A: TNP with a normal pupillary sph<strong>in</strong>cter with completely palsied extraocular muscles<br />
Type 4B: TNP with normal pupillary sph<strong>in</strong>cter and <strong>in</strong>complete palsied extraocular muscles<br />
Type 4C: TNP with subnormal pupillary sph<strong>in</strong>cter dysfunction and partial or complete extraocular<br />
muscle palsies<br />
Type 5: progressive or unresolved<br />
Patients with TNP that worsens after the acute stage (more than 2 weeks) or who develop new<br />
neurologic f<strong>in</strong>d<strong>in</strong>gs are considered to have progressive TNP; patients without resolution of TNP<br />
after 12 to 16 weeks are considered unresolved<br />
Type 6: signs of aberrant regeneration<br />
Is the TNP Due to a Nuclear Lesion?<br />
Lesions of the third nerve nucleus are rare and often associated with other signs of<br />
mesencephalic <strong>in</strong>volvement, especially vertical gaze impairment (Bengel, 1994;<br />
Bogousslavsky, 1994; Chee, 1990; Gaymard, 1990; Nakao, 1998; Saeki, 2000b). Nuclear<br />
lesions may be due to <strong>in</strong>farction, hemorrhage, tumor, <strong>in</strong>fection, or trauma and, thus,<br />
should be <strong>in</strong>vestigated by magnetic resonance imag<strong>in</strong>g (MRI). Paresis of an isolated<br />
muscle <strong>in</strong>nervated by the oculomotor nerve almost always results from a lesion <strong>in</strong> the<br />
orbit or from disease of the muscle or neuromuscular junction. For example, isolated<br />
<strong>in</strong>ferior rectus paresis may develop with trauma, myasthenia gravis, or vascular disease<br />
and may also occur on a congenital or idiopathic basis (von Noorden, 1991). Lesions of<br />
the <strong>in</strong>ferior rectus subnucleus, however, may also give rise to isolated weakness of the<br />
<strong>in</strong>ferior rectus muscle (Chou, 1998; Lee, 2000b; Tezer, 2000). Isolated <strong>in</strong>ferior rectus<br />
paresis may also occur on a supranuclear basis with a lesion selectively <strong>in</strong>terrupt<strong>in</strong>g<br />
fibers descend<strong>in</strong>g from the right medial longitud<strong>in</strong>al fasciculus (MLF) to the <strong>in</strong>ferior<br />
rectus subnucleus (Tezer, 2000). The levator palpebrae superioris muscles, the superior<br />
recti, and the constrictors of the pupils are affected bilaterally with nuclear lesions.<br />
Because medial rectus neurons probably lie at three different locations with<strong>in</strong> the<br />
oculomotor nucleus, it is unlikely that a medial rectus paralysis (unilateral or bilateral)<br />
would be the sole manifestation of a nuclear lesion (Umapathi, 2000). Most characteri-
Table 11–2. The Localization of TNP (Structure Involved: <strong>Cl<strong>in</strong>ical</strong> Manifestation)<br />
Lesions affect<strong>in</strong>g the third nerve nucleus<br />
Oculomotor nucleus: ipsilateral complete cranial nerve (CN) III palsy; contralateral ptosis<br />
and superior rectus paresis<br />
Oculomotor subnucleus: isolated muscle palsy (e.g., <strong>in</strong>ferior rectus)<br />
Isolated levator subnucleus: isolated bilateral ptosis<br />
Lesions affect<strong>in</strong>g the third nerve fasciculus<br />
Isolated fascicle: partial or complete isolated CN III palsy with or without pupil <strong>in</strong>volvement<br />
Paramedian mesencephalon: plus-m<strong>in</strong>us syndrome (ipsilateral ptosis and contralateral<br />
eyelid retraction)<br />
Fascicle, red nucleus=cerebellar peduncle: ipsilateral CN III palsy with contralateral ataxia<br />
and tremor (Claude)<br />
Fascicle and cerebral peduncle: ipsilateral CN III palsy with contralateral hemiparesis (Weber)<br />
Fascicle and red nucleus=substantia nigra: ipsilateral CN III palsy with contralateral<br />
choreiform movements (Benedikt)<br />
Lesions affect<strong>in</strong>g the third nerve <strong>in</strong> the subarachnoid space<br />
Oculomotor nerve: complete CN III palsy with or without other cranial nerve<br />
<strong>in</strong>volvement; superior or <strong>in</strong>ferior division palsy<br />
Lesions affect<strong>in</strong>g the third nerve <strong>in</strong> the cavernous s<strong>in</strong>us<br />
Cavernous s<strong>in</strong>us lesion: pa<strong>in</strong>ful or pa<strong>in</strong>less CN III palsy; with or without palsies of CN IV, VI,<br />
and V1; CN III palsy with small pupil (Horner syndrome); primary aberrant CN III<br />
regeneration<br />
Lesions affect<strong>in</strong>g the third nerve <strong>in</strong> the superior orbital fissure<br />
Superior orbital fissure lesion: CN III palsy with or without palsies of CN IV, VI, and V1;<br />
often with proptosis<br />
Lesion affect<strong>in</strong>g the third nerve <strong>in</strong> the orbit<br />
Oculomotor nerve: CN III palsy; superior or <strong>in</strong>ferior CN III branch palsy<br />
CN III and optic nerve or other orbital structures: visual loss; proptosis; swell<strong>in</strong>g of lids;<br />
chemosis<br />
Source: Modified from Brazis, 2001, with permission from Lipp<strong>in</strong>cott Williams & Wilk<strong>in</strong>s.<br />
Third Nerve Palsies 255<br />
stic of oculomotor nuclear <strong>in</strong>volvement is unilateral TNP, weakness of the ipsilateral<br />
and contralateral superior rectus, and bilateral <strong>in</strong>complete ptosis (Pratt, 1995). On rare<br />
occasions the ipsilateral superior rectus is spared while the contralateral superior rectus<br />
is paretic. Bilateral TNPs with spar<strong>in</strong>g of the lid levators may also be caused by nuclear<br />
lesions (the central caudal levator subnucleus is spared) (Bryan, 1992). Isolated bilateral<br />
ptosis with spar<strong>in</strong>g of the extraocular muscles and pupils may occur with lesions<br />
<strong>in</strong>volv<strong>in</strong>g the levator subnucleus and spar<strong>in</strong>g more rostral oculomotor subnuclei<br />
(Mart<strong>in</strong>, 1996). After surgery for a fourth ventricle ependymoma, bilateral nuclear<br />
oculomotor palsies affect<strong>in</strong>g only the levator and superior recti subnuclei have been<br />
described, result<strong>in</strong>g <strong>in</strong> third nerve paresis affect<strong>in</strong>g only the levators and superior recti<br />
bilaterally (Sanli, 1995). Bilateral total ophthalmoplegia, bilateral complete ptosis, and<br />
large, unreactive pupils have been described with midbra<strong>in</strong> hematoma (Worth<strong>in</strong>gton,<br />
1996). This constellation of f<strong>in</strong>d<strong>in</strong>gs was thought due to bilateral third nerve nuclear or<br />
fascicular damage or both, bilateral <strong>in</strong>volvement of the <strong>in</strong>terstitial nucleus of Cajal and<br />
the rostral nucleus of the MLF, and <strong>in</strong>volvement of bilateral horizontal saccadic and<br />
smooth pursuit pathways.
256 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 11–3. Etiologies of Third Nerve Palsy (TNP) by Topographical Localization<br />
Nuclear TNP<br />
Infarction or hemorrhage (Bengel, 1994; Bogousslavsky, 1994; Bryan, 1992; Chee, 1990; Gaymard,<br />
1990; Saeki, 2000a; Tezer, 2000; Worth<strong>in</strong>gton, 1996)<br />
Tumor (Chou, 1998; Nakao, 1998; Sanli, 1995)<br />
Infection<br />
Trauma<br />
Multiple sclerosis (Lee, 2000b)<br />
Fascicular TNP<br />
Infarction or hemorrhage (Breen, 1991; Castro, 1990; Gaymard, 1990, 2000; Getenet, 1994; Guy,<br />
1989a; Hopf, 1990; Kim, 1993; Ksiazek, 1994; Liu, 1991; Messe, 2001; Oishi, 1997; Onozu, 1998;<br />
Saeki, 1996, 2000a,b; Thömke, 1995; Umapathi, 2000)<br />
Tumor (<strong>An</strong>dreo, 1994; Barbas, 1995; Eggenberger, 1993; Ishikawa, 1997; Landolfi, 1998; Vetrugno,<br />
1997)<br />
Multiple sclerosis (Newman, 1990; Onozu, 1998; Thömke, 1997)<br />
Stereotactic surgery (Borras, 1997)<br />
Subarachnoid space<br />
<strong>An</strong>eurysms of the <strong>in</strong>ternal carotid–posterior communicat<strong>in</strong>g, superior cerebellar, basilar,<br />
or posterior cerebral arteries (Birchall, 1999; Branley, 1992; DiMario, 1992; Friedman, 2001;<br />
Giomb<strong>in</strong>i, 1991; Good, 1990; Greenspan, 1990; Griffiths, 1994; Horikoshi, 1999; Keane, 1996;<br />
McFadzean, 1998; Mudgil, 1999; Park-Matsumoto, 1997; Ranganadham, 1992; Renowden, 1993;<br />
Richards, 1992; Striph, 1993; Teasdale 1990; Tomsak, 1991; Tummala, 2001; Walter, 1994;<br />
We<strong>in</strong>berg, 1996; Wol<strong>in</strong>, 1992; Zimmer, 1991)<br />
Ectatic vessels (Hashimoto, 1998b; Nakagawa, 1991; Z<strong>in</strong>gale, 1993)<br />
Tumors, especially men<strong>in</strong>giomas, chordomas, metastases, or primary tumors of the third nerve<br />
(Cullom, 1993; Egan, 2001; Hardenack, 1994; Ide, 1997; Jacobson, 2001; Kadota, 1993; Kajiya,<br />
1995; Kawasaki, 1999; Kawase, 1996; Kaye-Wilson, 1994; Kodsi, 1992; Mehta, 1990; Norman,<br />
2001; Ogilvy, 1993; Reifenberger, 1996; Robertson, 1998; Sanchez Dalmau, 1998; Schultheiss,<br />
1993; Takano, 1990; W<strong>in</strong>terkorn, 2001)<br />
Infectious or <strong>in</strong>flammatory processes of the men<strong>in</strong>ges (e.g., sarcoidosis and Wegener’s) and<br />
carc<strong>in</strong>omatous or lymphomatous men<strong>in</strong>gitis (Balm, 1996; Galetta, 1992; Guar<strong>in</strong>o, 1995; Ing,<br />
1992; Ishibashi, 1998; Jacobson, 2001; Keane, 1993; Mark, 1992; McFadzean, 1998; Newman,<br />
1995; Renowden, 1993; Straube, 1993; Ueyama, 1997)<br />
Ophthalmoplegic migra<strong>in</strong>e (O’Hara, 2001)<br />
Subarachnoid hemorrhage with leukemia (Papke, 1993)<br />
Pseudotumor cerebri<br />
Spontaneous <strong>in</strong>tracranial hypotension (Ferrante, 1998)<br />
Trauma, especially dur<strong>in</strong>g neurosurgical procedures (Balcar, 1996; Hedges, 1993; Horikoshi, 1999;<br />
Kudo, 1990; Lepore, 1995)<br />
Nerve <strong>in</strong>farction from diabetes, atherosclerosis, giant cell arteritis, or systemic lupus erythematosus<br />
(nerve <strong>in</strong>farction may also occur <strong>in</strong> the cavernous s<strong>in</strong>us or anywhere along the course of<br />
nerve) (Berlit, 1991; Bondenson, 1997; Capo, 1992; Cullom, 1995; Davies, 1994;<br />
Jacobson, 1994, 1995, 1998a, 2001; Naghmi, 1990; Renowden, 1993; Richards, 1992)<br />
Uncal herniation<br />
Hydrocephalus<br />
Cavernous s<strong>in</strong>us=superior orbital fissure<br />
<strong>An</strong>eurysm of the <strong>in</strong>ternal carotid or posterior communicat<strong>in</strong>g artery (Hahn, 2000; Ikeda, 2001;<br />
Jacobson, 2001; Keane, 1996; Lanz<strong>in</strong>o, 1993; Silva, 1999; Z<strong>in</strong>gale, 1997)<br />
Dural carotid cavernous s<strong>in</strong>us fistula (Acierno, 1995; Brazis, 1994; Keane, 1996; Lee, 1996; Miyachi,<br />
1993; Perez Sempere, 1991; Uehara, 1998; Yen, 1998)<br />
ðcont<strong>in</strong>uedÞ
Table 11–3. (cont<strong>in</strong>ued)<br />
Third Nerve Palsies 257<br />
Ballon test occlusion of the cervical <strong>in</strong>ternal carotid artery (Lopes, 1998)<br />
Cavernous s<strong>in</strong>us thrombosis or <strong>in</strong>fection (e.g., tuberculoma); superior ophthalmic ve<strong>in</strong> thrombosis<br />
(Bikhazi, 1998; Grayeli, 1998; Holland, 1998; Polito, 1996)<br />
Tumors, <strong>in</strong>clud<strong>in</strong>g pituitary adenoma, men<strong>in</strong>gioma, esthesioneuroblastoma, arachnoid cyst,<br />
neur<strong>in</strong>oma, nasopharyngeal carc<strong>in</strong>oma, myeloma, lymphoma, Hodgk<strong>in</strong>’s disease, and metastases<br />
(Barr, 1999; Cullom, 1993; Ing, 1997; Kasner, 1996; Keane, 1996; Kurokawa, 1992; Lee,<br />
2000c; Liu, 1993; Manabe, 2000; Moster, 1996; North, 1993; Shen, 1993; Tao, 1992; Wake, 1993)<br />
Pituitary <strong>in</strong>farction or hemorrhage (pituitary apoplexy) (Lee, 2000c; Rob<strong>in</strong>son, 1990; Rossitch, 1992;<br />
Seyer, 1992)<br />
Gammopathy<br />
Intraneural hemorrhage (Miyao, 1993)<br />
Mucocele of the sphenoid s<strong>in</strong>us (Ashw<strong>in</strong>, 2001)<br />
Sphenoid s<strong>in</strong>usitis (Chotmongkol, 1999)<br />
Tolosa-Hunt syndrome, Wegener’s granulomatosis, or other granulomatous diseases (Herman,<br />
1999; Jacobson, 2001; Keane, 1996)<br />
Orbit<br />
Infections, <strong>in</strong>flammations, and granulomatous processes (e.g., orbital pseudotumor) (Kondoh,<br />
1998; Ohtsuka, 1997; Stefanis, 1993)<br />
Sphenoid s<strong>in</strong>us mucocele (Sethi, 1997)<br />
Tumors (Goldberg, 1990a,b)<br />
Dural arteriovenous malformation (Gray, 1999)<br />
Trauma<br />
Unknown localization<br />
Congenital (Good, 1991; Hamed, 1991; Ing, 1992; Mudgil, 1999; Parmeggiani 1992; Patel, 1993;<br />
Pratt, 1995; Schumacher-Feero, 1999; Tsaloumas, 1997; White, 1992)<br />
Migra<strong>in</strong>e (Mark, 1998; O’Halloran, 1999; Prats, 1999)<br />
Viral <strong>in</strong>fections (<strong>in</strong>clud<strong>in</strong>g herpes zoster ophthalmicus or Ramsay Hunt syndrome) and<br />
immunizations (Capoferri, 1997; Chang-God<strong>in</strong>ich, 1997; Mansour, 1997; Saeki, 2000c; Sood, 1999;<br />
Zurev<strong>in</strong>sky, 1993)<br />
Lyme disease (Savas, 1997)<br />
Diffuse neuropathic processes (e.g., Fisher’s syndrome and chronic <strong>in</strong>flammatory polyradiculoneuropathy<br />
(CIDP) (Arroyo, 1995; Nagaoka, 1996)<br />
Cervical carotid artery dissection, stenosis, or occlusion (Balcar, 1997; Holl<strong>in</strong>ger, 1999; Koennecke,<br />
1998; Mokri, 1996; Schiev<strong>in</strong>k, 1993)<br />
Subdural hematomas (Okuchi, 1999; Phookan, 1994)<br />
Glioblastoma multiforme (Al-Yamany, 1999)<br />
Un<strong>in</strong>tentional subdural catheter (Haughton, 1999)<br />
Submucosal diathermy to the <strong>in</strong>ferior turb<strong>in</strong>ates to improve the nasal airway (Green, 2000)<br />
Toxic effects of drugs (Pacifici, 1993; Soysal, 1993)<br />
Coca<strong>in</strong>e (Migita, 1998)<br />
Sildenafil citrate (Viagra) (Donahue, 1998)<br />
Internal carotid cisplat<strong>in</strong> <strong>in</strong>fusion (<strong>in</strong>ferolateral trunk carotid artery neurovascular toxicity)<br />
(Alderson, 1996; Wu, 1997)<br />
Dental anesthesia<br />
Radiation therapy (Ebner, 1995)<br />
Partial TNP associated with elevated anti-galactocerebroside and anti-GM 1 antibodies (Go, 2000)
258 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Is the TNP Due to a Fascicular Lesion?<br />
Lesions of the third nerve fascicle often accompany nuclear lesions because <strong>in</strong>farction is<br />
a common cause of a nuclear TNP, and the paramedian branches near the top of the<br />
basilar artery often feed both structures. For example, <strong>in</strong>farction of the dorsal paramedian<br />
midbra<strong>in</strong> may cause bilateral ptosis associated with unilateral paresis of all<br />
other muscles <strong>in</strong>nervated by the oculomotor nerve (pupil spared) with spar<strong>in</strong>g of the<br />
contralateral superior rectus muscle (Liu, 1991). These unique f<strong>in</strong>d<strong>in</strong>gs suggest a lesion<br />
of the proximal third nerve fascicles and the central caudal subnucleus. Third nerve<br />
fascicular lesions are often caused by <strong>in</strong>farction, hemorrhage, or demyel<strong>in</strong>ation. Pure<br />
fascicular lesions cause a unilateral peripheral type of oculomotor palsy. Involvement of<br />
bra<strong>in</strong>stem structures other than the fascicle of the third nerve identifies the mesencephalic<br />
location of the lesion (Liu, 1992). Concomitant damage of the red nucleus<br />
and superior cerebellar peduncle causes contralateral ataxia and outflow tract cerebellar<br />
tremor (Claude’s syndrome), whereas a more anterior lesion, affect<strong>in</strong>g the peduncle,<br />
gives rise to oculomotor palsy with contralateral hemiparesis (Weber’s syndrome). The<br />
TNP with Weber’s syndrome may affect or spare the pupillary fibers (Saeki, 1996).<br />
Larger lesions that affect the oculomotor fascicle and the red nucleus=substantia nigra<br />
region may produce TNP with contralateral choreiform movements or tremor<br />
(Benedikt’s syndrome) (Borras, 1997), sometimes associated with contralateral hemiparesis<br />
if the cerebral peduncle is also <strong>in</strong>volved. A pupil-spar<strong>in</strong>g TNP associated with<br />
b<strong>in</strong>ocular ocular torsion to the contralateral side—thereby <strong>in</strong>dicat<strong>in</strong>g a left-sided<br />
midbra<strong>in</strong> lesion that <strong>in</strong>cluded the fascicle of the third nerve and the supranuclear<br />
<strong>in</strong>tegration centers for torsional eye movements, the <strong>in</strong>terstitial nucleus of Cajal, and the<br />
rostral <strong>in</strong>terstitial nucleus of the MLF—has been described with a paramedian rostral<br />
midbra<strong>in</strong> <strong>in</strong>farction <strong>in</strong> a diabetic with giant cell arteritis (Dichgans, 1995). Ipsilateral<br />
TNP and contralateral downbeat nystagmus may be caused by unilateral paramedian<br />
thalamopeduncular <strong>in</strong>farction (Oishi, 1997).<br />
Rarely, a unilateral or bilateral fascicular third nerve lesion may occur <strong>in</strong> isolation<br />
without other ocular motor or neurologic signs or symptoms (see below) (<strong>An</strong>dreo, 1994;<br />
Barbas, 1995; Getenet, 1994; Kim, 1993; Newman, 1990; Thömke, 1995). Fascicular<br />
lesions, even when bilateral, may occasionally spare the pupil(s). Bilateral preganglionic<br />
<strong>in</strong>ternal ophthalmoplegia has been described with bilateral partial oculomotor fascicular<br />
lesions (Hashimoto, 1998a). Because of the <strong>in</strong>traaxial topographic arrangement of<br />
fibers, fascicular lesions may cause TNP limited to specific oculomotor-<strong>in</strong>nervated<br />
muscles (Ksiazek, 1994). Fascicular lesions have resulted <strong>in</strong> the follow<strong>in</strong>g:<br />
1. Isolated <strong>in</strong>ferior oblique paresis (Castro, 1990)<br />
2. Unilateral fixed, dilated pupil unassociated with other neurologic dysfunction<br />
(Shuaib, 1989)<br />
3. Paresis of the superior rectus and <strong>in</strong>ferior oblique without other evidence of<br />
oculomotor nerve <strong>in</strong>volvement (Gauntt, 1995)<br />
4. Paresis of the superior and medial rectus (Saeki, 2000a)<br />
5. Paresis of the levator muscle, superior rectus, and medial rectus (Onozu, 1998)<br />
6. Paresis of the <strong>in</strong>ferior oblique, superior rectus, medial rectus, and levator muscle with<br />
spar<strong>in</strong>g of the <strong>in</strong>ferior rectus muscle and pupil (Naudea, 1983; Schwartz, 1995;<br />
Shuaib, 1987)<br />
7. Paresis of the <strong>in</strong>ferior oblique, superior rectus, medial rectus, levator, and <strong>in</strong>ferior<br />
rectus with pupillary spar<strong>in</strong>g (Breen, 1991; Naudea, 1983)
8. Paresis of the left <strong>in</strong>ferior rectus, left pupil, right superior rectus, convergence, and<br />
left medial rectus (Umapathi, 2000).<br />
Based on these cl<strong>in</strong>ical studies, it has been proposed that <strong>in</strong>dividual third nerve<br />
fascicles <strong>in</strong> the ventral mesencephalon are arranged topographically from lateral to<br />
medial as follows: <strong>in</strong>ferior oblique, superior rectus, medial rectus and levator palpebrae,<br />
<strong>in</strong>ferior rectus, and pupillary fibers (Castro, 1990). A rostral-caudal topographic<br />
arrangement has also been suggested with pupillary fibers most superior, followed<br />
by fibers to the <strong>in</strong>ferior rectus, <strong>in</strong>ferior oblique, medial rectus, superior rectus, and<br />
levator, <strong>in</strong> that order (Saeki, 2000a; Schwartz, 1995). This model also accounts for the<br />
description of superior and <strong>in</strong>ferior division oculomotor palsies. The superior division<br />
paresis <strong>in</strong>volves the superior rectus and levator muscles without <strong>in</strong>volvement of other<br />
groups (Guy, 1989a; Hriso, 1990; Ksiazek, 1989). The <strong>in</strong>ferior division oculomotor<br />
palsies cause paresis of <strong>in</strong>ferior rectus, <strong>in</strong>ferior oblique, medial rectus, and pupillary<br />
fibers with spar<strong>in</strong>g of the superior rectus and levator (Abdollah, 1990; Eggenberger,<br />
1993; Ksiazek, 1989). Both divisional palsies may be associated with <strong>in</strong>traaxial midbra<strong>in</strong><br />
lesions. Thus, although superior and <strong>in</strong>ferior divisional TNP have classically been<br />
localized to anterior cavernous s<strong>in</strong>us or posterior orbital lesions, a divisional TNP may<br />
occur from damage at any location along the course of the oculomotor nerve, from the<br />
fascicle to the orbit (Ksiazek, 1989).<br />
Fascicular TNP may occasionally be associated with ipsilateral ptosis and contralateral<br />
eyelid retraction (plus-m<strong>in</strong>us lid syndrome) (Gaymard, 1992; Vetrugno, 1997).<br />
This syndrome occurs with a small lesion located <strong>in</strong> the paramedian mesencephalon.<br />
There is <strong>in</strong>volvement of the ipsilateral levator palpebrae fascicles as they emerge from<br />
the central caudal nucleus (the central caudal nucleus is spared), and the <strong>in</strong>hibitory<br />
pathways project<strong>in</strong>g on the levator palpebrae motor neurons immediately before<br />
their entrance <strong>in</strong> the central caudal nucleus. The plus-m<strong>in</strong>us syndrome has been<br />
described with bilateral glioma extend<strong>in</strong>g to the paramedian midbra<strong>in</strong> and thalamicmesencephalic<br />
<strong>in</strong>farction; it also may occur with peripheral processes such as peripheral<br />
TNP, myasthenia gravis, orbital myositis, congenital ptosis, or orbital trauma.<br />
Is the TNP Due to a Subarachnoid Lesion?<br />
Third Nerve Palsies 259<br />
<strong>An</strong> isolated pupil spared peripheral TNP is most often related to an ischemic neuropathy<br />
or a lesion affect<strong>in</strong>g its subarachnoid portion. Subarachnoid lesions may distort or<br />
<strong>in</strong>jure the bra<strong>in</strong>stem, and diffuse processes will show signs of men<strong>in</strong>geal irritation.<br />
Etiologies of TNP due to a subarachnoid lesion are outl<strong>in</strong>ed <strong>in</strong> Table 11–3. Third nerve<br />
schwannoma may cause a pa<strong>in</strong>ful relaps<strong>in</strong>g-remitt<strong>in</strong>g TNP mimick<strong>in</strong>g the cl<strong>in</strong>ical<br />
syndrome of ophthalmoplegic migra<strong>in</strong>e (Kawasaki, 1999). Monocular elevator paresis<br />
from isolated superior rectus and=or <strong>in</strong>ferior oblique dysfunction may occur <strong>in</strong><br />
neurofibromatosis type 2–related schwannoma (Egan, 2001). The third nerve is also<br />
susceptible to trauma <strong>in</strong> the subarachnoid space, especially dur<strong>in</strong>g neurosurgical<br />
procedures (Hedges, 1993; Horikoshi, 1999; Kudo, 1990). Closed head trauma may<br />
cause TNP due to shear<strong>in</strong>g <strong>in</strong>jury result<strong>in</strong>g <strong>in</strong> distal fascicular damage or partial root<br />
avulsion (Balcar, 1996). Walter et al described two patients with TNP precipitated by<br />
m<strong>in</strong>or head trauma with negative bra<strong>in</strong> computed tomography (CT) scans who were<br />
subsequently discovered to have ipsilateral posterior communicat<strong>in</strong>g artery aneurysms
260 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
(Walter, 1994). Park-Matsumoto and Tazawa described a similar case (Park-Matsumoto,<br />
1997).<br />
Compression of the third nerve by an aneurysm characteristically causes dilatation<br />
and unresponsiveness of the pupil. Compressive subarachnoid lesions may occasionally<br />
spare the pupil, however. Two explanations have been proposed: (1) compression may<br />
be evenly distributed and the relatively pressure-resistant, smaller-caliber pupillomotor<br />
fibers escape <strong>in</strong>jury; or (2) the lesion compresses only the <strong>in</strong>ferior portion of the nerve<br />
and spares the dorsally situated pupillomotor fibers. The TNP due to an aneurysm may<br />
be <strong>in</strong>complete with at least one element of nerve dysfunction (i.e., ptosis, mydriasis, or<br />
extraocular muscle weakness) be<strong>in</strong>g absent. Ptosis has been described <strong>in</strong> isolation as the<br />
sole manifestation of third nerve compression by a posterior communicat<strong>in</strong>g artery<br />
aneurysm (Good, 1990). Rarely, aneurysmal TNP may even be transient and clear<br />
spontaneously (Greenspan, 1990).<br />
A normal pupil <strong>in</strong> the sett<strong>in</strong>g of a complete somatic oculomotor paresis, however,<br />
essentially excludes a diagnosis of aneurysm (see below). A s<strong>in</strong>gle patient has been<br />
described <strong>in</strong> whom a pa<strong>in</strong>less, pupil-spar<strong>in</strong>g but otherwise complete oculomotor paresis<br />
was the only sign of an aneurysm aris<strong>in</strong>g from the basilar artery (Lustbader, 1988).<br />
Conversely, an isolated pupillary paralysis without ptosis or ophthalmoparesis is rarely<br />
caused by an aneurysm or other subarachnoid lesion (Kaye-Wilson, 1994; Wilhelm,<br />
1995). Koennecke and Seyfert reported a patient with a common carotid artery<br />
dissection from <strong>in</strong>traoperative trauma whose mydriasis preceded a complete TNP by<br />
12 hours (Koennecke, 1998).<br />
Is the TNP Due to a Cavernous S<strong>in</strong>us Lesion?<br />
Lesions of the third nerve <strong>in</strong> the cavernous s<strong>in</strong>us often also <strong>in</strong>volve the other ocular<br />
motor nerves, the ophthalmic branch of the trigem<strong>in</strong>al nerve, and sympathetic fibers.<br />
Sensory fibers from the ophthalmic division of the fifth cranial nerve jo<strong>in</strong> the oculomotor<br />
nerve with<strong>in</strong> the lateral wall of the cavernous s<strong>in</strong>us. The frontal-orbital pa<strong>in</strong><br />
experienced by patients with enlarg<strong>in</strong>g aneurysms could thus be caused by direct<br />
irritation of the third nerve (Lanz<strong>in</strong>o, 1993). Compressive cavernous s<strong>in</strong>us lesions may<br />
also spare the pupil because they often preferentially <strong>in</strong>volve only the superior division<br />
of the oculomotor nerve that carries no pupillomotor fibers (Silva, 1999) or the superior<br />
aspect of the nerve anterior to the po<strong>in</strong>t where the pupillomotor fibers descend <strong>in</strong> their<br />
course near the <strong>in</strong>ferior oblique muscle. The pupillary ‘‘spar<strong>in</strong>g’’ with anterior cavernous<br />
s<strong>in</strong>us lesions may be more apparent than real, result<strong>in</strong>g from simultaneous <strong>in</strong>jury<br />
of nerve fibers to both the pupillary sph<strong>in</strong>cter and dilator, caus<strong>in</strong>g a mid-position fixed<br />
pupil or from aberrant regeneration (see below). Ikeda et al described a patient with a<br />
pa<strong>in</strong>ful, ‘‘severe’’ TNP with normal pupils due to a cavernous s<strong>in</strong>us aneurysm (Ikeda,<br />
2001). Lesions <strong>in</strong> the neighborhood of the posterior cl<strong>in</strong>oid process may for some time<br />
affect only the third nerve as it pierces the dura (e.g., breast and prostatic carc<strong>in</strong>oma)<br />
(Cullom, 1993). Medial lesions <strong>in</strong> the cavernous s<strong>in</strong>us, such as a carotid artery<br />
aneurysm, may affect only the ocular motor nerves but spare the more laterally located<br />
ophthalmic branch of the trigem<strong>in</strong>al nerve, result<strong>in</strong>g <strong>in</strong> pa<strong>in</strong>less ophthalmoplegia.<br />
Lesions that beg<strong>in</strong> laterally present with retro-orbital pa<strong>in</strong> first, and only later<br />
does ophthalmoparesis supervene. Lesions located <strong>in</strong> the cavernous s<strong>in</strong>us caus<strong>in</strong>g<br />
TNP are outl<strong>in</strong>ed <strong>in</strong> Table 11–3. The cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs and etiologies for processes
located <strong>in</strong> the superior orbital fissure are similar to those of the cavernous s<strong>in</strong>us<br />
syndrome.<br />
Is the TNP Due to an Orbital Lesion?<br />
Lesions with<strong>in</strong> the orbit that produce third nerve dysfunction usually produce other<br />
ocular motor dysfunction as well as optic neuropathy and proptosis (Goldberg,<br />
1990a,b). Lesions may extend from the cavernous s<strong>in</strong>us to the orbital apex and vice<br />
versa so that a clear dist<strong>in</strong>ction between the two syndromes may be impossible. Isolated<br />
<strong>in</strong>volvement of the muscles <strong>in</strong>nervated by either the superior or the <strong>in</strong>ferior oculomotor<br />
branch has classically been localized to an orbital process: often trauma, tumor, or<br />
<strong>in</strong>fection, or a sphenocavernous lesion (Stefanis, 1993). However, as we noted, the<br />
functional division of the third nerve is present probably even at the fascicular level, and<br />
a divisional pattern may occur from damage anywhere along the course of the nerve.<br />
Superior division or <strong>in</strong>ferior division third nerve paresis may occur with subarachnoid<br />
lesions (Guy, 1985), and isolated superior division paresis has been described with a<br />
superior cerebellar–posterior cerebral artery junction aneurysm that compressed and<br />
flattened the <strong>in</strong>terpeduncular third nerve from below (Guy, 1989b). Superior branch<br />
palsy has also been described with basilar artery aneurysm, <strong>in</strong>tracavernous carotid<br />
aneurysm, migra<strong>in</strong>e, diabetes, lymphoma, sphenoidal abscess, sphenoid s<strong>in</strong>usitis,<br />
frontal s<strong>in</strong>us mucocele, viral illness, men<strong>in</strong>gitis, and after craniotomy (Chotmongkol,<br />
1992, 1999; Ehrenpries, 1995; Guy, 1989b; Manabe, 2000; O’Halloran, 1999; Saeki, 2000c;<br />
Silva, 1999; Stefanis, 1993). Even ophthalmoplegic migra<strong>in</strong>e may cause recurrent<br />
paroxysmal superior division oculomotor palsy. Isolated superior division-like paresis<br />
may be mimicked by myasthenia gravis (Dehaene, 1995). Isolated <strong>in</strong>ferior division<br />
<strong>in</strong>volvement has occurred with trauma, mesencephalic <strong>in</strong>farction and tumor (Abdollah,<br />
1990; Eggenberger, 1993; Ksiazek, 1989), basilar artery aneurysm (Kardon, 1991),<br />
parasellar tumors (e.g., men<strong>in</strong>gioma, schwannoma) (Carlow, 1990), viral illness, orbital<br />
dural arteriovenous malformation (Gray, 1999), as part of a more generalized vasculitic<br />
or demyel<strong>in</strong>at<strong>in</strong>g neuropathy (Cunn<strong>in</strong>gham, 1994), and <strong>in</strong> association with elevated<br />
antigalactocerebroside and anti-GM1 antibodies (Go, 2000). Inferior division <strong>in</strong>volvement<br />
with tumors may be pupil-spar<strong>in</strong>g, perhaps because of <strong>in</strong>sidious tumor growth<br />
spar<strong>in</strong>g pressure-resistant pupillomotor fibers.<br />
Partial or complete TNP may rarely follow dental anesthesia, presumably due to<br />
<strong>in</strong>advertent <strong>in</strong>jection of an anesthetic agent <strong>in</strong>to the <strong>in</strong>ferior dental artery or superior<br />
alveolar artery with subsequent retrograde flow <strong>in</strong>to the maxillary, middle men<strong>in</strong>geal,<br />
and f<strong>in</strong>ally the lacrimal branch of the ophthalmic artery.<br />
What Is the Evaluation of Nonisolated TNP?<br />
Third Nerve Palsies 261<br />
Nonisolated TNP should undergo neuroimag<strong>in</strong>g, with attention to areas suggested<br />
topographically by the associated neurologic signs and symptoms. Appropriate <strong>in</strong>vestigations<br />
and neuroimag<strong>in</strong>g studies are directed at the precise area of <strong>in</strong>terest, and this<br />
area is determ<strong>in</strong>ed by the associated localiz<strong>in</strong>g features with the TNP (Brazis, 1991; Lee,<br />
1999). In general, MRI with and without gadol<strong>in</strong>ium enhancement is the neuroimag<strong>in</strong>g<br />
modality of choice for all these processes (Renowden, 1993). Contrast-enhanced CT
262 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
scann<strong>in</strong>g with narrow (2-mm) collimation is reserved for those patients who cannot<br />
tolerate MRI or <strong>in</strong> whom MRI is contra<strong>in</strong>dicated (e.g., pacemaker, claustrophobia,<br />
metallic clips <strong>in</strong> head, etc.) (Renowden, 1993; Teasdale, 1990). CT scann<strong>in</strong>g is also the<br />
appropriate first-choice neuroimag<strong>in</strong>g study <strong>in</strong> patients be<strong>in</strong>g evaluated for acute head<br />
trauma or acute vascular events (<strong>in</strong>farction or hemorrhage). If there are cl<strong>in</strong>ical signs of<br />
a men<strong>in</strong>geal process, lumbar puncture should be performed. The evaluation of a patient<br />
with TNP is summarized <strong>in</strong> Figure 11–1.<br />
Is the TNP Due to Trauma?<br />
Traumatic isolated TNP (type 2) should undergo CT scann<strong>in</strong>g to search for associated<br />
central nervous system damage (e.g., subdural or <strong>in</strong>tracerebral hematoma) as <strong>in</strong>dicated<br />
by associated neurologic signs and symptoms (Balcar, 1996; Hedges, 1993; Kudo,<br />
1990; Lepore, 1995; Phookan, 1994). TNP after mild head trauma have been observed<br />
<strong>in</strong> association with otherwise asymptomatic lesions (e.g., cerebral aneurysm) (Park-<br />
Matsumoto, 1997; Walter, 1994). Although uncommon, neuroimag<strong>in</strong>g may be<br />
warranted <strong>in</strong> patients with TNP after m<strong>in</strong>imal or trivial trauma to exclude mass lesions<br />
or cerebral aneurysms (class III–IV, level C).<br />
Is the TNP Congential?<br />
Congenital isolated TNP (type 3) is rare, usually unilateral, and may occur <strong>in</strong> isolation or<br />
<strong>in</strong> association with other neurologic and systemic abnormalities, <strong>in</strong>clud<strong>in</strong>g congenital<br />
facial nerve palsies or other cranial neuropathies, facial capillary hemangioma, cerebellar<br />
hypoplasia, gaze palsy, ipsilateral nevus sebaceous of Jadassohn, mental retardation, and<br />
digital anomalies (Good, 1991; Hamed, 1991; Ing, 1992; Parmeggiani, 1992; Patel, 1993;<br />
Pratt, 1995; Shumacher-Feero, 1999; White, 1992). All patients have some degree of ptosis<br />
and ophthalmoplegia, and nearly all have pupillary <strong>in</strong>volvement. In most cases, the<br />
pupil is miotic rather than dilated, probably because of aberrant third nerve regeneration,<br />
and usually trace reactive or nonreactive to light. Rarely the pupil may be spared.<br />
Amblyopia is common (Schumacher-Feero, 1999). Most cases are spontaneous, but<br />
familial cases have been described. We recommend MRI <strong>in</strong> all patients with congenital<br />
TNPs, ma<strong>in</strong>ly to search for associated structural abnormalities of the bra<strong>in</strong> (class III–IV,<br />
level C).<br />
Is the Isolated TNP Acquired and<br />
Nontraumatic?<br />
Acquired, nontraumatic isolated TNP (type 4) may occur with lesions localized<br />
anywhere along the course of the third nerve from the fascicle to the orbit (Renowden<br />
1993). For cl<strong>in</strong>ical purposes, isolated TNP may be divided <strong>in</strong>to three types (types<br />
4A–4C) (Jacobson, 1999; Lee, 1999; Trobe 1985) (Table 11–1).
Figure 11–1. Evaluation of third nerve palsy (TNP).<br />
Third Nerve Palsies 263
264 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Does the Patient Have an Acquired Isolated<br />
TNP with a Normal Pupillary Sph<strong>in</strong>cter with<br />
Completely Palsied Extraocular Muscles<br />
(Type 4A TNP)?<br />
TNP with a normal pupillary sph<strong>in</strong>cter and completely palsied extraocular muscles is<br />
almost never due to an <strong>in</strong>tracranial aneurysm. However, a s<strong>in</strong>gle patient has been<br />
described <strong>in</strong> whom a pa<strong>in</strong>less, pupil-spar<strong>in</strong>g, but otherwise complete TNP was the only<br />
sign of an aneurysm aris<strong>in</strong>g from the basilar artery (Lustbader 1988). A similar pa<strong>in</strong>ful<br />
TNP palsy has been described with an aneurysm <strong>in</strong> the cavernous s<strong>in</strong>us (Ikeda, 2001),<br />
and pupillary spar<strong>in</strong>g may rarely occur with pituitary adenoma. This type of TNP is<br />
most commonly caused by ischemia, especially associated with diabetes mellitus. In a<br />
retrospective review of 34 consecutive cases of isolated atraumatic TNP, diabetes<br />
mellitus was the most common etiology account<strong>in</strong>g for 46% of the cases (Renowden<br />
1993). Ischemic TNP may also occur with giant cell arteritis (Berlit, 1991; Bondenson,<br />
1997; Davies, 1994; Renowden, 1993; Richards, 1992) and systemic lupus erythematosus.<br />
Pupil-spar<strong>in</strong>g TNP has also been reported with sildenafil citrate (Viagra) (Donahue,<br />
1998) and coca<strong>in</strong>e use (Migita, 1998). Significant risk factors for ischemic oculomotor<br />
nerve palsies <strong>in</strong>clude diabetes, left ventricular hypertrophy, and elevated hematocrit<br />
(Jacobson, 1994). Obesity, hypertension, and smok<strong>in</strong>g are also probable risk factors.<br />
Ischemic damage to the trigem<strong>in</strong>al fibers <strong>in</strong> the oculomotor nerve may be the source of<br />
pa<strong>in</strong> <strong>in</strong> ischemic-diabetic TNPs (Bortolami, 1993).<br />
Ischemic lesions of the oculomotor nerve often spare the pupil because the lesion is<br />
conf<strong>in</strong>ed to the core of the nerve and does not affect peripherally situated pupillomotor<br />
fibers. However, the pupil may be <strong>in</strong>volved <strong>in</strong> diabetic oculomotor palsies (Naghmi,<br />
1990), and diabetes may even cause a superior branch palsy of the oculomotor nerve.<br />
Pupil spar<strong>in</strong>g has been documented <strong>in</strong> 62 to 86% of TNPs due to ischemia (Jacobson,<br />
1998a). In a prospective study of 26 consecutive patients with diabetes-associated TNP,<br />
<strong>in</strong>ternal ophthalmoplegia occurred <strong>in</strong> 10 patients (38%) (Jacobson, 1998a). The size of<br />
anisocoria was 1 mm or less <strong>in</strong> most patients. Only two patients had anisocoria greater<br />
than 2.0 mm, and it was never greater than 2.5 mm. No patient had a fully dilated<br />
unreactive pupil. The author concluded that pupil <strong>in</strong>volvement <strong>in</strong> patients with<br />
diabetes-associated TNP occurs more often than has previously been recognized (14<br />
to 32% <strong>in</strong> other studies), although the degree of anisocoria <strong>in</strong> any one patient is usually<br />
1 mm or less. When comment<strong>in</strong>g on this study, Trobe stated, ‘‘We can presume that all<br />
patients who have oculomotor nerve palsies with anisocoria of greater than 2.0 mm are<br />
outliers for the diagnosis of ischemia’’ (Trobe, 1998).<br />
Postmortem exam<strong>in</strong>ations <strong>in</strong> three diabetic patients have demonstrated pathologic<br />
changes <strong>in</strong> the subarachnoid or cavernous s<strong>in</strong>us portion of the nerves. Ischemic TNP<br />
with pupillary spar<strong>in</strong>g, however, has also been reported due to fascicular damage with<br />
mesencephalic <strong>in</strong>farcts documented on MRI (Breen, 1991; Dichgans, 1995; Hopf, 1990;<br />
Murakami, 1994; Thömke, 1995). Keane and Ahmadi, however, noted that most diabetic<br />
TNP are peripheral (Keane, 1998). In their MRI study of 49 diabetic patients with<br />
isolated, unilateral TNPs, only one was found to have a bra<strong>in</strong>stem <strong>in</strong>farct. Of eight<br />
diabetics with midbra<strong>in</strong> <strong>in</strong>farcts and TNPs, seven had other central nervous system<br />
f<strong>in</strong>d<strong>in</strong>gs and five had bilateral TNPs.<br />
In a prospective study of 16 patients with ischemic TNPs, 11 (69%) had progression of<br />
ophthalmoplegia with a median time between reported onset and peak severity of
ophthalmoplegia of 10 days (Jacobson, 1995). Almost all patients with an ischemic TNP<br />
will improve with<strong>in</strong> 4 to 12 weeks of onset of symptoms (Capo, 1992).<br />
Sanders et al retrospectively studied 55 patients with vasculopathic TNP (Sanders,<br />
2001). Of these, 42 (76%) had normal pupillary function. Of these 42 patients, 23 (55%)<br />
demonstrated an <strong>in</strong>complete extraocular muscle palsy, def<strong>in</strong>ed as partially reduced<br />
ductions affect<strong>in</strong>g all third nerve <strong>in</strong>nervated extraocular muscles and levator (diffuse<br />
pattern) or partially reduced ductions that <strong>in</strong>volved only some third nerve <strong>in</strong>nervated<br />
muscles and levator (focal pattern). Twenty (87%) of these 23 patients showed a diffuse<br />
pattern or paresis and only three (13%) showed a focal pattern of paresis, one that<br />
affected only the superior rectus and levator muscles (superior division weakness).<br />
Based on their series, the authors noted that most patients with extraocular muscle and<br />
levator <strong>in</strong>volvement <strong>in</strong> pupil-spar<strong>in</strong>g, <strong>in</strong>complete TNPs of vasculopathic orig<strong>in</strong> have a<br />
diffuse pattern of paresis, whereas <strong>in</strong> the literature pupil-spar<strong>in</strong>g TNPs of aneurysmal<br />
orig<strong>in</strong> usually have a focal pattern of paresis.<br />
Adults who develop type 4A TNP do not need angiography (Jacobson, 1999; Miller,<br />
1999). <strong>An</strong> MRI scan need not be performed <strong>in</strong>itially, as the yield for detect<strong>in</strong>g a<br />
compressive lesion is very low, especially if the TNP resolves over time (class III–IV,<br />
level C). <strong>Neuro</strong>imag<strong>in</strong>g should be performed <strong>in</strong> patients with no vasculopathic risk<br />
factors or <strong>in</strong> patients who do not improve by 12 weeks of follow-up (class III–IV, level<br />
B). Patients with type 4A TNP should be observed for the first 24 to 48 hours dur<strong>in</strong>g the<br />
first week because some patients with aneurysms may develop delayed pupil <strong>in</strong>volvement.<br />
Patients who develop pupil <strong>in</strong>volvement should be reevaluated (see below).<br />
Vasculopathic risk factors, especially diabetes mellitus, hypertension, and <strong>in</strong>creased<br />
cholesterol, should be sought and controlled. Patients over the age of 55 years,<br />
especially those with other symptoms suggestive of giant cell arteritis (e.g., headache,<br />
jaw or tongue claudication, polymyalgia rheumatica symptoms), should have a<br />
sedimentation rate determ<strong>in</strong>ation (Bondenson, 1997; Davies 1994). Temporal artery<br />
biopsy should be performed if the sedimentation rate is elevated or other systemic<br />
symptoms are present (class III–IV, level C). Myasthenia gravis may rarely mimic this<br />
type of TNP, so an evaluation (e.g., Tensilon or Prostigm<strong>in</strong> test, antiacetylchol<strong>in</strong>e<br />
antibodies, etc.) should be considered, primarily <strong>in</strong> patients with fluctuat<strong>in</strong>g or<br />
fatigu<strong>in</strong>g ptosis or ophthalmoplegia (class III–IV, level C). If the complete, pupilspared<br />
TNP improves follow<strong>in</strong>g a period of observation, no neuroimag<strong>in</strong>g is required<br />
(class III–IV, level C). Some authors recommend non<strong>in</strong>vasive vascular studies (MRI with<br />
MR or CT angiography) <strong>in</strong> all patients with TNP, regardless of whether or not they have<br />
diabetes or any other systemic vasculopathy, with the one exception be<strong>in</strong>g patients with<br />
an otherwise complete TNP (i.e., complete ptosis, no adduction, no depression, no<br />
elevation) but normally reactive, isocoric pupils (Miller, 1999).<br />
Does the Patient Have an Acquired Isolated<br />
TNP with a Normal Pupillary Sph<strong>in</strong>cter and<br />
Incomplete Palsied Extraocular Muscles<br />
(Type 4B TNP)?<br />
Third Nerve Palsies 265<br />
Patients with an <strong>in</strong>complete motor TNP with pupillary spar<strong>in</strong>g require an MRI scan to<br />
rule out a mass lesion. If the MRI is normal, cerebral angiography should be considered<br />
to <strong>in</strong>vestigate the presence of an aneurysm, dural-cavernous s<strong>in</strong>us fistula, or high-grade
266 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
carotid stenosis. Three-dimensional time-of-flight MR angiography (MRA) or CT<br />
angiography may well reveal an aneurysm or other vascular malformation and may<br />
eventually take the place of arteriography (Jacobson, 1999; Kaufman, 1994; McFadzean,<br />
1998; Tomsak, 1991; We<strong>in</strong>berg, 1996); however, at this time, cerebral angiography is the<br />
‘‘gold standard’’ for the diagnosis of cerebral aneurysms (Davis, 1996; Trobe, 1998).<br />
Although MRA may be able to detect up to 95% of cerebral aneurysms that will bleed, it<br />
cannot exclude aneurysm as the etiology of a pupil-<strong>in</strong>volved TNP. Jacobson and Trobe<br />
addressed whether or not MRA was adequate for evaluat<strong>in</strong>g for aneurysms <strong>in</strong> patients<br />
with TNP (Jacobson, 1999). They noted that <strong>in</strong> 46 well-documented aneurysms of the<br />
posterior communicat<strong>in</strong>g artery caus<strong>in</strong>g TNP, the aneurysm diameters ranged from 3 to<br />
17 mm (median 8 mm); 42 of these (91.3%) measured 5 mm or more, and four (8.7%)<br />
measured less than 5 mm (Teasdale, 1990). They then <strong>in</strong>vestigated how sensitive MRA is<br />
<strong>in</strong> detect<strong>in</strong>g aneurysms and found that MRA detected 64 (97%) of 66 aneurysms 5 mm<br />
or greater <strong>in</strong> diameter but only 15 (53.6%) of 28 aneurysms less than 5 mm <strong>in</strong> diameter.<br />
The relationship between aneurysm size and risk of rupture was then assessed. Among<br />
the 115 aneurysms 5 mm or greater, 15 (13.0%) ruptured. None of the 40 aneurysms with<br />
a diameter of less than 5 mm ruptured. Comb<strong>in</strong><strong>in</strong>g these data, the authors estimated<br />
that properly performed MRA will overlook only 1.5% of aneurysms that cause TNP<br />
and that will go on to rupture dur<strong>in</strong>g the subsequent 8 years if untreated. The authors<br />
believe that MRA may assume an important role <strong>in</strong> the evaluation of patients with<br />
isolated TNP. When MRA is properly performed and <strong>in</strong>terpreted, the risk of overlook<strong>in</strong>g<br />
an aneurysm likely to rupture is nearly equal to the aggregate risk of stroke,<br />
myocardial <strong>in</strong>farction, or death associated with catheter angiography. Because of the<br />
potentially drastic consequences of overlook<strong>in</strong>g an aneurysm, however, the authors<br />
believe that MRA should be considered the def<strong>in</strong>itive screen<strong>in</strong>g test only <strong>in</strong> patients<br />
with a relatively low likelihood of harbor<strong>in</strong>g an aneurysm or relatively high likelihood<br />
of suffer<strong>in</strong>g a complication dur<strong>in</strong>g catheter angiography (e.g., age greater than 70,<br />
symptomatic atherosclerotic cardiovascular disease, significant cardiovascular or renal<br />
disease, Ehlers-Danlos syndrome). In patients with type 4B TNP (pupil-spar<strong>in</strong>g <strong>in</strong>complete<br />
TNP) (plus patient age greater than or equal to 40 years and vasculopathic factors<br />
present), these authors recommend MRI followed by MRA if MRI does not disclose a<br />
nonaneurysmal cause. Catheter angiography is recommended if (1) worsen<strong>in</strong>g of<br />
extraocular muscle or iris sph<strong>in</strong>cter impairment cont<strong>in</strong>ues beyond 14 days; (2) iris<br />
sph<strong>in</strong>cter impairment progresses to anisocoria >1 mm (Jacobson, 1998a); (3) no recovery<br />
of function occurs with<strong>in</strong> 12 weeks; or (4) signs of aberrant regeneration develop<br />
(Jacobson, 1999) (class IV, level U).<br />
Pupil <strong>in</strong>volvement is not diagnostic of aneurysmal compression, and up to 38% of<br />
presumed ischemic TNPs <strong>in</strong>volve the pupil (Jacobson, 1998a). Thus, a certa<strong>in</strong> number of<br />
negative cerebral angiograms would be expected <strong>in</strong> the evaluation of pupil <strong>in</strong>volved<br />
TNP. The 1 to 2% risk of catheter angiography, however, must be considered <strong>in</strong> the<br />
decision for angiography. MRI and MRA are especially warranted for superior division<br />
TNP. Myasthenia gravis may rarely mimic a superior division TNP, so a Tensilon test<br />
should be performed <strong>in</strong> these cases. If a patient with a partial TNP has signs of<br />
men<strong>in</strong>geal irritation, other cranial nerve palsies, or signs of more diffuse men<strong>in</strong>geal<br />
<strong>in</strong>volvement (e.g., radiculopathies), then a sp<strong>in</strong>al tap to <strong>in</strong>vestigate <strong>in</strong>fectious, <strong>in</strong>flammatory,<br />
or neoplastic men<strong>in</strong>gitis should be performed (class IV, level C). In cases of<br />
presumed or suspected subarachnoid hemorrhage, CT may be the preferred <strong>in</strong>itial<br />
imag<strong>in</strong>g study followed by cerebral angiography.
Does the Patient Have an Isolated Acquired<br />
TNP with Subnormal Pupillary Sph<strong>in</strong>cter<br />
Dysfunction and Partial or Complete<br />
Extraocular Muscle Palsies (Type 4C TNP)?<br />
Third Nerve Palsies 267<br />
Patients with a ‘‘relative pupil-spar<strong>in</strong>g’’ TNP should have MRI to rule out the possibility<br />
of a compressive lesion. Such patients should also have a CT scan if a subarachnoid<br />
hemorrhage is suspected and a subsequent cerebral angiogram if MRI is negative<br />
because of the possibility of a cerebral aneurysm. Cullom et al published a small<br />
prospective study of 10 patients with ‘‘relative pupillary-spar<strong>in</strong>g’’ TNP and none of the<br />
patients demonstrated aneurysms (Cullom, 1995). These authors suggested that the<br />
prevalence of aneurysm <strong>in</strong> patients with palsies of this type may be low enough to<br />
preclude rout<strong>in</strong>e angiography <strong>in</strong> this group. This report and subsequent recommendation,<br />
however, was based on an <strong>in</strong>adequate patient sample (class IV, level U). Jacobson<br />
reported 24 patients with relative pupil-spar<strong>in</strong>g TNP and found that 10 had nerve<br />
<strong>in</strong>farction, eight had parasellar tumors, two had <strong>in</strong>tracavernous carotid aneurysms, one<br />
had leptomen<strong>in</strong>geal carc<strong>in</strong>omatosis, one had Tolosa-Hunt syndrome, one had oculomotor<br />
neurilemmoma, and one had primary ocular neuromyotonia (Jacobson, 2001).<br />
Also, others have reported <strong>in</strong>ternal carotid, posterior communicat<strong>in</strong>g, and basilar artery<br />
aneurysms <strong>in</strong> isolated TNP with relative pupillary spar<strong>in</strong>g. Thus, cerebral angiography<br />
may still be warranted if MRI is negative (class IV, level C). Because 10 to 38% of<br />
patients with ischemic TNPs have pupillary dysfunction (Capo, 1992; Jacobson, 1998a),<br />
us<strong>in</strong>g these guidel<strong>in</strong>es there will be a certa<strong>in</strong> percentage of normal angiograms.<br />
In the Jacobson and Trobe study discussed above, <strong>in</strong> patients with the iris sph<strong>in</strong>cter<br />
partially impaired but with the extraocular muscle function totally impaired (relative<br />
pupil-spar<strong>in</strong>g complete TNP) plus patient age greater than or equal to 40 and vascular<br />
risk factors present, the authors recommended MRI followed by MRA if MRI does not<br />
show a nonaneurysmal cause (Jacobson, 1999). Catheter angiography may still be<br />
required <strong>in</strong> these patients (class IV, level U).<br />
In evaluat<strong>in</strong>g these patients, one must be cautious to avoid mistak<strong>in</strong>g ‘‘pseudo’’–<br />
pupil spar<strong>in</strong>g, due to aberrant regeneration (below) or coexistent Horner’s syndrome,<br />
from true relative pupil spar<strong>in</strong>g. In both of these conditions, a compressive lesion is<br />
likely localized <strong>in</strong> the cavernous s<strong>in</strong>us. Thus, pupil-spar<strong>in</strong>g or pseudo–pupil-spar<strong>in</strong>g<br />
TNPs may occur not only with extraaxial ischemic lesions but also <strong>in</strong> <strong>in</strong>traaxial<br />
(midbra<strong>in</strong>) lesions, <strong>in</strong> a small proportion of subarachnoid compressive lesions, and <strong>in</strong><br />
a high proportion of cavernous s<strong>in</strong>us compressive lesions (Naudea, 1983).<br />
Complete external and <strong>in</strong>ternal TNPs occurr<strong>in</strong>g <strong>in</strong> isolation are often due to<br />
compressive lesions or men<strong>in</strong>geal <strong>in</strong>filtration; thus, an MRI scan is <strong>in</strong>itially warranted.<br />
If this study is negative, a cerebral angiogram is necessary to <strong>in</strong>vestigate aneurysm or<br />
dural-cavernous s<strong>in</strong>us fistula. If men<strong>in</strong>geal signs are present, sp<strong>in</strong>al fluid evaluation is<br />
warranted. A CT scan should be performed for suspected subarachnoid hemorrhage. In<br />
patients with totally impaired iris sph<strong>in</strong>cter function and impairment of extraocular<br />
muscle function (‘‘pupil-blown TNP’’), Jacobson and Trobe recommend MRI followed<br />
by catheter angiography if MRI does not disclose a nonaneurysmal cause (Jacobson,<br />
1999). A fully dilated and nonreactive pupil occurs <strong>in</strong> up to 71% of patients with<br />
aneurysmal compression and TNP. <strong>An</strong>eurysms impair the pupil <strong>in</strong> 96% of TNP, and the<br />
rema<strong>in</strong><strong>in</strong>g 4% <strong>in</strong> which the pupil is spared have only partial TNP.
268 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What <strong>Neuro</strong>imag<strong>in</strong>g Procedures Should Be<br />
Considered <strong>in</strong> a Patient with an Isolated TNP?<br />
Lee et al reviewed the literature on MRI=MRA, CT and CT angiogram (CTA), and<br />
catheter angiography <strong>in</strong> the management of the isolated TNP, and proposed the<br />
follow<strong>in</strong>g guidel<strong>in</strong>es (Lee, 2002):<br />
1. Isolated complete or partial <strong>in</strong>ternal dysfunction (pupil dilated) with completely<br />
normal external function of the third nerve and no ptosis: The risk for aneurysm <strong>in</strong><br />
this sett<strong>in</strong>g is m<strong>in</strong>imal and neuroimag<strong>in</strong>g for aneurysm is probably not required. The<br />
papers that were reviewed <strong>in</strong> this manuscript, however, did not explicitly <strong>in</strong>clude or<br />
exclude isolated dilated pupils <strong>in</strong> their complete or <strong>in</strong>complete TNPs. The cl<strong>in</strong>ician<br />
should look for other etiologies for isolated pupil dysfunction (e.g., tonic pupil,<br />
pharmacologic, sph<strong>in</strong>cter damage). This represents a practice guidel<strong>in</strong>e of moderate<br />
certa<strong>in</strong>ty based on class III–IV evidence (level B).<br />
2. Partial external dysfunction TNP without <strong>in</strong>ternal dysfunction: The risk for<br />
aneurysm <strong>in</strong> patients with partial TNP is moderate (up to 30% of cases). Unfortunately,<br />
the risk for an <strong>in</strong>dividual patient is not well def<strong>in</strong>ed because other etiologies<br />
may cause a partial external dysfunction TNP with a normal pupil. For example,<br />
patients who have clear myasthenia gravis do not require additional aneurysm<br />
evaluation. Other nonaneurysmal etiologies <strong>in</strong>clud<strong>in</strong>g neoplastic, demyel<strong>in</strong>at<strong>in</strong>g,<br />
<strong>in</strong>filtrative, and ischemic etiologies may also cause a partial TNP without pupil<br />
<strong>in</strong>volvement and may require neuroimag<strong>in</strong>g. If the TNP is due to aneurysm, the TNP<br />
usually progresses over time to a complete TNP <strong>in</strong>clud<strong>in</strong>g pupil <strong>in</strong>volvement.<br />
Although there may not be <strong>in</strong>ternal dysfunction (pupil <strong>in</strong>volvement) <strong>in</strong> a partial<br />
external dysfunction TNP, the term pupil spar<strong>in</strong>g is probably not appropriate <strong>in</strong> this<br />
sett<strong>in</strong>g. That is, pupil <strong>in</strong>volvement may occur over time <strong>in</strong> patients with partial TNP<br />
due to aneurysm with <strong>in</strong>itially no <strong>in</strong>ternal dysfunction. Absence of pupil <strong>in</strong>volvement<br />
early <strong>in</strong> the course of a partial TNP may be due to <strong>in</strong>complete compression of<br />
the pupil fibers by the aneurysm.<br />
MRI with MRA or CTA <strong>in</strong> the acute sett<strong>in</strong>g is a reasonable screen <strong>in</strong> these cases.<br />
The patient should be followed cl<strong>in</strong>ically for progression or pupil <strong>in</strong>volvement <strong>in</strong> the<br />
first week. If the cranial MRI with MRA or CTA is negative and if the risk of<br />
angiography (e.g., elderly, severe cardiovascular disease, abnormal serum creat<strong>in</strong><strong>in</strong>e)<br />
is high, then observation alone is reasonable and the cl<strong>in</strong>ician should look for<br />
alternative etiologies for a partial external dysfunction TNP (e.g., myasthenia<br />
gravis). The cl<strong>in</strong>ician should still consider catheter angiography <strong>in</strong> these cases if<br />
the risk of aneurysm is higher than the risk of angiography (technically <strong>in</strong>adequate<br />
MRA, progression to complete TNP, pupil <strong>in</strong>volvement). The practice option for<br />
cranial MRI with MRA or CTA alone <strong>in</strong> this sett<strong>in</strong>g is of low certa<strong>in</strong>ty (level C) and is<br />
based on class III–IV evidence.<br />
3. Complete external dysfunction with completely normal <strong>in</strong>ternal function TNP: This<br />
cl<strong>in</strong>ical situation <strong>in</strong>dicates a very low risk for aneurysm, and the vasculopathic<br />
patient may be observed for improvement. The pupil should be reexam<strong>in</strong>ed with<strong>in</strong><br />
the first week. Patients who develop pupil <strong>in</strong>volvement should be evaluated us<strong>in</strong>g<br />
the recommendations outl<strong>in</strong>ed <strong>in</strong> the pupil-<strong>in</strong>volv<strong>in</strong>g TNP sections of this chapter. If<br />
the patient has no vasculopathic risk factors, or if there is no improvement after 4 to<br />
12 weeks, or if signs of aberrant regeneration develop, then cranial MRI with MRA or
Third Nerve Palsies 269<br />
CTA should be performed. This practice guidel<strong>in</strong>e is of moderate certa<strong>in</strong>ty based on<br />
the available evidence (level B). Evaluation for myasthenia gravis should be<br />
considered <strong>in</strong> pa<strong>in</strong>less, nonproptotic, pupil-spared ophthalmoplegia depend<strong>in</strong>g on<br />
the cl<strong>in</strong>ical situation.<br />
4. Partial external dysfunction with partial <strong>in</strong>ternal dysfunction TNP: <strong>An</strong> <strong>in</strong>itial cranial<br />
MRI with MRA (or CTA) is reasonable. If these studies are of excellent quality and<br />
negative, then the cl<strong>in</strong>ician should follow the patient for progression or complete<br />
<strong>in</strong>ternal dysfunction. The risk for aneurysm <strong>in</strong> this sett<strong>in</strong>g, however (even with a<br />
negative MRI=MRA), is uncerta<strong>in</strong>. Cl<strong>in</strong>icians should still consider catheter angiography<br />
if the risk of aneurysm <strong>in</strong> an <strong>in</strong>dividual patient is higher than the risk of<br />
angiography. This practice option is of low to moderate certa<strong>in</strong>ty <strong>in</strong> patients with low<br />
cl<strong>in</strong>ical risk for aneurysm based on class III–IV evidence (level C), and there is some<br />
disagreement among experts (level U).<br />
5. Complete external dysfunction with partial <strong>in</strong>ternal dysfunction TNP: The risk of<br />
aneurysm for complete external dysfunction with partial <strong>in</strong>ternal dysfunction (partial<br />
pupil or ‘‘relative pupil spar<strong>in</strong>g’’) is also unknown but probably lower than that for<br />
partial external dysfunction with or without partial <strong>in</strong>ternal dysfunction. The risk for<br />
aneurysm <strong>in</strong> this sett<strong>in</strong>g (even with a negative MRI=MRA or CTA) is uncerta<strong>in</strong>. The<br />
cl<strong>in</strong>ician should consider catheter angiography if the risk of aneurysm is deemed<br />
higher than risk of angiography. This practice option is of low to moderate certa<strong>in</strong>ty<br />
<strong>in</strong> patients with low cl<strong>in</strong>ical risk for aneurysm based on class III–IV evidence (level<br />
C), and there is significant disagreement among experts (level U).<br />
6. Isolated complete <strong>in</strong>ternal dysfunction with partial or complete external dysfunction<br />
TNP: This cl<strong>in</strong>ical situation has the highest risk for aneurysm (86 to 100% of<br />
aneurysmal TNPs have pupil <strong>in</strong>volvement). MRI with MRA or CTA of the head<br />
should be performed, but even with negative neuroimag<strong>in</strong>g there should be a strong<br />
consideration for catheter angiography. This practice guidel<strong>in</strong>e is of moderate<br />
certa<strong>in</strong>ty based on class III evidence and consensus expert op<strong>in</strong>ion (level B). There<br />
are <strong>in</strong>sufficient data to make a recommendation on whether a catheter angiogram<br />
must be performed <strong>in</strong> these cases (level U).<br />
7. <strong>An</strong>y patient with TNP and signs of subarachnoid hemorrhage (SAH): The presence<br />
of SAH (on unenhanced CT scan or lumbar puncture [LP]) essentially makes the<br />
issue of complete or <strong>in</strong>complete TNP as well as application of the ‘‘rule of the pupil’’<br />
moot. Unfortunately, most of the papers <strong>in</strong> the literature on aneurysm and TNP have<br />
<strong>in</strong>cluded nonneurologically isolated cases <strong>in</strong>clud<strong>in</strong>g SAH. In general, an <strong>in</strong>itial CT<br />
scan (with consideration for an LP) should be performed <strong>in</strong> patients with TNP and<br />
signs of SAH. The cl<strong>in</strong>ical picture of SAH (e.g., severe headache, men<strong>in</strong>gismus,<br />
altered consciousness) can be mimicked by other <strong>in</strong>tracranial etiologies such as<br />
pituitary apoplexy, and most cl<strong>in</strong>icians would consider a CT scan as an <strong>in</strong>itial<br />
neuroimag<strong>in</strong>g study prior to consideration of angiography. Patients with SAH on<br />
CT scan should probably undergo catheter angiography. Patients who cannot<br />
undergo a catheter angiogram (e.g., morbidly obese and unable to be placed on<br />
the angiography table) may have to undergo cranial CT and CTA alone prior to<br />
<strong>in</strong>tervention. In other cases of SAH, special MRI parameters <strong>in</strong>clud<strong>in</strong>g fluid attenuation<br />
<strong>in</strong>version recovery (FLAIR) MRI and MRA may be useful. Catheter angiography<br />
should be strongly considered even if the evaluations for SAH (e.g., CT, LP) are<br />
negative. This practice guidel<strong>in</strong>e is of strong certa<strong>in</strong>ty based on class II–III evidence<br />
and consensus expert op<strong>in</strong>ion (level B).
270 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
8. Patients who cannot undergo MRI or MRA: CT and CTA could be considered<br />
<strong>in</strong> selected cases especially if MRA is not available or <strong>in</strong> cases where MRI is<br />
contra<strong>in</strong>dicated (e.g., obesity, claustrophobia, pacemaker). Although CTA has some<br />
advantages over MRA (especially if the location of the aneurysm is known), the<br />
superior quality of MRI compared to CT <strong>in</strong> evaluat<strong>in</strong>g the entire course of the third<br />
nerve makes the comb<strong>in</strong>ation of MRI=MRA superior to CT=CTA as the screen<strong>in</strong>g<br />
study for TNP. There is <strong>in</strong>sufficient evidence to determ<strong>in</strong>e if a comb<strong>in</strong>ation of MRI<br />
and CTA would be superior to MRI=MRA <strong>in</strong> patients with TNP. At the time of this<br />
writ<strong>in</strong>g, the use of CT=CTA would be considered a practice option <strong>in</strong> the evaluation<br />
of TNP, and the recommendation is of low certa<strong>in</strong>ty based on limited class III<br />
evidence (level C).<br />
Is the TNP Progressive or Unresolved<br />
(Type 5 TNP)?<br />
Patients with TNP that worsens after the acute stage (greater than 2 weeks) or who<br />
develop new neurologic f<strong>in</strong>d<strong>in</strong>gs are considered to have progressive TNP. Patients<br />
without resolution of TNP after 12 to 16 weeks are considered unresolved. These<br />
patients require MRI and MRA and consideration for standard angiography. If signs of<br />
men<strong>in</strong>geal irritation or multiple cranial nerve palsies are present, LP is <strong>in</strong>dicated.<br />
Is the TNP Associated with Signs of Aberrant<br />
Regeneration (Type 6)?<br />
Months to years after the occurrence of a TNP, cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs of aberrant regeneration<br />
of the third nerve may be noted. They <strong>in</strong>clude elevation of the lid on downward gaze<br />
(pseudo–von Graefe phenomenon) or on adduction but lid depression dur<strong>in</strong>g abduction.<br />
Other f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clude limitation of elevation and depression of the eye with<br />
occasional eyeball retraction on attempted vertical gaze, adduction of the eye on<br />
attempted elevation or depression, and suppression of the vertical phase of the<br />
opticok<strong>in</strong>etic response. The pupil may be <strong>in</strong> a miotic or mid-dilated position; it may<br />
be fixed to light but may respond to near (near-light dissociation) or constrict on<br />
adduction or down-gaze. Lagophthalmos, presumably caused by co-contraction of the<br />
levator and superior rectus muscles dur<strong>in</strong>g Bell’s phenomenon, has also been described<br />
(Custer, 2000).<br />
Aberrant regeneration may be seen after TNP due to congenital causes, trauma,<br />
aneurysm, migra<strong>in</strong>e, and syphilis, but is very rarely, if ever, caused by ischemic<br />
neuropathy (Barr, 2000; Custer, 2000). A s<strong>in</strong>gle case of aberrant regeneration has been<br />
described after an ischemic stroke affect<strong>in</strong>g the third nerve fascicle <strong>in</strong> the cerebral<br />
peduncle (Messe, 2001). Misdirection of regenerat<strong>in</strong>g nerve fibers is likely the cause, but<br />
it has been postulated that the syndrome may be due to ephaptic neuron transmission<br />
of impulses or from chromatolysis-<strong>in</strong>duced reorganization of third nerve nuclear<br />
synapses. Ephaptic transmission would expla<strong>in</strong> the transient third nerve misdirection<br />
described with ophthalmoplegic migra<strong>in</strong>e, temporal arteritis, pituitary apoplexy, and<br />
non-Hodgk<strong>in</strong>’s lymphoma (Lee, 1992). Long-stand<strong>in</strong>g lesions, such as men<strong>in</strong>giomas of<br />
the cavernous s<strong>in</strong>us, trigem<strong>in</strong>al neuromas, large aneurysms, and pituitary tumors, may
present as primary aberrant regeneration of the third nerve without a history of<br />
previous TNP (Landau, 1997). Primary aberrant regeneration may rarely occur with<br />
extracavernous lesions, such as neurilemmoma, men<strong>in</strong>gioma, asymmetric mammillary<br />
body, or <strong>in</strong>tradural aneurysm (Varma, 1994). Bilateral primary aberrant regeneration<br />
may also occur with abetalipoprote<strong>in</strong>emia (Bassen-Kornzweig syndrome) (Cohen,<br />
1985). On rare occasions, the pseudo–von Graefe phenomenon may develop contralateral<br />
to a regenerat<strong>in</strong>g paretic third nerve (Guy, 1989b).<br />
All patients with nontraumatic TNP with aberrant regeneration (type 5) require MRI<br />
and MRA (and possibly angiography) to <strong>in</strong>vestigate the possibility of a compressive<br />
lesion. This is especially true if signs of aberrance develop <strong>in</strong> a patient with presumed<br />
‘‘ischemic’’ TNP or <strong>in</strong> patients with primary aberrant regeneration.<br />
Retrospective Review of TNP<br />
Third Nerve Palsies 271<br />
A previous retrospective study reviewed all patients with the diagnosis of TNP at a<br />
s<strong>in</strong>gle tertiary care referral center (Baylor College of Medic<strong>in</strong>e) from May 1992 to May<br />
1996 (Lee, 1999). N<strong>in</strong>ety-one patient records were reviewed. A complete (i.e., <strong>in</strong>volvement<br />
of all the somatic branches of the third cranial nerve) TNP was present <strong>in</strong> 79 cases<br />
(87%) and a partial TNP was present <strong>in</strong> 12 cases (13%). The pupil was completely<br />
un<strong>in</strong>volved (i.e., pupil-spared) <strong>in</strong> 49 cases (54%), <strong>in</strong>volved to some degree <strong>in</strong> 40 cases<br />
(44%), and <strong>in</strong>adequately documented <strong>in</strong> two cases (2%). The etiology of the TNP was<br />
presumed to be vasculopathic or ischemic <strong>in</strong> 16 cases (18%), <strong>in</strong>tracranial tumor <strong>in</strong> 15<br />
cases (16%), trauma <strong>in</strong> 14 cases (15%), congenital <strong>in</strong> n<strong>in</strong>e cases (10%), aneurysm <strong>in</strong> eight<br />
cases (9%), postsurgical or iatrogenic <strong>in</strong> eight cases (9%), cerebrovascular accident <strong>in</strong><br />
seven cases (8%), and miscellaneous or idiopathic <strong>in</strong> 14 cases (15%).<br />
Of the 91 cases, the TNP was not isolated <strong>in</strong> 38 (42%) and isolated <strong>in</strong> 53 cases (58%).<br />
Of the 38 nonisolated TNP (type 1), 35 (92%) underwent neuroimag<strong>in</strong>g. Two of the<br />
patients who were not imaged were referred for strabismus surgery for congenital TNP.<br />
They were presumably imaged elsewhere, although this was not documented <strong>in</strong> the<br />
record. The other patient went directly to angiogram and was diagnosed with an<br />
angiogram. Two of these 38 patients had CT scans that would not have been performed<br />
if the imag<strong>in</strong>g guide had been strictly followed, as neither patient had signs of<br />
subarachnoid hemorrhage or trauma, and both subsequently underwent appropriate<br />
MRI. No additional <strong>in</strong>formation was afforded by the CT scans.<br />
Of the 53 cases of isolated TNP, 23 were over the age of 55 years; eight of these 23<br />
(35%) underwent erythrocyte sedimentation rate test<strong>in</strong>g, which was normal. None of<br />
the patients <strong>in</strong> our series were diagnosed with giant cell arteritis. Thirty-two cases had<br />
isolated, pupil-spar<strong>in</strong>g ophthalmoplegia, and six of these patients underwent test<strong>in</strong>g<br />
with edrophonium chloride (Tensilon), none of which were positive. One of the patients<br />
<strong>in</strong> our series was diagnosed with myasthenia gravis by acetylchol<strong>in</strong>e receptor antibody<br />
test<strong>in</strong>g. Thirty-two patients had isolated, pupil-spared TNP. Twenty-five of these cases<br />
were complete TNP (type 4A), and seven were partial (type 4B). Of the 32 patients, 15<br />
cases had no known vasculopathic risk factors, and 17 cases (53%) had known<br />
vasculopathic risk factors. Of the 15 cases without a known vasculopathic risk factor,<br />
n<strong>in</strong>e underwent neuroimag<strong>in</strong>g. Five of these 15 cases were congenital TNP (type 3) and<br />
did not undergo neuroimag<strong>in</strong>g. One patient was sent for strabismus surgery for an<br />
idiopathic TNP, and there was no documentation <strong>in</strong> the record of previous imag<strong>in</strong>g
272 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
studies. One patient <strong>in</strong> this group underwent a CT scan that would not have been<br />
performed accord<strong>in</strong>g to the imag<strong>in</strong>g guide for the reasons mentioned above. This CT<br />
scan did not reveal an etiology for the TNP; however, a subsequent MRI scan showed a<br />
cavernous s<strong>in</strong>us tumor. Of the isolated pupil-spared TNPs that were presumed to be<br />
vasculopathic <strong>in</strong> nature, all demonstrated improvement or resolution of the TNP over<br />
time and none of these patients developed any new neurologic disease. Of these 17<br />
cases, 12 MRI scans and six CT scans were performed on these patients. Of the MRI<br />
scans, 10 would not have been performed accord<strong>in</strong>g to the imag<strong>in</strong>g guide. Six of these<br />
were vasculopathic, one patient had myasthenia gravis diagnosed by antiacetylchol<strong>in</strong>e<br />
receptor antibodies, and one patient had trauma and had undergone CT scan, LP, and<br />
cerebral angiogram. Two MRI scans actually revealed small midbra<strong>in</strong> strokes; however,<br />
this did not affect treatment and both patients showed spontaneous recovery. Of the CT<br />
scans, four would not have been performed, three were performed <strong>in</strong> vasculopathic<br />
patients, and one CT scan disclosed a tumor that would have undergone an <strong>in</strong>itial MRI<br />
scan accord<strong>in</strong>g to the imag<strong>in</strong>g guide.<br />
Twenty-one patients had isolated pupil-<strong>in</strong>volved TNP (type 4C). Of these 21 cases, all<br />
underwent neuroimag<strong>in</strong>g and 13 of 21 had an identifiable <strong>in</strong>tracranial etiology for the<br />
pupil-<strong>in</strong>volved TNP. Eight patients required further studies, of which six underwent<br />
standard cerebral angiography and two underwent MR angiography. Three of these<br />
eight patients were found to harbor an <strong>in</strong>tracranial aneurysm (posterior communicat<strong>in</strong>g<br />
artery). In this group, three MRI scans would not have been performed because each<br />
patient had signs of SAH and each underwent CT scan followed by angiogram. One CT<br />
scan would not have been performed because the patient had no signs of SAH and<br />
would have undergone an <strong>in</strong>itial MRI scan rather than a CT scan.<br />
In our series, patients with a nonisolated TNP thus had a significant chance of<br />
harbor<strong>in</strong>g an <strong>in</strong>tracranial lesion. Sixty percent of 38 patients had <strong>in</strong>tracranial pathology<br />
<strong>in</strong>clud<strong>in</strong>g tumor (48%), aneurysm (22%), stroke (16%), carotid cavernous fistula (4%),<br />
tuberculoma <strong>in</strong> the cavernous s<strong>in</strong>us (4%), and Tolosa-Hunt syndrome (4%).<br />
References<br />
Abdollah A, Francis GS. (1990). Intraaxial divisional oculomotor nerve paresis suggests <strong>in</strong>traaxial fascicular<br />
organization. <strong>An</strong>n <strong>Neuro</strong>l 28:589–590.<br />
Acierno MD, Trobe JD, Cornblath WT, Gebarski SS. (1995). Pa<strong>in</strong>ful oculomotor palsy caused by posterior-dra<strong>in</strong><strong>in</strong>g<br />
dural carotid cavernous fistulas. Arch Ophthalmol 113:1045–1049.<br />
Alderson LM, Noonan PT, Sup Choi I, Henson JW. (1996). Regional subacute cranial neuropathies follow<strong>in</strong>g<br />
<strong>in</strong>ternal carotid cisplat<strong>in</strong> <strong>in</strong>fusion. <strong>Neuro</strong>logy 47:1088–1090.<br />
Al-Yamany M, al-Shayji A, Bernste<strong>in</strong> M. (1999). Isolated oculomotor nerve palsy: an unusual presentation of<br />
glioblastoma multiforme. Case report and review of the literature. J <strong>Neuro</strong>oncol 41:77–80.<br />
<strong>An</strong>dreo LK, Gardner TA, Enzenauer RW. (1994). Third nerve palsy <strong>in</strong> an AIDS patient. Presented at the North<br />
American <strong>Neuro</strong>-Ophthalmology Society meet<strong>in</strong>g, Durango, Colorado, February 27–March 3.<br />
Arroyo JG, Horton JC. (1995). Acute, pa<strong>in</strong>ful, pupil-<strong>in</strong>volv<strong>in</strong>g third nerve palsy <strong>in</strong> chronic <strong>in</strong>flammatory<br />
demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy. <strong>Neuro</strong>logy 45:846–847.<br />
Ashw<strong>in</strong> PT, Mahmood S, Pollock WS. (2001). Sphenoid s<strong>in</strong>us mucocele mimick<strong>in</strong>g aneurysmal oculomotor nerve<br />
palsy. Eye 15:108–110.<br />
Balcar LJ, Galetta SL, Bagley LJ, Pakulo SJ. (1996). Localization of traumatic oculomotor nerve palsy to the<br />
midbra<strong>in</strong> exit site by magnetic resonance imag<strong>in</strong>g. Am J Ophthalmol 122:437–439.<br />
Balcar LJ, Galetta SL, Yousem DM, et al. (1997). Pupil-<strong>in</strong>volv<strong>in</strong>g third-nerve palsy and carotid stenosis: rapid<br />
recovery follow<strong>in</strong>g endarterectomy. <strong>An</strong>n <strong>Neuro</strong>l 41:273–276.<br />
Balm M, Hammack J. (1996). Leptomen<strong>in</strong>geal carc<strong>in</strong>omatosis. Present<strong>in</strong>g features and prognostic factors. Arch<br />
<strong>Neuro</strong>l 53:626–632.
Third Nerve Palsies 273<br />
Barbas NR, Hedges TR, Schwenn M. (1995). Isolated oculomotor nerve palsy due to neoplasm <strong>in</strong> <strong>in</strong>fancy.<br />
<strong>Neuro</strong>-ophthalmol 15:157–160.<br />
Barr D, Kupersmith MJ, P<strong>in</strong>to R, Turb<strong>in</strong> R. (1999). Arachnoid cyst of the cavernous s<strong>in</strong>us result<strong>in</strong>g <strong>in</strong> third nerve<br />
palsy. J <strong>Neuro</strong>-ophthalmol 19:249–251.<br />
Barr D, Kupersmith M, Turb<strong>in</strong> R, et al. (2000). Synk<strong>in</strong>esis follow<strong>in</strong>g diabetic third nerve palsy. Arch Ophthalmol<br />
118:132–134.<br />
Bengel D, Huffmann G. (1994). Oculomotor nuclear complex syndrome as a s<strong>in</strong>gle sign of midbra<strong>in</strong> hemorrhage.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 5:279–282.<br />
Berlit P. (1991). Isolated and comb<strong>in</strong>ed pareses of cranial nerves III, IV, and VI. A retrospective study of 412<br />
patients. J <strong>Neuro</strong>l Sci 103:10–15.<br />
Bikhazi NB, Sloan SH. (1998). Superior orbital fissure syndrome caused by <strong>in</strong>dolent Aspergillus sphenoid<br />
s<strong>in</strong>usitis. Otolaryngol Head Neck Surg 118:102–104.<br />
Birchall D, Khangure MS, McAuliffe W. (1999). Resolution of third nerve paresis after endovascular management<br />
of aneurysms of the posterior communicat<strong>in</strong>g artery. AJNR 20:411–413.<br />
Bogousslavsky J, Maeder P, Regli F, Meuli R. (1994). Pure midbra<strong>in</strong> <strong>in</strong>farction: cl<strong>in</strong>ical, MRI, and etiologic patterns.<br />
<strong>Neuro</strong>logy 44:2032–2040.<br />
Bondenson J, Asman P. (1997). Giant cell arteritis present<strong>in</strong>g with oculomotor nerve palsy. Scand J Rheumatol<br />
26:327–328.<br />
Borras JM, Salazaar FG, Grandas F. (1997). Oculomotor palsy and contralateral tremor (Benedikt’s syndrome)<br />
follow<strong>in</strong>g a stereotactic procedure. J <strong>Neuro</strong>l 244:272–274.<br />
Bortolami R, D’Alessandro R, Manni E. (1993). The orig<strong>in</strong> of pa<strong>in</strong> <strong>in</strong> ‘‘ischemic-diabetic’’ third-nerve palsy. Arch<br />
<strong>Neuro</strong>l 50:795.<br />
Branley MG, Wright KW, Borchert MS. (1992). Third nerve palsy due to cerebral artery aneurysm <strong>in</strong> a child. Aust<br />
NZ J Ophthalmol 20:137–140.<br />
Brazis PW. (1991). Subject review: localization of lesions of the oculomotor nerve: recent concepts. Mayo Cl<strong>in</strong> Proc<br />
66:1029–1035.<br />
Brazis PW, Capobianco DJ, Chang F-L F, et al. (1994). Low flow dural arteriovenous shunt: another cause of<br />
‘‘s<strong>in</strong>ister’’ Tolosa-Hunt syndrome. Headache 34:523–525.<br />
Brazis PW, Masdeu JC, Biller J. (2001). Localization <strong>in</strong> <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>logy. 4th ed. Philadelphia, Lipp<strong>in</strong>cott Williams &<br />
Wilk<strong>in</strong>s.<br />
Breen LA, Hopf HC, Farris RK, Gutman L. (1991). Pupil-spar<strong>in</strong>g oculomotor nerve palsy due to a midbra<strong>in</strong><br />
<strong>in</strong>farction. Arch <strong>Neuro</strong>l 48:105–106.<br />
Bryan JS, Hamed LM. (1992). Levator-spar<strong>in</strong>g nuclear oculomotor palsy. <strong>Cl<strong>in</strong>ical</strong> and magnetic resonance imag<strong>in</strong>g<br />
f<strong>in</strong>d<strong>in</strong>gs. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:26–30.<br />
Capo H, Warren F, Kupersmith MJ. (1992). Evolution of oculomotor nerve palsies. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
12:21–25.<br />
Capoferri C, Martor<strong>in</strong>a M, Menga M. (1997). Herpes Zoster ophthalmoplegia <strong>in</strong> two hemodialysis patients. <strong>Neuro</strong><strong>ophthalmology</strong><br />
17:49–51.<br />
Carlow TJ, Johnson JK. (1990). Parasellar tumors: isolated pupil-spar<strong>in</strong>g third nerve palsy. <strong>Neuro</strong>logy 40(suppl<br />
1):309.<br />
Castro O, Johnson LN, Mamourian AC. (1990). Isolated <strong>in</strong>ferior oblique paresis from bra<strong>in</strong>stem <strong>in</strong>farction.<br />
Perspective on oculomotor fascicular organization <strong>in</strong> the ventral midbra<strong>in</strong> tegmentum. Arch <strong>Neuro</strong>l 47:235–<br />
237.<br />
Chang-God<strong>in</strong>ich A, Lee AG, Brazis PW, et al. (1997). Complete ophthalmoplegia after zoster ophthalmicus.<br />
J <strong>Neuro</strong>-<strong>ophthalmology</strong> 17:262–265.<br />
Chee MW, Tan CB, Tjia HT. (1990). Nuclear third nerve palsy and somnolence due to stroke—a case report. <strong>An</strong>n<br />
Acad Med S<strong>in</strong>gapore 19:382–384.<br />
Chotmongkol V, Cha<strong>in</strong>unsamit S. (1999). Superior branch palsy of the oculomotor nerve caused by acute<br />
sphenoid s<strong>in</strong>usitis. J Med Assoc Thai 82:410–413.<br />
Chotmongkol V, Techasuknirun A. (1992). Superior division paresis of the oculomotor nerve casued by<br />
cryptococcal men<strong>in</strong>gitis. J Med Assoc Thai 75:548–550.<br />
Chou TM, Demer JL. (1998). Isolated <strong>in</strong>ferior rectus palsy caused by a metastasis to the oculomotor nucleus.<br />
Am J Ophthalmol 126:737–740.<br />
Cohen DA, Bosley TM, Sav<strong>in</strong>o PJ, et al. (1985). Primary aberrant regeneration of the oculomotor<br />
nerve—occurrence <strong>in</strong> a patient with abetalipoprote<strong>in</strong>emia. Arch <strong>Neuro</strong>l 42:821–823.<br />
Cox TA, Goldberg RA, Rootman J. (1991). Tonic pupil and Czarnecki’s sign follow<strong>in</strong>g third nerve palsy. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 11:55–56.
274 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Cullom ME, Sav<strong>in</strong>o PJ. (1993). Adenocarc<strong>in</strong>oma of the prostate present<strong>in</strong>g as a third nerve palsy. <strong>Neuro</strong>logy<br />
43:2146–2147.<br />
Cullom ME, Sav<strong>in</strong>o PJ, Sergott RC, Bosley TM. (1995). Relative pupillary spar<strong>in</strong>g third nerve palsies. To<br />
angiogram or not? J <strong>Neuro</strong>-ophthalmol 15:136–141.<br />
Cunn<strong>in</strong>gham ET, Good WV. (1994). Inferior branch oculomotor nerve palsy: a case report. J <strong>Neuro</strong>-ophthalmol<br />
14:21–23.<br />
Custer PL. (2000). Lagophthalmos: an unusual manifestation of oculomotor nerve aberrant regeneration.<br />
Ophthalmic Plastic Reconstruct Surg 16:50–51.<br />
Davies GE, Shakir RA. (1994). Giant cell arteritis present<strong>in</strong>g as oculomotor nerve palsy with pupillary dilatation.<br />
Postgrad Med J 70:298–299.<br />
Davis PC, Newman NJ. (1996). Perspective—advances <strong>in</strong> neuroimag<strong>in</strong>g of the visual pathways. Am J Ophthalmol<br />
121:690–705.<br />
Dehaene I, Van Zandijcke M. (1995). Isolated paralysis of the superior division of the ocular motor nerve<br />
mimicked by myasthenia gravis. <strong>Neuro</strong>-<strong>ophthalmology</strong> 5:257–258.<br />
Dichgans M, Dieterich M. (1995). Third nerve palsy with contralateral ocular torsion and b<strong>in</strong>ocular tilt of visual<br />
vertical, <strong>in</strong>dicat<strong>in</strong>g a midbra<strong>in</strong> lesion. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:315–320.<br />
DiMario FJ Jr, Rorke LB. (1992). Transient oculomotor nerve paresis <strong>in</strong> congenital distal basilar artery aneurysm.<br />
Pediatr <strong>Neuro</strong>l 8:303–306.<br />
Donahue SP, Taylor RJ. (1998). Pupil-spar<strong>in</strong>g third nerve palsy associated with sildenafil citrate (Viagra). Am<br />
J Ophthalmol 126:476–477.<br />
Ebner R, Slamovits TL, Friedlamd S, et al. (1995). Visual loss follow<strong>in</strong>g treatment of sphenoid s<strong>in</strong>us cancer. Surv<br />
Ophthalmol 40:62–68.<br />
Egan RA, Thompson CR, MacColl<strong>in</strong> M, Lessell S. (2001). Monocular elevator paresis <strong>in</strong> neurofibromatosis type 2.<br />
<strong>Neuro</strong>logy 56:1222–1224.<br />
Eggenberger ER, Miller NR, Hoffman PN, et al. (1993). Mesencephalic ependymal cyst caus<strong>in</strong>g an <strong>in</strong>ferior division<br />
paresis of the oculomotor nerve: case report. <strong>Neuro</strong>logy 43:2419–2420.<br />
Ehrenpries SJ, Biedl<strong>in</strong>gmaier JF. (1995). Isolated third-nerve palsy associated with frontal s<strong>in</strong>us mucocele. J <strong>Neuro</strong><strong>ophthalmology</strong><br />
15:105.<br />
Ferrante E, Sav<strong>in</strong>o A, Brioschi A, et al. (1998). Transient oculomotor cranial nerve palsy <strong>in</strong> spontaneous<br />
<strong>in</strong>tracranial hypotension. J <strong>Neuro</strong>surg Sci 42:177–179.<br />
Friedman JA, Piepgras DG, Pichelmann MA, et al. (2001). Small cerebral aneurysms present<strong>in</strong>g with symptoms<br />
other than rupture. <strong>Neuro</strong>logy 57:1212–1216.<br />
Galetta SL, Sergott RC, Wells GB, et al. (1992). Spontaneous remission of a third-nerve palsy <strong>in</strong> men<strong>in</strong>geal<br />
lymphoma. <strong>An</strong>n <strong>Neuro</strong>l 32:100–102.<br />
Gauntt CD, Kashii S, Nagata I. (1995). Monocular elevation paresis caused by an oculomotor fascicular lesion.<br />
J <strong>Neuro</strong>-ophthalmol 15:11–14.<br />
Gaymard B, Huynh C, Laffont I. (2000). Unilateral eyelid retraction. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 68:390–392.<br />
Gaymard B, Lafitte C, Gelot A, de Toffol B. (1992). Plus-m<strong>in</strong>us lid syndrome. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
55:846–848.<br />
Gaymard B, Larmande P, de Toffol B, Autret A. (1990). Reversible nuclear oculomotor nerve paralysis. Caused by<br />
a primary mesencephalic hemorrhage. Eur <strong>Neuro</strong>l 30:128–131.<br />
Getenet JC, Vighetto A, Nighoghossian N, Trouillas P. (1994). Isolated bilateral third nerve palsy caused by a<br />
mesencephalic hematoma. <strong>Neuro</strong>logy 44:981–982.<br />
Giomb<strong>in</strong>i S, Ferraresi S, Pluch<strong>in</strong>o F. (1991). Reversal of oculomotor disorders after <strong>in</strong>tracranial aneurysm surgery.<br />
Acta <strong>Neuro</strong>chir 112:19–24.<br />
Go T. (2000). Partial oculomotor nerve palsy associated with elevated anti-galactocerebroside and anti-GM1<br />
antibodies. J Pediatr 137:425–426.<br />
Goldberg RA, Rootman J. (1990a). <strong>Cl<strong>in</strong>ical</strong> characteristics of metastatic orbital tumors. Ophthalmology 47:620–624.<br />
Goldberg RA, Rootman J, Kl<strong>in</strong>e RA. (1990b). Tumors metastatic to the orbit: a chang<strong>in</strong>g picture. Surv Ophthalmol<br />
35:1–24.<br />
Good EF. (1990). Ptosis as the sole manifestation of compression of the oculomotor nerve by an aneurysm of the<br />
posterior communicat<strong>in</strong>g artery. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:59–61.<br />
Good WV, Barkovich AJ, Nickel BL, et al. (1991). Bilateral congenital oculomotor nerve palsy <strong>in</strong> a child with bra<strong>in</strong><br />
anomalies. Am J Ophthalmol 111:555–558.<br />
Gray LG, Galetta SL, Hershey B, et al. (1999). Inferior division third nerve paresis from an orbital dural<br />
arteriovenous malformation. J <strong>Neuro</strong>-Ophthalmol 19:46–48.<br />
Grayeli AB, Redondo A, Salama J, Rey A. (1998). Tuberculoma of the cavernous s<strong>in</strong>us: case report. <strong>Neuro</strong>surgery<br />
42:179–182.
Third Nerve Palsies 275<br />
Green KM, Board T, O’Keefe LJ. (2000). Oculomotor nerve palsy follow<strong>in</strong>g submucosal diathermy to the <strong>in</strong>ferior<br />
turb<strong>in</strong>ates. J Laryngol Otol 114:285–286.<br />
Greenspan BN, Reeves AG. (1990). Transient partial oculomotor nerve paresis with posterior communicat<strong>in</strong>g<br />
aneurysm—a case report. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:56–58.<br />
Griffiths PD, Gholkar A, Sengupta RP. (1994). Oculomotor nerve palsy due to thrombosis of a posterior<br />
communicat<strong>in</strong>g artery aneurysm follow<strong>in</strong>g diagnostic angiography. <strong>Neuro</strong>radiology 36:614–615.<br />
Guar<strong>in</strong>o M, Stracciari A, Cirignotta F, et al. (1995). Neoplastic men<strong>in</strong>gitis present<strong>in</strong>g with ophthalmoplegia, ataxia,<br />
and areflexia (Miller-Fisher syndrome). Arch <strong>Neuro</strong>l 52:443–444.<br />
Guy JR, Day AL. (1989a). Intracranial aneurysms with superior division paresis of the oculomotor nerve.<br />
Ophthalmology 96:1071–1076.<br />
Guy J, Engel HM, Lessner AM. (1989b). Acquired contralateral oculomotor synk<strong>in</strong>esis. Arch <strong>Neuro</strong>l 46:1021–1023.<br />
Guy J, Sav<strong>in</strong>o PJ, Schatz NJ, et al. (1985). Superior division paresis of the oculomotor nerve. Ophthalmology<br />
92:777–784.<br />
Hahn CD, Nicolle DA, Lownie SP, Drake CG. (2000). Giant cavernous carotid artery aneurysms: cl<strong>in</strong>ical<br />
presentation <strong>in</strong> fifty-seven cases. J <strong>Neuro</strong>-Ophthalmol 20:253–258.<br />
Hamed LM. (1991). Associated neurologic and ophthalmologic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> congenital oculomotor nerve palsy.<br />
Ophthalmology 98:708–714.<br />
Hardenack M, Volker A, Schroder JM, et al. (1994). Primary eos<strong>in</strong>ophilic granuloma of the oculomotor nerve.<br />
J <strong>Neuro</strong>surg 81:784–787.<br />
Hart AJ, Allibone J, Casey AT, Thomas DG. (1998). Malignant men<strong>in</strong>gioma of the oculomotor nerve without dural<br />
attachment. Case report and review of the literature. J <strong>Neuro</strong>surg 88:1104–1106.<br />
Hashimoto M, Ohtsuka K. (1998a). Bilateral <strong>in</strong>ternal ophthalmoplegia as a feature of oculomotor fascicular<br />
syndrome disclosed by magnetic resonance imag<strong>in</strong>g. Am J Ophthalmol 125:121–123.<br />
Hashimoto M, Ohtsuka K, Akiba H, Harada K. (1998b). Vascular compression of the oculomotor nerve disclosed<br />
by th<strong>in</strong>-sliced magnetic resonance imag<strong>in</strong>g. Am J Ophthalmol 125:881–882.<br />
Haughton AJ, Chalkiadis GA. (1999). Un<strong>in</strong>tentional paediatric subdural catheter with oculomotor and abducens<br />
nerve palsies. Paediatr <strong>An</strong>aesth 9:543–548.<br />
Hedges TR, Hirsh LF. (1993). Bilateral third nerve palsy from ‘‘m<strong>in</strong>or’’ head trauma. <strong>Neuro</strong>-<strong>ophthalmology</strong> 13:219.<br />
Herman M, Bobek-Billewicz B, Bullo B, Herman A. (1999). Wegener’s granulomatosis with unusual cavernous<br />
s<strong>in</strong>us and sella turcica extension. Eur Radiol 9:1859–1861.<br />
Holland NR, Deibert E. (1998). CNS Act<strong>in</strong>omycosis present<strong>in</strong>g with bilateral cavernous s<strong>in</strong>us syndrome. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 64:4.<br />
Holl<strong>in</strong>ger P, Sturzenegger M. (1999). Pa<strong>in</strong>ful oculomotor nerve palsy—a present<strong>in</strong>g sign of <strong>in</strong>ternal carotid artery<br />
stenosis. Cerebrovasc Dis 9:178–181.<br />
Hopf HC, Gutmann L. (1990). Diabetic third nerve palsy: evidence for a mesencephalic lesion. <strong>Neuro</strong>logy<br />
40:1041–1045.<br />
Horikoshi T, Nukui H, Yagishita T, et al. (1999). Oculomotor nerve palsy after surgery for upper basilar artery<br />
aneurysms. <strong>Neuro</strong>surgery 44:705–710.<br />
Hriso E, Miller A, Masdeu JC. (1990). Monocular elevation weakness and ptosis. <strong>Neuro</strong>logy 47(suppl 1):309.<br />
Ide C, De Coene B, Gilliard C, et al. (1997). Hemorrhagic arachnoid cyst with third nerve paresis: CT and MR<br />
f<strong>in</strong>d<strong>in</strong>gs. AJNR 18:1407–1410.<br />
Ikeda K, Tamura M, Iwasaki Y, K<strong>in</strong>oshita M. (2001). Relative pupil-spar<strong>in</strong>g third nerve palsy: etiology and cl<strong>in</strong>ical<br />
variables predictive of a mass. <strong>Neuro</strong>logy 57:1741–1742.<br />
Ing EB, Purv<strong>in</strong> V. (1997). Progressive visual loss and motility deficit. Surv Ophthalmol 41:488–492.<br />
Ing E, Sullivan TJ, Clarke MP, Buncic JR. (1992). Oculomotor nerve palsies <strong>in</strong> children. J Pediatr Ophthalmol<br />
Strabismus 29:331–336.<br />
Ishibashi A, Sueyoshi K, You M, Yokokura Y. (1998). MR f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> isolated oculomotor nerve palsy associated<br />
with <strong>in</strong>fectious mononucleosis caused by Epste<strong>in</strong>-Barr virus <strong>in</strong>fection. J Comput Assist Tomogr 22:995–997.<br />
Ishikawa H, Satoh H, Fujiwara M, et al. (1997). Oculomotor nerve palsy caused by lung cancer metastasis. Int Med<br />
36:301–303.<br />
Jacobson DM. (1998a). Pupil <strong>in</strong>volvement <strong>in</strong> patients with diabetes-associated oculomotor nerve palsy. Arch<br />
Ophthalmol 116:723–727.<br />
Jacobson DM. (1998b). Proptosis with acute oculomotor and abducens nerve palsies. J <strong>Neuro</strong>-Ophthalmol<br />
18:289–291.<br />
Jacobson DM. (2001). Relative pupil-spar<strong>in</strong>g third nerve palsy: etiology and cl<strong>in</strong>ical variables predictive of a mass.<br />
<strong>Neuro</strong>logy 56:797–798.<br />
Jacobson DM, Broste SK. (1995). Early progression of ophthalmoplegia <strong>in</strong> patients with ischemic oculomotor<br />
nerve palsies. Arch Ophthalmol 113:1535–1537.
276 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Jacobson DM, McCanna TD, Layde PM. (1994). Risk factors for ischemic ocular motor nerve palsies. Arch<br />
Ophthalmol 112:961–966.<br />
Jacobson DM, Trobe JD. (1999). The emerg<strong>in</strong>g role of magnetic resonance angiography <strong>in</strong> the management of<br />
patients with third cranial nerve palsy. Am J Ophthalmol 128:94–96.<br />
Kadota T, Miyawaki Y, Nakagawa H, et al. (1993). MR imag<strong>in</strong>g of oculomotor nerve neurilemmoma. Magn Reson<br />
Imag<strong>in</strong>g 11:1071–1075.<br />
Kajiya Y, Nakajo M, Kajiya Y, Miyaji N. (1995). Oculomotor nerve <strong>in</strong>vasion by lymphoma demonstrated by MRI. J<br />
Comput Assist Tomogr 19:502–504.<br />
Kardon RH, Traynelis VC, Biller J. (1991). Inferior division paresis of the oculomotor nerve caused by basilar<br />
artery aneurysm. Cerebrovasc Dis 1:171.<br />
Kasner SE, Galetta SL, Vaughn DJ. (1996). Cavernous s<strong>in</strong>us syndrome <strong>in</strong> Hodgk<strong>in</strong>’s disease. J <strong>Neuro</strong>-Ophthalmol<br />
16:204–207.<br />
Kaufman DI. (1994). Recent advances <strong>in</strong> neuro-imag<strong>in</strong>g and the impact on neuro-<strong>ophthalmology</strong>. Curr Op<strong>in</strong><br />
Ophthalmol 5:52–62.<br />
Kawasaki A. (1999). Oculomotor nerve schwannoma associated with ophthalmoplegic migra<strong>in</strong>e. Am J Ophthalmol<br />
128:658–660.<br />
Kawase T, Sone A, Igarashi Y, et al. (1996). Developmental patterns and characteristic symptoms of petroclival<br />
men<strong>in</strong>giomas. <strong>Neuro</strong>l Med Chir 36:1–6.<br />
Kaye-Wilson LG, Gibson R, Bell JE, Steers AJW. (1994). Oculomotor nerve neur<strong>in</strong>oma, early detection by magnetic<br />
resonance imag<strong>in</strong>g. <strong>Neuro</strong>-ophthalmol 14:37–41.<br />
Keane JR. (1993). Intermittent third nerve palsy with cryptococcal men<strong>in</strong>gitis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:124–126.<br />
Keane JR. (1996). Cavernous s<strong>in</strong>us syndrome. <strong>An</strong>alysis of 151 cases. Arch <strong>Neuro</strong>l 53:967–971.<br />
Keane JR, Ahmadi J. (1998). Most diabetic third nerve palsies are peripheral. <strong>Neuro</strong>logy 51:1510.<br />
Kim JS, Kang JK, Lee SA, Lee MC. (1993). Isolated or predom<strong>in</strong>ant ocular motor nerve palsy as a manifestation of<br />
bra<strong>in</strong> stem stroke. Stroke 24:581–586.<br />
Kodsi SR, Younge BR. (1992). Acquired oculomotor, trochlear, and abducent cranial nerve palsies <strong>in</strong> pediatric<br />
patients. Am J Ophthalmol 114:568–574.<br />
Koennecke H, Seyfert S. (1998). Mydriatic pupil as the present<strong>in</strong>g sign of common carotid artery dissection. Stroke<br />
29:2635–2655.<br />
Kondoh K, Ohtsuka K, Hashimoto M, Nakamura Y. (1998). Inferior branch palsy of the oculomotor nerve caused<br />
by EB virus <strong>in</strong>fection. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:36.<br />
Ksiazek SM, Repka MX, Maguire A, et al. (1989). Divisional oculomotor nerve paresis caused by <strong>in</strong>tr<strong>in</strong>sic<br />
bra<strong>in</strong>stem disease. <strong>An</strong>n <strong>Neuro</strong>l 26:714–718.<br />
Ksiazek SM, Slamovits TL, Rosen CE, et al. (1994). Fascicular arrangement <strong>in</strong> partial oculomotor paresis. Am<br />
J Ophthalmol 118:97–103.<br />
Kudo T. (1990). <strong>An</strong> operative complication <strong>in</strong> a patient with a true posterior communicat<strong>in</strong>g artery aneurysm: case<br />
report and review of the literature. <strong>Neuro</strong>surgery 27:650–653.<br />
Kurokawa Y, Uede T, Honda O, Honmou O. (1992). Successful removal of <strong>in</strong>tracavernous neur<strong>in</strong>oma orig<strong>in</strong>at<strong>in</strong>g<br />
from the oculomotor nerve—case report. <strong>Neuro</strong>l Med Chir 32:225–228.<br />
Landau K, Lepore FE. (1997). Discover<strong>in</strong>g a dys-cover<strong>in</strong>g lid. Surv Ophthalmol 42:87–91.<br />
Landolfi JC, Thaler HT, De <strong>An</strong>gelis LM. (1998). Adult bra<strong>in</strong>stem gliomas. <strong>Neuro</strong>logy 51:1136–1139.<br />
Lanz<strong>in</strong>o G, <strong>An</strong>dreoli A, Tognetti F, et al. (1993). Orbital pa<strong>in</strong> and unruptured carotid-posterior communicat<strong>in</strong>g<br />
artery aneurysms: the role of sensory fibers of the third cranial nerve. Acta <strong>Neuro</strong>chir 120:7–11.<br />
Lee AG. (1996). Third nerve palsy due to a carotid cavernous fistula without external eye signs. <strong>Neuro</strong><strong>ophthalmology</strong><br />
16:183–187.<br />
Lee AG, Hayman LA, Brazis PW. (2002). The evaluation of isolated third nerve palsy revisited: an update on the<br />
evolv<strong>in</strong>g role of magnetic resonance, computed tomography, and catheter angiography. Surv Ophthalmol<br />
47:137–157.<br />
Lee AG, Onan H, Brazis PW, Prager TC. (1999). <strong>An</strong> imag<strong>in</strong>g guide to the evaluation of third cranial nerve palsies.<br />
Strabismus 7:153–168.<br />
Lee AG, Tang RA. (2000a). Third nerve palsy as the present<strong>in</strong>g manifestation of esthesioneuroblastoma. J <strong>Neuro</strong>-<br />
Ophthalmol 20:20–21.<br />
Lee AG, Tang RA, Wong GG, et al. (2000b). Isolated <strong>in</strong>ferior rectus muscle palsy result<strong>in</strong>g from a nuclear third<br />
nerve lesion as the <strong>in</strong>itial manifestation of multiple sclerosis. J <strong>Neuro</strong>-Ophthalmol 20:246–247.<br />
Lee CC, Cho AS, Carter WA. (2000c). Emergency department presentation of pituitary apoplexy. Am J Emerg Med<br />
18:328–331.<br />
Lee SH, Yeow YK, Tan CB, Tjia H. (1992). Transient oculomotor nerve synk<strong>in</strong>esis <strong>in</strong> non-Hodgk<strong>in</strong>’s lymphoma.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:203–206.
Third Nerve Palsies 277<br />
Lepore FE. (1995). Disorders of ocular motility follow<strong>in</strong>g head trauma. Arch <strong>Neuro</strong>l 52:924–926.<br />
Liu GT, Carrazana EJ, Charness ME. (1991). Unilateral oculomotor palsy and bilateral ptosis from paramedian<br />
midbra<strong>in</strong> <strong>in</strong>farction. Arch <strong>Neuro</strong>l 48:983–986.<br />
Liu GT, Crenner CW, Logigian EL, et al. (1992). Midbra<strong>in</strong> syndromes of Benedikt, Claude, and Nothnagel: sett<strong>in</strong>g<br />
the record straight. <strong>Neuro</strong>logy 42:1820–1822.<br />
Liu GT, Kay MD, Byrne GE, et al. (1993). Ophthalmoparesis due to Burkitt’s lymphoma follow<strong>in</strong>g cardiac<br />
transplantation. <strong>Neuro</strong>logy 43:2147–2149.<br />
Lopes DK, Mericle RA, Wakhloo AK, et al. (1998). Cavernous s<strong>in</strong>us syndrome dur<strong>in</strong>g balloon test occlusion of the<br />
cervical <strong>in</strong>ternal carotid artery. Report of two cases. J <strong>Neuro</strong>surg 89:667–670.<br />
Lustbader JM, Miller NR. (1988). Pa<strong>in</strong>less, pupil-spar<strong>in</strong>g but otherwise complete oculomotor paresis caused by<br />
basilar artery aneurysm. Arch Ophthalmol 106:583–584.<br />
Manabe Y, Kurokawa K, Kashihara K, Abe K. (2000). Isolated oculomotor nerve palsy <strong>in</strong> lymphoma. <strong>Neuro</strong>l Res<br />
22:347–348.<br />
Mansour AM, Bailey BJ. (1997). Ocular f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Ramsay Hunt syndrome. J <strong>Neuro</strong>-Ophthalmol 17:199–201.<br />
Mark AS, Blake P, Atlas SW, et al. (1992). Gd-DTPA enhancement of the cisternal portion of the oculomotor nerve<br />
on MR imag<strong>in</strong>g. AJNR 13:1463–1470.<br />
Mark AS, Casselman J, Brown D, et al. (1998). Ophthalmoplegic migra<strong>in</strong>e: reversible enhancement and thicken<strong>in</strong>g<br />
of the cisternal segment of the oculomotor nerve on contrast-enhanced MR images. AJNR 19:1887–1891.<br />
Mart<strong>in</strong> TJ, Corbett JJ, Babikian PV, et al. (1996). Bilateral ptosis due to mesencephalic lesions with relative<br />
preservation of ocular motility. J <strong>Neuro</strong>-Ophthalmol 16:258–263.<br />
McFadzean RM, Teasdale EM. (1998). Computerized tomography angiography <strong>in</strong> isolated third nerve palsies.<br />
J <strong>Neuro</strong>surg 88:679–684, 1998.<br />
Mehta VS, S<strong>in</strong>gh RV, Misra NK, Choudry C. (1990). Schwannoma of the oculomotor nerve. Br J <strong>Neuro</strong>surg 4:69–72.<br />
Messe SR, Sh<strong>in</strong> RK, Liu GT, et al. (2001). Oculomotor synk<strong>in</strong>esis follow<strong>in</strong>g a midbra<strong>in</strong> stroke. <strong>Neuro</strong>logy<br />
57:1106–1107.<br />
Migita DS, Devereaux MW, Tomsak RL (1998). Coca<strong>in</strong>e and pupillary-spar<strong>in</strong>g oculomotor nerve paresis.<br />
<strong>Neuro</strong>logy 49:1466–1467.<br />
Miller NR. (1999). Unequal pupils can be seen <strong>in</strong> diabetic 3rd nerve palsy. Evidence-Based Eye Care 1:40–41.<br />
Miyachi S, Negoro M, Handa T, Sugita K. (1993). Dural carotid cavernous s<strong>in</strong>us fistula present<strong>in</strong>g as isolated<br />
oculomotor nerve palsy. Surg <strong>Neuro</strong>l 39:105–109.<br />
Miyao S, Takano A, Teramoto J, et al. (1993). Oculomotor nerve palsy due to <strong>in</strong>traneural hemorrhage <strong>in</strong> idiopathic<br />
thrombocytopenic purpura: a case report. Eur <strong>Neuro</strong>l 33:20–22.<br />
Mokri B, Silbert PL, Schiev<strong>in</strong>k WI, Piepgras DG. (1996). Cranial nerve palsy <strong>in</strong> spontaneous dissection of the<br />
extracranial <strong>in</strong>ternal carotid artery. <strong>Neuro</strong>logy 46:356–359.<br />
Moster ML, Scimeca GH, Romayananda N, et al. (1996). Mandibular ameloblastoma metastatic to the cavernous<br />
s<strong>in</strong>us. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:47–50.<br />
Mudgil AV, Repka MX. (1999). Ophthalmologic outcome after third cranial nerve palsy or paresis <strong>in</strong> childhood. J<br />
Am Assoc Pediatr Ophthalmol Strabismus 3:2–8.1<br />
Murakami M, Kitano I, Hitoshi Y, Ushio Y. (1994). Isolated oculomotor nerve palsy follow<strong>in</strong>g midbra<strong>in</strong> <strong>in</strong>farction.<br />
Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg 96:188–190.<br />
Nagaoka U, Kato T, Kurita K, et al. (1996). Cranial nerve enhancement on three-dimensional MRI <strong>in</strong> Miller Fisher<br />
syndrome. <strong>Neuro</strong>logy 47:1601–1502.<br />
Naghmi R, Subuhi R. (1990). Diabetic oculomotor mononeuropathy: <strong>in</strong>volvement of pupillomotor fibres with<br />
slow resolution. Horm Metab Res 22:38–40.<br />
Nakagawa H, Nakajima S, Nakajima Y, et al. (1991). Bilateral oculomotor nerve palsies due to posterior cerebral<br />
arterial compression relieved by microvascular decompression—case report. <strong>Neuro</strong>l Med Chir 31:45–48.<br />
Nakao H, Ohtsuka K, Hashimoto M. (1998). Nuclear oculomotor nerve palsy caused by metastatic tumor. <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:36.<br />
Naudea SE, Trobe JD. (1983). Pupil spar<strong>in</strong>g <strong>in</strong> oculomotor palsy: a brief review. <strong>An</strong>n <strong>Neuro</strong>l 13:143–148.<br />
Newman NJ, Lessell S. (1990). Isolated pupil-spar<strong>in</strong>g third-nerve palsy as the present<strong>in</strong>g sign of multiple sclerosis.<br />
Arch <strong>Neuro</strong>l 47:817–818.<br />
Newman NJ, Slamovits TL, Friedland S, Wilson WB. (1995). <strong>Neuro</strong>-ophthalmic manifestations of men<strong>in</strong>gocerebral<br />
<strong>in</strong>flammation from the limited form of Wegener’s granulomatosis. Am J Ophthalmol 120:613–621.<br />
Norman AA, Farris BK, Siatkowski RM. (2001). <strong>Neuro</strong>ma as a cause of oculomotor palsy <strong>in</strong> <strong>in</strong>fancy and early<br />
childhood. J Am Assoc Pediatr Ophthalmol Strabismus 5:9–12.<br />
North KN, <strong>An</strong>tony JH, Johnston IH. (1993). Dermoid of cavernous s<strong>in</strong>us result<strong>in</strong>g <strong>in</strong> isolated oculomotor nerve<br />
palsy. Pediatr <strong>Neuro</strong>l 9:221–223.
278 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Ogilvy CS, Pakzaban P, Lee JM. (1993). Oculomotor nerve cavernous angioma <strong>in</strong> a patient with Roberts<br />
syndrome. Surg <strong>Neuro</strong>l 40:39–42.<br />
O’Halloran HS, Lee WB, Baker RS, Pearson PA. (1999). Ophthalmoplegic migra<strong>in</strong>e with unusual features.<br />
Headache 39:670–673.<br />
O’Hara MA, <strong>An</strong>derson RT, Brown D. (2001). Magnetic resonance imag<strong>in</strong>g <strong>in</strong> ophthalmoplegic migra<strong>in</strong>e of<br />
children. J Am Assoc Pediatr Ophthalmol Strabismus 5:307–310.<br />
Ohtsuka K, Hashimoto M, Nakamura Y. (1997). Enhanced magnetic resonance imag<strong>in</strong>g <strong>in</strong> a patient with acute<br />
paralysis of the <strong>in</strong>ferior division of the oculomotor nerve. Am J Ophthalmol 124:406–409.<br />
Oishi M, Mochizuki Y. (1997). Ipsilateral oculomotor nerve palsy and contralateral downbeat nystagmus: a<br />
syndrome caused by unilateral paramedian thalamopeduncular <strong>in</strong>farction. J <strong>Neuro</strong>l 244:132–133.<br />
Okuchi K, Fujioka M, Maeda Y, et al. (1999). Bilateral chronic subdural hematomas result<strong>in</strong>g <strong>in</strong> unilateral<br />
oculomotor nerve paresis and bra<strong>in</strong> stem symptoms after operation—case report. <strong>Neuro</strong>l Med Chir<br />
39:367–371.<br />
Onozu H, Yamamoto S, Takou K, Hasyasaka S. (1998). Blepharoptosis <strong>in</strong> association with ipsilateral adduction<br />
and elevation palsy. A form of fascicular oculomotor palsy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:145–150.<br />
Pacifici L, Passarelli F, Papa G, et al. (1993). Acute third cranial nerve ophthalmoplegia: possible pathogenesis<br />
from alpha-II-<strong>in</strong>terferon treatment. Ital J <strong>Neuro</strong>l Sci 14:579–580.<br />
Papke K, Masur H, Mart<strong>in</strong>ez-Rubio A, et al. (1993). Complete bilateral oculomotor palsy: the only cl<strong>in</strong>ical sign of<br />
subarachnoid hemorrhage <strong>in</strong> leukemia. Acta <strong>Neuro</strong>l Scand 88:153–156.<br />
Park-Matsumoto YC, Tazawa T. (1997). Internal carotid-posterior communicat<strong>in</strong>g artery aneurysm manifest<strong>in</strong>g as<br />
an unusual ocular motor paresis after m<strong>in</strong>or head trauma—case report. <strong>Neuro</strong>l Med Chir 37:181–183.<br />
Parmeggiani A, Posar A, Leonardi M, et al. (1992). <strong>Neuro</strong>logic impairment <strong>in</strong> congenital bilateral ptosis with<br />
ophthalmoplegia. Bra<strong>in</strong> Dev 14:107–109.<br />
Patel CK, Taylor DS, Russell-Eggitt IM, et al. (1993). Congenital third nerve palsy associated with mid-trimester<br />
amniocentesis. Br J Ophthalmol 77:530–533.<br />
Perez Sempere A, Mart<strong>in</strong>ez Menendez B, Cabeza Alvarez C, Calandre Hoenigsfeld L. (1991). Isolated oculomotor<br />
nerve palsy due to dural cavernous s<strong>in</strong>us fistula. Eur <strong>Neuro</strong>l 31:186–187.<br />
Phookan G, Cameron M. (1994). Bilateral chronic subdural haematoma: an unusual presentation with isolated<br />
oculomotor nerve palsy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:1146.<br />
Polito E, Leccisotti A. (1996). Pa<strong>in</strong>ful ophthalmoplegia caused by superior ophthalmic ve<strong>in</strong> thrombosis. <strong>Neuro</strong><strong>ophthalmology</strong><br />
16:189–192.<br />
Prats JM, Mateos B, Garaizer C. (1999). Resolution of MRI abnormalities of the oculomotor nerve <strong>in</strong> childhood<br />
ophthalmoplegic migra<strong>in</strong>e. Cephalgia 19:655–659.<br />
Pratt DV, Orengo-Nania S, Horowitz BL, Oram O. (1995). Magnetic resonance f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a patient with nuclear<br />
oculomotor palsy. Arch Ophthalmol 113:141.<br />
Ranganadham P, D<strong>in</strong>akar I, Mohandas S, S<strong>in</strong>gh AK. (1992). A rare presentation of posterior communicat<strong>in</strong>g artery<br />
aneurysm. Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg 94:225–227.<br />
Reifenberger G, Bostrümj, Bettag M, et al. (1996). Primary glioblastoma multiforme of the oculomotor nerve. Case<br />
report. J <strong>Neuro</strong>surg 84:1062–1066.<br />
Renowden SA, Harris KM, Hourihan MD. (1993). Isolated atraumatic third nerve palsy: cl<strong>in</strong>ical features and<br />
imag<strong>in</strong>g techniques. Br J Radiol 66:1111–1117.<br />
Richards BW, Jones FR Jr, Younge BR. (1992). Causes and prognosis <strong>in</strong> 4,278 cases of paralysis of the oculomotor,<br />
trochlear, and abducens cranial nerves. Am J Ophthalmol 113:489–496.<br />
Robertson PL, Pavkovic I, Donovan C, Blaivas M. (1998). Immature teratoma of the leptomen<strong>in</strong>ges <strong>in</strong> an 8-yearold<br />
child: unusual presentation with recurrent transient oculomotor nerve palsies and rapid progression to<br />
diffuse bra<strong>in</strong> <strong>in</strong>farction. J Child <strong>Neuro</strong>l 13:143–145.<br />
Rob<strong>in</strong>son R, Toland J, Eustace P. (1990). Pituitary apoplexy. A cause for pa<strong>in</strong>ful third nerve palsy. <strong>Neuro</strong><strong>ophthalmology</strong><br />
10:257–260.<br />
Rossitch E Jr, Carrazana EJ, Black PM. (1992). Isolated oculomotor nerve palsy follow<strong>in</strong>g apoplexy of a pituitary<br />
adenoma. J <strong>Neuro</strong>surg Sci 36:103–105.<br />
Saeki N, Murai H, M<strong>in</strong>e S, Yamaura A. (2000a). Fascicular arrangement with<strong>in</strong> the oculomotor nerve. MRI<br />
analysis of a midbra<strong>in</strong> <strong>in</strong>farct. J Cl<strong>in</strong> <strong>Neuro</strong>sci 7:268–270.<br />
Saeki N, Murai N, Sunami K. (1996). Midbra<strong>in</strong> tegmental lesions affect<strong>in</strong>g or spar<strong>in</strong>g the pupillary fibres. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 61:401–402.<br />
Saeki N, Yamaura A, Sunami K. (2000b). Bilateral ptosis with pupil spar<strong>in</strong>g because of a discrete midbra<strong>in</strong> lesion:<br />
magnetic resonance imag<strong>in</strong>g evidence of topographic arrangement with<strong>in</strong> the oculomotor nerve. J <strong>Neuro</strong>-<br />
Ophthalmol 20:130–134.
Third Nerve Palsies 279<br />
Saeki N, Yotsukura J, Adachi E, Yamaura A. (2000c). Isolated superior division oculomotor palsy <strong>in</strong> a child with<br />
spontaneous recovery. J Cl<strong>in</strong> <strong>Neuro</strong>sci 7:62–64.<br />
Sanchez Dalmau BF, Abdul-Rahim AS, Zimmerman RA. (1998). Young boy with progressive double vision. Surv<br />
Ophthalmol 43:47–52.<br />
Sanders S, Kawasaki A, Purv<strong>in</strong> VA. (2001). Pattern of extraocular muscle weakness <strong>in</strong> vasculopathic pupilspar<strong>in</strong>g,<br />
<strong>in</strong>complete third nerve palsy. J <strong>Neuro</strong>-ophthalmol 21:256–259.<br />
Sanli M, Alt<strong>in</strong>urs N, Bavbek M. (1995). Partial bilateral oculomotor nucleus lesion follow<strong>in</strong>g surgery of a fourth<br />
ventricle ependymoma. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:103–105.<br />
Savas R, Sommer A, Gueckel F, Georgi M. (1997). Isolated oculomotor nerve paralysis <strong>in</strong> Lyme disease: MRI.<br />
<strong>Neuro</strong>radiology 39:139–141.<br />
Schiev<strong>in</strong>k WI, Mokri B, Garrity JA, et al. (1993). Ocular motor nerve palsies <strong>in</strong> spontaneous dissections of the<br />
cervical <strong>in</strong>ternal carotid artery. <strong>Neuro</strong>logy 43:1938–1941.<br />
Schultheiss R, Kristof R, Schramm J. (1993). Complete removal of an oculomotor nerve neur<strong>in</strong>oma without<br />
permanent functional deficit. Ger J Ophthalmol 2:228–233.<br />
Schumacher-Feero LA, Yoo KW, Mendiola Solari F, Biglan A. (1999). Third cranial nerve palsy <strong>in</strong> children. Am<br />
JOphthalmol 128:216–221.<br />
Schwartz TH, Lycette CA, Kargman DE. (1995). Cl<strong>in</strong>icoradiographic evidence for oculomotor fascicular anatomy.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 59:338.<br />
Sethi DS, Lau DP, Chan C. (1997). Sphenoid s<strong>in</strong>us mucocele present<strong>in</strong>g with isolated oculomotor nerve palsy.<br />
J Laryngol Otol 111:471–473.<br />
Seyer H, Kompf D, Fahlbusch R. (1992). Optomotor palsies <strong>in</strong> pituitary apoplexy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:217–224.<br />
Shen WC, Yang DY, Ho YJ, Lee SK. (1993). Neurilemmoma of the oculomotor nerve present<strong>in</strong>g as an orbital mass:<br />
MR f<strong>in</strong>d<strong>in</strong>gs. AJNR 14:1253–1254.<br />
Shuaib A, Murphy W. (1987). Mesencephalic hemorrhage and third nerve palsy. J Comput Assist Tomogr<br />
11:385–388.<br />
Shuaib A, Israelian G, Lee MA. (1989). Mesencephalic hemorrhage and unilateral pupillary deficit. J Cl<strong>in</strong> <strong>Neuro</strong>ophthal<br />
9:47–49.<br />
Silva MN, Saeki N, Hirai S, Yamaura A. (1999). Unusual cranial nerve palsy caused by cavernous s<strong>in</strong>us<br />
aneurysms. <strong>Cl<strong>in</strong>ical</strong> and anatomical considerations reviewed. Surg <strong>Neuro</strong>l 52:148–149.<br />
Sood A, Midha V, Sood N, Gupta D. (1999). Hepatitis B and pupil-spar<strong>in</strong>g oculomotor nerve paresis. Cl<strong>in</strong> Infect<br />
Dis 29:1330–1331.<br />
Soysal T, Ferhanoglu B, Bilir M, Akman N. (1993). Oculomotor nerve palsy associated with v<strong>in</strong>crist<strong>in</strong>e treatment.<br />
Acta Haematol 90:209–210.<br />
Stefanis L, Przedborski S. (1993). Isolated palsy of the superior branch of the oculomotor nerve due to chronic<br />
erosive sphenoid s<strong>in</strong>usitis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:229–231.<br />
Straube A, Bandmann O, Buttner U, Schmidt H. (1993). A contrast enhanced lesion of the III nerve on MR of a<br />
patient with ophthalmoplegic migra<strong>in</strong>e as evidence for a Tolosa-Hunt syndrome. Headache 33:446–448.<br />
Striph GG. (1993). Consecutive oculomotor nerve palsy from a de novo cerebral aneurysm. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
13:181–187.<br />
Takano S, Endo M, Miyasaka Y, et al. (1990). Neur<strong>in</strong>oma of the oculomotor nerve—case report. <strong>Neuro</strong>l Med Chir<br />
30:132–136.<br />
Tao ZD. (1992). Oculomotor neuropathy syndrome. A diagnostic challenge <strong>in</strong> nasopharyngeal carc<strong>in</strong>oma. Ch<strong>in</strong><br />
Med J 105:567–571.<br />
Teasdale E, Stratham P, Straiton J, MacPherson P. (1990). Non-<strong>in</strong>vasive radiological <strong>in</strong>vestigation for oculomotor<br />
palsy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 53:549–553.<br />
Tezer I, Dogulu CF, Kansu T. (2000). Isolated <strong>in</strong>ferior rectus palsy as a result of paramedian thalamopeduncular<br />
<strong>in</strong>farction. J <strong>Neuro</strong>-Ophthalmol 20:154–155.<br />
Thömke F, Lensch E, R<strong>in</strong>gel K, Hopf HC. (1997). Isolated cranial nerve palsies <strong>in</strong> multiple sclerosis. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 63:682–685.<br />
Thömke F, Tettenborn B, Hopf HC. (1995). Third nerve palsy as the sole manifestation of midbra<strong>in</strong> ischemia.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 15:327–335.<br />
Tomsak RL, Masaryk TJ, Bates JH. (1991). Magnetic resonance angiography (MRA) of isolated aneurysmal third<br />
nerve palsy. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:16–18.<br />
Trobe JD. (1985). Isolated pupil-spar<strong>in</strong>g third nerve palsy. Ophthalmology 92:58–61.<br />
Trobe JD. (1998). Manag<strong>in</strong>g oculomotor nerve palsy. Arch Ophthalmol 116:798.<br />
Tsaloumas MD, Willshaw HE. (1997). Congenital oculomotor palsy: associated neurological and ophthalmological<br />
f<strong>in</strong>d<strong>in</strong>gs. Eye 11:500–503.
280 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Tummala RP, Harrison A, Madison MT, and Nussbaum ES. (2001). Pseudomyasthenia result<strong>in</strong>g from a posterior<br />
carotid artery wall aneurysm: a novel presentation: case report. <strong>Neuro</strong>surgery 49:1466–1469.<br />
Uehara T, Tabuchi M, Kawaguchi T, Mori E. (1998). Spontaneous dural carotid cavernous s<strong>in</strong>us fistula present<strong>in</strong>g<br />
isolated ophthalmoplegia: evaluation with MR angiography. <strong>Neuro</strong>logy 50:814–816.<br />
Ueyama H, Kummamoto T, Fukuda S, et al. (1997). Isolated third nerve palsy due to sarcoidosis. Sarcoid Vasc<br />
Diffuse Lung Dis 14:169–170.<br />
Umapathi T, Koon SW, Eng BM, et al. (2000). Insight <strong>in</strong>to the three-dimensional structure of the oculomotor<br />
nuclear complex and fascicles. J <strong>Neuro</strong>-Ophthalmol 20:138–144.<br />
Varma R, Miller NR. (1994). Primary oculomotor nerve synk<strong>in</strong>esis caused by an extracavernous <strong>in</strong>tradural<br />
aneurysm. Am J Ophthalmol 118:83–87.<br />
Vetrugno R, Mascalchi M, Marulli D, et al. (1997). Plus m<strong>in</strong>us lid syndrome due to cerebral glioma. A case report.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 18:149–151.<br />
von Noorden GK, Hansell R. (1991). <strong>Cl<strong>in</strong>ical</strong> characteristics and treatment of isolated <strong>in</strong>ferior rectus paralysis.<br />
Ophthalmology 98:253–257.<br />
Wake A, Kak<strong>in</strong>uma A, Mori N, et al. (1993). <strong>An</strong>giotropic lymphoma of paranasal s<strong>in</strong>uses with <strong>in</strong>itial symptoms of<br />
oculomotor nerve palsy. Intern Med 32:237–242.<br />
Walter KA, Newman NJ, Lessell S. (1994). Oculomotor palsy from m<strong>in</strong>or head trauma: <strong>in</strong>itial sign of <strong>in</strong>tracranial<br />
aneurysm. <strong>Neuro</strong>logy 44:148–150.<br />
We<strong>in</strong>berg DA, Kaufman DI, Siebert JD, Pernicone JR. (1996). Negative MRI versus real disease. Surv Ophthalmol<br />
40:312–319.<br />
White WL, Mumma JV, Tomasovic JJ. (1992). Congenital oculomotor nerve palsy, cerebellar hypoplasia, and facial<br />
capillary hemangioma. Am J Ophthalmol 113:497–500.<br />
Wilhelm H, Klier R, Tùth B, Wilhelm B. (1995). Oculomotor nerve paresis start<strong>in</strong>g as isolated <strong>in</strong>ternal<br />
ophthalmoplegia. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:211–215.<br />
W<strong>in</strong>terkorn JMS, Bruno M. (2001). Relative pupil-spar<strong>in</strong>g oculomotor nerve palsy as the present<strong>in</strong>g sign of<br />
posterior fossa men<strong>in</strong>gioma. J <strong>Neuro</strong>-ophthalmol 21:207–209.<br />
Wol<strong>in</strong> MJ, Saunders RA. (1992). <strong>An</strong>eurysmal oculomotor nerve palsy <strong>in</strong> an 11-year-old boy. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
12:178–180.<br />
Worth<strong>in</strong>gton JM, Halmagyi GM. (1996). Bilateral total ophthalmoplegia due to midbra<strong>in</strong> hematoma. <strong>Neuro</strong>logy<br />
46:1176.<br />
Wu HM, Lee AG, Lehane DE, et al. (1997). Ocular and orbital complications of <strong>in</strong>traarterial cisplat<strong>in</strong>. A case<br />
report. J <strong>Neuro</strong>-Ophthalmol 17:195–198.<br />
Yen M-Y, Mu-Huo M, Wang A-G, Liu J-H. (1998). Isolated oculomotor palsy caused by dural carotid cavernous<br />
s<strong>in</strong>us fistula. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:38.<br />
Zimmer DV. (1991). Oculomotor nerve palsy from posterior communicat<strong>in</strong>g artery aneurysm. J La State Med Soc<br />
143:22–25.<br />
Z<strong>in</strong>gale A, Albanese V, Giuffrida A, et al. (1997). Pa<strong>in</strong>ful ophthalmoplegia syndrome (spheno-cavernous<br />
syndrome) caused by a ruptured posterior communicat<strong>in</strong>g artery aneurysm. A brief report. J <strong>Neuro</strong>surg<br />
Sci 41:299–301.<br />
Z<strong>in</strong>gale A, Chiarmonte I, Mancuso P, et al. (1993). Craniofacial pa<strong>in</strong> and <strong>in</strong>complete oculomotor palsy associated<br />
with ipsilateral primitive trigem<strong>in</strong>al artery. J <strong>Neuro</strong>surg Sci 37:251–255.<br />
Zurev<strong>in</strong>sky J. (1993). Ocular palsies <strong>in</strong> ophthalmic zoster. Am Orthop J 43:130–134.
12 r<br />
Fourth Nerve Palsies<br />
What Is the Topographic <strong>An</strong>atomy of the<br />
Fourth Nerve?<br />
The fourth nerve nucleus is located <strong>in</strong> the midbra<strong>in</strong> beneath the <strong>in</strong>ferior colliculus. The<br />
fourth nerve is the only cranial nerve that exits dorsally from the bra<strong>in</strong>stem, it has the<br />
longest <strong>in</strong>tracranial course, and it crosses <strong>in</strong> the anterior medullary velum. It passes<br />
between the superior cerebellar artery and the posterior cerebellar artery, runs <strong>in</strong> the<br />
subarachnoid space, travels with<strong>in</strong> the lateral wall of the cavernous s<strong>in</strong>us, and enters<br />
the orbit via the superior orbital fissure to <strong>in</strong>nervate the superior oblique muscle.<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of Fourth<br />
Nerve Palsies?<br />
Fourth cranial nerve palsies may cause the follow<strong>in</strong>g (von Noorden, 1986):<br />
1. Incomitant hypertropia is demonstrated with the three-step maneuver. The hypertropia<br />
<strong>in</strong>creases on head tilt toward the paralyzed side (positive Bielschowsky’s test).<br />
Usually the unaffected eye is fixat<strong>in</strong>g and the hypertropia occurs <strong>in</strong> the <strong>in</strong>volved<br />
eye. Hypotropia may occur <strong>in</strong> the normal eye if the affected eye is fixat<strong>in</strong>g. The<br />
hypertropia is usually most prom<strong>in</strong>ent <strong>in</strong> the field of gaze of the <strong>in</strong>volved superior<br />
oblique muscle, especially <strong>in</strong> cases of acute or recent onset. The hypertropia may also<br />
be most prom<strong>in</strong>ent <strong>in</strong> the field of gaze of the ipsilateral overact<strong>in</strong>g <strong>in</strong>ferior oblique<br />
muscle <strong>in</strong> subacute or chronic cases. In palsies of longer duration, the hypertropia<br />
may be relatively equal <strong>in</strong> the various gaze positions (spread of comitance).<br />
2. Duction test<strong>in</strong>g may variably reveal underaction of the ipsilateral superior oblique<br />
muscle, overaction of the ipsilateral <strong>in</strong>ferior oblique muscle, or overaction of the<br />
contralateral superior oblique muscle.<br />
281
282 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
3. Pseudo-overaction of the superior oblique <strong>in</strong> the un<strong>in</strong>volved eye may occur with<br />
spread of comitance. Secondary contracture of the superior rectus muscle <strong>in</strong> the<br />
<strong>in</strong>volved eye may cause hypertropia <strong>in</strong>volv<strong>in</strong>g the entire lower field of gaze. In a<br />
patient with a superior oblique muscle paralysis who habitually fixates with the<br />
paretic eye and <strong>in</strong> whom overaction of the ipsilateral <strong>in</strong>ferior oblique muscle has<br />
developed, less than the normal amount of <strong>in</strong>nervation will be required when the<br />
patient looks up and to the contralateral side. Because the <strong>in</strong>nervation flow<strong>in</strong>g to<br />
the opposite superior rectus is ‘‘determ<strong>in</strong>ed’’ by the overact<strong>in</strong>g ipsilateral <strong>in</strong>ferior<br />
oblique (Her<strong>in</strong>g’s law), the opposite superior rectus muscle will seem paretic<br />
(<strong>in</strong>hibitional palsy of the contralateral antagonist). In these cases, the head tilt test<br />
will correctly determ<strong>in</strong>e which of the two eyes is paretic.<br />
4. Excyclotropia due to loss of <strong>in</strong>cyclotorsion function of the superior oblique muscle.<br />
This torsion may be evident on fundus exam and can be measured us<strong>in</strong>g double<br />
Maddox rod test<strong>in</strong>g. The excyclotropia is usually symptomatic <strong>in</strong> acquired cases but<br />
is often asymptomatic <strong>in</strong> congenital cases.<br />
5. <strong>An</strong> anomalous head tilt elim<strong>in</strong>ates the hypertropia or less commonly the cyclotropia.<br />
This head tilt is present <strong>in</strong> approximately 70% of patients and is usually away from<br />
the <strong>in</strong>volved side but may be paradoxical (toward the <strong>in</strong>volved side) <strong>in</strong> about 3%.<br />
It is important to differentiate patients with decompensation of a congenital fourth<br />
nerve palsy (FNP) from those with an acquired FNP. In patients with congenital FNPs:<br />
1. Old photos may show a long-stand<strong>in</strong>g head tilt.<br />
2. Patients usually are noted to have cyclotropia on exam<strong>in</strong>ation but often do<br />
not compla<strong>in</strong> of cyclotropia (subjective image tilt<strong>in</strong>g) as do some patients with<br />
acquired FNPs.<br />
3. Large vertical fusional amplitudes (> 8 prism diopters) <strong>in</strong> primary gaze are characteristic<br />
of congenital cases.<br />
4. Facial asymmetry (hypoplasia on side of head turn) suggests a congenital lesion.<br />
Bilateral FNPs are suggested by the follow<strong>in</strong>g:<br />
1. A right hypertropia <strong>in</strong> left gaze and left hypertropia <strong>in</strong> right gaze (a revers<strong>in</strong>g<br />
hypertropia).<br />
2. A positive Bielschowsky test on tilt to either shoulder (‘‘double Bielschowsky test’’).<br />
3. Large excyclotropia (> 10 degrees).<br />
4. V-pattern esotropia (15 prism diopters or more difference <strong>in</strong> esotropia between<br />
upward and downward gaze). The V pattern is caused by a decrease of the<br />
abduct<strong>in</strong>g effect of the superior oblique(s) <strong>in</strong> depression and secondary overaction<br />
of the abduct<strong>in</strong>g effect of the <strong>in</strong>ferior oblique muscle(s).<br />
5. Underaction of both superior oblique muscles and=or overaction of both <strong>in</strong>ferior<br />
oblique muscles on duction test<strong>in</strong>g.<br />
6. In general, bilateral FNPs tend to have a smaller hypertropia <strong>in</strong> primary position<br />
than do unilateral FNPs.<br />
The criteria for the diagnosis of FNPs are listed <strong>in</strong> Table 12–1. FNP may be categorized<br />
as either isolated or nonisolated. For diagnostic classification based on topographic<br />
localization, nonisolated FNP may be grouped <strong>in</strong>to the follow<strong>in</strong>g four syndromes:<br />
1. Midbra<strong>in</strong> (nucleus=fascicle syndrome) FNP<br />
2. Subarachnoid space FNP<br />
3. Cavernous s<strong>in</strong>us FNP
Table 12–1. Criteria for the Diagnosis of Fourth Nerve Palsy<br />
4. Orbital FNP<br />
Nonisolated FNP (type 1; see below), with f<strong>in</strong>d<strong>in</strong>gs that localize to the bra<strong>in</strong>stem,<br />
subarachnoid space, cavernous s<strong>in</strong>us, or orbit, should undergo a directed neuroimag<strong>in</strong>g<br />
study (Berlit, 1991; Brazis, 1993; Burde, 1992; Celli, 1992; Elliot, 1991; Kim, 1992;<br />
Richards, 1992; Vanooteghem, 1992).<br />
Table 12–2 outl<strong>in</strong>es the cl<strong>in</strong>ical features of FNP by location of the responsible lesion.<br />
Table 12–3 lists the etiologies for an FNP based on cl<strong>in</strong>ical topographic localization.<br />
Is the FNP Due to a Midbra<strong>in</strong> Lesion?<br />
A midbra<strong>in</strong> (i.e., nuclear=fascicular) FNP is def<strong>in</strong>ed by the ‘‘company it keeps’’; other<br />
bra<strong>in</strong>stem signs usually present, <strong>in</strong>clud<strong>in</strong>g hemisensory loss, hemiparesis, a central<br />
Horner’s syndrome, or other bra<strong>in</strong>stem cranial neuropathies (e.g., third nerve palsy).<br />
The differential diagnosis <strong>in</strong>cludes midbra<strong>in</strong> ischemia, hemorrhage, demyel<strong>in</strong>ation, and<br />
neoplasm. <strong>Neuro</strong>imag<strong>in</strong>g (preferably magnetic resonance imag<strong>in</strong>g [MRI]) should be<br />
directed to the midbra<strong>in</strong> (class II–III, level B).<br />
Is the FNP the Result of a Subarachnoid<br />
Space Lesion?<br />
Lesions of the subarachnoid space are rarely associated with an isolated FNP. Patients<br />
with subarachnoid space lesions usually have associated signs and symptoms <strong>in</strong>clud<strong>in</strong>g<br />
headache, stiff neck, and other cranial neuropathies. <strong>Neuro</strong>imag<strong>in</strong>g (MRI) should be<br />
directed to the bra<strong>in</strong>stem and subarachnoid space. Computed tomography (CT)<br />
imag<strong>in</strong>g should be considered <strong>in</strong> cases of acute trauma, to evaluate bone lesions, or<br />
<strong>in</strong> the evaluation of acute vascular processes (e.g., subarachnoid hemorrhage). Lumbar<br />
puncture follow<strong>in</strong>g negative neuroimag<strong>in</strong>g should be considered <strong>in</strong> these cases<br />
(class II–III, level B).<br />
Is the FNP Due to a Cavernous S<strong>in</strong>us Lesion?<br />
Fourth Nerve Palsies 283<br />
B<strong>in</strong>ocular vertical and=or torsional diplopia or misalignment<br />
Ipsilateral hyperdeviation <strong>in</strong> primary position, worsened by contralateral gaze and ipsilateral<br />
head tilt (the three-step test)<br />
Variable ipsilateral excyclotorsion<br />
<strong>An</strong>omalous compensatory head or face position<br />
Weakness of the <strong>in</strong>volved superior oblique muscle on ductions<br />
Cavernous s<strong>in</strong>us lesions are usually associated with other cranial nerve signs (e.g., third,<br />
fifth, or sixth nerve paresis) or a Horner’s syndrome. <strong>Neuro</strong>imag<strong>in</strong>g (preferably MRI)<br />
should be directed to the cavernous s<strong>in</strong>us (class II–III, level B).
284 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 12–2. The Localization of Trochlear Nerve Lesions<br />
Structure Involved <strong>Cl<strong>in</strong>ical</strong> Manifestation<br />
A: Lesions affect<strong>in</strong>g the trochlear nucleus and=or fascicles (superior oblique palsy contralateral to lesions)<br />
Nucleus=fascicles alone Isolated trochlear palsy (rare)<br />
Pretectal region Vertical gaze palsy (dorsal midbra<strong>in</strong><br />
syndrome)<br />
Superior cerebellar peduncle Dysmetria on side of lesion<br />
Descend<strong>in</strong>g sympathetic fibers Horner’s syndrome on side of lesion<br />
Medial longitud<strong>in</strong>al fasciculus (MLF) Ipsilateral paresis of adduction with<br />
nystagmus of contralateral<br />
abduct<strong>in</strong>g eye<br />
Brachium of superior colliculus Contralateral relative afferent pupillary<br />
defect (RAPD) without visual<br />
impairment<br />
<strong>An</strong>terior medullary velum Bilateral trochlear nerve palsies<br />
B: Lesions affect<strong>in</strong>g the trochlear nerve with<strong>in</strong> the subarachnoid space (superior oblique palsy usually<br />
ipsilateral to lesion unless mesencephalon compressed)<br />
Trochlear nerve alone Isolated trochlear palsy<br />
Superior cerebellar peduncle Ipsilateral dysmetria<br />
Cerebral peduncle Contralateral hemiparesis<br />
C: Lesions affect<strong>in</strong>g the trochlear nerve with<strong>in</strong> the cavernous s<strong>in</strong>us and=or superior orbital fissure<br />
Trochlear nerve alone Isolated trochlear palsy (rare)<br />
Cranial nerves III, VI, sympathetic Ophthalmoplegia, pupil small, large, or<br />
spared, ptosis<br />
Cranial nerve V (ophthalmic division) Facial=retro-orbital pa<strong>in</strong>; sensory loss<br />
(forehead)<br />
Increased venous pressure<br />
D: Lesions affect<strong>in</strong>g the trochlear nerve with<strong>in</strong> the orbit<br />
Proptosis; chemosis<br />
Trochlear nerve, trochlea, superior oblique<br />
muscle or tendon<br />
Superior oblique palsy<br />
Mechanical restriction of superior oblique<br />
Brown’s superior oblique tendon sheath<br />
tendon<br />
syndrome<br />
Other ocular motor nerves=extraocular<br />
Ophthalmoplegia, ptosis, restricted ocular<br />
muscles<br />
movements<br />
Optic nerve Visual loss; optic disc swell<strong>in</strong>g=atrophy<br />
Mass effect Proptosis (occasionally enophthalmos),<br />
chemosis, eyelid swell<strong>in</strong>g, etc.<br />
Source: Modified from Brazis, 2001, with permission from Lipp<strong>in</strong>cott Williams & Wilk<strong>in</strong>s.<br />
Is the FNP Caused by an Orbital Lesion?<br />
Orbital lesions usually produce signs such as proptosis, chemosis, and orbital or<br />
conjunctival edema. <strong>Neuro</strong>imag<strong>in</strong>g (preferably MRI) should be directed to the orbit<br />
(class II–III, level B).<br />
We def<strong>in</strong>e six types of FNP, as shown <strong>in</strong> Table 12–4.
Fourth Nerve Palsies 285<br />
Table 12–3. Etiologies for a Fourth Nerve Palsy Based on <strong>Cl<strong>in</strong>ical</strong> Topographic<br />
Localization<br />
Midbra<strong>in</strong> (nuclear=fascicular) (Elliot, 1991; Thömke, 2000)<br />
Aplasia of the nucleus<br />
Arteriovenous malformation (Gonyea, 1990; Kim, 1992)<br />
Demyel<strong>in</strong>ation (Jacobson, 1999)<br />
Hemorrhage (Galetta, 1998; Kim, 1993; Mon, 1996; Müri, 1995; Tachibana, 1990;<br />
Thömke, 1999)<br />
Ischemia=<strong>in</strong>farction (Kim, 1993; Thömke, 1999; Ulrich, 1998)<br />
Tumor (e.g., glioma) (Barr, 1997; Landolfi, 1998; Mielke, 2001)<br />
Trauma (<strong>in</strong>clud<strong>in</strong>g surgical)<br />
Sarcoidosis (Leiba, 1996)<br />
Arachnoid cyst of quadrigem<strong>in</strong>al cistern (Ohtsuka, 1998)<br />
Subarachnoid space<br />
<strong>An</strong>eurysm (e.g., superior cerebellar artery) (Agost<strong>in</strong>is, 1992; Coll<strong>in</strong>s, 1992)<br />
Hydrocephalus<br />
Infections (mastoiditis, men<strong>in</strong>gitis) (Carter, 1997; Ferreira, 1997; Sadun, 1999)<br />
Wegener’s granulomatosis (Newman, 1995)<br />
Sarcoidosis (Frohman, 2001)<br />
Superficial siderosis of central nervous system (CNS) (Hashimoto, 1996; Sh<strong>in</strong>mei, 1997)<br />
Post–lumbar puncture or sp<strong>in</strong>al anesthesia<br />
Pseudotumor cerebri (Lee, 1995; Patton, 2000; Speer, 1999)<br />
Trauma, <strong>in</strong>clud<strong>in</strong>g surgical (Baker, 1991; Hara, 2001; Hoya, 2000; Jacobson 1995; Lepore,<br />
1995; Sabates, 1991)<br />
Neoplasm<br />
Carc<strong>in</strong>omatous men<strong>in</strong>gitis<br />
Cerebellar hemangioblastoma<br />
Ependymoma<br />
Men<strong>in</strong>gioma<br />
Metastasis<br />
<strong>Neuro</strong>lemmoma=schwannoma (Fe<strong>in</strong>berg, 1999; Gentry, 1991; Santoreneos, 1997)<br />
P<strong>in</strong>eal tumors<br />
Trochlear nerve sheath tumors<br />
Fisher’s syndrome (Tanaka, 1998)<br />
Churg-Strauss syndrome (Vitali, 1996)<br />
Cavernous s<strong>in</strong>us<br />
Neoplasm (e.g., men<strong>in</strong>gioma, pituitary adenoma) (Eisenberg, 1999; Keane, 1996;<br />
Petermann, 1999)<br />
Infectious: herpes zoster (Chang-God<strong>in</strong>ich, 1997), mucormycosis (Keane, 1996)<br />
Inflammation: Tolosa-Hunt syndrome, Wegener’s granulomatosis (Herman, 1999)<br />
Internal carotid artery aneurysm (Arruga, 1991; FitzSimon, 1995; Hahn, 2000;<br />
Keane, 1996; Shimo-oku, 1998)<br />
Dural carotid-cavernous s<strong>in</strong>us fistula (Tsai, 2000)<br />
Superior ophthalmic ve<strong>in</strong> thrombosis (Polito, 1996)<br />
Foramen ovale electrode placement (Herrendorf, 1997)<br />
Balloon test occlusion of cervical <strong>in</strong>ternal carotid artery (Lopes, 1998)<br />
(cont<strong>in</strong>ued)
286 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 12–3. (cont<strong>in</strong>ued)<br />
Orbit<br />
Neoplasm<br />
Infection<br />
Infiltration<br />
Waldenström’s macroglobul<strong>in</strong>emia<br />
Inflammation (orbital pseudotumor)<br />
Progressive systemic sclerosis<br />
Trauma (orbital floor fracture)<br />
Other<br />
Migra<strong>in</strong>e (Wong, 1996)<br />
Congenital (Botelho, 1996; Holmes, 1999)<br />
Congenital unmasked by botul<strong>in</strong>um tox<strong>in</strong> therapy for cervical torticollis)<br />
(Varrato, 2000)<br />
Cephalic tetanus (Orwitz, 1997)<br />
Table 12–4. Def<strong>in</strong>itions of the Six Types of Fourth Nerve Palsy (FNP)<br />
Type 1: nonisolated<br />
FNPs are considered nonisolated <strong>in</strong> the presence of the follow<strong>in</strong>g features:<br />
Other neurologic or neuro-ophthalmologic signs<br />
Evidence to suggest myasthenia gravis such as ptosis, or fatigability of the motility deficit<br />
Evidence for systemic <strong>in</strong>flammatory disorders such as giant cell arteritis<br />
Type 2: traumatic<br />
Isolated, unilateral, or bilateral FNPs that have a clearly established temporal relationship to<br />
previous head trauma and do not progress are considered traumatic <strong>in</strong> orig<strong>in</strong>; patients<br />
have no other neurologic deficits other than those associated with the <strong>in</strong>itial<br />
traumatic event<br />
Type 3: congenital<br />
Congenital FNPs may show the follow<strong>in</strong>g:<br />
Large vertical fusional amplitudes (greater than 8 prism diopters)<br />
Facial asymmetry or sternocleidomastoid muscle hypertrophy<br />
Long-stand<strong>in</strong>g anomalous head position that may be present <strong>in</strong> old photographs<br />
Type 4: vasculopathic<br />
Vasculopathic FNPs occur <strong>in</strong> patients older than 50 years of age with or without known<br />
hypertension or diabetes, or <strong>in</strong> younger patients with known vasculopathic risk factors<br />
Type 5: nonvasculopathic<br />
Patients without vasculopathic risk factors and not classified as any of the above types are<br />
classified nonvasculopathic FNP<br />
Type 6: progressive or unresolved<br />
FNPs that worsen after the acute stage (greater than 1 week) as def<strong>in</strong>ed by a significant<br />
<strong>in</strong>crease <strong>in</strong> the measured ocular vertical deviation are considered to be progressive, and<br />
patients without improvement <strong>in</strong> the measured ocular vertical deviation after 6 to 8 weeks<br />
are considered unresolved
Is the FNP Due to Trauma?<br />
At least 23 retrospective studies of traumatic (type 2) FNP have recommended that<br />
isolated, traumatic, unilateral, or bilateral FNP do not require additional neuroimag<strong>in</strong>g<br />
or further evaluation (Baker, 1991; Berlit, 1991; Burde, 1992; Hoya, 2000; Richards, 1992;<br />
Sabates, 1991). FNP after mild head trauma and out of proportion to the deficit have<br />
been observed <strong>in</strong> association with an underly<strong>in</strong>g asymptomatic basal <strong>in</strong>tracranial tumor<br />
<strong>in</strong> at least three reports (Jacobson, 1988; Miller, 1989; Neetens, 1981). Neetens reported<br />
three such cases, but two cases had other neuro-ophthalmologic signs as well (Neetens,<br />
1981). Although uncommon, neuroimag<strong>in</strong>g may be warranted <strong>in</strong> patients with FNP<br />
after m<strong>in</strong>imal or trivial head trauma to exclude a mass lesion (class III, level C).<br />
Is the FNP Congenital?<br />
Clearly congenital unilateral or bilateral FNP (type 3) are not associated with <strong>in</strong>tracranial<br />
lesions <strong>in</strong> isolation and therefore do not require further diagnostic evaluation<br />
such as neuroimag<strong>in</strong>g studies (Burde, 1992; Robb, 1990; von Noorden, 1986, 1994)<br />
(class III–IV, level C).<br />
Is the FNP Vasculopathic?<br />
Vasculopathic FNP (type 4) do not require any <strong>in</strong>itial neuroimag<strong>in</strong>g studies, and<br />
observation for improvement over the next 6 to 8 weeks is recommended (Burde,<br />
1992). Patients with vasculopathic FNP (type 4), often resolve spontaneously with<strong>in</strong> 4 to<br />
6 months. Rush reported a recovery rate for FNP of 53.5% <strong>in</strong> 172 nonselected cases, and<br />
a higher recovery rate of 71% <strong>in</strong> 166 patients with diabetes mellitus, hypertension, or<br />
atherosclerosis (Rush, 1981). <strong>An</strong>other report by Ksiazek et al described improvement <strong>in</strong><br />
90% of 39 patients with microvascular and idiopathic FNP with<strong>in</strong> 6 months (Ksiazek,<br />
1988). Vasculopathic FNP usually improves with<strong>in</strong> a few months (Burde, 1992; Rush,<br />
1981; von Noorden, 1986), and patients with progressive or unresolved FNP, or with<br />
new neurologic signs or symptoms, should have neuroimag<strong>in</strong>g (class II–III, level B)<br />
(Agost<strong>in</strong>is, 1992; Arruga, 1991; Burde, 1992; Gentry, 1991; Miller, 1989; Rush, 1981).<br />
Patients with spontaneously resolv<strong>in</strong>g palsies do not require any further neuroimag<strong>in</strong>g<br />
(class II–III, level B). It is recommended that elderly patients who present with headache,<br />
scalp tenderness, jaw claudication, or visual loss undergo an appropriate evaluation<br />
for giant cell arteritis, <strong>in</strong>clud<strong>in</strong>g an erythrocyte sedimentation rate and a temporal<br />
artery biopsy (Burde, 1992; Miller, 1989; Reich, 1990) (class III–IV, level B). There is<br />
<strong>in</strong>sufficient evidence to recommend evaluation for giant cell arteritis <strong>in</strong> every patient<br />
with motility suggest<strong>in</strong>g an isolated FNP (class IV, level U).<br />
What Is the Evaluation of Nonvasculopathic<br />
FNP?<br />
Fourth Nerve Palsies 287<br />
Nonvasculopathic FNP (type 5) may be observed for improvement over the next 6 to 8<br />
weeks (class III, level B). Patients with resolution of symptoms and signs do not require
288 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
further evaluation (class III, level B). Patients with progression or lack of resolution<br />
should undergo neuroimag<strong>in</strong>g (preferably MRI). Myasthenia gravis may mimic<br />
FNP, and patients with variable or fatigable motility f<strong>in</strong>d<strong>in</strong>gs and=or ptosis should be<br />
evaluated for myasthenia gravis (see Chapter 15) (Burde, 1992; Miller, 1989) (class III–IV,<br />
level B).<br />
Test<strong>in</strong>g for vasculopathic risk factors <strong>in</strong> type 4 or type 5 FNP should be considered,<br />
even <strong>in</strong> the absence of a history of previous diabetes or hypertension. Green et al<br />
reported an isolated third nerve palsy as the <strong>in</strong>itial cl<strong>in</strong>ical manifestation of diabetes <strong>in</strong><br />
almost half of 25 patients (Green, 1964). Shrader and Schlez<strong>in</strong>ger reported that almost<br />
50% of diabetic sixth nerve palsies were the present<strong>in</strong>g cl<strong>in</strong>ical manifestation of the<br />
disease (Shrader, 1960). The results of these studies concern<strong>in</strong>g vasculopathic third and<br />
sixth nerve palsies may well be applicable to vasculopathic FNP (class III, level C).<br />
Table 12–5 summarizes the etiologies of FNP <strong>in</strong> 11 large retrospective series (Ellis,<br />
1976; Harley, 1980; Keane, 1993; Ksiazek, 1988; Mittleman, 1976; Richards, 1992; Rucker,<br />
1956, 1958, 1966; von Noorden, 1986; Wright, 1977). Traumatic FNP occurred <strong>in</strong><br />
35%, idiopathic FNP <strong>in</strong> 34%, vasculopathic FNP <strong>in</strong> 16%, neoplasm was reported <strong>in</strong><br />
3%, aneurysm <strong>in</strong> 0.5%, and a wide variety of miscellaneous conditions <strong>in</strong>clud<strong>in</strong>g<br />
myasthenia gravis, <strong>in</strong>fections, thyroid disease, and <strong>in</strong>flammation <strong>in</strong> 11% of patients.<br />
Younger patients, or those without vasculopathic risk factors (type 5), may require<br />
<strong>in</strong>itial neuroimag<strong>in</strong>g, but the data suggest that observation for spontaneous improvement<br />
may be sufficient (class III, level C). Isolated, idiopathic FNPs very rarely have<br />
been found to have an underly<strong>in</strong>g etiology after prolonged follow-up, and most resolve<br />
spontaneously with<strong>in</strong> several weeks to months (Coppeto, 1978; Ksiazek, 1988; Nemet,<br />
1980). Two retrospective case series with follow-up greater than 6 months described the<br />
prognosis of isolated, idiopathic FNP. Coppeto et al reported that 12 of 15 cases had<br />
resolved by 4 months after a mean follow-up of 5.5 years (Coppeto, 1978). Nemet et al<br />
described 13 cases, with a follow-up rang<strong>in</strong>g from 4 to 7 years, and all had resolved by<br />
10 weeks (Nemet, 1980). None of the patients <strong>in</strong> either series developed new neurologic<br />
Table 12–5. Etiologies for Acquired Isolated Fourth Nerve Palsy<br />
Author Cases Trauma Tumor Vascular <strong>An</strong>eurysm Unknown Other<br />
Rucker, 1956 40 12 1 8 1 15 3<br />
Rucker, 1958 67 24 3 24 0 9 7<br />
Rucker, 1966 84 23 7 13 0 28 13<br />
Mittleman, 1976 64 22 – – – 42 –<br />
Ellis, 1976 104 32 0 – 1 63 8<br />
Wright, 1977 23 9 0 8 0 3 3<br />
Harley, 1980 18 5 0 0 0 12 1<br />
Richards, 1992 578 169 28 103 5 186 87<br />
von Noorden, 1986 141 73 – – 0 62 6*<br />
Ksiazek, 1988 88 24 2 39 – 23 –<br />
Keane, 1993 81 64 0 8 – – 9<br />
TOTAL 1288 457 41 203 7 443 137<br />
100% 35% 3% 16% 0.5% 34% 11%<br />
*‘‘Other’’ <strong>in</strong> this study <strong>in</strong>cluded tumor, vascular, and myasthenia gravis.
Table 12–6. ‘‘Isolated’’ Fourth Nerve Palsy Due to Intracranial Lesion<br />
Fourth Nerve Palsies 289<br />
Author Cases Pathology Other <strong>Neuro</strong>logic Signs<br />
Suzuki, 1962 4 P<strong>in</strong>ealomas Yes<br />
Rucker, 1956 2 Frontal lobe glioma Unknown<br />
<strong>An</strong>eurysm of circle of Willis Unknown<br />
Rucker, 1958 3 Primary bra<strong>in</strong> tumor (1) Unknown<br />
Metastatic (2) Unknown<br />
Wise, 1965 1 Persistent trigem<strong>in</strong>al artery Headache<br />
Rucker, 1966 7 Midbra<strong>in</strong> gliomas (2) Unknown<br />
Men<strong>in</strong>gioma (1) Unknown<br />
Primary bra<strong>in</strong> tumors (3) Unknown<br />
Khawam, 1967 1 ‘‘Bra<strong>in</strong> tumor’’ Unknown<br />
Burger, 1970 8 Cerebellopont<strong>in</strong>e angle (CPA)<br />
tumors (4)<br />
Yes<br />
Cerebellar tumor (1) Yes<br />
Nasopharyngeal cancer (1) Yes<br />
Metastatic lung cancer (1) Yes<br />
<strong>An</strong>eurysm (1) Yes<br />
Robert, 1973 2 Pituitary tumors Yes<br />
Ellis, 1976 1 ‘‘Intracranial aneurysm’’ Unknown<br />
K<strong>in</strong>g, 1976 1 Schwannoma No<br />
Scully, 1976 1 Medulloblastoma Yes<br />
Younge, 1977 4 Gliomas (2) Unknown<br />
Metastatic breast cancer (1) Unknown<br />
Metastatic ovarian cancer (1) Unknown<br />
Wray, 1977 2 Pituitary tumors Yes<br />
Coppeto, 1978 3 Ependymoma (1) Yes<br />
Medulloblastoma (1) Yes<br />
Acoustic neuroma (1) Yes<br />
Boggan, 1979 1 Schwannoma Yes<br />
Rush, 1981 10 Men<strong>in</strong>giomas (2) Unknown<br />
Primary bra<strong>in</strong> tumor (1) Unknown<br />
Metastatic tumors (4) Unknown<br />
Intracavernous aneurysm (1) Yes<br />
Basilar aneurysm (1) Unknown<br />
<strong>An</strong>eurysm=subarachnoid<br />
hemorrhage (1)<br />
Yes<br />
Ho, 1981 1 Schwannoma No<br />
Neetens, 1981 3 Skull base tumors Yes<br />
Krohel, 1982 1 Juvenile pilocytic astrocytoma Yes<br />
Leunda, 1982 1 Schwannoma Yes<br />
McK<strong>in</strong>na, 1983 3 <strong>An</strong>eurysms Unknown<br />
Re<strong>in</strong>ecke, 1986 1 Reported <strong>in</strong> Krohel, 1982 Yes<br />
Jacobson, 1988 1 Vascular malformation Yes<br />
Slav<strong>in</strong>, 1987 1 Cavernous men<strong>in</strong>gioma *<br />
Yamamoto, 1987 1 Schwannoma Headache<br />
(cont<strong>in</strong>ued)
290 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 12–6. (cont<strong>in</strong>ued)<br />
Ksiazek, 1988 2 ‘‘Compressive etiologies’’ Unknown<br />
Maurice-Williams, 1989 1 Intracavernous aneurysm Headache<br />
Gonyea, 1990 1 Bra<strong>in</strong>stem arteriovenous<br />
malformation (AVM)<br />
Headache<br />
Arruga, 1991 1 Intracavernous aneurysm No<br />
Agost<strong>in</strong>is, 1992 1 Superior cerebellar aneurysm Headache<br />
Coll<strong>in</strong>s, 1992 1 Superior cerebellar aneurysm Headache<br />
Richards, 1992 14 Men<strong>in</strong>gioma (7) Unknown<br />
Metastatic (1) Unknown<br />
Glioma (4) Unknown<br />
Acoustic neuroma (1) Unknown<br />
Other primary (1) Unknown<br />
Kim, 1993 1 Bra<strong>in</strong>stem stroke Yes<br />
Mon, 1996 1 Midbra<strong>in</strong> hemorrhage No<br />
Galetta, 1998 1 Midbra<strong>in</strong> hemorrhage No<br />
Petermann, 1999 1 Pituitary tumor Headache<br />
Fe<strong>in</strong>hers, 1999 6 Trochlear schwannoma No<br />
Thömke, 1999 3 Bra<strong>in</strong>stem lacunes (2) No<br />
Hemorrhage (1) No<br />
Mielke, 2001 1 Metastatic bronchial cancer No<br />
*Patient developed progression of deviation after 2 years.<br />
disease over an extensive follow-up period. Although type 5 patients who improve may<br />
not require neuroimag<strong>in</strong>g, the cl<strong>in</strong>ical certa<strong>in</strong>ty of such a recommendation is not<br />
sufficiently strong <strong>in</strong> our op<strong>in</strong>ion to obviate the need for neuroimag<strong>in</strong>g <strong>in</strong> these<br />
nonvasculopathic patients (class III, level U). However, neuroimag<strong>in</strong>g should be<br />
considered for patients who do not improvement <strong>in</strong> 2 months (class III, level C).<br />
Some reports have described aneurysm as an extremely rare cause for isolated FNP<br />
(Agost<strong>in</strong>is, 1992; Arruga, 1991; Coll<strong>in</strong>s, 1992; Richards, 1992; Rucker, 1956; Rush, 1981),<br />
and cerebral angiography is not recommended unless an aneurysm is suggested by<br />
other neuroimag<strong>in</strong>g studies (class III, level B). Agost<strong>in</strong>is et al and Coll<strong>in</strong>s et al reported<br />
isolated FNP due to superior cerebellar aneurysms, but both patients described headaches<br />
(Agost<strong>in</strong>is, 1992; Coll<strong>in</strong>s, 1992). In these cases, neuroimag<strong>in</strong>g studies confirmed<br />
the presence of the aneurysm before angiography. There are <strong>in</strong>sufficient data to make a<br />
comment on the usefulness of MR angiography <strong>in</strong> FNP (class III–IV, level U).<br />
Although MR scans are generally felt to be a more sensitive and specific than CT <strong>in</strong><br />
the evaluation of cranial neuropathies, no conclusive evidence demonstrates an<br />
<strong>in</strong>creased yield from perform<strong>in</strong>g an MR scan rather than a CT scan for the specific<br />
evaluation of FNP. Richards et al reported an etiologic diagnosis <strong>in</strong> 69 of 144 (48%) FNP<br />
us<strong>in</strong>g MRI and <strong>in</strong> 289 of 684 (42%) cases us<strong>in</strong>g CT. These authors felt that ‘‘multiplanar<br />
CT may be a sufficient non<strong>in</strong>vasive study, especially when cl<strong>in</strong>ical suspicion is high ...<br />
[or] <strong>in</strong> patients with other neurologic f<strong>in</strong>d<strong>in</strong>gs’’ (Richards, 1992). Nevertheless, we<br />
believe that MRI is the study of choice for patients with FNP (class II–III, level B).<br />
A number of cases have been reported <strong>in</strong> the literature document<strong>in</strong>g <strong>in</strong>tracranial<br />
lesions <strong>in</strong> patients with FNP. Table 12–6 summarizes 86 cases of ‘‘isolated’’ FNP due to
Figure 12–1. Evaluation of fourth nerve palsy (FNP).<br />
Fourth Nerve Palsies 291<br />
an <strong>in</strong>tracranial lesion. Of these 86 patients, only five (5.8%) did not have other<br />
neurologic signs or symptoms and thus would be considered truly isolated by our<br />
criteria. One developed other neurologic signs after a short follow-up period, and <strong>in</strong> the<br />
rema<strong>in</strong><strong>in</strong>g four patients persistence or progression of symptoms would have eventually<br />
resulted <strong>in</strong> a neuroimag<strong>in</strong>g study. Of the rema<strong>in</strong><strong>in</strong>g 81 patients, six had headache or
292 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
pa<strong>in</strong> (7%), 31 had other neurologic signs (38%), and the cl<strong>in</strong>ical <strong>in</strong>formation was<br />
<strong>in</strong>sufficient to determ<strong>in</strong>e if the FNP was truly isolated <strong>in</strong> 44 patients (54%). Keane<br />
reported <strong>in</strong>tracranial tumor as an etiology <strong>in</strong> 12 of 95 unilateral cases, but all 12 (100%)<br />
had other neuro-ophthalmic signs, and none of 81 isolated FNP later reported by Keane<br />
had an <strong>in</strong>tracranial tumor (Keane, 1993). This would suggest that the yield for<br />
evaluation of an isolated FNP is low (class III, level C).<br />
All patients with progressive FNP (type 6) should undergo neuroimag<strong>in</strong>g (preferably<br />
MRI). Lumbar puncture should be considered if neuroimag<strong>in</strong>g is normal or if there are<br />
signs or symptoms of men<strong>in</strong>geal irritation (class III, level C).<br />
<strong>An</strong> approach to FNP is outl<strong>in</strong>ed <strong>in</strong> Figure 12–1.<br />
References<br />
Agost<strong>in</strong>is C, Caverni L, Mosch<strong>in</strong>i L, et al. (1992). Paralysis of fourth cranial nerve due to superior cerebellar artery.<br />
<strong>Neuro</strong>logy 42:457–458.<br />
Arruga J, De Rivas P, Esp<strong>in</strong>et HL, et al. (1991). Chronic isolated trochlear nerve palsy produced by <strong>in</strong>tracavernous<br />
<strong>in</strong>ternal carotid artery aneurysm: report of a case. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:104–108.<br />
Baker RS, Epste<strong>in</strong> AD. (1991). Ocular motor abnormalities from head trauma. Surv Ophthalmol 35:245–267.<br />
Barr DB, McFadzean RM, Hadley D, et al. (1997). Acquired bilateral superior oblique palsy: a localiz<strong>in</strong>g sign <strong>in</strong> the<br />
dorsal midbra<strong>in</strong> syndrome. Eur J Ophthalmol 7:271–276.<br />
Berlit P. (1991). Isolated and comb<strong>in</strong>ed pareses of cranial nerves III, IV, and VI: a retrospective study of 412<br />
patients. J <strong>Neuro</strong>l Sci 103:10–15.<br />
Boggan JE, Rosenblum ML, Wilson CB. (1979). Neurilemmoma of the fourth cranial nerve. J <strong>Neuro</strong>surg 50:519–521.<br />
Botelho PJ, Giangiacomo JG. (1996). Autosomal-dom<strong>in</strong>ant <strong>in</strong>heritance of congenital superior oblique palsy.<br />
Ophthalmology 103:1508–1511.<br />
Brazis PW. (1993). Palsies of the trochlear nerve: diagnosis and localization-recent concepts. Mayo Cl<strong>in</strong> Proc 68:501.<br />
Brazis PW, Masdeu JC, Biller J. (2001). Localization <strong>in</strong> <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>logy. 4th ed. Philadelphia, Lipp<strong>in</strong>cott Williams &<br />
Wilk<strong>in</strong>s.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1992). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. St. Louis, Mosby Year<br />
Book, pp. 289–311.<br />
Burger LJ, Kalv<strong>in</strong> NH, Smith JL. (1970). Acquired lesions of the fourth cranial nerve. Bra<strong>in</strong> 93:567–574.<br />
Carter N, Miller NR. (1997). Fourth nerve palsy caused by Ehrlichia chaffeensis. J <strong>Neuro</strong>-Ophthalmol 17:47–50.<br />
Celli P, Ferrante L, Acqui M, et al. (1992). Neur<strong>in</strong>oma of the third, fourth, and sixth cranial nerves: a survey and<br />
report of a new fourth nerve case. Surg <strong>Neuro</strong>l 38:216–224.<br />
Chang-God<strong>in</strong>ich A, Lee AG, Brazis PW, et al. (1997). Complete ophthalmoplegia after zoster ophthalmicus. J<br />
<strong>Neuro</strong>-Ophthalmol 17:262–265.<br />
Coll<strong>in</strong>s TE, Mehalic TF, White TK, et al. (1992). Trochlear nerve palsy as the sole <strong>in</strong>itial sign of an aneurysm of the<br />
superior cerebellar artery. <strong>Neuro</strong>surgery 30:258–261.<br />
Coppeto JM, Lessell S. (1978). Cryptogenic unilateral paralysis of the superior oblique muscle. Arch Ophthalmol<br />
96:275–277.<br />
Eisenberg MB, Al-Mefty O, DeMonte F, Burson GT. (1999). Benign nonmen<strong>in</strong>geal tumors of the cavernous s<strong>in</strong>us.<br />
<strong>Neuro</strong>surgery 44:949–955.<br />
Elliot D, Cunn<strong>in</strong>gham Jr ET, Miller NR. (1991). Fourth nerve paresis and ipsilateral relative afferent pupillary<br />
defect without visual sensory disturbance: a sign of contralateral dorsal midbra<strong>in</strong> disease. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:169–172.<br />
Ellis FD, Helveston EM. (1976). Superior oblique palsy: diagnosis and classification. Int Ophthalmol Cl<strong>in</strong><br />
16:127–135.<br />
Fe<strong>in</strong>berg AS, Newman NJ. (1999). Schwannoma <strong>in</strong> patients with isolated trochlear nerve palsy. Am J Ophthalmol<br />
127:183–188.<br />
Ferreira R, Phan G, Bateman JB. (1997). Favorable visual outcome <strong>in</strong> cryptococcal men<strong>in</strong>gitis. Am J Ophthalmol<br />
124:558–560.<br />
FitzSimon JS, Toland J, Philips J, et al. (1995). Giant aneurysms <strong>in</strong> the cavernous s<strong>in</strong>us. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
15:59–65.<br />
Frohman LP, Grigorian R, Bielory L. (2001). <strong>Neuro</strong>-ophthalmic manifestations of sarcoidosis: cl<strong>in</strong>ical spectrum,<br />
evaluation, and management. J <strong>Neuro</strong>-Ophthalmol 21:132–137.
Fourth Nerve Palsies 293<br />
Galetta SL, Balcar LJ. (1998). Isolated fourth nerve palsy from midbra<strong>in</strong> hemorrhage. Case report. J <strong>Neuro</strong>-<br />
Ophthalmol 18:204–205.<br />
Gentry LR, Mehta RC, Appen RE, et al. (1991). MR imag<strong>in</strong>g of primary trochlear nerve neoplasms. AJNR 12:707–<br />
713.<br />
Gonyea EF. (1990). Superior oblique palsy due to a midbra<strong>in</strong> vascular malformation. <strong>Neuro</strong>logy 40:554–555.<br />
Green WR, Hackett ER, Schlez<strong>in</strong>ger NS. (1964). <strong>Neuro</strong>-ophthalmic evaluation of oculomotor nerve paralysis.<br />
Arch Ophthalmol 72:154–167.<br />
Hahn CD, Nicolle DA, Lownie SP, Drake CG. (2000). Giant cavernous carotid artery aneurysms: cl<strong>in</strong>ical<br />
presentation <strong>in</strong> fifty-seven cases. J <strong>Neuro</strong>-Ophthalmol 20:253–258.<br />
Hara N, Kan S, Simizu K. (2001). Localization of post-traumatic trochlear nerve palsy associated with hemorrhage<br />
at the subarachnoid space by magnetic resonance imag<strong>in</strong>g. Am J Ophthalmol 132:443–445.<br />
Harley RD. (1980). Paralytic strabismus <strong>in</strong> children: etiologic <strong>in</strong>cidence and management of the third, fourth, and<br />
sixth nerve palsies. Ophthalmology 87:24–43.<br />
Hashimoto M, Hoyt WF. (1996). Superficial siderosis and episodic fourth nerve paresis. Report of a case with<br />
cl<strong>in</strong>ical and magnetic resonance imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs. J <strong>Neuro</strong>-Ophthalmol 16:277–280.<br />
Herman M, Bobek-Billewicz B, Bullo B, et al. (1999). Wegener’s granulomatosis with unusual cavernous s<strong>in</strong>us and<br />
sella turcica extension. Eur Radiol 9:1859–1861.<br />
Herrendorf G, Ste<strong>in</strong>hoff BJ, Vadokas V, et al. (1997). Transitory fourth cranial nerve palsy due to foramen ovale<br />
electrode placement. Acta <strong>Neuro</strong>chur 139:789–790.<br />
Ho KL. (1981). Schwannoma of the trochlear nerve. J <strong>Neuro</strong>surg 55:132–135.<br />
Holmes JM, Mutyala S, Maus TL, et al. (1999). Pediatric third, fourth, and sixth nerve palsies: a population-based<br />
study. Am J Ophthalmol 127:388–392.<br />
Hoya K, Kir<strong>in</strong>o T. (2000). Traumatic trochlear nerve palsy follow<strong>in</strong>g m<strong>in</strong>or occipital impact—four case reports.<br />
<strong>Neuro</strong>l Med Chir 40:358–360.<br />
Jacobson DM, Moster ML, Eggenberger ER, et al. (1999). Isolated trochlear nerve palsy <strong>in</strong> patients with multiple<br />
sclerosis. <strong>Neuro</strong>logy 53:877–879.<br />
Jacobson DM, Warner JJ, Choucair AK, et al. (1988). Trochlear nerve palsy follow<strong>in</strong>g m<strong>in</strong>or head trauma: a sign of<br />
structural disorder. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 8:263–268.<br />
Jacobson DM, Warner JJ, Ruggles KH. (1995). Transient trochlear nerve palsy follow<strong>in</strong>g anterior temporal<br />
lobectomy for epilepsy. <strong>Neuro</strong>logy 45:1465.<br />
Khawam E, Scott AB, Jampolsky A. (1967). Acquired superior oblique palsy. Arch Ophthalmol 77:761–768.<br />
Keane JR. (1993). Fourth nerve palsy: historical review and study of 215 patients. <strong>Neuro</strong>logy 43:2439–2443.<br />
Keane JR. (1996). Cavernous s<strong>in</strong>us syndrome. <strong>An</strong>alysis of 151 cases. Arch <strong>Neuro</strong>l 53:967–971.<br />
Kim JS, Kang JK. (1992). Contralateral trochlear nerve palsy and facial sensory change due to a probable<br />
bra<strong>in</strong>stem vascular malformation. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:59–62.<br />
Kim JS, Kang JK, Lee SA, et al. (1993). Isolated or predom<strong>in</strong>ant ocular motor nerve palsy as a manifestation of<br />
bra<strong>in</strong> stem stroke. Stroke 24:581–586.<br />
K<strong>in</strong>g JS. (1976). Trochlear nerve sheath tumor. J <strong>Neuro</strong>surg 44:245–247.<br />
Krohel GB, Mansour AM, Petersen WL, et al. (1982). Isolated trochlear nerve palsy secondary to a juvenile<br />
pilocytic astrocytoma. J Cl<strong>in</strong> <strong>Neuro</strong>-Ophthalmol 2:119–123.<br />
Ksiazek S, Behar R, Sav<strong>in</strong>o PJ, et al. (1988). Isolated acquired fourth nerve palsies. <strong>Neuro</strong>logy 38(suppl 1):246.<br />
Landolfi JC, Thaler HT, De <strong>An</strong>gelis LM. (1998). Adult bra<strong>in</strong>stem gliomas. <strong>Neuro</strong>logy 51:1136–1139.<br />
Lee AG. (1995). Fourth nerve palsy <strong>in</strong> pseudotumor cerebri. Strabismus 3:57–59.<br />
Leiba H, Siatkowski RM, Culbertson WW, Glaser JS. (1996). <strong>Neuro</strong>sarcoidosis present<strong>in</strong>g as an <strong>in</strong>tracranial mass<br />
<strong>in</strong> childhood. J <strong>Neuro</strong>-ophthalmol 16:269–273.<br />
Lepore FE. (1995). Disorders of ocular motility follow<strong>in</strong>g head trauma. Arch <strong>Neuro</strong>l 52:924–926.<br />
Leunda G, Vaquero J, Cabezudo J, et al. (1982). Schwannoma of the oculomotor nerves: report of four cases. J<br />
<strong>Neuro</strong>surg 57:563–565.<br />
Lopes DK, Mericle RA, Wakhloo AK, et al. (1998). Cavernous s<strong>in</strong>us syndrome dur<strong>in</strong>g balloon test occlusion of the<br />
cervical <strong>in</strong>ternal carotid artery. Report of two cases. J <strong>Neuro</strong>surg 89:667–670.<br />
Mansour AM, Bailey BJ. (1997). Ocular f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Ramsay Hunt syndrome. J <strong>Neuro</strong>-Ophthalmology<br />
17:199–201.<br />
McK<strong>in</strong>na AJ. (1983). Eye signs <strong>in</strong> 611 cases of posterior fossa aneurysms: their diagnostic and prognostic value.<br />
Can J Ophthalmol 18:3–6.<br />
Mielke C, Alexander MSM, <strong>An</strong>and N. (2001). Isolated bilateral trochlear nerve palsy as the first cl<strong>in</strong>ical sign of a<br />
metastatic bronchial carc<strong>in</strong>oma. Am J Ophthalmol 132:593–594.<br />
Miller NR. (1989). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams & Wilk<strong>in</strong>s, p. 686.
294 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Mittleman D, Folk E. (1976). The evaluation and treatment of superior oblique muscle palsy. Trans Am Acad<br />
Ophthalmol Otolaryngol 81:893–898.<br />
Mon Y. (1996). Midbra<strong>in</strong> hemorrhage present<strong>in</strong>g with trochlear nerve palsy—-a case report. R<strong>in</strong>sho Sh<strong>in</strong>keigaki<br />
36:71–73.<br />
Müri RM, Baumgartner RW. (1995). Horner’s syndrome and contralateral trochlear nerve palsy. <strong>Neuro</strong><strong>ophthalmology</strong><br />
15:161.<br />
Neetens A, Van Aerde F. (1981). Extra-ocular muscle palsy from m<strong>in</strong>or head trauma. Initial sign of <strong>in</strong>tracranial<br />
tumor. Bull Soc Belge Ophthalmol 193:161–167.<br />
Nemet P, Godel V, Baruch E, et al. (1980). Benign palsy of the superior oblique. J Pediatr Ophthalmol Strabismus<br />
17:320–322.<br />
Newman NJ, Slamovits, Friedland S, et al. (1995). <strong>Neuro</strong>-ophthalmic manifestations of men<strong>in</strong>gocerebral<br />
<strong>in</strong>flammation from the limited form of Wegener’s granulomatosis. Am J Ophthalmol 120:613–621.<br />
Orwitz JI, Galetta SL, Teener JW. (1997). Bilateral trochlear nerve palsy and downbeat nystagmus <strong>in</strong> a patient with<br />
cephalic tetanus. <strong>Neuro</strong>logy 9:894–895.<br />
Ohtsuka K, Hashimoto M, Nakamura Y. (1998). Bilateral trochlear nerve palsy with arachnoid cyst of the<br />
quadrigem<strong>in</strong>al cistern. Am J Ophthalmol 125:268–270.<br />
Patton N, Beatty S, Lloyd IC. (2000). Bilateral sixth and fourth cranial nerve palsies <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension. J R Soc Med 93:80–81.<br />
Petermann SH, Newman NJ. (1999). Pituitary macroadenoma manifest<strong>in</strong>g as an isolated fourth nerve palsy. Am J<br />
Ophthalmol 127:235–236.<br />
Polito E, Leccisotti A. (1996). Pa<strong>in</strong>ful ophthalmoplegia caused by superior ophthalmic ve<strong>in</strong> thrombosis. <strong>Neuro</strong><strong>ophthalmology</strong><br />
16:189–192.<br />
Reich KA, Giansiracusa DR, Strongwater SL. (1990). <strong>Neuro</strong>logic manifestations of giant cell arteritis. Am J Med<br />
89:67–72.<br />
Richards BW, Jones FR Jr, Younge BR. (1992). Causes and prognosis <strong>in</strong> 4,278 cases of paralysis of the oculomotor,<br />
trochlear, and abducens cranial nerves. Am J Ophthalmol 113:489–496.<br />
Robb RM. (1990). Idiopathic superior oblique palsies <strong>in</strong> children. J Pediatr Ophthalmol Strabismus 27:66–69.<br />
Robert CM, Geigenbaum JA, Stern WE. (1973). Ocular palsy occurr<strong>in</strong>g with pituitary tumors. J <strong>Neuro</strong>surg<br />
38:17–19.<br />
Rucker CW. (1958). Paralysis of the third, fourth and sixth cranial nerves. Am J Ophthalmol 46:787–794.<br />
Rucker CW. (1966). The causes of paralysis of the third, fourth, and sixth cranial nerves. Am J Ophthalmol<br />
61:1293–1298.<br />
Rucker C, Dyer J, Smith D, et al. (1956). The causes of acquired paralysis of the ocular muscles. Am J Ophthalmol<br />
41:951–955.<br />
Rush JA, Younge BR. (1981). Paralysis of cranial nerves III, IV, and VI. Arch Ophthalmol 99:76–79.<br />
Sabates NR, Gonce MA, Farris BK. (1991). <strong>Neuro</strong>-ophthalmological f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> closed head trauma. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:273–277.<br />
Sadun F, De Negri AM, Santopadre P, Pezzi PP. (1999). Bilateral trochlear nerve palsy associated with cryptococcal<br />
men<strong>in</strong>gitis <strong>in</strong> human immunodeficiency virus <strong>in</strong>fection. J <strong>Neuro</strong>-Ophthalmol 19:118–119.<br />
Santoreneos S, Hanieh A, Jorgensen RE. (1997). Trochlear nerve schwannomas occurr<strong>in</strong>g <strong>in</strong> patients without<br />
neurofibromatosis: case report and review of the literature. <strong>Neuro</strong>surgery 41:282–287.<br />
Scully RE, Galdab<strong>in</strong>i JJ, McNeely BU. (1976). Case records of the Massachusetts General Hospital: weekly<br />
cl<strong>in</strong>icopathological exercises. Case 36-1976. N Engl J Med 295:553–561.<br />
Shimo-oku M, Izaki A, Shim-myo A. (1998). Fourth nerve palsy as an <strong>in</strong>itial sign of <strong>in</strong>ternal carotid-posterior<br />
communicat<strong>in</strong>g artery aneurysm. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:185–190.<br />
Sh<strong>in</strong>mei Y, Harada T, Ohashi T, et al. (1997). Trochlear nerve palsy associated with superficial siderosis of the<br />
central nervous system. Japanese J Ophthalmol 41:19–22.<br />
Shrader EC, Schlez<strong>in</strong>ger NS. (1960). <strong>Neuro</strong>-ophthalmologic evaluation of abducens nerve paralysis. Arch<br />
Ophthalmol 63:84–91.<br />
Slav<strong>in</strong> ML. (1987). Isolated trochlear nerve palsy secondary to cavernous s<strong>in</strong>us men<strong>in</strong>gioma. Am J Ophthalmol<br />
104:433–434.<br />
Speer C, Pearlman J, Phillips PH, et al. (1999). Fourth nerve palsy <strong>in</strong> pediatric pseudotumor cerebri. Am J<br />
Ophthalmol 127:236–237.<br />
Suzuki J, Wada T, Kowada M. (1962). <strong>Cl<strong>in</strong>ical</strong> observations on tumors of the p<strong>in</strong>eal region. J <strong>Neuro</strong>surg<br />
19:441–445.<br />
Tachibana H, Mimura O, Shiomi M, et al. (1990). Bilateral trochlear nerve palsies from a bra<strong>in</strong>stem hematoma. J<br />
Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:35–37.
Fourth Nerve Palsies 295<br />
Tanaka H, Yuki N, Hirata K. (1998). Trochlear nerve enhancement on three-dimensional magnetic resonance<br />
imag<strong>in</strong>g <strong>in</strong> Fisher syndrome. Am J Ophthalmol 126:322–324.<br />
Thömke F, Hopf HC. (2000). Isolated superior oblique palsies with electrophysiologically documented bra<strong>in</strong>stem<br />
lesions. Muscle Nerve 23:267–270.<br />
Thömke F, R<strong>in</strong>gel K. (1999). Isolated superior oblique palsies with bra<strong>in</strong>stem lesions. <strong>Neuro</strong>logy 53:1126–1127.<br />
Tsai RK, Chen HY, Wang HZ. (2000). Pa<strong>in</strong>ful fourth cranial nerve palsy caused by posteriorly-dra<strong>in</strong><strong>in</strong>g dural<br />
carotid-cavernous s<strong>in</strong>us fistula. J Formosan Med Assoc 99:730–732.<br />
Ulrich A, Kaiser HJ. (1998). Bilateral trochlear nerve palsy <strong>in</strong> systemic lupus erythematosus (SLE). <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:28.<br />
Vanooteghem P, Dehaene I, Van Zandycke M, et al. (1992). Comb<strong>in</strong>ed trochlear nerve palsy and <strong>in</strong>ternuclear<br />
ophthalmoplegia. Arch <strong>Neuro</strong>l 49:108–109.<br />
Varrato J, Galetta S. (2000). Fourth nerve palsy unmasked by botul<strong>in</strong>um tox<strong>in</strong> therapy for cervical torticollis.<br />
<strong>Neuro</strong>logy 55:896.<br />
Vitali C, Genovesi-Ebert F, Romani A, et al. (1996). Ophthalmological and neuro-ophthalmological <strong>in</strong>volvement <strong>in</strong><br />
Churg-Strauss syndrome: a case report. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 234:404–408.<br />
von Noorden GK, Helveston EM. (1994). Strabismus: A Decision Mak<strong>in</strong>g Approach. St. Louis, Mosby, pp. 162–169.<br />
von Noorden GK, Murray E, Wong SY. (1986). Superior oblique paralysis. A review of 270 cases. Arch Ophthalmol<br />
104:1771–1776.<br />
Wise BL, Palub<strong>in</strong>skas AJ. (1965). Persistent trigem<strong>in</strong>al artery (carotid-basilar anastomosis). J <strong>Neuro</strong>surg 21:199–206.<br />
Wong AMF, Sharpe JA. (1996). Fourth nerve palsy <strong>in</strong> migra<strong>in</strong>e. <strong>Neuro</strong>-Ophthalmology 16:51–54.<br />
Wray SH. (1977). <strong>Neuro</strong>-ophthalmologic manifestations of pituitary and parasellar lesions. Cl<strong>in</strong> <strong>Neuro</strong>surg<br />
24:86–117.<br />
Wright HG, Hansotia P. (1977). Isolated fourth cranial nerve palsies: etiology and prognosis. Wis Med J 76:26–28.<br />
Yamamoto M, Jimbo M, Ide M, et al. (1987). Trochlear neur<strong>in</strong>oma. Surg <strong>Neuro</strong>l 28:287–290.<br />
Younge BR, Sutula F. (1977). <strong>An</strong>alysis of trochlear nerve palsies. Mayo Cl<strong>in</strong> Proc 52:11–18.
13 r<br />
Sixth Nerve Palsies<br />
What is the <strong>An</strong>atomy of the Sixth Nerve?<br />
The paired abducens nuclei are located <strong>in</strong> the dorsal lower portion of the pons,<br />
separated from the floor of the fourth ventricle by the genu of the facial nerve (facial<br />
colliculus). The nucleus conta<strong>in</strong>s motor neurons for the lateral rectus muscle and<br />
<strong>in</strong>terneurons travel<strong>in</strong>g via the medial longitud<strong>in</strong>al fasciculus (MLF) to the contralateral<br />
medial rectus subnucleus of the third nerve. The sixth nerve nucleus thus conta<strong>in</strong>s all<br />
the neurons responsible for horizontal conjugate gaze. The nerve fascicle leaves the<br />
nucleus and travels with<strong>in</strong> the substance of the pont<strong>in</strong>e tegmentum, adjacent to<br />
the medial lemniscus and the corticosp<strong>in</strong>al tract. The sixth nerve leaves the bra<strong>in</strong>stem<br />
<strong>in</strong> the horizontal sulcus between the pons and medulla (lateral to the corticosp<strong>in</strong>al<br />
bundles). It enters the subarachnoid space, ascends along the base of the pons <strong>in</strong> the<br />
prepont<strong>in</strong>e cistern, courses nearly vertically along the clivus, and travels over the<br />
petrous apex of the temporal bone where it is tethered at the petrocl<strong>in</strong>oid (Grüber’s)<br />
ligament <strong>in</strong> Dorello’s canal beneath. It enters the substance of the cavernous s<strong>in</strong>us lateral<br />
to the <strong>in</strong>ternal carotid artery and medial to the ophthalmic division of the trigem<strong>in</strong>al<br />
nerve (V1) to enter the orbit via the superior orbital fissure. In their course from the<br />
pericarotid plexus to the ophthalmic branch of the trigem<strong>in</strong>al nerve, the pupil’s<br />
sympathetic fibers jo<strong>in</strong> the abducens nerve for a few millimeters.<br />
Based on this topographic anatomy, sixth nerve palsies (SNPs) may be categorized as<br />
isolated or nonisolated. The criteria for the diagnosis of an isolated SNP are listed <strong>in</strong><br />
Table 13–1. We def<strong>in</strong>e six types of SNP <strong>in</strong> Table 13–2. These types help to differentiate<br />
etiology and guide the management of SNP. The localization of nonisolated SNPs is<br />
outl<strong>in</strong>ed <strong>in</strong> Table 13–3.<br />
Is the Nonisolated SNP Due to a Pont<strong>in</strong>e<br />
(Lower Pons) Lesion?<br />
Sixth nerve nuclear lesions cause a horizontal gaze palsy, rather than an isolated<br />
abduction deficit. <strong>An</strong> ipsilateral facial palsy may occur because of the close proximity<br />
296
Table 13–1. Criteria for the Diagnosis of Unilateral Isolated Sixth Nerve Palsy (SNP)<br />
Ipsilateral abduction deficit<br />
Incomitant esodeviation that is typically worsened with gaze <strong>in</strong>to the field of the weak lateral<br />
rectus muscle (may become comitant over time)<br />
Exclusion of Duane’s retraction syndrome, spasm of the near reflex, and other causes of abduction<br />
deficits that can mimic an SNP, and exclusion of patients with signs of the follow<strong>in</strong>g:<br />
Orbital disease (e.g., chemosis, proptosis, lid swell<strong>in</strong>g, <strong>in</strong>jection, and positive forced ductions)<br />
Myasthenia gravis (e.g., ptosis, Cogan’s lid twitch sign, orbicularis oculi weakness, muscle<br />
fatigue or variability)<br />
Multiple cranial nerve palsies (<strong>in</strong>clud<strong>in</strong>g bilateral SNP) or radiculopathy<br />
Bra<strong>in</strong>stem signs (e.g., Horner’s syndrome, hemiplegia, cerebellar signs)<br />
Systemic, <strong>in</strong>fectious, or <strong>in</strong>flammatory risk factors for an SNP (e.g., history of previous malignancy,<br />
giant cell arteritis, collagen vascular disease)<br />
Exclusion of patients with severe headache<br />
of the facial and abducens nerve <strong>in</strong> the pons. Nuclear lesions are usually associated with<br />
other bra<strong>in</strong>stem signs (e.g., hemiparesis, hemisensory loss, a central Horner’s<br />
syndrome). Likewise, lesions of the sixth nerve fascicle <strong>in</strong>volve adjacent structures<br />
(e.g., cranial nerves V, VII, and VIII; cerebellar ataxia; a central Horner’s syndrome; or<br />
contralateral hemiplegia). Patients with a presumed nuclear or fascicular SNP should<br />
undergo neuroimag<strong>in</strong>g (usually magnetic resonance imag<strong>in</strong>g [MRI]) directed to the<br />
pons. The etiologies of nuclear or fascicular lesions <strong>in</strong> the pons are listed <strong>in</strong> Table 13–4.<br />
Table 13–2. Def<strong>in</strong>itions of the Six Types of Sixth Nerve Palsy<br />
Sixth Nerve Palsies 297<br />
Type 1: nonisolated<br />
SNP are considered nonisolated <strong>in</strong> the presence of the exclusionary conditions outl<strong>in</strong>ed <strong>in</strong> Table<br />
13–1<br />
Type 2: traumatic<br />
Isolated unilateral SNPs that have a clearly established temporal relationship to significant<br />
previous head trauma and do not progress are considered traumatic <strong>in</strong> orig<strong>in</strong>; patients with SNP<br />
follow<strong>in</strong>g m<strong>in</strong>or head trauma are excluded.<br />
Type 3: congenital<br />
Patients born with SNP<br />
Type 4: vasculopathic<br />
Vasculopathic SNPs occur <strong>in</strong> patients older than age 55 or those with known vasculopathic risk<br />
factors (e.g., hypertension or diabetes)<br />
Type 5: nonvasculopathic<br />
Patients without vasculopathic risk factors def<strong>in</strong>ed above are considered to have nonvasculopathic<br />
SNP<br />
Type 6: progressive (nonisolated) or unresolved<br />
SNPs that worsen after the acute stage (more than 2 weeks) as def<strong>in</strong>ed by a significant <strong>in</strong>crease <strong>in</strong><br />
the measured ocular deviation or who develop new neurologic f<strong>in</strong>d<strong>in</strong>gs are considered<br />
progressive or nonisolated; patients without resolution <strong>in</strong> the measured horizontal deviation<br />
after 12 to 16 weeks are considered unresolved
298 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 13–3. The Localization of Abducens Nerve Lesions<br />
Structure Involved <strong>Cl<strong>in</strong>ical</strong> Presentation<br />
Nuclear lesions<br />
Abducens nucleus Horizontal gaze palsy<br />
Möbius syndrome (gaze palsy with facial diplegia)<br />
Duane’s retraction syndrome (gaze palsy with globe<br />
retraction and narrow<strong>in</strong>g of palpebral fissure with<br />
adduction)<br />
Dorsolateral pons Ipsilateral gaze palsy, facial paresis, dysmetria;<br />
occasionally with contralateral hemiparesis (Foville<br />
syndrome)<br />
Lesions of the abducens fascicle<br />
Abducens fascicle Isolated CN VI palsy<br />
<strong>An</strong>terior paramedial pons Ipsilateral CN VI palsy, ipsilateral CN VII palsy,<br />
contralateral hemiparesis (Millard-Gubler)<br />
Prepont<strong>in</strong>e cistern May have contralateral hemiparesis<br />
Lesion of abducens nerve (subarachnoid, petrous)<br />
Petrous apex (Dorello’s canal) CN VI palsy, deafness, facial (especially retro-orbital) pa<strong>in</strong><br />
(Gradenigo)<br />
Cavernous s<strong>in</strong>us Isolated CN VI palsy; CN VI palsy plus Horner’s<br />
syndrome; also may affect CN III, IV, VI<br />
Superior orbital fissure syndrome CN VI palsy with variable affection of CN III, IV, VI;<br />
proptosis<br />
Orbit CN VI palsy; visual loss; variable proptosis, chemosis, lid<br />
swell<strong>in</strong>g<br />
Source: Modified from Brazis, 2001, with permission from Lipp<strong>in</strong>cott Williams & Wilk<strong>in</strong>s.<br />
Is the SNP Due to a Subarachnoid Space<br />
Lesion?<br />
Lesions of the subarachnoid space may result <strong>in</strong> unilateral or bilateral SNP. This SNP is<br />
a nonlocaliz<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>g because any cause of <strong>in</strong>creased <strong>in</strong>tracranial pressure may result<br />
<strong>in</strong> an SNP (see Table 13–3). Patients with a subarachnoid space lesion should undergo<br />
neuroimag<strong>in</strong>g directed to this location followed by a lumbar puncture (LP) as needed<br />
(class III–IV, level B).<br />
Is the SNP the Result of a Lesion of the<br />
Petrous Apex?<br />
Lesions of the petrous apex caus<strong>in</strong>g SNP are associated with other neurologic f<strong>in</strong>d<strong>in</strong>gs,<br />
<strong>in</strong>clud<strong>in</strong>g <strong>in</strong>volvement of other cranial nerves (e.g., fifth, seventh, and eighth) or facial<br />
pa<strong>in</strong>. <strong>Neuro</strong>imag<strong>in</strong>g should be directed toward the petrous apex (MRI or computed<br />
tomography [CT] for bone <strong>in</strong>volvement) (class III–IV, level B).
Table 13–4. Etiology of a Sixth Nerve Palsy by Topographical Localization<br />
Nuclear (horizontal gaze)<br />
Congenital (Carr, 1997)<br />
Möbius syndrome (Carr, 1997; Pedraza, 2000)<br />
Demyel<strong>in</strong>at<strong>in</strong>g<br />
Infarction or ischemia<br />
Neoplasm (pont<strong>in</strong>e and cerebellar)<br />
Glioma<br />
Metastasis<br />
Histiocytosis X<br />
Trauma<br />
Wernicke-Korsakoff syndrome<br />
Fascicular<br />
Demyel<strong>in</strong>ation (Thömke, 1997, 1998)<br />
Infarction (K<strong>in</strong>g, 1995; Lopez, 1996; Thömke, 1998)<br />
Neoplasm (Balcar, 1999; Landolfi, 1998; Thömke, 1998)<br />
Trauma<br />
Hematoma (Thömke, 1998)<br />
Migra<strong>in</strong>e (Lee, 2002)<br />
Subarachnoid<br />
<strong>An</strong>eurysm or vascular abnormality (K<strong>in</strong>g, 1995)<br />
Persistent primitive trigem<strong>in</strong>al artery<br />
Posterior <strong>in</strong>ferior cerebellar aneurysm<br />
Vertebral artery, <strong>in</strong>clud<strong>in</strong>g elongated vessel (Narai, 2000; Ohtsuka, 1996)<br />
Carc<strong>in</strong>omatous or leukemic men<strong>in</strong>gitis (Wolfe, 1994)<br />
Chiari malformation or basilar impression (Chavis, 1998; Hirose, 1998; K<strong>in</strong>g, 1995)<br />
Follow<strong>in</strong>g procedures<br />
Cervical traction<br />
Lumbar puncture (Thömke, 2000)<br />
Myelography (Bell, 1990)<br />
Postvacc<strong>in</strong>ation<br />
Radiculography (D<strong>in</strong>akaran, 1995; Lloyd, 1995)<br />
Shunt<strong>in</strong>g for hydrocephalus<br />
Sp<strong>in</strong>al or epidural anesthesia (De Veuster, 1994)<br />
Intrathecal glucocorticoid <strong>in</strong>jection (Dumont, 1998)<br />
Inflammatory<br />
Retropharyngeal space <strong>in</strong>flammation (Fanous, 1992)<br />
Necrotiz<strong>in</strong>g vasculitis<br />
Sarcoidosis (Frohman, 2001; Sachs, 1990)<br />
Systemic lupus erythematosus<br />
Wegener’s granulomatosis<br />
Fisher’s syndrome (Nagaoka, 1996)<br />
Infectious<br />
Lyme disease (Lesser, 1990; Mastrianni, 1994)<br />
Syphilis (Slav<strong>in</strong>, 1992; Stepper, 1998)<br />
Tuberculosis<br />
Sixth Nerve Palsies 299<br />
(cont<strong>in</strong>ued)
300 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 13–4. (cont<strong>in</strong>ued)<br />
Cryptococcal men<strong>in</strong>gitis<br />
Cysticercosis (Keane, 2000)<br />
HIV-CMV encephalitis<br />
Neoplasm (Hashimoto, 1998)<br />
Abducens nerve tumor (Ichimi, 1997; Okada, 1997; Tung, 1991)<br />
Cerebellopont<strong>in</strong>e angle tumor<br />
Clivus tumor (e.g., chordoma, chondrosarcoma, plasmacytoma) (Balcar, 1999; Forsyth, 1993;<br />
Harada, 1997; Mekari-Sabbagh, 2001; Movsas, 2000; Volpe, 1993b)<br />
Leukemia (Averbuch-Heller, 1994)<br />
Metastatic (O’Boyle, 1992)<br />
Skull base tumor (Keane, 2000; Volpe, 1993a)<br />
Nasopharyngeal carc<strong>in</strong>oma (Keane, 2000)<br />
Trigem<strong>in</strong>al nerve tumor<br />
Capillary hemangioma of Meckel’s cave (Brazis, 2000)<br />
Nonlocaliz<strong>in</strong>g sign of <strong>in</strong>creased <strong>in</strong>tracranial pressure (Aroichane, 1995)<br />
Pseudotumor cerebri (Krishna, 1998; Patton, 2000)<br />
Men<strong>in</strong>gitis or men<strong>in</strong>geal irritation of any type<br />
Intracranial tumor<br />
Venous s<strong>in</strong>us thrombosis (Biousse, 1999)<br />
Spontaneous cerebrosp<strong>in</strong>al fluid leak with <strong>in</strong>tracranial hypotension (Apte, 1999; Case Records of<br />
the Massachusetts General Hospital, 1998; Ferrante, 1998; Horton, 1994; Mokri, 1997; O’Carroll,<br />
1999; Schiev<strong>in</strong>k, 1996)<br />
Trauma (exclud<strong>in</strong>g surgical) (Hollis, 1997; Holmes, 1998; Lepore, 1995)<br />
Epidural hematoma of clivus (Mizushima, 1998)<br />
Petrous apex<br />
Neoplasm (e.g., nasopharyngeal carc<strong>in</strong>oma)<br />
Infection<br />
Complicated otitis media (Homer, 1996)<br />
Mastoiditis (Gradenigo’s syndrome) (Davé, 1997)<br />
Thrombosis of <strong>in</strong>ferior petrosal or transverse=sigmoid s<strong>in</strong>us (Kuehnen, 1998)<br />
Trauma (<strong>An</strong>toniades, 1993; Mutyala, 1996)<br />
Basilar skull fracture<br />
Inflammatory<br />
Cavernous s<strong>in</strong>us (Keane, 1996)<br />
Cavernous s<strong>in</strong>us thrombosis (Kriss, 1996)<br />
Cavernous s<strong>in</strong>us fistula (Eggenberger, 2000; K<strong>in</strong>g, 1995; Lee, 1998; Uehara, 1998)<br />
Superior ophthalmic ve<strong>in</strong> thrombosis (Polito, 1996)<br />
Neoplasm (Eisenberg, 1999)<br />
Nasopharyngeal carc<strong>in</strong>oma (Keane, 1996)<br />
Pituitary adenoma<br />
Plasmacytoma (Bachmeyer, 1997)<br />
Lymphoma (Keane, 1996; Liu, 1993; Roman-Goldste<strong>in</strong>, 1998)<br />
Hodgk<strong>in</strong>’s disease (Kasner, 1996)<br />
Hemangioma (Brazis, 2000; Lee, 1995)<br />
Hemangioendothelioma (Phookan, 1998)<br />
Men<strong>in</strong>gioma (Kawase, 1996)<br />
(cont<strong>in</strong>ued)
Table 13–4. (cont<strong>in</strong>ued)<br />
Sixth Nerve Palsies 301<br />
Rhabdomyosarcoma (Arita, 2001)<br />
Sixth nerve tumors<br />
Sphenoid s<strong>in</strong>us tumors<br />
Skull base tumors<br />
Squamous cell cancer of pterygopalat<strong>in</strong>e fossa<br />
Subarachnoid diverticulum<br />
Sphenoid s<strong>in</strong>us mucocele (Muneer, 1997)<br />
Ischemia<br />
Inflammatory or <strong>in</strong>fectious<br />
Herpes zoster (Chang-God<strong>in</strong>ich, 1997; Mansour, 1997; Smith, 1993)<br />
Act<strong>in</strong>omycoses (Holland, 1998)<br />
Tolosa-Hunt syndrome<br />
Internal carotid artery diseases<br />
<strong>An</strong>eurysm (FitzSimon, 1995; Hahn, 2000; Keane, 1996; Silva, 1999)<br />
Dissection (Kerty, 1999; Lemesle, 1998)<br />
Dolichoectasia (Blumenthal, 1997; Neugebauer, 2001)<br />
Balloon test occlusion (Lopes, 1998)<br />
Cisplat<strong>in</strong> <strong>in</strong>fusion (Alderson, 1996; Wu, 1997)<br />
Post–radiofrequency rhizotomy for trigem<strong>in</strong>al neuralgia (Harrigan, 1998)<br />
Orbital lesions<br />
Neoplastic (orbital schwannoma)<br />
Inflammation (orbital <strong>in</strong>flammatory pseudotumor)<br />
Infectious<br />
Traumatic (Lazow, 1995)<br />
Localization uncerta<strong>in</strong><br />
Infectious mononucleosis<br />
Mycoplasma pneumoniae <strong>in</strong>fection (Wang, 1998)<br />
Lyme disease (Mikkilä, 2000)<br />
Campylobacter jejuni enteritis (Roberts, 1995)<br />
Creutzfeldt-Jakob disease (Ifergane, 1998)<br />
Progressive multifocal leukoencephalopathy (PML) <strong>in</strong> AIDS (Ormerod, 1996)<br />
Lymphoma (Shaw, 1997)<br />
Bone marrow transplantation treatment with cyclospor<strong>in</strong>e and ganciclovir (Openshaw, 1997)<br />
3,4-Methyl-enedioxymetamphetam<strong>in</strong>e (MDMA, or ‘‘ecstasy’’) abuse (Schroeder, 2000)<br />
Guilla<strong>in</strong>-Barré syndrome (Ropper, 1993)<br />
Fisher’s syndrome (Al-D<strong>in</strong>, 1994; Chiba, 1992, 1993; Igarashi, 1992; Suzuki, 1998)<br />
Associated with anti-GQ1b IgG antibody (Sato, 2001)<br />
Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (CIDP) (Ropper, 1991; Wokke,<br />
1996)<br />
Un<strong>in</strong>tentional subdural catheter (Haughton, 1999)<br />
Pregnancy (Fung, 1999)
302 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Is the SNP Due to a Cavernous S<strong>in</strong>us Lesion?<br />
With lesions of the cavernous s<strong>in</strong>us, SNPs usually occur <strong>in</strong> association with other cranial<br />
neuropathies (e.g., third, fourth, or fifth nerves) or a Horner’s syndrome. <strong>Neuro</strong>imag<strong>in</strong>g<br />
(usually MRI) should be directed to the cavernous s<strong>in</strong>us (class III–IV, level B). Table 13–3<br />
lists the etiologies of a cavernous s<strong>in</strong>us lesion caus<strong>in</strong>g an SNP.<br />
Is the SNP Due to an Orbital Lesion?<br />
Lesions of the orbit caus<strong>in</strong>g SNP are usually associated with other orbital signs such as<br />
proptosis or chemosis. <strong>Neuro</strong>imag<strong>in</strong>g (preferably MRI) should be directed to the orbit<br />
(class III–IV, level B).<br />
What are the Recommendations for the<br />
Evaluation of SNP?<br />
1. Nonisolated SNP (type 1) should undergo neuroimag<strong>in</strong>g and further evaluation<br />
(class III–IV, level B). Special attention should be directed to areas suggested<br />
topographically by the associated neurologic signs or symptoms (see above)<br />
(Gailbraith, 1994; Nemzek, 1995; O’Boyle, 1992; Steel, 1994; Volpe, 1993).<br />
2. Traumatic SNP (type 2) should undergo the appropriate acute neuroimag<strong>in</strong>g (CT<br />
scann<strong>in</strong>g) as <strong>in</strong>dicated by the trauma associated neurologic signs and symptoms<br />
(class IV, level C). In acute traumatic SNP, failure to recover by 6 months after onset<br />
was associated <strong>in</strong>dependently with the <strong>in</strong>ability to abduct past midl<strong>in</strong>e at presentation<br />
and bilaterality (Holmes, 2001).<br />
3. Congenital SNPs (type 3) are rare, and there are <strong>in</strong>sufficient data from our review of<br />
the literature to make a strong recommendation for the management of congenital<br />
isolated SNPs (class IV, level U). Nevertheless, if the SNP can be clearly demonstrated<br />
to be congenital <strong>in</strong> orig<strong>in</strong>, additional neuroimag<strong>in</strong>g is not generally required (class IV,<br />
level U). Transient SNPs may occur follow<strong>in</strong>g birth trauma <strong>in</strong> newborns. Gailbraith<br />
reported the <strong>in</strong>cidence of SNP <strong>in</strong> a group of 6886 neonates as be<strong>in</strong>g 0.4% (Gailbraith,<br />
1994). All of these SNPs (type 3) resolved with<strong>in</strong> 6 weeks, and we recommend that<br />
imag<strong>in</strong>g may be deferred <strong>in</strong> these patients. The <strong>in</strong>cidence of SNP <strong>in</strong>creased with<br />
‘‘complexity of <strong>in</strong>strumentation,’’ with 0% prevalence for cesarean section, 0.1%<br />
prevalence for spontaneous vag<strong>in</strong>al delivery, 2.4% prevalence for forceps delivery,<br />
and 3.2% for vacuum extraction. Leung reported three cases of right SNP after<br />
vag<strong>in</strong>al delivery that all resolved after 4 to 12 weeks (Leung, 1987). Observation for<br />
improvement is a reasonable approach <strong>in</strong> these cases (class IV, level C).<br />
4. Isolated vasculopathic SNPs (type 4) may be observed (without neuroimag<strong>in</strong>g) for<br />
improvement for 4 to 12 weeks (class III, level C). Rush and Younge reported a<br />
recovery rate of 49.6% <strong>in</strong> 419 nonselected SNP cases, and a higher rate of 71% <strong>in</strong> 419<br />
patients with diabetes mellitus, hypertension, or atherosclerosis (Rush, 1981). Some<br />
authors have recommended observ<strong>in</strong>g vasculopathic isolated SNP beyond a 3-month<br />
<strong>in</strong>terval of recovery if the esotropia and the abduction deficit were decreas<strong>in</strong>g (Burde,<br />
1992). Elderly patients who present with an isolated SNP and headache, scalp
Sixth Nerve Palsies 303<br />
tenderness, jaw claudication, or visual loss should undergo an appropriate evaluation<br />
for giant cell arteritis (class IV, level C). We recommend check<strong>in</strong>g the erythrocyte<br />
sedimentation rate and, when cl<strong>in</strong>ically <strong>in</strong>dicated, do<strong>in</strong>g a temporal artery biopsy<br />
(Reich, 1990). Patients with progression or lack of improvement (type 6) should<br />
undergo neuroimag<strong>in</strong>g (class IV, level C).3.62<br />
It should be noted that early progression of paresis over the course of 1 week <strong>in</strong><br />
vasculopathic SNP is not uncommon (Jacobson, 1996). In one study, only two of 35<br />
patients with ischemic SNP had <strong>in</strong>itial complete abduction deficits (Jacobson, 1996).<br />
Of 33 patients with <strong>in</strong>itial <strong>in</strong>complete deficits, 18 (55%) showed progression over a 1week<br />
period. We do not consider progression over the first week after onset to be a<br />
sign of nonvasculopathic SNP (class IV, level C).<br />
5. We recommend that nonvasculopathic SNP (type 5) should undergo neuroimag<strong>in</strong>g<br />
(Fanous, 1992; Gailbraith, 1994; Hashimoto, 1998; Kuehnen, 1998; O’Boyle, 1992;<br />
Rush, 1981; Sachs, 1990; Slav<strong>in</strong>, 1992; Steel, 1994; Straussberg, 1993; Tung, 1991;<br />
Uehara, 1998) (class III–IV, level C). Younger patients, or those without vasculopathic<br />
risk factors (type 5), could also undergo a more extensive evaluation <strong>in</strong>clud<strong>in</strong>g a<br />
fast<strong>in</strong>g blood glucose, complete blood cell count, and a blood pressure check for<br />
underly<strong>in</strong>g vasculopathy (class IV, level C). Other test<strong>in</strong>g, <strong>in</strong>clud<strong>in</strong>g neuroimag<strong>in</strong>g<br />
(MRI) and if necessary LP, is recommended (class IV, level C). Type 5 SNPs have a<br />
significant (27%) chance of harbor<strong>in</strong>g an underly<strong>in</strong>g malignant neoplasm (Sav<strong>in</strong>o,<br />
1986). Evaluation for myasthenia gravis should also be considered <strong>in</strong> these patients<br />
(class IV, level C).<br />
6. Test<strong>in</strong>g for vasculopathic risk factors <strong>in</strong> type 4 or type 5 SNP should be performed,<br />
even <strong>in</strong> the absence of a previous history of diabetes or hypertension (Burde, 1992;<br />
Sav<strong>in</strong>o, 1982; Watanabe, 1990). Ocular motor cranial neuropathies may be the<br />
present<strong>in</strong>g sign or only sign of underly<strong>in</strong>g vasculopathy <strong>in</strong> these patients (class IV,<br />
level C).<br />
7. Patients with progressive or unresolved SNP (type 6) and patients with new<br />
neurologic signs or symptoms should undergo neuroimag<strong>in</strong>g (Burde, 1992; Moster,<br />
1984; Sav<strong>in</strong>o, 1982, 1986). Patients with progressive or unresolved SNP should<br />
probably undergo neuroimag<strong>in</strong>g (class IV, level C). Galetta and Smith described 13<br />
patients with chronic SNP, that is, an SNP last<strong>in</strong>g 6 months or longer. Of these, four<br />
were idiopathic, four due to tumor, two were traumatic, one was postsp<strong>in</strong>al<br />
anesthesia, one was temporal arteritis, and one was <strong>in</strong>tracavernous aneurysm<br />
(Galetta, 1989). Sav<strong>in</strong>o et al reviewed 38 patients with chronic SNP (Sav<strong>in</strong>o, 1982).<br />
Fourteen (37%) were discovered to have an <strong>in</strong>tracranial lesion. These authors<br />
specifically recommended neuroradiologic <strong>in</strong>vestigation at onset <strong>in</strong> any patient<br />
with a history of carc<strong>in</strong>oma.<br />
Moster et al commented on the lack of truly isolated SNP reported <strong>in</strong> the literature<br />
(Moster, 1984). Most reports do not dist<strong>in</strong>guish unilateral from bilateral SNP, or isolated<br />
SNP from those associated with other neurologic or cranial nerve defects (Sav<strong>in</strong>o, 1986).<br />
Our review of the literature on SNP revealed 31 case reports and case series describ<strong>in</strong>g<br />
237 patients with presumed isolated SNP. Of these 237 patients, 31 were traumatic, none<br />
were congenital, 60 were vasculopathic, 47 were idiopathic, and the rema<strong>in</strong>der had<br />
a number of miscellaneous etiologies (seven post-LP, 19 multiple sclerosis, two<br />
postimmunizations, five ‘‘<strong>in</strong>fectious,’’ five aneurysms, one sarcoid, six ‘‘presumed<br />
<strong>in</strong>flammation,’’ one orbital amyloidosis, and one diverticulum of the cavernous
304 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 13–1. Evaluation of sixth nerve palsy (SNP).
s<strong>in</strong>us). Fifty-two cases were the result of tumors (<strong>in</strong>clud<strong>in</strong>g chordomas, chondrosarcomas,<br />
men<strong>in</strong>giomas, cyl<strong>in</strong>droma, lymphomatous men<strong>in</strong>gitis, schwannomas, nasopharyngeal<br />
carc<strong>in</strong>oma, metastases, trigem<strong>in</strong>al neurilemmoma, pont<strong>in</strong>e glioma, pituitary<br />
adenomas, and miscellaneous tumors). The rema<strong>in</strong><strong>in</strong>g SNPs <strong>in</strong> the literature review<br />
were associated with other neurologic signs or symptoms, such as headache, t<strong>in</strong>nitus,<br />
disc edema, or nystagmus, or there were <strong>in</strong>sufficient cl<strong>in</strong>ical data <strong>in</strong> the report to<br />
determ<strong>in</strong>e if the SNP was truly isolated accord<strong>in</strong>g to our criteria <strong>in</strong> Table 13–1<br />
(<strong>An</strong>toniades, 1993; Averbuch-Heller, 1994; Barry-K<strong>in</strong>sella, 1994; Bell, 1990; Burde,<br />
1992; Depper, 1993; D<strong>in</strong>akaran, 1995; Fanous, 1992; Fujuoka, 1995; Gailbraith, 1994;<br />
Hamilton, 1993; Lee, 1992, 1994; Lesser, 1990; Leung, 1987; Lewis, 1995; Moster,<br />
1984; Nemzek, 1995; O’Boyle, 1992; Rush, 1981; Sachs, 1990; Sav<strong>in</strong>o, 1982, 1986;<br />
Simcock, 1994; Slav<strong>in</strong> 1992; Steel, 1994; Straussberg, 1993; Tung, 1991; Volpe, 1993a;<br />
Watanabe, 1990; Yang, 1991).<br />
SNPs that occur after LP, post–myelographic LP, and sp<strong>in</strong>al anesthesia have been<br />
reported <strong>in</strong> the literature (Simcock, 1994). Thorsen reported 229 cases of SNP after sp<strong>in</strong>al<br />
anesthesia and LP (Thorsen, 1947). Most of these SNPs occurred at the 10th day<br />
follow<strong>in</strong>g LP, were unilateral, associated with headache, and occurred <strong>in</strong> young patients<br />
(Simcock, 1994; Thorsen, 1947). These patients may be followed for resolution without<br />
imag<strong>in</strong>g (class IV, level C).<br />
<strong>An</strong>eurysm is a rare cause of acquired SNP. Rucker reported 924 cases of SNP, and<br />
only 31 (3.4%) were due to aneurysm (Rucker, 1966). Rush and Younge described 419<br />
cases of SNP, and only 15 (3.6%) were due to aneurysm (Rush, 1981). Other authors did<br />
not f<strong>in</strong>d any cases of aneurysm present<strong>in</strong>g with an isolated SNP <strong>in</strong> their series on<br />
cerebral aneurysms with ocular <strong>in</strong>volvement (Steel, 1994), and others have reported<br />
similar f<strong>in</strong>d<strong>in</strong>gs. We do not typically recommend evaluation for aneurysm <strong>in</strong> isolated<br />
SNP (class IV, level C), but aneurysm can cause SNP <strong>in</strong> patients with signs of<br />
subarachnoid hemorrhage, papilledema, or other cranial neuropathies.<br />
A suggested approach to the evaluation of SNP is presented <strong>in</strong> Figure 13–1.<br />
References<br />
Sixth Nerve Palsies 305<br />
Alderson LM, Noonan PT, Sup Choi I, Henson JW. (1996). Regional subacute cranial neuropathies follow<strong>in</strong>g<br />
<strong>in</strong>ternal carotid cisplat<strong>in</strong> <strong>in</strong>fusion. <strong>Neuro</strong>logy 47:1088–1090.<br />
Al-D<strong>in</strong> AN, <strong>An</strong>derson M, Eeg-Olofsson O, et al. (1994). <strong>Neuro</strong>-ophthalmic manifestations of the syndrome of<br />
ophthalmoplegia, ataxia, and areflexia. A review. Acta <strong>Neuro</strong>l Scand 89:157–163.<br />
<strong>An</strong>toniades K, Karakasis D, Taskos N. (1993). Abducens nerve palsy follow<strong>in</strong>g transverse fracture of the middle<br />
cranial fossa. J Craniomaxillofac Surg 21:172–175.<br />
Apte RS, Bartek W, Mello A, Haq A. (1999). Spontaneous <strong>in</strong>tracranial hypotension. Am J Ophthalmol 127:482–485.<br />
Arita K, Sugiyama K, Tom<strong>in</strong>aga A, Yamasaki F. (2001). Intrasellar rhabdomyosarcoma: case report. <strong>Neuro</strong>surgery<br />
48:677–680.<br />
Aroichane M, Repka MX. (1995). Outcome of sixth nerve palsy or paresis <strong>in</strong> young children. J Pediatr Ophthalmol<br />
32:152–156.<br />
Averbuch-Heller L, Gillis S, Ben-Hur T. (1994). Transient sixth-nerve palsy as the first presentation of acute<br />
leukemia. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:506.<br />
Bachmeyer C, Levy V, Carteret M, et al. (1997). Sphenoid s<strong>in</strong>us localization of multiple myeloma reveal<strong>in</strong>g<br />
evolution from benign gammopathy. Head Neck 19:347–350.<br />
Balcar LJ, Galetta SL, Cornblath WT, Liu GT. (1999). <strong>Neuro</strong>-ophthalmologic manifestations of Maffucci’s<br />
syndrome and Ollier’s disease. J <strong>Neuro</strong>-ophthalmol 19:62–66.<br />
Barry-K<strong>in</strong>sella C, Milner M, McCarthy N, et al. (1994). Sixth nerve palsy: an unusual manifestation of<br />
preeclampsia. Obstet Gynecol 83:849–850.
306 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Bell JA, Dowd TC, McIlwa<strong>in</strong>e GG, et al. (1990). Postmyelographic abducent nerve palsy <strong>in</strong> association with the<br />
contrast agent iopamidol. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:115–117.<br />
Biousse V, Ameri A, Bousser M-G. (1999). Isolated <strong>in</strong>tracranial hypertension as the only sign of cerebral venous<br />
thrombosis. <strong>Neuro</strong>logy 53:1537–1542.<br />
Blumenthal EZ, Gomori JM, Dotan S. (1997). Recurrent abducens nerve palsy caused by dolichoectasia of the<br />
cavernous <strong>in</strong>ternal carotid artery. J Ophthalmol 124:255–257.<br />
Brazis PW, Masdeu JC, Biller J. (2001). Localization <strong>in</strong> <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>logy. 4th ed. Philadelphia, Lipp<strong>in</strong>cott Williams<br />
& Wilk<strong>in</strong>s.<br />
Brazis PW, Wharen RE, Czervionke LF, et al. (2000). Hemangioma of the mandibular branch of the<br />
trigem<strong>in</strong>al nerve <strong>in</strong> the Meckel cave present<strong>in</strong>g with facial pa<strong>in</strong> and sixth nerve palsy. J <strong>Neuro</strong>-ophthalmol<br />
20:14–16.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1992). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. St. Louis, Mosby Year<br />
Book, pp. 289–311.<br />
Carr MM, Ross DA, Zuker RM. (1997). Cranial nerve defects <strong>in</strong> congenital facial palsy [review]. J Otolaryngol<br />
26:80–87.<br />
Case Records of the Massachusetts General Hospital. (1998). Case 2-1998. N Engl J Med 338:180–188.<br />
Chang-God<strong>in</strong>ich A, Lee AG, Brazis PW, et al. (1997). Complete ophthalmoplegia after zoster ophthalmicus.<br />
J <strong>Neuro</strong>-ophthalmol 17:262–265.<br />
Chavis PS, Mullaney PB, Bohlega S. (1998). Fluctuat<strong>in</strong>g oculomotor signs <strong>in</strong> Arnold-Chiari malformation.<br />
Diagnostic pitfalls. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:215–221.<br />
Chiba A, Kusunoki S, Obata H, et al. (1993). Serum anti-GB1b IgG antibody is associated with ophthalmoplegia <strong>in</strong><br />
Miller Fisher syndrome and Guilla<strong>in</strong>-Barré syndrome: cl<strong>in</strong>ical and immunohistochemical studies. <strong>Neuro</strong>logy<br />
43:1911–1917.<br />
Chiba A, Kusunoki S, Shimizu T, et al. (1992). Serum IgG antibody to ganglioside GQ1b is a possible marker of<br />
Miller Fisher syndrome. <strong>An</strong>n <strong>Neuro</strong>l 31:677–679.<br />
Davé AV, Diaz-Marchan PJ, Lee AG. (1997). <strong>Cl<strong>in</strong>ical</strong> and magnetic resonance imag<strong>in</strong>g features of Gradenigo<br />
syndrome. Am J Ophthalmol 124:568–570.<br />
Depper MH, Truwit CL, Dreisbach JN, et al. (1993). Isolated abducens nerve palsy: MR imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs. AJR<br />
160:837–841.<br />
De Veuster I, Smet H, Vercauteren M, Tassignon MJ. (1994). The time course of a sixth nerve paresis follow<strong>in</strong>g<br />
epidural anesthesia. Bull Soc Belge Ophtalmol 252:45–47.<br />
D<strong>in</strong>akaran S, Desai SP, Corney CE. (1995). Case report: sixth nerve palsy follow<strong>in</strong>g radiculography. Br J Radiol 68:424.<br />
Dumont D, Hariz H, Meynieu P, et al. (1998). Abducens palsy after an <strong>in</strong>trathecal glucocorticoid <strong>in</strong>jection.<br />
Evidence for a role of <strong>in</strong>tracranial hypotension. Rev Rhum (English edition) 65:352–354.<br />
Eggenberger E, Lee AG, Forget TR Jr, Rosenwasser R. (2000). A brutal headache and double vision. Surv<br />
Ophthalmol 45:147–153.<br />
Eisenberg MB, Al-Mefty O, DeMonte F, Burson GT. (1999). Benign nonmen<strong>in</strong>geal tumors of the cavernous s<strong>in</strong>us.<br />
<strong>Neuro</strong>surgery 44:949–955.<br />
Fanous MM, Margo CE, Hamed LM. (1992). Chronic idiopathic <strong>in</strong>flammation of the retropharyngeal space<br />
present<strong>in</strong>g with sequential abducens palsies. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:154–157.<br />
Ferrante E, Sav<strong>in</strong>o A, Brioschi A, et al. (1998). Transient oculomotor cranial nerve palsy <strong>in</strong> spontaneous<br />
<strong>in</strong>tracranial hypotension. J <strong>Neuro</strong>surg Sci 42:177–179.<br />
FitzSimon JS, Toland J, Philips, J, et al. (1995). Giant aneurysms <strong>in</strong> the cavernous s<strong>in</strong>us. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:59–65.<br />
Forsyth PA, Casc<strong>in</strong>o TL, Shaw EG, et al. (1993). Intracranial chordomas: a cl<strong>in</strong>icopathological and prognostic<br />
study of 51 cases. J <strong>Neuro</strong>surg 78:741–747.<br />
Frohman LP, Grigorian R, Bielory L. (2001). <strong>Neuro</strong>-ophthalmic manifestations of sarcoidosis: cl<strong>in</strong>ical spectrum,<br />
evaluation, and management. J <strong>Neuro</strong>-ophthalmol 21:132–137.<br />
Fujuoka T, Segawa F, Ogawa K, et al. (1995). Ischemic and hemorrhagic bra<strong>in</strong> stem lesions mimick<strong>in</strong>g diabetic<br />
ophthalmoplegia. Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg 97:167–171.<br />
Fung TY, Chung TK. (1999). Abducens nerve palsy complicat<strong>in</strong>g pregnancy: a case report. Eur J Obstet Gynecol<br />
Reprod Biol 83:223–224.<br />
Gailbraith RS. (1994). Incidence of neonatal sixth nerve palsy <strong>in</strong> relation to mode of delivery. Am J Obstet 170:1158.<br />
Galetta SL, Smith JL. (1989). Chronic isolated sixth nerve palsies. Arch <strong>Neuro</strong>l 46:79–82.<br />
Hahn CD, Nicolle DA, Lownie SP, Drake CG. (2000). Giant cavernous carotid artery aneurysms: cl<strong>in</strong>ical<br />
presentation <strong>in</strong> fifty-seven cases. J <strong>Neuro</strong>-ophthalmol 20:253–258.<br />
Hamilton SR, Smith CH, Lessell S. (1993). Idiopathic hypertrophic cranial pachymen<strong>in</strong>gitis. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
13:127–134.
Sixth Nerve Palsies 307<br />
Harada T, Ohashi T, Ohki K, et al. (1997). Clival chordoma present<strong>in</strong>g as acute esotropia due to bilateral abducens<br />
palsy. Ophthalmologica 211:109–111.<br />
Harrigan MR, Chandler WF. (1998). Abducens nerve palsy after radiofrequency rhizolysis for trigem<strong>in</strong>al<br />
neuralgia: case report. <strong>Neuro</strong>surgery 43:623–625.<br />
Hashimoto M, Ohtsuka K. (1998). Compressive lesions of the abducens nerve <strong>in</strong> the subarachnoid space disclosed<br />
by th<strong>in</strong>-slice magnetic resonance imag<strong>in</strong>g. Ophthalmologica 212:188–189.<br />
Haughton AJ, Chalkiadis GA. (1999). Un<strong>in</strong>tentional paediatric subdural catheter with oculomotor and abducens<br />
nerve palsies. Paediatr <strong>An</strong>aesth 9:543–548.<br />
Hirose Y, Sagoh M, Mayanagi K, Murakami H. (1998). Abducens nerve palsy caused by basilar impression<br />
associated with atlanto-occipital assimilation. <strong>Neuro</strong>l Med Chir 38:363–366.<br />
Holland NR, Deibert E. (1998). CNS Act<strong>in</strong>omycosis present<strong>in</strong>g with bilateral cavernous s<strong>in</strong>us syndrome. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 64:4.<br />
Hollis GJ. (1997). Sixth cranial nerve palsy follow<strong>in</strong>g closed head <strong>in</strong>jury <strong>in</strong> a child. J Accident Emerg Med<br />
14:172–175.<br />
Holmes JM, Beck RW, Kip KE, et al. (2001). Predictors of nonrecovery <strong>in</strong> acute traumatic sixth nerve palsy and<br />
paresis. Ophthalmology 108:1457–1460.<br />
Holmes JM, Droste PJ, Beck RW. (1998). The natural history of acute traumatic sixth nerve palsy or paresis. JAm<br />
Assoc Pediatr Ophthalmol Strabismus 2:265–268.<br />
Homer JJ, Johnson IJ, Jones NS. (1996). Middle ear <strong>in</strong>fection and sixth nerve palsy. J Laryngol Otol 110:872–874.<br />
Horton JC, Fishman RA. (1994). <strong>Neuro</strong>visual f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> the syndrome of spontaneous <strong>in</strong>tracranial hypotension<br />
from dural cerebrosp<strong>in</strong>al fluid leak. Ophthalmology 101:244–251.<br />
Ichimi K, Yoshida J, Inao S, Wakabayashi T. (1997). Abducens nerve neur<strong>in</strong>oma—case report. <strong>Neuro</strong>l Med Chur<br />
37:197–200.<br />
Ifergane G, Merk<strong>in</strong> S, Valdman I, et al. (1998). Ocular manifestations of Jakob-Creutzfeldt disease (CJD). <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:21.<br />
Igarashi Y, Takeda M, Maekawa H, et al. (1992). Fisher’s syndrome without total ophthalmoplegia. Ophthalmology<br />
205:163–167.<br />
Jacobson DM. (1996). Progressive ophthalmoplegia with acute ischemic abducens nerve palsies. Am J Ophthalmol<br />
122:278–279.<br />
Kasner SE, Galetta SL, Vaughn DJ. (1996). Cavernous s<strong>in</strong>us syndrome <strong>in</strong> Hodgk<strong>in</strong>’s disease. J <strong>Neuro</strong>-Ophthalmol<br />
16:204–207.<br />
Kawase T, Sone A, Igarashi Y, et al. (1996). Developmental patterns and characteristic symptoms of petroclival<br />
men<strong>in</strong>giomas. <strong>Neuro</strong>l Med Chir 36:1–6.<br />
Keane JR. (1996). Cavernous s<strong>in</strong>us syndrome. <strong>An</strong>alysis of 151 cases. Arch <strong>Neuro</strong>l 53:967–971.<br />
Keane JR. (2000). Comb<strong>in</strong>ed VIth and XIIth cranial nerve palsies: a clival syndrome. <strong>Neuro</strong>logy 54:1540–1541.<br />
Kerty E. (1999). The <strong>ophthalmology</strong> of <strong>in</strong>ternal carotid artery dissection. Acta Ophthalmol Scand 77:418–421.<br />
K<strong>in</strong>g AJ, Stacey E, Stephenson G, Trimble RB. (1995). Spontaneous recovery rates for unilateral sixth nerve palsies.<br />
Eye 9:476–478.<br />
Krishna R, Kosmorsky GS, Wright KW. (1998). Pseudotumor cerebri s<strong>in</strong>e papilledema with unilateral sixth nerve<br />
palsy. J <strong>Neuro</strong>-ophthalmol 18:53–55.<br />
Kriss TC, Martich Kriss V, Warf BC. (1996). Cavernous s<strong>in</strong>us thrombophlebitis: case report. <strong>Neuro</strong>surgery 39:385–<br />
389.<br />
Kuehnen J, Schwartz A, Neff W, Hennerici M. (1998). Cranial nerve syndrome <strong>in</strong> thrombosis of the transverse=sigmoid<br />
s<strong>in</strong>uses. Bra<strong>in</strong> 121:381–388.<br />
Landolfi JC, Thaler HT, De <strong>An</strong>gelis LM. (1998). Adult bra<strong>in</strong>stem gliomas. <strong>Neuro</strong>logy 51:1136–1139.<br />
Lazow SK, Izzo SR, Fe<strong>in</strong>berg ME, Berger JR. (1995). Bilateral abducens nerve palsy secondary to maxillofacial<br />
trauma: report of case with proposed mechanism of <strong>in</strong>jury. J Oral Maxillofac Surg 53:1197–1199.<br />
Lee AG, Miller NR, Brazis PW, Benson ML. (1995). Cavernous s<strong>in</strong>us hemangioma. <strong>Cl<strong>in</strong>ical</strong> and neuroimag<strong>in</strong>g<br />
features. J <strong>Neuro</strong>-ophthalmol 15:225–229.<br />
Lee J. (1992). Modern management of sixth nerve palsy. Aust NZ J Ophthalmol 20:41–46.<br />
Lee J, Harris S, Cohen J, et al. (1994). Results of a prospective randomized trial of botul<strong>in</strong>um tox<strong>in</strong> therapy <strong>in</strong> acute<br />
unilateral sixth nerve palsy. J Pediatr Ophthalmol Strabismus 31:283–286.<br />
Lee KY, Kim SM, Kim DI. (1998). Isolated bilateral abducens nerve palsy due to carotid cavernous dural<br />
arteriovenous fistula. Yonsei Med J 39:283–286.<br />
Lee TG, Choi W-S, Chung K-C. (2002). Ophthalmoplegic migra<strong>in</strong>e with reversible enhancement of <strong>in</strong>traparenchymal<br />
abducens nerve on MRI. Headache 42:140–141.<br />
Lemesle M, Beuriat P, Becker F, et al. (1998). Head pa<strong>in</strong> associated with sixth-nerve palsy: spontaneous dissection<br />
of the <strong>in</strong>ternal carotid artery. Cephalgia 18:112–114.
308 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Lepore FE. (1995). Disorders of ocular motility follow<strong>in</strong>g head trauma. Arch <strong>Neuro</strong>l 52:924–926.<br />
Lesser RL, Kornmehl EW, Pachner AR, et al. (1990). <strong>Neuro</strong>-ophthalmologic manifestations of Lyme disease.<br />
Ophthalmology 97:699–706.<br />
Leung AKC. (1987). Transient sixth cranial nerve palsy <strong>in</strong> newborn <strong>in</strong>fants. Br J Cl<strong>in</strong> Pract 41:717–718.<br />
Lewis AI, Tomsick TA, Tew JM. (1995). Management of 100 consecutive direct carotid-cavernous fistulas: results of<br />
treatment with detachable balloons. <strong>Neuro</strong>surgery 36:239–245.<br />
Liu GT, Kay MD, Byrne GE, et al. (1993). Ophthalmoparesis due to Burkitt’s lymphoma follow<strong>in</strong>g cardiac<br />
transplantation. <strong>Neuro</strong>logy 43:2147–2149.<br />
Lloyd MN. (1995). Sixth nerve palsy follow<strong>in</strong>g radiculography [letter; comment]. Br J Radiol 68:1039–1040.<br />
Lopes DK, Mericle RA, Wakhloo AK, et al. (1998). Cavernous s<strong>in</strong>us syndrome dur<strong>in</strong>g balloon test occlusion of the<br />
cervical <strong>in</strong>ternal carotid artery. Report of two cases. J <strong>Neuro</strong>surg 89:667–670.<br />
Lopez JM, Reigosa RP, Losada GA, et al. (1996). Bilateral <strong>in</strong>farction of the rostral pont<strong>in</strong>e tegmentum as a cause of<br />
isolated bilateral supranuclear sixth nerve palsy related to hypertension. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
60:235–236.<br />
Mansour AM, Bailey BJ. (1997). Ocular f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Ramsay Hunt syndrome. J <strong>Neuro</strong>-ophthalmol 17:199–201.<br />
Mastrianni JA, Galetta SL, Raps EC, Liu GT, Volpe NJ. (1994). Isolated fascicular abducens nerve palsy and Lyme<br />
disease. J <strong>Neuro</strong>-ophthalmol 14:2–5.<br />
Mekari-Sabbagh ON, DaCunha RP. (2001). Crossed eyes <strong>in</strong> a six-year-old girl. Surv Ophthalmol 45:331–334.<br />
Mikkilä HO, Seppälä IJT, Viljanen MK, et al. (2000). The expand<strong>in</strong>g cl<strong>in</strong>ical spectrum of ocular Lyme borreliosis.<br />
Ophthalmology 107:581–587.<br />
Mizushima H, Kobayashi N, Sawabe Y, et al. (1998). Epidural hematoma of the clivus. Case report. J <strong>Neuro</strong>surg<br />
88:590–593.<br />
Mokri B, Piepgras DG, Miller GM. (1997). Syndrome of orthostatic headaches and diffuse pachymen<strong>in</strong>geal<br />
gadol<strong>in</strong>ium enhancement. Mayo Cl<strong>in</strong> Proc 72:400–413.<br />
Moster ML, Sav<strong>in</strong>o PJ, Sergott RC, et al. (1984). Isolated sixth-nerve palsies <strong>in</strong> younger adults. Arch Ophthalmol<br />
102:1328–1330.<br />
Movsas TZ, Balcar LJ, Eggenberger ER, et al. (2000). Sixth nerve palsy as a present<strong>in</strong>g sign of <strong>in</strong>tracranial<br />
plasmacytoma and multiple myeloma. J <strong>Neuro</strong>-ophthalmol 20:242–245.<br />
Muneer A, Jones NS. (1997). Unilateral abducens nerve palsy: a present<strong>in</strong>g sign of sphenoid s<strong>in</strong>us mucoceles.<br />
J Laryngol Otol 111:644–646.<br />
Mutyala S, Holmes JM, Hodge DO, Younge BR. (1996). Spontaneous recovery rate <strong>in</strong> traumatic sixth-nerve palsy.<br />
Am J Ophthalmol 122:898–899.<br />
Nagaoka U, Kato T, Kurita K, et al. (1996). Cranial nerve enhancement on three-dimensional MRI <strong>in</strong> Miller Fisher<br />
syndrome. <strong>Neuro</strong>logy 47:1601–1502.<br />
Narai H, Manabe Y, Deguchi K, et al. (2000). Isolated abducens nerve palsy caused by vascular compression.<br />
<strong>Neuro</strong>logy 55:453–454.<br />
Nemzek W, Postma G, Poirier V, et al. (1995). MR features of pachymen<strong>in</strong>gitis present<strong>in</strong>g with sixth-nerve-palsy<br />
secondary to sphenoid s<strong>in</strong>usitis. AJNR 16:960–963.<br />
Neugebauer A, Kirsch A, Fricke J, Russmann W. (2001). New onset of crossed eyes <strong>in</strong> an adult. Surv Ophthalmol<br />
45:335–344.<br />
Newman NJ, Slamovits, Friedland S, et al. (1995). <strong>Neuro</strong>-ophthalmic manifestations of men<strong>in</strong>gocerebral<br />
<strong>in</strong>flammation from the limited form of Wegener’s granulomatosis. Am J Ophthalmol<br />
120:613–621.<br />
O’Boyle JE, Gardner TA, Oliva A, et al. (1992). Sixth nerve palsy as the <strong>in</strong>itial present<strong>in</strong>g sign of metastatic prostate<br />
cancer. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:149–153.<br />
O’Carroll CP, Brandt-Zawadski M. (1999). The syndrome of spontaneous <strong>in</strong>tracranial hypotension. Cephalgia<br />
19:80–87.<br />
Ohtsuka K, Sone A, Igarashi Y, et al. (1996). Vascular compressive abducens nerve palsy disclosed by magnetic<br />
resonance imag<strong>in</strong>g. Am J Ophthalmol 122:416–419.<br />
Okada Y, Shima T, Nishida M, Okita S. (1997). Large sixth nerve neuroma <strong>in</strong>volv<strong>in</strong>g the prepont<strong>in</strong>e region: case<br />
report. <strong>Neuro</strong>surgery 40:608–610.<br />
Ormerod LD, Rhodes RH, Gross SA, et al. (1996). Ophthalmologic manifestations of acquired immune deficiency<br />
syndrome-associated progressive multifocal leukoencephalopathy. Ophthalmology 103:899–906.<br />
Openshaw H, Slatk<strong>in</strong> NE, Smith E. (1997). Eye movement disorders <strong>in</strong> bone marrow transplant patients on<br />
cyclospor<strong>in</strong> and ganciclovir. Bone Marrow Transplant 19:503–505.<br />
Patton N, Beatty S, Lloyd IC. (2000). Bilateral sixth and fourth cranial nerve palsies <strong>in</strong> idiopathic <strong>in</strong>tracranial<br />
hypertension. J R Soc Med 93:80–81.
Sixth Nerve Palsies 309<br />
Pedraza S, Gamez J, Rovira A, et al. (2000). MRI f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Möbius syndrome: correlation with cl<strong>in</strong>ical features.<br />
<strong>Neuro</strong>logy 55:1058–1060.<br />
Phookan G, Davis AT, Holmes B. (1998). Hemangioendothelioma of the cavernous s<strong>in</strong>us: case report. <strong>Neuro</strong>surgery<br />
42:1153–1156.<br />
Polito E, Leccisotti A. (1996). Pa<strong>in</strong>ful ophthalmoplegia caused by superior ophthalmic ve<strong>in</strong> thrombosis. <strong>Neuro</strong><strong>ophthalmology</strong><br />
16:189–192.<br />
Reich KA, Giansiracusa DR, Strongwater SL. (1990). <strong>Neuro</strong>logic manifestations of giant cell arteritis. Am J Med<br />
89:67–72.<br />
Roberts BN, Mills PV, Hawksworth NJ. (1995). Bilateral ptosis, tonic pupils and abducens palsies follow<strong>in</strong>g<br />
Campylobacter jejuni enteritis. Eye 9:657–658.<br />
Roman-Goldste<strong>in</strong> SM, Jones A, Delashaw JB, et al. (1998). Atypical central nervous system lymphoma at the<br />
cranial base: report of four cases. <strong>Neuro</strong>surgery 43:613–616.<br />
Ropper AH. (1993). Four new variants of Guilla<strong>in</strong>-Barré syndrome. <strong>An</strong>n <strong>Neuro</strong>l 34:306.<br />
Ropper AH, Wijdisks EFM, Truax BT. (1991). Guilla<strong>in</strong>-Barre Syndrome. Philadelphia, FA Davis.<br />
Rucker CW. (1966). The causes of paralysis of the third, fourth, and sixth cranial nerves. Am J Ophthalmol<br />
61:1293–1298.<br />
Rush JA, Younge BR. (1981). Paralysis of cranial nerves III, IV, and VI. Arch Ophthalmol 99:76–79.<br />
Sachs R, Kashii S, Burde RM. (1990). Sixth nerve palsy as the <strong>in</strong>itial manifestation of sarcoidosis. Am J Ophthalmol<br />
110:438–440.<br />
Sato K, Yoshikawa H. (2001). Bilateral abducens nerve paresis associated with anti-GQ1b IgG antibody. Am J<br />
Ophthalmol 131:816–818.<br />
Sav<strong>in</strong>o PJ. (1986). Diplopia and sixth nerve palsies. Sem<strong>in</strong> <strong>Neuro</strong>l 6:142–146.<br />
Sav<strong>in</strong>o PJ, Hilliker JK, Casell GH, et al. (1982). Chronic sixth nerve palsies: are they really harb<strong>in</strong>gers of serious<br />
<strong>in</strong>tracranial disease? Arch Ophthalmol 100:1442–1444.<br />
Schiev<strong>in</strong>k WI, Meyer FB, Atk<strong>in</strong>son JLD, Mokri B. (1996). Spontaneous sp<strong>in</strong>al cerebrosp<strong>in</strong>al fluid leaks and<br />
<strong>in</strong>tracranial hypotension. J <strong>Neuro</strong>surg 84:598–605.<br />
Schroeder B, Brieden S. (2000). Bilateral sixth nerve palsy associated with MDMA (‘‘ecstasy’’) abuse. Am J<br />
Ophthalmol 129:408–409.<br />
Shaw JA, Strachnan FM, Sawers HA, Bevan JS. (1997). Non-Hodgk<strong>in</strong> lymphoma with panhypopituitarism,<br />
hyperprolact<strong>in</strong>aemia, and sixth nerve palsy. J R Soc Med 90:274–275.<br />
Silva MN, Saeki N, Hirai S, Yamaura A. (1999). Unusual cranial nerve palsy caused by cavernous s<strong>in</strong>us<br />
aneurysms. <strong>Cl<strong>in</strong>ical</strong> and anatomical considerations reviewed. Surg <strong>Neuro</strong>l 52:148–149.<br />
Simcock PR, Kelleher S, Dunne JA. (1994). <strong>Neuro</strong>-ophthalmic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> botulism type B. Eye 8:646–648.<br />
Slav<strong>in</strong> M, Haimovic I, Mahendra P. (1992). Sixth nerve palsy and pontocerebellar mass due to luetic men<strong>in</strong>goencephalitis.<br />
Arch Ophthalmol 110:322.<br />
Smith EF, Santamar<strong>in</strong>a L, Wol<strong>in</strong>tz AH. (1993). Herpes zoster ophthalmicus as a cause of Horner syndrome. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 13:250–253.<br />
Steel TR, Bentivoglio PB, Garrick R. (1994). Vascular neurofibromatosis affect<strong>in</strong>g the <strong>in</strong>ternal carotid artery: a case<br />
report. Br J <strong>Neuro</strong>surg 8:233–237.<br />
Stepper F, Schroth G, Sturzenegger M. (1998). <strong>Neuro</strong>syphilis mimick<strong>in</strong>g Miller-Fisher syndrome: a case report and<br />
MRI f<strong>in</strong>d<strong>in</strong>gs. <strong>Neuro</strong>logy 51:269–271.<br />
Straussberg R, Cohen AH, Amir J, et al. (1993). Benign abducens palsy associated with EBV <strong>in</strong>fection. J Pediatr<br />
Ophthalmol Strabismus 30:60.<br />
Suzuki T, Chiba A, Kusunoki S, et al. (1998). <strong>An</strong>ti-GQ1b ganglioside antibody and ophthalmoplegia of<br />
undeterm<strong>in</strong>ed cause. Br J Ophthalmol 82:916–918.<br />
Thömke F. (1998). Isolated abducens palsies due to pont<strong>in</strong>e lesions. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:91–100.<br />
Thömke F, Lensch E, R<strong>in</strong>gel K, Hopf HC. (1997). Isolated cranial nerve palsies <strong>in</strong> multiple sclerosis. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 63:682–685.<br />
Thömke F, Mika-Grüttner A, Visbeck A, Brühl K. (2000). The risk of abducens palsy after diagnostic lumbar<br />
puncture. <strong>Neuro</strong>logy 54:768–769.<br />
Thorsen G. (1947). <strong>Neuro</strong>logical complications after sp<strong>in</strong>al anesthesia. Acta Chir Scand 5(suppl 121):1–272.<br />
Tung H, Chen T, Weiss MH. (1991). Sixth nerve schwannomas. J <strong>Neuro</strong>surg 75:638–641.<br />
Uehara T, Tabuchi M, Kawaguchi T, Mori E. (1998). Spontaneous dural carotid cavernous s<strong>in</strong>us fistula present<strong>in</strong>g<br />
isolated ophthalmoplegia: evaluation with MR angiography. <strong>Neuro</strong>logy 50:814–816.<br />
Volpe NJ, Lessell S. (1993a). Remitt<strong>in</strong>g sixth nerve palsy <strong>in</strong> skull base tumors. Arch Ophthalmol 111:1391–1395.<br />
Volpe NJ, Liebsch NJ, Munzenrider JE, Lessell S. (1993b). <strong>Neuro</strong>-ophthalmologic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> chordoma and<br />
chondrosarcoma of the skull base. Am J Ophthalmol 115:97–104.
310 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Wang CH, Chou ML, Huang CH. (1998). Benign isolated abducens nerve palsy <strong>in</strong> Mycoplasma pneumoniae<br />
<strong>in</strong>fection. Pediatr <strong>Neuro</strong>l 18:71–72.<br />
Watanabe K, Hagura R, Akanuma Y, et al. (1990). Characteristics of cranial nerve palsies <strong>in</strong> diabetic patients.<br />
Diabetes Res Cl<strong>in</strong> Pract 10:19–27.<br />
Wokke JH, van den Berg LH, van Schaik JP. (1996). Sixth nerve palsy from a CNS lesion <strong>in</strong> chronic <strong>in</strong>flammatory<br />
demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:695–696.<br />
Wolfe GI, Galetta SL, Mollman, JE. (1994). Spontaneous remission of papilledema and sixth nerve palsy <strong>in</strong> acute<br />
lymphoblastic leukemia. J <strong>Neuro</strong>-Ophthalmol 14:91–94.<br />
Wu HM, Lee AG, Lehane DE, et al. (1997). Ocular and orbital complications of <strong>in</strong>traarterial cisplat<strong>in</strong>. A case<br />
report. J <strong>Neuro</strong>-Ophthalmol 17:195–198.<br />
Yang MC, Bateman JB, Yee RD, et al. (1991). Electrooculography an discrim<strong>in</strong>ant analysis <strong>in</strong> Duane’s syndrome<br />
and sixth-cranial-nerve palsy. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 229:52–56.
14 r<br />
Supranuclear Disorders of Gaze<br />
What Is the <strong>An</strong>atomy of Horizontal Conjugate<br />
Gaze?<br />
Supranuclear structures coord<strong>in</strong>ate the action of muscle groups and control two types of<br />
eye movements: conjugate movements (both eyes move <strong>in</strong> the same direction) and<br />
vergence movements (both eyes move <strong>in</strong> opposite directions). The vergence movements<br />
can either turn <strong>in</strong> (converge) or turn out (diverge) (Cassidy, 2000). All of the supranuclear<br />
components act through a ‘‘f<strong>in</strong>al common pathway’’ for horizontal conjugate<br />
gaze. This f<strong>in</strong>al common pathway starts <strong>in</strong> the abducens nucleus (composed of two<br />
types of <strong>in</strong>term<strong>in</strong>gled neurons: motor neurons and <strong>in</strong>ternuclear neurons). The axons of<br />
the <strong>in</strong>ternuclear neurons cross to the contralateral side <strong>in</strong> the lower pons, ascend <strong>in</strong> the<br />
medial longitud<strong>in</strong>al fasciculus (MLF), and synapse <strong>in</strong> the portion of the oculomotor<br />
nucleus that <strong>in</strong>nervates the medial rectus muscle. The f<strong>in</strong>al common pathway is<br />
modulated by several <strong>in</strong>puts: the vestibular, optok<strong>in</strong>etic, smooth pursuit, and saccadic<br />
systems. As an example, an excitatory horizontal vestibulo-ocular impulse orig<strong>in</strong>at<strong>in</strong>g<br />
<strong>in</strong> the horizontal canal is relayed from the ipsilateral medial vestibular nucleus to the<br />
contralateral abducens nucleus, result<strong>in</strong>g <strong>in</strong> conjugate horizontal deviation of the eyes<br />
to the contralateral side (Johnston, 1992; Pierrot-Deseilligny, 1995; Tusa, 1998).<br />
Where Are Lesions Caus<strong>in</strong>g Horizontal Gaze<br />
Palsies Located?<br />
A lesion located anywhere along the supranuclear, nuclear, and <strong>in</strong>franuclear pathways<br />
that control horizontal eye movements may cause a horizontal gaze palsy. Depend<strong>in</strong>g<br />
on the location of the lesion, horizontal pursuit or saccades, or both, may be impaired.<br />
Lesions caus<strong>in</strong>g defects <strong>in</strong> horizontal smooth pursuit are summarized <strong>in</strong> Table 14–1.<br />
Lesions caus<strong>in</strong>g defects <strong>in</strong> horizontal saccadic eye movements are summarized<br />
Table 14–2.<br />
311
312 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–1. Localization of Lesions Impair<strong>in</strong>g Horizontal Pursuit Eye Movements<br />
Frontal lobe: impaired ipsilateral horizontal smooth pursuit<br />
Posterior parietal cortex or temporo-occipito-parietal region decrease the amplitude and velocity of<br />
smooth pursuit toward lesion (Barton, 1996; Lekwuwa, 1996; Morrow, 1995)<br />
Occipitotemporal areas posteriorly, through the <strong>in</strong>ternal sagittal stratum, the posterior and anterior<br />
limbs of the <strong>in</strong>ternal capsule with adjacent striatum, to the dorsomedial frontal cortex anteriorly;<br />
ipsilesional pursuit deficits (Lekwuwa, 1996)<br />
Posterior thalamic hemorrhage: deficit <strong>in</strong> smooth pursuit toward lesion (Brigell, 1984)<br />
Unilateral midbra<strong>in</strong> or pont<strong>in</strong>e lesion: ipsilateral pursuit defects (Furman, 1991; Gaymard, 1993;<br />
Johkura, 1998; Johnston, 1992; Kato, 1990; Thier, 1991; Waespe, 1992)<br />
Unilateral cerebellar damage: transient impairment of pursuit <strong>in</strong> direction of <strong>in</strong>volved side<br />
Bilateral cerebellar damage: permanent impairment of smooth pursuit eye movements<br />
Posterior vermal lesion: may impair pursuit (Pierrot-Deseilligny, 1990)<br />
Middle cerebellar peduncle lesions or floccular lesions; ipsilateral pursuit defect (Kato, 1990; Waespe,<br />
1992)<br />
What Studies Are Indicated <strong>in</strong> a Patient with a<br />
Horizontal Gaze Palsy?<br />
In general, unilateral restriction of voluntary horizontal conjugate gaze to one side is<br />
usually due to contralateral frontal or ipsilateral pont<strong>in</strong>e damage. At the bedside,<br />
pont<strong>in</strong>e lesions can usually be differentiated from supranuclear lesions by associated<br />
neurologic f<strong>in</strong>d<strong>in</strong>gs and by the oculocephalic (doll’s-eyes) maneuver or caloric stimulation.<br />
These latter procedures will overcome gaze deviations <strong>in</strong>duced by supranuclear<br />
lesions but will not overcome gaze deviations caused by pont<strong>in</strong>e lesions. Structural<br />
lesions, such as <strong>in</strong>farction, hemorrhage, vascular malformations, tumors, demyel<strong>in</strong>ation,<br />
trauma, or <strong>in</strong>fections, are the usual causes of horizontal gaze palsies. As all processes<br />
caus<strong>in</strong>g horizontal gaze palsies directly or <strong>in</strong>directly damage <strong>in</strong>traparenchymal bra<strong>in</strong><br />
pathways, neuroimag<strong>in</strong>g studies are necessary <strong>in</strong> all patients. In the acute sett<strong>in</strong>g, <strong>in</strong><br />
patients with altered levels of consciousness, or <strong>in</strong> patients <strong>in</strong> whom magnetic resonance<br />
imag<strong>in</strong>g (MRI) is contra<strong>in</strong>dicated (e.g., patients with pacemakers), computed tomography<br />
(CT) is appropriate. Otherwise, MRI is the procedure of choice <strong>in</strong> evaluat<strong>in</strong>g<br />
patients with horizontal gaze palsies (class III, level B). In patients with evidence of<br />
cl<strong>in</strong>ical seizure activity, <strong>in</strong> patients with <strong>in</strong>termittent conjugate gaze deviation, or <strong>in</strong><br />
obtunded or comatose patients with horizontal gaze palsies and evidence for possible<br />
contralateral cortical lesions, an electroencephalogram is <strong>in</strong>dicted to evaluate the<br />
possibility a seizure disorder (e.g., status epilepticus) (class III, level C). The evaluation<br />
of patients with horizontal gaze palsies is outl<strong>in</strong>ed <strong>in</strong> Figure 14–1.<br />
What Is the <strong>An</strong>atomy of the Abducens<br />
Nucleus and Medial Longitud<strong>in</strong>al Fasciculus<br />
(MLF)?<br />
The abducens nucleus has two types of <strong>in</strong>term<strong>in</strong>gled neurons: motor neurons and<br />
<strong>in</strong>ternuclear neurons. The axons of the <strong>in</strong>ternuclear neurons cross to the contralateral<br />
side <strong>in</strong> the lower pons and ascend <strong>in</strong> the MLF to synapse <strong>in</strong> the portion of the<br />
oculomotor nucleus that <strong>in</strong>nervates the medial rectus muscle (Leigh, 1999). In pont<strong>in</strong>e
Supranuclear Disorders of Gaze 313<br />
Table 14–2. Localization of Lesions Caus<strong>in</strong>g Impaired Horizontal Conjugate Saccadic Eye<br />
Movements<br />
Frontal lobe lesions<br />
Transient neglect contralaterally<br />
Defect <strong>in</strong> generat<strong>in</strong>g voluntary saccades<br />
Transient horizontal gaze deviation ipsilaterally acutely<br />
Gaze palsy overcome with the oculocephalic maneuver or caloric stimulation<br />
Late disorders of saccades (contralateral more than ipsilateral) due to frontal eye field (FEF) lesions<br />
(Tijssen, 1993, 1994)<br />
Prolonged eye deviation after stroke implies large stroke or preexist<strong>in</strong>g damage to the contralateral<br />
frontal region (Ste<strong>in</strong>er, 1984)<br />
Impaired ability to make a remembered sequence of saccades to visible targets (supplementary eye<br />
field lesions)<br />
Impaired performance of antisaccade tasks (dorsolateral prefrontal lesions)<br />
Epileptogenic lesions <strong>in</strong> the frontal eye fields<br />
Transient deviation of the eyes and head to the contralateral side (Godoy, 1990)<br />
Ipsiversive head and eye movements dur<strong>in</strong>g a seizure may also occur<br />
Initial forced turn<strong>in</strong>g (versive) head and eye movements usually correspond to a contralateral<br />
epileptiform focus, but these <strong>in</strong>itial contraversive movements may be followed by late<br />
ipsiversive or contraversive nonforced movements dur<strong>in</strong>g the secondary generalization<br />
(Kernan, 1993)<br />
Unilateral parietal lesions<br />
Ipsilateral horizontal gaze preference with acute lesions contralateral <strong>in</strong>attention with right-sided<br />
lesions<br />
Unilateral or bilateral <strong>in</strong>creased saccade latencies<br />
Hypometria for contralateral saccades<br />
Saccadic slow<strong>in</strong>g<br />
Bilateral parietal lesions<br />
Acquired ocular motor apraxia (Dehaene, 1991; Harris, 1996; Prasad, 1994; Shawkat, 1996)<br />
Lesions <strong>in</strong> the corona radiata adjacent to the genu of the <strong>in</strong>ternal capsule<br />
Contralateral selective saccadic palsy (Fukutake, 1993)<br />
Lesion (e.g., hemorrhage) deep <strong>in</strong> a cerebral hemisphere, particularly the thalamus<br />
Eye deviation to the side of the hemiparesis (‘‘wrong-way eyes’’) (Tijssen, 1994)<br />
Paresis of contralateral saccades<br />
Supranuclear contralateral gaze palsies associated with ipsilateral oculomotor palsies<br />
Pont<strong>in</strong>e lesions affect<strong>in</strong>g the abducens nucleus and=or the paramedian pont<strong>in</strong>e reticular formation<br />
(PPRF)<br />
Ipsilateral conjugate gaze palsy (Kataoka, 1997)<br />
Ipsilateral horizontal gaze palsy with ipsilateral esotropia (Coats, 1998)<br />
Acutely, eyes deviated contralaterally<br />
Doll’s-eye maneuver or cold caloric stimulation usually does not overcome gaze palsy<br />
Saccades toward side of lesion are present <strong>in</strong> contralateral hemifield but are slow with abducens<br />
nuclear lesions; ipsilaterally directed saccades from opposite field are small and slow or absent<br />
with PPRF lesions (Leigh, 1999)<br />
Horizontal gaze-evoked nystagmus on look<strong>in</strong>g contralaterally<br />
Bilateral horizontal gaze palsies with bilateral lesions (Shimura, 1997)<br />
Selective saccadic palsy (bilateral lesions of pons)<br />
(cont<strong>in</strong>ued)
314 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–2. (cont<strong>in</strong>ued)<br />
Voluntary saccades <strong>in</strong> both horizontal and vertical planes slow<br />
Smooth pursuit, the vestibulo-ocular reflex, the ability to hold steady eccentric gaze, and vergence<br />
eye movements preserved<br />
Paraneoplastic loss of horizontal voluntary eye movements or slow horizontal saccades (Baloh, 1993)<br />
Associated with persistent muscle spasms of the face, jaw, and pharynx<br />
Associated prostate carc<strong>in</strong>oma<br />
Congenital and familial bilateral horizontal gaze palsy (Aribal, 1998; Harris, 1996; Shawkat, 1996;<br />
Stavrou, 1999)<br />
May occur <strong>in</strong> isolation<br />
May be associated with kyphoscoliosis and facial contraction with myokymia<br />
MRI may reveal absence of abducens nuclei bilaterally (Aribal, 1998)<br />
Pseudo–horizontal gaze palsy with pont<strong>in</strong>e lesions<br />
Damag<strong>in</strong>g the medial longitud<strong>in</strong>al fasciculus on one side and the contralateral abducens nerve<br />
fascicle<br />
Bilateral <strong>in</strong>ternuclear ophthalmoplegia with the process subsequently extend<strong>in</strong>g laterally to the<br />
regions of the abducens fascicles bilaterally, result<strong>in</strong>g <strong>in</strong> complete horizontal gaze paralysis due<br />
to multiple sclerosis (Milea, 2001)<br />
lesions affect<strong>in</strong>g the abducens nucleus and=or the paramedian pont<strong>in</strong>e reticular formation<br />
(PPRF), a conjugate horizontal gaze palsy to the ipsilateral side occurs. Lesions<br />
of the MLF result <strong>in</strong> <strong>in</strong>ternuclear ophthalmoplegia (INO), whereas lesions of the MLF<br />
plus the ipsilateral abducens nucleus and=or PPRF result <strong>in</strong> the one-and-a-half<br />
syndrome. The cl<strong>in</strong>ical characteristics of these latter two syndromes and their evaluation<br />
are reviewed below.<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of INO?<br />
<strong>Cl<strong>in</strong>ical</strong>ly, an INO is characterized by adduction weakness on the side of the MLF lesion<br />
and monocular horizontal nystagmus of the opposite abduct<strong>in</strong>g eye. Convergence is<br />
usually preserved unless the responsible lesion is high <strong>in</strong> the midbra<strong>in</strong>. Often patients<br />
with INO have no visual symptoms, but some compla<strong>in</strong> of diplopia (due to skew<br />
deviation or limitation of adduction) or oscillopsia. INO is most evident dur<strong>in</strong>g<br />
horizontal saccadic eye movements, and the ‘‘adduction lag’’ is best detected dur<strong>in</strong>g<br />
optok<strong>in</strong>etic test<strong>in</strong>g us<strong>in</strong>g a tape or drum. For example, with a right INO when the drum<br />
is rotated to the right the amplitude and velocity of the adduct<strong>in</strong>g quick phase of the<br />
right eye is smaller and slower than that of the abduct<strong>in</strong>g saccades <strong>in</strong> the left eye. The<br />
pathogenesis of the nystagmus <strong>in</strong> the abduct<strong>in</strong>g eye is unclear but is likely a normal<br />
adaptive process that helps overcome the adduct<strong>in</strong>g weakness of the fellow eye<br />
(Getenet, 1993; Thömke, 1996). Other cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs noted with unilateral and<br />
bilateral INO are outl<strong>in</strong>ed <strong>in</strong> Table 14–3.<br />
Vitam<strong>in</strong> E deficiency syndrome (abetalipoprote<strong>in</strong>emia) may cause an eye movement<br />
disorder that superficially resembles the wall-eyed bilateral <strong>in</strong>ternuclear ophthalmoplegia<br />
(WEBINO) syndrome (Yee, 1976). In both of these syndromes, patients demonstrate<br />
exotropia with associated adduction limitation and dissociated horizontal nystagmus
Figure 14–1. Evaluation of horizontal gaze palsy.<br />
Supranuclear Disorders of Gaze 315<br />
on lateral gaze. In vitam<strong>in</strong> E deficiency, however, saccades are slower <strong>in</strong> the abduct<strong>in</strong>g<br />
eye than <strong>in</strong> the adduct<strong>in</strong>g eye, and the dissociated nystagmus is of greater amplitude <strong>in</strong><br />
the adduct<strong>in</strong>g eye. This motility impairment is especially noted with abetalipoprote<strong>in</strong>emia,<br />
with other f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clud<strong>in</strong>g ataxia, weakness, posterior column dysfunction,<br />
and pigmentary ret<strong>in</strong>opathy.<br />
What used to be called Lutz posterior INO is now known as INO of abduction<br />
(Oliveri, 1997; Thömke, 1992b). In this rare syndrome, abduction is restricted on volition<br />
but can be fully effected by reflex maneuvers, such as cold caloric stimulation. Unilateral<br />
or bilateral INO of abduction, occasionally associated with adduction nystagmus of the<br />
contralateral eye, has been described with ipsilateral rostral pont<strong>in</strong>e or mesencephalic<br />
lesions (Thömke, 1992b).
316 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–3. <strong>Cl<strong>in</strong>ical</strong> F<strong>in</strong>d<strong>in</strong>gs Noted with Internuclear Ophthalmoplegia (INO)<br />
Unilateral INO<br />
Ipsilateral adduction weakness, especially slow or fractionated adduct<strong>in</strong>g saccades (‘‘adduction<br />
lag’’), and monocular nystagmus <strong>in</strong> contralateral abduct<strong>in</strong>g eye<br />
May have esophoria acutely, suggest<strong>in</strong>g <strong>in</strong>creased vergence tone<br />
Convergence usually spared<br />
Skew deviation with the higher eye on the side of the lesion<br />
Vertical gaze-evoked nystagmus and impaired vestibular and pursuit vertical eye movements (i.e.,<br />
dissociated vertical nystagmus) (Leigh, 1999)<br />
Ipsilateral downbeat nystagmus and contralateral <strong>in</strong>cyclorotatory (torsional) nystagmus (Marshall,<br />
1991)<br />
Transient (disappear<strong>in</strong>g with<strong>in</strong> 3 days) torsional nystagmus, which is clockwise (exam<strong>in</strong>er’s view)<br />
<strong>in</strong> cases of left INO and counterclockwise <strong>in</strong> right INO (Fant<strong>in</strong>, 1995)<br />
Normal vertical saccades<br />
Rare exotropia (wall-eyed monocular <strong>in</strong>ternuclear ophthalmoplegia [WEMINO] syndrome)<br />
(Johnston, 1994)<br />
Rare exotropia <strong>in</strong> contralateral eye due to overexcitation of contralateral PPRF when fixat<strong>in</strong>g with<br />
paretic eye (Komiyama, 1998)<br />
Bilateral INO<br />
Bilateral adduction paresis or lag with the eyes generally aligned <strong>in</strong> primary gaze<br />
Exotropia, with both eyes deviated laterally (wall-eyed-bilateral <strong>in</strong>ternuclear ophthalmoplegia<br />
[WEBINO] syndrome) (Fay, 1999; Flitcroft, 1996)<br />
Vertical gaze-evoked nystagmus (on look<strong>in</strong>g up or down) and impaired vestibular and pursuit<br />
vertical eye movements (Leigh, 1999)<br />
Impaired vertical gaze hold<strong>in</strong>g<br />
What Is the Cause of the INO?<br />
INO is due to pathologic processes affect<strong>in</strong>g the medial pont<strong>in</strong>e or midbra<strong>in</strong> parenchyma.<br />
Often there are associated bra<strong>in</strong>stem symptoms and signs, although occasionally<br />
unilateral or bilateral INO may occur <strong>in</strong> isolation. The nature of the responsible<br />
pathologic process is suggested by the temporal mode of onset of the INO, the general<br />
cl<strong>in</strong>ical circumstances, and associated signs on neurologic and neuro-ophthalmologic<br />
exam<strong>in</strong>ation. Etiologies for INO are listed <strong>in</strong> Table 14–4.<br />
Although bilateral INO is more common with multiple sclerosis than with vascular<br />
<strong>in</strong>sults, bilateral INO may occur with stroke as well as many other pathologic processes,<br />
and thus the presence of a unilateral or bilateral INO cannot be used as a differential<br />
feature for etiologic diagnosis (class III–IV, level C). For example, <strong>in</strong> a series of 100<br />
patients with multiple sclerosis, 34 had INO, which was bilateral <strong>in</strong> 14 and unilateral <strong>in</strong><br />
20 (Muri, 1985). In another study of 51 patients with INO, 28 had multiple sclerosis and<br />
23 had <strong>in</strong>farction; INO was bilateral <strong>in</strong> 33 patients and unilateral <strong>in</strong> 28 (Hopf, 1991).<br />
Most patients with nutritional, metabolic, degenerative, and drug-<strong>in</strong>duced <strong>in</strong>toxication<br />
have bilateral INOs. Bilateral MLF <strong>in</strong>volvement with the pathologic process subsequently<br />
extend<strong>in</strong>g laterally to the region of the two abducens fascicles has been<br />
described as expla<strong>in</strong><strong>in</strong>g complete bilateral horizontal gaze paralysis <strong>in</strong> two patients<br />
with multiple sclerosis (Milea, 2001). The pattern of extraocular muscle weakness with<br />
myasthenia gravis (<strong>in</strong>clud<strong>in</strong>g penicillam<strong>in</strong>e-<strong>in</strong>duced myasthenia) can mimic INO
Table 14–4. Etiologies of Internuclear Ophthalmoplegia<br />
Supranuclear Disorders of Gaze 317<br />
Multiple sclerosis (Flitcroft, 1996; Frohman, 2001a; Gass, 1997; Hopf, 1991; Milea, 2001)<br />
Bra<strong>in</strong>stem <strong>in</strong>farction (Alexander, 1991; Getenet, 1993; Hopf, 1991; Kataoka, 1997; Marshall, 1991;<br />
Nagasaka, 1999; Okuda, 1993)<br />
Giant cell arteritis (Ahmad, 1999; Askari, 1993; Hughes, 1994; Johnston, 1990; Trend, 1990)<br />
Systemic lupus erythematosus<br />
<strong>Neuro</strong>-Behçet’s disease (Masai, 1995)<br />
Sickle cell trait (Leavitt, 1994)<br />
Polyarteritis nodosa (Kirkali, 1991)<br />
Eales’ disease (Ataby, 1992)<br />
Pyoderma gangrenosum (Lana, 1990)<br />
Sneddon’s syndrome (Rehany, 1998)<br />
Complication of angiography<br />
Complication of cardiac catheterization (Mihaescu, 2000)<br />
D-penicillam<strong>in</strong>e–<strong>in</strong>duced cerebral vasculitis (Pless, 1997)<br />
Bra<strong>in</strong>stem hemorrhage, <strong>in</strong>clud<strong>in</strong>g hemorrhage due to ‘‘crack’’ coca<strong>in</strong>e use (Diaz-Calderon, 1991)<br />
Bra<strong>in</strong>stem and fourth ventricular tumors (Arnold, 1990)<br />
Infections<br />
Cryptococcal men<strong>in</strong>gitis (Fay, 1999; Sung, 1991)<br />
Tuberculosis (granuloma or <strong>in</strong>fectious vasculitis)<br />
Viral and bacterial men<strong>in</strong>goencephalitis (Luis Guerrero-Peral, 2001)<br />
Syphilis<br />
Poliomyelitis (Wasserstrom, 1992)<br />
AIDS (Cacciatori, 1997)<br />
Jakob-Creutzfeldt disease (Billette de Villemeur, 1996)<br />
Head trauma (Chan, 2001; Haller, 1990; Hsu, 2001; Mueller, 1993; Strauss, 1995)<br />
Cervical <strong>in</strong>jury by hyperextension or manipulation<br />
Cancer-related<br />
Carc<strong>in</strong>omatous men<strong>in</strong>gitis<br />
Remote effect of cancer<br />
Bra<strong>in</strong>stem demyel<strong>in</strong>ation due to chemotherapy=radiation therapy<br />
Nutritional and metabolic disorders<br />
Wernicke’s encephalopathy (De la Paz, 1992)<br />
Pernicious anemia<br />
Hepatic encephalopathy<br />
Maple syrup ur<strong>in</strong>e disease<br />
Abetalipoprote<strong>in</strong>emia<br />
Fabry’s disease<br />
Hexosam<strong>in</strong>idase A deficiency (Barnes, 1991)<br />
Degenerative diseases<br />
Progressive supranuclear palsy (Friedman, 1992)<br />
Familial sp<strong>in</strong>ocerebellar degeneration (Senanayake, 1992)<br />
Arnold-Chiari malformation and associated hydrocephalus or syr<strong>in</strong>gobulbia (Arnold, 1990b; Chavis,<br />
1998; Lewis, 1996)<br />
(cont<strong>in</strong>ued)
318 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–4. (cont<strong>in</strong>ued)<br />
Drug <strong>in</strong>toxications<br />
Narcotics<br />
Phenothiaz<strong>in</strong>es<br />
Tricyclic antidepressants<br />
Propranolol<br />
Barbiturates<br />
Lithium<br />
<strong>An</strong>tiobesity treatments (Lledo Carreres, 1992)<br />
Toluene (Hunnewell, 1998)<br />
Miscellaneous causes<br />
Hydrocephalus<br />
Pseudotumor cerebri (Friedman, 1998)<br />
Mesencephalic midl<strong>in</strong>e clefts (Lagreze, 1996)<br />
Subdural hematoma<br />
Subdural hygroma with an arachnoid cyst <strong>in</strong> the middle cranial fossa (M<strong>in</strong>amori, 1992)<br />
After external ventricular dra<strong>in</strong>age of a benign aqueductal cyst (Sh<strong>in</strong>, 2000)<br />
Supratentorial arteriovenous malformations<br />
Partial seizures<br />
Pseudo-INO<br />
Myasthenia gravis (may be associated with downshoot of adduct<strong>in</strong>g eye) (Ito, 1997)<br />
Fisher syndrome (Al-D<strong>in</strong>, 1994; Mori, 2001; Ortuno, 1990)<br />
Guilla<strong>in</strong>-Barré syndrome<br />
Myotonic muscular dystrophy (Azuara-Bianco, 1997; Verhagen, 1998)<br />
Surgical paresis of the medial rectus muscle<br />
(pseudo-INO) (Ito, 1997). Myasthenic pseudo-INO is not uncommon and may be<br />
associated with downshoot <strong>in</strong> the adduct<strong>in</strong>g eye (Ito, 1997). Other etiologies of<br />
pseudo-INO are listed <strong>in</strong> Table 14–4.<br />
What Studies Should Be Ordered <strong>in</strong> a Patient<br />
with INO?<br />
In general, the <strong>in</strong>vestigation of a patient with INO depends on the cl<strong>in</strong>ical circumstances.<br />
For example, <strong>in</strong> a patient with known multiple sclerosis, the appearance of INO<br />
as part of an exacerbation of the disease may not require neuroimag<strong>in</strong>g (class IV,<br />
level C), whereas INO <strong>in</strong> isolation or with associated unexpla<strong>in</strong>ed bra<strong>in</strong>stem signs and<br />
symptoms usually requires neuroimag<strong>in</strong>g (class III–IV, level C). If there is variability of<br />
the adduction deficit, associated fluctuat<strong>in</strong>g ptosis, or other variable ocular motor signs<br />
suggestive of myasthenia gravis, a myasthenic pseudo-INO should be considered (class<br />
III–IV, level C). If the evaluation <strong>in</strong>clud<strong>in</strong>g a Tensilon test is normal or if the cl<strong>in</strong>ical<br />
situation does not suggest myasthenia and there are no signs of an associated<br />
degenerative process (e.g., progressive supranuclear palsy) on cl<strong>in</strong>ical exam<strong>in</strong>ation,<br />
neuroimag<strong>in</strong>g is usually warranted (class III–IV, level C).
MRI is superior to CT scan <strong>in</strong> evaluat<strong>in</strong>g patients with INO (Alexander, 1991; Atlas,<br />
1987; Frohman, 2001a; Haller, 1990; Hopf, 1991; Strauss, 1995). For example, <strong>in</strong> a study<br />
of 11 patients with INO (n<strong>in</strong>e with multiple sclerosis and two with <strong>in</strong>farct), CT <strong>in</strong> all<br />
n<strong>in</strong>e tested failed to show a responsible lesion, whereas appropriate MRI abnormalities<br />
were documented <strong>in</strong> 10 of 11 patients (Atlas, 1987). In another study, CT did not detect<br />
abnormalities of the MLF <strong>in</strong> two patients with INO who had abnormal bra<strong>in</strong>stem<br />
hypersignals on MRI (Awerbach, 1990). In a study of chronic INO <strong>in</strong> 58 multiple<br />
sclerosis patients, proton density imag<strong>in</strong>g (PDI) revealed a hyper<strong>in</strong>tensity <strong>in</strong> the MLF <strong>in</strong><br />
all patients, whereas T2-weighted imag<strong>in</strong>g and fluid-attenuated <strong>in</strong>version recovery<br />
(FLAIR) imag<strong>in</strong>g showed these lesions <strong>in</strong> 88% and 48% of patients, respectively<br />
(Frohman, 2001a). Thus, PDI imag<strong>in</strong>g may show the MLF lesion <strong>in</strong> patients with<br />
multiple sclerosis and INO. CT imag<strong>in</strong>g <strong>in</strong> a patient with INO is reserved only for acute<br />
situations (e.g., bra<strong>in</strong>stem hemorrhage) or for patients <strong>in</strong> whom MRI is contra<strong>in</strong>dicated<br />
(e.g., pacemakers, etc.). MRI may give useful diagnostic data by also giv<strong>in</strong>g <strong>in</strong>formation<br />
about supratentorial processes likely to be <strong>in</strong>volved <strong>in</strong> the etiology of the INO, such as<br />
multiple sclerosis, multiple cerebral <strong>in</strong>farcts, etc. If an <strong>in</strong>farct is detected as the cause of<br />
INO <strong>in</strong> a patient older than 50 years of age, giant cell arteritis should be considered as<br />
an etiology, especially if other stroke risk factors are not evident (class III, level C). Thus,<br />
a sedimentation rate is warranted, and if it is elevated or the patient has other systemic<br />
symptoms of giant cell arteritis (e.g., jaw claudications, headache, polymyalgia rheumatic<br />
symptoms, etc.), a temporal artery biopsy should be obta<strong>in</strong>ed. There is <strong>in</strong>sufficient<br />
evidence to recommend an evaluation for giant cell arteritis <strong>in</strong> every INO <strong>in</strong> the elderly<br />
(class III–IV, level U).<br />
If MRI <strong>in</strong> nontraumatic cases is normal, then rarer etiologies for the INO should be<br />
considered (class III–IV, level C). If the INO is bilateral, drug <strong>in</strong>toxication should<br />
be suspected. Because pernicious anemia has rarely been reported to cause INO, a<br />
B12 level should be considered (class IV, level C). Syphilis may rarely cause INO, so<br />
serology for syphilis should be considered (class III–IV, level U). If MRI reveals<br />
men<strong>in</strong>geal enhancement or if men<strong>in</strong>geal signs or symptoms are present, sp<strong>in</strong>al fluid<br />
exam<strong>in</strong>ation is warranted to search for <strong>in</strong>fectious or carc<strong>in</strong>omatous men<strong>in</strong>gitis (class III–<br />
IV, level C). The suggested evaluation of a patient with INO is outl<strong>in</strong>ed <strong>in</strong> Figure 14–2.<br />
What Is the One-and-a-Half Syndrome?<br />
Supranuclear Disorders of Gaze 319<br />
In the one-and-a-half syndrome, there is a conjugate gaze palsy to one side (‘‘one’’) and<br />
impaired adduction on look<strong>in</strong>g to the other side (‘‘and-a-half’’) (Bronste<strong>in</strong>, 1990;<br />
Çelebisoy, 1996; Wall, 1983). As a result, the only horizontal movement rema<strong>in</strong><strong>in</strong>g is<br />
abduction of one eye, which may exhibit nystagmus <strong>in</strong> abduction. The responsible<br />
lesion <strong>in</strong>volves the PPRF or abducens nucleus and the adjacent MLF on the side of the<br />
complete gaze palsy. Patients with the one-and-a-half syndrome often have exotropia of<br />
the eye opposite the side of the lesion (paralytic pont<strong>in</strong>e exotropia). Rarely, a primary<br />
position esotropia occurs, most likely due to <strong>in</strong>volvement of the abducens nerve fascicle<br />
superimposed upon lesions of the PPRF and MLF (Wall, 1983).<br />
The one-and-a-half syndrome may be associated with ocular bobb<strong>in</strong>g and, more<br />
often, facial nerve palsy (the ‘‘eight-and-a-half syndrome’’) (Eggenberger, 1998). Patients<br />
with the one-and-a-half syndrome and facial nerve palsies may develop oculopalatal<br />
myoclonus months to years after the onset of the ocular motility problem (Wol<strong>in</strong>, 1996).
320 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 14–2. Evaluation of <strong>in</strong>ternuclear ophthalmoplegia (INO) and the one-and-a-half syndrome.<br />
The one-and-a-half syndrome may also be associated with supranuclear facial weakness<br />
on the same side as the gaze palsy and an INO. The lesion is <strong>in</strong> the paramedian aspect of<br />
the dorsal pont<strong>in</strong>e tegmentum, provid<strong>in</strong>g evidence for the existence of corticofugal<br />
fibers that extend to the facial nucleus <strong>in</strong> the dorsal paramedian pont<strong>in</strong>e tegmentum<br />
(<strong>An</strong>derson, 1999).<br />
<strong>An</strong>other type of one-and-a-half syndrome may result from two separate lesions<br />
<strong>in</strong>volv<strong>in</strong>g both MLFs and the fascicles of the abducens nerve on the side of the unilateral<br />
horizontal ‘‘gaze’’ palsy. In this case, however, if the ‘‘gaze’’ palsy is <strong>in</strong>complete, the eyes<br />
would move disconjugately <strong>in</strong> the direction of the gaze palsy (Pierrot-Deseilligny, 1981).<br />
A true gaze palsy due to unilateral PPRF damage causes concomitant paresis of both<br />
eyes. <strong>An</strong>other form of one-and-a-half syndrome consists of a horizontal conjugate gaze<br />
palsy to one side (e.g., to the left). This is associated with abduction paralysis of the eye<br />
on the side opposite to the gaze palsy (e.g., the right eye) and adduction nystagmus <strong>in</strong><br />
the eye on the side of the gaze palsy (e.g., <strong>in</strong> left eye) (Çelebisoy, 1996). This form of the<br />
one-and-a-half syndrome may be seen with rostral bra<strong>in</strong>stem <strong>in</strong>farction. A fourth type<br />
of (pseudo) one-and-a-half syndrome has been described <strong>in</strong> a patient with mucormycosis<br />
of the cavernous s<strong>in</strong>us (Carter, 1994). The patient had an ipsilateral sixth nerve<br />
palsy due to cavernous s<strong>in</strong>us <strong>in</strong>volvement and a contralateral horizontal gaze palsy due<br />
to simultaneous carotid artery occlusion with <strong>in</strong>farction of the frontal lobe. Contrary to<br />
the pont<strong>in</strong>e one-and-a-half syndrome, <strong>in</strong> which abduction <strong>in</strong> one eye is the preserved<br />
horizontal movement, this patient had only preserved adduction <strong>in</strong> one eye (contralateral<br />
to the sixth nerve palsy). Myasthenia gravis or the Miller Fisher syndrome may
produce f<strong>in</strong>d<strong>in</strong>gs that mimic a one-and-a-half syndrome (‘‘pseudo-one-and-a-half<br />
syndrome’’).<br />
What Etiologies Should Be Considered as a<br />
Cause of the One-and-a-Half Syndrome and<br />
What Evaluation Is Indicated?<br />
Etiologies of the one-and-a-half syndrome are listed <strong>in</strong> Table 14–5. Bra<strong>in</strong>stem <strong>in</strong>farction<br />
is the most common cause of the one-and-a-half syndrome <strong>in</strong> the elderly, whereas<br />
multiple sclerosis is the most common cause of the one-and-a-half syndrome <strong>in</strong> young<br />
adults. In fact, the one-and-a-half syndrome may be the present<strong>in</strong>g sign of multiple<br />
sclerosis (Martyn, 1988).<br />
Most patients with a one-and-a-half syndrome have other signs and symptoms of<br />
bra<strong>in</strong>stem <strong>in</strong>volvement. A patient with f<strong>in</strong>d<strong>in</strong>gs suggestive of a one-and-a-half<br />
syndrome with variable ocular motor paresis or ptosis should be evaluated for<br />
myasthenia gravis (class III, level C). All others should have MRI with attention to<br />
posterior fossa structures, to <strong>in</strong>vestigate structural etiologies (Hirose, 1993; Martyn,<br />
1988; Ohta, 1994). The evaluation of patients with the one-and-a-half syndrome is<br />
outl<strong>in</strong>ed <strong>in</strong> Figure 14–2 (class III–IV, level B).<br />
What Is the <strong>An</strong>atomy of Vertical Gaze?<br />
The ocular motor neurons concerned with vertical gaze and torsional eye movements lie<br />
<strong>in</strong> the oculomotor and trochlear nuclei. These nuclei receive afferents from the<br />
vestibular, smooth pursuit, optok<strong>in</strong>etic, and saccadic systems (Leigh, 1999; Tusa, 1998).<br />
Where Are the Lesions Responsible for<br />
Vertical Gaze Palsies Localized?<br />
The localization of lesions caus<strong>in</strong>g vertical gaze palsies is outl<strong>in</strong>ed <strong>in</strong> Table 14–6. The<br />
constellation of neuro-ophthalmologic f<strong>in</strong>d<strong>in</strong>gs seen with pretectal lesions has been<br />
Table 14–5. Etiologies of the One-and-a-Half Syndrome<br />
Supranuclear Disorders of Gaze 321<br />
Bra<strong>in</strong>stem <strong>in</strong>farction (<strong>An</strong>derson, 1999; Çelebisoy, 1996; Kataoka, 1997; Ohta, 1994; Wall, 1983; Wol<strong>in</strong>,<br />
1996; Yigit, 1996)<br />
Bra<strong>in</strong>stem hemorrhage<br />
Multiple sclerosis<br />
Tumors (primary or metastatic) of the bra<strong>in</strong>stem, fourth ventricle, or cerebellum (Newton, 1991)<br />
Postoperatively after the removal of tumors of the posterior fossa (Newton, 1991)<br />
Basilar artery aneurysms or bra<strong>in</strong>stem arteriovenous malformations (Wall, 1983)<br />
Trauma<br />
Mucormycosis of the cavernous s<strong>in</strong>us<br />
Pseudo–one-and-a-half syndrome (myasthenia gravis, Miller Fisher syndrome) (Band<strong>in</strong>i, 2001)
322 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–6. The Localization of Lesions Caus<strong>in</strong>g Vertical Gaze Palsies<br />
Unilateral right hemispheral lesions (Averbuch-Heller, 1996)<br />
Upgaze palsy, associated with bilateral ptosis<br />
May reflect the special contribution that the nondom<strong>in</strong>ant hemisphere makes to attention<br />
Thalamic lesions may be associated with vertical gaze palsies<br />
Likely due to concomitant midbra<strong>in</strong> <strong>in</strong>volvement<br />
Occasionally due to medial thalamic lesions without midbra<strong>in</strong> <strong>in</strong>volvement (Clark, 1995; Deleu,<br />
1997; Onder, 2000)<br />
Unilateral lesions of the rostral <strong>in</strong>terstitial nucleus of the medial longitud<strong>in</strong>al fasciculus (riMLF)<br />
(Bogousslavsky, 1990; Leigh, 1999)<br />
Slow<strong>in</strong>g of downward saccades or downgaze palsy<br />
Defect of torsional saccades (e.g., lesion of right riMLF impairs extorsion of right eye and <strong>in</strong>torsion<br />
of left eye) (Riordan-Eva, 1996)<br />
Torsional nystagmus beat<strong>in</strong>g contralesionally<br />
Pseudo–abducens palsy <strong>in</strong> opposite eye (Pullic<strong>in</strong>o, 2000)<br />
Bilateral lesions of the riMLF (Green, 1993; Hommel, 1990; Leigh, 1999)<br />
Downgaze saccadic palsy<br />
Paralysis of upward and downward saccades<br />
Lesions of the <strong>in</strong>terstitial nucleus of Cajal (INC) (Leigh, 1999)<br />
Impaired vertical gaze hold<strong>in</strong>g<br />
Impaired vertical saccades, especially upward<br />
Vertical gaze palsy, especially downward gaze, with bilateral lesions (Ohashi, 1998b)<br />
Ocular tilt reaction with unilateral lesions<br />
Upbeat nystagmus and neck retroflexion with bilateral lesions<br />
Posterior commissure lesions (Green, 1993; Hommel, 1990; Keane, 1990; Leigh, 1999)<br />
Dorsal midbra<strong>in</strong> syndrome (see Table 14–7)<br />
Paresis of upward gaze<br />
Paresis of downward gaze<br />
Paresis of upward and downward gaze<br />
Unilateral mesencephalic lesions (probably damage afferent and efferent connections to posterior<br />
commissure) (Albera, 1993; Hommel, 1990)<br />
Bilateral upgaze palsy (Hommel, 1990)<br />
Palsy of upward and downward saccades (Hommel, 1990)<br />
Palsy of upward and downward gaze<br />
variously designated as Par<strong>in</strong>aud’s syndrome, the sylvian aqueduct syndrome, the<br />
pretectal syndrome, the dorsal midbra<strong>in</strong> syndrome, and the Koerber-Salus-Elschnig<br />
syndrome. Ophthalmic f<strong>in</strong>d<strong>in</strong>gs of this syndrome are outl<strong>in</strong>ed <strong>in</strong> Table 14–7.<br />
What Etiologies Cause Vertical Gaze<br />
Impairment?<br />
Impaired upward gaze often occurs as a ‘‘physiologic’’ f<strong>in</strong>d<strong>in</strong>g <strong>in</strong> the elderly. Etiologies<br />
caus<strong>in</strong>g impaired vertical gaze are outl<strong>in</strong>ed <strong>in</strong> Table 14–8.
Table 14–7. Ophthalmic F<strong>in</strong>d<strong>in</strong>gs with the Dorsal Midbra<strong>in</strong> Syndrome<br />
What Studies Are Indicated for the Evaluation<br />
of a Patient with Impaired Vertical Gaze?<br />
Supranuclear Disorders of Gaze 323<br />
Vertical gaze abnormalities, especially upgaze limitation, with or without associated limitation of<br />
downgaze<br />
Downward vestibulo-ocular movements may be spared<br />
Bell’s phenomenon may be spared<br />
Downward gaze preference or a tonic downward deviation of the eyes (‘‘sett<strong>in</strong>g sun sign’’)<br />
Primary position downbeat nystagmus.<br />
Impaired convergence and divergence; the patient thus may be exotropic or esotropic with A or V<br />
patterns<br />
Excessive convergence tone may result <strong>in</strong> slow or restricted abduction (‘‘midbra<strong>in</strong> pseudo–sixth<br />
palsy’’) dur<strong>in</strong>g horizontal refixations<br />
Convergence-retraction nystagmus, with quick adduct<strong>in</strong>g-retraction jerks predom<strong>in</strong>antly on upgaze<br />
Pretectal pseudobobb<strong>in</strong>g (nonrhythmic, rapid comb<strong>in</strong>ed downward and adduct<strong>in</strong>g movements,<br />
often preceded by a bl<strong>in</strong>k, with movement followed by slow return to midl<strong>in</strong>e)<br />
Skew deviation often with the higher eye on the side of the lesion<br />
Alternat<strong>in</strong>g adduction hypertropia or alternat<strong>in</strong>g adduction hypotropia<br />
Fixation <strong>in</strong>stability with square wave jerks<br />
Eyelid abnormalities<br />
Bilateral upper eyelid retraction, bar<strong>in</strong>g the sclera above the cornea (Collier’s ‘‘tucked lid’’ sign)<br />
Bilateral ptosis (lesion of ventral caudal nucleus of third nerve)<br />
Pupillary abnormalities (large with light-near dissociation)<br />
Occasionally, peripheral eye movement abnormalities, such as myasthenia gravis,<br />
Lambert-Eaton myasthenic syndrome, thyroid eye disease, or the Miller Fisher variant<br />
of Guilla<strong>in</strong>-Barré syndrome may simulate upgaze palsy or even convergence nystagmus<br />
(Keane, 1990). Retractory nystagmus, for example, may be mimicked by bilateral<br />
dysthyroid orbitopathy with bilateral <strong>in</strong>volvement of both medial recti and <strong>in</strong>ferior<br />
recti; saccadic upgaze attempts may cause convergence and retraction due to limitation<br />
of eye movements (Burde, 1985). Most of these peripheral processes are associated with<br />
other peripheral neurologic f<strong>in</strong>d<strong>in</strong>gs suggest<strong>in</strong>g the appropriate localization. If vertical<br />
gaze paresis fluctuates and there are no other signs of neurologic or systemic disease, a<br />
Tensilon test should be considered (class III–IV, level C).<br />
The evaluation of patients with vertical gaze impairments due to supranuclear<br />
etiologies depends on the cl<strong>in</strong>ical situation, especially if signs or symptoms of neurologic<br />
or systemic disease are present. For example, the presence of isolated impaired<br />
upward gaze <strong>in</strong> an elderly <strong>in</strong>dividual is a common ‘‘physiologic’’ f<strong>in</strong>d<strong>in</strong>g and requires<br />
no further evaluation. Vertical gaze disorders <strong>in</strong> the sett<strong>in</strong>g of other cl<strong>in</strong>ical manifestations<br />
of metabolic diseases of childhood or adolescence require appropriate biochemical<br />
<strong>in</strong>vestigation of the metabolic derangement. Also, if a vertical gaze impairment occurs<br />
as part of degenerative process (e.g., progressive supranuclear palsy), further neuroimag<strong>in</strong>g<br />
may not be required (class III, level C).<br />
If there is no evidence of a generalized metabolic or degenerative process on cl<strong>in</strong>ical<br />
exam<strong>in</strong>ation to expla<strong>in</strong> the f<strong>in</strong>d<strong>in</strong>gs, then further evaluation is typically warranted.
324 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 14–8. Etiologies of Vertical Gaze Impairment<br />
Primary and secondary tumors of the p<strong>in</strong>eal, thalamus, midbra<strong>in</strong>, aqueduct of Sylvius, or third<br />
ventricle (Chang, 1995; Keane, 1990)<br />
Midbra<strong>in</strong> or thalamic <strong>in</strong>farction or hemorrhage (<strong>An</strong>derson, 1998; Bogousslavsky, 1990; Clark, 1995;<br />
Deleu, 1997; Green, 1993; Hommel, 1990; Keane, 1990; Lee, 1996; Onder, 2000; Pullic<strong>in</strong>o, 2000;<br />
Tatemichi, 1992; Yijssen, 1996)<br />
Hydrocephalus, especially when dilatation of the third ventricle and aqueduct or enlargement of the<br />
suprap<strong>in</strong>eal recess cause pressure on and deformity of the posterior commissure (Bleasel, 1992;<br />
C<strong>in</strong>alli, 1999; Katz, 1994; Keane, 1990; Suzuki, 2000)<br />
Infectious or <strong>in</strong>flammatory etiologies<br />
Encephalitis (Green, 1993; Keane, 1990)<br />
Syphilis<br />
Sarcoidosis (Frohman, 2001b)<br />
Toxoplasmosis (Keane, 1990)<br />
Dissem<strong>in</strong>ated histoplasmosis (Perry, 1999)<br />
Tuberculosis (Keane, 1990)<br />
Whipple’s disease (Averbuch-Heller, 1999)<br />
Jakob-Creutzfeldt disease (Grant, 1993; Ifergane, 1998)<br />
Multiple sclerosis (Qu<strong>in</strong>t, 1993)<br />
Degenerative diseases<br />
Progressive supranuclear palsy (Bhidayasiri, 2001; Coll<strong>in</strong>s, 1995)<br />
Corticobasal ganglionic (corticobasal) degeneration (Riley, 1990)<br />
Hunt<strong>in</strong>gton’s disease<br />
Diffuse Lewy body disease (Brett, 2002; Fearnley, 1991; Lewis, 1990)<br />
Olivopontocerebellar degeneration (Wessel, 1998)<br />
Sp<strong>in</strong>ocerebellar atrophy type 1 (Klostermann, 1997)<br />
Postencephalitic park<strong>in</strong>sonism (Wenn<strong>in</strong>g, 1997)<br />
Motor neuron disease (Averbuch-Heller, 1998; Okuda, 1992)<br />
Idiopathic striopallidodentate calcifications syndrome (Saver, 1994)<br />
Lytico-Bodig (amyotrophic lateral sclerosis=park<strong>in</strong>sonism–dementia complex of Guam)<br />
Park<strong>in</strong>sonism, dementia, and vertical gaze palsy <strong>in</strong> Guamian with atypical neuroglial degeneration<br />
(Oyanagi, 2000)<br />
Progressive autosomal dom<strong>in</strong>ant park<strong>in</strong>sonism with pallido-ponto-nigral degeneration (Wszolek,<br />
1992)<br />
Joubert syndrome (Garbutt, 2000)<br />
Arteriovenous malformations and posterior fossa aneurysms (Keane, 1990)<br />
Metabolic diseases<br />
Bassen-Kornzweig syndrome (Keane, 1990)<br />
Niemann-Pick C disease and variants, <strong>in</strong>clud<strong>in</strong>g sea-blue histiocytosis syndrome, juvenile dystonic<br />
lipidoses, and the DAF (downgaze paralysis, ataxia=athetosis, and foam cells) syndrome<br />
(Garbutt, 2000; Lossos, 1997)<br />
Tay-Sachs disease<br />
Gaucher’s disease (Garbutt, 2000)<br />
Maple syrup ur<strong>in</strong>e disease<br />
(cont<strong>in</strong>ued)
Table 14–8. (cont<strong>in</strong>ued)<br />
Supranuclear Disorders of Gaze 325<br />
Hyperglyc<strong>in</strong>uria (Night<strong>in</strong>gale, 1991)<br />
Hexosam<strong>in</strong>idase A deficiency<br />
Wilson’s disease (Lee, 1999)<br />
Kernicterus (Keane, 1990)<br />
Wernicke’s syndrome (Keane, 1990)<br />
Vitam<strong>in</strong> B 12 deficiency<br />
Leigh disease (Garbutt, 2000)<br />
Trauma, <strong>in</strong>clud<strong>in</strong>g neurosurgical procedures from catheter compression (Keane, 1990; Shults, 1993)<br />
Drugs<br />
Barbiturates<br />
<strong>Neuro</strong>leptics<br />
Carbamazep<strong>in</strong>e<br />
Drugs most often affect vertical gaze by caus<strong>in</strong>g oculogyric crisis, an episodic, spasmodic,<br />
conjugate ocular deviation that usually occurs <strong>in</strong> an upward and lateral direction<br />
Miscellaneous causes<br />
Subdural hematoma<br />
Superficial central nervous system (CNS) siderosis with hydrocephalus (Janss, 1993)<br />
Pseudotumor cerebri (Friedman, 1998)<br />
Tentorial herniation (Keane, 1990)<br />
Paraneoplastic encephalomyelitis (e.g., sem<strong>in</strong>oma with positive anti-Ta antibody and encephalomyelitis<br />
with anti-Hu antibodies) (Bennett, 1999; Cr<strong>in</strong>o, 1996; Schiff, 1996; W<strong>in</strong>gerchuck, 1999)<br />
Congenital defects (Keane, 1990; Magli, 1991)<br />
Cerebral palsy (Garbutt, 2000)<br />
Wolfram’s syndrome (hereditary diabetes mellitus with bra<strong>in</strong>stem and optic atrophy, diabetes<br />
<strong>in</strong>sipidus, and deafness) (Scold<strong>in</strong>g, 1996)<br />
Benign transient form <strong>in</strong> childhood (benign paroxysmal tonic upgaze of neonates and children); may<br />
be associated with developmental delay, <strong>in</strong>tellectual disability, or language delay (Campistol,<br />
1993; Gieron, 1993; Hayman, 1998)<br />
Migra<strong>in</strong>e<br />
Subdural fluid collection over the cerebellar hemisphere (Rismondo, 1992)<br />
Mesencephalic clefts (Lagreze, 1996)<br />
Miller Fisher syndrome (Al-D<strong>in</strong>, 1994; Mori, 2001)<br />
Vertical gaze impairment, either <strong>in</strong> isolation or with other neurologic f<strong>in</strong>d<strong>in</strong>gs localized<br />
to the meso-diencephalon, generally requires cranial MRI with contrast (class IV,<br />
level C). If a pretectal syndrome develops <strong>in</strong> a patient with shunted hydrocephalus<br />
and neuroimag<strong>in</strong>g reveals no ventricular dilatation, shunt dysfunction should still be<br />
suspected, and neurosurgical consultation should be obta<strong>in</strong>ed to consider shunt<br />
revision or third ventriculostomy (C<strong>in</strong>alli, 1999) (class III–IV, level C). If MRI is<br />
normal and there are signs suggestive of <strong>in</strong>fection, especially signs of men<strong>in</strong>geal<br />
irritation, or if MRI reveals diffuse men<strong>in</strong>geal enhancement by contrast agent, then a<br />
lumbar puncture should be considered (class IV, level C). F<strong>in</strong>ally, if MRI is normal and<br />
no other etiologies are evident, a B12 level should be obta<strong>in</strong>ed and thiam<strong>in</strong>e supplementation<br />
considered (class III–IV, level C). Whipple’s disease (e.g., biopsy of <strong>in</strong>test<strong>in</strong>e
326 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
for histology and polymerase cha<strong>in</strong> reaction [PCR] or cerebrosp<strong>in</strong>al fluid PCR for T.<br />
whippelii) (Lynch, 1997; Von Herbay, 1997), syphilis, or a paraneoplastic process<br />
(e.g., sem<strong>in</strong>oma with anti-Ta or anti-Ma2 antibodies) (Bennett, 1999; Voltz, 1999)<br />
should be considered (class III–IV, level C). A proposed evaluation of the patient with<br />
vertical gaze impairment is outl<strong>in</strong>ed <strong>in</strong> Figure 14–3.<br />
Figure 14–3. Evaluation of vertical gaze palsy.
What Are the Characteristics of Supranuclear<br />
Monocular Elevation Paresis, the Vertical Oneand-a-Half<br />
Syndrome, and Skew Deviation?<br />
Supranuclear Disorders of Gaze 327<br />
Monocular elevation paresis (‘‘double elevator palsy’’) may occur on a peripheral basis<br />
(e.g., due to primary <strong>in</strong>ferior rectus restriction, primary superior rectus palsy, myasthenia<br />
gravis, or a fascicular third nerve lesion) or with pretectal supranuclear lesions.<br />
Supranuclear monocular elevation paresis may be congenital (Bell, 1990; Ziffer, 1992) or<br />
acquired. Acquired lesions contralateral or ipsilateral to the paretic eye <strong>in</strong>terrupt<br />
efferents from the rostral <strong>in</strong>terstitial nucleus of the MLF to the superior rectus and<br />
<strong>in</strong>ferior oblique subnuclei (often Bell’s phenomenon is <strong>in</strong>tact) (Hommel, 1990; Thömke,<br />
1992a). Double elevator palsy may be an asymmetric upgaze palsy that cl<strong>in</strong>ically<br />
presents as monocular elevation paresis <strong>in</strong> the more severely affected eye (Thömke,<br />
1992a).<br />
A vertical one-and-a-half syndrome, with vertical upgaze palsy and monocular<br />
paresis of downgaze on the side of the lesion or contralateral to the lesion, has been<br />
described with thalamomesencephalic <strong>in</strong>farction, best expla<strong>in</strong>ed by selective damage to<br />
supranuclear pathways or partial nuclear <strong>in</strong>volvement (Hommel, 1990; Tatemichi,<br />
1992). <strong>An</strong>other vertical one-and-a-half syndrome due to bilateral mesodiencephalic<br />
<strong>in</strong>farcts has been described. There is impairment of all downward rapid eye movements<br />
(<strong>in</strong>clud<strong>in</strong>g the vestibulo-ocular reflex) and downward smooth pursuit (nondissociated<br />
downgaze paralysis) associated with monocular paralysis of elevation (Deleu, 1991).<br />
Monocular elevation paresis of the right eye with contralateral paresis of downward<br />
gaze (‘‘crossed vertical gaze paresis’’) has been described with an <strong>in</strong>farct <strong>in</strong>volv<strong>in</strong>g the<br />
left mesodiencephalic junction and medial thalamus (Wiest, 1996). F<strong>in</strong>ally, a coexist<strong>in</strong>g<br />
vertical and horizontal one-and-a-half syndrome has been described with an <strong>in</strong>farct<br />
<strong>in</strong>volv<strong>in</strong>g the right medial thalamus, left dorsal upper midbra<strong>in</strong>, and left cerebellum<br />
(Terao, 2000). The right eye could abduct and had monocular horizontal nystagmus, but<br />
the left eye could gaze down only.<br />
A patient with locked-<strong>in</strong> syndrome due to pont<strong>in</strong>e <strong>in</strong>farction had dysconjugate<br />
vertical and torsional ocular movements (Park, 2001). When the patient was asked to<br />
look to the right, the right eye moved upward with <strong>in</strong>torsion and the left eye moved<br />
downward with extorsion. When the patient was asked to look to the left, the reversal<br />
cycle, with the left eye mov<strong>in</strong>g upward with <strong>in</strong>torsion and the right eye mov<strong>in</strong>g<br />
downward with extorsion, was observed. Horizontal gaze was limited to m<strong>in</strong>imal<br />
movement. It was thought that this <strong>in</strong>termittent dysconjugate abnormality was<br />
mediated by the <strong>in</strong>terstitial nucleus of Cajal.<br />
The term skew deviation is reserved for vertical misalignment result<strong>in</strong>g from supranuclear<br />
derangements. This skew deviation may be constant or transient. For example,<br />
epileptic skew deviation has been described (Galimberti, 1998). It occurs whenever<br />
peripheral or central lesions cause an imbalance of graviceptive bra<strong>in</strong>stem pathways<br />
and can accompany lesions at different areas of the bra<strong>in</strong>stem (mesencephalon to<br />
medulla) or cerebellum (Brandt, 1993, 1994, 1996; Halmagyi, 1990; Hamed, 1996;<br />
Suzuki, 2001). In a study of patients with unilateral bra<strong>in</strong>stem <strong>in</strong>farcts present<strong>in</strong>g<br />
with skew deviation and ocular torsion, all skew deviations were ipsiversive (ipsilateral<br />
eye was undermost) with caudal pontomedullary lesions, and contraversive (contralateral<br />
eye was lowermost) with rostral pontomesencephalic lesions (Brandt, 1993).
328 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Otolith <strong>in</strong>puts to the <strong>in</strong>terstitial nucleus of Cajal (INC) from the contralateral vestibular<br />
(especially lateral vestibular) nuclei and motor outputs from the INC to cervical and<br />
ocular motoneurons are likely <strong>in</strong>volved. In some patients, skew deviation may be<br />
associated with ocular torsion and head tilt (the ocular tilt reaction [OTR]) (Brandt, 1993,<br />
1994, 1996, 1998; Halmagyi, 1990). In the OTR, the head tilt, conjugate eye torsion, and<br />
hypotropia are all to the same side, suggest<strong>in</strong>g that this reaction is a motor compensation<br />
of a lesion-<strong>in</strong>duced apparent eye-head tilt; the contralateral head tilt represents a<br />
compensatory response to the perceived tilt of the subjective visual vertical.<br />
A left OTR could be due to a lesion of the left labyr<strong>in</strong>th, left vestibular nerve, left<br />
vestibular nucleus (e.g., Wallenberg syndrome), or right mesodiencephalon, suggest<strong>in</strong>g<br />
the existence of a crossed graviceptive pathway (possibly the MLF) between the<br />
vestibular nucleus and the contralateral INC (Brazis, 1992; Halmagyi, 1990; Keane,<br />
1992). OTRs have been reported <strong>in</strong> multiple conditions <strong>in</strong>clud<strong>in</strong>g vestibular nerve <strong>in</strong>jury<br />
(e.g., unilateral vestibular neurectomy and labyr<strong>in</strong>thectomy), herpes zoster of the<br />
vestibular nerve, auditory trauma, Wallenberg syndrome, lateral medullary compression,<br />
pontomedullary ischemia, and mesodiencephalic lesions (<strong>An</strong>derson, 1998;<br />
Arbusow, 1998; Averbuch-Heller, 1997; Brandt, 1993, 1994, 1996, 1998; Brazis, 1992;<br />
Dieterich, 1993; Halmagyi, 1990; Keane, 1992; Ohashi, 1998a; Riordan-Eva, 1997; Safran,<br />
1994; Vibert, 1996). The absence of bra<strong>in</strong>stem signs <strong>in</strong> peripheral OTR helps to exclude a<br />
central cause for the vertical diplopia. OTR has been described secondary to<br />
paraneoplastic encephalitis <strong>in</strong> a patient with sem<strong>in</strong>oma and anti-Ta antibodies<br />
(Bennett, 1999).<br />
A contraversive OTR may rarely be due to unilateral cerebellar lesions (the OTR may<br />
be under <strong>in</strong>hibitory control by the posterior cerebellum, possibly the nodulus)<br />
(Mossman, 1997). Occasionally, <strong>in</strong>creased <strong>in</strong>tracranial pressure (e.g., from benign<br />
<strong>in</strong>tracranial hypertension or pseudotumor cerebri), Fisher’s syndrome (Esaki, 1992),<br />
or hepatic coma may cause skew deviation. F<strong>in</strong>ally, a patient with a dorsal midbra<strong>in</strong><br />
syndrome with an ipsilateral skew deviation has been described due to a right<br />
paramedian thalamic <strong>in</strong>farct that perhaps impaired the tonic <strong>in</strong>put of the thalamus<br />
on the INC (<strong>An</strong>derson, 1998).<br />
A tonic OTR may simulate superior oblique palsy (Donahue, 1999). Five patients with<br />
OTR had a three-step test suggest<strong>in</strong>g superior oblique palsy (bilateral <strong>in</strong> one patient).<br />
No patient, however, had the expected excyclotorsion of the hypertropic eye. Two<br />
patients had conjugate ocular torsion (<strong>in</strong>torsion of the hypertropic eye and extorsion of<br />
the hypotropic eye) and two patients had only <strong>in</strong>torsion of the hypotropic eye. All had<br />
neurologic deficits consistent with more widespread bra<strong>in</strong>stem disease. The authors<br />
concluded that vertical ocular deviations that three-step to a superior oblique palsy are<br />
not always caused by fourth nerve weakness. When a patient with an apparent fourth<br />
nerve palsy has ocular torsion <strong>in</strong>consistent with a superior oblique (SO) palsy, OTR<br />
should be suspected, especially if posterior fossa or vestibular dysfunction coexist.<br />
Because results of the Bielschowsky head tilt test may be positive <strong>in</strong> patients with<br />
the OTR, the feature dist<strong>in</strong>guish<strong>in</strong>g OTR from SO palsy is the direction of torsion. The<br />
authors advocate a fourth step—evaluation of ocular torsion—<strong>in</strong> addition to the<br />
standard three steps.<br />
References<br />
Ahmad I, Zaman M. (1999). Bilateral <strong>in</strong>ternuclear ophthalmoplegia: an <strong>in</strong>itial present<strong>in</strong>g sign of giant cell arteritis.<br />
J Am Geriatr Soc 47:734–736.
Supranuclear Disorders of Gaze 329<br />
Albera R, Magnano M, Lacilla M, et al. (1993). Vascular dorsal midbra<strong>in</strong> syndrome. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
13:207–213.<br />
Al-D<strong>in</strong> AN, <strong>An</strong>derson M, Eeg-Olofsson O, et al. (1994). <strong>Neuro</strong>-ophthalmic manifestations of the syndrome of<br />
ophthalmoplegia, ataxia, and areflexia. A review. Acta <strong>Neuro</strong>l Scand 89:157–163.<br />
Alexander JA, Castillo M, Hoffman JC Jr. (1991). Magnetic resonance f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a patient with <strong>in</strong>ternuclear<br />
ophthalmoplegia. <strong>Neuro</strong>radiological-cl<strong>in</strong>ical correlation. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:58–61.<br />
<strong>An</strong>derson CA, Sanberg E, Filley CM, et al. (1999). One and one-half syndrome with supranuclear facial weakness.<br />
Arch <strong>Neuro</strong>l 56:1509–1511.<br />
<strong>An</strong>derson DF, Morris RJ. (1998). Par<strong>in</strong>aud’s syndrome and ipsilateral tonic ocular skew deviation from unilateral<br />
right paramedian thalamic <strong>in</strong>farct. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:13–15.<br />
Arbusow V, Dieterich M, Strupp M, et al. (1998). Herpes zoster neuritis <strong>in</strong>volv<strong>in</strong>g superior and <strong>in</strong>ferior parts of the<br />
vestibular nerve caus<strong>in</strong>g ocular tilt reaction. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:17–22.<br />
Aribal ME, Karaman ZC, Özkan SB, Söylev MF. (1998) Bilateral congenital horizontal gaze palsy: MR f<strong>in</strong>d<strong>in</strong>gs.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 19:69–74.<br />
Arnold AC. (1990a). Internuclear ophthalmoplegia from <strong>in</strong>tracranial tumor. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:278–286.<br />
Arnold AC, Baloh RW, Yee RD, Hepler RS. (1990b). Internuclear ophthalmoplegia <strong>in</strong> the Chiari type 2<br />
malformation. <strong>Neuro</strong>logy 40:1850–1854.<br />
Askari A, Jolobe OM, Sheperd DI. (1993). Internuclear ophthalmoplegia and Horner’s syndrome due to presumed<br />
giant cell arteritis. J R Soc Med 86:362.<br />
Ataby C, Erdem E, Kansu T, Eldem B. (1992). Eales’ disease with <strong>in</strong>ternuclear ophthalmoplegia. <strong>An</strong>n Ophthalmol<br />
24:267–269.<br />
Atlas SW, Grossman RI, Sav<strong>in</strong>o PJ, et al. (1987). Internuclear ophthalmoplegia: MR-anatomic correlation. AJNR<br />
8:243–247.<br />
Averbuch-Heller L, Helmchen C, Horn AKE, et al. (1998). Slow vertical saccades <strong>in</strong> motor neuron disease:<br />
correlation of structure and function. <strong>An</strong>n <strong>Neuro</strong>l 44:641–648.<br />
Averbuch-Heller L, Paulson GW, Daroff RB, Leigh RJ. (1999). Whipple’s disease mimick<strong>in</strong>g progressive supranuclear<br />
palsy: the diagnostic value of eye movement record<strong>in</strong>g. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 66:532–535.<br />
Averbuch-Heller L, Rottach KG, Zivotofsky AZ, et al. (1997). Torsional eye movements <strong>in</strong> patients with skew<br />
deviation and spasmodic torticollis: responses to static and dynamic head roll. <strong>Neuro</strong>logy 48:506–514.<br />
Averbuch-Heller L, Stahl JS, Remler BF, Leigh RJ. (1996). Bilateral ptosis and upgaze palsy with right hemispheric<br />
lesions. <strong>An</strong>n <strong>Neuro</strong>l 49:465–468.<br />
Awerbach G, Brown M, Lev<strong>in</strong> JR. (1990). Magnetic resonance imag<strong>in</strong>g of <strong>in</strong>ternuclear ophthalmoplegia. Int J<br />
<strong>Neuro</strong>sci 52:39–43.<br />
Azuara-Bianco A, Katz LJ, Arkfeld DF, Walsh TJ. (1997). Myotonic dystrophy mimick<strong>in</strong>g bilateral <strong>in</strong>ternuclear<br />
ophthalmoplegia. <strong>Neuro</strong>-<strong>ophthalmology</strong> 17:11–14.<br />
Baloh RW, DeRossett SE, Cloughesy TF, et al. (1993). Novel bra<strong>in</strong>stem syndrome associated with prostate<br />
carc<strong>in</strong>oma. <strong>Neuro</strong>logy 43:2591–2596.<br />
Band<strong>in</strong>i F, Faga D, Simonetti S. (2001). Ocular myasthenia mimick<strong>in</strong>g a one-and-a-half syndrome. J <strong>Neuro</strong>ophthalmol<br />
21:210–211.<br />
Barnes D, Misra VP, Young EP, et al. (1991). <strong>An</strong> adult onset hexosam<strong>in</strong>idase syndrome with sensory neuropathy<br />
and <strong>in</strong>ternuclear ophthalmoplegia. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:1112–1113.<br />
Barton JJS, Sharpe JA, Raymond JE. (1996). Directional defects <strong>in</strong> pursuit and motion perception <strong>in</strong> humans with<br />
unilateral cerebral lesions. Bra<strong>in</strong> 119:1535–1550.<br />
Bell JA, Fielder AR, V<strong>in</strong>ey S. (1990). Congenital double elevator palsy <strong>in</strong> identical tw<strong>in</strong>s. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol<br />
10:32–34.<br />
Bennett JL, Galetta SL, Frohman LP, et al. (1999). <strong>Neuro</strong>-ophthalmologic manifestations of a paraneoplastic<br />
syndrome and testicular carc<strong>in</strong>oma. <strong>Neuro</strong>logy 52:864–867.<br />
Bhidayasiri R, Riley DE, Somers JT, et al. (2001). Pathophysiology of slow vertical saccades <strong>in</strong> progressive<br />
supranuclear palsy. <strong>Neuro</strong>logy 57:2070–2077.<br />
Billette de Villemeur T, Deslys J-P, Pradel A, et al. (1996). Creutzfeldt-Jacob disease from contam<strong>in</strong>ated growth<br />
hormone extracts <strong>in</strong> France. <strong>Neuro</strong>logy 47:690–695.<br />
Bleasel AF, Ell JJ, Johnston I. (1992). Pretectal syndrome and shunt dysfunction. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:193–196.<br />
Bogousslavsky J, Miklossy J, Regli F, Janzer R. (1990). Vertical gaze palsy and selective unilateral <strong>in</strong>farction of the<br />
rostral <strong>in</strong>terstitial nucleus of the medial longitud<strong>in</strong>al fasciculus (riMLF). J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
53:67–71.<br />
Brandt T, Dieterich M. (1993). Skew deviation with ocular torsion: a vestibular bra<strong>in</strong>stem sign of topographic<br />
diagnostic value. <strong>An</strong>n <strong>Neuro</strong>l 33:528–534.<br />
Brandt T, Dieterich M. (1994). Vestibular syndromes <strong>in</strong> the roll plane: topographic diagnosis from bra<strong>in</strong>stem to<br />
cortex. <strong>An</strong>n <strong>Neuro</strong>l 36:337–347.
330 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Brandt T, Dieterich M. (1996). Central vestibular syndromes <strong>in</strong> the roll, pitch, and yaw planes. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
15:291–303.<br />
Brandt T, Dieterich M. (1998). Two types of ocular tilt reaction: the ‘‘ascend<strong>in</strong>g’’ pontomedullary VOR-OTR and<br />
the ‘‘descend<strong>in</strong>g’’ mesencephalic <strong>in</strong>tegrator-OTR. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:83–92.<br />
Brazis PW. (1992). Ocular motor abnormalities <strong>in</strong> Wallenberg’s lateral medullary syndrome. Mayo Cl<strong>in</strong> Proc<br />
67:365–368.<br />
Brett FM, Henson C, Stauton H. (2002). Familial diffuse Lewy body disease, eye movement abnormalities, and<br />
distribution of pathology. Arch <strong>Neuro</strong>l 59:44–467.<br />
Brigell M, Babikian V, Goodw<strong>in</strong> JA. (1984). Hypometric saccades and low-ga<strong>in</strong> pursuit result<strong>in</strong>g from a thalamic<br />
hemorrhage. <strong>An</strong>n <strong>Neuro</strong>l 15:374–378.<br />
Bronste<strong>in</strong> AM, Rudge P, Gresty MA, et al. (1990). Abnormalities of horizontal gaze. <strong>Cl<strong>in</strong>ical</strong>, oculographic and<br />
magnetic resonance imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs. II. Gaze palsy and <strong>in</strong>ternuclear ophthalmoplegia. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 53:200–207.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1985). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-Ophthalmology. 2nd ed. St. Louis, Mosby, pp.<br />
204–205.<br />
Cacciatori M, Dhillon B. (1997). Bilateral <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> AIDS. <strong>Neuro</strong>-<strong>ophthalmology</strong> 17:219–222.<br />
Campistol J, Prats JM, Garaizar C. (1993). Benign paroxysmal tonic upgaze of childhood with ataxia. A neuroophthalmological<br />
syndrome of familial orig<strong>in</strong>? Dev Med Child <strong>Neuro</strong>l 35:436–439.<br />
Carter JE, Rauch RA. (1994). One-and-a-half syndrome, type II. Arch <strong>Neuro</strong>l 51:87–89.<br />
Cassidy L, Taylor D, Harris C. (2000). Abnormal supranuclear eye movements <strong>in</strong> the child: a practical guide to<br />
exam<strong>in</strong>ation and <strong>in</strong>terpretation. Surv Ophthalmol 44:479–506.<br />
Çelebisoy N, Akyürekli Ö. (1996). One-and-a-half syndrome, type II: a case with rostral bra<strong>in</strong> stem <strong>in</strong>farction.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 16:373–377.<br />
Chan JW. (2001). Isolated unilateral post-traumatic <strong>in</strong>ternuclear ophthalmoplegia. J <strong>Neuro</strong>-ophthalmol 21:212–213.<br />
Chang SM, Lillis-Hearne PK, Larson DA, et al. (1995). P<strong>in</strong>eal blastoma <strong>in</strong> adults. <strong>Neuro</strong>surgery 37:383–391.<br />
Chavis PS, Mullaney PB, Bohlega S. (1998). Fluctuat<strong>in</strong>g oculomotor signs <strong>in</strong> Arnold-Chiari malformation.<br />
Diagnostic pitfalls. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:215–221.<br />
C<strong>in</strong>alli G, Sa<strong>in</strong>te-Rose C, Simon I, et al. (1999). Sylvian aqueduct syndrome and global rostral midbra<strong>in</strong><br />
dysfunction associated with shunt malfunction. J <strong>Neuro</strong>surg 90:227–236.<br />
Clark JM, Albers GW. (1995). Vertical gaze palsies from medial thalamic <strong>in</strong>farctions without midbra<strong>in</strong> <strong>in</strong>volvement.<br />
Stroke 26:1467–1470.<br />
Coats DK, Avilla CW, Lee AG, Paysse EA. (1998). Etiology and surgical management of horizontal pont<strong>in</strong>e gaze<br />
palsy with ipsilateral esotropia. J Am Assoc Pediatr Ophthalmol Strabismus 2:293–297.<br />
Coll<strong>in</strong>s SJ, Ahlskog JE, Parisi JE, Maraganore DM. (1995). Progressive supranuclear palsy: neuropathologically<br />
based diagnostic cl<strong>in</strong>ical criteria. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 58:167–173.<br />
Cr<strong>in</strong>o PB, Galetta SL, Sater RA, et al. (1996). Cl<strong>in</strong>icopathologic study of paraneoplastic bra<strong>in</strong>stem encephalitis and<br />
ophthalmoparesis. J <strong>Neuro</strong>-ophthalmol 16:44–48.<br />
Dehaene I, Lammens M. (1991). Acquired ocular motor apraxia. A cl<strong>in</strong>icopathologic study. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
11:117–122.<br />
De la Paz MA, Chung SM, McCrary JA III. (1992). Bilateral <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> a patient with<br />
Wernicke’s encephalopathy. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:116–120.<br />
Deleu D. (1997). Selective vertical saccadic palsy from unilateral medial thalamic <strong>in</strong>farction: cl<strong>in</strong>ical, neurophysiologic<br />
and MRI correlates. Acta <strong>Neuro</strong>l Scand 96:332–336.<br />
Deleu D, Eb<strong>in</strong>ger G. (1991). Vertical one-and-a-half syndrome. <strong>Cl<strong>in</strong>ical</strong>, oculographic and radiologic f<strong>in</strong>d<strong>in</strong>gs.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 11:99–101.<br />
Diaz-Calderon E, Del Brutto OH, Aguire R, Alarcon TA. (1991). Bilateral <strong>in</strong>ternuclear ophthalmoplegia after<br />
smok<strong>in</strong>g ‘‘crack’’ coca<strong>in</strong>e. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:297–299.<br />
Dieterich M, Brandt T. (1993). Ocular torsion and tilt of subjective visual are sensitive bra<strong>in</strong>stem signs. <strong>An</strong>n <strong>Neuro</strong>l<br />
33:292–299.<br />
Donahue SP, Lev<strong>in</strong> PJM, Hamed LM. (1999). Tonic ocular tilt reaction simulat<strong>in</strong>g a superior oblique palsy.<br />
Diagnostic confusion with the 3-step test. Arch Ophthalmol 117:347–352.<br />
Eggenberger EJ. (1998). Eight-and-a-half syndrome: one-and-a-half syndrome plus cranial nerve VII palsy. <strong>Neuro</strong><strong>ophthalmology</strong><br />
18:114–116.<br />
Esaki H, Sh<strong>in</strong>ji O. (1992). Skew deviation <strong>in</strong> Fisher’s syndrome. <strong>Neuro</strong>-ophthalmol Jpn 9:66.<br />
Fant<strong>in</strong> A. (1995). Torsional nystagmus <strong>in</strong> unilateral <strong>in</strong>ternuclear ophthalmoplegia. Presented at the annual<br />
meet<strong>in</strong>g of the North American <strong>Neuro</strong>-Ophthalmology Society, Tuscon, Arizona.<br />
Fay PM, Strom<strong>in</strong>ger MB. (1999). Wall-eyed bilateral <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> central nervous system<br />
cryptococcosis. J <strong>Neuro</strong>-ophthalmol 19:131–135.
Supranuclear Disorders of Gaze 331<br />
Fearnley JM, Revesz T, Brooks DJ, et al. (1991). Diffuse Lewy body disease present<strong>in</strong>g with a supranuclear gaze<br />
palsy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:159–161.<br />
Flitcroft DI, Saidléar CA, Stack JP, Eustace P. (1996). A proposed neuroanatomical and neurophysiological basis<br />
for WEBINO. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:280.<br />
Friedman DI, Forman S, Levi L, et al. (1998). Unusual ocular motility disturbances with <strong>in</strong>creased <strong>in</strong>tracranial<br />
pressure. <strong>Neuro</strong>logy 50:1893–1896.<br />
Friedman DI, Jankovic J, McCrary JA. (1992). <strong>Neuro</strong>-ophthalmic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> progressive supranuclear palsy. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 12:104–109.<br />
Frohman EM, Zhang H, Lramer PD, et al. (2001a). MRI characteristics of the MLF <strong>in</strong> MS patients with chronic<br />
<strong>in</strong>ternuclear ophthalmoplegia. <strong>Neuro</strong>logy 57:762–768.<br />
Frohman LP, Grigorian R, Bielory L. (2001b). <strong>Neuro</strong>-ophthalmic manifestations of sarcoidosis: cl<strong>in</strong>ical spectrum,<br />
evaluation, and management. J <strong>Neuro</strong>-ophthalmol 21:132–137.<br />
Fukutake T, Hirayama K, Sakakibara R. (1993). Contralateral selective saccadic palsy after a small haematoma <strong>in</strong><br />
the corona radiata adjacent to the genu of the <strong>in</strong>ternal capsule. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 56:221.<br />
Furman JMR, Hurtt MR, Hirsch WL. (1991). Asymmetrical ocular pursuit with posterior fossa tumors. <strong>An</strong>n <strong>Neuro</strong>l<br />
30:208–211.<br />
Galimberti CA, Vers<strong>in</strong>o M, Sartori I, et al. (1998). Epileptic skew deviation. <strong>Neuro</strong>logy 50:1469–1472.<br />
Garbutt S, Harris CM. (2000). Abnormal vertical optok<strong>in</strong>etic nystagmus <strong>in</strong> <strong>in</strong>fants and children. Br J Ophthalmol<br />
84:451–455.<br />
Gass A, Hennerici MG. (1997). Bilateral <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> multiple sclerosis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 63:564.<br />
Gaymard B, Pierrot-Deseilligny C, Rivaud S, Velut S. (1993). Smooth pursuit eye movement deficits after pont<strong>in</strong>e<br />
nuclei lesions <strong>in</strong> man. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 56:799–807.<br />
Getenet JC, Ventre J, Vighetto A, Tadary B. (1993). Saccades <strong>in</strong> <strong>in</strong>ternuclear ophthalmoplegia: are abduction<br />
disorders related to <strong>in</strong>terocular disconjugacy? J <strong>Neuro</strong>l Sci 114:160–164.<br />
Gieron MA, Korthals JK. (1993). Benign paroxysmal tonic upward gaze. Pediatr <strong>Neuro</strong>l 9:154–155.<br />
Godoy J, Lüders H, D<strong>in</strong>ner DS, et al. (1990). Versive eye movements elicited by cortical stimulation of the human<br />
bra<strong>in</strong>. <strong>Neuro</strong>logy 40:296–299.<br />
Grant MP, Cohen M, Peterson RB, et al. (1993). Abnormal eye movements <strong>in</strong> Creutzfeldt-Jacob disease. <strong>An</strong>n <strong>Neuro</strong>l<br />
34:192–197.<br />
Green JP, Newman NJ, W<strong>in</strong>terkorn JS. (1993). Paralysis of down gaze <strong>in</strong> two patients with cl<strong>in</strong>ical-radiologic<br />
correlation. Arch Ophthalmol 111:219–222.<br />
Haller KA, Miller-Meeks M, Kardon R. (1990). Early magnetic resonance imag<strong>in</strong>g <strong>in</strong> acute traumatic <strong>in</strong>ternuclear<br />
ophthalmoplegia. Ophthalmology 97:1162–1165.<br />
Halmagyi GM, Brandt T, Dieterich M, et al. (1990). Tonic contraversive ocular tilt reaction due to unilateral mesodiencephalic<br />
lesion. <strong>Neuro</strong>logy 40:1503–1509.<br />
Hamed L, Maria BL, Briscoe ST, et al. (1996). Intact b<strong>in</strong>ocular function and absent ocular torsion <strong>in</strong> children with<br />
alternat<strong>in</strong>g skew on lateral gaze. J Pediatr Ophthalmol Strabismus 33:164–166.<br />
Harris CM, Shawkat F, Russell-Eggitt I, et al. (1996). Intermittent horizontal saccade failure (‘‘ocular motor<br />
apraxia’’) <strong>in</strong> children. Br J Ophthalmol 80:151–158.<br />
Hayman M, Harvey AS, Hopk<strong>in</strong>s IJ, et al. (1998). Paroxysmal tonic upgaze: a reappraisal of outcome. <strong>An</strong>n <strong>Neuro</strong>l<br />
43:514–520.<br />
Hirose G, Furui K, Yoshioka A, Sakai K. (1993). Unilateral conjugate gaze palsy due to a lesion of the abducens<br />
nucleus. <strong>Cl<strong>in</strong>ical</strong> and neuroradiological correlation. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:54–58.<br />
Hommel M, Bogousslavsky DC. (1990). The spectrum of vertical gaze palsy follow<strong>in</strong>g unilateral bra<strong>in</strong>stem stroke.<br />
<strong>Neuro</strong>logy 41:1229–1234.<br />
Hopf HC, Thomke F, Gutmann L. (1991). Midbra<strong>in</strong> vs. pont<strong>in</strong>e medial longitud<strong>in</strong>al fasciculus lesions: the<br />
utilization of masseter and bl<strong>in</strong>k reflexes. Muscle Nerve 14:326–330.<br />
Hsu H-C, Chen HiJ, Lu K, Liang C-L. (2001). Reversible bilateral <strong>in</strong>ternuclear ophthalmoplegia follow<strong>in</strong>g head<br />
<strong>in</strong>jury. Acta Ophthalmol Scand 79:57–59.<br />
Hughes TA, Wiles CM, Hourihan M. (1994). Cervical radiculopathy and bilateral <strong>in</strong>ternuclear ophthalmoplegia<br />
caused by temporal arteritis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:764–765.<br />
Hunnewell J, Miller NR. (1998). Bilateral <strong>in</strong>ternuclear ophthalmoplegia related to chronic toluene abuse. J <strong>Neuro</strong>ophthalmol<br />
18:277–280.<br />
Ifergane G, Merk<strong>in</strong> S, Valdman I, et al. (1998). Ocular manifestations of Jakob-Creutzfeldt disease (CJD). <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:21.<br />
Ito K, Mizutani J, Murofushi T, Mizuno M. (1997). Bilateral pseudo-<strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> myasthenia<br />
gravis. J ORL Related Specialties 59:122–126.
332 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Janss AJ, Galetta SL, Freese A, et al. (1993). Superficial siderosis of the central nervous system: magnetic resonance<br />
imag<strong>in</strong>g and pathological correlation. Case report. J <strong>Neuro</strong>surg 79:756–760.<br />
Johkura K, Matsumoto S, Komiyama A, et al. (1998). Unilateral saccadic pursuit <strong>in</strong> patients with sensory stroke.<br />
Sign of a pont<strong>in</strong>e tegmentum lesion. Stroke 29:2377–2380.<br />
Johnston JL, Sharpe JA. (1994). The WEMINO syndrome—wall-eyed monocular <strong>in</strong>ternuclear ophthalmoplegia: an<br />
oculographic and neuropathologic characterization. <strong>Neuro</strong>logy 44(suppl 2):A311.<br />
Johnston JL, Sharpe JA, Morrow MJ. (1992). Paresis of contralateral smooth pursuit and normal vestibular smooth<br />
eye movements after unilateral bra<strong>in</strong>stem lesions. <strong>An</strong>n <strong>Neuro</strong>l 31:495–502.<br />
Johnston JL, Thomson GT, Sharpe JA, Inman RD. (1990). Internuclear ophthalmoplegia <strong>in</strong> giant cell arteritis.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 55:84–85.<br />
Kataoka S, Hori A, Shirakawa T, et al. (1997). Paramedian pont<strong>in</strong>e <strong>in</strong>farction: neurological=topographical<br />
correlation. Stroke 28:809–815.<br />
Kato I, Watanabe J, Nakamura T, et al. (1990). Mapp<strong>in</strong>g of bra<strong>in</strong>stem lesions by the comb<strong>in</strong>ed use of tests of<br />
visually-<strong>in</strong>duced eye movements. Bra<strong>in</strong> 113:921–935.<br />
Katz DM, Trobe JD, Muraszko KM, Dauser RC. (1994). Shunt failure without ventriculomegaly proclaimed by<br />
ophthalmic f<strong>in</strong>d<strong>in</strong>gs. J <strong>Neuro</strong>surg 81:721–725.<br />
Keane JR. (1990). The pretectal syndrome: 206 patients. <strong>Neuro</strong>logy 40:684–690.<br />
Keane JR. (1992). Ocular tilt reaction follow<strong>in</strong>g lateral pontomedullary <strong>in</strong>farction. <strong>Neuro</strong>logy 42:259–260.<br />
Kernan JC, Dev<strong>in</strong>sky O, Luciano DJ, et al. (1993). Lateraliz<strong>in</strong>g significance of head and eye deviation <strong>in</strong> secondary<br />
generalized tonic-clonic seizures. <strong>Neuro</strong>logy 43:1308–1310.<br />
Kirkali P, Topaloglu R, Kansu T, Bakkaloglu A. (1991). Third nerve palsy and <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong><br />
periarteritis nodosa. J Pediatr Ophthalmol Strabismus 28:45–46.<br />
Klostermann W, Zuhlke C, Heide W, et al. (1997). Slow saccades and other eye movement disorders <strong>in</strong><br />
sp<strong>in</strong>ocerebellar atrophy type 1. J <strong>Neuro</strong>l 244:105–111.<br />
Komiyama A, Takamatsu K, Johkura K, et al. (1998). Internuclear ophthalmoplegia and contralateral exotropia.<br />
Nonparalytic pont<strong>in</strong>e exotropia and WEBINO syndrome. <strong>Neuro</strong>-<strong>ophthalmology</strong> 19:33–44.<br />
Lagreze W-D, Warner JEA, Zamani AA, et al. (1996). Mesencephalic clefts associated with eye movement<br />
disorders. Arch Ophthalmol 114:429–432.<br />
Lana MA, Moreira PR, Nerves LB. (1990). Wall-eyed bilateral <strong>in</strong>ternuclear ophthalmoplegia (Web<strong>in</strong>o syndrome)<br />
and myelopathy <strong>in</strong> pyoderma gangrenosum. Arq <strong>Neuro</strong>psiquiatr 48:497–501.<br />
Leavitt JA, Butrus SI. (1994). Internuclear ophthalmoplegia <strong>in</strong> sickle cell trait. J <strong>Neuro</strong>-ophthalmol 14:49–51.<br />
Lee AG, Brown DG, Diaz PJ. (1996). Dorsal midbra<strong>in</strong> syndrome due to mesencephalic hemorrhage. Case report<br />
with serial imag<strong>in</strong>g. J <strong>Neuro</strong>-ophthalmol 16:281–285.<br />
Lee MS, Kim YD, Lyoo CH. (1999). Oculogyric crisis as an <strong>in</strong>itial manifestation of Wilson’s disease. <strong>Neuro</strong>logy<br />
52:1714–1715.<br />
Leigh RJ, Zee DS. (1999). The <strong>Neuro</strong>logy of Eye Movements. 3rd ed. New York, Oxford University Press.<br />
Lekwuwa GU, Barnes GR. (1996). Cerebral control of eye movements. I. The relationship between cerebral lesion<br />
sites and smooth pursuit deficits. Bra<strong>in</strong> 119:473–490.<br />
Lewis AJ, Gawel MJ. (1990). Diffuse Lewy body disease with dementia and oculomotor dysfunction. Mov Disord<br />
5:143–147.<br />
Lewis AR, Kl<strong>in</strong>e LB, Sharpe JA. (1996). Acquired esotropia due to Arnold-Chiari I malformation. J <strong>Neuro</strong>ophthalmol<br />
16:49–54.<br />
Lledo Carreres M, Lajo Garrido JL, Gonzalez Rico M, et al. (1992). Toxic <strong>in</strong>ternuclear ophthalmoplegia related to<br />
antiobesity treatment. <strong>An</strong>n Pharmacother 26:1457–1458.<br />
Lossos A, Schles<strong>in</strong>ger I, Okon E, et al. (1997). Adult-onset Niemann-Pick type C disease. <strong>Cl<strong>in</strong>ical</strong>, biochemical, and<br />
genetic study. Arch <strong>Neuro</strong>l 54:1536–1541.<br />
Luis Guerrero-Peral A, Mohamed Buskri A, <strong>An</strong>gel Ponce Villares M, Bueno V. (2001). Internuclear ophthalmoplegia<br />
as a presentation of men<strong>in</strong>geal <strong>in</strong>fection by the varicella virus. Med Cl<strong>in</strong> 116:36.<br />
Lynch T, Ode J, Fredericks DN, Louis ED, et al. (1997). Polymerase cha<strong>in</strong> reaction-based detection of Tropheryma<br />
whippelii <strong>in</strong> central nervous system Whipple’s disease. <strong>An</strong>n <strong>Neuro</strong>l 42:120–124.<br />
Magli A, DeMarco R, DiMaio S, et al. (1991). A case of Par<strong>in</strong>aud’s syndrome <strong>in</strong> a boy with delayed puberty.<br />
Ophthalmologica 202:132–137.<br />
Malessa S, Gaymard GD, Rivaud S, et al. (1994). Role of pont<strong>in</strong>e nuclei damage <strong>in</strong> smooth pursuit impairment of<br />
progressive supranuclear palsy: a cl<strong>in</strong>ical-pathologic study. <strong>Neuro</strong>logy 44:716–721.<br />
Marshall RS, Sacco RL, Krueger R, et al. (1991). Dissociated vertical nystagmus and <strong>in</strong>ternuclear ophthalmoplegia<br />
from a midbra<strong>in</strong> <strong>in</strong>farction. Arch <strong>Neuro</strong>l 48:1304–1305.<br />
Martyn CN, Keane D. (1988). The one-and-a-half syndrome. <strong>Cl<strong>in</strong>ical</strong> correlation with a pont<strong>in</strong>e lesion demonstrated<br />
by nuclear magnetic resonance imag<strong>in</strong>g <strong>in</strong> a case of multiple sclerosis. Br J Ophthalmol 72:515–517.
Supranuclear Disorders of Gaze 333<br />
Masai H, Kashii S, Kimura H, Fukuyama H. (1995). <strong>Neuro</strong>-Behçet disease present<strong>in</strong>g with <strong>in</strong>ternuclear<br />
ophthalmoplegia. Am J Ophthalmol 122:897–898.<br />
Merrill PT, Paige GD, Abrams RA, et al. (1991). Ocular motor abnormalities <strong>in</strong> human immunodeficiency virus<br />
<strong>in</strong>fection. <strong>An</strong>n <strong>Neuro</strong>l 30:130–138.<br />
Mihaescu M, Brillman J, Rothfus W. (2000). Midbra<strong>in</strong> ptosis caused by periaqueductal <strong>in</strong>farct follow<strong>in</strong>g cardiac<br />
catheterization: early detection with diffusion-weighted imag<strong>in</strong>g. J <strong>Neuro</strong>imag<strong>in</strong>g 10:187–189.<br />
Milea D, Napolitano M, Dechy H, et al. (2001). Complete bilateral horizontal gaze paralysis disclos<strong>in</strong>g multiple<br />
sclerosis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 70:252–255.<br />
M<strong>in</strong>amori Y, Yamamoto M, Tanaka A, et al. (1992). Medial longitud<strong>in</strong>al fasciculus syndrome associated with a<br />
subdural hygroma and an arachnoid cyst <strong>in</strong> the middle cranial fossa. Intern Med 31:1286–1290.<br />
Mori M, Kuwabara S, Fukutake T, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> features and prognosis of Miller Fisher syndrome.<br />
<strong>Neuro</strong>logy 56:1104–1106.<br />
Morrow MJ, Sharpe JA. (1993). Ret<strong>in</strong>otopic and directional deficits of smooth pursuit <strong>in</strong>itiation after posterior<br />
cerebral hemispheric lesions. <strong>Neuro</strong>logy 43:595–603.<br />
Morrow MJ, Sharpe JA. (1995). Deficits <strong>in</strong> smooth-pursuit eye movements after unilateral frontal lobe lesions. <strong>An</strong>n<br />
<strong>Neuro</strong>l 37:443–451.<br />
Mossman S, Halmagyi GM. (1997). Partial ocular tilt reaction due to unilateral cerebellar lesion. <strong>Neuro</strong>logy<br />
49:491–493.<br />
Mueller C, Koch S, 0Toifl K. (1993). Transient bilateral <strong>in</strong>ternuclear ophthalmoplegia after m<strong>in</strong>or head-trauma. Dev<br />
Med Child <strong>Neuro</strong>l 35:163–166.<br />
Muri RM, Meienberg O. (1985). The cl<strong>in</strong>ical spectrum of <strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> multiple sclerosis. Arch<br />
<strong>Neuro</strong>l 42:851–855.<br />
Nagasaka S, Fukushima T, Utsumomiya H, et al. (1999). Internuclear ophthalmoplegia caused by a lesion <strong>in</strong> the<br />
isthmus of the midbra<strong>in</strong>. <strong>Neuro</strong>-<strong>ophthalmology</strong> 21:113–116.<br />
Newton HB, M<strong>in</strong>er ME. (1991). ‘‘One-and-a-half’’ syndrome after a resection of a midl<strong>in</strong>e cerebellar astrocytoma:<br />
case report and discussion of the literature. <strong>Neuro</strong>surgery 29:768–772.<br />
Night<strong>in</strong>gale S, Barton ME. (1991). Intermittent vertical supranuclear ophthalmoplegia and ataxia. Mov Disord<br />
6:76–78.<br />
Ohashi T, Fukushima K, Ch<strong>in</strong> S, et al. (1998a). Ocular tilt reaction with vertical eye movement palsy caused by<br />
localized unilateral midbra<strong>in</strong> lesion. J <strong>Neuro</strong>-ophthalmol 18:40–42.<br />
Ohashi T, Nakano T, Harada T, et al. (1998b). Downward gaze palsy caused by bilateral lesions of the rostral<br />
mesencephalon. Ophthalmologica 212:212–214.<br />
Ohta K, Gotoh F, Fukuuchi Y, et al. (1994). Midpont<strong>in</strong>e tegmentum <strong>in</strong>farction with ‘‘one-and-a-half syndrome’’<br />
demonstrated by magnetic resonance imag<strong>in</strong>g. Keio J Med 43:164–165.<br />
Okuda B, Tachibana H, Sugita M, Maeda Y. (1993). Bilateral <strong>in</strong>ternuclear ophthalmoplegia, ataxia, and tremor<br />
from a midbra<strong>in</strong> <strong>in</strong>farction. Stroke 24:481–482.<br />
Okuda B, Yamamoto T, Yamasaki M, et al. (1992). Motor neuron disease with slow eye movements and vertical<br />
gaze palsy. Acta <strong>Neuro</strong>l Scand 85:71–76.<br />
Oliveri RL, Bono F, Quattrone A. (1997). Pont<strong>in</strong>e lesion of the abducens fasciculus produc<strong>in</strong>g so-called posterior<br />
<strong>in</strong>ternuclear ophthalmoplegia. Eur <strong>Neuro</strong>l 37:67–69.<br />
Onder F, Can I, Cosar CB, Kural G. (2000). Correlation of cl<strong>in</strong>ical and neuroradiological f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> down-gaze<br />
palsy. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 238:369–371.<br />
Ortuno AD, Maeztu C, Munoz JA, et al. (1990). Miller Fisher syndrome associated with Q fever. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 53:615–616.<br />
Oyanagi K, Chen KM, Craig KM, et al. (2000). Park<strong>in</strong>sonism, dementia and vertical gaze palsy <strong>in</strong> a Guamian with<br />
atypical neuroglial degeneration. Acta <strong>Neuro</strong>pathol 99:73–80.<br />
Park S-H, Na DL, Kim M. (2001). Disconjugate vertical ocular movement <strong>in</strong> a patient with locked-<strong>in</strong> syndrome.<br />
Br J Ophthalmol 85:496.<br />
Perry JD, Girk<strong>in</strong> CA, Miller NR, Mann RB. (1999). Dissem<strong>in</strong>ated histoplasmosis caus<strong>in</strong>g reversible gaze palsy and<br />
optic neuropathy. J <strong>Neuro</strong>-ophthalmol 19:140–143.<br />
Pierrot-Deseilligny C, Amarenco P, Roullet E, Marteau R. (1990). Vermal <strong>in</strong>farct with pursuit eye movement<br />
disorders. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 53:519–521.<br />
Pierrot-Deseilligny C, Cha<strong>in</strong> F, Serdaru M, et al. (1981). The one and a half syndrome. Electro-oculographic<br />
analysis of five cases with deductions about the physiological mechanisms of lateral gaze. Bra<strong>in</strong> 104:665–699.<br />
Pierrot-Deseilligny C, Rivaud S, Gaymard B, et al. (1995). Cortical control of saccades. <strong>An</strong>n <strong>Neuro</strong>l 37:557–567.<br />
Pless M, Sandson T. (1997). Chronic <strong>in</strong>ternuclear ophthalmoplegia. A manifestation of D-penicillam<strong>in</strong>e cerebral<br />
vasculitis. J <strong>Neuro</strong>-ophthalmol 17:44–46.
334 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Prasad P, Nair S. (1994). Congenital ocular motor apraxia: sporadic and familial. Support for natural resolution.<br />
J <strong>Neuro</strong>-ophthalmol 14:102–104.<br />
Pullic<strong>in</strong>o P, L<strong>in</strong>coff N, Truax BT. (2000). Abnormal vergence with upper bra<strong>in</strong>stem <strong>in</strong>farcts. Pseudoabducens palsy.<br />
<strong>Neuro</strong>logy 55:32–358.<br />
Qu<strong>in</strong>t DJ, Cornblath WT, Trobe JD. (1993). Multiple sclerosis present<strong>in</strong>g as Par<strong>in</strong>aud syndrome. AJNR<br />
14:1200–1202.<br />
Rehany U, Kassif Y, Rumelt S. (1998). Sneddon’s syndrome: neuro-ophthalmologic manifestations <strong>in</strong> a possible<br />
autosomal recessive pattern. <strong>Neuro</strong>logy 51:1185–1187.<br />
Riley DE, Lang AE, Lewis A, et al. (1990). Cortical-basal ganglionic degeneration. <strong>Neuro</strong>logy 40:1203–1212.<br />
R<strong>in</strong>ne JO, Lee MS, Thompson PD, Marsden CD. (1994). Corticobasal degeneration. A cl<strong>in</strong>ical study of 36 cases.<br />
Bra<strong>in</strong> 117:1183–1196.<br />
Riordan-Eva P, Faldon M, Büttner-Ennever JA, et al. (1996). Abnormalities of torsional fast phase eye movements<br />
<strong>in</strong> unilateral rostral midbra<strong>in</strong> disease. <strong>Neuro</strong>logy 47:201–207.<br />
Riordan-Eva P, Jarcourt JP, Faldon M, et al. (1997). Skew deviation follow<strong>in</strong>g vestibular nerve surgery. <strong>An</strong>n <strong>Neuro</strong>l<br />
41:94–99.<br />
Rismondo V, Borchert M. (1992). Position-dependent Par<strong>in</strong>aud’s syndrome. Am J Ophthalmol 114:107–<br />
108.<br />
Rosenberg ML, Jabbari B. (1991). Miosis and <strong>in</strong>ternuclear ophthalmoplegia as a manifestation of partial seizures.<br />
<strong>Neuro</strong>logy 41:737–739.<br />
Safran AB, Vibert D, Issoua D, Hausler A. (1994). Skew deviation after vestibular neuronitis. Am J Ophthalmol<br />
118:238–245.<br />
Saver JL, Liu GT, Charness ME. (1994). Idiopathic striopalidodentate calcification with prom<strong>in</strong>ent supranuclear<br />
abnormality of eye movement. J <strong>Neuro</strong>-ophthalmol 14:29–33.<br />
Schiff ND, Moore DF. (1996). Predom<strong>in</strong>ant downgaze ophthalmoparesis <strong>in</strong> anti-Hu encephalomyelitis. J <strong>Neuro</strong>ophthalmol<br />
16:302–303.<br />
Scold<strong>in</strong>g NJ, Kellar-Wood HF, Shaw C, et al. (1996). Wolfram syndrome: hereditary diabetes mellitus with<br />
bra<strong>in</strong>stem and optic atrophy. <strong>An</strong>n <strong>Neuro</strong>l 39:352–360.<br />
Senanayake N. (1992). A syndrome of early onset sp<strong>in</strong>ocerebellar ataxia with optic atrophy, <strong>in</strong>ternuclear<br />
ophthalmoplegia, dementia, and startle myoclonus <strong>in</strong> a Sri Lanken family. J <strong>Neuro</strong>l 239:293–294.<br />
Shawkat FS, Harris CM, Taylor DSI, Kriss A. (1996). The role of ERG=VEP and eye movement record<strong>in</strong>gs <strong>in</strong><br />
children with ocular motor apraxia. Eye 10:53–60.<br />
Sherman MD, All<strong>in</strong>son RW, Obbens EA, Darragh JM, Simons KB. Internuclear ophthalmoplegia <strong>in</strong> acquired<br />
immunodeficiency syndrome. <strong>An</strong>n Ophthalmol 21:294–295.<br />
Shimura M, Kiyosawa M, Tom<strong>in</strong>aga T, Tamai M. (1997). Bilateral horizontal gaze palsy with pont<strong>in</strong>e cavernous<br />
hemangioma: a case report. Ophthalmologica 211:320–322.<br />
Sh<strong>in</strong> M, Nishihara T, Iai S, Eguchi T. (2000). Benign aqueductal cyst caus<strong>in</strong>g bilateral <strong>in</strong>ternuclear ophthalmoplegia<br />
after external ventricular dra<strong>in</strong>age. Case report. J <strong>Neuro</strong>surg 92:490–492.<br />
Shults WT, Hamby S, Corbett JJ, et al. (1993). <strong>Neuro</strong>-ophthalmic complications of <strong>in</strong>tracranial catheters.<br />
<strong>Neuro</strong>surgery 33:135–138.<br />
Stavrou P, Willshaw HE. (1999). Familial congenital horizontal gaze palsy. J Pediatr Ophthalmol Strabismus<br />
36:47–49.<br />
Ste<strong>in</strong>er I, Melamed E. (1984). Conjugate eye deviation after acute hemispheric stroke: delayed recovery after<br />
previous contralateral frontal lobe damage. <strong>An</strong>n <strong>Neuro</strong>l 16:509–511.<br />
Strauss C, Ganslandt O, Huk WJ, Jonas JB. (1995). Isolated unilateral <strong>in</strong>ternuclear ophthalmoplegia follow<strong>in</strong>g<br />
head <strong>in</strong>jury. F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> magnetic resonance imag<strong>in</strong>g. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:15–19.<br />
Sung JY, Cheng PN, Lai KN. (1991). Internuclear ophthalmoplegia <strong>in</strong> cryptococcal men<strong>in</strong>gitis. J Trop Med Hyg<br />
94:116–117.<br />
Suzuki H, Matsubara T, Kanamaru K, Kojima T. (2000). Chronic hydrocephalus present<strong>in</strong>g with bilateral ptosis<br />
after m<strong>in</strong>or head <strong>in</strong>jury: case report. <strong>Neuro</strong>surgery 47:977–980.<br />
Suzuki T, Nishio M, Chikuda M, Takayanagi K. (2001). Skew deviation as a complication of cardiac catheterization.<br />
Am J Ophthalmol 132:282–283.<br />
Tatemichi TK, Ste<strong>in</strong>ke W, Duncan C, et al. (1992). Paramedian thalamopeduncular <strong>in</strong>farction: cl<strong>in</strong>ical syndromes<br />
and magnetic resonance imag<strong>in</strong>g. <strong>An</strong>n <strong>Neuro</strong>l 32:162–171.<br />
Terao S, Osano Y, Fukuoka T, et al. (2000). Coexist<strong>in</strong>g vertical and horizontal one and a half syndrome. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 69:401–402.<br />
Thier P, Bachor A, Faiss J, et al. (1991). Selective impairment of smooth-pursuit eye movements due to an ischemic<br />
lesion of the basal pons. <strong>An</strong>n <strong>Neuro</strong>l 29:443–448.
Supranuclear Disorders of Gaze 335<br />
Thömke F. (1996). Some observations on abduction nystagmus <strong>in</strong> <strong>in</strong>ternuclear ophthalmoplegia. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
16:27–38.<br />
Thömke F, Hopf HC. (1992a). Acquired monocular elevation paresis. <strong>An</strong> asymmetric up-gaze palsy. Bra<strong>in</strong><br />
115:1901–1910.<br />
Thömke F, Hopf HC, Kramer G. (1992b). Internuclear ophthalmoplegia of abduction: cl<strong>in</strong>ical and electrophysiological<br />
data on the existence of an abduction paresis of prenuclear orig<strong>in</strong>. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
55:105–111.<br />
Tijssen CC. (1994). Contralateral conjugate eye deviation <strong>in</strong> acute supratentorial lesion. Stroke 25:1516–1519.<br />
Tijssen CC, van Gisbergen JAM. (1993). Conjugate eye deviation after hemispheric stroke. A contralateral saccadic<br />
palsy? <strong>Neuro</strong>-<strong>ophthalmology</strong> 13:107–118.<br />
Trend P, Graham E. (1990). Internuclear ophthalmoplegia <strong>in</strong> giant-cell arteritis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
53:532–533.<br />
Tusa RJ, Ungerleider LG. (1998). Fiber pathways of cortical areas mediat<strong>in</strong>g smooth pursuit eye movements <strong>in</strong><br />
monkeys. <strong>An</strong>n <strong>Neuro</strong>l 23:174–183.<br />
Verhagen WIM, Huygen PLM. (1998). Myotonic dystrophy mimick<strong>in</strong>g INO. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:101–102.<br />
Vibert D, Häusler R, Safran AB, et al. (1996). Diplopia from skew deviation <strong>in</strong> unilateral peripheral vestibular<br />
lesions. Acta Otolaryngol 116:170–176.<br />
Voltz R, Gultek<strong>in</strong> SH, Rosenfeld MR, et al. (1999). A serologic marker of paraneoplastic limbic and bra<strong>in</strong>-stem<br />
encephalitis <strong>in</strong> patients with testicular cancer. N Engl J Med 340:1788–1795.<br />
Von Herbay A, Ditton H-J, Schuhmacher F, Maiwald M. (1997). Whipple’s disease: stag<strong>in</strong>g and monitor<strong>in</strong>g by<br />
cytology and polymerase cha<strong>in</strong> reaction analysis of cerebrosp<strong>in</strong>al fluid. Gastroenterology 113:434–441.<br />
Waespe W. (1992). Deficits of smooth-pursuit eye movements <strong>in</strong> two patients with a lesion <strong>in</strong> the para-floccular or<br />
dorsolateral pont<strong>in</strong>e region. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:91–96.<br />
Wall M, Wray SH. (1983). The one-and-a-half syndrome—a unilateral disorder of the pont<strong>in</strong>e tegmentum: a study<br />
of 20 cases and review of the literature. <strong>Neuro</strong>logy 33:971–980.<br />
Wasserstrom R, Mamourian AC, McGary CT, Miller G. (1992). Bulbar poliomyelitis: MR f<strong>in</strong>d<strong>in</strong>gs with pathologic<br />
correlation. AJNR 13:371–373.<br />
Waterston JA, Barnes GR, Grealy MA. (1992). A quantitative study of eye and head movements dur<strong>in</strong>g smooth<br />
pursuit <strong>in</strong> patients with cerebellar disease. Bra<strong>in</strong> 115:1343–1358.<br />
Wenn<strong>in</strong>g GK, Jell<strong>in</strong>ger K, Litvan I. (1997). Supranuclear gaze palsy and eyelid apraxia <strong>in</strong> postencephalitic<br />
park<strong>in</strong>sonism. J Neural Transm 104:845–865.<br />
Wessel K, Moschner C, Wand<strong>in</strong>ger K-P, et al. (1998). Oculomotor test<strong>in</strong>g <strong>in</strong> the differential diagnosis of<br />
degenerative ataxic disorders. Arch <strong>Neuro</strong>l 55:949–956.<br />
Wiest G, Baumgartner C, Schnider P, et al. (1996). Monocular elevation paresis and contralateral downgaze paresis<br />
from unilateral mesodiencephalic <strong>in</strong>farction. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:579–581.<br />
W<strong>in</strong>gerchuk DM, Noseworthy JH, Kimmel DW. (1999). Paraneoplastic encephalitis and sem<strong>in</strong>oma: importance of<br />
testicular ultrasonography. <strong>Neuro</strong>logy 51:1504–1507.<br />
Wol<strong>in</strong> MJ, Trent RG, Lav<strong>in</strong> PJM, Cornblath WT. (1996). Oculopalatal myoclonus after the one-and-a-half<br />
syndrome with facial nerve palsy. Ophthalmology 103:177–180.<br />
Wszolek ZK, Pfeiffer RF, Bhatt MH, et al. (1992). Rapidly progressive autosomal dom<strong>in</strong>ant park<strong>in</strong>sonism and<br />
dementia with pallido-ponto-nigral degeneration. <strong>An</strong>n <strong>Neuro</strong>l 32:31–320.<br />
Yee RD, Cogan DG, Zee DS. (1976). Ophthalmoplegia and dissociated nystagmus <strong>in</strong> abetalipoprote<strong>in</strong>emia. Arch<br />
Ophthalmol 94:571–575.<br />
Yee RD, Farlow MA, Suzuki DA, et al. (1992). Abnormal eye movements <strong>in</strong> Gerstmann-Straussler-Sche<strong>in</strong>ker<br />
disease. Arch Ophthalmol 110:68–74.<br />
Yigit A, B<strong>in</strong>göl A, Mutluer N, Taçilar N. (1996). The one-and-a-half syndrome <strong>in</strong> systemic lupus erythematosus.<br />
J <strong>Neuro</strong>-ophthalmol 16:274–276.<br />
Yijssen CC, De Letter MACJ, Op de Coul AAW. (1996). Convergence-retraction nystagmus. <strong>Neuro</strong>-<strong>ophthalmology</strong><br />
16:215–218<br />
Ziffer AJ, Rosenbaum AL, Demer JL, Yee RD. (1992). Congenital double elevator palsy: vertical saccadic velocity<br />
utiliz<strong>in</strong>g the scleral search coil technique. J Pediatr Ophthalmol Strabismus 29:142–149.
This page <strong>in</strong>tentionally left blank
15 r<br />
Ocular Myasthenia Gravis<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Myasthenia<br />
Gravis?<br />
Myasthenia gravis (MG) is a chronic disorder of neuromuscular transmission characterized<br />
cl<strong>in</strong>ically by vary<strong>in</strong>g degrees of weakness and fatigue of voluntary muscles. MG<br />
is caused by an acquired autoimmunity to the motor end plate and is associated with<br />
antibodies that block or cause <strong>in</strong>creased degradation of acetylchol<strong>in</strong>e receptors<br />
(AChRs). There is abnormal weakness <strong>in</strong> some or all voluntary muscles. The most<br />
commonly affected muscles are the levator palpebrae superioris, the extraocular<br />
muscles, the orbicularis oculi, triceps, quadriceps, and the tongue. Other<br />
voluntary muscles <strong>in</strong>nervated by cranial nerves (facial, masticatory, pharyngeal, and<br />
laryngeal muscles) and cervical, pectoral girdle, and hip flexor muscles are also<br />
frequently affected. The weakness <strong>in</strong>creases with repeated or susta<strong>in</strong>ed exertion and<br />
over the course of the day, but is improved by rest; it also may be worsened by elevation<br />
of body temperature and is often improved by cold (Engel, 1994; We<strong>in</strong>berg, 1994).<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features of Ocular<br />
Myasthenia Gravis and Generalized<br />
Myasthenia Gravis?<br />
The levator palpebrae superioris and extraocular muscles are <strong>in</strong>volved <strong>in</strong>itially <strong>in</strong><br />
approximately 50 to 70% of cases, and these muscles are eventually affected <strong>in</strong> about<br />
90% of patients. Ocular myasthenia (OM) is a form of MG conf<strong>in</strong>ed to the extraocular,<br />
levator palpebrae superioris, and=or orbicularis oculi muscles. Approximately 50% of<br />
patients <strong>in</strong>itially present with OM, but only 12 to 50% of these rema<strong>in</strong> ocular (Bever,<br />
1983; Oosterhuis, 1982). Of the 50 to 80% of patients with purely ocular symptoms and<br />
signs at onset that go on to develop generalized MG, most, but not all, develop<br />
generalized symptoms with<strong>in</strong> 2 to 3 years of onset of the disorder. Bever et al performed<br />
337
338 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
a retrospective study and found that 226 (84%) of 269 myasthenics displayed ocular<br />
f<strong>in</strong>d<strong>in</strong>gs at onset of disease and 142 (53%) demonstrated only ocular <strong>in</strong>volvement<br />
(Bever, 1983). Follow-up (average 14 years, range 1 to 39 years) of 108 patients with MG<br />
who had only ocular symptoms and signs at onset showed that 43 (40%) rema<strong>in</strong>ed<br />
ocular and 53 (49%) became generalized. Of the 53 patients who became generalized, 44<br />
(83%) did so with<strong>in</strong> 2 years of onset of the disease. Age of onset <strong>in</strong> their patients was of<br />
prognostic significance. Patients older than 50 years of age at onset had a greater risk of<br />
generalized MG and severe complications, whereas patients who were younger at onset<br />
had a more benign outcome. In another study of 1487 myasthenic patients, 53%<br />
presented with ocular MG and 202 (4%) cont<strong>in</strong>ued to demonstrate purely ocular<br />
<strong>in</strong>volvement for up to 45 years of follow-up (mean, 17 years) (Oosterhuis, 1982). Of<br />
those patients with strictly ocular signs and symptoms dur<strong>in</strong>g the first month after<br />
onset (40% of the 1487 patients), 66% subsequently developed cl<strong>in</strong>ically generalized<br />
disease; of these, 78% became generalized with<strong>in</strong> 1 year after onset of symptoms and<br />
94% with<strong>in</strong> 3 years.<br />
Ptosis <strong>in</strong> MG may occur as an isolated sign or <strong>in</strong> association with extraocular muscle<br />
<strong>in</strong>volvement. Evoli et al studied 48 patients with OM and noted that 10% had ptosis<br />
only, 90% had ptosis and extraocular muscle <strong>in</strong>volvement, and 25% had weakness of the<br />
orbicularis oculi (Evoli, 1988). The ptosis may be unilateral or bilateral and, when<br />
bilateral, is usually asymmetric. The ptosis may be absent when the patient awakens<br />
and appear later <strong>in</strong> the day, becom<strong>in</strong>g more pronounced as the day progresses.<br />
Prolonged upward gaze may <strong>in</strong>crease the ptosis. Enhanced or seesaw ptosis may be<br />
demonstrated (i.e., a worsen<strong>in</strong>g of ptosis on one side when the opposite eyelid is<br />
elevated and held <strong>in</strong> a fixed position). Enhancement of ptosis is not specific for MG and<br />
may rarely be seen with the Lambert-Eaton myasthenic syndrome, senile ptosis, ocular<br />
myopathy, Fisher’s syndrome, and even third nerve palsy (Averbuch-Heller, 1995;<br />
Brazis, 1997; Ishikawa, 1990). Dur<strong>in</strong>g refixation (a vertical saccade) from down to the<br />
primary position, the upper eyelid may either slowly beg<strong>in</strong> to droop or twitch several<br />
times before settl<strong>in</strong>g <strong>in</strong> a stable position (Cogan’s lid-twitch sign). This sign is<br />
characteristic, but not diagnostic, of MG (Phillips, 1997; Ragge, 1992). For example,<br />
Kao et al described two patients with fatigable ptosis due to <strong>in</strong>tracranial mass lesions<br />
(hematoma and metastasis) likely caus<strong>in</strong>g compression of the central caudal nucleus of<br />
the dorsal midbra<strong>in</strong> (Kao, 1999). MG may also be associated with three types of eyelid<br />
retraction (Miller, 1985): (1) contralateral eyelid retraction due to bilateral excessive<br />
<strong>in</strong>nervation (Her<strong>in</strong>g’s law) to raise the ptotic lid; (2) brief eyelid retraction last<strong>in</strong>g only<br />
seconds follow<strong>in</strong>g a saccade from downgaze to primary position (Cogan’s lid twitch<br />
sign); and (3) transient eyelid retraction last<strong>in</strong>g seconds or m<strong>in</strong>utes after star<strong>in</strong>g straight<br />
ahead or look<strong>in</strong>g upward for several seconds.<br />
Involvement of extraocular muscles with MG usually occurs <strong>in</strong> association with<br />
ptosis, though not always. MG should be considered <strong>in</strong> any case of ocular motor<br />
weakness without pupil <strong>in</strong>volvement because MG may mimic any pattern of neurogenic<br />
paresis. <strong>An</strong>y extraocular muscle may be selectively impaired, especially the<br />
medial rectus, and weakness characteristically <strong>in</strong>creases with susta<strong>in</strong>ed effort (Miller,<br />
1985; Odel, 1992; We<strong>in</strong>berg, 1994). Myasthenia can mimic pupil-spar<strong>in</strong>g third nerve<br />
palsies, superior division third nerve palsies, and fourth or sixth nerve palsies (Dehaene,<br />
1995; Miller, 1985; We<strong>in</strong>berg, 1994). Myasthenia may produce a false <strong>in</strong>ternuclear<br />
ophthalmoplegia (Ito, 1997), the one-and-a-half syndrome (Band<strong>in</strong>i, 2001), horizontal<br />
or vertical gaze palsy (Miller, 1985), divergence paresis (Lepore, 1999), double elevator<br />
palsy, and complete external ophthalmoplegia. MG may also be associated with
abnormalities of saccadic eye movements (Miller, 1985) <strong>in</strong>clud<strong>in</strong>g (1) hypermetric<br />
saccades; (2) hypometric saccades that beg<strong>in</strong> with normal velocity but ultimately show a<br />
decrease <strong>in</strong> velocity (<strong>in</strong>tersaccadic fatigue) and undershoot the target; (3) small, jerky,<br />
quiver<strong>in</strong>g eye movements; and (4) gaze-evoked nystagmus. Patients with MG often<br />
have weakness of the orbicularis oculi muscles. In some cases, a ‘‘peek sign’’ may occur.<br />
In an attempt to susta<strong>in</strong> forceful eye closure, the orbicularis oculi may fatigue, result<strong>in</strong>g<br />
<strong>in</strong> the patient ‘‘peek<strong>in</strong>g’’ through the partially opened palpebral fissure. Lower eyelid<br />
ectropion may occur <strong>in</strong> myasthenic patients, and become especially noticeable as the<br />
day progresses (Miller, 1985). F<strong>in</strong>ally, although abnormalities of pupillary function and<br />
accommodation have been described <strong>in</strong> MG, this dysfunction is not cl<strong>in</strong>ically significant<br />
(Miller, 1985; We<strong>in</strong>berg, 1994).<br />
In a study of 25 children with MG, more than half had had ocular symptoms<br />
(Mullaney, 2000). Generalization occurred <strong>in</strong> 5 of the 14 patients; ocular progression<br />
to systemic <strong>in</strong>volvement developed on average <strong>in</strong> 7.8 months (range 1 to 23 months).<br />
Long-term permanent damage to the extraocular muscles as a result of juvenile MG is<br />
rare.<br />
What Studies Are Suggested to Diagnosis<br />
Ocular Myasthenia Gravis?<br />
The diagnosis of OM is based on the cl<strong>in</strong>ical history and exam (fatigue, rest or sleep<br />
test), pharmacologic test<strong>in</strong>g (e.g., Tensilon), serologic test<strong>in</strong>g (e.g., antibody test<strong>in</strong>g), and<br />
electrophysiology (e.g., electromyography [EMG]). EMG <strong>in</strong>vestigations <strong>in</strong>clude study of<br />
the decremental response, conventional needle EMG, and s<strong>in</strong>gle-fiber record<strong>in</strong>gs. In<br />
some <strong>in</strong>stances, <strong>in</strong> vitro microelectrode studies of neuromuscular transmission and<br />
ultrastructural studies of the neuromuscular junction may be required to establish the<br />
diagnosis (Engel, 1994). In general, microelectrode and ultrastructural studies are<br />
reserved for patients with generalized MG and are not discussed here.<br />
The diagnosis of OM should be considered <strong>in</strong> any patient with ptosis and=or ocular<br />
motor weakness without pupillary <strong>in</strong>volvement. Weakness and fatigue conf<strong>in</strong>ed to the<br />
extraocular muscles or lids comb<strong>in</strong>ed with orbicularis oculi paresis is especially<br />
suggestive of OM. Significant cl<strong>in</strong>ical <strong>in</strong>volvement of the pupil, eye pa<strong>in</strong> or headaches,<br />
proptosis, visual loss, or <strong>in</strong>volvement of trigem<strong>in</strong>al sensation are not seen <strong>in</strong> MG.<br />
What Studies Are Used <strong>in</strong> the Pharmacologic<br />
Test<strong>in</strong>g for Ocular MG?<br />
Ocular Myasthenia Gravis 339<br />
A positive Tensilon (edrophonium hydrochloride) or Prostigm<strong>in</strong> (neostigm<strong>in</strong>e methylsulfate)<br />
test is usually, but not always, <strong>in</strong>dicative of ocular myasthenia. The improvement<br />
of extraocular muscle function should be quantified with prisms, a Hess screen, or<br />
the Lancaster red-green test (Coll, 1992). Ptosis tends to respond better to antichol<strong>in</strong>esterases<br />
than does ophthalmoparesis (Miller, 1985). Evoli studied 43 OM patients with<br />
both ptosis and diplopia and found that Tensilon relieved only the ptosis <strong>in</strong> 15 (35%)<br />
patients (Evoli, 1988). False-positive responses to antichol<strong>in</strong>esterases have been<br />
described with bra<strong>in</strong>stem and parasellar tumors, aneurysms, metastasis to the orbital
340 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
apex, multiples sclerosis, Lambert-Eaton myasthenic syndrome, poliomyelitis, Guilla<strong>in</strong>-<br />
Barré syndrome, motor neuron disease, botulism, orbital myositis, congenital ptosis,<br />
snake bites, diabetic sixth nerve palsy, and dermatomyositis (Miller, 1985; Ragge, 1992;<br />
Shams, 2002; Straube, 1990; We<strong>in</strong>berg, 1994). In most of these reports, the correct<br />
diagnosis was evident by associated neurologic signs and symptoms. Moorthy et al,<br />
however, described eight cases orig<strong>in</strong>ally diagnosed as hav<strong>in</strong>g MG <strong>in</strong> whom an<br />
<strong>in</strong>tracranial lesion <strong>in</strong>stead of, or <strong>in</strong> addition to, MG was later identified (Moorthy,<br />
1989). Four of these patients probably had both MG and an <strong>in</strong>tracranial lesion, but the<br />
other four had only <strong>in</strong>tracranial lesions with cl<strong>in</strong>ical ‘‘pseudo-myasthenic’’ features,<br />
<strong>in</strong>clud<strong>in</strong>g fatigable weakness, Cogan’s lid twitch sign, and positive Tensilon or<br />
Prostigm<strong>in</strong> tests. Three had pupil-spar<strong>in</strong>g third nerve palsies and one had a third<br />
nerve palsy associated with a sixth nerve palsy. These authors suggested that patients<br />
with cl<strong>in</strong>ical features consistent with MG restricted to the ocular or cranial muscles<br />
should be carefully evaluated for <strong>in</strong>tracranial lesions us<strong>in</strong>g computed tomography (CT)<br />
or magnetic resonance imag<strong>in</strong>g (MRI). We do not rout<strong>in</strong>ely perform neuroimag<strong>in</strong>g on<br />
all patients with OM (class IV, level C). Miller suggests that it is advisable to rule out an<br />
<strong>in</strong>tracranial lesion by CT or MR imag<strong>in</strong>g <strong>in</strong> all patients with isolated, unilateral, pupilspar<strong>in</strong>g<br />
ophthalmoparesis even when the diagnosis of MG seems assured by a positive<br />
Tensilon or Prostigm<strong>in</strong> test or other studies (Miller, 1985) (class IV, level C).<br />
A negative Tensilon or Prostigm<strong>in</strong> test does not rule out MG (Evoli, 1988; Miller, 1985;<br />
We<strong>in</strong>berg, 1994). For example, Spector and Daroff noted negative responses to Tensilon<br />
<strong>in</strong> 2 of 11 (18%) OM and <strong>in</strong> 6 of 21 (29%) of patients with both OM and generalized MG<br />
(Spector, 1976). Paradoxical responses to Tensilon may also occur <strong>in</strong> OM patients,<br />
<strong>in</strong>clud<strong>in</strong>g paresis of previously nonparetic muscles and <strong>in</strong>creased eye misalignment due<br />
to further weaken<strong>in</strong>g of paretic muscles.<br />
What Nonpharmacologic Test<strong>in</strong>g Is Helpful <strong>in</strong><br />
the Diagnosis of Myasthenia Gravis?<br />
The ‘‘sleep test’’ may also be <strong>in</strong>corporated to demonstrate objective improvement <strong>in</strong> MG<br />
symptoms after rest (Odel, 1991). The patient is kept <strong>in</strong> a quiet, darkened room and<br />
<strong>in</strong>structed to close the eyes and rest for 30 m<strong>in</strong>utes. The ptosis and ocular motility are<br />
quantified before and after the rest period. This study may be positive <strong>in</strong> some Tensilonnegative<br />
MG but may also be negative <strong>in</strong> Tensilon-positive patients (We<strong>in</strong>berg, 1994).<br />
<strong>An</strong>other non<strong>in</strong>vasive test is the ice-pack test, which may be useful <strong>in</strong> the diagnosis of<br />
OM <strong>in</strong> the patient with ptosis (Golnik, 1999; Kubis, 2000; Sethi, 1987). Ice <strong>in</strong> a surgical<br />
glove is placed over one lightly closed eye for 2 m<strong>in</strong>utes or to the limit of patient<br />
tolerance. In cases of bilateral ptosis, the opposite (uncooled) eye serves as control. The<br />
palpebral fissures are measured before and after the ice is applied. Sethi et al noted<br />
improvement of ptosis <strong>in</strong> 8 of 10 MG patients (Sethi, 1987), and Golnik et al found the<br />
test to be positive <strong>in</strong> 16 of 20 (80%) of patients with MG and none of 20 patients with<br />
ptosis not due to MG (Golnik, 1999). In four patients with MG and complete ptosis,<br />
however, the ice pack test was negative, and thus the sensitivity of the test <strong>in</strong> patients<br />
with complete ptosis decreases considerably. It is thought that the decreased<br />
temperature may <strong>in</strong>hibit acetylchol<strong>in</strong>esterase function (Golnik, 1999). Improvement of<br />
eyelid elevation after the ice test is <strong>in</strong> part caused by rest, but the ice significantly<br />
improved ptosis more than rest alone <strong>in</strong> one study (Kubis, 2000). In another study,
however, myasthenic ptosis was markedly improved <strong>in</strong> four patients regardless of local<br />
cool<strong>in</strong>g or warm<strong>in</strong>g, with the common denom<strong>in</strong>ator of these tests be<strong>in</strong>g rest rather than<br />
temperature per se (Movaghar, 2000).<br />
Electrophysiologic test<strong>in</strong>g might establish the diagnosis of MG. EMG f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong>clude<br />
fluctuations <strong>in</strong> the amplitude and duration of motor unit potentials recorded dur<strong>in</strong>g<br />
voluntary activity; decremental responses of evoked compound muscle action potentials<br />
to repetitive supramaximal motor nerve stimulation; and s<strong>in</strong>gle-fiber electromyography<br />
(SFEMG) abnormalities (e.g., impulse block<strong>in</strong>g and <strong>in</strong>creased ‘‘jitter’’) (Engel,<br />
1994; Hermann, 1996; Miller, 1985; Odel, 1991). Repetitive stimulation studies must<br />
<strong>in</strong>clude proximal and facial nerves to <strong>in</strong>crease diagnostic yield. The diagnostic yield of<br />
these studies <strong>in</strong> patients with OM is unknown, but Sanders and Howard observed<br />
decremental responses <strong>in</strong> hand or shoulder muscles <strong>in</strong> only 10% of patients with OM<br />
(Sanders, 1986).<br />
If repetitive nerve stimulation studies are negative <strong>in</strong> a patient with suspected MG,<br />
SFEMG studies might be useful. SFEMG is positive <strong>in</strong> 75% of myasthenic patients <strong>in</strong><br />
remission, 80 to 88% of those with ocular signs and symptoms only, 91 to 100% of<br />
patients with generalized symptoms, and 88 to 94% of patients with myasthenia overall<br />
(Emeryk, 1990; Sanders, 1986; We<strong>in</strong>berg, 1994). For example, <strong>in</strong> one study SFEMG <strong>in</strong><br />
limb muscles was abnormal <strong>in</strong> 17 of 20 patients with OM (Emeryk, 1990). SFEMG is<br />
quite sensitive for detect<strong>in</strong>g abnormalities of the neuromuscular junction but is not<br />
specific for MG. In another study of OM, SFEMG showed the highest sensitivity (100%),<br />
whereas acetylchol<strong>in</strong>e receptor antibodies studies showed the highest specificity (100%)<br />
for diagnosis (Padua, 2000). SFEMG of the frontalis muscle may be a sensitive technique<br />
for the diagnosis of OM (Valls-Canals, 2000).<br />
What Is the Diagnostic Utility of<br />
<strong>An</strong>tiacetylchol<strong>in</strong>e Receptor <strong>An</strong>tibodies <strong>in</strong> the<br />
Diagnosis of Myasthenia Gravis?<br />
AChR antibody titers are quite useful <strong>in</strong> the diagnosis of MG. In one large and<br />
representative study, the percentage of positive tests <strong>in</strong> different cl<strong>in</strong>ical forms of MG<br />
were as follows: remission, 24%; ocular, 50%; mild generalized, 80%; moderately severe<br />
or acutely severe, 100%; chronic severe, 89% (T<strong>in</strong>dall, 1981). Overall, AChR antibodies<br />
are positive <strong>in</strong> 80 to 95% of patients with generalized MG and 34 to 56% of those with<br />
OM (Evoli, 1988; Oosterhuis, 1988; We<strong>in</strong>berg, 1994). Test<strong>in</strong>g for AChR b<strong>in</strong>d<strong>in</strong>g, block<strong>in</strong>g,<br />
and modulat<strong>in</strong>g antibodies <strong>in</strong>creases the assay yield <strong>in</strong> patients with generalized<br />
MG and OM. In OM, the antibody titer tends to be low, and the serum antibody titer<br />
correlates poorly with the severity of MG when a group of patients is studied (Engel,<br />
1994).<br />
Summary of Test<strong>in</strong>g for Ocular Myasthenia<br />
Gravis<br />
Ocular Myasthenia Gravis 341<br />
No test is specific for OM, and its diagnosis should not be based exclusively on any<br />
s<strong>in</strong>gle test. Kelly et al advised that all patients with suspected MG should have serum
342 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
assays of AChR antibodies, repetitive stimulation studies, and SFEMG, <strong>in</strong> addition to a<br />
Tensilon or Prostigm<strong>in</strong> test (Kelly, 1982). These procedures confirm the diagnosis <strong>in</strong> at<br />
least 95% of patients. Muscle biopsy with receptor assay, <strong>in</strong> addition to these studies,<br />
should diagnose close to 100% of patients, <strong>in</strong>clud<strong>in</strong>g those with OM (Miller, 1985). Oh<br />
et al studied 20 patients diagnosed with OM and found SFEMG positive <strong>in</strong> 80%, AChR<br />
antibodies present <strong>in</strong> 70%, and repetitive nerve stimulation studies positive <strong>in</strong> 35 to 45%<br />
(Oh, 1992). These authors advised <strong>in</strong>itial AChR antibody assay and repetitive nerve<br />
stimulation studies, and follow-up SFEMG if the first two studies are normal. Evoli et al<br />
studied 48 patients with OM and found Tensilon tests positive <strong>in</strong> 47, repetitive<br />
nerve stimulation of the limb muscles positive <strong>in</strong> 50% (24 of 48), and elevated AChR<br />
antibody titers <strong>in</strong> 45% (20 of 44) (Evoli, 1988). In another study of 19 Tensilon-positive<br />
OM patients, Tsujihata et al found that 6 of 16 (38%) were seronegative for AChR<br />
antibodies. Eight of 13 (62%) had normal SFEMG of arm muscles, and 15 of 17 (88%)<br />
had normal repetitive nerve stimulation study of the facial nerve to the orbicularis oculi<br />
muscles (Tsujihata, 1989).<br />
Should CT Imag<strong>in</strong>g of the Chest for<br />
Thymoma Be Performed <strong>in</strong> MG?<br />
Because there is an <strong>in</strong>creased risk of thymoma <strong>in</strong> patients with MG, all patients with the<br />
diagnosis of MG should undergo CT or MRI of the mediast<strong>in</strong>um. Thymoma occurs <strong>in</strong> 5<br />
to 20% of myasthenic patients overall, and about one third to one half of those with<br />
thymoma have MG. The risk of thymoma <strong>in</strong> patients with OM is probably lower: 4% <strong>in</strong><br />
patients with OM compared to 12% <strong>in</strong> those with generalized MG <strong>in</strong> one series<br />
(Papetestas, 1971). Thymoma is more common <strong>in</strong> older patients and <strong>in</strong> patients with<br />
high AChR antibody titers (Oger, 1993). In a large series of patients with MG, striated<br />
muscle antibodies were present <strong>in</strong> 84% of patients with thymoma (Limburg, 1983). In<br />
those without thymoma, striational antibodies were found <strong>in</strong> 5% or 47%, respectively, of<br />
patients <strong>in</strong> whom the onset of MG was before or after the age of 40. Thyroid disease<br />
may be associated with MG and sensitive thyroid-stimulat<strong>in</strong>g hormone (TSH) levels<br />
might detect subcl<strong>in</strong>ical or asymptomatic associated thyroid disease (class IV, level C).<br />
<strong>An</strong> approach to the diagnosis and evaluation of patients with possible OM is outl<strong>in</strong>ed<br />
<strong>in</strong> Figure 15–1.<br />
What Is the Suggested Management of OM?<br />
Patients with pure OM must be warned of the possibility of generalization of the disease<br />
process and should specifically be <strong>in</strong>structed to <strong>in</strong>form their physician immediately if<br />
symptoms such as dysphagia, respiratory <strong>in</strong>volvement, or extremity weakness develop.<br />
Good diet (e.g., potassium), adequate rest, and avoidance of precipitants (e.g., medications<br />
that worsen MG) are reasonable.<br />
For patients with OM, if the diplopia or ptosis is mild, then observation or patch<strong>in</strong>g<br />
one eye may be sufficient. Ptosis may be elim<strong>in</strong>ated <strong>in</strong> some patients by hav<strong>in</strong>g a crutch<br />
attachment placed on a spectacle frame for one or both eyes, although this often causes<br />
irritation of the eyes from exposure (Miller, 1985). Ptosis surgery may be performed <strong>in</strong><br />
some patients, particularly those who are refractory to medical therapy or <strong>in</strong> whom
Figure 15–1. Evaluation of ocular myasthenia (OM) gravis.<br />
Ocular Myasthenia Gravis 343<br />
ptosis is a predom<strong>in</strong>ant f<strong>in</strong>d<strong>in</strong>g (Miller, 1985). In some patients, prisms can alleviate<br />
diplopia, particularly when there is a relatively comitant deviation.<br />
For more severe ocular motor weakness, antichol<strong>in</strong>esterase agents, such as pyridostigm<strong>in</strong>e<br />
bromide (Mest<strong>in</strong>on), are warranted, although these agents often do not<br />
succeed <strong>in</strong> correct<strong>in</strong>g the diplopia. Diplopia is often more refractory to treatment than
344 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
ptosis. If moderate or large doses of antichol<strong>in</strong>esterase drugs fail or cannot be tolerated<br />
and symptoms are troublesome, then corticosteroids, often at relatively low alternateday<br />
doses, are usually effective <strong>in</strong> correct<strong>in</strong>g the diplopia (Agius, 2000; Evoli, 1988;<br />
Kupersmith, 1996). Some authors, however, suggest that corticosteroids be used for OM<br />
only if patients demand their use, or if there is severe bilateral ptosis or severe<br />
ophthalmoplegia that precludes useful vision (Kam<strong>in</strong>ski, 2000). Some authors have<br />
suggested azathiopr<strong>in</strong>e for MG that is <strong>in</strong>adequately controlled on low-dose steroids or<br />
for those patients with <strong>in</strong>tolerable steroid side effects (We<strong>in</strong>berg, 1994). Cyclophosphamide,<br />
cyclospor<strong>in</strong>e, <strong>in</strong>travenous immunoglobul<strong>in</strong>, and plasmapheresis have also<br />
been used but are not usually recommended for OM because their benefit-risk ratios<br />
have not been adequately studied (class IV, level U).<br />
What About Thymectomy for OM?<br />
The presence of a thymoma <strong>in</strong> any patient with MG is an <strong>in</strong>dication for thymectomy<br />
(Engel, 1994) (class III, level B). Patients with OM should be evaluated with mediast<strong>in</strong>al<br />
CT or MRI. Although thymectomy can be effective <strong>in</strong> OM without thymoma and may<br />
prevent generalization of the disease, most cl<strong>in</strong>icians are reluctant to recommend this<br />
procedure for purely ocular symptoms (Lanska, 1990). Transsternal thymectomy was<br />
studied <strong>in</strong> 22 cases of purely OM. Remission was def<strong>in</strong>ed as complete freedom from<br />
symptoms without medications for more than 3 months. The remission rates <strong>in</strong>creased<br />
with time from 11.8% at 3 years to 23.1% at 5 years and 33.3% at 10 years (Nakamura,<br />
1996). Those patients undergo<strong>in</strong>g thymectomy with<strong>in</strong> 12 months of symptom onset<br />
showed a significantly earlier and better chance of remission compared to patients<br />
undergo<strong>in</strong>g thymectomy longer than 12 months after symptom onset. The authors<br />
concluded that thymectomy for OM <strong>in</strong> the earlier stages of the disease is the preferred<br />
treatment, just as for generalized MG. <strong>An</strong>other study reviewed 61 patients with OM<br />
who underwent thymectomy and who were followed for a mean of 9 years (Roberts,<br />
2001). Thymoma was present <strong>in</strong> 12 patients. Overall, 71% were cured (51%) or improved<br />
(20%) by thymectomy, with 16 patients (26%) unchanged, one worsen<strong>in</strong>g, and one<br />
dy<strong>in</strong>g <strong>in</strong> the postoperative period. Improvement or cure was noted <strong>in</strong> 67% of the<br />
thymoma group. The authors concluded that thymectomy is safe and effective for OM.<br />
We do not generally recommend thymectomy (without thymoma) for OM (class IV,<br />
level C). From 20 to 50% of patients with OM go <strong>in</strong>to remission without thymectomy,<br />
and no controlled studies have compared this to a surgical group. The argument for<br />
thymectomy will rema<strong>in</strong> unconv<strong>in</strong>c<strong>in</strong>g unless a prospective trial compar<strong>in</strong>g<br />
thymectomy to medical management is performed.<br />
What Is the Prognosis of OM? Does the Use of<br />
Corticosteroids Alter the Prognosis?<br />
About 10 to 20% of patients with OM undergo spontaneous remission that may be<br />
temporary or permanent. Although corticosteroid treatment produces a higher <strong>in</strong>cidence<br />
of remission and improvement, there is no evidence that antichol<strong>in</strong>esterase agents<br />
affect the course of the disease (Kupersmith, 1996). Kupersmith et al reported a<br />
retrospective review of 32 patients with OM who were treated with corticosteroids<br />
and followed for a m<strong>in</strong>imum of 2 years (Kupersmith, 1996). Diplopia was <strong>in</strong>itially
present <strong>in</strong> the primary position <strong>in</strong> 29 patients and <strong>in</strong> downgaze position <strong>in</strong> 26. Ptosis<br />
was present <strong>in</strong> 24 patients (unilateral <strong>in</strong> 13 and bilateral <strong>in</strong> 11). Tensilon test was positive<br />
<strong>in</strong> 31 patients (one patient did not have a Tensilon test but had marked fatigability of the<br />
lids and ocular muscles that recovered with rest). SFEMG or repetitive stimulation<br />
studies were abnormal <strong>in</strong> six of 19 patients, and AChR antibodies were elevated <strong>in</strong> 10 of<br />
28 patients. CT scan of the chest revealed no thymomas <strong>in</strong> any of the 32 patients.<br />
Patients were treated with one or more courses of daily prednisone (the highest <strong>in</strong>itial<br />
dose, 40 to 80 mg) gradually withdrawn over 4 to 6 weeks. Subsequently, <strong>in</strong> six patients,<br />
2.5 to 20 mg of prednisone was given on alternate days for more than 6 months. No<br />
patients had major steroid complications. Two years after diagnosis, diplopia was found<br />
<strong>in</strong> primary position <strong>in</strong> 11 patients and <strong>in</strong> downward gaze <strong>in</strong> 11 patients (9 had diplopia<br />
<strong>in</strong> primary gaze), and 66% of patients had normal ocular function. Ptosis was found <strong>in</strong><br />
seven patients. Generalized MG had developed <strong>in</strong> three patients (9.4%) at 2 years;<br />
elevated AChR antibody levels and abnormal electrophysiologic studies were not<br />
predictive of worsen<strong>in</strong>g. Of the 16 patients who had follow-up for 3 years and the 13<br />
for 4 or longer, ocular motility was normal <strong>in</strong> 56% at 3 years and 62% at 4 years, with 2<br />
additional patients develop<strong>in</strong>g generalized MG at 4 years. The authors concluded that<br />
moderate-dose daily prednisone for 4 to 6 weeks, followed by low-dose, alternate-day<br />
therapy as needed, can control the diplopia of OM, and that the frequency of<br />
deterioration to generalized MG at 2 years may be reduced.<br />
Sommer et al retrospectively analyzed 78 patients with OM with a mean disease<br />
duration of 8.3 years (Sommer, 1997). In 54 patients (69%), symptoms and signs<br />
rema<strong>in</strong>ed conf<strong>in</strong>ed to the extraocular muscles dur<strong>in</strong>g the observation period, whereas<br />
the rema<strong>in</strong><strong>in</strong>g 24 patients (31%) developed symptoms of generalized myasthenia (50%<br />
of them with<strong>in</strong> 2 years and 75% with<strong>in</strong> 4 years after onset). There was a slightly reduced<br />
risk of generalization for patients with mild symptoms, normal repetitive stimulation<br />
studies, and low or absent AChR antibodies. Patients receiv<strong>in</strong>g immunosuppressive<br />
treatment (corticosteroids and=or azathiopr<strong>in</strong>e) rarely developed generalized MG (6 of<br />
50 [12%]). Those without such treatment, usually due to uncerta<strong>in</strong> diagnosis and late<br />
referral, converted <strong>in</strong>to generalized MG significantly more often (18 of 28 [64%]). The<br />
authors concluded that short-term corticosteroids and long-term azathiopr<strong>in</strong>e seemed<br />
adequate for achiev<strong>in</strong>g remission <strong>in</strong> most patients. The proportion of patients develop<strong>in</strong>g<br />
generalized MG was smaller <strong>in</strong> this population compared to previously<br />
published groups and early immunosuppressive treatment was thought to be at least<br />
partially responsible for this f<strong>in</strong>d<strong>in</strong>g. Thymectomy (performed <strong>in</strong> 12 patients with an<br />
abnormal chest CT) also correlated with a good outcome, but had no apparent<br />
advantage over medical treatment alone (Sommer, 1997). Until a prospective cl<strong>in</strong>ical<br />
trial of corticosteroids or other immunosuppressive is performed <strong>in</strong> patients with OM,<br />
the value of these agents <strong>in</strong> the prevention of the development of generalized MG<br />
rema<strong>in</strong>s undef<strong>in</strong>ed (class III–IV, level C). F<strong>in</strong>ally, stable disabl<strong>in</strong>g diplopia may<br />
occasionally respond favorably to strabismus surgery or <strong>in</strong>jection of the extraocular<br />
muscle with botul<strong>in</strong>um (Bentley, 2001).<br />
References<br />
Ocular Myasthenia Gravis 345<br />
Agius MA. (2000). Treatment of ocular myasthenia with corticosteroids—yes. Arch <strong>Neuro</strong>l 57:750–751.<br />
Averbuch-Heller L, Poonyathalang A, von Maydell RD, Remler BF. (1995). Her<strong>in</strong>g’s law for eyelids: still valid.<br />
<strong>Neuro</strong>logy 45:1781–1783.
346 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Band<strong>in</strong>i F, Faga D, Simonetti S. (2001). Ocular myasthenia mimick<strong>in</strong>g a one-and-a-half syndrome. J <strong>Neuro</strong>ophthalmol<br />
21:210–211.<br />
Bentley CR, Dawson E, Lee JP. (2001). Active management <strong>in</strong> patients with ocular manifestations of myasthenia<br />
gravis. Eye 15:18–22.<br />
Bever CT Jr, Aqu<strong>in</strong>o AV, Penn AS, et al. (1983). Prognosis of ocular myasthenia gravis. <strong>An</strong>n <strong>Neuro</strong>l 14:516–519.<br />
Brazis PW. (1997). Enhanced ptosis <strong>in</strong> Lambert-Eaton myasthenic syndrome. J <strong>Neuro</strong>-ophthalmol 17:202–203.<br />
Coll GE, Demer JL. (1992). The Edrophonium-Hess screen test <strong>in</strong> the diagnosis of ocular myasthenia gravis. Am<br />
J Ophthalmol 114:489–493.<br />
Dehaene I, van Zandijcke M. (1995). Isolated paralysis of the superior division of the ocular motor nerve<br />
mimicked by myasthenia gravis. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:257–258.<br />
Emeryk B, Row<strong>in</strong>ska-Marc<strong>in</strong>ska K, Nowak-Michalska T. (1990). Pseudoselectivity of the neuromuscular block <strong>in</strong><br />
ocular myasthenia: a SFEMG study. Electromyograph Cl<strong>in</strong> <strong>Neuro</strong>physiol 30:53–59.<br />
Engel AG. (1994). Disturbances of neuromuscular transmission. Acquired autoimmune myasthenia gravis.<br />
In: Engel AG, Franz<strong>in</strong>i-Armstrong C, eds. Myology. Basic and <strong>Cl<strong>in</strong>ical</strong>. 2nd ed. New York, McGraw-Hill,<br />
pp. 1769–1797.<br />
Evoli A, Tonali P, Bartoccioni AP, Lo Monaco M. (1988). Ocular myasthenia: diagnostic and therapeutic problems.<br />
Acta <strong>Neuro</strong>l Scand 77:31–35.<br />
Golnik KC, Pena R, Lee AG, Eggenberger ER. (1999). <strong>An</strong> ice test for the diagnosis of myasthenia gravis.<br />
Ophthalmology 106:1282–1286.<br />
Hermann RC Jr. (1996). Repetitive stimulation studies. In: Daube JR, ed. <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>physiology. Philadelphia, FA<br />
Davis, pp. 237–247.<br />
Ishikawa H, Wakakura M, Ishikawa S. (1990). Enhanced ptosis <strong>in</strong> Fisher’s syndrome after Epste<strong>in</strong>-Barr virus<br />
<strong>in</strong>fection. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:197–200.<br />
Ito K, Mizutani J, Murofushi T, Mizuno M. (1997). Bilateral pseudo-<strong>in</strong>ternuclear ophthalmoplegia <strong>in</strong> myasthenia<br />
gravis. J ORL Related Specialties 59:122–126.<br />
Kam<strong>in</strong>ski HJ, Daroff RB. (2000). Treatment of ocular myasthenia. Steroids only when compelled. Arch <strong>Neuro</strong>l<br />
57:752–753.<br />
Kao Y-F, Lan M-Y, Chou M-S, Chen W-H. (1999). Intracranial fatigable ptosis. J <strong>Neuro</strong>-ophthalmol 19:257–259.<br />
Kelly JJ Jr, Daube JR, Lennon VA, et al. (1982). The laboratory diagnosis of mild myasthenia gravis. <strong>An</strong>n <strong>Neuro</strong>l<br />
12:238–242.<br />
Kubis KC, Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Sergott RC. (2000). The ice test versus rest test <strong>in</strong> myasthenia gravis.<br />
Ophthalmology 107:1995–1998.<br />
Kupersmith MJ, Moster M, Bhuiyan S, et al. (1996). Beneficial effects of corticosteroids on ocular myasthenia<br />
gravis. Arch <strong>Neuro</strong>l 53:802–804.<br />
Lanska DJ. (1990). Indications for thymectomy <strong>in</strong> myasthenia gravis. <strong>Neuro</strong>logy 40:1828–1829.<br />
Lepore FE. (1999). Divergence paresis: a nonlocaliz<strong>in</strong>g cause of diplopia. J <strong>Neuro</strong>-ophthalmol 19:242–245.<br />
Limburg PC, The TH, Hummel-Tappel E, Oosterhuis HJ. (1983). <strong>An</strong>ti-acetylchol<strong>in</strong>e receptor antibodies <strong>in</strong><br />
myasthenia gravis. Part 1: relation to cl<strong>in</strong>ical parameters <strong>in</strong> 250 patients. J <strong>Neuro</strong>l Sci 58:357–370.<br />
Litchy WJ. (1996). Quantitative electromyography and s<strong>in</strong>gle-fiber electromyography. In: Daube JR, ed. <strong>Cl<strong>in</strong>ical</strong><br />
<strong>Neuro</strong>physiology. Philadelphia, FA Davis, pp. 282–300.<br />
Miller NR. (1985). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams & Wilk<strong>in</strong>s, pp. 841–891.<br />
Moorthy G, Behrens MM, Drachman DB, et al. (1989). Ocular pseudomyasthenia or ocular myasthenia ‘‘plus’’: a<br />
warn<strong>in</strong>g to cl<strong>in</strong>icians. <strong>Neuro</strong>logy 39:1150–1154.<br />
Movaghar M, Slav<strong>in</strong> ML. (2000). Effect of local heat versus ice on blepharoptosis result<strong>in</strong>g from ocular<br />
myasthenia. Ophthalmology 107:2209–2214.<br />
Mullaney P, Vajsar J, Smith R, Buncic JR. (2000). The natural history and ophthalmic <strong>in</strong>volvement <strong>in</strong> childhood<br />
myasthenia gravis at the Hospital for Sick Children. Ophthalmology 107:504–510.<br />
Nakamura H, Taniguchi Y, Suzuki Y, et al. (1996). Delayed remission after thymectomy for myasthenia gravis of<br />
the purely ocular type. J Thorac Cardiovasc Surg 112:371–375.<br />
Odel J. (1992). Ocular myasthenia. Presented at the North American <strong>Neuro</strong>-Ophthalmology Society meet<strong>in</strong>g,<br />
Rancho Bernardo, California, February.<br />
Odel J, W<strong>in</strong>terkorn J, Behrens M. (1991). The sleep test for myasthenia gravis—a safe alternative to Tensilon. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 11:288–292.<br />
Oger JJF. (1993). Thymus histology and acetylchol<strong>in</strong>e receptor antibodies <strong>in</strong> generalized myasthenia gravis. <strong>An</strong>n<br />
NY Acad Sci 681:110–112.<br />
Oh SJ, Kim DE, Kuruoglu A, et al. (1992). Diagnostic sensitivity of the laboratory tests <strong>in</strong> myasthenia gravis.<br />
Muscle Nerve 15:720–724.
Ocular Myasthenia Gravis 347<br />
Oosterhuis HJGH. (1982). The ocular signs and symptoms of myasthenia gravis. Doc Ophthalmol 52:363–378.<br />
Oosterhuis HJGH. (1988). Long-term effects of treatment <strong>in</strong> 374 patients with myasthenia gravis. Monogr Allergy<br />
25:75–85.<br />
Padua L, Stalberg E, LoMonaco M, et al. (2000). SFEMG <strong>in</strong> ocular myasthenia gravis diagnosis. Cl<strong>in</strong> <strong>Neuro</strong>physiol<br />
111:1203–1207.<br />
Papetestas AE, Genk<strong>in</strong>s G, Horowitz SH, Kornfield P. (1971). Studies <strong>in</strong> myasthenia gravis: effects of thymectomy.<br />
Results <strong>in</strong> 1985 patients with nonthymomatous and thymomatous myasthenia gravis, 1941–1969. Am J Med<br />
50:465–476.<br />
Phillips PH, Newman NJ. (1997). Here today ... gone tomorrow. Surv Ophthalmol 41:354–356.<br />
Ragge NK, Hoyt WF. (1992). Midbra<strong>in</strong> myasthenia: fatiguable ptosis, ‘‘lid twitch’’ sign, and ophthalmoparesis<br />
from a dorsal midbra<strong>in</strong> glioma. <strong>Neuro</strong>logy 42:917–919.<br />
Roberts PF, Venuto F, Rend<strong>in</strong>a E, et al. (2001). Thymectomy <strong>in</strong> the treatment of ocular myasthenia gravis. J Thorac<br />
Cardiovasc Surg 122:562–568.<br />
Sanders DB, Howard JF. (1986). S<strong>in</strong>gle fiber EMG <strong>in</strong> myasthenia gravis. Muscle Nerve 9:809–819.<br />
Sethi KD, Rivner MH, Thomas TR. (1987). Ice pack test for myasthenia gravis. <strong>Neuro</strong>logy 37:1383–1385.<br />
Shams PN, Waldman A, Plant GT. (2002). B cell lymphoma of the bra<strong>in</strong> stem masquerad<strong>in</strong>g as myasthenia. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 72:271–273.<br />
Sommer N, Sigg B, Melms A, et al. (1997). Ocular myasthenia gravis: response to long term immunosuppressive<br />
treatment. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 62:156–162.<br />
Spector RH, Daroff RB. (1976). Edrophonium <strong>in</strong>frared optok<strong>in</strong>etic nystagmography <strong>in</strong> the diagnosis of myasthenia<br />
gravis. <strong>An</strong>n NY Acad Sci 274:642–651.<br />
Straube A, Witt T. (1990). Oculo-bulbar myasthenic symptoms as the sole sign of tumour <strong>in</strong>volv<strong>in</strong>g or<br />
compress<strong>in</strong>g the bra<strong>in</strong>stem. J <strong>Neuro</strong>l 237:369–371.<br />
T<strong>in</strong>dall RSA. (1981). Humoral immunity <strong>in</strong> myasthenia gravis: biochemical characterization of acquired antireceptor<br />
antibodies and cl<strong>in</strong>ical correlation. <strong>An</strong>n <strong>Neuro</strong>l 10:437–447.<br />
Tsujihata M, Yoshimura T, Satoh A, et al. (1989). Diagnostic significance of IgG, C3, and C9 at the limb muscle<br />
motor end-plate <strong>in</strong> m<strong>in</strong>imal myasthenia gravis. <strong>Neuro</strong>logy 39:1359–1363.<br />
Valls-Canals J, Montero J, Pradas J. (2000). Stimulated s<strong>in</strong>gle fiber EMG of the frontalis muscle <strong>in</strong> the diagnosis of<br />
ocular myasthenia gravis. Muscle Nerve 23:779–783.<br />
We<strong>in</strong>berg DA, Lesser RL, Vollmer TL. (1994). Ocular myasthenia: a protean disorder. Surv Ophthalmol 39:169–210.
This page <strong>in</strong>tentionally left blank
16 r<br />
Thyroid Eye Disease: Graves’<br />
Ophthalmopathy<br />
What Are the Typical <strong>Cl<strong>in</strong>ical</strong> Features of<br />
Graves’ Ophthalmopathy?<br />
Graves’ ophthalmopathy (GO) is characterized cl<strong>in</strong>ically by the signs listed <strong>in</strong><br />
Table 16–1. Patients without the typical features of GO should undergo further<br />
evaluation for other etiologies of their signs: proptosis (e.g., orbital tumor or pseudotumor),<br />
strabismus (e.g., myasthenia gravis), and lid retraction (see Chapter 19).<br />
Although we do not typically image GO without compressive optic neuropathy, we<br />
do recommend neuroimag<strong>in</strong>g and orbital imag<strong>in</strong>g for patients with atypical features for<br />
GO (class IV, level C).<br />
What Are the Imag<strong>in</strong>g F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Graves’<br />
Ophthalmopathy?<br />
Orbital imag<strong>in</strong>g, such as computed tomography (CT) and magnetic resonance imag<strong>in</strong>g<br />
(MRI) scans, often demonstrate proptosis, extraocular muscle (EOM) enlargement<br />
spar<strong>in</strong>g the tendons (Just, 1991; Ozgen, 1999), <strong>in</strong>creased orbital fat volume (Chang,<br />
1990; Char, 1991; Firbank, 2000), and sometimes engorgement of the superior ophthalmic<br />
ve<strong>in</strong>. MRI may be superior to CT scan <strong>in</strong> differentiat<strong>in</strong>g EOM edema (with elevated<br />
T2 relaxation times) from fibrosis (Just, 1991; Muller-Forell, 1999; Nagy, 2000). Serial<br />
short tau <strong>in</strong>version recovery (STIR) sequence MRI correlates with the cl<strong>in</strong>ical activity<br />
score (Mayer, 2001). Ultrasonography of the orbit can also demonstrate EOM enlargement<br />
consistent with GO. MRI, however, is usually more costly than CT imag<strong>in</strong>g.<br />
Apical compression of the optic nerve <strong>in</strong> compression optic neuropathy (CON) may<br />
be seen on CT or MRI. Coronal as well as axial images are useful <strong>in</strong> the radiographic<br />
diagnosis of CON <strong>in</strong> GO (So, 2000). We recommend orbital imag<strong>in</strong>g <strong>in</strong> patients with<br />
349
350 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 16–1. Typical Features of Graves’ Ophthalmopathy (GO)<br />
Eyelid signs<br />
Lid retraction (the most common cl<strong>in</strong>ical feature of GO) (Bartley, 1994, 1995, 1996)<br />
Stare<br />
Lid lag <strong>in</strong> downgaze<br />
Exophthalmos<br />
Enlargement of extraocular muscles<br />
Increased orbital fat volume (Hudson, 1991)<br />
Increased <strong>in</strong>traocular pressure (Danesh-Meyer, 2001; Kalmann, 1998)<br />
Diplopia=ophthalmoplegia secondary to extraocular muscle <strong>in</strong>flammation or fibrosis<br />
Visual loss<br />
Exposure keratopathy<br />
Compressive optic neuropathy (CON) from extraocular muscle <strong>in</strong>volvement <strong>in</strong> the orbital apex<br />
(Liu, 1992; Mourits, 1990a,b; Nagy, 2000; Trobe, 1978)<br />
Stretch<strong>in</strong>g of the optic nerve due to proptosis (Liu, 1992)<br />
Flash<strong>in</strong>g lights (Danks, 1998)<br />
Signs and symptoms of orbital congestion<br />
Due to proptosis with or without venous outflow obstruction<br />
Conjunctival <strong>in</strong>jection and chemosis<br />
Eyelid and periorbital edema<br />
Tear<strong>in</strong>g, photophobia, and orbital discomfort (Bahn, 1990; Nunery, 1991; Srivastava, 2000)<br />
cl<strong>in</strong>ical evidence of an optic neuropathy and <strong>in</strong> cases where the diagnosis is uncerta<strong>in</strong> or<br />
atypical features are present (class IV, level C).<br />
Newer technologies such as 1H-magnetic resonance spectroscopy of the retrobulbar<br />
tissues have been used to estimate the concentration of chondroit<strong>in</strong> sulfate proteoglycan<br />
<strong>in</strong> retrobulbar tissue. Because the concentration of glycosam<strong>in</strong>oglycans is <strong>in</strong>creased <strong>in</strong><br />
patients with GO, this cl<strong>in</strong>ical tool may thus assist <strong>in</strong> the evaluation of patients with<br />
thyroid orbitopathy (Ohtsuka, 1999a). Octreotide sc<strong>in</strong>tigraphy may also be a useful test<br />
for determ<strong>in</strong><strong>in</strong>g activity of disease by demonstrat<strong>in</strong>g orbital uptake <strong>in</strong> GO (Gerd<strong>in</strong>g,<br />
1999; Krassas, 1999a,b).<br />
What Is the Relationship between Graves’<br />
Disease and Systemic Thyroid Status?<br />
Although GO is often associated with systemic hyperthyroidism, GO may occur <strong>in</strong><br />
primary hypothyroidism, Hashimoto’s thyroiditis, and sometimes <strong>in</strong> euthyroid <strong>in</strong>dividuals<br />
(Gleeson, 1999; Salvi, 1990; Weetman, 2000).<br />
What Is the Treatment for Graves’<br />
Ophthalmopathy?<br />
Treatment of the underly<strong>in</strong>g systemic thyroid abnormalities is the logical first step <strong>in</strong> the<br />
management of thyroid disease. The evidence is controversial regard<strong>in</strong>g the effect of the
Thyroid Eye Disease: Graves’ Ophthalmopathy 351<br />
degree of thyroid abnormality or the speed, type (medical or surgical), or completeness<br />
of systemic therapy (Feldon, 1990) on the <strong>in</strong>cidence or severity of GO (class III–IV,<br />
level C). Nevertheless, we recommend that systemic thyroid control be achieved and<br />
this may improve the signs and symptoms of GO (Prummel, 1990; Tallstedt, 1992) (class<br />
III–IV, level B). Prummel et al studied 90 patients with GO and hyperthyroidism <strong>in</strong><br />
whom the severity of GO and thyroid function were assessed. Patients were assigned to<br />
four groups with <strong>in</strong>creas<strong>in</strong>gly severe GO. More dysthyroid patients were <strong>in</strong> the groups<br />
with severe GO than <strong>in</strong> the other groups (Prummel, 1990). Other uncontrolled studies,<br />
however, failed to show regression of GO after careful treatment of hyperthyroidism<br />
(class III–IV, level C).<br />
The relation between therapy for hyperthyroidism and the course of GO was studied<br />
by Bartalena et al (Bartalena, 1998a). Patients with Graves’ hyperthyroidism and slight<br />
or no GO (443 patients) were randomly assigned to receive radioactive iod<strong>in</strong>e (RAI),<br />
RAI followed by a 3-month course of prednisone, or methimazole for 18 months. The<br />
patients were evaluated at <strong>in</strong>tervals of 1 to 2 months for 12 months. Among the 150<br />
patients treated with RAI, GO developed or worsened <strong>in</strong> 23 (15%) at 2 to 6 months after<br />
treatment. The change was transient <strong>in</strong> 15 patients, but it persisted <strong>in</strong> 8 (5%), who<br />
subsequently required treatment for GO. None of the 55 other patients <strong>in</strong> this group<br />
who had GO at basel<strong>in</strong>e had improvement. Among the 145 patients treated with RAI<br />
and prednisone, 50 (67%) of the 75 with GO at basel<strong>in</strong>e had improvement and no<br />
patient had progression. The effects of RAI on thyroid function were similar <strong>in</strong> these<br />
two groups. Among the 148 patients treated with methimazole, three (2%) who had GO<br />
at basel<strong>in</strong>e improved, four (3%) had worsen<strong>in</strong>g of eye disease, and the rema<strong>in</strong><strong>in</strong>g 141<br />
had no change. The authors concluded that RAI therapy for Graves’ hyperthyroidism is<br />
followed by the appearance or worsen<strong>in</strong>g of GO more than is therapy with methimazole.<br />
Worsen<strong>in</strong>g of GO after RAI therapy is often transient and might be prevented by<br />
the adm<strong>in</strong>istration of low-dose prednisone (Dietle<strong>in</strong>, 1999; Marcocci, 1999a). The<br />
authors concluded there was worsen<strong>in</strong>g of GO <strong>in</strong> 15% of the patients treated with<br />
RAI, but <strong>in</strong> none of those treated with RAI and prednisone. Only 3% of those treated<br />
with methimazole experienced any worsen<strong>in</strong>g of GO (Keltner, 1998).<br />
Bartalena et al studied 26 patients treated with RAI alone and 26 treated with RAI<br />
and systemic prednisone for 4 months (Bartalena, 1989). The <strong>in</strong>itial dose of prednisone<br />
was 0.4 to 0.5 mg=kg of body weight for 1 month, with a gradual taper over 3 months.<br />
Before RAI, 15 patients had no evidence for GO and none of these developed GO after<br />
RAI. Of the patients treated with RAI alone with <strong>in</strong>itial GO, 56% worsened and 44%<br />
were unchanged <strong>in</strong> soft tissue abnormalities and EOM function. Conversely, there was<br />
an improvement <strong>in</strong> GO <strong>in</strong> 52% and no change <strong>in</strong> 48% of RAI patients treated with<br />
steroids. These authors and others (Bartalena, 1989; Rasmussen, 2000) have recommended<br />
systemic corticosteroid treatment to prevent exacerbation of GO <strong>in</strong> patients<br />
undergo<strong>in</strong>g RAI who have some degree of ocular <strong>in</strong>volvement before treatment, but<br />
there is some disagreement with this recommendation (Beck, 1990). We recommend a<br />
short course of oral prednisone dur<strong>in</strong>g RAI therapy for GO (class III–IV, level C).<br />
Some authors believe that patients may experience worsen<strong>in</strong>g of GO after any<br />
systemic thyroid treatments (e.g., thyroid surgery, RAI, and neck radiotherapy for<br />
nonthyroidal neoplasms). The presumed mechanism for worsen<strong>in</strong>g GO is leakage of<br />
thyroid antigens and an <strong>in</strong>crease <strong>in</strong> circulat<strong>in</strong>g thyroid autoantibodies. In contrast to<br />
RAI, Marcocci et al did not f<strong>in</strong>d any effect on GO of near-total thyroidectomy <strong>in</strong> patients<br />
with nonsevere or absent GO (Marcocci, 1999b).
352 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Several studies have shown that smok<strong>in</strong>g is associated with worsen<strong>in</strong>g GO, and we<br />
recommend discont<strong>in</strong>u<strong>in</strong>g tobacco to all of our patients with GO (class III, level B)<br />
(Balazs, 1990; Bartalena, 1998b; Mann, 1999; Nunery, 1993; Pfeilschifter, 1996; Prummel,<br />
1993; Sh<strong>in</strong>e, 1990; Solberg, 1998; Tallstedt, 1993; Tellez, 1992). Insul<strong>in</strong>-dependent<br />
diabetes mellitus is also a risk factor for GO, and optic neuropathy occurs much<br />
more frequently (33.3%) <strong>in</strong> patients with GO and diabetes (and seems to have a worse<br />
prognosis) than <strong>in</strong> a total group of patients with GO (3.9%) (Kalmann, 1999).<br />
The natural history of the GO is variable, and although most GO appears with<strong>in</strong> a few<br />
months of the diagnosis of hyperthyroidism, it may develop many months to years before<br />
or after the onset of the systemic diagnosis of thyroid abnormality. Some patients never<br />
show cl<strong>in</strong>ical or laboratory evidence for systemic thyroid abnormalities (euthyroid GO). In<br />
many patients GO is a self-limited disease that may not require any therapy and the<br />
disease often stabilizes with<strong>in</strong> 1 to 3 years. Therefore, treatment is usually directed at shortterm<br />
control of the <strong>in</strong>flammatory component of the disease (usually with<strong>in</strong> the first 6 to 36<br />
months); acute <strong>in</strong>tervention for vision-threaten<strong>in</strong>g proptosis or CON; and long-term<br />
reconstructive management of lid retraction, strabismus, and proptosis.<br />
Medical and other conservative therapy should generally precede consideration of<br />
surgical <strong>in</strong>tervention. Shorr and Seif described a logical stepwise approach to<br />
the surgical rehabilitation of GO (Shorr, 1986). These authors proposed the follow<strong>in</strong>g<br />
four stages for GO: (1) orbital decompression, (2) strabismus surgery, (3) lid marg<strong>in</strong><br />
reposition<strong>in</strong>g surgery, and (4) blepharoplasty.<br />
The rationale for this sequential approach to GO is that orbital decompression often<br />
results <strong>in</strong> worsen<strong>in</strong>g, new, or changed EOM dysfunction as well as changes <strong>in</strong> lid<br />
position. Therefore, orbital decompression should precede strabismus and lid surgery<br />
<strong>in</strong> patients who require all three surgeries (class III–IV, level C). Patients with CON<br />
should undergo treatment to preserve or improve vision (class III–IV, level B). Trobe<br />
summarized the natural course of untreated CON <strong>in</strong> three series of 32 eyes. In this<br />
report, 21% of these eyes were left with a visual acuity of 20=100 or less, <strong>in</strong>clud<strong>in</strong>g a f<strong>in</strong>al<br />
vision of count<strong>in</strong>g f<strong>in</strong>gers to no light perception <strong>in</strong> five eyes (Trobe, 1978).<br />
What Therapies Are Suggested for Local<br />
Ocular and Orbital Inflammatory Signs?<br />
Patients with lid or ocular irritation, mild <strong>in</strong>flammation, or exposure keratopathy may<br />
benefit from conservative treatments, such as topical artificial tears and=or lubricat<strong>in</strong>g<br />
o<strong>in</strong>tments, t<strong>in</strong>ted or wrap-around glasses, elevation of the head of the bed, or tap<strong>in</strong>g the<br />
eyelids shut dur<strong>in</strong>g sleep (Liu, 1992). Although some authors have advocated the use of<br />
topical, peribulbar, or retrobulbar steroids, we do not usually employ these routes of<br />
steroid therapy for periorbital swell<strong>in</strong>g (class IV, level C). The evidence to support the<br />
use of steroids <strong>in</strong> this manner is anecdotal at best, and these treatments may be<br />
associated with complications, such as secondary <strong>in</strong>creased <strong>in</strong>traocular pressure (class<br />
III–IV, level U).<br />
Should Immunosuppressive Therapy Be<br />
Considered <strong>in</strong> GO?<br />
Although systemic corticosteroids have been employed for GO, there is limited<br />
evidence outl<strong>in</strong><strong>in</strong>g the specific <strong>in</strong>dications for their use or the results of treatment,
Thyroid Eye Disease: Graves’ Ophthalmopathy 353<br />
except <strong>in</strong> patients with CON (Liu, 1992). In addition, although medical therapy usually<br />
consists of prednisone, other immunosuppressive agents (e.g., azathiopr<strong>in</strong>e [Imuran],<br />
methotrexate, cyclophosphamide [Cytoxan], or cyclospor<strong>in</strong>e) have also been<br />
recommended. Experience with these agents is limited (class IV, level U). Perros et al<br />
reported negative results with azathiopr<strong>in</strong>e for moderate GO <strong>in</strong> a matched study of 20<br />
patients (Perros, 1990). Other studies have reported little effect with azathiopr<strong>in</strong>e,<br />
methotrexate (Smith, 2001), or cimexone (Kahaly, 1990). <strong>An</strong>ecdotal success has been<br />
reported with plasmapheresis and bromocript<strong>in</strong>e, but the relative efficacy of these<br />
therapies compared with traditional treatment for GO rema<strong>in</strong>s to be def<strong>in</strong>ed.<br />
Prummel et al reported a s<strong>in</strong>gle-bl<strong>in</strong>d randomized cl<strong>in</strong>ical trial compar<strong>in</strong>g the efficacy<br />
of prednisone (60 mg=day with a taper<strong>in</strong>g dose) versus cyclospor<strong>in</strong>e (7.5 mg=kg of body<br />
weight=day) (Prummel, 1989). Dur<strong>in</strong>g the 12-week treatment period, 11 prednisonetreated<br />
and 4 cyclospor<strong>in</strong>e-treated patients responded to therapy (61% vs. 22%;<br />
p ¼ 0.018) as def<strong>in</strong>ed by decrease <strong>in</strong> EOM enlargement and proptosis, improved<br />
vision, and subjective eye scores. There were no differences at basel<strong>in</strong>e between the<br />
patients who responded later and those who did not, but the prednisone was less well<br />
tolerated than the cyclospor<strong>in</strong>e, and comb<strong>in</strong>ation therapy was better tolerated than<br />
prednisone alone. These authors concluded that s<strong>in</strong>gle-drug therapy with prednisone<br />
was more effective than cyclospor<strong>in</strong>e <strong>in</strong> patients with severe GO, but that the<br />
comb<strong>in</strong>ation may be effective <strong>in</strong> patients who did not respond to either drug alone<br />
(Prummel, 1989).<br />
Corticosteroids have been shown to reduce EOM enlargement <strong>in</strong> CON (Liu, 1992).<br />
Many authors recommend relatively high doses of prednisone (1 to 1.5 mg=kg=day) for<br />
the treatment of CON (Liu, 1992). Kazim et al reviewed 84 cases of acute GO treated<br />
with either high-dose systemic steroids or radiotherapy (RT) and reported that RT (1 of<br />
29 required decompression) was more effective than corticosteroids (6 of 16 patients<br />
required decompression) and that RT had fewer complications than steroids (Kazim,<br />
1991). Guy et al reported the use of high-dose pulse <strong>in</strong>travenous corticosteroids (Guy,<br />
1989). Macchia et al compared oral (prednisone 60 to 80 mg=d) and high-dose <strong>in</strong>travenous<br />
(two weekly <strong>in</strong>jections of 1 g for 6 weeks) corticosteroid therapy for GO <strong>in</strong> 25<br />
patients. High-dose IV steroids were better tolerated but all patients showed significant<br />
improvement <strong>in</strong> proptosis, <strong>in</strong>flammation, and diplopia (Macchia, 2001). Many authors<br />
believe that improvement <strong>in</strong> CON follow<strong>in</strong>g prednisone therapy is usually evident<br />
with<strong>in</strong> the first few weeks of treatment and that there is no justification for ma<strong>in</strong>ta<strong>in</strong><strong>in</strong>g<br />
patients with CON on prolonged corticosteroid regimens (Trokel, 1993).<br />
Claridge et al <strong>in</strong>vestigated comb<strong>in</strong>ed immunosuppression with primary bilateral<br />
orbital radiotherapy (20 Gy <strong>in</strong> 10 fractions). These authors studied RT with azathiopr<strong>in</strong>e<br />
(up to 3 mg=kg=d) and low-dose prednisolone (maximum dose 40 mg=kg=d)<br />
(Claridge, 1997). Forty consecutive patients with active GO were recruited. Before<br />
treatment, 15 had CON, 35 had significant motility restriction, and 38 had marked soft<br />
tissue signs. On average, GO became <strong>in</strong>active after 1.2 years of immunosuppression,<br />
and treatment was well tolerated. Compared with previously reported treatments, the<br />
authors thought that this therapy regimen was more effective than either treatment<br />
alone and led to fewer side effects than high-dose steroids. In particular, there was<br />
more than a fourfold reduction <strong>in</strong> the requirement for orbital decompression and<br />
strabismus surgery.<br />
Baschieri et al performed a prospective nonrandomized study of <strong>in</strong>travenous<br />
immunoglobul<strong>in</strong> (IVIG) versus corticosteroids for GO (Baschieri, 1997). Twenty-seven
354 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
patients treated with IVIG were followed for an average of 21 months (12 to 48 months).<br />
Soft tissue <strong>in</strong>volvement improved or disappeared <strong>in</strong> 32 of 35 patients (91%) treated with<br />
IVIG and 25 of 27 (93%) of patients treated with steroids. Diplopia improved or<br />
disappeared <strong>in</strong> 22 of 29 (76%) patients treated with IVIG and 16 of 20 (80%) of patients<br />
treated with steroids. A significant reduction of EOM thickness on CT imag<strong>in</strong>g<br />
was observed after treatment <strong>in</strong> both groups. Proptosis improved or disappeared <strong>in</strong><br />
20 of 31 (65%) of patients treated with IVIG and <strong>in</strong> 15 of 24 (63%) of patients treated<br />
with steroids. The authors suggest that IVIG is safe and effective <strong>in</strong> reduc<strong>in</strong>g the<br />
eye changes <strong>in</strong> patients with GO. Prophet et al used immunoadsorption therapy (20<br />
sessions of Plasmaselect=Therasorp <strong>An</strong>ti-IgG) <strong>in</strong> two patients with refractory GO<br />
(Prophet, 2001).<br />
Balazs et al performed a pilot study of pentoxifyll<strong>in</strong>e on moderately severe GO <strong>in</strong> 10<br />
patients (Balazs, 1997). At 12 weeks, 80% of patients had improvement of soft tissue<br />
<strong>in</strong>volvement, but not of proptosis or EOM <strong>in</strong>volvement. This agent may have a future<br />
role <strong>in</strong> the treatment of GO.<br />
Krassas and Heufelder reviewed the immunosuppressive treatment available for GO,<br />
and concluded that cyclospor<strong>in</strong> A, azathiopr<strong>in</strong>e, cyclophosphamide, and cimexone had<br />
modest results but with unfavorable risk-benefit ratios (Krassas, 2001). Somatostat<strong>in</strong><br />
analogs, octreotide, and lanreotide are emerg<strong>in</strong>g therapies but rema<strong>in</strong> costly and as yet<br />
unproven alternatives (Krassas, 2001). Uysal et al reported a positive response to<br />
octreotide <strong>in</strong> seven of n<strong>in</strong>e patients with GO (1999).<br />
We recommend immunosuppression therapy only for GO <strong>in</strong> the active phase. Usually<br />
a short course (2- to 4-week trial) of oral corticosteroids (prednisone 1 mg=kg per day)<br />
would be a reasonable first-l<strong>in</strong>e therapy (class IV, level C). We generally do not<br />
recommend long-term corticosteroid therapy <strong>in</strong> GO because of the systemic side effects.<br />
Concomitant corticosteroid treatment, however, may be useful as an adjunct to radiotherapy<br />
(see below). The evidence for us<strong>in</strong>g other immunosuppressive agents <strong>in</strong> GO is<br />
not sufficient to support a def<strong>in</strong>itive recommendation on the <strong>in</strong>dications, drug, dosage,<br />
or duration of therapy (class IV, level U).<br />
What Is the Role of Radiotherapy <strong>in</strong> GO?<br />
Low-dose orbital RT has been reported with good results for the treatment of acute<br />
<strong>in</strong>flammation (soft tissue signs) and=or CON due to GO. The typical cumulative RT<br />
dose of 2000 cGy is well tolerated and generally considered to be safe (class III, level B).<br />
RT may also be useful <strong>in</strong> reduc<strong>in</strong>g the dosage or elim<strong>in</strong>at<strong>in</strong>g the need for corticosteroid<br />
or other immunosuppressive treatments (Kazim, 1991; Petersen, 1990; Rush, 2000).<br />
Table 16–2 summarizes selected cases of orbital RT <strong>in</strong> the treatment of GO.<br />
Marcocci et al compared the efficacy and tolerance of <strong>in</strong>travenous or oral glucocorticoids<br />
and orbital RT <strong>in</strong> a prospective, s<strong>in</strong>gle-bl<strong>in</strong>d, randomized study of 82 patients with<br />
severe GO (Marcocci, 2001). There was a significant reduction <strong>in</strong> proptosis, diplopia,<br />
and CON <strong>in</strong> both groups. IV steroids were tolerated better than oral steroids. The<br />
cl<strong>in</strong>ical activity score was significantly lower <strong>in</strong> the IV group than the oral group. These<br />
authors concluded that high-dose (15 mg=kg for four cycles, then 7.5 mg=kg for four<br />
cycles, each cycle consisted of two <strong>in</strong>fusions on alternate days at 2-week <strong>in</strong>tervals) IV<br />
steroids and oral steroids (prednisone 100 mg=d, withdrawal after 5 months) associated<br />
with orbital RT were effective <strong>in</strong> severe GO. IV steroids, however, were more effective,
Thyroid Eye Disease: Graves’ Ophthalmopathy 355<br />
Table 16–2. Treatment of GO with Orbital RT: Summary of Selected Studies<br />
Author No. of Patients Response Rate (%) Comments<br />
Donaldson, 1973 80 67% >1 year follow-up<br />
Rav<strong>in</strong>, 1975 9 with optic All 9 improved vision Little effect on soft<br />
neuropathy<br />
tissue abnormalities<br />
Brennan, 1983 14 13=14 (93%) reduced Myopathy showed<br />
soft tissue <strong>in</strong>flammation the least improvement<br />
Hurbli, 1985 62 34=46 (74%) improved Patients with<br />
motility and<br />
duration < 6 months<br />
10=14 (71%)<br />
CON improved<br />
responded better<br />
Wiers<strong>in</strong>ga, 1988 39 25 (64%) improved<br />
Sandler, 1989 35 71% improved<br />
Lloyd, 1992 36 33=36 (92%) improved<br />
Kazim, 1991 29 28=29 (97%) improved 1 required decompression<br />
Mourits, 2000 30 60% improved Placebo controlled<br />
Placebo (31%) improved Improved diplopia<br />
Rush, 2000 10 CON 8=10 (80%) improved Improved vision<br />
Tsuj<strong>in</strong>o, 2000 121 Limited proptosis<br />
Excellent (14%),<br />
response<br />
good (54%), fair (25%)<br />
Van Ruyven, 2000 111 No change <strong>in</strong> proptosis Improved motility;<br />
improved soft<br />
tissue signs<br />
Gorman, 2001 42 No beneficial response Randomized trial<br />
had fewer side effects, and were better tolerated than oral steroids (Marcocci, 2001).<br />
Tsuj<strong>in</strong>o et al reported 121 patients with GO treated with orbital RT (20 Gy <strong>in</strong> 10<br />
fractions). The cl<strong>in</strong>ical response was excellent <strong>in</strong> 14%, good <strong>in</strong> 54%, fair <strong>in</strong> 25%, no<br />
response <strong>in</strong> 6%, and worse <strong>in</strong> 1% (Tsuj<strong>in</strong>o, 2000). Kahaly et al reported improvement<br />
us<strong>in</strong>g three RT dos<strong>in</strong>g protocols: group A (1 Gy weekly for 20 weeks), group B (10<br />
fractions of 1 Gy), and group C (2 Gy daily over 2 weeks). Improvement was noted <strong>in</strong> 12<br />
patients <strong>in</strong> group A (67%), 13 <strong>in</strong> group B (59%), and 12 <strong>in</strong> group C (55%). They<br />
concluded that the response rates were similar <strong>in</strong> low and high RT dose treatment but<br />
that the 1 Gy per week protocol was most effective and better tolerated (Kahaly, 2000).<br />
Most authors do not believe that RT is <strong>in</strong>dicated for patients with mild GO or for<br />
long-stand<strong>in</strong>g, fibrotic, non<strong>in</strong>flammatory GO (class IV, level C). Previous head or orbit<br />
RT is probably a contra<strong>in</strong>dication to further RT for GO. No significant morbidity has<br />
been reported <strong>in</strong> patients with appropriate RT dos<strong>in</strong>g for GO, although there is a<br />
theoretical risk of RT-<strong>in</strong>duced cataracts or neoplasms, radiation optic neuropathy, or<br />
radiation ret<strong>in</strong>opathy (class III–IV, level C). Several cases of radiation ret<strong>in</strong>opathy have<br />
been described <strong>in</strong> patients with <strong>in</strong>appropriate RT dos<strong>in</strong>g (K<strong>in</strong>youn, 1984). We recommend<br />
that only centers with considerable experience with RT perform this treatment for<br />
GO (Char, 1991).<br />
Gorman et al performed a prospective, randomized, double-bl<strong>in</strong>d, placebo-controlled<br />
study of orbital radiotherapy for GO (Gorman, 2001). The patients had symptomatic GO<br />
without optic neuropathy. Forty-two of 53 eligible consecutive patients were treated
356 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
(20 Gy of external beam therapy to one orbit with sham therapy to other side, followed <strong>in</strong><br />
6 months with reversal of the therapies). Every 3 months for 1 year, the authors measured<br />
the volume of the EOM and of fat, proptosis, range of EOM motion, area of diplopia fields,<br />
and lid fissure width. No cl<strong>in</strong>ical statistically significant difference between the treated<br />
and untreated orbit was observed <strong>in</strong> any of the outcome measures at 6 months. At 12<br />
months, muscle volume and proptosis improved slightly more <strong>in</strong> the orbit that was treated<br />
first. The authors concluded that <strong>in</strong> this group of patients they were unable to demonstrate<br />
any beneficial therapeutic effects (Gorman, 2001). The usefulness of this study has been<br />
criticized, however, because of its broad patient <strong>in</strong>clusion criteria that lacked rigor <strong>in</strong><br />
controll<strong>in</strong>g the issues of tim<strong>in</strong>g of therapy, the cl<strong>in</strong>ical variability <strong>in</strong> presentation of<br />
the patients, and multiple treatment methods used for <strong>in</strong>dividual patients (Feldon,<br />
2001). We recommend that low-dose orbital RT still be considered a valid treatment<br />
option for the treatment of active <strong>in</strong>flammatory GO (class III–IV, level C). Unfortunately,<br />
the assessment of which patients have active GO rema<strong>in</strong>s difficult and controversial<br />
despite multiple proposed grad<strong>in</strong>g schemes (Cockerham, 2002; Dick<strong>in</strong>son, 2001).<br />
What Is the Treatment for Lid Retraction <strong>in</strong><br />
GO?<br />
Lid retraction may be due to Müller’s muscle (sympathetic) or levator overaction, levator<br />
contraction (degeneration of the muscle or aponeurosis), levator adhesions, or pseudoretraction<br />
(Lemke, 1991). Occasionally eyelid retraction spontaneously resolves (von<br />
Brauchitsch, 1999). Upper eyelid retraction may be treated surgically by a number of<br />
approaches (Maarten, 1999; Oliver, 1998b), <strong>in</strong>clud<strong>in</strong>g levator marg<strong>in</strong>al myotomies,<br />
Müller’s muscle excision, levator stripp<strong>in</strong>g, and levator spacers placed <strong>in</strong>to the upper<br />
eyelid to create length. Detailed descriptions of these procedures are beyond the scope of<br />
this text. Patients with lesser degrees of proptosis may benefit from eyelid procedures<br />
more than orbital procedures. Char recommended that patients with exophthalmometry<br />
read<strong>in</strong>gs of under 23 or 24 with good motility are probably better treated with eyelid<br />
procedures (Char, 1991, 1992). The use of topical a-adrenergic agents, such as guanethid<strong>in</strong>e,<br />
has been advocated by some authors, but significant corneal toxicity usually limits<br />
the use of these agents. Botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jections <strong>in</strong>to the lids may also transiently<br />
relieve lid retraction (Ceisler, 1995; Oliver, 1998a; Ozkan, 1997; Traisk, 2001). We generally<br />
recommend lid surgery be considered for patients with <strong>in</strong>active and stable disease who<br />
do not have evidence for optic neuropathy and who are not go<strong>in</strong>g to undergo orbital<br />
decompression or strabismus surgery <strong>in</strong> the near future (class IV, level C).<br />
What Treatments Should Be Considered for<br />
Strabismus Due to GO?<br />
Strabismus <strong>in</strong> GO may be treated with patch<strong>in</strong>g, prism therapy, or strabismus surgery<br />
(Prendiville, 2000). Patients with difficulty <strong>in</strong> downgaze and the read<strong>in</strong>g position may<br />
benefit from simply occlud<strong>in</strong>g the lower segment of their bifocal, rais<strong>in</strong>g the bifocal<br />
height, or us<strong>in</strong>g two pairs of spectacles (one pair of s<strong>in</strong>gle-vision glasses for read<strong>in</strong>g and<br />
one pair for distance). The surgical techniques to correct strabismus will vary depend<strong>in</strong>g
on the severity and distribution of extraocular <strong>in</strong>volvement. A detailed description of<br />
these procedures is beyond the scope of this text (Mourits, 1990a,b). Limited anecdotal<br />
success has been reported with botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jections <strong>in</strong>to EOMs, but we do not<br />
generally recommend botul<strong>in</strong>um tox<strong>in</strong> treatment for GO (class IV, level C). Surgical<br />
treatment for strabismus should be deferred until after the acute <strong>in</strong>flammatory phase of<br />
GO has been treated adequately (class III–IV, level C). Although successful long-term<br />
alignment may be achieved with strabismus surgery dur<strong>in</strong>g the active phase of GO <strong>in</strong><br />
selected patients with marked disability (Coats, 1999), we generally advocate stable<br />
measurements, good thyroid control, and <strong>in</strong>active disease before proceed<strong>in</strong>g with<br />
strabismus surgery (class IV, level C).<br />
What Treatments Are Suggested for Proptosis<br />
and=or Compressive Optic <strong>Neuro</strong>pathy Due to<br />
GO?<br />
Thyroid Eye Disease: Graves’ Ophthalmopathy 357<br />
The natural history of CON is poorly documented but presumably variable (class IV).<br />
Carter et al reviewed the comb<strong>in</strong>ed reports of 16 untreated patients (26 eyes). There was<br />
spontaneous visual improvement to 20=50 acuity or better <strong>in</strong> 19 eyes (73%), but 6 eyes<br />
(23%) did not improve (range, count<strong>in</strong>g f<strong>in</strong>gers to no light perception) (Carter, 1991).<br />
CON may be treated with systemic corticosteroids, orbital RT, or orbital surgical<br />
decompression (Kazim, 1991; Kubis, 2000; Trobe, 1978). Kazim et al retrospectively<br />
reviewed 84 cases of acute GO and reported that only 1 of 29 patients with CON treated<br />
with RT required surgical decompression versus 6 of 16 treated with corticosteroids<br />
(Kazim, 1991). Nevertheless, oral or IV corticosteroids may be the first-l<strong>in</strong>e treatment for<br />
CON (Guy, 1989). Guy et al reported the use of pulse IV methylprednisolone (1 g daily<br />
for 3 days) <strong>in</strong> five patients with CON. Oral corticosteroids and orbital RT allowed the<br />
treatment response to be ma<strong>in</strong>ta<strong>in</strong>ed <strong>in</strong> all five patients for several months (Guy, 1989).<br />
Patients with CON who fail or cannot tolerate steroid treatment and=or RT should be<br />
considered for orbital decompression.<br />
A wide variety of surgical approaches for orbital decompression have been advocated,<br />
<strong>in</strong>clud<strong>in</strong>g one, two, three, and even four wall decompressions (<strong>An</strong>tozyck, 1992;<br />
Carter, 1991; Coday, 1998; Fatourechi, 1993; Garrity, 1993; Goldberg, 1997; Gormley,<br />
1997; Graham, 1997; Hurwitz, 1992; Kalmann, 1997; Kennedy, 1990; Kulw<strong>in</strong>, 1990;<br />
Leatherbarrow, 1991; Lund, 1997; May, 2000; McNab, 1997; Mourits, 1990b; Ohtsuka,<br />
1999b; Olivari, 1991; Paridaens, 1998, 2000; Ruttum, 2000; Sillers, 1997; Thaller, 1990;<br />
West, 1997; Wilson, 1991). Wulc et al also advocated lateral wall advancement as an<br />
adjunct to orbital decompression to enhance the decompressive effect and provide a<br />
potential space for lateral expansion (Wulc, 1990). Golken et al described microsurgical<br />
liposuction <strong>in</strong> GO via a lateral canthotomy (Golken, 2000). Kazim et al performed<br />
orbital fat decompression <strong>in</strong>stead of orbital bone decompression on five patients (eight<br />
eyes) with dysthyroid optic neuropathy who had an enlarged orbital fat compartment<br />
(Kazim, 2000). These patients did not have EOM enlargement as the solitary cause of the<br />
optic neuropathy. The optic neuropathy was reversed <strong>in</strong> all of the patients, and there<br />
was no postoperative diplopia, enophthalmos, globe ptosis, or sensory loss. Table 16–3<br />
summarizes several studies concern<strong>in</strong>g surgical orbital decompression <strong>in</strong> the treatment<br />
of proptosis and=or CON <strong>in</strong> GO. Although there is no class I evidence, there is
358 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 16–3. Orbital Decompression (Results <strong>in</strong> Selected Series)<br />
Author=Year<br />
Number of Eyes<br />
or Patients Surgical Approach Comment<br />
Algvere, 1973 22 eyes Pterional 91% improved<br />
Trobe, 1978 9 eyes (6 patients) 4 temporal<br />
decompression,<br />
3 temporal and<br />
orbital floor,<br />
2 transfrontal<br />
66% improved,<br />
3=4 steroid failures,<br />
3=5 primary surgery<br />
L<strong>in</strong>berg, 1981 11 eyes Transorbital 82% improved<br />
Leone, 1981 14 eyes Inferior orbital 100% improved<br />
McCord, 1981 11 patients <strong>An</strong>tral ethmoidal 55% improved with<br />
surgery alone,<br />
45% improved after<br />
radiotherapy (RT)<br />
Hurwitz, 1985 27 eyes Inferomedial approach 81% improved<br />
Lamberg, 1985 27 bilateral and<br />
3 unilateral<br />
Transantral approach 93% improved<br />
Shorr, 1986 28 patients Transantral approach 100% improved<br />
Hall<strong>in</strong>, 1988 25 patients (48 eyes) Transantral approach 77% improved<br />
visual acuity<br />
Hart<strong>in</strong>g, 1989 28 patients Variable techniques 50% improved<br />
Leone, 1989 2 patients Medial and lateral wall 100% improved<br />
Warren, 1989 305 patients Transantral 95% improved<br />
Kennedy, 1990 4 patients Endoscopic transnasal 75% improved<br />
Mourits, 1990a,b 25 patients 13 <strong>in</strong>feromedial,<br />
5 <strong>in</strong>feromedial<br />
þ lateral, 7 coronal<br />
76% improved<br />
Carter, 1991 30 (52 orbits) Transantral-ethmoidal 92% improved<br />
Leatherbarrow, 1991 4 patients 3 wall coronal 50% improved<br />
Olivari, 1991 10 patients Intraorbital fat removal 60% improved<br />
Hurwitz, 1992 25 patients (46 orbits) Ethmoidectomy<br />
(medial wall)<br />
86% improved<br />
<strong>An</strong>tozyck, 1992 5 orbits Transorbital 3 wall 80% improved<br />
Garrity, 1993 217 patients Transantral 89% improved<br />
visual acuity;<br />
91% improved<br />
visual field<br />
Neugenbauer, 1996 21 Endoscopic-endonasal 95% improved vision<br />
West, 1997 22 4 wall coronal<br />
Goldberg, 1997 20 3 wall coronal<br />
Kalmann, 1997 125 3 wall coronal<br />
May, 1999 17 patients (27 orbits) Microsurgical endonasal Proptosis reduction<br />
4.1 mm<br />
Ohtsuka, 1999b 4 patients Transmedial-canthal<br />
ethmoidal (one-wall)<br />
All improved<br />
Ulualp, 1999 28 orbits Transnasal endoscopic Vision improved<br />
9=15 (60%)<br />
Eloy, 2000 16 patients, 27 orbits Endoscopic endonasal Proptosis reduction<br />
3.17 mm<br />
(cont<strong>in</strong>ued)
Table 16–3. (cont<strong>in</strong>ued)<br />
May, 2000 19 patients, 29 orbits Endonasal<br />
Proptosis reduction<br />
microsurgery<br />
4.2 mm<br />
Paridaens, 2000 19 patients (35 orbits), 1, 2, or 3 wall by 6 CON improved<br />
6 with CON<br />
lateral canthotomy<br />
and lower<br />
fornix <strong>in</strong>cision<br />
Tallstedt 2000 63 patients Transantral Proptosis reduction<br />
3.2 mm, 20=21 (95%)<br />
vision improved<br />
Michel, 2001 78 patients (145 eyes) Transnasal orbital 4 cases required<br />
repeat surgery<br />
L<strong>in</strong>net 2001 50 eyes Transcranial 2 wall 87% improved vision<br />
consensus class III evidence that orbital decompression is an effective treatment for<br />
CON <strong>in</strong> GO (class III–IV, level B).<br />
What Is the Treatment for Proptosis without<br />
Optic <strong>Neuro</strong>pathy <strong>in</strong> GO?<br />
Thyroid Eye Disease: Graves’ Ophthalmopathy 359<br />
McCord <strong>in</strong> 1985 reported that more than 60% of orbital decompressions were performed<br />
for mild to severe exophthalmos to correct corneal exposure or disfigurement, based on a<br />
survey of members of the American Society of Ophthalmic Plastic and Reconstructive<br />
Surgeons and the Orbital Society. The majority of decompressions were via an antralethmoidal<br />
decompression and translid or fornix approach (McCord, 1985). Kennerdell et<br />
al suggested that patients with proptosis of less than 25 to 26 mm, especially if<br />
accompanied by lid retraction but without CON, could be treated with lid lengthen<strong>in</strong>g<br />
procedures alone to disguise the proptosis (Kennerdell, 1987). Lyons and Rootman<br />
reported orbital decompression on 65 orbits (34 patients) for disfigur<strong>in</strong>g exophthalmos<br />
and achieved a mean retroplacement of 4 mm (range 21.0 to 10 mm). Diplopia, however,<br />
arose de novo <strong>in</strong> five (18%) previously asymptomatic patients <strong>in</strong> this series (Lyons, 1994).<br />
Fatourechi et al described 34 patients with GO who underwent transantral orbital<br />
decompression primarily for cosmetic reasons (Fatourechi, 1994). There was a mean<br />
reduction <strong>in</strong> proptosis of 5.2 mm. Persistent diplopia developed <strong>in</strong> 73% of 15 patients<br />
who were asymptomatic preoperatively. Other reports have confirmed that although<br />
orbital decompression for primarily cosmetic reasons is successful, ocular motility<br />
deficits and diplopia may occur postdecompression <strong>in</strong> a significant number of patients<br />
(Carter, 1991; Fatourechi, 1994; McNab, 1997). For example, <strong>in</strong> a study of 81 patients with<br />
GO who underwent orbital decompression, 8 of 41 coronal patients (20%) and 4 of 29<br />
translid patients (14%) experienced aggravation of their motility impairment (Paridaens,<br />
1998). Goldberg et al reported that lateral wall decompression produced less new-onset,<br />
persistent postoperative strabismus than balanced medial and lateral wall decompression<br />
for GO (Goldberg, 2000). Seiff et al reported modified orbital decompression with<br />
preservation of the anterior periorbita reduced the risk of postoperative diplopia (Seiff,
360 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 16–1. Evaluation and treatment of thyroid eye disease.
2000). Bersani and Jian-Seyed-Ahmadi reported surgical placement of a lateral orbital<br />
rim implant as an effective method of orbital volume expansion <strong>in</strong> GO (1999).<br />
In addition to diplopia, orbital decompression may result <strong>in</strong> loss of vision, globe or<br />
eyelid damage, worsen<strong>in</strong>g lid retraction, cerebrosp<strong>in</strong>al fluid leakage, <strong>in</strong>fraorbital<br />
anesthesia, or s<strong>in</strong>us complications. Therefore, patients should be counseled appropriately<br />
preoperatively regard<strong>in</strong>g these possible complications.<br />
Trokel et al performed orbital fat removal on 81 patients (158 decompressions)<br />
(Trokel, 1993). These authors reported an average reduction of proptosis of 1.8 mm<br />
(range 0 to 6.0 mm). The greatest reduction <strong>in</strong> proptosis (3.3 mm) was produced <strong>in</strong><br />
patients with more than 25 mm of proptosis (Hertel measurements). Trokel et al reserve<br />
decompression with bone removal for patients with CON who are unresponsive to<br />
medical therapy or patients with persistent deform<strong>in</strong>g exophthalmos follow<strong>in</strong>g primary<br />
orbital fat removal. We recommend orbital decompression for proptosis <strong>in</strong> patients who<br />
have vision-threaten<strong>in</strong>g exposure keratopathy or other significant symptoms (e.g., pa<strong>in</strong>,<br />
pressure, severe exophthalmos) related to the proptosis (class III–IV, level C).<br />
<strong>An</strong> approach to the patient with thyroid eye disease is outl<strong>in</strong>ed <strong>in</strong> Figure 16–1.<br />
References<br />
Thyroid Eye Disease: Graves’ Ophthalmopathy 361<br />
Algvere P, Almqvist S, Backlund EO. (1973). Pterional orbital decompression <strong>in</strong> progressive ophthalmopathy of<br />
Graves’ disease II: a follow-up study. Acta Ophthalmol (Copenh) 51:475–482.<br />
<strong>An</strong>tozyck JH, Tucker N, Codere F. (1992). Orbital decompression for Graves’ disease: exposure through a<br />
modified blepharoplasty <strong>in</strong>cision. Ophthalmic Surg 23:516–521.<br />
Bahn RS, Garrity JA, Gorman CA. (1990). Diagnosis and management of Graves’ ophthalmopathy. J Cl<strong>in</strong><br />
Endocr<strong>in</strong>ol Metab 71:559–563.<br />
Bahn RS, Heufelder AE. (1993). Pathogenesis of Graves’ ophthalmopathy. N Engl J Med 329:1468–1475.<br />
Balazs C, Kiss E, Vamos A, et al. (1997). Beneficial effect of pentoxifyll<strong>in</strong>e on thyroid associated ophthalmopathy. J<br />
Cl<strong>in</strong> Endocr<strong>in</strong>ol Metab 82:1999–2002.<br />
Balazs C, Stenszky V, Farid NR. (1990). Association between Graves’ ophthalmopathy and smok<strong>in</strong>g. Lancet<br />
336:754.<br />
Bartalena L, Marcocci C, Bogazzi F, et al. (1989). Use of corticosteroids to prevent progression of Graves’<br />
ophthalmopathy after radioiod<strong>in</strong>e therapy for hyperthyroidism. N Engl J Med 321:1349–1352.<br />
Bartalena L, Marcocci C, Bogazzi F, et al. (1990). Use of corticosteroids to prevent progression of Graves’<br />
ophthalmopathy. Arch Intern Med 150:1098–1101.<br />
Bartalena L, Marcocci C, Bogazzi F, et al. (1998a). Relation between therapy for hyperthyroidism and the course of<br />
Graves’ ophthalmopathy. N Engl J Med 338:73–78.<br />
Bartalena L, Marcocci C, Tanda ML, et al. (1998b). Cigarette smok<strong>in</strong>g and treatment outcomes <strong>in</strong> Graves<br />
ophthalmopathy. <strong>An</strong>n Intern Med 129:632–635.<br />
Bartalena L, P<strong>in</strong>chera A, Marcocci C. (2000). Management of Graves’ ophthalmopathy. Reality and perspectives.<br />
Endocr Rev 21:168–199.<br />
Bartley GB. (1994). The epidemiologic characteristics and cl<strong>in</strong>ical course of ophthalmopathy associated with<br />
autoimmune thyroid disease <strong>in</strong> Olmstead County, M<strong>in</strong>nesota. Trans Am Ophthalmol Soc 92:477–588.<br />
Bartley GB. (1995). Evolution and classification systems for Graves’ ophthalmopathy. Ophthalmic Plast Reconstr<br />
Surg 11:229–237.<br />
Bartley GB. (1996). The differential diagnosis and classification of eyelid retraction. Ophthalmology 103:168–176.<br />
Bartley GB, Fatourechi V, Kadrmas EF, et al. (1995). The <strong>in</strong>cidence of Graves’ ophthalmopathy <strong>in</strong> Olmsted County,<br />
M<strong>in</strong>nesota. Am J Ophthalmol 120:511–517.<br />
Baschieri L, <strong>An</strong>tonelli A, Nardi S, et al. (1997). Intravenous immunoglobul<strong>in</strong> versus corticosteroid <strong>in</strong> treatment of<br />
Graves’ ophthalmopathy. Thyroid 7:579–585.<br />
Beck RW, DiLoreto DA. (1990). Treatment of Graves’ ophthalmopathy. N Engl J Med 322:1088–1089.<br />
Bersani T, Jian-Seyed-Ahmadi A. (1999). Orbital volume expansion of dysthyroid ophthalmopathy by surgical<br />
placement of lateral rim implants: a case study. Ophthalmic Plast Reconstr Surg 15:429–431.
362 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Brennan MW, Leone CR Jr, Janaki L. (1983). Radiation therapy for Graves’ disease. Am J Ophthalmol 96:195–199.<br />
Carter KD, Frueh BR, Hessburg TP, Musch DC. (1991). Long-term efficacy of orbital decompression for<br />
compressive optic neuropathy of Graves’ eye disease. Ophthalmology 98:1435–1442.<br />
Ceisler EJ, Bilyk JA, Rub<strong>in</strong> PA, et al. (1995). Results of Mullerectomy and levator aponeurosis transposition for the<br />
correction of upper eyelid retraction <strong>in</strong> Graves’ disease. Ophthalmology 102:483–492.<br />
Chang TC, Huang KM, Chang TJ, L<strong>in</strong> SL. (1990). Correlation of orbital computed tomography and antibodies <strong>in</strong><br />
patients with hyperthyroid Graves’ disease. Cl<strong>in</strong> Endocr<strong>in</strong>ol 32:551–558.<br />
Char DH. (1991). The ophthalmopathy of Graves’ disease. Med Cl<strong>in</strong> North Am 75:97–119.<br />
Char DH. (1992). Advances <strong>in</strong> thyroid orbitopathy. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:25–39.<br />
Claridge KG, Ghabrial R, Davis G, et al. (1997). Comb<strong>in</strong>ed radiotherapy and medical immunosuppression <strong>in</strong> the<br />
management of thyroid eye disease. Eye 11:717–722.<br />
Coats DK, Paysse EA, Plager DA, Wallace DK. (1999). Early strabismus surgery for thyroid ophthalmopathy.<br />
Ophthalmology 106:324–329.<br />
Cockerham K, Kennerdell JS, Mourits MP, McNab AA. (2002). Does radiotherapy have a role <strong>in</strong> the management<br />
of thyroid orbitopathy? Br J Ophthalmol 86:102–107.<br />
Coday MP, Dallow RL.(1998). Manag<strong>in</strong>g Graves’ orbitopathy. Int Ophthalmol Cl<strong>in</strong> 38:103–115.<br />
Danesh-Meyer HV, Sav<strong>in</strong>o PJ, Deramo V, et al. (2001). Intraocular pressure changes after treatment for Graves’<br />
orbitopathy. Ophthalmology 108:145–150.<br />
Danks JJ, Harrad RA. (1998). Flash<strong>in</strong>g lights <strong>in</strong> thyroid eye disease: a new symptom described and possibly<br />
expla<strong>in</strong>ed. Br J Ophthalmol 82:1309–1311.<br />
Dick<strong>in</strong>son AJ, Perros P. (2001). Controversies <strong>in</strong> the cl<strong>in</strong>ical evaluation of thyroid-associated orbitopathy: use of a<br />
detailed protocol with comparative photographs for objective assessment. Cl<strong>in</strong> Endocr<strong>in</strong>ol 55:283–303.<br />
Dietle<strong>in</strong> M, Dederichs B, Weigand A, Schicha H. (1999). Radioiod<strong>in</strong>e therapy and thyroid-associated<br />
orbitopathy: risk factors and preventive effects of glucocorticoids. Exp Cl<strong>in</strong> Endocr Diabetes 107 (suppl<br />
5):S190–194.<br />
Donaldson SS, Bagshaw MA, Kriss JP. (1973). Supervoltage orbital radiotherapy for Graves’ ophthalmopathy.<br />
J Cl<strong>in</strong> Endocr<strong>in</strong>ol Metab 37:276–285.<br />
Eloy P, Trussart C, Jouzdani E, et al. (2000). Transnasal endoscopic orbital decompression and Graves’<br />
ophthalmopathy. Acta Otorh<strong>in</strong>olaryngol Belg 54:165–174.<br />
Fatourechi V, Garrity JA, Bartley GB, Bergstrahl EJ, DeSanto LW, Gorman CA. (1994). Graves’ ophthalmopathy:<br />
results of transantral orbital decompression performed primarily for cosmetic <strong>in</strong>dications. Ophthalmology<br />
101:1341–1346.<br />
Fatourechi V, Garrity JA, Bartley GB, Bergstrahl EJ, Gorman CA. (1993). Orbital decompression <strong>in</strong> Graves’<br />
ophthalmopathy associated with pretibial myxedema. J Endocr<strong>in</strong>ol Invest 16:433–437.<br />
Feldon SE. (1990). Graves’ ophthalmopathy: is it really thyroid disease? Arch Intern Med 150:948–950.<br />
Feldon S. (1993). Classification of Graves’ ophthalmopathy [letter]. Thyroid 3:171.<br />
Feldon SE. (2001). Radiation therapy for Graves’ ophthalmopathy: trick or treat? Ophthalmology 108:<br />
1521–1522.<br />
Firbank MJ, Coulthard A. (2000). Evaluation of a technique for estimation of extraocular muscle volume us<strong>in</strong>g 2D<br />
MRI. Br J Radiol 73:1282–1289.<br />
Frueh BR. (1992). Why the NOSPECS classification of Graves’ eye disease should be abandoned, with suggestions<br />
for the characterization of this disease. Thyroid 2:85–88.<br />
Garrity JA, Fatourechi V, Bergstralh EJ, et al. (1993). Results of transantral orbital decompression <strong>in</strong> 428 patients<br />
with severe Graves’ ophthalmopathy. Am J Ophthalmol 116:533–547.<br />
Gerd<strong>in</strong>g MN, van der Zant FM, van Royen EA, et al. (1999). Octreotide-sc<strong>in</strong>tigraphy is a disease-activity<br />
parameter <strong>in</strong> Graves’ ophthalmopathy. Cl<strong>in</strong> Endocr<strong>in</strong>ol 50:373–379.<br />
Gleeson H, Kelly W, Toft A, et al. (1999). Severe thyroid eye disease associated with primary hypothyroidism and<br />
thyroid-associated dermopathy. Thyroid 9:1115–1118.<br />
Goldberg RA, Perry JD, Hortaleza V, Tong JT. (2000). Strabismus after balanced medial plus lateral wall versus<br />
lateral wall only decompression for dysthyroid orbitopathy. Ophthalmic Plast Reconstr Surg 16:271–277.<br />
Goldberg RA, We<strong>in</strong>berg DA, Shorr N, Wirta D. (1997). Maximal, three-wall, orbital decompression through a<br />
coronal approach. Ophthalmic Surg Lasers 28:832–843.<br />
Golken R, W<strong>in</strong>ter R, Sistani F, et al. (2000). M<strong>in</strong>imal <strong>in</strong>vasive decompression of the orbit <strong>in</strong> Graves’ orbitopathy.<br />
Strabismus 8:251–259.<br />
Gorman CA. (1991). Clever is not enough: NOSPECS is form <strong>in</strong> search of function. Thyroid 1:353–355.<br />
Gorman CA, Garrity JA, Fatourechi V, et al. (2001). A prospective, randomized, double-bl<strong>in</strong>d, placebo-controlled<br />
study of orbital radiotherapy for Graves’ ophthalmopathy. Ophthalmology 108:1523–1534.
Thyroid Eye Disease: Graves’ Ophthalmopathy 363<br />
Gormley PD, Bowyer J, Jones NS, Downes RN. (1997). The sphenoidal s<strong>in</strong>us <strong>in</strong> optic nerve decompression. Eye<br />
11:723–726.<br />
Graham SM, Carter KD. (1997). Comb<strong>in</strong>ed endoscopic and subciliary orbital decompression for thyroid-related<br />
compressive optic neuropathy. Rh<strong>in</strong>ology 35:103–107.<br />
Guy JR, Fagien S, Donovan JP, Rub<strong>in</strong> ML. (1989). Methylprednisolone pulse therapy <strong>in</strong> severe dysthyroid optic<br />
neuropathy. Ophthalmology 96:1048–1053.<br />
Hart<strong>in</strong>g R, Koornneef L, Peeters HJF, Gillissen JPA. (1989). Decompression surgery <strong>in</strong> Graves’ orbitopathy: a<br />
review of 14 years’ experience at the Orbita Centrum, Amsterdam. Dev Ophthalmol 20:185–198.<br />
Hudson HL, Lev<strong>in</strong> L, Feldon SE. (1991). Graves’ exophthalmos unrelated to extraocular muscle enlargement.<br />
Ophthalmology 98:1495–1499.<br />
Hurbli T, Char DH, Harris J, Weaver K, Greenspan F, Shel<strong>in</strong>e G. (1985). Radiation therapy for thyroid eye diseases.<br />
Am J Ophthalmol 99:633–637.<br />
Hurwitz JJ, Freeman JL, Eplett CJ, et al. (1992). Ethmoidectomy decompression for the treatment of Graves’ optic<br />
neuropathy. Can J Ophthalmol 27:283–287.<br />
Just M, Kahaly G, Higer HP, et al. (1991). Graves’ ophthalmopathy: role of MR imag<strong>in</strong>g <strong>in</strong> radiation therapy.<br />
Radiology 179:187–190.<br />
Kahaly G, Lieb W, Müller-Forell W, et al. (1990). Ciamexone <strong>in</strong> endocr<strong>in</strong>e orbitopathy. Acta Endocr<strong>in</strong>ol<br />
122:13–21.<br />
Kahaly G, Rosler HP, Pitz S, Hommel G (2000). Low- versus high-dose radiotherapy for Graves’ ophthalmopathy:<br />
a randomized, s<strong>in</strong>gle bl<strong>in</strong>d trial. J Cl<strong>in</strong> Endocr<strong>in</strong>ol Metab 85:102–108.<br />
Kalmann R, Mourits MP. (1998). Prevalence and management of elevated <strong>in</strong>traocular pressure <strong>in</strong> patients with<br />
Graves’ orbitopathy. Br J Ophthalmol 82:754–757.<br />
Kalmann R, Mourits MP. (1999). Diabetes mellitus: a risk factor <strong>in</strong> patients with Graves’ orbitopathy. Br J<br />
Ophthalmol 83:463–465.<br />
Kalmann R, Mourits MP, van der Pol JP, Koormeef L. (1997). Coronal approach for rehabilitative orbital<br />
decompression <strong>in</strong> Graves’ ophthalmopathy. Br J Ophthalmol 81:41–45.<br />
Kazim M, Trokel SL, Acaroglu G, Elliott A. (2000). Reversal of dysthyroid optic neuropathy follow<strong>in</strong>g orbital fat<br />
decompression. Br J Ophthalmol 84:600–605.<br />
Kazim M, Trokel S, Moore S. (1991). Treatment of acute Graves’ orbitopathy. Ophthalmology 98:1443–1448.<br />
Keltner JL. (1998). Is Graves ophthalmopathy a preventable disease? Arch Ophthalmol 116:1106–1107.<br />
Kendler DL, Lippa J, Rootman J. (1993). The <strong>in</strong>itial cl<strong>in</strong>ical characteristics of Graves’ orbitopathy vary with age<br />
and sex. Arch Ophthalmol 111:197–201.<br />
Kennedy DW, Goldste<strong>in</strong> ML, Miller NR, Z<strong>in</strong>reich SJ. (1990). Endoscopic transnasal orbital decompression. Arch<br />
Otolaryngol Head Neck Surg 116:275–282.<br />
Kennerdell JS, Maroon JC, Buerger GF. (1987). Comprehensive surgical management of proptosis <strong>in</strong> dysthyroid<br />
orbitopathy. Orbit 6:153–179.<br />
K<strong>in</strong>youn JL, Kal<strong>in</strong>a RE, Brower SA, et al. (1984). Radiation ret<strong>in</strong>opathy after orbital irradiation for Graves’<br />
ophthalmopathy. Arch Ophthalmol 102:1473–1476.<br />
Krassas GE, Doumas A, Kaltsas T, et al. (1999a). Somatostat<strong>in</strong> receptor sc<strong>in</strong>tigraphy before and after treatment<br />
with somatostat<strong>in</strong> analogues <strong>in</strong> patients with thyroid eye disease. Thyroid 9:47–52.<br />
Krassas GE, Heufelder AE. (2001). Immunosuppressive therapy <strong>in</strong> patients with thyroid eye disease: an overview<br />
of current concepts. Eur J Endocr<strong>in</strong>ol 144:311–318.<br />
Krassas GE, Kahaly GJ. (1999b). The role of octreoscan <strong>in</strong> thyroid eye disease. Eur J Endocr<strong>in</strong>ol 140:373–375.<br />
Kubis KC, Danesh-Meyer H, Pribitk<strong>in</strong> EA, et al. (2000). Progressive visual loss and ophthalmoplegia. Survey<br />
Ophthalmol 44:433–441.<br />
Kulw<strong>in</strong> DR, Cotton RT, Kersten RC. (1990). Comb<strong>in</strong>ed approach to orbital decompression. Otolaryngol Cl<strong>in</strong> North<br />
Am 23:381–390.<br />
Lamberg BA, Grahne B, Tommila V, et al. (1985). Orbital decompression <strong>in</strong> endocr<strong>in</strong>e exophthalmos of Graves’<br />
disease. Acta Endocr<strong>in</strong>ol (Copenh) 109:335–340.<br />
Leatherbarrow B, Lendrum J, Mahaffey PJ, et al. (1991). Three wall orbital decompression for Graves’<br />
ophthalmopathy via a coronal approach. Eye 5:456–465.<br />
Lemke BN. (1991). Management of thyroid eyelid retraction. Focal Po<strong>in</strong>t 9:Module 6, 1–9.<br />
Leone CR Jr, Bajandas FJ. (1981). Inferior orbital decompression for dysthyroid optic neuropathy. Ophthalmology<br />
88:525–532.<br />
Leone CR Jr, Piest KL, Newman RJ. (1989). Medial and lateral wall decompression for thyroid ophthalmopathy.<br />
Am J Ophthalmol 108:160–166.<br />
L<strong>in</strong>berg JV, <strong>An</strong>derson RL. (1981). Transorbital decompression: <strong>in</strong>dications and results. Arch Ophthalmol 99:113–119.
364 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
L<strong>in</strong>net J, Hegedus L, Bjerre P. (2001). Results of a neurosurgical two-wall orbital decompression <strong>in</strong> the treatment of<br />
severe thyroid associated ophthalmopathy. Acta Ophthalmol Scand 79:49–52.<br />
Liu D, Feldon SE. (1992). Thyroid ophthalmopathy. Ophthalmol Cl<strong>in</strong> North Am 5:597–622.<br />
Lloyd WC, Leone CR. (1992). Supervoltage orbital radiotherapy <strong>in</strong> 36 cases of Graves’ disease. Am J Ophthalmol<br />
113:374–380.<br />
Lund VJ, Lark<strong>in</strong> G, Felis P, Asams G. (1997). Orbital decompression for thyroid eye disease: a comparison of<br />
external and endoscopic techniques. J Laryngol Otol 111:1051–1055.<br />
Lyons CJ, Rootman J. (1994). Orbital decompression for disfigur<strong>in</strong>g exophthalmos <strong>in</strong> thyroid ophthalmopathy.<br />
Ophthalmology 101:223–230.<br />
Maarten P, Sasim IV. (1999). A s<strong>in</strong>gle technique to correct various degrees of upper lid retraction <strong>in</strong> patients with<br />
Graves’ orbitopathy. Br J Ophthalmol 83:81–84.<br />
Macchia PE, Bagatt<strong>in</strong>i M, Lupoli G, et al. (2001). High-dose <strong>in</strong>travenous corticosteroid therapy for Graves’<br />
ophthalmopathy. J Endocr<strong>in</strong>ol Invest 24:152–158.<br />
Mann K. (1999). Risk of smok<strong>in</strong>g <strong>in</strong> thyroid-associated orbitopathy. Exp Cl<strong>in</strong> Endocr<strong>in</strong>ol Diabetes. 107 (suppl<br />
5):S164–167.<br />
Mann W, Kahaly G, Lieb W, Rothoff T, Spr<strong>in</strong>gborn S. (1993). Orbital decompression for endocr<strong>in</strong>e ophthalmopathy:<br />
the endonasal approach. Dev Ophthalmol 25:142–150.<br />
Marcocci C, Bartalena L, Bogazzi F, Bruno-Bossio G, Lepri A, P<strong>in</strong>crea A. (1991). Orbital radiotherapy comb<strong>in</strong>ed<br />
with high dose systemic glucocorticoids for Graves’ ophthalmopathy is more effective than radiotherapy<br />
alone: results of a prospective randomized study. J Endocr<strong>in</strong>ol Invest 14:853–860.<br />
Marcocci C, Bartalena L, Tanda ML, et al. (1999a). Graves’ ophthalmopathy and 131I therapy. Q J Nucl Med<br />
43:307–312.<br />
Marcocci C, Bartalena L, Tanda ML, et al. (2001). Comparison of the effectiveness and tolerability of <strong>in</strong>travenous<br />
or oral glucocorticoids associated with orbital radiotherapy <strong>in</strong> the management of severe Graves’ ophthalmopathy:<br />
results of a prospective, s<strong>in</strong>gle bl<strong>in</strong>d randomized study. J Cl<strong>in</strong> Endocr<strong>in</strong>ol Metab 86:3562–3567.<br />
Marcocci C, Bruno-Bossio G, Manetti L, et al. (1999b). The course of Graves’ ophthalmopathy is not <strong>in</strong>fluenced by<br />
near total thyroidectomy: a case control study. Cl<strong>in</strong> Endocr<strong>in</strong>ol 51:503–508.<br />
May A, Fries U, Reimold I, Weber A. (1999). Microsurgical endonasal decompression <strong>in</strong> thyroid orbitopathy. Acta<br />
Otolaryngol 119:826–831.<br />
May A, Fries U, von Ilberg C, Weber A. (2000). Indication and technique of transnasal microscopic orbital<br />
decompression for endocr<strong>in</strong>e ophthalmopathy. J Otorh<strong>in</strong>olaryngol Related Specialties 62:128–133.<br />
Mayer E, Herdman G, Burnett C, et al. (2001). Serial STIR magnetic resonance imag<strong>in</strong>g correlates with cl<strong>in</strong>ical<br />
score of activity <strong>in</strong> thyroid disease. Eye 15:313–318.<br />
McCord CD Jr. (1981). Orbital decompression for Graves’ disease: exposure through lateral canthal and <strong>in</strong>ferior<br />
fornix <strong>in</strong>cision. Ophthalmology 88:533–541.<br />
McCord CD Jr. (1985). Current trends <strong>in</strong> orbital decompression. Ophthalmology 92:21–33.<br />
McNab AA. (1997). Orbital decompression for thyroid orbitopathy. Aust N Z J Ophthalmol 25:55–61.<br />
Michel O, Oberlander N, Neugebauer P, et al. (2001). Follow-up of transnasal orbital decompression <strong>in</strong> severe<br />
Graves’ ophthalmopathy. Ophthalmology 108:400–404.<br />
Mourits M, Koorneef L, Van Mourik-Noordenbos AM, et al. (1990a). Extraocular muscle surgery for Graves’<br />
ophthalmopathy: does prior treatment <strong>in</strong>fluence surgical outcomes? Br J Ophthalmol 74:481–483.<br />
Mourits MP, Koorneef L, Wiers<strong>in</strong>ga WM, Prummell MF, Berghout A, van der Gaag R. (1990b). Orbital<br />
decompression for Graves’ ophthalmopathy by <strong>in</strong>feromedial plus lateral and coronal approach. Ophthalmology<br />
97:636–641.<br />
Mourits M, Suttorp-Schulten MSA, Tijssen RO, Apkarian P. (1990c). Contrast sensitivity and the diagnosis of<br />
dysthyroid optic neuropathy. Doc Ophthalmol 74:329–335.<br />
Mourits M, van Kempen-Harteveld ML, Garcia MB, et al. (2000). Radiotherapy for Graves’ orbitopathy:<br />
randomized placebo-controlled study. Lancet 355:1505–1509.<br />
Muller-Forell W, Pitz S, Mann W, Kahaly GJ. (1999). <strong>Neuro</strong>radiological diagnosis <strong>in</strong> thyroid-associated orbitopathy.<br />
Exp Cl<strong>in</strong> Endocr<strong>in</strong>ol Diabetes 107 (suppl 5):S177–183.<br />
Nagy EV, Toth J, Kaldi I, et al. (2000). Graves’ ophthalmopathy: eye muscle <strong>in</strong>volvement <strong>in</strong> patients with diplopia.<br />
Eur J Endocr<strong>in</strong>ol 142:591–597.<br />
Neugenbauer A, Nish<strong>in</strong>o K, Neugebauer P, Konen W, Michel O. (1996). Effects of bilateral orbital decompression<br />
by an endoscopic endonasal approach <strong>in</strong> dysthyroid orbitopathy. Br J Ophthalmol 80:58–62.<br />
Nunery WR. (1991). Ophthalmic Graves’ disease: a dual theory of pathogenesis. Ophthalmol Cl<strong>in</strong> North Am 4:73–<br />
87.<br />
Nunery WR, Mart<strong>in</strong> RT, He<strong>in</strong>z GW, Gav<strong>in</strong> TJ. (1993). The association of cigarette smok<strong>in</strong>g with cl<strong>in</strong>ical subtypes<br />
of ophthalmic Graves’ disease. Ophthalmic Plast Reconstr Surg 9:77–82.
Thyroid Eye Disease: Graves’ Ophthalmopathy 365<br />
Ohtsuka K, Hashimoto M. (1999a). 1H-Magnetic resonance spectroscopy of retrobulbar tissue <strong>in</strong> Graves’<br />
ophthalmopathy. Am J Ophthalmol 128:715–719.<br />
Ohtsuka K, Nakamura Y. (1999b). Results of transmedial-canthal ethmoidal decompression for severe dysthyroid<br />
optic neuropathy. Jpn J Ophthalmol 43:426–432.<br />
Olivari N. (1991). Transpalpebral decompression of endocr<strong>in</strong>e ophthalmopathy (Graves’ disease) by removal of<br />
<strong>in</strong>traorbital fat: experience with 147 operations over 5 years. Plast Reconstr Surg 87:627–643.<br />
Oliver JM. (1998a). Botul<strong>in</strong>um tox<strong>in</strong> A treatment of overactive corrugator supercilii <strong>in</strong> thyroid eye disease. Br J<br />
Ophthalmol 82:528–533.<br />
Oliver JM, Rose GE, Khaw PT, Coll<strong>in</strong> JRO. (1998b). Correction of lower eyelid retraction <strong>in</strong> thyroid eye disease: a<br />
randomized trial of retractor tenotomy with adjuvant antimetabolite versus scleral graft. Br J Ophthalmol<br />
82:174–180.<br />
Ozgen A, Alp MN, Ariyurek M, et al. (1999). Quantitative CT or the orbit <strong>in</strong> Graves’ disease. Br J Radiol 72:757–<br />
762.<br />
Ozkan SB, Can D, Soylev MF, et al. (1997). Chemodenervation <strong>in</strong> treatment of upper eyelid retraction.<br />
Ophthalmologica 211:387–390.<br />
Paridaens D, Hans K, Van Buitenen S, Mourits MP. (1998). The <strong>in</strong>cidence of diplopia follow<strong>in</strong>g coronal and<br />
translid orbital decompression <strong>in</strong> Graves orbitopathy. Eye 12:800–805.<br />
Paridaens DA, Verhoeff K, Bouwens D, van den Bosch WA. (2000). Transconjunctival orbital decompression <strong>in</strong><br />
Graves’ ophthalmopathy: lateral wall approach ab <strong>in</strong>terno. Br J Ophthalmol 84:775–781.<br />
Perros P, Weightman DR, Crombie AL, Kendall-Taylor P. (1990). Azathiopr<strong>in</strong>e <strong>in</strong> the treatment of thyroidassociated<br />
ophthalmopathy. Acta Endocr<strong>in</strong>ol 122:8–12.<br />
Petersen IA, Kriss JP, McDougall IR, Donaldson SS. (1990). Prognostic factors <strong>in</strong> the radiotherapy of Graves’<br />
ophthalmopathy. Int J Radiat Oncol Biol Phys 19:259–264.<br />
Pfeilschifter J, Ziegler R. (1996). Smok<strong>in</strong>g and endocr<strong>in</strong>e ophthalmopathy: impact of smok<strong>in</strong>g severity and current<br />
vs lifetime cigarette consumption. Cl<strong>in</strong> Endocr<strong>in</strong>ol 45:477–481.<br />
Prendiville P, Chopra M, Gauderman WJ, Feldon SE. (2000). The role of restricted motility <strong>in</strong> determ<strong>in</strong><strong>in</strong>g<br />
outcomes for vertical strabismus surgery <strong>in</strong> Graves’ ophthalmopathy. Ophthalmology 107:545–549.<br />
Prophet H, Matic GB, W<strong>in</strong>kler RE, et al. (2001). Two cases of refractory endocr<strong>in</strong>e ophthalmopathy successfully<br />
treated with extracorporeal immunoadsorption. Ther Aphersis 5:142–146.<br />
Prummel MF, Mourits MP, Berghout A, et al. (1989). Prednisone and cyclospor<strong>in</strong>e <strong>in</strong> the treatment of severe<br />
Graves’ ophthalmopathy. N Engl J Med 321:1353–1359.<br />
Prummel MF, Wiers<strong>in</strong>ga WM. (1993). Smok<strong>in</strong>g and risk of Graves’ disease. JAMA 269:479–482.<br />
Prummel MF, Wiers<strong>in</strong>ga WM, Mourits MP, et al. (1990). Effect of abnormal thyroid function on the severity of<br />
Graves’ ophthalmopathy. Arch Intern Med 150:1098–1101.<br />
Rasmussen AK, Nygaard B, Feldt-Rasmussen U. (2000). I131 and thyroid-associated ophthalmopathy. Eur J<br />
Endocr<strong>in</strong>ol 143:155–160.<br />
Rav<strong>in</strong> JG, Sisson JC, Knapp WT. (1975). Orbital radiation for the ocular changes of Graves’ disease. Am J<br />
Ophthalmol 79:285–288.<br />
Rush S, W<strong>in</strong>terkorn JM, Zak R. (2000). Objective evaluation of improvement <strong>in</strong> optic neuropathy follow<strong>in</strong>g<br />
radiation therapy for thyroid eye disease. Int J Radiat Oncol Biol Phys 47:191–194.<br />
Ruttum MS. (2000). Effect of prior orbital decompression on outcome of strabismus surgery <strong>in</strong> patients with<br />
thyroid ophthalmopathy. J Am Assoc Pediatr Ophthalmol Strabismus 4:102–105.<br />
Salvi M, Zhang ZG, Haegart D, et al. (1990). Patients with endocr<strong>in</strong>e ophthalmopathy not associated with overt<br />
thyroid disease have multiple thyroid immunological abnormalities. J Cl<strong>in</strong> Endocr<strong>in</strong>ol Metab 70:89–94.<br />
Sandler HM, Rubenste<strong>in</strong> JH, Fowble BL, Sergott RC, Sav<strong>in</strong>o PJ, Bosley TM. (1989). Results of radiotherapy for<br />
thyroid ophthalmopathy. Int J Radiat Oncol Biol Phys 17:823–827.<br />
Seiff SR, Tovilla JL, Carter SR, Choo PH. (2000). Modified orbital decompression for dysthyroid orbitopathy.<br />
Ophthalmic Plast Reconstr Surg 16:62–66.<br />
Sh<strong>in</strong>e B, Fells P, Edwards OM, Weetman AP. (1990). Association between Graves’ ophthalmopathy and smok<strong>in</strong>g.<br />
Lancet 335:1261–1263.<br />
Shorr N, Seiff SR. (1986). The four stages of surgical rehabilitation of the patient with dysthyroid ophthalmopathy.<br />
Ophthalmology 93:476–483.<br />
Shulka R, Kur<strong>in</strong>czuk JJ. (1990). Graves’ ophthalmopathy and smok<strong>in</strong>g. Lancet 336:184.<br />
Sillers MJ, Cuilty-Siller C, Kuhn FA, et al. (1997). Transconjunctival endoscopic orbital decompression. Otolaryngol<br />
Head Neck Surg 117:S137-S141.<br />
Smith JR, Rosenbaum JT. (2001). A role for methotrexate <strong>in</strong> the management of non-<strong>in</strong>fectious orbital <strong>in</strong>flammatory<br />
disease. Br J Ophthalmol 85:1220–1224.
366 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
So NM, Lam WW, Cheng G, et al. (2000). Assessment of optic nerve compression <strong>in</strong> Graves’ ophthalmopathy. The<br />
usefulness of a quick T1 weighted sequence. Acta Radiol 41:559–561.<br />
Solberg Y, Rosner M, Belk<strong>in</strong> M. (1998). The association between cigarette smok<strong>in</strong>g and ocular disease. Surv<br />
Ophthalmol 42:535–547.<br />
Srivastava SK, Newman NJ. (2000). Pseudo-pseudotumor. Surv Ophthalmol 45:135–138.<br />
Tallstedt L, Lundell G, Taube A. (1993). Graves’ ophthalmopathy and tobacco smok<strong>in</strong>g. Acta Endocr<strong>in</strong>ol 129:147–150.<br />
Tallstedt L, Lundell G, Tjrr<strong>in</strong>g O, et al. (1992). Occurrence of ophthalmopathy after treatment for Graves’<br />
hyperthyroidism. N Engl J Med 326:1733–1738.<br />
Tallstedt L, Paptziamos G, Lundbland L, <strong>An</strong>ggard A. (2000). Results of transantral orbital decompression <strong>in</strong><br />
patients with thyroid-associated ophthalmopathy. Acta Ophthalmol Scand 78:206–210.<br />
Tellez M, Cooper J, Edmonds C. (1992). Graves’ ophthalmopathy <strong>in</strong> relation to cigarette smok<strong>in</strong>g and ethnic<br />
orig<strong>in</strong>. Cl<strong>in</strong> Endocr<strong>in</strong>ol 36:291–294.<br />
Thaller SR, Kawamoto HK. (1990). Surgical correction of exophthalmos secondary to Graves’ disease. Plast<br />
Reconstrr Surg 86:411–417.<br />
Traisk F, Tallstedt L. (2001). Thyroid associated ophthalmopathy: botul<strong>in</strong>um tox<strong>in</strong> A <strong>in</strong> the treatment of upper<br />
eyelid retraction—a pilot study. Acta Ophthalmol Scand 79:585–588.<br />
Trobe JD, Glaser JS, Laflamme P. (1978). Dysthyroid optic neuropathy: cl<strong>in</strong>ical profile and rationale for management.<br />
Arch Ophthalmol 96:1199–1209.<br />
Trokel S, Kazim M, Moore S. (1993). Orbital fat removal: decompression for Graves’ orbitopathy. Ophthalmology<br />
100:674–682.<br />
Tsuj<strong>in</strong>o K, Hirota S, Hagiwara M, et al. (2000). <strong>Cl<strong>in</strong>ical</strong> outcomes of orbital irradiation comb<strong>in</strong>ed with or without<br />
systemic high-dose or pulsed corticosteroids for Graves’ ophthalmopathy. Int J Radiat Oncol Biol Phys 48:857–<br />
864.<br />
Ulualp SO, Massaro BM, Toohill RJ. (1999). Course of proptosis <strong>in</strong> patients with Graves’ disease after endoscopic<br />
orbital decompression. Laryngoscope 109:1217–1222.<br />
Uysal AR, Corapciouglu D, Tonyukuk VC, et al. (1999). Effect of octreotide treatment on Graves’ ophthalmopathy.<br />
Endocr J 46:573–577.<br />
Van der Wal KG, de Visscher JG, Boukes RJ, Smed<strong>in</strong>g B. (2001). Surgical treatment of Graves’ orbitopathy: a<br />
modified balanced approach. Int J Oral Maxillofacial Surg 30:254–258.<br />
Van Ruyven RL, Van Den Bosch WA, et al. (2000). The effect of retrobulbar irradiation on exophthalmos, ductions<br />
and soft tissue signs <strong>in</strong> Graves’ ophthalmopathy: a retrospective analysis of 90 cases. Eye 14:761–764.<br />
von Brauchitsch DK, Egbert J, Kersten RC, Kulw<strong>in</strong> DR. (1999). Spontaneous resolution of upper eyelid retraction<br />
<strong>in</strong> thyroid orbitopathy. J <strong>Neuro</strong>-ophthalmol 19:122–124.<br />
Warren JD, Spector JG, Burde R. (1989). Long-term follow-up and recent observations on 305 cases of orbital<br />
decompression for dysthyroid orbitopathy. Laryngoscope 99:35–40.<br />
Weetman AP. (2000). Graves’ disease. N Engl J Med 343:1236–1248.<br />
Weetman AP, Wiers<strong>in</strong>ga WM. (1998). Current management of thyroid-associated ophthalmopathy <strong>in</strong> Europe.<br />
Results of an <strong>in</strong>ternational survey. Cl<strong>in</strong> Endocr<strong>in</strong>ol 49:21–28.<br />
West M, Stranc M. (1997). Long term results of four-wall orbital decompression for Graves’ ophthalmopathy. Br J<br />
Plast Surg 50:507–516.<br />
Wiers<strong>in</strong>ga WM, Prummel MF. (2000). <strong>An</strong> evidence-based approach to the treatment of Graves’ ophthalmopathy.<br />
Endocr<strong>in</strong>ol Metab Cl<strong>in</strong> North Am 29:297–319.<br />
Wiers<strong>in</strong>ga WM, Smit T, Schuster-Uittenhoeve AL, van der Gaag R, Koornneef L. (1988). Therapeutic outcome of<br />
prednisone medication and of orbital irradiation <strong>in</strong> patients with Graves’ ophthalmopathy. Ophthalmologica<br />
197:75–84.<br />
Wilson WB, Manke WF. (1991). Orbital decompression <strong>in</strong> Graves’ disease: the predictability of reduction of<br />
proptosis. Arch Ophthalmol 109:343–345.<br />
Wulc AE, Popp JC, Bartlett SP. (1990). Lateral wall advancement <strong>in</strong> orbital decompression. Ophthalmology 97:1358–<br />
1369.
17 r<br />
Nystagmus and Other Ocular<br />
Oscillations<br />
Nystagmus is a rhythmic, repetitive, to-and-fro movement of the eyes that <strong>in</strong>cludes<br />
smooth s<strong>in</strong>usoidal oscillations (pendular nystagmus) and alternation of slow drift and<br />
corrective quick phase (jerk nystagmus) (Leigh, 1999). If the size of the oscillation differs<br />
<strong>in</strong> each eye, the abnormal movements are referred to as dissociative nystagmus. If the<br />
direction of the oscillations <strong>in</strong> each eye is the same, the nystagmus is conjugate, and if<br />
they differ, the nystagmus is dysconjugate. Nystagmus <strong>in</strong>duced by optok<strong>in</strong>etic or<br />
vestibular stimuli is physiologic but can be affected by pathologic processes. Nystagmus<br />
<strong>in</strong> extreme lateral or vertical gaze (end-po<strong>in</strong>t nystagmus) can also be found as a normal<br />
variant. End-po<strong>in</strong>t nystagmus tends to wane easily and belongs to the variety described<br />
below as ‘‘gaze-evoked’’ nystagmus. These physiologic forms of nystagmus may be<br />
pathologic if they persist beyond a few beats <strong>in</strong> end gaze.<br />
<strong>An</strong> approach to pathologic nystagmus is presented <strong>in</strong> this chapter. The slow phase<br />
reflects the underly<strong>in</strong>g abnormality caus<strong>in</strong>g the nystagmus. The slow component may<br />
have a uniform velocity or may reduce or ga<strong>in</strong> speed as the eyes move <strong>in</strong> the direction<br />
of the slow component. This slow-phase abnormality is usually due to disruption of the<br />
mechanisms that normally function to hold gaze steady. Thus, disorders of the<br />
vestibular system, the gaze-hold<strong>in</strong>g mechanisms (e.g., the neural <strong>in</strong>tegrator), and<br />
visual stabilization and pursuit systems may lead to nystagmus (Leigh, 1999).<br />
Vestibular tone imbalance results <strong>in</strong> an asymmetric <strong>in</strong>put to the horizontal gaze<br />
generator; vestibular nystagmus shows l<strong>in</strong>ear, constant velocity slow phases reflect<strong>in</strong>g a<br />
persistent drive of the eyes toward the damaged vestibular apparatus (labyr<strong>in</strong>th, nerve,<br />
nuclei). <strong>An</strong> impaired neural <strong>in</strong>tegrator (‘‘leaky’’ <strong>in</strong>tegrator) may cause gaze-evoked<br />
nystagmus with a negative exponential slow phase. The velocity of the slow component<br />
decreases as the eyes move from the periphery of the orbit, where the pull due to the<br />
viscosity of the orbital tissues is greatest, toward rest<strong>in</strong>g <strong>in</strong> primary position. The<br />
<strong>in</strong>ability of the gaze hold<strong>in</strong>g mechanisms to keep the eyes eccentric <strong>in</strong> the orbit is often<br />
present with central or peripheral lesions caus<strong>in</strong>g weakness of eye movements. For this<br />
367
368 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
reason, this type of nystagmus is sometimes referred to as ‘‘gaze-paretic’’ nystagmus.<br />
High ga<strong>in</strong> <strong>in</strong>stability of slow eye movement subsystems (e.g., the pursuit system) may<br />
also cause nystagmus, with the nystagmus slow phase hav<strong>in</strong>g an exponentially<br />
<strong>in</strong>creas<strong>in</strong>g time course (‘‘runaway’’ movements). Such nystagmus <strong>in</strong> the horizontal<br />
plane is seen <strong>in</strong> congenital nystagmus and <strong>in</strong> the vertical plane is seen with cerebellar<br />
disease. High ga<strong>in</strong> <strong>in</strong>stability may also result <strong>in</strong> congenital or acquired pendular<br />
nystagmus. The types of nystagmus and other ocular oscillations are outl<strong>in</strong>ed <strong>in</strong><br />
Figure 17–1.<br />
Are the Abnormal Eye Movements B<strong>in</strong>ocular<br />
and Symmetric, B<strong>in</strong>ocular and Asymmetric, or<br />
Monocular?<br />
The oscillations may be conf<strong>in</strong>ed to one eye (monocular), <strong>in</strong>volve ma<strong>in</strong>ly one eye<br />
(b<strong>in</strong>ocular asymmetric or dissociated), or <strong>in</strong>volve both eyes symmetrically (b<strong>in</strong>ocular<br />
symmetric) (Burde, 1991).<br />
What Causes Monocular Eye Oscillations and<br />
Asymmetric B<strong>in</strong>ocular Eye Oscillations?<br />
Monocular eye oscillations and asymmetric b<strong>in</strong>ocular eye oscillations may be due to<br />
spasmus nutans and its mimickers, monocular visual deprivation or loss, monocular<br />
pendular nystagmus, <strong>in</strong>ternuclear ophthalmoplegia and its mimickers, partial paresis of<br />
extraocular muscles, restrictive syndromes of extraocular muscles, or superior oblique<br />
myokymia.<br />
Spasmus nutans is a benign syndrome characterized by a triad of head nodd<strong>in</strong>g,<br />
nystagmus, and abnormal head posture (Gottlob, 1995b; Young, 1997). The onset is<br />
typically <strong>in</strong> the first year of life and remits spontaneously with<strong>in</strong> 1 month to several (up<br />
to 8) years. The syndrome is occasionally familial and has been reported <strong>in</strong> monozygotic<br />
tw<strong>in</strong>s. The s<strong>in</strong>usoidal nystagmus is often <strong>in</strong>termittent, asymmetric, or unilateral, and of<br />
high frequency and small amplitude with a ‘‘shimmer<strong>in</strong>g’’ quality. The nystagmus is<br />
usually horizontal but may have a vertical or torsional component. It may be<br />
accentuated by near effort and is usually greater <strong>in</strong> an abduct<strong>in</strong>g eye. Rarely, convergence<br />
nystagmus may occur (Massry, 1996). The irregular head nodd<strong>in</strong>g with spasmus<br />
nutans has horizontal, vertical, or mixed components. Patients often also demonstrate a<br />
head turn or tilt.<br />
In children with spasmus nutans, monocular nystagmus, or asymmetric pendular<br />
nystagmus, one must consider tumor of the anterior visual pathway (e.g., optic nerve,<br />
chiasm, third ventricle, or thalamus) (Arnoldi, 1995; Gottlob, 1990; Newman, 1990).<br />
These latter patients may also have visual loss, optic atrophy, or other signs of tumor.<br />
Other spasmus nutans mimickers <strong>in</strong>clude arachnoid cyst, Leigh’s subacute necrotiz<strong>in</strong>g<br />
encephalomyelopathy, congenital stationary night bl<strong>in</strong>dness (Gottlob, 1995a; Lambert,<br />
1993), ret<strong>in</strong>al dystrophy (Smith, 2000), and Bardet-Biedl syndrome (characterized by<br />
polydactyly, obesity, cognitive delay, and ret<strong>in</strong>al degeneration) (Gottlob, 1999).<br />
All children with monocular nystagmus or spasmus nutans should undergo a<br />
complete ophthalmologic exam<strong>in</strong>ation. We recommend neuroimag<strong>in</strong>g (preferably
Nystagmus and Other Ocular Oscillations 369<br />
Figure 17–1. Nystagmus and other ocular oscillations.
370 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
magnetic resonance imag<strong>in</strong>g [MRI]) <strong>in</strong> patients with monocular or predom<strong>in</strong>antly<br />
monocular oscillations, spasmus nutans, or a spasmus nutans–like cl<strong>in</strong>ical picture<br />
(class IV, level C). Although most cases of spasmus nutans are benign, atypical features<br />
should prompt further evaluation <strong>in</strong>clud<strong>in</strong>g older age of onset, associated visual loss, or<br />
persistence of symptoms (Gottlob, 1990; Newman, 1990). Some authors, however, have<br />
stated that the estimated prevalence of tumor <strong>in</strong> spasmus nutans is less than 1.4%, and<br />
have suggested that without other evidence of an <strong>in</strong>tracranial mass lesion, neuroimag<strong>in</strong>g<br />
of <strong>in</strong>fants <strong>in</strong>itially diagnosed with spasmus nutans may not be immediately<br />
warranted (Arnoldi, 1995). Electrophysiologic test<strong>in</strong>g should be considered for a<br />
myopic child suspected of hav<strong>in</strong>g spasmus nutans to exclude the diagnosis of<br />
congenital stationary night bl<strong>in</strong>dness (Lambert, 1993) or ret<strong>in</strong>al dystrophy (Smith,<br />
2000) (class IV, level C).<br />
Monocular nystagmus may occur <strong>in</strong> adults or children with acquired monocular<br />
visual loss, and consists of small, slow vertical pendular oscillations <strong>in</strong> primary position<br />
of gaze. It may develop years after uniocular visual loss (Heimann-Bielschowsky<br />
phenomenon) and may improve if vision is corrected. Monocular, small-amplitude,<br />
fast frequency, and predom<strong>in</strong>antly horizontal nystagmus <strong>in</strong> children may be caused by<br />
unilateral anterior visual pathway disease (Davey, 1998; Good, 1993).<br />
Acquired monocular pendular nystagmus may also occur with multiple sclerosis,<br />
neurosyphilis, and bra<strong>in</strong>stem <strong>in</strong>farct (thalamus and upper midbra<strong>in</strong>) and may be<br />
vertical, horizontal, or multivectorial. Stahl et al (2000) reported that servo-controlled<br />
optics could reduce oscillopsia <strong>in</strong> acquired pendular nystagmus.<br />
Vertical pendular nystagmus, with greater amplitude <strong>in</strong> the <strong>in</strong>volved eye, has been<br />
described <strong>in</strong> a patient with chronic monocular myositis of the medial and lateral rectus<br />
muscles (Goldberg, 1978). Monocular downbeat nystagmus may occur with acute<br />
<strong>in</strong>farction of the medial thalamus and upper midbra<strong>in</strong> and with pontocerebellar<br />
degeneration; this abnormality is likely due to dysfunction of the ipsilateral brachium<br />
conjunctivum. Contralateral unilateral downbeat nystagmus has been described with a<br />
paramedian thalamopeduncular <strong>in</strong>farction (Oishi, 1997). Monocular rotatory nystagmus<br />
may occur with bra<strong>in</strong>stem lesions. Congenital nystagmus may rarely be uniocular. One<br />
patient has been described who developed ictal monocular horizontal nystagmus<br />
dur<strong>in</strong>g a generalized seizure triggered by photic stimulation (Jacome, 1982). We<br />
recommend that neuroimag<strong>in</strong>g be performed <strong>in</strong> monocular nystagmus (class IV,<br />
level C).<br />
Dissociated nystagmus occurs <strong>in</strong> the abduct<strong>in</strong>g eye <strong>in</strong> <strong>in</strong>ternuclear ophthalmoplegia<br />
(INO) and <strong>in</strong> pseudo-INO syndromes. These entities and their evaluation are discussed<br />
<strong>in</strong> Chapter 14. In patients with partial paresis of one of the extraocular muscles, a<br />
monocular oscillation may occur <strong>in</strong> the <strong>in</strong>volved eye or its yoke dur<strong>in</strong>g an ocular<br />
movement <strong>in</strong>to the field of action of the <strong>in</strong>volved muscle (Burde, 1991). Monocular<br />
oscillations may also occur <strong>in</strong> restrictive syndromes (e.g., thyroid ophthalmopathy) <strong>in</strong><br />
the field of action <strong>in</strong> which the tether<strong>in</strong>g is occurr<strong>in</strong>g (Burde, 1991).<br />
Superior oblique myokymia (SOM) is a disorder of unknown etiology characterized<br />
symptomatically by oscillopsia, vertical or torsional diplopia, or both. Affected patients<br />
show bursts of rotary oscillations of the eye of small amplitude and high frequency,<br />
slow-frequency large-amplitude <strong>in</strong>torsional movements, or a comb<strong>in</strong>ation of these<br />
paroxysms. Most patients with SOM compla<strong>in</strong> of brief episodes of rapid vertical or<br />
torsional movements of the environment or shimmer<strong>in</strong>g sensations, usually last<strong>in</strong>g only<br />
a few seconds.
Nystagmus and Other Ocular Oscillations 371<br />
<strong>Neuro</strong>-ophthalmologic exam<strong>in</strong>ation of SOM patients often reveals brief episodes of<br />
rapid, f<strong>in</strong>e, torsional movements of one eye that are best seen us<strong>in</strong>g either the slit-lamp<br />
biomicroscope or the direct ophthalmoscope. The abnormal movements can be <strong>in</strong>duced<br />
<strong>in</strong> some patients by movement of the affected eye down and outward, by a head tilt<br />
toward the side of the affected eye, by convergence effort, or by movement of the eye<br />
downward and back to primary position.<br />
Patients with SOM are usually young adults who are otherwise healthy. Most patients<br />
report no precipitat<strong>in</strong>g event for the onset of their symptoms. Several cases have<br />
followed ipsilateral trochlear nerve palsies, lead<strong>in</strong>g some authors to suggest that SOM<br />
might be associated with the recovery stage of <strong>in</strong>jury to this nerve (Komai, 1992). SOM<br />
has occurred several months after removal of a cerebellar tumor. In addition, two cases<br />
of SOM have occurred <strong>in</strong> patients with posterior fossa tumors (one an astrocytoma of<br />
the rostral cerebellar vermis with midbra<strong>in</strong> tectal compression and the other a pilocytic<br />
astrocytoma expand<strong>in</strong>g with<strong>in</strong> the fourth ventricle and compress<strong>in</strong>g the midbra<strong>in</strong><br />
tectum) (Haene, 1993; Morrow, 1990). The rare association of SOM with bra<strong>in</strong>stem<br />
tectal disease has caused some authors to recommend neuroimag<strong>in</strong>g exam<strong>in</strong>ation of the<br />
course of the trochlear nerve <strong>in</strong> all patients with this diagnosis (Morrow, 1990); however,<br />
the association of SOM with a posterior fossa tumor is extremely uncommon (Brazis,<br />
1994) (class IV, level U). In one reported case, SOM may have been due to vascular<br />
compression of the trochlear nerve by a branch of the posterior cerebral artery noted on<br />
th<strong>in</strong>-slice MRI (Hashimoto, 2001). In another study of six patients with SOM, neurovascular<br />
contact at the root exit zone of the trochlear nerve was identified <strong>in</strong> all patients,<br />
suggest<strong>in</strong>g that SOM may be a neovascular compression syndrome (Yousry, 2002). SOM<br />
has been described <strong>in</strong> a patient with a dural arteriovenous fistula (Geis, 1996), and<br />
Neetens and Mart<strong>in</strong> described two cases of SOM, one associated with lead <strong>in</strong>toxication<br />
and the other with adrenoleukodystrophy (Neetens, 1983). Some of these associations<br />
may well have been co<strong>in</strong>cidental (class IV). We do not recommend neuroimag<strong>in</strong>g for<br />
typical isolated SOM but consider MRI scan <strong>in</strong> patients with atypical features (class IV,<br />
level C).<br />
Rosenberg and Glaser obta<strong>in</strong>ed from 1 to 19 years (average 8 years) of follow-up for<br />
n<strong>in</strong>e patients with SOM (Rosenberg, 1983). These authors noted that the natural history<br />
of the disorder is one of spontaneous remissions and exacerbations, with untreated<br />
patients frequently enjoy<strong>in</strong>g months or even years of remission before subsequent<br />
relapses. Indeed, seven of their n<strong>in</strong>e patients cont<strong>in</strong>ued to have some symptoms after<br />
prolonged follow-up.<br />
The treatment for a majority of patients with SOM is reassurance, because most are<br />
not significantly disabled by their visual symptoms. If the condition disrupts the<br />
patient’s work and lifestyle, medications such as carbamazep<strong>in</strong>e or propranolol<br />
(Tyler, 1990) may be considered. In Rosenberg and Glaser’s series, 7 of 11 patients<br />
were tried on carbamazep<strong>in</strong>e, and 6 noted a prompt decrease or cessation of ocular<br />
symptoms (Rosenberg, 1983). All experienced at least one subsequent relapse days to<br />
months after the <strong>in</strong>itial improvement, however, and only three chose to cont<strong>in</strong>ue the<br />
medication. We have tried gabapent<strong>in</strong> <strong>in</strong> one patient with SOM without subjective or<br />
objective improvement.<br />
Brazis et al <strong>in</strong>vestigated the cl<strong>in</strong>ical presentations and long-term course of 16 patients<br />
with SOM (Brazis, 1994). Follow-up <strong>in</strong>formation was obta<strong>in</strong>ed for 14 of the 16 patients<br />
with time from onset of symptoms to most recent contact 3 to 29 years (mean, 12 years).<br />
The SOM gradually improved or resolved, at least temporarily, without treatment <strong>in</strong> a
372 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
significant number of patients. Three of the patients had complete spontaneous<br />
resolution of symptoms for periods of 6 to 12 months without recurrence. Six of 7<br />
patients treated with carbamazep<strong>in</strong>e reported no significant response. One patient<br />
rema<strong>in</strong>ed on the medication for 3 years with only rare symptoms that worsen when<br />
attempts were made to taper the drug. Two of the patients treated with propranolol<br />
reported no significant benefit, and a third noted dramatic but transient improvement <strong>in</strong><br />
symptoms. Four of the patients were cured by superior oblique tenectomy comb<strong>in</strong>ed<br />
with <strong>in</strong>ferior oblique myectomy. All four surgical patients experienced disappearance of<br />
oscillopsia, although one patient developed postoperative vertical diplopia that gradually<br />
resolved. Brazis et al concluded that because SOM is a much more chronic disease<br />
than formerly realized and because of the poor long-term effects and potential side<br />
effects of the medications used for treatment, medical treatment of SOM, at least with<br />
currently available medications, may not be the optimum way to manage the disease.<br />
They believe that surgery is the treatment of choice when symptoms of SOM are<br />
<strong>in</strong>tolerable to the patient. Other authors have also reported successful treatment of SOM<br />
with surgery (de Sa, 1992; Hayakawa, 2000). For example, Kosmorsky et al performed a<br />
Harada-Ito procedure on a woman with SOM (Kosmorsky, 1995). This procedure<br />
<strong>in</strong>volves nasally transpos<strong>in</strong>g the anterior portion of the superior oblique tendon,<br />
which is responsible for the cyclorotation, to create an effective weaken<strong>in</strong>g of the<br />
anterior portion of the tendon <strong>in</strong>stead of temporal displacement utilized for superior<br />
oblique paresis. The SOM was abolished and vertical eye movements, <strong>in</strong>clud<strong>in</strong>g<br />
saccades, were unaffected. Samil et al reported one patient with SOM who responded<br />
to microvascular decompression of the fourth nerve at the root exit zone (Samil, 1998).<br />
The treatment of SOM is usually reassurance and the condition may be self-limit<strong>in</strong>g.<br />
When symptoms are <strong>in</strong>tolerable, medical or surgical therapy may be considered. A<br />
weaken<strong>in</strong>g procedure of the affected superior oblique muscle comb<strong>in</strong>ed with a weaken<strong>in</strong>g<br />
procedure of the ipsilateral <strong>in</strong>ferior oblique muscle or the Harada-Ito procedure is<br />
an effective treatment for SOM after failure of medical treatment or as an alternative to<br />
such treatment, and should be considered <strong>in</strong> patients with unacceptable visual symptoms.<br />
Microvascular decompression of the fourth cranial nerve at the root exit zone<br />
may be another approach, but so far there has been little experience with this procedure<br />
for SOM.<br />
The evaluation of monocular or asymmetric b<strong>in</strong>ocular oscillations is outl<strong>in</strong>ed <strong>in</strong><br />
Figure 17–2.<br />
What Are the Causes of Dysconjugate<br />
Bilateral Symmetric Eye Oscillations?<br />
If the ocular oscillations <strong>in</strong>volve both eyes to a relatively equal degree, the next step <strong>in</strong><br />
evaluation <strong>in</strong>volves determ<strong>in</strong><strong>in</strong>g whether the eye movements are disconjugate (the eyes<br />
mov<strong>in</strong>g <strong>in</strong> opposite directions) or conjugate (both eyes mov<strong>in</strong>g <strong>in</strong> the same direction)<br />
(Burde, 1991). When the oscillations are disconjugate, the exam<strong>in</strong>er should determ<strong>in</strong>e<br />
whether the oscillations are vertical or horizontal. Vertical disconjugate eye oscillations<br />
are usually due to seesaw nystagmus. Horizontal disconjugate eye oscillations <strong>in</strong>clude<br />
convergence-retraction nystagmus (nystagmus retractorius), divergence nystagmus,<br />
repetitive divergence, and oculomasticatory myorhythmia.
Figure 17–2. Evaluation of monocular or asymmetric oscillations.<br />
What Are the <strong>Cl<strong>in</strong>ical</strong> Features and Etiologies<br />
of Seesaw Nystagmus?<br />
Nystagmus and Other Ocular Oscillations 373<br />
Seesaw nystagmus is a cyclic movement of the eyes with a conjugate torsional<br />
component and a dysjunctive vertical component. While one eye rises and <strong>in</strong>torts, the<br />
other falls and extorts; the vertical and torsional movements are then reversed,<br />
complet<strong>in</strong>g the cycle. This nystagmus is usually pendular and may be due to a large<br />
suprasellar lesion compress<strong>in</strong>g or <strong>in</strong>vad<strong>in</strong>g the bra<strong>in</strong>stem bilaterally at the mesodien-
374 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
cephalic junction. Pendular seesaw nystagmus may also be congenital (May, 1997).<br />
Seesaw nystagmus may also have a underly<strong>in</strong>g jerk waveform, often due to a <strong>in</strong>tr<strong>in</strong>sic<br />
focal bra<strong>in</strong>stem lesion, either <strong>in</strong> the lateral medulla (usually on the side opposite the<br />
torsional quick phases) or <strong>in</strong> the mesodiencephalon on the same side as the quick phases<br />
(Halmagyi, 1991, 1994). Jerk seesaw nystagmus has a slow phase correspond<strong>in</strong>g to one<br />
half-cycle of seesaw nystagmus and is thus often called hemi-seesaw nystagmus.<br />
Seesaw nystagmus likely represents oscillations <strong>in</strong>volv<strong>in</strong>g central otolithic connections,<br />
especially the <strong>in</strong>terstitial nucleus of Cajal (Halmagyi, 1991). Seesaw nystagmus<br />
may also be <strong>in</strong> part due to an unstable visuovestibular <strong>in</strong>teraction control system.<br />
Lesions <strong>in</strong> the optic pathways may prevent ret<strong>in</strong>al error signals, essential for vestibuloocular<br />
reflex adaptation, from reach<strong>in</strong>g the cerebellar flocculus and <strong>in</strong>ferior olivary<br />
nucleus, thereby mak<strong>in</strong>g the system less stable. Etiologies responsible for seesaw<br />
nystagmus are outl<strong>in</strong>ed <strong>in</strong> Table 17–1.<br />
Chiari malformation type I may be associated with nystagmus of skew <strong>in</strong> which one<br />
eye beats upward while the other eye beats downward (Pieh, 2000). The evaluation of a<br />
patient with seesaw nystagmus <strong>in</strong>cludes a complete ophthalmologic and neurologic<br />
exam<strong>in</strong>ation. Patients with parasellar lesions often have bitemporal field defects and<br />
‘‘bow-tie’’ optic atrophy associated with pendular seesaw nystagmus. Jerk seesaw<br />
nystagmus usually is associated with other bra<strong>in</strong>stem signs. We recommend neuroimag<strong>in</strong>g<br />
(preferably MRI attend<strong>in</strong>g to parasellar and posterior fossa regions) for patients<br />
with seesaw nystagmus, with particular attention to the third ventricle=<br />
parasellar area (class IV, level C). The presence of this nystagmus with a skew deviation<br />
requires MRI studies for a Chiari malformation (class IV, level C). The treatment of<br />
seesaw nystagmus is directed at the responsible lesion. One patient with <strong>in</strong>termittent<br />
seesaw nystagmus responded to clonazepam, and the nystagmus did not recur after<br />
withdrawal of the medication (Coch<strong>in</strong>, 1995). Also, baclofen, with and without<br />
clonazepam, improved both nystagmus and associated oscillopsia <strong>in</strong> another patient,<br />
suggest<strong>in</strong>g a possible g-am<strong>in</strong>obutyric acid (GABA)-ergic mechanism <strong>in</strong>fluenc<strong>in</strong>g the<br />
<strong>in</strong>terstitial nucleus of Cajal.<br />
Table 17–1. Etiologies of Seesaw Nystagmus<br />
Parasellar masses (Barton, 1995)<br />
Bra<strong>in</strong>stem and thalamic stroke (Halmagyi, 1991, 1994)<br />
Multiple sclerosis (Samkoff, 1994)<br />
Trauma<br />
Chiari malformation<br />
Hydrocephalus<br />
Syr<strong>in</strong>gobulbia<br />
Paraneoplastic encephalitis (with testicular cancer and anti-Ta antibodies) (Bennett, 1999)<br />
Whole bra<strong>in</strong> irradiation and <strong>in</strong>trathecal methotrexate (Epste<strong>in</strong>, 2001)<br />
Septo-optic dysplasia, ret<strong>in</strong>itis pigmentosa, and cone degeneration (May, 1997)<br />
Congenital seesaw nystagmus* (Rambold, 1998)<br />
*Congenital seesaw nystagmus may lack the torsional component or even present with an opposite<br />
pattern, that is, extorsion with eye elevation and <strong>in</strong>torsion with eye depression. With congenital cases,<br />
the b<strong>in</strong>ocular torsional eye movements may be <strong>in</strong> phase with cl<strong>in</strong>ically visible head oscillations (i.e.,<br />
head movements are not compensatory for the torsional eye movements) (Rambold, 1998).
What Are the Causes of Horizontal<br />
Dysconjugate Eye Oscillations?<br />
Nystagmus and Other Ocular Oscillations 375<br />
Convergence may evoke various forms of nystagmus (i.e., convergence-evoked nystagmus;<br />
see below). Convergence-retraction nystagmus is a disorder of ocular motility <strong>in</strong><br />
which repetitive adduct<strong>in</strong>g saccades, which are often accompanied by retraction of the<br />
eyes <strong>in</strong>to the orbit, occur spontaneously or on attempted upgaze (Pullic<strong>in</strong>o, 2000).<br />
Rotat<strong>in</strong>g an optok<strong>in</strong>etic tape or drum downward may elicit the movements.<br />
Convergence-retraction nystagmus is primarily a saccadic disorder as the convergence<br />
movements are not normal vergence movements but asynchronous, adduct<strong>in</strong>g<br />
saccades. Other authors feel that convergence-retraction nystagmus is a disorder of<br />
vergence rather than of oppos<strong>in</strong>g adduct<strong>in</strong>g saccades (Rambold, 2001b). Mesencephalic<br />
lesions affect<strong>in</strong>g the pretectal region are most likely to cause this type of nystagmus,<br />
which is often associated with abnormalities of vertical gaze. The localization and<br />
evaluation of these vertical gaze abnormalities and convergence-retraction nystagmus<br />
are discussed <strong>in</strong> Chapter 14. Convergence nystagmus has been described without<br />
vertical gaze abnormalities <strong>in</strong> patients with dorsal midbra<strong>in</strong> stroke and <strong>in</strong> patients<br />
with Chiari malformation (Mossman, 1990; Schnyder, 1996). Whipple’s disease may also<br />
cause convergence nystagmus at approximately 1 Hz (pendular vergence oscillations)<br />
(Selhorst, 1987). Convergence nystagmus has been described <strong>in</strong> a patient with spasmus<br />
nutans (Massry, 1996).<br />
Divergence nystagmus (with divergent quick phases) may occur with h<strong>in</strong>dbra<strong>in</strong><br />
abnormalities (e.g., Chiari malformation) and is associated with downbeat nystagmus.<br />
These patients have slow phases directed upward and <strong>in</strong>ward. Repetitive divergence<br />
consists of a slow divergent movement followed by a rapid return to the primary<br />
position at regular <strong>in</strong>tervals (Noda, 1987). This rare disorder has been described with<br />
coma from hepatic encephalopathy. A similar disorder, probably related to seizures, was<br />
reported <strong>in</strong> a neonate <strong>in</strong> association with burst-suppression patterns of the electroencephalogram<br />
(Nelson, 1986).<br />
Oculomasticatory myorhythmia refers to acquired pendular vergence oscillations<br />
associated with concurrent contraction of the masticatory muscles (Adler, 1990; Louis,<br />
1996). If nonfacial skeletal muscles are <strong>in</strong>volved, it is called oculofacial-skeletal myorhythmia.<br />
There is a smooth, rhythmic eye convergence, which cycles at a frequency of<br />
approximately 1 Hz, followed by divergence back to the primary position. Rhythmic<br />
elevation and depression of the mandible is synchronous with the ocular oscillations<br />
that persist <strong>in</strong> sleep and are unaltered by stimuli. The masticatory <strong>in</strong>volvement may<br />
occasionally consist of a permanent bruxism lead<strong>in</strong>g to severe tooth abrasions (Tison,<br />
1992). Patients with oculomasticatory myorhythmia may also have paralysis of vertical<br />
gaze, progressive somnolence, and <strong>in</strong>tellectual deterioration. This dist<strong>in</strong>ct movement<br />
disorder has been recognized only <strong>in</strong> Whipple’s disease (class III–IV, level B).<br />
Therefore, if this condition is diagnosed, empiric antibiotic treatment should be<br />
considered and tissue diagnosis should be attempted. Whipple’s disease may be<br />
diagnosed by endoscopically guided biopsy of multiple jejunal sites (Adler, 1990;<br />
Louis, 1996). Electron microscopy and polymerase cha<strong>in</strong> reaction (PCR)-based test<strong>in</strong>g<br />
on <strong>in</strong>test<strong>in</strong>al or extra<strong>in</strong>test<strong>in</strong>al tissue may also confirm the diagnosis (Louis, 1996; Tison,<br />
1992). PCR can also be performed on cerebrosp<strong>in</strong>al fluid <strong>in</strong> central nervous system<br />
(CNS) Whipple’s disease (Lynch, 1997; Von Herbay, 1997).<br />
The evaluation of disconjugate bilateral symmetric eye oscillations is outl<strong>in</strong>ed <strong>in</strong><br />
Figure 17–3.
376 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 17–3. Evaluation of disconjugate bilateral symmetric oscillations.<br />
What Are the Causes of B<strong>in</strong>ocular Symmetric<br />
Conjugate Eye Oscillations?<br />
B<strong>in</strong>ocular symmetric conjugate eye oscillations may be divided <strong>in</strong>to pendular nystagmus,<br />
jerk nystagmus, and saccadic <strong>in</strong>trusions (Burde, 1991).
What Are the Causes of B<strong>in</strong>ocular Symmetric<br />
Pendular Conjugate Eye Oscillations?<br />
Nystagmus and Other Ocular Oscillations 377<br />
B<strong>in</strong>ocular symmetric pendular conjugate eye oscillations may be due to congenital<br />
nystagmus, pendular nystagmus, oculopalatal myoclonus, spasmus nutans (discussed<br />
above), and visual deprivation nystagmus.<br />
Congenital nystagmus may be noted at birth or <strong>in</strong> early <strong>in</strong>fancy, or may emerge or<br />
enhance <strong>in</strong> teenage or adult life (Dell’Osso, 1993; Gresty, 1991; Hertle, 1999). It may be<br />
familial, hereditary (X-l<strong>in</strong>ked, autosomal dom<strong>in</strong>ant, autosomal recessive) (Kerrison,<br />
1999; Oett<strong>in</strong>g, 2000) or idiopathic. Metabolic derangements and structural anomalies of<br />
the bra<strong>in</strong>, <strong>in</strong>clud<strong>in</strong>g abnormalities of the anterior and posterior visual pathways, may be<br />
responsible (Jacobson, 1998). More important, when it is found later <strong>in</strong> life it must be<br />
dist<strong>in</strong>guished from other forms of nystagmus that have a potentially treatable cause.<br />
Congenital nystagmus may be wholly pendular or have both pendular and jerk<br />
components. Congenital jerk nystagmus has a slow phase with a velocity that <strong>in</strong>creases<br />
exponentially as the eyes move <strong>in</strong> the direction of the slow phase. Occasionally<br />
congenital nystagmus may be purely vertical or torsional, and although these f<strong>in</strong>d<strong>in</strong>gs<br />
usually implicate an <strong>in</strong>tracranial lesion, these forms of nystagmus may occur <strong>in</strong> sensory<br />
congenital nystagmus (Shawkat, 2000). Although irregular, congenital nystagmus is<br />
generally conjugate and horizontal, even on upgaze or downgaze (uniplanar), visual<br />
fixation accentuates it and active eyelid closure or convergence attenuates it (Gresty,<br />
1991). The nystagmus decreases <strong>in</strong> an eye position (‘‘null region’’) that is specific for<br />
each patient. Despite the constant eye motion, these patients do not experience<br />
oscillopsia. When they are tested with a hand-held optok<strong>in</strong>etic tape or drum, the<br />
quick phase of the elicited nystagmus generally follows the direction of the tape<br />
(reversed optok<strong>in</strong>etic nystagmus).<br />
Symptomatic oscillopsia <strong>in</strong> patients with congenital nystagmus is unusual but may be<br />
precipitated after visual maturation by new or chang<strong>in</strong>g associated visual sensory<br />
conditions (e.g., decompensat<strong>in</strong>g strabismus or ret<strong>in</strong>al degeneration) (Hertle, 2001).<br />
Congenital nystagmus has been associated with many disease processes affect<strong>in</strong>g the<br />
visual afferent system <strong>in</strong>clud<strong>in</strong>g ocular and oculocutaneous alb<strong>in</strong>ism, achromatopsia,<br />
optic nerve hypoplasia, Leber’s amaurosis, coloboma, aniridia, cone dystrophies,<br />
corectopia, congenital stationary night bl<strong>in</strong>dness, Chédiak-Higashi syndrome, Joubert<br />
syndrome, and peroxisomal disorders. It has also been associated with hypothyroidism.<br />
The evaluation of children with congenital nystagmus thus should <strong>in</strong>clude a complete<br />
ophthalmologic exam<strong>in</strong>ation, especially attend<strong>in</strong>g to symptoms of photophobia and<br />
paradoxical pupillary constriction <strong>in</strong> darkness, and thyroid functions. <strong>An</strong> electroret<strong>in</strong>ogram<br />
(ERG) may be helpful even with a normal afferent exam (Cibis, 1993). For<br />
example, 56% of 105 consecutive patients with congenital nystagmus were found to<br />
have ret<strong>in</strong>al disease when tested with ERG (Cibis, 1993).<br />
Congenital nystagmus often decreases <strong>in</strong> an eye position (‘‘null region’’) that is<br />
specific for each patient, and convergence often attenuates the nystagmus. Prisms can be<br />
used to take advantage of the dampen<strong>in</strong>g effect of convergence and the null region–lens<br />
comb<strong>in</strong>ations can be adjusted so that an asymmetric arrangement of base-out prisms<br />
both converge the eyes and turn them toward the null angle. Leigh et al suggest<br />
7.00-diopter base-out prisms with 1.00-diopter spheres added to compensate for<br />
accommodation (Leigh, 1994). Contact lenses may improve vision <strong>in</strong> patients with
378 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
congenital nystagmus, possibly due to tactile feedback. <strong>An</strong>other approach for the<br />
treatment of severe nystagmus <strong>in</strong> general <strong>in</strong>volves employ<strong>in</strong>g an optical system to<br />
stabilize images on the ret<strong>in</strong>a (Leigh, 1994; Yaniglos, 1992). The comb<strong>in</strong>ation of high<br />
‘‘plus’’ (i.e., converg<strong>in</strong>g) spectacle lenses with high ‘‘m<strong>in</strong>us’’ (i.e., diverg<strong>in</strong>g) contact<br />
lenses is used with the converg<strong>in</strong>g system focus<strong>in</strong>g the image at the center of eye<br />
rotation (thus, stabiliz<strong>in</strong>g the image) and the diverg<strong>in</strong>g system mov<strong>in</strong>g the image back<br />
to focus on the ret<strong>in</strong>a. The contact lens moves with the eye so it does not negate the<br />
effect of image stabilization produced by the spectacle lens. This imag<strong>in</strong>g system is<br />
theoretically beneficial but difficult to ma<strong>in</strong>ta<strong>in</strong> <strong>in</strong> practice, especially as the system<br />
disables the vestibulo-ocular reflex and is thus only useful when the patient is<br />
stationary.<br />
Congenital nystagmus may also be treated with botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jections <strong>in</strong>to the<br />
extraocular muscles or surgery. Acuity was restored <strong>in</strong> four patients, to the extent that<br />
they were able to receive daytime drivers licenses, by multiple horizontal recti <strong>in</strong>jections<br />
of botul<strong>in</strong>um tox<strong>in</strong> (Carruthers, 1995). Surgical procedures effectively control congenital<br />
nystagmus by attempt<strong>in</strong>g to move the attachments of the extraocular muscles so that<br />
the null angle corresponds to the new primary position (the null region is shifted and<br />
broadened), to decrease nystagmus outside the null region, and to prolong foveation<br />
time by chang<strong>in</strong>g the waveform and dampen<strong>in</strong>g the nystagmus (Atilla, 1999; Bilska,<br />
1995; Helveston, 1991; von Noorden, 1991; Zubkov, 1993). Procedures used <strong>in</strong>clude the<br />
<strong>An</strong>derson-Kestenbaum procedure, which moves the eyes to the null region, divergence<br />
procedures, large recessions of the horizontal rectus muscles, and comb<strong>in</strong>ed procedures<br />
(Lee, 2000; Leigh, 1994). F<strong>in</strong>ally, biofeedback has been reported to help some patients<br />
with this disorder. Evans et al performed a randomized, double-masked, placebocontrolled<br />
trial of various treatments for congenital nystagmus and concluded that these<br />
putative therapies should be assumed to be placebos until proven otherwise by<br />
randomized trial (class III–IV, level C) (Evans, 1998).<br />
Latent nystagmus is common and generally congenital (Gresty, 1992; Wagner, 1990;<br />
Zubkov, 1990). It appears when one eye is covered. Both eyes then develop conjugate<br />
jerk nystagmus, with the view<strong>in</strong>g eye hav<strong>in</strong>g a slow phase directed toward the nose<br />
(i.e., the quick phase of both eyes beat toward the side of the fixat<strong>in</strong>g eye). Although<br />
present at birth, latent nystagmus is often not recognized until later <strong>in</strong> life, when an<br />
attempt is made to determ<strong>in</strong>e monocular visual acuity dur<strong>in</strong>g vision screen<strong>in</strong>g at<br />
school. Latent nystagmus is usually associated with strabismus, especially esotropia;<br />
amblyopia may occur and b<strong>in</strong>ocular vision with normal stereopsis is rare. In addition to<br />
horizontal strabismus, upward deviation of the covered eye (dissociated vertical<br />
deviation or alternat<strong>in</strong>g sursumduction) and a torsional, occasionally pendular, component<br />
to the nystagmus may occur. Latent nystagmus is a marker for congenital<br />
ocular motor disturbance and does not <strong>in</strong>dicate progressive structural bra<strong>in</strong> disease<br />
(Burde, 1991).<br />
Manifest latent nystagmus is an oscillation that occurs <strong>in</strong> patients with strabismus or<br />
acquired visual loss who have a jerk nystagmus <strong>in</strong> the direction of the fix<strong>in</strong>g eye<br />
(i.e., right-beat<strong>in</strong>g nystagmus when fix<strong>in</strong>g with the right eye and left-beat<strong>in</strong>g nystagmus<br />
when fix<strong>in</strong>g with the left eye) (Burde, 1991). Patients with <strong>in</strong>fantile uniocular bl<strong>in</strong>dness<br />
may have a bilateral horizontal nystagmus that represents a manifest nystagmus of the<br />
latent type (Kushner, 1995). These patients often have a family history of strabismus; the<br />
monocular bl<strong>in</strong>dness (opacity of the media or suppression) acts as an occluder, mak<strong>in</strong>g<br />
manifest what would have been latent nystagmus. Therapy for latent nystagmus
Nystagmus and Other Ocular Oscillations 379<br />
consists of measures to improve vision, such as patch<strong>in</strong>g for amblyopia <strong>in</strong> children or<br />
surgical correction of strabismus (Zubkov, 1993).<br />
Voluntary nystagmus (psychogenic flutter) occurs <strong>in</strong> normal subjects, sometimes as a<br />
familial trait, and consists of bursts of high-frequency horizontal oscillations composed<br />
of back-to-back saccades (Lee, 1993; Sato, 1999). The movements may be vertical or<br />
torsional as well. This movement will completely disappear if patients are forced to<br />
keep their eyes open, because it requires tremendous volitional effort and cannot be<br />
susta<strong>in</strong>ed for prolonged periods of time (Burde, 1991). Voluntary nystagmus is often<br />
accompanied by a ‘‘fixed look’’ required to produce the symptoms, eyelid flutter,<br />
and convergence. Voluntary nystagmus may be associated with spasm of the near<br />
reflex (Sato, 1999) and has been described as a component of nonepileptic seizures<br />
(Davis, 2000).<br />
Although pendular nystagmus is often congenital, acquired forms exist. Acquired<br />
pendular nystagmus may be wholly horizontal, wholly vertical, or have mixed<br />
components (circular, elliptical, or w<strong>in</strong>dmill pendular nystagmus). Pendular nystagmus<br />
may be symmetric, dissociated, or even monocular and often causes distress<strong>in</strong>g<br />
oscillopsia and decreased visual acuity (Averbuch-Heller, 1995b; Barton, 1993; Lopez,<br />
1996). Damage to the dentatorubro-olivary pathways (Guilla<strong>in</strong>-Mollaret triangle) is<br />
found <strong>in</strong> some cases of acquired pendular nystagmus, which is most often caused by<br />
multiple sclerosis, stroke, or tumor of the bra<strong>in</strong>stem or other posterior fossa structures<br />
(Averbuch-Heller, 1995b; Barton, 1993, 1999; Lopez, 1996; Revol, 1990; Schon, 1999;<br />
Starck, 1997; Talks, 1997). In multiple sclerosis, pendular nystagmus may be a sign of<br />
cerebellar nuclear <strong>in</strong>volvement or result from optic neuropathy, but the most consistent<br />
f<strong>in</strong>d<strong>in</strong>g on MRI is a lesion <strong>in</strong> the dorsal pont<strong>in</strong>e tegmentum, perhaps affect<strong>in</strong>g the<br />
central tegmental tract (Barton, 1993). In a study of 27 patients with acquired pendular<br />
nystagmus, MRI f<strong>in</strong>d<strong>in</strong>gs were characterized by multiple areas of abnormal signal with<br />
statistically significant ones occurr<strong>in</strong>g <strong>in</strong> areas conta<strong>in</strong><strong>in</strong>g the red nucleus, the central<br />
tegmental tract, the medial vestibular nucleus, and the <strong>in</strong>ferior olive (Lopez, 1996).<br />
The abundance of abnormal MRI signals, predom<strong>in</strong>antly <strong>in</strong> the pons but also <strong>in</strong> the<br />
midbra<strong>in</strong> and the medulla, suggests that large or multiple structural lesions may be<br />
required to elicit pendular nystagmus. Acquired convergence-<strong>in</strong>duced pendular<br />
nystagmus may occur with multiple sclerosis (Barton, 1999) and we recommend<br />
neuroimag<strong>in</strong>g (e.g., cranial MRI) for all unexpla<strong>in</strong>ed cases of acquired pendular<br />
nystagmus (class III–IV, level B).<br />
Other causes of acquired b<strong>in</strong>ocular pendular nystagmus <strong>in</strong>clude Pelizaeus-<br />
Merzbacher disease, mitochondrial cytopathy, Cockayne’s syndrome, neonatal adrenoleukodystrophy<br />
(a peroxisomal disorder), and toluene addiction (Kori, 1998; Maas,<br />
1991; Trobe, 1991). Spontaneous horizontal pendular nystagmus <strong>in</strong> a patient with a<br />
surgically acquired perilymph fistula was found related to the heart rate and may have<br />
been caused by pressure transfer of blood pulses to the labyr<strong>in</strong>th (Rambold, 2001a).<br />
Congenital, familial, or acquired bilateral paralysis of horizontal gaze may be associated<br />
with pendular nystagmus; the familial type may also be associated with progressive<br />
scoliosis and facial contractures with myokymia. Pendular nystagmus may also appear<br />
with bl<strong>in</strong>dness or monocular loss of vision; <strong>in</strong> the latter case, it may be monocular (see<br />
above). B<strong>in</strong>ocular visual loss may cause nystagmus that has both horizontal and vertical<br />
components that change direction over seconds or m<strong>in</strong>utes (i.e., a wander<strong>in</strong>g null po<strong>in</strong>t)<br />
(Good, 1990). Bl<strong>in</strong>d patients may have w<strong>in</strong>dmill nystagmus, <strong>in</strong> which there are repeated
380 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
oscillations <strong>in</strong> the vertical plane alternat<strong>in</strong>g with repeated oscillations <strong>in</strong> the horizontal<br />
plane.<br />
Horizontal pendular pseudonystagmus has been described <strong>in</strong> patients with horizontal<br />
essential head tremor and bilateral vestibular dysfunction (Bronste<strong>in</strong>, 1992;<br />
Verhagen, 1994). The deficient vestibulo-ocular reflex results <strong>in</strong> ocular oscillations <strong>in</strong><br />
space when the head oscillates, and funduscopy reveals a f<strong>in</strong>e pendular motion of the<br />
eyes that is reduced by firm support of the head. The oscillopsia improves with<br />
treatment of the tremor with propranolol. Yen et al described two renal transplant<br />
patients who developed pseudonystagmus and oscillopsia caused by immunosuppressant<br />
(tacrolimus)-<strong>in</strong>duced head tremor and gentamic<strong>in</strong>-<strong>in</strong>duced vestibulopathy<br />
(Yen, 1999). Although the patients were <strong>in</strong>itially thought to have nystagmus, closer<br />
observation revealed no true nystagmus but corrective saccades compensat<strong>in</strong>g for an<br />
absent vestibulo-ocular reflex dur<strong>in</strong>g the head tremor (pseudonystagmus). Typically<br />
patients with vestibulo-ocular impairment have only head movement–<strong>in</strong>duced oscillopsia,<br />
but these patients had constant oscillopsia because the visual track<strong>in</strong>g system<br />
(smooth pursuit) could not compensate for the loss of vestibular function at immunosuppressant-<strong>in</strong>duced<br />
head oscillation greater than 1 Hz. Vestibular rehabilitation helped<br />
one of these patients.<br />
Palatal myoclonus is a cont<strong>in</strong>uous rhythmic <strong>in</strong>voluntary movement of the soft palate<br />
that may be accompanied by synchronous movements of other adjacent structures, such<br />
as the face, pharynx, larynx, or diaphragm. The association of pendular nystagmus with<br />
palatal myoclonus is not <strong>in</strong>frequent, and the condition is then termed oculopalatal<br />
myoclonus or oculopalatal tremor (Eggenberger, 2001; Talks, 1997). Oculopalatal<br />
myoclonus may be of two types (Nakada, 1986):<br />
1. A lateral form, consist<strong>in</strong>g of jerky, nystagmoid movements with simultaneous<br />
oblique and rotatory components associated (and synchronous) with lateralized<br />
palatal myoclonus (<strong>in</strong> this form, the eye on the side of the myoclonus <strong>in</strong>torts as it<br />
rises and extorts as it falls, whereas the opposite eye extorts as it rises and <strong>in</strong>torts as it<br />
falls).<br />
2. A midl<strong>in</strong>e form <strong>in</strong> which vertical to-and-fro pendular eye movements occur<br />
synchronous with symmetric bilateral palatal myoclonus.<br />
Oculopalatal myoclonus <strong>in</strong>volves vestibulo-ocular reflex adaption mediated by the<br />
cerebellar flocculus, and floccular <strong>in</strong>tegrity is preserved <strong>in</strong> most patients (Nakada, 1986).<br />
The lateral form implies unilateral disease, whereas the midl<strong>in</strong>e form <strong>in</strong>dicates bilateral<br />
disease. Damage to the dentatorubro-olivary pathways (Guilla<strong>in</strong>-Mollaret triangle) is<br />
found <strong>in</strong> cases of oculopalatal myoclonus, which is most often caused by multiple<br />
sclerosis or vascular lesions of the bra<strong>in</strong>stem. MRI often shows enlargement of the<br />
<strong>in</strong>ferior olivary nuclei (Talks, 1997).<br />
There may be an association between the one-and-a-half syndrome (see Chapter 14)<br />
and oculopalatal myoclonus (Wol<strong>in</strong>, 1996). In five patients with one-and-a-half<br />
syndrome and facial nerve palsy, oculopalatal myoclonus developed <strong>in</strong> 4 months to 3<br />
years. Involvement of the facial nerve may predict subsequent development of<br />
oculopalatal myoclonus. Oculopalatal tremor may be associated with delayed (tardive)<br />
ataxia (Eggenberger, 2001).<br />
The evaluation of the patient with pendular nystagmus depends on the cl<strong>in</strong>ical<br />
circumstances and associated neurologic f<strong>in</strong>d<strong>in</strong>gs. In patients with multiple sclerosis,
Nystagmus and Other Ocular Oscillations 381<br />
the diagnosis is usually obvious by a history of remissions and exacerbations of<br />
neurologic signs and symptoms associated with abnormalities on neurologic exam<strong>in</strong>ation,<br />
suggest<strong>in</strong>g a dissem<strong>in</strong>ated process. Bra<strong>in</strong>stem stroke or tumor is diagnosed by<br />
mode of onset of symptoms, associated neurologic signs and symptoms, and MRI.<br />
Ophthalmologic exam will reveal bl<strong>in</strong>dness as a cause for the nystagmus <strong>in</strong> some<br />
patients. MRI is warranted <strong>in</strong> all patients with palatal myoclonus (class III–IV, level B).<br />
The neurotransmitters <strong>in</strong>volved <strong>in</strong> pendular nystagmus are unknown, but chol<strong>in</strong>ergic<br />
and GABA-ergic pathways may be <strong>in</strong>volved. <strong>An</strong>tichol<strong>in</strong>ergic agents have produced<br />
variable treatment results (Barton, 1994; Leigh, 1991). In a randomized, double-bl<strong>in</strong>d<br />
study, trihexyphenidyl improved only one of five patients with pendular elliptical<br />
nystagmus. In another double-bl<strong>in</strong>d study, <strong>in</strong>travenous scopolam<strong>in</strong>e reduced nystagmus<br />
and improved vision <strong>in</strong> five patients (Barton, 1994; Jabbari, 1987). Isoniazid<br />
relieved nystagmus and oscillopsia <strong>in</strong> two of three patients with pendular elliptical<br />
nystagmus due to multiple sclerosis, but others have not found this drug to be helpful<br />
(Leigh, 1994; Traccis, 1990). Memant<strong>in</strong>e (a glutamate antagonist) caused complete<br />
cessation of nystagmus <strong>in</strong> 11 of 14 patients with acquired pendular nystagmus due to<br />
multiple sclerosis (Starck, 1997). These 11 responders had fixation pendular nystagmus<br />
(i.e., nystagmus <strong>in</strong>creased with fixation). A dramatic suppression of pendular nystagmus<br />
<strong>in</strong> a patient with multiple sclerosis was described after smok<strong>in</strong>g cannabis, but not<br />
by tak<strong>in</strong>g orally adm<strong>in</strong>istered capsules conta<strong>in</strong><strong>in</strong>g cannabis oil (Schon, 1999).<br />
Although the mechanism of action of gabapent<strong>in</strong> is unknown, Stahl et al have<br />
measured the effects of this agent on vision and eye movements <strong>in</strong> acquired pendular<br />
nystagmus <strong>in</strong> two patients with multiple sclerosis and one with bra<strong>in</strong>stem stroke (Stahl,<br />
1996). <strong>An</strong> oral dose of 600 mg produced improvement of vision due to changes <strong>in</strong> ocular<br />
oscillations <strong>in</strong> all three patients. The drug was well tolerated and was cont<strong>in</strong>ued at 900<br />
to 1500 mg daily <strong>in</strong> divided doses with long-term benefit. All the patients reported<br />
useful visual improvement that enabled them to read, watch television, and recognize<br />
faces. In other studies, gabapent<strong>in</strong> improved acquired pendular nystagmus <strong>in</strong> 10 of 15<br />
patients (Averbuch-Heller, 1997) and 3 of 8 patients (Band<strong>in</strong>i, 2001).<br />
Several reports have suggested that <strong>in</strong>jection of botul<strong>in</strong>um tox<strong>in</strong> either <strong>in</strong>to selected<br />
extraocular muscles or <strong>in</strong>to the retrobulbar space might be effective <strong>in</strong> the treatment of<br />
acquired nystagmus (Carruthers, 1995; Leigh, 1992; Repka, 1994; Ruben, 1994a,b; Talks,<br />
1997; Tomsak, 1995). Leigh et al <strong>in</strong>jected the horizontal rectus muscles of the right eye of<br />
two patients with acquired pendular nystagmus (Leigh, 1992). The treatment effectively<br />
abolished the horizontal component of the nystagmus <strong>in</strong> the <strong>in</strong>jected eyes of both<br />
patients for 2 months. However, side effects <strong>in</strong>clud<strong>in</strong>g diplopia, ptosis, and worsen<strong>in</strong>g<br />
of the oscillopsia <strong>in</strong> the un<strong>in</strong>jected eye (attributed to plastic-adaptive changes <strong>in</strong><br />
response to paresis caused by the botul<strong>in</strong>um tox<strong>in</strong>) limited the effectiveness of the<br />
treatment. In another study, botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jection <strong>in</strong>to the retrobulbar space of three<br />
patients with acquired pendular nystagmus abolished or reduced all components of the<br />
nystagmus (Tomsak, 1995). Aga<strong>in</strong>, side effects of the treatment seem to be the limit<strong>in</strong>g<br />
factor. Others have reported variable improvement <strong>in</strong> visual function and oscillopsia<br />
with retrobulbar or horizontal recti botul<strong>in</strong>um <strong>in</strong>jection, with transient ptosis the most<br />
common side effect (Ruben, 1994a,b). Repka et al <strong>in</strong>jected 25 to 30 units of botul<strong>in</strong>um<br />
tox<strong>in</strong> <strong>in</strong>to the retrobulbar space of six adults with acquired nystagmus (Repka, 1994).<br />
Each patient had subjective and objective improvement of distance visual acuity<br />
follow<strong>in</strong>g <strong>in</strong>jection with reduction of the amplitude but not the frequency of<br />
the nystagmus. Visual improvement lasted no more than 8 weeks but persisted
382 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
for 6 months <strong>in</strong> two patients with oculopalatal myoclonus. The authors concluded that<br />
retrobulbar botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jection may improve visual function for patients with<br />
acquired nystagmus and that improvement seemed to be longer for patients<br />
with oculopalatal myoclonus. Further studies on the safety and efficacy of botul<strong>in</strong>um<br />
tox<strong>in</strong> <strong>in</strong>jection for acquired nystagmus are warranted (class IV, level U).<br />
Lesions of the Guilla<strong>in</strong>-Mollaret triangle are thought to <strong>in</strong>duce chol<strong>in</strong>ergic denervation<br />
supersensitivity of the <strong>in</strong>ferior olive, which results <strong>in</strong> the oculopalatal myoclonus.<br />
<strong>An</strong>tichol<strong>in</strong>ergic agents (trihexyphenidyl) have thus been tried effectively <strong>in</strong> four<br />
patients with palatal myoclonus without ocular <strong>in</strong>volvement (Jabbari, 1987) and <strong>in</strong><br />
one patient with vertical pendular nystagmus identical to that seen with oculopalatal<br />
myoclonus but without palatal <strong>in</strong>volvement (Herishanu, 1986). Valproate and carbamazep<strong>in</strong>e<br />
have each been reported to reduce the nystagmus of palatal myoclonus.<br />
F<strong>in</strong>ally, as noted above, the nystagmus <strong>in</strong> patients with oculopalatal myoclonus may be<br />
especially sensitive to retrobulbar botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jection (Repka, 1994).<br />
The evaluation and treatment of pendular nystagmus is outl<strong>in</strong>ed <strong>in</strong> Figure 17–4.<br />
What Are the Causes of B<strong>in</strong>ocular Symmetric<br />
Jerk Nystagmus?<br />
B<strong>in</strong>ocular symmetric conjugate jerk nystagmus may be divided <strong>in</strong>to that which is<br />
present spontaneously and that which is <strong>in</strong>duced (Burde, 1991). Spontaneous jerk<br />
nystagmus may be further divided <strong>in</strong>to forms present <strong>in</strong> primary position and forms<br />
present predom<strong>in</strong>antly on eccentric gaze.<br />
Spontaneous symmetric conjugate jerk nystagmus that occurs <strong>in</strong> primary position<br />
may be predom<strong>in</strong>antly horizontal, predom<strong>in</strong>antly torsional, or predom<strong>in</strong>antly vertical.<br />
Spontaneous symmetric conjugate jerk nystagmus <strong>in</strong> primary gaze that is predom<strong>in</strong>antly<br />
horizontal <strong>in</strong>cludes congenital nystagmus (above), latent nystagmus (above),<br />
vestibular nystagmus, periodic alternat<strong>in</strong>g nystagmus, drug-<strong>in</strong>duced nystagmus, and<br />
epileptic nystagmus. Spontaneous symmetric conjugate jerk nystagmus <strong>in</strong> primary gaze<br />
that is purely torsional is a form of central vestibular nystagmus. Spontaneous<br />
symmetric conjugate jerk nystagmus <strong>in</strong> primary gaze that is predom<strong>in</strong>antly vertical<br />
<strong>in</strong>cludes upbeat nystagmus and downbeat nystagmus.<br />
Horizontal nystagmus <strong>in</strong> the primary position is often the result of peripheral<br />
vestibular disease. Vestibular nystagmus has a l<strong>in</strong>ear (constant velocity) slow phase.<br />
The horizontal component is dim<strong>in</strong>ished when the patient lies with the <strong>in</strong>tact ear down<br />
and is exacerbated with the affected ear down. Peripheral vestibular lesions <strong>in</strong>duce a<br />
tendency for the eyes to drift <strong>in</strong> a direction parallel to the plane <strong>in</strong> which the diseased<br />
canal lies. Horizontal nystagmus with the slow component toward the lesion (the<br />
opposite vestibular nuclei drive the eyes toward the diseased side) results from<br />
unilateral horizontal canal or total labyr<strong>in</strong>th<strong>in</strong>e destruction. In the latter case there is<br />
a torsional slow component caus<strong>in</strong>g the upper part of the globe to rotate toward the<br />
lesioned side. Although constant for a particular position of gaze, the slow-phase<br />
velocity is greater when the eyes are turned <strong>in</strong> the direction of the quick component<br />
(Alexander’s law). Nystagmus due to peripheral vestibular disease is most prom<strong>in</strong>ent,<br />
or only becomes apparent, when fixation is prevented. Both peripheral and central<br />
vestibular nystagmus may vary with head position and movement, but peripheral
Figure 17–4. Evaluation of pendular nystagmus.<br />
Nystagmus and Other Ocular Oscillations 383<br />
nystagmus changes after a latency period follow<strong>in</strong>g the postural change and tends to<br />
fatigue.<br />
Peripheral vestibular disease is suspected when the nystagmus is associated with<br />
subjective vertigo. Central vestibular disease (e.g., bra<strong>in</strong>stem <strong>in</strong>farction) is suspected<br />
when associated neurologic signs and symptoms of bra<strong>in</strong>stem dysfunction are present.<br />
We recommend otolaryngologic consultation for peripheral vestibular disease and MRI<br />
for central vestibular disorders (class III–IV, level C).<br />
With periodic alternat<strong>in</strong>g nystagmus (PAN), the eyes exhibit primary position<br />
nystagmus, which, after 60 to 120 seconds, stops for a few seconds and then starts<br />
beat<strong>in</strong>g <strong>in</strong> the opposite direction (Shallo-Hoffmann, 1999). Horizontal jerk nystagmus <strong>in</strong><br />
the primary position not associated with vertigo is usually periodic alternat<strong>in</strong>g<br />
nystagmus (Burde, 1991). This disorder may be associated with periodic alternat<strong>in</strong>g<br />
oscillopsia, periodic alternat<strong>in</strong>g gaze, or periodic alternat<strong>in</strong>g skew deviation (Troost,<br />
1990). PAN may be congenital or acquired. It may be caused by craniocervical junction<br />
disease. The nodulus and uvula of the cerebellum ma<strong>in</strong>ta<strong>in</strong> <strong>in</strong>hibitory control over<br />
vestibular rotational responses by us<strong>in</strong>g GABA. Follow<strong>in</strong>g ablation of these structures,<br />
the postrotational response is excessively prolonged, so that normal vestibular repair
384 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
mechanisms act to reverse the direction of the nystagmus, which results <strong>in</strong> PAN (Leigh,<br />
1991). PAN is thus thought to be produced by dysfunction of the GABA-ergic velocitystorage<br />
mechanism and may be controlled <strong>in</strong> most, but not all, patients by the GABA-B<br />
agonist baclofen (Furman, 1990b; Troost, 1990). Patients with congenital PAN may also<br />
respond to baclofen or benefit from horizontal recti recessions (Gradste<strong>in</strong>, 1997).<br />
Etiologies for periodic alternat<strong>in</strong>g nystagmus are listed <strong>in</strong> Table 17–2.<br />
The evaluation of a patient with PAN <strong>in</strong>cludes a complete neurologic and ophthalmologic<br />
exam. In many patients, the etiology of the nystagmus is evident by history<br />
(e.g., congenital onset, associated alb<strong>in</strong>ism, family history of cerebellar degeneration,<br />
anticonvulsant use, history of remissions and exacerbations of neurologic signs and<br />
symptoms, acute onset of bra<strong>in</strong>stem signs and symptoms, severe visual impairment,<br />
etc.). Otherwise, the evaluation should <strong>in</strong>clude MRI with attention to the craniocervical<br />
junction (class III–IV, level B). If MRI is normal and the patient has a history of the<br />
subacute onset of progressive cerebellar signs and symptoms, Jakob-Creutzfeldt disease<br />
should be suspected (class IV, level C). Serology for syphilis and hepatic function studies<br />
could be considered (class IV, level C).<br />
Drug-<strong>in</strong>duced nystagmus may be predom<strong>in</strong>antly horizontal, predom<strong>in</strong>antly vertical,<br />
predom<strong>in</strong>antly rotatory, or (most commonly) mixed. It is most often seen with<br />
tranquiliz<strong>in</strong>g medications and anticonvulsants. Although drug-<strong>in</strong>duced nystagmus is<br />
more often evident with eccentric gaze (see below), it may also be evident <strong>in</strong> primary<br />
gaze (Burde, 1991; Remler, 1990).<br />
Nystagmus may occur as an epileptic phenomena. Epileptic nystagmus is usually<br />
horizontal, may be seen with epileptiform activity ipsilateral or contralateral to the<br />
direction of the slow component of the nystagmus, and often is associated with altered<br />
states of consciousness, although consciousness may be preserved dur<strong>in</strong>g the attacks<br />
(Furman, 1990a; Harris, 1997; Kaplan, 1993; Stolz, 1991; Tusa, 1990). There are two<br />
postulated mechanisms for the eye deviation <strong>in</strong> epileptic nystagmus (Furman, 1990a;<br />
Harris, 1997; Kaplan, 1993; Tusa, 1990). Ipsiversive eye deviation, with eye movement<br />
record<strong>in</strong>gs and EEG show<strong>in</strong>g seizure-<strong>in</strong>duced ipsilateral l<strong>in</strong>ear slow phases, is postu-<br />
Table 17–2. Etiologies of Periodic Alternat<strong>in</strong>g Nystagmus (PAN)<br />
Congenital (may be associated with alb<strong>in</strong>ism) (Abadi, 1994; Gradste<strong>in</strong>, 1997; Huygen, 1995; Ito, 2000;<br />
Shallo-Hoffmann, 1999)<br />
Arnold-Chiari malformation and other malformations of the craniocervical junction (Furman, 1990b)<br />
Cerebellar degenerations (Furman, 1990; Sakakibara, 1993)<br />
Ataxia-telangiectasia<br />
Cerebellar masses, <strong>in</strong>clud<strong>in</strong>g tumors, abscesses, and cysts<br />
Bra<strong>in</strong>stem <strong>in</strong>farction (Furman, 1990b)<br />
Cerebellar <strong>in</strong>fections, <strong>in</strong>clud<strong>in</strong>g syphilis and Jakob-Creutzfeldt disease (Grant, 1993; Leigh, 1991;<br />
Yokota, 1991)<br />
Hepatic encephalopathy (Averbuch-Heller, 1995a)<br />
Trauma (Leigh, 1991)<br />
Multiple sclerosis (Leigh, 1991; Matsumoto, 2001)<br />
<strong>An</strong>ticonvulsant medications<br />
Follow<strong>in</strong>g visual loss (e.g., due to cataract or vitreous hemorrhage)<br />
Epileptic PAN (after hypoxic encephalopathy) (Moster, 1998)
lated to result from stimulation of the smooth pursuit region <strong>in</strong> the temporo-occipital<br />
cortex. If eye velocity is high or the eye reaches a far eccentric portion <strong>in</strong> the orbit, a<br />
normal resett<strong>in</strong>g quick phase eye movement occurs after each slow phase, result<strong>in</strong>g <strong>in</strong><br />
nystagmus. Contraversive eye deviations, with eye movement record<strong>in</strong>gs and electroencephalogram<br />
(EEG) show<strong>in</strong>g seizure-<strong>in</strong>duced contralateral quick phases, is thought<br />
due to stimulation of the saccade-controll<strong>in</strong>g regions of the temporo-occipital or frontal<br />
cortex. If gaze-hold<strong>in</strong>g is defective (e.g., the neural <strong>in</strong>tegration is ‘‘leaky’’), then velocitydecreas<strong>in</strong>g<br />
slow phases br<strong>in</strong>g the eyes back to the midl<strong>in</strong>e after each quick phase,<br />
result<strong>in</strong>g <strong>in</strong> nystagmus. Epileptic PAN has been described (after hypoxic encephalopathy)<br />
(Moster, 1998).<br />
Epileptic nystagmus is rare and usually seen <strong>in</strong> patients with a history of epilepsy and<br />
<strong>in</strong> those with the nystagmus associated with altered levels of consciousness (Gire, 2001).<br />
Electroencephalography should be considered <strong>in</strong> patients with episodic nystagmus and<br />
oscillopsia, especially if other f<strong>in</strong>d<strong>in</strong>gs suggest a seizure disorder as a diagnostic<br />
possibility. Episodic vertigo with nystagmus may also be due to a form of migra<strong>in</strong>e<br />
(Dieterich, 1999).<br />
Spontaneous jerk nystagmus that is purely torsional is a rare form of central<br />
vestibular nystagmus. Often it is difficult to detect except by observation of<br />
the conjunctival vessels or by not<strong>in</strong>g the direction of ret<strong>in</strong>al movements on either side<br />
of the fovea. Purely torsional nystagmus may be present <strong>in</strong> primary gaze or elicited<br />
by head position<strong>in</strong>g or gaze deviation (Lopez, 1992). Purely torsional nystagmus may<br />
be seen with bra<strong>in</strong>stem and posterior fossa lesions, such as tumors, syr<strong>in</strong>gobulbia,<br />
syr<strong>in</strong>gomyelia with Arnold-Chiari malformation, lateral medullary syndrome, multiple<br />
sclerosis, trauma, vascular anomalies, postencephalitis, and sarcoidosis, and the stiffperson<br />
syndrome (Lopez, 1992; Stearns, 1993).<br />
Contralesionally beat<strong>in</strong>g torsional nystagmus may be due to a midbra<strong>in</strong> lesion<br />
<strong>in</strong>volv<strong>in</strong>g the rostral <strong>in</strong>terstitial nucleus of the medial longitud<strong>in</strong>al fasciculus (MLF),<br />
whereas lesions of the <strong>in</strong>terstitial nucleus of Cajal <strong>in</strong> the midbra<strong>in</strong> cause ipsilesional<br />
torsional nystagmus (Helmchen, 1996). Torsional nystagmus occurr<strong>in</strong>g only dur<strong>in</strong>g<br />
vertical pursuit has been described with cavernous angiomas of the middle cerebellar<br />
peduncle (FitzGibbon, 1996). We recommend MRI for unexpla<strong>in</strong>ed purely torsional<br />
nystagmus (class III–IV, level B). Nonrhythmic but cont<strong>in</strong>uous torsional eye movements<br />
have been reported as a paraneoplastic process (Rosenthal, 1987).<br />
What Are the Causes of Predom<strong>in</strong>antly<br />
Vertical Jerk Nystagmus?<br />
Nystagmus and Other Ocular Oscillations 385<br />
Spontaneous jerk nystagmus <strong>in</strong> primary gaze that is predom<strong>in</strong>antly vertical <strong>in</strong>cludes<br />
upbeat nystagmus and downbeat nystagmus (Buttner, 1995). Downbeat nystagmus is<br />
usually present <strong>in</strong> primary position, but is greatest when the patient looks down<br />
(Alexander’s law) and laterally. On upward gaze, the nystagmus is less pronounced or<br />
disappears completely. Downbeat nystagmus is often associated with horizontal<br />
gaze-evoked nystagmus. Convergence may <strong>in</strong>crease, suppress, or convert the<br />
nystagmus to upbeat nystagmus. The nystagmus may be dysjunctive, more vertical<br />
<strong>in</strong> one eye and torsional <strong>in</strong> the other eye. There may be an <strong>in</strong>ternuclear ophthalmoplegia<br />
(see Chapter 14). Downbeat nystagmus may occur with cervicomedullary junction
386 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
disease, midl<strong>in</strong>e medullary lesions, posterior midl<strong>in</strong>e cerebellar lesions, or diffuse<br />
cerebellar disease (Buttner, 1995; Walker, 1999). Most responsible lesions affect the<br />
vestibulocerebellum (flocculus, paraflocculus, nodulus, and uvula) and the underly<strong>in</strong>g<br />
medulla. Deficient drive by the posterior semicircular canals, whose central projections<br />
cross <strong>in</strong> the floor of the fourth ventricle, has been postulated as an explanation for<br />
downbeat nystagmus. Interruption of downward vestibulo-ocular reflex pathways,<br />
which synapse <strong>in</strong> the medial vestibular nucleus and cross <strong>in</strong> the medulla (beneath the<br />
nucleus prepositus hypoglossi) to reach the contralateral medial longitud<strong>in</strong>al fasciculus,<br />
would result <strong>in</strong> upward smooth eye drift and a downward corrective saccade.<br />
Cerebellar, especially floccular and uvulonodular, lesions may cause this nystagmus<br />
by dis<strong>in</strong>hibition of the cerebellar effect on the vestibular nuclei. The cerebellar flocculus<br />
conta<strong>in</strong>s Purk<strong>in</strong>je cells that send <strong>in</strong>hibitory projections to the anterior canal but not<br />
posterior canal central pathways; therefore, dis<strong>in</strong>hibition would lead to downbeat<br />
nystagmus. Damage to the nuclei propositus hypoglossi and the medial vestibular<br />
nuclei (the neural <strong>in</strong>tegrator) <strong>in</strong> the medulla has also been suggested as the cause of<br />
the nystagmus. A patient with acute multiple sclerosis with a lesion of the caudal<br />
medulla (which conta<strong>in</strong>s Roller’s nucleus and nucleus <strong>in</strong>tercalatus) developed downbeat<br />
nystagmus upon horizontal head oscillations (perverted head-shak<strong>in</strong>g nystagmus)<br />
(M<strong>in</strong>agar, 2001). Etiologies of downbeat nystagmus are listed <strong>in</strong> Table 17–3.<br />
The evaluation of downbeat nystagmus depends on the cl<strong>in</strong>ical circumstances and<br />
associated neurologic f<strong>in</strong>d<strong>in</strong>gs. We recommend MRI <strong>in</strong> patients with unexpla<strong>in</strong>ed<br />
downbeat nystagmus (Himi, 1995) (class IV, level C). MRI is normal or shows diffuse<br />
cerebellar atrophy <strong>in</strong> patients with familial cerebellar degenerations. In patients tak<strong>in</strong>g<br />
anticonvulsants or lithium, drug levels should be measured and adjusted as needed<br />
(class IV, level C). If MRI is normal, B12 and magnesium levels should be considered<br />
(class IV, level C). Thiam<strong>in</strong>e therapy for selected cases should be considered and the<br />
possibility of alcohol or toluene abuse <strong>in</strong>vestigated (class IV, level C). If there are signs<br />
suggestive of CNS <strong>in</strong>fection, a sp<strong>in</strong>al tap may be warranted. In a patient with downbeat<br />
nystagmus with the acute or subacute onset of cerebellar signs and symptoms, a<br />
paraneoplastic process must be considered, especially due to small cell lung cancer,<br />
testicular cancer, gynecologic cancers (especially ovarian and breast cancer), and<br />
Hodgk<strong>in</strong>’s disease. The workup of these patients might <strong>in</strong>clude serum anti-Yo (anti-<br />
Purk<strong>in</strong>je cell) antibodies, serum anti-Hu antibodies (ant<strong>in</strong>euronal nuclear antibodies<br />
type 1 or ANNA type 1), serum anti-Ta antibodies, chest x-ray and chest computed<br />
tomography (CT) imag<strong>in</strong>g, gynecologic exam<strong>in</strong>ation, CT or MRI of the abdomen and<br />
pelvis, mammography, and possibly hematologic consultation for bone marrow biopsy<br />
(class IV, level C).<br />
F<strong>in</strong>ally, <strong>in</strong> a significant number of <strong>in</strong>dividuals, no etiology for the downbeat<br />
nystagmus will be discovered. Young and Huang reported the use of clonazepam<br />
(1.0 mg twice daily) <strong>in</strong> five idiopathic cases of downbeat nystagmus (Young, 2001).<br />
Damage to the central projections of the anterior semicircular canals, which tend to<br />
deviate the eyes superiorly, has been suggested to expla<strong>in</strong> upbeat nystagmus. Upbeat<br />
nystagmus is usually worse <strong>in</strong> upgaze (Alexander’s law) and, unlike downbeat<br />
nystagmus, it usually does not <strong>in</strong>crease on lateral gaze. Convergence may <strong>in</strong>crease or<br />
decrease the nystagmus, or convert downbeat nystagmus to upbeat nystagmus (Hirose,<br />
1991). Damage to the ventral tegmental pathways, which may l<strong>in</strong>k the superior<br />
vestibular nuclei to the superior rectus and <strong>in</strong>ferior oblique subnuclei of the oculomotor<br />
nuclei, may cause the eyes to glide down, result<strong>in</strong>g <strong>in</strong> upbeat nystagmus. Medullary
Table 17–3. Etiologies of Downbeat Nystagmus<br />
Nystagmus and Other Ocular Oscillations 387<br />
Craniocervical anomalies, <strong>in</strong>clud<strong>in</strong>g cerebellar ectopia, Chiari malformation, platybasia,<br />
basilar <strong>in</strong>vag<strong>in</strong>ation, and Paget’s disease (Pieh, 2000; Russell, 1992)<br />
Familial cerebellar degenerations <strong>in</strong>clud<strong>in</strong>g sp<strong>in</strong>ocerebellar ataxia 6 (Harada, 1998)<br />
Multiple system atrophy (Bertholon, 2002)<br />
Posterior fossa tumors (Chan, 1991)<br />
Increased <strong>in</strong>tracranial pressure (e.g., due to supratentorial mass) and hydrocephalus (Chan, 1991)<br />
Bra<strong>in</strong>stem or cerebellar <strong>in</strong>farction, anoxia, or hemorrhage (Olson, 2001; Rousseaux, 1991)<br />
Dolichoectasia of the vertebrobasilar artery (Gans, 1990; Himi, 1995; Krespi, 1995; Lee, 2001)<br />
Intermittent vertebral artery compression by an osteophyte (Rosengart, 1993)<br />
Encephalitis, <strong>in</strong>clud<strong>in</strong>g herpes simplex encephalitis and human T-cell leukemia virus 1 (HTLV-1)<br />
<strong>in</strong>fection (Waragai, 1995; Yoshimoto, 1991)<br />
Heat stroke (Van Stavern, 2000)<br />
Cephalic tetanus (Orwitz, 1997)<br />
Multiple sclerosis and other leukodystrophies (M<strong>in</strong>agar, 2001)<br />
Syr<strong>in</strong>gomyelia=syr<strong>in</strong>gobulbia (Rowlands, 2000)<br />
Trauma<br />
Alcohol, <strong>in</strong>clud<strong>in</strong>g alcohol-<strong>in</strong>duced cerebellar degeneration<br />
Wernicke’s encephalopathy<br />
Thiam<strong>in</strong>e deficiency (Mulder, 1999)<br />
Alcoholics<br />
Nonalcoholics (vomit<strong>in</strong>g, drastic weight reduction diet, colonic surgery, chronic hemodialysis)<br />
(Merk<strong>in</strong>-Zaborsky, 2000)<br />
Paraneoplastic cerebellar degeneration (<strong>in</strong>clud<strong>in</strong>g testicular cancer with anti-Ta antibody) (Bennett,<br />
1999; Hammack, 1992; Peterson, 1992)<br />
Superificial siderosis of the central nervous system (CNS) (Pelak, 1999)<br />
Congenital<br />
Vitam<strong>in</strong> B12 deficiency<br />
Magnesium deficiency (Du Pasquier, 1998)<br />
Drugs, <strong>in</strong>clud<strong>in</strong>g lithium, toluene, <strong>in</strong>travenous or epidural narcotics, and anticonvulsants (e.g.,<br />
phenyto<strong>in</strong>, carbamazep<strong>in</strong>e, felbamate) (Henderson, 2000; Hwang, 1995; Monteiro, 1993)<br />
Transient f<strong>in</strong>d<strong>in</strong>g <strong>in</strong> otherwise normal <strong>in</strong>fants<br />
Idiopathic (Olson, 2001)<br />
disease may cause upbeat nystagmus as may lesions of the anterior cerebellar vermis,<br />
perihypoglossal and <strong>in</strong>ferior olivary nuclei of the medulla, pont<strong>in</strong>e tegmentum,<br />
brachium conjunctivum, midbra<strong>in</strong>, and bra<strong>in</strong>stem diffusely (Buttner, 1995; Kanaya,<br />
1994; Munro, 1993). Medullary lesions <strong>in</strong>variably <strong>in</strong>volve the perihypoglossal nucleus<br />
and adjacent medial vestibular nucleus, nucleus <strong>in</strong>tercalatus, and ventral tegmentum,<br />
which conta<strong>in</strong> projections from vestibular nuclei that receive <strong>in</strong>puts from the anterior<br />
semicircular canals. Primary position upbeat nystagmus may occur with unilateral<br />
medial medullary <strong>in</strong>farction, likely due to impairment of the vertical position-tovelocity<br />
neural <strong>in</strong>tegrator <strong>in</strong> the nucleus <strong>in</strong>tercalatus of Stader<strong>in</strong>i, a structure <strong>in</strong> the<br />
paramedian caudal medulla located caudal to the vestibular nuclei and to the most<br />
rostral of the perihypoglossal nuclei (nucleus prepositus hypoglossi and nucleus of<br />
Roller) (Hirose, 1998; Janssen, 1998). Lesions of this structure may cause primary
388 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
position upbeat nystagmus <strong>in</strong>creased <strong>in</strong> downward gaze (Ohkoshi, 1998). Bow-tie<br />
nystagmus, <strong>in</strong> which quick phases are directed obliquely upward with horizontal<br />
components alternat<strong>in</strong>g to the right and left, is probably a variant of upbeat nystagmus.<br />
Etiologies of upbeat nystagmus are outl<strong>in</strong>ed <strong>in</strong> Table 17–4.<br />
The evaluation of upbeat nystagmus <strong>in</strong>cludes a complete neurologic and ophthalmologic<br />
exam<strong>in</strong>ation. MRI is warranted <strong>in</strong> most cases to <strong>in</strong>vestigate the presence of a<br />
structural lesion (class III–IV, level B). In children, MRI is <strong>in</strong>dicated to <strong>in</strong>vestigate not<br />
only posterior fossa lesions but also lesions of the anterior visual pathways. If imag<strong>in</strong>g is<br />
normal <strong>in</strong> children, then ERG should be considered (class IV, level C). Sp<strong>in</strong>al tap is<br />
<strong>in</strong>dicated <strong>in</strong> patients with signs or symptoms suggestive of men<strong>in</strong>geal irritation or<br />
central nervous system (CNS) <strong>in</strong>fection. In adults with negative neuroimag<strong>in</strong>g studies,<br />
organophosphate or anticonvulsant <strong>in</strong>toxication should be <strong>in</strong>vestigated. Testicular<br />
cancer is a consideration <strong>in</strong> men (anti-Ta antibodies) (Bennett, 1999) (class IV, level C).<br />
The treatment of vertical nystagmus is directed at the etiology (e.g., surgical<br />
correction of Arnold-Chiari malformation). <strong>Cl<strong>in</strong>ical</strong> evidence suggests <strong>in</strong>volvement of<br />
GABA-ergic pathways and chol<strong>in</strong>ergic transmission <strong>in</strong> vertical vestibulo-ocular reflexes<br />
(Leigh, 1994). GABA agonists and chol<strong>in</strong>ergic drugs have thus been tried to relieve the<br />
visual impairment with vertical nystagmus. Clonazepam, a GABA-A agonist, and<br />
baclofen, a GABA-B agonist, have been shown to reduce nystagmus velocity and<br />
oscillopsia <strong>in</strong> some patients with downbeat or upbeat nystagmus (Averbuch-Heller,<br />
1997; Dieterich, 1991). Gabapent<strong>in</strong> may occasionally <strong>in</strong>duce a response (Averbuch-<br />
Heller, 1997; Band<strong>in</strong>i, 2001) <strong>in</strong> acquired nystagmus <strong>in</strong> multiple sclerosis. Intravenous<br />
physostigm<strong>in</strong>e, an acetylchol<strong>in</strong>esterase <strong>in</strong>hibitor, worsened vertical nystagmus <strong>in</strong> five<br />
Table 17–4. Etiologies of Upbeat Nystagmus<br />
Primary cerebellar degenerations and atrophies (Fujikane, 1992; Mizuno, 1990)<br />
Arnold-Chiari malformation<br />
Posterior fossa tumors<br />
Bra<strong>in</strong>stem or cerebellum <strong>in</strong>farction or hemorrhage (Hirose, 1990, 1998; Munro, 1993; Rousseaux,<br />
1991)<br />
Multiple sclerosis (Hirose, 1990, 1991; Ohkoshi, 1998)<br />
Men<strong>in</strong>gitis and bra<strong>in</strong>stem encephalitis<br />
Thalamic arteriovenous malformation<br />
Wernicke’s encephalopathy<br />
Behçet’s syndrome<br />
Congenital, <strong>in</strong>clud<strong>in</strong>g cases associated with Leber’s congenital amaurosis and other congenital<br />
anterior visual pathway disorders (Good, 1990, 1993)<br />
Pelizaeus-Merzbacher disease (Trobe, 1991)<br />
Fisher’s syndrome (ataxia, areflexia, and ophthalmoplegia) (Yamazaki, 1994)<br />
Middle ear disease<br />
Organophosphate poison<strong>in</strong>g<br />
Tobacco-<strong>in</strong>duced<br />
<strong>An</strong>ticonvulsant <strong>in</strong>toxication<br />
Cyclospor<strong>in</strong> A (Albera, 1997)<br />
Paraneoplastic syndrome with testicular cancer and anti-Ta antibodies (Bennett, 1999)<br />
Transient f<strong>in</strong>d<strong>in</strong>g <strong>in</strong> otherwise healthy neonates (Goldblum, 1994)
patients (Dieterich, 1991), whereas <strong>in</strong>travenous scopolam<strong>in</strong>e, an antichol<strong>in</strong>ergic drug,<br />
reduced nystagmus and oscillopsia <strong>in</strong> two patients with downbeat nystagmus (Barton,<br />
1994). <strong>An</strong>tichol<strong>in</strong>ergic drugs may thus be considered for patients with upbeat or<br />
downbeat nystagmus. F<strong>in</strong>ally, downbeat nystagmus usually is present <strong>in</strong> primary<br />
position but is greatest when the patient looks down (Alexander’s law) and subsides<br />
<strong>in</strong> upgaze. Patients may therefore benefit from symmetric base-down prisms that turn<br />
the eyes up.<br />
What Are the Causes of B<strong>in</strong>ocular Symmetric<br />
Jerk Nystagmus Present <strong>in</strong> Eccentric Gaze or<br />
Induced by Various Maneuvers?<br />
Nystagmus and Other Ocular Oscillations 389<br />
Spontaneous b<strong>in</strong>ocular conjugate symmetric jerk nystagmus that is <strong>in</strong>duced by eccentric<br />
gaze (gaze-evoked nystagmus) <strong>in</strong>cludes nystagmus due to bra<strong>in</strong>stem=cerebellar disease,<br />
Bruns’ nystagmus, drug-<strong>in</strong>duced nystagmus, physiologic nystagmus, rebound<br />
nystagmus, and convergence-<strong>in</strong>duced nystagmus. Downbeat nystagmus and upbeat<br />
nystagmus may occur only on downward or upward gaze, respectively (see above).<br />
With gaze-evoked nystagmus, the eyes fail to rema<strong>in</strong> <strong>in</strong> an eccentric position of gaze<br />
but drift to midposition. The velocity of the slow component decreases exponentially as<br />
the eyes approach midposition. A ‘‘leaky’’ neural <strong>in</strong>tegrator or cerebellar (especially<br />
vestibulocerebellar) lesion may result <strong>in</strong> this type of nystagmus, which is more<br />
pronounced when the patient looks toward the lesion. Cerebellopont<strong>in</strong>e angle tumors<br />
may cause Bruns’ nystagmus, a comb<strong>in</strong>ation of ipsilateral large-amplitude, lowfrequency<br />
nystagmus that is due to impaired gaze hold<strong>in</strong>g, and contralateral smallamplitude,<br />
high-frequency nystagmus that is due to vestibular impairment (Leigh,<br />
1991). Gaze-evoked nystagmus may be a side effect of medications, <strong>in</strong>clud<strong>in</strong>g anticonvulsants,<br />
sedatives, and alcohol. Gaze-evoked nystagmus has been described with<br />
adult-onset Alexander’s disease with <strong>in</strong>volvement of the middle cerebellar peduncles<br />
and dentate nuclei (Martidis, 1999) and is also a feature of familial episodic vertigo and<br />
ataxia type 2 that is responsive to acetazolamide (Baloh, 1991, 1997; Brandt, 1997).<br />
Physiologic or end-po<strong>in</strong>t nystagmus is a benign low-amplitude jerk nystagmus with the<br />
fast component directed toward the field of gaze. It usually ceases when the eyes are<br />
brought to a position somewhat less than the extremes of gaze.<br />
Rebound nystagmus is seen <strong>in</strong> some patients with bra<strong>in</strong>stem and=or cerebellar<br />
disease (e.g., olivocerebellar atrophy, bra<strong>in</strong>stem=cerebellar tumor or <strong>in</strong>farction,<br />
Mar<strong>in</strong>esco-Sjögren syndrome, Dandy-Walker cyst, Gerstmann-Straussler-Sche<strong>in</strong>ker<br />
disease, adult-onset Alexander’s disease, etc.) (L<strong>in</strong>, 1999; Martidis, 1999; Yee, 1992).<br />
The orig<strong>in</strong>al gaze-evoked nystagmus may wane and actually reverse direction so that<br />
the slow component is directed centrifugally (centripetal nystagmus). Rebound nystagmus<br />
probably reflects an attempt by bra<strong>in</strong>stem or cerebellar mechanisms to correct for<br />
the centripetal drift of gaze-evoked nystagmus (Leigh, 1999).<br />
Patients with gaze-evoked nystagmus who are tak<strong>in</strong>g anticonvulsant or sedative<br />
medications and those with physiologic nystagmus require no further evaluation.<br />
Otherwise, patients with gaze-evoked or rebound nystagmus, especially if associated<br />
symptoms or signs of bra<strong>in</strong>stem or cerebellar dysfunction or auditory impairment exist,
390 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
require MRI, with special attention to the cerebellum and cerebello-pont<strong>in</strong>e angle (L<strong>in</strong>,<br />
1999) (class IV, level C).<br />
Convergence may change nystagmus by convert<strong>in</strong>g downbeat to upbeat, upbeat to<br />
downbeat, or pendular to upbeat. Convergence-evoked nystagmus is usually vertical<br />
(upbeat is more common than downbeat) and seen most commonly with multiple<br />
sclerosis or bra<strong>in</strong>stem <strong>in</strong>farction (Oliva, 1990). MRI is thus warranted <strong>in</strong> patients with<br />
convergence-<strong>in</strong>duced nystagmus (class IV, level C). Convergence may also <strong>in</strong>crease or<br />
decrease the amplitude of nystagmus and may evoke horizontal (congenital or acquired<br />
pendular and jerk) or vertical (upbeat or downbeat) nystagmus. Convergence-<strong>in</strong>duced<br />
pendular nystagmus has been described as a congenital phenomenon (conjugate) and as<br />
an acquired phenomenon (disjunctive) with multiple sclerosis (Barton, 1999; Oliva,<br />
1990). Base-<strong>in</strong> prisms have been used to alleviate the symptoms of oscillopsia and<br />
improve read<strong>in</strong>g acuity <strong>in</strong> patients with acquired convergence-evoked pendular<br />
nystagmus due to multiple sclerosis (Barton, 1999). The effects of convergence on<br />
nystagmus are not to be confused with convergence nystagmus <strong>in</strong> which a slow<br />
abduction of the eyes is followed by quick adduction (see above).<br />
B<strong>in</strong>ocular symmetric conjugate jerk nystagmus that is <strong>in</strong>duced <strong>in</strong>cludes optok<strong>in</strong>etic<br />
nystagmus, rotational=caloric vestibular nystagmus, positional nystagmus, Valsalva<strong>in</strong>duced<br />
nystagmus, and hyperventilation-<strong>in</strong>duced nystagmus (Burde, 1991; Leigh,<br />
1991). The first two types of <strong>in</strong>duced nystagmus are physiologic and, although<br />
abnormalities of these responses may aid <strong>in</strong> cl<strong>in</strong>ical diagnosis, they are not further<br />
discussed here.<br />
Positional vertigo of the benign paroxysmal type, also known as benign paroxysmal<br />
position<strong>in</strong>g vertigo or positional nystagmus, is usually idiopathic and possibly related<br />
to degeneration of the macula of the otolith organ or to lesions of the posterior<br />
semicircular canal (Baloh, 1995; Brandt, 1990, 1991; Furman, 1999; Lawden, 1995;<br />
Weider, 1994). It has been proposed that otoconia detached from the otoconial layer<br />
(by degeneration or trauma) gravitate and settle on the cupula of the posterior canal<br />
caus<strong>in</strong>g it to become heavier than the surround<strong>in</strong>g endolymph and thus sensitive to<br />
changes <strong>in</strong> the direction of gravity (with positional change). After rapid head tilt toward<br />
the affected ear or follow<strong>in</strong>g head extension, when the posterior semicircular canal is<br />
moved <strong>in</strong> the specific plane of stimulation, an ampullofugal deflection of the cupula<br />
occurs, with a rotational vertigo and concomitant nystagmus. Some patients show a<br />
strong horizontal nystagmus <strong>in</strong>duced by lateral head position<strong>in</strong>g suggest<strong>in</strong>g lateral<br />
(rather than posterior) semicircular canal irritation (lateral canal or horizontal canal<br />
variant of benign paroxysmal positional vertigo) (Baloh, 1993; De la Meilleure, 1996).<br />
Other causes of positional vertigo <strong>in</strong>clude trauma, <strong>in</strong>fection, labyr<strong>in</strong>th<strong>in</strong>e fistula,<br />
ischemia, demyel<strong>in</strong>at<strong>in</strong>g disease, Arnold-Chiari malformation, and, rarely, posterior<br />
fossa tumors or vascular malformations (Lawden, 1995; Sakata, 1991).<br />
Besides paroxysmal positional nystagmus, patients often also exhibit static (persistent)<br />
positional nystagmus while ly<strong>in</strong>g <strong>in</strong> a lateral position. This static nystagmus is<br />
predom<strong>in</strong>antly horizontal with m<strong>in</strong>imal vertical component. Paroxysmal vertigo<br />
<strong>in</strong>duced by certa<strong>in</strong> head positions is the most common compla<strong>in</strong>t; the patient is<br />
asymptomatic between bouts. The Nylen-Barany (Dix-Hallpike) maneuver (briskly<br />
tilt<strong>in</strong>g the patient’s head backward and turn<strong>in</strong>g it 45 degrees to one side) allows<br />
differentiat<strong>in</strong>g a peripheral from a central orig<strong>in</strong> for positional vertigo.<br />
With peripheral lesions, severe rotational vertigo associated with nausea (occasionally<br />
vomit<strong>in</strong>g) and nystagmus appear several seconds (2 to 15 seconds) after the head
position is changed. In benign paroxysmal positional (position<strong>in</strong>g) vertigo, it is unusual<br />
for the vertigo to have a duration of more than 1 m<strong>in</strong>ute. Cochlear or neurologic<br />
symptoms are typically absent. The nystagmus is usually torsional, with the upper pole<br />
of the eye beat<strong>in</strong>g toward the ground. The vertigo and nystagmus then fatigue and<br />
abate with<strong>in</strong> 10 seconds after appearance, and when the patient is rapidly brought back<br />
to a sitt<strong>in</strong>g position, vertigo recurs and nystagmus develops <strong>in</strong> the opposite direction<br />
(rebound). With repetition of the maneuver, the symptoms and nystagmus become<br />
progressively less severe (habituation), and the reproducibility of the abnormalities is<br />
<strong>in</strong>constant. Mild transient nystagmus <strong>in</strong> one head position may occasionally be elicited<br />
<strong>in</strong> normal subjects. Patients with benign paroxysmal positional vertigo are often treated<br />
successfully with canalith reposition<strong>in</strong>g procedures (Weider, 1994).<br />
A central lesion should be suspected when (1) the maneuver is positive with the head<br />
turned to either side; (2) the nystagmus is direction chang<strong>in</strong>g rather than fixed,<br />
appear<strong>in</strong>g immediately after the shift <strong>in</strong> position and rema<strong>in</strong><strong>in</strong>g for as long as the<br />
head is down; (3) the nystagmus is unaccompanied by nausea or a sense of discomfort;<br />
if present, vertigo is mild and lasts no longer than 60 seconds; and (4) repetition does<br />
not cause blunt<strong>in</strong>g of the effects. Typically, there are other associated CNS f<strong>in</strong>d<strong>in</strong>gs.<br />
Patients with the central form of positional nystagmus require MRI to <strong>in</strong>vestigate<br />
structural posterior fossa lesions (class III–IV, level B). Occasionally, patients with<br />
benign paroxysmal positional vertigo will demonstrate f<strong>in</strong>d<strong>in</strong>gs dur<strong>in</strong>g the<br />
Nylen-Barany maneuver similar to those documented <strong>in</strong> patients with central lesions<br />
(Baloh, 1995).<br />
Nystagmus <strong>in</strong>duced by the Valsalva maneuver may occur with Chiari malformation<br />
or perilymph fistulas (Leigh, 1991). Hyperventilation may <strong>in</strong>duce nystagmus <strong>in</strong> patients<br />
with tumors of the eighth cranial nerve (e.g., acoustic neuroma or epidermoid tumors),<br />
after vestibular neuritis, or with central demyel<strong>in</strong>at<strong>in</strong>g lesions (Leigh, 1991; M<strong>in</strong>or,<br />
1999). Hyperventilation-<strong>in</strong>duced nystagmus has the slow phase away from the side of<br />
the lesion (an excitatory or recovery nystagmus) and is likely due to the effect of<br />
hyperventilation upon serum pH and calcium concentration, which improves nerve<br />
conduction <strong>in</strong> a marg<strong>in</strong>ally functional, demyel<strong>in</strong>ated nerve (Leigh, 1991; M<strong>in</strong>or, 1999).<br />
The evaluation of patients with jerk nystagmus is outl<strong>in</strong>ed <strong>in</strong> Figure 17–5.<br />
What Are the Characteristics of Saccadic<br />
Intrusions?<br />
Nystagmus and Other Ocular Oscillations 391<br />
Inappropriate saccades, or saccadic <strong>in</strong>trusions, <strong>in</strong>terfere with macular fixation of an<br />
object of <strong>in</strong>terest. The essential difference between nystagmus and saccadic <strong>in</strong>trusions<br />
lies <strong>in</strong> the <strong>in</strong>itial eye movement that takes the l<strong>in</strong>e of sight away from the object of<br />
regard (Leigh, 1999). For nystagmus, it is a slow drift or slow phase as opposed to an<br />
<strong>in</strong>appropriate saccadic movement that <strong>in</strong>trudes on steady fixation. There are several<br />
types of saccadic <strong>in</strong>trusions.<br />
Square-wave jerks take the eyes off the target and are followed after about 200 msec<br />
by a corrective saccade (Shallo-Hoffmann, 1990). They may appear normally <strong>in</strong> the<br />
young and the elderly, but when larger than 1 or 2 degrees they are pathologic, result<strong>in</strong>g<br />
from disorders <strong>in</strong>clud<strong>in</strong>g a variety of cerebral or cerebellar lesions, progressive supranuclear<br />
palsy, Hunt<strong>in</strong>gton’s chorea, Park<strong>in</strong>son’s disease, Wernicke-Korsakoff’s
392 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 17–5. Evaluation of jerk nystagmus.<br />
syndrome, Friedreich’s ataxia, AIDS-dementia complex, Gerstmann-Straussler-<br />
Sche<strong>in</strong>ker disease, adult-onset Alexander’s disease, carbohydrate-deficient glycoprote<strong>in</strong><br />
syndrome type 1a, and schizophrenia (Friedman, 1992; Martidis, 1999; Rascol, 1991;<br />
Stark, 2000; Yee, 1992). They may also occur with lithium or tobacco use. <strong>An</strong> <strong>in</strong>creased<br />
frequency of square wave jerks may be noted after unilateral pallidotomy for<br />
Park<strong>in</strong>son’s disease (Averbuch-Heller, 1999). Very frequent square-wave jerks (squarewave<br />
oscillations) may be mistaken for nystagmus and may occur with cerebellar<br />
disease, progressive supranuclear palsy, and cerebral hemispheral disease (Friedman,<br />
1992; Leigh, 1991).
Nystagmus and Other Ocular Oscillations 393<br />
Macro–square-wave jerks are similar to square-wave jerks but are of larger amplitude<br />
(20 to 40 degrees). They are occasionally present <strong>in</strong> the vertical plane and have been<br />
noted <strong>in</strong> multiple sclerosis, cerebellar hemorrhage, olivopontocerebellar atrophy, multiple<br />
systems atrophy, and Arnold-Chiari malformation (Leigh, 1991; Yamamoto, 1992).<br />
Macrosaccadic oscillations are different from square-wave jerks and consist of eye<br />
oscillations around the fixation angle with <strong>in</strong>tersaccadic <strong>in</strong>tervals approximately<br />
200 msec (Leigh, 1991). They are usually conjugate, horizontal, and symmetric <strong>in</strong> both<br />
directions of gaze, but may occur <strong>in</strong> torsional or vertical planes. Macrosaccadic<br />
oscillations occur <strong>in</strong> patients with cerebellar disease, especially affect<strong>in</strong>g the cerebellar<br />
midl<strong>in</strong>e and underly<strong>in</strong>g nuclei (e.g., cerebellar hemorrhage or sp<strong>in</strong>ocerebellar degenerations).<br />
Macrosaccadic oscillations after pont<strong>in</strong>e trauma may have been due to<br />
dysfunction of pont<strong>in</strong>e omnipause neurons, and thus dis<strong>in</strong>hibition of saccadic burst<br />
neurons (Averbuch-Heller, 1996). Macrosaccadic oscillations may be <strong>in</strong>duced by edrophonium<br />
(Tensilon) <strong>in</strong> patients with profound ophthalmoplegia from myasthenia gravis<br />
(Komiyama, 1999a).<br />
Square-wave jerks, square-wave oscillations, macro–square-wave jerks, and macrosaccadic<br />
oscillations usually occur <strong>in</strong> the context of otherwise evident neurologic<br />
diseases. If the nature of the causal degenerative process account<strong>in</strong>g for these <strong>in</strong>trusions<br />
is not evident on cl<strong>in</strong>ical history and neurologic exam, MRI may be needed (class IV,<br />
level C). Macrosaccadic oscillations and high-amplitude square-wave jerks may be<br />
treated with GABA-A agonists, benzodiazep<strong>in</strong>es, and barbiturates. Square-wave jerks<br />
and square-wave oscillations may improve with valproic acid (2000 mg=day), which<br />
may restore GABA-ergic tonic <strong>in</strong>hibitory action from the substantia nigra pars reticulata<br />
to the superior colliculus (Traccis, 1997).<br />
Occasionally, otherwise normal <strong>in</strong>dividuals show <strong>in</strong>termittent, 15- to 30-Hz<br />
frequency, low-amplitude (0.1 to 0.5 degrees) horizontal oscillations (not detected on<br />
visual <strong>in</strong>spection but seen with the ophthalmoscope) termed microsaccadic flutter (Ashe,<br />
1991). Patients with microsaccadic flutter often compla<strong>in</strong> of ‘‘shimmer<strong>in</strong>g,’’ ‘‘jiggl<strong>in</strong>g,’’<br />
‘‘wavy,’’ or ‘‘laser beams’’ with paroxysms of visual disturbances last<strong>in</strong>g seconds to<br />
hours. Dizz<strong>in</strong>ess or dysequilibrium often accompanies the visual symptoms. Most<br />
patients are otherwise normal, although one patient had multiple sclerosis. Clonazepam,<br />
propranolol, and verapamil may reduce visual symptoms <strong>in</strong> some patients (Ashe,<br />
1991).<br />
Ocular flutter is a burst of to-and-fro horizontal saccades without an <strong>in</strong>tersaccadic<br />
<strong>in</strong>terval. Opsoclonus (saccadomania) is similar to ocular flutter, except that <strong>in</strong> opsoclonus<br />
there are conjugate, <strong>in</strong>voluntary, large amplitude saccades <strong>in</strong> all directions. Like<br />
ocular flutter, opsoclonus <strong>in</strong>dicates bra<strong>in</strong>stem, especially mesencephalic or pont<strong>in</strong>e, or<br />
cerebellar disease (Hattori, 1993). Opsoclonus evident only dur<strong>in</strong>g eye closure has been<br />
described with hereditary cerebellar ataxia (Hattori, 1993). Etiologies for ocular flutter<br />
and opsoclonus are outl<strong>in</strong>ed <strong>in</strong> Table 17–5.<br />
In patients with viral encephalitis, men<strong>in</strong>gitis, and other <strong>in</strong>fections, the opsoclonus<br />
may occur after a prodromal illness, <strong>in</strong>clud<strong>in</strong>g gastro<strong>in</strong>test<strong>in</strong>al tract symptoms, upper<br />
respiratory symptoms, malaise, and fever (Sheth, 1995; Tabarki, 1998; Wiest, 1997). The<br />
opsoclonus <strong>in</strong> these patients is often associated with truncal ataxia and other cerebellar<br />
signs, long tract signs, tremulousness, and myoclonus of the trunk and limbs. Sp<strong>in</strong>al<br />
fluid studies often show <strong>in</strong>creased prote<strong>in</strong> and a mononuclear pleocytosis. The illness<br />
usually resolves <strong>in</strong> a few weeks or months, although the course may be protracted and<br />
recovery <strong>in</strong>complete, especially <strong>in</strong> children (K<strong>in</strong>sbourne’s myoclonic encephalopathy or
394 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 17–5. Etiologies of Ocular Flutter and Opsoclonus<br />
Viral encephalitis, men<strong>in</strong>gitis, and other <strong>in</strong>fections (Connolly, 1997; Sheth, 1995; Tabarki, 1998;<br />
Vers<strong>in</strong>o, 1999; Wiest, 1997)<br />
K<strong>in</strong>sbourne’s myoclonic encephalopathy or danc<strong>in</strong>g eyes and danc<strong>in</strong>g feet or <strong>in</strong>fantile<br />
polymyoclonia<br />
<strong>Neuro</strong>blastoma (Fisher, 1994; Mitchell, 1990)<br />
Paraneoplastic effect of other tumors, especially small-cell lung cancer and breast cancer<br />
(Aggarwal, 1997; Bataller, 2001; Cav<strong>in</strong>ess, 1995; Corcia, 1997; Honnorat, 1997; Hormigo, 1994;<br />
Koukoulis, 1998; Luque, 1991; Mitoma, 1996; Schwartz, 1990; Tzuzaka, 1993)<br />
Intracranial tumors or cysts (Corcia, 1997;Tzuzaka, 1993)<br />
Trauma<br />
Hydrocephalus<br />
Hereditary cerebellar degeneration (Hattori, 1993)<br />
Thalamic hemorrhage, pont<strong>in</strong>e hemorrhage, or vertebrobasilar vascular <strong>in</strong>sufficiency<br />
Multiple sclerosis (Schon, 2001)<br />
Hyperosmolar stupor and coma<br />
Side effects of drugs, <strong>in</strong>clud<strong>in</strong>g lithium, amitriptyl<strong>in</strong>e, coca<strong>in</strong>e, phenyto<strong>in</strong> and diazepam, phenelz<strong>in</strong>e<br />
and imipram<strong>in</strong>e (Fisher, 1990)<br />
Toxic exposures, <strong>in</strong>clud<strong>in</strong>g thallium, toluene, chlordecone, strychn<strong>in</strong>e, DDT, and organophosphates<br />
Associated with other miscellaneous diseases, <strong>in</strong>clud<strong>in</strong>g acute polyradiculoneuritis, viral hepatitis,<br />
cherry-red spot myoclonus syndrome, carbohydrate-deficient glycoprote<strong>in</strong> syndrome type 1a,<br />
sarcoidosis, and AIDS (Gizzi, 1990; Kam<strong>in</strong>ski, 1991; Stark, 2000)<br />
In neonates, as either a transient benign phenomenon or related to bra<strong>in</strong> <strong>in</strong>jury due to anoxia,<br />
<strong>in</strong>tracranial hemorrhage, or Leber’s congenital amaurosis<br />
Idiopathic (Bataller, 2001)<br />
danc<strong>in</strong>g eyes and danc<strong>in</strong>g feet or <strong>in</strong>fantile polymyoclonia). Opsoclonus occurs <strong>in</strong> 2% of<br />
children with neuroblastoma, and conversely 50% of children with opsoclonus=<br />
myoclonus have neuroblastoma (Fisher, 1994; Mitchell, 1990). Opsoclonus appears<br />
before the discovery of a neuroblastoma <strong>in</strong> over 50% of the cases, and neuroblastomas<br />
associated with opsoclonus have a tendency to be located with<strong>in</strong> the thorax. Opsoclonus<br />
may also develop as a paraneoplastic effect of other tumors, especially small-cell<br />
lung cancer and breast cancer (Aggarwal, 1997; Bataller, 2001; Cav<strong>in</strong>ess, 1995; Corcia,<br />
1997; Hormigo, 1994; Koukoulis, 1998; Luque, 1991; Mitoma, 1996; Schwartz, 1990;<br />
Tzuzaka, 1993; Vigliani, 2001). Approximately 20% of patients with opsoclonusmyoclonus<br />
<strong>in</strong> adults have an underly<strong>in</strong>g tumor. In patients with small-cell lung<br />
cancer, opsoclonus usually antedates the diagnosis of the neoplasm, whereas <strong>in</strong> patients<br />
with breast cancer, opsoclonus develops before the diagnosis of the tumor <strong>in</strong> only<br />
half the patients. Although opsoclonus <strong>in</strong> these patients may occur as an isolated sign, it<br />
is more often associated with myoclonus, ataxia, and encephalopathy. The cerebrosp<strong>in</strong>al<br />
fluid may show an elevated prote<strong>in</strong> and a mild pleocytosis. Patients with breast cancer<br />
and opsoclonus=myoclonus may demonstrate an anti-Ri antibody (ant<strong>in</strong>euronal nuclear<br />
antibody 2 [ANNA-2]) <strong>in</strong> their serum.<br />
Bataller et al analyzed a series of 24 adult patients with idiopathic (10 cases) and<br />
paraneoplastic (14 cases) opsoclonus-myoclonus syndrome (OMS) to ascerta<strong>in</strong> possible<br />
differences <strong>in</strong> cl<strong>in</strong>ical course and response to immunotherapies between both groups<br />
(Bataller, 2001). Associated tumors were small-cell lung cancer (SCLC) (n<strong>in</strong>e patients),
Nystagmus and Other Ocular Oscillations 395<br />
non-SCLC (one patient), breast carc<strong>in</strong>oma (two patients), gastric adenocarc<strong>in</strong>oma (one<br />
patient), and kidney carc<strong>in</strong>oma (one patient). Patients with paraneoplastic OMS were<br />
older (median age: 66 years versus 40 years of those with idiopathic OMS) and had a<br />
higher frequency of encephalopathy (64% versus 10%). Serum from 10=10 idiopathic<br />
and 12=14 paraneoplastic OMS patients showed no specific immunoreactivity on rat or<br />
human bra<strong>in</strong>stem or cerebellum, lacked specific ant<strong>in</strong>euronal antibodies (Hu, Yo, Ri, Tr,<br />
glutamic acid decarboxylase, amphiphys<strong>in</strong>, or CV2) and did not conta<strong>in</strong> antibodies to<br />
voltage-gated calcium channels. The two paraneoplastic exceptions were a patient with<br />
SCLC, whose serum conta<strong>in</strong>ed both anti-Hu and antiamphiphys<strong>in</strong> antibodies, and a<br />
patient with breast cancer who had serum anti-Ri antibodies. The cl<strong>in</strong>ical course of<br />
idiopathic OMS was monophasic except <strong>in</strong> two elderly women who had relapses of the<br />
opsoclonus and mild residual ataxia. Most idiopathic OMS patients made a good<br />
recovery, but residual gait ataxia tended to persist <strong>in</strong> older patients.<br />
Immunotherapy (ma<strong>in</strong>ly <strong>in</strong>travenous immunoglobul<strong>in</strong>s or corticosteroids) seemed to<br />
accelerate recovery. Paraneoplastic OMS had a more severe cl<strong>in</strong>ical course, despite<br />
treatment with <strong>in</strong>travenous immunoglobul<strong>in</strong>s or corticosteroids, and was the cause of<br />
death <strong>in</strong> five patients whose tumors were not treated. By contrast, the eight patients<br />
whose tumors were treated showed a complete or partial neurologic recovery. The<br />
authors concluded that idiopathic OMS occurs <strong>in</strong> younger patients, the cl<strong>in</strong>ical<br />
evolution is more benign, and the effect of immunotherapy appears more effective<br />
than <strong>in</strong> paraneoplastic OMS. In patients aged 50 years and older with OMS who<br />
develop encephalopathy, early diagnosis and treatment of a probable underly<strong>in</strong>g<br />
tumour, usually SCLC, is <strong>in</strong>dicated to <strong>in</strong>crease the chances of neurologic recovery. At<br />
present, there are no immunologic markers to identify the adult patients with<br />
paraneoplastic OMS.<br />
The evaluation of ocular flutter and opsoclonus depends on the age of the patient and<br />
the cl<strong>in</strong>ical circumstances. In children or adults with prodromal symptoms and signs of<br />
systemic or CNS <strong>in</strong>fection, neuroimag<strong>in</strong>g (preferably MRI) followed by sp<strong>in</strong>al tap are<br />
warranted (class IV, level C). In children without apparent <strong>in</strong>fection, a search for occult<br />
neuroblastoma is <strong>in</strong>dicated, with studies variably <strong>in</strong>clud<strong>in</strong>g chest x-ray, CT, or MRI of<br />
chest and abdomen, skeletal survey, <strong>in</strong>travenous pyelogram, bone marrow biopsy, and<br />
determ<strong>in</strong>ation of ur<strong>in</strong>ary catecholam<strong>in</strong>es (class III–IV, level C). In adults, without signs<br />
of <strong>in</strong>fection, an occult malignancy should be sought with workup <strong>in</strong>clud<strong>in</strong>g gynecologic<br />
exam<strong>in</strong>ation; chest x-ray; CT or MRI of the chest, abdomen, and pelvis; mammogram;<br />
possible bone marrow biopsy; and serum anti-Ri antibody (ANNA-2), anti-Hu antibody,<br />
and antiamphiphys<strong>in</strong> antibody studies (class III–IV, level C). MRI of the bra<strong>in</strong> is<br />
warranted for most patients (class III–IV, level B). A history of drug or toxic exposures<br />
should be sought <strong>in</strong> all patients. In some circumstances, other blood studies to be<br />
considered <strong>in</strong>clude serum osmolality, HIV titers, and angiotens<strong>in</strong>-convert<strong>in</strong>g enzyme<br />
(ACE) levels for sarcoidosis (class IV, level C).<br />
The treatment of ocular flutter and opsoclonus is <strong>in</strong>itially directed at the underly<strong>in</strong>g<br />
etiology (e.g., underly<strong>in</strong>g neoplasm) as some patients improve with tumor removal<br />
(Vigliani, 2001). Symptomatic reduction of the eye movements has been reported with<br />
adrenocorticotropic hormone (ACTH), corticosteroids, clonazepam, baclofen, propranolol,<br />
thiam<strong>in</strong>e, reserp<strong>in</strong>e, and valproic acid (Leigh, 1991). High-dose <strong>in</strong>travenous<br />
immunoglobul<strong>in</strong> has been successful <strong>in</strong> several cases (Fisher, 1994; Pless, 1996).<br />
The evaluation and treatment of opsoclonus are outl<strong>in</strong>ed <strong>in</strong> Figure 17–6.
396 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 17–6. Evaluation of opsoclonus.<br />
What Are the Characteristics of Spontaneous<br />
Eye Oscillations <strong>in</strong> Stuporous and Comatose<br />
Patients?<br />
In comatose patients, if the bra<strong>in</strong>stem is <strong>in</strong>tact, the eyelids are closed, and the eyes,<br />
slightly divergent, drift slowly from side to side (rov<strong>in</strong>g eye movements). The rov<strong>in</strong>g<br />
eye movements of light coma cannot be voluntarily executed and are therefore
<strong>in</strong>compatible with the diagnosis of feigned unresponsiveness. As coma deepens, rov<strong>in</strong>g<br />
eye movements disappear.<br />
Other spontaneous eye movements seen <strong>in</strong> comatose patients <strong>in</strong>clude the follow<strong>in</strong>g<br />
(see Table 17–6):<br />
1. Short-cycle periodic alternat<strong>in</strong>g gaze (P<strong>in</strong>g-Pong gaze), which consists of rov<strong>in</strong>g of<br />
the eyes from one extreme of horizontal gaze to the other and back with each<br />
oscillat<strong>in</strong>g cycle tak<strong>in</strong>g 2.5 to 8 seconds (Ishikawa, 1993). This f<strong>in</strong>d<strong>in</strong>g usually<br />
<strong>in</strong>dicates bilateral cerebral damage (e.g., bilateral cerebral <strong>in</strong>farcts) with an <strong>in</strong>tact<br />
bra<strong>in</strong>stem, but has also been described with posterior fossa hemorrhage, basal<br />
ganglia <strong>in</strong>farcts, bilateral cerebral peduncle lesions, hydrocephalus, hepatic encephalopathy,<br />
diffuse cerebral hypoxia, carbon monoxide <strong>in</strong>toxication, and overdose<br />
of the monoam<strong>in</strong>e oxidase <strong>in</strong>hibitor trancyprom<strong>in</strong>e (Crevits, 1992; Ishikawa, 1993;<br />
Johkura, 1998). The disorder may occasionally occur <strong>in</strong> coma with no structural<br />
hemispheric lesion. Saccadic (versus smooth waveform) p<strong>in</strong>g-pong gaze may<br />
<strong>in</strong>dicate less extensive bra<strong>in</strong> damage (Johkura, 1998).<br />
2. P<strong>in</strong>g-pong gaze must be differentiated from periodic alternat<strong>in</strong>g gaze deviation,<br />
which is an alternat<strong>in</strong>g horizontal conjugate gaze deviation last<strong>in</strong>g 1 to 2 m<strong>in</strong>utes <strong>in</strong><br />
each direction. Periodic alternat<strong>in</strong>g gaze deviation usually occurs <strong>in</strong> alert patients<br />
with structural lesions <strong>in</strong>volv<strong>in</strong>g the cerebellum and bra<strong>in</strong>stem, such as the Arnold-<br />
Chiari malformation or medulloblastoma, but has been described <strong>in</strong> obtunded or<br />
comatose patients with hepatic encephalopathy (Averbuch-Heller, 1995a). Jakob-<br />
Table 17–6. Spontaneous Eye Movements <strong>in</strong> Comatose Patients<br />
Movement Description Localization<br />
Periodic alternat<strong>in</strong>g gaze<br />
(p<strong>in</strong>g-pong gaze)<br />
Repetitive divergence Slow deviation out,<br />
Monocular nystagmoid Vertical, horizontal,<br />
Nystagmus and Other Ocular Oscillations 397<br />
Cyclic horizontal rov<strong>in</strong>g Bilateral cerebral damage,<br />
rarely posterior fossa lesion,<br />
hepatic, hypoxic,<br />
carbon monoxide,<br />
drug <strong>in</strong>toxication<br />
Metabolic encephalopathy<br />
rapid return to primary<br />
Middle or low pont<strong>in</strong>e lesion<br />
or rotatory movements<br />
Status epilepticus Small-amplitude vertical<br />
(occasionally horizontal)<br />
movements<br />
Diffuse encephalopathy (hypoxic)<br />
Ocular bobb<strong>in</strong>g Fast down, slow up Pont<strong>in</strong>e lesion, extraaxial<br />
posterior fossa mass, diffuse<br />
encephalopathy<br />
Inverse ocular bobb<strong>in</strong>g Slow down, fast up <strong>An</strong>oxia, post–status epilepticus<br />
(ocular dipp<strong>in</strong>g)<br />
(diffuse encephalopathy)<br />
Reverse ocular bobb<strong>in</strong>g Fast up, slow down Diffuse encephalopathy,<br />
rarely pont<strong>in</strong>e<br />
Slow-upward ocular Slow up, fast down Diffuse encephalopathy bobb<strong>in</strong>g<br />
Pretectal pseudobobb<strong>in</strong>g ‘‘V-pattern’’; down and <strong>in</strong> Pretectal (hydrocephalus)<br />
Vertical ocular myoclonus Pendular, vertical isolated Pont<strong>in</strong>e<br />
Source: Modified from Brazis, 2001, with permission from Lipp<strong>in</strong>cott Williams & Wilk<strong>in</strong>s.
398 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Creutzfeldt disease may be associated with geotropic ocular deviation with skew<br />
deviation and absence of saccades (Grant, 1993). When the head is turned to one<br />
side, the eyes very slowly deviate to that side, while the abduct<strong>in</strong>g eye moves<br />
upward and the adduct<strong>in</strong>g eye moves downward. These spontaneous ocular<br />
movements are slow with no saccadic component.<br />
3. Repetitive divergence is rarely seen <strong>in</strong> patients with coma from metabolic encephalopathy<br />
(e.g., hepatic encephalopathy) (Noda, 1987). With this disorder, the eyes are<br />
midposition or slightly divergent at rest. They then slowly deviate out, become fully<br />
deviated for a brief period, and then rapidly return to primary position before<br />
repeat<strong>in</strong>g the cycle. These motions are synchronous <strong>in</strong> the two eyes.<br />
4. Nystagmoid jerk<strong>in</strong>g of a s<strong>in</strong>gle eye, <strong>in</strong> a vertical, horizontal, or rotatory fashion,<br />
may occur with mid- to lower pont<strong>in</strong>e damage. Pont<strong>in</strong>e lesions occasionally give<br />
rise to disconjugate rotatory and vertical movements of the eyes, <strong>in</strong> which one eye<br />
may rise and <strong>in</strong>tort as the other falls and extorts. This type of movement should not<br />
be confused with seesaw nystagmus, which is very seldom seen <strong>in</strong> comatose<br />
patients.<br />
5. Electrographic status epilepticus without appendicular motor manifestations, due<br />
to anoxia, may result <strong>in</strong> brisk, small-amplitude, ma<strong>in</strong>ly vertical (occasionally<br />
horizontal) eye movements detectable by passive lid elevation (Simon, 1986).<br />
6. Ocular bobb<strong>in</strong>g refers to <strong>in</strong>termittent, often conjugate, brisk, bilateral downward<br />
movement of the eyes with slow return to midposition. Ocular bobb<strong>in</strong>g has been<br />
associated with <strong>in</strong>tr<strong>in</strong>sic pont<strong>in</strong>e lesions (e.g., hemorrhage, tumor, <strong>in</strong>farction, central<br />
pont<strong>in</strong>e myel<strong>in</strong>olysis, etc.), extraaxial posterior fossa masses (e.g., aneurysm rupture<br />
or cerebellar hemorrhage or <strong>in</strong>farction), diffuse encephalitis, Jakob-Creutzfeldt<br />
disease, and toxic-metabolic encephalopathies (e.g., acute organophosphate poison<strong>in</strong>g).<br />
‘‘Typical’’ ocular bobb<strong>in</strong>g, which is associated with preserved horizontal eye<br />
movements, is thought to be specific but not pathognomonic of acute pont<strong>in</strong>e <strong>in</strong>jury,<br />
whereas ‘‘atypical’’ ocular bobb<strong>in</strong>g, which is associated with absent horizontal eye<br />
movements, is thought to be less helpful <strong>in</strong> predict<strong>in</strong>g the site of abnormality.<br />
Monocular bobb<strong>in</strong>g (paretic bobb<strong>in</strong>g), which consists of a quick downward movement<br />
of one eye and <strong>in</strong>torsion or no movement <strong>in</strong> the other eye, may occur if there is<br />
a coexistent unilateral fascicular oculomotor nerve palsy (Dehaene, 1993). Disconjugate<br />
ocular bobb<strong>in</strong>g, with movements <strong>in</strong>volv<strong>in</strong>g sometimes one eye and sometimes<br />
the other, may also occur without oculomotor nerve palsy (Gaymard, 1993).<br />
7. Inverse ocular bobb<strong>in</strong>g (ocular dipp<strong>in</strong>g or fast-upward ocular bobb<strong>in</strong>g) consists of a<br />
slow-downward eye movement with fast return to midposition, which may occur <strong>in</strong><br />
anoxic coma or after prolonged status epilepticus. Ocular dipp<strong>in</strong>g has also been<br />
described associated with deafness <strong>in</strong> a patient with p<strong>in</strong>ealoblastoma (Toshniwal,<br />
1986). Inverse=reverse ocular bobb<strong>in</strong>g consists of <strong>in</strong>verse ocular bobb<strong>in</strong>g <strong>in</strong> which<br />
the eyes do not stop on rapidly return<strong>in</strong>g to primary position but shoot <strong>in</strong>to upgaze<br />
and slowly return to midposition.<br />
8. Reverse ocular bobb<strong>in</strong>g (fast-upward ocular bobb<strong>in</strong>g) consists of fast-upward eye<br />
movement with a slow return to midposition, which may occur <strong>in</strong> patients with<br />
metabolic encephalopathy, viral encephalitis, or pont<strong>in</strong>e hemorrhage. It has been<br />
described with coma due to comb<strong>in</strong>ed phenothiaz<strong>in</strong>e and benzodiazep<strong>in</strong>e poison<strong>in</strong>g<br />
(Lennox, 1993). Occasionally ocular bobb<strong>in</strong>g, ocular dipp<strong>in</strong>g, and reverse<br />
bobb<strong>in</strong>g may occur at different times <strong>in</strong> the same patient.
9. Slow-upward ocular bobb<strong>in</strong>g (converse ocular bobb<strong>in</strong>g or reverse ocular dipp<strong>in</strong>g) is<br />
characterized by slow-upward eye movements followed by a fast return to<br />
midposition. This eye movement disorder has been described with pont<strong>in</strong>e <strong>in</strong>farction<br />
and with metabolic or viral encephalopathy (i.e., diffuse cerebral dysfunction).<br />
10. Pretectal pseudobobb<strong>in</strong>g has been described with acute hydrocephalus (Keane,<br />
1985) and consists of arrhythmic, repetitive downward and <strong>in</strong>ward (‘‘V-pattern’’)<br />
eye movements at a rate rang<strong>in</strong>g from one per 3 seconds to two per second and an<br />
amplitude of one fifth to one half of the full voluntary range. These movements may<br />
be mistaken for ocular bobb<strong>in</strong>g, but their V pattern, their faster rate, and their<br />
pretectal rather than pont<strong>in</strong>e-associated signs dist<strong>in</strong>guish them from true pont<strong>in</strong>e<br />
bobb<strong>in</strong>g. Thus, patients with pretectal pseudobobb<strong>in</strong>g may have abnormal pupillary<br />
light reactions, <strong>in</strong>tact horizontal eye movements, open and often retracted<br />
eyelids, a bl<strong>in</strong>k frequently preced<strong>in</strong>g each eye movement, and a mute or stuporous<br />
rather than a comatose state. Pretectal pseudobobb<strong>in</strong>g probably represents a variety<br />
of convergence nystagmus, and its presence usually <strong>in</strong>dicates the need for prompt<br />
surgical attention (e.g., hydrocephalus decompression) (Keane, 1985). Pretectal<br />
pseudobobb<strong>in</strong>g has also been described with an expand<strong>in</strong>g posterior fossa cyst<br />
(Komiyama, 1999b). It is possible that some cases of ‘‘ocular bobb<strong>in</strong>g’’ associated<br />
with thalamic hemorrhage or tentorial herniation may actually be cases of pretectal<br />
pseudobobb<strong>in</strong>g.<br />
11. Vertical ocular myoclonus consists of pendular, vertical isolated movements of the<br />
eyes noted <strong>in</strong> patients either locked-<strong>in</strong> or comatose after severe pont<strong>in</strong>e strokes<br />
(Keane, 1986). Their frequency is 2 Hz, and other rhythmic body movements at a<br />
similar frequency occur after a 6-week to 9-month delay. These movements are<br />
generally associated with palatal myoclonus (palatal tremor), with which they share<br />
a common mechanism (Keane, 1986).<br />
References<br />
Nystagmus and Other Ocular Oscillations 399<br />
Abadi RV, Pascal E. (1994). Periodic alternat<strong>in</strong>g nystagmus <strong>in</strong> humans with alb<strong>in</strong>ism. Invest Ophthalmol Vis Sci<br />
35:4080–4086.<br />
Abrusow V, Strupp M, Brandt T. (1998). Amiodarone-<strong>in</strong>duced severe prolonged head-positional vertigo and<br />
vomit<strong>in</strong>g. <strong>Neuro</strong>logy 51:917.<br />
Adler CH, Galetta SL. (1990). Oculo-facial-skeletal myorhythmia <strong>in</strong> Whipple disease: treatment with ceftriaxone.<br />
<strong>An</strong>n Intern Med 112:467–469.<br />
Aggarwal A, Williams D. (1997). Opsoclonus as a paraneoplastic manifestation of pancreatic carc<strong>in</strong>oma. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 63:687–688.<br />
Albera R, Luda E, Canale G, et al. (1997). Cyclospor<strong>in</strong>e A as a possible cause of upbeat<strong>in</strong>g nystagmus. <strong>Neuro</strong><strong>ophthalmology</strong><br />
17:163–168.<br />
Arnoldi KA, Tychsen L. (1995). Prevalence of <strong>in</strong>tracranial lesions <strong>in</strong> children <strong>in</strong>itially diagnosed with disconjugate<br />
nystagmus (spasmus nutans). J Pediatr Ophthalmol Strabismus 32:296–301.<br />
Ashe J, Ha<strong>in</strong> TC, Zee DS, Schatz NJ. (1991). Microsaccadic flutter. Bra<strong>in</strong> 114:461–472.<br />
Atilla H, Erkam N, Isikçelik Y. (1999). Surgical treatment <strong>in</strong> nystagmus. Eye 13:11–15.<br />
Averbuch-Heller L, Kori AA, Rottach KG, et al. (1996). Dysfunction of pont<strong>in</strong>e omnipause neurons causes<br />
impaired fixation: macrosaccadic oscillations with a unilateral pont<strong>in</strong>e lesion. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:99–106.<br />
Averbuch-Heller L, Me<strong>in</strong>er Z. (1995a). Reversible periodic alternat<strong>in</strong>g gaze deviation <strong>in</strong> hepatic encephalopathy.<br />
<strong>Neuro</strong>logy 45:191–192.<br />
Averbuch-Heller L, Stahl JS, Hlav<strong>in</strong> ML, Leigh RJ. (1999). Square-wave jerks <strong>in</strong>duced by pallidotomy <strong>in</strong><br />
park<strong>in</strong>sonism patients. <strong>Neuro</strong>logy 52:185–188.
400 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Averbuch-Heller L, Tusa RJ, Fuhry L, et al. (1997). A double-bl<strong>in</strong>d controlled study of Gabapent<strong>in</strong> and Baclofen as<br />
treatment for acquired nystagmus. <strong>An</strong>n <strong>Neuro</strong>l 41:818–85.<br />
Averbuch-Heller L, Zivotofsky AZ, Das VE, et al. (1995b). Investigations of the pathogenesis of acquired pendular<br />
nystagmus. Bra<strong>in</strong> 118:369–378.<br />
Baloh RW, Jacobson K, Honrubia V. (1993). Horizontal semicircular canal variant of benign positional vertigo.<br />
<strong>Neuro</strong>logy 43:2542–2549.<br />
Baloh RW, W<strong>in</strong>der A. (1991). Acetazolamide-responsive vestibulo-cerebellar syndrome: cl<strong>in</strong>ical and oculographic<br />
features. <strong>Neuro</strong>logy 41:429–433.<br />
Baloh RW, Yue Q, Furman JM, Nelson SF. (1997). Familial episodic ataxia: cl<strong>in</strong>ical heterogeneity <strong>in</strong> four families<br />
l<strong>in</strong>ked to chromosome 19p. <strong>An</strong>n <strong>Neuro</strong>l 41:8–16.<br />
Baloh RW, Yue Q, Jacobson KM, Honrubia V. (1995). Persistent direction-chang<strong>in</strong>g positional nystagmus: another<br />
variant of benign positional nystagmus? <strong>Neuro</strong>logy 45:1297–1301.<br />
Band<strong>in</strong>i F, Castello E, Mazzella L, et al. (2001). Gabapent<strong>in</strong> but not vigabatr<strong>in</strong> is effective <strong>in</strong> the treatment of<br />
acquired nystagmus <strong>in</strong> multiple sclerosis: how valid is the GABAergic hypothesis? J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 71:107–110.<br />
Barton JJS. (1995). Bl<strong>in</strong>k- and saccade-<strong>in</strong>duced seesaw nystagmus. <strong>Neuro</strong>logy 45:831.<br />
Barton JJS, Cox TA. (1993). Acquired pendular nystagmus <strong>in</strong> multiple sclerosis: cl<strong>in</strong>ical observations and role of<br />
optic neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 56:262–267.<br />
Barton JJS, Cox TA, Digre K. (1999). Acquired convergence-evoked pendular nystagmus <strong>in</strong> multiple sclerosis.<br />
J <strong>Neuro</strong>-ophthalmol 19:34–38.<br />
Barton JJS, Huaman AG, Sharpe JA. (1994). Muscar<strong>in</strong>ic antagonists <strong>in</strong> the treatment of acquired pendular<br />
nystagmus: a double-bl<strong>in</strong>d, randomized trial of three <strong>in</strong>travenous drugs. <strong>An</strong>n <strong>Neuro</strong>l 35:319–325.<br />
Bataller L, Graus F, Saiz A, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> outcome <strong>in</strong> adult onset idiopathic or paraneoplastic opsoclonusmyoclonus.<br />
Bra<strong>in</strong> 124:437–443.<br />
Bennett JL, Galetta SL, Frohman LP, et al. (1999). <strong>Neuro</strong>-ophthalmologic manifestations of a paraneoplastic<br />
syndrome and testicular carc<strong>in</strong>oma. <strong>Neuro</strong>logy 52:864–867.<br />
Bertholon P, Bronste<strong>in</strong> AM, Davies RA, et al. (2002). Positional down beat<strong>in</strong>g nystagmus <strong>in</strong> 50 patients: cerebellar<br />
disorders and possible anterior semicircular canalithiasis. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 72:366–372.<br />
Bilska C, Pociej-Zero M, Krzystkowa KM. (1995). Surgical treatment of congenital nystagmus <strong>in</strong> 463 children<br />
(Polish). Kl<strong>in</strong> Oczna 97:140–141.<br />
Brandt T. (1990). Positional and position<strong>in</strong>g vertigo and nystagmus. J <strong>Neuro</strong>l Sci 95:3–28.<br />
Brandt T. (1991). Man <strong>in</strong> motion. Historical and cl<strong>in</strong>ical aspects of vestibular function. Bra<strong>in</strong> 114:2159–2174.<br />
Brandt T, Strupp M. (1997). Episodic ataxia type 1 and 2 (familial periodic ataxia=vertigo). Audiol <strong>Neuro</strong>-otol<br />
2:373–383.<br />
Brazis PW, Miller NR, Henderer JD, Lee AG. (1994). The natural history and results of treatment of superior<br />
oblique myokymia. Arch Ophthalmol 112:1063–1067.<br />
Brazis PW, Masdeu JC, Biller J. (2001). Localization <strong>in</strong> <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>logy. 4th ed. Philadelphia, Lipp<strong>in</strong>cott Williams<br />
& Wilk<strong>in</strong>s.<br />
Bronste<strong>in</strong> AM, Gresty MA, Mossman SS. (1992). Pendular pseudonystagmus aris<strong>in</strong>g as a comb<strong>in</strong>ation of head<br />
tremor and vestibular failure. <strong>Neuro</strong>logy 42:1527–1531.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1991). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. St Louis, Mosby<br />
Yearbook, pp. 289–320.<br />
Buttner U, Helmchen C, Buttner-Ennever JA. (1995). The localiz<strong>in</strong>g value of nystagmus <strong>in</strong> bra<strong>in</strong>stem disorders.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 15:283–290.<br />
Carruthers J. (1995). The treatment of congenital nystagmus with Botox. J Pediatr Ophthalmol Strabismus<br />
32:306–308.<br />
Cav<strong>in</strong>ess JN, Forsyth PA, Layton DD, McPhee TJ. (1995). The movement disorder of adult opsoclonus. Mov Dis<br />
10:22–27.<br />
Chan T, Logan P, Eustace P. (1991). Intermittent downbeat nystagmus secondary to vermian arachnoid cyst with<br />
associated obstructive hydrocephalus. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 11:293–296.<br />
Cibis GW, Fitzgerald KM. (1993). Electroret<strong>in</strong>ography <strong>in</strong> congenital idiopathic nystagmus. Pediatr <strong>Neuro</strong>l<br />
9:369–371.<br />
Coch<strong>in</strong> JP, Hannequ<strong>in</strong> D, Do Marcol<strong>in</strong>o C, et al. (1995). Intermittent seesaw nystagmus successfully treated with<br />
clonazepam (French). Rev <strong>Neuro</strong>l (Paris) 151:60–62.<br />
Connolly AM, Pestronk A, Mehta S, et al. (1997). Serum autoantibodies <strong>in</strong> childhood opsoclonus-myoclonus<br />
syndrome: an analysis of antigenic targets <strong>in</strong> neural tissue. J Pediatr 130:878–884.<br />
Corcia P, De Toffol B, Hommet C, et al. (1997). Paraneoplastic opsoclonus associated with cancer of the gall<br />
bladder. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:293.
Nystagmus and Other Ocular Oscillations 401<br />
Crevits L, Decruyenaere J. (1992). ‘‘P<strong>in</strong>g-pong’’ gaze. <strong>Neuro</strong>-<strong>ophthalmology</strong> 12:121–123.<br />
Davey K, Kowal L, Fril<strong>in</strong>g R, et al. (1998) The Heimann-Bielschowlsky phenomenon: dissociated vertical<br />
nystagmus. Aust NZ J Ophthalmol 26:237–240.<br />
Davis BJ. (2000). Voluntary nystagmus as a component of a nonepileptic seizure. <strong>Neuro</strong>logy 55:1937.<br />
De la Meilleure G, Dehaene I, Depondt M, et al. (1996). Benign paroxysmal positional vertigo of the horizontal<br />
canal. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 60:68–71.<br />
de Sa LC, Good WV, Hoyt CS. (1992). Surgical management of myokymia of the superior oblique muscle.<br />
Am J Ophthalmol 114:693–696.<br />
Dehaene I, Lammens M, Marchau M. (1993). Paretic ocular bobb<strong>in</strong>g. A cl<strong>in</strong>icopathological study of two cases.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 13:143–146.<br />
Dell’Osso LF, Weissman BM, Leigh RJ, et al. (1993). Hereditary congenital nystagmus and gaze-hold<strong>in</strong>g failure:<br />
the role of the neural <strong>in</strong>tegrator. <strong>Neuro</strong>logy 43:1741–1749.<br />
Dieterich M, Brandt T. (1999). Episodic vertigo related to migra<strong>in</strong>e (90 cases): vestibular migra<strong>in</strong>e? J <strong>Neuro</strong>l<br />
246:883–892.<br />
Dieterich M, Straube A, Brandt T, et al. (1991). The effects of baclofen and chol<strong>in</strong>ergic drugs on upbeat and<br />
downbeat nystagmus. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:627–632.<br />
Du Pasquier R, V<strong>in</strong>gerhoets F, Safran AB, Landis T. (1998). Periodic downbeat nystagmus. <strong>Neuro</strong>logy 51:1478–<br />
1480.<br />
Eggenberger E, Cornblath W, Stewart DH. (2001). Oculopalatal tremor with tardive ataxia. J <strong>Neuro</strong>-Ophthalmol<br />
21:83–86.<br />
Epste<strong>in</strong> JA, Moster ML, Spiritos M. (2001). Seesaw nystagmus follow<strong>in</strong>g whole bra<strong>in</strong> irradiation and <strong>in</strong>trathecal<br />
methotrexate. J <strong>Neuro</strong>-Ophthalmol 21:264–265.<br />
Evans BJ, Evans BV, Jordahl-Moroz J, Nabee M. (1998). Randomized double-masked placebo-controlled trial of a<br />
treatment for congenital nystagmus. Vis Res 38:2193–2202.<br />
Fisher CM. (1990). Ocular flutter. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:155–156.<br />
Fisher PG, Wechsler DS, S<strong>in</strong>ger HS. (1994). <strong>An</strong>ti-Hu antibody <strong>in</strong> a neuroblastoma-associated paraneoplastic<br />
syndrome. Pediatr <strong>Neuro</strong>l 10:309–312.<br />
FitzGibbon EJ, Calvert PC, Dieterich M, et al. (1996). Torsional nystagmus dur<strong>in</strong>g vertical pursuit. J <strong>Neuro</strong>ophthamol<br />
16:79–90.<br />
Friedman DI, Jankovic J, McCrary JA 3rd. (1992). <strong>Neuro</strong>-ophthalmic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> progressive supranuclear palsy.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:104–109.<br />
Fujikane M, Katayama S, Hirata K, Sunami S. (1992). Central diabetes <strong>in</strong>sipidus complicated with upbeat<br />
nystagmus and cerebellar ataxia (Japanese). R<strong>in</strong>sho Sh<strong>in</strong>keigaku 32:68–72.<br />
Furman JM, Cass SP. (1999). Benign paroxysmal positional vertigo. N Engl J Med 341:1590–1596.<br />
Furman JM, Crumr<strong>in</strong>e PK, Re<strong>in</strong>muth OM. (1990a). Epileptic nystagmus. <strong>An</strong>n <strong>Neuro</strong>l 27:686–688.<br />
Furman JMR, Wall C III, Pang D. (1990b). Vestibular function <strong>in</strong> periodic alternat<strong>in</strong>g nystagmus. Bra<strong>in</strong><br />
113:1425–1439.<br />
Gans MS, Melmed CA. (1990). Downbeat nystagmus associated with dolichoectasia of the vertebrobasilar artery.<br />
Arch <strong>Neuro</strong>l 47:843.<br />
Gaymard B. (1993). Disconjugate ocular bobb<strong>in</strong>g. <strong>Neuro</strong>logy 43:2151.<br />
Geis TC, Newman NJ, Dawson RC. (1996). Superior oblique myokymia associated with a dural arteriovenous<br />
fistula. J <strong>Neuro</strong>-ophthalmol 16:41–43.<br />
Gire C, Somma-Mauvais H, Nicaise C, et al. (2001). Epileptic nystagmus: electrocl<strong>in</strong>ical study of a case. Epileptic<br />
Dis 3:33–37.<br />
Gizzi M, Randolph S, Perakis A. (1990). Ocular flutter <strong>in</strong> vidarab<strong>in</strong>e toxicity. Am J Ophthalmol 109:105.<br />
Goldberg RT. (1978). Vertical pendular nystagmus <strong>in</strong> chronic myositis of medial and lateral rectus. <strong>An</strong>n Ophthalmol<br />
10:1697–1702.<br />
Goldblum TA, Effron LA. (1994). Upbeat nystagmus associated with tonic downward deviation <strong>in</strong> healthy<br />
neonates. J Pediatr Ophthalmol Strabismus 31:334–335.<br />
Good WV, Brodsky MC, Hoyt CS, Ahn JC. (1990). Upbeat<strong>in</strong>g nystagmus <strong>in</strong> <strong>in</strong>fants: a sign of anterior visual<br />
pathway disease. B<strong>in</strong>ocular Vis Q 5:13–18.<br />
Good WV, Koch TS, Jan JE. (1993). Monocular nystagmus caused by unilateral anterior visual-pathway lesions.<br />
Dev Med Child <strong>Neuro</strong>l 35:1106–1110.<br />
Gottlob I, Helbl<strong>in</strong>g A. (1999). Nystagmus mimick<strong>in</strong>g spasmus nutans as the present<strong>in</strong>g sign of Bardet-Biedl<br />
syndrome. Am J Ophthalmol 128:770–772.<br />
Gottlob I, Wizov SS, Re<strong>in</strong>cke RD. (1995a). Quantitative eye and head movement record<strong>in</strong>gs of ret<strong>in</strong>al disease<br />
mimick<strong>in</strong>g spasmus nutans. Am J Ophthalmol 119:374–376.
402 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Gottlob I, Wizov SS, Re<strong>in</strong>cke RD. (1995b). Spasmus nutans. A long-term follow-up. Invest Ophthalmol Vis Sci<br />
36:2768–2771.<br />
Gottlob I, Zubcov A, Catalan RA, et al. (1990). Signs dist<strong>in</strong>guish<strong>in</strong>g spasmus nutans (with and without central<br />
nervous system lesions) from <strong>in</strong>fantile nystagmus. Ophthalmology 97:1166–1175.<br />
Gradste<strong>in</strong> L, Re<strong>in</strong>ecke RD, Wizov SS, Goldste<strong>in</strong> HP. (1997). Congenital periodic alternat<strong>in</strong>g nystagmus. Diagnosis<br />
and management. Ophthalmology 104:918–929.<br />
Grant MP, Cohen M, Petersen RB, et al. (1993). Abnormal eye movements <strong>in</strong> Creutzfeldt-Jacob disease. <strong>An</strong>n <strong>Neuro</strong>l<br />
34:192–197.<br />
Gresty MA, Bronste<strong>in</strong> AM, Page NG, Rudge P. (1991). Congenital-type nystagmus emerg<strong>in</strong>g <strong>in</strong> later life. <strong>Neuro</strong>logy<br />
41:653–656.<br />
Gresty MA, Metcalfe T, Timms C, et al. (1992). <strong>Neuro</strong>logy of latent nystagmus. Bra<strong>in</strong> 115:1303–1321.<br />
Haene I, Casselman J. (1993). Left superior oblique myokymia and right superior oblique paralysis due to a<br />
posterior fossa tumor. <strong>Neuro</strong>-<strong>ophthalmology</strong> 13:13–16.<br />
Halmagyi GM, Aw ST, Dehaene I, et al. (1994). Jerk-waveform seesaw nystagmus due to unilateral mesodiencephalic<br />
lesion. Bra<strong>in</strong> 117:789–803.<br />
Halmagyi GM, Hoyt WF. (1991). Seesaw nystagmus due to unilateral mesodiencephalic lesion. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
11:79–84.<br />
Hammack JE, Kotanides H, Rosenblum MK, Posner JB. (1992). Paraneoplastic cerebellar degeneration. II. <strong>Cl<strong>in</strong>ical</strong><br />
and immunologic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> 21 patients with Hodgk<strong>in</strong>’s disease. <strong>Neuro</strong>logy 42:1938–1943.<br />
Harada H, Tamaoka A, Watanabe M, et al. (1998). Downbeat nystagmus <strong>in</strong> two sibl<strong>in</strong>gs with sp<strong>in</strong>ocerebellar<br />
ataxia type 6 (SCA 6). J <strong>Neuro</strong>l Sci 160:161–163.<br />
Harris CM, Boyd S, Ch<strong>in</strong>g K, et al. (1997). Epileptic nystagmus <strong>in</strong> <strong>in</strong>fancy. J <strong>Neuro</strong>l Sci 151:111–114.<br />
Hashimoto M, Ohtsuka K, Hoyt WF. (2001). Vascular compression as a cause of superior oblique myokymia<br />
disclosed by th<strong>in</strong>-sliced magnetic resonance imag<strong>in</strong>g. Am J Ophthalmol 131:676–677.<br />
Hattori T, Takaya Y, Tsuboi Y, Hirayama K. (1993). Opsoclonus show<strong>in</strong>g only dur<strong>in</strong>g eye closure <strong>in</strong> hereditary<br />
cerebellar ataxia. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 56:1037–1038.<br />
Hayakawa Y, Takagi M, Hasebe H, et al. (2000). A case of superior oblique myokymia observed by an imageanalysis<br />
system. J <strong>Neuro</strong>-Ophthalmol 20:163–165.<br />
Helmchen C, Glasauer S, Bartl K, et al. (1996). Contralesionally beat<strong>in</strong>g torsional nystagmus <strong>in</strong> a unilateral rostral<br />
midbra<strong>in</strong> lesion. <strong>Neuro</strong>logy 47:482–486.<br />
Helveston EM, Ellis FD, Plager DA. (1991). Large recession of the horizontal recti for treatment of nystagmus.<br />
Ophthalmology 98:1302–1305.<br />
Henderson RD, Wijdicks WF. (2000). Downbeat nystagmus associated with <strong>in</strong>travenous patient-controlled<br />
adm<strong>in</strong>istration of morph<strong>in</strong>e. <strong>An</strong>esth <strong>An</strong>alg 91:691–692.<br />
Herishanu Y, Louzoun Z. (1986). Trihexyphenidyl treatment of vertical pendular nystagmus. <strong>Neuro</strong>logy 36:82–84.<br />
Hertle RW, Dell’Osso LF. (1999). <strong>Cl<strong>in</strong>ical</strong> and ocular motor analysis of congenital nystagmus <strong>in</strong> <strong>in</strong>fancy. JAAPOS<br />
3:70–79.<br />
Hertle RW, FitzGibbon EJ, Avallone JM, et al. (2001). Onset of oscillopsia after visual maturation <strong>in</strong> patients with<br />
congenital nystagmus. Ophthalmology 108:2301–2308.<br />
Himi T, Katura A, Tokuda S, et al. (1995). Downbeat nystagmus with compression of the medulla oblongata by<br />
the dolicoectatic vertebral arteries. Am J Otol 16:377–381.<br />
Hirose G, Kawada J, Tsukada K, et al. (1991). Upbeat nystagmus: cl<strong>in</strong>icopathological and pathophysiological<br />
considerations. J <strong>Neuro</strong>l Sci 105:159–167.<br />
Hirose G, Kawada J, Yoshioka A. (1990). Primary position upbeat nystagmus: cl<strong>in</strong>icopathologic study <strong>in</strong> three<br />
patients. <strong>Neuro</strong>logy 40(suppl):312.<br />
Hirose G, Ogasawara T, Shirakawa T, et al. (1998). Primary position upbeat nystagmus due to unilateral medial<br />
medullary <strong>in</strong>farction. <strong>An</strong>n <strong>Neuro</strong>l 43:403–406.<br />
Honnorat J, Trillet M, <strong>An</strong>to<strong>in</strong>e JC, et al. (1997). Paraneoplastic opsomyoclonus, cerebellar ataxia and encephalopathy<br />
associated with anti-Purk<strong>in</strong>je cell antibodies. J <strong>Neuro</strong>l 244:333–339.<br />
Hormigo A, Rosenblum MK, River ME, Posner JB. (1994). Immunological and pathological study of anti-<br />
Ri-associated encephalopathy. <strong>An</strong>n <strong>Neuro</strong>l 36:896–902<br />
Huygen PLM, Verhagen WIM, Cruysberg JRM, Koch PAM. (1995). Familial congenital periodic alternat<strong>in</strong>g<br />
nystagmus with presumably X-l<strong>in</strong>ked dom<strong>in</strong>ant <strong>in</strong>heritance. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:149–155.<br />
Hwang TL, Still CN, Jones JE. (1995). Reversible downbeat nystagmus and ataxia <strong>in</strong> felbamate <strong>in</strong>toxication.<br />
<strong>Neuro</strong>logy 45:846.<br />
Ishikawa H, Ishikawa S, Mukuno K. (1993). Short-cycle periodic alternat<strong>in</strong>g (p<strong>in</strong>g-pong) gaze. <strong>Neuro</strong>logy<br />
43:1067–1070.
Nystagmus and Other Ocular Oscillations 403<br />
Ito K, Murofuchi T, Mizuno M. (2000). Periodic alternat<strong>in</strong>g nystagmus and congenital nystagmus: similarities <strong>in</strong><br />
possibly <strong>in</strong>herited cases. J Otorh<strong>in</strong>olarnygol Related Specialties 62:53–56.<br />
Jabbari B, Rosenberg M, Scherokman B, et al. (1987). Effectiveness of trihexyphenidyl aga<strong>in</strong>st pendular nystagmus<br />
and palatal myoclonus: evidence of chol<strong>in</strong>ergic dysfunction. Mov Disord 2:93–98.<br />
Jacobson L, Ygge J, Flodmark O. (1998). Nystagmus <strong>in</strong> periventricular leukomalacia. Br J Ophthalmol 82:1026–1032.<br />
Jacome DE, FitzGerald R. (1982). Monocular ictal nystagmus. Arch <strong>Neuro</strong>l 39:653–656.<br />
Janssen JC, Larner AJ, Morris H, et al. (1998). Upbeat nystagmus: cl<strong>in</strong>icoanatomical correlation. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 65:380–381.<br />
Johkura K, Komiyama A, Tobita M, Hasegawa O. (1998). Saccadic p<strong>in</strong>g-pong gaze. J <strong>Neuro</strong>-ophthalmol 18:43–46.<br />
Kam<strong>in</strong>ski HJ, Zee DS, Leigh RJ, et al. (1991). Ocular flutter and ataxia associated with AIDS-related complex.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 11:163–167.<br />
Kanaya T, Nonaka S, Kamito M, et al. (1994). Primary position upbeat nystagmus—localiz<strong>in</strong>g value.<br />
J Otorh<strong>in</strong>olaryngol Related Specialties 56:236–238.<br />
Kaplan PW, Tusa RJ. (1993). <strong>Neuro</strong>physiologic and cl<strong>in</strong>ical correlations of epileptic nystagmus. <strong>Neuro</strong>logy<br />
43:2508–2514.<br />
Keane JR. (1985). Pretectal pseudobobb<strong>in</strong>g: five patients with ‘‘V’’-pattern convergence nystagmus. Arch <strong>Neuro</strong>l<br />
42:592–594.<br />
Keane JR. (1986). Acute vertical ocular myoclonus. <strong>Neuro</strong>logy 36:86–89.<br />
Kerrison JB, Vagefi MR, Barmada MM, Maumenee IH. (1999). Congenital motor nystagmus l<strong>in</strong>ked to Xq26-q27.<br />
Am J Hum Genet 64:600–607.<br />
Komai K, Mimura O, Uyama J, et al. (1992). <strong>Neuro</strong>-ophthalmological evaluation of superior oblique myokymia.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 12:135–140.<br />
Komiyama A, Toda H, Johkura K. (1999a). Edrophonium-<strong>in</strong>duced macrosaccadic oscillations <strong>in</strong> myasthenia<br />
gravis. <strong>An</strong>n <strong>Neuro</strong>l 45:522–525.<br />
Komiyama A, Toda H, Johkura K, et al. (1999b). Pretectal pseudobobb<strong>in</strong>g associated with an expand<strong>in</strong>g posterior<br />
fossa cyst <strong>in</strong> tectocerebellar dysraphia: an electro-oculographic study. J <strong>Neuro</strong>l 146:221–223.<br />
Kori AA, Rob<strong>in</strong> NH, Jacobs JB, et al. (1998). Pendular nystagmus <strong>in</strong> patients with peroxisomal assembly disorder.<br />
Arch <strong>Neuro</strong>l 55:554–558.<br />
Kosmorsky GS, Ellis BD, Fogt N, Leigh RJ. (1995). The treatment of superior oblique myokymia utiliz<strong>in</strong>g the<br />
Harada-Ito procedure. J <strong>Neuro</strong>-ophthalmol 15:142–146.<br />
Koukoulis A, Cimas I, Gómara S. (1998). Paraneoplastic opsoclonus associated with papillary renal cell<br />
carc<strong>in</strong>oma. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 64:137–138.<br />
Krespi Y, Vertichel P, Masson C, Cambier J. (1995). Downbeat nystagmus and vertebrobasilar arterial dolichoectasia<br />
(French). Rev <strong>Neuro</strong>l 151:196–197.<br />
Kushner BJ. (1995). Infantile uniocular bl<strong>in</strong>dness with bilateral nystagmus. A syndrome. Arch Ophthalmol<br />
113:1298–1300.<br />
Lambert SR, Newman NJ. (1993). Ret<strong>in</strong>al disease masquerad<strong>in</strong>g as spasmus nutans. <strong>Neuro</strong>logy 43:1607–1609.<br />
Lawden MC, Bronste<strong>in</strong> AM, Kennard C. (1995). Repetitive paroxysmal nystagmus and vertigo. <strong>Neuro</strong>logy<br />
45:276–280.<br />
Lee AG. (2001). Downbeat nystagmus associated with caudal bra<strong>in</strong>stem compression by the vertebral artery.<br />
J <strong>Neuro</strong>-Ophthalmol 21:219–220.<br />
Lee IS, Lee JB, Kim HS, et al. (2000). Modified Kestenbaum surgery for correction of abnormal head posture <strong>in</strong><br />
<strong>in</strong>fantile nystagmus: outcome <strong>in</strong> 63 patients with graded augmentation. B<strong>in</strong>ocular Vis Strabismus Q 15:53–58.<br />
Lee J, Gresty M. (1993). A case of ‘‘voluntary nystagmus’’ and head tremor. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
56:1321–1322.<br />
Leigh RJ, Averbuch-Heller L, Tomsak RL, et al. (1994). Treatment of abnormal eye movements that impair vision:<br />
strategies based on current concepts of physiology and pharmacology. <strong>An</strong>n <strong>Neuro</strong>l 36:129–141.<br />
Leigh RJ, Burnst<strong>in</strong>e TH, Ruff RL, Kasmer RJ. (1991). The effect of antichol<strong>in</strong>ergic agents upon acquired pendular<br />
nystagmus. A double-bl<strong>in</strong>d study of trihexyphenidyl and tridihexethyl chloride. <strong>Neuro</strong>logy 41:1737–1741.<br />
Leigh RJ, Tomsak RL, Grant MP, et al. (1992). Effectiveness of botul<strong>in</strong>um tox<strong>in</strong> adm<strong>in</strong>istration to abolish acquired<br />
nystagmus. <strong>An</strong>n <strong>Neuro</strong>l 32:633–642.<br />
Leigh RJ, Zee DS. (1999). The <strong>Neuro</strong>logy of Eye Movements. 3rd ed. New York, Oxford University Press.<br />
Lennox G. (1993). Reverse ocular bobb<strong>in</strong>g due to comb<strong>in</strong>ed phenothiaz<strong>in</strong>e and benzodiazep<strong>in</strong>e poison<strong>in</strong>g. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 56:1137–1137.<br />
L<strong>in</strong> CY, Young YH. (1999). <strong>Cl<strong>in</strong>ical</strong> significance of rebound nystagmus. Laryngoscope 109:1803–1805.<br />
Lopez L, Bronste<strong>in</strong> AM, Gresty MA, et al. (1992). Torsional nystagmus. A neuro-otological and MRI study of<br />
thirty-five cases. Bra<strong>in</strong> 115:1107–1124.
404 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Lopez LI, Bronste<strong>in</strong> AM, Gresty MA, et al. (1996). <strong>Cl<strong>in</strong>ical</strong> and MRI correlates <strong>in</strong> 27 patients with acquired<br />
pendular nystagmus. Bra<strong>in</strong> 119:465–472.<br />
Louis ED, Lynch T, Kaufmann P, et al. (1996). Diagnostic guidel<strong>in</strong>es <strong>in</strong> central nervous system Whipple’s disease.<br />
<strong>An</strong>n <strong>Neuro</strong>l 40:561–568.<br />
Luque FA, Furneaux HM, Ferziger R, et al. (1991). <strong>An</strong>ti-Ri: an antibody associated with paraneoplastic opsoclonus<br />
and breast cancer. <strong>An</strong>n <strong>Neuro</strong>l 29:241–251.<br />
Lynch T, Ode J, Fredericks DN, Louis ED, et al. (1997). Polymerase cha<strong>in</strong> reaction-based detection of Tropheryma<br />
whippelii <strong>in</strong> central nervous system Whipple’s disease. <strong>An</strong>n <strong>Neuro</strong>l 42:120–124.<br />
Maas EF, Ashe J, Spiegel P, et al. (1991). Acquired pendular nystagmus <strong>in</strong> toluene addiction. <strong>Neuro</strong>logy 41:282–285.<br />
Martidis A, Yee RD, Azzarelli B, Biller J. (1999). <strong>Neuro</strong>-ophthalmic, radiographic, and pathologic manifestations of<br />
adult-onset Alexander disease. <strong>Neuro</strong>logy 117:256–267.<br />
Massry GG, Bloom JN, Cruz OA. (1996). Convergence nystagmus associated with spasmus nutans. J <strong>Neuro</strong>ophthalmol<br />
16:196–198.<br />
Matsumoto S, Ohyagi Y, Inoue I, et al. (2001). Periodic alternat<strong>in</strong>g nystagmus <strong>in</strong> a patient with MS. <strong>Neuro</strong>logy<br />
56:276–277.<br />
May EF, Truxal AR. (1997). Loss of vision alone may result <strong>in</strong> seesaw nystagmus. J <strong>Neuro</strong>-ophthalmol 17:84–85.<br />
Merk<strong>in</strong>-Zaborsky H, Ifergane G, Frischer S, et al. (2000). Thiam<strong>in</strong>e-responsive acute neurological disorders <strong>in</strong> nonalcoholic<br />
patients. Eur <strong>Neuro</strong>l 45:34–37.<br />
M<strong>in</strong>agar A, Sheremata WA, Tusa RJ. (2001). Perverted head-shak<strong>in</strong>g nystagmus: a possible mechanism. <strong>Neuro</strong>logy<br />
57:887–889.<br />
M<strong>in</strong>or LB, Haslwanter T, Straumann D, Zee DS. (1999). Hyperventilation-<strong>in</strong>duced nystagmus <strong>in</strong> patients with<br />
vestibular schwannoma. <strong>Neuro</strong>logy 53:2158–2168.<br />
Mitchell WG, Snodgrass SR. (1990). Opsoclonus-ataxia due to childhood neural crest tumors: a chronic neurologic<br />
syndrome. J Child <strong>Neuro</strong>l 5:153–158.<br />
Mitoma H, Orimo S, Sodeyama N, Tamaki M. (1996). Paraneoplastic opsoclonus-myoclonus syndrome and<br />
neurofibrosarcoma. Eur <strong>Neuro</strong>l 36:322.<br />
Mizuno M, Kudo Y, Yamane M. (1990). Upbeat nystagmus <strong>in</strong>fluenced by posture: report of two cases. Auris Nasus<br />
Larynx 16:215–221.<br />
Monteiro ML, Sampaio CM. (1993). Lithium-<strong>in</strong>duced downbeat nystagmus <strong>in</strong> a patient with Arnold-Chiari<br />
malformation. Am J Ophthalmol 116:648–649.<br />
Morrow MJ, Sharpe JA, Ranalli PJ. (1990). Superior oblique myokymia associated with a posterior fossa tumor:<br />
oculographic correlation with an idiopathic case. <strong>Neuro</strong>logy 40:367–370.<br />
Mossman SS, Bronste<strong>in</strong> AM, Gresty MA, et al. (1990). Convergence nystagmus associated with Arnold-Chiari<br />
malformation. Arch <strong>Neuro</strong>l 47:357–359.<br />
Moster ML, Schnayder E. (1998). Epileptic periodic alternat<strong>in</strong>g nystagmus. J <strong>Neuro</strong>-ophthalmol 18:292–293.<br />
Mulder AH, Raemaekers JM, Boerman RH, Mattijssen V. (1999). Downbeat nystagmus caused by thiam<strong>in</strong>e<br />
deficiency: an unusual presentation of CNS localization of large cell anaplastic CD 30-positive non-<br />
Hodgk<strong>in</strong>’s lymphoma. <strong>An</strong>n Hematol 78:105–107.<br />
Munro NAR, Gaymard B, Rivaud S, et al. (1993). Upbeat nystagmus <strong>in</strong> a patient with a small medullary <strong>in</strong>farct.<br />
J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 56:1126–1128.<br />
Nakada T, Kwee IL. (1986). Oculopalatal myoclonus. Bra<strong>in</strong> 109:431–441.<br />
Neetens A, Mart<strong>in</strong> JJ. (1983). Superior oblique myokymia <strong>in</strong> a case of adrenoleukodystrophy and <strong>in</strong> a case of lead<br />
<strong>in</strong>toxication. <strong>Neuro</strong>-<strong>ophthalmology</strong> 3:103–107.<br />
Nelson KR, Brenner RP, Carlow T. (1986). Divergent-convergence eye movements and transient eyelid open<strong>in</strong>g<br />
associated with an EEG burst-suppression pattern. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 6:43–46.<br />
Newman SA, Hedges TR, Wall M, Sedwick LA. (1990). Spasmus nutans: or is it? Surv Ophthalmol 34:453–456.<br />
Noda S, Ide K, Umezaki H, et al. (1987). Repetitive divergence. <strong>An</strong>n <strong>Neuro</strong>l 21:109–110.<br />
Oett<strong>in</strong>g WS, Armstrong CM, Holleschau AM, et al. (2000). Evidence for genetic heterogeneity <strong>in</strong> families with<br />
congenital motor nystagmus. Ophthalmic Genet 21:227–233.<br />
Ohkoshi N, Komatsu Y, Mizusawa H, Kanazawa I. (1998). Primary position upbeat nystagmus <strong>in</strong>creased on<br />
downgaze: cl<strong>in</strong>icopathologic study of a patient with multiple sclerosis. <strong>Neuro</strong>logy 50:551–553.<br />
Oishi M, Mochizuki Y. (1997). Ipsilateral oculomotor nerve palsy and contralateral downbeat nystagmus: a<br />
syndrome caused by unilateral paramedian thalamopeduncular <strong>in</strong>farction. J <strong>Neuro</strong>l 244:132–133.<br />
Oliva A, Rosenberg ML. (1990). Convergence-evoked nystagmus. <strong>Neuro</strong>logy 40:161–162.<br />
Olson JL, Jacobson DM. (2001). Comparison of cl<strong>in</strong>ical associations of patients with vasculopathic and idiopathic<br />
downbeat nystagmus. J <strong>Neuro</strong>-Ophthalmol 21:39–41.<br />
Orwitz JI, Galetta SL, Teener JW. (1997). Bilateral trochlear nerve palsy and downbeat nystagmus <strong>in</strong> a patient with<br />
cephalic tetanus. <strong>Neuro</strong>logy 9:894–895.
Nystagmus and Other Ocular Oscillations 405<br />
Pelak VS, Galetta SL, Grossman RI, et al. (1999). Evidence for preganglionic pupillary <strong>in</strong>volvement <strong>in</strong> superficial<br />
siderosis. <strong>Neuro</strong>logy 53:1130–1132.<br />
Peterson K, Rosenblum MK, Kotanides H, Posner JB. (1992). Paraneoplastic cerebellar degeneration. I. A cl<strong>in</strong>ical<br />
analysis of 55 anti-Yo antibody-positive patients. <strong>Neuro</strong>logy 42:1931–1938.<br />
Pieh C, Gottlob I. (2000). Arnold-Chiari malformation and nystagmus of skew. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
69:124–126.<br />
Pless M, Ronthal M. (1996). Treatment of opsoclonus-myoclonus with high-dose <strong>in</strong>travenous immunoglobul<strong>in</strong>.<br />
<strong>Neuro</strong>logy 46:583–584.<br />
Pullic<strong>in</strong>o P, L<strong>in</strong>coff N, Truax BT. (2000). Abnormal vergence with upper bra<strong>in</strong>stem <strong>in</strong>farcts. Pseudoabducens palsy.<br />
<strong>Neuro</strong>logy 55:32–358.<br />
Rambold H, Heide W, Sprenger A, et al. (2001a). Perilymph fistula associated with pulse-synchronous eye<br />
oscillations. <strong>Neuro</strong>logy 56:1769–1771.<br />
Rambold H, Helmchen C, Straube A, Büttner U. (1998). Seesaw nystagmus associated with <strong>in</strong>voluntary torsional<br />
head oscillations. <strong>Neuro</strong>logy 51:831–837.<br />
Rambold H, Kompf D, Helmchen C. (2001b). Convergence retraction nystagmus: a disorder of vergence? <strong>An</strong>n<br />
<strong>Neuro</strong>l 50:677–681.<br />
Rascol O, Sabat<strong>in</strong>i U, S<strong>in</strong>onetta-Moreau M, et al. (1991). Square-wave jerks <strong>in</strong> Park<strong>in</strong>sonian syndromes. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 54:599–602.<br />
Remler BF, Leigh RJ, Osoria I, et al. (1990). The characteristics and mechanisms of visual disturbance associated<br />
with anticonvulsant therapy. <strong>Neuro</strong>logy 40:791–796.<br />
Repka MX, Sav<strong>in</strong>o PJ, Re<strong>in</strong>cke RD. (1994). Treatment of acquired nystagmus with botul<strong>in</strong>um neurotox<strong>in</strong> A. Arch<br />
Ophthalmol 112:1320–1324.<br />
Revol A, Vighetto A, Confavreux C, et al. (1990). Oculo-palatal myoclonus and multiple sclerosis (French).<br />
Rev <strong>Neuro</strong>l 146:518–521.<br />
Rosenberg ML, Glaser JS. (1983). Superior oblique myokymia. <strong>An</strong>n <strong>Neuro</strong>l 13:667–669.<br />
Rosengart A, Hedges TR 3rd, Teal PA, et al. (1993). Intermittent downbeat nystagmus due to vertebral artery<br />
compression. <strong>Neuro</strong>logy 43:216–218.<br />
Rosenthal JG, Selhorst JB. (1987). Cont<strong>in</strong>uous non-rhythmic cycloversion. A possible paraneoplastic disorder.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 7:291–295.<br />
Rousseaux M, Dupard T, Leso<strong>in</strong> F, et al. (1991). Upbeat and downbeat nystagmus occurr<strong>in</strong>g successively <strong>in</strong> a<br />
patient with posterior medullary haemorrhage. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 54:367–369.<br />
Rowlands A, Sgouros S, Williams B. (2000). Ocular manifestations of h<strong>in</strong>dbra<strong>in</strong>-related syr<strong>in</strong>gomyelia and<br />
outcome follow<strong>in</strong>g craniovertebral decompression. Eye 14:884–888.<br />
Ruben S, Dunlop IS, Elston J. (1994a). Retrobulbar botul<strong>in</strong>um tox<strong>in</strong> for treatment of oscillopsia. Aust NZ J<br />
Ophthalmol 22:65–67.<br />
Ruben S, Lee JP, O’Neill D, et al. (1994b). The use of botul<strong>in</strong>um tox<strong>in</strong> for treatment of acquired nystagmus and<br />
oscillopsia. Ophthalmology 101:783–787.<br />
Russell GE, Wick B, Tang RA. (1992). Arnold-Chiari malformation. Optom Vis Sci 69:242–247.<br />
Sakakibara R, Hirayama K, Takaya Y, et al. (1993). Periodic alternat<strong>in</strong>g nystagmus <strong>in</strong> familial congenital cerebellar<br />
ataxia (Japanese). R<strong>in</strong>sho Sh<strong>in</strong>keigaku 33:1–7.<br />
Sakata E, Ohtsu K, Itoh Y. (1991). Positional nystagmus of benign paroxysmal type (BPPN) due to cerebellar<br />
vermis lesions. Pseudo-BPPN. Acta Otolaryngol Suppl 481:254–257.<br />
Samil M, Rosahl SK, Carvalho GA, Krizizk T. (1998). Microvascular decompression for superior oblique<br />
myokymia. Case report. J <strong>Neuro</strong>surg 89:1020–1024.<br />
Samkoff LM, Smith CR. (1994). Seesaw nystagmus <strong>in</strong> a patient with cl<strong>in</strong>ically def<strong>in</strong>ite MS. Eur <strong>Neuro</strong>l 34:228–229.<br />
Sato M, Kurachi T, Arai M, Abel LA. (1999). Voluntary nystagmus associated with accommodation spasm. Jpn J<br />
Ophthalmol 43:1–4.<br />
Schnyder H, Bassetti C. (1996). Bilateral convergence nystagmus <strong>in</strong> unilateral dorsal midbra<strong>in</strong> stroke due to<br />
occlusion of the superior cerebellar artery. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:59–63.<br />
Schon F, Hart PE, Hodgson TL, et al. (1999). Suppression of pendular nystagmus by smok<strong>in</strong>g cannabis <strong>in</strong> a patient<br />
with multiple sclerosis. <strong>Neuro</strong>logy 53:2209–2210.<br />
Schon F, Hodgson TL, Mort D, Kennard C. (2001). Ocular flutter with a localized lesion <strong>in</strong> the paramedian pont<strong>in</strong>e<br />
reticular formation. <strong>An</strong>n <strong>Neuro</strong>l 50:413–416.<br />
Schwartz M, Sharf B, Zidan J. (1990). Opsoclonus as a present<strong>in</strong>g symptom <strong>in</strong> thymic cancer. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 53:534.<br />
Selhorst JB. (1987). Pendular vergence oscillations. In: Ishikawa H, ed. Highlights <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>.<br />
Proceed<strong>in</strong>gs of the Sixth Meet<strong>in</strong>g of the International <strong>Neuro</strong>-Ophthalmology Society. Amsterdam, Aeolus,<br />
pp. 153–162.
406 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Shallo-Hoffmann J, Faldon M, Tusa RJ. (1999). The <strong>in</strong>cidence and waveform characteristics of periodic alternat<strong>in</strong>g<br />
nystagmus <strong>in</strong> congenital nystagmus. Invest Ophthalmol Vis Sci 40:2546–2553.<br />
Shallo-Hoffmann J, Sendler B, Muhlendyck H. (1990). Normal square wave jerks <strong>in</strong> differ<strong>in</strong>g age groups. Invest<br />
Ophthalmol Vis Sci 31:1649–1652.<br />
Shawkat FS, Kriss A, Thompson D, et al. (2000). Vertical or asymmetric nystagmus need not imply neurological<br />
disease. Br J Ophthalmol 84:175–180.<br />
Sheth RD, Horwitz SJ, Aronoff S, et al. (1995). Opsoclonus myoclonus syndrome secondary to Epste<strong>in</strong>-Barr virus<br />
<strong>in</strong>fection. J Child <strong>Neuro</strong>l 10:297–299.<br />
Simon RP, Am<strong>in</strong>off MJ. (1986). Electrographic status epilepticus <strong>in</strong> fatal anoxic coma. <strong>An</strong>n <strong>Neuro</strong>l 20:351–355.<br />
Smith DE, Fitzgerald K, Strass-Isern M, Cibis GW. (2000). Electroret<strong>in</strong>ography is necessary for spasmus nutans<br />
diagnosis. Pediatr <strong>Neuro</strong>l 23:33–36.<br />
Stahl JS, Averbuch-Heller L, Leigh RJ. (2000). Acquired nystagmus. Arch Ophthalmol 118:544–549.<br />
Stahl JS, Rottach KG, Averbuch-Heller L, et al. (1996). A pilot study of gabapent<strong>in</strong> as treatment for acquired<br />
nystagmus. <strong>Neuro</strong>-<strong>ophthalmology</strong> 16:107–113.<br />
Starck M, Albrecht H, Pollmann W, et al. (1997). Drug therapy for acquired pendular nystagmus <strong>in</strong> multiple<br />
sclerosis. J <strong>Neuro</strong>l 244:9–16.<br />
Stark KL, Gibson JB, Hertle RW, Brodsky MC. (2000). Ocular motor signs <strong>in</strong> an <strong>in</strong>fant with carbohydrate-deficient<br />
glycoprote<strong>in</strong> syndrome type 1a. Am J Ophthalmol 130:533–535.<br />
Stearns MQ, S<strong>in</strong>off SE, Rosenberg ML. (1993). Purely torsional nystagmus <strong>in</strong> a patient with stiff-man syndrome: a<br />
case report. <strong>Neuro</strong>logy 43:220.<br />
Stolz SE, Chatrian GE, Spence AM. (1991). Epileptic nystagmus. Epilepsia 32:910–918.<br />
Tabarki B, Palmer P, Lebon P, Sebire G. (1998). Spontaneous recovery of opsoclonus-myoclonus syndrome caused<br />
by enterovirus <strong>in</strong>fection. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 64:406–407.<br />
Talks SJ, Elston JS. (1997). Oculopalatal myoclonus: eye movement studies, MRI f<strong>in</strong>d<strong>in</strong>gs, and the difficulty of<br />
treatment. Eye 11:19–24.<br />
Tison F, Louvet-Giendaj C, Henry P, et al. (1992). Permanent bruxism as a manifestation of the oculo-facial<br />
syndrome related to systemic Whipple’s disease. Mov Disord 7:82–85.<br />
Tomsak RL, Remler BF, Averbuch-Heller L, et al. (1995). Unsatisfactory treatment of acquired nystagmus with<br />
retrobulbar <strong>in</strong>jection of botul<strong>in</strong>um tox<strong>in</strong>. Am J Ophthalmol 119:489–496.<br />
Toshniwal P, Yadava R, Goldbarg H. (1986). Presentation of p<strong>in</strong>ealoblastoma with ocular dipp<strong>in</strong>g and deafness.<br />
J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 6:128–136.<br />
Traccis S, Marras MA, Puliga MV, et al. (1997). Square-wave jerks and square wave oscillations: treatment with<br />
valproic acid. <strong>Neuro</strong>-<strong>ophthalmology</strong> 18:51–58.<br />
Traccis S, Rosati G, Monaco MF, et al. (1990). Successful treatment of acquired pendular elliptical nystagmus <strong>in</strong><br />
multiple sclerosis with isoniazid and base-out prisms. <strong>Neuro</strong>logy 40:492–494.<br />
Trobe JD, Sharpe JA, Hirsh DK, Gebarski SS. (1991). Nystagmus <strong>in</strong> Pelizaeus-Merzbacher disease. A magnetic<br />
search-coil study. Arch <strong>Neuro</strong>l 48:87–91.<br />
Troost BT, Janton F, Weaver R. (1990). Periodic alternat<strong>in</strong>g oscillopsia: a symptom of periodic alternat<strong>in</strong>g<br />
nystagmus abolished by baclofen. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:273–277.<br />
Tsuzaka K, Aimoto Y, M<strong>in</strong>ami N, Moriwaka F, Tashiro K. (1993). A case of primary <strong>in</strong>tracranial malignant<br />
lymphoma present<strong>in</strong>g as opsoclonus-myoclonus syndrome. R<strong>in</strong>sho Sh<strong>in</strong>keigaku 33:194–198.<br />
Tusa RJ, Kaplan PW, Ha<strong>in</strong> TC, Naidu S. (1990). Ipsiversive eye deviation and epileptic nystagmus. <strong>Neuro</strong>logy<br />
40:662–665.<br />
Tyler TD, Ruiz RS. (1990). Propranolol <strong>in</strong> the treatment of superior oblique myokymia. Arch Ophthalmol<br />
108:175–176.<br />
Van Stavern GP, Biousse V, Newman NJ, Le<strong>in</strong>gang JC. (2000). Downbeat nystagmus from heat stroke. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 69:403–404.<br />
Verhagen WIM, Huygen PLM, Nicolasen MCM. (1994). Pendular pseudonystagmus. <strong>Neuro</strong>logy 44:1188.<br />
Vers<strong>in</strong>o M, Mascolo A, Piccolo G, et al. (1999). Opsoclonus <strong>in</strong> a patient with cerebellar dysfunction. J <strong>Neuro</strong>ophthalmol<br />
19:229–231.<br />
Vigliani MC, Palmucci L, Polo P, et al. (2001). Paraneoplastic opsoclonus-myoclonus associated with renal cell<br />
carc<strong>in</strong>oma and responsive to tumour ablation. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 70:814–815.<br />
Von Herbay A, Ditton H-J, Schuhmacher F, Maiwald M. (1997). Whipple’s disease: Stag<strong>in</strong>g and monitor<strong>in</strong>g by<br />
cytology and polymerase cha<strong>in</strong> reaction analysis of cerebrosp<strong>in</strong>al fluid. Gastroenterology 113:434–441.<br />
Von Noorden GK, Sprunger DT. (1991). Large rectus muscle recession for the treatment of congenital nystagmus.<br />
Arch Ophthalmol 109:221–224.<br />
Wagner RS, Caputo AR, Reynolds RD. (1990). Nystagmus <strong>in</strong> Down’s syndrome. Ophthalmology 97:1439–1444.
Nystagmus and Other Ocular Oscillations 407<br />
Walker MF, Zee DS. (1999). The effect of hyperventilation on downbeat nystagmus <strong>in</strong> cerebellar disorders.<br />
<strong>Neuro</strong>logy 53:1576–1579.<br />
Waragai M, Takaya Y, Hayashi M. (1995). HTLV-1 associated myelopathy (HAM) with cerebellar atrophy<br />
present<strong>in</strong>g as down-beat nystagmus (Japanese). R<strong>in</strong>sho Sh<strong>in</strong>keigaku 35:549–552.<br />
Weider DJ, Ryder CJ, Stram JR. (1994). Benign paroxysmal positional vertigo: analysis of 44 cases treated by the<br />
canalith reposition<strong>in</strong>g procedure of Epley. Am J Otol 15:321–326.<br />
Wiest G, Safoschnik G, Schnaberth G, Mueller C. (1997). Ocular flutter and truncal ataxia may be associated with<br />
enterovius <strong>in</strong>fection. J <strong>Neuro</strong>l 244:288–292.<br />
Wol<strong>in</strong> MJ, Trent RG, Lav<strong>in</strong> PJM, Cornblath WT. (1996). Oculopalatal myoclonus after the one-and-a-half<br />
syndrome with facial nerve palsy. Ophthalmology 103:177–180.<br />
Yamamoto K, Fukusako T, Nogaki H, Morimatsu M. (1992). Multiple system atrophy with macro square wave<br />
jerks and pendular nystagmus (Japanese). R<strong>in</strong>sho Sh<strong>in</strong>keigaku 32:1261–1265.<br />
Yamazaki K, Katayama S, Ishihara T, Hirata K. (1994). A case of Fisher’s syndrome with upbeat nystagmus<br />
(Japanese). R<strong>in</strong>sho Sh<strong>in</strong>keigaku 34:489–492.<br />
Yaniglos SS, Leigh RJ. (1992). Ref<strong>in</strong>ement of an optical device that stabilizes vision <strong>in</strong> patients with nystagmus.<br />
Optom Vis Sci 69:447–450.<br />
Yee RD, Farlow MR, Suzuki DA, et al. (1992). Abnormal eye movements <strong>in</strong> Gerstmann-Straussler-Sche<strong>in</strong>ker<br />
disease. Arch Ophthalmol 110:68–74,178.<br />
Yen MT, Herdman SJ, Tusa RJ. (1999). Oscillopsia and pseudonystagmus <strong>in</strong> kidney transplant patients. Am J<br />
Opthalmol 128:768–770.<br />
Yokota T, Tsuchiya K, Yamane M, et al. (1991). Geotrophic ocular deviation with skew and absence of saccades <strong>in</strong><br />
Creutzfeldt-Jacob disease. J <strong>Neuro</strong>l Sci 106:175–178.<br />
Yoshimoto Y, Koyama S. (1991). A case of acquired nystagmus alternans associated with acute cerebellitis. Acta<br />
Otolaryngol Suppl 481:371–373.<br />
Young YH, Huang TW. (2001). Role of clonazepam <strong>in</strong> the treatment of idiopathic downbeat nystagmus.<br />
Laryngoscope 111:1490–1493.<br />
Young TL, Weis JR, Summers G, Egbert JE. (1997). The association of strabismus, amblyopia, and refractive errors<br />
<strong>in</strong> spasmus nutans. Ophthalmology 104:112–117.<br />
Yousry I, Dieterich M, Naidich TP, et al. (2002). Superior oblique myokymia: magnetic resonance imag<strong>in</strong>g support<br />
for the neurovascular compression hypothesis. <strong>An</strong>n <strong>Neuro</strong>l 51:361–368.<br />
Zubkov AA, Re<strong>in</strong>encke RD, Gottlob I, et al. (1990). Treatment of manifest latent nystagmus. Am J Ophthalmol<br />
110:160–167.<br />
Zubkov AA, Staark N, Weber A, et al. (1993). Improvement of visual acuity after surgery for nystagmus.<br />
Ophthalmology 100:1488–1497.
This page <strong>in</strong>tentionally left blank
18 r<br />
Ptosis<br />
Ptosis is present when the upper eyelid is less than 2 mm from the center of the pupil.<br />
The causes of ptosis may be classified as mechanical, neurogenic, myogenic, and<br />
neuromuscular junctional (Kersten, 1995; Oosterhuis, 1996).<br />
What Dist<strong>in</strong>guishes Ptosis from Pseudoptosis?<br />
A number of conditions may cause downward displacement of the eyelid without true<br />
ptosis (pseudoptosis). These conditions are listed <strong>in</strong> Table 18–1.<br />
What Is Blepharospasm or Hemifacial Spasm?<br />
Blepharospasm or hemifacial spasm may produce a pseudoptosis due to the <strong>in</strong>termittent<br />
(voluntary or <strong>in</strong>voluntary) closure (spasm) of the upper and lower eyelid(s)<br />
result<strong>in</strong>g from contraction of the orbicularis oculi muscles. The lid position dur<strong>in</strong>g the<br />
Table 18–1. Causes of Pseudoptosis<br />
Aberrant regeneration of the facial nerve<br />
<strong>An</strong>ophthalmic socket<br />
Apraxia of lid open<strong>in</strong>g<br />
Downgaze paralysis and pseudoblepharoptosis<br />
Blepharospasm or hemifacial spasm<br />
Hypertropia or hyperglobus (Kratky, 1992; Lyon, 1993; Meyer, 1991)<br />
Lid retraction <strong>in</strong> the contralateral eye<br />
Microphthalmia or phthisis bulbi<br />
Psychogenic pseudoptosis (all show depression of eyebrow on affected side) (Hop, 1997)<br />
409
410 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
periods without contraction of the orbicularis muscles is normal. Voluntary lid closure<br />
may create pseudoptosis and occur as a conversion reaction.<br />
What Is Apraxia of Eyelid Open<strong>in</strong>g?<br />
Apraxia of eyelid open<strong>in</strong>g is a supranuclear <strong>in</strong>ability to open the eyelids voluntarily.<br />
Spontaneous and reflex eyelid open<strong>in</strong>g are normal, and patients may manually open the<br />
lids or employ a head thrust as a compensatory movement.<br />
Aramideh et al correlated the cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs of apraxia of eye open<strong>in</strong>g with<br />
synchronous levator palpebrae (LP) and orbicularis oculi (Ooc) electromyographic<br />
(EMG) record<strong>in</strong>gs (Aramideh, 1995). EMG was characterized by either <strong>in</strong>termittent<br />
LP <strong>in</strong>hibition (ILPI) or a cont<strong>in</strong>uation of Ooc activity (Tozlovanu, 2001) follow<strong>in</strong>g<br />
voluntary closure of the eyes (pretarsal motor persistence [PMP]). This study demonstrated<br />
the follow<strong>in</strong>g:<br />
1. In some patients there may be <strong>in</strong>termittent <strong>in</strong>voluntary eye closure as a result of ILPI.<br />
Persistence of ILPI follow<strong>in</strong>g eye closure would <strong>in</strong>terfere with eye open<strong>in</strong>g. When<br />
there is no ILPI, these patients have no difficulty open<strong>in</strong>g their eyes at will follow<strong>in</strong>g<br />
voluntary closure.<br />
2. In other patients, closure of the eyes due to ILPI may activate Ooc. These patients<br />
have PMP <strong>in</strong> addition to ILPI and are unable to open their eyes at will follow<strong>in</strong>g<br />
voluntary closure.<br />
3. Patients who have PMP alone may be unable to open their eyes at will follow<strong>in</strong>g<br />
voluntary closure. Once open, the eyes do not have the tendency to close by<br />
themselves.<br />
Unilateral or bilateral hemispheric disease and extrapyramidal disease may produce<br />
apraxia of lid open<strong>in</strong>g. The etiologies of apraxia of eye open<strong>in</strong>g are listed <strong>in</strong> Table 18–2.<br />
What Is Hypertropia or Hyperglobus?<br />
Hypertropia or hyperglobus may result <strong>in</strong> an abnormal position of the eye under a<br />
normal eyelid. Fixation with the hypertropic eye may elim<strong>in</strong>ate the pseudoptosis.<br />
Likewise, contralateral eyelid retraction (e.g., thyroid eye disease) may produce an<br />
apparent ptosis <strong>in</strong> the fellow (normal) eye (Kratky, 1992; Lyon, 1993; Meyer, 1991).<br />
Is the Ptosis Acquired or Congenital?<br />
Ptosis may occur on a congenital or acquired basis. Long-stand<strong>in</strong>g isolated and static<br />
congenital ptosis with no other signs of systemic neurogenic (e.g., third nerve palsy),<br />
myogenic (e.g., chronic progressive external ophthalmoplegia), or neuromuscular<br />
disease (e.g., myasthenia gravis) does not require additional evaluation. Other forms<br />
of congenital nonisolated ptosis necessitate the same evaluation as the patients with<br />
acquired nonisolated ptosis. Table 18–3 lists the various forms of congenital ptosis.<br />
Congenital ptosis may be associated with other ocular and nonocular defects.<br />
Although children with congenital ptosis <strong>in</strong> isolation do not usually require any<br />
further evaluation, they may need amblyopia treatment or surgical therapy to avoid
Table 18–2. Etiologies of Apraxia of Eyelid Open<strong>in</strong>g<br />
Extrapyramidal disease<br />
Park<strong>in</strong>son’s disease<br />
MPTP-<strong>in</strong>duced park<strong>in</strong>sonism<br />
Progressive autosomal-dom<strong>in</strong>ant park<strong>in</strong>sonism and dementia<br />
with pallido-ponto-nigral degeneration<br />
Hunt<strong>in</strong>gton’s disease<br />
Multiple systems atrophy (e.g., Shy-Drager syndrome)<br />
Progressive supranuclear palsy<br />
Wilson’s disease<br />
<strong>Neuro</strong>acanthocytosis<br />
Cortical-basal ganglionic degeneration<br />
Adult-onset Hallervordan-Spatz syndrome<br />
Unilateral (especially nondom<strong>in</strong>ant hemisphere) or bilateral hemispheric lesions<br />
Focal <strong>in</strong>ferior and lateral frontal lobe cortical degeneration<br />
Motor neuron disease<br />
Post–bilateral stereotactic subthalamotomy<br />
Post–implantation of bilateral subthalamic nucleus electrical stimulators<br />
for Park<strong>in</strong>son’s disease<br />
Unilateral putam<strong>in</strong>al hemorrhage<br />
Isolated f<strong>in</strong>d<strong>in</strong>g (may be levodopa responsive)<br />
Source: Abe, 1995; Adair, 1995; Aramideh, 1995; Averbuch-Heller, 1998; Boghen, 1997;<br />
Dewey, 1994; Jancovic, 1995; Klostermann, 1997; Krack, 1994; Limous<strong>in</strong>, 1999; Nazarian,<br />
1998; Riley, 1990; Verghese, 1999; Wszolek, 1992.<br />
MPTP, 1-methyl-4-phenyl-1,2,3,6 tetrahydropyrid<strong>in</strong>e.<br />
Table 18–3. Congenital Ptosis<br />
Ptosis 411<br />
Isolated<br />
Nonisolated<br />
Associated other ocular defects<br />
Congenital cataracts<br />
Epicanthus<br />
Punctal abnormalities<br />
Refractive error=anisometropia<br />
Strabismus and amblyopia<br />
Associated nonocular defects (e.g., skeletal, central nervous system [CNS])<br />
Blepharophimosis syndrome<br />
Synk<strong>in</strong>etic ptosis (e.g., Marcus-Gunn jaw-w<strong>in</strong>k<strong>in</strong>g ptosis)<br />
Myogenic (e.g., congenital fibrosis)<br />
<strong>Neuro</strong>genic (e.g., third nerve palsy)<br />
<strong>Neuro</strong>muscular junction (e.g., myasthenia gravis)<br />
Comb<strong>in</strong>ed valproate and hydanto<strong>in</strong> embryopathy with anomalous septum pellucidum<br />
(Gigantelli, 2000)<br />
X-l<strong>in</strong>ked congenital isolated ptosis (McMullan, 2000, 2001)
412 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
amblyopia (Cibis, 1995; Gusek-Schneider, 2000; Hornblass, 1995; McCulloch, 1993;<br />
Steel, 1996). Intermittent lid retraction of a ptotic eyelid may occur dur<strong>in</strong>g chew<strong>in</strong>g,<br />
jaw movement, or suck<strong>in</strong>g <strong>in</strong> a child due to aberrant <strong>in</strong>nervation of the oculomotor<br />
(levator) and trigem<strong>in</strong>al nerves (the Marcus-Gunn jaw w<strong>in</strong>k<strong>in</strong>g phenomenon). No<br />
evaluation is required <strong>in</strong> children with the jaw-w<strong>in</strong>k<strong>in</strong>g phenomenon (class IV, level<br />
C). Patients with acquired forms of ptosis should undergo further diagnostic evaluation<br />
(class III–IV, level B).<br />
Is the Ptosis Isolated or Nonisolated?<br />
A nonisolated acquired ptosis is def<strong>in</strong>ed as ptosis associated with other f<strong>in</strong>d<strong>in</strong>gs that<br />
may suggest a specific etiology as listed <strong>in</strong> Table 18–4.<br />
Is the Ptosis Due to Mechanical Causes?<br />
<strong>An</strong>y mechanical disturbance of the upper eyelid may result <strong>in</strong> ptosis. Mechanical causes<br />
of ptosis are listed <strong>in</strong> Table 18–5. Patients with an underly<strong>in</strong>g mechanical ptosis that<br />
might be due to an underly<strong>in</strong>g mass or <strong>in</strong>filtrative lesion should undergo imag<strong>in</strong>g of the<br />
orbit (e.g., magnetic resonance imag<strong>in</strong>g of the head and orbit with fat suppression and<br />
gadol<strong>in</strong>ium contrast) (class IV, level C).<br />
Is the Ptosis Due to Myogenic Causes?<br />
Chronic progressive external ophthalmoplegia (CPEO) <strong>in</strong>cludes a spectrum of disorders<br />
that may result <strong>in</strong> a syndrome of pa<strong>in</strong>less, pupil-spar<strong>in</strong>g, slowly progressive, and<br />
generally symmetric ophthalmoplegia. One subset of CPEO, the Kearns-Sayre<br />
syndrome, is characterized by the cl<strong>in</strong>ical triad of early-onset (usually before the age<br />
of 20 years) CPEO, pigmentary degeneration of the ret<strong>in</strong>a, and cardiac abnormalities<br />
(e.g., <strong>in</strong>traventricular conduction defects, bundle branch block, and complete heart<br />
block). Other features of Kearns-Sayre syndrome are elevation of cerebrosp<strong>in</strong>al fluid<br />
prote<strong>in</strong>, other neurologic dysfunction (e.g., cerebellar, auditory, and vestibular dysfunction),<br />
cognitive dysfunction, short stature, and developmental delay. Muscle biopsy<br />
may show ‘‘ragged red fibers’’ or other changes <strong>in</strong> mitochondria (Gross-Jendroska, 1992;<br />
Simonsz, 1992). Pathologic exam<strong>in</strong>ation of the bra<strong>in</strong> may demonstrate spongiform<br />
degeneration.<br />
Table 18–4. Etiologies of Nonisolated Ptosis<br />
Mechanical (e.g., lid mass, <strong>in</strong>fection, or <strong>in</strong>flammation)<br />
Myogenic (e.g., external ophthalmoplegia)<br />
<strong>Neuro</strong>genic (e.g., ocular motor deficit and=or diplopia, other neurologic f<strong>in</strong>d<strong>in</strong>gs,<br />
Horner’s syndrome)<br />
<strong>Neuro</strong>muscular junction disease (e.g., signs of myasthenia gravis such as fatigue,<br />
enhancement, variability, Cogan’s lid twitch sign)
Table 18–5. Mechanical Causes of Ptosis<br />
Other systemic and ocular myopathies may cause ptosis but are usually associated<br />
with myopathic signs and symptoms due to <strong>in</strong>volvement of extraocular and other<br />
muscles (Parmeggiani, 1992). Detailed discussion of these myogenic forms of ptosis is<br />
beyond the scope of this text. Myopathies that may cause ptosis are listed <strong>in</strong> Table 18–6.<br />
Is the Ptosis Due to a <strong>Neuro</strong>genic Cause?<br />
Ptosis 413<br />
Redundant sk<strong>in</strong> or fat on the upper eyelid (e.g., dermatochalasis)<br />
Tumors or cysts of the conjunctiva or eyelid (Avisar, 1991)<br />
Infection (e.g., preseptal or orbital cellulitis)<br />
Cicatricial scarr<strong>in</strong>g (e.g., posttraumatic, postsurgical, or post<strong>in</strong>flammatory) (S<strong>in</strong>gh, 1997)<br />
Inflammation and edema<br />
Graves’ disease<br />
Blepharochalasis<br />
Dermatomyositis<br />
Infiltration<br />
Amyloid<br />
Sarcoid<br />
Neoplastic (e.g., breast cancer) (Po, 1996)<br />
Waldenström’s macroglobul<strong>in</strong>emia (Klapper, 1998)<br />
Primary or metastatic tumors or orbital pseudotumor (Kersten, 1995; Po, 1996)<br />
<strong>Neuro</strong>fibroma<br />
Hemangioma<br />
Dermoid<br />
Lymphoma<br />
Contact lenses related (Kersten, 1995; Levy, 1992; van der Bosch, 1992)<br />
Foreign body reaction<br />
Giant papillary conjunctivitis (GPC)<br />
Contact lens migration (Patel, 1998)<br />
Dis<strong>in</strong>sertion of the levator from excessive eyelid manipulation<br />
The neurogenic causes of ptosis are listed <strong>in</strong> Table 18–7. Denervation of the levator<br />
muscle due to a third nerve palsy may result <strong>in</strong> partial or complete ptosis. Levator<br />
excursion is decreased <strong>in</strong> all of these patients. As noted <strong>in</strong> Chapter 11, nuclear third<br />
nerve palsies result <strong>in</strong> bilateral ptosis or no ptosis because both levator muscles are<br />
<strong>in</strong>nervated by a s<strong>in</strong>gle central caudal nucleus. This type of ptosis is usually associated<br />
with other features of a third nerve palsy (e.g., pupil <strong>in</strong>volvement, extraocular muscle<br />
dysfunction), but may rarely occur with other m<strong>in</strong>imal third nerve signs (Good, 1990;<br />
Mart<strong>in</strong>, 1996). Midbra<strong>in</strong> lesions may result <strong>in</strong> ptosis with or without downgaze<br />
paralysis, fatigable ptosis (Kao, 1999; Ragge, 1992), or pseudoptosis (Galetta, 1993;<br />
Johnson, 1992; Lagreze, 1996; Tomecek, 1994).<br />
Ptosis and ipsilateral miosis may be due to a Horner’s syndrome. The associated<br />
features of the Horner’s syndrome are discussed <strong>in</strong> more detail <strong>in</strong> Chapter 20.
414 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 18–6. Myopathies Associated with Ptosis<br />
Chronic progressive external ophthalmoplegia (Rowland, 1997)<br />
Congenital fibrosis (Engle, 1995; Gillies, 1995; Tandon, 1993)<br />
Congenital myopathy<br />
Central core myopathy<br />
Centronuclear myopathy<br />
Multicore myopathy<br />
Nemal<strong>in</strong>e myopathy<br />
Fiber-type disproportion (Ohtaki, 1990)<br />
Congenital muscular dystrophy<br />
Diabetes (possibly due to hypoxia to levator)<br />
Familial periodic paralysis<br />
Myotonia congenita and myotonic dystrophy (Ashizawa, 1992)<br />
Oculopharyngeal dystrophy (Blumen, 1993; Lacomis, 1991; Rowland, 1997)<br />
Rapidly progressive adolescent-onset oculopharyngeal somatic syndrome with<br />
rimmed vacuoles (Rose, 1997)<br />
Inflammatory and <strong>in</strong>filtrative myopathies<br />
Hypothyroid myopathy<br />
Table 18–7. <strong>Neuro</strong>genic Conditions Associated with Ptosis<br />
Third nerve palsy<br />
With or without partial or complete ophthalmoplegia (Tummala, 2001)<br />
With or without aberrant regeneration<br />
Alternat<strong>in</strong>g ptosis <strong>in</strong> abetalipoprote<strong>in</strong>emia<br />
Bra<strong>in</strong>stem <strong>in</strong>farct (Mihaescu, 2000; Saeki, 2000)<br />
Horner’s syndrome<br />
Fisher syndrome (Al-D<strong>in</strong>, 1994; Berlit, 1992; Ishikawa, 1990; Mori, 2001; Yip, 1991)<br />
Acute <strong>in</strong>flammatory polyradiculoneuropathy (Guilla<strong>in</strong>-Barré syndrome)<br />
Chronic <strong>in</strong>flammatory polyradiculoneuropathy (CIDP)<br />
Cerebral ptosis (Afifi, 1990; Averbuch-Heller, 1996, 2002; Barton, 1995; Zachariah, 1994)<br />
Cerebellar ptosis follow<strong>in</strong>g craniovertebral decompression of Chiari I malformation<br />
(Holly, 2001)<br />
M<strong>in</strong>or head trauma <strong>in</strong> patient with chronic hydrocephalus (supranuclear ptosis)<br />
(Suzuki, 2000)<br />
Putam<strong>in</strong>al hemorrhage (Verghese, 1999)<br />
Paradoxic supranuclear <strong>in</strong>hibition of levator tonus<br />
Seizure-<strong>in</strong>duced<br />
Thiam<strong>in</strong>e deficiency<br />
Wernicke’s disease<br />
Recurrent isolated ptosis (last<strong>in</strong>g 6 to 10 weeks) <strong>in</strong> presumed ophthalmoplegic migra<strong>in</strong>e of<br />
childhood (Stidham, 2000)<br />
Relaps<strong>in</strong>g alternat<strong>in</strong>g ptosis (episodes last<strong>in</strong>g days)—reported <strong>in</strong> two sibl<strong>in</strong>gs (Sieb, 2000)
The Fisher variant of Guilla<strong>in</strong>-Barré syndrome may present with supranuclear ptosis<br />
ophthalmoplegia, ataxia, and areflexia (Al-D<strong>in</strong>, 1994; Berlit, 1992; Ishikawa, 1990;<br />
Yip, 1991).<br />
Unilateral or bilateral hemispheric dysfunction (e.g., stroke, arteriovenous malformation,<br />
seizure) may produce ptosis (Afifi, 1990; Averbuch-Heller, 1996, 2002; Barton, 1995;<br />
Zachariah, 1994).<br />
Is <strong>Neuro</strong>muscular Junction Disease<br />
Responsible for the Ptosis?<br />
Myasthenia gravis may result <strong>in</strong> a ptosis that is often variable and may worsen after<br />
susta<strong>in</strong>ed effort or fatigue. Fatigable ptosis, however, has been reported <strong>in</strong> patients with<br />
<strong>in</strong>tracranial etiologies (e.g., hematoma, metastasis) (Kao, 1999). As noted <strong>in</strong> Chapter 15,<br />
myasthenia gravis may result <strong>in</strong> ptosis with or without other extraocular muscle<br />
dysfunction. Other rare causes of neuromuscular junction ptosis <strong>in</strong>clude wound<br />
botulism, Eaton-Lambert syndrome (Brazis, 1997), and topical neuromuscular blockade<br />
(e.g., topical timolol). We recommend that myasthenia gravis be considered <strong>in</strong> every<br />
case of unexpla<strong>in</strong>ed, pa<strong>in</strong>less, unilateral or bilateral ptosis with or without ophthalmoplegia<br />
(class IV, level C).<br />
What Are the Causes of Isolated Ptosis?<br />
The causes of isolated ptosis <strong>in</strong>clude congenital ptosis, trauma (<strong>in</strong>clud<strong>in</strong>g surgery),<br />
steroid-<strong>in</strong>duced ptosis, and aponeurotic ptosis.<br />
Is the Ptosis Secondary to Trauma?<br />
Trauma may damage the sk<strong>in</strong>, soft tissues, muscles, levator complex, or nerve supply of<br />
the eyelid (Keane, 1993). Surgical trauma and=or myotoxicity from the local anesthetic<br />
agents dur<strong>in</strong>g orbital or ocular (<strong>in</strong>clud<strong>in</strong>g strabismus, ret<strong>in</strong>al, corneal, cataract, glaucoma,<br />
and refractive) surgery may also damage the eyelid structures and cause ptosis<br />
(Feibel, 1993; Liu, 1992; Loeffler, 1990; Ropo, 1992). Kaplan et al performed a prospective<br />
analysis of ptosis and cataract surgery, and found that trauma to the superior rectus<br />
complex was the most critical factor (Kaplan, 1985).<br />
Table 18–8. <strong>Cl<strong>in</strong>ical</strong> Features of Aponeurotic<br />
Ptosis<br />
Acquired<br />
Good to excellent levator function ( > 12 mm)<br />
Elevated or absent lid crease<br />
Th<strong>in</strong>n<strong>in</strong>g of the eyelid above the tarsus<br />
Bilateral but may be unilateral or asymmetric<br />
Elderly population<br />
Ptosis 415
416 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 18–1. Evaluation of ptosis.
Is the Ptosis Steroid Induced?<br />
Topical steroids have been implicated <strong>in</strong> some cases of ptosis. Discont<strong>in</strong>uation of the<br />
steroids may reverse the ptosis <strong>in</strong> these cases.<br />
Is Aponeurotic Ptosis Present?<br />
Levator aponeurosis th<strong>in</strong>n<strong>in</strong>g and=or dehiscence may occur as a result of trauma,<br />
surgery, lid swell<strong>in</strong>g, patch<strong>in</strong>g, or, most commonly, as an age-related phenomenon<br />
(Frueh, 1996; Liu, 1993; Older, 1995). The characteristic features of aponeurotic ptosis are<br />
listed <strong>in</strong> Table 18–8.<br />
Patients with aponeurotic ptosis may have significant ptosis <strong>in</strong> downgaze more than<br />
primary position (Dryden, 1992; Wojno, 1993). In the absence of f<strong>in</strong>d<strong>in</strong>gs to suggest<br />
mechanical, neurogenic, myogenic, or neuromuscular etiologies for ptosis, no further<br />
evaluation is necessary. Superior visual field loss may occur due to ptosis and may be an<br />
<strong>in</strong>dication for surgical correction. The surgical treatment of aponeurotic ptosis is well<br />
described <strong>in</strong> the literature and is not reviewed here (Frueh, 1996; Liu, 1993; Older, 1995).<br />
Aponeurotic ptosis does not require any neuroimag<strong>in</strong>g (class III–IV, level B).<br />
<strong>An</strong> approach to the evaluation of ptosis is outl<strong>in</strong>ed <strong>in</strong> Figure 18–1.<br />
References<br />
Ptosis 417<br />
Abe K, Fujimura H, Tatsumi C, et al. (1995). Eyelid ‘‘apraxia’’ <strong>in</strong> patients with motor neuron disease. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 59:629.<br />
Adair JC, Williamson DJG, Heilman KM. (1995). Eyelid open<strong>in</strong>g apraxia <strong>in</strong> focal cortical degeneration. J. <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 58:508–509.<br />
Afifi AK, Corbett JJ, Thompson HS, Wells KK. (1990). Seizure-<strong>in</strong>duced miosis and ptosis: association with<br />
temporal lobe magnetic resonance imag<strong>in</strong>g abnormalities. J Child <strong>Neuro</strong>l 5:142–146.<br />
Al-D<strong>in</strong> AN, <strong>An</strong>derson M, Eeg-Olofsson O, et al. (1994). <strong>Neuro</strong>-ophthalmic manifestations of the syndrome of<br />
ophthalmoplegia, ataxia, and areflexia. A review. Acta <strong>Neuro</strong>l Scand 89:157–163.<br />
Aramideh M, Ongerboer de Visser BW, Koelman JHTM, Speelman JD. (1995). Motor persistence of orbicularis<br />
oculi muscle <strong>in</strong> eyelid-open<strong>in</strong>g disorders. <strong>Neuro</strong>logy 45:897–902.<br />
Ashizawa T, Hejtmancki JF, Liu J, Perryman MB, Epste<strong>in</strong> MF, Koch DD. (1992). Diagnostic value of ophthalmologic<br />
f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> myotonic dystrophy: comparison with risks calculated by haplotype analysis of closely<br />
l<strong>in</strong>ked restriction fragment length polymorphisms. Am J Med Genet 42:55–60.<br />
Averbuch-Heller L, Helmchen C, Horn AKE, et al. (1998). Slow vertical saccades <strong>in</strong> motor neuron disease:<br />
correlation of structure and function. <strong>An</strong>n <strong>Neuro</strong>l 44:641–648.<br />
Averbuch-Heller L, Leigh RJ, Mermelste<strong>in</strong> V, et al. (2002). Ptosis <strong>in</strong> patients with hemispheric stroke. <strong>Neuro</strong>logy<br />
58:620–624.<br />
Averbuch-Heller L, Stahl JS, Remler BR, Leigh RJ. (1996). Bilateral ptosis and upgaze palsy with right hemispheric<br />
lesions. <strong>An</strong>n <strong>Neuro</strong>l 40:465–468.<br />
Avisar R, Leshem Y, Savir H. (1991). Unilateral congenital ptosis due to plexiform neurofibroma, caus<strong>in</strong>g<br />
refraction error and secondary amblyopia. Metab Pediatr Syst Ophthalmol 14:62–63.<br />
Barton JJ, Kardon RH, Slagel D, Thompson HS. (1995). Bilateral central ptosis <strong>in</strong> acquired immunodeficiency<br />
syndrome (review). Can J <strong>Neuro</strong>l Sci 22:52–55.<br />
Berlit P, Rakicky J. (1992). The Miller Fisher syndrome: review of the literature. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:57–63.<br />
Blumen SC, Nisipeanu P, Sadeh M, Asherov A, Tome F, Korczyn AD. (1993). <strong>Cl<strong>in</strong>ical</strong> features of oculopharyngeal<br />
muscular dystrophy among Bukhara Jews. <strong>Neuro</strong>musc Dis 3:575–577.<br />
Boghen D. (1997). Apraxia of lid open<strong>in</strong>g: a review. <strong>Neuro</strong>logy 48:1491–1503.<br />
Brazis PW. (1997). Enhanced ptosis <strong>in</strong> Lambert-Eaton myasthenic syndrome. J <strong>Neuro</strong>-ophthalmol 17:202–203.
418 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Cibis GW, Fitzgerald KM. (1995). Amblyopia <strong>in</strong> unilateral congenital ptosis: early detection by sweep visual<br />
evoked potential. Graefes Arch Cl<strong>in</strong> Exp Ophthalmol 233:605–609.<br />
Dewey RB Jr, Maraganore DM. (1994). Isolated eyelid open<strong>in</strong>g apraxia: report of a new levodopa-responsive<br />
syndrome. <strong>Neuro</strong>logy 44:1752–1754.<br />
Dryden RM, Kahanic DA. (1992). Worsen<strong>in</strong>g of blepharoptosis <strong>in</strong> downgaze. Ophthalmic Plast Reconstr Surg<br />
8:126–129.<br />
Engle EC, Marondel I, Houtman WA, et al. (1995). Congenital fibrosis of the extraocular muscles (autosomal<br />
dom<strong>in</strong>ant congenital external ophthalmoplegia): genetic homogeneity, l<strong>in</strong>kage ref<strong>in</strong>ement, and physical<br />
mapp<strong>in</strong>g on chromosome 12. Am J Hum Genet 57:1086–1094.<br />
Feibel RM, Custer PL, Gordon MO. (1993). Postcataract ptosis: a randomized, double-masked comparison of<br />
peribulbar and retrobulbar anesthesia. Ophthalmology 100:660–665.<br />
Frueh BR, Musch DC. (1996). Evaluation of levator muscle <strong>in</strong>tegrity <strong>in</strong> ptosis with levator force measurement.<br />
Ophthalmology 103:244–250.<br />
Galetta SL, Gray LG, Raps EC, Grossman RI, Schatz NJ. (1993). Unilateral ptosis and contralateral eyelid<br />
retraction from a thalamic-midbra<strong>in</strong> <strong>in</strong>farction. Magnetic resonance imag<strong>in</strong>g correlation. J Cl<strong>in</strong> <strong>Neuro</strong>ophthalmol<br />
13:221–224.<br />
Gigantelli JW, Braddock SR, Johnson LN. (2000). Blepharoptosis and central nervous system abnormalities <strong>in</strong><br />
comb<strong>in</strong>ed valproate and hydanto<strong>in</strong> embryopathy. Ophthalmic Plast Reconstr Surg 16:52–54.<br />
Gillies WE, Harris AJ, Brooks AM, Rivers MR, Wolfe RJ. (1995). Congenital fibrosis of the vertically act<strong>in</strong>g<br />
extraocular muscles. A new group of dom<strong>in</strong>antly <strong>in</strong>herited ocular fibrosis with radiologic f<strong>in</strong>d<strong>in</strong>gs.<br />
Ophthalmology 102:607–612.<br />
Good EF. (1990). Ptosis as the sole manifestation of compression of the oculomotor nerve by an aneurysm of the<br />
posterior communicat<strong>in</strong>g artery. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:59–61.<br />
Gross-Jendroska M, Schatz H, McDonald HR, Johnson RN. (1992). Kearns-Sayre syndrome: a case report and<br />
review (review). Eur J Ophthalmol 2:15–20.<br />
Gusek-Schneider GC, Martus P. (2000). Stimulus deprivation amblyopia <strong>in</strong> human congenital ptosis: a study of<br />
100 patients. Strabismus 8:261–270.<br />
Holly LT, Batzdorf U. (2001). Management of cerebellar ptosis follow<strong>in</strong>g craniovertebral decompression for Chiari<br />
I malformation. J <strong>Neuro</strong>surg 94:21–26.<br />
Hop JW, Frijns CJ, van Gijn J. (1997). Psychogenic pseudoptosis. J <strong>Neuro</strong>l 244:623–624.<br />
Hornblass A, Kass LG, Ziffer AJ. (1995). Amblyopia <strong>in</strong> congenital ptosis. Ophthalmic Surg 26:334–337.<br />
Ishikawa H, Wakakura M, Ishikawa S. (1990). Enhanced ptosis <strong>in</strong> Fisher’s syndrome after Epste<strong>in</strong>-Barr virus<br />
<strong>in</strong>fection. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:197–200.<br />
Jancovic J. (1995). Apraxia of lid open<strong>in</strong>g. Mov Disord 10:5.<br />
Johnson LN, Castro D. (1992). Monocular elevation paresis and <strong>in</strong>complete ptosis due to midbra<strong>in</strong> <strong>in</strong>farction<br />
<strong>in</strong>volv<strong>in</strong>g the fascicular segment of the oculomotor nerve (letter). J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:73.<br />
Kao Y-F, Lan M-Y, Chou M-S, Chen W-H. (1999). Intracranial fatigable ptosis. J <strong>Neuro</strong>-Ophthalmol 19:257–259.<br />
Kaplan LJ, Jaffe NS, Clayman HM. (1985). Ptosis and cataract surgery. A multivariant computer analysis of a<br />
prospective study. Ophthalmology 92:237–242.<br />
Keane JR. (1993). Ptosis and levator paralysis caused by orbital roof fractures. Three cases with subfrontal epidural<br />
hematomas. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:225–228.<br />
Kersten RC, De Concillis C, Kulw<strong>in</strong> DR. (1995). Acquired ptosis <strong>in</strong> the young and middle-aged adult population.<br />
Ophthalmology 102:924–928.<br />
Klapper SR, Jordan DR, Pelletier C, et al. (1998). Ptosis <strong>in</strong> Waldenstrom’s macroglobul<strong>in</strong>emia. Am J Ophthalmol<br />
126:315–317.<br />
Klostermann W, Viereege P, Kömpf D. (1997). Apraxia of eyelid open<strong>in</strong>g after bilateral stereotaxic subthalamotomy.<br />
J <strong>Neuro</strong>-Ophthalmol 17:122–123.<br />
Krack P, Marion MH. (1994). ‘‘Apraxia of lid open<strong>in</strong>g,’’ a focal dystonia: cl<strong>in</strong>ical study of 32 patients. Mov Disord<br />
9:610–615.<br />
Kratky V, Harvey JT. (1992). Tests for contralateral pseudoretraction <strong>in</strong> blepharoptosis. Ophthalmic Plast Reconstr<br />
Surg 8:22–25.<br />
Lacomis D, Kupsky WJ, Kuban KK, Specht LA. (1991). Childhood onset oculopharyngeal muscular dystrophy.<br />
Pediatr <strong>Neuro</strong>l 7:382–384.<br />
Lagreze WD, Warner JE, Zamani AA, Gouras GK, Koralnik IJ, Bienfang DC. (1996). Mesencephalic clefts with<br />
associated eye movement disorders. Arch Ophthalmol 114:429–432.<br />
Levy B, Stamper RL. (1992). Acute ptosis secondary to contact lens wear. Optom Vis Sci 69:565–566.<br />
Limous<strong>in</strong> P, Krack P, Pollak P, et al. (1998). Electrical stimulation of the subthalamic nucleus <strong>in</strong> advanced<br />
Park<strong>in</strong>son’s disease. N Engl J Med 339:1105–1111.
Ptosis 419<br />
Liu D. (1993). Ptosis repair by s<strong>in</strong>gle suture aponeurotic tuck. Surgical technique and long-term results.<br />
Ophthalmology 100:251–259.<br />
Liu D, Bachynski BN. (1992). Complete ptosis as a result of removal of epibulbar lipodermoid. Ophthalmic Plast<br />
Reconstr Surg 8:134–136.<br />
Loeffler M, Solomon LD, Renaud M. (1990). Postcataract extraction ptosis: effect of the bridle suture. J Cataract<br />
Refract Surg 16:501–504.<br />
Lyon DB, Gonner<strong>in</strong>g RS, Dorztbach RK, Lemke BN. (1993). Unilateral ptosis and eye dom<strong>in</strong>ance. Ophthalmic Plast<br />
Reconstr Surg 9:237–240.<br />
Mart<strong>in</strong> JJ Jr, Tenzel RR. (1992). Acquired ptosis: dehiscences and dis<strong>in</strong>sertions. Are they real or iatrogenic?<br />
Ophthalmic Plast Reconstr Surg 8:130–132.<br />
Mart<strong>in</strong> TJ, Corbett JJ, Babikian PV, et al. (1996). Bilateral ptosis due to mesencephalic lesions with relative<br />
preservation of ocular motility. J <strong>Neuro</strong>-Ophthalmol 16:258–263.<br />
McCulloch DL, Wright KW. (1993). Unilateral congenital ptosis: compensatory head postur<strong>in</strong>g and amblyopia.<br />
Ophthalmic Plast Reconstr Surg 9:196–200.<br />
McMullan TF, Coll<strong>in</strong>s AR, Tyers AG, Rob<strong>in</strong>son DO. (2000). A novel X-l<strong>in</strong>ked dom<strong>in</strong>ant condition: X-l<strong>in</strong>ked<br />
congenital isolated ptosis. Am J Hum Genet 66:1455–1460.<br />
McMullan TF, Tyers AG. (2001). X-l<strong>in</strong>ked dom<strong>in</strong>ant congenital isolated bilateral ptosis: the def<strong>in</strong>ition and<br />
characterization of a new condition. Br J Ophthalmol 85:70–73.<br />
Meyer DR, Wobig JL. (1991). Detection of contralateral eyelid retraction associated with blepharoptosis.<br />
Ophthalmology 98:366–375.<br />
Mihaescu M, Brillman J, Rothfus W. (2000). Midbra<strong>in</strong> ptosis caused by periaqueductal <strong>in</strong>farct follow<strong>in</strong>g cardiac<br />
catheterization: early detection with diffusion-weighted imag<strong>in</strong>g. J <strong>Neuro</strong>imag<strong>in</strong>g 10:187–189.<br />
Mori M, Kuwabara S, Fukutake T, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> features and prognosis of Miller Fisher syndrome.<br />
<strong>Neuro</strong>logy 56:1104–1106.<br />
Nazarian SM, Amiri M. (1998). Apraxia of eyelid open<strong>in</strong>g <strong>in</strong> right hemisphere stroke. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:25.<br />
Ohtaki E, Yamaguch Y, Yamashita Y, et al. (1990). Complete external ophthalmoplegia <strong>in</strong> a patient with congenital<br />
myopathy without specific features (m<strong>in</strong>imal change myopathy). Bra<strong>in</strong> Dev 12:427–430.<br />
Older JJ. (1995). Ptosis repair and blepharoplasty <strong>in</strong> the adult (review). Ophthalmic Surg 26:304–308.<br />
Oosterhuis HJ. (1996). Acquired blepharoptosis. Cl<strong>in</strong> <strong>Neuro</strong>l <strong>Neuro</strong>surg 98:1–7.<br />
Parmeggiani A, Posar A, Leonardi M, Rossi PG. (1992). <strong>Neuro</strong>logical impairment <strong>in</strong> congenital bilateral ptosis<br />
with ophthalmoplegia. Bra<strong>in</strong> Dev 14:107–109.<br />
Patel NP, Sav<strong>in</strong>o PJ, We<strong>in</strong>berg DA. (1998). Unilateral eyelid ptosis and a red eye. Surv Ophthalmol 43:182–187.<br />
Po SM, Custer PL, Smith ME. (1996). Bilateral lagophthalmos. <strong>An</strong> unusual presentation of metastatic breast<br />
carc<strong>in</strong>oma. Arch Ophthalmol 114:1139–1141.<br />
Ragge NK, Hoyt WF. (1992). Midbra<strong>in</strong> myasthenia: fatiguable ptosis, lid twitch sign, and ophthalmoparesis from<br />
a dorsal midbra<strong>in</strong> glioma. <strong>Neuro</strong>logy 42:917–919.<br />
Riley DE, Lang AE, Lewis A, et al. (1990). Cortical-basal ganglionic degeneration. <strong>Neuro</strong>logy 40:1203–1212.<br />
Ropo A, Ruusuvaara P, Nikki P. (1992). Ptosis follow<strong>in</strong>g periocular or general anesthesia <strong>in</strong> cataract surgery. Acta<br />
Ophthalmol 70:262–265.<br />
Rose M, Landon D, Papadimitrious A, et al. (1997). A rapidly progressive adolescent-onset oculopharyngeal<br />
somatic syndrome with rimmed vacuoles <strong>in</strong> two sibl<strong>in</strong>gs. <strong>An</strong>n <strong>Neuro</strong>l 41:25–31.<br />
Rowland LP, Hirano M, DiMauro S, Schon EA. (1997). Oculopharyngeal muscular dystrophy, other ocular<br />
myopathies, and progressive external ophthalmoplegia. <strong>Neuro</strong>mus Disord 7(suppl 1):S15–S21.<br />
Saeki N, Yamaura A, Sunami K. (2000). Bilateral ptosis with pupil spar<strong>in</strong>g because of a discrete midbra<strong>in</strong> lesion:<br />
magnetic resonance imag<strong>in</strong>g evidence of topographic arrangement with<strong>in</strong> the oculomotor nerve. J <strong>Neuro</strong>ophthalmol<br />
20:130–134.<br />
Sieb JP, Hartmann A. (2000). Relaps<strong>in</strong>g alternat<strong>in</strong>g ptosis <strong>in</strong> two sibl<strong>in</strong>gs. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 69:282–283.<br />
Simonsz HJ, Barlocher K, Rotig A. (1992). Kearns-Sayre syndrome develop<strong>in</strong>g <strong>in</strong> a boy who survived Pearson’s<br />
syndrome caused by mitochondrial DNA deletion. Doc Ophthalmol 82:73–79.<br />
S<strong>in</strong>gh SK, Sekhar GC, Gupta S. (1997). Etiology of ptosis after cataract surgery. J Cataract Refract Surg<br />
23:1409–1413.<br />
Steel DH, Harrad RA. (1996). Unilateral congenital ptosis with ipsilateral superior rectus muscle overaction.<br />
Am J Ophthalmol 122:550–556.<br />
Stidham DB, Butler IJ. (2000). Recurrent isolated ptosis <strong>in</strong> presumed ophthalmoplegic migra<strong>in</strong>e of childhood.<br />
Ophthalmology 107:1476–1478.<br />
Suzuki H, Matsubara T, Kanamaru K, Kojima T. (2000). Chronic hydrocephalus present<strong>in</strong>g with bilateral ptosis<br />
after m<strong>in</strong>or head <strong>in</strong>jury: case report. <strong>Neuro</strong>surgery 47:977–980.
420 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Tandon RK, Burke JP, Strachan IM. (1993). Unilateral congenital fibrosis syndrome present<strong>in</strong>g with hypertropia.<br />
Acta Ophthalmol 71:860–862.<br />
Tomecek FJ, Morgan JK. (1994). Ophthalmoplegia with bilateral ptosis secondary to midbra<strong>in</strong> hemorrhage. A case<br />
with cl<strong>in</strong>ical and radiologic correlation. Surg <strong>Neuro</strong>l 41:131–136.<br />
Tozlovanu V, Forget R, Iancu A, Boghen D. (2001). Prolonged orbicularis oculi activity. A major factor <strong>in</strong> apraxia of<br />
lid open<strong>in</strong>g. <strong>Neuro</strong>logy 57:1013–1018.<br />
Tummala RP, Harrison A, Madison MT, Nussbaum ES. (2001). Pseudomyasthenia result<strong>in</strong>g from a posterior<br />
carotid artery wall aneurysm—a novel presentation: case report. <strong>Neuro</strong>surgery 49:1466–1469.<br />
van der Bosch WA, Lemij HG. (1992). Blepharoptosis <strong>in</strong>duced by prolonged hard contact lens wear. Ophthalmology<br />
99:1759–1765.<br />
Verghese J, Mill<strong>in</strong>g C, Rosenbaum DM. (1999). Ptosis, blepharospasm, and apraxia of eyelid open<strong>in</strong>g secondary to<br />
putam<strong>in</strong>al hemorrhage. <strong>Neuro</strong>logy 53:652.<br />
Wojno TH. (1993). Downgaze ptosis. Ophthalmic Plast Reconstr Surg 9:83–89.<br />
Wszolek ZK, Pfeiffer RF, Bhatt MH, et al. (1992). Rapidly progressive autosomal dom<strong>in</strong>ant park<strong>in</strong>sonism and<br />
dementia with pallido-ponto-nigral degeneration. <strong>An</strong>n <strong>Neuro</strong>l 32:31–320.<br />
Yip PK. (1991). Bilateral ptosis, ataxia and areflexia—a variant of Fisher’s syndrome (letter). J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 54:1121.<br />
Zachariah SB, Wilson MC, Zachariah B. (1994). Bilateral lid ptosis on a supranuclear basis <strong>in</strong> the elderly. JAm<br />
Geriatr Soc 42:215–217.
19 r<br />
Lid Retraction and Lid Lag<br />
What Is the <strong>An</strong>atomy of the Eyelids and What<br />
Bra<strong>in</strong>stem Structures Control Lid Elevation?<br />
In normal adults, the upper lid just covers the superior cornea (1–2 mm) and the lower<br />
lid lies slightly below the <strong>in</strong>ferior corneal marg<strong>in</strong>. Eyelid elevation occurs with<br />
contraction of the levator palpebrae superioris (LPS) muscle <strong>in</strong>nervated by the oculomotor<br />
nerve. Accessory muscles <strong>in</strong>clude Müller’s muscle (sympathetic <strong>in</strong>nervation),<br />
which is embedded <strong>in</strong> the LPS and <strong>in</strong>serts ma<strong>in</strong>ly on the tarsal plate, and the frontalis<br />
muscle (<strong>in</strong>nervated by the temporal branch of the facial nerve), which helps to retract<br />
the lid <strong>in</strong> extreme upgaze (Schmidtke, 1992). Tone <strong>in</strong> the LPS normally parallels that of<br />
the superior rectus muscle, and <strong>in</strong> extreme downgaze both muscles are completely<br />
<strong>in</strong>hibited. However, there is an <strong>in</strong>verse relationship between the LPS and the superior<br />
rectus dur<strong>in</strong>g forced lid closure where the eye elevates (Bell’s phenomenon). The motor<br />
neurons for both levator muscles are <strong>in</strong> the unpaired central caudal nucleus (CCN),<br />
located at the dorsal caudal pole of the oculomotor complex adjacent to the medial<br />
rectus and superior rectus subdivisions. With<strong>in</strong> the CCN, motor neurons of both LPS<br />
muscles are <strong>in</strong>termixed. The region of the nuclear complex of the posterior commissure<br />
is <strong>in</strong>volved <strong>in</strong> lid-eye movement coord<strong>in</strong>ation (Schmidtke, 1992).<br />
The upper lid position is abnormal if it exposes a white band of sclera between the lid<br />
marg<strong>in</strong> and the upper corneal limbus. This may be due to lid retraction (related to<br />
overactivity of the LPS, contracture of the LPS, or hyperactivity of Müller’s muscle), or<br />
lid lag, which is noted on attempted downgaze. Bartley divided lid retraction <strong>in</strong>to four<br />
categories: neurogenic, myogenic (<strong>in</strong>clud<strong>in</strong>g disease processes affect<strong>in</strong>g the neuromuscular<br />
junction), mechanical, and miscellaneous (Bartley, 1996). This chapter adopts this<br />
classification, discusses the etiologies of lid lag and lid retraction, and suggests a<br />
diagnostic approach.<br />
421
422 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What Are the <strong>Neuro</strong>genic Causes of Lid<br />
Retraction and Lid Lag?<br />
<strong>Neuro</strong>genic eyelid retraction and lid lag may be due to supranuclear, nuclear, or<br />
<strong>in</strong>franuclear lesions affect<strong>in</strong>g the LPS or conditions that produce hyperactivity of the<br />
sympathetically <strong>in</strong>nervated Müller’s muscle (Miller, 1985). Preterm <strong>in</strong>fants may have a<br />
benign transient conjugate downward gaze deviation with eyelid retraction thought to<br />
be due to immature myel<strong>in</strong>ation of vertical eye movement control pathways (Kle<strong>in</strong>man,<br />
1994; Miller, 1985). Approximately 80% of normal <strong>in</strong>fants of 14 to 18 weeks of age may<br />
demonstrate bilateral transient lid retraction (‘‘eye-popp<strong>in</strong>g reflex’’) when ambient light<br />
levels are suddenly reduced. Both of these phenomena are usually benign and typically<br />
require no evaluation if transient and <strong>in</strong> isolation (class IV, level C).<br />
Dorsal mesencephalic supranuclear lesions may result <strong>in</strong> eyelid retraction, which is<br />
noted when the eyes are <strong>in</strong> the primary position of gaze or on look<strong>in</strong>g upward (Collier’s<br />
sign or posterior fossa stare). Unlike the retraction from thyroid orbitopathy (see below),<br />
with midbra<strong>in</strong> lid retraction there is typically no retraction <strong>in</strong> downgaze. Patients with<br />
dorsal mesencephalic lesions often have associated vertical gaze palsies and other<br />
dorsal midbra<strong>in</strong> f<strong>in</strong>d<strong>in</strong>gs. The etiologies of the dorsal midbra<strong>in</strong> syndrome and the<br />
workup of these patients are discussed <strong>in</strong> Chapter 14. Spells of lid retraction last<strong>in</strong>g 20<br />
to 30 seconds that may be seen with impend<strong>in</strong>g tentorial bra<strong>in</strong> herniation may be due to<br />
a dorsal mesencephalic mechanism (Miller, 1985).<br />
Lesions of the medial and=or pr<strong>in</strong>cipal portion of the nuclear complex of the posterior<br />
commissure (NPC) are <strong>in</strong>volved <strong>in</strong> lid–eye coord<strong>in</strong>ation and provide <strong>in</strong>hibitory modulation<br />
of levator motor neuronal activity (Schmidtke, 1992). <strong>Cl<strong>in</strong>ical</strong> and experimental<br />
evidence suggests an <strong>in</strong>hibitory premotor network <strong>in</strong> the periaqueductal gray (the<br />
supraoculomotor area or supra III) that is dorsal to the third cranial nerve nucleus and<br />
projects from the NPC to the central caudal subnucleus (Galetta, 1993a,b, 1996;<br />
Schmidtke, 1992). Lesions <strong>in</strong> the region of NPC may produce excessive <strong>in</strong>nervation to<br />
the lids with lid retraction <strong>in</strong> primary position. Bilateral eyelid retraction and eyelid lag<br />
with m<strong>in</strong>imal impairment of vertical gaze has been described with a circumscribed<br />
unilateral lesion immediately rostral and dorsal to the red nucleus <strong>in</strong>volv<strong>in</strong>g the lateral<br />
periaqueductal gray area <strong>in</strong> the region of the NPC (Galetta, 1993a,b, 1996). Eyelid lag<br />
without retraction has also been described <strong>in</strong> pretectal disease, imply<strong>in</strong>g that these lid<br />
signs may have separate neural mechanisms (Galetta, 1996). Vertical gaze paralysis<br />
without eyelid retraction may occur. In these cases the fibers and nucleus of the<br />
posterior commissure are spared and the lesions <strong>in</strong>volve the rostral <strong>in</strong>terstitial nucleus<br />
of the medial longitud<strong>in</strong>al fasciculus (MLF), the <strong>in</strong>terstitial nucleus of Cajal, and the<br />
periaqueductal gray area (Schmidtke, 1992). Ipsilateral ptosis and contralateral superior<br />
eyelid retraction may be due to a nuclear oculomotor nerve syndrome (plus-m<strong>in</strong>us lid<br />
syndrome) (Galetta, 1993b; Gaymard, 1992; Vertrugno, 1997). The plus-m<strong>in</strong>us syndrome<br />
results from a unilateral lesion of the third nerve fascicle with extension rostrally and<br />
dorsally to <strong>in</strong>volve the nucleus of the posterior commissure or its connections. The plusm<strong>in</strong>us<br />
syndrome has been described with glioma, third nerve palsy, orbital myositis,<br />
myasthenia gravis, congenital ptosis, and orbital trauma (Vertrugno, 1997). Also, a<br />
patient has been described with a nuclear third nerve palsy, spar<strong>in</strong>g the caudal central<br />
nucleus and its efferent fibers, who had no ipsilateral ptosis but had contralateral lid<br />
retraction (Gaymard, 2000). The contralateral eyelid retraction was thought to be due to<br />
damage to fibers from the NPC, most probably <strong>in</strong> the region of the supraoculomotor<br />
area, and it is <strong>in</strong>ferred from this case that <strong>in</strong>hibitory connections between the NPC and
Lid Retraction and Lid Lag 423<br />
the central caudal nucleus are unilateral and crossed. A similar crossed pattern may also<br />
exist for excitatory afferents to the central caudal nucleus as hemispheric lesions result<br />
<strong>in</strong> contralateral ptosis.<br />
Paroxysmal superior rectus with LPS spasm is a rare and unique disorder described<br />
<strong>in</strong> a s<strong>in</strong>gle patient with multiple sclerosis (Ezra, 1996). Paroxysms of vertical diplopia<br />
and lid retraction <strong>in</strong> this patient lasted 3 to 4 seconds, and exam<strong>in</strong>ation revealed<br />
<strong>in</strong>termittent right hypertropia, lid retraction, and restriction of downgaze. Magnetic<br />
resonance imag<strong>in</strong>g (MRI) revealed multiple lesions consistent with multiple sclerosis,<br />
<strong>in</strong>clud<strong>in</strong>g a lesion <strong>in</strong> the midbra<strong>in</strong> <strong>in</strong> the region of the third nerve fascicle. Carbamazep<strong>in</strong>e<br />
stopped all the symptoms that were thought due to spontaneous spasm of the<br />
superior rectus=levator complex.<br />
Bilateral episodic retraction of the eyelids may occur as a manifestation of epileptic<br />
discharges associated with petit mal or myoclonic seizures or due to ‘‘levator spasms’’<br />
dur<strong>in</strong>g an oculogyric crisis (Miller, 1985). Lid lag may occur on a supranuclear basis <strong>in</strong><br />
progressive supranuclear palsy, likely due to defective <strong>in</strong>hibition of the levator nuclei<br />
dur<strong>in</strong>g downward gaze (Friedman, 1992; Miller, 1985). Lid lag may occur <strong>in</strong> the acute<br />
phases of Guilla<strong>in</strong>-Barré syndrome (Tan, 1990), and lid retraction may also occur with<br />
park<strong>in</strong>sonism (Miller, 1985; Tan, 1990). Lid retraction has also been described with<br />
Fisher syndrome (Al-D<strong>in</strong>, 1994) and POEMS (peripheral neuropathy, organomegaly,<br />
endocr<strong>in</strong>opathy, M-prote<strong>in</strong>, and sk<strong>in</strong> changes) syndrome (Gheradi, 1994).<br />
Rhythmic upward jerk<strong>in</strong>g of the lids (eyelid nystagmus) refers to eyelid twitches that<br />
are synchronous with the fast phase of horizontal nystagmus on lateral gaze. It has been<br />
ascribed to lateral medullary disease where it may be <strong>in</strong>hibited by near effort. Lid<br />
nystagmus may also be provoked by convergence (Pick’s sign) <strong>in</strong> cerebellar or<br />
medullary pathology. There is a slow down drift of the lid corrected by an upward<br />
flick. Rhythmic upward jerk<strong>in</strong>g of the eyelids may also be associated with vertical<br />
nystagmus, palatal myoclonus, or convergence-retraction nystagmus (Miller, 1985) (see<br />
Chapters 14 and 17).<br />
Eyelid retraction may also occur from paradoxic levator excitation that may be<br />
congenital or acquired supranuclear, nuclear, or <strong>in</strong>franuclear lesions (Miller, 1985).<br />
Paradoxic lid retraction may occur with jaw movement or swallow<strong>in</strong>g (the Marcus<br />
Gunn jaw-w<strong>in</strong>k<strong>in</strong>g phenomenon). This trigem<strong>in</strong>o-oculomotor synk<strong>in</strong>esis occurs on a<br />
congenital basis. Levator contraction with contraction of the external pterygoid muscle<br />
is the most common form of trigem<strong>in</strong>o-oculomotor synk<strong>in</strong>esis (Miller, 1985). The<br />
<strong>in</strong>volved eyelid is usually ptotic, but may be normal or even retracted while the jaw<br />
muscles are <strong>in</strong>active. Elevation of the lid occurs when the mandible is moved to the<br />
opposite side, when the mandible is projected forward or the tongue protruded, or on<br />
wide open<strong>in</strong>g of the mouth. These patients commonly have other associated ocular<br />
abnormalities <strong>in</strong>clud<strong>in</strong>g strabismus (e.g., double elevator palsy or superior rectus<br />
palsy), amblyopia, and anisometropia (Miller, 1985). <strong>An</strong>other rare form of trigem<strong>in</strong>ooculomotor<br />
synk<strong>in</strong>esis is levator contraction with contraction of the <strong>in</strong>ternal pterygoid<br />
muscle (i.e., eyelid elevation with closure of the mouth or clench<strong>in</strong>g of the teeth).<br />
Treatment of cases of Marcus Gunn jaw-w<strong>in</strong>k<strong>in</strong>g phenomenon <strong>in</strong>cludes occlusion<br />
therapy for amblyopia, strabismus surgery, and surgery to correct the ptosis or<br />
retraction (Miller, 1985). Paradoxical eyelid retraction may also occur ipsilaterally <strong>in</strong><br />
congenital or acquired horizontal gaze or abducens palsies (Miller, 1985).<br />
Eyelid retraction may also occur with aberrant regeneration of the third nerve. The lid<br />
may elevate when the eye adducts, elevates, or depresses (Stout, 1993) (see Chapter 11).
424 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Partial paresis of the superior rectus muscle (Mauriello, 1993) or orbital floor ‘‘blowout’’<br />
fractures with globe hypotropia may produce an appearance of lid retraction. Secondary<br />
eyelid retraction (pseudoretraction) may also occur if there is ptosis of the opposite<br />
eyelid (especially when the ptosis is due to disease at or distal to the neuromuscular<br />
junction) when fixat<strong>in</strong>g with the eye with the unilateral ptosis (due to Her<strong>in</strong>g’s<br />
law). Compensatory unilateral orbicularis oculi contraction may mask lid retraction;<br />
therefore, if the orbicularis oculi muscle is also weakened as <strong>in</strong> myasthenia gravis,<br />
contralateral lid retraction becomes more evident. Occlusion of the eye on the side of the<br />
ptosis restores the retracted eyelid to a normal position.<br />
Sympathetic overactivity may cause lid retraction by contraction of Müller’s muscle.<br />
Intermittent oculosympathetic irritation may cause cyclic sympathetic spasm. The pupil<br />
dilates for 40 to 60 seconds and may be associated with lid retraction, facial hyperhidrosis,<br />
and headache (Claude-Bernard syndrome) (Burde, 1992). Sympathetic overactivity<br />
may also play a role <strong>in</strong> the lid retraction rarely noted <strong>in</strong> ipsilateral orbital<br />
blowout fractures. Sympathomimetic drops used <strong>in</strong> rout<strong>in</strong>e dilation of the pupils for<br />
ophthalmoscopy (e.g., phenylephr<strong>in</strong>e) may also cause lid retraction. F<strong>in</strong>ally, volitional<br />
bilateral lid retraction may occur <strong>in</strong> anxious or psychotic patients (Burde, 1992).<br />
What Are the <strong>Neuro</strong>muscular and Myopathic<br />
Causes of Lid Retraction and Lid Lag?<br />
Congenital maldevelopment or fibrosis of the LPS muscle or tendon may cause eyelid<br />
retraction or entropion at birth (Coll<strong>in</strong>, 1990; Gillies, 1995; Stout, 1993). This eyelid<br />
retraction may be unilateral or bilateral and may be associated with congenital<br />
abnormalities (Miller, 1985). Other causes of congenital eyelid retraction <strong>in</strong>clude<br />
maternal hyperthyroidism (transient), congenital myotonia, and myotonic dystrophy.<br />
Dysthyroid disease (Graves’ ophthalmopathy) with <strong>in</strong>volvement of the LPS is the<br />
most common cause of acquired unilateral or bilateral susta<strong>in</strong>ed eyelid retraction<br />
(Burde, 1992; Miller, 1985). Patients may show retraction of the upper eyelid associated<br />
with <strong>in</strong>frequent or <strong>in</strong>complete bl<strong>in</strong>k<strong>in</strong>g (Stellwag’s sign) and abnormal widen<strong>in</strong>g of the<br />
palpebral fissure (Dalrymple’s sign). When the patient looks downward, there is often<br />
lid lag; the upper eyelid pauses and then <strong>in</strong>completely follows the eye down (Graefe’s<br />
sign). The retracted upper eyelid rema<strong>in</strong>s elevated <strong>in</strong> downgaze <strong>in</strong> dysthyroid disease;<br />
this differentiates dysthyroid eyelid retraction from dorsal midbra<strong>in</strong> eyelid retraction<br />
(Collier’s sign), where the eyelids are also retracted <strong>in</strong> primary position but are typically<br />
normal <strong>in</strong> downgaze (Burde, 1992).<br />
Eyelid retraction <strong>in</strong> patients with thyroid ophthalmopathy may result from excessive<br />
sympathetic activity, LPS fibrosis, local adhesions of the LPS to fixed orbital tissues, or<br />
contracture of the <strong>in</strong>ferior rectus muscle (Feldon, 1990). The lid retraction may be<br />
controlled by botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jection <strong>in</strong>to the LPS (Biglan, 1994; Ebner, 1993). Surgical<br />
procedures are available to improve eyelid retraction with options <strong>in</strong>clud<strong>in</strong>g lateral<br />
tarsorrhaphy, Müller’s muscle and LPS muscle lengthen<strong>in</strong>g, lower eyelid elevation, and<br />
blepharoplasty with orbital fat excision (Ceisler, 1995). Orbital decompression may<br />
improve lid retraction that is due to distortion from the proptotic globe. Strabismus<br />
surgery may relieve the compensatory component of lid retraction related to restrictive<br />
extraocular muscles, but recessions of the <strong>in</strong>ferior rectus muscle often worsen the eyelid
etraction. Therefore, the order of surgery for patients with thyroid ophthalmopathy<br />
who require different surgical procedures should <strong>in</strong> general be first orbital decompression<br />
followed by strabismus surgery and then lid surgery (class IV, level C).<br />
Myasthenia gravis may also be associated with three types of eyelid retraction:<br />
(1) unilateral ptosis and contralateral eyelid retraction due to <strong>in</strong>nervation to elevate the<br />
ptotic lid; (2) ptosis and brief eyelid retraction last<strong>in</strong>g only seconds follow<strong>in</strong>g a saccade from<br />
downgaze to primary position (Cogan’s lid twitch sign); and (3) transient eyelid retraction<br />
last<strong>in</strong>g seconds or m<strong>in</strong>utes after star<strong>in</strong>g straight ahead or look<strong>in</strong>g upward for several<br />
seconds (possibly due to post-tetanic facilitation of the levator muscle) (Miller, 1985).<br />
Other myopathic causes of lid retraction <strong>in</strong>clude hypokalemic or hyperkalemic<br />
periodic paralysis, myotonic muscular dystrophy, after botul<strong>in</strong>um tox<strong>in</strong> <strong>in</strong>jections of<br />
the eyelids, and after eye surgery, <strong>in</strong>clud<strong>in</strong>g superior rectus recession, ptosis repair, and<br />
enucleation (Bartley, 1996; Leatherbarrow, 1994; Miller, 1985).<br />
What Are the Mechanical and Miscellaneous<br />
Etiologies of Lid Lag and Lid Retraction?<br />
Lid Retraction and Lid Lag 425<br />
The eyelid retraction noted with mechanical causes often responds to correction of the<br />
underly<strong>in</strong>g abnormality (Bartley, 1996). Prom<strong>in</strong>ence of the globe, such as may occur<br />
with severe myopia, buphthalmos, proptosis, cherubism, craniosynostosis, or Paget’s<br />
disease, may cause apparent lid retraction (Bartley, 1996; Leatherbarrow, 1994;<br />
Mauriello, 1993; Stout, 1993). Cicatricial scarr<strong>in</strong>g of the eyelid and LPS fibrosis from<br />
eyelid tumors, hemangioma of the orbit, herpes zoster ophthalmicus, atopic dermatitis,<br />
scleroderma, or thermal or chemical burns may also mechanically retract or distort the<br />
eyelids (Bartley, 1996; Burde, 1992; Stout, 1993). Blowout fractures of the orbital floor<br />
may cause upper eyelid retraction on either a neurogenic or mechanistic basis;<br />
hypotropia of the globe can stimulate <strong>in</strong>creased <strong>in</strong>nervation of the superior rectus,<br />
and LPS or traction on the connective sheath of the LPS can elevate the upper eyelid<br />
mechanically (Bartley, 1996). Contact lens wear may also cause upper eyelid retraction,<br />
presumably by mechanical irritation of the palpebral conjunctiva (Bartley, 1996). Lid<br />
retraction due to a lost hard contact lens becom<strong>in</strong>g embedded <strong>in</strong> the upper eyelid has<br />
also been described (We<strong>in</strong>ste<strong>in</strong>, 1993).<br />
Eyelid retraction, often associated with enophthalmos and hypoglobus, may occur<br />
with chronic maxillary s<strong>in</strong>usitis, maxillary s<strong>in</strong>us hypoplasia, and orbital floor resorption<br />
(silent s<strong>in</strong>us syndrome) (Rub<strong>in</strong>, 1994; Soparker, 1994). Radiation or trauma to the orbit<br />
or s<strong>in</strong>us may also be associated with eyelid retraction (Smitt, 1993). A retracted eyelid<br />
may also be a complication of surgical procedures, <strong>in</strong>clud<strong>in</strong>g trabeculectomy for<br />
glaucoma, scleral buckle, frontal s<strong>in</strong>usotomy, blepharoplasty, orbicularis myectomy,<br />
and cataract extraction (Bartley, 1996; Mauriello, 1993; Miller, 1985).<br />
Other miscellaneous entities that have been reported to be associated with eyelid<br />
retraction <strong>in</strong>clude optic nerve hypoplasia, microphthalmos, Down syndrome, hypertension,<br />
men<strong>in</strong>gitis, sphenoid w<strong>in</strong>g men<strong>in</strong>gioma, and superior cul-de-sac lymphoma<br />
(Bartley, 1996; Stout, 1993). Bilateral upper and lower lid retraction may occur with<br />
hepatic cirrhosis (Summerskill’s sign). The existence of this sign has been questioned, as<br />
many of the orig<strong>in</strong>al patients described may well have had Graves’ ophthalmopathy <strong>in</strong><br />
addition to liver disease, but rare cases without thyroid disease have been documented
426 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 19–1. Etiologies of Upper Lid Retraction and Lid Lag<br />
<strong>Neuro</strong>genic<br />
Benign transient lid retraction <strong>in</strong> preterm <strong>in</strong>fants (‘‘eye-popp<strong>in</strong>g reflex’’ <strong>in</strong> <strong>in</strong>fants)<br />
Dorsal midbra<strong>in</strong> syndrome<br />
Paroxysmal superior rectus and levator spasm <strong>in</strong> multiple sclerosis<br />
Seizures (petit mal or myoclonic)<br />
Oculogyric crisis<br />
Progressive supranuclear palsy<br />
Autosomal-dom<strong>in</strong>ant cerebellar ataxias<br />
Park<strong>in</strong>son’s disease<br />
Guilla<strong>in</strong>-Barré syndrome (<strong>in</strong>clud<strong>in</strong>g Fisher syndrome)<br />
POEMS (polyneuropathy, organomegaly, endocr<strong>in</strong>opathy, M-prote<strong>in</strong>, sk<strong>in</strong> changes) syndrome<br />
Lid nystagmus<br />
Cerebellar or medullary lesions<br />
Associated with vertical nystagmus<br />
Associated with palatal myoclonus<br />
Associated with convergence-retraction nystagmus<br />
Paradoxic levator excitation<br />
Marcus Gunn jaw-w<strong>in</strong>k<strong>in</strong>g phenomenon<br />
Abducens nerve palsy<br />
Aberrant regeneration of the third nerve<br />
Partial superior rectus paresis<br />
Orbital floor fracture<br />
Pseudoretraction<br />
Sympathetic overactivity<br />
Claude-Bernard syndrome<br />
Sympathomimetic drops<br />
Volitional lid retraction<br />
<strong>Neuro</strong>muscular and myopathic<br />
Congenital<br />
Congenital maldevelopment or fibrosis of the levator<br />
Maternal hyperthyroidism<br />
Congenital myotonia<br />
Myotonic dystrophy<br />
Graves’ ophthalmopathy<br />
Hypokalemic or hyperkalemic periodic paralysis<br />
Myotonic muscular dystrophy<br />
After botul<strong>in</strong>um <strong>in</strong>jection <strong>in</strong>to lids<br />
After eye surgery<br />
Superior rectus recession<br />
Ptosis repair<br />
Enucleation<br />
Mechanical<br />
Prom<strong>in</strong>ence of the globe<br />
Myopia<br />
Buphthalmos<br />
(cont<strong>in</strong>ued)
Table 19–1. (cont<strong>in</strong>ued)<br />
Proptosis<br />
Cherubism<br />
Craniosynostosis<br />
Paget’s disease<br />
Cicatricial scarr<strong>in</strong>g and fibrosis<br />
Eyelid tumors<br />
Hemangioma of the orbit<br />
Herpes zoster ophthalmicus<br />
Atopic dermatitis<br />
Scleroderma<br />
Thermal or chemical burns<br />
Blowout fracture of the orbital floor<br />
Contact lens wear<br />
Contact lens embedded <strong>in</strong> upper lid<br />
Enophthalmos and hypoglobus<br />
Silent s<strong>in</strong>us syndrome (Kubis, 2000; Wan, 2000)<br />
Radiation therapy<br />
Trauma<br />
Surgical procedures<br />
Trabeculectomy<br />
Scleral buckle<br />
Frontal s<strong>in</strong>us surgery<br />
Blepharoplasty<br />
Orbicularis myectomy<br />
Cataract extraction<br />
Miscellaneous associations<br />
Optic nerve hypoplasia<br />
Microphthalmos<br />
Down’s syndrome<br />
Hypertension<br />
Men<strong>in</strong>gitis<br />
Sphenoid w<strong>in</strong>g men<strong>in</strong>gioma<br />
Superior cul-de-sac lymphoma<br />
Hepatic cirrhosis<br />
Source: Repr<strong>in</strong>ted from Bartley, 1996, with permission from Elsevier Science.<br />
Lid Retraction and Lid Lag 427<br />
(Bartley, 1991, 1996; Miller, 1985, 1991). Etiologies of upper lid retraction and lid lag are<br />
listed <strong>in</strong> Table 19–1.<br />
<strong>An</strong> approach to the diagnosis of unilateral or bilateral upper eyelid retraction is<br />
outl<strong>in</strong>ed <strong>in</strong> Figure 19–1. <strong>An</strong> adequate history, ophthalmologic exam<strong>in</strong>ation, and<br />
neurologic exam<strong>in</strong>ation should be able to dist<strong>in</strong>guish the major causes of lid retraction<br />
(class IV, level C).
428 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Figure 19–1. Evaluation of lid retraction.<br />
What Are the Etiologies of Lower Eyelid<br />
Retraction?<br />
Like upper eyelid retraction, retraction of the lower eyelid may be due to neurogenic,<br />
myogenic, and mechanical causes (Brazis, 1991). Congenital paradoxical lower eyelid<br />
retraction on upgaze and unilateral congenital lower eyelid retraction, due to the lid<br />
be<strong>in</strong>g tethered to the orbital marg<strong>in</strong>, have been described. Lower eyelid retraction may<br />
be the earliest cl<strong>in</strong>ical lid sign of a lesion of the facial nerve, and facial nerve lesions are<br />
the most common cause of lower lid retraction (Brazis, 1991). Flaccidity of the lower lid<br />
may be an early manifestation of facial muscle paresis <strong>in</strong> myasthenia and myopathies.<br />
Lower lid retraction may occur with the follow<strong>in</strong>g:<br />
Dysthyroid orbitopathy (with or without proptosis)<br />
Proptosis
Table 19–2. Lower Eyelid Retraction<br />
Senile entropion or ectropion<br />
Enophthalmos<br />
After eye muscle (e.g., <strong>in</strong>ferior rectus recession) or orbital surgery (e.g., orbital floor<br />
‘‘blowout’’ fracture repair, orbitotomy, or maxillectomy)<br />
After scarr<strong>in</strong>g and contraction of lid tissue (e.g., from burns, tumors, trauma,<br />
granulomas of the orbital septum, dermatoses, or surgery) (Bartley, 1996)<br />
With a hypertropia, the ipsilateral lid may appear to be retracted, whereas with a<br />
hypotropia there may be an illusion of contralateral lid retraction. Lid retraction may be<br />
due to elevation of the contralateral lower eyelid with facial contracture follow<strong>in</strong>g Bell’s<br />
palsy, spastic-paretic facial contracture with myokymia, hemifacial spasm, enophthalmos,<br />
or Horner’s syndrome ‘‘upside-down’’ ptosis.<br />
The etiologies of lower eyelid retraction are outl<strong>in</strong>ed <strong>in</strong> Table 19–2.<br />
References<br />
Lid Retraction and Lid Lag 429<br />
<strong>Neuro</strong>genic causes<br />
Congenital paradoxical lower eyelid retraction on upgaze<br />
Unilateral congenital lower eyelid retraction due to the lid be<strong>in</strong>g tethered to the orbital marg<strong>in</strong><br />
Lesion of the facial nerve<br />
Myogenic causes<br />
Myasthenia<br />
Myopathies<br />
Dysthyroid orbitopathy<br />
Mechanical causes<br />
Proptosis<br />
Senile entropion or ectropion<br />
Enophthalmos<br />
After eye muscle or orbital surgery, <strong>in</strong>clud<strong>in</strong>g <strong>in</strong>ferior rectus recession, orbital floor blowout<br />
fracture repair, orbitotomy, or maxillectomy<br />
With scarr<strong>in</strong>g and contraction of lid tissue (e.g., burns, tumors, granulomas of the orbital septum,<br />
dermatoses, or surgery)<br />
Apparent lid retraction<br />
Ipsilateral with hypertropia<br />
Contralateral with hypotropia<br />
With elevation of the contralateral lower eyelid from:<br />
Facial contracture follow<strong>in</strong>g Bell’s palsy (Meadows, 2000)<br />
Spastic-paretic facial contracture with myokymia<br />
Hemifacial spasm<br />
Enophthalmos<br />
Horner’s syndrome (‘‘upside-down’’ ptosis)<br />
Al-D<strong>in</strong> AN, <strong>An</strong>derson M, Eeg-Olofsson O, et al. (1994). <strong>Neuro</strong>-ophthalmic manifestations of the syndrome of<br />
ophthalmoplegia, ataxia, and areflexia. A review. Acta <strong>Neuro</strong>l Scand 89:157–163.<br />
Bartley GB. (1996). The differential diagnosis and classification of eyelid retraction. Ophthalmology 103:168–176.
430 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Bartley GB, Gorman CA. (1991). Hepatic cirrhosis as a doubtful cause of eyelid retraction. Am J Ophthalmol<br />
111:109–110.<br />
Biglan AW. (1994). Control of eyelid retraction associated with Graves’ disease with botul<strong>in</strong>um A tox<strong>in</strong>.<br />
Ophthalmic Surg 25:186–188.<br />
Brazis PW, Vogler JB, Shaw KE. (1991). The ‘‘numb-cheek-limp lower lid’’ syndrome. <strong>Neuro</strong>logy 41:327–328.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1992). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 2nd ed. St. Louis, Mosby,<br />
pp. 362–364.<br />
Ceisler EJ, Bilyk JA, Rub<strong>in</strong> PA, et al. (1995). Results of Mullerectomy and levator aponeurosis transposition for the<br />
correction of upper eyelid retraction <strong>in</strong> Graves’ disease. Ophthalmology 102:483–492.<br />
Coll<strong>in</strong> JR, Allen L, Castronuovo S. (1990). Congenital eyelid retraction. Br J Ophthalmol 74:542–544.<br />
Ebner A. (1993). Botul<strong>in</strong>um tox<strong>in</strong> type A <strong>in</strong> upper lid retraction of Graves’ ophthalmopathy. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 13:258–261.<br />
Ezra E, Plant GT. (1996). Paroxysmal superior rectus and levator palpabrae spasm: a unique presentation of<br />
multiple sclerosis. Br J Ophthalmol 80:187–188.<br />
Feldon SE, Lev<strong>in</strong> L. (1990). Graves’ ophthalmopathy: V. Aetiology of upper eyelid retraction <strong>in</strong> Graves’<br />
ophthalmopathy. Br J Ophthalmol 74:484–485.<br />
Friedman DI, Jankovic J, McCrary JA. (1992). <strong>Neuro</strong>-ophthalmic f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> progressive supranuclear palsy. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 12:104–109.<br />
Galetta SL, Gray LG, Raps EC, Schatz NJ. (1993a). Pretectal eyelid retraction and lag. <strong>An</strong>n <strong>Neuro</strong>l 33:554–557.<br />
Galetta SL, Gray LG, Raps EC, et al. (1993b). Unilateral ptosis and contralateral eyelid retraction from a thalamicmidbra<strong>in</strong><br />
<strong>in</strong>farction. Magnetic resonance imag<strong>in</strong>g correlation. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:221–224.<br />
Galetta SL, Raps EC, Liu GT, et al. (1996). Eyelid lag without retraction <strong>in</strong> pretectal disease. J <strong>Neuro</strong>-ophthalmol<br />
16:96–98.<br />
Gaymard B, Huynh C, Laffont I. (2000). Unilateral eyelid retraction. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 68:390–<br />
392.<br />
Gaymard B, Lafitte C, Gelot A, de Toffol BL. (1992). Plus-m<strong>in</strong>us syndrome. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
55:846–848.<br />
Gheradi RK, Chouaub S, Malapert D, et al. (1994). Early weight loss and high serum tumor necrosis factor-alpha<br />
<strong>in</strong> polyneuropathy, organomegaly, endocr<strong>in</strong>opathy, M prote<strong>in</strong>, sk<strong>in</strong> changes syndrome. <strong>An</strong>n <strong>Neuro</strong>l<br />
35:501–505.<br />
Gillies WE, Harris AJ, Brooks AM, et al. (1995). Congenital fibrosis of the vertically act<strong>in</strong>g extraocular muscles: a<br />
new group of dom<strong>in</strong>antly <strong>in</strong>herited ocular fibrosis with radiologic f<strong>in</strong>d<strong>in</strong>gs. Ophthalmology 102:607–612.<br />
Kle<strong>in</strong>man MD, DiMario FJ Jr, Leconche DA, Zalneraitis EL. (1994). Benign transient downward gaze <strong>in</strong> preterm<br />
<strong>in</strong>fants. Pediatr <strong>Neuro</strong>l 10:313–316.<br />
Kubis KC, Danesh-Meyer H, Bilyk JR, et al. (2000). Unilateral lid retraction dur<strong>in</strong>g pregnancy. Surv Ophthalmol<br />
45:69–76.<br />
Leatherbarrow B, Kwartz J, Sunderland S, et al. (1994). The ‘‘baseball’’ orbital implant: a prospective study. Eye<br />
8:569–576.<br />
Mauriello JA Jr, Palydowycz SB. (1993). Upper eyelid retraction after ret<strong>in</strong>al detachment repair. Ophthalmic Surg<br />
24:694–697.<br />
Meadows A, Hall N, Shah-Desai S, et al. (2000). The House-Brackmann system and assessment of corneal risk <strong>in</strong><br />
facial nerve palsy. Eye 14:353–357.<br />
Miller NR. (1985). <strong>An</strong>atomy and physiology of normal and abnormal eyelid position and movement. In: Miller<br />
NR, ed. Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams & Wilk<strong>in</strong>s, pp. 932–995.<br />
Miller NR. (1991). Hepatic cirrhosis as a cause of eyelid retraction. Am J Ophthalmol 112:94–95.<br />
Rub<strong>in</strong> PAD, Bilyk JR, Shore JW. (1994). Orbital reconstruction us<strong>in</strong>g porous polyethylene sheets. Ophthalmology<br />
101:1679–1708.<br />
Schmidtke K, Buttner-Ennever JA. (1992). Nervous control of eyelid function. A review of cl<strong>in</strong>ical, experimental<br />
and pathologic data. Bra<strong>in</strong> 115:227–247.<br />
Smitt MC, Donaldson SS. (1993). Radiotherapy is successful treatment of orbital lymphoma. Int J Radiat Oncol Biol<br />
Phys 26:59–66.<br />
Soparker CNS, Patr<strong>in</strong>ely JR, Cuaycong MJ, et al. (1994). The silent s<strong>in</strong>us syndrome. A cause of spontaneous<br />
enophthalmos. Ophthalmology 101:772–778.<br />
Stout AU, Borchert M. (1993). Etiology of eyelid retraction <strong>in</strong> children: a retrospective study. J Pediatr Ophthalmol<br />
Strabismus 30:96–99.<br />
Tan E, Kansu T, Kirkali P, Zileli T. (1990). Lid lag and the Guilla<strong>in</strong>-Barré syndrome. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:121–<br />
123.
Lid Retraction and Lid Lag 431<br />
Vetrugno R, Mascalchi M, Marulli D, et al. (1997). Plus m<strong>in</strong>us lid syndrome due to cerebral glioma. A case report.<br />
<strong>Neuro</strong>-<strong>ophthalmology</strong> 18:149–151.<br />
Wan MK, Francis IC, Carter PR, et al. (2000). The spectrum of presentation of silent s<strong>in</strong>us syndrome. J <strong>Neuro</strong><strong>ophthalmology</strong><br />
20:207–212.<br />
We<strong>in</strong>ste<strong>in</strong> GS, Myers BB. (1993). Eyelid retraction as a complication of an embedded hard contact lens. Am<br />
J Ophthalmol 116:102–103.
This page <strong>in</strong>tentionally left blank
20 r<br />
<strong>An</strong>isocoria and Pupillary<br />
Abnormalities<br />
Careful exam<strong>in</strong>ation of pupillary reaction to light and near stimuli, the difference <strong>in</strong><br />
anisocoria <strong>in</strong> light and dark, and attention to dist<strong>in</strong>ctive associated signs and symptoms<br />
facilitate differentiat<strong>in</strong>g the abnormalities <strong>in</strong> pupil size and response to stimuli. Old<br />
photographs may be helpful <strong>in</strong> def<strong>in</strong><strong>in</strong>g the duration of anisocoria. Generally, the<br />
history and exam<strong>in</strong>ation help dist<strong>in</strong>guish the major entities caus<strong>in</strong>g an abnormal large<br />
pupil (e.g., third nerve palsy, tonic pupil, iris damage, pharmacologic dilation, or<br />
sympathetic irritation) or small pupil (e.g., Horner’s syndrome, simple anisocoria,<br />
pharmacologic miosis). Pharmacologic test<strong>in</strong>g confirms the diagnosis and facilitates<br />
topographic localization <strong>in</strong> many cases. Our algorithm cannot account for patients with<br />
multiple causes for anisocoria. For example, Slav<strong>in</strong> reported a case of physiologic<br />
anisocoria with Horner’s syndrome and equal-sized pupils (Slav<strong>in</strong>, 2000).<br />
Is the <strong>An</strong>isocoria More Apparent <strong>in</strong> the Light<br />
or <strong>in</strong> the Dark?<br />
If the anisocoria is greater <strong>in</strong> dim light (stimulates dilation of the pupils), then the defect<br />
is <strong>in</strong> the sympathetic <strong>in</strong>nervation of the pupil. If the anisocoria is greater <strong>in</strong> bright light<br />
(stimulates constriction of the pupil), then the lesion is <strong>in</strong> the parasympathetic <strong>in</strong>nervation<br />
of the pupil. If a large pupil is poorly reactive to light and the visual afferent system<br />
is normal, then a defect <strong>in</strong> the efferent parasympathetic <strong>in</strong>nervation to this pupil is likely<br />
(Burde, 1992). If the light reaction is difficult to compare to the fellow eye, then a<br />
measurement of the anisocoria <strong>in</strong> light and dark may help determ<strong>in</strong>e the pupillary<br />
abnormality.<br />
433
434 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Is Light-Near Dissociation Present?<br />
If the light reaction is poor <strong>in</strong> both eyes but the near reaction is <strong>in</strong>tact, the patient has<br />
bilateral light-near dissociation of the pupils. Table 20–1 lists the causes of light-near<br />
dissociation.<br />
Argyll Robertson pupils are small and irregular and are characterized by light-near<br />
dissociation, variable iris atrophy, and normal afferent visual function. They are<br />
classically described with neurosyphilis, and the lesion is with<strong>in</strong> the rostral midbra<strong>in</strong><br />
and pretectal oculomotor light reflex fibers on the dorsal side of the Ed<strong>in</strong>ger-Westphal<br />
nucleus. There is spar<strong>in</strong>g of the near fibers that approach this nucleus more ventrally.<br />
The pupils are small because supranuclear adrenergic <strong>in</strong>hibitory fibers to the Ed<strong>in</strong>ger-<br />
Westphal nucleus are blocked. Patients with diabetes may also have small, poorly<br />
reactive pupils with light-near dissociation that may appear similar to the Argyll<br />
Robertson pupil.<br />
Is There Other Evidence for a Third Nerve<br />
Palsy?<br />
Patients with anisocoria and a poorly reactive pupil should be evaluated for ipsilateral<br />
third nerve palsy. Sunderland and Hughes suggested that an extraaxial lesion compress<strong>in</strong>g<br />
the third nerve (e.g., unruptured <strong>in</strong>tracranial aneurysm) may cause a dilated pupil<br />
<strong>in</strong> isolation or with m<strong>in</strong>imal ocular motor nerve paresis. <strong>An</strong>isocoria or a dilated pupil <strong>in</strong><br />
the absence of an extraocular motility deficit and=or ptosis, however, is rarely due to a<br />
third nerve paresis (Sunderland, 1952). Intracranial aneurysms (e.g., posterior commu-<br />
Table 20–1. Etiologies of Light-Near Dissociation<br />
Bilateral afferent disease<br />
Bilateral anterior visual pathway (optic nerve, chiasm, tract)<br />
Bilateral ret<strong>in</strong>opathy<br />
Midbra<strong>in</strong> lesions<br />
Dorsal midbra<strong>in</strong> syndrome (Par<strong>in</strong>aud’s syndrome)<br />
Encephalitis=men<strong>in</strong>gitis<br />
Wernicke’s encephalopathy and alcoholism<br />
Demyel<strong>in</strong>ation<br />
P<strong>in</strong>eal tumors<br />
Vascular disease<br />
Argyll Robertson pupil<br />
Diabetes (autonomic neuropathy)<br />
Tonic pupils (e.g., local orbital, neuropathic, Adie’s pupil)<br />
Aberrant third nerve regeneration (not spar<strong>in</strong>g of near but ‘‘restor<strong>in</strong>g’’ of near)<br />
Syr<strong>in</strong>gomyelia (rare)<br />
Familial amyloidosis<br />
Sp<strong>in</strong>ocerebellar ataxia type 1 (SCA-1) (Mabuchi, 1998)
<strong>An</strong>isocoria and Pupillary Abnormalities 435<br />
nicat<strong>in</strong>g artery-<strong>in</strong>ternal carotid artery junction) often produce a fixed and dilated pupil<br />
(pupil-<strong>in</strong>volved third nerve palsy), but this is almost always associated with other signs<br />
of a third nerve palsy (Miller, 1985). Walsh and Hoyt reported a patient with headache<br />
and a unilateral dilated pupil who was found to have an aneurysm at the junction of the<br />
superior cerebellar artery and basilar artery (Walsh, 1969). One week later, however, the<br />
patient developed other signs of a third nerve palsy. Payne and Adamkiewicz reported<br />
a case of unilateral <strong>in</strong>ternal ophthalmoplegia with a posterior communicat<strong>in</strong>g<br />
aneurysm, but this patient also had an <strong>in</strong>termittent exotropia and variable ptosis<br />
(Payne, 1969). Crompton and Moore reported two cases of isolated pupil dilation due<br />
to aneurysm, but these patients developed severe headache and eventual signs of a<br />
third nerve palsy (Crompton, 1981). Fujiwara et al reviewed 26 patients with an<br />
oculomotor palsy due to cerebral aneurysm and reported three with only ptosis and<br />
anisocoria (Fujiwara, 1989).<br />
Basilar aneurysms can produce isolated <strong>in</strong>ternal ophthalmoplegia, but this f<strong>in</strong>d<strong>in</strong>g is<br />
rare and usually the patient rapidly develops signs of external ophthalmoplegia due to<br />
third nerve dysfunction. Gale and Crockard observed transient unilateral mydriasis <strong>in</strong> a<br />
patient with a basilar aneurysm (Gale, 1982). Miller reported an isolated <strong>in</strong>ternal<br />
ophthalmoplegia <strong>in</strong> a patient with a basilar aneurysm (Miller, 1985). Wilhelm et al<br />
described an oculomotor nerve paresis that began as an isolated <strong>in</strong>ternal ophthalmoplegia<br />
<strong>in</strong> 1979 and then developed <strong>in</strong>to a more typical third nerve palsy <strong>in</strong> 1993 due to a<br />
neur<strong>in</strong>oma of the third nerve (Wilhelm, 1995). Kaye-Wilson et al. also described a<br />
patient who <strong>in</strong>itially had only m<strong>in</strong>imal pupil signs due to a neur<strong>in</strong>oma of the third<br />
nerve (Kaye-Wilson, 1994). A mydriatic pupil was the present<strong>in</strong>g sign of a common<br />
carotid artery dissection with the pupil dilation preced<strong>in</strong>g other signs and symptoms of<br />
a third nerve palsy and cerebral ischemia (Koennecke, 1998). These cases are uncommon<br />
presentations, and <strong>in</strong> general an isolated dilated pupil is more likely to be due to local<br />
iris abnormalities, the tonic pupil syndrome, or pharmacologic dilation than third nerve<br />
palsy (class IV, level C).<br />
Other rare cases of <strong>in</strong>terpeduncular cyst, mesencephalic hemorrhage, presumed<br />
ocular motor nerve <strong>in</strong>flammation due to men<strong>in</strong>gitis (e.g., bacterial, cryptococcal or<br />
tuberculous basal), and direct head trauma to the third nerve at the posterior<br />
petrocl<strong>in</strong>oid ligament have been described that presented with an isolated, unilateral,<br />
fixed, and dilated pupil. Other neurologic signs of a third nerve palsy, however, were<br />
present or appeared over time <strong>in</strong> almost all these patients. Unilateral pupillary<br />
<strong>in</strong>volvement from probable preganglionic oculomotor nerve dysfunction (normal<br />
ductions but pupil m<strong>in</strong>imally reactive to light; however, reacted well to near stimuli)<br />
has also been described with superficial siderosis of the central nervous system (CNS)<br />
with selective <strong>in</strong>volvement of the superficially located pupillary fibers (Pelak, 1999). In a<br />
patient with an isolated dilated pupil <strong>in</strong> the presence of normal extraocular motility, a<br />
third nerve palsy can be safely excluded <strong>in</strong> almost every circumstance simply with close<br />
follow-up (class IV, level C).<br />
In <strong>in</strong>determ<strong>in</strong>ate cases, topical pilocarp<strong>in</strong>e 1% can be used as a simple test for third<br />
nerve palsy versus pharmacologic blockade (see below). A pupil dilated from a third<br />
nerve palsy will constrict to pilocarp<strong>in</strong>e 1%, but one with a parasympathetic pharmacologic<br />
blockade will not.
436 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Is There Evidence for Pharmacologic (or Toxic)<br />
Mydriasis or Miosis?<br />
A careful history is usually all that is required for patients with <strong>in</strong>advertent or<br />
<strong>in</strong>tentional (e.g., glaucoma medication, treatment with topical cycloplegics for uveitis)<br />
exposure to agents that may affect pupil size (e.g., mydriatics or miotics). Table 20–2<br />
lists some medications and environmental agents that may result <strong>in</strong> mydriasis or miosis.<br />
Pharmacologically <strong>in</strong>duced pupil abnormalities may produce a large pupil due to<br />
<strong>in</strong>creased sympathetic tone with dilator stimulation (e.g., ocular decongestants, adrenergic<br />
<strong>in</strong>halants <strong>in</strong> the <strong>in</strong>tensive care unit, etc.) or decreased parasympathetic tone with<br />
sph<strong>in</strong>cter block (e.g., belladonna alkaloids, scopolam<strong>in</strong>e patch, antichol<strong>in</strong>ergic <strong>in</strong>halents,<br />
topical gentamic<strong>in</strong>, lidoca<strong>in</strong>e <strong>in</strong>jection <strong>in</strong> orbit, etc.). Small pupils might <strong>in</strong>dicate<br />
decreased sympathetic tone or <strong>in</strong>creased parasympathetic stimulation (e.g., pilocarp<strong>in</strong>e<br />
glaucoma drops, antichol<strong>in</strong>esterases such as flea collar or <strong>in</strong>secticides, etc.).<br />
Nurses, physicians, and other health care workers are particularly prone to <strong>in</strong>advertent<br />
or <strong>in</strong>tentional exposure to pharmacologic mydriatics. The pupil size of patients<br />
with pharmacologic sph<strong>in</strong>cter blockade is often quite large (8 to 12 mm <strong>in</strong> diameter).<br />
This large, dilated pupil is much greater than the mydriasis usually seen <strong>in</strong> typical third<br />
nerve palsy or tonic pupil syndromes. The pupils are evenly affected for 360 degrees,<br />
unlike the irregular pupil seen <strong>in</strong> the tonic pupil or iris trauma. Topical pilocarp<strong>in</strong>e 1%<br />
can be used as a simple test for pharmacologic blockade. A pupil dilated from a third<br />
nerve palsy will constrict to pilocarp<strong>in</strong>e 1%, but a pupil with a parasympathetic<br />
pharmacologic blockade will constrict poorly or not at all to topical miotics. <strong>An</strong> acute<br />
tonic pupil may be unreactive to either light or near stimuli and may be difficult to<br />
dist<strong>in</strong>guish from a pharmacologically dilated pupil or acute traumatic iridoplegia.<br />
Adrenergic pharmacologic mydriasis (e.g., phenylephr<strong>in</strong>e) typically produces<br />
blanched conjunctival vessels, reta<strong>in</strong>s residual light reaction, and produces a retracted<br />
upper lid due to sympathetic stimulation of the upper lid retractor muscle. Most ‘‘eyewhiten<strong>in</strong>g’’<br />
over-the-counter eyedrops (e.g., oxymetazol<strong>in</strong>e, phenylephr<strong>in</strong>e) conta<strong>in</strong><br />
sympathomimetics too weak to dilate the pupil unless the corneal epithelium is<br />
breached (e.g., contact lens wear). Exposure to antichol<strong>in</strong>esterases can result <strong>in</strong> a<br />
miotic pupil (Apt, 1995; Ellenberg, 1992). For cases of presumed isolated dilated or<br />
constricted pupils due to pharmacologic exposure, we recommend close follow-up to<br />
ensure that the pupil returns to normal size. Confirmatory pharmacologic test<strong>in</strong>g could<br />
be considered <strong>in</strong> atypical or persistent cases (class IV, level C).<br />
Are Intermittent or Transient Pupillary<br />
Phenomena Present?<br />
Transient mydriasis or miosis has been reported <strong>in</strong> the follow<strong>in</strong>g conditions: cluster or<br />
migra<strong>in</strong>e headaches (Drummond, 1991); migra<strong>in</strong>e aura without headache (Soriani,<br />
1996); astrocytoma (Berreen, 1990); Horner’s syndrome after carotid puncture; dur<strong>in</strong>g<br />
or after seizure activity (Masjuan, 1997); after reduction of bilateral orbital floor<br />
fractures (Stromberg, 1988); and <strong>in</strong> normal <strong>in</strong>dividuals. Episodic miosis with ptosis<br />
accompanied by ipsilateral nasal stuff<strong>in</strong>ess may occur without headache (cluster s<strong>in</strong>e<br />
headache) (Salveson, 2000). Tadpole-shaped pupils due to segmental spasm of the pupil<br />
sph<strong>in</strong>cter may also be related to a partial postganglionic Horner’s syndrome or
<strong>An</strong>isocoria and Pupillary Abnormalities 437<br />
Table 20–2. Medications and Environmental Agents Associated with Mydriasis or Miosis<br />
Ocular mydriatics<br />
Medications<br />
Topical<br />
Topical parasympatholytics (antichol<strong>in</strong>ergics)<br />
Atrop<strong>in</strong>e<br />
Cyclopentolate (Cyclogyl)<br />
Eucatrop<strong>in</strong>e<br />
Homatrop<strong>in</strong>e<br />
Oxyphenonium<br />
Scopolam<strong>in</strong>e<br />
Tropicamide (Mydriacyl)<br />
Gentamic<strong>in</strong><br />
Topical sympathomimetics (adrenergic)<br />
Apraclonid<strong>in</strong>e (a-adrenergic agonist) (Morales, 2000)<br />
Ep<strong>in</strong>ephr<strong>in</strong>e<br />
Dipivalyl ep<strong>in</strong>ephr<strong>in</strong>e (Prop<strong>in</strong>e)<br />
Phenylephr<strong>in</strong>e (NeoSynephr<strong>in</strong>e)<br />
Coca<strong>in</strong>e (e.g., topical placed <strong>in</strong> nose may back up <strong>in</strong>to conjunctival sac)<br />
Ocular decongestants (tetrahydrozol<strong>in</strong>e hydrochloride, phenerim<strong>in</strong>e maleate,<br />
chlorpheniram<strong>in</strong>e maleate) (Gelmi, 1994)<br />
Topical dexamethasone (vehicle <strong>in</strong> Decadron)<br />
Topical apraclonid<strong>in</strong>e<br />
Aerosolized albuterol sulfate—ipratropium bromide (antichol<strong>in</strong>ergic) given by loosely fitt<strong>in</strong>g<br />
mask (Goldste<strong>in</strong>, 1997)<br />
Adrenergic drugs given <strong>in</strong> a mist for pulmonary therapy may escape around mask and<br />
condense <strong>in</strong> conjunctival sac<br />
<strong>An</strong>esthetic agents for the airway<br />
Phenylephr<strong>in</strong>e=lidoca<strong>in</strong>e spray (Prielipp, 1994)<br />
Nasal vasoconstrictor (phenylephr<strong>in</strong>e)<br />
Aerosolized atrop<strong>in</strong>e (Nakagawa, 1993)<br />
Local and systemic mydriatics (Miller, 1985)<br />
Atrop<strong>in</strong>e (IV) <strong>in</strong> general anesthesia<br />
Benztrop<strong>in</strong>e<br />
Barracuda meat<br />
Calcium<br />
Coca<strong>in</strong>e (Stewart, 1999)<br />
Diphenhydram<strong>in</strong>e<br />
Ep<strong>in</strong>ephr<strong>in</strong>e (Perlman, 1991)<br />
Fenfluram<strong>in</strong>e=norfenfluram<strong>in</strong>e<br />
Glutethimide<br />
Levodopa<br />
Lidoca<strong>in</strong>e local <strong>in</strong>jection (e.g., orbital <strong>in</strong>jection)<br />
Lysergic acid diethylamide<br />
Magnesium<br />
Nalorph<strong>in</strong>e<br />
Nutmeg (? may not have an effect on pupils)<br />
(cont<strong>in</strong>ued)
438 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–2. (cont<strong>in</strong>ued)<br />
Pimozide (? patient also treated with benztrop<strong>in</strong>e)<br />
Propanthel<strong>in</strong>e bromide (Pro-Banth<strong>in</strong>e)<br />
Scopolam<strong>in</strong>e methylbromide (Nussdorf, 2000)<br />
Thiopental<br />
Transdermal scopolam<strong>in</strong>e patches<br />
Tricyclic antidepressants<br />
Plants<br />
Alkaloids (belladonna alkaloids) (antichol<strong>in</strong>ergic effect)<br />
Jimson weed (Datura stramonium)<br />
Blue nightshade or European bittersweet (Solanum dulcamara)<br />
Deadly nightshade (Atropa belladona)<br />
Henbane (Hyoscamus niger)<br />
Moonflower (Datura wrightii or D. meteloides)<br />
Other Datura species (D. suaveolans [angel’s trumpet], aurea, candida, sangu<strong>in</strong>ea, stramonium,<br />
wrightii) (Wilhelm, 1994)<br />
Others<br />
Siderosis bulbi=iron mydriasis—occult <strong>in</strong>traocular iron foreign body (Monteiro, 1993; Scotcher,<br />
1995)<br />
Hypromellose viscoelastic <strong>in</strong> cataract surgery (Tan, 1993)<br />
Ocular miotics<br />
Medications<br />
Topical<br />
Parasympathomimetics (chol<strong>in</strong>ergic)<br />
Aceclid<strong>in</strong>e<br />
Carbachol<br />
Methachol<strong>in</strong>e (Mecholyl)<br />
Organophosphate esters<br />
Physostigm<strong>in</strong>e (eser<strong>in</strong>e)<br />
Pilocarp<strong>in</strong>e<br />
Sympatholytics (antiadrenergic)<br />
Adrenergic blockers<br />
Thymoxam<strong>in</strong>e hydrochloride<br />
Dapiprazole (‘‘RevEyes’’)<br />
Dibenzyl<strong>in</strong>e (hemoxybenzam<strong>in</strong>e)<br />
Phentolam<strong>in</strong>e (Regit<strong>in</strong>e)<br />
Tolazol<strong>in</strong>e (Priscol<strong>in</strong>e)<br />
Guanethid<strong>in</strong>e<br />
Timolol with ep<strong>in</strong>ephr<strong>in</strong>e<br />
Systemic miotics<br />
Adrenergic blockers<br />
Chlorpromaz<strong>in</strong>e<br />
Hero<strong>in</strong><br />
Lidoca<strong>in</strong>e (extradural anesthesia)<br />
Marijuana<br />
Methadone<br />
Morph<strong>in</strong>e and other narcotics<br />
(cont<strong>in</strong>ued)
Table 20–2. (cont<strong>in</strong>ued)<br />
Phenothiaz<strong>in</strong>es<br />
Other<br />
Flea collar (antichol<strong>in</strong>esterase)<br />
Pyrithens and piperonyl butoxide (<strong>in</strong>secticide) (antichol<strong>in</strong>esterase)<br />
migra<strong>in</strong>e phenomenon (occurs between rather than with headache attacks). Some of<br />
these phenomena represent true sympathetic irritation or excess, but the mechanism<br />
rema<strong>in</strong>s controversial. If the transient or <strong>in</strong>termittent nature of the mydriasis can be<br />
firmly established, then these patients should not undergo arteriography or other<br />
test<strong>in</strong>g and should simply be followed for 24 to 48 hours, at which po<strong>in</strong>t improvement<br />
would <strong>in</strong>dicate the benign nature of the mydriasis.<br />
Jacobson reported 24 patients with benign episodic unilateral mydriasis (Jacobson,<br />
1995). The median age of the patients was 31 (range, 14 to 50) and the median duration<br />
of events was 12 hours (range, 10 m<strong>in</strong>utes to 7 days). Associated symptoms <strong>in</strong>cluded<br />
visual blur, headache, orbital pa<strong>in</strong>, monocular photophobia, monocular red eye,<br />
monocular diplopia, and monocular positional transient obscurations. Some cases<br />
were thought due to parasympathetic <strong>in</strong>sufficiency of the iris sph<strong>in</strong>cter. These patients<br />
had associated impaired near vision, impaired accommodative function, and the<br />
anisocoria <strong>in</strong>creased with added ambient light. Other patients had sympathetic hyperactivity<br />
of the iris dilator associated with normal near vision and normal reaction of the<br />
pupil dur<strong>in</strong>g the attack. No associated neurologic disorders were found <strong>in</strong> these<br />
patients. We do not recommend any further evaluation for isolated transient unilateral<br />
mydriasis (class IV, level C).<br />
Is a Structural Iris Abnormality Present?<br />
Careful slit-lamp biomicroscopy of the iris should be performed <strong>in</strong> all patients with<br />
anisocoria to exclude structural iris abnormalities or damage. In many cases, the pupil is<br />
irregular and the structural abnormality can easily be identified. Table 20–3 outl<strong>in</strong>es the<br />
cl<strong>in</strong>ical features of structural iris abnormality and Table 20–4 lists some etiologies of<br />
structural damage to the iris. Abnormalities of the iris are a common cause of<br />
anisocoria. False-positive pharmacologic test<strong>in</strong>g may result <strong>in</strong> patients with structural<br />
abnormalities of the iris that prevent dilation or constriction to pharmacologic agents. In<br />
these cases, it may be necessary to test the <strong>in</strong>tegrity of the pupil dilation or constriction<br />
Table 20–3. <strong>Cl<strong>in</strong>ical</strong> Characteristics of Abnormalities of the Iris Structure<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 439<br />
No associated ptosis or ocular motility disturbance (vs. third nerve palsy)<br />
Pupil often irregular with disruption of pupillary marg<strong>in</strong> due to tears <strong>in</strong> iris sph<strong>in</strong>cter (vs. the smooth<br />
marg<strong>in</strong> seen <strong>in</strong> drug-related pupillary abnormalities)<br />
Irregular contraction of the pupil to light<br />
Eventually iris atrophy may occur<br />
Poor or no response to direct parasympathomimetic (e.g., 1% pilocarp<strong>in</strong>e)
440 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–4. Etiologies of Abnormalities of Iris Structure<br />
Congenital aplasia of the iris sph<strong>in</strong>cter and dilator muscles (Buys, 1993)<br />
Increased <strong>in</strong>traocular pressure due to acute angle closure glaucoma (sph<strong>in</strong>cter paresis due to iris<br />
ischemia)<br />
Intraocular <strong>in</strong>flammation (e.g., iritis)<br />
Ischemia (e.g., ocular ischemic syndrome, iris ischemia after anterior chamber air=gas <strong>in</strong>jection after<br />
deep lamellar keratoplasty for keratoconus) (Maur<strong>in</strong>o, 2002)<br />
Mechanical (e.g., iris tumor, <strong>in</strong>traocular lens)<br />
Surgical (e.g., iridectomy, iridotomy, iris damage)<br />
Trauma<br />
Blunt trauma (traumatic iridoplegia)<br />
Sph<strong>in</strong>cter tears at the pupillary marg<strong>in</strong><br />
Atonic pupil after cataract extraction (Behndig, 1998)<br />
capacity by apply<strong>in</strong>g a topical direct sympathomimetic or parasympathomimetic (class<br />
IV, level C).<br />
Is a Tonic Pupil Present?<br />
The typical presentation of the tonic pupil is isolated anisocoria that is greater <strong>in</strong> light.<br />
Patients often present with acute awareness of the dilated pupil. The cl<strong>in</strong>ical features of<br />
a tonic pupil are listed <strong>in</strong> Table 20–5.<br />
Pharmacologic test<strong>in</strong>g with low-dose pilocarp<strong>in</strong>e (1=8%) may demonstrate chol<strong>in</strong>ergic<br />
supersensitivity <strong>in</strong> the tonic pupil (a more miotic response than the fellow eye).<br />
Leavitt et al suggested a solution of 0.0625% pilocarp<strong>in</strong>e (Leavitt, 2002). Unfortunately,<br />
chol<strong>in</strong>ergic supersensitivity is not uniformly present <strong>in</strong> tonic pupils (80% with topical<br />
pilocarp<strong>in</strong>e test<strong>in</strong>g) and is not specific for postganglionic parasympathetic denervation.<br />
Supersensitivity has been reported after oculomotor nerve palsy (Cox, 1991; Jacobson,<br />
1990, 1994). In addition, larger-sized pupils normally constrict more than smaller pupils<br />
to the same dose of topical chol<strong>in</strong>ergics. Jacobson recommends evaluat<strong>in</strong>g chol<strong>in</strong>ergic<br />
supersensitivity responses <strong>in</strong> darkness to m<strong>in</strong>imize the mechanical resistance factors of<br />
large and small pupil size (Jacobson, 1990, 1994). A larger pupil that becomes the<br />
Table 20–5. <strong>Cl<strong>in</strong>ical</strong> Features of a Tonic Pupil<br />
Poor pupillary light reaction<br />
Segmental palsy of the sph<strong>in</strong>cter<br />
Tonic pupillary near response with light-near dissociation (near response not ‘‘spared’’ but<br />
‘‘restored’’ due to aberrant regeneration)<br />
Chol<strong>in</strong>ergic supersensitivity of the denervated muscles<br />
Accommodation paresis (that tends to recover)<br />
Induced astigmatism at near<br />
Tonicity of accommodation<br />
Occasional ciliary cramp with near work<br />
Occasionally regional corneal anesthesia (trigem<strong>in</strong>al ophthalmic division fibers <strong>in</strong> ciliary ganglion<br />
damaged)
smaller pupil <strong>in</strong> darkness after topical chol<strong>in</strong>ergics is more likely a supersensitive<br />
response (Jacobson, 1990, 1994).<br />
Is the Tonic Pupil Isolated?<br />
The history and exam<strong>in</strong>ation should be able to differentiate the various associations of<br />
secondary pupils from idiopathic Adie’s tonic pupil syndrome. Table 20–6 lists the<br />
causes of a tonic pupil.<br />
Is This Adie’s Tonic Pupil Syndrome?<br />
The cl<strong>in</strong>ical features of Adie’s tonic pupil syndrome, based on Thompson’s extensive<br />
review (Thompson, 1977a,b) and the literature, is reported <strong>in</strong> Table 20–7. With the tonic<br />
pupil, the iris sph<strong>in</strong>cter and ciliary muscles become supersensitive to acetylchol<strong>in</strong>e, and<br />
thus when they are stimulated their response is strong and tonic and their relaxation is<br />
slow and susta<strong>in</strong>ed. Initially there is an isolated <strong>in</strong>ternal ophthalmoplegia, and <strong>in</strong> the<br />
acute stage there is no reaction to light or near stimuli at all. The diagnosis of a tonic<br />
pupil can usually be made on cl<strong>in</strong>ical grounds alone (class IV, level B).<br />
What Causes the Adie’s Tonic Pupil<br />
Syndrome?<br />
The pathophysiology of Adie’s tonic pupil is damage to the ciliary ganglion (Kardon,<br />
1998; Phillips, 1996; Soylev, 1997). More than 90% of the ciliary ganglion cells normally<br />
serve the ciliary body and only 3% serve the iris sph<strong>in</strong>cter. After damage to the ciliary<br />
ganglion, aberrant regeneration of fibers orig<strong>in</strong>ally dest<strong>in</strong>ed for the ciliary body now<br />
<strong>in</strong>nervate the iris sph<strong>in</strong>cter. The <strong>in</strong>itially mydriatic pupil may become smaller over time<br />
(‘‘little old Adie’s’’) and <strong>in</strong>deed Adie’s tonic pupil may present as a miotic pupil (acute<br />
awareness rather than acute onset of anisocoria). Although most Adie’s tonic pupils<br />
present unilaterally, bilateral <strong>in</strong>volvement may develop at a rate of 4% per year<br />
(Thompson, 1977a). Thompson reviewed 220 cases from the literature and reported<br />
that 20% were bilateral (Thompson, 1977a). Rarely, Adie’s syndrome may be associated<br />
with a chronic cough likely related to vagal <strong>in</strong>volvement (Kimber, 1998).<br />
Should <strong>Neuro</strong>imag<strong>in</strong>g Studies Be Performed <strong>in</strong><br />
Adie’s Syndrome?<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 441<br />
Once the diagnosis of the Adie’s tonic pupil is confirmed cl<strong>in</strong>ically and=or pharmacologically,<br />
no neuroimag<strong>in</strong>g studies are required (class III–IV, level C).
442 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–6. Etiologies of a Tonic Pupil<br />
Local (ocular or orbital) lesion affect<strong>in</strong>g ciliary ganglion or nerve<br />
Infection (Capputo, 1992)<br />
Campylobacter jejuni enteritis (Roberts, 1995)<br />
Cellulitis<br />
Chickenpox<br />
Choroiditis<br />
Diphtheria<br />
Herpes simplex virus<br />
Herpes zoster virus<br />
HTLV-II (Hjelle, 1992)<br />
Influenza<br />
Measles<br />
Parvovirus B19 (Corridan, 1991)<br />
Pertussis<br />
Scarlet fever<br />
S<strong>in</strong>usitis<br />
Syphilis<br />
Varicella virus (Hodgk<strong>in</strong>s, 1993)<br />
Viral hepatitis<br />
Inflammation<br />
Iritis=uveitis damage to ciliary ganglion<br />
Rheumatoid arthritis<br />
Sarcoidosis<br />
Vogt-Koyanagi-Harada syndrome (Kim, 2001a)<br />
Ischemia<br />
Orbital vasculitis<br />
Lymphomatoid granulomatosis (Haider, 1993)<br />
Migra<strong>in</strong>e (Purv<strong>in</strong>, 1995)<br />
Giant cell arteritis<br />
Orbital or choroidal tumor (Haider, 1993)<br />
Polyarteritis nodosa (Bennett, 1999)<br />
Local anesthesia (Perlman, 1991)<br />
Inferior dental block<br />
Injection of retrobulbar alcohol<br />
Surgery (Bodker, 1993; Golnik, 1995; Halpern, 1995; Saiz, 1991)<br />
Cataract surgery (Monson, 1992; Saiz, 1991)<br />
Cryotherapy<br />
Diathermy<br />
Penetrat<strong>in</strong>g keratoplasty<br />
Ret<strong>in</strong>al surgery<br />
Strabismus surgery<br />
Orbital surgery (Bodker, 1993)<br />
Laser therapy<br />
(cont<strong>in</strong>ued)
Table 20–6. (cont<strong>in</strong>ued)<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 443<br />
Toxicity<br />
Qu<strong>in</strong><strong>in</strong>e<br />
Trichloroethylene<br />
Trauma (nonsurgical)<br />
Blunt trauma to ciliary plexus<br />
Orbital floor fracture<br />
Retrobulbar hemorrhage<br />
Damage to short ciliary nerves<br />
<strong>Neuro</strong>pathic<br />
Peripheral or autonomic neuropathy<br />
Amyloidosis (Davies, 1999)<br />
Diabetes<br />
Alcohol-related<br />
Familial dysautonomia<br />
Hereditary neuropathy (e.g., Charcot-Marie-Tooth disease)<br />
Guilla<strong>in</strong>-Barré syndrome<br />
Fisher syndrome (<strong>in</strong>clud<strong>in</strong>g isolated bilateral <strong>in</strong>ternal ophthalmoplegia with IgG anti-GQ1b<br />
antibodies) (Berlit, 1992; Caccavale, 2000; Cher, 1993; Igarishi, 1992; Ishikawa, 1990;<br />
Mori, 2001; Radziwill, 1998; Sawada, 1990)<br />
Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyradiculoneuropathy (Midroni, 1996)<br />
Acute sensorimotor polyneuropathy with tonic pupils and abduction deficit with polyarteritis<br />
nodosa (Bennett, 1999)<br />
Pandysautonomia<br />
Progressive autonomic failure<br />
Shy-Drager syndrome<br />
Ross’ syndrome (tonic pupil, hyporeflexia segmental anhidrosis) (Sh<strong>in</strong>, 2000; Weller, 1992; Wolfe,<br />
1995)<br />
Sjögren’s syndrome (Bachmeyer, 1997; Vetrugno, 1997)<br />
Systemic <strong>in</strong>fectious (e.g., syphilis)<br />
Paraneoplastic<br />
Eaton-Lambert syndrome (Wirtz, 2001)<br />
Carc<strong>in</strong>omatous neuropathy<br />
Congenital neuroblastoma with Hirschsprung disease and central hypoventilation syndrome<br />
(Lambert, 2000)<br />
Unilateral Adie pupil <strong>in</strong> patient with small cell lung cancer and anti-Hu antibodies (Kimura<br />
Bruno, 2000)<br />
Congenital neuroblastoma, Hirschsprung disease, central hypoventilation syndrome<br />
(Lambert, 2000)<br />
Follow<strong>in</strong>g oculomotor nerve palsy (Cox, 1991)<br />
Adie’s tonic pupil syndrome
444 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–7. <strong>Cl<strong>in</strong>ical</strong> Features of Adie’s Syndrome<br />
Prevalence 2 cases per 1000 population<br />
Mean age 32 years<br />
Female to male ratio 2.6 : 1<br />
Unilateral 80%<br />
Reduced deep tendon reflexes 89%<br />
Sector palsy 100%*<br />
Accommodative paresis 66%<br />
Bilateral 4% per year<br />
Chol<strong>in</strong>ergic supersensitivity 80%<br />
Decreased regional corneal sensation 90%<br />
Prognosis Accommodative paresis resolves over months<br />
Pupil light reaction usually does not recover<br />
Pupil smaller with time (‘‘little old Adie’s’’)<br />
Most symptoms resolve spontaneously<br />
*In patients with some degree of light reaction.<br />
What Treatment Is Recommended for Adie’s<br />
Syndrome?<br />
Patients with Adie’s syndrome often compla<strong>in</strong> of difficulty read<strong>in</strong>g due to accommodative<br />
paresis. The treatment of Adie’s tonic pupil is usually reassurance alone. Unequal<br />
bifocal read<strong>in</strong>g aids or a unilateral frosted bifocal segment may be needed for patients<br />
with accommodative paresis. The use of topical low-dose pilocarp<strong>in</strong>e or eser<strong>in</strong>e has<br />
been suggested by some authors for Adie’s syndrome, but may precipitate ciliary<br />
spasm, <strong>in</strong>duce myopia, cause browache, or worsen anisocoria due to miosis (Thompson,<br />
1977a,b). We do not generally recommend treatment for Adie’s tonic pupil (class IV,<br />
level C).<br />
When Does One Perform Syphilis Serology <strong>in</strong><br />
Bilateral, Tonic or Miotic, Irregular Pupils<br />
with Light-Near Dissociation?<br />
Thompson recommends that all patients with bilateral tonic pupils should have<br />
serologic test<strong>in</strong>g for syphilis (Thompson, 1977a). Fletcher and Sharpe reported that<br />
five of 60 consecutive patients with tonic pupils had positive serology for syphilis<br />
(Fletcher, 1986). Of these patients, all were bilateral tonic pupils and none presented<br />
with acute mydriasis or cycloplegia. We recommend syphilis serology for unexpla<strong>in</strong>ed<br />
bilateral tonic pupils (class IV, level C).<br />
The Argyll Robertson pupil consists of bilateral, miotic, irregular pupils with lightnear<br />
dissociation. Although classically described with neurosyphilis, other entities may<br />
produce a similar cl<strong>in</strong>ical syndrome. These etiologies <strong>in</strong>clude diabetes, chronic alcoholism,<br />
encephalitis, multiple sclerosis, degenerative diseases of the CNS (e.g., Charcot-
Marie-Tooth), rare midbra<strong>in</strong> tumors, herpes zoster, neurosarcoidosis, and lymphocytic<br />
men<strong>in</strong>goradiculitis.<br />
Is the Pupillary Light Reaction Normal?<br />
If the pupillary light reaction is normal <strong>in</strong> both eyes, then physiologic (simple)<br />
anisocoria (Lam, 1996), a Horner’s syndrome, sympathetic irritation, or pharmacologic<br />
mydriasis should be considered.<br />
Is the <strong>An</strong>isocoria Isolated?<br />
If the patient has an isolated anisocoria (e.g., no ptosis or dilation lag, no evidence of iris<br />
<strong>in</strong>jury or drugs, and not related to Adie’s tonic pupil or other <strong>in</strong>nervational defects),<br />
then simple (physiologic or central) anisocoria is likely to be present (Lam, 1996). Simple<br />
anisocoria may have a prevalence of up to 21% (range 1 to 90% <strong>in</strong> various studies), and<br />
most of these patients have an anisocoria of less than 0.4 mm that is usually only<br />
<strong>in</strong>termittently present (Lam, 1996). The anisocoria tends to be equal <strong>in</strong> light or dark.<br />
Topical coca<strong>in</strong>e will dilate both pupils equally (see ‘‘What Is Pharmacologic Localization<br />
of HS,’’ below). It is assumed that <strong>in</strong> these patients <strong>in</strong>hibition of the sph<strong>in</strong>cter nuclei <strong>in</strong><br />
the midbra<strong>in</strong> is not ‘‘balanced’’ with any precision that is necessary for clear b<strong>in</strong>ocular<br />
vision.<br />
Is a Horner’s Syndrome Present?<br />
Interruption of the ocular sympathetic pathway is known as a Horner’s syndrome (HS).<br />
HS is characterized cl<strong>in</strong>ically by the signs listed <strong>in</strong> Table 20–8.<br />
HS may result from a lesion anywhere along a three-neuron pathway that arises as a<br />
first-order (central) neuron from the posterolateral hypothalamus, descends <strong>in</strong> the<br />
Table 20–8. <strong>Cl<strong>in</strong>ical</strong> F<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> Horner’s Syndrome<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 445<br />
Ipsilateral mild (usually < 2 mm) ptosis (due to denervation of the Müller’s muscle of the upper<br />
eyelid)<br />
‘‘Upside down ptosis’’ (from sympathetic denervation to the lower eyelid retractors)<br />
Apparent enophthalmos<br />
<strong>An</strong>isocoria due to ipsilateral miosis<br />
Dilation lag (slow dilation of the pupil after the lights are dimmed)<br />
Increased accommodative amplitude or accommodative paresis (Miller, 1985)<br />
Transient (acute phase) ocular hypotony and conjunctival hyperemia<br />
Variable ipsilateral facial anhidrosis<br />
Ipsilateral straight hair <strong>in</strong> congenital cases<br />
Heterochromia of the iris (usually congenital but rarely acquired) (Dissenhouse, 1992;<br />
Miller, 1985)<br />
Rarely, neurotrophic corneal endothelial failure with pa<strong>in</strong> and stromal edema (Zamir, 1999)
446 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
bra<strong>in</strong>stem and lateral column of the sp<strong>in</strong>al cord to exit at the cervical (C8) and thoracic<br />
(T1-T2) levels (ciliosp<strong>in</strong>al center of Budge) of the sp<strong>in</strong>al cord as a second-order neuron.<br />
This second-order (<strong>in</strong>termediate) preganglionic neuron exits the ventral root and arches<br />
over the apex of the lung to ascend <strong>in</strong> the cervical sympathetic cha<strong>in</strong>. The second-order<br />
neurons synapse <strong>in</strong> the superior cervical ganglion and exit as a third-order neuron. The<br />
neural fibers for sweat<strong>in</strong>g of the face travel with the external carotid artery. The thirdorder<br />
postganglionic neuron travels with the carotid artery <strong>in</strong>to the cavernous s<strong>in</strong>us.<br />
With<strong>in</strong> the cavernous s<strong>in</strong>us, the sympathetic fibers jo<strong>in</strong> the abducens nerve for a short<br />
course and then travel with the ophthalmic division of the trigem<strong>in</strong>al nerve and jo<strong>in</strong> the<br />
nasociliary branch of the trigem<strong>in</strong>al nerve. The fibers pass through the ciliary ganglion<br />
and to the eye as the long and short ciliary nerves (Burde, 1992; Miller, 1985).<br />
The evaluation of HS <strong>in</strong>cludes two stages (Burde, 1992; Miller, 1985): (1) recognition of<br />
the cl<strong>in</strong>ical syndrome, and (2) confirmation and localization by pharmacologic test<strong>in</strong>g.<br />
Is the HS Isolated?<br />
Nonisolated HS should undergo imag<strong>in</strong>g with attention to the topographic localization<br />
of the cl<strong>in</strong>ical f<strong>in</strong>d<strong>in</strong>gs.<br />
Is a Central HS Present?<br />
Patients with a central HS can usually be identified by the presence of associated<br />
hypothalamic or bra<strong>in</strong>stem signs or symptoms (e.g., contralateral fourth nerve palsy,<br />
diabetes <strong>in</strong>sipidus, disturbed temperature or sleep regulation, men<strong>in</strong>geal signs, vertigo,<br />
sensory deficits, anhidrosis of the body, etc.). The etiologies of central HS are listed <strong>in</strong><br />
Table 20–9.<br />
Is a Preganglionic (Intermediate) HS Present?<br />
The preganglionic (<strong>in</strong>termediate) HS patient may have neck or arm pa<strong>in</strong>, anhidrosis<br />
<strong>in</strong>volv<strong>in</strong>g the face and neck, brachial plexopathy, vocal cord paralysis, or phrenic nerve<br />
palsy (Burde, 1992). The etiologies of preganglionic <strong>in</strong>termediate HS are listed <strong>in</strong><br />
Table 20–10.<br />
Is a Postganglionic HS Present?<br />
The postganglionic HS patient may have ipsilateral pa<strong>in</strong> and other symptoms suggestive<br />
of cluster or migra<strong>in</strong>e headaches (e.g., tear<strong>in</strong>g, facial flush<strong>in</strong>g, rh<strong>in</strong>orrhea)<br />
(DeMar<strong>in</strong>is, 1994; Manzoni, 1991). <strong>An</strong>hidrosis <strong>in</strong> postganglionic HS is often absent<br />
(Thompson, 1977b). Sweat glands of the forehead are supplied by the term<strong>in</strong>al branches<br />
of sympathetics to the <strong>in</strong>ternal carotid, and <strong>in</strong>volvement of these fibers after they have<br />
separated from the rema<strong>in</strong><strong>in</strong>g facial sweat fibers may expla<strong>in</strong> the occurrence of<br />
anhidrosis of the forehead with spar<strong>in</strong>g of the rest of the face <strong>in</strong> these patients.<br />
Postganglionic HS due to cavernous s<strong>in</strong>us lesions (e.g., thrombosis, <strong>in</strong>fection, neoplasm)
Table 20–9. Central Causes of Horner’s Syndrome<br />
Neoplasm<br />
Hypothalamic=pituitary<br />
Third ventricle<br />
Bra<strong>in</strong>stem<br />
Sp<strong>in</strong>al cord<br />
Infection<br />
Syphilis<br />
Poliomyelitis<br />
Men<strong>in</strong>gitis<br />
Demyel<strong>in</strong>ation<br />
Inflammation (e.g., sarcoid)<br />
Trauma (Worth<strong>in</strong>gton, 1998)<br />
Hemorrhage (Müri, 1995)<br />
Ischemia or <strong>in</strong>farction<br />
Midbra<strong>in</strong> (Bassetti, 1995)<br />
Hypothalamic (Aust<strong>in</strong>, 1991; Mutschler, 1994)<br />
Wallenberg syndrome (Kim, 1994)<br />
Giant cell arteritis (unilateral <strong>in</strong>ternuclear ophthalmoplegia with ipsilateral Horner’s syndrome)<br />
(Askari, 1993)<br />
<strong>An</strong>terior sp<strong>in</strong>al artery thrombosis (Smith, 1999)<br />
Syr<strong>in</strong>gomyelia (? if central or preganglionic Horner’s) (Kerrison, 2000)<br />
Source: Aust<strong>in</strong>, 1991; Burde, 1992; Everett, 1999; Miller, 1985; Mutschler, 1994.<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 447<br />
usually is associated with other localiz<strong>in</strong>g signs such as ipsilateral third, fourth, or sixth<br />
nerve palsy or trigem<strong>in</strong>al nerve dysfunction (Miller, 1985).<br />
Dissection of the <strong>in</strong>ternal carotid artery (e.g., traumatic, spontaneous) may result <strong>in</strong><br />
HS. Biousse et al, for example, studied 146 patients with <strong>in</strong>ternal carotid artery<br />
dissections and found that 28% (41 of 146) had a pa<strong>in</strong>ful HS that was isolated <strong>in</strong> half<br />
of the cases (32 of 65) (Biousse, 1998b). Kerty noted HS <strong>in</strong> 23 of 28 patients with <strong>in</strong>ternal<br />
carotid artery dissection (Kerty, 1999). A third-order HS and orbital and=or ipsilateral<br />
head pa<strong>in</strong> or neck pa<strong>in</strong> of acute onset is diagnostic of <strong>in</strong>ternal carotid artery dissection<br />
unless proven otherwise (Biousse, 1998b). Table 20–11 lists the associated signs and<br />
symptoms of a possible carotid artery dissection (Baumgartner, 2001; Bilbao, 1997;<br />
Biousse, 1998b; Brown, 1995; Burde, 1992; C<strong>in</strong>tron, 1995; Cullom, 1994, 1995; Grau,<br />
1997; Kerty, 1999; Leira, 1998; Purv<strong>in</strong>, 1997; Schiev<strong>in</strong>k, 1998; Venketasubramanian,<br />
1998). Patients with these signs should undergo imag<strong>in</strong>g of the head and neck. We<br />
recommend magnetic resonance (MR) imag<strong>in</strong>g and MR angiography of the head and<br />
neck and consideration for carotid angiography <strong>in</strong> cases of HS due to suspected carotid<br />
dissection (class III–IV, level B). Other etiologies of a postganglionic HS are listed <strong>in</strong><br />
Table 20–12.<br />
Although facial sweat<strong>in</strong>g abnormalities may be helpful <strong>in</strong> localiz<strong>in</strong>g a HS, the<br />
performance of cl<strong>in</strong>ical test<strong>in</strong>g with starch and iod<strong>in</strong>e (e.g., thermoregulatory sweat<br />
test) as described by some authors is somewhat time consum<strong>in</strong>g, messy, and may be<br />
difficult to perform <strong>in</strong> the outpatient sett<strong>in</strong>g. Other tests of facial sweat<strong>in</strong>g may not add<br />
to the cl<strong>in</strong>ical or pharmacologic localization of HS.
448 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–10. Etiologies of Preganglionic Intermediate Horner’s Syndrome<br />
Neoplasm (<strong>in</strong>clud<strong>in</strong>g neck, head, brachial plexus, lung)<br />
Glomus tumors<br />
Breast cancer<br />
Sarcomas<br />
Lung cancer<br />
Lymphoreticular neoplasms (Emir, 2000)<br />
<strong>Neuro</strong>fibroma<br />
<strong>Neuro</strong>blastoma (Simon, 2001)<br />
Thyroid adenoma (Freeman, 1997)<br />
Syr<strong>in</strong>gomyelia (? if central or preganglionic Horner’s) (Kerrison, 2000)<br />
Mediast<strong>in</strong>al or neck lymphadenopathy<br />
Cervicothoracic abnormalities<br />
Cervical rib<br />
Pachymen<strong>in</strong>gitis<br />
Hypertrophic sp<strong>in</strong>al arthritis<br />
Foram<strong>in</strong>al osteophyte<br />
Ruptured <strong>in</strong>tervertebral disc<br />
Thoracic aneurysm<br />
Herpes zoster <strong>in</strong> T3-T4 distribution (Poole, 1997)<br />
Cont<strong>in</strong>uous thoracic epidural analgesia (Aronson, 2000; Liu, 1998; Menendez, 2000)<br />
Neck, brachial plexus or lung trauma or surgery (Oono, 1999)<br />
Carotid endarterectomy (Perry, 2001)<br />
Birth trauma (Klumpke’s paralysis)<br />
Surgical or procedural trauma (Naimer, 2000)<br />
Upper cervical sympathectomies (Smith, 1999)<br />
<strong>An</strong>terior C3-C6 fusion<br />
Radical thyroid surgery<br />
Chest trauma (Hassan, 2000)<br />
Implantation of vagus nerve stimulator for epilepsy (Kim, 2001b)<br />
Internal jugular ve<strong>in</strong> thrombosis <strong>in</strong> polycythemia vera (Glemarec, 1998)<br />
Thoracic aneurysms (Delabrousse, 2000)<br />
Infection or <strong>in</strong>flammation<br />
Migration of foreign body from pharynx to soft tissues of neck (Scaglione, 1999)<br />
Source: Attar, 1998; Burde, 1992; Miller, 1985.<br />
What Is Alternat<strong>in</strong>g HS?<br />
HS that alternates from one eye to the other (usually over days to weeks) is an<br />
uncommon f<strong>in</strong>d<strong>in</strong>g but has been reported <strong>in</strong> multiple system atrophy (Shy-Drager<br />
syndrome) and <strong>in</strong> cervical sp<strong>in</strong>al cord lesions. Tan et al reported a case and reviewed 25<br />
cases from the literature (one vertebral luxation, 14 cervical cord <strong>in</strong>juries, eight Shy-<br />
Drager syndromes, one syr<strong>in</strong>gomyelia, one unknown, and one radiation myelopathy)<br />
(Tan, 1990). Generalized peripheral or autonomic neuropathies (e.g., diabetes, Fisher’s<br />
syndrome, Shy-Drager syndrome) may also result <strong>in</strong> HS (Miller, 1985).
Is the HS Related to Trauma?<br />
Patients with a clear temporal association of the onset of HS with surgical or<br />
nonsurgical trauma to the sympathetic cha<strong>in</strong> <strong>in</strong> the neck or chest do not require<br />
additional evaluation. Pharmacologic test<strong>in</strong>g may aid <strong>in</strong> localization and confirmation<br />
of the diagnosis (class IV, level C). The etiologies of traumatic HS are listed <strong>in</strong><br />
Table 20–13.<br />
What Is Congenital HS?<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 449<br />
Table 20–11. Associated Signs and Symptoms of Carotid Artery Dissection<br />
Ipsilateral orbital, facial, or neck pa<strong>in</strong> (present <strong>in</strong> 90% of cases; ipsilateral to <strong>in</strong>volved vessel <strong>in</strong> 80%)<br />
Diplopia (transient or persistent)<br />
May be due to cavernous carotid <strong>in</strong>volvement<br />
More likely due to transient or permanent impairment of blood supply through <strong>in</strong>ferolateral<br />
trunk supply<strong>in</strong>g third, fourth, and sixth cranial nerves<br />
Also possible due to orbital (extraocular muscle) ischemia or ophthalmic artery occlusion<br />
May have third, fourth, and=or sixth cranial nerve palsies<br />
Transient carotid distribution ischemic attacks (e.g., amaurosis fugax), sometimes evoked by<br />
changes <strong>in</strong> posture<br />
Transient monocular ‘‘sc<strong>in</strong>tillations’’ or ‘‘flash<strong>in</strong>g lights,’’ often related to postural changes or<br />
exposure to bright lights (possible choroidal ischemia)<br />
Visual loss<br />
<strong>An</strong>terior (AION) or posterior (PION) ischemic optic neuropathy<br />
Central ret<strong>in</strong>al artery occlusion (CRAO), branch ret<strong>in</strong>al artery occlusion (BRAO)<br />
Ophthalmic artery occlusion (often associated with head or neck pa<strong>in</strong>)<br />
Ocular ischemic syndrome<br />
Horner’s syndrome (third order, often pa<strong>in</strong>ful)<br />
Transient unilateral mydriasis (rare) (Inzelberg, 2000)<br />
Neck bruit or swell<strong>in</strong>g<br />
Other neurologic deficits<br />
Dysgeusia<br />
T<strong>in</strong>nitus (often pulsatile)<br />
Syncope<br />
Other cranial neuropathy (VI, IX, X, XI, XII)<br />
Sk<strong>in</strong> biopsies might show ultrastructural connective tissue abnormalities (36 of 65 patients<br />
studied <strong>in</strong> one series) usually without other cl<strong>in</strong>ical manifestations of a connective<br />
tissue disease (Brandt, 2001)<br />
Source: Baumgartner, 2001; Bilbao, 1997; Biousse, 1998b; Brandt, 2001; Brown, 1995; Burde, 1992; C<strong>in</strong>tron,<br />
1995; Cullom, 1995; Grau, 1997; Kerty, 1999; Leira, 1998; Mokhtari, 2000; Purv<strong>in</strong>, 1997; Schiev<strong>in</strong>k, 1998;<br />
Venketasubramanian, 1998.<br />
We<strong>in</strong>ste<strong>in</strong> et al reported 11 patients with congenital HS and divided them <strong>in</strong>to three<br />
groups based on cl<strong>in</strong>ical and pharmacologic test<strong>in</strong>g (We<strong>in</strong>ste<strong>in</strong>, 1980):<br />
1. Obstetric per<strong>in</strong>atal forceps (high forceps and rotation for fetal malposition) trauma to<br />
the carotid sympathetic plexus
450 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Table 20–12. Etiologies of a Postganglionic Horner’s Syndrome.<br />
Cavernous s<strong>in</strong>us lesions (Miller, 1985)<br />
Infection (e.g., thrombosis, herpes zoster) (Smith, 1993)<br />
Inflammatory (e.g., sarcoid, Tolosa-Hunt syndrome)<br />
Intracavernous aneurysm<br />
Ophthalmic artery aneurysm (Pritz, 1999)<br />
Neoplasm (e.g., men<strong>in</strong>gioma, metastatic)<br />
Headache syndromes (e.g., cluster or migra<strong>in</strong>e) (DeMar<strong>in</strong>is, 1994, 1998; Manzoni, 1991)<br />
Inflammatory lesions of adjacent structures<br />
Cervical lymphadenopathy (Bollen, 1998)<br />
Otitis media (caroticotympanic plexus)<br />
Petrositis<br />
Sphenoid s<strong>in</strong>us mucocele<br />
Infectious<br />
Severe purulent otitis media (caroticotympanic plexus)<br />
Herpetic geniculate neuralgia<br />
Men<strong>in</strong>gitis<br />
S<strong>in</strong>usitis<br />
Neoplasm<br />
Cavernous s<strong>in</strong>us<br />
Cervical node metastasis<br />
Cervical sympathetic cha<strong>in</strong> schwannoma or neurilemommas (Ganesan, 1997; Hamza, 1997)<br />
Metastatic<br />
Orbital<br />
Systemic peripheral or autonomic disorders<br />
Diabetes (Smith, 1999)<br />
Amyloidosis (Davies, 1999; Smith, 1999)<br />
Ross’ syndrome (Sh<strong>in</strong>, 2000)<br />
Fisher’s syndrome<br />
Mononeuritis multiplex due to cytomegalovirus (CMV) <strong>in</strong> patient with AIDS (Harada, 1998)<br />
Pure autonomic failure (Smith, 1999)<br />
Hereditary sensory and autonomic neuropathy (HSAN) type III (Smith, 1999)<br />
Familial dysautonomia (Smith, 1999)<br />
Dopam<strong>in</strong>e b-hydroxylase deficiency (Smith, 1999)<br />
Multiple systems atrophy (Shy-Drager syndrome) (Smith, 1999)<br />
Trauma <strong>in</strong>clud<strong>in</strong>g surgery<br />
Basilar skull fracture<br />
Orbital fractures<br />
Radical middle ear surgery<br />
Injection or surgery of the gasserian ganglion<br />
Intraoral trauma to <strong>in</strong>ternal carotid sympathetic plexus<br />
Tonsillectomy<br />
Prolonged abnormal posture dur<strong>in</strong>g coma (Thompson, 1998)<br />
Head trauma with <strong>in</strong>tracranial carotid artery <strong>in</strong>jury (Fujisawa, 2001)<br />
Vascular abnormalities of the <strong>in</strong>ternal carotid artery<br />
Congenital anomalies (e.g., congenital agenesis of <strong>in</strong>ternal carotid artery) (Ryan, 2000)<br />
(cont<strong>in</strong>ued)
Table 20–12. (cont<strong>in</strong>ued)<br />
Fibromuscular dysplasia<br />
Carotid artery aneurysms or dissection (Assaf, 1993; Cullom, 1995; Foster, 1991; Mokri, 1992;<br />
Vighetto, 1990) (see Table 20–11)<br />
Arteriosclerosis or thrombosis of the <strong>in</strong>ternal carotid artery (Koivunen, 1999)<br />
Giant cell arteritis (Pascual-Sedano, 1998)<br />
Source: Burde, 1992; Miller, 1985.<br />
2. Presumed superior cervical ganglion lesions (postganglionic lesions with facial<br />
anhidrosis)<br />
3. Surgical (thoracic) or obstetric trauma (brachial plexus) to the preganglionic pathway<br />
Congenital HS (Weissberg, 2001) may result <strong>in</strong> heterochromia of the irides as the<br />
sympathetic <strong>in</strong>nervation of the iris determ<strong>in</strong>es iris pigmentation. Acquired HS,<br />
however, has also been rarely reported to cause iris heterochromia (Dissenhouse,<br />
1992; Miller, 1985).<br />
Table 20–13. Traumatic Horner’s Syndrome<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 451<br />
Direct or <strong>in</strong>direct trauma to the sympathetic cha<strong>in</strong> (Oono, 1999)<br />
Medical procedures<br />
Chest tube above the third posterior rib (Burde, 1992; Gasch, 1996; Miller, 1985; Resnick, 1993)<br />
Extradural analgesia<br />
Lumbar epidural anesthesia (Biousse, 1998a; Hered, 1998; Jeret, 1995; Paw, 1998)<br />
Thoracic epidural analgesia (Liu, 1998)<br />
Percutaneous catheterization of the <strong>in</strong>ternal jugular ve<strong>in</strong> (Gomez, 1993; Guccione, 1992;<br />
Peake, 1990; Reddy, 1998; Vaswani, 1991; Zamir, 1999; Zelligowsky, 1991)<br />
Swan-Ganz catheterization via the <strong>in</strong>ternal jugular ve<strong>in</strong><br />
Carotid artery damage (e.g., carotid angiography) (Maloney, 1980)<br />
Intraoral anesthesia (Penarrocha-Diago, 2000)<br />
Surgery (Burde, 1992; Gasch, 1996; Hamza, 1997; Resnick, 1993; Miller, 1985)<br />
Cardiac surgery (Barbut, 1996)<br />
Median sternotomy<br />
Intentional surgical damage (e.g., sympathectomy)<br />
Thoracic esophageal surgery (Szawlowski, 1991)<br />
<strong>An</strong>terior cervical sp<strong>in</strong>e surgery (Ebraheim, 2000)<br />
Cervical sympathetic cha<strong>in</strong> schwannoma resection (Hood, 2000)<br />
Other<br />
After patient malposition<strong>in</strong>g (Thompson, 1998)<br />
Interscalene brachial plexus block<br />
Stereotactic thalamotomy<br />
Intrathecal Bicill<strong>in</strong> <strong>in</strong>jections <strong>in</strong> the neck<br />
Traumatic <strong>in</strong>ternal carotid dissection (Schiev<strong>in</strong>k, 1998)<br />
Injection <strong>in</strong>to the carotid artery of hero<strong>in</strong> by a drug addict
452 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
What Is Pharmacologic Localization of HS?<br />
Patients with HS that cannot be localized by cl<strong>in</strong>ical exam<strong>in</strong>ation alone should undergo<br />
pharmacologic studies to confirm the diagnosis of HS and localize it to the preganglionic<br />
or postganglionic levels (class III–IV, level B). Although the cl<strong>in</strong>ical features of HS<br />
are classic, they are not pathognomonic. Ipsilateral ptosis and miosis may occur <strong>in</strong><br />
patients without HS (e.g., levator dehiscence and physiologic anisocoria). Pharmacologic<br />
confirmation is relatively easy to perform and is more specific and sensitive than<br />
cl<strong>in</strong>ical diagnosis alone.<br />
Coca<strong>in</strong>e <strong>in</strong>hibits the reuptake of norep<strong>in</strong>ephr<strong>in</strong>e at the neuromuscular junction.<br />
Therefore, topical 5 to 10% coca<strong>in</strong>e dilates a normal pupil (the mydriatic effect is<br />
small and usually about 1 mm) but does not dilate a pupil with HS (regardless of the<br />
location of the affected sympathetic neuron) as well as it dilates a normal pupil.<br />
Therefore, there is an <strong>in</strong>crease <strong>in</strong> the degree of anisocoria after the coca<strong>in</strong>e test <strong>in</strong> a<br />
patient with HS. M<strong>in</strong>imal dilation of the pupil may occur <strong>in</strong> patients with partial<br />
disruption of the oculosympathetic pathway or first-order neuron <strong>in</strong>volvement (Burde,<br />
1992; Miller, 1985). M<strong>in</strong>imal or no dilation of the pupil after topical coca<strong>in</strong>e confirms<br />
that HS exists, but does not localize the responsible process to a preganglionic or<br />
postganglionic location. Friedman et al reported the response to topical coca<strong>in</strong>e 10% <strong>in</strong><br />
24 normal volunteers and thought that 0.5 mm or more of anisocoria was necessary for<br />
the diagnosis of HS (Friedman, 1984). Van der Wiel and Van Gijn compared 12 patients<br />
with HS and 20 normals and found that an anisocoria of 1.0 mm after topical 5% coca<strong>in</strong>e<br />
was sufficient to diagnose HS (Van der Wiel, 1986). Kardon et al adm<strong>in</strong>istered the<br />
coca<strong>in</strong>e test to 50 normals and 119 patients with HS (Kardon, 1990). A post–coca<strong>in</strong>e test<br />
anisocoria value of 1.0 mm gave a mean odds ratio us<strong>in</strong>g logistic regression analysis of<br />
about 5990 : 1 that HS was present (lower 95% confidence limit 37 : 1). These authors<br />
stated that simply measur<strong>in</strong>g the post–coca<strong>in</strong>e test anisocoria (versus measur<strong>in</strong>g the net<br />
change <strong>in</strong> anisocoria) was the best predictor of HS (Kardon, 1990). The amount of<br />
post–coca<strong>in</strong>e test anisocoria and the mean odds (that a patient has HS) are listed <strong>in</strong><br />
Table 20–14.<br />
Hydroxyamphetam<strong>in</strong>e releases stored norep<strong>in</strong>ephr<strong>in</strong>e from the postganglionic adrenergic<br />
nerve end<strong>in</strong>gs at the dilator muscle of the pupil. Therefore, a preganglionic HS<br />
(with <strong>in</strong>tact postganglionic third-order neuron) dilates after adm<strong>in</strong>istration of topical<br />
hydroxyamphetam<strong>in</strong>e 1% (Paredr<strong>in</strong>e), whereas a postganglionic HS pupil does not<br />
dilate (no norep<strong>in</strong>ephr<strong>in</strong>e stores). It should be noted that a false-negative Paredr<strong>in</strong>e test<br />
may occur with postganglionic HS dur<strong>in</strong>g the first week after <strong>in</strong>jury (Donahue, 1996).<br />
The suggested procedure for pharmacologic test<strong>in</strong>g for HS is outl<strong>in</strong>ed <strong>in</strong> Table 20–15<br />
(class III–IV, level C).<br />
A positive test result is noted if the anisocoria <strong>in</strong>creases after the test versus a negative<br />
result if the anisocoria is dim<strong>in</strong>ished or unchanged (this measurement accounts for any<br />
preexist<strong>in</strong>g anisocoria and psychosensory transient dilation effects) (Cremer, 1990a,b).<br />
In <strong>in</strong>termediate and central preganglionic lesions, the affected pupil usually dilates<br />
more <strong>in</strong> response to hydroxyamphetam<strong>in</strong>e possibly because of enhanced receptor<br />
sensitivity at the dilator muscle (Cremer, 1990a,b). There is no effective pharmacologic<br />
test to differentiate central from <strong>in</strong>termediate preganglionic HS. The hydroxyamphetam<strong>in</strong>e<br />
test should be deferred for 24 to 48 hours follow<strong>in</strong>g the coca<strong>in</strong>e test because<br />
coca<strong>in</strong>e will block the effects of the hydroxyamphetam<strong>in</strong>e (Cremer, 1990a,b). Topical<br />
pharmacologic test<strong>in</strong>g should be performed <strong>in</strong> both eyes (the fellow eye serves as a
Table 20–14. Post–Coca<strong>in</strong>e Test <strong>An</strong>isocoria<br />
and the Mean Odds of Horner’s<br />
Syndrome<br />
<strong>An</strong>isocoria (mm) Mean Odds<br />
0.0 1 : 1<br />
0.1 2 : 1<br />
0.2 6 : 1<br />
0.3 14 : 1<br />
0.4 32 : 1<br />
0.5 77 : 1<br />
0.6 185 : 1<br />
0.7 441 : 1<br />
0.8 1050 : 1<br />
0.9 2510 : 1<br />
1.0 5990 : 1<br />
Source: Kardon, 1990.<br />
control) and iatrogenic disruption of the corneal epithelium (e.g., applanation tonometry<br />
or corneal sensitivity test<strong>in</strong>g) should be avoided prior to test<strong>in</strong>g. Patients with congenital<br />
HS may fail to dilate after topical hydroxyamphetam<strong>in</strong>e due to orthograde transsynaptic<br />
dysgenesis of the postganglionic neuron and may <strong>in</strong> reality have a preganglionic<br />
lesion. Friedman et al noted that the pupils of black patients (with heavily pigmented<br />
irides) dilated poorly with coca<strong>in</strong>e, and therefore the test should be <strong>in</strong>terpreted with<br />
more caution <strong>in</strong> black patients (Friedman, 1984; Kardon, 1990). Patients undergo<strong>in</strong>g<br />
topical pharmacologic test<strong>in</strong>g should be <strong>in</strong>formed that ur<strong>in</strong>e drug screen<strong>in</strong>g tests (for<br />
occupational hir<strong>in</strong>g reasons) rema<strong>in</strong> positive for 24 to 48 hours follow<strong>in</strong>g topical test<strong>in</strong>g.<br />
Maloney et al reviewed the cl<strong>in</strong>ical accuracy of the pharmacologic localization of HS<br />
<strong>in</strong> 267 patients (Maloney, 1980). The hydroxyamphetam<strong>in</strong>e test correctly localized<br />
peripheral postganglionic HS <strong>in</strong> 75 (84%) of 89 patients. The reported sensitivity for<br />
identification of a postganglionic HS by hydroxyamphetam<strong>in</strong>e was 96% (Maloney,<br />
1980). Van der Wiel and Van Gijn reported a sensitivity of only 40% (Van der Wiel, 1983),<br />
but their study had a relatively smaller number of patients and excluded patients with<br />
cluster headache. Cremer et al described the results of hydroxyamphetam<strong>in</strong>e test<strong>in</strong>g <strong>in</strong><br />
54 patients with HS and reported a sensitivity of 93% and specificity of 83% (Cremer,<br />
1990a,b). Patients with an isolated postganglionic HS usually have a benign HS,<br />
whereas patients with a preganglionic HS are at risk for harbor<strong>in</strong>g an underly<strong>in</strong>g<br />
Table 20–15. Pharmacologic Test<strong>in</strong>g for Horner’s Syndrome<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 453<br />
One drop of 1% hydroxyamphetam<strong>in</strong>e or coca<strong>in</strong>e (4% or 10%) is <strong>in</strong>stilled <strong>in</strong> the conjunctival sac<br />
of each eye<br />
Both eyes are wiped with a tissue<br />
20 to 40 seconds later a second drop is applied to each eye (to balance the dosage)<br />
The amount of mydriasis (dilation) is measured and the difference <strong>in</strong> the amount of dilation<br />
between the eyes compared<br />
The coca<strong>in</strong>e test and the hydroxyamphetam<strong>in</strong>e test cannot be given on the same day
454 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
malignancy. Grimson and Thompson described 67 patients with HS (Grimson, 1975).<br />
The <strong>in</strong>cidence of malignant neoplasm <strong>in</strong> the preganglionic HS was almost 50% versus<br />
2% <strong>in</strong> postganglionic HS. Some authors have recommended a screen<strong>in</strong>g chest radiograph<br />
for all cases of HS of undeterm<strong>in</strong>ed etiology due to the small risk of misdiagnosis<br />
of a preganglionic HS by the hydroxyamphetam<strong>in</strong>e test (Gasch, 1996). Wilhelm et al<br />
reviewed 90 cases of HS and reported a specificity of 90% for postganglionic HS and<br />
88% for preganglionic HS (Wilhelm, 1992).<br />
Grimson and Thompson reported 120 patients with HS (Grimson, 1979). Of these 120<br />
patients, 41% were preganglionic, and one half of these were due to underly<strong>in</strong>g<br />
neoplasm (Grimson, 1979). Maloney et al reported an etiology <strong>in</strong> 270 (60%) of 450<br />
cases of HS (Maloney, 1980). Of the 180 cases without a def<strong>in</strong>ed etiology, 65 (36%) were<br />
reexam<strong>in</strong>ed (6 months to 28 years later) without a def<strong>in</strong>ite etiology, and the authors thus<br />
felt this <strong>in</strong>dicated a benign and stable orig<strong>in</strong> of the HS. The etiology of the rema<strong>in</strong><strong>in</strong>g 270<br />
cases was as follows: 60 (22%) tumors (23 benign lesions and 37 malignant lesions); 54<br />
(20%) cluster headaches; 45 (16%) iatrogenic cases (e.g., neck surgery and carotid<br />
angiography); 18 (7%) Raeder’s syndromes; 18 (7%) trauma; 13 (5%) cervical disc<br />
protrusions; 13 (5%) congenital cases; 13 (5%) vascular occlusions; 9 (3%) vascular<br />
anomalies, and 27 (10%) miscellaneous (e.g., pneumothorax, herpes zoster, cervical rib,<br />
and mediast<strong>in</strong>al lymphadenopathy) cases. Of these 270 cases, 34 (13%) were central<br />
preganglionic HS, 120 (44%) were <strong>in</strong>termediate preganglionic HS, and 116 (43%) were<br />
peripheral postganglionic HS. Of particular <strong>in</strong>terest, 13 patients <strong>in</strong> this series had<br />
undetected malignancy, and 10 were due to primary or metastatic tumor <strong>in</strong>volv<strong>in</strong>g<br />
the pulmonary apex. N<strong>in</strong>e of these 10 (90%) patients had arm pa<strong>in</strong> (due to presumed<br />
<strong>in</strong>volvement of the adjacent sympathetic cha<strong>in</strong> and C8-T2 nerves).<br />
Giles and Henderson reported a 35.6% <strong>in</strong>cidence (77 cases) of HS due to underly<strong>in</strong>g<br />
neoplasm (Giles, 1958). Of these 77 cases, 58 were malignant (mostly bronchogenic<br />
carc<strong>in</strong>oma and metastatic disease) and 19 were benign (e.g., neurofibroma and thyroid<br />
adenoma) (Giles, 1958).<br />
Is the Evaluation of HS Different <strong>in</strong> Children?<br />
Giles and Henderson reported birth trauma to be the most common etiology of HS <strong>in</strong><br />
children (Giles, 1958). In children, cervical or thoracic tumors (e.g., neuroblastoma,<br />
neurilemmoma, and other congenital or acquired tumors) may cause HS. We recommend<br />
a complete evaluation <strong>in</strong>clud<strong>in</strong>g imag<strong>in</strong>g (e.g., computed tomography scan) of<br />
the cervicothoracic region <strong>in</strong> all children with unexpla<strong>in</strong>ed HS (e.g., no history of birth<br />
trauma to the brachial plexus or other iatrogenic etiology) (Burde, 1992; Gibbs, 1992;<br />
Miller, 1985). Murasella et al reviewed 405 children with neuroblastoma and 14 had HS;<br />
9 of these 14 patients presented with HS (Murasella, 1984). Woodruff et al reported that<br />
two out of 10 children with HS had neuroblastoma (Woodruff, 1988). Sauer and<br />
Lev<strong>in</strong>sohn described seven patients (younger than 11 years old) with HS due to<br />
sp<strong>in</strong>al cord tumor, traumatic brachial plexus palsy, <strong>in</strong>trathoracic aneurysm, embryonal<br />
cell carc<strong>in</strong>oma, neuroblastoma, rhabdomyosarcoma, and thrombosis of the <strong>in</strong>ternal<br />
carotid artery (Sauer, 1976). Iris coloration is not established until several months of age,<br />
and therefore iris heterochromia is not a helpful differential feature of HS <strong>in</strong> these<br />
patients after the per<strong>in</strong>atal period (Burde, 1992). Patients with a substantial history of<br />
per<strong>in</strong>atal head trauma, such as forceps delivery or with evidence of brachial plexus
<strong>in</strong>jury (Klumpke’s paralysis), and pharmacologic evidence of a postganglionic HS do<br />
not require additional evaluation. Childhood HS without a history of clear trauma<br />
(<strong>in</strong>clud<strong>in</strong>g surgical and birth trauma) to the sympathetic cha<strong>in</strong> often have a preganglionic<br />
(<strong>in</strong>termediate) lesion, and therefore should undergo evaluation for an underly<strong>in</strong>g<br />
neoplasm such as neuroblastoma (Burde, 1992; Miller, 1985; Sauer, 1976; Woodruff,<br />
1988). Other etiologies of congenital HS <strong>in</strong>clude viral <strong>in</strong>fections (e.g., cytomegalovirus or<br />
varicella); fibromuscular dysplasia of the <strong>in</strong>ternal carotid artery (possibly posttraumatic);<br />
and HS <strong>in</strong> association with other congenital anomalies (e.g., facial hemiatrophy,<br />
enterogenous cyst, and cervical vertebral anomaly) (Miller, 1985).<br />
What Are the Indications for Imag<strong>in</strong>g Based<br />
on <strong>Cl<strong>in</strong>ical</strong> and Pharmacologic Localization?<br />
Digre et al prospectively performed MR imag<strong>in</strong>g studies <strong>in</strong> 33 patients with HS (Digre,<br />
1992). Of these 33 patients, 13 were preganglionic HS and 20 were postganglionic HS.<br />
Figure 20–1. Evaluation of anisocoria.<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 455
456 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Patients with preganglionic HS without bra<strong>in</strong>stem signs or symptoms underwent T1weighted<br />
sagittal imag<strong>in</strong>g of the entire neck, offset to the ipsilateral side; coronal<br />
imag<strong>in</strong>g of the posterior sp<strong>in</strong>al cord through anterior neck; and axial T1- and T2weighted<br />
imag<strong>in</strong>g from cervical level 2 (C2) to thoracic level 6 (T6). Preganglionic HS<br />
patients with bra<strong>in</strong>stem signs or symptoms underwent extensive imag<strong>in</strong>g of the<br />
sympathetic axis <strong>in</strong>clud<strong>in</strong>g (1) sagittal imag<strong>in</strong>g of the entire bra<strong>in</strong>; (2) axial T1- and<br />
T2-weighted sagittal bra<strong>in</strong> and upper cervical sp<strong>in</strong>e; (3) imag<strong>in</strong>g offset to the side of<br />
<strong>in</strong>terest; (4) coronal T2-weighted imag<strong>in</strong>g of the carotid and cavernous s<strong>in</strong>uses; and<br />
(5) axial T1- and T2-weighted images from the optic chiasm to C4. Four patients had a<br />
lateral medullary <strong>in</strong>farct out of six patients with central preganglionic HS; two patients<br />
had sp<strong>in</strong>al cord=root compression secondary to disc disease, one had apical Pancoast<br />
lung tumor, and one had paravertebral metastatic mass out of seven patients with<br />
preganglionic HS. There were three carotid dissections out of 20 postganglionic HS.<br />
Table 20–16. Pupillary Signs <strong>in</strong> the ICU<br />
Unilateral large poorly reactive pupil<br />
Third nerve palsy<br />
Contusion of eye<br />
Accidental exposure to aerosolized antichol<strong>in</strong>ergics or spill<strong>in</strong>g of atrop<strong>in</strong>e droplets dur<strong>in</strong>g<br />
preparation of the syr<strong>in</strong>ge<br />
Transient (ipsilateral or contralateral) dur<strong>in</strong>g focal seizure or as part of an absence seizure<br />
Oval unilateral nonreactive pupil—transitory appearance <strong>in</strong> bra<strong>in</strong> death<br />
Bilateral mydriasis with normal reaction to light<br />
<strong>An</strong>xiety, delirium, pa<strong>in</strong><br />
Dur<strong>in</strong>g seizure<br />
Botulism<br />
Drugs—systemic atrop<strong>in</strong>e, aerosolized albuterol, amyl nitrate, magnesium sulfate,<br />
norep<strong>in</strong>ephr<strong>in</strong>e, dopam<strong>in</strong>e, am<strong>in</strong>oglycoside, polypeptide, tetracycl<strong>in</strong>e overdose<br />
Bilateral midposition and fixed to light—bra<strong>in</strong> death<br />
Unilateral small, reactive—Horner’s syndrome<br />
Traumatic carotid dissection<br />
Brachial plexopathy<br />
Internal jugular ve<strong>in</strong> catheterization<br />
Extensive thoracic surgery<br />
Spastic miosis <strong>in</strong> acute corneal penetration <strong>in</strong>jury<br />
Bilateral miosis (reaction present but may be difficult to see even with magnify<strong>in</strong>g glass)<br />
Narcotic agents (e.g., morph<strong>in</strong>e)<br />
<strong>An</strong>y metabolic encephalopathy<br />
Respiratory distress with hypercapnea and tachypnea<br />
Bilateral p<strong>in</strong>po<strong>in</strong>t, reactive<br />
Acute pont<strong>in</strong>e lesion, especially hemorrhage<br />
Nonketonic hyperglycemia
What Is the Evaluation for an Isolated<br />
Postganglionic HS?<br />
We do not typically recommend any evaluation for isolated postganglionic HS (class IV,<br />
level C) (Burde, 1992). A number of headache syndromes may be associated with a<br />
postganglionic HS, <strong>in</strong>clud<strong>in</strong>g cluster headache, migra<strong>in</strong>e (Drummond, 1991), and<br />
Raeder’s syndrome (Pimental, 1993). Cluster headache is typically characterized by<br />
the follow<strong>in</strong>g ipsilateral cl<strong>in</strong>ical manifestations <strong>in</strong> addition to headache: conjunctival<br />
<strong>in</strong>jection, tear<strong>in</strong>g, miosis or mydriasis, ptosis, bradycardia, nasal stuff<strong>in</strong>ess, rh<strong>in</strong>orrhea,<br />
facial hyperhidrosis, or flush<strong>in</strong>g. These cluster accompaniments are related to a<br />
comb<strong>in</strong>ation of sympathetic hypofunction and parasympathetic hyperfunction<br />
(e.g., tear<strong>in</strong>g and rh<strong>in</strong>orrhea). Cremer et al reported that 19 of 39 (49%) postganglionic<br />
HS were due to cluster headache (Cremer, 1990a,b). The headache and facial pa<strong>in</strong> of<br />
Raeder’s syndrome can be mimicked by <strong>in</strong>ternal carotid artery dissection however<br />
(Dihne, 2000), and patients suspected of harbor<strong>in</strong>g a dissection should undergo<br />
appropriate imag<strong>in</strong>g of the carotid artery (class IV, level C).<br />
<strong>An</strong> approach to anisocoria is outl<strong>in</strong>ed <strong>in</strong> Figure 20–1. Table 20–16 reviews pupillary<br />
signs of importance <strong>in</strong> the <strong>in</strong>tensive care unit (ICU) sett<strong>in</strong>g.<br />
References<br />
<strong>An</strong>isocoria and Pupillary Abnormalities 457<br />
Abramson SJ, Berdon WE, Ruzal-Shapiro C, Stolar C, Garv<strong>in</strong> J. (1993). Cervical neuroblastoma <strong>in</strong> eleven <strong>in</strong>fants—<br />
a tumor with favorable prognosis. <strong>Cl<strong>in</strong>ical</strong> and radiologic (US, CT, MRI) f<strong>in</strong>d<strong>in</strong>gs. Pediatr Radiol 23:253–257.<br />
Apt L. (1995). Flea collar anisocoria. Arch Ophthalmol 113:403–404.<br />
Aronson LA, Parker GC, Valley R, Norfleet EA. (2000). Acute Horner syndrome due to thoracic epidural analgesia<br />
<strong>in</strong> a paediatric patient. Paediatr <strong>An</strong>aesth 10:89–91.<br />
Askari A, Jolobe OM, Shepherd DI. (1993). Internuclear ophthalmoplegia and Horner’s syndrome due to<br />
presumed giant cell arteritis. J R Soc Med 86:362.<br />
Assaf M, Sweeney PJ, Kosmorsky G, Masaryk T. (1993). Horner’s syndrome secondary to angiogram negative<br />
subadventitial carotid artery dissection. Can J <strong>Neuro</strong>l Sci 20:62–64.<br />
Attar S, Krasna MJ, Sonett JR, et al. (1998). Superior sulcus (Pancoast) tumor: experience with 105 patients.<br />
<strong>An</strong>n Thorac Surg 66:193–198.<br />
Aust<strong>in</strong> CP, Lessel S. (1991). Horner’s syndrome from hypothalamic <strong>in</strong>farction. Arch <strong>Neuro</strong>l 48:332–334.<br />
Bachmeyer C, Zuber M, Dupont S, et al. (1997). Adie syndrome as the <strong>in</strong>itial sign of primary Sjögren syndrome.<br />
Am J Ophthalmol 123:691–692.<br />
Barbut D, Gold JP, He<strong>in</strong>emann MH, et al. (1996). Horner’s syndrome after coronary artery bypass surgery. Urology<br />
46:181.<br />
Bassetti C, Staikov IN. (1995). Hemiplegia vegetativa alterna (ipsilateral Horner’s syndrome and contralateral<br />
hemihyperhidrosis) follow<strong>in</strong>g proximal posterior cerebral artery occlusion. Stroke 26:702–704.<br />
Baumgartner RW, Arnold M, Baumgartner I, et al. (2001). Carotid dissection with and without ischemic events.<br />
Local symptoms and cerebral artery f<strong>in</strong>d<strong>in</strong>gs. <strong>Neuro</strong>logy 57:827–832.<br />
Behndig A. (1998). Small <strong>in</strong>cision s<strong>in</strong>gle-suture-loop pupilloplasty for postoperative atonic pupil. J Cataract Refract<br />
Surg 24:1429–1431.<br />
Bennett JL, Pelak VA, Mourelatos Z, et al. (1999). Acute sensorimotor polyneuropathy with tonic pupils and<br />
abduction deficit: an unusual presentation of polyarteritis nodosa. Surv Ophthalmol 43:341–344.<br />
Berlit P, Rakicky J. (1992). The Miller Fisher syndrome: review of the literature. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 12:57–63.<br />
Berreen JP, Vrabec MP, Penar PL. (1990). Intermittent pupillary dilatation associated with astrocytoma (letter). Am<br />
J Ophthalmol 109:237–239.<br />
Bilbao R, Amoros S, Murube J. (1997). Horner syndrome as an isolated manifestation of an <strong>in</strong>trapetrous <strong>in</strong>ternal<br />
carotid artery dissection. Am J Ophthalmol 123:562–564.<br />
Biousse V, Guevara RA, Newman NJ. (1998a). Transient Horner’s syndrome after lumbar epidural anesthesia.<br />
<strong>Neuro</strong>logy 51:1473–1475.
458 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Biousse V, Touboul P-J, D’<strong>An</strong>glejan-Chatillon J, et al. (1998b). Ophthalmic manifestations of <strong>in</strong>ternal carotid<br />
dissection. Am J Ophthalmol 126:565–577.<br />
Bodker FS, Cytryn AS, Putterman AM, Marschall MA. (1993). Postoperative mydriasis after repair of orbital floor<br />
fracture. Am J Ophthalmol 115:372–375.<br />
Bollen AE, Krikke AP, de Jager AEJ. (1998). Pa<strong>in</strong>ful Horner syndrome due to arteritis of the <strong>in</strong>ternal carotid artery.<br />
<strong>Neuro</strong>logy 51:1471–1472.<br />
Brandt T, Orberk E, Weber R, et al. (2001). Pathogenesis of cervical artery dissections. Association with connective<br />
tissue abnormalities. <strong>Neuro</strong>logy 57:24–30.<br />
Brown J, Danielson R, Donahue SP, Thompson HS. (1995). Horner’s syndrome <strong>in</strong> subadventitial carotid artery<br />
dissection and the role of magnetic resonance angiography. Am J Ophthalmol 119:811–813.<br />
Burde RM, Sav<strong>in</strong>o PJ, Trobe JD. (1992). <strong>Cl<strong>in</strong>ical</strong> Decisions <strong>in</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. St. Louis, Mosby Year Book,<br />
pp. 326–327, 330–332.<br />
Burde RM, Thompson HS. (1991). Hydroxyamphetam<strong>in</strong>e. A good drug lost? (editorial). Am J Ophthalmol<br />
111:100–102.<br />
Buys Y, Buncic JR, Enzenauer RW, Mednick E, O’Keefe M. (1993). Congenital aplasia of the iris sph<strong>in</strong>cter and<br />
dilator muscles. Can J Ophthalmol 28:72–75.<br />
Caccavale A, Mignemi L. (2000). Acute onset of a bilateral mydriasis <strong>in</strong> Miller-Fisher syndrome: a rare neuroophthalmologic<br />
disease. J <strong>Neuro</strong>-ophthalmol 20:61–62.<br />
Capputo AR, Mickey KJ, Guo S. (1992). A varicella-<strong>in</strong>duced pupil abnormality (letter). Pediatrics 89:685–686.<br />
Cher LM, Merory JM. (1993). Miller Fisher syndrome mimick<strong>in</strong>g stroke <strong>in</strong> immunosuppressed patient with<br />
rheumatoid arthritis respond<strong>in</strong>g to plasma exchange. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:138–140.<br />
C<strong>in</strong>tron R, Kattah J. (1995). Oculosympathetic paresis and hemicrania <strong>in</strong> spontaneous dissection of the <strong>in</strong>ternal<br />
carotid artery. Four cases and review of the literature. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:241–248.<br />
Corridan PG, Laws DR, Morrell AJ, Murray PI. (1991). Tonic pupils and human parvovirus (B19) <strong>in</strong>fection. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 11:109–110.<br />
Cox TA, Goldberg RA, Rootman J. (1991). Tonic pupil and Czarnecki’s sign follow<strong>in</strong>g third nerve palsy. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 11:55.<br />
Cremer SA, Thompson HS, Digre KB, Kardon RH. (1990a). Hydroxyamphetam<strong>in</strong>e mydriasis <strong>in</strong> normal subjects.<br />
Am J Ophthalmol 110:66–70.<br />
Cremer SA, Thompson HS, Digre KB, Kardon RH. (1990b). Hydroxyamphetam<strong>in</strong>e mydriasis <strong>in</strong> Horner’s<br />
syndrome. Am J Ophthalmol 110:71–76.<br />
Crompton JL, Moore CF. (1981). Pa<strong>in</strong>ful third nerve palsy: how not to miss an <strong>in</strong>tracranial aneurysm. Aust<br />
J Ophthalmol 9:113–115.<br />
Cullom RD Jr, Cullom ME, Kardon R, Digre K. (1995). Two neuro-ophthalmic episodes separated <strong>in</strong> time and<br />
space. Surv Ophthalmol 40:217–224.<br />
Davies DR, Smith SE. (1999). Pupil abnormality <strong>in</strong> amyloidosis with autonomic neuropathy. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 67:819–822.<br />
Delabrousse E, Kastler B, Bernard Y, et al. (2000). MR diagnosis of a congenital abnormality of the thoracic aorta<br />
with an aneurysm of the right subclavian artery present<strong>in</strong>g as a Horner’s syndrome <strong>in</strong> an adult. Eur Radiol<br />
10:650–652.<br />
DeMar<strong>in</strong>is M. (1994). Pupillary abnormalities due to sympathetic dysfunction <strong>in</strong> different forms of idiopathic<br />
headache. Cl<strong>in</strong> Autonomic Res 4:331–338.<br />
DeMar<strong>in</strong>is M, Assenza S, Carletto F. (1998). Oculosympathetic alterations <strong>in</strong> migra<strong>in</strong>e patients. Cephalgia 18:77–84.<br />
Digre KB, Smoker WRK, Johnston P, et al. (1992). Selective MR imag<strong>in</strong>g approach for evaluation of patients with<br />
Horner’s syndrome. AJNR 13:223–227.<br />
Dihne M, Block F, Thron A, Kuker W. (2000). Raeder’s syndrome: a rare presentation of <strong>in</strong>ternal carotid artery<br />
dissection. Cerebrovasc Dis 10:159–160.<br />
Dissenhouse MC, Palay DA, Newman NJ, To K, Albert DM. (1992). Acquired heterochromia with Horner’s<br />
syndrome <strong>in</strong> two adults. Ophthalmology 99:1815–1817.<br />
Donahue SP, Lav<strong>in</strong> PJM, Digre K. (1996). False-negative hydroxyamphetam<strong>in</strong>e (Paredr<strong>in</strong>e) test <strong>in</strong> acute Horner’s<br />
syndrome. Am J Ophthalmol 122:900–901.<br />
Drummond PD. (1991). Cervical sympathetic deficit <strong>in</strong> unilateral migra<strong>in</strong>e headache. Headache 31:669–672.<br />
Ebraheim NA, Lu J, Yang H, et al. (2000). Vulnerability of the sympathetic trunk dur<strong>in</strong>g anterior approach to the<br />
lower cervical sp<strong>in</strong>e. Sp<strong>in</strong>e 25:1603–1606.<br />
Ellenberg DJ, Spector LD, Lee A. (1992). Flea collar pupil (letter). <strong>An</strong>n Emerg Med 21:1170.<br />
Emir S, Kutluk MT, Gogus S, Buyukpamukcu M. (2000). Paraneoplastic cerebellar degeneration and Horner<br />
syndrome: association of two uncommon f<strong>in</strong>d<strong>in</strong>gs <strong>in</strong> a child with Hodgk<strong>in</strong> disease. J Pediatr Hematol Oncol<br />
22:158–161.
<strong>An</strong>isocoria and Pupillary Abnormalities 459<br />
Everett CM, Gutowski NJ. (1999). Prostate carc<strong>in</strong>oma present<strong>in</strong>g as bra<strong>in</strong> stem dysfunction. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 66:546.<br />
Fletcher WA, Sharpe JA. (1986). Tonic pupils <strong>in</strong> neurosyphilis. <strong>Neuro</strong>logy 36:188–192.<br />
Foster RE, Kosmorsky GS, Sweeney PJ, Masaryk TJ. (1991). Horner’s syndrome secondary to spontaneous carotid<br />
artery dissection with normal angiographic f<strong>in</strong>d<strong>in</strong>gs (letter). Arch Ophthalmol 109:1499–1500.<br />
Freeman JL, van den Brekel MW, Brown D. (1997). Carc<strong>in</strong>oma of the thyroid present<strong>in</strong>g as Horner’s syndrome.<br />
J Otolaryngol 26:387–388.<br />
Friedman JR, Whit<strong>in</strong>g DW, Kosmorsky GS, Burde RM. (1984). The coca<strong>in</strong>e test <strong>in</strong> normal patients. Am J Ophthalmol<br />
98:808–810.<br />
Fujisawa H, Marukawa K, Kida S, et al. (2001). Abducens nerve palsy and ipsilateral Horner syndrome: a<br />
predict<strong>in</strong>g sign of <strong>in</strong>tracranial carotid <strong>in</strong>jury <strong>in</strong> a head trauma patient. J Traum Injury Infect Crit Care<br />
50:554–556.<br />
Fujiwara S, Fujii K, Nishio S, Matsushima T, Fukui M. (1989). Oculomotor nerve palsy <strong>in</strong> patients with cerebral<br />
aneurysms. <strong>Neuro</strong>surg Rev 12:123–132.<br />
Gale A, Crockard HA. (1982). Transient unilateral mydriasis with basilar aneurysm. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
45:565–566.<br />
Ganesan S, Harar RP, Owen RA, et al. (1997). Horner’s syndrome: a rare presentation of cervical sympathetic<br />
cha<strong>in</strong> schwannoma. J Laryngol Otol 111:493–495.<br />
Gasch AT. (1996). Horner’s syndrome secondary to chest tube placement. <strong>An</strong>n Ophthalmol 28:235–239.<br />
Gelmi C, Ceccuzzi R. (1994). Mydriatic effects of ocular decongestants studied by pupillography. Ophthalmologica<br />
208:243–246.<br />
Gibbs J, Appleton RE, Mart<strong>in</strong> J, F<strong>in</strong>dlay G. (1992). Congenital Horner syndrome associated with non-cervical<br />
neuroblastoma. Dev Med Child <strong>Neuro</strong>l 34:642–644.<br />
Giles CL, Henderson JW. (1958). Horner’s syndrome: an analysis of 216 cases. Am J Ophthalmol 46:289–296.<br />
Glemarec J, Berthelot JM, Chevalet P, et al. (1998). Brachial plexopathy and Horner’s syndrome as the first<br />
manifestation of <strong>in</strong>ternal jugular ve<strong>in</strong> thrombosis <strong>in</strong>augurat<strong>in</strong>g polycythemia vera. Rev Rhum (English<br />
Version) 65:358–359.<br />
Goldste<strong>in</strong> JB, Biousse V, Newman NJ. (1997). Unilateral pharmacologic mydriasis <strong>in</strong> a patient with respiratory<br />
compromise. Arch Ophthalmol 115:806.<br />
Golnik KC, Hund PW III, Apple DJ. (1995). Atonic pupil after cataract surgery. J Cataract Refract Surg 21:<br />
170–175.<br />
Gomez ME, Gonzales I, Lo<strong>in</strong>az C, et al. (1993). <strong>Neuro</strong>logic complications <strong>in</strong> liver transplantation. Acta <strong>Neuro</strong>l<br />
Scand 87:25–31.<br />
Grau AJ, Brandt T, Forst<strong>in</strong>g M, et al. (1997). Infection-associated cervical artery dissection. Three cases.<br />
Stroke 28:453–455.<br />
Grimson BS, Thompson HS. (1975). Drug test<strong>in</strong>g <strong>in</strong> Horner’s syndrome. In: Glaser JS, et al, eds. <strong>Neuro</strong><strong>ophthalmology</strong><br />
Symposium of the University of Miami and the Bascom Palmer Eye Institute. Vol. 8. St. Louis,<br />
Mosby Year Book, p. 265.<br />
Grimson BS, Thompson HS. (1979). Horner’s syndrome: overall view of 120 cases. In. Thompson HS, ed. Topics <strong>in</strong><br />
<strong>Neuro</strong>-<strong>ophthalmology</strong>. Baltimore, Williams and Wilk<strong>in</strong>s, pp. 151–156.<br />
Guccione P, Gagliardi G, Bevilacqua M, Parisi F, Mar<strong>in</strong>o B. (1992). Cardiac catheterization through the <strong>in</strong>ternal<br />
jugular ve<strong>in</strong> <strong>in</strong> pediatric patients. Chest 101:1512–1514.<br />
Haider S. (1993). Tonic pupil <strong>in</strong> lymphomatoid granulomatosis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:38–39.<br />
Halpern BL, Pavilack MA, Gallagher SP. (1995). The <strong>in</strong>cidence of atonic pupil follow<strong>in</strong>g cataract surgery. Arch<br />
Ophthalmol 113:448–450.<br />
Hamza A, Fagan JJ, Weissman JL, Myers EN. (1997). Neurilemomas of the parapharyngeal space. Arch<br />
Otlolaryngol Head Neck Surg 123:622–626.<br />
Harada H, Tamaoka A, Yoshida H, et al. (1998). Horner’s syndrome associated with mononeuritis multiplex due<br />
to cytomegalovirus as the <strong>in</strong>itial manifestation <strong>in</strong> a patient with AIDS. J <strong>Neuro</strong>l Sci 154:91–93.<br />
Hassan AN, Ballester J, Slater N. (2000). Bilateral first rib fractures associated with Horner’s syndrome. Injury<br />
31:273–274.<br />
Hered RW, Cumm<strong>in</strong>gs RJ, Helffrich R. (1998). Persistent Horner’s syndrome after sp<strong>in</strong>al fusion and epidural<br />
analgesia. A case report. Sp<strong>in</strong>e 23:387–390.<br />
Hjelle B, Appenzeller O, Mills R, et al. (1992). Chronic neurodegenerative disease associated with HTLV-II<br />
<strong>in</strong>fection. Lancet 339:645–646.<br />
Hodgk<strong>in</strong>s PR, Luff AJ, Absolon MJ. (1993). Internal ophthalmoplegia—a compilation of ocular varicella. Aust NZ J<br />
Ophthalmol 21:53–54.
460 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Hood RJ, Reibel JF, Jensen ME, Lev<strong>in</strong>e PA. (2000). Schwannoma of the cervical sympathetic cha<strong>in</strong>. <strong>An</strong>n Otol Rh<strong>in</strong>ol<br />
Laryngol 109:48–51.<br />
Igarishi Y, Takeda M, Maekawa H, et al. (1992). Fisher’s syndrome without total ophthalmoplegia. Ophthalmology<br />
205:163–167.<br />
Inzelberg R, Nisipeanu P, Blumen SC, et al. (2000). Transient unilateral mydriasis as the present<strong>in</strong>g sign of aortic<br />
and carotid dissection. <strong>Neuro</strong>logy 55:1934–1935.<br />
Ishikawa H, Wakakura M, Ishikawa S. (1990). Enhanced ptosis <strong>in</strong> Fisher’s syndrome after Epste<strong>in</strong>-Barr virus<br />
<strong>in</strong>fection. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 10:197–200.<br />
Jacobson DM. (1990). Pupillary responses to dilute pilocarp<strong>in</strong>e <strong>in</strong> preganglionic third nerve disorders. <strong>Neuro</strong>logy<br />
40:804–808.<br />
Jacobson DM. (1994). A prospective evaluation of chol<strong>in</strong>ergic supersensitivity of the iris sph<strong>in</strong>cter <strong>in</strong> patients with<br />
oculomotor nerve palsies. Am J Ophthalmol 118:377–383.<br />
Jacobson DM. (1995). Benign episodic unilateral mydriasis. Ophthalmology 102:1623–1627.<br />
Jeret JS, Mazurek AA. (1995). Acute postpartum Horner’s syndrome due to epidural anesthesia. Arch Ophthalmol<br />
113:560.<br />
Johkura K, Hasegawa O, Kuroiwa Y. (2001). Episodic encephalopathy with dilated pupils. <strong>Neuro</strong>logy 56:1115–1116.<br />
Kardon RH, Corbett JJ, Thompson HS. (1998). Segmental denervation and re<strong>in</strong>nervation of the iris sph<strong>in</strong>cter as<br />
shown by <strong>in</strong>frared videographic transillum<strong>in</strong>ation. Ophthalmology 105:313–321.<br />
Kardon RH, Denison CE, Brown CK, Thompson HS. (1990). Critical evaluation of the coca<strong>in</strong>e test <strong>in</strong> the diagnosis<br />
of Horner’s syndrome. Arch Ophthalmol 108:384–387.<br />
Kaye-Wilson LG, Gibson R, Bell JE, Steers AJW. (1994). Oculomotor nerve neur<strong>in</strong>oma, early detection by magnetic<br />
resonance imag<strong>in</strong>g. <strong>Neuro</strong>-<strong>ophthalmology</strong> 14:37–41.<br />
Kerrison JB, Biousse V, Newman NJ. (2000). Isolated Horner’s syndrome and syr<strong>in</strong>gomyelia. J <strong>Neuro</strong>l <strong>Neuro</strong>surg<br />
Psychiatry 69:131–132.<br />
Kerty E. (1999). The <strong>ophthalmology</strong> of <strong>in</strong>ternal carotid artery dissection. Acta Ophthalmol Scand 77:418–421.<br />
Kim JS, Lee JH, Suh DC, Lee MC. (1994). Spectrum of lateral medullary syndrome. Correlation between cl<strong>in</strong>ical<br />
f<strong>in</strong>d<strong>in</strong>gs and magnetic resonance imag<strong>in</strong>g <strong>in</strong> 33 subjects. Stroke 25:1405–1410.<br />
Kim JS, Yu CH, Moon CS. (2001a). Bilateral tonic (Adie’s) pupils <strong>in</strong> Vogt-Koyanagi-Harada syndrome. J <strong>Neuro</strong>ophthalmol<br />
21:205–206.<br />
Kim W, Clancy RR, Liu GT. (2001b). Horner syndrome associated with implantation of a vagus nerve stimulator.<br />
Am J Ophthalmol 131:383–384.<br />
Kimber J, Mitchell D, Mathias CJ. (1998). Chronic cough <strong>in</strong> the Holmes-Adie’s syndrome: association <strong>in</strong> five cases<br />
with autonomic dysfunction. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 65:583–586.<br />
Kimura Bruno M, W<strong>in</strong>terkorn JMS, Edgar MA, et al. (2000). Unilateral Adie pupil as sole ophthalmic sign of<br />
anti-Hu paraneoplastic syndrome. J <strong>Neuro</strong>-Ophthalmol 20:248–249.<br />
Koennecke H-C, Seyfert S. (1998). Mydriatic pupil as the present<strong>in</strong>g sign of common carotid artery dissection.<br />
Stroke 29:2653–2655.<br />
Koivunen P, Lopponen H. (1999). Internal carotid artery thrombosis and Horner’s syndrome as complications of<br />
parapharyngeal abscess. Otolaryngol Head Neck Surg 121:160–162.<br />
Lam BL, Thompson HS, Walls RC. (1996). Effect of light on the prevalence of simple anisocoria. Ophthalmology<br />
103:790–793.<br />
Lambert SR, Yang LLH, Stone C. (2000). Tonic pupil associated with congenital neuroblastoma, Hirschsprung<br />
disease, and central hypoventilation syndrome. Am J Ophthalmol 130:238–240.<br />
Leavitt JA, Wayman LL, Hodge DO, et al. (2002). Pupillary response to four concentrations of pilocarp<strong>in</strong>e <strong>in</strong><br />
normal subjects: application of test<strong>in</strong>g for Adie tonic pupil. Am J Ophthalmol 133:333–336.<br />
Leira EC, Bendixen BH, Kardon RH, Adams HP Jr. (1998). Brief, transient Horner’s syndrome can be the hallmark<br />
of carotid artery dissection. <strong>Neuro</strong>logy 50:289–290.<br />
Lepore FE. (1993). Amaurotic mydriasis. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:200.<br />
Liu M, Kim PS, Chen CK, Smythe WR. (1998). Delayed Horner’s syndrome as a complication of cont<strong>in</strong>uous<br />
thoracic epidural analgesia. J Cardiothorac Vasc <strong>An</strong>esth 12:195–196.<br />
Mabuchi K, Yoshikawa H, Takamori M, et al. (1998). Pseudo-Argyll Robertson pupil of patients with sp<strong>in</strong>ocerebellar<br />
ataxia type 1 (SCA1). J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 65:612–613.<br />
Maloney WF, Younge BR, Moyer NJ. (1980). Evaluation of the causes and accuracy of pharmacologic localization<br />
<strong>in</strong> Horner’s syndrome. Am J Ophthalmol 90:394–402.<br />
Manzoni GC, Micieli G, Zanferrari S, et al. (1991). Cluster headache. Recent developments <strong>in</strong> cl<strong>in</strong>ical characterization<br />
and pathogenesis. Acta <strong>Neuro</strong>l 13:506–513.<br />
Masjuan J, García-Segovia J, Barón M, Alvarez-Cermeno JC. (1997). Ipsilateral mydriasis <strong>in</strong> focal occipitotemporal<br />
seizures. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 63:810–811.
<strong>An</strong>isocoria and Pupillary Abnormalities 461<br />
Maur<strong>in</strong>o V, Allan BDS, Stevens JD, Tuft SJ. (2002). Fixed dilated pupil (Urrets-Zavalia syndrome) after air=gas<br />
<strong>in</strong>jection after deep lamellar keratoplasty for keratoconus. Am J Ophthalmol 133:266–268.<br />
Menendez C, MacMillan DT, Britt LD. (2000). Transient Horner’s syndrome <strong>in</strong> a trauma patient with thoracic<br />
epidural analgesia: a case report. Am Surg 66:756–758.<br />
Midroni G, Dyck PJ. (1996). Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy: unusual cl<strong>in</strong>ical features and<br />
therapeutic responses. <strong>Neuro</strong>logy 46:1206–1212.<br />
Miller NR. (1985). Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-<strong>ophthalmology</strong>. 4th ed. Baltimore, Williams and Wilk<strong>in</strong>s,<br />
pp. 425–428, 500–511, 703–705, 1012–1015.<br />
Mokhtari F, Mass<strong>in</strong> P, Paques M, et al. (2000). Central ret<strong>in</strong>al artery occlusion associated with head or neck pa<strong>in</strong><br />
reveal<strong>in</strong>g spontaneous <strong>in</strong>ternal carotid artery dissection. Am J Ophthalmol 129:108–109.<br />
Mokri B, Schiev<strong>in</strong>k WI, Olsen KD, Piepgras DG. (1992). Spontaneous dissection of the cervical <strong>in</strong>ternal carotid<br />
artery. Presentation with lower cranial nerve palsies. Arch Otolaryngol Head Neck Surg 118:431–435.<br />
Monson MC, Mamalis N, Olson RJ. (1992). Toxic anterior segment <strong>in</strong>flammation follow<strong>in</strong>g cataract surgery.<br />
J Cataract Refract Surg 18:184–189.<br />
Monteiro ML, Coppeto JR, Milani JA. (1993). Iron mydriasis. Pupillary paresis from occult <strong>in</strong>traocular foreign<br />
body. J Cl<strong>in</strong> <strong>Neuro</strong>-ophthalmol 13:254–257.<br />
Morales J, Brown SM, Abdul-Rahim AS, Crosson CE. (2000). Ocular effects of apraclonid<strong>in</strong>e <strong>in</strong> Horner syndrome.<br />
Arch Ophthalmol 118:951–954.<br />
Mori M, Kuwabara S, Fukutake T, et al. (2001). <strong>Cl<strong>in</strong>ical</strong> features and prognosis of Miller Fisher syndrome.<br />
<strong>Neuro</strong>logy 56:1104–1106.<br />
Murasella MA, Chan HS, DeBoer G, Gallie BL. (1984). Ocular <strong>in</strong>volvement <strong>in</strong> neuroblastoma: prognostic<br />
implications. Ophthalmology 91:936–940.<br />
Müri RM, Baumgartner RW. (1995). Horner’s syndrome and contralateral trochlear nerve palsy. <strong>Neuro</strong><strong>ophthalmology</strong><br />
15:161–163.<br />
Mutschler V, Sellal F, Maillot C, Maton B, Collard M. (1994). Horner’s syndrome and thalamic lesions. <strong>Neuro</strong><strong>ophthalmology</strong><br />
14:231–236.<br />
Naimer SA, We<strong>in</strong>ste<strong>in</strong> O, Rosenthal G. (2000). Congenital Horner syndrome: a rare though significant complication<br />
of subclavian flap aortoplasty. J Thorac Cardiovasc Surg 120:419–421.<br />
Nakagawa TA, Geurra L, Storgion SA. (1993). Aerosolized atrop<strong>in</strong>e as an unusual cause of anisocoria <strong>in</strong> a child<br />
with asthma. Pediatr Emerg Care 9:153–154.<br />
Nish<strong>in</strong>o H, Rub<strong>in</strong>o FA. (1993). Horner’s syndrome <strong>in</strong> Wegener’s granulomatosis. Report of four cases. J <strong>Neuro</strong>l<br />
<strong>Neuro</strong>surg Psychiatry 56:897.<br />
Nussdorf JD, Berman EL. (2000). <strong>An</strong>isocoria associated with the medical treatment of irritable bowel syndrome.<br />
J <strong>Neuro</strong>-ophthalmol 20:100–101.<br />
Oono S, Saito I, Inukai G, Morisawa K. (1999). Traumatic Horner syndrome without anhidrosis. J <strong>Neuro</strong>-ophthalmol<br />
19:148–151.<br />
Pascual-Sedano B, Roig C. (1998). Horner’s syndrome due to giant cell arteritis. <strong>Neuro</strong>-<strong>ophthalmology</strong> 20:<br />
75–77.<br />
Paw HG. (1998). Horner’s syndrome follow<strong>in</strong>g low-dose epidural <strong>in</strong>fusion for labour; a cautionary tale. Eur<br />
J <strong>An</strong>esth 15:110–111.<br />
Payne JW, Adamkiewicz J. (1969). Unilateral <strong>in</strong>ternal ophthalmoplegia with <strong>in</strong>tracranial aneurysm. Am<br />
J Ophthalmol 68:349–352.<br />
Peake ST, Bollen B. (1990). Unilateral fixed dilated pupil after aortic valve replacement: an unusual comb<strong>in</strong>ation of<br />
causes. J Cardiothorac <strong>An</strong>esth 4:737–739.<br />
Pelak VS, Galetta SL, Grossman RI, et al. (1999). Evidence for preganglionic pupillary <strong>in</strong>volvement <strong>in</strong> superficial<br />
siderosis. <strong>Neuro</strong>logy 53:1130–1132.<br />
Pelton JJ, Ratner IA. (1990). <strong>Neuro</strong>blastoma of the thoracic <strong>in</strong>let. J Pediatr Surg 25:547–549.<br />
Penarrocha-Diago M, Sanchis-Bielsa JM. (2000). Ophthalmologic complications after <strong>in</strong>traoral local anesthesia<br />
with artica<strong>in</strong>e. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 90:21–24.<br />
Perk<strong>in</strong> GD. (1994). The pupil. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 57:1179.<br />
Perlman JP, Conn H. (1991). Transient <strong>in</strong>ternal ophthalmoplegia dur<strong>in</strong>g blepharoplasty. A report of three cases.<br />
Adv Ophthalmic Plast Reconstr Surg 7:141–143.<br />
Perry C, Wixon JD, Mills J, Eriksen C. (2001). Horner’s syndrome after carotid endarterectomy—a case report.<br />
Vasc Surg 35:325–327.<br />
Phillips PH, Newman NJ. (1996). Tonic pupil <strong>in</strong> child. J Pediatr Ophthalmol Strabismus 33:331–332.<br />
Pimentel J, Mart<strong>in</strong>s IP. (1993). Raeder’s syndrome. A case with an unusual localization. Cephalgia 13:135.<br />
Poole TR, Acheson JF, Smith SE, Steiger MJ. (1997). Horner’s syndrome due to herpes zoster <strong>in</strong> the T3-T4<br />
dermatome. J R Soc Med 90:395–396.<br />
Prielipp RC. (1994). Unilateral mydriasis after <strong>in</strong>duction of anaesthesia. Can J <strong>An</strong>aesth 41:140–143.
462 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Pritz MB. (1999). Ophthalmic artery aneurysm associated with Horner’s syndrome. Acta <strong>Neuro</strong>chir 141:<br />
891–892.<br />
Purv<strong>in</strong> VA. (1995). Adie’s tonic pupil secondary to migra<strong>in</strong>e. J <strong>Neuro</strong>-ophthalmol 15:43–44.<br />
Purv<strong>in</strong> V, Wall M, Slav<strong>in</strong> M. (1997). Unilateral headache and ptosis <strong>in</strong> a 30-year-old woman. Surv Ophthalmol<br />
42:163–168<br />
Radziwill AJ, Steck AJ, Borruat F-X, Bogousslavsky J. (1998). Isolated <strong>in</strong>ternal ophthalmoplegia associated with<br />
IgG anti-GQ1b antibody. <strong>Neuro</strong>logy 50:307.<br />
Reddy G, Coombes A, Hubbard AD. (1998). Horner’s syndrome follow<strong>in</strong>g <strong>in</strong>ternal jugular ve<strong>in</strong> cannulation.<br />
Intensive Care Med 24:194–196.<br />
Resnick DK. (1993). Delayed pulmonary perforation. A rare complication of tube thoracostomy. Chest 103:311–313.<br />
Roarty JD, Keltner JL. (1990). Normal pupil size and anisocoria <strong>in</strong> newborn <strong>in</strong>fants. Arch Ophthalmol 108:94–95.<br />
Roberts BN, Mills PV, Hawksworth NJ. (1995). Bilateral ptosis, tonic pupils and abducens palsies follow<strong>in</strong>g<br />
Campylobacter jejuni enteritis. Eye 9:657–658.<br />
Ryan FH, Kl<strong>in</strong>e LB, Gomez C. (2000). Congenital Horner’s syndrome result<strong>in</strong>g from agenesis of the <strong>in</strong>ternal<br />
carotid artery. Ophthalmology 107:185–188.<br />
Saiz A, <strong>An</strong>gulo S, Fernandez M. (1991). Atonic pupil: an unusual complication of cataract surgery. Ophthalmic<br />
Surg 22:20–22.<br />
Salveson R. (2000). Cluster headache s<strong>in</strong>e headache: case report. <strong>Neuro</strong>logy 55:451.<br />
Sauer C, Lev<strong>in</strong>sohn MW. (1976). Horner’s syndrome <strong>in</strong> childhood. <strong>Neuro</strong>logy 26:216–220.<br />
Sawada T, Kimura T, Kimura W, et al. (1990). Two cases of Fisher’s syndrome with tectal pupil. Folia Ophthalmol<br />
Jpn 41:1833–1838<br />
Scaglione M, P<strong>in</strong>to F, Grassi R, et al. (1999). Migration of a foreign body from the pharynx to the soft tissues of the<br />
neck: delayed presentation with Horner’s syndrome. AJR 172:1131–1132.<br />
Schiev<strong>in</strong>k WI, Atk<strong>in</strong>son JL, Bartleson JD, Whisnant JP. (1998). Traumatic <strong>in</strong>ternal carotid artery dissections caused<br />
by blunt softball <strong>in</strong>juries. Am J Emerg Med 16:179–182.<br />
Scotcher SM, Cann<strong>in</strong>g CR, Dorrell D. (1995). Siderosis bulbi: an unusual cause of a unilaterally dilated pupil. Br<br />
J Hosp Med 54:110–111.<br />
Sh<strong>in</strong> RK, Galetta SL, T<strong>in</strong>g TY, et al. (2000). Ross syndrome plus. Beyond Horner, Holmes-Adie, and harlequ<strong>in</strong>.<br />
<strong>Neuro</strong>logy 55:1841–1846.<br />
Simon T, Voth E, Berthold F. (2001). Asymmetric salivary gland 123I-meta-iodobenzylguanid<strong>in</strong>e uptake <strong>in</strong> a<br />
patient with cervical neuroblastoma and Horner syndrome. Med Pediatr Oncol 36:489–490.<br />
Slav<strong>in</strong> ML. (2000). Horner syndrome with equal sized pupils <strong>in</strong> a case with underly<strong>in</strong>g physiologic anisocoria.<br />
J <strong>Neuro</strong>-ophthalmol 20:1–2.<br />
Smith EF, Santamar<strong>in</strong>a L, Wol<strong>in</strong>tz AH. (1993). Herpes zoster ophthalmicus as a cause of Horner syndrome. J Cl<strong>in</strong><br />
<strong>Neuro</strong>-ophthalmol 13:250–253.<br />
Smith SA, Smith SE. (1999). Bilateral Horner’s syndrome: detection and occurrence. J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry<br />
66:48–51.<br />
Soriani S, Scarpa P, Arnaldi C, et al. (1996). Migra<strong>in</strong>e aura without headache and ictal fast EEG activity <strong>in</strong> an<br />
11-year-old boy. Eur J Pediatr 155:126–129.<br />
Soylev MF, Saatci O, Kavukcu S, Erg<strong>in</strong> M. (1997). Adie’s syndrome <strong>in</strong> childhood. Acta Paediatr Jpn 39:395–396.<br />
Stewart D, Simpson GT, Nader ND. (1999). Postoperative anisocoria <strong>in</strong> a patient undergo<strong>in</strong>g endoscopic s<strong>in</strong>us<br />
surgery. Reg <strong>An</strong>esth Pa<strong>in</strong> Med 24:467–469.<br />
Stromberg BC, Knibbe M. (1988). <strong>An</strong>isocoria follow<strong>in</strong>g reduction of bilateral orbital floor fractures. <strong>An</strong>n Plast Surg<br />
21:486–488.<br />
Sunderland S. (1952). Mechanism responsible for changes <strong>in</strong> the pupil unaccompanied by disturbances of<br />
extraocular muscle function. Br J Ophthalmol 36:638–644.<br />
Szawlowski AW, Falkowski S, Morys<strong>in</strong>ski T, et al. (1991). Preoperative concurrent chemotherapy and radiotherapy<br />
for local-regional and advanced squamous cell carc<strong>in</strong>oma of the thoracic oesophagus: prelim<strong>in</strong>ary<br />
results of a pilot study. Eur J Surg Oncol 17:575–580.<br />
Tan AK, Humphry RC. (1993). The fixed pupil after cataract surgery: is it related to <strong>in</strong>traocular use of<br />
hypromellose? Br J Ophthalmol 77:639–641.<br />
Tan E, Kansu T, Saygi S, Zileli T. (1990). Alternat<strong>in</strong>g Horner’s syndrome. A case report and review of the<br />
literature. <strong>Neuro</strong>-<strong>ophthalmology</strong> 10:19–22.<br />
Thompson CG. (1998). Horner’s syndrome result<strong>in</strong>g from a prolonged abnormal posture dur<strong>in</strong>g a coma. Aust N Z<br />
J Ophthalmol 26:165–167.<br />
Thompson HS. (1977a). Adie’s syndrome: some new observations. Trans Am Ophthalmol Soc 75:587–626.<br />
Thompson HS. (1977b). Diagnos<strong>in</strong>g Horner’s syndrome. Trans Am Acad Ophthalmol Otolaryngol 83:840–842.
<strong>An</strong>isocoria and Pupillary Abnormalities 463<br />
Van der Wiel HL, Van Gijn J. (1983). Localization of Horner’s syndrome: use and limitations of the hydroxyamphetam<strong>in</strong>e<br />
test. J <strong>Neuro</strong>l Sci 59:229–235.<br />
Van der Wiel HL, Van Gijn J. (1986). The diagnosis of Horner’s syndrome: use and limitations of the coca<strong>in</strong>e test.<br />
J <strong>Neuro</strong>l Sci 73:311–316.<br />
Vaswani S, Garv<strong>in</strong> L, Matuschak GM. (1991). Postganglionic Horner’s syndrome after <strong>in</strong>sertion of a pulmonary<br />
artery catheter through the <strong>in</strong>ternal jugular ve<strong>in</strong>. Crit Care Med 19:1215–1216.<br />
Venketasubramanian N, S<strong>in</strong>gh J, Hui F, Lim MK. (1998). Carotid artery dissection present<strong>in</strong>g as a pa<strong>in</strong>less<br />
Horner’s syndrome <strong>in</strong> a pilot: fit tofly? Aviat Space Environ Med 69:307–310.<br />
Vetrugno R, Liguori R, Cevoli S, et al. (1997). Adie’s tonic pupil as a manifestation of Sjögren’s syndrome. Ital<br />
J <strong>Neuro</strong>l Sci 18:293–295.<br />
Vighetto A, Lisovoski F, Revol A, Trillet M, Almard G. (1990). Internal carotid artery dissection and ipsilateral<br />
hyoglossal nerve palsy (letter). J <strong>Neuro</strong>l <strong>Neuro</strong>surg Psychiatry 53:530–531.<br />
Walsh and Hoyt’s <strong>Cl<strong>in</strong>ical</strong> <strong>Neuro</strong>-Ophthalmology. (1969). Baltimore, Williams and Wilk<strong>in</strong>s.<br />
We<strong>in</strong>ste<strong>in</strong> JM, Zweifel TJ, Thompson HS. (1980). Congenital Horner’s syndrome. Arch Ophthalmol 98:1074–1078.<br />
Weissberg D. (2001). Congenital Horner syndrome. J Thorac Cardiovasc Surg 121:819–820.<br />
Weller M, Wilhelm H, Sommer N, Dichgans J, Wietholter H. (1992). Tonic pupil, areflexia, and segmental<br />
anhidrosis: two additional cases of Ross syndrome and review of the literature. J <strong>Neuro</strong>l 239:231–234.<br />
Wijdicks EFM. (1995). <strong>Neuro</strong>logy of Critical Illness. Philadelphia, FA Davis.<br />
Wilhelm H. (1994). Pupil exam<strong>in</strong>ation and evaluation of pupillary disorders. <strong>Neuro</strong>-<strong>ophthalmology</strong> 14:283–295.<br />
Wilhelm H, Klier R, Toth B, Wilhelm B. (1995). Oculomotor nerve paresis start<strong>in</strong>g as isolated <strong>in</strong>ternal<br />
ophthalmoplegia. <strong>Neuro</strong>-<strong>ophthalmology</strong> 15:211–215.<br />
Wilhelm H, Ochsner H, Kopycziok E, et al. (1992). Horner’s syndrome: a retrospective analysis of 90 cases and<br />
recommendations for cl<strong>in</strong>ical handl<strong>in</strong>g. German J Ophthalmol 1:96–102.<br />
Wirtz PW, de Keizer RJW, de Visser M, et al. (2001). Tonic pupils <strong>in</strong> Lambert-Eaton myasthenic syndrome. Muscle<br />
Nerve 24:444–445.<br />
Wolfe GI, Galetta SL, Teener JW, Katz JS, Bird SJ. (1995). Site of autonomic dysfunction <strong>in</strong> a patient with Ross’<br />
syndrome and postganglionic Horner’s syndrome. <strong>Neuro</strong>logy 45:2094–2096.<br />
Woodruff G, Buncic JR, Mor<strong>in</strong> JD. (1988). Horner’s syndrome <strong>in</strong> children. J Pediatr Ophthalmol Strabismus 25:40–44.<br />
Worth<strong>in</strong>gton JP, Snape L. (1998). Horner’s syndrome secondary to a basilar skull fracture after maxillofacial<br />
trauma. J Oral Maxillofac Surg 56:996–1000.<br />
Zamir E, Chowers I, Ban<strong>in</strong> E, Frucht-Pery J. (1999). <strong>Neuro</strong>trophic corneal endothelial failure complicat<strong>in</strong>g acute<br />
Horner syndrome. Ophthalmology 106:1692–1696.<br />
Zander DR, Just N, Schipper HM. (1998). <strong>An</strong>eurysm of the <strong>in</strong>trapetrous <strong>in</strong>ternal carotid artery present<strong>in</strong>g as<br />
isolated Horner’s syndrome: case report. Can Assoc Radiol J 49:46–48.<br />
Zelligowsky A, Szold A, Seror D, Vromen A, Pffeffermann R. (1991). Horner syndrome: a rare complication of<br />
<strong>in</strong>ternal jugular ve<strong>in</strong> cannulation. J Parenter Enter Nutr 15:199.
Index r<br />
Page numbers <strong>in</strong> italic <strong>in</strong>dicate that the entry on that page is <strong>in</strong> a figure or table.<br />
Abducens nerve paresis. See also Sixth<br />
nerve palsies (SNP)<br />
divergence <strong>in</strong>sufficiency/paralysis,<br />
229–230<br />
localization, 297<br />
one-and-a-half syndrome, 320–321<br />
Abducens nucleus, anatomy,<br />
312, 314<br />
Aberrant regeneration, third nerve palsy<br />
(TNP), 270–271<br />
Abetalipoprote<strong>in</strong>emia, horizontal gaze<br />
palsy, 314–315<br />
Acetazolamide, idiopathic pseudotumor<br />
cerebri therapy, 146, 147<br />
Adie’s tonic pupil syndrome<br />
cl<strong>in</strong>ical features, 441, 444<br />
etiology, 441<br />
neuroimag<strong>in</strong>g studies, 441<br />
therapeutic strategies, 444<br />
Adrenocorticotropic hormone (ACTH),<br />
optic neuritis (ON) therapy,<br />
47–49, 48<br />
Alcohol amblyopia, nutritional optic<br />
neuropathy, cl<strong>in</strong>ical features,<br />
9, 12–14, 17<br />
Alexia, homonymous hemianopsia, optic<br />
radiations, 202<br />
Alzheimer’s disease, homonymous<br />
hemianopsia, 206<br />
<strong>An</strong>eurysm<br />
acquired isolated third nerve palsies<br />
(TNP)<br />
neuroimag<strong>in</strong>g protocols, 268–270<br />
normal sph<strong>in</strong>cter<br />
464<br />
complete extraocular muscle palsy<br />
(type 4A TNP), 264–265<br />
<strong>in</strong>complete extraocular muscle<br />
palsy (type 4B TNP), 266<br />
acquired sixth nerve palsy (SNP), 304<br />
third nerve palsies (TNP),<br />
subarachnoid lesion, 260<br />
<strong>An</strong>gle-closure glaucoma, horizontal<br />
diplopia, 223–226<br />
<strong>An</strong>isocoria<br />
Adie’s syndrome, 441, 444, 444<br />
evaluation protocol, 456, 456<br />
Horner’s syndrome, 445, 445–456<br />
alternat<strong>in</strong>g Horner’s syndrome, 448<br />
central Horner’s syndrome, 446, 447<br />
congenital Horner’s syndrome, 449<br />
imag<strong>in</strong>g and evaluation protocols,<br />
455<br />
isolated Horner’s syndrome, 446<br />
isolated postganglionic Horner’s<br />
syndrome, 455–456<br />
pediatric patients, 454–455<br />
pharmacologic localization, 452–454,<br />
453<br />
postganglionic Horner’s syndrome,<br />
446–447, 449–451<br />
preganglionic (<strong>in</strong>termediate)<br />
Horner’s syndrome, 446, 448<br />
trauma etiology, 449, 451<br />
<strong>in</strong>termittent/transient pupillary<br />
phenomenon, 435, 439<br />
iris structural abnormalities, 439,<br />
439–440<br />
isolated etiology, 445
isolated anisocoria, cl<strong>in</strong>ical evaluation,<br />
445<br />
light-near dissociation, 434, 434<br />
syphilis serology evaluation, 444–445<br />
light reaction, 433<br />
normal pupil, 445<br />
pharmacologic mydriasis/miosis, 435,<br />
437–439<br />
third nerve palsies and, 434–435<br />
tonic pupil, 440–441, 440–443<br />
<strong>An</strong>terior choroidal artery occlusion,<br />
homonymous hemianopsia,<br />
lateral geniculate body<br />
lesions, 200<br />
<strong>An</strong>terior ischemic optic neuropathy<br />
(AION)<br />
associated conditions, 75, 76–78<br />
cl<strong>in</strong>ical presentation, 3<br />
atypical features, 75, 78–79, 79<br />
differential diagnosis, 2<br />
evaluation and treatment, 79–80<br />
future research issues, 80–81<br />
giant cell arteritis (GCA)<br />
cl<strong>in</strong>ical features, 93, 94<br />
atypical features, 98–99, 100–101<br />
corticosteroid therapy, 107–108<br />
diagnostic criteria, 94, 94–96, 96–98,<br />
98–99<br />
ESR elevation, 99, 105<br />
ESR normal values, 99<br />
evaluation flowchart, 109<br />
hematologic test<strong>in</strong>g, 99, 101, 102<br />
laboratory abnormalities, 99, 102, 105<br />
patient evaluation protocols, 104–105,<br />
105<br />
TAB procedures, 101–104<br />
therapeutic regimen, 106–110<br />
oral vs. IV corticosteroids, 106–108<br />
steroid complications, 108, 110<br />
nonarteritic ischemic optic neuropathy,<br />
cl<strong>in</strong>ical features, 73–75, 74–75<br />
<strong>An</strong>terior ischemic optic neuropathy of<br />
the young (AIONY), cl<strong>in</strong>ical<br />
features, 75<br />
<strong>An</strong>terior knee, visual field defects,<br />
junctional characteristics,<br />
192–194<br />
<strong>An</strong>tiacetylchol<strong>in</strong>e receptor antibodies<br />
myasthenia gravis (MG) diagnosis, 341<br />
Index 465<br />
ocular myasthenia gravis (OM)<br />
diagnosis, 341–342<br />
<strong>An</strong>tichol<strong>in</strong>esterase agents, ocular<br />
myasthenia gravis (OM)<br />
management, 343–344<br />
Aponeurotic ptosis<br />
etiology, 415, 415<br />
evaluation protocol, 416, 417<br />
Apraxia of eyelid open<strong>in</strong>g, ptosis<br />
etiology, 410, 411<br />
Argyll Robertson pupil, syphilis serology<br />
test<strong>in</strong>g, 444–445<br />
Arnold-Chiari malformation, seesaw<br />
nystagmus,<br />
374<br />
Arteriorvenous malformations (AVMs),<br />
b<strong>in</strong>ocular transient visual loss,<br />
180–184<br />
Aspir<strong>in</strong> therapy, nonarteritic anterior<br />
ischemic optic neuropathy<br />
(NA-AION), 81–82<br />
Asymmetric b<strong>in</strong>ocular eye oscillations,<br />
nystagmus etiology, 368,<br />
370–372, 373<br />
Ataxia, periodic alternat<strong>in</strong>g esotropia<br />
(PAE), 228<br />
Atherosclerotic disease, monocular TVL<br />
with, 171–172<br />
cl<strong>in</strong>ical features, 173–179<br />
Atypical optic neuritis, cl<strong>in</strong>ical<br />
presentation, 38, 39<br />
Atypical/unexpla<strong>in</strong>ed optic neuropathy,<br />
14, 22<br />
Basilar aneurysms, anisocoria etiology,<br />
435<br />
Bielschowsky’s test, fourth nerve palsies<br />
(FNP), 281<br />
B<strong>in</strong>asal hemianopsia, visual field defects,<br />
196, 198<br />
B<strong>in</strong>ocular diplopia<br />
evaluation protocol, 217<br />
horizontal (esotropia/exotropia),<br />
etiology, 219–220,<br />
221–222<br />
vertical diplopia<br />
cl<strong>in</strong>ical evaluation, 233–234<br />
etiology, 234, 235
466 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
B<strong>in</strong>ocular symmetric conjugate eye<br />
oscillations<br />
etiology, 377–382<br />
evaluation, 376<br />
B<strong>in</strong>ocular symmetric jerk nystagmus<br />
eccentric gaze/maneuver <strong>in</strong>duction,<br />
389–391, 392<br />
etiology, 382–385<br />
B<strong>in</strong>ocular transient visual loss<br />
cl<strong>in</strong>ical signs, 179–183<br />
evaluation protocols, 169–170, 183<br />
Biopsy, horizontal diplopia, 224–226<br />
Bitemporal hemianopsia, visual field<br />
defects, 194, 195–196, 196, 197<br />
Blepharospasm, ptosis, 409–410<br />
Blood pressure monitor<strong>in</strong>g, papilledema<br />
evaluation, 133, 135, 135<br />
Botul<strong>in</strong>um tox<strong>in</strong> therapy<br />
congenital nystagmus, 378<br />
Graves’ ophthalmopathy (GO)<br />
lid retraction, 356<br />
strabismus, 356–357<br />
pendular nystagmus, 381–382<br />
Botulism, vertical diplopia, 238–239<br />
Branstem structures, lid retraction/lid<br />
lag and, 421<br />
Brown’s superior oblique tendon sheath<br />
syndrome, vertical diplopia,<br />
241–242<br />
B-scan echography, papilledema<br />
differential diagnosis, 130<br />
Calcific emboli, monocular TVL, 173<br />
Carbonic anhydrase <strong>in</strong>hibitors,<br />
idiopathic pseudotumor<br />
cerebri therapy, 146,<br />
147<br />
Carotid artery dissection<br />
Horner’s syndrome, cl<strong>in</strong>ical evaluation<br />
and etiology, 447, 449<br />
monocular TVL with, 173–179<br />
Carotid endarterectomy, transient visual<br />
loss risk and, 177–178<br />
Cataract surgery, vertical diplopia,<br />
243–244<br />
Cat-scratch disease, neuroret<strong>in</strong>itis and<br />
optic disc edema with macular<br />
star (ODEMS), 65–66, 67<br />
Cavernous s<strong>in</strong>us lesion<br />
fourth nerve palsy (FNP), 283<br />
sixth nerve palsies (SNP), 297, 301<br />
third nerve palsies (TNP), 260–261<br />
Central caudal nucleus (CCN), lid<br />
retraction/lid lag, 421<br />
Central disorders<br />
convergence spasm, 230–231<br />
horizontal diplopia, 227–228<br />
Central Horner’s syndrome, cl<strong>in</strong>ical<br />
evaluation, 446, 447<br />
Central ret<strong>in</strong>al artery occlusion (CRAO),<br />
giant cell arteritis (GCA)<br />
cl<strong>in</strong>ical diagnosis, 95–96, 96–98, 98–99<br />
corticosteroid therapy, 107–108<br />
Central ret<strong>in</strong>al venous occlusion (CRVO),<br />
venous stasis ret<strong>in</strong>opathy,<br />
differential diagnosis, 171–179<br />
Central venous thrombosis (CVT),<br />
pseudotumor cerebri<br />
syndrome, 136–137<br />
Cerebral bl<strong>in</strong>dness, visual field defects,<br />
homonymous hemianopsia,<br />
204–205<br />
Cerebral <strong>in</strong>farction<br />
cerebral polyopia, 217<br />
homonymous hemianopsia, 206<br />
Cerebral ischemia, transient visual loss,<br />
176–179<br />
Cerebral polyopia, monocular diplopia,<br />
differential diagnosis, 214, 217<br />
Cerebral structural lesions, b<strong>in</strong>ocular<br />
transient visual loss, 180<br />
Cerebrosp<strong>in</strong>al fluid (CSF)<br />
optic neuritis (ON)<br />
lumbar punctures, 46<br />
neuroimag<strong>in</strong>g studies, 44–46<br />
papilledema, cl<strong>in</strong>ical evaluation, 135<br />
pseudotumor cerebri syndrome,<br />
136–137<br />
lumboperitoneal shunt (LSP)<br />
complications and, 148–155,<br />
149<br />
Chiasmal syndromes, visual field<br />
defects, 194, 195–196, 196, 197<br />
Childhood strabismus syndromes,<br />
esotropia/exotropia etiology,<br />
220, 222, 223<br />
Cholesterol emboli, monocular TVL, 172
Chronic progressive external<br />
ophthalmoplegia (CPEO),<br />
ptosis etiology, 412–413,<br />
414<br />
Claude’s syndrome, third nerve palsies<br />
(TNP), fascicular lesion,<br />
258–259<br />
Coca<strong>in</strong>e, pharmacological localization of<br />
Horner’s syndrome, 452–454,<br />
453<br />
Comatose patients, spontaneous eye<br />
oscillations, 396–399,<br />
397–398<br />
Compressive optic neuropathy (CON)<br />
cl<strong>in</strong>ical presentation, 3, 5, 5–12<br />
Graves’ ophthalmopathy (GO)<br />
imag<strong>in</strong>g f<strong>in</strong>d<strong>in</strong>gs, 349–350<br />
therapeutic strategies, 353, 357–360,<br />
358–359<br />
Computed tomography (CT)<br />
acquired isolated third nerve palsies<br />
(TNP), neuroimag<strong>in</strong>g<br />
protocols, 268–270<br />
gaze-evoked transient visual loss<br />
(TVL), 170–171<br />
Graves’ ophthalmopathy (GO), 349<br />
horizontal gaze palsies, 312<br />
<strong>in</strong>ternuclear ophthalmoplegia (INO),<br />
319<br />
isolated fourth nerve palsies (TNP),<br />
289–290, 290–292<br />
myasthenia gravis (MG) diagnosis, 342<br />
optic neuritis, 44–46<br />
papilledema<br />
cl<strong>in</strong>ical evaluation, 133, 135, 135<br />
differential diagnosis, 130<br />
third nerve palsies (TNP), retrospective<br />
studies, 271–272<br />
Congenital Horner’s syndrome,<br />
evaluation and etiology, 449<br />
Congenital nystagmus, cl<strong>in</strong>ical features<br />
and classification, 377–378<br />
Congenital ptosis, etiology, 410, 411, 412<br />
Congenital syndromes<br />
fourth nerve palsies (FNP), 287<br />
sixth nerve palsy (SNP), evaluation<br />
protocols, 301<br />
vertical diplopia, 239–245<br />
Consecutive esotropia, etiology, 222<br />
Index 467<br />
Contralateral eyelid retraction, third<br />
nerve palsies (TNP), fascicular<br />
lesion, 259<br />
Contralesionally beat<strong>in</strong>g torsional<br />
nystagmus, cl<strong>in</strong>ical features<br />
and etiology, 385<br />
Convergence <strong>in</strong>sufficiency/paralysis<br />
b<strong>in</strong>ocular symmetric jerk nystagmus,<br />
390–391<br />
cl<strong>in</strong>ical features and etiology, 231<br />
downbeat nystagmus, 385–389, 387<br />
horizontal dysconjugate eye<br />
oscillations, 375,<br />
376<br />
Convergence-retraction nystagmus,<br />
horizontal dysconjugate eye<br />
oscillations, 375, 376<br />
Convergence spasm, cl<strong>in</strong>ical features and<br />
etiology, 230–231<br />
Cortical bl<strong>in</strong>dness, visual field defects,<br />
homonymous hemianopsia,<br />
204–205<br />
Corticosteroids<br />
giant cell arteritis (GCA)<br />
complications, 108–110, 111<br />
oral vs. IV therapy, 106–108<br />
treatment protocols, 106<br />
Graves’ ophthalmopathy (GO),<br />
352–353<br />
idiopathic pseudotumor cerebri<br />
therapy, 146, 147<br />
nonarteritic anterior ischemic optic<br />
neuropathy (NA-AION), 80<br />
ocular myasthenia gravis (OM)<br />
management, 344<br />
prognosis, 344–345<br />
optic neuritis (ON) therapy, 47–49, 48<br />
traumatic optic neuropathy (TON)<br />
management, 121–123<br />
Cranial nerve impairment<br />
fourth nerve palsies (FNP)<br />
cavernous s<strong>in</strong>us lesion, 283–284<br />
classifications, 284, 286<br />
cl<strong>in</strong>ical features, 281–283, 284<br />
congenital orig<strong>in</strong>, 287<br />
diagnostic criteria, 282–283, 283<br />
etiologies, 283, 285–286<br />
<strong>in</strong>tracranial lesions, 289–290<br />
midbra<strong>in</strong> lesions, 283
468 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Cranial nerve impairment (cont.)<br />
nonvasculopathic, evaluation<br />
protocols, 287–288, 288–291,<br />
290–292<br />
orbital lesions, 284<br />
subarachnoid space lesion, 283<br />
topographic anatomy, 281<br />
trauma, 287<br />
vasculopathic etiology, 287<br />
horizontal diplopia, 226–227<br />
sixth nerve palsies (SNP)<br />
anatomy, 295, 296–297<br />
cavernous s<strong>in</strong>us lesion, 298–300, 301<br />
evaluation guidel<strong>in</strong>es, 301–304, 303<br />
localization and classification,<br />
298–300<br />
nonisolated SNP, pont<strong>in</strong>e (lower<br />
pons) lesion, 295–296, 298–300<br />
orbital lesion, 301<br />
petrous apex lesion, 297<br />
subarachnoid space lesion, 297, 297<br />
third nerve palsies (TNP)<br />
aberrant regeneration (type 6),<br />
270–271<br />
cavernous s<strong>in</strong>us lesion, 260–261<br />
classification, 253, 254<br />
cl<strong>in</strong>ical features, 253<br />
congenital orig<strong>in</strong>s, 262<br />
fascicular lesion, 258–259<br />
isolated acquired TNP<br />
neuroimag<strong>in</strong>g protocols, 268–270<br />
nontraumatic, 262<br />
normal pupillary sph<strong>in</strong>cter,<br />
extraocular muscle <strong>in</strong>complete<br />
palsy (type 4B), 265–266<br />
normal pupillary sph<strong>in</strong>cter,<br />
extraocular muscle palsy (type<br />
4A), 264–265<br />
subnormal pupillary sph<strong>in</strong>cter<br />
dysfunction, extraocular<br />
muscle palsies (type 4C), 267<br />
localization etiologies, 253, 256–257<br />
nonisolated TNP, cl<strong>in</strong>ical evaluation,<br />
261–262, 263<br />
nuclear lesion, 254–255<br />
orbital lesion, 261<br />
progressive/unresolved (type 5<br />
TNP), 270<br />
retrospective review, 271–272<br />
subarachnoid lesion, 259–260<br />
trauma, 262<br />
vertical diplopia, 237–238<br />
C-reactive prote<strong>in</strong>, giant cell arteritis<br />
(GCA), diagnosis, 99, 101, 102<br />
‘‘Crowded’’ hyperoptic disks,<br />
papilledema, differential<br />
diagnosis, 131, 131<br />
Cyclic esotropia, cl<strong>in</strong>ical features, 228<br />
Cyclodeviation, vertical diplopia,<br />
233–234<br />
Cyclospor<strong>in</strong>, Graves’ ophthalmopathy<br />
(GO) therapy, 353<br />
Dandy criteria, pseudotumor cerebri<br />
syndrome, 135<br />
Decompensation syndromes<br />
esotropia/exotropia etiology, 220, 222,<br />
223<br />
fourth nerve palsies (FNP), 282<br />
vertical diplopia, 239<br />
Decompressions, Graves’<br />
ophthalmopathy (GO),<br />
358–359, 359–361<br />
Deep sclerotomy with collagen implant<br />
(DSCI), transient visual loss,<br />
175<br />
Dental anesthesia, third nerve palsies<br />
(TNP), orbital lesion, 261<br />
Devic’s disease, cl<strong>in</strong>ical features, 39, 42<br />
Diabetes<br />
acquired isolated third nerve palsies<br />
(TNP), normal sph<strong>in</strong>cter,<br />
complete extraocular muscle<br />
palsy (type 4A TNP), 264–265<br />
papillopathy, anterior ischemic optic<br />
neuropathy (AIONY) and, 75,<br />
75<br />
Digox<strong>in</strong>, idiopathic pseudotumor cerebri<br />
therapy, 147<br />
Diplopia<br />
acquired motor fusion deficiency,<br />
231–232<br />
b<strong>in</strong>ocular diplopia, 217<br />
horizontal (esotropia/exotropia),<br />
etiology, 219–220, 221–222<br />
childhood strabismus syndromes,<br />
esotropia/exotropia, 220, 222,<br />
223
convergence <strong>in</strong>sufficiency and<br />
paralysis, 231<br />
convergence spasm, 230–231<br />
cyclic esotropia, 228<br />
divergence <strong>in</strong>sufficiency and paralysis,<br />
228–230<br />
evaluation protocol, 215, 216<br />
foveal displacement syndrome, 245<br />
Graves’ ophthalmopathy (GO), 359,<br />
361<br />
hemifield slide phenomenon, 232<br />
horizontal diplopia<br />
central nerve disorders, 227–228<br />
cranial nerve disorders, 226–227<br />
extraocular muscle disorders,<br />
223–226, 225–226<br />
monocular diplopia, etiology and<br />
evaluation, 215, 217, 217<br />
mysathenia gravis, 218<br />
ocular motor cranial neuropathy, 218<br />
ocular myasthenia gravis (OM),<br />
management of, 342–344<br />
periodic alternat<strong>in</strong>g esotropia, 228<br />
phorias and tropias, functional<br />
assessment,<br />
219<br />
restrictive ophthalmoplegia with<br />
orbital disease, 218<br />
sensory esotropia/exotropia, 222<br />
supranuclear ophthalmoplegia, 219<br />
thyroid eye disease and, 218–219<br />
transient vs. persistent etiologies, 217,<br />
218<br />
vertical diplopia<br />
cranial nerve impairment, 237–238<br />
etiology, 234, 235–236<br />
exam<strong>in</strong>ation protocol, 232–234<br />
extraocular muscle disease, 238–240<br />
mechanical misalignment processes,<br />
240–245<br />
neuromuscular junction diseases,<br />
238<br />
supranuclear processes, 234, 236–237<br />
vertical/horizontal deviation, 219<br />
vitreous hemorrhage, secondary<br />
exotropia, 232<br />
Divergence <strong>in</strong>sufficiency/paralysis<br />
esotropia, 228–230<br />
neurologic disease, 229–230<br />
Index 469<br />
Divergence nystagmus, horizontal<br />
dysconjugate eye oscillations,<br />
375, 376<br />
Doll’s-head maneuver, diplopia<br />
evaluation, supranuclear<br />
process, 219<br />
Dom<strong>in</strong>ant optic atrophy, cl<strong>in</strong>ical features,<br />
19, 20<br />
Dorsal mesencephalic supranuclear<br />
lesions, lid retraction/lid lag,<br />
422<br />
Dorsal mid-bra<strong>in</strong> syndrome, vertical<br />
gaze palsy, 322, 323<br />
Double elevator palsy<br />
cl<strong>in</strong>ical features, 327–328<br />
vertical diplopia, 234, 236–237<br />
congenital conditions, 240<br />
Double Maddox rod test, vertical<br />
diplopia, 233–234<br />
cranial nerve impairment, 238<br />
Downbeat nystagmus, cl<strong>in</strong>ical features<br />
and etiology, 370, 385–389, 387<br />
Drug-<strong>in</strong>duced nystagmus, cl<strong>in</strong>ical<br />
features, 384<br />
Duane’s retraction syndrome, esotropia/<br />
exotropia etiology, 220, 222,<br />
223<br />
Duction movements<br />
diplopia assessment, 219<br />
fourth nerve palsies (FNP), cl<strong>in</strong>ical<br />
evaluation, 281<br />
Dysconjugate bilateral symmetric eye<br />
oscillations, etiology, 372<br />
Eccentric gaze, b<strong>in</strong>ocular symmetric jerk<br />
nystagmus, 389–391, 392<br />
Electrographic status epilepticus,<br />
stuporous/comatose patients,<br />
398–399<br />
Electromyography<br />
apraxia of eyelid open<strong>in</strong>g, 410<br />
myasthenia gravis (MG) diagnosis, 341<br />
Electrophysiologic test<strong>in</strong>g, myasthenia<br />
gravis (MG) diagnosis, 341<br />
Enophthalmos, lid retraction/lid lag, 425<br />
‘‘Entomopia,’’ monocular diplopia, 214,<br />
217<br />
Epileptic nystagmus, cl<strong>in</strong>ical features,<br />
384–385
470 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Erythrocyte sedimentation rate<br />
(ESR), giant cell arteritis<br />
(GCA)<br />
cl<strong>in</strong>ical diagnosis, 94, 94–96, 98–99<br />
corticosteroid therapy, 107–108<br />
elevation patterns, 99<br />
marked elevation, evaluation<br />
protocols, 105, 105<br />
normal values, 99<br />
Esotropia (ET)<br />
cyclic esotropia, 228<br />
divergence <strong>in</strong>sufficiency/paralysis,<br />
228–230<br />
etiology, 219–220, 221–223<br />
childhood strabismus syndromes,<br />
220, 222, 223<br />
periodic alternat<strong>in</strong>g esotropia<br />
(PAE), 228<br />
sensory esotropia, 222<br />
thyroid eye disease, 224–226<br />
Ethambutol exposure, toxic optic<br />
neuropathy, cl<strong>in</strong>ical features,<br />
10–12<br />
Exophthalmos, gaze-evoked transient<br />
visual loss (TVL), 170–171<br />
Exotropia (XT)<br />
cranial nerve lesions, 227<br />
etiology, 219–220, 221–223<br />
childhood strabismus syndromes,<br />
220, 222, 223<br />
isolated medial rectus paresis, 226<br />
orbital trauma, 226<br />
secondary exotropia, vitrous<br />
hemorrhage, 232<br />
sensory exotropia, 222<br />
vitreous hemorrhage, 227–228, 232<br />
Extraocular muscular disorders<br />
acquired isolated third nerve palsies<br />
(TNP)<br />
normal sph<strong>in</strong>cter, complete extraocular<br />
muscle palsy (type 4A TNP),<br />
264–265<br />
normal sph<strong>in</strong>cter, <strong>in</strong>complete<br />
extraocular muscle palsy (type<br />
4B TNP), 265–266<br />
subnormal pupillary sph<strong>in</strong>cter<br />
dysfunction, extraocular<br />
muscle palsies (type 4C TNP),<br />
267<br />
Graves’ ophthalmopathy (GO),<br />
radiotherapy, 355–356<br />
Graves’ ophthalmopathy (GO) and,<br />
therapeutic strategies, 351, 353<br />
horizontal diplopia, 223–226<br />
myasthemia gravis (MG) vs. ocular<br />
myasthenia gravis (OMG),<br />
differential diagnosis,<br />
338–341<br />
vertical diplopia, 238–239<br />
Eyelid nystagmus, etiology, 423–424<br />
Eyelids. See also Lid retraction/lid lag<br />
anatomy, 421<br />
Facial nerve palsy, one-and-a-half<br />
syndrome, 319–320<br />
Fallen eye syndrome, vertical diplopia,<br />
242<br />
Fascicular lesion, third nerve palsies<br />
(TNP), 258–259<br />
Fisher’s syndrome, vertical diplopia, 238<br />
Fluid-attenuated <strong>in</strong>version recovery<br />
(FLAIR) imag<strong>in</strong>g, <strong>in</strong>ternuclear<br />
ophthalmoplegia (INO), 319<br />
Foster Kennedy syndrome, pseudotumor<br />
cerebri syndrome, differential<br />
diagnosis, 143–144<br />
Fourth nerve palsies (FNP)<br />
acquired isolated fourth nerve palsies<br />
(FNP), evaluation protocols,<br />
288, 288–292<br />
cavernous s<strong>in</strong>us lesion, 283–284<br />
classifications, 284, 286<br />
cl<strong>in</strong>ical features, 281–283, 284<br />
congenital orig<strong>in</strong>, 287<br />
diagnostic criteria, 282–283, 283<br />
etiologies, 283, 285–286<br />
<strong>in</strong>tracranial lesions, 289–290<br />
localization and classification, 284,<br />
285–286<br />
midbra<strong>in</strong> lesions, 283<br />
nonvasculopathic, evaluation<br />
protocols, 287–288, 288–291,<br />
290–292<br />
orbital lesions, 284<br />
subarachnoid space lesion, 283<br />
topographic anatomy, 281<br />
trauma, 287<br />
vasculopathic etiology, 287
Foveal displacement syndrome,<br />
b<strong>in</strong>ocular diplopia, 245<br />
Frisen papilledema grad<strong>in</strong>g scale, 131,<br />
134<br />
Fusion procedures, psoria/tropia<br />
management, 219<br />
GABA agonists, vertical nystagmus<br />
management, 388–389<br />
Gabapent<strong>in</strong>, pendular nystagmus<br />
therapy, 381–382<br />
Gaze disorders<br />
abducens nucleus anatomy, 312, 314<br />
horizontal conjugate gaze, anatomy,<br />
311<br />
horizontal gaze palsies<br />
cl<strong>in</strong>ical evaluation, 312, 315<br />
lesion localization and classification,<br />
311, 312–314<br />
<strong>in</strong>ternuclear ophthalmoplegia (INO)<br />
cl<strong>in</strong>ical features, 314–315, 316<br />
etiology, 316, 317–318<br />
evaluation protocols, 318–319<br />
medial longitud<strong>in</strong>al fasciculus (MLF)<br />
anatomy, 312, 314<br />
one-and-a-half syndrome<br />
cl<strong>in</strong>ical evaluation, 319–321, 321<br />
etiologies, 321, 321<br />
p<strong>in</strong>g-pong gaze, stuporous/comatose<br />
patients, 397–399<br />
short-cycle periodic alternat<strong>in</strong>g gaze,<br />
stuporous/comatose patients,<br />
397–399<br />
skew deviation, cl<strong>in</strong>ical features,<br />
327–328<br />
supranuclear monocular elevation<br />
paresis, cl<strong>in</strong>ical features,<br />
327–328<br />
vertical gaze impairment<br />
anatomy, 321<br />
etiology, 322, 324–325<br />
evaluation protocols, 323, 325–326,<br />
326<br />
lesion localization of palsies, 321–322,<br />
322–323<br />
vertical one-and-a-half syndrome,<br />
cl<strong>in</strong>ical features, 327–328<br />
‘‘Gaze-evoked’’ nystagmus, def<strong>in</strong>ed, 367<br />
Index 471<br />
Gaze-evoked nystagmus, etiology,<br />
389–391<br />
Gaze-evoked transient visual loss (TVL),<br />
evaluation of, 169, 170–171<br />
Giant cell arteritis (GCA)<br />
b<strong>in</strong>ocular transient visual loss, 182–183<br />
cl<strong>in</strong>ical features, 93, 94<br />
atypical features, 98–99, 100–101<br />
diagnostic suspicion, 94, 94–96, 96–98,<br />
98–99<br />
ESR elevation, 99, 105<br />
ESR normal values, 99<br />
evaluation flowchart, 109<br />
hematologic test<strong>in</strong>g, 99, 101, 102<br />
laboratory abnormalities, 99, 102, 105<br />
low cl<strong>in</strong>ical suspicion, evaluation<br />
criteria, 105, 105<br />
moderate cl<strong>in</strong>ical suspicion, evaluation<br />
criteria, 104–105<br />
patient evaluation protocols, 104–105,<br />
105<br />
TAB procedures, 101–104<br />
therapeutic regimen, 106–110<br />
complications, 108–110, 111<br />
oral vs. IV corticosteroids, 106–108<br />
steroid complications, 108, 110<br />
transient visual loss, 175<br />
Giant cell myocarditis, horizontal<br />
diplopia, 224–226<br />
Glaucoma, optic disc cupp<strong>in</strong>g, optic<br />
neuropathy, differential<br />
diagnosis, 2<br />
Gliomas, anterior visual pathway, optic<br />
neuropathy, 5, 9<br />
Glucocorticoids, Graves’<br />
ophthalmopathy (GO)<br />
therapy, 354–356, 355<br />
Graves’ ophthalmopathy (GO)<br />
cl<strong>in</strong>ical features, 349, 350<br />
decompression studies, 358–359<br />
evaluation and treatment flowchart,<br />
360<br />
imag<strong>in</strong>g studies, 349–350<br />
immunosuppressive therapy, 352–354<br />
lid retraction, treatment protocols, 356<br />
lid retraction/lid lag, 424–425<br />
ocular and orbital <strong>in</strong>flammation, 352<br />
proptosis and/or compressive optic<br />
neuropathy, 357, 359
472 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Graves’ ophthalmopathy (GO) (cont.)<br />
proptosis without optic neuropathy,<br />
359, 361<br />
radiotherapy, 354–356<br />
strabismus management, 356–357<br />
systemic thyroid status, 350<br />
therapeutic management, 350–352,<br />
355<br />
vertical diplopia, 241<br />
Guilla<strong>in</strong>-Barré syndrome, vertical<br />
diplopia, 238<br />
Guilla<strong>in</strong>-Mollaret triangle lesions,<br />
pendular nystagmus, 382<br />
‘‘Half-moon syndrome,’’ visual field<br />
defects, monocular temporal<br />
crescent, 192<br />
Hematologic test<strong>in</strong>g, giant cell arteritis<br />
(GCA), 99, 101, 102<br />
Hemianopic anosognosia, visual field<br />
defects, occipital lesions,<br />
203–205<br />
Hemifacial spasm, ptosis, 409–410<br />
Hemifield slide phenomenon<br />
b<strong>in</strong>ocular diplopia, 245<br />
cl<strong>in</strong>ical features, 232<br />
Hereditary optic neuropathy, cl<strong>in</strong>ical<br />
features, 19, 20–22, 22<br />
Her<strong>in</strong>g’s law, pseudo-overaction, fourth<br />
nerve palsies (FNP), 282<br />
1H-magnetic resonance spectroscopy,<br />
Graves’ ophthalmopathy<br />
(GO), 350<br />
Homonymous hemianopsia, visual field<br />
defects, 198<br />
lateral geniculate body lesion, 199–200<br />
normal neuroimag<strong>in</strong>g, 205–207<br />
occipital lesions, 202–205<br />
optic radiation, 201–202<br />
optic tract lesions, 198–199<br />
therapeutic management, 207<br />
Horizontal diplopia<br />
b<strong>in</strong>ocular horizontal diplopia, etiology,<br />
219–220, 221–222<br />
central nerve disorders, 227–228<br />
cranial nerve disorders, 226–227<br />
extraocular muscle disorders, 223–226,<br />
225–226<br />
Horizontal dysconjugate eye oscillations,<br />
evaluation and etiology, 375,<br />
376<br />
Horizontal gaze palsies<br />
cl<strong>in</strong>ical evaluation, 312, 315<br />
lesion localization and classification,<br />
311, 312–314<br />
one-and-a-half syndrome, 320–321<br />
Horizontal nystagmus, etiology,<br />
382–383<br />
Horizontal pendular nystagmus,<br />
diagnosis and etiology, 380<br />
Horizontal smooth pursuit defects, lesion<br />
localization, 311, 312–314<br />
Horner’s syndrome<br />
anisocoria, 445, 445–456<br />
alternat<strong>in</strong>g Horner’s syndrome,<br />
448<br />
central Horner’s syndrome, 446, 447<br />
congenital Horner’s syndrome, 449<br />
imag<strong>in</strong>g and evaluation protocols,<br />
455<br />
isolated Horner’s syndrome, 446<br />
isolated postganglionic Horner’s<br />
syndrome, 455–456<br />
pediatric patients, 454–455<br />
pharmacologic localization, 452–454,<br />
453<br />
postganglionic Horner’s syndrome,<br />
446–447, 449–451<br />
preganglionic (<strong>in</strong>termediate)<br />
Horner’s syndrome, 446, 448<br />
trauma etiology, 449, 451<br />
fourth nerve palsy (FNP), cavernous<br />
s<strong>in</strong>us lesion, 283<br />
monocular TVL with, 173–179<br />
ptosis etiology, 413, 414, 415<br />
Hydrocephalus, periodic alternat<strong>in</strong>g<br />
esotropia (PAE), 228<br />
Hydroxyamphetam<strong>in</strong>es,<br />
pharmacological localization<br />
of Horner’s syndrome,<br />
452–454, 453<br />
Hyperdeviation, vertical diplopia,<br />
congenital conditions, 239–240<br />
Hyperglobus, ptosis etiology, 410<br />
Hyperthyroidism, Graves’<br />
ophthalmopathy (GO),<br />
therapeutic strategies, 351
Hypertropia<br />
fourth nerve palsies (FNP), 281–283,<br />
284<br />
ptosis etiology, 410<br />
thyroid eye disease, 224–226<br />
Hypoglobus, lid retraction/lid lag, 425<br />
Ice-pack test, myasthenia gravis (MG)<br />
diagnosis, 340–341<br />
Immunoglobul<strong>in</strong> therapy<br />
Graves’ ophthalmopathy (GO),<br />
353–354<br />
optic neuritis (ON), 49<br />
Immunosuppressive therapy<br />
Graves’ ophthalmopathy (GO),<br />
352–353<br />
saccadic <strong>in</strong>trusions, classification and<br />
etiology, 395<br />
Infectious disease<br />
optical disc edema with macular star<br />
(ODEMA)<br />
cl<strong>in</strong>ical features, 64<br />
differential diagnosis, 64–66, 67<br />
saccadic <strong>in</strong>trusions, classification and<br />
etiology, 393–395<br />
Inferior rectus fibrosis syndrome, vertical<br />
diplopia, 243<br />
Inferior rectus paresis, isolated, third<br />
nerve palsies (TNP), 254–255<br />
Infiltrative optic neuropathy, cl<strong>in</strong>ical<br />
features, 5–6, 8, 13–14<br />
Inflammatory optic neuropathy, cl<strong>in</strong>ical<br />
features, 56, 8, 13–14<br />
‘‘Insect eye,’’ monocular diplopia, 214,<br />
217<br />
Interferon therapy, optic neuritis (ON),<br />
49<br />
Intermittent angle closure glaucoma<br />
monocular transient visual loss,<br />
175–176<br />
read<strong>in</strong>g-evoked TVL, 171<br />
Intermittent LP <strong>in</strong>hibition (ILPI), apraxia<br />
of eyelid open<strong>in</strong>g, 410<br />
Internal carotid artery (ICA),<br />
atherosclerotic disease,<br />
monocular TVL with, 173–179<br />
Internuclear ophthalmoplegia (INO)<br />
cl<strong>in</strong>ical features, 314–315, 316<br />
etiology, 316, 317–318, 318<br />
Index 473<br />
evaluation protocols, 318–319, 320<br />
horizontal gaze palsy, 314<br />
nystagmus etiology, 370<br />
Internuclear ophthalmoplegia (INO) of<br />
abduction, 315<br />
Intracranial lesions, isolated fourth nerve<br />
palsies (TNP), 289–290, 290291<br />
Intracranial pressure<br />
papilledema evaluation, 132–133, 135,<br />
135<br />
pseudotumor cerebri syndrome,<br />
136–137<br />
lumboperitoneal shunt (LSP)<br />
complications and, 148–155,<br />
149<br />
vertical diplopia, 237–238<br />
Ipsilateral ptosis, third nerve palsies<br />
(TNP), fascicular lesion, 259<br />
Iris abnormalities, structural<br />
abnormalities, anisocoria, 439,<br />
439–440<br />
Jakob-Creutzfeldt disease, Heidenha<strong>in</strong><br />
variant, homonymous<br />
hemianopsia, 205–206<br />
Junctional scotoma, visual field defects,<br />
193–194<br />
Kearns-Sayre syndrome, ptosis etiology,<br />
412–413, 414<br />
Kjer’s hereditary optic neuropathy,<br />
cl<strong>in</strong>ical features, 19, 20<br />
Latent nystagmus, diagnosis and<br />
management, 378–379<br />
Lateral geniculate body lesions,<br />
homonymous hemianopsia,<br />
199–200<br />
Leber’s hereditary optic neuropathy<br />
(LHON), cl<strong>in</strong>ical features, 19,<br />
20–21, 22<br />
Levator palpebrae superioris (LPS)<br />
lid retraction/lid lag and, 421, 423–424<br />
vertical diplopia and spasm of, 236<br />
Levodopa, nonarteritic anterior ischemic<br />
optic neuropathy (NA-AION)<br />
therapy, 82–83<br />
Lewy body disease, homonymous<br />
hemianopsia, 206
474 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Lid retraction/lid lag<br />
evaluation protocol, 427, 428<br />
eyelid anatomy, 421<br />
Graves’ ophthalmopathy (GO),<br />
management options, 356<br />
lower eyelid retraction, etiology,<br />
428–429, 429<br />
miscellaneous etiologies, 425, 426–427,<br />
427<br />
neurogenic etiology, 422–424<br />
neuromuscular/myopathic etiology,<br />
424–425<br />
Light-near dissociation<br />
anisocoria, etiology, 434, 434<br />
pupillary abnormalities, syphilis<br />
serology test<strong>in</strong>g, 444–445<br />
Light reaction, anisocoria, 433<br />
normal pupil, 445<br />
Locked-<strong>in</strong> syndrome, cl<strong>in</strong>ical evaluation,<br />
327–328<br />
Lower eyelid retraction, etiologies,<br />
428–429, 429<br />
Lumbar punctures<br />
optic neuritis (ON), <strong>in</strong>dications for, 46<br />
papilledema, cl<strong>in</strong>ical evaluation, 135<br />
progressive/unresolved sixth nerve<br />
palsy (SNP), 304<br />
pseudotumor cerebri therapy, 147<br />
Lumboperitoneal shunt (LPS),<br />
pseudotumor cerebri therapy,<br />
148–155, 149<br />
Lutz posterior INO. See INO of<br />
abduction<br />
Lyme disease, neuroret<strong>in</strong>itis and optic<br />
disc edema with macular star<br />
(ODEMS), 66<br />
Macro-square-wave jerks, saccadic<br />
<strong>in</strong>trusions, classification and<br />
etiology, 393–395<br />
Magnetic resonance imag<strong>in</strong>g (MRI)<br />
acquired isolated third nerve palsies<br />
(TNP), neuroimag<strong>in</strong>g<br />
protocols, 268–270<br />
gaze-evoked transient visual loss<br />
(TVL), 170–171<br />
Graves’ ophthalmopathy (GO), 349<br />
horizontal gaze palsies, 312<br />
Horner’s syndrome evaluation, 455<br />
<strong>in</strong>ternuclear ophthalmoplegia (INO),<br />
319<br />
isolated fourth nerve palsies (TNP),<br />
289–290, 290–292<br />
monocular TVL assessment, 172<br />
nonarteritic anterior ischemic optic<br />
neuropathy (NA-AION),<br />
80–81<br />
optic neuritis, 44–46<br />
multiple sclerosis risk assessment,<br />
51–53<br />
papilledema, cl<strong>in</strong>ical evaluation, 133,<br />
135,<br />
135<br />
pseudotumor cerebri syndrome,<br />
136–137<br />
third nerve palsies (TNP), retrospective<br />
studies, 271–272<br />
transient visual loss assessment,<br />
178–179<br />
vertical gaze palsy, 325–326<br />
Medial longitud<strong>in</strong>al fasciculus (MLF)<br />
anatomy, 312, 314<br />
lid retraction/lid lag etiology, 422–424<br />
one-and-a-half syndrome, 319–321<br />
Medial rectus paresis, isolated<br />
exotropia (XT), 226<br />
third nerve palsies (TNP), 254–255<br />
Men<strong>in</strong>giomas<br />
compressive optic neuropathy, cl<strong>in</strong>ical<br />
features, 5, 67<br />
monocular TVL with, 171–172<br />
Meso-diencephalon disorders, vertical<br />
gaze palsy, 322<br />
Methylprednisolone<br />
giant cell arteritis (GCA) therapy,<br />
106–108<br />
optic neuritis (ON) therapy, 47–49, 48<br />
traumatic optic neuropathy (TON),<br />
122–123<br />
Meyer’s loop, homonymous<br />
hemianopsia, lateral<br />
geniculate body lesions,<br />
199–200<br />
Midbra<strong>in</strong> lesion<br />
fourth nerve palsy (FNP), 283<br />
ptosis etiology, 413, 414, 415<br />
Migra<strong>in</strong>es<br />
b<strong>in</strong>ocular transient visual loss, 179–184
monocular TVL with, 171–172<br />
third nerve palsies (TNP),<br />
subarachnoid lesion, 256–257,<br />
259260<br />
transient visual loss, vasospasm,<br />
175–176<br />
vertical diplopia, 237–238<br />
Miller Fisher syndrome, one-and-a-half<br />
syndrome, 320–321<br />
Morbidity<br />
idiopathic pseudotumor cerebri,<br />
weight fluctuation and<br />
therapy for, 145–147, 146<br />
pseudotumor cerebri syndrome, visual<br />
field loss, 144<br />
Motor fusion deficiency, acquired motor<br />
fusion deficiency, cl<strong>in</strong>ical<br />
features, 231–232<br />
Müller muscle contraction, lid<br />
retraction/lid lag and, 424<br />
Multiple sclerosis (MS)<br />
<strong>in</strong>ternuclear ophthalmoplegia (INO),<br />
316, 317–318, 318<br />
optic neuritis and<br />
lumbar puncture results, 46<br />
neuroimag<strong>in</strong>g studies, 44–46<br />
risk assessment, 50–53, 51–52<br />
therapeutic strategies, 47–49, 48<br />
pendular nystagmus, 379–381<br />
vertical diplopia syndromes, 236–237<br />
Myasthenia gravis (MG). See also Ocular<br />
myasthenia gravis<br />
acquired isolated third nerve palsies<br />
(TNP), normal sph<strong>in</strong>cter,<br />
<strong>in</strong>complete extraocular muscle<br />
palsy (type 4B TNP), 266<br />
antiacetylchol<strong>in</strong>e receptor antibody<br />
test<strong>in</strong>g, 341<br />
diplopia and, 218<br />
horizontal diplopia, 225<br />
lid retraction/lid lag, 425<br />
non-pharmacologic diagnosis,<br />
340–341<br />
ocular myasthenia gravis, differential<br />
diagnosis, 337–341<br />
one-and-a-half syndrome, 320–321<br />
ptosis etiology, 415<br />
thymoma, CT imag<strong>in</strong>g for, 342, 343<br />
vertical diplopia, 238–239<br />
Index 475<br />
Myasthenic pseudo-INO<br />
cl<strong>in</strong>ical features, 318<br />
evaluation protocols, 318<br />
Myopathies<br />
lid retraction/lid lag and, 424–425<br />
ptosis etiology, 412–413, 414<br />
Nerve palsies, convergence spasm,<br />
230–231<br />
<strong>Neuro</strong>fibromatosis-2, papilledema,<br />
135–136<br />
<strong>Neuro</strong>genic disorders<br />
lid retraction/lid lag, 422–424<br />
ptosis etiology, 413, 414, 415<br />
<strong>Neuro</strong>imag<strong>in</strong>g<br />
acquired isolated third nerve palsies<br />
(TNP)<br />
normal sph<strong>in</strong>cter, complete<br />
extraocular muscle palsy (type<br />
4A TNP), 264–265<br />
normal sph<strong>in</strong>cter, <strong>in</strong>complete<br />
extraocular muscle palsy (type<br />
4B TNP), 266<br />
subnormal pupillary sph<strong>in</strong>cter<br />
dysfunction, extraocular<br />
muscle palsies (type 4C TNP),<br />
267<br />
fourth nerve palsy (FNP),<br />
subarachnoid space lesion,<br />
283<br />
homonymous hemianopsia, 205–207<br />
isolated fourth nerve palsies (TNP),<br />
289–290,<br />
290–292<br />
isolated third nerve palsies (TNP),<br />
268–270<br />
<strong>Neuro</strong>muscular junction diseases<br />
ptosis etiology, 415<br />
vertical diplopia, 238<br />
<strong>Neuro</strong>myelitis optica. See Devic’s<br />
disease<br />
<strong>Neuro</strong>ret<strong>in</strong>itis<br />
cl<strong>in</strong>ical features, 63–64<br />
evaluation flowchart, 67, 68<br />
<strong>in</strong>fectious agents, 64–66, 67<br />
optical disc edema with macular star<br />
(ODEMS), differential<br />
diagnosis, 64–67, 65, 67–68
476 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Nonarteritic anterior ischemic optic<br />
neuropathy<br />
(NA-AION)<br />
cl<strong>in</strong>ical features, 73–75, 74–75<br />
atypical features, 75, 78–79, 79<br />
evaluation and treatment, 78–79<br />
future research issues, 80–81<br />
giant cell arteritis (GCA), differential<br />
diagnosis, 95–96, 96–98,<br />
98–99<br />
therapeutic management<br />
aspir<strong>in</strong> therapy, 81–82<br />
experimental treatments, 82–83<br />
flowchart for, 84, 85<br />
optic nerve sheath fenestration, 83,<br />
83–85<br />
Nonketotic hyperglycemia,<br />
homonymous hemianopsia,<br />
206<br />
Nuclear complex of the posterior<br />
commissure (NPC), lid<br />
retraction/lid lag, 422–424<br />
Nuclear lesion, third nerve palsies (TNP),<br />
254–255<br />
Nutritional optic neuropathy<br />
cl<strong>in</strong>ical features, 9, 12–14, 17<br />
Leber’s hereditary optic neuropathy<br />
(LHON), differential<br />
diagnosis, 22<br />
Nylen-Barany maneuver, b<strong>in</strong>ocular<br />
symmetric jerk nystagmus,<br />
391<br />
Nystagmoid jerk<strong>in</strong>g, stuporous/<br />
comatose patients, 398–399<br />
Nystagmus<br />
acquired b<strong>in</strong>ocular pendular<br />
nystagmus, diagnosis and<br />
etiology, 379–380<br />
asymmetric b<strong>in</strong>ocular oscillations,<br />
etiology, 368, 370–372, 373<br />
b<strong>in</strong>ocular/symmetric, b<strong>in</strong>ocular/<br />
asymmetric, or monocular<br />
oscillations, 368<br />
b<strong>in</strong>ocular symmetric conjugate<br />
oscillations, 376<br />
b<strong>in</strong>ocular symmetric jerk nystagmus<br />
eccentric gaze/maneuvered<br />
<strong>in</strong>duction, 389–391<br />
etiology, 382–385<br />
b<strong>in</strong>ocular symmetric pendular<br />
conjugate oscillations,<br />
etiology, 377–382, 383<br />
cl<strong>in</strong>ical features, 367–368, 369<br />
downbeat nystagmus, 385–387, 387<br />
dysconjugate bilateral symmetric eye<br />
oscillations, 372<br />
horizontal dysconjugate oscillations,<br />
etiology, 375, 376<br />
monocular oscillations, etiology, 368,<br />
370–372, 373<br />
periodic alternat<strong>in</strong>g nystagmus (PAN),<br />
384,<br />
384–385<br />
predom<strong>in</strong>antly vertical jerk nystagmus,<br />
385–389<br />
saccadic <strong>in</strong>trusions, 391–395<br />
seesaw nystagmus, etiology, 373–374,<br />
374<br />
spontaneous oscillations, stuporous<br />
and comatose patients,<br />
396–399, 397<br />
upbeat nystagmus, 388, 388–389<br />
Obesity, idiopathic pseudotumor cerebri,<br />
140–142, 141<br />
therapeutic management protocols,<br />
145–147, 146<br />
Occipital epilepsy, idiopathic, b<strong>in</strong>ocular<br />
transient visual loss, 181<br />
Occipital hypoperfusion, b<strong>in</strong>ocular<br />
transient visual loss, migra<strong>in</strong>e<br />
and, 180<br />
Occipital ischemia, bilateral, b<strong>in</strong>ocular<br />
transient visual loss, 182<br />
Occipital lesions, visual field defects,<br />
202–205<br />
Occipital lobe tumors, b<strong>in</strong>ocular<br />
transient visual loss, 180–181<br />
Ocular bobb<strong>in</strong>g<br />
one-and-a-half syndrome, 319–320<br />
stuporous/comatose patients, 398–399<br />
Ocular flutter, saccadic <strong>in</strong>trusions,<br />
classification and etiology,<br />
393–395<br />
Ocular hypoperfusion, monocular TVL<br />
and, 173–179<br />
Ocular <strong>in</strong>flammation, Graves’<br />
ophthalmopathy (GO), 352
Ocular ischemic syndrome, transient<br />
visual loss and, 175–179<br />
Ocular motor cranial neuropathy,<br />
diplopia and, 218<br />
Ocular myasthenia gravis (OM)<br />
evaluation flowchart, 342, 343<br />
management protocols, 342–344<br />
myasthemia gravis (MG), differential<br />
diagnosis, 337–341<br />
non-pharmacologic diagnosis,<br />
340–341<br />
prognosis, 344–345<br />
test<strong>in</strong>g summary, 341–342<br />
thymectomy, <strong>in</strong>dications for, 344<br />
thymoma, CT imag<strong>in</strong>g for, 342, 343<br />
Ocular neuromyotonia (ONM),<br />
horizontal diplopia, 227<br />
Ocular tilt reaction (OTR)<br />
vertical diplopia, 234, 236–237<br />
vertical gaze disorders, 328<br />
Ocular torsion, vertical diplopia, 233–234<br />
Oculomasticatory myorhythmia,<br />
horizontal dysconjugate eye<br />
oscillations, 375,<br />
376<br />
Oculomotor nerve. See also Third nerve<br />
palsies (TNP)<br />
acquired isolated third nerve palsies<br />
(TNP), normal sph<strong>in</strong>cter,<br />
complete extraocular muscle<br />
palsy (type 4A TNP), 264–265<br />
anatomy, 253<br />
Oculopalatal myoclonus<br />
one-and-a-half syndrome, 319–320<br />
pendular nystagmus, 380<br />
One-and-a-half syndrome<br />
cl<strong>in</strong>ical features, 319–321<br />
etiologies and evaluation protocols,<br />
320–321, 321<br />
vertical gaze palsy with, 327–328<br />
Ophthalmoplegia, bilateral preganglionic<br />
<strong>in</strong>ternal, third nerve palsies<br />
(TNP), fascicular lesion,<br />
258–259<br />
Ophthalmoplegic migra<strong>in</strong>e<br />
third nerve palsies (TNP),<br />
subarachnoid lesion, 256–257,<br />
259–260<br />
vertical diplopia, 237–238<br />
Index 477<br />
Opsoclonus-myoclonus syndrome<br />
(OMS), saccadic <strong>in</strong>trusions,<br />
classification and etiology,<br />
394–395, 396<br />
Optic chiasm, visual field defects<br />
chiasmal syndromes, 194, 195–196, 196,<br />
197<br />
junctional characteristics, 192<br />
Optic disc cupp<strong>in</strong>g, optic neuropathy<br />
diagnosis, 2<br />
Optic disc drusen<br />
evaluation protocols, 130<br />
papilledema differential diagnosis,<br />
130<br />
Optic disc edema<br />
differential diagnosis,<br />
pseudopapilledema, 131, 134<br />
evaluation protocol, 154<br />
papilledema, 129<br />
differential diagnosis, optic<br />
neuropathy, 131, 131<br />
pseudopapilledma, differential<br />
diagnosis, 131, 131<br />
Optic disc edema with macular star<br />
(ODEMS)<br />
cl<strong>in</strong>ical presentation, 3, 63–64, 64<br />
evaluation flowchart, 67, 68<br />
<strong>in</strong>fectious agents, 64–66, 67<br />
neuroret<strong>in</strong>itis, differential diagnosis,<br />
64–67, 65, 67–68<br />
prognosis, 67, 69<br />
Optic glioma, compressive optic<br />
neuropathy, cl<strong>in</strong>ical features,<br />
5, 7–8<br />
Optic nerve, visual field defects,<br />
junctional characteristics,<br />
192–194<br />
Optic nerve sheath fenestration (ONSF)<br />
nonarteritic anterior ischemic optic<br />
neuropathy (NA-AION), 83,<br />
8385<br />
pseudotumor cerebri therapy, 148–155,<br />
153<br />
complications, 152–154, 153<br />
Optic neuritis (ON)<br />
anterior ischemic optic neuropathy<br />
(AION), differential diagnosis,<br />
73–75, 74–75<br />
associated disorders, 38, 40–42
478 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Optic neuritis (ON) (cont.)<br />
atypical optic neuritis, cl<strong>in</strong>ical<br />
presentation, 38,<br />
39<br />
cl<strong>in</strong>ical presentation, 3, 35, 36–38<br />
evaluation protocol, 39, 42<br />
lumbar punctures, <strong>in</strong>dications<br />
for, 46<br />
multiple sclerosis (MS) and<br />
lumbar puncture results, 46<br />
neuroimag<strong>in</strong>g studies, 44–46<br />
risk assessment, 50–53, 51–52<br />
therapeutic strategies, 47–49, 48<br />
neuroimag<strong>in</strong>g techniques, 44–46<br />
pediatric patients, cl<strong>in</strong>ical features,<br />
38–39, 43<br />
treatment strategies<br />
immunoglobul<strong>in</strong> therapy, 49<br />
<strong>in</strong>terferon therapy, 49<br />
steroids, 47–49, 48<br />
vision prognosis, 49–50<br />
visual field defects, 35–36<br />
visually evoked potentials (VEPs), 47<br />
Optic neuritis treatment trial (ONTT)<br />
study<br />
cl<strong>in</strong>ical results, 43–44, 44<br />
multiple sclerosis risk assessment,<br />
50–53, 51–52<br />
neuroimag<strong>in</strong>g techniques, multiple<br />
sclerosis lesions, 44–46<br />
therapeutic strategies, 47–49, 48<br />
vision prognosis measurements, 49–50<br />
Optic neuropathies<br />
diagnosis<br />
anterior ischemic optic neuropathy<br />
(AION), 3<br />
atypical/unexpla<strong>in</strong>ed optic<br />
neuropathy, 14, 22<br />
cl<strong>in</strong>ical evaluation flowchart, 4<br />
cl<strong>in</strong>ical features and etiology, 1, 12<br />
compressive optic neuropathy<br />
(CON), 3, 5, 58<br />
hereditary optic neuropathy, 19,<br />
20–22, 22<br />
<strong>in</strong>filtrative/<strong>in</strong>flammatory optic<br />
neuropathy, 56, 7, 8,13<br />
optic disc edema with macular star<br />
(ODEMS), 3<br />
optic neuritis, 3<br />
radiation exposure (RON), 14, 16–17,<br />
18–19<br />
toxic/nutritional optic neuropathy,<br />
914, 15–17<br />
traumatic optic neuropathy (TON), 9<br />
papilledema, differential diagnosis,<br />
131, 131<br />
visual field defects, 191–192<br />
Optic radiations, homonymous<br />
hemianopsia, 201–202<br />
Optic tract lesions, homonymous<br />
hemianopsia, visual field<br />
defects, 198–199<br />
Orbital blow-fracture, vertical diplopia,<br />
242<br />
Orbital disease<br />
diplopia and, restrictive<br />
ophthalmoplegia, 218<br />
horizontal diplopia, orbital<br />
pseudotumor/orbital<br />
myositis, 223–226<br />
Orbital fourth nerve palsies (FNP),<br />
cl<strong>in</strong>ical evaluation, 283<br />
Orbital <strong>in</strong>flammation, Graves’<br />
ophthalmopathy (GO), 352<br />
Orbital lesion<br />
fourth nerve palsies (FNP), 284<br />
sixth nerve palsies (SNP), 301<br />
third nerve palsies (TNP), 261<br />
Orbital myositis<br />
differential diagnosis, 224–226, 226<br />
horizontal diplopia, 223–226<br />
Orbital pseudotumor<br />
differential diagnosis, 224, 225<br />
horizontal diplopia, 223–226<br />
Orbital trauma. See also Traumatic optic<br />
neuropathy (TON)<br />
exotropia (XT), orbital trauma, 226<br />
horizontal diplopia, 225–226<br />
orbital blow-fracture, vertical diplopia,<br />
242<br />
third nerve palsy (TNP), 262<br />
Palatal myoclonus, pendular nystagmus,<br />
380<br />
Papilledema<br />
bilateral<br />
cl<strong>in</strong>ical evaluation, 132–133, 135, 135<br />
etiology, 131, 132
cl<strong>in</strong>ical features, 130–131, 131–134<br />
def<strong>in</strong>ed, 129<br />
differential diagnosis<br />
optic neuropathy, 131, 131<br />
pseudopapilledema, 129–130<br />
etiology, 132<br />
evaluation protocols, 130<br />
monocular TVL with, 171–172<br />
neuroimag<strong>in</strong>g studies, 132–133, 135,<br />
135<br />
pseudotumor cerebri syndrome<br />
cl<strong>in</strong>ical signs, 143–144<br />
def<strong>in</strong>ed, 135–137<br />
stages of, 131, 133<br />
unilateral papilledema, etiology,<br />
131<br />
Papilllomacular bundle, visual field<br />
defects, optic neuropathies,<br />
191–192<br />
Papillomacular <strong>in</strong>volvement, optic<br />
neuritis (ON), 36, 38<br />
Paramedian pont<strong>in</strong>e reticular formation<br />
(PPRF)<br />
horizontal gaze palsy, 314<br />
one-and-a-half syndrome, 319–321<br />
Paroxysmal superior rectus, vertical<br />
diplopia, 236<br />
Pediatric patients<br />
Horner’s syndrome evaluation,<br />
454–455<br />
optic neuritis, cl<strong>in</strong>ical features, 38–39,<br />
43<br />
pseudotumor cerebri syndrome, optic<br />
nerve sheath fenestration<br />
(ONSF) therapy <strong>in</strong>,<br />
151–155<br />
transient visual loss assessment, 179<br />
Pendular nystagmus<br />
acquired monocular variation, etiology,<br />
370<br />
diagnosis and management, 379–382,<br />
383<br />
Pentoxifyll<strong>in</strong>e, Graves’ ophthalmopathy<br />
(GO) therapy, 354<br />
Periodic alternat<strong>in</strong>g esotropia (PAE),<br />
cl<strong>in</strong>ical features, 228<br />
Periodic alternat<strong>in</strong>g nystagmus (PAN),<br />
cl<strong>in</strong>ical features and etiology,<br />
383–385, 384<br />
Index 479<br />
Peripheral ganglion cells, visual field<br />
defects, optic neuropathies,<br />
191–192<br />
Peripheral vestibular disease, horizontal<br />
nystagmus, 382–383<br />
Peripheral visual defects, cl<strong>in</strong>ical<br />
presentation, 36<br />
Pernicious anemia, nutritional optic<br />
neuropathy, cl<strong>in</strong>ical features,<br />
9, 12–14, 17<br />
Petrous apex lesion, sixth nerve palsies<br />
(SNP), 297<br />
Pharmacological localization, Horner’s<br />
syndrome, 452–454, 453,<br />
455<br />
Pharmacological mydriasis/miosis,<br />
anisocoria, 436, 437–439<br />
Phoria syndromes, vertical diplopia,<br />
239<br />
Phorias, diplopia evaluation, 219<br />
‘‘Pie-<strong>in</strong>-the-sky’’ defects, homonymous<br />
hemianopsia, optic radiations,<br />
201–202<br />
‘‘Pie-on-the-floor’’ defects, homonymous<br />
hemianopsia, optic radiations,<br />
201–202<br />
Pilocarp<strong>in</strong>e test<strong>in</strong>g<br />
anisocoria, 435<br />
tonic pupil, 440–441, 442–443<br />
P<strong>in</strong>g-pong gaze, stuporous/comatose<br />
patients, 397–399<br />
P<strong>in</strong>hole device, monocular diplopia, 215,<br />
217, 217<br />
Platelet-fibr<strong>in</strong> emboli, monocular TVL,<br />
173<br />
Plus-m<strong>in</strong>us syndrome, third nerve<br />
palsies (TNP), 259<br />
Polymerase cha<strong>in</strong> reaction (PCR), vertical<br />
gaze palsy evaluation, 326<br />
Pont<strong>in</strong>e (lower pons) lesion<br />
horizontal gaze palsies, 312, 315<br />
sixth nerve palsies (SNP), 295–296,<br />
298–300<br />
vertical gaze palsy, locked-<strong>in</strong><br />
syndrome, 327–328<br />
Posterior cerebral artery <strong>in</strong>farction,<br />
visual field defects,<br />
homonymous hemianopsia,<br />
204–205
480 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Posterior ischemic optic neuropathy<br />
(PION)<br />
associated conditions, 75, 76–78<br />
cl<strong>in</strong>ical features, 74, 7475<br />
Postganglionic Horner’s syndrome<br />
cl<strong>in</strong>ical evaluation and etiology,<br />
446–447, 450–451<br />
evaluation protocols, 455–456<br />
pharmacologic etiology, 454<br />
Postprandial transient visual loss,<br />
evaluation, 174<br />
Prednisone<br />
Graves’ ophthalmopathy (GO) therapy,<br />
351, 353<br />
optic neuritis (ON) therapy, 47–49, 48<br />
Preganglionic (<strong>in</strong>termediate) Horner’s<br />
syndrome<br />
cl<strong>in</strong>ical evaluation and etiology, 446,<br />
448<br />
pharmacologic etiology, 454<br />
Pretarsal motor persistence (PMP),<br />
apraxia of eyelid open<strong>in</strong>g, 410<br />
Pretectal pseudobobb<strong>in</strong>g, stuporous/<br />
comatose patients, 399<br />
Proptosis, Graves’ ophthalmopathy<br />
(GO), treatment alternatives,<br />
357–358, 358–360, 361<br />
Prostigm<strong>in</strong> test, myasthemia gravis (MG)<br />
vs. ocular myasthenia gravis<br />
(OMG), 338–341<br />
Proton density imag<strong>in</strong>g (PDI),<br />
<strong>in</strong>ternuclear ophthalmoplegia<br />
(INO), 319<br />
Pseudo-<strong>in</strong>ternuclear ophthalmoplegia<br />
(INO), nystagmus etiology,<br />
370<br />
Pseudo-one-and-a-half syndrome,<br />
cl<strong>in</strong>ical features, 321<br />
Pseudopapilledema, differential<br />
diagnosis<br />
papilledema, 129–130<br />
true optic disc edema, 131, 134<br />
Pseudoptosis, ptosis, differential<br />
diagnosis, 409, 409<br />
Pseudotumor cerebri syndrome<br />
associated systemic disease, 136,<br />
138–139, 140<br />
cl<strong>in</strong>ical signs, 143–144<br />
def<strong>in</strong>ed, 135–137<br />
drug toxicity and, 137<br />
etiologies, 138–139<br />
evaluation protocols, 144–145<br />
gaze-evoked transient visual loss<br />
(TVL), 170–171<br />
idiopathic pseudotumor cerebri,<br />
140, 141<br />
diagnostic criteria, 140, 141<br />
risk factors and cl<strong>in</strong>ical<br />
characteristics, 140–142<br />
symptoms, 142, 143<br />
therapeutic management protocols,<br />
145–147, 146<br />
symptoms, 142, 143<br />
therapeutic management protocols,<br />
145–147, 146<br />
lumbar punctures, 147<br />
surgical procedures, 148–155,<br />
149, 154<br />
Pseudo-von Graefe phenomenon, third<br />
nerve palsy (TNP), aberrant<br />
regeneration, 270–271<br />
Ptosis<br />
acquired vs. congenital, 410, 411,<br />
412<br />
acquired, etiology, 410, 411, 412<br />
aponeurotic ptosis, 417<br />
apraxia of eyelid open<strong>in</strong>g, 410, 411<br />
blepharospasm, 409–410<br />
evaluation protocol, 416<br />
hemifacial spasm, 409–410<br />
hypertropia/hyperglobus, 410<br />
isolated, classification, 412, 412<br />
isolated, etiology, 415, 415<br />
mechanical etiology, 412, 413<br />
myasthemia gravis (MG) vs. ocular<br />
myasthenia gravis (OMG),<br />
differential diagnosis,<br />
338–341<br />
myogenic etiology, 412–413, 414<br />
neurogenic etiology, 413, 414, 415<br />
neuromuscular junction disease, 415<br />
nonisolated, etiology, 412, 412<br />
ocular myasthenia gravis (OM),<br />
management of, 342–344<br />
pseudoptosis, differential diagnosis,<br />
409, 409<br />
steroid-<strong>in</strong>duced, 417<br />
trauma and, 415
Pupillary abnormalities<br />
<strong>in</strong>termittent/transient phenomenon,<br />
etiology, 436, 439<br />
syphilis serology test<strong>in</strong>g, 444–445<br />
Pupillary sph<strong>in</strong>cter, acquired isolated<br />
third nerve palsies (TNP)<br />
complete extraocular muscle palsy<br />
(type 4A TNP), 264–265<br />
<strong>in</strong>complete extraocular muscle palsy<br />
(type 4B TNP), 265–266<br />
subnormal dysfunction, extraocular<br />
muscle palsies (type 4C TNP),<br />
267<br />
Purtscher’s ret<strong>in</strong>opathy, venous stasis<br />
ret<strong>in</strong>opathy, transient visual<br />
loss, differential diagnosis, 174<br />
Quadrantanopias, homonymous<br />
hemianopsia<br />
occipital lesions, 202–205<br />
optic radiations, 201–202<br />
Quadruple sectoranopia, homonymous<br />
hemianopsia, lateral<br />
geniculate body lesions, 200<br />
Radiation optic neuropathy (RON),<br />
cl<strong>in</strong>ical features, 14, 16–17,<br />
18–19, 19<br />
Radioactive iod<strong>in</strong>e (RAI) therapy,<br />
Graves’ ophthalmopathy<br />
(GO), 351, 353<br />
Radiotherapy, Graves’ ophthalmopathy<br />
(GO), 354–356, 355<br />
Read<strong>in</strong>g-evoked transient visual loss<br />
(TVL), evaluation, 171<br />
Rebound nystagmus, etiology, 389–391<br />
Relative afferent pupillary defect<br />
(RAPD), optic tract lesions,<br />
homonymous hemianopsia,<br />
198–199<br />
Repetitive divergence, stuporous/<br />
comatose patients, 398–399<br />
Restrictive ophthalmopathy, vertical<br />
diplopia, 240–242<br />
Restrictive ophthalmoplegia, orbital<br />
disease, diplopia and, 218<br />
Ret<strong>in</strong>al emboli, monocular TVL and,<br />
172–173, 177, 177–179<br />
Index 481<br />
Ret<strong>in</strong>al fibers, visual field defects, optic<br />
neuropathies, 192<br />
Ret<strong>in</strong>al lesions, visual field defects, 189,<br />
191, 191<br />
Rubeosis iridis, transient visual loss and,<br />
ocular ischemic syndrome,<br />
175–179<br />
Saccadic <strong>in</strong>trusions, classification and<br />
etiology, 391–395, 394<br />
Sc<strong>in</strong>tillat<strong>in</strong>g scotoms, b<strong>in</strong>ocular transient<br />
visual loss, 180–182<br />
Seesaw nystagmus, cl<strong>in</strong>ical features and<br />
etiologies, 373–374, 374<br />
Seizure disorders<br />
b<strong>in</strong>ocular transient visual loss, 180–181<br />
epileptic nystagmus, 384–385<br />
lid retraction/lid lag and, 423–424<br />
Sensory esotropia, etiology, 222<br />
Sensory exotropia, etiology, 222<br />
Short-cycle periodic alternat<strong>in</strong>g gaze,<br />
stuporous/comatose patients,<br />
397–399<br />
Shy-Drager syndrome, alternat<strong>in</strong>g<br />
Horner’s syndrome, 448<br />
Silent s<strong>in</strong>us syndrome, vertical diplopia,<br />
242<br />
S<strong>in</strong>gle-fiber electromyography (SFEMG)<br />
abnormalities<br />
myasthenia gravis (MG) diagnosis, 341<br />
ocular myasthenia gravis (OM)<br />
diagnosis, 341–342<br />
S<strong>in</strong>gle photon emission tomography<br />
(SPECT), b<strong>in</strong>ocular transient<br />
visual loss, migra<strong>in</strong>e and, 180<br />
Sixth nerve palsies (SNP)<br />
anatomy, 295, 296–297<br />
cavernous s<strong>in</strong>us lesion, 298–300, 301<br />
evaluation guidel<strong>in</strong>es, 301–304, 303<br />
isolated vasculopathic, evaluation<br />
protocols, 301–302<br />
localization and classification, 296,<br />
298–300<br />
nonisolated SNP, pont<strong>in</strong>e (lower pons)<br />
lesion, 295–296, 298–300<br />
nonvasculopathic, evaluation<br />
protocols, 302<br />
orbital lesion, 301<br />
petrous apex lesion, 297
482 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Sixth nerve palsies (SNP) (cont.)<br />
progressive/unresolved SNP,<br />
evaluation protocols, 302<br />
subarachnoid space lesion, 297, 297<br />
unilateral isolated SNP, diagnostic<br />
criteria, 296<br />
Skew deviation, cl<strong>in</strong>ical evaluation,<br />
327–328<br />
‘‘Sleep test,’’ myasthenia gravis (MG)<br />
diagnosis, 340–341<br />
Small-cell lung cancer (SCLC), saccadic<br />
<strong>in</strong>trusions, classification and<br />
etiology, 394–395<br />
Smok<strong>in</strong>g<br />
Graves’ ophthalmopathy (GO) therapy,<br />
352<br />
nutritional optic neuropathy, cl<strong>in</strong>ical<br />
features, 9, 12–14, 17<br />
Spasmus nutans, nystagmus etiology,<br />
368, 370<br />
Spontaneous anterior chamber<br />
hemorrhage, transient visual<br />
loss, 175<br />
Spontaneous eye oscillations, stuporous/<br />
comatose patients, 396–399,<br />
397–398<br />
Spontaneous jerk nystagmus, cl<strong>in</strong>ical<br />
features and etiology, 385<br />
Square-wave jerks, saccadic <strong>in</strong>trusions,<br />
classification and etiology,<br />
391–395, 394<br />
Steroids<br />
Graves’ ophthalmopathy (GO) therapy,<br />
354–356, 355<br />
ptosis etiology, 417<br />
Strabismus<br />
divergence <strong>in</strong>sufficiency/paralysis,<br />
228–230<br />
Graves’ ophthalmopathy (GO),<br />
treatment alternatives,<br />
356–357<br />
vertical diplopia, 239–245<br />
Striate cortex lesions, visual field defects,<br />
homonymous hemianopsia,<br />
204–205<br />
Stroke patients, transient visual loss risk<br />
and, 176–179<br />
Stuporous patients, spontaneous eye<br />
oscillations, 396–399, 397–398<br />
Subarachnoid space lesion<br />
fourth nerve palsy (FNP), 283<br />
sixth nerve palsies (SNP), 297, 297<br />
third nerve palsies (TNP), 256–257,<br />
259–260<br />
neuroimag<strong>in</strong>g protocols, 269–270<br />
Superior oblique click syndrome, vertical<br />
diplopia, 241–242<br />
Superior oblique myokymia (SOM)<br />
nystagmus etiology, 370–372<br />
vertical diplopia, 237–238<br />
Superior oblique (SO) palsy<br />
fascicular lesion, 259<br />
pseudo-overaction, fourth nerve<br />
palsies (FNP),<br />
282<br />
vertical diplopia, 236<br />
Superior rectus palsy, isolated, vertical<br />
diplopia, 238–239<br />
Supranuclear gaze disorders<br />
abducens nucleus anatomy, 312,<br />
314<br />
horizontal conjugate gaze, anatomy,<br />
311<br />
horizontal gaze palsies<br />
cl<strong>in</strong>ical evaluation, 312, 315<br />
lesion localization and classification,<br />
311, 312–314<br />
<strong>in</strong>ternuclear ophthalmoplegia (INO)<br />
cl<strong>in</strong>ical features, 314–315, 316<br />
etiology, 316, 317–318<br />
evaluation protocols, 318–319<br />
medial longitud<strong>in</strong>al fasciculus (MLF)<br />
anatomy, 312, 314<br />
one-and-a-half syndrome<br />
cl<strong>in</strong>ical evaluation, 319–321, 321<br />
etiologies, 321, 321<br />
skew deviation, cl<strong>in</strong>ical features,<br />
327–328<br />
supranuclear monocular elevation<br />
paresis, cl<strong>in</strong>ical features,<br />
327–328<br />
vertical gaze impairment<br />
anatomy, 321<br />
etiology, 322, 324–325<br />
evaluation protocols, 323, 325–326,<br />
326<br />
lesion localization of palsies, 321–322,<br />
322–323
vertical one-and-a-half syndrome,<br />
cl<strong>in</strong>ical features, 327–328<br />
Supranuclear monocular elevation<br />
paresis, cl<strong>in</strong>ical features,<br />
327–328<br />
Supranuclear ophthalmoplegia<br />
diplopia and, 219<br />
vertical diplopia, 234, 236–237<br />
Surgical procedures<br />
Graves’ ophthalmopathy (GO)<br />
lid retraction treatments, 356<br />
proptosis and/or compressive optic<br />
neuropathy (CON), 353,<br />
357–358, 358–359, 361<br />
traumatic optic neuropathy (TON),<br />
124, 125, 126<br />
Syphilitic men<strong>in</strong>gitis, neuroret<strong>in</strong>itis and<br />
optic disc edema with macular<br />
star (ODEMS), 65–66<br />
Systemic disease, pseudotumor cerebri<br />
syndrome, 136–137<br />
Systemic lupus erythematosus, transient<br />
visual loss, 175<br />
Systemic thyroid status, Graves’<br />
ophthalmopathy (GO), 350<br />
Temporal artery biopsy (TAB), giant cell<br />
arteritis (GCA)<br />
cl<strong>in</strong>ical diagnosis, 94, 94–96, 96–98,<br />
98–99<br />
<strong>in</strong>dications for, 101–104<br />
therapeutic complications, 108–110, 111<br />
unilateral vs. bilateral TAB, 101–104<br />
Tensilon test, myasthemia gravis (MG)<br />
vs. ocular myasthenia gravis<br />
(OMG), differential diagnosis,<br />
338–341<br />
Third nerve palsies (TNP)<br />
aberrant regeneration (type 6), 270–271<br />
acquired isolated TNP<br />
neuroimag<strong>in</strong>g protocols, 268–270<br />
nontraumatic (type 4), 262<br />
normal pupillary sph<strong>in</strong>cter,<br />
extraocular muscle <strong>in</strong>complete<br />
palsy (type 4B), 265–266<br />
normal pupillary sph<strong>in</strong>cter,<br />
extraocular muscle palsy (type<br />
4A), 264–265<br />
Index 483<br />
subnormal pupillary sph<strong>in</strong>cter<br />
dysfunction, extraocular<br />
muscle palsies (type 4C), 267<br />
anisocoria, 434–435<br />
bilateral, nuclear lesions, 255<br />
cavernous s<strong>in</strong>us lesion, 260–261<br />
classification, 253, 254<br />
cl<strong>in</strong>ical features, 253<br />
congenital orig<strong>in</strong>s, 262<br />
fascicular lesion, 258–259<br />
lid retraction/lid lag and, 423–424<br />
localization etiologies, 253, 256–257<br />
monocular elevator paresis,<br />
subarachnoid lesion, 259–260<br />
nonisolated TNP, cl<strong>in</strong>ical evaluation,<br />
261–262, 263<br />
nuclear lesion, 254–255<br />
orbital lesion, 261<br />
progressive/unresolved (type 5 TNP),<br />
evaluation protocols, 270<br />
ptosis etiology, 413, 414, 415<br />
retrospective review, 271–272<br />
subarachnoid lesion, 259–260<br />
trauma, 262<br />
unilateral TNP, nuclear lesions, 255<br />
Thromboembolic disease, monocular<br />
TVL and, 172–179<br />
Thymectomy, ocular myasthenia gravis<br />
(OM), <strong>in</strong>dications for, 344<br />
Thymoma evaluation, myasthenia gravis<br />
(MG) diagnosis, 342<br />
Thyroid eye disease<br />
diplopia and, 218–219<br />
Graves’ ophthalmopathy<br />
cl<strong>in</strong>ical features, 349, 350<br />
decompression studies, 358–359<br />
evaluation and treatment flowchart,<br />
360<br />
imag<strong>in</strong>g studies, 349–350<br />
immunosuppressive therapy, 352–354<br />
lid retraction, treatment protocols,<br />
356<br />
ocular and orbital <strong>in</strong>flammation, 352<br />
proptosis and/or compressive optic<br />
neuropathy, 357, 359<br />
proptosis without optic neuropathy,<br />
359, 361<br />
radiotherapy, 354–356<br />
strabismus management, 356–357
484 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Thyroid eye disease (cont.)<br />
systemic thyroid status, 350<br />
therapeutic management, 350–352,<br />
355<br />
horizontal diplopia, 224–226, 226<br />
lid retraction/lid lag, 424–425<br />
vertical diplopia, 241<br />
Thyroid ophthalmopathy, b<strong>in</strong>ocular<br />
transient visual loss, 182<br />
Tilted optic disc, papilledema,<br />
differential diagnosis, 131, 131<br />
Tonic pupil<br />
Adie’s tonic pupil syndrome<br />
cl<strong>in</strong>ical features, 441, 444<br />
etiology, 441<br />
neuroimag<strong>in</strong>g studies, 441<br />
therapeutic strategies, 444<br />
anisocoria, 440–441, 442–443<br />
isolated, differential diagnosis, 441,<br />
442–443<br />
syphilis serology test<strong>in</strong>g, 444–445<br />
Toxic optic neuropathy, cl<strong>in</strong>ical features,<br />
914, 15–16<br />
Transient diplopia, etiology, 217, 218<br />
Transient visual loss (TVL)<br />
b<strong>in</strong>ocular TVL<br />
etiology, 179–183<br />
evaluation, 167, 169–170, 183<br />
etiology, 171–179, 177<br />
monocular TVL<br />
duration of episodes, read<strong>in</strong>g-evoked<br />
TVL, 171–179<br />
gaze positions, 168, 168169, 171<br />
patient history, 167<br />
prolonged read<strong>in</strong>g, 171<br />
Transurethral prostatic resection (TURP)<br />
syndrome, b<strong>in</strong>ocular transient<br />
visual loss, 183<br />
Traquair’s junctional scotoma, visual<br />
field defects, 193–194<br />
Trauma. See Orbital trauma<br />
fourth nerve palsies (FNP), 287<br />
Horner’s syndrome and, 449, 451<br />
ptosis etiology, 415<br />
sixth nerve palsy (SNP), evaluation<br />
protocols,<br />
301<br />
Traumatic optic neuropathy (TON)<br />
cl<strong>in</strong>ical features, 9, 119, 119<br />
evaluation and treatment flow chart,<br />
125<br />
grad<strong>in</strong>g and classification, 120, 121<br />
neuroimag<strong>in</strong>g, 119–120<br />
pathogenesis, 119, 120<br />
therapeutic management strategies,<br />
120–126, 125<br />
corticosteroid therapy, 122–123<br />
protocols and classification, 123, 123<br />
surgical management, 124–126<br />
Trigem<strong>in</strong>o-oculomotor synk<strong>in</strong>esis, lid<br />
retraction/lid lag and,<br />
423–424<br />
Trochlear nerve. See also Fourth nerve<br />
palsies (FNP)<br />
localization, 284<br />
Tropias, diplopia evaluation, 219<br />
T2-weighted imag<strong>in</strong>g, <strong>in</strong>ternuclear<br />
ophthalmoplegia (INO), 319<br />
Upbeat nystagmus, cl<strong>in</strong>ical features and<br />
etiology, 386–389, 388<br />
Uveitis-glaucoma-hyphema (UGH)<br />
syndrome, transient visual<br />
loss, 175, 177<br />
Valsalva maneuver<br />
b<strong>in</strong>ocular symmetric jerk nystagmus,<br />
391<br />
gaze-evoked transient visual loss<br />
(TVL), 170–171<br />
Vasculopathic fourth nerve palsies<br />
(FNP), evaluation protocols,<br />
287<br />
Vasculopathic sixth nerve palsy (SNP)<br />
evaluation protocols, 301–302<br />
risk factors, 302<br />
Vasospasm, transient visual loss, 175–176<br />
Venereal Disease Research Laboratory<br />
(VDRL) test, papilledema,<br />
cl<strong>in</strong>ical evaluation, 135<br />
Venous occlusive disease, pseudotumor<br />
cerebri syndrome, evaluation<br />
protocols, 144–145<br />
Venous stasis ret<strong>in</strong>opathy, transient<br />
visual loss, 174–179<br />
Vertebrobasilar transient ischemic<br />
attacks, b<strong>in</strong>ocular transient<br />
visual loss, 182
evaluation protocol, 183<br />
Vertical diplopia<br />
cranial nerve impairment, 237–238<br />
etiology, 234, 235–236<br />
exam<strong>in</strong>ation protocol, 232–234<br />
extraocular muscle disease, 238–240<br />
mechanical misalignment processes,<br />
240–245<br />
monocular elevator paresis, 234,<br />
236–237<br />
neuromuscular junction diseases, 238<br />
supranuclear processes, 234, 236–237<br />
Vertical gaze impairment<br />
anatomy, 321<br />
etiology, 322, 324–325<br />
evaluation protocols, 323, 325–326, 326<br />
lesion localization of palsies, 321–322,<br />
322–323<br />
Vertical jerk nystagmus, cl<strong>in</strong>ical features<br />
and etiology, 385–389<br />
Vertical ocular myoclonus, stuporous/<br />
comatose patients, 399<br />
Vertical pendular nystagmus, etiology,<br />
370<br />
Vertigo, b<strong>in</strong>ocular symmetric jerk<br />
nystagmus, 390–391<br />
Vistibular tone imbalance, nystagmus<br />
etiology, 367368 i368<br />
Visual acuity<br />
optical disc edema with macular star<br />
(ODEMA), 64<br />
pseudotumor cerebri syndrome, optic<br />
nerve sheath fenestration<br />
(ONSF) and, 150–155<br />
traumatic optic neuropathy (TON),<br />
121–122<br />
Visual field defects<br />
bilateral defects, cl<strong>in</strong>ical features, 192,<br />
193<br />
b<strong>in</strong>asal hemianospia, 196, 198<br />
bitemporal hemianopsia, 194, 195–196,<br />
196<br />
evaluation protocols, 190<br />
homonymous hemianopsia, 198<br />
lateral geniculate body lesion,<br />
199–200<br />
normal neuroimag<strong>in</strong>g, 205–207<br />
optic radiation, 201–202<br />
optic tract lesions, 198–199<br />
Index 485<br />
therapeutic management, 207<br />
junctional defects, 192–194<br />
monocular temporal cresent, 192<br />
occipital lesions, 202–205<br />
optic neuritis (ON), 35–36<br />
optic neuropathy, 191–192<br />
pseudotumor cerebri syndrome, 144<br />
ret<strong>in</strong>al lesions, 189, 191, 191<br />
topographical diagnosis, 189, 190<br />
unexpla<strong>in</strong>ed defects, 207–208<br />
unilateral defects, 189<br />
Visual loss<br />
giant cell arteritis (GCA), cl<strong>in</strong>ical<br />
diagnosis, 95–96, 96–98,<br />
98–99<br />
monocular nystagmus, 370<br />
transient visual loss (TVL)<br />
b<strong>in</strong>ocular TVL<br />
etiology, 179–183<br />
evaluation, 167, 169–170, 183<br />
etiology, 171–179, 177<br />
monocular TVL, duration of episodes,<br />
171–179<br />
monocular TVL, gaze positions, 168,<br />
168–169, 171<br />
patient history, 167<br />
prolonged read<strong>in</strong>g, 171<br />
Visually evoked potentials (VEPs), optic<br />
neuritis (ON), <strong>in</strong>dications for,<br />
47<br />
Visual prognosis<br />
nonarteritic anterior ischemic optic<br />
neuropathy (NA-AION), 79<br />
levodopa therapy, 82–83<br />
optic nerve sheath fenestration, 83,<br />
83–85<br />
optic disc edema with macular<br />
(ODEMS), 67, 69<br />
optic neuritis (ON), 49–50<br />
Visual recovery<br />
giant cell arteritis (GCA), therapeutic<br />
protocols, 106–108<br />
optic neuritis (ON), 49–50<br />
traumatic optic neuropathy (TON)<br />
corticosteroid therapy, 122–123<br />
natural history and progression,<br />
120–122<br />
Vitam<strong>in</strong> A, idiopathic pseudotumor<br />
cerebri etiology, 140–142, 141
486 <strong>Cl<strong>in</strong>ical</strong> <strong>Pathways</strong> <strong>in</strong> <strong>Neuro</strong>-Ophthalmology, second edition<br />
Vitam<strong>in</strong> E deficiency syndrome,<br />
horizontal gaze palsy, 314–315<br />
Vitreous hemorrhage, secondary<br />
exotropia, 232<br />
Voluntary nystagmus, diagnosis and<br />
management, 379<br />
Wall-eyed bilateral <strong>in</strong>ternuclear<br />
ophthalmoplegia (WEBINO)<br />
syndrome, horizontal gaze<br />
palsy, 314<br />
Weber’s syndrome, third nerve palsies<br />
(TNP), fascicular lesion,<br />
258–259<br />
Weight reduction programs, idiopathic<br />
pseudotumor cerebri therapy,<br />
145–147, 146<br />
Wernicke’s syndrome, vertical diplopia,<br />
236<br />
Whipple’s disease, vertical gaze palsy,<br />
325–326<br />
Wilbrand’s knee, visual field defects,<br />
junctional characteristics,<br />
192–194









![SISTEM SENSORY [Compatibility Mode].pdf](https://img.yumpu.com/20667975/1/190x245/sistem-sensory-compatibility-modepdf.jpg?quality=85)