Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
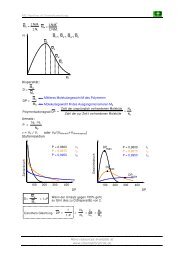
Synthesis of JASMONE<br />
Br<br />
Br<br />
Br<br />
+<br />
CO 2Et<br />
CO 2Et<br />
CO2Et CO2Et S N2'<br />
K2CO3 CO2Et ?<br />
DMF CO2Et CO 2Et<br />
CO 2Et<br />
∆<br />
150°C<br />
CO2Et ?<br />
CO2Et Industrial Synthesis (Firmenich) Helv. Chim. Acta 1978, 61, 2524-2529<br />
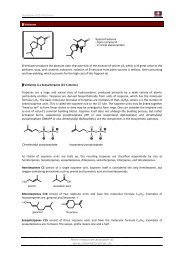
O<br />
+ Br 2 (hν)<br />
(heat in a radical<br />
fashion)<br />
Br<br />
Br<br />
+<br />
Br<br />
Br<br />
Br<br />
Br<br />
E/Z mixture<br />
and..<br />
+<br />
Br<br />
Br<br />
via the<br />
enolate<br />
More resources available at<br />
www.chemistforchrist.de<br />
O<br />
O<br />
> 150°C<br />
The Problem is that during the radical bromination epoxides are involved due to the presence of air and one<br />
day the plant that produced <strong>Jasmone</strong> via this pathway was blown up and since then this reaction is not used<br />
industrially anymore.