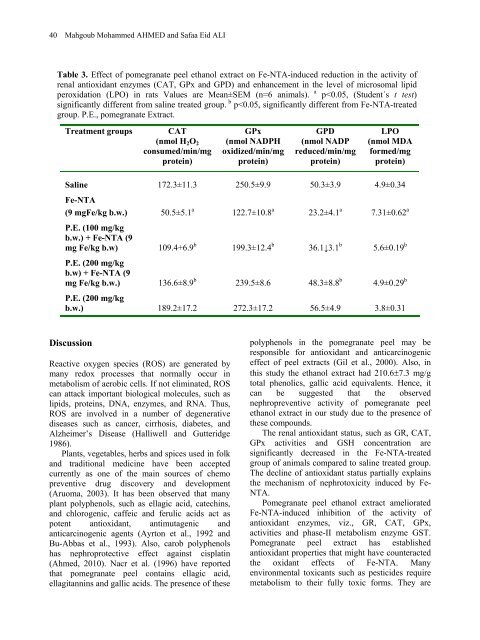
40 Mahgoub Mohammed AHMED <strong>and</strong> Safaa Eid ALI Table 3. Effect <strong>of</strong> pomegranate peel ethanol extract on Fe-NTA-induced reduction in the activity <strong>of</strong> renal antioxidant enzymes (CAT, GPx <strong>and</strong> GPD) <strong>and</strong> enhancement in the level <strong>of</strong> microsomal lipid peroxidation (LPO) in rats Values are Mean±SEM (n=6 animals). a p
<strong>of</strong>ten metabolized to proximate toxicants by phase I enzymes, e.g., cytochrome P450 which catalyze oxidative reactions. The oxidized metabolites <strong>of</strong> potentially toxic xenobiotics are then detoxified by Phase II metabolizing enzymes into the forms that are relatively inert <strong>and</strong> more easily excreted (Talalay et al., 1995). GSH depletion increases the sensitivity <strong>of</strong> organ to oxidative <strong>and</strong> chemical injury. Studies with a number <strong>of</strong> models show that the metabolism <strong>of</strong> xenobiotics <strong>of</strong>ten produced GSH depletion (Mitchell et al., 1973 <strong>and</strong> Ahmed <strong>and</strong> Zaki, 2009). The depletion <strong>of</strong> GSH, also, seems to be the prime factor that permits lipid peroxidation in the Fe- NTA treated group. Pretreatment <strong>of</strong> pomegranate peel extract reduced the depletion <strong>of</strong> GSH levels <strong>and</strong> provided protection to the kidney. The protection <strong>of</strong> GSH is by forming the substrate for GPx activity that can react directly with various aldehydes produced from the peroxidation <strong>of</strong> membrane lipid. Pomegranate peel extract pretreatment also reduced the elevated levels <strong>of</strong> serum urea <strong>and</strong> ceatinine that are marker parameters <strong>of</strong> kidney toxicity. In conclusion, we can say that, the high antioxidant <strong>and</strong> nephropreventive effect <strong>of</strong> the pomegranate peel extract appeared to be attributed to its high phenolics content. The mechanism <strong>of</strong> action <strong>of</strong> pomegranate peel extract may be through induction <strong>of</strong> various antioxidant <strong>and</strong> phase II enzymes, <strong>and</strong> scavenging reactive oxygen species. Thus our data suggest that pomegranate peel ethanol extract is a potent nephropreventive agent. Further work is required for the isolation <strong>and</strong> characterization <strong>of</strong> individual phenolic compounds present in peel ethanol extract <strong>and</strong> to determine the mechanisms involved in the nephropreventive effect <strong>of</strong> pomegranate peel extract. References Ahmed MM <strong>and</strong> Zaki NI. Assessment the ameliorative effect <strong>of</strong> pomegranate <strong>and</strong> rutin on chlorpyrifos-ethyl-induced oxidative stress in rats. Nature <strong>and</strong> Science. 7(10): 49-61, 2009. Ahmed MM. Biochemical studies on nephroprotective effect <strong>of</strong> carob (Ceratonia siliqua L.) growing in Egypt. Nature <strong>and</strong> Science. 8 (3): 41-47, 2010. Ajaikumar K.B, Asheef, M, Babu BH <strong>and</strong> Padikkala J. The inhibition <strong>of</strong> gastric mucosal injury by Punica granatum L. (pomegranate) Pomegranate peel extract against renal oxidative damage 41 methanolic extract. <strong>Journal</strong> <strong>of</strong> Ethnopharma. 96: 171–176, 2005. American Institute <strong>of</strong> Nutrition. Report <strong>of</strong> the American Institute <strong>of</strong> Nutrition. Ad Hoc Committee J Nutr. 110: 1340-1348, 1980. Aruoma OI. Methodological considerations for characterizing potential antioxidant actions <strong>of</strong> bioactive components in plant foods. Mutation Research. (523-524): 9-20, 2003. Athar M <strong>and</strong> Iqbal M. Ferric nitrilotriacetate promotes N-diethylnitrosamine-induced renal tumorigenesis in the rat: implications for the involvement <strong>of</strong> oxidative stress. Carcinogenesis. 19 (6):1133-1139, 1998 Ayrton AD, Lewis DF, Waker R. <strong>and</strong> Ioannides C. Antimutagenicity <strong>of</strong> ellagic acid towards the food mutagen IQ: investigation into possible mechanisms <strong>of</strong> action. Food <strong>and</strong> Chemical Toxicology, 3D: 289-295, 1992. Bartles H, Bohmer M <strong>and</strong> Heieri C. Serum keratinin bestmmung ohno enteeissen. Clinical Chimca Acta. 37: 139-197, 1972. Bu-Abbas A, Clifford MN, Walker R <strong>and</strong> Ioannides C. Marker antimutagenic potential <strong>of</strong> aquous green tea extracts: Mechanism <strong>of</strong> action. Mutagenesis. 9: 325-331, 1994. Carlberg I <strong>and</strong> Mannervik B. Glutathione level in rat brain. J Biological Chemistry. 250: 4480- 4575, 1975. De Flora S <strong>and</strong> Ramel C. Mechanism <strong>of</strong> inhibition <strong>of</strong> mutagenesis <strong>and</strong> carcinogenesis: classification <strong>and</strong> overview. Mutation Research. 202: 285-306, 1988. De Freitas JM <strong>and</strong> Meneghini R. Iron <strong>and</strong> its sensitive balance in the cell. Mutation Research. 475: 153-159, 2001. Deiana M, Aruma OI, Rosa A, Crobu V, Piga R <strong>and</strong> Derri MA. The effect <strong>of</strong> ferricnitrilotriacetic acid on the pr<strong>of</strong>ile <strong>of</strong> polyunsaturated fatty acids in the kidney <strong>and</strong> liver <strong>of</strong> rats. Toxicol Letters. 123: 125-133, 2001. Dhawan BN, Patnaik GK, Rastogi RP, Singh KK <strong>and</strong> T<strong>and</strong>on JS. Screening <strong>of</strong> Indian plants for biological activity. Indian J. Exp. Biol. 15: 208- 219, 1977. Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 82: 70-77, 1959. Gil MI, Tomas-Barberan FA, Hess Pierce B, Holcr<strong>of</strong>t, DM. <strong>and</strong> Kader AA. Antioxidant