Sucrose
Sucrose
Sucrose
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Sucrose</strong><br />
430. 300.<br />
425.5 298.15<br />
422.50 297.0<br />
Finegold, Franks, et al.,<br />
1989<br />
Anderson, Higbie, et al.,<br />
1950<br />
Parks, Huffman, et al.,<br />
1933<br />
Reaction thermochemistry data<br />
http://webbook.nist.gov/cgi/cbook.cgi?Name=sucrose&Units=SI&cTC=on&cTR=on#ref-3<br />
T(glass) = 330 K.; DH<br />
T = 298 to 363 K.; DH<br />
02/23/2007 05:54 AM<br />
T = 94 to 297 K. Value is unsmoothed experimental<br />
datum.; DH<br />
Go To: Top, Condensed phase thermochemistry data, References, Notes / Error Report<br />
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights<br />
reserved.<br />
Data compiled by: H.Y. Afeefy, J.F. Liebman, and S.E. Stein<br />
Note: Please consider using the reaction search for this species. This page allows searching of all reactions involving<br />
this species. A general reaction search form is also available. Future versions of this site may rely on reaction search<br />
pages in place of the enumerated reaction displays seen below.<br />
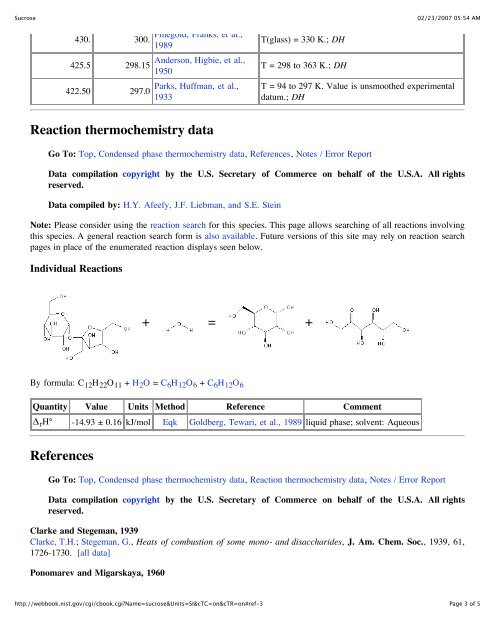
Individual Reactions<br />
+ = +<br />
By formula: C 12H 22O 11 + H 2O = C 6H 12O 6 + C 6H 12O 6<br />
Quantity Value Units Method Reference Comment<br />
Δ rH° -14.93 ± 0.16 kJ/mol Eqk Goldberg, Tewari, et al., 1989 liquid phase; solvent: Aqueous<br />
References<br />
Go To: Top, Condensed phase thermochemistry data, Reaction thermochemistry data, Notes / Error Report<br />
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights<br />
reserved.<br />
Clarke and Stegeman, 1939<br />
Clarke, T.H.; Stegeman, G., Heats of combustion of some mono- and disaccharides, J. Am. Chem. Soc., 1939, 61,<br />
1726-1730. [all data]<br />
Ponomarev and Migarskaya, 1960<br />
Page 3 of 5


![Experiment 9 Research Into Alternate Syntheses of [Cr(en)3]Cl3 ...](https://img.yumpu.com/18756539/1/190x245/experiment-9-research-into-alternate-syntheses-of-cren3cl3-.jpg?quality=85)