n - Wits Structural Chemistry
n - Wits Structural Chemistry
n - Wits Structural Chemistry
- No tags were found...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
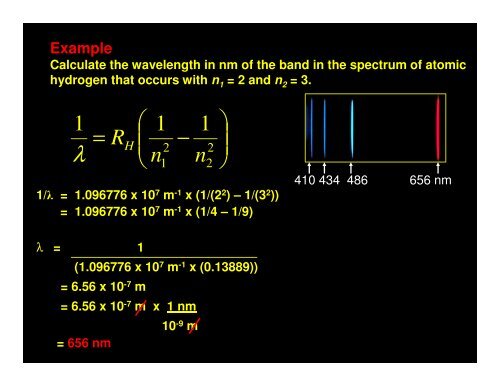
ExampleCalculate the wavelength in nm of the band in the spectrum of atomichydrogen that occurs with n 1 = 2 and n 2 = 3.1λ⎛ 1 1= R ⎜−H 2 2⎝ n n1 21/λ = 1.096776 x 10 7 m -1 x (1/(2 2 ) – 1/(3 2 ))= 1.096776 x 10 7 m -1 x (1/4 – 1/9)⎞⎟⎠410 434 486 656 nmλ = 1(1.096776 x 10 7 m -1 x (0.13889))= 6.56 x 10 -7 m= 6.56 x 10 -7 m x 1 nm10 -9 m= 656 nm