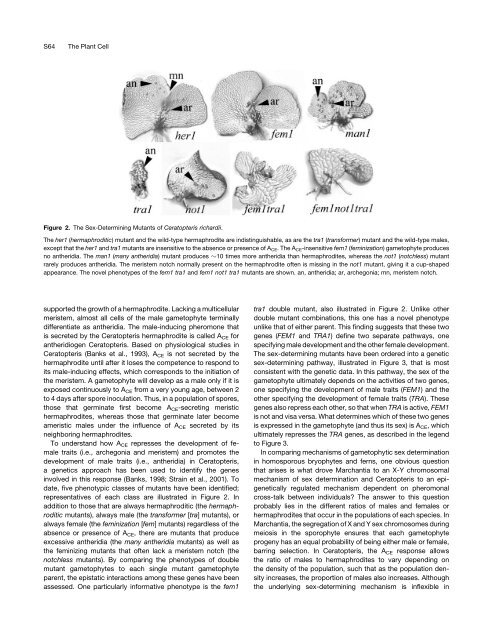
S64The Plant CellFigure 2. The <strong>Sex</strong>-<strong>Determ<strong>in</strong><strong>in</strong>g</strong> Mutants of Ceratopteris richardii.The her1 (hermaphroditic) mutant and the wild-type hermaphrodite are <strong>in</strong>dist<strong>in</strong>guishable, as are the tra1 (transformer) mutant and the wild-type males,except that the her1 and tra1 mutants are <strong>in</strong>sensitive to the absence or presence of A CE .TheA CE -<strong>in</strong>sensitive fem1 (fem<strong>in</strong>ization) gametophyte producesno antheridia. The man1 (many antheridia) mutant produces 10 times more antheridia than hermaphrodites, whereas the not1 (notchless) mutantrarely produces antheridia. The meristem notch normally present on the hermaphrodite often is miss<strong>in</strong>g <strong>in</strong> the not1 mutant, giv<strong>in</strong>g it a cup-shapedappearance. The novel phenotypes of the fem1 tra1 and fem1 not1 tra1 mutants are shown. an, antheridia; ar, archegonia; mn, meristem notch.supported the growth of a hermaphrodite. Lack<strong>in</strong>g a multicellularmeristem, almost all cells of the male gametophyte term<strong>in</strong>allydifferentiate as antheridia. The male-<strong>in</strong>duc<strong>in</strong>g pheromone thatis secreted by the Ceratopteris hermaphrodite is called A CE forantheridiogen Ceratopteris. Based on physiological studies <strong>in</strong>Ceratopteris (Banks et al., 1993), A CE is not secreted by thehermaphrodite until after it loses the competence to respond toits male-<strong>in</strong>duc<strong>in</strong>g effects, which corresponds to the <strong>in</strong>itiation ofthe meristem. A gametophyte will develop as a male only if it isexposed cont<strong>in</strong>uously to A CE from a very young age, between 2to 4 days after spore <strong>in</strong>oculation. Thus, <strong>in</strong> a population of spores,those that germ<strong>in</strong>ate first become A CE -secret<strong>in</strong>g meristichermaphrodites, whereas those that germ<strong>in</strong>ate later becomeameristic males under the <strong>in</strong>fluence of A CE secreted by itsneighbor<strong>in</strong>g hermaphrodites.To understand how A CE represses the development of femaletraits (i.e., archegonia and meristem) and promotes thedevelopment of male traits (i.e., antheridia) <strong>in</strong> Ceratopteris,a genetics approach has been used to identify the genes<strong>in</strong>volved <strong>in</strong> this response (Banks, 1998; Stra<strong>in</strong> et al., 2001). Todate, five phenotypic classes of mutants have been identified;representatives of each class are illustrated <strong>in</strong> Figure 2. Inaddition to those that are always hermaphroditic (the hermaphroditicmutants), always male (the transformer [tra] mutants), oralways female (the fem<strong>in</strong>ization [fem] mutants) regardless of theabsence or presence of A CE , there are mutants that produceexcessive antheridia (the many antheridia mutants) as well asthe fem<strong>in</strong>iz<strong>in</strong>g mutants that often lack a meristem notch (thenotchless mutants). By compar<strong>in</strong>g the phenotypes of doublemutant gametophytes to each s<strong>in</strong>gle mutant gametophyteparent, the epistatic <strong>in</strong>teractions among these genes have beenassessed. One particularly <strong>in</strong>formative phenotype is the fem1tra1 double mutant, also illustrated <strong>in</strong> Figure 2. Unlike otherdouble mutant comb<strong>in</strong>ations, this one has a novel phenotypeunlike that of either parent. This f<strong>in</strong>d<strong>in</strong>g suggests that these twogenes (FEM1 and TRA1) def<strong>in</strong>e two separate pathways, onespecify<strong>in</strong>g male development and the other female development.The sex-determ<strong>in</strong><strong>in</strong>g mutants have been ordered <strong>in</strong>to a geneticsex-determ<strong>in</strong><strong>in</strong>g pathway, illustrated <strong>in</strong> Figure 3, that is mostconsistent with the genetic data. In this pathway, the sex of thegametophyte ultimately depends on the activities of two genes,one specify<strong>in</strong>g the development of male traits (FEM1) and theother specify<strong>in</strong>g the development of female traits (TRA). Thesegenes also repress each other, so that when TRA is active, FEM1is not and visa versa. What determ<strong>in</strong>es which of these two genesis expressed <strong>in</strong> the gametophyte (and thus its sex) is A CE , whichultimately represses the TRA genes, as described <strong>in</strong> the legendto Figure 3.In compar<strong>in</strong>g mechanisms of gametophytic sex determ<strong>in</strong>ation<strong>in</strong> homosporous bryophytes and ferns, one obvious questionthat arises is what drove Marchantia to an X-Y chromosomalmechanism of sex determ<strong>in</strong>ation and Ceratopteris to an epigeneticallyregulated mechanism dependent on pheromonalcross-talk between <strong>in</strong>dividuals? The answer to this questionprobably lies <strong>in</strong> the different ratios of males and females orhermaphrodites that occur <strong>in</strong> the populations of each species. InMarchantia, the segregation of X and Y sex chromosomes dur<strong>in</strong>gmeiosis <strong>in</strong> the sporophyte ensures that each gametophyteprogeny has an equal probability of be<strong>in</strong>g either male or female,barr<strong>in</strong>g selection. In Ceratopteris, the A CE response allowsthe ratio of males to hermaphrodites to vary depend<strong>in</strong>g onthe density of the population, such that as the population density<strong>in</strong>creases, the proportion of males also <strong>in</strong>creases. Althoughthe underly<strong>in</strong>g sex-determ<strong>in</strong><strong>in</strong>g mechanism is <strong>in</strong>flexible <strong>in</strong>
<strong>Sex</strong> Determ<strong>in</strong>ationS65Figure 3. The Genetic <strong>Sex</strong>-<strong>Determ<strong>in</strong><strong>in</strong>g</strong> Pathways <strong>in</strong> the Fern Ceratopteris,the Fly Drosophila melanogaster, and the Nematode Caenorhabditiselegans.The genetic model of sex determ<strong>in</strong>ation <strong>in</strong> Ceratopteris (Stra<strong>in</strong> et al.,2001) is dependent on two genes, FEM1 and TRA (there are at least twoTRA genes), which promote the differentiation of male (antheridia) andfemale (meristem and archegonia) traits, respectively. FEM1 and TRAalso antagonize each other such that if FEM1 is active, TRA is not, andvice versa. What determ<strong>in</strong>es which of these two genes prevails <strong>in</strong> thegametophyte and thus its sex is the pheromone A CE , which activates theHER genes, of which there are at least five, and sets <strong>in</strong>to motion a seriesof switches that ultimately result <strong>in</strong> male development (i.e., FEM1 on andTRA off). These switches are thrown <strong>in</strong> the opposite direction whenspores germ<strong>in</strong>ate <strong>in</strong> the absence of A CE . Although FEM1 represses TRAand TRA represses FEM1, they do not do so directly. TRA activatesMAN1, which represses FEM1, and FEM1 activates NOT1, whichrepresses TRA. Because TRA and FEM1 are the primary regulators ofsex, NOT1 and MAN1 are considered regulators of the regulators. Thesex determ<strong>in</strong>ation pathway <strong>in</strong> C. elegans (Hodgk<strong>in</strong>, 1987; Villeneuve andMeyer, 1990) is l<strong>in</strong>ear and consists of a series of negative controlswitches. The state of the <strong>in</strong>itial switch gene (xol-1) is dependent on theratio of X to autosomal (A) chromosomes. If the ultimate downstreamgene <strong>in</strong> this pathway (TRA-1) is high, the nematode develops asa hermaphrodite. If TRA-1 is low, it develops as a male. The l<strong>in</strong>ear sexdeterm<strong>in</strong>ation pathway shown for Drosophila is from the mid-1980s(Baker and Ridge, 1980; Cl<strong>in</strong>e, 1983). There are actually other sexdeterm<strong>in</strong><strong>in</strong>gpathways that account for most aspects of sexualphenotype (reviewed by Oliver, 2002); the pathway shown is the somaticpathway. In the soma, Sxl is the key regulator of sex, and its state ofactivity is determ<strong>in</strong>ed by the X:A ratio. The dsx gene is the downstreamregulatory gene that ultimately determ<strong>in</strong>es whether male or female genesare expressed <strong>in</strong> the soma.Marchantia, it is flexible enough <strong>in</strong> Ceratopteris to allow each<strong>in</strong>dividual to determ<strong>in</strong>e its sex accord<strong>in</strong>g to the size of thepopulation <strong>in</strong> which it resides and the speed at which it germ<strong>in</strong>atesrelative to its neighbors. The flexibility of the Ceratopterissex-determ<strong>in</strong><strong>in</strong>g mechanism is reflected <strong>in</strong> its sex-determ<strong>in</strong><strong>in</strong>gpathway, and this becomes especially apparent compared withthe sex-determ<strong>in</strong><strong>in</strong>g pathways known from other organisms,<strong>in</strong>clud<strong>in</strong>g Drosophila melanogaster and Caenorhabditis elegans,which are illustrated <strong>in</strong> Figure 3. In both of these animals, an<strong>in</strong>dividual’s sex (male or female <strong>in</strong> D. melanogaster and male orhermaphrodite <strong>in</strong> C. elegans) is determ<strong>in</strong>ed genetically by theratio of X to autosomal chromosomes. This ratio is read andeither activates or represses the activities of downstream genes<strong>in</strong> each pathway. In both animals, the sex ultimately depends onthe state of the term<strong>in</strong>al gene <strong>in</strong> each l<strong>in</strong>ear pathway, TRA1 <strong>in</strong> thecase of C. elegans. The Ceratopteris sex-determ<strong>in</strong><strong>in</strong>g pathway isdist<strong>in</strong>ctly different from those of D. melanogaster and C. elegans<strong>in</strong> that it is not l<strong>in</strong>ear and there are two sex-determ<strong>in</strong><strong>in</strong>g genes,one for male and one for female development. Their ability torepress each other endows each Ceratopteris spore with theflexibility to determ<strong>in</strong>e its sex upon germ<strong>in</strong>ation based onenvironmental cues.So why would a flexible mechanism of sex determ<strong>in</strong>ation thatallows sex ratios to vary be adaptive <strong>in</strong> ferns but not <strong>in</strong>bryophytes? The answer to this question may lie <strong>in</strong> the ephemeralnature of the fern gametophyte. Although the gametophytes ofbryophytes are persistent, the gametophytes of ferns are not. InCeratopteris, for example, gametophytes reach sexual maturityonly 14 days after spore <strong>in</strong>oculation and die once they arefertilized. The limited time that a fern gametophyte is able to becrossed by another might favor a sex-determ<strong>in</strong><strong>in</strong>g system thatwould promote outcross<strong>in</strong>g by <strong>in</strong>creas<strong>in</strong>g the proportion ofmales when population densities are high and ensur<strong>in</strong>g a highproportion of hermaphrodites capable of self-fertilization whenpopulation densities are low. Because there are a variety of sexdeterm<strong>in</strong><strong>in</strong>gmutants available <strong>in</strong> Ceratopteris, the hypothesisrelated to the consequences of variable versus fixed sex ratioscan be tested easily, at least under def<strong>in</strong>ed laboratory conditions.Future studies to clone the sex-determ<strong>in</strong><strong>in</strong>g genes <strong>in</strong> Ceratopteriswill be necessary to understand their biochemicalfunctions and to test their <strong>in</strong>teractions predicted by the geneticmodel. Although the size of the Ceratopteris genome is probablyvery large (n ¼ 37), the ability to <strong>in</strong>activate genes <strong>in</strong> theCeratopteris gametophyte by RNA <strong>in</strong>terference (Stout et al.,2003; G. Rutherford, M. Tanurdzic, and J.A. Banks, unpublishedobservations) provides an important means to study the effectsof <strong>in</strong>activat<strong>in</strong>g potential sex-determ<strong>in</strong><strong>in</strong>g genes <strong>in</strong> the Ceratopterisgametophyte.THE FLOWERING PLANTSAlthough unisexuality is very common <strong>in</strong> animals, hermaphroditismis the rule <strong>in</strong> angiosperms. Approximately 90% of allangiosperm species have perfect flowers with specializedorgans produc<strong>in</strong>g microspores or megaspores from which themale or female gametophytes develop. Of the rema<strong>in</strong><strong>in</strong>g species,approximately half are monoecious, produc<strong>in</strong>g unisexual flowersof both sexes on the same <strong>in</strong>dividual, and the other half aredioecious, with unisexual male and female flowers aris<strong>in</strong>g onseparate <strong>in</strong>dividuals (Yampolsky and Yampolsky, 1922). Thedistribution of dioecy and monoecy with<strong>in</strong> the angiospermphylogenetic tree strongly favors the evolutionary scenario <strong>in</strong>which unisexual flowers evolved from perfect flowers multipletimes <strong>in</strong> the angiosperm l<strong>in</strong>eage (Lebel-Hardenack and Grant,1997; Charlesworth, 2002). Not surpris<strong>in</strong>gly, there are a variety ofsex determ<strong>in</strong>ation mechanisms <strong>in</strong> the angiosperms. For organizationalpurposes only, sex determ<strong>in</strong>ation <strong>in</strong> monoecious anddioecious species are treated separately <strong>in</strong> this review.