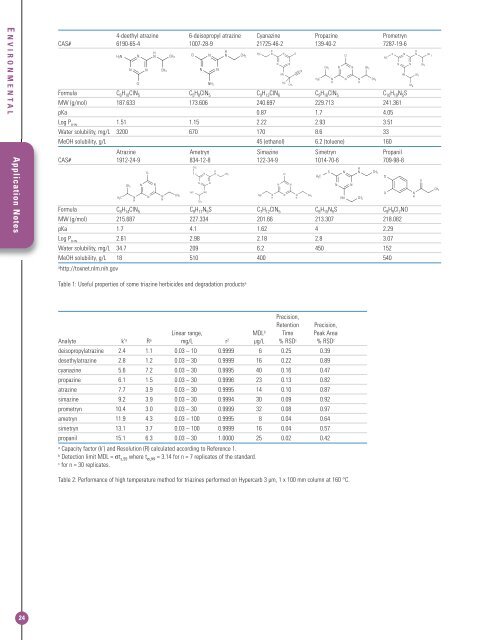
E NVIRONMENTALApplication Notes4-deethyl atrazine 6-deisopropyl atrazine Cyanazine Propazine PrometrynCAS# 6190-65-4 1007-28-9 21725-46-2 139-40-2 7287-19-6Formula C 6 H 10 ClN 5 C 5 H 8 ClN 5 C 9 H 13 ClN 6 C 9 H 16 ClN 5 C 10 H 19 N 5 SMW (g/mol) 187.633 173.606 240.697 229.713 241.361pKa 0.87 1.7 4.05Log P o-w 1.51 1.15 2.22 2.93 3.51Water solubility, mg/L 3200 670 170 8.6 33MeOH solubility, g/L 45 (ethanol) 6.2 (toluene) 160Atrazine Ametryn Simazine Simetryn PropanilCAS# 1912-24-9 834-12-8 122-34-9 1014-70-6 709-98-8Formula C 8 H 14 ClN 5 C 9 H 17 N 5 S C 7 H 12 ClN 5 C 8 H 15 N 5 S C 9 H 9 Cl 2 NOMW (g/mol) 215.687 227.334 201.66 213.307 218.082pKa 1.7 4.1 1.62 4 2.29Log P o-w 2.61 2.98 2.18 2.8 3.07Water solubility, mg/L 34.7 209 6.2 450 152MeOH solubility, g/L 18 510 400 540ahttp://toxnet.nlm.nih.govTable 1: Useful properties of some triazine herbicides and degradation products aPrecision,Retention Precision,Linear range, MDL b Time Peak AreaAnalyte k’ a R a mg/L r 2 µg/L % RSD c % RSD cdeisopropylatrazine 2.4 1.1 0.03 – 10 0.9999 6 0.25 0.39desethylatrazine 2.8 1.2 0.03 – 30 0.9999 16 0.22 0.89cyanazine 5.6 7.2 0.03 – 30 0.9995 40 0.16 0.47propazine 6.1 1.5 0.03 – 30 0.9996 23 0.13 0.82atrazine 7.7 3.9 0.03 – 30 0.9995 14 0.10 0.87simazine 9.2 3.9 0.03 – 30 0.9994 30 0.09 0.92prometryn 10.4 3.0 0.03 – 30 0.9999 32 0.08 0.97ametryn 11.9 4.3 0.03 – 100 0.9995 8 0.04 0.64simetryn 13.1 3.7 0.03 – 100 0.9999 16 0.04 0.57propanil 15.1 6.3 0.03 – 30 1.0000 25 0.02 0.42aCapacity factor (k’) and Resolution (R) calculated according to Reference 1.bDetection limit MDL = σt s,99 where t σ,99 = 3.14 for n = 7 replicates of the standard.cfor n = 30 replicates.Table 2: Performance of high temperature method for triazines performed on <strong>Hypercarb</strong> 3 µm, 1 x 100 mm column at 160 °C.24
SamplesSamples of surface water (Salinas River, Monterey County,CA), ground water (domestic well, Santa Cruz county, CA),and drinking water (San Jose, CA tap water) were collectedin accordance with established procedures, stored at 4-8 °C,and were filtered through a 0.45 µm nylon syringe filterinto a glass autosampler vial before analysis.System PreparationTo ensure good performance of this application, prepare thesystem as directed in Appendix A.ResultsSeparation of seven triazine herbicides, two triazinedegradation products often found in environmental samples,and propanil is shown in Figure 1. To optimize this separation,we adjusted the mobile phase composition to elute the firstanalyte with a capacity factor k’ > 2, thereby improvingresolution of the target analytes from sample matrix junk.Analytes spanning a wide range of polarity are well resolvedby the combination of high temperature, solvent gradient,and the selectivity of the <strong>Hypercarb</strong> stationary phase. Notethat because of the reduced viscosity of the mobile phaseat 160 °C, this separation occurs at a linear flow rate of15 mm/s – equivalent to a flow rate of over 10 mL/min on a4.6 mm i.d. column. The system backpressure under theseconditions is less than 4000 psi (272 bar).Method performance is characterized by peak resolution,linear calibration range, limits of detection, and precision ofretention time and peak area, as summarized in Table 2.MDLs for each analyte were determined by performingseven replicate injections of LC/MS-grade water fortified at aconcentration of three to five times the estimated instrumentdetection limits, calculating the standard deviation of themeasured concentration, and using the equation given inthe figure caption. Note the good precision of retentiontime and peak area, as this reflects the temperaturestability maintained by the high temperature oven.We analyzed several environmental water samples todemonstrate the efficacy of this method with real matrices.Samples of surface, ground and municipal drinking waterwere analyzed before and after fortification with a knownamount of each target analyte. Spike recovery was calculatedas the amount of each analyte found in the spiked sampledivided by the amount expected (i.e., the amount determinedin the blank plus the amount added in the spike). Even withthe dirtiest matrix, Salinas River water, the target analytesare well separated from the early eluting matrix peaks(Figure 2) and recovery of the spiked analytes exceeds 80%(Table 3).E NVIRONMENTALApplication NotesColumn: <strong>Hypercarb</strong> 3 µm, 100 x 1 mmTemperature: 160 °CFlow Rate: 500 µL/minDetector: Accela PDA at 215 nm, 20 Hz, 0s rise timeInjection: 2 µL partial loop from 5 µL loopSolvents: A: WaterB: AcetonitrileGradient: Time (min) A% B%0.00 75 251.00 70 302.20 15 852.10 75 254.00 75 25Samples: triazines and propanil in 20% acetonitrilePeaks: 1. Melamine 7. Prometryn2. Unknown 8. Atrazine3. Deisopropylatrazine 9. Ametryn4. Desethylatrazine 10. Simazine5. Cyanazine 11. Simetryn6. Propazine 12. PropanilFigure 1: Separation of triazine herbicides, degradation products, and propanilon the Accela High Speed LC by reversed-phase chromatography with UVabsorbance detection at 215 nm. Peaks: see Figure. Sample: overlay of 30injections of triazines in HPLC-grade water with 20% acetonitrile.Figure 2: Chromatograms of four environmental water samples spiked with200 µg/L each of triazines and propanil. Chromatograms obtained on theAccela High Speed LC by reversed phase chromatography with UV absorbancedetection at 215 nm. Peaks: see Figure 1. Samples: top trace A, surface water(Salinas River); trace B, ground water (Simoes’ well); trace C, drinking water(San Jose tap); bottom trace D, HPLC-grade water. Conditions: see text for details.25