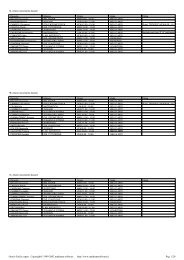
3020Phase diagram for NaClFreeze pointSalt solution-1010SolidIce +salt+-15Temp. Salt solutionSalt Temp.solution( ° F)0( ° C)-6-10-205Eutectic temperature of salt10Solid salt+ Ice23 25solution to form. This first step is relatively slow, but then the growingsolution continues to dissolve more salt <strong>and</strong> melt more ice. Passingvehicles warm the slush through friction, which speeds the dissolutionof the ice <strong>and</strong> may crush the salt <strong>and</strong> ice together, which will increasethe surface areas of the particles in contact with each other. Somecommunities use “prewetted” salt (usually rock salt with a CaCl 2 solutionsprayed on it) to speed the process.Road salt history, fun facts,<strong>and</strong> current technology1520Solution concentration (% by weight)0-5-20-21.1This phase diagram illustrates the impact of NaCl concentration <strong>and</strong>temperature on the phase of an aqueous NaCl solution.-25-30MIKE CIESIELSKIPutting salt on the roads to lessen the buildup of snow <strong>and</strong> icebegan in the 1930s, <strong>and</strong> by the 1960s, it was used by most communitieswhere snow <strong>and</strong> ice are a problem. Concerns about the effect of theuse of sodium chloride (common table salt) on the environment haveprompted some state <strong>and</strong> local road crews to explore the use of moreenvironmentally friendly salts, such as magnesium chloride <strong>and</strong> calciumchloride. These two salts have the advantage of being more effective atlowering the freezing point <strong>and</strong> there is some interestingchemistry behind this benefit.As you probably know, sodium chloride has the formulaNaCl. When it dissolves in water, it dissociates into its ions:Na + <strong>and</strong> Cl , producing two ions in solution for every NaCl formulaunit. Magnesium chloride (MgCl 2) <strong>and</strong> calcium chloride (CaCl 2)dissociate to three ions each because the metal has a 2 + charge <strong>and</strong>there are two chlorides per metal ion:MgCl 2(s) ➞ Mg 2+ (aq) + 2Cl - (aq)CaCl 2(s) ➞ Ca 2+ (aq) + 2Cl - (aq)It is the number of dissolved particles that determine the extent ofthe lowering of the freezing point of a solution. So although NaCl producestwo ions, MgCl 2 <strong>and</strong> CaCl 2 each produce three, making themmore effective. Other variations include mixtures of the magnesium <strong>and</strong>calcium chlorides, as well as magnesium <strong>and</strong> calcium acetates,Mg(C 2H 3O 2) 2 <strong>and</strong> Ca(C 2H 3O 2) 2.Practical Melt Temp. Eutectic Temp.CaCl 2 32°C 56°CMgCl 2 15°C 33°CThe technology of salting roads has become fairly sophisticated.Often, these salts are dissolved in water or some other solvent so thatthey can be sprayed onto the road surface. Having the deicing substancein a solution (i.e., fluid) form makes it possible to pump throughhoses, which allows for a more targeted application. In addition, variousanticorrosive substances are added to protect highways <strong>and</strong> cars fromthe damage the salts can cause over time. Some 15 million tons of deicingsalt is used each year in the United States <strong>and</strong> about 4–5 milliontons in Canada.You may have seen signs that warn about bridges freezing beforeroad surfaces. This is because bridges are more exposed <strong>and</strong> not insulatedby the ground from underneath like the rest of the highway. Somehigh-tech highway bridges have been constructed with deicingsprayers built right into the pavement, complete with sensors thatdetect when conditions are right (e.g., cold temperatures, high windspeeds, high humidity) for ice to form. The sensors detect the possibleformation of ice, <strong>and</strong> the deicing sprayers go to work to keep the roadwayfrom freezing.Highway engineers have been working withother interesting variations. One deicing materialthat is currently on the market mixes magnesiumchloride with sugar cane or sugar beet molasses.The sticky molasses keeps the magnesium chloridefrom getting blown or washed off the road surface.There are also substances that are added directly tothe top layer of concrete or asphalt when the road isbuilt or repaved that help prevent ice from forming.Highway workers can then get away with using less saltthan before, which is cheaper, easier on the environment,<strong>and</strong> helpful in preventing corrosion. Scientists<strong>and</strong> engineers continue to develop new ways to keepwinter highways safe while minimizing expense <strong>and</strong>environmental harm. Just another way that chemistryis keeping you out of harm’s way.Doris Kimbrough teaches chemistry at the University of Colorado-Denver. Herlast article, “Einstein’s Miraculous Year”, appeared in the December 2005 issue.16 ChemMatters, FEBRUARY 2006 http://chemistry.org/education/chemmatters.html
From Ferraris to Ford Pintos,almost every car is fightinga losing battle to rust.TBy Christen Brownleehe old-style Volkswagen Beetle: Isit a classic car, or an endangeredspecies? The answer depends onwhere you live. Although there arefew classic cars hanging aroundthe northern states or on thecoastlines, plenty of vintage automobiles stillexist in the mild southern climates <strong>and</strong> inAmerica’s interior states.The reason that Volkswagen Bugs <strong>and</strong>other older cars are dropping like flies isn’t thetypical habitat loss or human encroachmentthat's plaguing other endangered species.Classic car fleets are constantly shrinking dueto a chemical reaction that you’re no doubtalready familiar with: rusting.But why does rust unequally strike carsin the snowy states <strong>and</strong> coastal towns butleave vehicles elsewhere virtually untouched?And more importantly, how can you keepyour beloved grocery-getter safe, no matterwhat parking place you call home? Read onto get the lowdown on how rust works <strong>and</strong>what measures you can take to stop corrosionin its tracks.Electron swap meetLike all types of corrosion, rust isactually a chemical bargain, withtwo reactions in one: reduction,in which someatoms gain electrons,<strong>and</strong> oxidation, inwhich other atomslose electrons. Withall those electronsflowing from oneplace to another,rust-making is alsoconsidered an electrochemicalreaction.According to John Scully, aChemMatters, FEBRUARY 2006 17