The Nutrition of Almonds
The%20Nutrition%20of%20Almonds_HR_tcm583-241357
The%20Nutrition%20of%20Almonds_HR_tcm583-241357
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Nitrogen<br />
<strong>Almonds</strong> are among the most efficient crops studied<br />
in terms <strong>of</strong> nitrogen (N) uptake. <strong>The</strong> requirement for<br />
N fertiliser application is based on crop demand which<br />
is driven by crop yield. Hence, higher yields require<br />
more N than lower yielding crops. General fertiliser N<br />
applications rates should be 0.1 Kg N / Kg <strong>of</strong> targeted<br />
nut yield. Rates may need to be increased if trees are<br />
still actively expanding, as additional N will be needed<br />
for permanent tissue or if there are other losses/<br />
inefficiencies. Shortages <strong>of</strong> N will reduce growth and<br />
yields.<br />
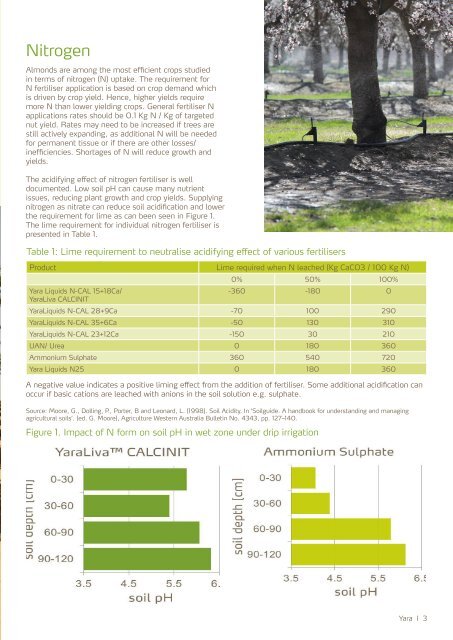
<strong>The</strong> acidifying effect <strong>of</strong> nitrogen fertiliser is well<br />
documented. Low soil pH can cause many nutrient<br />
issues, reducing plant growth and crop yields. Supplying<br />
nitrogen as nitrate can reduce soil acidification and lower<br />
the requirement for lime as can been seen in Figure 1.<br />
<strong>The</strong> lime requirement for individual nitrogen fertiliser is<br />
presented in Table 1.<br />
Table 1: Lime requirement to neutralise acidifying effect <strong>of</strong> various fertilisers<br />
Product Lime required when N leached (Kg CaCO3 / 100 Kg N)<br />
Yara Liquids N-CAL 15+18Ca/<br />
YaraLiva CALCINIT<br />
0% 50% 100%<br />
-360 -180 0<br />
YaraLiquids N-CAL 28+9Ca -70 100 290<br />
YaraLiquids N-CAL 35+6Ca -50 130 310<br />
YaraLiquids N-CAL 23+12Ca -150 30 210<br />
UAN/ Urea 0 180 360<br />
Ammonium Sulphate 360 540 720<br />
Yara Liquids N25 0 180 360<br />
A negative value indicates a positive liming effect from the addition <strong>of</strong> fertiliser. Some additional acidification can<br />
occur if basic cations are leached with anions in the soil solution e.g. sulphate.<br />
Source: Moore, G., Dolling, P., Porter, B and Leonard, L. (1998). Soil Acidity. In ‘Soilguide. A handbook for understanding and managing<br />
agricultural soils’. (ed. G. Moore), Agriculture Western Australia Bulletin No. 4343, pp. 127–140.<br />
Figure 1. Impact <strong>of</strong> N form on soil pH in wet zone under drip irrigation<br />
Yara I 3