Maladies Rares : attentes et préoccupations des acteurs ... - Leem
Maladies Rares : attentes et préoccupations des acteurs ... - Leem
Maladies Rares : attentes et préoccupations des acteurs ... - Leem
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
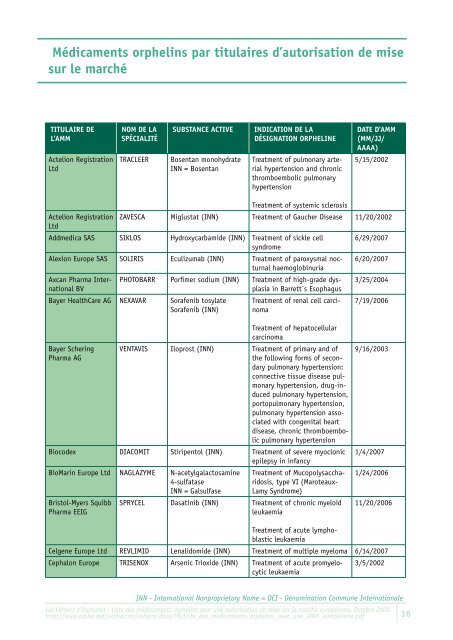
Médicaments orphelins par titulaires d’autorisation de mise<br />
sur le marché<br />
tituLaire de<br />
L’aMM<br />
Actelion Registration<br />
Ltd<br />
NoM de La<br />
spéciaLité<br />
substaNce active iNdicatioN de La<br />
désigNatioN orpheLiNe<br />
TRACLEER Bosentan monohydrate<br />
INN = Bosentan<br />
Treatment of pulmonary arterial<br />
hypertension and chronic<br />
thromboembolic pulmonary<br />
hypertension<br />
date d’aMM<br />
(MM/JJ/<br />
aaaa)<br />
5/15/2002<br />
Actelion Registration<br />
Ltd<br />
ZAVESCA Miglustat (INN)<br />
Treatment of systemic sclerosis<br />
Treatment of Gaucher Disease 11/20/2002<br />
Addmedica SAS SIKLOS Hydroxycarbamide (INN) Treatment of sickle cell<br />
syndrome<br />
6/29/2007<br />
Alexion Europe SAS SOLIRIS Eculizumab (INN) Treatment of paroxysmal nocturnal<br />
haemoglobinuria<br />
6/20/2007<br />
Axcan Pharma Inter- PHOTOBARR Porfimer sodium (INN) Treatment of high-grade dys- 3/25/2004<br />
national BV<br />
plasia in Barr<strong>et</strong>t´s Esophagus<br />
Bayer HealthCare AG NEXAVAR Sorafenib tosylate Treatment of renal cell carci- 7/19/2006<br />
Sorafenib (INN) noma<br />
Bayer Schering<br />
Pharma AG<br />
VENTAVIS Iloprost (INN)<br />
Treatment of hepatocellular<br />
carcinoma<br />
Treatment of primary and of<br />
the following forms of secondary<br />
pulmonary hypertension:<br />
connective tissue disease pulmonary<br />
hypertension, drug-induced<br />
pulmonary hypertension,<br />
portopulmonary hypertension,<br />
pulmonary hypertension associated<br />
with congenital heart<br />
disease, chronic thromboembolic<br />
pulmonary hypertension<br />
Biocodex DIACOMIT Stiripentol (INN) Treatment of severe myoclonic<br />
epilepsy in infancy<br />
BioMarin Europe Ltd NAGLAZYME N-ac<strong>et</strong>ylgalactosamine<br />
4-sulfatase<br />
INN = Galsulfase<br />
Bristol-Myers Squibb<br />
Pharma EEIG<br />
Treatment of Mucopolysaccharidosis,<br />
type VI (Maroteaux-<br />
Lamy Syndrome)<br />
SPRYCEL Dasatinib (INN) Treatment of chronic myeloid<br />
leukaemia<br />
9/16/2003<br />
1/4/2007<br />
1/24/2006<br />
11/20/2006<br />
Celgene Europe Ltd REVLIMID Lenalidomide (INN)<br />
Treatment of acute lymphoblastic<br />
leukaemia<br />
Treatment of multiple myeloma 6/14/2007<br />
Cephalon Europe TRISENOX Arsenic Trioxide (INN) Treatment of acute promyelocytic<br />
leukaemia<br />
3/5/2002<br />
INN - International Nonpropri<strong>et</strong>ary Name = DCI - Dénomination Commune Internationale<br />
Les Cahiers d’Orphan<strong>et</strong> - Liste <strong>des</strong> médicaments orphelins avec une autorisation de mise sur le marché européenne. Octobre 2008<br />
http://www.orpha.n<strong>et</strong>/orphacom/cahiers/docs/FR/Liste_<strong>des</strong>_medicaments_orphelins_avec_une_AMM_europeenne.pdf 16