Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
O<br />
ll<br />
O<br />
(CH3CH2) 2CuLi + CH ether ll<br />
3CH2C-Cl CH3CH2CCH2CH3 O<br />
ll<br />
C-Cl + (CH3) 2CuLi<br />
ether<br />
-78 o C<br />
-78 o C<br />
O<br />
ll<br />
(CH3CH2CH2) 2CuLi + CH3C-Cl ether<br />
-78 o C<br />
O<br />
ll<br />
C-CH3 กกก<br />
กกก<br />
กก<br />
alkyne กก<br />
<br />
ก H2O ก<br />
alkyne ก Markovnikov <br />
H-atom <br />
H กก<br />
R-C≡CH + H 2O HgSO 4<br />
H 2SO 4<br />
H<br />
R-C-CH<br />
ll<br />
O H<br />
6. . Ozonolysis of alkene<br />
R1 R<br />
C=C<br />
2 R4<br />
R 3 + O3<br />
Zn<br />
H 2O<br />
H<br />
R-C=CH<br />
OH<br />
R 1<br />
R 2<br />
tautomerization<br />
C=O +<br />
37<br />
H<br />
R-C−CH<br />
OH<br />
+<br />
O=C<br />
R3 R4<br />
R = alkyl, aryl H<br />
+ Zn(OH) 2<br />
H3C C=C<br />
H<br />
H3C CH3 Zn<br />
H2O C=O O=C H<br />
<br />
+ O3 2-methyl-2-butene<br />
H3C H<br />
+<br />
3C CH3 acetone acetaldehyde<br />
39<br />
41<br />
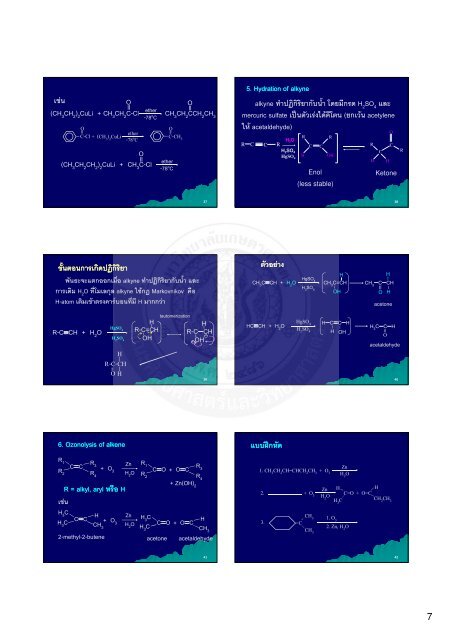
5. . Hydration of alkyne<br />
alkyne กก<br />
ก H2SO4 <br />
mercuric sulfate (ก acetylene<br />
acetaldehyde)<br />
R C C R<br />
<br />
H 2O<br />
H 2SO 4<br />
HgSO 4<br />
HgSO4 HC≡CH + H2O H2SO4 R<br />
H<br />
C C<br />
Enol<br />
R<br />
OH<br />
(less stable)<br />
H<br />
HgSO4 CH3C≡CH + H2O CH<br />
H 3C=CH<br />
2SO4 OH<br />
ก ก<br />
1. CH 3CH 2CH=CHCH 2CH 3 + O 3<br />
2. + O 3<br />
3.<br />
C CH 3<br />
CH 3<br />
H−C=C−H<br />
H OH<br />
Zn<br />
H 2O<br />
Zn<br />
H 2O<br />
R<br />
C<br />
H H<br />
O<br />
C<br />
Ketone<br />
H<br />
CH3−C−CH ll<br />
O H<br />
acetone<br />
38<br />
H 3C−C−H<br />
ll<br />
O<br />
acetaldehyde<br />
H<br />
H<br />
C=O + O=C<br />
H3C CH2CH3 1. O 3<br />
2. Zn, H 2O<br />
40<br />
42<br />
R<br />
7