HOW A MARKET ENTRY REWARD FOR ANTIBIOTICS WOULD WORK 55 Market Entry Rewards Global panel specifies the antibiotics we need Money in Global access with stewardship for antibiotics Money out Funders Product developers National purchasing arrangements for antimicrobials End users
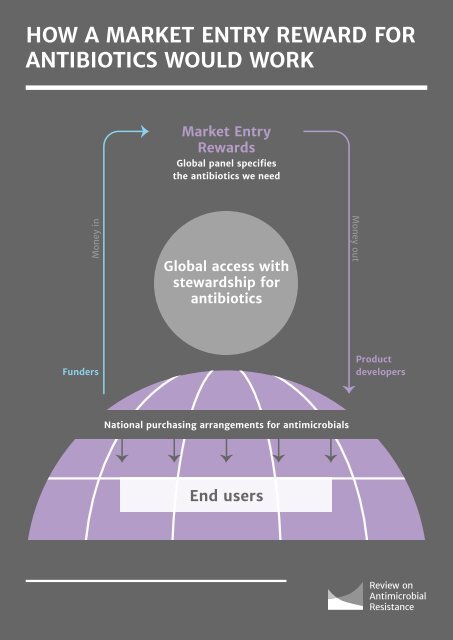
56 investment decisions by companies need to be influenced by this intervention, therefore funding commitments must be made over the long-term, without the risk associated with political cycles at country level. supporting professional and public education, and efforts to monitor use and resistance. The ability to ‘claw back’ all or part of a market entry reward in circumstances where there are egregious breaches of such conditions should be part of the award process. “Funding commitments must be made over the long‐term, without the risk associated with political cycles at country level. ” • Rewards should be linked to a product’s value to society. Drugs that meet the most acute unmet medical needs, most effectively, should be the most generously rewarded. At the same time, value criteria need to be transparently and objectively designed to provide certainty for product developers and global authorities alike. A points system, which addresses issues such as the level of unmet need, toxicity, efficacy, ability to counter resistance, etc., offers a promising basis for such an approach and ideas for implementing this system have started to emerge in the literature96. • The payment should come as soon after a product reaches market as possible, but this may not be immediate and may not come all at once. The high discount rate used by companies when calculating the current value of future payments means that this system will deliver better value for money the sooner after launch a reward is paid. However, some of the criteria for the points-based system described above may take some time to evaluate – meaning that a payment may only be made two or more years after a product reaches market, and may (in some instances) need to be staggered over a longer period. • There should be strings attached for recipients of the payouts. A key quid pro quo for receiving a lump sum payment should be a broad commitment to continued development post-approval and responsible sale and marketing of the product. Central to this would be commitments to ensuring global, affordable access to the product – either directly or through licensing arrangements such as the Medicines Patent Pool (MPP) – see box below. Such commitments could go further, for instance by barring recipient companies from giving out financial incentives to their own salesforce or clinicians, linked to the volume of the antibiotic sold. Recipients could also be asked to commit to 96 Rex JH, Outterson K, Antibiotic reimbursement in a model delinked from sales: a benchmark-based worldwide approach, The Lancet Infectious Diseases, 2016, 16, 500-505. • Leaving control in the hands of the developer brings significant advantages. From a public health purist’s perspective, a ‘buyout’ model, whereby a commercial antibiotic developer cedes control of their new product to a global public body, might be optimal for stewardship and access. However, such an approach brings with it significant delivery risks, not least as commercial operators possess substantial advantages over bureaucratic entities to oversee complex global pharmaceutical supply chains. • This should be administered at a global level. The pharmaceutical industry is global, with R&D conducted in all corners of the world and the demand for antibiotics coming from all regions. An intervention to deliver a stimulus to this market is therefore most efficient and effective if it is provided at a global level or by a critical mass of countries. We believe this would provide the best of both worlds, encouraging the private sector to innovate while ensuring research priorities are aligned to public need. Such an approach will help stimulate the market for antibiotics, ensuring that there is better commercial reward for antibiotic development without relying on high prices or large sales volumes. By paying for successful end products, rather than subsidising antibiotic R&D directly, the problems of governments or bureaucracies being asked to ‘pick winners’ are avoided. Judgement about the scientific merits of a product remain with developers, who are best-placed to make such decisions. Companies will properly make decisions not to advance projects which are scientifically unpromising, but will no longer cancel antibiotic programmes that show scientific potential but are not looking promising commercially based on projected volume of sales during patent life. “A key quid pro quo for receiving a lump sum payment should be a broad commitment to continued development post-approval and responsible sale and marketing of the product. Central to this would be commitments to ensuring global, affordable access to the product. ”