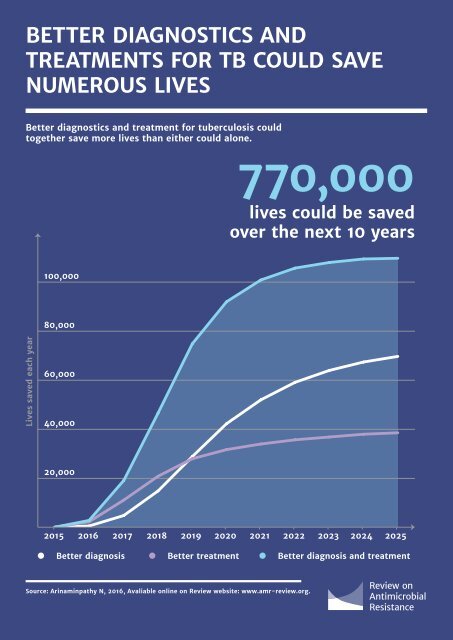
BETTER DIAGNOSTICS <strong>AND</strong> TREATMENTS FOR TB COULD SAVE NUMEROUS LIVES 61 Better diagnostics and treatment for tuberculosis could together save more lives than either could alone. 770,000 lives could be saved over the next 10 years 100,000 80,000 Lives saved each year 60,000 40,000 20,000 2015 2016 2017 2018 2019 2020 2021 2022 2023 2024 2025 Better diagnosis Better treatment Better diagnosis and treatment Source: Arinaminpathy N, 2016, Avaliable online on Review website: www.amr-review.org.
62 of a new diagnostic (with no new treatment regimen) has a less immediate impact, but has a greater impact on transmission rates and therefore a more significant effect than a new treatment alone over time. After six years, a new treatment by itself would reduce deaths from TB by 34,000 each year, whilst the introduction of just the new diagnostic would be saving 52,000 lives. This represents a decrease in MDR-TB deaths of 18 and 29 percent respectively. Most impressively, the results showed that the combined impact of introducing both a new diagnostic and a new treatment regimen would save 100,000 lives annually after six years, reducing death rates by 56 percent against their current trend. Over a decade, 770,000 lives would be saved by the combined interventions, underlining how significant a breakthrough the advent of new TB treatments and diagnostics could be. TB drug development represents particular challenges This modelling, and the rising burden of MDR-TB, together underline the pressing need for a strong pipeline of new treatments. This development pipeline is inextricably linked with that for antibiotics, as drugs for treating TB will by and large originate from the same processes by which antibiotics for use against other bacterial infections would be discovered. In common with wider antibiotic development efforts, the TB drug development field suffers from a prolonged period of disinvestment by commercial product developers, other than a few exceptions, leaving a perilously thin pipeline of products under development. There is a critical need to take action to reverse this and support the development of new and more effective TB treatment regimens, alongside better TB diagnostics and the continued search for new TB vaccines. Although the TB market shares some key characteristics with that for antibiotics, it represents a distinct challenge on account of two factors: 1. Products need to be developed as combinations. TB treatments must be delivered as combination therapies – usually using three or more antibiotics together – to prevent the development of resistance during prolonged treatment. The complex interaction of medicines means that the individual drugs making up these regimens should ideally be developed as combinations from early on during clinical testing, rather than once they are finished and licensed single products. This poses technical as well as commercial challenges. Furthermore, a new treatment should preferably be composed of multiple new products, rather than new products combined with those already in use which may hasten the development of resistance and offer limited value for the treatment of MDR-TB. 2. The low incomes of most TB sufferers limit commercial potential. Unlike conventional antibiotics, where the size of the market for new drugs is the key commercial challenge, the addressable market for a new TB regimen is huge. However, although not exclusively confined to low and middle-income countries, the greatest burden of TB falls on the poorest parts of the world, meaning that the principal purchasers of TB products will often be public or philanthropic donors. These features are specific to TB drugs and need to guide what interventions are chosen to stimulate the development of new TB treatments. More detailed thinking than this Review can offer needs to be done to design the detailed mechanisms. At a high level it is clear that TB drug development needs sustained ‘push’ funding especially for early clinical work and better market ‘pull mechanisms’ to incentivise developers. The world needs to sustain grant funding for TB research In the absence of an effectively functioning market for new TB products, much R&D in this space is given vital support by the work of non-profit product development partnerships such as the TB Alliance, Aeras and other organisations looking to develop a new TB vaccine. The Bill and Melinda Gates Foundation’s ‘TB accelerator’ is also becoming a critical tool to increase the number of promising molecules at an early stage of development. This type of funding needs to be sustained and increased where possible. We also need much better ‘pull’ incentives to reward the development of TB treatments In addition to traditional grant funding, there is a wide recognition that novel mechanisms are also needed to support the development of new TB treatments, including so-called ‘pull’ incentives that reward products for achieving market entry or important milestones along the drug development pipeline. Médecins Sans Frontières (MSF) in collaboration with other public health organisations, have developed a proposal referred to as the ‘3Ps’, for ‘Push, Pull, and Pool’. It uses a combination of milestone payments, R&D ‘push’ funding and pooling of intellectual property to overcome the key challenges of TB regimen development, by incentivising the early collaboration and open research needed to develop fixed dose combinations