The preparation of salicylic acid from phenol
The preparation of salicylic acid from phenol
The preparation of salicylic acid from phenol
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
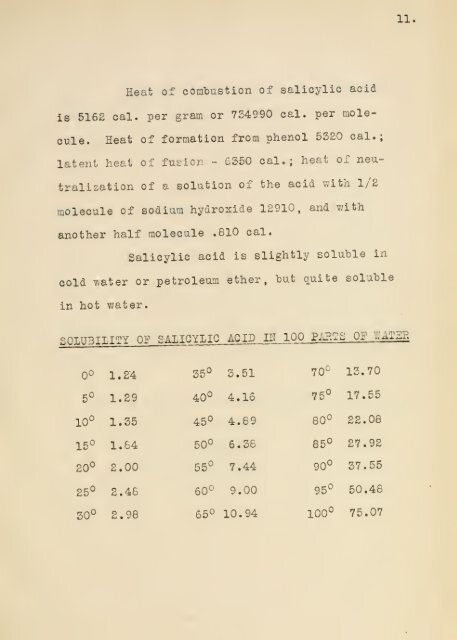
Heat <strong>of</strong> comTDustion <strong>of</strong> <strong>salicylic</strong> <strong>acid</strong><br />
is 5162 cal. per gram or 754990 cal. per mole-<br />
cule. Heat <strong>of</strong> formation <strong>from</strong> <strong>phenol</strong> 5520 cal.;<br />
latent heat <strong>of</strong> futic/. - 6550 cal.; heat <strong>of</strong> neu-<br />
tralisation <strong>of</strong> a solution <strong>of</strong> the <strong>acid</strong> with l/E<br />
molecule <strong>of</strong> sodium hydroxide 12910, and v.-ith<br />
another half molecule .810 cal.<br />
Salicylic <strong>acid</strong> is slightly soluble in<br />
cold vvater or petroleuoi ether, hut (luite soluble<br />
in hot water.<br />
0°<br />
11.