〈85〉 bacterial endotoxins test - US Pharmacopeial Convention
〈85〉 bacterial endotoxins test - US Pharmacopeial Convention
〈85〉 bacterial endotoxins test - US Pharmacopeial Convention
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Interim Revision Announcement<br />
4 〈85〉 Bacterial Endotoxins Test Official April 1, 2011<br />
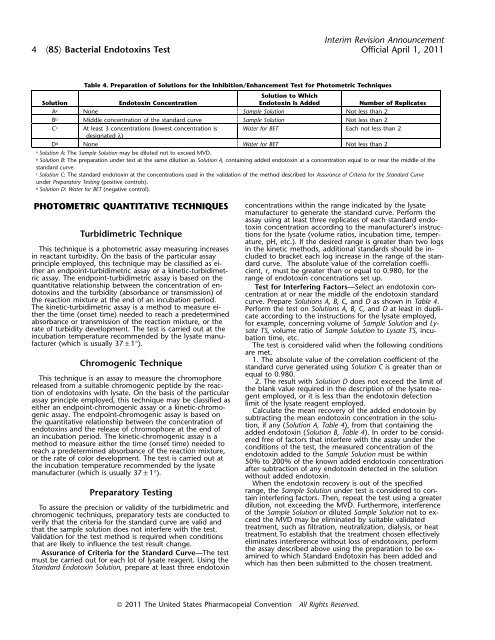
Table 4. Preparation of Solutions for the Inhibition/Enhancement Test for Photometric Techniques<br />
Solution to Which<br />
Solution Endotoxin Concentration Endotoxin Is Added Number of Replicates<br />
A a None Sample Solution Not less than 2<br />
B b Middle concentration of the standard curve Sample Solution Not less than 2<br />
C c At least 3 concentrations (lowest concentration is Water for BET Each not less than 2<br />
designated λ)<br />
D d None Water for BET Not less than 2<br />
a Solution A: The Sample Solution may be diluted not to exceed MVD.<br />
b Solution B: The preparation under <strong>test</strong> at the same dilution as Solution A, containing added endotoxin at a concentration equal to or near the middle of the<br />
standard curve.<br />
c Solution C: The standard endotoxin at the concentrations used in the validation of the method described for Assurance of Criteria for the Standard Curve<br />
under Preparatory Testing (positive controls).<br />
d Solution D: Water for BET (negative control).<br />
PHOTOMETRIC QUANTITATIVE TECHNIQUES<br />
Turbidimetric Technique<br />
concentrations within the range indicated by the lysate<br />
manufacturer to generate the standard curve. Perform the<br />
assay using at least three replicates of each standard endotoxin<br />
concentration according to the manufacturer’s instructions<br />
for the lysate (volume ratios, incubation time, temperature,<br />
pH, etc.). If the desired range is greater than two logs<br />
This technique is a photometric assay measuring increases in the kinetic methods, additional standards should be inin<br />
reactant turbidity. On the basis of the particular assay cluded to bracket each log increase in the range of the stanprinciple<br />
employed, this technique may be classified as ei- dard curve. The absolute value of the correlation coeffither<br />
an endpoint-turbidimetric assay or a kinetic-turbidimet- cient, r, must be greater than or equal to 0.980, for the<br />
ric assay. The endpoint-turbidimetric assay is based on the range of endotoxin concentrations set up.<br />
quantitative relationship between the concentration of <strong>endotoxins</strong><br />
and the turbidity (absorbance or transmission) of<br />
the reaction mixture at the end of an incubation period.<br />
The kinetic-turbidimetric assay is a method to measure either<br />
the time (onset time) needed to reach a predetermined<br />
absorbance or transmission of the reaction mixture, or the<br />
rate of turbidity development. The <strong>test</strong> is carried out at the<br />
incubation temperature recommended by the lysate manufacturer<br />
(which is usually 37 ± 1°).<br />
Test for Interfering Factors—Select an endotoxin con-<br />
centration at or near the middle of the endotoxin standard<br />
curve. Prepare Solutions A, B, C, and D as shown in Table 4.<br />
Perform the <strong>test</strong> on Solutions A, B, C, and D at least in dupli-<br />
cate according to the instructions for the lysate employed,<br />
for example, concerning volume of Sample Solution and Ly-<br />
sate TS, volume ratio of Sample Solution to Lysate TS, incu-<br />
bation time, etc.<br />
The <strong>test</strong> is considered valid when the following conditions<br />
are met.<br />
Chromogenic Technique<br />
1. The absolute value of the correlation coefficient of the<br />
standard curve generated using Solution C is greater than or<br />
This technique is an assay to measure the chromophore<br />
released from a suitable chromogenic peptide by the reaction<br />
of <strong>endotoxins</strong> with lysate. On the basis of the particular<br />
assay principle employed, this technique may be classified as<br />
either an endpoint-chromogenic assay or a kinetic-chromogenic<br />
assay. The endpoint-chromogenic assay is based on<br />
the quantitative relationship between the concentration of<br />
<strong>endotoxins</strong> and the release of chromophore at the end of<br />
an incubation period. The kinetic-chromogenic assay is a<br />
method to measure either the time (onset time) needed to<br />
reach a predetermined absorbance of the reaction mixture,<br />
or the rate of color development. The <strong>test</strong> is carried out at<br />
the incubation temperature recommended by the lysate<br />
manufacturer (which is usually 37 ± 1°).<br />
equal to 0.980.<br />
2. The result with Solution D does not exceed the limit of<br />
the blank value required in the description of the lysate rea-<br />
gent employed, or it is less than the endotoxin detection<br />
limit of the lysate reagent employed.<br />
Calculate the mean recovery of the added endotoxin by<br />
subtracting the mean endotoxin concentration in the solu-<br />
tion, if any (Solution A, Table 4), from that containing the<br />
added endotoxin (Solution B, Table 4). In order to be consid-<br />
ered free of factors that interfere with the assay under the<br />
conditions of the <strong>test</strong>, the measured concentration of the<br />
endotoxin added to the Sample Solution must be within<br />
50% to 200% of the known added endotoxin concentration<br />
after subtraction of any endotoxin detected in the solution<br />
without added endotoxin.<br />
When the endotoxin recovery is out of the specified<br />
Preparatory Testing<br />
range, the Sample Solution under <strong>test</strong> is considered to contain<br />
interfering factors. Then, repeat the <strong>test</strong> using a greater<br />
To assure the precision or validity of the turbidimetric and<br />
chromogenic techniques, preparatory <strong>test</strong>s are conducted to<br />
verify that the criteria for the standard curve are valid and<br />
that the sample solution does not interfere with the <strong>test</strong>.<br />
Validation for the <strong>test</strong> method is required when conditions<br />
that are likely to influence the <strong>test</strong> result change.<br />
Assurance of Criteria for the Standard Curve—The <strong>test</strong><br />
must be carried out for each lot of lysate reagent. Using the<br />
Standard Endotoxin Solution, prepare at least three endotoxin<br />
dilution, not exceeding the MVD. Furthermore, interference<br />
of the Sample Solution or diluted Sample Solution not to ex-<br />
ceed the MVD may be eliminated by suitable validated<br />
treatment, such as filtration, neutralization, dialysis, or heat<br />
treatment.To establish that the treatment chosen effectively<br />
eliminates interference without loss of <strong>endotoxins</strong>, perform<br />
the assay described above using the preparation to be examined<br />
to which Standard Endotoxin has been added and<br />
which has then been submitted to the chosen treatment.<br />
© 2011 The United States <strong>Pharmacopeial</strong> <strong>Convention</strong> All Rights Reserved.