- Page 1 and 2: New synthesis routes for production
- Page 3 and 4: This work reported here has been ca
- Page 5 and 6: Contents 1. Introduction and Object
- Page 7 and 8: Contents 5.2.6 Effect of H2O2/cyclo
- Page 9 and 10: Abbreviations Part from chemical sy
- Page 11 and 12: 1. Introduction and Objectives 1. I
- Page 13 and 14: 1. Introduction and Objectives econ
- Page 15 and 16: 2. General Part 2. General Part 2.1
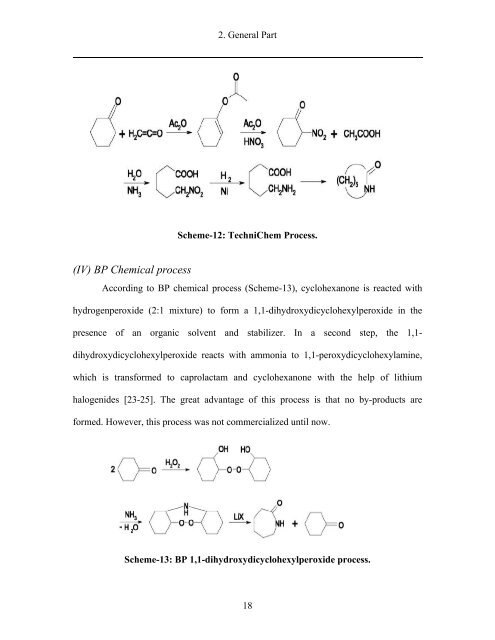
- Page 17 and 18: 2. General Part Scheme 1: Current c
- Page 19 and 20: Step 1: Step 2: Cyclohexanone 2 NO
- Page 21 and 22: 2. General Part alumina carrier. Th
- Page 23 and 24: +HNO3 -H 2O 2. General Part 13 NO 2
- Page 25 and 26: 2. General Part (NH4)2SO4 could be
- Page 27: (II) UCC Process 2. General Part Sc
- Page 31 and 32: 2. General Part selectivity for cap
- Page 33 and 34: 2. General Part polymeric species a
- Page 35 and 36: 2. General Part by oxidation with e
- Page 37 and 38: 2. General Part different research
- Page 39 and 40: 2. General Part The first step in t
- Page 41 and 42: 2. General Part cyclohexanone oxime
- Page 43 and 44: 2. General Part alkylbenzenes, prod
- Page 45 and 46: 2. General Part Figure -2: Possible
- Page 47 and 48: 2. General Part Figure-3: Possible
- Page 49 and 50: 3. Gas phase Beckmann rearrangement
- Page 51 and 52: 3.1. Catalysts characterization res
- Page 53 and 54: 3.1. Catalysts characterization res
- Page 55 and 56: 3.1.2 N2 Physisorption 3.1. Catalys
- Page 57 and 58: 3.1. Catalysts characterization res
- Page 59 and 60: 3.1. Catalysts characterization res
- Page 61 and 62: 3.1. Catalysts characterization res
- Page 63 and 64: 3.1. Catalysts characterization res
- Page 65 and 66: 3.1. Catalysts characterization res
- Page 67 and 68: 3.1. Catalysts characterization res
- Page 69 and 70: 3.1. Catalysts characterization res
- Page 71 and 72: 3.1. Catalysts characterization res
- Page 73 and 74: 3.1. Catalysts characterization res
- Page 75 and 76: 3.1. Catalysts characterization res
- Page 77 and 78: 3.1.9. Pyridine FT-IR 3.1. Catalyst
- Page 79 and 80:
3.1. Catalysts characterization res
- Page 81 and 82:
3.1. Catalysts characterization res
- Page 83 and 84:
3.1. Catalysts characterization res
- Page 85 and 86:
3.2. Catalytic results and discussi
- Page 87 and 88:
C.hexanone oxime conversion % 100 9
- Page 89 and 90:
Cyclohexanone oxime conversion % Cy
- Page 91 and 92:
3.2. Catalytic results and discussi
- Page 93 and 94:
Cyclohexanone oxime conversion % 10
- Page 95 and 96:
Conversion/ selectivity% 100 95 90
- Page 97 and 98:
Conversion/selectivity% 100 90 80 7
- Page 99 and 100:
Conversion/ selectivity (%) 100 80
- Page 101 and 102:
3.2. Catalytic results and discussi
- Page 103 and 104:
3.2. Catalytic results and discussi
- Page 105 and 106:
Uv/mg Weight loss % 0.2 0.1 0.0 -0.
- Page 107 and 108:
DESIGN-EXPERT Plot Actual Factors:
- Page 109 and 110:
3.2. Catalytic results and discussi
- Page 111 and 112:
DESIGN-EXPERT Plot Actual Factors:
- Page 113 and 114:
3.2. Catalytic results and discussi
- Page 115 and 116:
4. Summary and Outlook 4. Summary a
- Page 117 and 118:
4. Summary and Outlook indicated th
- Page 119 and 120:
4. Summary and Outlook loss of the
- Page 121 and 122:
5. Liquid phase ammoximation reacti
- Page 123 and 124:
5. Liquid phase ammoximation reacti
- Page 125 and 126:
5. Liquid phase ammoximation reacti
- Page 127 and 128:
5. Liquid phase ammoximation reacti
- Page 129 and 130:
% 5. Liquid phase ammoximation reac
- Page 131 and 132:
5. Liquid phase ammoximation reacti
- Page 133 and 134:
5. Liquid phase ammoximation reacti
- Page 135 and 136:
5. Liquid phase ammoximation reacti
- Page 137 and 138:
5. Liquid phase ammoximation reacti
- Page 139 and 140:
% 5. Liquid phase ammoximation reac
- Page 141 and 142:
5. Liquid phase ammoximation reacti
- Page 143 and 144:
5. Liquid phase ammoximation reacti
- Page 145 and 146:
% 5. Liquid phase ammoximation reac
- Page 147 and 148:
5. Liquid phase ammoximation reacti
- Page 149 and 150:
% 5. Liquid phase ammoximation reac
- Page 151 and 152:
5. Liquid phase ammoximation reacti
- Page 153 and 154:
5. Liquid phase ammoximation reacti
- Page 155 and 156:
6. Summary and Outlook 6. Summary a
- Page 157 and 158:
6. Summary and Outlook. oligomers a
- Page 159 and 160:
6. Summary and Outlook. Ammonia TPD
- Page 161 and 162:
6. Summary and Outlook. Reaction re
- Page 163 and 164:
7. Materials and Methods 7. Materia
- Page 165 and 166:
7. Materials and Methods 7.1.1 Hydr
- Page 167 and 168:
7. Materials and Methods 7.1.4 Hydr
- Page 169 and 170:
7. Materials and Methods mixed and
- Page 171 and 172:
7. Materials and Methods for the mo
- Page 173 and 174:
7. Materials and Methods equilibrat
- Page 175 and 176:
7. Materials and Methods NH3-TPD of
- Page 177 and 178:
7. Materials and Methods Carrier ga
- Page 179 and 180:
7. Materials and Methods 7.3.2 Ammo
- Page 181 and 182:
7. Materials and Methods Figure: 2-
- Page 183 and 184:
8. References 8. References [1] P.
- Page 185 and 186:
8. References [33] S. Sato, K. Urab
- Page 187 and 188:
8. References [66] S. Van Donk, A.
- Page 189 and 190:
8. References [98] C. Yan, J. Fraga
- Page 191 and 192:
8. References [134] J. M. R. Gallo,
- Page 193 and 194:
8. References [168] G. P. Heitmann,
- Page 195 and 196:
8. References [195] Beck, J.S., Var