SRK1 PHENYLATION OF NITRILE CARBANIONS J. Org. Chem., Vol. 38, No. 24, 1973 4157 TABLE I REACTIONS OF MONOSUBSTITUTED BENZENES WITH CYANOMETHYL ANIONS, PROVOKED Run no. Registry no. Substituent PhX, CHzCNK. mmol mmol K, mmol "3, ml BY POTASSIUM METAL IN LIQUID Temp, Ether, ------ Yield, %------- OC Methoda ml Toluene PAN' DPW e DPEd e CeHs Aniline 1 462-06-6 F 25 115 63 150 -33 Af 80 44 2 Trace 8 31 -1 2 F 14 76 40 110 -33 Ab' 65 30 3 0 7 3 44 h 3 F 40 123 107 250% -78 AB! 49 3 4 23 10 2 4 5 108-90-7 C1 c1 22 60 87 150 55 69 130 50 -33 -33 Ak A' 70 40 18 7 31 2 -2 12 5 20 17 -2 m 6 c1 64 160 95 50 -33 B 24 24 -3 9 17 m 7 8 c1 c1 63 165 75 165 110 330 50 50 -33 -78 B B 16 14 19 19 5 3 3 5 2 7 30 m 9 c1 67 80 81 50 -78 B 31 18 5 14 26 m 10 c1 30 88 37 (15)* 60 -78 B 27 12 2 40 8 11 C1 64 80 87 (34)' 60 (150) -78 AB 35 22 7 12 18 12 c1 146 49 254 (85)" 50 (250) -33 AB 46 3 3 7 22* lop 13 108-86-1 Br 27 106 69 150 -33 B 70 26 5 3 14 43 -1 14 591-50-4 I 25 95 64 150 -33 B 70 14 5 4 25 32 m 15 2510-86-3 OPO(0Et)z 50 80 90 (2Qr 50 -78 AB 43 3 1 7 11 -38 16 98-04-4 N(CHa)a-I- 63 78 85 50 -78 ABt 26 5 4 19 21 -7 AMMONIA a Methods: A, the monosubstituted benzene, neat or in solution, was slowly added to a solution <strong>of</strong> CH2CNK <strong>and</strong> K metal; AB, the monosubstituted benzene, neat or in solution, was slowly added to a solution <strong>of</strong> CHZCSK, <strong>and</strong> K metal was added slowly in small portions so as to keep K metal in more or less constant excess; B, to a solution <strong>of</strong> CHzCSK <strong>and</strong> the monosubstituted benzene, a small piece <strong>of</strong> K metal was added <strong>and</strong> then, after the blue color had vanished, another small piece, etc. PAN = phenylacetonitrile. DPM = diphenylmethane. DPE = IJ2-diphenylethane. 6 The yield calculation takes account <strong>of</strong> the fact that 2 mol <strong>of</strong> C&X are required to form 1 mol <strong>of</strong> DPM or DPE. f C&F in ether (5 ml) added during 30 min; quenched with CeHjCOO?u'a, then NHaC1. 0 CsH& in ether (40 ml) added. la Not sought. Sot distilled before use. 3 C&sF in SH3 (150 ml) added during 90 min; quenched with C6H;COONaJ then "$1. k CsH&l in ether (5 ml) added during 40 min. C6HsCl added as fast as possible (during 5 min); vigorous refluxing. m A tiny peak at the retention time for aniline. fi After addition <strong>of</strong> 37 mmol <strong>of</strong> K in especially small pieces, ice was added until the orange color faded, <strong>and</strong> then a further 15 mmol <strong>of</strong> K. 0 Half the C6H;Cl was added, then about 44 mmol <strong>of</strong> K, then the rest <strong>of</strong> the C&&l, then about 43 mmol <strong>of</strong> K, then 390 mmol <strong>of</strong> ice (causing the orange color to fade), then 34 mmol <strong>of</strong> K. p After addition <strong>of</strong> 254 mmol <strong>of</strong> K, 670 mmol <strong>of</strong> ice <strong>and</strong> 200 ml <strong>of</strong> ammonia were added, <strong>and</strong> then 85 mmol <strong>of</strong> K (with shaking <strong>of</strong> the thick slurry). 8 47, <strong>of</strong> phenol also obtained. Based on C&Cl. r After addition <strong>of</strong> 90 mmol <strong>of</strong> K, ice was added <strong>and</strong> then 25 mmol <strong>of</strong> K. K added in two portions, each followed by half the substrate. TSBLE 11 REACTIONS OF MONOSUBSTITUTED BENZENES WITH ANIONS FROM NITRILES OF STRUCTURE KCHzCS, PROVOKED BY POTASSIUM AIETAL IN LIQUID AMMONIA 7-- Yield, %--------- [RCH- Ph- Run no. R X <strong>of</strong> CeH6X Registry no. CN--l, [C6H5X1, IM 1M K, M' Temp, Ph- PhCH- 'C Methoda CHzR RCN Phz- ,--CHCRHRPhC- CHR Meso Rae CsH6 Aniline 17 CHa c1 42117-12-4 1.38 0.87 2.0 (0.65) -78 -4B 22 d d 5 5 3 4 5 18 CHa c1 1.40 2.80 3.98 -33 AB 34 6 7 6 6 3Se loe,' 19 20 CHs CHa OPO(0Et)r N( CHa) 3 'I - 1.16 1.07 0.73 0.67 1.7 (0.1)v 1.26 -78 -78 AB ABi 14 8 d d d d 7 8 7 8 3 35 6 5h 3 21 CtHs F 42117-13-5 1.28 0.43 1.09 -78 ABi 28 4 d 13 13 35 13 22 CiHa c1 1.37 4.1 6.04 -33 ABk 37 0.5 13 278 3' 23 C2H5 I 0.30 0.16 0.20 -33 A 6 29 2 12 12 27 1 24 CzHs OPO(0Et)z 1.04 0.15 0.30 -33 B 28 4 0.2 1 1 20 n 25 (CH3)Z0 Br 42117-14-6 0.98 2.94 5.3 -33 AB 27 31 7 26 n-CsHi C1 42117-15-7 1.09 3.30 6.3 (1.7) -33 AB 56 d m m m 38' 14' 27 n-CaH7 OPO(0Et)Z 1.58 0.60 1.45 (0.2) -33 BAP 38 10 d d d 2 7 q 28 (CHa)zCH C1 42117-16-8 1.64 3.2 4.9 -33 AB 37 19' 12 d d 31e -5' 29 (CHdzCH OPO(0Et)z 1.80 0.72 1.48 -78 B 20 7 36 -2' 30 (CHa)zCH OPO(0Et)z 1.30 0.52 0.85 (0.23) -33 BAP 31 10 d d d 3 2 d 31 (CHdzCH OPO(0Et)z 0.63 0.20 0.70 -78 ABi 22 15 d d d 4 1 2 32 CsHs OPO(0Et)z 18802-89-6 1.44 1.12 1.47 -78 AB 12 3t 6 25 1" 33 C6H5 N(CHs)a+I- 1.50 1.19 2.08 -78 AB 17 12 43 18 a See footnote a, Table 1. Concentration that would have prevailed had there been no reaction; the K indicated in parentheses was added after ice. The glpc peaks for racemic <strong>and</strong> meso stereoisomers were not fully resolved, but were approximately equal in area; the sum <strong>of</strong> the two could be measured accurately, <strong>and</strong> was arbitrarily allocated in equal parts to meso <strong>and</strong> racemic. A tiny peak at the expected retention time. e Based on CaH6Cl. f 4% <strong>of</strong> CsHsC1 recovered. b' The K indicated in parentheses was added after ice <strong>and</strong> 1.50 ml <strong>of</strong> ether. Phenol (2%) also formed. Excess K present. at the end was destroyed by addition <strong>of</strong> ice. j The CaF& in I50 ml <strong>of</strong> "I, was added during 90 min. After reaction, SH3 was added to increase the volume from ca. 50 ml to 200 ml, followed by 10 g <strong>of</strong> ice <strong>and</strong> 75 mmol <strong>of</strong> K. 12% <strong>of</strong> CsHJ recovered. m Detected qualitatively. fi Phenol (29%) <strong>and</strong> some 4-amino-3-cyano-3heptene also formed. Isobutyronitrile. p Method BA: the C&jX <strong>and</strong> K metal were both added in portions, but so as always to keep Cs&X in excess. Phenol (8%) also formed. I^ After decyanation <strong>of</strong> the isolated product mixture, 507, <strong>of</strong> isobutylbenzene <strong>and</strong> a mere trace <strong>of</strong> 2-phenyl-3-methylbutanonitrile were obtained. Phenol (6%) also formed. Identification solely by glpc retention time. Phenol (12%) also formed; 80% <strong>of</strong> PAS recovered. 76% <strong>of</strong> PAS recovered. the variation is <strong>of</strong>ten diEcult to correlate with differ- ences in reaction participants or conditions. Alkyl- benzenes, PhCH2R, corresponding to toluene in Table I, are prominent products <strong>of</strong> most runs, as is benzene. Phenyl derivatives <strong>of</strong> the starting nitriles are formed in significant amounts in some runs but only in trace amounts in others. l,l-Diphenylalkanes, Ph&HR, appear to a significant extent in only a few runs, most <strong>of</strong> which involve the aromatic substrate in excess. Dimeric products, meso <strong>and</strong> racemic PhCHRCHRPh, are formed in amounts totalling as high as 26% in reactions <strong>of</strong> the anions from propio- <strong>and</strong> butyronitrile, but only in minor amounts in the other runs. The yields <strong>of</strong> aniline are variable, but never large. Photostimulated <strong>Reactions</strong>. -Inasmuch as SRN~ reactions <strong>of</strong> halobenzenes with acetone enolate ion
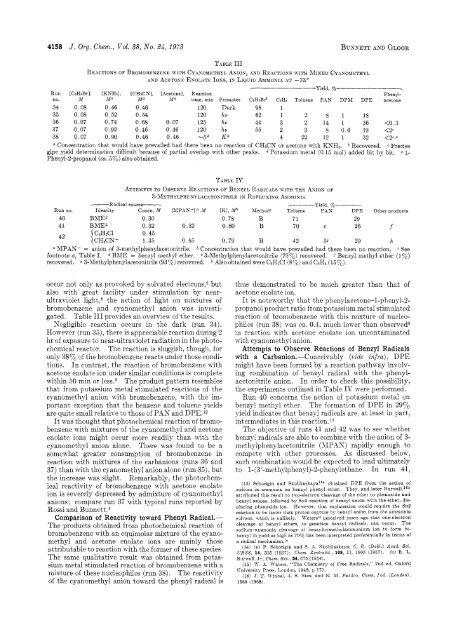
4158 J. Org. Chem., Vol. 58, No. 24, 1975 BUNNETT AND GLOOR TABLE 111 REACTIONS OF BROMOBENZENE WITH CYASOMETHYL ANIOS, AND REACTIONS WITH MIXED CYANOMETHYL AND ACETOSE ENOLATE IONS, IS LIQUID AMMOSIA AT -33" '---------- Yield, yo --. Run no. [CsHsBr], M [KNH21, .Ma [CHaCNI, Ma [Acetonel, .IfQ Reaction time, min Promoter CsHsBrb C6He Toluene PAN DPM DPE Phenylacetone 34 0.08 0.46 0.46 120 Dark 98 1 35 0.08 0.52 0.54 120 hv 62 1 2 8 1 18 36 0.07 0.74 0.68 0.07 125 hv 44 3 2 14 1 36