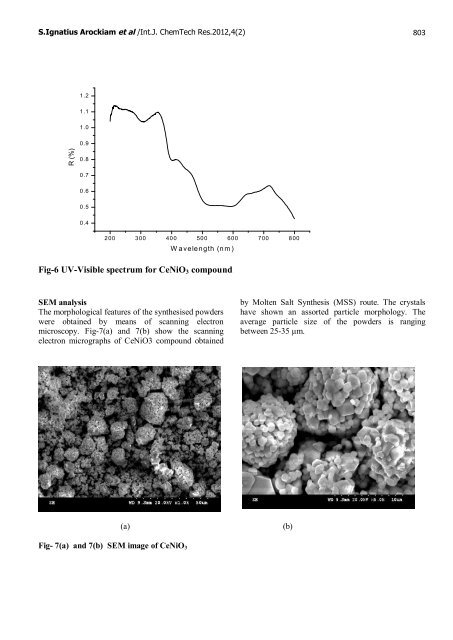
S.Ignatius Arockiam et al /Int.J. ChemTech Res.2012,4(2) 803 R (%) 1.2 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 200 300 400 500 600 700 800 Wavelength (nm) Fig-6 UV-Visible spectrum for CeNiO3 compound SEM analysis The morphological features <strong>of</strong> the synthesised powders were obtained by means <strong>of</strong> scanning electron microscopy. Fig-7(a) <strong>and</strong> 7(b) show the scanning electron micrographs <strong>of</strong> CeNiO3 compound obtained Fig- 7(a) <strong>and</strong> 7(b) SEM image <strong>of</strong> CeNiO3 (a) (b) by Molten Salt <strong>Synthesis</strong> (MSS) route. The crystals have shown an assorted particle morphology. The average particle size <strong>of</strong> the powders is ranging between 25-35 μm.
S.Ignatius Arockiam et al /Int.J. ChemTech Res.2012,4(2) 804 Conclusions Fine <strong>crystalline</strong> CeNiO3 powders are successfully synthesized using low temperature molten salt technique. The XRD analysis confirms that the compound has the cubic structure. FTIR spectrum reveals the Ce-O, Ni-O b<strong>and</strong> positions in CeNiO3 compound. CHNS analysis shows the compound has minor impurities such as carbon, hydrogen <strong>and</strong> sulphur. From the UV reflectance spectrum, the b<strong>and</strong> References [1] F. Damay, N. Nguyen, A. Maignan, M. Hervieu, B. Raveau,Solid State Commun. 98 (1996) 997. [2] Th. Lagouri, Sp. Dedoussis, M. Chardalas, <strong>and</strong> A. Liolios, Phys. Lett. A, 229 (1997) 259 [3] Junmo Koo, Jae Hyeok Jang, <strong>and</strong> Byeong- Soo Bae J. Am. Ceram. Soc.84 (2001) 193. [4] M. Panneerselvam <strong>and</strong> K. J. Rao J. Mater. Chem. 13 (2003) 596. [5] C. Suddhakar <strong>and</strong> M. A. Vannice, Appl. Catal., 14, (1985), 47. [6] C.Suddhakar <strong>and</strong>M. A.Vannice,J.Catal.,95,(1985), 227. [7] M. Mendelovici <strong>and</strong> M. Steinberg, J. Catal., 96, (1985), 285. [8] C. Padeste, N. W. Cant <strong>and</strong> D. L. Trimm, Catal. Lett., 18, (1993), 305. [9] H. Diaz, J. P. Marcq, M. Pinabiau <strong>and</strong> Y. Barbaux, Eur. Pat. 8,508, (1985), 210. [10] M. G. Sanchez <strong>and</strong> J. L. Gazquez, J. Catal., 104, (1987), 120. [11] H. Wan, Z. Chao, W. Weng, X. Zhou, J. Cai <strong>and</strong> K. Tsai, Catal.Today, 30, (1996), 67. [12] J. M. Herrmann, C. Hoang-Van, L. Dibansa <strong>and</strong> R. Hari-vololonola, J. Catal., 159, (1996), 361. [13] C. De Leitenburg, A. Trovarelli, J. Llorca, F. Cavani <strong>and</strong> G. Bini,Appl. Catal., 139, (1996) 161 [14] S. Bae, K. S. Han <strong>and</strong> J. H. Adair, J. Mater. Chem., 12, (2002), 3117 [15]R. Alexendrescu, I. Morjan, F. Dumitrache, M. Scarisoreanu, I. Soare, C. Fleaca, R. Birjega, E. Popovici, L. Gavrila, G. Prodan, V. Ciupina, G. Filoti, V. Kuncser <strong>and</strong> L.Vekas, Int. J. Photoenergy, (2008),11 [16]O. Michitaka <strong>and</strong> M. Shusaku, Nippon Kagakkai Koen Yokoshu, 83(1), 378 (2008). ***** gap value is determined <strong>and</strong> found to be eg = 3.18 eV. The EPR spectrum reveals that the value <strong>of</strong> g factor is g=2. The SEM image reveals that the particles have assorted crystal morphology. The average particle size is found to be 20 - 35 μm. From the above studies, it has been concluded that the fine <strong>crystalline</strong> cerium nickelate compound can be synthesized by low temperature molten salt technique. [17]. J. Tao, J. Ma, Y. Wang, X. Zhu, J. Liu, X. Jiang, B. Lin <strong>and</strong> Y. Ren, J. Amer. Cer.Soc., 89(II), (2008),3554 [18] Siegel et al.,United States Patent, No. 5, 128, 081. [19]. Peterson et al., United States Patent No. 6, 580, 051. [20]Y. Mao, T.-J Park, F. Zhang, H. Zhou, S. S. Wong, Small 3 (2007) 1122. [21] K. H Yoon, C. Y. S, D. H. Kang, J. Mater. Sci. 33 (1998) 2977. [22] Yuanbing Mao, Xia Guo, Jian Y. Huang, Kang L. Wang, <strong>and</strong> Jane P. Chang, J. Phys. Chem.C 113 (2009) 1204. [23] B. L Cushing, V. L Kolesnichenko, C.O’Connor, J. Chem. Rev. 104 (2004) 3893. [24] Jansen,M.Angew. Chem., Int. Ed. 41 (2002) 3746. [25] X Weng, P. Boldrin, I. Abrahams, S. J Skinner, Darr, J. A.Chem. Mater. 19 (2007) 4382. [26] M. Helan,L.John Berchmans, journal <strong>of</strong> rare earths, 28 (2010) 255 [27]M. Helan, L. J. Berchmans, V. S. Syamala Kumari, R. RaviSankar <strong>and</strong> V. M.Shanmugam Materials Research Innovations 15, 2(2011)130 [28] M. Helan, L. John Berchmans, Timy P. Joseb, A.Visuvasam, S. Angappan Materials Chemistry <strong>and</strong> Physics 124 (2010) 439–442 [29]S.Ignatius Arockiam, L. John Berchmans, S.Angappan, A. Visuvasam,V. Mani Materials Science Forum 699 (2012) 67-78 [30] Z-R Silva, J.D.G Fern<strong>and</strong>es, D.M.A. Melo, C. Alves, Jr. E.R. Leite, C.A. Paskocimas, E.Longo, M.I.B. Bernardi, Mater. lett. 56 (2002) 232. [31]J.D.G. Fern<strong>and</strong>es, D.M.A. Melo, L.B. Zinner, C.M. Salustiano, Z.R. Silva, A.E. Martinelli,M. Cerqueira, C. Alves Ju´nior, E. Longo, M.I. B.Bernardi, Mater. Lett. 53 (2002) 122