Recommended Liquid–Liquid Equilibrium Data. Part 4. 1-Alkanol ...
Recommended Liquid–Liquid Equilibrium Data. Part 4. 1-Alkanol ...
Recommended Liquid–Liquid Equilibrium Data. Part 4. 1-Alkanol ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
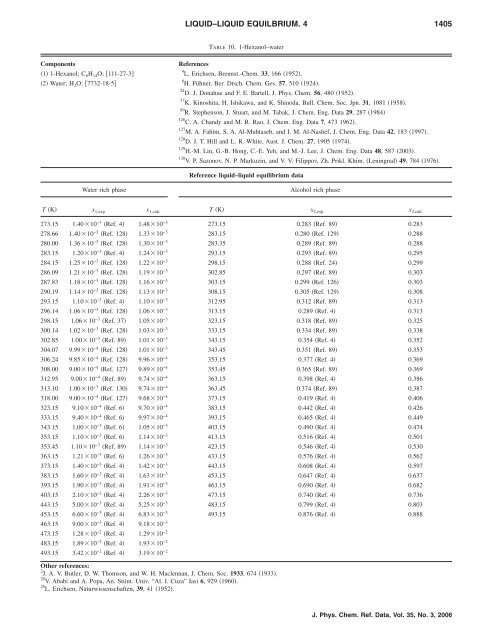
TABLE 10. 1-Hexanol–water<br />
Components References<br />
1 1-Hexanol; C6H14O; 111-27-3<br />
4<br />
L. Erichsen, Brennst.-Chem. 33, 166 1952.<br />
2 Water; H2O; 7732-18-5<br />
6<br />
H. Fühner, Ber. Dtsch. Chem. Ges. 57, 510 192<strong>4.</strong><br />
24<br />
D. J. Donahue and F. E. Bartell, J. Phys. Chem. 56, 480 1952.<br />
37 K. Kinoshita, H. Ishikawa, and K. Shinoda, Bull. Chem. Soc. Jpn. 31, 10811958.<br />
89 R. Stephenson, J. Stuart, and M. Tabak, J. Chem. Eng. <strong>Data</strong> 29, 287 1984<br />
126 C. A. Chandy and M. R. Rao, J. Chem. Eng. <strong>Data</strong> 7, 473 1962.<br />
127 M. A. Fahim, S. A. Al-Muhtaseb, and I. M. Al-Nashef, J. Chem. Eng. <strong>Data</strong> 42, 1831997.<br />
128 D. J. T. Hill and L. R. White, Aust. J. Chem. 27, 1905 197<strong>4.</strong><br />
129 H.-M. Lin, G.-B. Hong, C.-E. Yeh, and M.-J. Lee, J. Chem. Eng. <strong>Data</strong> 48, 587 2003.<br />
130 V. P. Sazonov, N. P. Markuzin, and V. V. Filippov, Zh. Prikl. Khim. Leningrad 49, 784 1976.<br />
Reference liquid–liquid equilibrium data<br />
Water rich phase Alcohol rich phase<br />
T K x 1,exp x 1,calc T K x 2,exp x 2,calc<br />
273.15 1.4010−3 Ref. 4 1.4810−3 273.15 0.283 Ref. 89 0.283<br />
278.66 1.4010−3 Ref. 128 1.3310−3 283.15 0.280 Ref. 129 0.288<br />
280.00 1.3610−3 Ref. 128 1.3010−3 283.35 0.289 Ref. 89 0.288<br />
283.15 1.2010−3 Ref. 4 1.2410−3 293.15 0.293 Ref. 89 0.295<br />
28<strong>4.</strong>15 1.2510−3 Ref. 128 1.2210−3 298.15 0.288 Ref. 24 0.299<br />
286.09 1.2110−3 Ref. 128 1.1910−3 302.85 0.297 Ref. 89 0.303<br />
287.83 1.1810−3 Ref. 128 1.1610−3 303.15 0.299 Ref. 126 0.303<br />
290.19 1.1410−3 Ref. 128 1.1310−3 308.15 0.305 Ref. 129 0.308<br />
293.15 1.1010−3 Ref. 4 1.1010−3 312.95 0.312 Ref. 89 0.313<br />
296.14 1.0610−3 Ref. 128 1.0610−3 313.15 0.289 Ref. 4 0.313<br />
298.15 1.0610−3 Ref. 37 1.0510−3 323.15 0.318 Ref. 89 0.325<br />
300.14 1.0210−3 Ref. 128 1.0310−3 333.15 0.334 Ref. 89 0.338<br />
302.85 1.0010−3 Ref. 89 1.0110−3 343.15 0.354 Ref. 4 0.352<br />
30<strong>4.</strong>07 9.9910−4 Ref. 128 1.0110−3 343.45 0.351 Ref. 89 0.353<br />
306.24 9.8510−4 Ref. 128 9.9610−4 353.15 0.377 Ref. 4 0.369<br />
308.00 9.0010−4 Ref. 127 9.8910−4 353.45 0.365 Ref. 89 0.369<br />
312.95 9.0010−4 Ref. 89 9.7410−4 363.15 0.398 Ref. 4 0.386<br />
313.10 1.0010−3 Ref. 130 9.7410−4 363.45 0.374 Ref. 89 0.387<br />
318.00 9.0010−4 Ref. 127 9.6810−4 373.15 0.419 Ref. 4 0.406<br />
323.15 9.1010−4 Ref. 6 9.7010−4 383.15 0.442 Ref. 4 0.426<br />
333.15 9.4010−4 Ref. 6 9.9710−4 393.15 0.465 Ref. 4 0.449<br />
343.15 1.0010−3 Ref. 6 1.0510−3 403.15 0.490 Ref. 4 0.474<br />
353.15 1.1010−3 Ref. 6 1.1410−3 413.15 0.516 Ref. 4 0.501<br />
353.45 1.1010−3 Ref. 89 1.1410−3 423.15 0.546 Ref. 4 0.530<br />
363.15 1.2110−3 Ref. 6 1.2610−3 433.15 0.576 Ref. 4 0.562<br />
373.15 1.4010−3 Ref. 4 1.4210−3 443.15 0.608 Ref. 4 0.597<br />
383.15 1.6010−3 Ref. 4 1.6310−3 453.15 0.647 Ref. 4 0.637<br />
393.15 1.9010−3 Ref. 4 1.9110−3 463.15 0.690 Ref. 4 0.682<br />
403.15 2.1010−3 Ref. 4 2.2610−3 473.15 0.740 Ref. 4 0.736<br />
443.15 5.0010−3 Ref. 4 5.2510−3 483.15 0.799 Ref. 4 0.803<br />
453.15 6.6010−3 Ref. 4 6.8310−3 493.15 0.876 Ref. 4 0.888<br />
463.15 9.0010−3 Ref. 4 9.1810−3 473.15 1.2810 −2 Ref. 4 1.2910 −2<br />
483.15 1.8910 −2 Ref. 4 1.9310 −2<br />
493.15 3.4210 −2 Ref. 4 3.1910 −2<br />
Other references:<br />
2 J. A. V. Butler, D. W. Thomson, and W. H. Maclennan, J. Chem. Soc. 1933, 674 1933.<br />
20 V. Ababi and A. Popa, An. Stiint. Univ. “Al. I. Cuza” Iasi 6, 929 1960.<br />
26 L. Erichsen, Naturwissenschaften, 39, 411952.<br />
LIQUID–LIQUID EQUILBRIUM. 4 1405<br />
J. Phys. Chem. Ref. <strong>Data</strong>, Vol. 35, No. 3, 2006