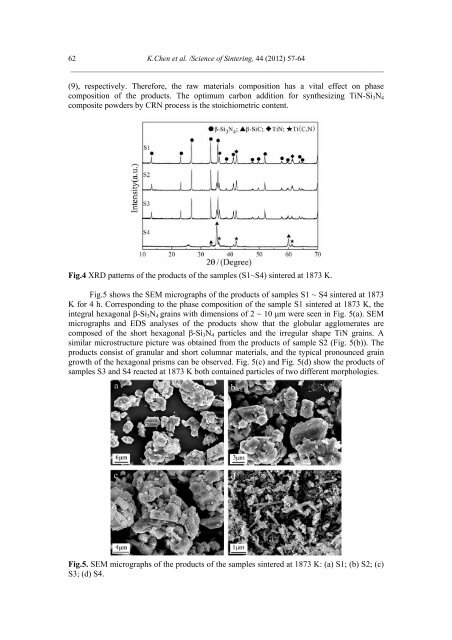
62 K.Chen et al. /Science <strong>of</strong> Sintering, 44 (2012) 57-64 ___________________________________________________________________________ (9), respectively. Therefore, the raw materials composition has a vital effect on phase composition <strong>of</strong> the products. The optimum carbon addition for synthesizing <strong>TiN</strong>-Si 3 N 4 composite powders by CRN process is the stoichiometric content. Fig.4 XRD patterns <strong>of</strong> the products <strong>of</strong> the samples (S1~S4) sintered at 1873 K. Fig.5 shows the SEM micrographs <strong>of</strong> the products <strong>of</strong> samples S1 ~ S4 sintered at 1873 K for 4 h. Corresponding to the phase composition <strong>of</strong> the sample S1 sintered at 1873 K, the integral hexagonal β-Si 3 N 4 grains with dimensions <strong>of</strong> 2 ~ 10 μm were seen in Fig. 5(a). SEM micrographs <strong>and</strong> EDS analyses <strong>of</strong> the products show that the globular agglomerates are composed <strong>of</strong> the short hexagonal β-Si 3 N 4 particles <strong>and</strong> the irregular shape <strong>TiN</strong> grains. A similar microstructure picture was obtained <strong>from</strong> the products <strong>of</strong> sample S2 (Fig. 5(b)). The products consist <strong>of</strong> granular <strong>and</strong> short columnar materials, <strong>and</strong> the typical pronounced grain growth <strong>of</strong> the hexagonal prisms can be observed. Fig. 5(c) <strong>and</strong> Fig. 5(d) show the products <strong>of</strong> samples S3 <strong>and</strong> S4 reacted at 1873 K both contained particles <strong>of</strong> two different morphologies. Fig.5. SEM micrographs <strong>of</strong> the products <strong>of</strong> the samples sintered at 1873 K: (a) S1; (b) S2; (c) S3; (d) S4.
K. Chen et al./Science <strong>of</strong> Sintering, 44 (2012) 57-64 63 ___________________________________________________________________________ The major part <strong>of</strong> the products had rod-like structure β-SiC <strong>of</strong> 0.1 ~ 0.4 μm in diameter <strong>and</strong> 2 ~ 10 μm in length, <strong>and</strong> the remainder consisted <strong>of</strong> granular agglomerates with dimensions <strong>of</strong> 0.1 ~ 1 μm. The formation <strong>and</strong> growth <strong>of</strong> rod-like β-SiC can be explained in term <strong>of</strong> the vapor-solid (VS) mechanism [20]. It also can be found that more rod-like grains were obtained in the product <strong>of</strong> sample S4 than sample S3. This indicated that the increase <strong>of</strong> carbon addition promoted the formation <strong>of</strong> β-SiC. 4. Conclusion A low cost preparation method <strong>of</strong> the <strong>TiN</strong>-Si 3 N 4 composite powders were obtained <strong>from</strong> rutile <strong>and</strong> quartz. The optimum parameters for synthesizing <strong>TiN</strong>-Si 3 N 4 by CRN process are carbon addition <strong>of</strong> stoichiometric content, temperature <strong>of</strong> 1873 K for 4 h <strong>and</strong> nitrogen pressure <strong>of</strong> 0.2 MPa. The produced <strong>TiN</strong>-Si 3 N 4 in this experiment exist in granular <strong>and</strong> hexagonal columnar shape, <strong>and</strong> the average particle size <strong>of</strong> the synthesized powders is 2 ~ 10 μm. Acknowledgements The authors greatly appreciate the National Natural Science Foundation <strong>of</strong> China for financial support (Grant Nos. 51032007 <strong>and</strong> 50972134). We also thank the Fundamental Research Funds for the Central Universities (Grant Nos. 2010ZD12 <strong>and</strong> 2011PY0174) <strong>and</strong> the New Star Technology Plan <strong>of</strong> Beijing (Grant No. 2007A080). Z.H. Huang also thanks the Gledden Visiting Senior Fellowship <strong>from</strong> University <strong>of</strong> Western Australia in 2010. 5. References 1. 1. F. L. Riley, J. Am. Ceram. Soc., 83 (2000) 245-265. 2. H. Klemm, J. Am. Ceram. Soc., 93 (2010) 1501-1522. 3. T. Yano, J. Tatami, K. Komeya, T. Meguro, J. Ceram. Soc. Jpn., 109 (2001) 396- 400. 4. J.L. Huang, M.T. Lee, H.H. Lu, D.F. Li, Mater. Chem. Phys., 45 (1996) 203-210. 5. C.Y. Tian, H. Jiang, N. Liu, Int. J. Refract. Met. H., 29 (2011) 14-20. 6. L. Gao, J.G. Li, T. Kusunose, K. Niihara, J. Eur. Ceram. Soc., 24 (2004) 381-386. 7. T. Hirai, S. Hayashi, J. Mater. Sci., 17 (1982) 1320-1328. 8. Z.M. Zheng, Y.M. Li, Y.M. Luo, S.X. Su, Z.J. Zhang, S.Y. Yang, W. Gao, Z.M. Xie, J. Appl. Polym. Sci., 92 (2004) 2733-2739. 9. Y.M. Luo, Z.M. Zheng, C.H. Xu, X.N. Mei, Ceram. Int., 35 (2009) 1301-1303. 10. W.B. He, B.L. Zhang, H.R. Zhuang, W.L. Li, Ceram. Int., 30 (2004) 2211-2214. 11. G. Blugan, K.V. Manukyan, S.L. Kharatyan, J. Kuebler, Ceram. Int., 33 (2007) 379-383. 12. C.U. Bang, Y.C. Hong, H.S. Uhm, Surf. Coat Tech., 201 (2007) 5007-5011. 13. D.P. Xiang, Y. Liu, M.J. Tu, Y.Y. Li, W.P. Chen, Int. J. Refract. Met. H., 27 (2009) 111-114. 14. S.L. Chung, C.W. Chang, J. Mater. Sci., 44 (2009) 3784-3792. 15. D.P. Xiang, Y. Liu, S.J. Gao, M.J. Tu, Mater. Charact., 59 (2008) 241-244. 16. G.A. Swift, R. Koc, J. Mater. Sci., 34 (1999) 3083-3093. 17. L.M. Berger, W. Gruner, E. Langholf, S. Stolle, Int. J. Refract. Met. H., 17 (1999) 235-243.