E - Glenmark
E - Glenmark
E - Glenmark
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
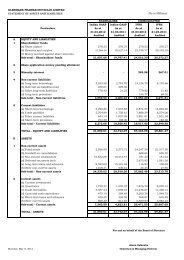
Active Pharmaceutical Ingredients<br />
Status in FY 2006<br />
Plan going forward…<br />
Revenue decline (12%) driven by:<br />
capacity diversion for captive APIs<br />
across US, India and LRM<br />
decline in prices in some key APIs<br />
Revenue growth in excess of 30%<br />
Complete capacity expansion in<br />
Ankleshwar, start construction in<br />
Aurangabad<br />
discontinuation of Valdecoxib<br />
File 12-14 DMFs per year<br />
Mix of revenues from segments:<br />
India and co-marketing – 48%<br />
Regulated APIs – 22%<br />
Commercial supplies to regulated<br />
markets to increase dramatically with<br />
several ANDA’s/Dossiers filed and<br />
pending from out 3rd party customers<br />
Rest of world – 30%<br />
Commenced commercial supplies of<br />
DMFs during year; several samples,<br />
validation, pivotal batches to global<br />
generic players for their ANDA/Dossier<br />
filings<br />
Develop several APIs annually to<br />
provide cost and timing advantage to<br />
complement our US and EU generic<br />
filings<br />
Continue to be a preferred 3rd party API<br />
supplier to the generic industry<br />
Filed 7 DMFs in the year<br />
Capacity de-bottlenecking initiated for<br />
Ankleshwar, commenced preconstruction<br />
work at new site<br />
Aurangabad





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)




![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

