Industry guideline for the Compliance of Paper & Board ... - cepi
Industry guideline for the Compliance of Paper & Board ... - cepi
Industry guideline for the Compliance of Paper & Board ... - cepi
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
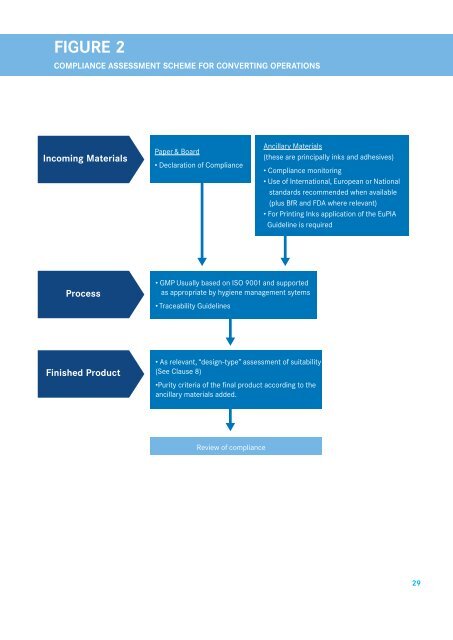
Figure 2<br />
<strong>Compliance</strong> Assessment Scheme <strong>for</strong> converting operations<br />
Incoming Materials<br />
<strong>Paper</strong> & <strong>Board</strong><br />
• Declaration <strong>of</strong> <strong>Compliance</strong><br />
Ancillary Materials<br />
(<strong>the</strong>se are principally inks and adhesives)<br />
• <strong>Compliance</strong> monitoring<br />
• Use <strong>of</strong> International, European or National<br />
standards recommended when available<br />
(plus BfR and FDA where relevant)<br />
• For Printing Inks application <strong>of</strong> <strong>the</strong> EuPIA<br />
Guideline is required<br />
Process<br />
• GMP Usually based on ISO 9001 and supported<br />
as appropriate by hygiene management sytems<br />
• Traceability Guidelines<br />
Finished Product<br />
• As relevant, “design-type” assessment <strong>of</strong> suitability<br />
(See Clause 8)<br />
•Purity criteria <strong>of</strong> <strong>the</strong> final product according to <strong>the</strong><br />
ancillary materials added.<br />
Review <strong>of</strong> compliance<br />
29