8. Acids, Bases and Salts (41.1 MB)
8. Acids, Bases and Salts (41.1 MB)
8. Acids, Bases and Salts (41.1 MB)
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Acids</strong>, <strong>Bases</strong> <strong>and</strong> <strong>Salts</strong><br />
The temperature is not allowed to rise beyond 100°C otherwise whole of water of<br />
crystallization is lost <strong>and</strong> anhydrous calcium sulphate is produced which is called<br />
‘dead burnt’ as it does not have the property to set after mixing with water.<br />
(b) Uses<br />
1. In making casts for manufacture of toys <strong>and</strong> statues.<br />
2. In medicine for making plaster casts to hold fractured bones in place while they<br />
set. It is also used for making casts in dentistry.<br />
3. For making the surface of walls <strong>and</strong> ceiling smooth.<br />
4. For making decorative designs on ceilings, walls <strong>and</strong> pillars.<br />
5. For making‘ chalk’ for writing on blackboard.<br />
6. For making fire proof materials.<br />
MODULE - 2<br />
Matter in our Surroundings<br />
Notes<br />
<strong>8.</strong>6.4 Bleaching Powder<br />
Have you ever wondered at the whiteness of a new white cloth? How is it made<br />
so white? It is done by bleaching of the cloth at the time of its manufacture. Bleaching<br />
is a process of removing colour from a cloth to make it whiter. Bleaching powder<br />
has been used for this purpose since long. Chemically, it is calcium oxychloride,<br />
CaOCl 2 .<br />
(a) Manufacture<br />
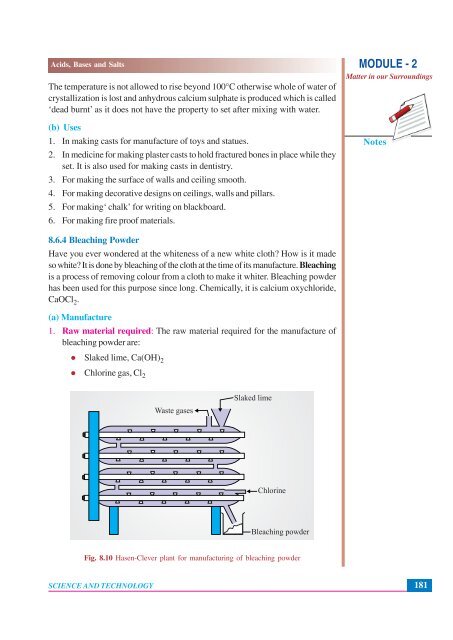
1. Raw material required: The raw material required for the manufacture of<br />
bleaching powder are:<br />
Slaked lime, Ca(OH) 2<br />
Chlorine gas, Cl 2<br />
Waste gases<br />
Slaked lime<br />
Chlorine<br />
Bleaching powder<br />
Fig. <strong>8.</strong>10 Hasen-Clever plant for manufacturing of bleaching powder<br />
SCIENCE AND TECHNOLOGY<br />
181