IMPLANON IMPLANT FOR SUBDERMAL USE ... - epgonline.org
IMPLANON IMPLANT FOR SUBDERMAL USE ... - epgonline.org
IMPLANON IMPLANT FOR SUBDERMAL USE ... - epgonline.org
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
ORGANON EU SmPC<br />
RA 0450 EU S6 (REF 6.0) FINAL<br />
14<br />
<strong>IMPLANON</strong><br />
<strong>IMPLANT</strong> <strong>FOR</strong> <strong>SUBDERMAL</strong> <strong>USE</strong><br />
carefully followed. Based on the available data, Implanon may be used during<br />
lactation.<br />
4.7 Effects on ability to drive and use machines<br />
On the basis of the pharmacodynamic profile, Implanon is expected to have no<br />
or negligible influence on the ability to drive or use machines.<br />
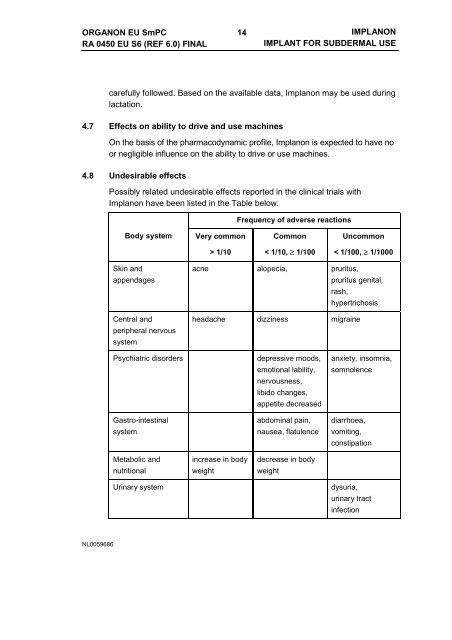
4.8 Undesirable effects<br />
Possibly related undesirable effects reported in the clinical trials with<br />
Implanon have been listed in the Table below.<br />
Frequency of adverse reactions<br />
Body system<br />
Very common<br />
Common<br />
Uncommon<br />
> 1/10<br />
< 1/10, ≥ 1/100<br />
< 1/100, ≥ 1/1000<br />
Skin and<br />
appendages<br />
Central and<br />
peripheral nervous<br />
system<br />
acne alopecia, pruritus,<br />
pruritus genital,<br />
rash,<br />
hypertrichosis<br />
headache dizziness migraine<br />
Psychiatric disorders<br />
Gastro-intestinal<br />
system<br />
depressive moods,<br />
emotional lability,<br />
nervousness,<br />
libido changes,<br />
appetite decreased<br />
abdominal pain,<br />
nausea, flatulence<br />
anxiety, insomnia,<br />
somnolence<br />
diarrhoea,<br />
vomiting,<br />
constipation<br />
Metabolic and<br />
nutritional<br />
increase in body<br />
weight<br />
decrease in body<br />
weight<br />
Urinary system<br />
dysuria,<br />
urinary tract<br />
infection<br />
NL0059686