Part III - BEBAC ⢠Consultancy Services for Bioequivalence
Part III - BEBAC ⢠Consultancy Services for Bioequivalence
Part III - BEBAC ⢠Consultancy Services for Bioequivalence
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Taking a Biostatistical Approach to Designing a <strong>Bioequivalence</strong>B<br />
Study: Ensuring Success through Effective Planning (3/3)<br />
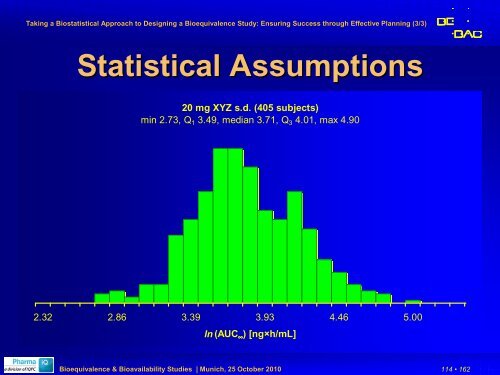
Statistical Assumptions<br />
20 mg XYZ s.d. (405 subjects)<br />
min 2.73, Q 1 3.49, median 3.71, Q 3 4.01, max 4.90<br />
2.32 2.86 3.39 3.93 4.46 5.00<br />
ln (AUC ∞ ) [ng×h/mL]<br />
<strong>Bioequivalence</strong> & Bioavailability Studies | Munich, 252<br />
October 2010<br />
114 • 162