Download - Veterinary Medicines Directorate - Defra
Download - Veterinary Medicines Directorate - Defra
Download - Veterinary Medicines Directorate - Defra
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
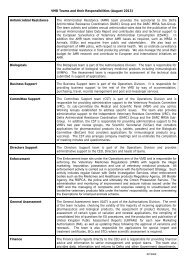
VMGN No. 11<br />
QUICK START GUIDE<br />
This <strong>Veterinary</strong> <strong>Medicines</strong> Guidance Note (VMGN) is aimed primarily at Marketing<br />
Authorisation Holders (MAH) and is intended to provide guidance on pharmacovigilance<br />
and the reporting of adverse events.<br />
The quick start guide is a summary of the provisions of the <strong>Veterinary</strong> <strong>Medicines</strong><br />
Regulations (VMR); detailed information is found in the body of the guidance note.<br />
MAHs have a legal obligation to record any information which they receive about adverse<br />
events involving their veterinary medicinal products. Adverse events associated with<br />
veterinary medicines include adverse reactions in animals which occur after use in<br />
accordance with the advice on the label or following off-label use, and suspected lack of<br />
expected efficacy after use in accordance with the label, and adverse reactions in humans<br />
following exposure to a veterinary medicine or a treated animal.<br />
MAHs must report any animal adverse events involving death, permanent disability, lifethreatening<br />
illness or congenital abnormality, and all human reactions, to the relevant<br />
competent authority within 15 days of their receipt of the information. For adverse events<br />
which occur in the UK, the VMD is the relevant competent authority.<br />
The scope of pharmacovigilance includes incidents arising from exposure to a veterinary<br />
medicine present in the environment, and issues concerning the validity of withdrawal<br />
periods.<br />
MAHs must include reports of all adverse events and other pharmacovigilance issues<br />
involving a veterinary medicine in the Periodic Safety Update Report (PSUR) for the<br />
product. A scientific evaluation of the risk-benefit balance of the veterinary medicine is an<br />
important aspect of a PSUR, which should also include an estimation of exposure to the<br />
veterinary medicine and the incidence of adverse events associated with it.<br />
The VMD should be informed immediately about any concerns relating to the safety of a<br />
veterinary medicine arising from pharmacovigilance or resulting from quality issues.<br />
FURTHER INFORMATION<br />
For more information on pharmacovigilance please contact the VMD‟s pharmacovigilance<br />
team on 01932 338427 or alternatively contact VMD reception on 01932 336911 and<br />
quote “pharmacovigilance”.<br />
2