Rapid Diagnostic Tests - NVBDCP
Rapid Diagnostic Tests - NVBDCP
Rapid Diagnostic Tests - NVBDCP
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
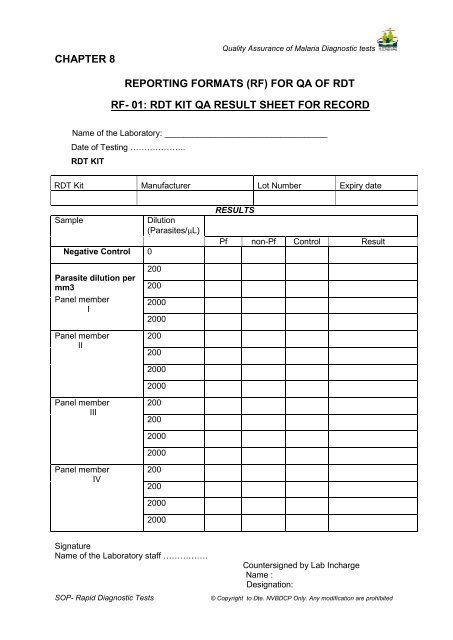
CHAPTER 8<br />
Quality Assurance of Malaria <strong>Diagnostic</strong> tests<br />
REPORTING FORMATS (RF) FOR QA OF RDT<br />
RF- 01: RDT KIT QA RESULT SHEET FOR RECORD<br />
Name of the Laboratory: ___________________________________<br />
Date of Testing ..<br />
RDT KIT<br />
RDT Kit Manufacturer Lot Number Expiry date<br />
Sample<br />
Negative Control 0<br />
Dilution<br />
(Parasites/µL)<br />
RESULTS<br />
Pf non-Pf Control Result<br />
Parasite dilution per<br />
mm3<br />
Panel member<br />
I<br />
Panel member<br />
II<br />
200<br />
200<br />
2000<br />
2000<br />
200<br />
200<br />
2000<br />
2000<br />
Panel member<br />
III<br />
200<br />
200<br />
2000<br />
2000<br />
Panel member<br />
IV<br />
200<br />
200<br />
2000<br />
2000<br />
Signature<br />
Name of the Laboratory staff .<br />
SOP- <strong>Rapid</strong> <strong>Diagnostic</strong> <strong>Tests</strong><br />
Countersigned by Lab Incharge<br />
Name :<br />
Designation:<br />
© Copyright to Dte. <strong>NVBDCP</strong> Only. Any modification are prohibited