Course Objectives - Institute for Clinical Research Education
Course Objectives - Institute for Clinical Research Education
Course Objectives - Institute for Clinical Research Education
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
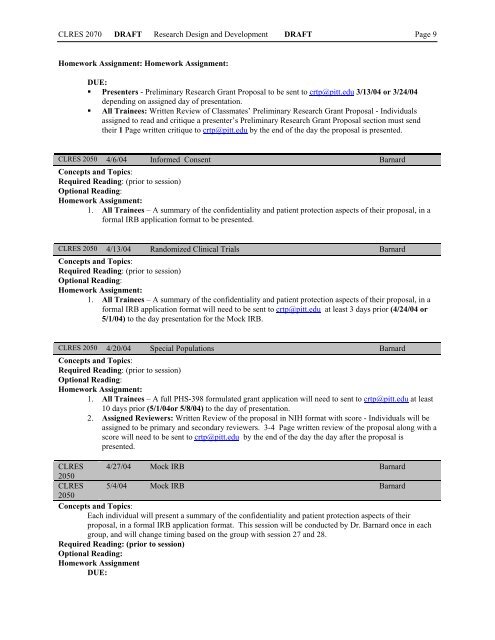
CLRES 2070 DRAFT <strong>Research</strong> Design and Development DRAFT Page 9<br />
Homework Assignment: Homework Assignment:<br />
DUE:<br />
• Presenters - Preliminary <strong>Research</strong> Grant Proposal to be sent to crtp@pitt.edu 3/13/04 or 3/24/04<br />
depending on assigned day of presentation.<br />
• All Trainees: Written Review of Classmates’ Preliminary <strong>Research</strong> Grant Proposal - Individuals<br />
assigned to read and critique a presenter’s Preliminary <strong>Research</strong> Grant Proposal section must send<br />
their 1 Page written critique to crtp@pitt.edu by the end of the day the proposal is presented.<br />
CLRES 2050 4/6/04 In<strong>for</strong>med Consent Barnard<br />
Concepts and Topics:<br />
Required Reading: (prior to session)<br />
Optional Reading:<br />
Homework Assignment:<br />
1. All Trainees – A summary of the confidentiality and patient protection aspects of their proposal, in a<br />
<strong>for</strong>mal IRB application <strong>for</strong>mat to be presented.<br />
CLRES 2050 4/13/04 Randomized <strong>Clinical</strong> Trials Barnard<br />
Concepts and Topics:<br />
Required Reading: (prior to session)<br />
Optional Reading:<br />
Homework Assignment:<br />
1. All Trainees – A summary of the confidentiality and patient protection aspects of their proposal, in a<br />
<strong>for</strong>mal IRB application <strong>for</strong>mat will need to be sent to crtp@pitt.edu at least 3 days prior (4/24/04 or<br />
5/1/04) to the day presentation <strong>for</strong> the Mock IRB.<br />
CLRES 2050 4/20/04 Special Populations Barnard<br />
Concepts and Topics:<br />
Required Reading: (prior to session)<br />
Optional Reading:<br />
Homework Assignment:<br />
1. All Trainees – A full PHS-398 <strong>for</strong>mulated grant application will need to sent to crtp@pitt.edu at least<br />
10 days prior (5/1/04or 5/8/04) to the day of presentation.<br />
2. Assigned Reviewers: Written Review of the proposal in NIH <strong>for</strong>mat with score - Individuals will be<br />
assigned to be primary and secondary reviewers. 3-4 Page written review of the proposal along with a<br />
score will need to be sent to crtp@pitt.edu by the end of the day the day after the proposal is<br />
presented.<br />
CLRES 4/27/04 Mock IRB Barnard<br />
2050<br />
CLRES 5/4/04 Mock IRB Barnard<br />
2050<br />
Concepts and Topics:<br />
Each individual will present a summary of the confidentiality and patient protection aspects of their<br />
proposal, in a <strong>for</strong>mal IRB application <strong>for</strong>mat. This session will be conducted by Dr. Barnard once in each<br />
group, and will change timing based on the group with session 27 and 28.<br />
Required Reading: (prior to session)<br />
Optional Reading:<br />
Homework Assignment<br />
DUE: