86 J. Phys. Chem. A, Vol. 110, No. 1, 2006 Huang et al.chemisorption <strong>of</strong> O 2 on W 3 O 8 <strong>and</strong> may serve as importantintermediate in catalytic oxidation reactions.2. Experimental <strong>and</strong> Theoretical Methods2.1. Photoelectron Spectroscopy. The experiments werecarried out using a magnetic-bottle photoelectron spectroscopy(PES) apparatus equipped with a laser vaporization supersoniccluster source. 30,31 Briefly, W m O n - cluster anions were producedby laser vaporization <strong>of</strong> a pure tungsten target in <strong>the</strong> presence<strong>of</strong> helium carrier gas seeded with 0.5% O 2 <strong>and</strong> were analyzedusing a time-<strong>of</strong>-flight mass spectrometer. The W 3 O n - (n )7-10) clusters <strong>of</strong> current interest were each mass selected <strong>and</strong>decelerated before being photodetached with 157-nm photonsfrom an F 2 excimer laser. Photoelectrons were collected at nearly100% efficiency by <strong>the</strong> magnetic bottle <strong>and</strong> analyzed in a 3.5m long electron flight tube. Photoelectron spectra were calibratedusing <strong>the</strong> known spectrum <strong>of</strong> Rh - , <strong>and</strong> <strong>the</strong> energy resolution<strong>of</strong> <strong>the</strong> apparatus was ∆E k /E k ≈ 2.5%, that is, ∼25 meV for 1eV electrons.2.2. Density Functional Calculations. The <strong>the</strong>oretical calculationswere performed at <strong>the</strong> DFT level using <strong>the</strong> B3LYPhybrid functional. 32-34 A number <strong>of</strong> structural c<strong>and</strong>idates wereevaluated, <strong>and</strong> <strong>the</strong> search for <strong>the</strong> global minima was performedusing analytical gradients with <strong>the</strong> Stuttgart 14-valence-electronpseudopotentials <strong>and</strong> <strong>the</strong> valence basis sets 35,36 augmented withtwo f-type <strong>and</strong> one g-type polarization functions [ζ(f) ) 0.256,0.825; ζ(g) ) 0.627] for tungsten as recommended by Martin<strong>and</strong> Sundermann 37 <strong>and</strong> <strong>the</strong> aug-cc-pVTZ basis set for oxygen.38,39 Scalar relativistic effects, i.e., <strong>the</strong> mass-velocity <strong>and</strong>Darwin effects, were taken into account via <strong>the</strong> quasirelativisticpseudopotentials. Since we were mainly interested in explaining<strong>the</strong> experimentally observed PES spectra, no fur<strong>the</strong>r effort wasdevoted to resolve <strong>the</strong> spin-orbit coupled fine structures in <strong>the</strong>calculated spectra. Our previous results on tungsten oxidesshowed that spin-orbit coupling effects would shift <strong>the</strong> orbitalenergies by up to a few tenth <strong>of</strong> an eV, which would not affect<strong>the</strong> spectral assignment. 40 <strong>On</strong>ly a selected set <strong>of</strong> optimizedstructures (<strong>the</strong> ground state <strong>and</strong> a few low-lying isomers),considered to be important in interpreting <strong>the</strong> experimental data,are reported. Vibrational frequency calculations were performedat <strong>the</strong> same level <strong>of</strong> <strong>the</strong>ory to verify <strong>the</strong> nature <strong>of</strong> <strong>the</strong> stationarypoints. The vertical detachment energies (VDEs) were calculatedusing a combined ∆SCF-TDDFT approach, as we previouslyoutlined. 41,42 In this approach, <strong>the</strong> ground-state energies <strong>of</strong> <strong>the</strong>anions <strong>and</strong> <strong>the</strong> neutrals were calculated from <strong>the</strong> ∆SCF energydifference at <strong>the</strong> B3LYP level, whereas <strong>the</strong> excited states <strong>of</strong><strong>the</strong> one-electron-detached species were obtained from TDDFTcalculations <strong>of</strong> <strong>the</strong> neutrals. For all calculations, <strong>the</strong> extra fineintegration grid was used to obtain highly accurate DFT results.All calculations were accomplished using <strong>the</strong> NWChem 4.6program <strong>and</strong> <strong>the</strong> Molecular Science Computing Facility locatedat <strong>the</strong> Environmental Molecular Sciences Laboratory. 43 TheExtensible Computational Chemistry Environment s<strong>of</strong>tware wasused to generate <strong>the</strong> three-dimensional contours <strong>of</strong> <strong>the</strong> calculatedKohn-Sham orbitals. 443. Experimental ResultsThe PES spectra <strong>of</strong> W 3 O n - (n ) 7-10) measured at 157 nmare shown in Figure 1. The observed spectral b<strong>and</strong>s are labeledwith letters (X, A, B, C). The low binding energy features inW 3 O n - (n ) 7-9) are exp<strong>and</strong>ed to illustrate <strong>the</strong> spectral details.The obtained VDEs <strong>and</strong> ground-state adiabatic detachmentenergies (ADEs) are given in Table 1.Figure 1. Photoelectron spectra <strong>of</strong> W 3O n- (n ) 7-10) at 157 nm(7.866 eV).The spectrum <strong>of</strong> W 3 O - 7 (Figure 1a) shows two relativelyweak b<strong>and</strong>s (X <strong>and</strong> A) at low binding energies. More intensesignals above 6 eV appear to be congested <strong>and</strong> continuous. Theground-state b<strong>and</strong> X has a VDE <strong>of</strong> 3.9 eV with a long tail downto ∼3.3 eV ei<strong>the</strong>r due to vibrational hot b<strong>and</strong>s or a largegeometry change between <strong>the</strong> anion <strong>and</strong> neutral ground states.The ADE <strong>of</strong> <strong>the</strong> X b<strong>and</strong> was estimated to be 3.5 ( 0.1 eV,which defines <strong>the</strong> electron affinity (EA) <strong>of</strong> neutral W 3 O 7 . Thelarge uncertainty was a result <strong>of</strong> <strong>the</strong> low energy tail, which madeit difficult to define <strong>the</strong> ADE. B<strong>and</strong> A with a VDE <strong>of</strong> 5.04 eVis relatively sharp, which should correspond to <strong>the</strong> first excitedstate <strong>of</strong> W 3 O 7 . A shoulder seems to be present at <strong>the</strong> lowerbinding energy side <strong>of</strong> b<strong>and</strong> A. The spectrum beyond ∼6.0 isfeatureless, which is labeled with B <strong>and</strong> C simply to denotetwo spectral regions.The spectrum <strong>of</strong> W 3 O - 8 (Figure 1b) displays relatively sharpfeatures. A weak b<strong>and</strong> X with a VDE <strong>of</strong> 4.62 eV is followedby a large energy gap <strong>and</strong> three well-defined spectral features(A, B, C) at high binding energies. The ground-state b<strong>and</strong> Xyields an ADE <strong>of</strong> 4.37 eV, which defines <strong>the</strong> EA <strong>of</strong> neutralW 3 O 8 . The first excited-state b<strong>and</strong> (A) at a VDE <strong>of</strong> 6.78 eVgives a X-A gap <strong>of</strong> 2.16 eV. The VDEs <strong>of</strong> b<strong>and</strong>s B <strong>and</strong> C aremeasured to be 7.23 <strong>and</strong> 7.65 eV, respectively.The PES spectrum <strong>of</strong> <strong>the</strong> stoichiometric W 3 O - 9 cluster isshown in Figure 1c. The ground-state X has weak intensity <strong>and</strong>is very broad, suggesting a large geometry change between <strong>the</strong>anion <strong>and</strong> neutral ground state. The X b<strong>and</strong> yields a VDE <strong>of</strong>4.2 eV <strong>and</strong> an ADE <strong>of</strong> 3.55 eV. The X b<strong>and</strong> is followed by alarge energy gap <strong>and</strong> <strong>the</strong> first excited-state b<strong>and</strong> (A) is locatedat a high binding energy with an estimated onset <strong>of</strong> ∼7 eV. Nodistinct features were observed in <strong>the</strong> higher binding energy
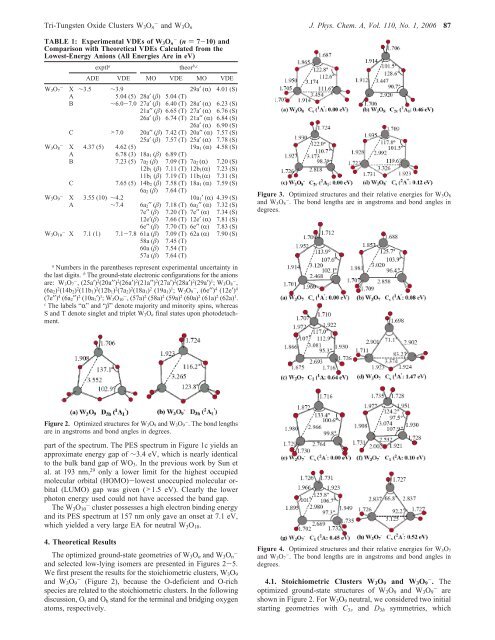
<strong>Tri</strong>-<strong>Tungsten</strong> <strong>Oxide</strong> Clusters W 3 O n - <strong>and</strong> W 3 O n J. Phys. Chem. A, Vol. 110, No. 1, 2006 87TABLE 1: Experimental VDEs <strong>of</strong> W 3 O n- (n ) 7-10) <strong>and</strong>Comparison with Theoretical VDEs Calculated from <strong>the</strong>Lowest-Energy Anions (All Energies Are in eV)exptl a<strong>the</strong>or b,cADE VDE MO VDE MO VDEW 3O - 7 X ∼3.5 ∼3.9 29a′ (R) 4.01 (S)A 5.04 (5) 28a′ (β) 5.04 (T)B ∼6.0-7.0 27a′ (β) 6.40 (T) 28a′ (R) 6.23 (S)21a′′ (β) 6.65 (T) 27a′ (R) 6.76 (S)26a′ (β) 6.74 (T) 21a′′ (R) 6.84 (S)26a′ (R) 6.90 (S)C >7.0 20a′′ (β) 7.42 (T) 20a′′ (R) 7.57 (S)25a′ (β) 7.57 (T) 25a′ (R) 7.78 (S)W 3O - 8 X 4.37 (5) 4.62 (5) 19a 1 (R) 4.58 (S)A6.78 (3) 18a 1 (β) 6.89 (T)B 7.23 (5) 7a 2 (β) 7.09 (T) 7a 2 (R) 7.20 (S)12b 1 (β) 7.11 (T) 12b 1(R) 7.23 (S)11b 1 (β) 7.19 (T) 11b 1(R) 7.31 (S)C 7.65 (5) 14b 2 (β) 7.58 (T) 18a 1 (R) 7.59 (S)6a 2 (β) 7.64 (T)W 3O - 9 X 3.55 (10) ∼4.2 10a 1′ (R) 4.39 (S)A ∼7.4 6a 2′′ (β) 7.18 (T) 6a 2′′ (R) 7.32 (S)7e′′ (β) 7.20 (T) 7e′′ (R) 7.34 (S)12e′(β) 7.66 (T) 12e′ (R) 7.81 (S)6e′′ (β) 7.70 (T) 6e′′ (R) 7.83 (S)W 3O - 10 X 7.1 (1) 7.1-7.8 61a (β) 7.09 (T) 62a (R) 7.90 (S)58a (β) 7.45 (T)60a (β) 7.54 (T)57a (β) 7.64 (T)aNumbers in <strong>the</strong> paren<strong>the</strong>ses represent experimental uncertainty in<strong>the</strong> last digits. b The ground-state electronic configurations for <strong>the</strong> anionsare: W 3O 7- , (25a′) 2 (20a′′) 2 (26a′) 2 (21a′′) 2 (27a′) 2 (28a′) 2 (29a′) 1 ;W 3O 8- ,(6a 2) 2 (14b 2) 2 (11b 1) 2 (12b 1) 2 (7a 2) 2 (18a 1) 2 (19a 1) 1 ;W 3O 9- , (6e′′) 4 (12e′) 4(7e′′) 4 (6a 2′′) 2 (10a 1′) 1 ;W 3O 10- , (57a) 2 (58a) 2 (59a) 2 (60a) 2 (61a) 2 (62a) 1 .cThe labels “R” <strong>and</strong> “β” denote majority <strong>and</strong> minority spins, whereasS <strong>and</strong> T denote singlet <strong>and</strong> triplet W 3O n final states upon photodetachment.Figure 3. Optimized structures <strong>and</strong> <strong>the</strong>ir relative energies for W 3O 8<strong>and</strong> W 3O 8- . The bond lengths are in angstroms <strong>and</strong> bond angles indegrees.Figure 2. Optimized structures for W 3O 9 <strong>and</strong> W 3O 9- . The bond lengthsare in angstroms <strong>and</strong> bond angles in degrees.part <strong>of</strong> <strong>the</strong> spectrum. The PES spectrum in Figure 1c yields anapproximate energy gap <strong>of</strong> ∼3.4 eV, which is nearly identicalto <strong>the</strong> bulk b<strong>and</strong> gap <strong>of</strong> WO 3 . In <strong>the</strong> previous work by Sun etal. at 193 nm, 29 only a lower limit for <strong>the</strong> highest occupiedmolecular orbital (HOMO)-lowest unoccupied molecular orbital(LUMO) gap was given (>1.5 eV). Clearly <strong>the</strong> lowerphoton energy used could not have accessed <strong>the</strong> b<strong>and</strong> gap.The W 3 O 10 - cluster possesses a high electron binding energy<strong>and</strong> its PES spectrum at 157 nm only gave an onset at 7.1 eV,which yielded a very large EA for neutral W 3 O 10 .4. Theoretical ResultsThe optimized ground-state geometries <strong>of</strong> W 3 O n <strong>and</strong> W 3 O- n<strong>and</strong> selected low-lying isomers are presented in Figures 2-5.We first present <strong>the</strong> results for <strong>the</strong> stoichiometric clusters, W 3 O 9<strong>and</strong> W 3 O - 9 (Figure 2), because <strong>the</strong> O-deficient <strong>and</strong> O-richspecies are related to <strong>the</strong> stoichiometric clusters. In <strong>the</strong> followingdiscussion, O t <strong>and</strong> O b st<strong>and</strong> for <strong>the</strong> terminal <strong>and</strong> bridging oxygenatoms, respectively.Figure 4. Optimized structures <strong>and</strong> <strong>the</strong>ir relative energies for W 3O 7<strong>and</strong> W 3O 7- . The bond lengths are in angstroms <strong>and</strong> bond angles indegrees.4.1. Stoichiometric Clusters W 3 O 9 <strong>and</strong> W 3 O 9 - . Theoptimized ground-state structures <strong>of</strong> W 3 O 9 <strong>and</strong> W 3 O 9 - areshown in Figure 2. For W 3 O 9 neutral, we considered two initialstarting geometries with C 3V <strong>and</strong> D 3h symmetries, which