Guidelines for Drugs of Abuse Testing vers 2012 - Brenneisen
Guidelines for Drugs of Abuse Testing vers 2012 - Brenneisen
Guidelines for Drugs of Abuse Testing vers 2012 - Brenneisen
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
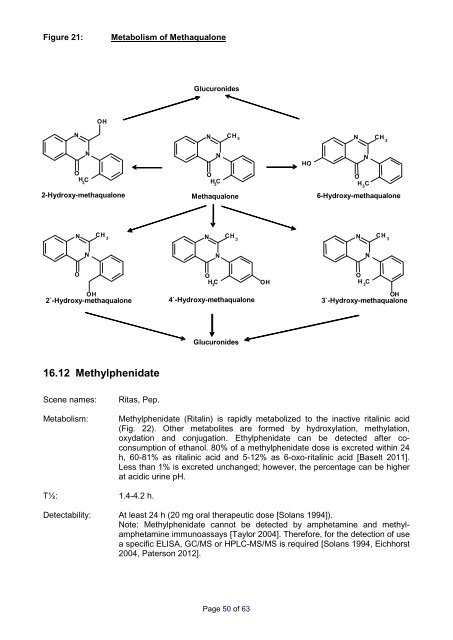
Figure 21: Metabolism <strong>of</strong> Methaqualone<br />
N<br />
N<br />
O<br />
H C 3<br />
N<br />
N<br />
O<br />
H C 3<br />
2-Hydroxy-methaqualone Methaqualone<br />
6-Hydroxy-methaqualone<br />
N<br />
O<br />
N<br />
OH<br />
CH 3<br />
OH<br />
2`-Hydroxy-methaqualone<br />
16.12 Methylphenidate<br />
Scene names: Ritas, Pep.<br />
Glucuronides<br />
N<br />
N<br />
O<br />
H C 3<br />
CH 3<br />
CH 3<br />
4`-Hydroxy-methaqualone<br />
Glucuronides<br />
Page 50 <strong>of</strong> 63<br />
OH<br />
HO<br />
N CH 3<br />
N<br />
O<br />
H C 3<br />
N<br />
O<br />
H 3<br />
N<br />
C<br />
CH 3<br />
OH<br />
3`-Hydroxy-methaqualone<br />
Metabolism: Methylphenidate (Ritalin) is rapidly metabolized to the inactive ritalinic acid<br />
(Fig. 22). Other metabolites are <strong>for</strong>med by hydroxylation, methylation,<br />
oxydation and conjugation. Ethylphenidate can be detected after coconsumption<br />
<strong>of</strong> ethanol. 80% <strong>of</strong> a methylphenidate dose is excreted within 24<br />
h, 60-81% as ritalinic acid and 5-12% as 6-oxo-ritalinic acid [Baselt 2011].<br />
Less than 1% is excreted unchanged; however, the percentage can be higher<br />
at acidic urine pH.<br />
T½: 1.4-4.2 h.<br />
Detectability: At least 24 h (20 mg oral therapeutic dose [Solans 1994]).<br />
Note: Methylphenidate cannot be detected by amphetamine and methylamphetamine<br />
immunoassays [Taylor 2004]. There<strong>for</strong>e, <strong>for</strong> the detection <strong>of</strong> use<br />
a specific ELISA, GC/MS or HPLC-MS/MS is required [Solans 1994, Eichhorst<br />
2004, Paterson <strong>2012</strong>].