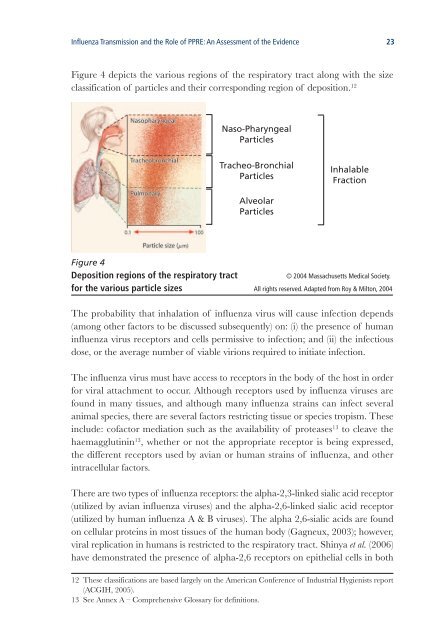
Influenza Tr<strong>an</strong>smission <strong>an</strong>d <strong>the</strong> Role <strong>of</strong> PPRE: An Assessment <strong>of</strong> <strong>the</strong> Evidence 23Figure 4 depicts <strong>the</strong> various regions <strong>of</strong> <strong>the</strong> respiratory tract along with <strong>the</strong> sizeclassification <strong>of</strong> particles <strong>an</strong>d <strong>the</strong>ir corresponding region <strong>of</strong> deposition. 12Naso-PharyngealParticlesTracheo-BronchialParticlesInhalableFractionAlveolarParticlesFigure 4Deposition regions <strong>of</strong> <strong>the</strong> respiratory tract© 2004 Massachusetts Medical Society.for <strong>the</strong> various particle sizes All rights reserved. Adapted from Roy & Milton, 2004The probability that inhalation <strong>of</strong> influenza virus will cause infection depends(among o<strong>the</strong>r factors to be discussed subsequently) on: (i) <strong>the</strong> presence <strong>of</strong> hum<strong>an</strong>influenza virus receptors <strong>an</strong>d cells permissive to infection; <strong>an</strong>d (ii) <strong>the</strong> infectiousdose, or <strong>the</strong> average number <strong>of</strong> viable virions required to initiate infection.The influenza virus must have access to receptors in <strong>the</strong> body <strong>of</strong> <strong>the</strong> host in orderfor viral attachment to occur. Although receptors used by influenza viruses arefound in m<strong>an</strong>y tissues, <strong>an</strong>d although m<strong>an</strong>y influenza strains c<strong>an</strong> infect several<strong>an</strong>imal species, <strong>the</strong>re are several factors restricting tissue or species tropism. Theseinclude: c<strong>of</strong>actor mediation such as <strong>the</strong> availability <strong>of</strong> proteases 13 to cleave <strong>the</strong>haemagglutinin 13 , whe<strong>the</strong>r or not <strong>the</strong> appropriate receptor is being expressed,<strong>the</strong> different receptors used by avi<strong>an</strong> or hum<strong>an</strong> strains <strong>of</strong> influenza, <strong>an</strong>d o<strong>the</strong>rintracellular factors.There are two types <strong>of</strong> influenza receptors: <strong>the</strong> alpha-2,3-linked sialic acid receptor(utilized by avi<strong>an</strong> influenza viruses) <strong>an</strong>d <strong>the</strong> alpha-2,6-linked sialic acid receptor(utilized by hum<strong>an</strong> influenza A & B viruses). The alpha 2,6-sialic acids are foundon cellular proteins in most tissues <strong>of</strong> <strong>the</strong> hum<strong>an</strong> body (Gagneux, 2003); however,viral replication in hum<strong>an</strong>s is restricted to <strong>the</strong> respiratory tract. Shinya et al. (2006)have demonstrated <strong>the</strong> presence <strong>of</strong> alpha-2,6 receptors on epi<strong>the</strong>lial cells in both12 These classifications are based largely on <strong>the</strong> Americ<strong>an</strong> Conference <strong>of</strong> Industrial Hygienists report(ACGIH, 2005).13 See Annex A – Comprehensive Glossary for definitions.
24 Influenza Tr<strong>an</strong>smission <strong>an</strong>d <strong>the</strong> Role <strong>of</strong> PPRE: An Assessment <strong>of</strong> <strong>the</strong> Evidence<strong>the</strong> upper <strong>an</strong>d lower respiratory tract, <strong>an</strong>d that hum<strong>an</strong> influenza viruses c<strong>an</strong> infectepi<strong>the</strong>lial cells in <strong>the</strong> nasal epi<strong>the</strong>lium, <strong>the</strong> tracheobronchial tree <strong>an</strong>d <strong>the</strong> alveolarepi<strong>the</strong>lium. Mastrosovich et al. (2004) demonstrated that hum<strong>an</strong> influenza virusespreferentially infected non-ciliated epi<strong>the</strong>lial cells, in correlation with <strong>the</strong> receptordensity. This latter result was obtained on epi<strong>the</strong>lial cells cultured in vitro <strong>an</strong>d maynot represent entirely accurately <strong>the</strong> situation in vivo. Shinya et al. (2006) <strong>an</strong>dv<strong>an</strong> Riel et al. (2006) have also shown that some epi<strong>the</strong>lial cells in <strong>the</strong> alveoli <strong>an</strong>d <strong>the</strong>respiratory bronchioles, identified as pneumocytes type II, express <strong>the</strong> alpha-2,3receptor <strong>an</strong>d allow for <strong>the</strong> replication <strong>of</strong> avi<strong>an</strong> influenza A (H5N1).A common me<strong>an</strong>s <strong>of</strong> evaluating <strong>the</strong> infectious dose for <strong>an</strong>y given disease is bydetermining <strong>the</strong> amount <strong>of</strong> virus required to infect 50 per cent <strong>of</strong> a samplepopulation (ei<strong>the</strong>r in vitro or in vivo). This is referred to as <strong>the</strong> “tissue cultureinfectious dose 50 per cent” (TCID50) for in-vitro studies or <strong>the</strong> “hum<strong>an</strong> infectiousdose 50 per cent” (HID50) for in-vivo, hum<strong>an</strong> studies. Alford et al. (1966) showed thatfor alveolar-sized particles <strong>the</strong> HID50 was in <strong>the</strong> r<strong>an</strong>ge <strong>of</strong> 0.6 to 3.0 TCID50. Couchet al. reported that <strong>the</strong> dose by intr<strong>an</strong>asal drop inoculation (which initiates infectionat <strong>the</strong> same site as nasopharyngeal sized-particles) required to cause infection was100 times greater th<strong>an</strong> that required to infect with alveolar-sized particles (Couch,1971; Couch, 1974). The viral concentration from nasal washes in infected personshas been shown to be as high as 10 7 TCID50 per ml. Since in a single sneeze, <strong>the</strong>volume <strong>of</strong> all <strong>the</strong> particles 10 µm or less (alveolar-sized particles) is estimated at1.2 x 10 -5 ml (Nicas, 2005), <strong>the</strong>se alveolar-sized particles would contain 120 TCID50,well above <strong>the</strong> HID50 calculated by Alford. Also, since 99.9 per cent <strong>of</strong> <strong>the</strong> volumein a sneeze is comprised <strong>of</strong> particles greater th<strong>an</strong> 8 µm (Nicas, 2005), <strong>the</strong> infectiousdose contained in nasopharyngeal-sized particles would also exceed <strong>the</strong> requireddose for infection.Long-R<strong>an</strong>ge Inhalation Tr<strong>an</strong>smissionFor this report, long-r<strong>an</strong>ge tr<strong>an</strong>smission has been defined as inhalation tr<strong>an</strong>smission<strong>of</strong> <strong>the</strong> virus at dist<strong>an</strong>ces greater th<strong>an</strong> approximately two metres. Once small particleshave been produced by <strong>an</strong> infected person, <strong>the</strong> likelihood <strong>of</strong> long-r<strong>an</strong>ge, inhalationtr<strong>an</strong>smission depends on <strong>the</strong> duration <strong>of</strong> viral viability in <strong>the</strong> air, <strong>the</strong> environmentalconditions to which <strong>the</strong> virus is exposed, <strong>an</strong>d <strong>the</strong> effects <strong>of</strong> dilution <strong>an</strong>d air currents.The persistence <strong>of</strong> <strong>the</strong> infectivity <strong>of</strong> influenza virus in aerosols has been studied in<strong>the</strong> laboratory. In experiments that used homogeneous aerosolized influenza virussuspensions (me<strong>an</strong> diametre <strong>of</strong> 6 µm), virus infectivity at a fixed relative humidityin <strong>the</strong> r<strong>an</strong>ge <strong>of</strong> 15 to 40 per cent undergoes slow, exponential decay (Hemmes,1960; Hemmes, 1962). These results are consistent with those <strong>of</strong> <strong>an</strong> older study inwhich infectious influenza viruses in <strong>an</strong> aerosol could be demonstrated to persist