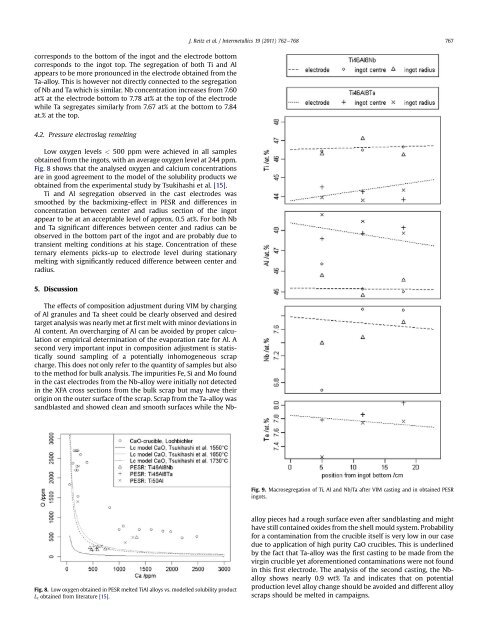
J. Reitz et al. / Intermetallics 19 (2011) 762e768 767corresponds to the bottom <strong>of</strong> the ingot and the electrode bottomcorresponds to the ingot top. The segregation <strong>of</strong> both Ti and Alappears to be more pronounced in the electrode obtained <strong>from</strong> theTa-alloy. This is however not directly connected to the segregation<strong>of</strong> Nb and Ta which is similar. Nb concentration increases <strong>from</strong> 7.60at% at the electrode bottom to 7.78 at% at the top <strong>of</strong> the electrodewhile Ta segregates similarly <strong>from</strong> 7.67 at% at the bottom to 7.84at.% at the top.4.2. Pressure electroslag remeltingLow oxygen levels < 500 ppm were achieved in all samplesobtained <strong>from</strong> the ingots, with an average oxygen level at 244 ppm.Fig. 8 shows that the analysed oxygen and calcium concentrationsare in good agreement to the model <strong>of</strong> the solubility products weobtained <strong>from</strong> the experimental study by Tsukihashi et al. [15].Ti and Al segregation observed in the cast electrodes wassmoothed by the backmixing-effect in PESR and differences inconcentration between center and radius section <strong>of</strong> the ingotappear to be at an acceptable level <strong>of</strong> approx. 0.5 at%. For both Nband Ta significant differences between center and radius can beobserved in the bottom part <strong>of</strong> the ingot and are probably due totransient melting conditions at his stage. Concentration <strong>of</strong> theseternary elements picks-up to electrode level during stationarymelting with significantly reduced difference between center andradius.5. DiscussionFig. 9. Macrosegregation <strong>of</strong> Ti, Al and Nb/Ta after VIM casting and in obtained PESRingots.Fig. 8. Low oxygen obtained in PESR melted TiAl alloys vs. modelled solubility productL c obtained <strong>from</strong> literature [15].The effects <strong>of</strong> composition adjustment during VIM by charging<strong>of</strong> Al granules and Ta sheet could be clearly observed and desiredtarget analysis was nearly met at first melt with minor deviations inAl content. An overcharging <strong>of</strong> Al can be avoided by proper calculationor empirical determination <strong>of</strong> the evaporation rate for Al. Asecond very important input in composition adjustment is statisticallysound sampling <strong>of</strong> a potentially inhomogeneous <strong>scrap</strong>charge. This does not only refer to the quantity <strong>of</strong> samples but alsoto the method for bulk analysis. The impurities Fe, Si and Mo foundin the cast electrodes <strong>from</strong> the Nb-alloy were initially not detectedin the XFA cross sections <strong>from</strong> the bulk <strong>scrap</strong> but may have theirorigin on the outer surface <strong>of</strong> the <strong>scrap</strong>. Scrap <strong>from</strong> the Ta-alloy wassandblasted and showed clean and smooth surfaces while the Nballoypieces had a rough surface even after sandblasting and mighthave still contained oxides <strong>from</strong> the shell mould system. Probabilityfor a contamination <strong>from</strong> the crucible itself is very low in our casedue to application <strong>of</strong> high purity CaO crucibles. This is underlinedby the fact that Ta-alloy was the first casting to be made <strong>from</strong> thevirgin crucible yet aforementioned contaminations were not foundin this first electrode. The analysis <strong>of</strong> the second casting, the Nballoyshows nearly 0.9 wt% Ta and indicates that on potentialproduction level alloy change should be avoided and different alloy<strong>scrap</strong>s should be melted in campaigns.
768J. Reitz et al. / Intermetallics 19 (2011) 762e768The obtained oxygen levels in the PESR ingots are highly reliablewith mean errors below 10% upon triple analysis. This howeverdoes not hold for the determination <strong>of</strong> calcium concentration.A detection <strong>of</strong> calcium below 800 ppm by XFA is questionablebecause <strong>of</strong> the high signal to background noise ratios and thealternative to dissolve TiAl in acid for ICP analysis results ina relatively large mean error <strong>of</strong> 60%. This is due to the fact that HF isa necessary component for dissolution <strong>of</strong> the sample but should bekept to a minimum as the solubility product for CaF 2 is so low thatdissolved Ca precipitates as fluoride which leads to an analyticalerror. However within the achievable accuracy, the obtainedresults for the CaO equilibrium fit very well to the theoreticalexpectations.6. ConclusionsThe recycling <strong>of</strong> g-TiAl alloys by consolidative melting via VIM ina CaO crucible and subsequent deoxidation by PESR has beendemonstrated to prove the high potential <strong>of</strong> such a process chain.Alloy composition was successfully adjusted and the major processmechanisms are understood. A correct sampling <strong>of</strong> <strong>scrap</strong> charges ina statistical meaningful manner and a unified standard for chemicalanalysis <strong>of</strong> TiAl are to be improved in order to obtain a more precisecontrol <strong>of</strong> alloy composition and impurity content as necessary forthe sensitive TiAl alloys.PESR <strong>of</strong>fers efficient deoxidation <strong>of</strong> TiAl even down to 250 ppmif correctly controlled. This <strong>of</strong>fers an interesting starting point forreuse <strong>of</strong> the material and might reveal so far unseen materialproperties. Macrosegregation in the obtained ingots is minimal andsegregation effects <strong>from</strong> the VIM cast electrodes could be homogenisedby PESR. After the first two processing steps, the materialcontains residual Ca in a concentration <strong>of</strong> 500e1000 ppm which isprobably too much for direct reuse in an <strong>investment</strong> casting processdue to strong evaporation tendencies. However calculations indicatethat it should be completely removed by subsequent VAR andtrials on the obtained material will be carried out in the near futurefor experimental pro<strong>of</strong>.AcknowledgementsThe presented experimental works were carried out in theframework <strong>of</strong> the EU FP6 project IMPRESS (contract number NMP3-CT-2004-500635). The authors would like to thank the Europeancommission for funding <strong>of</strong> their work, as well as the Germanministry <strong>of</strong> education and research (BMBF) and the Deutsche Forschungsgemeinschaft(DFG) who delivered funding for elaboration<strong>of</strong> the basic principles <strong>of</strong> the presented work in earlier projects. Wewould further like to acknowledge the support <strong>of</strong> GfE Gesellschaftfür Elektrometallurgie mbH, Nürnberg, Germany, ACCESS e.V.,Aachen, Germany, IRC Birmingham, UK and Doncasters Settas,Jumet, Belgium for providing <strong>scrap</strong> material for trials on TiAlrecycling. The authors gratefully acknowledge the support <strong>of</strong>ACCESS e.V., Aachen, Germany for assistance in preparation andinterpretation REM micrographs.References[1] Hammerschmidt J. Entwicklung einer prozessroute zur herstellung von<strong>gamma</strong>-tial-legierungen durch aluminothermie und schutzgas-elektroschlackeumschmelzen.IME Metallurgische Prozesstechnik und Metallrecycling,RWTH Aachen University; 2003, Ph.D. thesis.[2] Stoephasius J. Elektroschlackeraffination aluminothermisch hergestellter<strong>gamma</strong>-titan<strong>aluminide</strong>. IME Metallurgische Prozesstechnik und Metallrecycling,RWTH Aachen University; 2006, Ph.D. thesis.[3] Morscheiser J, Friedrich B, Lochbichler C, Aachen. Potential <strong>of</strong> ceramic cruciblesfor melting <strong>of</strong> <strong>titanium</strong>-alloys and <strong>gamma</strong>-<strong>titanium</strong>aluminde. Proceedings<strong>of</strong> the 51st international colloquium on refractories. 978-3-00-025895-4.Forschungsgemeinschaft Feuerfest e.V; 2008.[4] Nafziger R. Slag compositions for <strong>titanium</strong> electroslag remelting; 1969. Secondinternational symposium on electroslag remelting technology.[5] Armantrout C, Nafziger R. Proceedings <strong>of</strong> the seventy-third annual meeting <strong>of</strong>the American foundryman’s society. Development <strong>of</strong> electroslag meltingtechnique for <strong>titanium</strong>: selected properties <strong>of</strong> fabricated material, vol. 77;1969. pp. 353e359.[6] Benz M, Ryabtsev A. ESR as a fast technique to dissolve nitrogen rich inclusionsin <strong>titanium</strong>. Materials Research Innovations; 1999:364e8.[7] Nafziger R. Electroslag melting <strong>of</strong> <strong>titanium</strong> and aluminium. The Electroslagremelting process. U.S. Dept. <strong>of</strong> the Interior, Bureau <strong>of</strong> Mines; 1976.[8] Choudhury A, Scholz H, Ludwid N. Electroslag remelting <strong>of</strong> <strong>titanium</strong>. Conferenceproceedings; 1993.[9] Paton B, Medovar B, Benz M, Nafziger R, Medovar L. Esr for <strong>titanium</strong>:yesterday, today, tomorrow. 9th world conference on <strong>titanium</strong>. St. Petersburg;1999.[10] Stoephasius J, Reitz J, Friedrich B. ESR refining potential for <strong>titanium</strong> alloysusing a CaF 2 -based active slag. Advanced Engineering Materials 2007;9.doi:10.1002/adem.200700009.[11] Reitz J, Stoephasius J, Friedrich B. Tracing ca and f during remelting <strong>of</strong><strong>titanium</strong>-<strong>aluminide</strong>s in ESR and VAR. Titanium 2009 e conferenceproceedings. International <strong>titanium</strong> association. In: Association I.T, editor,editor; 2009.[12] Jarvis D, Voss D. Impress integrated projectean overview paper. MaterialsScience and Engineering A 2005;413e414:583e91.[13] Guo J, Jia J, Liu Y, Liu G, Su Y, Ding H. Evaporation behavior <strong>of</strong> aluminumduring the cold crucible induction skull melting <strong>of</strong> <strong>titanium</strong> aluminum alloys.Metallurgical and Materials Transactions B 2000;31(4):837e44.[14] Kobayashi Y, Tsukihashi F. Thermodynamics <strong>of</strong> yttrium and oxygen in moltenTi, Ti 3 Al, and TiAl. Metallurgical and Materials Transactions B 1998;29(5):1037e42.[15] Tsukihashi F, Hatta T, Tawara E. Thermodynamics <strong>of</strong> calcium and oxygen inmolten <strong>titanium</strong> and <strong>titanium</strong>ealuminum alloy. Metallurgical and MaterialsTransactions B 1996;27(6):967e72.[16] Renjie C, Ming G, Hu Z, Shengkai G. Interactions between TiAl alloys and yttriarefractory material in casting process. Journal <strong>of</strong> Materials Processing Technology2010;210(9):1190e6.[17] Sakamoto K, Yoshikawa K, Kusamichi T, Onoye T. Changes in oxygen contents<strong>of</strong> <strong>titanium</strong> <strong>aluminide</strong>s by vaccum induction, cold crucible induction andelectron beam melting. ISIJ International 1992;32(5).