Solubility of Copper (II) Citraconate
Research paper (pdf) - Harlem Children Society
Research paper (pdf) - Harlem Children Society
- No tags were found...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
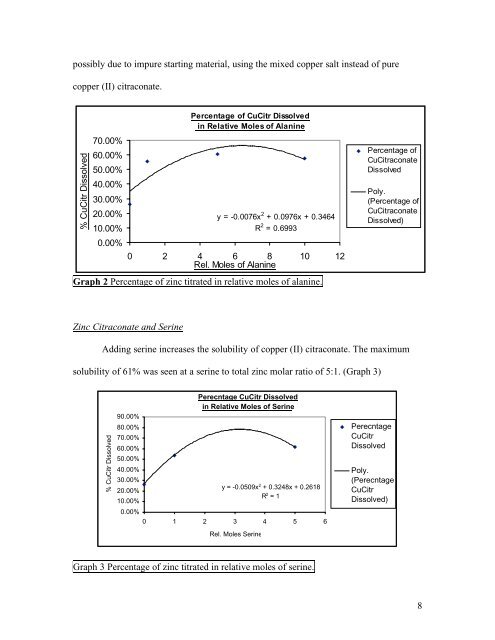
possibly due to impure starting material, using the mixed copper salt instead <strong>of</strong> pure<br />
copper (<strong>II</strong>) citraconate.<br />
Percentage <strong>of</strong> CuCitr Dissolved<br />
in Relative Moles <strong>of</strong> Alanine<br />
% CuCitr Dissolved<br />
70.00%<br />
60.00%<br />
50.00%<br />
40.00%<br />
30.00%<br />
20.00%<br />
10.00%<br />
y = -0.0076x 2 + 0.0976x + 0.3464<br />
R 2 = 0.6993<br />
Percentage <strong>of</strong><br />
Cu<strong>Citraconate</strong><br />
Dissolved<br />
Poly.<br />
(Percentage <strong>of</strong><br />
Cu<strong>Citraconate</strong><br />
Dissolved)<br />
0.00%<br />
0 2 4 6 8 10 12<br />
Rel. Moles <strong>of</strong> Alanine<br />
Graph 2 Percentage <strong>of</strong> zinc titrated in relative moles <strong>of</strong> alanine.<br />
Zinc <strong>Citraconate</strong> and Serine<br />
Adding serine increases the solubility <strong>of</strong> copper (<strong>II</strong>) citraconate. The maximum<br />
solubility <strong>of</strong> 61% was seen at a serine to total zinc molar ratio <strong>of</strong> 5:1. (Graph 3)<br />
% CuCitr Dissolved<br />
Perecntage CuCitr Dissolved<br />
in Relative Moles <strong>of</strong> Serine<br />
90.00%<br />
80.00%<br />
70.00%<br />
60.00%<br />
50.00%<br />
40.00%<br />
30.00%<br />
y = -0.0509x 2 + 0.3248x + 0.2618<br />
20.00%<br />
R 2 = 1<br />
10.00%<br />
0.00%<br />
0 1 2 3 4 5 6<br />
Rel. Moles Serine<br />
Perecntage<br />
CuCitr<br />
Dissolved<br />
Poly.<br />
(Perecntage<br />
CuCitr<br />
Dissolved)<br />
Graph 3 Percentage <strong>of</strong> zinc titrated in relative moles <strong>of</strong> serine.<br />
8