Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
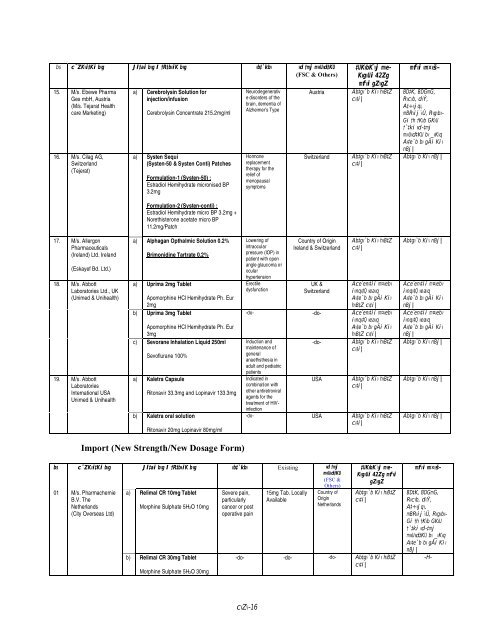
s cÖ¯‘ZKvi‡Ki bvg Jl‡ai bvg I †R‡bwiK bvg wb‡`©kbv wd« †mj mvwU©wd‡KU<br />
(FSC & Others)<br />
15. M/s. Ebewe Pharma<br />
Ges mbH, Austria<br />
(M/s. Tejarat Health<br />
care Marketing)<br />
16. M/s. Cilag AG,<br />
Switzerland<br />
(Tejerat)<br />
17. M/s. Allergon<br />
Pharmaceuticals<br />
(Ireland) Ltd. Ireland<br />
(Eskayef Bd. Ltd.)<br />
18. M/s. Abbott<br />
Laboratories Ltd., UK<br />
(Unimed & Unihealth)<br />
19. M/s. Abbott<br />
Laboratories<br />
International USA<br />
Unimed & Unihealth<br />
a) Cerebrolysin Solution for<br />
injection/infusion<br />
Cerebrolysin Concentrate 215.2mg/ml<br />
a) Systen Sequi<br />
(Systen-50 & Systen Conti) Patches<br />
Formulation-1 (Systen-50) :<br />
Estradiol Hemihydrate micronised BP<br />
3.2mg<br />
Formulation-2 (Systen-conti) :<br />
Estradiol Hemihydrate micro BP 3.2mg +<br />
Norethisterone acetate micro BP<br />
11.2mg/Patch<br />
a) Alphagan Opthalmic Solution 0.2%<br />
Brimonidine Tartrate 0.2%<br />
a) Uprima 2mg Tablet<br />
Apomorphine HCl Hemihydrate Ph. Eur<br />
2mg<br />
b) Uprima 3mg Tablet<br />
Apomorphine HCl Hemihydrate Ph. Eur<br />
3mg<br />
c) Sevorane Inhalation Liquid 250ml<br />
Sevoflurane 100%<br />
a) Kaletra Capsule<br />
Ritonavir 33.3mg and Lopinavir 133.3mg<br />
b) Kaletra oral solution<br />
Ritonavir 20mg Lopinavir 80mg/ml<br />
Import (New Strength/New Dosage Form)<br />
Neurodegenerativ<br />
e disorders of the<br />
brain, dementia of<br />
Alzheimer’s Type<br />
Hormone<br />
replacement<br />
therapy for the<br />
relief of<br />
menopausal<br />
symptoms<br />
Lowering of<br />
intraocular<br />
pressure (IOP) in<br />
patient with open<br />
angle glaucoma or<br />
ocular<br />
hypertension<br />
Erectile<br />
dysfunction<br />
cvZv-16<br />
‡UKwbK¨vj mve-<br />
KwgwUi 42Zg<br />
mfvi gZvgZ<br />
Austria Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
Switzerland Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
Country of Origin<br />
Ireland & Switzerland<br />
UK &<br />
Switzerland<br />
Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
Ace¨env‡ii m¤¢vebv<br />
iwnqv‡Q weavq<br />
Av‡e`b bv gÄyi Kiv<br />
hvB‡Z cv‡i|<br />
-do- -do- Ace¨env‡ii m¤¢vebv<br />
iwnqv‡Q weavq<br />
Av‡e`b bv gÄyi Kiv<br />
hvB‡Z cv‡i|<br />
Induction and<br />
maintenance of<br />
general<br />
anaethsthesia in<br />
adult and pediatric<br />
patients<br />
Indicated in<br />
combination with<br />
other antiretroviral<br />
agents for the<br />
treatment of HIVinfection<br />
-do- Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
USA Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
-do- USA Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
bs cÖ¯‘ZKvi‡Ki bvg Jl‡ai bvg I †R‡bwiK bvg wb‡`©kbv Existing wd« †mj<br />
mvwU©wd‡KU<br />
(FSC &<br />
Others)<br />
01 M/s. Pharmachemie<br />
B.V. The<br />
Netherlands<br />
(City Overseas Ltd)<br />
a) Relimal CR 10mg Tablet<br />
Morphine Sulphate 5H2O 10mg<br />
b) Relimal CR 30mg Tablet<br />
Morphine Sulphate 5H2O 30mg<br />
Severe pain,<br />
particularly<br />
cancer or post<br />
operative pain<br />
15mg Tab. Locally<br />
Available<br />
Country of<br />
Origin<br />
Netherlands<br />
‡UKwbK¨vj mve-<br />
KwgwUi 42Zg mfvi<br />
gZvgZ<br />
Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
-do- -do- -do- Aby‡gv`b Kiv hvB‡Z<br />
cv‡i|<br />
mfvi wm×vš—<br />
BD‡K, BDGmG,<br />
Rvcvb, d«vÝ,<br />
A‡÷ªwjqv,<br />
myBRvij¨vÛ, Rvg©vbx-<br />
Gi †h †Kvb GKwU<br />
†`‡ki wd«-‡mj<br />
mvwU©wd‡KU bv _vKvq<br />
Av‡e`b bv gÄyi Kiv<br />
nBj|<br />
Aby‡gv`b Kiv nBj|<br />
Aby‡gv`b Kiv nBj|<br />
Ace¨env‡ii m¤¢vebv<br />
iwnqv‡Q weavq<br />
Av‡e`b bv gÄyi Kiv<br />
nBj|<br />
Ace¨env‡ii m¤¢vebv<br />
iwnqv‡Q weavq<br />
Av‡e`b bv gÄyi Kiv<br />
nBj|<br />
Aby‡gv`b Kiv nBj|<br />
Aby‡gv`b Kiv nBj|<br />
Aby‡gv`b Kiv nBj|<br />
mfvi wm×vš—<br />
BD‡K, BDGmG,<br />
Rvcvb, d«vÝ,<br />
A‡÷ªwjqv,<br />
myBRvij¨vÛ, Rvg©vbx-<br />
Gi †h †Kvb GKwU<br />
†`‡ki wd«-‡mj<br />
mvwU©wd‡KU bv _vKvq<br />
Av‡e`b bv gÄyi Kiv<br />
nBj|<br />
-H-