Clean, Fast Organic Chemistry - LaborPraxis
Clean, Fast Organic Chemistry - LaborPraxis
Clean, Fast Organic Chemistry - LaborPraxis
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
C(O)CH 3 is an electronwithdrawing<br />
group. Its presence<br />
on the aryl halide makes the<br />
aromatic ring electron-poor.<br />
This makes the carbonbromine<br />
bond weak, and thus,<br />
facilitates palladium insertion.<br />
This makes the coupling easier<br />
than with the other two<br />
substrates used.<br />
Refer to Chapter 3 for more<br />
detailed instructions on setting<br />
up and safely securing your<br />
HP-500 vessel, as well as<br />
information on programming<br />
the MARS System.<br />
The vessel is vented by loosening the blue nut. It is then removed from the frame<br />
and its protective sleeve. The reaction mixture is transferred from the microwave<br />
vessel into a separatory funnel to perform an extraction. Ethyl acetate (30 mL) is<br />
used to rinse the reaction vessel and is added to the separatory funnel along with<br />
water (30 mL). Any solids remaining in the microwave vessel are scraped into the<br />
funnel using a spatula. After stoppering the funnel, the solution is repeatedly<br />
shaken and vented. The layers are then allowed to separate. The aqueous layer is<br />
removed to an Erlenmeyer flask and the organic layer is washed with saturated<br />
sodium chloride solution and dried over sodium sulfate. The ethyl acetate is<br />
removed on a rotary evaporator, leaving a crystalline product. The yield of the<br />
product is determined and the melting point compared to the literature value. The<br />
chemical structure of the product is confirmed by IR, 1H-NMR and/or 13C-NMR<br />
spectroscopy.<br />
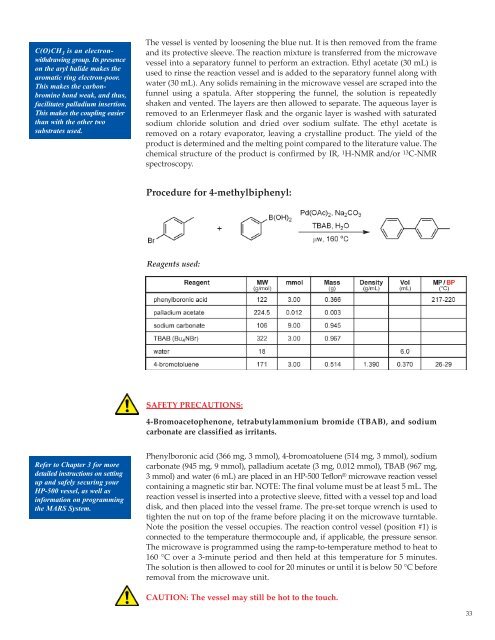
Procedure for 4-methylbiphenyl:<br />
Reagents used:<br />
SAFETY PRECAUTIONS:<br />
4-Bromoacetophenone, tetrabutylammonium bromide (TBAB), and sodium<br />
carbonate are classified as irritants.<br />
Phenylboronic acid (366 mg, 3 mmol), 4-bromoatoluene (514 mg, 3 mmol), sodium<br />
carbonate (945 mg, 9 mmol), palladium acetate (3 mg, 0.012 mmol), TBAB (967 mg,<br />
3 mmol) and water (6 mL) are placed in an HP-500 Teflon ® microwave reaction vessel<br />
containing a magnetic stir bar. NOTE: The final volume must be at least 5 mL. The<br />
reaction vessel is inserted into a protective sleeve, fitted with a vessel top and load<br />
disk, and then placed into the vessel frame. The pre-set torque wrench is used to<br />
tighten the nut on top of the frame before placing it on the microwave turntable.<br />
Note the position the vessel occupies. The reaction control vessel (position #1) is<br />
connected to the temperature thermocouple and, if applicable, the pressure sensor.<br />
The microwave is programmed using the ramp-to-temperature method to heat to<br />
160 °C over a 3-minute period and then held at this temperature for 5 minutes.<br />
The solution is then allowed to cool for 20 minutes or until it is below 50 °C before<br />
removal from the microwave unit.<br />
CAUTION: The vessel may still be hot to the touch.<br />
33