RILSAN® Polyamide 11 in Oil & Gas Off - HCL Fasteners Ltd
RILSAN® Polyamide 11 in Oil & Gas Off - HCL Fasteners Ltd
RILSAN® Polyamide 11 in Oil & Gas Off - HCL Fasteners Ltd
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
3.3 Chemical ag<strong>in</strong>g<br />
In offshore applications, certa<strong>in</strong> offshore<br />
fluids and chemicals can have a detrimental<br />
effect on polyamide <strong>11</strong> performance. For<br />
each application, the specific chemicals<br />
should be reviewed <strong>in</strong> order to estimate<br />
service life.<br />
<strong>Polyamide</strong>s, and <strong>in</strong> particular polyamide<br />
<strong>11</strong>, are very resistant to many types of<br />
chemicals. <strong>Polyamide</strong> <strong>11</strong> is very resistant<br />
to oils and hydrocarbons as well as to a<br />
large variety of solvents. In contrast to<br />
standard polyamides 6 and 66; polyamide<br />
<strong>11</strong> shows only little absorption of water<br />
and is also resistant to diluted acids and<br />
bases. Due to its <strong>in</strong>creased flexibility and<br />
molecular structure, it is also highly resistant<br />
to stress crack<strong>in</strong>g, unlike most other thermoplastics.<br />
<strong>Polyamide</strong> <strong>11</strong> can be used <strong>in</strong> conjunction<br />
with a great variety of standard offshore<br />
chemicals. A detailed description of compatibilities<br />
is given <strong>in</strong> sections 3.8 and 3.9.<br />
Because chemical species attack thermoplastic<br />
res<strong>in</strong>s when they are absorbed,<br />
diffusion and solubility play important roles<br />
<strong>in</strong> the assessment of chemical compatibility.<br />
There are two effects <strong>in</strong>duced by absorbed<br />
species – an <strong>in</strong>fluence on the mechanical<br />
properties due to plasticization, and a<br />
chemical effect lead<strong>in</strong>g to loss of material<br />
performance.<br />
Specific examples of absorption and<br />
plasticizer extraction are given <strong>in</strong> sections<br />
3.6 and 3.7 on methanol-and glycol-based<br />
hydraulic liquids.<br />
The ma<strong>in</strong> chemical effect is reduction <strong>in</strong><br />
polymer molecular weight due to hydrolysis.<br />
Hydrolysis is the reverse reaction of the<br />
cha<strong>in</strong>-form<strong>in</strong>g polycondensation reaction.<br />
It can be <strong>in</strong>duced by water at elevated<br />
temperatures and is accelerated by acids<br />
and, to some extent, also by bases. Due to<br />
the importance of hydrolysis <strong>in</strong> ag<strong>in</strong>g related<br />
to offshore applications, section 3.5<br />
describes the phenomenon <strong>in</strong> detail.<br />
ELONGATION AT BREAK (%)<br />
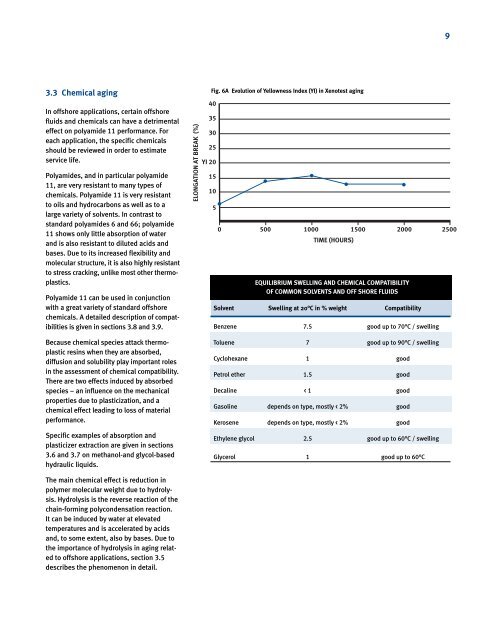
Fig. 6A Evolution of Yellowness Index (YI) <strong>in</strong> Xenotest ag<strong>in</strong>g<br />
40<br />
35<br />
30<br />
25<br />
YI 20<br />
15<br />
10<br />
5<br />
•<br />
0<br />
•<br />
•<br />
500 1000 1500 2000 2500<br />
TIME (HOURS)<br />
EQUILIBRIUM SWELLING AND CHEMICAL COMPATIBILITY<br />
OF COMMON SOLVENTS AND OFF SHORE FLUIDS<br />
Solvent Swell<strong>in</strong>g at 20°C <strong>in</strong> % weight Compatibility<br />
Benzene 7.5 good up to 70°C / swell<strong>in</strong>g<br />
Toluene 7 good up to 90°C / swell<strong>in</strong>g<br />
Cyclohexane 1 good<br />
Petrol ether 1.5 good<br />
Decal<strong>in</strong>e < 1 good<br />
<strong>Gas</strong>ol<strong>in</strong>e depends on type, mostly < 2% good<br />
Kerosene depends on type, mostly < 2% good<br />
Ethylene glycol 2.5 good up to 60°C / swell<strong>in</strong>g<br />
Glycerol 1 good up to 60°C<br />
•<br />
•<br />
9