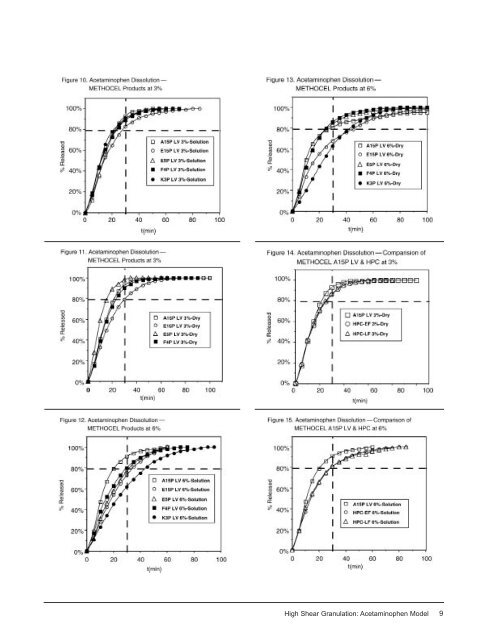
The dry binder addition method (see Table 4) also produced tablets <strong>with</strong> excellent hardness overall. The results were somewhat mixed concerning which binder level gave the harder tablets (in 5 of the 9 cases, the tablets at 3% binder were harder). At the 3% binder concentration, the hardest tablets were made <strong>with</strong> the PVP (K90), METHOCEL F4P LV, HPC-LF, and PVP (K29-32) materials. Note that these are essentially the same binders and essentially the same hardness values that were obtained by the solution addition method. This was a somewhat surprising result, since it is generally considered that binders are more effective when they are placed in solution prior to the granulation operation. Similarly, at the 6% level, the best binders were PVP (K29-32), PVP (K90) ≈ HPC-LF, HPC- EF, and METHOCEL K3P LV. In terms of friability, the lowest losses were predominantly from tablets containing 3% binders, namely 3% METHOCEL A15P LV, 3% METHOCEL F4P LV ≈ 3% and 6% HPC-EF, and 3% PVP (K90). The highest friabilities, which were still less than 0.50%, resulted from the use of 6% METHOCEL E15P LV and A15P LV. The statistical significance of differences in the two binder addition methods was examined. Due to the differences in the formulations, only comparisons between 3% solution/3% dry and 6% solution/6% dry were made. Of the 17 sets of data (results of the 3% METHOCEL K3P LV excluded), the dry binder addition method was statistically harder in 7 cases: 3% METHOCEL A15P LV, E15P LV, and F4P LV, 6% HPC-EF and -LF, 6% PVP (K29-32), and 3% PVP (K90). The solution binder addition method produced 8 <strong>High</strong> <strong>Shear</strong> <strong>Granulation</strong>: Acetaminophen Model statistically harder tablets in 4 cases: 6% METHOCEL A15P LV and E15P LV, and 3% HPC-EF and -LF. The remaining 6 cases showed no statistically significant difference in hardness between the binder addition methods. As mentioned on page 3, the formulations were compressed at 1000, 2000, and 3000 lbs. total compression force. For both binder addition methods, tablet hardness increased <strong>with</strong> force <strong>with</strong> very few exceptions, those being 6% acacia (hardness was essentially constant), 10% pregelatinized starch (tablets compressed at 3000 lbs. were about 5 SCU softer than those compressed at 2000 lbs.), and the 3% K3P LV (solution) and 3% PVP (K29-32) (dry) tablets (which had slightly softer tablets produced at 3000 lbs. compression compared to 2000 lbs. compression). The friabilities were measured at 2, 4, and 6 minutes at each force. At a given force, the percent weight loss naturally increased <strong>with</strong> time. At a given time (e.g., 4 minutes), there was the expected decrease in friability as the compression force, and thereby the tablet hardness increased. There was a proportionally smaller decrease in friability between 2000 lbs. and 3000 lbs. than there was between 1000 lbs. and 2000 lbs. There were a few cases in which the friability of tablets compressed at 3000 lbs. was greater than those compressed at 2000 lbs. despite the fact that the one compressed at the higher force had a higher hardness. One example of this behavior is 3% PVP (K29-32) (solution), where the hardness and friability at 2000 lbs. compression force were 20.9 SCU and 0.37%, while the hardness and friability at 3000 lbs. compression force were 21.9 SCU and 0.75%, respectively. The weight and thickness variation of the tablets for each formulation and binder addition method were also measured (n = 20). The thickness variation in all cases was excellent, varying from a low of 0.12% relative standard deviation (RSD, equal to the standard deviation/mean x 100) for the 6% METHOCEL F4P LV (solution method) formulation, to 0.84% RSD for the 3% HPC-EF (solution) formula; the range of thickness variation for the dry addition method was 0.18–0.67% RSD. Similarly, the weight variation was very good for the majority of the formulations, varying from 0.47% RSD for the 3% METHOCEL F4P LV (dry) formula, to 1.39% RSD for the 6% METHOCEL E15P LV (solution) case. The binders that performed well in one of the binder addition methods tended to perform well in the other addition method as well. Tablet Dissolution Properties The in vitro dissolution properties varied both as a function of the nature of the binder and the amount of binder in the formulation, but relatively little as a function of the binder addition method. The USP dissolution conditions for acetaminophen tablets were used (Type 2 apparatus at 50 rpm, 900 mL of pH 5.8 phosphate buffer). The time to reach 80% dissolved is designated by t 80% . A number of these dissolutions are presented in Figures 10-18.
<strong>High</strong> <strong>Shear</strong> <strong>Granulation</strong>: Acetaminophen Model 9