Brassica campestris L. ssp. chinensis M
Brassica campestris L. ssp. chinensis M
Brassica campestris L. ssp. chinensis M
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
452<br />
plants. Control of pollen acceptance by adhesion is<br />
particularly important in species with a dry stigma,<br />
such as members of the <strong>Brassica</strong>ceae. In addition,<br />
PCPs are an extensive family characterized by high<br />
polymorphism and are gametophysically expressed<br />
small cysteine-rich proteins. To date, PCP Class A<br />
(PCP-A) is the only group of pollen coat proteins<br />
identified that is suggested to play a key role in the<br />
pollen–stigma interaction and pollen recognition. It<br />
is also highly polymorphic around the conserved<br />
cysteine backbone (Doughty et al., 2000). In this<br />
study, we isolated and characterized a novel<br />
member of the PCP-A family featuring eight<br />
conservative cysteine residues from the GMS A/B<br />
line ‘ZUBajh97-01A/B,’ named as BcMF5. The<br />
size of its intron and its deduced amino acid<br />
sequence were found to be identical to PCPA2 and<br />
ARTICLE IN PRESS<br />
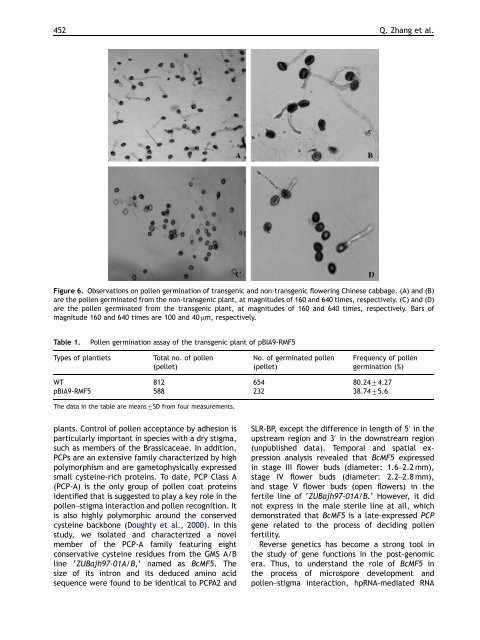
Figure 6. Observations on pollen germination of transgenic and non-transgenic flowering Chinese cabbage. (A) and (B)<br />
are the pollen germinated from the non-transgenic plant, at magnitudes of 160 and 640 times, respectively. (C) and (D)<br />
are the pollen germinated from the transgenic plant, at magnitudes of 160 and 640 times, respectively. Bars of<br />
magnitude 160 and 640 times are 100 and 40 mm, respectively.<br />
Table 1. Pollen germination assay of the transgenic plant of pBIA9-RMF5<br />
Types of plantlets Total no. of pollen<br />
(pellet)<br />
No. of germinated pollen<br />
(pellet)<br />
Frequency of pollen<br />
germination (%)<br />
WT 812 654 80.2474.27<br />
pBIA9-RMF5 588 232 38.7475.6<br />
The data in the table are means7SD from four measurements.<br />
Q. Zhang et al.<br />
SLR-BP, except the difference in length of 5 0 in the<br />
upstream region and 3 0 in the downstream region<br />
(unpublished data). Temporal and spatial expression<br />
analysis revealed that BcMF5 expressed<br />
in stage III flower buds (diameter: 1.6–2.2 mm),<br />
stage IV flower buds (diameter: 2.2–2.8 mm),<br />
and stage V flower buds (open flowers) in the<br />
fertile line of ‘ZUBajh97-01A/B.’ However, it did<br />
not express in the male sterile line at all, which<br />
demonstrated that BcMF5 is a late-expressed PCP<br />
gene related to the process of deciding pollen<br />
fertility.<br />
Reverse genetics has become a strong tool in<br />
the study of gene functions in the post-genomic<br />
era. Thus, to understand the role of BcMF5 in<br />
the process of microspore development and<br />
pollen–stigma interaction, hpRNA-mediated RNA