Download PDF - SynCardia Systems, Inc.
Download PDF - SynCardia Systems, Inc.
Download PDF - SynCardia Systems, Inc.
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
2<br />
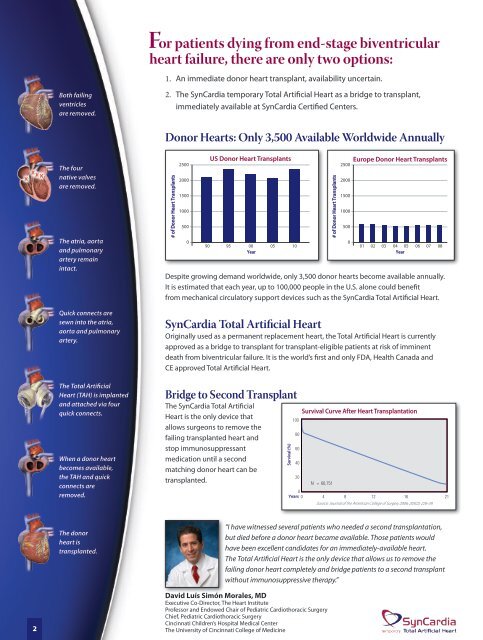
Both failing<br />
ventricles<br />
are removed.<br />
The four<br />
native valves<br />
are removed.<br />
The atria, aorta<br />
and pulmonary<br />
artery remain<br />
intact.<br />
Quick connects are<br />
sewn into the atria,<br />
aorta and pulmonary<br />
artery.<br />
The Total Artificial<br />
Heart (TAH) is implanted<br />
and attached via four<br />
quick connects.<br />
When a donor heart<br />
becomes available,<br />
the TAH and quick<br />
connects are<br />
removed.<br />
The donor<br />
heart is<br />
transplanted.<br />
For patients dying from end-stage biventricular<br />
heart failure, there are only two options:<br />
1. An immediate donor heart transplant, availability uncertain.<br />
2. The <strong>SynCardia</strong> temporary Total Artificial Heart as a bridge to transplant,<br />
Donor Hearts: Only 3,500 Available Worldwide Annually<br />
# of Donor Heart Transplants<br />
immediately available at <strong>SynCardia</strong> Certified Centers.<br />
2500 –<br />
2000 –<br />
1500 –<br />
1000 –<br />
500 –<br />
US Donor Heart Transplants<br />
0 – 90 95 00 05 10<br />
Year<br />
Despite growing demand worldwide, only 3,500 donor hearts become available annually.<br />
It is estimated that each year, up to 100,000 people in the U.S. alone could benefit<br />
from mechanical circulatory support devices such as the <strong>SynCardia</strong> Total Artificial Heart.<br />
<strong>SynCardia</strong> Total Artificial Heart<br />
Originally used as a permanent replacement heart, the Total Artificial Heart is currently<br />
approved as a bridge to transplant for transplant-eligible patients at risk of imminent<br />
death from biventricular failure. It is the world’s first and only FDA, Health Canada and<br />
CE approved Total Artificial Heart.<br />
Bridge to Second Transplant<br />
The <strong>SynCardia</strong> Total Artificial<br />
Heart is the only device that<br />
allows surgeons to remove the<br />
failing transplanted heart and<br />
stop immunosuppressant<br />
medication until a second<br />
matching donor heart can be<br />
transplanted.<br />
“I have witnessed several patients who needed a second transplantation,<br />
but died before a donor heart became available. Those patients would<br />
have been excellent candidates for an immediately-available heart.<br />
The Total Artificial Heart is the only device that allows us to remove the<br />
failing donor heart completely and bridge patients to a second transplant<br />
without immunosuppressive therapy.”<br />
David Luís Simón Morales, MD<br />
Executive Co-Director, The Heart Institute<br />
Professor and Endowed Chair of Pediatric Cardiothoracic Surgery<br />
Chief, Pediatric Cardiothoracic Surgery<br />
Cincinnati Children’s Hospital Medical Center<br />
The University of Cincinnati College of Medicine<br />
Survival (%)<br />
100<br />
80<br />
60<br />
40<br />
20<br />
Survival Curve After Heart Transplantation<br />
N = 66,751<br />
# of Donor Heart Transplants<br />
Europe Donor Heart Transplants<br />
2500 –<br />
2000 –<br />
1500 –<br />
1000 –<br />
500 –<br />
0 – 01 02 03 04 05 06 07 08<br />
Year<br />
0<br />
Years 0 4 8 12 16 21<br />
Source: Journal of the American College of Surgery 2006; 203(2): 226-39.