The Principal Investigator's Responsibilities in Research
The Principal Investigator's Responsibilities in Research
The Principal Investigator's Responsibilities in Research
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
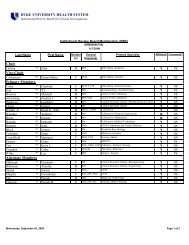
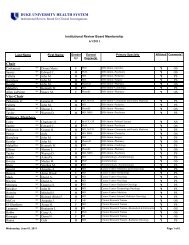
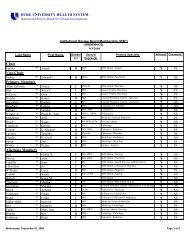
DUHS PI <strong>Responsibilities</strong><br />
15. Will conduct research <strong>in</strong>volv<strong>in</strong>g FDA-regulated FDA regulated products<br />
under an IND application or IDE,<br />
Will comply with all applicable FDA regulations and fulfill<br />
all <strong>in</strong>vestigator responsibilities [or <strong>in</strong>vestigator-sponsor<br />
<strong>in</strong>vestigator sponsor<br />
responsibilities, where appropriate], <strong>in</strong>clud<strong>in</strong>g those<br />
described at 21 CFR 312 and 812, the text of which is<br />
found at:<br />
http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr<br />
/cfrsearch.cfm





![Human Research Protection Program [Dr. Wesley Byerly]](https://img.yumpu.com/50293157/1/190x143/human-research-protection-program-dr-wesley-byerly.jpg?quality=85)