Ketamine in Chronic Pain - McMaster University
Ketamine in Chronic Pain - McMaster University
Ketamine in Chronic Pain - McMaster University
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
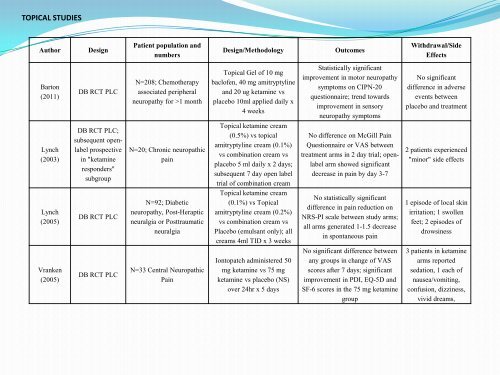
TOPICAL STUDIES<br />
Author Design<br />
Barton<br />
(2011)<br />
Lynch<br />
(2003)<br />
Lynch<br />
(2005)<br />
Vranken<br />
(2005)<br />
DB RCT PLC<br />
DB RCT PLC;<br />
subsequent openlabel<br />
prospective<br />
<strong>in</strong> "ketam<strong>in</strong>e<br />
responders"<br />
subgroup<br />
DB RCT PLC<br />
DB RCT PLC<br />
Patient population and<br />
numbers<br />
N=208; Chemotherapy<br />
associated peripheral<br />
neuropathy for >1 month<br />
N=20; <strong>Chronic</strong> neuropathic<br />
pa<strong>in</strong><br />
N=92; Diabetic<br />
neuropathy, Post-Heraptic<br />
neuralgia or Posttraumatic<br />
neuralgia<br />
N=33 Central Neuropathic<br />
Pa<strong>in</strong><br />
Design/Methodology Outcomes<br />
Topical Gel of 10 mg<br />
baclofen, 40 mg amitryptyl<strong>in</strong>e<br />
and 20 ug ketam<strong>in</strong>e vs<br />
placebo 10ml applied daily x<br />
4 weeks<br />
Topical ketam<strong>in</strong>e cream<br />
(0.5%) vs topical<br />
amitryptyl<strong>in</strong>e cream (0.1%)<br />
vs comb<strong>in</strong>ation cream vs<br />
placebo 5 ml daily x 2 days;<br />
subsequent 7 day open label<br />
trial of comb<strong>in</strong>ation cream<br />
Topical ketam<strong>in</strong>e cream<br />
(0.1%) vs Topical<br />
amitryptyl<strong>in</strong>e cream (0.2%)<br />
vs comb<strong>in</strong>ation cream vs<br />
Placebo (emulsant only); all<br />
creams 4ml TID x 3 weeks<br />
Iontopatch adm<strong>in</strong>istered 50<br />
mg ketam<strong>in</strong>e vs 75 mg<br />
ketam<strong>in</strong>e vs placebo (NS)<br />
over 24hr x 5 days<br />
Statistically significant<br />
improvement <strong>in</strong> motor neuropathy<br />
symptoms on CIPN-20<br />
questionnaire; trend towards<br />
improvement <strong>in</strong> sensory<br />
neuropathy symptoms<br />
No difference on McGill Pa<strong>in</strong><br />
Questionnaire or VAS between<br />
treatment arms <strong>in</strong> 2 day trial; openlabel<br />
arm showed significant<br />
decrease <strong>in</strong> pa<strong>in</strong> by day 3-7<br />
No statistically significant<br />
difference <strong>in</strong> pa<strong>in</strong> reduction on<br />
NRS-PI scale between study arms;<br />
all arms generated 1-1.5 decrease<br />
<strong>in</strong> spontaneous pa<strong>in</strong><br />
No significant difference between<br />
any groups <strong>in</strong> change of VAS<br />
scores after 7 days; significant<br />
improvement <strong>in</strong> PDI, EQ-5D and<br />
SF-6 scores <strong>in</strong> the 75 mg ketam<strong>in</strong>e<br />
group<br />
Withdrawal/Side<br />
Effects<br />
No significant<br />
difference <strong>in</strong> adverse<br />
events between<br />
placebo and treatment<br />
2 patients experienced<br />
"m<strong>in</strong>or" side effects<br />
1 episode of local sk<strong>in</strong><br />
irritation; 1 swollen<br />
feet; 2 episodes of<br />
drows<strong>in</strong>ess<br />
3 patients <strong>in</strong> ketam<strong>in</strong>e<br />
arms reported<br />
sedation, 1 each of<br />
nausea/vomit<strong>in</strong>g,<br />
confusion, dizz<strong>in</strong>ess,<br />
vivid dreams,