Sucrose
Sucrose
Sucrose
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Sucrose</strong><br />
Go To: Top, Reaction thermochemistry data, References, Notes / Error Report<br />
http://webbook.nist.gov/cgi/cbook.cgi?Name=sucrose&Units=SI&cTC=on&cTR=on#ref-3<br />
02/23/2007 05:54 AM<br />
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights<br />
reserved.<br />
Data compiled as indicated in comments:<br />
ALS - H.Y. Afeefy, J.F. Liebman, and S.E. Stein<br />
DH - E.S. Domalski and E.D. Hearing<br />
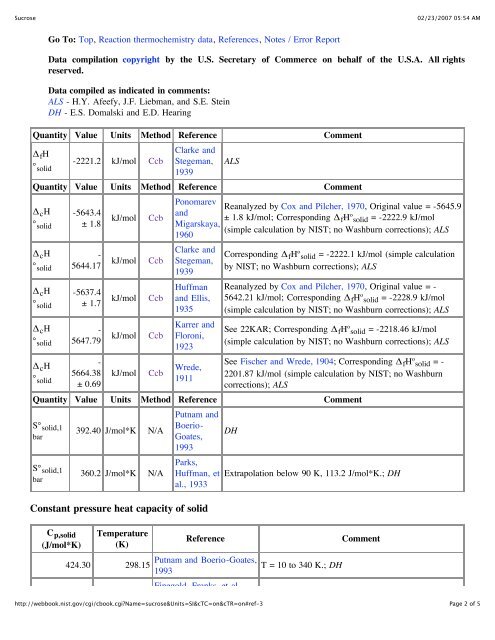
Quantity Value Units Method Reference Comment<br />
Δ fH<br />
° solid<br />
-2221.2 kJ/mol Ccb<br />
Clarke and<br />
Stegeman,<br />
1939<br />
Quantity Value Units Method Reference Comment<br />
Δ cH<br />
° solid<br />
Δ cH<br />
° solid<br />
ΔcH<br />
° solid<br />
Δ cH<br />
° solid<br />
Δ cH<br />
° solid<br />
-5643.4<br />
± 1.8<br />
-<br />
5644.17<br />
-5637.4<br />
± 1.7<br />
-<br />
5647.79<br />
-<br />
5664.38<br />
± 0.69<br />
kJ/mol Ccb<br />
kJ/mol Ccb<br />
kJ/mol Ccb<br />
kJ/mol Ccb<br />
Ponomarev<br />
and<br />
Migarskaya,<br />
1960<br />
Clarke and<br />
Stegeman,<br />
1939<br />
Huffman<br />
and Ellis,<br />
1935<br />
Karrer and<br />
Floroni,<br />
1923<br />
kJ/mol Ccb Wrede,<br />
1911<br />
ALS<br />
Reanalyzed by Cox and Pilcher, 1970, Original value = -5645.9<br />
± 1.8 kJ/mol; Corresponding Δ fHº solid = -2222.9 kJ/mol<br />
(simple calculation by NIST; no Washburn corrections); ALS<br />
Corresponding Δ fHº solid = -2222.1 kJ/mol (simple calculation<br />
by NIST; no Washburn corrections); ALS<br />
Reanalyzed by Cox and Pilcher, 1970, Original value = -<br />
5642.21 kJ/mol; Corresponding Δ fHº solid = -2228.9 kJ/mol<br />
(simple calculation by NIST; no Washburn corrections); ALS<br />
See 22KAR; Corresponding Δ fHº solid = -2218.46 kJ/mol<br />
(simple calculation by NIST; no Washburn corrections); ALS<br />
See Fischer and Wrede, 1904; Corresponding Δ fHº solid = -<br />
2201.87 kJ/mol (simple calculation by NIST; no Washburn<br />
corrections); ALS<br />
Quantity Value Units Method Reference Comment<br />
S° solid,1<br />
bar<br />
S° solid,1<br />
bar<br />
392.40 J/mol*K N/A<br />
360.2 J/mol*K N/A<br />
Putnam and<br />
Boerio-<br />
Goates,<br />
1993<br />
Parks,<br />
Huffman, et<br />
al., 1933<br />
Constant pressure heat capacity of solid<br />
C p,solid<br />
(J/mol*K)<br />
Temperature<br />
(K)<br />
424.30 298.15<br />
DH<br />
Extrapolation below 90 K, 113.2 J/mol*K.; DH<br />
Reference Comment<br />
Putnam and Boerio-Goates,<br />
1993<br />
Finegold, Franks, et al.,<br />
T = 10 to 340 K.; DH<br />
Page 2 of 5


![Experiment 9 Research Into Alternate Syntheses of [Cr(en)3]Cl3 ...](https://img.yumpu.com/18756539/1/190x245/experiment-9-research-into-alternate-syntheses-of-cren3cl3-.jpg?quality=85)