Facility Name Address Phone_____ - Nevada State Health Division
Facility Name Address Phone_____ - Nevada State Health Division
Facility Name Address Phone_____ - Nevada State Health Division
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
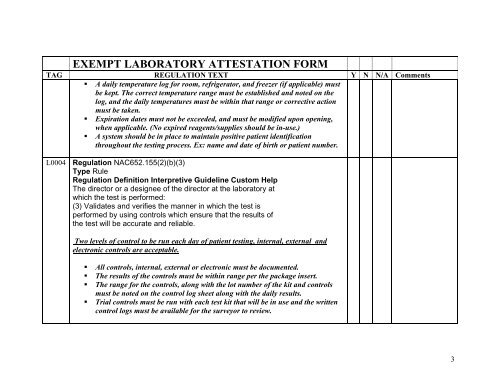
EXE MPT LABORATORY ATTESTATION FORM<br />
TAG REGULATION TEXT<br />
Y N N/A Comments<br />
A daily temperature log for room, refrigerator, and freezer (if applicable) must<br />
be kept. The correct temperature range must be established and noted on the<br />
log, and the daily temperatures must be within that range or corrective action<br />
must be taken.<br />
Expiration dates must not be exceeded, and must be modified upon opening,<br />
when applicable. (No expired reagents/supplies should be in-use.)<br />
A system should be in place to maintain positive patient identification<br />
throughout the testing process. Ex: name and date of birth or patient number.<br />
L0004 Regulation NAC652.155(2)(b)(3)<br />
Type Rule<br />
Regulation Definition Interpretive Guideline Custom Help<br />
The director or a designee of the director at the laboratory at<br />
which the test is performed:<br />
(3) Validates and verifies the manner in which the test is<br />
performed by using controls which ensure that the results of<br />
the test will be accurate and reliable.<br />
Two levels of control to be run each day of patient testing, internal, external and<br />
electronic controls are acceptable.<br />
All controls, internal, external or electronic must be documented.<br />
The results of the controls must be within range per the package insert.<br />
The range for the controls, along with the lot number of the kit and controls<br />
must be noted on the control log sheet along with the daily results.<br />
Trial controls must be run with each test kit that will be in use and the written<br />
control logs must be available for the surveyor to review.<br />
3