White Paper on Dietary Supplements
White Paper on Dietary Supplements
White Paper on Dietary Supplements
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
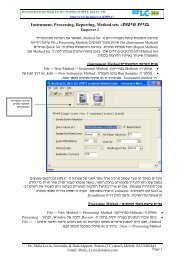
Final FDA Rule <strong>on</strong> <strong>Dietary</strong> <strong>Supplements</strong> (21 CFR Part 111)<br />
<str<strong>on</strong>g>White</str<strong>on</strong>g> <str<strong>on</strong>g>Paper</str<strong>on</strong>g><br />
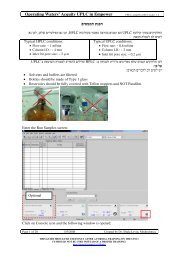
Benefits of customer complaint software: A customer complaint software soluti<strong>on</strong> automates the<br />
process of documenting, reviewing and, when necessary, investigating customer complaints. The<br />
process of investigating complaints is greatly expedited by the ability to launch a review/approval<br />
routing (with all relevant documents attached). This prevents delays in resolving complaints due to the<br />
paperwork not being complete.<br />
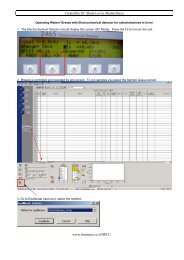
Another benefit is that complaint records stored in secure, centralized electr<strong>on</strong>ic files can be easily<br />
retrieved by authorized users. They can also be automatically retrieved by the system to print reports,<br />
perform analyses, etc., which help companies to see trends and resp<strong>on</strong>d proactively.<br />
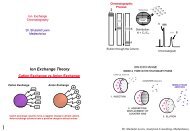
In summary, an automated document c<strong>on</strong>trol/management system and automated processes can help<br />
assure quality and regulatory compliance, proper documentati<strong>on</strong>, and greatly speed up the processes<br />
involved in the producti<strong>on</strong> of dietary supplements.<br />
References<br />
1. FDA informati<strong>on</strong> about the recall can be found at the following URL, downloaded March 25, 2008,<br />
from http://www.fda.gov/oc/po/firmrecalls/nasutra09_06.html<br />
2.<br />
FDA informati<strong>on</strong> about the recall can be found at the following URL, downloaded March 25, 2008,<br />
from http://www.fda.gov/oc/po/firmrecalls/fiberchoice03_07.html<br />
3. <strong>Dietary</strong> Supplement Current Good Manufacturing Practices (CGMPs) and Interim Final Rule (IFR)<br />
Facts. Downloaded March 25, 2008, from the following URL: www.cfsan.fda.gov/~dms/dscgmps6.<br />
html<br />
4. Mei-Chih Lin, Yoe-Ray Ku, Yi-Chu Liu, Jer-Huei Lin. P-090: Isolati<strong>on</strong> and Identificati<strong>on</strong> of a<br />
novel Sildenafil analogue adulterated in herbal supplements. Bureau of Food and Drug Analysis,<br />
Department of Health, Taipei, Taiwan. Retrieved: March 31, 2008 from http://www.phcog.org/<br />
AnnualMtg/2006/papers/P_090.pdf<br />
5. Guidance for Industry: Questi<strong>on</strong>s and Answers Regarding Adverse Event Reporting and<br />
Recordkeeping for <strong>Dietary</strong> <strong>Supplements</strong> as Required by the <strong>Dietary</strong> Supplement and<br />
N<strong>on</strong>prescripti<strong>on</strong> Drug C<strong>on</strong>sumer Protecti<strong>on</strong> Act. Downloaded <strong>on</strong> March 25, 2008, from http://www.<br />
cfsan.fda.gov/~dms/dsaergui.html#def<br />
6. Adverse event reporting form. Retrieved March 31, 2008 from the following URL: http://www.fda.<br />
gov/medwatch/getforms.htm.<br />
10