Pharmacy
Pharmacy
Pharmacy
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
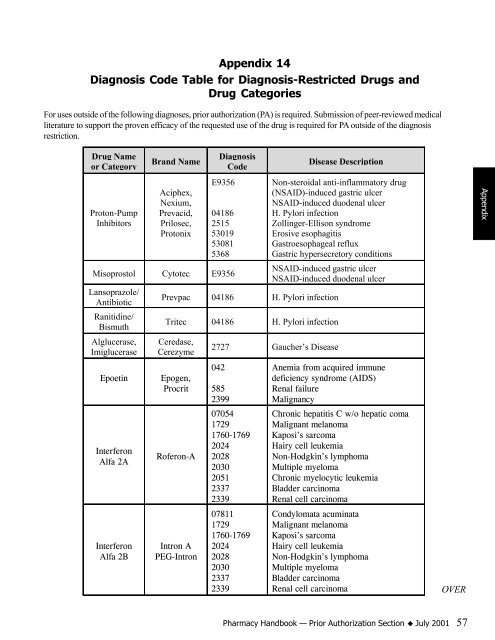
Appendix 14<br />
Diagnosis Code Table for Diagnosis-Restricted Drugs and<br />
Drug Categories<br />
For uses outside of the following diagnoses, prior authorization (PA) is required. Submission of peer-reviewed medical<br />
literature to support the proven efficacy of the requested use of the drug is required for PA outside of the diagnosis<br />
restriction.<br />
Drug Name<br />
or Category<br />
Proton-Pump<br />
Inhibitors<br />
Brand Name<br />
Aciphex,<br />
Nexium,<br />
Prevacid,<br />
Prilosec,<br />
Protonix<br />
Diagnosis<br />
Code<br />
E9356<br />
04186<br />
2515<br />
53019<br />
53081<br />
5368<br />
Misoprostol Cytotec E9356<br />
Lansoprazole/<br />
Antibiotic<br />
Ranitidine/<br />
Bismuth<br />
Alglucerase,<br />
Imiglucerase<br />
Disease Description<br />
Non-steroidal anti-inflammatory drug<br />
(NSAID)-induced gastric ulcer<br />
NSAID-induced duodenal ulcer<br />
H. Pylori infection<br />
Zollinger-Ellison syndrome<br />
Erosive esophagitis<br />
Gastroesophageal reflux<br />
Gastric hypersecretory conditions<br />
NSAID-induced gastric ulcer<br />
NSAID-induced duodenal ulcer<br />
Prevpac 04186 H. Pylori infection<br />
Tritec 04186 H. Pylori infection<br />
Ceredase,<br />
Cerezyme<br />
Epoetin Epogen,<br />
Procrit<br />
Interferon<br />
Alfa 2A<br />
Interferon<br />
Alfa 2B<br />
Roferon-A<br />
Intron A<br />
PEG-Intron<br />
2727 Gaucher’s Disease<br />
042<br />
585<br />
2399<br />
07054<br />
1729<br />
1760-1769<br />
2024<br />
2028<br />
2030<br />
2051<br />
2337<br />
2339<br />
07811<br />
1729<br />
1760-1769<br />
2024<br />
2028<br />
2030<br />
2337<br />
2339<br />
Anemia from acquired immune<br />
deficiency syndrome (AIDS)<br />
Renal failure<br />
Malignancy<br />
Chronic hepatitis C w/o hepatic coma<br />
Malignant melanoma<br />
Kaposi’s sarcoma<br />
Hairy cell leukemia<br />
Non-Hodgkin’s lymphoma<br />
Multiple myeloma<br />
Chronic myelocytic leukemia<br />
Bladder carcinoma<br />
Renal cell carcinoma<br />
Condylomata acuminata<br />
Malignant melanoma<br />
Kaposi’s sarcoma<br />
Hairy cell leukemia<br />
Non-Hodgkin’s lymphoma<br />
Multiple myeloma<br />
Bladder carcinoma<br />
Renal cell carcinoma<br />
OVER<br />
<strong>Pharmacy</strong> Handbook — Prior Authorization Section K July 2001 57<br />
Appendix