Download pdf of current issue - GGH Journal
Download pdf of current issue - GGH Journal
Download pdf of current issue - GGH Journal
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
GROWTH, GENETICS & HORMONES<br />
*<br />
Mnll<br />
insulin-like growth factor<br />
(IGF)-I in the 2 families<br />
may be related to the<br />
well-known limitations<br />
<strong>of</strong> the <strong>current</strong> methods<br />
<strong>of</strong> clinical investigation.<br />
This study, along with the<br />
mutated murine models,<br />
supports the (debated)<br />
view that ghrelin and<br />
GH/IGF-I interact in the<br />
control <strong>of</strong> growth.<br />
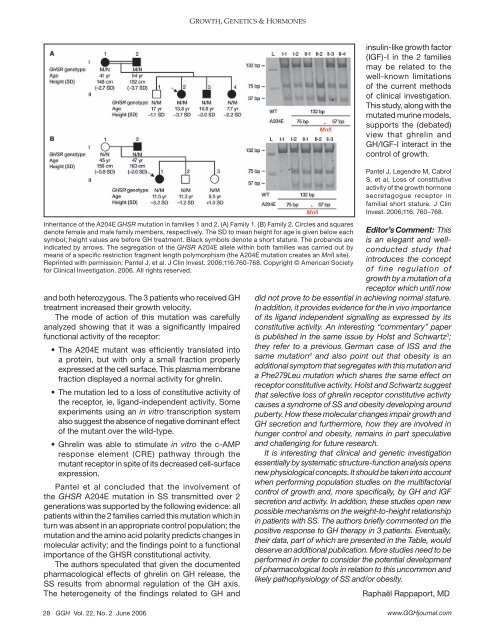
Inheritance <strong>of</strong> the A204E GHSR mutation in families 1 and 2. (A) Family 1. (B) Family 2. Circles and squares<br />
denote female and male family members, respectively. The SD to mean height for age is given below each<br />
symbol; height values are before GH treatment. Black symbols denote a short stature. The probands are<br />
indicated by arrows. The segregation <strong>of</strong> the GHSR A204E allele within both families was carried out by<br />
means <strong>of</strong> a specific restriction fragment length polymorphism (the A204E mutation creates an MnlI site).<br />
Reprinted with permission: Pantel J, et al. J Clin Invest. 2006;116:760-768. Copyright © American Society<br />
for Clinical Investigation. 2006. All rights reserved.<br />
and both heterozygous. The 3 patients who received GH<br />
treatment increased their growth velocity.<br />
The mode <strong>of</strong> action <strong>of</strong> this mutation was carefully<br />
analyzed showing that it was a significantly impaired<br />
functional activity <strong>of</strong> the receptor:<br />
• The A204E mutant was efficiently translated into<br />
a protein, but with only a small fraction properly<br />
expressed at the cell surface. This plasma membrane<br />
fraction displayed a normal activity for ghrelin.<br />
• The mutation led to a loss <strong>of</strong> constitutive activity <strong>of</strong><br />
the receptor, ie, ligand-independent activity. Some<br />
experiments using an in vitro transcription system<br />
also suggest the absence <strong>of</strong> negative dominant effect<br />
<strong>of</strong> the mutant over the wild-type.<br />
• Ghrelin was able to stimulate in vitro the c-AMP<br />
response element (CRE) pathway through the<br />
mutant receptor in spite <strong>of</strong> its decreased cell-surface<br />
expression.<br />
Pantel et al concluded that the involvement <strong>of</strong><br />
the GHSR A204E mutation in SS transmitted over 2<br />
generations was supported by the following evidence: all<br />
patients within the 2 families carried this mutation which in<br />
turn was absent in an appropriate control population; the<br />
mutation and the amino acid polarity predicts changes in<br />
molecular activity; and the findings point to a functional<br />
importance <strong>of</strong> the GHSR constitutional activity.<br />
The authors speculated that given the documented<br />
pharmacological effects <strong>of</strong> ghrelin on GH release, the<br />
SS results from abnormal regulation <strong>of</strong> the GH axis.<br />
The heterogeneity <strong>of</strong> the findings related to GH and<br />
Pantel J, Legendre M, Cabrol<br />
S, et al. Loss <strong>of</strong> constitutive<br />
activity <strong>of</strong> the growth hormone<br />
secretagogue receptor in<br />
familial short stature. J Clin<br />
Invest. 2006;116: 760−768.<br />
Editor’s Comment: This<br />
is an elegant and wellconducted<br />
study that<br />
introduces the concept<br />
<strong>of</strong> fine regulation <strong>of</strong><br />
growth by a mutation <strong>of</strong> a<br />
receptor which until now<br />
did not prove to be essential in achieving normal stature.<br />
In addition, it provides evidence for the in vivo importance<br />
<strong>of</strong> its ligand independent signalling as expressed by its<br />
constitutive activity. An interesting “commentary” paper<br />
is published in the same <strong>issue</strong> by Holst and Schwartz 3 ;<br />
they refer to a previous German case <strong>of</strong> ISS and the<br />
same mutation 4 and also point out that obesity is an<br />
additional symptom that segregates with this mutation and<br />
a Phe279Leu mutation which shares the same effect on<br />
receptor constitutive activity. Holst and Schwartz suggest<br />
that selective loss <strong>of</strong> ghrelin receptor constitutive activity<br />
causes a syndrome <strong>of</strong> SS and obesity developing around<br />
puberty. How these molecular changes impair growth and<br />
GH secretion and furthermore, how they are involved in<br />
hunger control and obesity, remains in part speculative<br />
and challenging for future research.<br />
It is interesting that clinical and genetic investigation<br />
essentially by systematic structure-function analysis opens<br />
new physiological concepts. It should be taken into account<br />
when performing population studies on the multifactorial<br />
control <strong>of</strong> growth and, more specifically, by GH and IGF<br />
secretion and activity. In addition, these studies open new<br />
possible mechanisms on the weight-to-height relationship<br />
in patients with SS. The authors briefly commented on the<br />
positive response to GH therapy in 3 patients. Eventually,<br />
their data, part <strong>of</strong> which are presented in the Table, would<br />
deserve an additional publication. More studies need to be<br />
performed in order to consider the potential development<br />
<strong>of</strong> pharmacological tools in relation to this uncommon and<br />
likely pathophysiology <strong>of</strong> SS and/or obesity.<br />
Raphaël Rappaport, MD<br />
28 <strong>GGH</strong> Vol. 22, No. 2 June 2006 www.<strong>GGH</strong>journal.com<br />
*<br />
Mnll