Acanthosis Nigricans: A New Manifestation of Insulin Resistance in ...
Acanthosis Nigricans: A New Manifestation of Insulin Resistance in ...
Acanthosis Nigricans: A New Manifestation of Insulin Resistance in ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
1 MARCH<br />
Correspondence<br />
<strong>Acanthosis</strong> <strong>Nigricans</strong>:<br />
A <strong>New</strong> <strong>Manifestation</strong><br />
<strong>of</strong> <strong>Insul<strong>in</strong></strong> <strong>Resistance</strong><br />
<strong>in</strong> Patients Receiv<strong>in</strong>g<br />
Treatment with Protease<br />
Inhibitors<br />
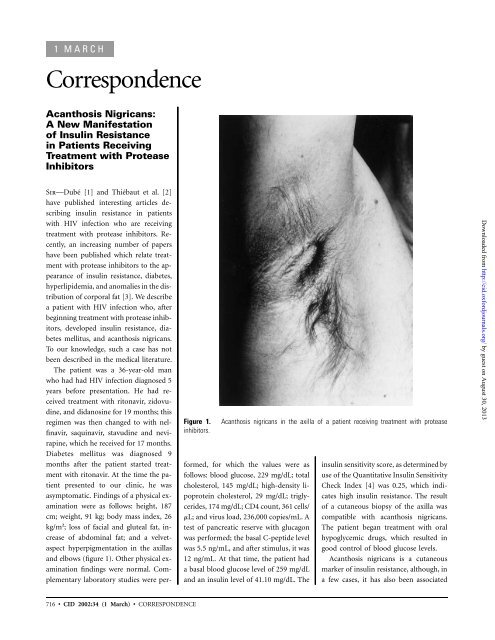
Figure 1.<br />
<strong>in</strong>hibitors.<br />
<strong>Acanthosis</strong> nigricans <strong>in</strong> the axilla <strong>of</strong> a patient receiv<strong>in</strong>g treatment with protease<br />
Sir—Dubé [1] and Thiébaut et al. [2]<br />
have published <strong>in</strong>terest<strong>in</strong>g articles describ<strong>in</strong>g<br />
<strong>in</strong>sul<strong>in</strong> resistance <strong>in</strong> patients<br />
with HIV <strong>in</strong>fection who are receiv<strong>in</strong>g<br />
treatment with protease <strong>in</strong>hibitors. Recently,<br />
an <strong>in</strong>creas<strong>in</strong>g number <strong>of</strong> papers<br />
have been published which relate treatment<br />
with protease <strong>in</strong>hibitors to the appearance<br />
<strong>of</strong> <strong>in</strong>sul<strong>in</strong> resistance, diabetes,<br />
hyperlipidemia, and anomalies <strong>in</strong> the distribution<br />
<strong>of</strong> corporal fat [3]. We describe<br />
a patient with HIV <strong>in</strong>fection who, after<br />
beg<strong>in</strong>n<strong>in</strong>g treatment with protease <strong>in</strong>hibitors,<br />
developed <strong>in</strong>sul<strong>in</strong> resistance, diabetes<br />
mellitus, and acanthosis nigricans.<br />
To our knowledge, such a case has not<br />
been described <strong>in</strong> the medical literature.<br />
The patient was a 36-year-old man<br />
who had had HIV <strong>in</strong>fection diagnosed 5<br />
years before presentation. He had received<br />
treatment with ritonavir, zidovud<strong>in</strong>e,<br />
and didanos<strong>in</strong>e for 19 months; this<br />
regimen was then changed to with nelf<strong>in</strong>avir,<br />
saqu<strong>in</strong>avir, stavud<strong>in</strong>e and nevirap<strong>in</strong>e,<br />
which he received for 17 months.<br />
Diabetes mellitus was diagnosed 9<br />
months after the patient started treatment<br />
with ritonavir. At the time the patient<br />
presented to our cl<strong>in</strong>ic, he was<br />
asymptomatic. F<strong>in</strong>d<strong>in</strong>gs <strong>of</strong> a physical exam<strong>in</strong>ation<br />
were as follows: height, 187<br />
cm; weight, 91 kg; body mass <strong>in</strong>dex, 26<br />
kg/m 2 ; loss <strong>of</strong> facial and gluteal fat, <strong>in</strong>crease<br />
<strong>of</strong> abdom<strong>in</strong>al fat; and a velvetaspect<br />
hyperpigmentation <strong>in</strong> the axillas<br />
and elbows (figure 1). Other physical exam<strong>in</strong>ation<br />
f<strong>in</strong>d<strong>in</strong>gs were normal. Complementary<br />
laboratory studies were performed,<br />
for which the values were as<br />
follows: blood glucose, 229 mg/dL; total<br />
cholesterol, 145 mg/dL; high-density lipoprote<strong>in</strong><br />
cholesterol, 29 mg/dL; triglycerides,<br />
174 mg/dL; CD4 count, 361 cells/<br />
mL; and virus load, 236,000 copies/mL. A<br />
test <strong>of</strong> pancreatic reserve with glucagon<br />
was performed; the basal C-peptide level<br />
was 5.5 ng/mL, and after stimulus, it was<br />
12 ng/mL. At that time, the patient had<br />
a basal blood glucose level <strong>of</strong> 259 mg/dL<br />
and an <strong>in</strong>sul<strong>in</strong> level <strong>of</strong> 41.10 mg/dL. The<br />
<strong>in</strong>sul<strong>in</strong> sensitivity score, as determ<strong>in</strong>ed by<br />
use <strong>of</strong> the Quantitative <strong>Insul<strong>in</strong></strong> Sensitivity<br />
Check Index [4] was 0.25, which <strong>in</strong>dicates<br />
high <strong>in</strong>sul<strong>in</strong> resistance. The result<br />
<strong>of</strong> a cutaneous biopsy <strong>of</strong> the axilla was<br />
compatible with acanthosis nigricans.<br />
The patient began treatment with oral<br />
hypoglycemic drugs, which resulted <strong>in</strong><br />
good control <strong>of</strong> blood glucose levels.<br />
<strong>Acanthosis</strong> nigricans is a cutaneous<br />
marker <strong>of</strong> <strong>in</strong>sul<strong>in</strong> resistance, although, <strong>in</strong><br />
a few cases, it has also been associated<br />
Downloaded from http://cid.oxfordjournals.org/ by guest on August 30, 2013<br />
716 • CID 2002:34 (1 March) • CORRESPONDENCE
with malignant diseases, receipt <strong>of</strong> certa<strong>in</strong><br />
medications, and uncommon illnesses<br />
[5]. It is characterized by the appearance<br />
<strong>of</strong> papillomatosis with hyperpigmented<br />
and hyperkeratosis plaques that have a<br />
velvet texture and a grayish-brown coloration.<br />
These lesions are distributed<br />
symmetrically and affect flexural areas,<br />
<strong>in</strong>clud<strong>in</strong>g the neck, axilla, gro<strong>in</strong>, antecubital<br />
and popliteal fossa, and the periumbilical<br />
region; occasionally, they can<br />
affect the mucous areas [6]. It is believed<br />
that hyper<strong>in</strong>sul<strong>in</strong>emia favors the bond<br />
between <strong>in</strong>sul<strong>in</strong> and growth factor receptors<br />
similar to <strong>in</strong>sul<strong>in</strong> receptors, which<br />
stimulate the proliferation <strong>of</strong> kerat<strong>in</strong>ocytes<br />
and fibroblasts <strong>in</strong> the dermis. The<br />
pathologic diagnosis <strong>in</strong>cludes hyperkeratosis<br />
and slight acanthosis with dermal<br />
papillomatosis.<br />
<strong>Insul<strong>in</strong></strong> resistance appears <strong>in</strong> 61% <strong>of</strong><br />
patients with HIV <strong>in</strong>fection who are receiv<strong>in</strong>g<br />
treatment with protease <strong>in</strong>hibitors<br />
[7], but acanthosis nigricans has only<br />
been described as associated with HIV<br />
<strong>in</strong>fection <strong>in</strong> 1 patient with opportunistic<br />
<strong>in</strong>fections [8]. It has not, to our knowledge,<br />
been described as associated with<br />
protease <strong>in</strong>hibitor treatments, although it<br />
is not <strong>in</strong>frequent <strong>in</strong> other situations <strong>of</strong><br />
<strong>in</strong>sul<strong>in</strong> resistance [9]. <strong>Acanthosis</strong> nigricans<br />
should be considered a new manifestation<br />
<strong>of</strong> <strong>in</strong>sul<strong>in</strong> resistance syndrome<br />
<strong>in</strong> patients with HIV <strong>in</strong>fection who are<br />
receiv<strong>in</strong>g treatment with protease <strong>in</strong>hibitors;<br />
the sk<strong>in</strong> <strong>of</strong> such patients should be<br />
carefully exam<strong>in</strong>ed.<br />
Susana Mellor-Pita, Miguel Yebra-Bango,<br />
Joaquín Alfaro-Martínez, and Emilio Suárez<br />
Servicio de Medic<strong>in</strong>a Interna 1, Cl<strong>in</strong>ica Puerta<br />
de Hierro Universidad Autónoma de Madrid,<br />
Madrid, Spa<strong>in</strong><br />
References<br />
1. Dubé MP. Disorders <strong>of</strong> glucose metabolism <strong>in</strong><br />
patients <strong>in</strong>fected with human immunodeficiency<br />
virus. Cl<strong>in</strong> Infect Dis 2000; 31:1467–75.<br />
2. Thiébaut R, Daucourt V, Mercié P, et al. Lipodystrophy,<br />
metabolic disorders, and human<br />
immunodeficiency virus <strong>in</strong>fection: Aquita<strong>in</strong>e<br />
cohort, France, 1999. Cl<strong>in</strong> Infect Dis 2000;<br />
31:1482–7.<br />
3. Carr A, Samaras K, Burton S, et al. A syndrome<br />
<strong>of</strong> peripheral lipodystrophy, hyperlipidaemia<br />
and <strong>in</strong>sul<strong>in</strong> resistance <strong>in</strong> patients receiv<strong>in</strong>g<br />
HIV protease <strong>in</strong>hibitors. AIDS 1998;<br />
12:F51–58.<br />
4. Katz A, Nambi SS, Mather K, Baron AD, et<br />
al. Quantitative <strong>in</strong>sul<strong>in</strong> sensitivity check <strong>in</strong>dex:<br />
a simple, accurate method for assess<strong>in</strong>g<br />
<strong>in</strong>sul<strong>in</strong> sensitivity <strong>in</strong> humans. J Cl<strong>in</strong> Endocr<strong>in</strong>ol<br />
Metab 2000; 85:2402–10.<br />
5. Houpt KR, Cruz PD. <strong>Acanthosis</strong> nigricans. In:<br />
Freedberg IM, Eisen AZ, Wolff K, et al., eds.<br />
Fitzpatrick’s dermatology <strong>in</strong> general medic<strong>in</strong>e.<br />
5th ed. <strong>New</strong> York: McGraw-Hill, 1999:<br />
2121–6.<br />
6. Flier JS. Metabolic importance <strong>of</strong> acanthosis<br />
nigricans. Arch Dermatol 1985; 121:193–4.<br />
7. Walli R, Herfort O, Gerl<strong>in</strong>de M, et al. Treatment<br />
with protease <strong>in</strong>hibitors associated with<br />
peripheral <strong>in</strong>sul<strong>in</strong> resistance and impaired<br />
oral glucose tolerance <strong>in</strong> HIV-1 <strong>in</strong>fected patients.<br />
AIDS 1998; 12:F167–73.<br />
8. Maltez, F, Mart<strong>in</strong>s T, Morgado A, Proença R.<br />
Será a canthosis nigricans uma nova manifestação<br />
cutâtanea de <strong>in</strong>fecção pelo vírus da<br />
imunodeficiência humana? Acta Médica Portuguesa<br />
1997; 10:493–5.<br />
9. Stuart CA, Pate CJ, Peters EJ. Prevalence <strong>of</strong><br />
acanthosis nigricans <strong>in</strong> an unselected population.<br />
Am J Med 1989; 87:269–72.<br />
Repr<strong>in</strong>ts or correspondence: Dr. Susana Mellor-Pita, Servicio<br />
de Medic<strong>in</strong>a Interna 1, Cl<strong>in</strong>ica Puerta de Hierro Universidad<br />
Autónoma de Madrid, San Martín de Porres, 4, 28035 Madrid,<br />
Spa<strong>in</strong>.<br />
Cl<strong>in</strong>ical Infectious Diseases 2002; 34:716–7<br />
2002 by the Infectious Diseases Society <strong>of</strong> America. All<br />
rights reserved. 1058-4838/2002/3405-0027$03.00<br />
Seroreversion from<br />
Hepatitis C after<br />
Needlestick Injury<br />
Sir—We read with great <strong>in</strong>terest the case<br />
report by Morand et al. [1], which described<br />
the lack <strong>of</strong> seroconversion after<br />
early treatment <strong>of</strong> acute hepatitis C follow<strong>in</strong>g<br />
needlestick <strong>in</strong>jury, despite the<br />
normal cellular and humoral responses<br />
<strong>of</strong> the host. Because few reports are available<br />
on the course <strong>of</strong> the immune response<br />
to hepatitis C virus (HCV) after<br />
early treatment <strong>of</strong> acute hepatitis C, we<br />
present a case report that may contribute<br />
to discussion <strong>of</strong> this topic.<br />
In January 1997, a 34-year-old nurse<br />
who had tested negative for both HIV<br />
and HCV susta<strong>in</strong>ed a superficial wound<br />
caused by a venipuncture needle. The donor<br />
patient was HIV negative, was <strong>in</strong>fected<br />
with an HCV isolate <strong>of</strong> genotype<br />
1a (identified by Inno-LiPA HCV II;<br />
InGeN), and had a high virus load (7.00<br />
log 10 copies/mL) detected <strong>in</strong> serum samples<br />
(Cobas Monitor HCV; Roche Diagnostic<br />
System).<br />
Follow-up exam<strong>in</strong>ation <strong>of</strong> the nurse<br />
<strong>in</strong>cluded determ<strong>in</strong>ation <strong>of</strong> the alan<strong>in</strong>e<br />
am<strong>in</strong>otransferase (ALT) level, monthly<br />
test<strong>in</strong>g for HCV RNA by the use <strong>of</strong> reverse-transcriptase<br />
PCR (Cobas Amplicor<br />
HCV; Roche Diagnostic System),<br />
and anti-HCV test<strong>in</strong>g with the use <strong>of</strong> 2<br />
third-generation ELISAs (AxSYM HCV,<br />
version 3.0 [Abbott Laboratories], and<br />
Monolisa HCV Plus [Bio-Rad]). Specific<br />
antibody response was analyzed by strip<br />
immunoassay (SIA); analysis was done <strong>in</strong><br />
1997, by use <strong>of</strong> the Deciscan assay (Bio-<br />
Rad), and then retrospectively, by use <strong>of</strong><br />
the RIBA HCV 3.0 assay (Chiron). The<br />
results <strong>of</strong> these analyses are shown <strong>in</strong> table<br />
1.<br />
Dur<strong>in</strong>g the first month after exposure<br />
(month 1), HCV RNA was detected <strong>in</strong><br />
the nurse’s serum, and her ALT level <strong>in</strong>creased<br />
to up to 3.5 times greater than<br />
the normal level. The nurse was found<br />
to have the same HCV genotype as the<br />
donor patient. Treatment with IFN-a<br />
(3 million U given 3 times per week for<br />
3 months) was <strong>in</strong>itiated immediately.<br />
Treatment cont<strong>in</strong>ued pragmatically for a<br />
total <strong>of</strong> 6 months, because HCV RNA was<br />
still detectable after 1 month and because<br />
it became undetectable only after 3<br />
months <strong>of</strong> therapy. A prolonged response<br />
was obta<strong>in</strong>ed; a normal ALT level and the<br />
absence <strong>of</strong> detectable RNA were noted 4<br />
years after the needlestick <strong>in</strong>jury. Anti-<br />
HCV antibodies were detected by ELISA<br />
only at and after month 2. An isolated<br />
reactivity aga<strong>in</strong>st HCV core prote<strong>in</strong> was<br />
detected by RIBA dur<strong>in</strong>g month 1, and<br />
the response <strong>in</strong>creased until month 4.<br />
Specific anti–HCV core prote<strong>in</strong> response<br />
rapidly decreased at the end <strong>of</strong> treatment.<br />
Four years after the needlestick <strong>in</strong>jury oc-<br />
Downloaded from http://cid.oxfordjournals.org/ by guest on August 30, 2013<br />
CORRESPONDENCE • CID 2002:34 (1 March) • 717