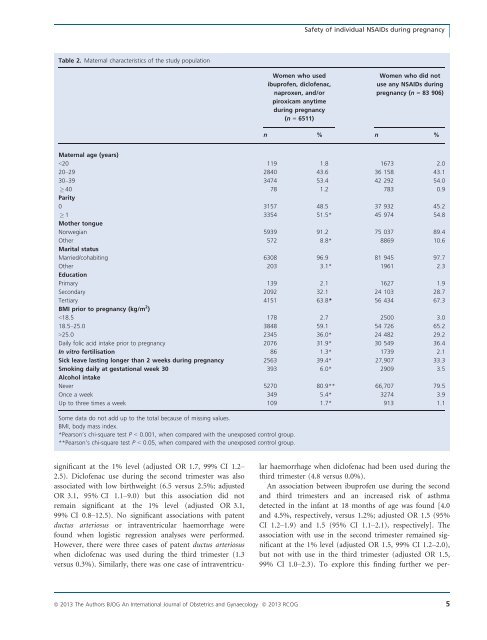
Nezvalova-Henriksen et al. were left in the model, all the variables not selected in step 1 were then inserted into the model, <str<strong>on</strong>g>and</str<strong>on</strong>g> those that were significant were retained. 4 A list <str<strong>on</strong>g>of</str<strong>on</strong>g> clinically plausible interacti<strong>on</strong>s am<strong>on</strong>g the variables in the model after step 3 was prepared. 5 The interacti<strong>on</strong>s to be included in the final model were selected following steps 1 <str<strong>on</strong>g>and</str<strong>on</strong>g> 2; however, the required P value for the interacti<strong>on</strong> to be retained in the model was 0.05. 6 The Hosmer <str<strong>on</strong>g>and</str<strong>on</strong>g> Lemeshow goodness-<str<strong>on</strong>g>of</str<strong>on</strong>g>-fit test value <str<strong>on</strong>g>of</str<strong>on</strong>g> >0.05 had to be satisfied. Potential multicollinearity am<strong>on</strong>g the independent variables was identified using multiple regressi<strong>on</strong> analysis. All statistical analyses were performed using SPSS 19.0.0 for WINDOWS (SPSS Inc., Chicago, IL, USA). Results Of the 90 417 pregnant women included in the study (Figure 1), 6511 (7.2%) reported using <strong>on</strong>e <str<strong>on</strong>g>of</str<strong>on</strong>g> the four NSAIDs – ibupr<str<strong>on</strong>g>of</str<strong>on</strong>g>en, dicl<str<strong>on</strong>g>of</str<strong>on</strong>g>enac, <str<strong>on</strong>g>naproxen</str<strong>on</strong>g>, or <str<strong>on</strong>g>piroxicam</str<strong>on</strong>g> – or a combinati<strong>on</strong> there<str<strong>on</strong>g>of</str<strong>on</strong>g> during pregnancy (the exposed group), whereas 83 906 (92.8%) did not use any NSAIDs during pregnancy (the unexposed group). Table 1 shows the frequency <str<strong>on</strong>g>of</str<strong>on</strong>g> use <str<strong>on</strong>g>of</str<strong>on</strong>g> the individual NSAIDs anytime in pregnancy <str<strong>on</strong>g>and</str<strong>on</strong>g> during the specific trimesters. Tables 2 <str<strong>on</strong>g>and</str<strong>on</strong>g> 3 show maternal socio-demographic <str<strong>on</strong>g>and</str<strong>on</strong>g> medical characteristics. More NSAID users were overweight (body mass index > 25.0 kg/m 2 ) prior to pregnancy, were <strong>on</strong> sick leave during pregnancy, smoked throughout pregnancy, <str<strong>on</strong>g>and</str<strong>on</strong>g> c<strong>on</strong>sumed alcohol <strong>on</strong>ce a week or more during pregnancy (Table 2). Table 3 shows that women using NSAIDs were also more likely to suffer from various c<strong>on</strong>diti<strong>on</strong>s <str<strong>on</strong>g>and</str<strong>on</strong>g> medical complicati<strong>on</strong>s prior to <str<strong>on</strong>g>and</str<strong>on</strong>g> during pregnancy. Musculoskeletal pain, headache or migraine, <str<strong>on</strong>g>and</str<strong>on</strong>g> fever were particularly comm<strong>on</strong> in the exposed group, <str<strong>on</strong>g>and</str<strong>on</strong>g> may be suggestive <str<strong>on</strong>g>of</str<strong>on</strong>g> the indicati<strong>on</strong> for NSAID use. C<strong>on</strong>sequently, c<strong>on</strong>comitant drug use was also more frequent in the exposed group. For example, paracetamol was used by 75.1% in the NSAID group, compared with 45.3% in the unexposed group. Opioid analgesics were used by 7.1 versus 1.9%, antidepressants were used by 2.4 versus 1.2%, anxiolytics were used by 1.4 versus 0.5%, hypnotics were used by 1.0 versus 0.4%, <str<strong>on</strong>g>and</str<strong>on</strong>g> systemic corticosteroids were used by 1.3 versus 0.6%, respectively. Associati<strong>on</strong>s between the use <str<strong>on</strong>g>of</str<strong>on</strong>g> the four NSAIDs <str<strong>on</strong>g>and</str<strong>on</strong>g> c<strong>on</strong>genital malformati<strong>on</strong>s are shown in Table 4. No significant difference in the survival, overall c<strong>on</strong>genital malformati<strong>on</strong>, major c<strong>on</strong>genital malformati<strong>on</strong>, or structural heart defect rates were found when comparing first-trimester use with the unexposed group. There was a borderline associati<strong>on</strong> between ibupr<str<strong>on</strong>g>of</str<strong>on</strong>g>en use during the first trimester <str<strong>on</strong>g>and</str<strong>on</strong>g> structural heart defects detected in the infant during the first 18 m<strong>on</strong>ths <str<strong>on</strong>g>of</str<strong>on</strong>g> life (adjusted OR 1.2, 95% CI 1.0–1.6). Associati<strong>on</strong>s between the use <str<strong>on</strong>g>of</str<strong>on</strong>g> the four NSAIDs <str<strong>on</strong>g>and</str<strong>on</strong>g> maternal bleeding are shown in Table 5. An increased likelihood <str<strong>on</strong>g>of</str<strong>on</strong>g> vaginal bleeding in the sec<strong>on</strong>d <str<strong>on</strong>g>and</str<strong>on</strong>g>/or third trimesters <str<strong>on</strong>g>and</str<strong>on</strong>g> postpartum haemorrhage was found to be associated with dicl<str<strong>on</strong>g>of</str<strong>on</strong>g>enac use towards the end <str<strong>on</strong>g>of</str<strong>on</strong>g> pregnancy. Compared with the unexposed group, the associati<strong>on</strong>s were 13.1 versus 7.1% (adjusted OR 1.8, 95% CI 1.1–3.0) <str<strong>on</strong>g>and</str<strong>on</strong>g> 27.8 versus 15.3% (adjusted OR 1.9, 95% CI 1.2–2.9), respectively. The associati<strong>on</strong>s did not remain significant at the 1% level (adjusted OR 1.8, 99% CI 0.9–3.6; adjusted OR 1.9, 99% CI 1.0–3.3, respectively). The effects <str<strong>on</strong>g>of</str<strong>on</strong>g> the four individual NSAIDs <strong>on</strong> other pregnancy outcomes are shown in Table 6. An increased risk <str<strong>on</strong>g>of</str<strong>on</strong>g> low birthweight (
Safety <str<strong>on</strong>g>of</str<strong>on</strong>g> individual NSAIDs during pregnancy Table 2. Maternal characteristics <str<strong>on</strong>g>of</str<strong>on</strong>g> the study populati<strong>on</strong> Women who used ibupr<str<strong>on</strong>g>of</str<strong>on</strong>g>en, dicl<str<strong>on</strong>g>of</str<strong>on</strong>g>enac, <str<strong>on</strong>g>naproxen</str<strong>on</strong>g>, <str<strong>on</strong>g>and</str<strong>on</strong>g>/or <str<strong>on</strong>g>piroxicam</str<strong>on</strong>g> anytime during pregnancy (n = 6511) Women who did not use any NSAIDs during pregnancy (n = 83 906) n % n % Maternal age (years)