Medeiros et al Am J Surg Pathol • Volume 28, Number 7, July 2004 MATERIALS AND METHODS The study group included 25 <strong>tumors</strong> that were typical of GIST clinically and histologically, but were <strong>KIT</strong> <strong>negative</strong> by immunohistochemistry in formalin-fixed, paraffin-embedded tissue. Among 495 consecutive GISTs diagnosed between 1999 and 2002 at the Brigham and Women’s Hospital, 20 <strong>tumors</strong> (4%) were <strong>KIT</strong> <strong>negative</strong> by immunohistochemistry. The remaining 5 <strong>tumors</strong> were obtained from consultation files of the Department of Pathology at Oregon Health & Science University. Five of the 25 cases were included in a prior study. 9 Only surgical excision specimens were included to minimize nonrepresentative sampling. Patients previously treated with imatinib mesylate were excluded from the study to avoid treatment effect as a cause of <strong>KIT</strong> negativity. Clinical data, including age, gender, and tumor location, were obtained in all cases. An average of five hematoxylineosin archival slides per case was available for histopathologic review. Paraffin-embedded, formalin-fixed tissue was used for immunohistochemical analysis. Tissue sections were cut at 4 µm and incubated with the primary antibodies for 40 minutes at room temperature. Immunohistochemistry was performed for <strong>KIT</strong> (Dako Corporation, Carpinteria, CA; polyclonal A4502, 1:250) in all cases without epitope retrieval, as previously described. 12 Selected cases were also stained for CD34 (Dako Corporation; clone Qbend10, 1:400), desmin (Dako Corporation; D33, 1:500), S-100 (Dako Corporation; polyclonal, 1:3000), -smooth-muscle actin (Sigma, St. Louis, MO; clone 1A4, 1:20,000), and keratins (Dako Corporation; AE1/AE3; 1:200) with the Envison+ avidin-biotin peroxidase kit (Dako Corporation) according to manufacturer’s specifications. <strong>KIT</strong>positive GISTs were used as positive controls. Negative controls consisted of substituting normal serum for the primary antibody, which resulted in no staining of the tissues. Cytogenetic analysis was performed in 4 cases according to standard procedures. 7 Immunoblotting was performed using total cell lysates from snap-frozen GIST specimens, as described elsewhere. 8,17 Mutational analyses were performed on DNA extracted from paraffin-embedded tumor tissue using a combination of PCR amplification, denaturing high performance liquid chromatography screening, and automated sequencing, as described previously. 2,9,17 RESULTS There were 17 males and 8 females (2:1). The median age at diagnosis was 56 years (range 29–79 years). Most <strong>tumors</strong> originated in the stomach (N = 14, 56%), followed by omentum/mesentery (N = 5) and small bowel (N = 1). In 5 cases (20%), there was an intraabdominal mass, but no distinct primary site could be identified at the time of presentation, suggesting possible peritoneal origin. Tumor size ranged from 4 to 38 cm (median, 8.5 cm) (Table 1). All <strong>tumors</strong> exhibited classic histologic features for GIST, being composed of cellular sheets, fascicles, or nests of cells that lacked significant nuclear pleomorphism. Tumor cell cytoplasm was eosinophilic and slightly fibrillary with illdefined cytoplasmic borders, producing a somewhat syncytial appearance. Nuclei were spindled, ovoid, or rounded and had a uniform appearance with evenly distributed chromatin. The majority of cases showed epithelioid cell morphology (N = 13, 52%) (Fig. 1). Eight <strong>tumors</strong> were of mixed (epithelioid and spindle) cell type (32%) and four were composed of spindle cells only (16%). Mitoses ranged from 1 to 52 per 50 high power fields (median 8) (Table 1). All cases were classified as intermediate or high risk for aggressive behavior based on tumor size and mitotic count. 5 All <strong>tumors</strong> completely lacked <strong>KIT</strong> staining by immunohistochemistry (Fig. 1). Complementary immunostains were performed in 23 cases to exclude other <strong>tumors</strong> in the differential diagnosis. Eleven and 10 cases were positive for CD34 and smooth muscle actin, respectively. Focal S-100 protein positivity was detected in one tumor. All cases that were evaluated for desmin and keratin were <strong>negative</strong> for these markers. Cytogenetic analysis revealed noncomplex karyotypes and a typical loss of chromosome 14 in all four cases evaluated. One tumor also showed deletion of chromosome 22 (Table 2). Mutational analysis revealed <strong>KIT</strong> mutations in 4 cases (16%) and PDGFRA mutations in 18 cases (72%) (Table 1). In only 3 cases, no <strong>KIT</strong> or PDGFRA mutations were identified. Most PDGFRA mutations involved exon 18 (15 of 18, 83%), including 11 GISTs with missense mutations leading to a substitution of valine (N = 9) or tyrosine (N = 2) for aspartic acid 842 (D842V and D842Y, respectively). The D842Y mutation is a novel mutation, whereas the D842V mutation has been reported previously. 9 The remaining 4 GISTs with PDGFRA exon 18 mutations had in-frame deletions. Two GISTs had PDGFRA mutations in exon 12 (N = 2), encoding the PDGFRA juxtamembrane region, and one GIST had a previously undescribed point mutation in PDGFRA exon 14 (N659K). Four GISTs had <strong>KIT</strong> mutations involving either exon 11 (N = 3) or exon 9 (N = 1) (Table 1). No tumor had mutations of both PDGFRA and <strong>KIT</strong> or more than one mutation in either of these genes. Immunoblotting, performed in 3 cases with available snap-frozen tissue, confirmed absence of <strong>KIT</strong> protein expression (Fig. 2). Two of these GISTs expressed phosphorylated and total PDGFRA strongly and had PDGFRA oncogenic mutations (Fig. 2; Table 1). The third case expressed neither <strong>KIT</strong> nor PDGFRA but had a <strong>KIT</strong> exon 11 mutation (Fig. 2; Table 1). DISCUSSION GIST is a mesenchymal neoplasm that exhibits morphologic and immunophenotypic features similar to the interstitial cells of Cajal, which are pacemaker cells regulating <strong>gastrointestinal</strong> peristalsis. 13 A characteristic feature of GISTs, similar to the Cajal cells, is expression of the protein tyrosine kinase <strong>KIT</strong>, which is readily detected by immunohistochemistry and 890 © 2004 Lippincott Williams & Wilkins
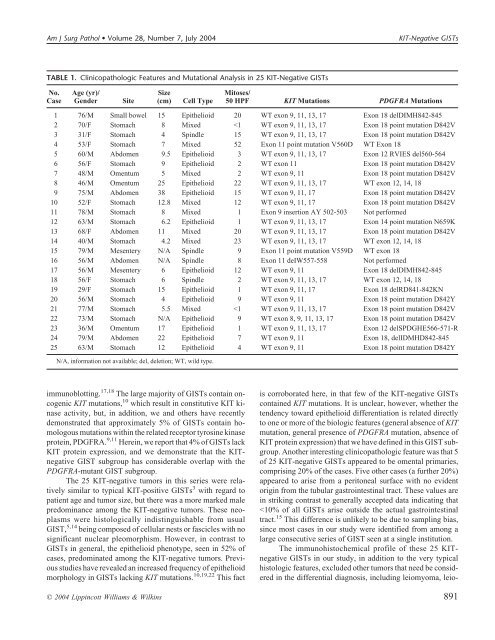
Am J Surg Pathol • Volume 28, Number 7, July 2004 <strong>KIT</strong>-Negative GISTs TABLE 1. Clinicopathologic Features and Mutational Analysis in 25 <strong>KIT</strong>-Negative GISTs No. Case Age (yr)/ Gender Site Size (cm) Cell Type Mitoses/ 50 HPF <strong>KIT</strong> Mutations PDGFRA Mutations 1 76/M Small bowel 15 Epithelioid 20 WT exon 9, 11, 13, 17 Exon 18 delDIMH842-845 2 70/F Stomach 8 Mixed