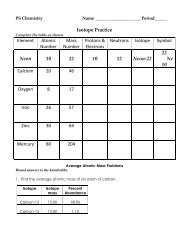
PS Chemistry: Chapter 23, Sections 1 & 2 Acids & Bases ...
PS Chemistry: Chapter 23, Sections 1 & 2 Acids & Bases ...
PS Chemistry: Chapter 23, Sections 1 & 2 Acids & Bases ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>PS</strong> <strong>Chemistry</strong>: <strong>Chapter</strong> <strong>23</strong>, <strong>Sections</strong> 1 & 2 <strong>Acids</strong> & <strong>Bases</strong><br />
Name__________________Period____<br />
Identify: For each item below, indicate if it is an acid (A), base (B), or both (AB).<br />
_____ 1. sour taste<br />
_____ 2. bitter taste<br />
_____ 3. produces hydrogen ions in solution<br />
_____ 4. is often corrosive<br />
_____ 5. is slippery<br />
_____ 6. can cause severe burns<br />
and tissue damage<br />
_____ 7. produces hydroxide ions in solution<br />
_____ 8. reacts with a predictable indicator<br />
to produce a color change<br />
_____ 9. soaps are an example<br />
_____ 10. gastric juice in stomach<br />
_____ 11. produces hydronium ions<br />
_____ 12. HCl is an example<br />
_____ 13. ammonia is a common example<br />
_____ 14. a solution that contains more<br />
H 3 O + ions than OH - ions<br />
Matching: Match the following acids and/or bases with their common use or where they are found.<br />
_____ 1. acetic acid<br />
_____ 2. ascorbic acid<br />
_____ 3. carbonic acid<br />
_____ 4. hydrochloric acid<br />
_____ 5. sulfuric acid<br />
_____ 6. lactic acid<br />
_____ 7. citric acid<br />
_____ 8. aluminum hydroxide<br />
_____ 9. magnesium hydroxide<br />
A. car batteries<br />
B. soda pop<br />
C. laxative<br />
D. citrus fruit<br />
E. vinegar<br />
F. soap making<br />
G. antacid<br />
H. vitamin C<br />
I. digestion in stomach<br />
J. yogurt<br />
_____ 10. sodium hydroxide<br />
Short Answer: Answer the following question.<br />
1. What makes an acid strong?<br />
2. What makes a base strong?
Labeling: The pH values of several common substances are listed below. Place each item from the<br />
list on the pH scale in its proper location. The first one has been done for you.<br />
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14<br />
l l l l l l l l l l l l l l l<br />
Pure water 7.0<br />
Ocean water 8.5<br />
Tomatoes 4.0<br />
Lye 13.8<br />
Stomach acid 1.0<br />
Lemons 2.5<br />
Shampoo 5.8<br />
Bananas 5.2<br />
Blood 7.2<br />
Milk of magnesia 10.5<br />
Ammonia 11.5<br />
Eggs 7.8<br />
Soap 10.0<br />
Vinegar 3.0<br />
Battery acid 1.5<br />
Completion: Complete the table below by writing the name of each substance above under the<br />
proper heading. Place substances with a pH lower than 3.0 in the strong acid column. Place<br />
substances with a pH higher than 10.0 in the strong bases column.<br />
Strong acids Weak acids Weak bases Strong bases<br />
Short Answer: Answer the following question.<br />
1. Is pure water acidic, basic, or neutral substance?<br />
2. Is the pH of a strong acid higher or lower than the pH of a weak acid of the same<br />
concentration?<br />
3. Is the pH of a strong base higher or lower than the pH of a weak acid of the same<br />
concentration?<br />
4. On the pH scale, what are the values of acids and what are the values of bases?